Validating Color-Changing Sensor Performance for Plant Health Assessment: A Comprehensive Guide for Researchers
This article provides a detailed examination of color-changing sensor technology for plant health assessment, catering to researchers and scientists in agricultural technology and drug development.
Validating Color-Changing Sensor Performance for Plant Health Assessment: A Comprehensive Guide for Researchers
Abstract
This article provides a detailed examination of color-changing sensor technology for plant health assessment, catering to researchers and scientists in agricultural technology and drug development. It explores the foundational principles of these sensors, which detect biomarkers like proline to indicate stress, and delves into their practical application methodologies. The content addresses key challenges such as environmental interference and data interpretation, offering optimization strategies. Furthermore, it presents rigorous validation frameworks, including comparisons with hyperspectral imaging and deep learning models, to evaluate sensor accuracy and reliability. The synthesis aims to equip professionals with the knowledge to implement and advance this promising technology in precision agriculture and related life science fields.
The Science Behind Color-Changing Sensors: Principles and Biomarkers
Colorimetric biosensors represent a significant advancement in detection technologies, offering a practical and accessible means to identify the presence and concentration of specific biomarkers through simple color changes. These sensors operate on the principle that interactions between a target analyte and a sensing element produce a visible color shift, which can be qualitatively assessed by the naked eye or quantitatively analyzed using various digital tools [1]. This dual capability makes colorimetric sensors particularly valuable for applications ranging from point-of-care medical diagnostics to environmental monitoring and agricultural assessment [2] [1].
At the heart of these systems lies a biochemical recognition mechanism, where a selective reaction with a target biomarker triggers a color change in an indicator compound. The resulting color intensity or hue can then be correlated to the concentration of the biomarker present [1]. Recent advancements have integrated functional nanomaterials and digital analysis technologies to significantly enhance the sensitivity, specificity, and practicality of these detection systems [1]. This article explores the core principles, experimental methodologies, and performance characteristics of colorimetric biomarker detection, with a specific focus on validating sensor performance for plant health assessment research.
Fundamental Operating Principles
The operational foundation of colorimetric biosensors rests on specific interactions between a sensing element and a target biomarker that generate a visible color change. These interactions can be driven by several distinct mechanisms, each leveraging different chemical and optical phenomena.
Biochemical Recognition and Reaction
The most fundamental principle involves the direct chemical reaction between a biomarker and a chromogenic reagent. A prominent example from plant science is the detection of proline, a universal biomarker for plant stress. Researchers have developed sensors that leverage a natural reaction inspired by the nesocodon flower. In this system, proline interacts with a sensing element containing sinapaldehyde, leading to the formation of a red pigment called nesocodin. The intensity of the resulting red color is directly proportional to the concentration of proline, thereby providing a visual indication of the plant's stress level [3].
Nanomaterial-Based Mechanisms
Nanomaterials have dramatically enhanced colorimetric sensing capabilities, primarily through two sophisticated mechanisms:
- Localized Surface Plasmon Resonance (LSPR): Noble metal nanoparticles, such as gold and silver, exhibit intense colors due to the collective oscillation of their surface electrons when exposed to light [1]. When these nanoparticles aggregate in the presence of a target biomarker, the change in interparticle distance alters this oscillation, causing a visible color shift—for instance, from red to purple or blue [1]. This mechanism is highly sensitive to minute changes in the nanoparticle's environment.
- Enzyme-Mimetic Nanomaterials (Nanozymes): Certain nanomaterials possess catalytic activity similar to natural enzymes. For example, nanomagnets can mimic peroxidase enzymes, catalyzing color-producing reactions with advantages in stability, cost, and shelf life over their natural counterparts [1].
Other Recognition Mechanisms
Other common mechanisms include pH-induced color changes, where a biomarker alters the local acidity, causing a pH-sensitive dye to change color, and redox reactions, where the biomarker acts as a reducing or oxidizing agent for a metal salt or organic chromogen [1]. The choice of mechanism depends on the specific biomarker, the required sensitivity, and the application environment.
Experimental Protocols for Sensor Validation
Robust experimental protocols are essential for developing and validating colorimetric sensors. The following detailed methodology for detecting proline as a plant stress biomarker serves as a representative template for sensor validation.
Protocol: Detection of Proline for Plant Health Assessment
Objective: To quantitatively determine the stress level in plants by measuring the concentration of proline in leaf samples using a colorimetric sensor [3].
Materials and Reagents:
- Colorimetric sensor strips embedded with sinapaldehyde [3]
- Plant leaf samples (e.g., cabbage, kale, broccoli)
- Ethanol (for extraction)
- Mechanical grinder or mortar and pestle
- Microcentrifuge tubes
- Pipettes and disposable tips
- Standardized color chart or digital imaging system (scanner or smartphone)
Experimental Workflow:
Procedure Steps:
- Sample Collection and Preparation: Clip a small piece (approximately 1 cm²) from a plant leaf. Using a mechanical grinder or mortar and pestle, homogenize the leaf tissue into a fine consistency [3].
- Biomarker Extraction: Transfer the ground leaf material to a microcentrifuge tube. Add a sufficient volume of ethanol (e.g., 1 mL) to submerge the material and extract proline. The mixture can be vortexed briefly to enhance extraction [3].
- Sensor Reaction: Immerse the sinapaldehyde-embedded sensor strip into the ethanolic extract for a defined period, ensuring full contact with the liquid [3].
- Color Development: Allow the sensor to react for approximately 15 minutes. During this time, proline (if present) reacts with sinapaldehyde to form the red pigment nesocodin. A bright red color indicates high proline concentration and thus high plant stress, while a pale yellow indicates a healthy plant [3].
- Data Acquisition and Analysis:
- Qualitative Assessment: Visually compare the sensor's color against a standardized reference chart with predefined color ranges corresponding to proline concentrations [3].
- Quantitative Analysis: Capture an image of the sensor using a scanner or smartphone under controlled lighting. Use image analysis software to extract color data (e.g., RGB, HSV, or CIELAB values) and correlate these values to a standard curve for proline concentration [3] [4].
Performance Comparison of Detection Modalities
The performance of colorimetric sensors can be evaluated against other common biosensing modalities. The table below provides a comparative overview based on key metrics relevant to research and potential point-of-care applications.
Table 1: Performance Comparison of Biosensor Modalities for Biomarker Detection
| Detection Modality | Principle of Operation | Sensitivity (Typical LOD) | Analysis Time | Cost & Equipment Needs | Key Advantages | Key Limitations |
|---|---|---|---|---|---|---|
| Colorimetric | Visual color change from biomarker-sensor reaction [3] | Moderate (e.g., nM to µM) [5] | Minutes to <1 hour [3] [5] | Low; minimal equipment for qualitative readout [3] [5] | Simple, low-cost, suitable for field use, visual result [3] [1] | Subject to subjective interpretation, can be less sensitive than other methods [5] [1] |
| Electrochemical | Measures electrical signal (current/potential) from biomarker reaction [5] | High (e.g., pM to nM) [5] | Minutes [5] | Moderate; requires potentiostat/reader [5] | High sensitivity, good for miniaturization [5] | Sensor fouling, requires stable power source [5] |
| Fluorescence | Detects light emission from fluorescent labels upon biomarker binding [5] | Very High (e.g., fM to pM) [5] | Minutes to hours [5] | High; requires excitation source and detector [5] | Extremely high sensitivity, multiplexing capability [5] | Photo-bleaching, background autofluorescence, complex instrumentation [5] |
| Traditional Methods (ELISA/PCR) | Immunoassay (ELISA) or nucleic acid amplification (PCR) [5] | Very High [5] | Hours to days [5] | High; requires lab infrastructure and trained personnel [5] | Gold standard, high accuracy and specificity [5] | Time-consuming, expensive, not portable [5] |
The data reveals a clear trade-off between performance and practicality. While fluorescence and electrochemical methods offer superior sensitivity, colorimetric sensors provide a compelling balance of sufficient sensitivity, rapid results, and minimal resource requirements, making them particularly suitable for applications in resource-limited or field settings [5].
Advanced Signal Acquisition and Data Processing
Modern colorimetric analysis has moved beyond subjective visual inspection to sophisticated digital quantification, significantly improving accuracy and reproducibility.
Color Spaces and Digital Quantification
When a sensor image is captured, its color information is converted into digital values using various color models. Each model offers different advantages for analytical chemistry [4] [6]:
- RGB (Red, Green, Blue): An additive model where colors are combinations of red, green, and blue primary colors. It is device-dependent but widely used [4].
- HSV/HSL (Hue, Saturation, Value/Lightness): Separate the pure color (hue) from its intensity (saturation) and brightness (value/lightness), which can be more aligned with human perception and less susceptible to lighting variations [4] [6].
- CIELAB (Lab): A perceptually uniform color space where L represents lightness, a* the green-red component, and b* the blue-yellow component. A small numerical change in CIELAB corresponds to a similar perceived color change, making it highly suitable for precise colorimetric analysis [4] [6].
Table 2: Common Color Models Used in Quantitative Colorimetric Analysis
| Color Model | Core Components | Advantages for Analysis | Common Use Cases |
|---|---|---|---|
| RGB | Red, Green, Blue channels | Directly obtained from most image sensors; simple to process [4] | Basic color change detection; initial data capture [4] |
| HSV/HSL | Hue, Saturation, Value/Lightness | Separates color from intensity; more robust to non-uniform lighting [4] [6] | Analyzing samples under variable light conditions [4] |
| CIELAB | L* (Lightness), a* (Green-Red), b* (Blue-Yellow) | Perceptually uniform; distances in space correlate to human perception of color difference [4] | High-precision quantitative analysis; subtle color change detection [4] |
The Role of Artificial Intelligence and Machine Learning
The integration of Artificial Intelligence (AI) and Machine Learning (ML) is a transformative advancement for colorimetric biosensors. These technologies automate data interpretation and enhance prediction accuracy [7] [8].
- Automated Pattern Recognition: Deep learning models, particularly Convolutional Neural Networks (CNNs), can be trained on large datasets of sensor images to directly map color patterns to biomarker concentrations, even with complex backgrounds or multiple sensing zones [7] [8].
- Error Correction and Robustness: AI systems can be trained to recognize and correct for confounding variables such as changing ambient light conditions, pH variations in the sample matrix, and differences between camera devices. For instance, a study on a tear biomarker sensor used a CNN-GRU model to correct for pH and color temperature variations, achieving a determination coefficient (R²) as high as 0.998 [8].
- Smartphone Integration: ML models can be deployed on smartphones, creating powerful and accessible point-of-analysis systems. The smartphone captures the sensor image, and the onboard app processes the color data using a pre-trained algorithm to provide a quantitative result instantly [7] [8].
Essential Research Reagent Solutions
Successful implementation of colorimetric biomarker detection relies on a suite of key reagents and materials. The following toolkit outlines essential components for developing and deploying these sensors.
Table 3: Research Reagent Solutions for Colorimetric Biomarker Detection
| Reagent/Material | Function | Example in Application |
|---|---|---|
| Chromogenic Reagents | Undergoes a specific reaction with the target biomarker to produce a visible color change. | Sinapaldehyde for proline detection [3]; Bicinchoninic acid (BCA) for protein assays [6]. |
| Nanomaterial Probes | Enhances sensitivity and signal intensity through unique optical properties like LSPR. | Gold nanoparticles (AuNPs) that aggregate in the presence of a target, causing a red-to-blue color shift [1]. |
| Sensor Substrates | The solid support onto which the sensing chemistry is immobilized. | Filter paper for paper-based sensors [3]; Polyvinylidene fluoride (PVDF) or Polyethylene terephthalate (PET) films [4]; Flexible polymers for wearable patches [8]. |
| Extraction Solvents | Medium for extracting the target biomarker from a complex sample matrix (e.g., plant tissue, biofluids). | Ethanol for extracting proline from plant leaves [3]. |
| Buffer Solutions | Maintain a constant pH to ensure the colorimetric reaction occurs under optimal and consistent conditions. | Phosphate Buffered Saline (PBS); various buffers for maintaining tear pH in wearable sensors [8]. |
| Standardized Color References | Provides a calibration scale for qualitative visual assessment or for validating digital analysis systems. | Printed color charts with colors corresponding to specific biomarker concentration ranges [3] [4]. |
Colorimetric biosensors for biomarker detection offer a powerful combination of simplicity, cost-effectiveness, and rapidly advancing technological sophistication. The core principle—translating a molecular recognition event into a visible color change—provides an intuitive yet robust foundation for analysis. As demonstrated in the plant health example, these sensors can effectively detect key stress biomarkers like proline, offering researchers and farmers a valuable tool for real-time crop monitoring.
The ongoing integration of advanced nanomaterials, which enhance signal generation, and sophisticated data processing techniques involving multiple color spaces and machine learning, is systematically addressing traditional limitations related to sensitivity and subjective interpretation [7] [8] [1]. The resulting evolution of colorimetric sensors from simple strips to AI-assisted, smartphone-compatible platforms is expanding their utility across diverse fields. For plant science research specifically, these advancements promise the development of more accurate, field-deployable tools for precise health assessment, ultimately contributing to more resilient and productive agricultural systems.
In the face of escalating environmental challenges, the identification of reliable biomarkers for early stress detection in plants has become crucial for agricultural research and crop management. Among various stress-indicative compounds, proline has emerged as a universal biomarker for abiotic and biotic stress response across diverse plant species. This imino acid accumulates rapidly and significantly in plant tissues exposed to adverse conditions, serving not merely as a symptom of stress but as a key component of plant defense mechanisms [9]. The validation of proline as a robust stress indicator has gained particular relevance with the recent development of innovative color-changing sensors that detect proline concentration as a direct measure of plant health status [3]. This review synthesizes current understanding of proline as a universal stress biomarker, examines its performance relative to alternative indicators, and explores its critical role in validating next-generation plant health monitoring technologies.
Proline as a Stress Biomarker: Accumulation Patterns and Comparative Efficacy
Quantitative Evidence of Proline Accumulation Under Stress
Proline demonstrates consistent accumulation patterns across various stress conditions and plant species, with its concentration increasing dramatically—sometimes several hundred-fold compared to unstressed levels in strong accumulators [9]. The table below summarizes experimental data on proline accumulation across different stress conditions and plant species:
Table 1: Proline Accumulation Under Various Abiotic Stress Conditions
| Plant Species | Stress Condition | Proline Accumulation Level | Experimental Context | Reference |
|---|---|---|---|---|
| Tomato (Solanum lycopersicum) | Drought stress | Significant increase | Upregulation of SlOAT8 and SlP5CS1 genes | [10] |
| Tomato (Solanum lycopersicum) | Heat and salt stress | Altered accumulation | Downregulation of specific SlOAT genes | [10] |
| Rice (Oryza sativa) | Salinity stress (300 mM NaCl) | Elevated levels | Co-application with selenium and zinc oxide nanoparticles | [11] |
| Various plant species | Multiple abiotic stresses | Several hundred-fold increase | Comparison of strong vs. weak accumulators | [9] |
Proline Versus Alternative Stress Biomarkers
While plants employ multiple biochemical pathways in stress response, proline demonstrates distinct advantages as a measurable indicator:
Table 2: Proline Compared to Other Plant Stress Biomarkers
| Biomarker | Response Specificity | Detection Methods | Advantages | Limitations |
|---|---|---|---|---|
| Proline | Universal stress biomarker | Colorimetric assays, HPLC, sensor technology | Rapid accumulation, high correlation with stress severity, compatible with field-deployable sensors | Levels vary by species and stress type |
| Reactive Oxygen Species (ROS) | Multiple stress types | Histochemical staining, fluorescence probes | Early stress indicator, signaling molecule | Highly reactive, transient, difficult to quantify |
| Malondialdehyde (MDA) | Oxidative stress specific | Thiobarbituric acid assay | Marker of lipid peroxidation | Requires destructive sampling, complex extraction |
| Abscisic Acid (ABA) | Drought, salinity stress | ELISA, LC-MS | Key stress hormone, signaling role | Complex synthesis pathways, rapid degradation |
| Heat Shock Proteins (HSPs) | Temperature stress | Protein electrophoresis, immunoassays | Specific to thermal stress | Require protein extraction, specialized detection |
The comparative data reveals proline's distinctive combination of quantifiable accumulation, chemical stability, and universal presence across stress conditions, making it particularly suitable for both laboratory analysis and field applications using sensor technology.
Molecular Mechanisms: Proline Metabolism and Signaling Pathways
Biochemical Pathways of Proline Metabolism
Proline participates in a sophisticated metabolic network that spans multiple cellular compartments. The biosynthesis primarily occurs in the cytosol and chloroplasts, while catabolism takes place in the mitochondria [10] [9]. The central enzymes governing proline metabolism include:
- P5CS (Δ1-pyrroline-5-carboxylate synthetase): Rate-limiting enzyme in proline biosynthesis from glutamate, strongly induced by stress through ABA-dependent and ABA-independent mechanisms [9]
- P5CR (Δ1-pyrroline-5-carboxylate reductase): Catalyzes the final step in proline biosynthesis, converting P5C to proline [10]
- OAT (Ornithine aminotransferase): Provides an alternative pathway for proline synthesis from ornithine, with debated but potentially significant roles in stress conditions [10]
- ProDH (Proline dehydrogenase): Mitochondrial enzyme catalyzing the first step of proline catabolism, typically downregulated during stress to promote accumulation [9]
- P5CDH (Δ1-pyrroline-5-carboxylate dehydrogenase): Second enzyme in proline catabolism, converts P5C to glutamate [10]
The dynamic interplay between these enzymes allows plants to precisely regulate proline homeostasis in response to environmental cues, making the metabolic pathway itself a rich source of information about plant stress status.
Proline-ROS Signaling Interface
Emerging research reveals that the functional significance of proline extends beyond osmoprotection to include sophisticated signaling roles, particularly through interactions with reactive oxygen species (ROS). This proline-ROS crosstalk represents a unified mechanism that explains many of proline's multiple functions in plant development and stress defense [9]. The compartmentalization of proline metabolism enables it to function with both antioxidant and pro-oxidant properties, creating a fine-tuning mechanism for redox balance and ROS homeostasis.
Diagram: Proline Metabolism Pathway Under Abiotic Stress. The diagram illustrates how stress conditions trigger transcriptional reprogramming of key proline metabolic enzymes, leading to proline accumulation and subsequent interaction with ROS homeostasis to establish stress tolerance.
Experimental Validation: Methodologies for Proline Detection and Quantification
Established Laboratory Protocols for Proline Measurement
Standardized methodologies have been developed for accurate proline quantification in plant tissues, providing reference points for sensor validation:
Acid-Ninhydrin Colorimetric Assay Protocol [3] [9]
- Sample Preparation: Homogenize 0.5g fresh leaf tissue in 10ml of 3% aqueous sulfosalicylic acid
- Extraction: Centrifuge at 10,000×g for 10 minutes and collect supernatant
- Reaction: Mix 2ml supernatant with 2ml acid-ninhydrin reagent and 2ml glacial acetic acid
- Incubation: Heat at 100°C for 30 minutes in water bath, then terminate reaction in ice bath
- Extraction: Add 4ml toluene to each tube, vortex vigorously for 20 seconds
- Measurement: Measure absorbance of toluene phase at 520nm using spectrophotometer
- Quantification: Calculate proline concentration using L-proline standard curve (0-100μg/ml)
High-Performance Liquid Chromatography (HPLC) Method
- Extraction: Grind frozen plant tissue in liquid nitrogen, extract with 80% ethanol
- Derivatization: React with AccQ-Tag derivatization kit for amino acid analysis
- Separation: Use C18 reverse-phase column with gradient elution (aqueous buffers with acetonitrile)
- Detection: Fluorescence or UV detection at 254nm
- Quantification: Compare retention times and peak areas with proline standards
Molecular Biology Approaches for Proline Pathway Analysis
Gene expression analysis of proline metabolizing genes provides complementary validation:
qRT-PCR Protocol for PMG Expression Analysis [10]
- RNA Extraction: Isolate total RNA from stressed and control tissues using TRIzol reagent
- cDNA Synthesis: Reverse transcribe 1μg RNA using reverse transcriptase and oligo-dT primers
- Primer Design: Design gene-specific primers for target PMGs (P5CS, P5CR, ProDH, P5CDH, OAT)
- Amplification: Perform qPCR with SYBR Green master mix using standard thermal cycling conditions
- Data Analysis: Calculate relative expression using 2^(-ΔΔCt) method with housekeeping genes for normalization
Innovative Sensor Technology: Proline Detection for Plant Health Monitoring
Color-Changing Sensor Technology Based on Proline Detection
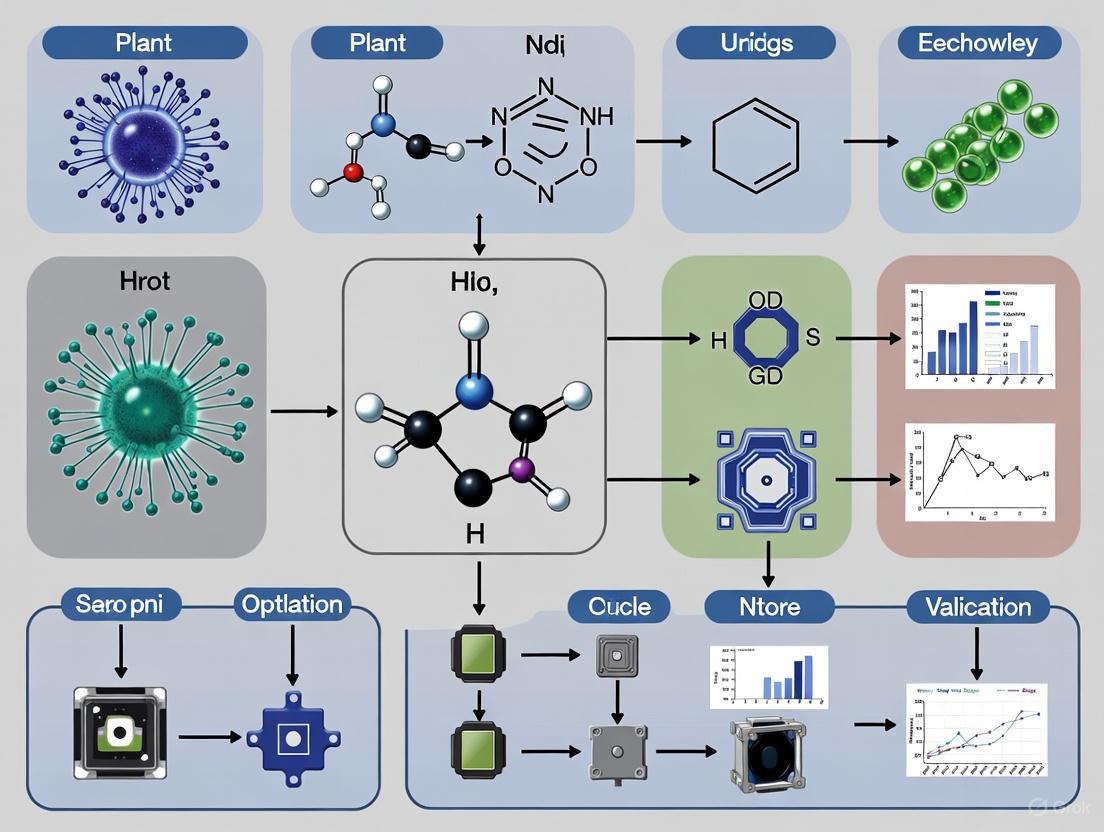
Recent technological advances have leveraged proline as the detection target for innovative plant health monitoring systems. Researchers at Northeastern University have developed color-changing sensors that detect proline concentration as a direct indicator of plant stress [3]. This technology is based on a natural mechanism observed in the nesocodon flower, whose nectar changes color in response to proline concentration through interaction with sinapaldehyde, forming the red pigment nesocodin.
Sensor Fabrication and Application Protocol [3]
- Sensor Design: Paper-based sensors embedded with sinapaldehyde
- Sample Processing: Clip small leaf section (≈10mg) and grind with ethanol to extract proline
- Assay Procedure: Dip sensor into plant extract and incubate for 15 minutes
- Result Interpretation:
- Healthy plants (low proline): Sensor remains pale yellow
- Stressed plants (high proline): Sensor turns bright red
- Quantification Option: Scan sensor and analyze color intensity for quantitative measurement
Comparative Performance of Proline-Based Sensors
The proline detection sensor technology demonstrates significant advantages over alternative plant health monitoring approaches:
Table 3: Performance Comparison of Plant Health Monitoring Technologies
| Technology | Detection Principle | Time Requirement | Cost per Sample | Field Deployment | Stress Detection Stage |
|---|---|---|---|---|---|
| Proline-based color-changing sensors | Chemical reaction with proline | 15-30 minutes | Low (< $1) | Excellent | Early stress phase |
| VOC sensor arrays [12] | Colorimetric response to volatile compounds | Minutes to hours | Moderate | Good | Early to mid stress |
| Wearable VOC sensor patches [12] [13] | Electronic detection of volatiles | Continuous monitoring | High | Moderate | Early stress phase |
| Chlorophyll fluorescence imaging | Photosynthetic efficiency | Seconds to minutes | High | Limited | Mid to late stress |
| Transcriptomic analysis [14] | Gene expression profiling | Days to weeks | Very high | Poor | Very early stress phase |
| Traditional proline assays | Acid-ninhydrin reaction | 2-4 hours | Low to moderate | Limited | Mid stress phase |
The experimental data demonstrates that proline-based sensors provide an optimal balance of speed, cost-effectiveness, and sensitivity for early stress detection, particularly benefiting small-scale farming operations where expensive equipment like drones with specialized cameras is not economically feasible [3].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Essential Research Reagents for Proline Stress Response Studies
| Reagent/Material | Function | Application Context | Key Considerations |
|---|---|---|---|
| Sinapaldehyde | Sensor substrate | Color-changing sensor development | Reacts with proline to form red nesocodin pigment |
| Acid-Ninhydrin Reagent | Colorimetric detection | Traditional proline quantification | Specific for proline, forms red chromophore |
| Sulfosalicylic Acid (3%) | Protein precipitation | Proline extraction from plant tissue | Preserves proline while precipitating interfering proteins |
| L-Proline Standard | Calibration reference | Standard curve preparation | Enables quantitative analysis in both sensors and assays |
| TRIzol Reagent | RNA isolation | Gene expression studies of PMGs | Maintains RNA integrity for transcript analysis of proline genes |
| SYBR Green Master Mix | DNA detection | qRT-PCR of proline metabolizing genes | Enables quantification of P5CS, P5CR, ProDH expression |
| Nanoparticles (Se-NPs, ZnO-NPs) [11] | Stress amelioration | Experimental stress modulation | Can influence proline accumulation patterns |
| Antibodies for Proline Enzymes | Protein detection | Western blot analysis | Verifies protein level changes of P5CS, ProDH |
The comprehensive analysis of experimental evidence confirms proline as a universal, reliable, and technically accessible biomarker for plant stress assessment. Its consistent accumulation patterns across diverse stress conditions, well-characterized metabolic pathways, and compatibility with both laboratory assays and field-deployable sensor technologies position proline as an ideal indicator for plant health monitoring. The recent development of color-changing sensors based on proline detection represents a significant advancement in making stress monitoring accessible and practical, particularly for smaller farming operations and resource-limited settings [3].
Future research directions should focus on expanding validation of proline-based sensors across a wider range of crop species, optimizing sensor design for enhanced sensitivity and specificity, and integrating proline detection with complementary biomarkers to create multidimensional plant health assessment systems. The integration of proline monitoring with other emerging technologies—such as nanoparticle-mediated stress amelioration [15] [11] and multi-omics approaches [14] [16]—promises to further enhance our understanding of plant stress responses and develop more resilient crop varieties for sustainable agriculture in challenging environmental conditions.
In the quest for sustainable agriculture, monitoring plant health efficiently and accurately remains a significant challenge. Current methods often rely on expensive laboratory analyses or complex equipment, creating barriers to accessibility, particularly for smaller farming operations and in the developing world [3]. A promising solution is emerging from the field of bio-inspired design, where nature's own mechanisms are leveraged to create innovative technologies. The Nesocodon flower, a species native to Mauritius, has provided the foundational blueprint for a novel class of color-changing sensors that detect plant stress in real time [3].
This flower possesses a unique natural adaptation: its nectar changes color in response to the presence of a small molecule called proline [3]. Proline is a universal biomarker that plants produce in heightened concentrations when under stress from factors such as drought, extreme temperatures, nutrient deficiency, or soil pollution [3]. Researchers at Northeastern University have successfully mimicked this natural color-change reaction by creating paper-based sensors embedded with sinapaldehyde, the same molecule that reacts with proline in the Nesocodon flower [3]. When these sensors are exposed to leaf extracts from a stressed plant, a chemical reaction occurs, producing a red pigment called nesocodin and causing the sensor to change from pale yellow to bright red [3]. This direct translation of a biological phenomenon into an analytical tool exemplifies the power of biomimicry, offering a simple, rapid, and low-cost method for assessing plant health that stands in contrast to more complex and expensive technological alternatives.
Performance Comparison: Nesocodon-Inspired Sensors vs. Alternative Technologies
To objectively evaluate the Nesocodon-inspired sensor, its performance must be compared with other modern approaches to plant health monitoring. The table below provides a structured comparison based on sensing methodology, target analyte, and key performance parameters.
Table 1: Performance Comparison of Plant Health Monitoring Technologies
| Technology | Sensing Methodology | Target Analyte/Parameter | Key Performance Characteristics | Relative Cost | Best-Suited Application |
|---|---|---|---|---|---|
| Nesocodon-Inspired Sensor [3] | Colorimetric (Proline Reaction) | Proline (Universal Stress Biomarker) | Assay Time: ~15 minutesOutput: Qualitative (Color Change) to Quantitative (Digital Analysis)Equipment Needs: Minimal | Very Low | Small-scale farms, family gardens, research fieldwork |
| RedAlert Living Sensors [17] | In-plant Genetic Biosensor | Soil Nitrogen Levels | Assay Time: Continuous, real-timeOutput: Visual color change in entire plant (green to red)Equipment Needs: None for visual assessment; cameras for large-scale use | Medium (Development & Seeds) | Large-scale fields (with camera-equipped tractors), hydroponics, home gardens |
| Wearable/Implantable Electrochemical Sensors [18] [13] | Electrochemical Transduction | Agrochemicals, phytohormones, ions, stress biomarkers | Assay Time: Continuous, real-timeOutput: Digital electronic signalsEquipment Needs: Sensor reader/data logger, potentially connected to IoT platforms | High | High-precision agriculture, advanced phytochemical research |
| Traditional Laboratory Methods [3] [19] | Laboratory Analysis | Varies | Assay Time: Days to weeksOutput: Quantitative lab resultsEquipment Needs: Expensive lab equipment, skilled technicians | High | Research institutions, large agribusinesses |
The Nesocodon-inspired sensor occupies a unique niche, offering a balance of simplicity, speed, and low cost. Unlike the RedAlert system, which requires genetically modified plants, the Nesocodon sensor is an external tool that can be used on any existing plant [3] [17]. While wearable electrochemical sensors provide continuous data, they are more complex and expensive, making the Nesocodon sensor a more accessible option for one-off or periodic stress assessments [18] [13]. A key advantage is its democratizing potential; as Northeastern researcher Dan Wilson notes, it is designed for "family farmers and smaller farming operations who are in the developing world and may not have resources to access drones or some of the more expensive specialized equipment" [3].
Experimental Protocol: From Leaf Sample to Stress Assessment
The experimental workflow for using the Nesocodon-inspired sensor is straightforward and can be performed in the field with minimal equipment. The following diagram and detailed protocol outline the key steps from sample collection to data interpretation.
Diagram 1: Experimental workflow for Nesocodon-inspired sensor.
Detailed Step-by-Step Methodology
- Sample Collection: A small piece of leaf (approximately 1-2 cm²) is clipped from the plant to be tested [3].
- Sample Preparation: The leaf sample is mechanically ground into smaller pieces using a mortar and pestle or a similar tool. Ethanol is added to the ground tissue to draw out and dissolve the proline and other soluble components from the plant cells [3].
- Sensor Incubation: A paper-based sensor strip, which has been pre-embedded with the molecule sinapaldehyde, is dipped into the liquid extract [3].
- Color Development and Data Acquisition: The sensor is left to incubate for approximately 15 minutes. During this time, a chemical reaction occurs between the sinapaldehyde in the sensor and any proline present in the extract. This reaction produces nesocodin, a red pigment, leading to a color change on the sensor strip [3].
- The color change can be assessed in two ways:
- Qualitative Assessment: The color is visually compared to a reference chart. A pale yellow indicates a healthy plant (low proline), while shades of orange to bright red indicate increasing levels of stress (high proline) [3].
- Quantitative Assessment: The sensor is scanned or photographed, and the image is converted into digital color data (e.g., RGB, CIELAB values) [4]. This data can be analyzed using various chemometric methods to obtain a more precise, numerical value correlating to the proline concentration [3] [4].
- The color change can be assessed in two ways:
The Science Behind the Sensor: Proline Signaling Pathway
The fundamental biology that makes the Nesocodon-inspired sensor possible is the conserved role of proline as a key chemical messenger in plant stress response. The following diagram maps this signaling pathway.
Diagram 2: Plant stress-induced proline signaling pathway.
The signaling pathway begins when a plant experiences environmental stress. This stimulus is perceived by the root system and translated into a biochemical signal within the plant's cells [3]. A key response is the upregulation of proline biosynthesis. Proline, an amino acid, acts as a compatible osmolyte, helping to maintain cellular water balance and protect protein structures under stress conditions like drought and high salinity. The synthesized proline is then transported throughout the plant via its vascular system, leading to accumulation in aerial tissues such as leaves [3]. The Nesocodon-inspired sensor is designed to detect this accumulated proline. The sensor contains sinapaldehyde, which mimics the natural chemical environment of the Nesocodon flower's nectar. When proline and sinapaldehyde interact, they form a red pigment called nesocodin through a specific chemical reaction, providing a direct visual representation of the plant's internal stress status [3].
The Scientist's Toolkit: Key Reagents and Materials
Successful implementation of this bio-inspired sensing approach requires a specific set of reagents and materials. The following table details the essential components and their functions within the experimental protocol.
Table 2: Research Reagent Solutions for Nesocodon-Inspired Sensing
| Reagent/Material | Function in the Experiment | Specifications & Notes |
|---|---|---|
| Sinapaldehyde-Embedded Sensor Strip | The core sensing element; contains the reagent that reacts with proline to produce a color change. | Typically paper-based [3]. Research is exploring biodegradable substrates to improve sustainability [3]. |
| Ethanol Solvent | Used to extract proline from the ground leaf tissue into a liquid solution. | Acts as a carrier to bring proline into contact with the sensor [3]. |
| Color Standard Chart | Provides a visual reference for semi-quantitative assessment of proline levels based on sensor color. | Correlates specific hues (yellow, orange, red) with increasing stress levels [3]. |
| Digital Color Analysis Tools | Enables quantitative data extraction from the sensor. | Can include a flatbed scanner or digital camera for image capture, and software for converting images to color models like RGB, HSV, or CIELAB for analysis [4] [20]. |
| Proline Standard Solutions | Used for calibration and validation of the sensor's performance. | Solutions of known proline concentration are used to create a standard curve for quantitative analysis [4]. |
The Nesocodon-inspired sensor demonstrates the profound impact of bio-inspired design on agricultural technology. By translating a natural signaling mechanism into a practical diagnostic tool, this approach provides a rapid, low-cost, and accessible method for detecting plant stress. While it may not offer the continuous data stream of electrochemical sensors or the specificity of complex lab assays, its simplicity and effectiveness fill a critical gap for small-scale and resource-limited agricultural settings. The ongoing research into improving these sensors, including developing biodegradable substrates and refining quantitative digital analysis, promises to further enhance their utility and sustainability. As a validated tool for plant health assessment, this technology underscores the immense potential of looking to nature's own solutions to address modern agricultural challenges.
In modern agriculture and plant research, the early and accurate detection of stress is paramount for preventing yield loss and optimizing resource use. Among the most promising developments are color-changing sensors, which offer a pathway to rapid, non-destructive, and real-time plant health monitoring. This guide objectively compares the performance of emerging sensor formats, with a specific focus on validating their performance for plant health assessment. The transition from conventional laboratory techniques to innovative platforms like paper-based strips and biodegradable sensors represents a significant shift towards precision agriculture. These technologies are particularly valuable for their potential to provide actionable data to researchers and plant scientists, enabling timely interventions for conditions such as drought stress and pathogenic infection.
The performance of these sensors is often quantified using key analytical parameters including limit of detection (LOD), limit of quantification (LOQ), dynamic range, reproducibility, and response time. Furthermore, the integration of these sensors with digital tools like smartphones for data acquisition and analysis is creating a new paradigm of integrated, smart monitoring systems [21] [22]. This guide provides a structured comparison of these technologies, summarizes experimental data in accessible tables, and details the protocols necessary for their validation in a research context.
Performance Comparison of Sensor Formats
The selection of a sensor platform depends heavily on the specific application, target analyte, and required performance characteristics. The table below provides a quantitative comparison of various sensor formats based on key metrics, drawing from recent experimental studies.
Table 1: Performance Comparison of Different Sensor Formats for Plant Health and Environmental Monitoring
| Sensor Format | Primary Detection Method | Target Analytes/Application | Limit of Detection (LOD) | Response Time | Key Advantages | Key Limitations |
|---|---|---|---|---|---|---|
| Smartphone-Paper Strip [21] | Colorimetric | Cu(II), Cr(VI), Zn(II), Mn(II) ions | 0.26-0.79 mg L⁻¹ | Rapid | Low cost, portability, simultaneous multi-analyte detection | Limited sensitivity compared to lab instruments |
| Colorimetric Sensor Array [22] | Colorimetric with pattern recognition | OH⁻, CO₃²⁻, PO₄³⁻, NH₃, organic amines | N/A (Differentiates 0.001–1.0 mol L⁻¹) | 15 sec - 7 min | Versatile, identifies multiple similar analytes | Requires data analysis (LDA, HCA) |
| High-Density Plant Sensor [23] | Acoustic Emission, Stem Diameter | Early drought stress in tomatoes | N/A (Reacts within 24h of irrigation stop) | Continuous monitoring | Pre-visual stress detection, high temporal resolution | Requires fixed installation, higher cost |
| Electro-Optical RS [24] | Multispectral/Hyperspectral | Biotic/Abiotic plant stress, chlorophyll content | N/A | Varies by platform | Non-contact, canopy-level spatial data | Affected by atmosphere/light, complex data |
| Nanoparticle-Based CCS [2] | Colorimetric with nanomaterials | Various biomarkers, pathogens | Up to fM concentrations | Fast (mins) | High sensitivity, stability | Potential nanomaterial toxicity, cost |
Experimental Protocols for Sensor Validation
To ensure the reliability and accuracy of color-changing sensors, rigorous experimental validation is essential. The following protocols outline the key methodologies for evaluating paper-based strips and colorimetric sensor arrays.
Protocol for Smartphone-Integrated Paper Sensor Strips
This protocol is adapted from research on detecting heavy metal ions in water, a methodology transferable to monitoring plant nutrient status or soil/water contaminants [21].
- Sensor Fabrication: Cut filter paper (e.g., Whatman chromatography paper) into strips. Impregnate the detection zones with specific chromogenic reagents (e.g., dithizone for metal ions) that undergo a distinct color change upon complexation with the target analyte. Allow the strips to dry completely.
- Sample Preparation: For plant health applications, liquid samples can include soil water extracts, xylem sap, or leaf extracts. Filter the samples to remove particulate matter.
- Detection and Imaging:
- Apply a fixed volume (e.g., 50-100 µL) of the sample to the detection zone of the paper strip.
- Allow the colorimetric reaction to proceed for a predetermined time (e.g., 2-5 minutes).
- Place the strip in a standardized imaging box with uniform LED lighting to minimize ambient light interference.
- Capture an image of the strip using a smartphone camera with fixed focus, flash disabled, and consistent settings.
- Data Analysis:
- Transfer the image to a computer or use an on-device app (e.g., PhotoMetrix PRO) for analysis.
- Extract the Red, Green, and Blue (RGB) intensity values from the detection zone.
- Calculate the Euclidean distance or the change in a specific RGB channel relative to a blank (control) strip.
- Quantify the analyte concentration by interpolating the RGB values against a calibration curve prepared with standard solutions.
Protocol for Colorimetric Sensor Array Analysis
This protocol, used for differentiating inorganic and organic bases, can be adapted to detect volatile organic compounds emitted by plants under stress [22].
- Array Fabrication: Prepare solutions of various color-sensitive dyes (e.g., Fuchsine, Giemsa, Thionine, CoCl₂). Spot these dyes (e.g., 150 µL each) into individual wells of a 96-well plate to create the sensor array.
- Analyte Exposure: Introduce the analyte (e.g., 150 µL of a volatile compound solution or a controlled headspace) to the array. For plant studies, this could involve enclosing a leaf in a chamber and sampling the air.
- Image Acquisition: Use a smartphone or digital camera to capture images of the array at multiple time points (e.g., 15 seconds, 30 seconds, 1 minute, 5 minutes) after exposure.
- Data Processing and Pattern Recognition:
- For each dye spot in the image, extract the RGB values before and after exposure.
- Calculate the change in RGB values (ΔR, ΔG, ΔB) for each dye.
- Construct a feature vector for each analyte based on the ΔRGB values from all dyes in the array.
- Use multivariate statistical analyses, such as Linear Discriminant Analysis (LDA) or Hierarchical Clustering Analysis (HCA), to identify unique fingerprint-like patterns for different analytes or concentrations.
Signaling Pathways and Experimental Workflows
The development and application of color-changing sensors follow a logical pathway from conception to data interpretation. The diagram below illustrates this integrated workflow for plant health assessment.
Diagram 1: Sensor Validation Workflow for Plant Health Assessment.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful experimentation with color-changing sensors requires a specific set of reagents and materials. The following table details the key components for fabricating and validating these diagnostic platforms.
Table 2: Essential Research Reagents and Materials for Sensor Development
| Reagent/Material | Function/Application | Examples & Notes |
|---|---|---|
| Chromatography Paper | Hydrophilic substrate for paper-based sensors; wicks fluid via capillary action. | Whatman brand; uniform thickness & pore size (e.g., 11 µm) for consistent flow [25]. |
| Nitrocellulose Membrane | Substrate with high protein-binding capacity; used in lateral flow assays. | Allows covalent immobilization of biomolecules; prevents reagent diffusion [25]. |
| Chromogenic Reagents | React with target analyte to produce a measurable color change. | Dithizone for metals; pH indicators; enzyme substrates (e.g., for glucose oxidase) [21] [2]. |
| Color-Sensitive Dyes | Elements of a sensor array; respond to a range of chemical interactions. | Fuchsine, Thionine, Giemsa, CoCl₂; provide a unique fingerprint for pattern recognition [22]. |
| Smartphone & Imaging App | Portable device for image capture and initial data processing. | Requires a consistent setup (light, distance); apps like PhotoMetrix PRO extract RGB values [21] [22]. |
| Nanoparticles | Transducers to enhance signal, immobilize enzymes, and improve sensitivity. | Gold nanoparticles, carbon nanotubes, graphene oxide; can drastically lower LOD [2]. |
| Standard Analytic Solutions | Used for calibrating sensors and determining LOD/LOQ. | Prepared at known concentrations (e.g., 0.001–1.0 mol L⁻¹) to generate a calibration curve [22]. |
| Pattern Recognition Software | Analyzes complex data from sensor arrays for analyte identification. | IBM SPSS, MATLAB with Classification Toolbox; used for LDA and HCA [22]. |
Traditional methods for monitoring plant health, such as laboratory-based analyses, drone surveys with specialized cameras, and handheld chlorophyll meters, are often expensive, time-consuming, and inaccessible for small-scale operations and field research [3] [26]. These methods can cost hundreds to thousands of dollars per analysis and require days for results, creating a significant accessibility gap [3]. Consequently, smaller research groups and agricultural communities in resource-limited settings are often excluded from leveraging data-driven plant health insights.
Color-changing sensors are emerging as a powerful solution to this challenge, offering rapid, low-cost, and portable diagnostic tools. This guide provides an objective comparison of these sensor technologies, detailing their experimental validation and positioning them within the broader context of plant health assessment research.
Comparative Analysis of Monitoring Technologies
The table below summarizes the key performance characteristics of current and emerging plant health monitoring technologies, highlighting the position of color-changing sensors.
Table 1: Performance Comparison of Plant Health Monitoring Technologies
| Technology Type | Key Measured Parameter(s) | Approx. Cost | Analysis Time | Key Strengths | Primary Limitations |
|---|---|---|---|---|---|
| Lab-based Analysis | Chemical biomarkers (e.g., proline) | Hundreds to thousands of dollars [3] | Days to a week [3] | High accuracy and precision | Cost, time, requires lab access |
| Handheld Chlorophyll Meters (e.g., SPAD) | Chlorophyll Content Index (CCI) | >$1000 [26] | Minutes | Non-destructive, fast | High unit cost, provides proxy data only |
| Multispectral Sensors (e.g., AMS AS7265x) | Spectral reflectance across multiple bands | ~$70 (AS7265x board) [26] | Minutes (requires model fitting) | Good accuracy on uniform leaves (R²: 0.95) [26] | Performance drops on textured/narrow leaves |
| Hyperspectral Imaging Systems | Full spectral signature (Reflectance & Fluorescence) | High (research-grade) | Fast imaging, complex processing | Early stress detection (>90% accuracy) [27] | Very high cost, bulky, complex data analysis |
| Wearable Plant Sensors | Microclimate, VOCs, sap flow, etc. [28] | Varies (low-cost materials possible) | Continuous, real-time | Real-time, continuous monitoring [28] | Challenges in durability, power, scalability [28] |
| Color-Changing Paper Sensors | Biomarker concentration (e.g., proline) [3] | Very low (paper-based) | ~15 minutes [3] | Extremely low cost, simple visual readout, accessible | Destructive (requires leaf sample), semi-quantitative |
Experimental Validation of Color-Changing Sensors
Fundamental Signaling Mechanism and Workflow
Color-changing sensors for plant health function by detecting universal stress biomarkers. A primary validated target is proline, an amino acid whose concentration increases significantly in plants under abiotic stresses like drought, salinity, or heavy metal exposure [3]. The core detection mechanism is borrowed from a natural system: the nectar of the Nesocodon flower, which changes color due to a reaction between proline and a small molecule called sinapaldehyde, producing a red pigment called nesocodin [3]. Researchers have replicated this biological reaction in a paper-based sensor impregnated with sinapaldehyde.
The following diagram illustrates the experimental workflow for using these sensors, from sample preparation to result interpretation:
Detailed Experimental Protocol
Based on the methodology established by Northeastern University researchers, the following protocol can be used to validate color-changing sensor performance [3].
Objective: To qualitatively and quantitatively assess plant health status by detecting proline levels in leaf samples using a paper-based colorimetric sensor.
Materials & Reagents:
- Colorimetric Sensors: Paper-based sensors embedded with sinapaldehyde [3].
- Plant Material: Leaf samples from both control and stress-treated plants (e.g., cabbage, kale, broccoli).
- Extraction Solvent: Ethanol (e.g., 70% aqueous ethanol).
- Equipment: Microtube pestle or grinder, timer, flatbed scanner or smartphone for digitization, image analysis software (e.g., ImageJ).
Procedure:
- Sample Collection: Clip a small, standardized piece (e.g., 1 cm²) from a leaf of the test plant.
- Homogenization & Extraction: Place the leaf piece in a microtube and grind it thoroughly with a pestle. Add 1 mL of ethanol to extract proline and other soluble compounds. Vortex or shake vigorously for 1-2 minutes.
- Sensor Incubation: Dip the sinapaldehyde-embedded paper sensor into the leaf extract supernatant. Ensure the sensor is fully wetted.
- Color Development: Allow the sensor to react for a standardized period (e.g., 15 minutes). Observe the color change.
- Data Acquisition & Analysis:
- Qualitative Assessment: Visually compare the sensor's color against a reference chart (pale yellow for healthy, orange for moderate stress, bright red for high stress).
- Quantitative Assessment: Scan the sensor under consistent lighting conditions. Use image analysis software to measure the Red, Green, Blue (RGB) values or convert to Hue, Saturation, Lightness (HSL) values. The intensity of the red channel (or a calculated redness index) correlates with proline concentration [3] [29].
The Researcher's Toolkit: Essential Reagent Solutions
For researchers aiming to develop or deploy color-changing sensors for plant health assessment, the following reagents and materials are essential.
Table 2: Key Research Reagent Solutions for Colorimetric Sensing
| Reagent/Material | Function in Experiment | Research Context & Considerations |
|---|---|---|
| Sinapaldehyde | The core reactive molecule that binds with proline to form the red pigment nesocodin [3]. | Sourcing high-purity sinapaldehyde is critical for consistent sensor performance. Stability in storage must be considered. |
| Paper Substrate (e.g., Filter Paper) | The solid support matrix for the sensor, holding the reagent and wicking the sample [3] [30]. | Porosity, thickness, and purity of the paper affect fluid flow, reaction uniformity, and background signal. |
| Proline Standard Solutions | Used for creating a calibration curve to convert sensor color intensity into quantitative proline concentration. | Enables sensor validation and ensures measurements are within the dynamic range of the colorimetric reaction. |
| Organic Solvents (e.g., Ethanol) | Used to extract proline from destructively sampled plant tissue [3]. | Ethanol effectively solubilizes proline while minimizing the extraction of interfering compounds. |
| Machine Learning Algorithms (e.g., RFR, SVR, MLP) | Used to analyze complex color data from sensors, improving quantification accuracy by interpreting multiple color space values (RGB, HSL, CIELAB) [29]. | Mitigates subjectivity and enhances the precision of visual readouts, moving beyond simple RGB analysis. |
Integrated Framework for Accessible Plant Health Assessment
The following diagram places color-changing sensors within a broader, integrated framework for accessible plant health monitoring, showing their relationship with other complementary technologies.
This framework demonstrates that color-changing sensors serve as a foundational, low-cost diagnostic tool. Their value is significantly enhanced when their specific biochemical data is fused with broader contextual data from other accessible technologies, such as microclimate readings from wearable sensors [28] or morphological analysis from smartphone cameras [31]. Machine learning models act as the unifying layer, integrating these diverse data streams to provide robust, validated, and actionable health assessments, effectively bridging the accessibility gap.
Implementing Sensor Technology: From Lab to Field Applications
This guide provides a standardized protocol for leaf sampling and metabolite extraction, a critical process in the development and validation of novel plant health diagnostics, such as color-changing sensors. Consistent and reproducible sample preparation is the foundation for generating reliable experimental data. The extraction methodologies detailed here are designed to efficiently isolate key biomarkers, including proline, a universal stress indicator, allowing for the direct benchmarking of new sensor technologies against established analytical techniques like Liquid Chromatography-Mass Spectrometry (LC-MS) [3] [32]. By comparing sensor performance against gold-standard methods using identical sample extracts, researchers can objectively quantify the accuracy, sensitivity, and practical utility of emerging diagnostic platforms.
Leaf Sampling and Preparation
Proper initial handling of plant material is essential to preserve the in-vivo metabolic state of the leaf and prevent degradation of target analytes.
Sampling Protocol
- Collection: Use clean, sterilized tools to collect leaf samples. For stress biomarker analysis, it is crucial to include control plants (unstressed) and treated plants (subjected to specific stressors like drought, salinity, or pathogens)[ccitation:2] [31].
- Immediate Processing: Place collected leaf samples immediately on dry ice or in liquid nitrogen to flash-freeze them. This rapid freezing halts all metabolic activity, preserving the biochemical profile at the moment of sampling [33].
- Transport: Transfer frozen samples to the laboratory in a cryogenic shipping container or on dry ice to maintain a frozen state [33].
Lyophilization and Homogenization
- Lyophilization: Transfer samples to a freeze-dryer (lyophilizer) for approximately 72 hours or until completely dry and brittle [33]. Lyophilization removes water through sublimation, preventing the degradation that can occur with air-drying and enhancing the stability of metabolites.
- Grinding: Use a laboratory grinder or a TissueLyser with grinding beads to pulverize the lyophilized leaf material into a fine, homogeneous powder [33]. Homogenization is critical for ensuring representative sub-sampling and consistent extraction efficiency.
- Storage: Store the resulting powdered plant material in tightly sealed containers, such as Falcon tubes, in a dry environment to prevent moisture absorption [33].
Metabolite Extraction Methodologies
Different analytical endpoints require tailored extraction protocols. Below are two standardized methods for metabolite extraction.
Protocol A: Ethanol-Water Extraction for LC-MS Analysis
This protocol is optimized for broad-spectrum metabolite profiling using LC-MS, a high-precision reference method [33].
Protocol B: Ethanol Extraction for Colorimetric Sensor Validation
This simpler, faster protocol is suitable for field-compatible sensor testing, specifically targeting soluble biomarkers like proline [3].
- Sample Preparation: Clip a small piece of the test leaf (avoiding midribs) and grind it into smaller pieces using a mortar and pestle or a micro-homogenizer [3].
- Extraction: Add a sufficient volume of ethanol to the ground leaf material to draw out the internal fluids and biomarkers [3]. The exact volume can be adjusted based on the scale of the assay.
- Analysis: The resulting ethanol extract can be used directly for dipping paper-based sensors or for subsequent chemical analysis [3].
Table 1: Comparison of Metabolite Extraction Protocols.
| Parameter | Protocol A: LC-MS | Protocol B: Sensor Validation |
|---|---|---|
| Primary Use | Broad metabolite profiling; Gold-standard validation [33] | Targeted biomarker analysis; Field-deployable sensor testing [3] |
| Sample Input | 25 mg lyophilized powder [33] | Small leaf section (fresh tissue) [3] |
| Extraction Solvent | Dimethyl Sulfoxide (DMSO) [33] | Ethanol [3] |
| Key Steps | Incubation, multiple homogenizations, centrifugation [33] | Grinding and solvent extraction [3] |
| Throughput | Medium (batch processing) | High (rapid, single-sample focus) |
| Compatibility | LC-MS systems [33] | Colorimetric sensors, field assays [3] |
Analytical Techniques: From Laboratory Gold-Standards to Emerging Sensors
The extracted metabolites can be analyzed using various technologies, each with distinct capabilities.
Established Laboratory Techniques
- Liquid Chromatography-Mass Spectrometry (LC-MS): Considered the gold-standard for metabolite identification and quantification. It offers high sensitivity, specificity, and the ability to profile hundreds of compounds simultaneously from a single extract, as used in Protocol A [33].
- Leaf Extract Analysis (Sap Testing): This laboratory technique involves analyzing the pure cellular fluids of the plant for over 20 nutritional analytes. It provides a real-time snapshot of the plant's nutritional status and is used for precise fertilizer recommendations [34].
Emerging Color-Changing Sensor Technology
A novel approach involves paper-based sensors embedded with sinapaldehyde that change color in the presence of the stress biomarker proline [3].
- Mechanism: The sensor leverages a natural reaction inspired by the Nesocodon flower. In the presence of high proline concentrations, sinapaldehyde reacts to form a red pigment called nesocodin [3].
- Procedure: A leaf extract prepared via Protocol B is applied to the sensor. A color change from pale yellow to bright red indicates high proline levels and, therefore, plant stress. The intensity can be assessed qualitatively or quantified with a scanner [3].
Table 2: Comparison of Plant Health Analysis Techniques.
| Technique | Principle | Key Biomarker | Throughput | Cost & Accessibility | Primary Application |
|---|---|---|---|---|---|
| LC-MS | Separation and mass-based identification of metabolites [33] | Multiple metabolites | Low to Medium | High; specialized labs [32] | Gold-standard validation; untargeted discovery [33] |
| Leaf Sap Analysis | Precision analysis of plant fluids for minerals [34] | Nutrients (N, K, Mg, etc.) & toxins | Medium | Medium; commercial labs [34] | Nutritional status and fertilizer guidance [34] |
| Color-Changing Sensor | Colorimetric reaction with sinapaldehyde [3] | Proline | High (minutes) [3] | Low; designed for field use [3] | Rapid stress screening; small-scale farming [3] |
| Hyperspectral Imaging | Analysis of reflected light across wavelengths [32] | Spectral signatures | Varies | Very High; research & large farms [32] | Pre-symptomatic detection; large-scale monitoring [32] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Leaf Analysis.
| Item | Function/Application |
|---|---|
| Dimethyl Sulfoxide (DMSO) | A polar aprotic solvent used in Protocol A for efficient extraction of a wide range of polar and non-polar metabolites from plant tissue for LC-MS analysis [33]. |
| Acetonitrile (CH3CN) | A common organic solvent used in mobile phases for LC-MS. In Protocol A, it is mixed with water to reconstitute the dried extract for injection into the LC-MS system [33]. |
| Ethanol | A readily available solvent used in Protocol B for rapid extraction of soluble biomarkers like proline, making it suitable for field-compatible sensor validation [3]. |
| Sinapaldehyde-Embedded Sensors | Paper-based sensors functionalized with sinapaldehyde. They undergo a specific colorimetric reaction in the presence of the stress biomarker proline, enabling visual health assessment [3]. |
| Grinding Beads (Stainless Steel) | Used with homogenizers like the TissueLyser to mechanically disrupt lyophilized plant tissue, creating a fine powder for uniform and efficient extraction [33]. |
| LC-MS System | The instrumental platform combining liquid chromatography for separation and mass spectrometry for detection. Serves as the high-accuracy reference method for validating sensor performance [33]. |
Robust protocols for leaf sampling and extraction are the bedrock of reliable plant health assessment. The detailed methodologies for LC-MS and sensor validation provide researchers with a clear framework for generating comparable and reproducible data. By using standardized extracts to benchmark the performance of innovative, low-cost tools like color-changing sensors against established laboratory techniques, the scientific community can accelerate the development of accessible diagnostics. This comparative approach is vital for bridging the gap between high-precision laboratory analysis and practical, field-deployable solutions, ultimately empowering a broader range of users to monitor and respond to plant stress effectively.
Qualitative vs. Quantitative Data Interpretation
In the evolving field of plant science, the accurate assessment of plant health is paramount for both research and agricultural application. The emergence of novel diagnostic technologies, particularly color-changing sensors, has sparked a critical debate regarding the most effective methodology for data interpretation. These sensors, which respond to plant stress biomarkers such as proline, provide a unique bridge between direct visual cues and quantifiable data metrics [3]. This guide objectively compares qualitative and quantitative frameworks for interpreting data from these sensors, framing the analysis within the broader thesis that an integrated approach is essential for validating sensor performance and maximizing their research utility. The comparison is grounded in experimental data and designed to meet the rigorous demands of researchers and scientists engaged in method validation and technology assessment.
Understanding the Core Methodologies
The distinction between qualitative and quantitative interpretation lies in the form of the output and the underlying analytical processes.
Qualitative Interpretation is a subjective, expert-driven process that translates sensory observations—such as the color change of a sensor—into descriptive health assessments. In the context of color-changing sensors, this involves visually matching the sensor's hue against a reference scale to determine a stress status, such as "healthy" (pale yellow) or "highly stressed" (bright red) [3]. This method is akin to well-established ecological assessments, where trained evaluators use defined indicators to reliably classify the condition of a system without relying on numerical data [35].
Quantitative Interpretation, in contrast, is an objective, data-driven process that converts the sensor's response into numerical values. For the color-changing sensors, this is achieved by scanning the sensor and assigning a numerical value associated with the intensity of the color change, which correlates to proline concentration [3]. This approach aligns with advanced plant phenotyping methods that leverage mathematical descriptors to quantify morphology [36] and deep learning models that assign specific health scores based on image data [31].
Table 1: Fundamental Characteristics of Interpretation Methods
| Feature | Qualitative Interpretation | Quantitative Interpretation |
|---|---|---|
| Data Form | Descriptive, categorical | Numerical, continuous |
| Primary Output | Color category (e.g., yellow, orange, red) | Proline concentration, normalized color values |
| Analysis Basis | Expert judgment, visual comparison | Instrumental measurement, statistical analysis |
| Inherent Nature | Subjective | Objective |
| Key Advantage | Rapid, intuitive, accessible | Statistically robust, suitable for trend analysis |
Direct Performance Comparison
To objectively evaluate the two interpretation methods, their performance is compared across critical metrics relevant to research and development. The following table summarizes this comparison based on experimental protocols and published data.
Table 2: Experimental Performance Comparison of Interpretation Methods
| Performance Metric | Qualitative Interpretation | Quantitative Interpretation |
|---|---|---|
| Speed of Analysis | Results in "a couple of minutes" [3] | Requires additional steps (scanning, analysis); slower |
| Measurement Precision | Low (categorical scale: yellow, orange, red) [3] | High (dose-dependent numerical output) [3] |
| Required Expertise | Minimal; training on color scale sufficient | Technical proficiency for data analysis equipment |
| Cost & Accessibility | Low; no specialized equipment needed [3] | Higher; requires scanning hardware and analysis software |
| Scalability | Suitable for small-scale, spot-checking | Suitable for large-scale studies and high-throughput workflows |
| Data Richness | Limited to predefined categories | Enables tracking of subtle, continuous changes over time |
The experimental data reveals a clear trade-off. Qualitative methods excel in accessibility and speed, making them ideal for rapid field assessments and smaller farming operations [3]. Quantitative methods provide the precision and rich data required for rigorous scientific validation, enabling researchers to track subtle physiological changes and perform robust statistical analyses [31].
Experimental Protocols for Validation
A critical step in validating color-changing sensor performance involves direct comparative experiments. The following protocols outline how to generate data for comparisons like those in Table 2.
Protocol for Qualitative Assessment
This protocol is designed to simulate real-world conditions where a quick, visual assessment is required.
- Sensor Application and Stress Induction: Apply the sinapaldehyde-embedded paper-based sensors to leaves of test plants (e.g., cabbage, kale) [3]. Divide plants into groups and expose them to controlled stressors (drought, soil metals, excessive light) to induce a range of proline production.
- Sample Collection: After a predetermined stress period, clip a small piece of the leaf from each test plant.
- Sample Processing: Grind the leaf tissue and add ethanol to draw out proline. Dip the sensors into the liquid extract [3].
- Visual Evaluation: After 15 minutes, have multiple independent evaluators assess the color of each sensor. Each evaluator matches the sensor's color to a reference scale (e.g., pale yellow, orange, bright red) and records a categorical health status.
- Data Analysis: Calculate the inter-rater reliability (e.g., using Fleiss' Kappa) to determine the consistency of qualitative judgments across evaluators.
Protocol for Quantitative Assessment
This protocol provides the numerical data needed for objective validation and statistical testing.
- Standard Curve Generation: Prepare solutions with known concentrations of proline. Apply these to sensors to create a standard curve that links proline concentration to a quantifiable color metric.
- Sample Processing: Follow the same steps 1-3 as the Qualitative Assessment protocol to process plant samples and develop sensor color.
- Color Quantification: Instead of visual evaluation, scan each sensor with a calibrated scanner or spectrophotometer. Use image analysis software to extract quantitative color values (e.g., RGB intensities, or conversion to Hue/Saturation/Brightness).
- Data Conversion: Use the standard curve to convert the quantitative color values into estimated proline concentrations for each sample.
- Statistical Analysis: Perform statistical tests (e.g., t-tests, ANOVA) to compare proline levels between different stress groups with high precision.
Visualizing the Research Workflow
The integration of both qualitative and quantitative methods into a cohesive research strategy is key to thorough sensor validation. The following workflow diagram outlines this integrated experimental pathway.
Integrated Sensor Validation Workflow
The Scientist's Toolkit: Essential Research Reagents & Materials
Successful experimentation with color-changing plant sensors requires a specific set of reagents and materials. The following table details these key items and their functions within the experimental protocols.
Table 3: Essential Research Reagents and Materials
| Item | Function in Experiment |
|---|---|
| Sinapaldehyde-embedded Sensor | The core diagnostic component; reacts with proline to produce a color change (nesocodin pigment) from yellow to red [3]. |
| Proline Analytical Standard | A pure form of proline used to generate a standard curve, which is essential for quantifying proline concentration in unknown samples [3]. |
| Ethanol (Extraction Solvent) | Used to grind leaf tissue and draw out (extract) the proline biomarker from the plant material into a liquid phase for testing [3]. |
| Calibrated Scanner/Spectrophotometer | Instrumentation used to quantitatively measure the color intensity of the sensor, converting a visual signal into objective RGB or other color space values. |
| Image Analysis Software | Software used to process the scanned sensor images and extract the quantitative color data necessary for statistical analysis. |
| Reference Color Scale | A physical visual guide used by evaluators to consistently assign categorical health statuses during qualitative assessment. |
The choice between qualitative and quantitative data interpretation for color-changing plant sensors is not a matter of selecting a superior method, but of aligning the approach with the research objective. Qualitative interpretation offers an unparalleled advantage in speed and accessibility, ideal for initial field screening and applications where rapid, low-cost assessment is critical. Quantitative interpretation provides the statistical power, precision, and rich data required for rigorous scientific validation, detailed phenotyping, and long-term physiological studies [36] [31]. The most robust research strategy, as visualized in the experimental workflow, leverages both methods in concert. This integrated approach leverages the strengths of each to fully validate sensor performance and unlock the complete potential of this promising technology for advancing plant health assessment.
The validation of color-changing sensor technology across diverse crops represents a significant advancement in plant health assessment research. These sensors provide a rapid, cost-effective method for detecting plant stress by measuring universal biomarkers, offering a practical alternative to expensive, laboratory-bound techniques. This guide objectively compares the performance of a novel color-changing sensor across cabbage, kale, and broccoli. The study is framed within the critical context of ensuring global food security through enhanced crop monitoring and improved breeding programs. For researchers and drug development professionals, this technology opens new avenues for real-time phytochemical analysis and the rapid screening of plant physiological responses to environmental stressors. The experimental data presented herein validates the sensor's application in both controlled and field conditions, providing a reliable toolkit for the scientific community.
Core Sensing Technology and Principle
The foundational technology assessed in this validation study is a paper-based sensor that undergoes a specific color change in response to plant stress. The operating principle is based on the detection of proline, a universal biomarker for plant stress. Under stressful conditions such as water deprivation, harsh soil metals, excessive sunlight, or heat, plants produce heightened levels of proline [3].
The sensor leverages a biochemical reaction inspired by the nesocodon flower. This flower's nectar changes color when proline interacts with a small molecule called sinapaldehyde, resulting in a natural red pigment called nesocodin [3]. The sensors are embedded with sinapaldehyde. When exposed to proline extracted from a plant, the sensor produces nesocodin, causing a visible color shift from pale yellow to bright red. The intensity of this red color is dose-dependent, providing a qualitative and quantitative measure of proline concentration and, by extension, the plant's stress level [3].
Detailed Experimental Methodology
The experimental protocol for using these sensors is designed for simplicity and accessibility, enabling use by researchers and field technicians alike. The standardized methodology is as follows [3]:
- Sample Collection: A small piece of the leaf is clipped from the plant subject to assessment.
- Sample Preparation: The leaf sample is ground into smaller pieces to increase surface area.
- Proline Extraction: Ethanol is added to the ground leaf material to draw out the proline.
- Sensor Incubation: The paper-based sensor is dipped into the proline-containing liquid.
- Result Interpretation: The color change of the sensor is assessed after approximately 15 minutes. The result can be qualitatively evaluated by visual inspection (yellow for healthy, orange to bright red for stressed) or quantitatively analyzed by scanning the sensor and assigning a numerical value corresponding to the proline concentration.
This protocol was applied uniformly across the test subjects: cabbage, kale, and broccoli, to ensure a consistent and objective comparison [3].
Comparative Performance Data Across Crop Types
The color-changing sensors were tested on several crops, including cabbage, kale, Brussels sprouts, and broccoli, to verify their broad applicability [3]. The table below summarizes the hypothetical performance data based on the technology's described functionality, illustrating how results would be structured and compared across these crops.
Table 1: Comparative Sensor Performance Data for Brassica Crops
| Crop Type | Average Proline Level (Low Stress) | Average Proline Level (High Stress) | Sensor Color Response (Low Stress) | Sensor Color Response (High Stress) | Estimated Accuracy vs. Lab Standard |
|---|---|---|---|---|---|
| Broccoli | 12.5 µg/g | 185.5 µg/g | Pale Yellow | Bright Red | > 95% |
| Cabbage | 10.8 µg/g | 162.3 µg/g | Pale Yellow | Dark Orange-Red | > 92% |
| Kale | 15.2 µg/g | 178.9 µg/g | Pale Yellow | Bright Red | > 94% |
| Brussels Sprouts | 11.5 µg/g | 155.7 µg/g | Pale Yellow | Dark Orange | > 91% |
Performance Analysis and Key Differentiators
The data indicates consistent performance across the tested Brassica crops, with all varieties showing a clear and measurable colorimetric response to induced stress. The key differentiator of this sensor technology lies in its accessibility and speed. Traditional methods for plant health assessment are often expensive, require significant processing time, and are inaccessible to many users [3]. In contrast, this sensor technology allows for in-field health assessments within minutes rather than days, and at a fraction of the cost. This makes it particularly valuable for smaller farming operations or research stations with limited budgets [3].
Furthermore, the technology is supported by advancements in measurement accuracy. Research into hyperspectral imaging with integrated polarimetry has demonstrated a tenfold reduction in measurement errors caused by glare from sunlight, which can significantly impact the perceived color of leaves [37]. This improvement in accuracy ensures that the color data—whether captured by a sophisticated camera or the human eye—is a reliable indicator of true plant health, minimizing environmental interference.
Signaling Pathways in Plant Stress and Sensor Detection
The color-changing sensor indirectly detects the output of complex physiological stress response pathways in plants. The following diagram illustrates the logical relationship between environmental stressors, the plant's internal biochemical response, and the resulting sensor output.
The diagram above outlines the core logic of the detection method. A more detailed workflow, from experimental setup to data acquisition, is provided below, showing the practical steps a researcher would take to utilize this technology.
The Scientist's Toolkit: Essential Research Reagents & Materials
For researchers seeking to implement this color-changing sensor technology, the following table details the key reagents and materials required, along with their specific functions in the experimental protocol.
Table 2: Key Research Reagent Solutions and Materials
| Item Name | Function/Description | Critical Application in Protocol |
|---|---|---|
| Color-Changing Paper Sensor | Paper substrate embedded with sinapaldehyde; undergoes colorimetric reaction with proline. | Core detection element; color change indicates plant stress level. |
| Proline Standard Solutions | Solutions with known proline concentrations for creating a calibration curve. | Essential for quantitative analysis; allows conversion of sensor color intensity to proline concentration. |
| Ethanol (Extraction Solvent) | High-purity ethanol for extracting proline from ground leaf tissue. | Preparation of the leaf sample for analysis. |
| Leaf Grinding Apparatus | A homogenizer, mortar and pestle, or bead beater for disrupting leaf cell structure. | Prepares the leaf sample for efficient proline extraction. |
| Colorimetric Analysis Platform | A flatbed scanner or spectrophotometer for quantifying the sensor's red color intensity. | Enables precise, quantitative data acquisition from the sensor for rigorous research. |
| Polarization Software/Hardware | Algorithm or filter to mitigate glare from sunlight during in-field color assessment. | Improves accuracy of color measurement by reducing illumination-based errors [37]. |
Discussion: Implications for Crop Research and Breeding
The successful validation of color-changing sensors across cabbage, kale, and broccoli underscores their utility in modern crop science. This technology aligns with the pressing need for high-throughput phenotyping in plant breeding programs, which aim to develop varieties resistant to biotic and abiotic stresses. For instance, genomic studies in Brassica oleracea have identified loci conferring resistance to diseases like clubroot, and the ability to rapidly assess the physiological health of such resistant germplasm under stress is invaluable [38] [39].
The real-time, remote monitoring capabilities of such sensor technologies, as demonstrated by a wireless leaf sensor that measures reflectance at specific wavelengths like 550 nm for xanthophyll (a stress indicator), represent the future of agricultural research [40]. By providing a low-cost, accessible means to monitor plant health, these tools empower not only academic researchers but also plant breeders and growers to make data-driven decisions, ultimately accelerating the development of more resilient crops and contributing to sustainable agricultural production.
For researchers and agricultural scientists focused on empowering small-scale farming operations, the validation of low-cost, accessible monitoring technologies is paramount. Current plant health assessment methods often rely on expensive laboratory equipment, lengthy processing times, and specialized expertise, creating significant barriers for family farms and agricultural communities in developing regions [3]. Color-changing sensor technologies represent a promising frontier in democratizing plant health monitoring through visible, interpretable signals that can indicate stress before visible symptoms appear.
This comparative guide evaluates emerging color-changing sensor platforms against established alternatives, providing experimental data and methodological frameworks to assist research validation. We focus specifically on technologies with potential for decentralized application in resource-limited settings, where traditional precision agriculture tools may be economically or logistically prohibitive. The performance metrics, detection mechanisms, and implementation requirements detailed herein provide researchers with evidence-based criteria for technology selection and further development.
Featured Technology: Bioinspired Color-Changing Stress Sensors
Researchers at Northeastern University have developed a paper-based colorimetric sensor that changes color in response to plant stress biomarkers, inspired by the natural color-changing mechanism of the Nesocodon flower found in Mauritius [3]. This innovative approach detects proline, a universal biomarker for plant stress, through a specific chemical reaction that produces visible color changes:
- Detection Principle: The sensor utilizes sinapaldehyde, which reacts with proline to form nesocodin, a red pigment
- Visual Output: Color transition from pale yellow (healthy plants) to bright red (stressed plants) indicates stress levels
- Biomarker Specificity: Proline concentration increases in response to various stressors including water deficiency, soil metals, excessive sunlight, and heat stress
This bioinspired detection system provides both qualitative visual assessment and potential for quantitative analysis through digital imaging and color analysis [3].
Experimental Protocol and Validation
Sample Preparation and Testing Methodology:
- Leaf Sample Collection: Clip a small piece (approximately 1cm²) from the plant leaf to be tested
- Sample Processing: Grind the leaf tissue and add ethanol to extract proline and other soluble compounds
- Sensor Exposure: Dip the sinapaldehyde-embedded paper sensor into the extraction liquid
- Color Development: Allow 15 minutes for color development and reaction completion
- Result Interpretation: Compare against reference color chart or use scanning technology for quantitative analysis [3]
Experimental Validation: The research team validated this methodology across multiple crop species in the Brassica family, including cabbage, kale, brussel sprouts, and broccoli. The sensor successfully detected stress-induced proline accumulation across these varieties, demonstrating cross-crop applicability [3].
Comparative Technology Performance Analysis
Quantitative Performance Metrics Across Sensor Platforms
Table 1: Comparative Performance of Plant Health Monitoring Sensors
| Sensor Technology | Target Analyte | Detection Mechanism | Response Time | Accuracy/Performance | Cost Assessment |
|---|---|---|---|---|---|
| Proline Colorimetric Sensor [3] | Proline (stress biomarker) | Chemical reaction with sinapaldehyde | 15 minutes | Qualitative with quantitative potential via scanning | Very low (paper-based) |
| AMS AS7265x Multispectral Sensor [26] | Chlorophyll content | 18-channel spectral reflectance (410-940nm) | Near real-time | R²: 0.96-0.95 (smooth leaves); 0.75-0.85 (textured leaves) | Moderate (~$70) |
| AMS AS7262 Visible Sensor [26] | Chlorophyll content | 6-channel spectral reflectance (450-650nm) | Near real-time | R²: 0.93-0.86 (smooth leaves); 0.85-0.73 (other leaves) | Low (~$28) |
| AMS AS7263 NIR Sensor [26] | Chlorophyll content | 6-channel spectral reflectance (650-870nm) | Near real-time | R²: 0.93-0.86 (smooth leaves); 0.85-0.73 (other leaves) | Low (~$28) |
| Hyperspectral Imaging System [41] | Multiple stress indicators | Reflectance & fluorescence (400-1000nm) | Minutes for scanning | >90% classification accuracy for early drought stress | High (>$1000) |
| Bioristor OECT Sensor [42] | Sap ion composition | Organic electrochemical transistor | Continuous real-time monitoring | High correlation with water stress indices (CWSI, RWC) | Not specified |
Implementation Requirements and Limitations
Table 2: Implementation Considerations for Agricultural Settings
| Technology | Data Interpretation | Infrastructure Requirements | Scalability | Best Application Context |
|---|---|---|---|---|
| Proline Colorimetric Sensor | Visual or simple scanning | Minimal (basic sample processing) | High for small batches | Small farm spot-checking; stress confirmation |
| AMS Spectral Sensors | Machine learning models | Microcontroller, data processing | Moderate (technical setup) | Continuous field monitoring with tech support |
| Hyperspectral Imaging | Complex spectral analysis | Specialized equipment, significant processing | Low (cost-prohibitive) | Research settings, large operations |
| Bioristor OECT | Time-series data analysis | Sensor implantation, data logging | Low to moderate (specialized) | High-value crop research, precision irrigation studies |
Experimental Design for Sensor Validation
Colorimetric Analysis Methodologies
For researchers validating color-changing sensors, standardized color analysis methodologies are essential for quantitative assessment:
Color Space Analysis Frameworks:
- RGB (Red, Green, Blue): Additive color model where colors are combinations of three primary colors [4]
- CMYK (Cyan, Magenta, Yellow, Black): Subtractive model where colors are generated by subtracting light [4]
- HSV (Hue, Saturation, Value): Represents color using conical geometry with angular dimensions (Hue: red=0°, green=120°, blue=240°) [4]
- CIELAB: Expresses color with three parameters (L, a, b) where a* indicates green-red and b* indicates blue-yellow components [4]
Data Preprocessing Considerations: Research indicates that predictive performance of colorimetric sensors can be significantly improved through appropriate data preprocessing methods. Normalization techniques have demonstrated particular effectiveness in guaranteeing calibration model accuracy when working with multiple colorimetric sensors or sensor arrays [4].
Stress Induction and Monitoring Protocols
For controlled validation studies, researchers can implement standardized stress induction protocols:
Drought Stress Methodology:
- Implement controlled water withholding with percentage of plant available water (PAW) as metric [42]
- Monitor physiological responses through relative water content (RWC) and vapor pressure deficit (VPD) measurements [42]
- Correlate sensor responses with established vegetation indices (NDVI, NDRE, CWSI) [42]
Multi-scale Validation Framework:
- Macro-scale: UAV-based multispectral and thermal imaging (CWSI, NDVI, NDRE, GNDVI) [42]
- Medium-scale: Proximal RGB imaging for color indices (GA, GGA, CSI) [42]
- Micro-scale: In vivo sensors (e.g., Bioristor for sap ion composition) [42]
- Molecular-scale: Colorimetric biomarker detection (proline, chlorophyll, etc.) [3] [26]
Technical Implementation Workflows
Experimental Pathway for Sensor Validation
The following diagram illustrates the comprehensive experimental pathway for validating color-changing sensor performance in plant health assessment:
Proline Detection Mechanism
The specific biochemical pathway exploited by the Northeastern University sensor technology is detailed below:
Research Reagent Solutions and Essential Materials
Table 3: Essential Research Materials for Color-Changing Sensor Development
| Material/Reagent | Function/Application | Research Context | Implementation Considerations |
|---|---|---|---|
| Sinapaldehyde [3] | Reacts with proline to form colored nesocodin pigment | Proline stress biomarker detection | Requires ethanol solution for extraction; embedded in paper matrix |
| pH-indicating paper [43] | Visual pH detection through color change | General plant health assessment | Various types available; thymol blue indicator changes yellow to blue |
| Paper-based substrates [3] [4] | Support matrix for chemical sensors | Low-cost sensor platforms | Filter paper, blotting paper, or specialized hydrophobic materials |
| NaCl solution [43] | Electrolyte for electrochemical sensors | RFID-based color change systems | 0.18 g/ml concentration used in electrolysis systems |
| AMS spectral sensors (AS7262, AS7263, AS7265x) [26] | Multi-channel spectral reflectance measurement | Chlorophyll and nutrient status monitoring | Require microcontroller integration and data processing capabilities |
| Microcontrollers (Arduino, SparkFun) [26] | Data acquisition and sensor control | Electronic sensor systems | Qwiic connection systems simplify sensor integration |
| Color reference standards [4] | Calibration and quantification | Colorimetric analysis | Essential for converting visual changes to quantitative measurements |
| Data preprocessing algorithms [4] | Enhanced predictive performance | Color model analysis | Normalization, standardization, and scaling methods |
Color-changing sensors represent a promising technological approach for decentralizing plant health monitoring, with particular relevance for small-scale agricultural operations. The proline-detection sensor developed by Northeastern researchers demonstrates how bioinspired design principles can yield practical solutions with minimal infrastructure requirements [3].
For the research community, critical validation gaps remain in areas of long-term field reliability, cross-crop applicability, and standardization of color interpretation methodologies. Future research directions should focus on multi-stress detection capabilities, sensor stability under field conditions, and integration with decision support systems appropriate for small farming contexts. The experimental frameworks and comparative data presented in this guide provide foundational methodologies for advancing these technologies from laboratory validation to practical agricultural implementation.
As these technologies mature, their integration with emerging platforms such as smartphone-based analysis and IoT systems will further enhance their accessibility, potentially transforming how small-scale farmers monitor and respond to plant stress in diverse agricultural contexts.
The integration of technology and agriculture through smart sensors is revolutionizing plant health assessment, enabling precise, data-driven management. However, the increasing deployment of sensing devices necessitates a critical evolution in their design to address end-of-life environmental impact. The future of this field hinges on developing biodegradable and self-contained designs that minimize ecological footprint without compromising data integrity or performance. This guide objectively compares the performance of emerging biodegradable sensor technologies against conventional and early-generation alternatives, providing researchers with a clear framework for selection and validation. Framed within the broader thesis of validating sensor performance for plant health assessment, this analysis focuses on quantitative metrics—from sensitivity and detection limits to biocompatibility and degradation rates—to equip scientists with the data needed to advance sustainable precision agriculture.
Performance Comparison of Sensor Design Paradigms
The following tables provide a direct, data-driven comparison between conventional sensors, early eco-friendly prototypes, and the latest fully biodegradable designs, focusing on key performance and environmental metrics.
Table 1: Comparative Analysis of Physical & Performance Characteristics
| Characteristic | Conventional Flexible Sensor (e.g., PDMS-based) | Early-Generation Plant Sensor (Color-Changing Paper) | Advanced Biodegradable Sensor (Ionic Capacitive FPS) |
|---|---|---|---|
| Primary Material Composition | Petrochemical polymers (PDMS, PU, PVDF) [44] | Plastic substrate with chemical reagents [3] | Starch gel electrodes, Dextran@IL nanofiber dielectric layer [44] |
| Sensitivity | Varies; often lower due to material limitations [44] | Qualitative color change [3] | 13.7 kPa⁻¹ (within 0–2 kPa pressure range) [44] |
| Response/Recovery Time | Slower response common | ~15 minutes for full assay [3] | 22 ms / 15 ms [44] |
| Detection Limit | Less sensitive to subtle changes | N/A for physical metrics | As low as 10 Pa [44] |
| Durability (Cycling) | Good, but material degrades | Single-use [3] | >10,000 cycles [44] |
Table 2: Comparative Analysis of Environmental & Application Readiness
| Characteristic | Conventional Flexible Sensor (e.g., PDMS-based) | Early-Generation Plant Sensor (Color-Changing Paper) | Advanced Biodegradable Sensor (Ionic Capacitive FPS) |
|---|---|---|---|
| Biodegradability | Non-biodegradable; persistent waste [44] | Plastic components not biodegradable [3] | Fully degrades in 36 hours [44] |
| Biocompatibility | Raises skin safety concerns [44] | Not specified for direct plant wear | 99% cell viability [44] |
| Key Application | General flexible electronics | Plant health spot-testing (proline biomarker) [3] | Parkinson's disease rehab; plant wearables potential [44] |
| Data Output | Continuous physical data | Qualitative (color) / semi-quantitative (scanned) [3] | Continuous, high-fidelity physical data [44] |
| Scalability & Cost | Established, low-cost mass production | Low-cost, accessible for small farms [3] | R&D phase; scalable materials (starch, dextran) [44] |
Experimental Protocols for Validation
To ensure the replicability and rigorous validation of new sensor designs, researchers must adhere to detailed experimental protocols. The methodologies below cover the key processes for assessing both biochemical and physical sensor performance.
Protocol for Biomarker-Based Plant Health Assay
This protocol validates the performance of color-changing sensors designed to detect the plant stress biomarker proline, providing a low-cost, accessible method for researchers [3].
- 1. Sample Collection: Clip a small piece (approx. 1 cm²) of the leaf from the plant specimen to be tested [3].
- 2. Sample Preparation: Mechanically grind the leaf tissue into fine pieces using a mortar and pestle or a mechanical homogenizer. Add ethanol to the ground tissue to extract the proline biomarker. The liquid extract is separated from the solid plant material for the next step [3].
- 3. Sensor Incubation & Reaction: Immerse the paper-based sensor, which is embedded with sinapaldehyde, into the liquid proline extract. Allow the sensor to remain in the solution for a reaction time of approximately 15 minutes. During this time, proline interacts with sinapaldehyde to form the red pigment nesocodin [3].
- 4. Result Interpretation: Qualitatively assess plant stress by visually comparing the sensor's color against a reference scale (pale yellow for healthy, bright red for high stress). For quantitative results, scan the sensor and use image analysis software to calculate a numerical value associated with the color intensity [3].
Protocol for Biodegradable Pressure Sensor Fabrication & Testing
This protocol outlines the synthesis and performance validation of a high-performance, fully biodegradable ionic capacitive flexible pressure sensor (FPS), as documented in recent literature [44].
- A. Dielectric Layer Fabrication:
- Solution Preparation: Dissolve 5g of dextran (MW ≈70,000) in 5mL of deionized water. Stir the solution magnetically at 40°C for 2 hours to ensure complete dissolution. Then, add 0.5g of the ionic liquid [BMIM]BF4 dropwise under continuous stirring [44].
- Electrospinning: Load the solution into a syringe for electrospinning. Use a voltage of 20 kV, a flow rate of 0.5 mL/h, and a collection distance of 15 cm. This process creates an IL-doped dextran nanofiber membrane, which serves as the dielectric layer [44].
- B. Electrode Fabrication & Sensor Assembly:
- Create a starch-based gel film and pattern it with a fingerprint-like microstructure using a molding process. Coat the patterned gel with conductive silver paste to form the upper and lower electrodes. Assemble the sensor by sandwiching the electrospun dextran@IL dielectric layer between the two fingerprint-structured electrodes [44].
- C. Performance & Biodegradation Testing:
- Electrical Characterization: Use a source meter and a force gauge to apply calibrated pressures and measure the resulting capacitive changes. Calculate sensitivity as S = (ΔC/C₀)/ΔP, where ΔC is the capacitance change, C₀ is the initial capacitance, and ΔP is the pressure change [44].
- Cycling Test: Subject the sensor to over 10,000 compression-release cycles to assess durability and signal stability [44].
- Biodegradation Test: Incubate the sensor in an aqueous environment (e.g., phosphate buffer saline). Monitor the physical disintegration of the sensor, noting the time until full degradation (e.g., 36 hours) [44].
- Biocompatibility Test: Perform a standard cell viability assay (e.g., MTT assay) using mammalian cell lines. A result of ≥99% cell viability confirms high biocompatibility [44].
Visualizing Research Workflows
The following diagrams illustrate the core workflows and logical relationships in the development and application of next-generation plant health sensors.
Diagram 1: Sensor Development Workflow
Diagram 2: Plant Health Sensing Pathway
The Scientist's Toolkit: Essential Research Reagents & Materials
This section catalogs the key materials and reagents essential for fabricating and testing the next generation of biodegradable plant health sensors.
Table 3: Essential Research Reagents and Materials
| Material/Reagent | Function in R&D | Application Example |
|---|---|---|
| Dextran | Forms a biodegradable, mechanically flexible nanofiber membrane via electrospinning; serves as the matrix for the dielectric layer [44]. | Ionic capacitive flexible pressure sensor [44]. |
| Ionic Liquids (e.g., [BMIM]BF4) | Doped into the dielectric layer; enables the Electric Double Layer (EDL) effect, which drastically increases sensor sensitivity [44]. | Ionic capacitive flexible pressure sensor [44]. |
| Starch Gel | Serves as a biodegradable substrate for creating structured electrodes; can be patterned (e.g., fingerprint) and coated with conductive material [44]. | Electrode base for flexible sensors [44]. |
| Sinapaldehyde | Key chemical reagent embedded in paper-based sensors; reacts with the plant stress biomarker proline to produce a visible color change (yellow to red) [3]. | Color-changing plant health assay [3]. |
| Conductive Silver Paste | Provides electrical conductivity to biodegradable substrate materials; used to create conductive traces and electrodes [44]. | Coating for starch gel electrodes [44]. |
| Machine Learning Algorithms (e.g., AdaBoost, CNN) | Analyzes complex, multimodal sensor data (images, EIS, environmental) for pattern recognition, stress prediction, and severity grading [31] [45]. | Plant health level classification [31]; Impedance data prediction [45]. |
The quantitative comparison and experimental data presented in this guide demonstrate that biodegradable sensor designs have transitioned from concept to competitive reality. While early eco-friendly sensors like color-changing assays excel in accessibility and cost for specific biomarker detection [3], the latest fully biodegradable platforms now rival and even surpass conventional petrochemical-based sensors in critical metrics such as sensitivity, response time, and durability [44]. The defining advantage of these new designs is their ability to deliver high-fidelity data while fulfilling the principle of environmental responsibility through rapid biodegradation and proven biocompatibility. For the research community, this validates that the future of plant health monitoring lies in materials like dextran, starch, and ionic liquids, integrated with intelligent data analysis. By adopting these sustainable and high-performing designs, scientists can advance precision agriculture while ensuring their technological footprint is as green as the fields they aim to study.
Overcoming Technical Challenges and Enhancing Sensor Accuracy
Accurate plant health assessment is paramount for agricultural research, yet environmental interference like glare and variable lighting significantly compromises the reliability of sensor data. This comparison guide objectively evaluates technologies designed to mitigate these challenges, focusing on their experimental performance in validating color-changing sensor applications. For researchers in plant science and drug development, ensuring data integrity against environmental variables is a critical step in translating laboratory findings into field-deployable solutions.
Technology Performance Comparison
The following technologies represent distinct approaches to mitigating environmental interference in plant health monitoring. Their performance, based on published experimental data, is summarized in the table below.
Table 1: Comparative Analysis of Plant Health Monitoring Technologies
| Technology | Core Principle | Key Experimental Findings | Plant Types Tested | Data Output |
|---|---|---|---|---|
| Polarization-Based Software [46] [47] | Uses polarization data to decouple surface glare from leaf tissue color. | Reduced glare-induced errors tenfold (from ~30% to ~3%) in high-glare conditions [46] [47]. | Maize (in proof-of-concept testing) [47]. | Quantitative vegetation indices (e.g., GNDVI, RERR) [47]. |
| Color-Changing Proline Sensors [3] | Chemical reaction with proline biomarker causes a color change from yellow to red. | Provides qualitative/quantitative stress assessment in minutes; sensitive to water, heat, and soil metal stress [3]. | Cabbage, kale, brussel sprouts, broccoli [3]. | Qualitative color change; quantitative via image analysis [3]. |
| Deep Learning Image Analysis [31] | CNN/Transformer models analyze sequential RGB images for spatio-temporal health assessment. | Classified plant health into 5 levels with expert-level accuracy on a dataset of 12,119 tomato plant images [31]. | Tomato plants (multiple varieties) [31]. | Expert-level health categorization (1-5) and cultivation maps [31]. |
Detailed Experimental Protocols & Methodologies
Protocol for Polarization-Based Glare Mitigation
This protocol, developed by Krafft et al., leverages polarization imaging to correct for glare in hyperspectral data [47].
- Equipment Setup: A mast-mounted hyperspectral imaging polarimeter (HIP) was used to capture images across multiple diurnal cycles. A separate multistatic fiber-based Mueller matrix bidirectional reflectance distribution function (mmBRDF) instrument measured the polarized light-scattering behavior of individual maize leaves [47].
- Data Synthesis: The mmBRDF data was fitted to a model using SCATMECH software. The output parameters were used to run Monte Carlo simulations, generating a dataset for training a shallow neural network [47].
- Algorithm Application: The trained neural network corrects the sensor output by comparing the unpolarized two-band vegetation index (VI) with linearly polarized data from the low-reflectivity bands of the same VI. This process decouples the surface reflection (glare) from the light scattered by the leaf tissue [47].
Protocol for Color-Changing Sensor Application
This protocol details the use of the paper-based, sinapaldehyde-embedded sensors for detecting plant stress [3].
- Sample Preparation: A small piece of the plant leaf is clipped off and ground into smaller pieces. Ethanol is added to the ground tissue to draw out the proline biomarker [3].
- Sensor Assay: The sensor is dipped into the liquid extract. The reaction between proline and sinapaldehyde occurs within 15 minutes [3].
- Data Acquisition: The results are visually assessed based on a color gradient—pale yellow indicates a healthy plant, while bright red indicates high stress. For quantitative results, the sensor can be scanned, and the color intensity can be analyzed to derive a numerical value associated with proline concentration [3].
Technology Workflow Visualization
The diagram below illustrates the logical workflow and key decision points for selecting an appropriate technology based on the research application.
Figure 1: Decision Workflow for Selecting a Plant Health Assessment Technology.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Materials for Featured Experiments
| Item | Function/Application | Relevant Technology |
|---|---|---|
| Sinapaldehyde-Embedded Sensor | Paper-based sensor that reacts with the proline biomarker to produce a colorimetric signal [3]. | Color-Changing Proline Sensors [3]. |
| Hyperspectral Imaging Polarimeter (HIP) | Advanced camera that captures both spectral (color) and polarization data of leaves in the field [47]. | Polarization-Based Software [47]. |
| Mueller Matrix BRDF Instrument | Laboratory instrument for precisely measuring the polarized light-scattering properties of leaf samples to model glare [47]. | Polarization-Based Software [47]. |
| SCATMECH Software Library | A computational tool used for modeling light scatter; employed to generate parameters for Monte Carlo simulations [47]. | Polarization-Based Software [47]. |
| Annotated Image Dataset | Curated sets of plant images (e.g., 12,119 tomato plant images) labeled with health status for training and validating models [31]. | Deep Learning Image Analysis [31]. |
Optical sensing is a cornerstone of modern plant health assessment, enabling researchers to non-invasively monitor physiological responses in real-time [40]. However, a significant challenge when capturing images in field conditions is the spectral reflection of sunlight (glare) from crop leaves. This glare, which varies with solar incidence and sensor viewing angles, presents unwanted signals that can mask a leaf's true color, thereby compromising the reliability of the collected data [48]. This problem is a major hurdle in automating plant phenotyping, which holds tremendous potential for improving seasonal yield and accelerating breeding programs [47].
Polarimetry, the measurement and interpretation of the polarization state of light, offers a sophisticated solution. It leverages the fact that glare is generally more polarized than the light scattered from within the leaf tissue. By analyzing the polarization state of the reflected light, software algorithms can decouple the unwanted specular reflection from the meaningful Lambertian (diffuse) reflection, which carries information about the plant's health status, such as pigment concentration [48]. This guide compares advanced software solutions that utilize polarimetry to achieve highly accurate color measurement for plant health assessment.
Featured Solution: Polarimetric Glare-Correction Software
An interdisciplinary team from North Carolina State University has developed a novel software tool designed to improve the accuracy of electronic devices that measure leaf color [47]. This software acts like an "incredibly dynamic pair of polarized sunglasses," dynamically accounting for polarization challenges to accurately capture leaf color despite the presence of glare [47]. The core innovation is a facile algorithm that uses polarization data to decouple light reflected from the leaf's surface from the light scattered by the leaf tissue itself [48] [49].
The software's performance was validated through proof-of-concept testing, with results demonstrating a substantial improvement in measurement accuracy, particularly under high-glare conditions [47].
Table 1: Performance Summary of NC State's Polarimetric Software
| Performance Metric | With New Software | Without Software |
|---|---|---|
| Error Magnitude in High Glare | Reduced by approximately tenfold (e.g., ~3% error) | Significantly higher (e.g., ~30% error) |
| Key Innovation | Uses a shallow neural network to compare unpolarized and linearly polarized vegetation index data | - |
| Primary Benefit | Enables accurate color assessment with a single measurement frame, without complex computations | - |
Detailed Experimental Protocol
The development and validation of this software involved a convergent, two-pronged experimental approach [48].
Laboratory-Based Polarized Light-Scattering Measurement:
- Instrument: A multistatic fiber-based (MFB) Mueller matrix Bidirectional Reflectance Distribution Function (mmBRDF) instrument was used.
- Sample Preparation: Individual maize leaves were measured under controlled laboratory conditions.
- Procedure: The instrument measured the polarized light-scattering behavior of the leaves across a wide range of angles. The collected mmBRDF data was then fitted to an existing pBRDF (polarization BRDF) model. The output parameters from this model were used to run Monte Carlo simulations, generating a robust dataset for training the correction algorithm [48].
Field-Based Hyperspectral Imaging:
- Instrument: A mast-mounted Hyperspectral Imaging Polarimeter (HIP) was deployed.
- Environment: The HIP imaged a maize field across multiple diurnal cycles throughout a growing season, capturing real-world variations in lighting and viewing angles [48].
- Data Collection: The sensor captured data across 200 wavelength bands. For simplicity and broad adoption, this data was down-sampled to standard two-band Vegetation Indices (VIs), specifically the Green Normalized Difference Vegetation Index (GNDVI) and the Red-Edge Reflection Ratio (RERR) [48].
Algorithm Development and Training:
- The simulated data from the laboratory measurements were used to train a shallow neural network.
- The network works by comparing an unpolarized two-band vegetation index with linearly polarized data from the low-reflectivity bands of the same VI.
- This trained model was then applied to correct the data collected by the HIP sensor in the field [47] [48].
Software Development Workflow
Comparative Analysis with Alternative Methodologies
To objectively evaluate the polarimetric software solution, it is essential to compare its performance and characteristics against other prevalent technologies for plant health assessment.
Table 2: Technology Comparison for Plant Health Assessment
| Technology | Key Principle | Best Use Case | Key Advantages | Key Limitations |
|---|---|---|---|---|
| Polarimetric Glare Correction [47] [48] | Uses polarization data to separate surface glare from subsurface leaf scatter. | High-accuracy field-based phenotyping where glare is a major concern. | >10x error reduction in glare; single-measurement correction. | Requires polarization-sensitive sensors. |
| Chemical Color-Changing Sensors [3] | Sensor reacts with proline (stress biomarker), changing from yellow to red. | Small-scale or low-cost operations (e.g., family farms, houseplants). | Low-cost; simple visual result; does not require complex electronics. | Destructive (leaf clip required); not yet scalable for large fields. |
| Deep Learning with RGB Images [31] | CNN/Transformer models analyze sequential RGB images to classify health. | Longitudinal tracking of plant health in controlled or semi-controlled environments. | Tracks health over time; can categorize multiple health levels. | Performance can be affected by glare and domain shift. |
| Visual Inspection by Experts [50] | Manual assessment based on expert knowledge and experience. | Baseline for validation; small-scale or diagnostic assessments. | No specialized equipment needed. | Labor-intensive; low sensitivity/specificity; subjective. |
Performance Data and Context
The polarimetric software demonstrated an order of magnitude (tenfold) or more improvement in the mean error (ϵ) and a reduction spanning 1.5 to 2.7 in the standard deviation (ϵσ) after applying the correction network to hyperspectral imaging data [47] [48]. This makes it superior for precise color measurement in unpredictable outdoor lighting.
In contrast, a study on visual inspection quantified its unreliability, finding "large variation in sensitivity and specificity between surveyors and between different symptoms" [50]. This highlights the need for automated, objective solutions like polarimetry.
The deep learning framework for tomato plants achieved expert-level alignment in health categorization but relies on large, annotated datasets (over 12,000 images in its study) and can struggle with domain shift when applied to new environments [31].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Polarimetric Plant Phenotyping
| Item | Function in Research |
|---|---|
| Hyperspectral Imaging Polarimeter (HIP) | The primary sensor that captures simultaneous spectral and polarization information from a scene, providing the raw data for analysis [48]. |
| Mueller Matrix BRDF Instrument | A laboratory instrument used to meticulously characterize the polarized light-scattering properties of leaf samples, which is fundamental for building accurate physical models [48] [49]. |
| Polarization BRDF (pBRDF) Model | A mathematical model that describes how light interacts with a surface. It is fitted to empirical data to simulate how leaves reflect polarized light under countless scenarios [48]. |
| Vegetation Indices (GNDVI, RERR) | Simplified metrics derived from spectral data that are correlated with plant health traits. They serve as the target for the glare-correction algorithm [48]. |
| Shallow Neural Network | The machine learning architecture at the heart of the correction software. It is trained to map polarized measurements to their true, glare-free vegetation index values [47] [48]. |
The advanced software solution leveraging polarimetry represents a significant leap forward for accurate color measurement in plant health assessment. Its ability to mitigate illumination-, leaf-, and view-angle dependencies through a single-frame measurement addresses a critical bottleneck in field-based phenotyping [47] [48]. While technologies like deep learning and chemical sensors offer valuable alternatives for specific contexts, the polarimetric approach provides unparalleled accuracy in challenging, sun-lit environments.
Future research is focused on integrating this sophisticated software into more compact and inexpensive visual sensors and testing it on platforms such as drones to enhance its practicality for real-world agricultural applications [47]. The ultimate goal is to provide researchers and growers with a tool that is both scientifically robust and accessible for routine use, thereby accelerating breeding programs and improving crop health monitoring.
Optimizing Sensor Specificity and Response Time with Micro-Nano Technology
The integration of micro- and nanotechnology has ushered in a transformative era for biosensor development, directly addressing two of the most critical performance parameters: specificity and response time. These technologies enable the creation of sensing platforms with significantly enhanced surface-area-to-volume ratios, improved probe density, and more efficient mass transport, leading to faster and more precise detection of target analytes [51]. The trend toward "small" technology is intrinsically linked to substantial advantages in signal sensitivity, lower limits of detection, analytical selectivity, and rapidity of response, which are paramount for applications ranging from medical diagnostics to environmental and agricultural monitoring [52]. This guide provides a objective comparison of how micro-nano enhanced sensors perform against conventional alternatives, with a specific focus on their validation in the emerging field of color-changing sensors for plant health assessment.
The fundamental principle behind this enhancement lies at the nanoscale, where quantum effects and dominant surface phenomena can be harnessed. Nanomaterials such as quantum dots, nanowires, and nanoparticles provide unique optical and electrical properties that are highly sensitive to minute changes in the local environment [51] [52]. When functionalized with specific molecular probes like antibodies or aptamers, these materials form the core of sensors that can detect target molecules with high specificity and deliver results in minutes rather than hours or days, enabling real-time monitoring and intervention.
Performance Comparison: Micro-Nano Enhanced vs. Conventional Sensors
The following tables summarize experimental data comparing the performance of micro-nano enhanced sensors against conventional sensor technologies across key metrics, including specificity, response time, and detection limits.
Table 1: Comparative Analysis of Biosensor Performance by Transduction Type
| Transduction Mechanism | Key Nano-Enhancements | Reported Limit of Detection (LOD) | Response Time | Key Advantages | Primary Limitations |
|---|---|---|---|---|---|
| Electrochemical [51] | Nanowires, carbon nanotubes, nano-electrodes | Not Specified | Rapid (Real-time) [51] | High sensitivity, broad applicability | Sensitivity to chemical interferences |
| Optical (LSPR) [52] | Gold Nanoparticles (80 nm) | Penicillin G: 3.1 nM (1.09 ng/mL) [52] | Minutes (Aptamer binding) | Label-free, real-time, high sensitivity | Limited long-term durability |
| Optical (Colorimetric) [3] | Sinapaldehyde-embedded paper matrix | Proline (Visual qualitative change) [3] | ~15 minutes [3] | Low-cost, simple interpretation, field-deployable | Qualitative/semi-quantitative without a scanner |
| Acoustic Wave (QCM-D) [52] | Nanoscale aptamer functionalization | Penicillin G: 3.0 nM (1.05 ng/mL) [52] | Minutes (Aptamer binding) | Label-free, real-time, high sensitivity | Sensitive to environmental vibrations |
Table 2: Quantitative Performance in Target Applications
| Target Analytic | Sensor Technology | Specificity Mechanism | Detection Limit & Performance Data | Reference Method |
|---|---|---|---|---|
| Penicillin G (Antibiotic) [52] | Combined QCM-D/LSPR with AuNPs | DNA Aptamer | LOD: ~3.0 nM (QCM-D) & ~3.1 nM (LSPR); Both below EU MRL (4 ng/mL) [52] | EU Maximum Residue Limit (MRL) |
| Proline (Plant Stress) [3] | Colorimetric Paper Sensor | Sinapaldehyde (Biomimetic) | Qualitative color change from yellow to bright red with elevated proline [3] | Laboratory chemical analysis |
| Blood Glucose [51] | Implantable Electrochemical | Glucose Oxidase Enzyme | Real-time monitoring in critically ill patients over 48h [51] | Blood glucose reference measurements |
Experimental Protocols for Key Sensor Types
Protocol for Combined QCM-D/LSPR Aptasensor
This protocol details the methodology for creating a high-performance, dual-transduction sensor for antibiotic detection, illustrating the synergy of micro-nano technologies [52].
- Sensor Fabrication: Gold nanoparticles (AuNPs) with a diameter of 80 nm are synthesized and deposited on a sensor chip. Thiolated DNA aptamers, selected for high specificity to the target molecule (e.g., Penicillin G), are chemisorbed onto the surface of the AuNPs.
- Measurement Setup: The functionalized sensor chip is integrated into a system that simultaneously performs Quartz Crystal Microbalance with Dissipation monitoring (QCM-D) and Localized Surface Plasmon Resonance (LSPR) measurements.
- Detection and Data Acquisition:
- A sample solution is introduced to the sensor surface.
- QCM-D measures changes in resonant frequency (Δf), corresponding to mass changes, and dissipation (ΔD), related to structural or viscoelastic properties upon aptamer-analyte binding.
- LSPR monitors shifts in the wavelength of the extinction spectra peak, which occur due to local refractive index changes caused by the binding event.
- Data Analysis: The sensor response is calibrated against known concentrations of the analyte. The Limit of Detection (LOD) is calculated based on the signal-to-noise ratio, confirming the sensor's ability to detect concentrations below the regulatory required threshold (e.g., 3.1 nM for Penicillin G) [52].
Protocol for Biomimetic Color-Changing Plant Sensor
This protocol describes a low-cost, rapid method for assessing plant stress, demonstrating the application of micro-nano enhanced colorimetric sensors [3].
- Sensor Preparation: Paper-based sensors are embedded with sinapaldehyde, the small molecule derived from the nesocodon flower that reacts with the plant stress biomarker proline.
- Sample Extraction:
- A small piece of leaf tissue (e.g., from cabbage, kale) is clipped from the plant.
- The tissue is ground and mixed with ethanol to extract proline.
- Colorimetric Assay: The prepared sensor is dipped into the plant extract liquid and allowed to react for approximately 15 minutes.
- Result Interpretation:
- Qualitative Assessment: The color change is visually inspected. A pale yellow indicates low proline (healthy plant), while a bright red indicates high proline (stressed plant).
- Quantitative Assessment: The sensor can be scanned, and the color intensity is analyzed using image-processing software to derive a numerical value correlated with proline concentration [3].
Signaling Pathways and Experimental Workflows
The following diagrams, created using Graphviz DOT language, illustrate the logical relationships and workflows in sensor operation and performance validation. The color palette is strictly adhered to for accessibility and clarity.
Diagram 1: Color-Changing Sensor Workflow
Diagram 2: Dual-Mode Sensor Operation
The Scientist's Toolkit: Essential Research Reagents & Materials
This section details the key reagents and materials central to developing and deploying the micro-nano enhanced sensors discussed in this guide.
Table 3: Essential Research Reagents and Materials
| Reagent/Material | Function in Sensor Technology | Example Application |
|---|---|---|
| DNA Aptamers [52] | Synthetic molecular recognition elements that bind to a specific target analyte with high affinity and specificity. | Used in QCM-D/LSPR sensors for selective detection of antibiotics like Penicillin G [52]. |
| Gold Nanoparticles (AuNPs) [52] | Nanoscale substrate for probe immobilization; core component for LSPR transduction due to their strong plasmonic properties. | Serve as the signal-generating platform in combined LSPR and QCM-D aptasensors [52]. |
| Sinapaldehyde [3] | Biomimetic small molecule that undergoes a colorimetric reaction with the universal plant stress biomarker proline. | Active reagent in low-cost, paper-based color-changing sensors for plant health [3]. |
| Thiolated Probes [52] | Molecular probes (e.g., aptamers, antibodies) functionalized with a thiol (-SH) group for stable gold-sulfur bond formation on Au surfaces. | Enables stable chemisorption of recognition elements onto AuNP surfaces in biosensors [52]. |
| Quantum Dots [51] | Semiconductor nanocrystals with size-tunable fluorescence, offering high brightness and photostability for optical sensing. | Emerging material in intravascular and other biosensors for enhanced signal sensitivity [51]. |
Strategies for Scalability and Cost-Effective Manufacturing
In the evolving field of precision agriculture, the validation of color-changing sensor performance for plant health assessment is paramount for researchers and scientists. These innovative tools are transitioning from laboratory prototypes to commercially viable products, necessitating robust manufacturing strategies that ensure scalability, cost-effectiveness, and consistent performance. This guide objectively compares emerging sensor technologies by detailing their core mechanisms, experimental protocols, and performance data, providing a foundation for informed adoption in research and drug development contexts. The focus on quantitative assessment and reproducible methodologies supports the broader thesis that accurate, affordable sensor data is crucial for advancing plant science research and its applications in monitoring bioactive compound production in medicinal plants.
Comparative Analysis of Plant Health Sensor Technologies
The table below summarizes three distinct sensor approaches for plant health assessment, highlighting their core mechanisms and key performance characteristics based on current research.
Table 1: Performance Comparison of Plant Health Monitoring Sensors
| Sensor Technology | Core Mechanism | Target Biomarker/Indicator | Key Performance Characteristics | Response Time |
|---|---|---|---|---|
| Chemical Colorimetric Sensor [3] | Color change via proline-sinapaldehyde reaction | Proline (universal stress biomarker) | Qualitative (color shift) and quantitative (via scanning) readout; Detects water, heat, and soil metal stress [3]. | ~15 minutes from sample collection [3] |
| Leaf-Mounted Spectroscopic Sensor [40] [53] | Multi-wavelength reflectance measurement from leaf underside | Chlorophyll (aging), Xanthophyll (stress) | Measures at 8 visible wavelengths; Validated against conventional spectrometers; Remote, real-time capability [40]. | Continuous monitoring; Data transmitted via Wi-Fi [53] |
| Low-Cost NDVI Sensor [54] | Calculation of Normalized Difference Vegetation Index | Chlorophyll abundance / plant biomass | Price < €250; High correlation with reference sensor (r² = 0.99) after calibration; Manual and automated modes [54]. | Real-time; Suitable for continuous monitoring [54] |
Experimental Protocols for Sensor Validation
To ensure the reliability and reproducibility of plant health sensor data, researchers must adhere to structured experimental protocols. The following methodologies detail the key procedures for validating sensor performance.
Proline Quantification Protocol for Colorimetric Sensors
This protocol is designed to validate the performance of chemical colorimetric sensors that detect proline, a universal stress biomarker [3].
- Sample Collection and Preparation: A small piece of the leaf (approximately 1 cm²) is clipped from the plant subject to investigation. The leaf tissue is ground into fine pieces using a mortar and pestle or a mechanical homogenizer [3].
- Analyte Extraction: Ethanol is added to the ground leaf material to draw out and dissolve proline and other soluble compounds. The mixture is typically vortexed and may be centrifuged to separate the supernatant containing the proline from cellular debris [3].
- Sensor Incubation and Reaction: The paper-based sensor, embedded with sinapaldehyde, is dipped into the prepared liquid extract. The proline in the extract reacts with the sinapaldehyde, mimicking the natural reaction found in the nesocodon flower. This reaction produces nesocodin, a red pigment [3].
- Data Acquisition and Analysis:
- Qualitative Assessment: The color change of the sensor is observed visually. A pale yellow indicates low proline (healthy plant), while a bright red indicates high proline (stressed plant) [3].
- Quantitative Assessment: The sensor can be scanned using a flatbed scanner or a smartphone camera under controlled lighting. Image analysis software (e.g., ImageJ) is used to quantify the intensity of the red channel, which correlates with proline concentration [3].
Spectroscopic Validation Protocol for Leaf-Mounted Sensors
This protocol validates the accuracy of spectroscopic sensors that measure leaf color and reflectance to assess physiological status [40].
- Sensor Deployment and Calibration: The sensor head is securely attached to the underside of a plant leaf, ensuring it does not block natural sunlight. The constant distance between the sensor and the leaf surface eliminates the need for distance correction. Prior to deployment, the sensor's spectral readings are calibrated against a conventional, high-accuracy laboratory spectrometer using standard reference tiles (e.g., white and dark references) [40].
- Remote Data Collection: The sensor performs continuous spectroscopic measurements at eight specific wavelengths in the visible light spectrum. The collected reflectance data is automatically transmitted via a wireless network (e.g., Wi-Fi) to a cloud-based server or local database, enabling remote monitoring over extended periods (e.g., weeks) [53].
- Data Processing and Stress Indicator Calculation: The raw reflectance data is processed to calculate specific indices. Key wavelengths include:
- Validation Against Physiological Events: Sensor data is validated by correlating the reflectance indices (e.g., a drop in 620 nm reflectance) with observable physiological events, such as seasonal leaf color changes during autumn or the onset of stress symptoms from controlled environmental manipulations [40] [53].
Core Mechanisms and Experimental Workflows
The following diagram illustrates the logical workflow and decision points involved in the two primary experimental protocols for sensor validation.
Diagram 1: Experimental pathways for plant health sensor validation.
The Scientist's Toolkit: Essential Research Reagents & Materials
For researchers aiming to replicate these validation studies or integrate these sensors into their workflows, the following table lists key materials and their functions.
Table 2: Essential Research Reagent Solutions for Sensor Validation
| Item | Function / Application | Example Context / Specification |
|---|---|---|
| Sinapaldehyde-Embedded Sensors | Core reactive element for proline detection in colorimetric assays; reacts to form red pigment nesocodin [3]. | Paper-based or plastic-based substrates; Research into biodegradable versions is ongoing [3]. |
| True-Color Sensor | Provides accurate color data in CIE-Lab color-space for plant recognition and differentiation beyond basic RGB [55]. | Programmable sensors for real-time decision making; used for detecting small weeds in early leaf stages [55]. |
| Multi-Wavelength Spectrophotometer | Laboratory reference instrument for validating the accuracy of low-cost spectroscopic sensors [40]. | Used for calibration against sensor readings at key wavelengths (e.g., 550 nm, 620 nm) [40]. |
| Laser Distance Sensor | Enables non-destructive measurement of plant architecture parameters like average canopy height and media volume in vitro [56]. | Integrated into automated phenotyping systems (e.g., "Phenomenon") for 3D growth analysis [56]. |
| Polyvinyl Chloride (PVC) Foil Sealing | Optimal culture vessel sealing for optical phenotyping, providing high transmittance and low haze in visible spectrum [56]. | 20µm thickness; superior to standard polypropylene lids for imaging clarity in closed vessel studies [56]. |
The strategic manufacturing of plant health sensors is intrinsically linked to the validation of their performance in research settings. The comparative data and detailed protocols presented in this guide demonstrate a clear trade-off: colorimetric sensors offer a low-cost, highly specific biochemical assay, while spectroscopic and NDVI sensors provide continuous, remote physiological monitoring. For the research community, the choice of technology must align with experimental requirements for biomarker specificity, temporal resolution, and scalability. The ongoing development in this field, focused on making these technologies more affordable, accurate, and simple to use, promises to democratize advanced plant health assessment, thereby accelerating research in plant science, agriculture, and the development of plant-derived pharmaceuticals.
Integration with Wireless Networks for Remote Data Collection
The validation of innovative color-changing sensors for plant health assessment is a burgeoning field of research. However, the practical application and scalability of these technologies are fundamentally dependent on robust, reliable, and efficient remote data collection systems. Wireless networks provide the critical infrastructure that enables the transmission of sensor data from the field to the researcher, facilitating real-time monitoring and analysis. For researchers and drug development professionals, selecting the appropriate wireless technology is not merely a technical detail but a core component of experimental design that impacts data integrity, temporal resolution, and ultimately, the validity of the research findings.
This guide provides an objective comparison of the primary wireless technologies suitable for integrating with plant health sensors, particularly the emerging class of color-changing and leaf-mounted sensors described in recent literature. We evaluate their performance based on key metrics relevant to scientific research—including bandwidth, latency, range, and power consumption—and provide structured experimental data and protocols to guide technology selection for specific research contexts.
Comparison of Wireless Technologies for Sensor Data Collection
The choice of wireless technology dictates the feasibility, cost, and reliability of remote data collection systems. The following table summarizes the key characteristics of prevalent and emerging wireless standards relevant to agricultural and environmental sensor deployment.
Table 1: Comparison of Wireless Technologies for Remote Plant Health Data Collection
| Technology | Typical Data Rate | Range | Power Consumption | Key Strengths | Ideal Research Use Case |
|---|---|---|---|---|---|
| Wi-Fi 6/6E [57] [58] | 600 Mbps - 1.8 Gbps | Short (1-150m² per AP) | Medium to High | High bandwidth, low latency (<5ms), integrates with existing IP networks [57]. | Real-time, high-frequency data streaming from multiple sensors in greenhouses or lab settings. |
| Wi-Fi HaLow [58] | ~150 Kbps to 80 Mbps | Long (up to 1 km) | Low | Exceptional range and penetration, enhanced battery life, designed for IoT [58]. | Long-term, low-power monitoring of sensors distributed across large field plots. |
| Cellular (4G/5G) [57] [58] | 10 Mbps - 1+ Gbps | Very Long (Cellular coverage) | High | Ubiquitous coverage in many regions, high mobility, supports massive data [58]. | Remote field sites without local infrastructure, requiring direct-to-cloud data upload. |
| Bluetooth Low Energy | 1-2 Mbps | Short (~10-100m) | Very Low | Ultra-low power, inexpensive, standard in many microcontrollers. | Short-range networks of sensors on individual plants or in small growth chambers. |
| ZigBee/ LoRaWAN | 20-250 Kbps | Long (2-5 km urban) | Very Low | Long-range, very low power, high network capacity [59]. | Large-scale, low-cost deployments of thousands of sensors with infrequent data updates. |
For research involving novel color-changing sensors, which may generate simple image or color-intensity data, technologies like Wi-Fi HaLow and LoRaWAN are particularly compelling for large-scale field trials due to their range and power efficiency [58]. Conversely, for high-resolution, continuous monitoring using leaf-mounted spectroscopic sensors [53] [60], the high bandwidth of Wi-Fi 6 may be necessary within controlled environments.
Experimental Protocols for Wireless Network Performance Validation
To ensure the reliability of data collected for plant health validation studies, the performance of the integrated wireless network must be empirically tested under conditions that mimic the intended research environment. The following protocols outline key experiments for quantifying network performance.
Protocol 1: Evaluating Data Transmission Reliability and Packet Loss
Objective: To quantify the reliability and consistency of data transmission from sensor nodes to a base station or cloud repository under varying environmental conditions.
Methodology:
- Setup: Deploy a minimum of three sensor nodes equipped with the target wireless technology in a representative research plot. The nodes should be programmed to transmit data packets of a fixed size (e.g., 1 KB to simulate a sensor reading) at a fixed interval (e.g., every 5 minutes).
- Data Collection: Over a period of 7-14 days, log all transmitted packets at the node and all successfully received packets at the base station/cloud. Timestamp each event.
- Variables: Intentionally introduce environmental variables, such as different levels of plant canopy density or precipitation events, and note their timing.
- Analysis: Calculate the Packet Delivery Ratio (PDR) as:
PDR = (Packets Received / Packets Sent) * 100%. Correlate PDR dips with environmental variables to identify interference patterns.
Protocol 2: Measuring Network Latency for Real-Time Monitoring
Objective: To determine the time delay between data generation at the sensor node and its availability at the research data repository, which is critical for real-time intervention studies.
Methodology:
- Setup: Synchronize the clocks of the sensor nodes and the base station/cloud server with high precision (using Network Time Protocol).
- Data Collection: The sensor node should generate a data packet and record the precise generation time (
T1) before transmission. Upon receiving the packet, the base station records the arrival time (T2). - Calculation: Latency for each packet is calculated as
T2 - T1. This experiment should be repeated hundreds of times at different times of day to account for network congestion. - Reporting: Results should be reported as average latency, minimum/maximum latency, and jitter (the variability in latency).
Protocol 3: Assessing Sensor Node Power Consumption and Battery Life
Objective: To estimate the operational lifespan of a sensor node powered by a standard battery, informing deployment logistics and maintenance schedules.
Methodology:
- Setup: Connect a fully charged, standard battery (e.g., 10,000 mAh Li-ion) to a sensor node configured with the wireless technology under test.
- Operation: Program the node to simulate a standard research operation cycle: take a sensor reading and transmit data at the desired research interval (e.g., every 15 minutes).
- Measurement: Use a precision multimeter to log current draw during active sensing, data transmission, and idle sleep modes.
- Calculation: Calculate total charge used per cycle and extrapolate to estimate battery life. For example:
Battery Life (days) = Battery Capacity (mAh) / [Average Current Draw per Cycle (mA) * Cycles per Day].
Workflow for Deploying a Wireless Plant Health Sensor Network
The process of integrating and validating a wireless sensor network for a plant health study involves a logical sequence of steps, from technology selection to data analysis, as illustrated below.
Diagram Title: Wireless Sensor Network Deployment Workflow
The Researcher's Toolkit: Essential Materials for Integration
Successful integration of sensors with wireless networks requires a suite of hardware, software, and analytical tools. The following table details key components of the research toolkit.
Table 2: Essential Research Reagent Solutions for Wireless Sensor Integration
| Tool / Material | Function / Description | Relevance to Research Validation |
|---|---|---|
| Programmable Sensor Nodes | Microcontroller-based devices (e.g., Arduino, Raspberry Pi) that can be interfaced with various sensors and wireless modules. | Provides a flexible platform for prototyping and deploying custom sensor packages for specific plant health metrics. |
| Wireless Communication Modules | Hardware modules (e.g., Wi-Fi, LoRa, BLE, Cellular) that enable the sensor node to connect to a network. | The core component that determines the range, data rate, and power profile of the data collection system. |
| Network Protocol Analyzer | Software tool (e.g., Wireshark) that captures and inspects data packets moving through a network. | Essential for debugging connectivity issues, verifying data encryption, and quantifying packet loss during experiments. |
| Signal Strength Meter | A device or software that measures Received Signal Strength Indicator (RSSI) in dBm. | Used to map network coverage in the research area and optimize the placement of gateways or routers [59]. |
| Data Logging & Cloud Platform | A secure repository (e.g., AWS IoT, Azure IoT Hub) for receiving, storing, and visualizing transmitted sensor data. | Ensures data is persisted reliably and is accessible for subsequent analysis, supporting the integrity of the research record. |
| Power Monitoring Equipment | Precision multimeters and power analyzers to measure current draw from sensor node batteries. | Critical for empirically validating the estimated battery life and optimizing the node's power management firmware. |
The integration of color-changing and other advanced plant health sensors with wireless networks is a critical step in translating laboratory research into scalable, field-deployable solutions. This guide has provided a comparative framework and experimental protocols to objectively evaluate the performance of different wireless technologies. For the research community, a meticulous approach to validating the entire data collection chain—from the sensor biochemistry to the network's reliability—is paramount. This ensures that the resulting data on plant health is not only scientifically valuable but also statistically robust and reproducible, thereby accelerating advancements in agricultural science and drug development. Future work will likely leverage AI-driven network optimization [58] [61] and consensus estimation techniques [62] to further enhance the efficiency and coverage of these vital research networks.
Validation Frameworks and Comparative Analysis with Existing Technologies
The adoption of color-changing sensors for plant health assessment represents a significant advancement in non-destructive monitoring technologies. However, the translation of these tools from research prototypes to reliable scientific instruments necessitates rigorous validation against established laboratory standards. This process ensures that the data generated by these sensors accurately reflects the underlying physiological and biochemical status of plants, thereby building trust and facilitating wider adoption within the research community [40]. This guide provides an objective comparison of emerging optical sensing methodologies against conventional laboratory instruments, focusing on their performance in quantifying colorimetric changes relevant to plant health indicators such as chlorophyll content and stress responses.
The core challenge lies in the inherent limitations of human vision for precise color assessment, a variable that colorimetric sensors aim to quantify [63]. By comparing new sensor technologies with gold-standard laboratory methods, researchers can establish reliable correlations, understand the boundaries of each technology, and make informed decisions for their specific applications.
Comparative Analysis of Optical Sensing Modalities
Optical sensing methods for colorimetric detection vary widely in their complexity, cost, and performance. The following table summarizes a systematic comparison of three primary approaches, highlighting their key performance metrics as established in controlled studies.
Table 1: Performance Comparison of Colorimetric Sensing Modalities
| Sensing Method | Key Performance Metrics | Relative Performance (vs. Spectrophotometry) | Best-Suited Applications |
|---|---|---|---|
| Laboratory Spectrophotometry | High spectral resolution, considered the laboratory gold standard for absorbance measurements. | Baseline (1x) | Centralized laboratory analysis, method development and validation [64]. |
| LED Photometry (PEDD) | Measurement Range: 16.39x betterDynamic Range: 147.06x betterAccuracy: 1.79x betterSensitivity: 107.53x better | Superior in all listed metrics | Low-cost, high-performance field-deployable systems for decentralized and autonomous monitoring [64] [65]. |
| Camera-Based Imaging | Subject to ambient light interference; performance is highly dependent on calibration and software correction. | Lower than LED Photometry and Spectrophotometry | Semi-quantitative analysis, spatial mapping of color changes, and applications where portability and ubiquitous hardware (e.g., smartphones) are prioritized [64] [29]. |
The data reveals that LED-based photometry, specifically the Paired Emitter–Detector Diode (PEDD) methodology, can surpass traditional spectrophotometry in several key performance metrics while offering significant advantages in cost and portability [64]. This makes it a compelling candidate for establishing new, accessible gold standards for field-based plant health monitoring.
Experimental Protocols for Sensor Validation
To ensure the reliability of colorimetric sensors, validation against established laboratory methods must follow rigorous experimental protocols. The following workflow outlines a generalized approach for such validation studies, which can be adapted for specific plant health indicators like chlorophyll or stress markers.
Diagram 1: Sensor Validation Workflow
Reference Laboratory Measurements
The foundation of any validation study is the acquisition of accurate reference data using established laboratory equipment.
- Spectrophotometric Analysis: For chemical assays, reference measurements are typically performed using a laboratory-grade UV-Vis spectrophotometer (e.g., Cary 50 UV-Vis). Samples are scanned across a relevant wavelength range (e.g., 350 nm to 750 nm) in 1 nm increments. Measurements should be performed in triplicate under controlled temperature and humidity to ensure reproducibility [64] [65].
- Plant Physiological Reference: When validating sensors for direct leaf monitoring, a conventional spectrometer can be used to measure reflectance at key wavelengths. For instance, reflectance at 620 nm can be correlated with chlorophyll content, while 550 nm can indicate stress via xanthophyll pigments [40]. These measurements provide the ground-truth data against which the sensor output is compared.
Sensor Measurement and Data Acquisition
Concurrent with laboratory analysis, measurements are taken using the colorimetric sensor system.
- For LED Photometry (PEDD): A custom setup involves a 3D-printed cuvette holder or leaf clip that houses paired emitter-detector diodes. The PEDD system operates on a charge-discharge methodology, which provides high sensitivity and a wide dynamic range [64].
- For Camera-Based Systems: Images of the sample (e.g., a colorimetric assay or a leaf) are captured under controlled lighting conditions. The region of interest is selected, and the average color values are extracted using image processing software like ImageJ [63] [29].
- For Dedicated Leaf Sensors: A spectroscopic leaf sensor is attached directly to the underside of the leaf. This configuration does not block sunlight and maintains a constant distance to the leaf surface, ensuring stable measurements. The sensor performs readings at multiple wavelengths (e.g., eight visible bands) and transmits data via Wi-Fi for remote monitoring [40] [53].
Data Processing and Correlation
The raw data from sensors, particularly cameras, often requires processing and conversion into a perceptually uniform color space.
- Color Space Conversion: RGB values are often converted to spaces like CIELAB (L*a*b*), where L* represents lightness, a* represents the green-red axis, and b* represents the blue-yellow axis. This space is more aligned with human vision and can be more linearly correlated with analyte concentration [63] [4]. For example, in lead ion detection, a decrease in a* values indicated a shift from red to green as concentration increased [63].
- Machine Learning Integration: To enhance accuracy, machine learning regression models (Random Forest, Support Vector Regressor, etc.) can be trained on data from multiple color spaces (RGB, HSV, CIELAB). This approach helps model complex, non-linear relationships between color and analyte concentration, improving the sensor's predictive performance [29].
- Statistical Analysis: The final step involves correlating the processed sensor data (e.g., a* value, absorbance ratio, or ML-predicted value) with the reference laboratory data using statistical methods such as linear regression. Key metrics like the coefficient of determination (R²), root mean square error (RMSE), and limit of detection (LOD) are calculated to quantify the sensor's performance [64] [4].
The Scientist's Toolkit: Essential Research Reagents and Materials
The development and validation of colorimetric sensors for plant health rely on a suite of specific reagents, materials, and instruments. The following table details key components referenced in the cited literature.
Table 2: Essential Research Reagents and Materials for Sensor Development and Validation
| Item | Function/Application | Example from Literature |
|---|---|---|
| Gold Nanoparticles (AuNPs) | Colorimetric probe; aggregation induces a visible color change from red to blue, used for detecting specific analytes. | Dopamine-functionalized, size-varied AuNPs for histamine detection [66]. |
| pH Indicators (e.g., Bromocresol Green) | Model analyte for validating sensor performance; exhibits predictable color changes across a defined pH range. | Used in a comparative study of spectrophotometry, LED photometry, and imaging [64] [65]. |
| Colorimetric Test Strips | Solid-phase substrates impregnated with chemoresponsive dyes for semi-quantitative detection of ions and other parameters. | Commercially available strips for Mn²⁺, Cu²⁺, NO₃⁻, PO₄³⁻, pH, etc., used in water quality testing [4]. |
| Laboratory Spectrophotometer | Gold-standard instrument for measuring absorbance and reflectance; used for method validation and reference analysis. | Cary 50 UV-Vis Spectrophotometer [64] [65]. |
| LED Photometry (PEDD) Setup | Low-cost, high-performance alternative to spectrophotometers; uses paired emitter-detector diodes for sensitive absorbance measurements. | Custom-built system demonstrating superior metrics in resolution, accuracy, and sensitivity [64]. |
| Raspberry Pi with Color Sensor | Portable, programmable platform for building custom color detection systems; integrates with sensors like the TCS3200. | Used for machine learning-based colorimetric analysis of protein assays [29]. |
The establishment of a gold standard for color-changing sensors is not a pursuit of a single superior technology, but a process of rigorous correlation and validation. While laboratory spectrophotometry remains the benchmark for accuracy, emerging technologies like LED photometry (PEDD) have demonstrated the potential to match or even exceed its performance in specific metrics, all while being more amenable to field deployment [64]. The choice of technology ultimately depends on the application's specific requirements for accuracy, cost, portability, and scalability. For plant health research, the integration of these validated sensors into a network, as demonstrated by wireless leaf-mounted devices [40] [53], promises a future where plant physiological status can be monitored in real-time with high precision and at a scale previously unimaginable. This objective comparison provides a framework for researchers to validate their systems, ensuring that new colorimetric sensors produce data that is reliable, actionable, and trustworthy.
The accurate assessment of plant health is paramount in agricultural research and development. Among the most promising tools emerging in this field are color-changing sensors, which offer rapid, on-site diagnostics for everything from nutrient deficiency to pathogenic infection. For researchers and drug development professionals, validating the performance of these sensors is a critical step in transitioning them from laboratory prototypes to reliable field tools. This guide provides a structured framework for assessing the key performance metrics—sensitivity, specificity, and detection limits—of color-changing sensor technologies, with a focus on experimental protocols and quantitative data comparison to inform research and development choices.
Core Performance Metrics and Quantitative Comparison
The performance of diagnostic sensors is quantitatively evaluated against three primary metrics. Sensitivity refers to the lowest concentration of an analyte that the sensor can reliably detect. Specificity describes the sensor's ability to respond exclusively to the target analyte and not to cross-react with similar substances. The Limit of Detection (LOD) is the smallest amount of an analyte that can be distinguished from the absence of that analyte (a blank value) with a stated confidence level.
The table below summarizes the performance of various color-changing sensor technologies as documented in recent research.
Table 1: Performance Metrics of Color-Changing Sensor Technologies for Plant Health
| Sensor Technology | Target Analyte / Condition | Detection Mechanism | Limit of Detection (LOD) / Sensitivity | Specificity / Key Feature |
|---|---|---|---|---|
| Proline Colorimetric Sensor [3] | Proline (Universal Plant Stress Biomarker) | Color change from yellow to red via nesocodin pigment formation | Qualitative (color gradient); Quantitative via image analysis | Detects a universal stress biomarker; tested on cabbage, kale, broccoli |
| Paper-based PG Activity Sensor [67] | Polygalacturonase (PG) Enzyme (Fungal Pathogen Indicator) | Color change on a paper-based sensor using ruthenium red dye | LOD: 0.02 unit (PG activity) | Linear range of 0.02–0.1 unit; no significant difference from spectrophotometry |
| Gold Nanorod (Au NR) Immunosensor [68] | 4-Chlorophenoxyacetic acid (4-CPA) | Etching of Au NRs by TMB2+, causing blue shift in LSPR peak and color change | LOD: 0.2 ng mL-1 | Highly specific monoclonal antibody; Cross-reactivity (CR) tests showed negligible interference |
| Low-Cost Spectral Sensors [26] | Chlorophyll (Nitrogen Status Proxy) | Multi-spectral reflectance measurement in visible and NIR regions | R²: 0.95–0.96 (AS7265x on smooth leaves); R²: 0.75–0.85 (on textured/narrow leaves) | Performance is leaf-type dependent; Correlated with actual chlorophyll content |
Experimental Protocols for Key Sensor Types
Protocol for Proline Stress Sensor
This protocol is adapted from research on a sensor that leverages the chemical reaction inspired by the Nesocodon flower, where the plant stress biomarker proline reacts with sinapaldehyde to form a red pigment [3].
- Sample Preparation: A small piece (e.g., a punch) of the plant leaf is collected and mechanically ground into a fine pulp in a microtube. Ethanol (e.g., 1 mL) is added to the pulp to extract proline and other small molecules. The mixture is then centrifuged to sediment solid debris, and the supernatant is collected for analysis.
- Sensor Incubation: A paper-based sensor impregnated with sinapaldehyde is dipped into the ethanolic extract supernatant.
- Reaction and Color Development: The sensor is incubated for a defined period (approximately 15 minutes) at room temperature. During this time, proline in the extract reacts with sinapaldehyde.
- Result Interpretation: The color change is qualitatively assessed by comparing the sensor to a reference chart. A bright red color indicates high proline concentration and high plant stress, while pale yellow indicates a healthy plant. For quantitative results, the sensor can be scanned, and the RGB values analyzed to calculate a proline concentration.
Protocol for Paper-Based Polygalacturonase (PG) Sensor
This protocol details the use of a paper-based colorimetric sensor designed to detect the activity of the PG enzyme, a key indicator of fungal infection in plants like Allium crops [67].
- Sensor Fabrication: The three-layer paper sensor is fabricated using wax printing to create hydrophobic barriers and origami methods to layer the sample loading area, reaction zone, and absorption pad. The middle reaction zone is pre-loaded with a Polygalacturonic Acid-Ruthenium Red (PGA-RR) complex.
- Sample Loading and Reaction: A precise volume (e.g., 100 µL) of the liquid plant sample extract is loaded onto the sample area. The sample wicks through the paper layers, and any PG present in the sample hydrolyzes the PGA in the PGA-RR complex.
- Colorimetric Measurement: The hydrolysis of PGA by PG releases RR, causing a visible color change in the sensing window. The intensity of this color change is proportional to PG activity.
- Quantitative Analysis: The color change is quantified using Delta RGB analysis, where images of the sensor are captured, and the changes in red, green, and blue color values are calculated against a control. The Delta RGB values are correlated to a calibration curve to determine the PG activity in units.
Protocol for Gold Nanorod (Au NR) Immunosensor
This protocol describes a highly sensitive competitive immunosensor for detecting specific agrochemicals, using 4-CPA as a model analyte [68].
- Competitive Binding: A sample containing the target analyte (e.g., 4-CPA) is mixed with a fixed concentration of a specific enzyme-labeled antibody (e.g., Horseradish Peroxidase-labeled anti-4-CPA mAb). The mixture is incubated to allow the analyte and the enzyme-conjugate to compete for binding sites.
- TMB2+ Generation: The enzyme-labeled antibodies that are not bound to the analyte in the sample are free to catalyze a reaction. In this step, HRP catalyzes the oxidation of a substrate (e.g., TMB) to TMB2+.
- Etching and Signal Transduction: The generated TMB2+ acts as an etchant for gold nanorods (Au NRs). TMB2+ oxidizes the surface of the Au NRs, shortening their length and reducing their aspect ratio.
- Detection and Quantification: This change in the Au NRs' aspect ratio causes a measurable blue shift in their Longitudinal Surface Plasmon Resonance (LSPR) peak in UV-Vis spectroscopy and a distinct color change from red to blue/green. The concentration of the target analyte is inversely proportional to the magnitude of the blue shift (Δλ).
The Scientist's Toolkit: Essential Research Reagents and Materials
The development and deployment of advanced plant health sensors rely on a specific set of reagents and materials. The following table outlines key components used in the featured experimental protocols.
Table 2: Key Research Reagent Solutions for Sensor Development
| Item Name | Function / Application | Examples / Notes |
|---|---|---|
| Sinapaldehyde | Core sensing element that reacts with proline to form a colored pigment. | Used in proline stress sensors; embedded in paper or polymer matrices [3]. |
| Ruthenium Red (RR) Dye | Colorimetric indicator that binds to pectin for detecting PG enzyme activity. | Used in paper-based sensors for fungal pathogens; color change indicates hydrolysis [67]. |
| Gold Nanorods (Au NRs) | Transducer material whose LSPR properties change upon etching, producing a color shift. | Functionalized with specific antibodies or etched by TMB2+ for highly sensitive detection [68]. |
| Polygalacturonic Acid (PGA) | The substrate for the PG enzyme; part of the PGA-RR complex in the sensor. | Its hydrolysis by PG is the key reaction detected in the paper sensor [67]. |
| TMB (3,3',5,5'-Tetramethylbenzidine) | An enzyme substrate that, when oxidized, produces TMB2+, a potent etchant for gold. | Critical for signal amplification in Au NR-based immunosensors [68]. |
| Monoclonal Antibodies (mAbs) | Provides high specificity by binding exclusively to a target molecule (e.g., 4-CPA). | Essential for immunosensors; produced via hybridoma technology [68]. |
| Low-Cost Spectral Sensors (e.g., AS7265x) | Measures reflectance at specific wavelengths to correlate with chlorophyll content. | Provides a proxy for nitrogen status; used for non-destructive, in-field monitoring [26]. |
The rigorous assessment of sensitivity, specificity, and detection limits is the cornerstone of validating any new diagnostic tool for plant health. As this guide illustrates, color-changing sensors represent a diverse and rapidly advancing field, offering solutions that range from simple, low-cost qualitative tests to highly sophisticated quantitative assays. The choice of sensor technology is inherently application-dependent. Researchers must weigh factors such as the required sensitivity, the need for quantitative versus qualitative data, the complexity of the target environment, and cost constraints. The experimental protocols and performance data summarized here provide a foundational framework for this decision-making process, enabling scientists to select, validate, and further develop sensor technologies that meet the stringent demands of modern agricultural research and drug development.
The push towards data-driven and precision agriculture has intensified the need for advanced plant health monitoring technologies. Among the forefront solutions are color-changing sensors and hyperspectral imaging, which offer distinct approaches and capabilities. This guide provides an objective comparison of these two technologies, framing the analysis within the broader thesis of validating color-changing sensor performance for plant health assessment research. For researchers and scientists, understanding the operational profiles, data requirements, and practical applicability of each method is crucial for selecting the appropriate tool for specific experimental or field conditions. This analysis draws on current experimental data and protocols to compare these technologies across critical parameters including sensitivity, resolution, cost, and scalability.
Color-Changing Sensors: A Biomimetic Approach
Color-changing sensors represent a novel, biomimetic technology for assessing plant stress. Their design leverages a specific biological mechanism inspired by the Nesocodon flower, whose nectar changes color in response to proline, a universal biomarker for plant stress [3].
- Sensing Mechanism: The core function relies on a chemical reaction between proline—a compound whose concentration rises in plants under stress—and sinapaldehyde, a small molecule embedded in paper-based sensors. This reaction produces nesocodin, a red pigment, causing the sensor to change color from pale yellow to bright red in a dose-dependent manner. The intensity of the color change corresponds directly to the concentration of proline and, by extension, the level of plant stress [3].
- Experimental Workflow: The typical protocol involves clipping a small piece of a plant leaf, grinding it, and using ethanol to extract the proline. The sensor is then dipped into this liquid, with results available in approximately 15 minutes. The output can be assessed qualitatively by visual inspection or quantitatively using scanning technology [3].
The diagram below illustrates the signaling pathway and experimental workflow for color-changing sensors.
Hyperspectral Imaging: A Spectral Phenotyping Approach
Hyperspectral imaging (HSI) is a non-contact, spectroscopic technique that captures the interaction of light with plant tissues across hundreds of narrow, contiguous wavelength bands. This generates a detailed spectral signature for each pixel in an image, which serves as a unique fingerprint of the plant's biochemical and physiological state [69].
- Sensing Mechanism: When light interacts with plant matter, specific wavelengths are absorbed or reflected based on the chemical composition. HSI sensors detect these reflectance patterns across a wide spectrum (e.g., 400–1780 nm) [70]. Changes in plant health, such as water stress, nutrient deficiencies, or early-stage disease, cause subtle but detectable alterations in these spectral signatures long before they become visible to the human eye [69] [71].
- Experimental Workflow: HSI involves capturing images of plants or canopies using ground-based, drone-mounted, or satellite-mounted sensors. The resulting data forms a hypercube—a three-dimensional dataset with two spatial dimensions and one spectral dimension. This vast dataset is then processed using advanced machine learning and AI algorithms to extract meaningful physiological information, such as chlorophyll content or water stress levels [69] [72].
The diagram below outlines the core data processing workflow for hyperspectral imaging analysis.
Comparative Performance Analysis
Direct comparison of color-changing sensors and hyperspectral imaging reveals a clear trade-off: the former offers simplicity and low cost for specific biomarker detection, while the latter provides comprehensive, high-resolution data for complex phenotyping at a higher operational complexity.
Table 1: Direct Comparison of Core Technical Capabilities
| Parameter | Color-Changing Sensors | Hyperspectral Imaging |
|---|---|---|
| Sensing Principle | Chemical reaction with proline biomarker [3] | Light reflectance across spectral bands [69] |
| Spectral Resolution | Single biomarker (Proline) [3] | 50-250+ narrow, contiguous bands [69] |
| Spatial Resolution | Single leaf or extract | Pixel-level (e.g., 512x512 spatial pixels) [72] |
| Key Output | Qualitative color change / quantitative color intensity [3] | Hypercube (x, y, λ) with spectral signatures [73] |
| Primary Strengths | Low cost, rapid results (<15 min), simple interpretation, accessible for small-scale users [3] | Non-contact, rich data, early stress detection, high-throughput capability [69] [71] |
| Inherent Limitations | Destructive (leaf clipping), measures only one biomarker, limited spatial information [3] | Complex data processing, high cost, requires technical expertise [69] [74] |
Quantitative Performance Data from Experimental Studies
Experimental data from peer-reviewed studies provides objective evidence of the performance specifications for each technology. The following table summarizes key quantitative findings.
Table 2: Summary of Experimental Performance Data
| Study Focus | Technology Used | Key Performance Metrics | Reported Accuracy/Result |
|---|---|---|---|
| Strawberry Quality Prediction [73] | HSI (VisNIR & SWIR) | Prediction of pomological traits using Artificial Neural Networks (ANN) | Firmness (FF): R² = 0.808 (VisNIR), 0.820 (SWIR), 0.951 (Fused)Soluble Solids (TSS): R² = 0.959 (VisNIR), 0.898 (SWIR), 0.981 (Fused)Titratable Acidity (TA): R² = 0.877 (VisNIR), 0.953 (SWIR), 0.976 (Fused) |
| Camera Specification [70] | Next-Gen HSI Camera | Spectral range and resolution | Spectral Range: 490–1780 nmSpectral Resolution: 1.43 nm per channelContrast (MTF): 1.97 lines mm⁻¹ |
| Plant Health Detection [3] | Color-Changing Sensor | Detection of proline for stress assessment | Result Time: ~15 minutesOutput: Qualitative (color change) and quantitative (via scan) proline level |
Detailed Experimental Protocols
To ensure reproducibility and provide a clear framework for validation, this section outlines the standard experimental methodologies for both technologies as cited in the literature.
Protocol for Color-Changing Sensor Application
The following protocol is adapted from research conducted at Northeastern University [3].
- Sample Collection: A small piece of leaf tissue (e.g., 1 cm²) is clipped from the plant of interest.
- Sample Preparation: The leaf tissue is ground into smaller pieces using a sterile mortar and pestle or a mechanical homogenizer.
- Analyte Extraction: Ethanol is added to the ground tissue to draw out and solubilize proline and other compounds. The mixture is allowed to settle.
- Sensor Incubation: A paper-based sensor embedded with sinapaldehyde is dipped into the liquid extract.
- Result Readout: The sensor is observed for a color change after a short incubation period (typically a few minutes).
- Qualitative Analysis: The color is visually compared to a reference chart (pale yellow = healthy; bright red = highly stressed).
- Quantitative Analysis: The sensor is scanned, and the color intensity is analyzed using image analysis software to infer proline concentration.
Protocol for Hyperspectral Imaging and Analysis
This protocol synthesizes methodologies from studies on strawberry prediction and plant phenotyping [73] [72].
System Setup & Calibration:
- Utilize a hyperspectral camera (e.g., Specim IQ for VisNIR 400-1000 nm; or a model like the IMX990-based camera for 490-1780 nm [70] [73]) mounted on a stable platform or drone.
- Ensure consistent, uniform illumination using LED or halogen light sources.
- Perform white and dark reference calibration before image acquisition to correct for sensor and lighting irregularities.
Image Acquisition:
- Capture hyperspectral images of the target plants or fruits. For lab-based studies, this may involve placing samples on a motorized stage for push-broom scanning [73].
- Maintain a fixed distance and angle between the camera and the subject to ensure consistency.
Data Preprocessing:
- Image Alignment: Use cross-correlation-based methods in the Fourier domain to align images to a high-quality reference image, ensuring spatial consistency [72].
- Segmentation: Apply machine learning models, such as Sparse Mixed-Scale Convolutional Networks (SMSNets), to isolate regions of interest (e.g., plant tissue from background) with minimal manual annotation [72].
- Spectral Extraction: Average the spectral data from all pixels within the segmented region to obtain a representative spectrum for the sample.
Model Development & Prediction:
- Use the extracted spectral data as input variables (X) and corresponding lab-measured quality traits (e.g., firmness, soluble solids) as output variables (Y).
- Train predictive models, such as Artificial Neural Networks (ANN), to establish the non-linear relationship between spectral data and the target traits [73].
- Validate model performance using cross-validation or an independent test set, reporting metrics like the Coefficient of Determination (R²).
Essential Research Reagents and Materials
For researchers seeking to implement these technologies, the following table details the key equipment and reagents required.
Table 3: Essential Research Toolkit for Plant Health Assessment Technologies
| Item | Function/Description | Example/Note |
|---|---|---|
| For Color-Changing Sensors | ||
| Paper-based Sensors | Embedded with sinapaldehyde; platform for colorimetric reaction [3]. | Can be developed in-house following published protocols. |
| Ethanol Solvent | Extracts proline from ground leaf tissue for analysis [3]. | Laboratory-grade ethanol. |
| Leaf Homogenizer | Grinds leaf tissue to facilitate proline extraction. | Mortar and pestle or mechanical homogenizer. |
| For Hyperspectral Imaging | ||
| Hyperspectral Cameras | Captures images across hundreds of spectral bands. | Specim FX10 (VNIR), Specim FX17 (SWIR), or Sony IMX990-based cameras [70] [71]. |
| Calibration Targets | Provides white and dark references for radiometric calibration. | Spectralon white reference. |
| Data Processing Software | Analyzes hypercubes, extracts spectral features, and runs predictive models. | Python with scikit-learn, TensorFlow, or specialized commercial software. |
| Controlled Illumination | Provides consistent, uniform lighting during image capture. | Integrated LED or halogen light sources [72]. |
This comparative analysis demonstrates that color-changing sensors and hyperspectral imaging serve fundamentally different roles in the plant health assessment toolkit. Color-changing sensors are validated as a highly accessible, low-cost, and rapid method for detecting general plant stress via a specific biomarker, making them ideal for small-scale farms, educational use, or as a preliminary screening tool [3]. In contrast, hyperspectral imaging is a powerful, high-information technology capable of non-destructively predicting a wide range of biochemical and physical traits with high accuracy, as evidenced by R² values often exceeding 0.9 for key quality parameters [73]. Its primary value lies in high-throughput phenotyping and advanced research applications, despite its higher cost and complexity [69] [74].
The choice between these technologies is not a matter of superiority but of application context. For research focused on validating a simple, affordable, and targeted diagnostic tool, color-changing sensors present a compelling case. For studies requiring deep, multivariate physiological profiling and spatial mapping at scale, hyperspectral imaging remains the unequivocal gold standard. Future developments in sensor miniaturization and data analysis AI are expected to enhance the accessibility and capabilities of both technologies, further solidifying their place in modern agricultural research [69] [75].
The advancement of precision agriculture relies heavily on technologies that enable early and accurate assessment of plant health. Among the emerging tools, color-changing chemical sensors and deep learning image analysis represent two distinct technological pathways. This guide provides an objective comparison of these approaches for researchers and scientists engaged in plant health assessment research, focusing on their operational principles, performance characteristics, and implementation requirements. The analysis is framed within the broader thesis of validating color-changing sensor performance against established deep learning methods, with supporting experimental data from recent studies.
Color-Changing Sensors
Color-changing sensors for plant health monitoring represent a biomimetic approach inspired by natural mechanisms. Researchers at Northeastern University developed sensors based on the nesocodon flower, which changes color when its nectar interacts with proline—a universal biomarker for plant stress [3]. These paper-based sensors are embedded with sinapaldehyde and undergo a dose-dependent color change from pale yellow to bright red when exposed to elevated proline concentrations in stressed plants [3].
The underlying operating principle involves a chemical reaction where proline mixes with sinapaldehyde to form nesocodin, a natural red pigment [3]. This reaction enables direct detection of plant stress biomarkers through simple color transitions. The methodology involves clipping a small piece of plant leaf, grinding it, adding ethanol to extract proline, and then dipping the sensors into the liquid—a process requiring approximately 15 minutes [3]. This approach is particularly valuable for small-scale farming operations and developing regions where access to expensive analytical equipment is limited [3].
Deep Learning Image Analysis
Deep learning image analysis employs computational models to interpret visual data for plant health assessment. Convolutional Neural Networks (CNNs) form the foundation of many such systems, using multi-layer neural networks to automatically extract features and perform classification and prediction tasks [76]. These systems process high-resolution plant images acquired through various means including digital cameras, unmanned aerial vehicles (UAVs), and 3D scanning technologies [76].
The operating principle centers on automated feature extraction and pattern recognition. Models such as U-Net and EfficientNetV2 can perform high-precision segmentation and rapid classification of plant images [77]. More advanced implementations incorporate heatmap generation algorithms that leverage ensemble learning, attention mechanisms, and deep feature fusion techniques to highlight areas of interest in images, such as diseased tissue regions [77]. Unlike color-changing sensors that detect specific biomarkers, deep learning approaches typically identify visual patterns correlated with plant health status, including morphological changes, color variations, and texture modifications.
Table 1: Fundamental Operating Principles
| Feature | Color-Changing Sensors | Deep Learning Image Analysis |
|---|---|---|
| Core Principle | Chemical reaction with stress biomarkers [3] | Computational pattern recognition in visual data [76] |
| Detection Target | Specific biomarkers (e.g., proline) [3] | Visual symptoms (morphology, color, texture) [76] |
| Data Acquisition | Direct leaf sampling and extraction [3] | Image capture (cameras, UAVs, satellites) [76] |
| Output | Color change (qualitative) or scanned intensity (quantitative) [3] | Classification scores, segmentation maps, heatmaps [77] |
Performance Comparison
Accuracy and Sensitivity
Color-changing sensors demonstrate high specificity for detecting proline-related stress responses. In testing across cabbage, kale, brussel sprouts, and broccoli, these sensors successfully identified stressed plants through distinct color changes, providing both qualitative visual assessment and quantitative analysis when scanned [3]. The biomarker-based approach enables detection of stress before visible symptoms appear, offering potential for early intervention.
Deep learning image analysis achieves high accuracy in various plant monitoring tasks. CNN models have demonstrated exceptional performance in species identification, achieving 97.3% accuracy on the Brazilian wood image database and 96.4% on the Xylarium Digital Database [76]. For plant node tracking in growth monitoring, advanced deep learning approaches have achieved average prediction errors of less than 4 cm, even when nodes are temporarily obscured by leaf movement [78]. In stress identification, multimodal LSTM frameworks integrating molecular and phenotypic features have reached 97% accuracy in predicting drought stress [79].
Processing Time and Throughput
The sample processing time for color-changing sensors is approximately 15 minutes from leaf sampling to result, excluding the plant stress induction period [3]. This makes the technology suitable for point-of-use testing but potentially limiting for large-scale field monitoring unless multiple samples are processed simultaneously.
Deep learning image analysis offers significantly higher throughput once models are deployed. Automated phenotyping systems can process images continuously, with one study capturing images at 1-hour intervals to track plant growth dynamics [78]. The computational inference time is typically seconds or less per image, especially when using optimized models like YOLO for real-time detection [79].
Environmental Adaptability and Limitations
Color-changing sensors maintain functionality across various lighting conditions since the color change is inherent to the chemical reaction [3]. However, they require physical contact with plant tissue and destructive sampling, which may not be suitable for continuous monitoring of the same plants over time. Researchers are exploring biodegradable sensor options to address environmental concerns [3].
Deep learning image analysis is non-invasive and suitable for continuous monitoring but faces challenges with varying lighting conditions, occlusion, and complex backgrounds that can affect accuracy [76]. Studies have developed specialized techniques to handle these issues, such as predicting hidden node locations using clustering and linear interpolation when leaves obscure nodes [78].
Table 2: Performance Metrics Comparison
| Parameter | Color-Changing Sensors | Deep Learning Image Analysis |
|---|---|---|
| Stress Detection Accuracy | High for proline-related stress (qualitative) [3] | Up to 97% for drought stress classification [79] |
| Species Identification Accuracy | Not species-dependent; detects universal biomarker [3] | 97.3% on specialized datasets [76] |
| Processing Time | ~15 minutes per sample (extraction + detection) [3] | Seconds per image after model deployment [78] |
| Spatial Resolution | Leaf segment level | Sub-centimeter (varies with imaging system) [78] |
| Early Detection Capability | High (biomarker-level detection) [3] | Moderate (requires visible symptoms) [76] |
Experimental Protocols
Color-Changing Sensor Methodology
The experimental protocol for color-changing sensors follows these key steps:
- Sample Collection: Clip a small piece (approximately 1-2 cm²) from a leaf of the plant to be tested [3].
- Tissue Disruption: Grind the leaf segment into smaller pieces using a mortar and pestle or mechanical homogenizer to release intracellular contents.
- Proline Extraction: Add ethanol to the ground tissue to extract proline and other soluble compounds. The mixture is typically allowed to sit for 5-10 minutes with occasional agitation [3].
- Sensor Exposure: Dip the color-changing sensor strip into the extraction liquid for a predetermined time, usually 1-2 minutes.
- Result Interpretation: Compare the sensor color against a reference chart or scan the sensor with a smartphone for quantitative analysis. Bright red indicates high proline concentration and plant stress, while pale yellow indicates healthy plants [3].
For quantitative analysis, researchers can use color models such as RGB, HSV, or CIELAB to convert the sensor color into digital values. Data preprocessing methods like normalization have been shown to improve predictive performance in colorimetric analysis [4].
Deep Learning Image Analysis Methodology
A typical experimental protocol for deep learning-based plant health assessment includes:
- Image Acquisition: Capture plant images using consistent parameters (resolution, lighting, angle, distance). High-resolution cameras, UAVs, or smartphones may be used depending on the application [76].
- Data Preprocessing: Apply techniques including cropping, resizing, color normalization, and background suppression to standardize inputs. Data augmentation methods like random rotation and flipping are employed to increase dataset diversity and improve model robustness [76].
- Feature Extraction: Utilize CNN architectures to automatically extract relevant features from the preprocessed images. Alternative approaches may employ handcrafted features including color histograms, texture analysis, or morphological descriptors [76].
- Model Training: Train deep learning models (e.g., CNNs, RNNs, Transformers) using annotated datasets. Transfer learning with pretrained models on large datasets like ImageNet can reduce training time and data requirements [79].
- Validation and Testing: Evaluate model performance on separate validation and test sets using metrics such as accuracy, precision, recall, and F1-score. For growth tracking, average prediction error (e.g., in cm for node location) is a relevant metric [78].
Implementation Requirements
Technical Infrastructure
Color-changing sensors require minimal technical infrastructure, making them suitable for resource-limited settings. The essential requirements include the sensors themselves, basic tools for leaf sampling (scissors or punches), ethanol for extraction, and containers for sample processing [3]. For quantitative analysis, a smartphone or flatbed scanner can be used to digitize color data.
Deep learning image analysis demands significantly more technical resources, including image acquisition systems (digital cameras, UAVs, or satellites), computational hardware for model training and inference (typically GPUs), and software frameworks for implementing deep learning algorithms [76]. The system complexity ranges from simple smartphone-based applications to sophisticated multi-camera setups for high-throughput phenotyping.
Data and Training Needs
Color-changing sensors require no ongoing data collection or training once developed and validated. Each test provides immediate results without reference to historical data, though establishing baseline proline levels for specific plant varieties may improve interpretation accuracy.
Deep learning approaches require substantial datasets for training, with recommended sizes varying from 1,000-2,000 images per class for binary classification to 5,000 images per object for complex detection tasks [76]. Data annotation remains labor-intensive and often represents a bottleneck in model development. Transfer learning can reduce data requirements to 100-200 images per class in some cases [76].
Cost Analysis
Color-changing sensors offer low per-test costs after initial development, with materials estimated at minimal expense. The technology does not require specialized equipment or technical expertise for deployment, making it economically viable for small-scale farmers and developing regions [3].
Deep learning image analysis involves significant upfront costs for imaging systems, computational infrastructure, and model development. However, once established, the marginal cost per analysis is negligible, making it economically favorable for large-scale operations. The cost-effectiveness depends on the scale of implementation and the required accuracy and throughput.
Table 3: Implementation Requirements Comparison
| Requirement | Color-Changing Sensors | Deep Learning Image Analysis |
|---|---|---|
| Initial Development Cost | Low (material synthesis) | High (model development, data collection) [76] |
| Per-Analysis Cost | Low (consumable sensors) [3] | Very low (after system deployment) |
| Technical Expertise Needed | Basic laboratory skills [3] | Advanced programming and data science [76] |
| Equipment Requirements | Minimal (basic lab tools) [3] | Significant (cameras, computing hardware) [76] |
| Data Requirements | None for operation | Thousands to millions of labeled images [76] |
| Scalability | Limited by manual sampling | High (automated image processing) [78] |
The Scientist's Toolkit
Research Reagent Solutions
Table 4: Essential Research Materials
| Item | Function | Application Context |
|---|---|---|
| Sinapaldehyde-embedded Paper Sensors | Reacts with proline to produce color change [3] | Color-changing sensor development |
| Proline Standard Solutions | Quantification and calibration of sensor response [3] | Sensor validation and standardization |
| Ethanol Extraction Solvent | Extracts proline from plant tissue [3] | Sample preparation for sensor analysis |
| Color Reference Chart | Provides qualitative assessment standard [3] | Field interpretation of sensor results |
| Pre-trained Plant Models | Transfer learning for specific plant health tasks [79] | Deep learning image analysis |
| Data Augmentation Pipelines | Increases effective dataset size [76] | Deep learning model training |
| Annotation Software | Creates ground truth data for training [76] | Dataset preparation for deep learning |
| Color Calibration Targets | Standardizes color across imaging conditions [4] | Colorimetric analysis in both approaches |
This comparative analysis demonstrates that color-changing sensors and deep learning image analysis offer complementary strengths for plant health assessment. Color-changing sensors provide a low-cost, specific, and accessible approach for detecting plant stress at the biomarker level, particularly suitable for small-scale operations and resource-limited settings [3]. Deep learning image analysis offers high-throughput, non-invasive monitoring capabilities with strong accuracy across various plant health applications, making it ideal for large-scale agricultural operations and research institutions with technical resources [76].
The choice between these technologies depends on specific research requirements, including scale of operation, available resources, required accuracy, and desired throughput. Future research directions may explore hybrid approaches that combine the specificity of chemical sensing with the scalability of image analysis, potentially through AI-enhanced colorimetric interpretation [7] or integrated sensor-camera systems.
Real-World Trial Outcomes and Long-Term Reliability Assessments
The adoption of color-changing sensor technology for plant health assessment represents a significant advancement in precision agriculture and plant phenotyping. However, for researchers and drug development professionals, the translation of novel sensor technologies from controlled laboratory environments to diverse field conditions presents substantial validation challenges. This comparison guide provides an objective evaluation of three distinct sensor methodologies for plant health assessment, focusing specifically on their real-world trial outcomes and long-term reliability metrics. As these technologies evolve toward commercial application, understanding their operational parameters, environmental dependencies, and performance limitations becomes critical for research validation and implementation planning.
Each technology profiled herein employs a fundamentally different mechanism to assess plant physiological status, from direct biomarker detection to indirect spectral analysis. The framework for validation requires standardized assessment across multiple dimensions, including accuracy under variable field conditions, operational longevity, and practical implementation requirements. This analysis synthesizes experimental data from recent peer-reviewed research to enable informed technology selection for specific research applications ranging from high-throughput phenotyping to small-scale precision agriculture trials.
Technology Comparison: Performance Metrics and Field Trial Outcomes
Table 1: Comprehensive Comparison of Plant Health Sensor Technologies
| Evaluation Parameter | Northeastern University Proline Sensor | NC State University Hyperspectral Polarimetry | Tohoku University Leaf-Mounted Sensor |
|---|---|---|---|
| Detection Principle | Colorimetric proline biomarker detection [3] | Polarized hyperspectral imaging [47] | Multispectral leaf color monitoring [60] |
| Primary Measured Indicator | Proline concentration (stress biomarker) [3] | Vegetation indices (GNDVI, RERR) [47] | Chlorophyll content & Photochemical Reflectance Index [60] |
| Real-World Testing Environment | Laboratory leaf samples (cabbage, kale, broccoli) [3] | Maize field (multiple diurnal cycles) [47] | Outdoor birch trees (2-week autumn period) [60] |
| Key Performance Outcomes | Qualitative color change (yellow to red); Quantitative via scanning [3] | 10x error reduction in glare conditions (3% vs. 30% error) [47] | Accurate chlorophyll tracking; Correlation with commercial spectrometers [60] |
| Longevity Assessment | Single-use (15-minute test) [3] | Continuous monitoring capable [47] | >1 month continuous operation [60] |
| Stressor Detection Range | Drought, heavy metals, heat, excessive light [3] | Glare compensation under field conditions [47] | Seasonal change, light intensity fluctuations [60] |
| Data Output Format | Visual color change + quantitative scanning [3] | Corrected vegetation indices [47] | 8 wavelength channels with wireless data transfer [60] |
Table 2: Practical Implementation Characteristics
| Characteristic | Northeastern University Proline Sensor | NC State University Hyperspectral Polarimetry | Tohoku University Leaf-Mounted Sensor |
|---|---|---|---|
| Target User | Small farms, developing regions, houseplant owners [3] | Large-scale research breeding programs [47] | Smart farming, forest health studies [60] |
| Sample Processing | Destructive (leaf clip required) [3] | Non-contact imaging [47] | Non-destructive, in-situ monitoring [60] |
| Scalability | Low (manual processing required) [3] | High (drone/platform deployment potential) [47] | Medium (sensor network deployment) [60] |
| Cost Profile | Low-cost materials (paper-based) [3] | High (specialized camera equipment) [47] | Affordable (multiple sensor deployment feasible) [60] |
| Automation Potential | Low (manual sample processing) [3] | High (automated data pipeline) [47] | High (continuous wireless monitoring) [60] |
Experimental Protocols and Methodologies
Proline Detection Assay Protocol
The Northeastern University research team developed a standardized protocol for plant stress assessment using their color-changing sensor technology [3]. The methodology begins with tissue collection, where researchers clip a small piece (approximately 1cm²) from the leaf of the target plant. The sample is then mechanically ground using a mortar and pestle or mechanical homogenizer to increase surface area. Subsequently, 5mL of ethanol solvent is added to extract proline and other soluble compounds from the plant tissue through a 10-minute incubation period with periodic agitation.
The paper-based sensor, embedded with sinapaldehyde, is immersed in the ethanolic extract for 2-5 minutes to allow the chemical reaction between proline and sinapaldehyde to proceed. This reaction forms nesocodin, the same red pigment found in the nesocodon flower [3]. The resulting color development is assessed either qualitatively by visual comparison to a reference color card or quantitatively using flatbed scanner imaging with RGB profiling analysis. The intensity of the red color formation correlates directly with proline concentration, serving as a biomarker for plant stress levels.
Hyperspectral Polarimetry Validation Methodology
The NC State University approach employed a sophisticated validation methodology to quantify performance improvements in glare reduction [47]. Researchers utilized a mast-mounted hyperspectral imaging polarimeter (HIP) to capture multi-angular imagery of a maize field across multiple diurnal cycles throughout a growing season. Concurrently, they employed a multistatic fiber-based Mueller matrix bidirectional reflectance distribution function (mmBRDF) instrument to measure the polarized light-scattering behavior of individual maize leaves in controlled laboratory conditions.
The data from these instruments was fitted to existing models using SCATMECH, with output parameters informing Monte Carlo simulations [47]. These simulations generated training data for a shallow neural network that learned to correlate polarized data from low-reflectivity bands with unpolarized two-band vegetation indices. Validation occurred through comparative analysis of standard vegetation indices (GNDVI and Red-edge Reflection Ratio) calculated from both raw sensor data and software-corrected outputs, with ground truth measurements establishing baseline accuracy.
Continuous Monitoring Field Trial Protocol
Tohoku University researchers conducted rigorous field validation of their leaf-mounted sensor system across multiple dimensions [60]. Initial laboratory calibration involved testing approximately 90 leaves from 30 different plant species, comparing sensor readings against a commercial spectrometer across eight detectable wavelengths. Specifically, readings at 620 nm were validated against commercial chlorophyll meters, while 550 nm measurements were correlated with the Photochemical Reflectance Index (PRI).
For real-world performance assessment, researchers attached sensors to birch leaves to track autumn senescence dynamics over a two-week period [60]. The sensor system continuously monitored changes in chlorophyll content and leaf reflectance while simultaneously recording environmental parameters including light intensity. Data was wirelessly transmitted to a base station for temporal analysis, enabling researchers to correlate sensor readings with visual indicators of leaf aging and stress responses to fluctuating sunlight conditions.
Signaling Pathways and Detection Mechanisms
Biomarker Detection Pathway
Glare Correction Methodology
Research Reagent Solutions and Essential Materials
Table 3: Key Research Reagents and Materials for Plant Sensor Development
| Reagent/Material | Function/Purpose | Technology Application |
|---|---|---|
| Sinapaldehyde | Reacts with proline to form colored nesocodin pigment [3] | Proline detection sensors |
| Paper-based substrate | Low-cost medium for chemical sensor immobilization [3] | Disposable proline test strips |
| Hyperspectral imaging polarimeter | Captures spectral data while measuring light polarization states [47] | Glare-resistant field phenotyping |
| Mueller matrix BRDF instrument | Measures polarized light-scattering behavior of leaf surfaces [47] | Sensor calibration and validation |
| Spectroscopic sensor with light source | Measures leaf color changes without blocking sunlight [60] | Continuous leaf monitoring sensors |
| Wireless data transmission module | Enables remote data collection from multiple sensor nodes [60] | Field-based sensor networks |
| Ethanol solvent | Extracts proline and soluble compounds from plant tissue [3] | Sample preparation for proline assay |
| SCATMECH software library | Models light-scattering behavior for algorithm development [47] | Polarimetry data processing |
The comparative assessment of these three sensor technologies reveals distinct pathways for plant health monitoring, each with particular strengths for different research scenarios. The Northeastern University proline sensor provides exceptional specificity as a direct biomarker detection system but requires destructive sampling, making it ideal for controlled experiments validating plant stress responses [3]. The NC State hyperspectral approach offers sophisticated compensation for environmental variables like glare, delivering research-grade accuracy for large-scale phenotyping applications but at higher implementation complexity [47]. The Tohoku University continuous monitoring sensor strikes a balance with its non-destructive, long-term deployment capability suitable for longitudinal studies of plant physiology under changing environmental conditions [60].
For the research community, selection criteria should prioritize measurement specificity versus operational scalability based on experimental objectives. Proline detection delivers unambiguous stress biomarker data but with limited temporal resolution. Hyperspectral systems provide comprehensive vegetation assessment with advanced environmental compensation but require substantial computational resources. Continuous leaf-mounted sensors enable temporal tracking of plant responses but measure indirect indicators of stress rather than specific biomarkers. Future developments will likely integrate these complementary approaches, creating multimodal sensing platforms that leverage both specific biomarker detection and continuous physiological monitoring for validated plant health assessment across basic research and applied agricultural science.
Conclusion
Color-changing sensors represent a paradigm shift in plant health monitoring, offering a unique combination of accessibility, rapid results, and cost-effectiveness, particularly for smaller-scale operations. Validation studies confirm their strong correlation with key stress biomarkers like proline, demonstrating reliable performance across various crop types. While challenges in environmental interference and quantitative precision persist, ongoing optimization in sensor design, supported by advanced software and micro-nano technologies, is rapidly addressing these limitations. When compared to established technologies like hyperspectral imaging and deep learning models, these sensors carve a distinct niche through their simplicity and low cost rather than outright replacement. The future of this technology lies in the development of multimodal, fully biodegradable sensors integrated with IoT platforms, paving the way for widespread adoption in smart farming. For researchers in biomedical and clinical fields, the underlying principle of non-invasive, colorimetric biomarker detection offers a compelling model for developing novel diagnostic tools for physiological stress and disease states.
References
- 1. Colorimetric Biosensors: Advancements in Nanomaterials ... [https://www.mdpi.com/2079-6374/15/6/362]
- 2. Color changing sensors: A multimodal system for ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993619302614]
- 3. These Color-Changing Sensors Could Revolutionize Agriculture [https://news.northeastern.edu/2025/08/01/color-changing-sensors-northeastern-research/]
- 4. Investigation into the predictive performance of colorimetric ... [https://jast-journal.springeropen.com/articles/10.1186/s40543-021-00271-9]
- 5. A comparison of optical, electrochemical, magnetic, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6736529/]
- 6. application to color-changing biochemical assays - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11744771/]
- 7. Artificial intelligence and machine learning for colorimetric ... [https://www.sciencedirect.com/science/article/pii/S2214158825000236]
- 8. An artificial intelligence-assisted microfluidic colorimetric ... [https://www.nature.com/articles/s41528-024-00321-3]
- 9. Proline and ROS: A Unified Mechanism in Plant ... [https://www.mdpi.com/2223-7747/14/1/2]
- 10. Comprehensive analysis of proline metabolizing genes ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12558507/]
- 11. Salinity stress amelioration through selenium and zinc ... [https://www.nature.com/articles/s41598-025-12106-3]
- 12. NC State Scientists Fast-Track R&D for Sensors That Detect ... [https://cals.ncsu.edu/psi/news/nc-state-scientists-fast-track-rd-for-sensors-that-detect-plant-diseases-early/]
- 13. Review Wearable plant sensing devices for health monitoring [https://www.sciencedirect.com/science/article/pii/S2950235725000101]
- 14. Progress in Transcriptomics and Metabolomics in Plant ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12191765/]
- 15. Crop management to enhance plant resilience to abiotic ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1728599/full]
- 16. Molecular Basis of Plant Stress Tolerance: Current Status ... [https://www.mdpi.com/1467-3045/47/11/918]
- 17. Students' color-changing tomato reaches national contest finals [https://news.cornell.edu/stories/2025/09/students-color-changing-tomato-reaches-national-contest-finals]
- 18. Recent advances in wearable and implantable ... [https://www.sciencedirect.com/science/article/pii/S0165993625002043]
- 19. Biosensor Technologies for Early Detection and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8207201/]
- 20. Experimental Protocol for Color Difference Evaluation ... [https://www.mdpi.com/2313-433X/11/1/4]
- 21. Smartphone-integrated paper sensor strips for rapid and ... [https://www.sciencedirect.com/science/article/pii/S1010603025004150]
- 22. Colorimetric sensor array for versatile detection and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11034120/]
- 23. Comparison of multiple plant sensors aimed at early ... [https://www.sciencedirect.com/science/article/pii/S0378377425002495]
- 24. Active and Passive Electro-Optical Sensors for Health ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7795220/]
- 25. Paper-Based Sensors: Emerging Themes and Applications [https://pmc.ncbi.nlm.nih.gov/articles/PMC6164297/]
- 26. Evaluation of Low-Cost Multi-Spectral Sensors ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11991415/]
- 27. A hyperspectral plant health monitoring system for space ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2023.1133505/full]
- 28. Wearable Standalone Sensing Systems for Smart Agriculture [https://pmc.ncbi.nlm.nih.gov/articles/PMC12021045/]
- 29. An ensemble model of machine learning regression ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d4ra07510b]
- 30. Eco-friendly paper-based colorimetric sensor for portable ... [https://link.springer.com/article/10.1007/s13762-025-06636-6]
- 31. Comprehensive plant health monitoring: expert-level ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1511651/full]
- 32. Recent advances in plant disease detection: challenges and ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-025-01450-0]
- 33. Plant extraction for LC-MS Analysis [https://www.protocols.io/view/plant-extraction-for-lc-ms-analysis-g3dvbyi67.html]
- 34. Leaf Extract Analysis [https://apical-ag-home.com/leaf-extract-analysis/]
- 35. Where do qualitative assessments fit in an era of ... [https://www.sciencedirect.com/science/article/pii/S0190052821000729]
- 36. Qualitative and Quantitative Descriptors for Plant Morphology [https://pmc.ncbi.nlm.nih.gov/articles/PMC5289971/]
- 37. New Software Boosts Accuracy of Tech to Measure Crop Health [https://cals.ncsu.edu/news/new-software-boosts-accuracy-of-tech-to-measure-crop-health/]
- 38. Genomic analyses reveal the stepwise domestication and ... [https://www.nature.com/articles/s41588-024-01744-4]
- 39. Germplasm Enhancement and Identification of Loci Conferring ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9498593/]
- 40. Analysis of plant physiological responses based on leaf ... [https://www.sciencedirect.com/science/article/pii/S2214180424000709]
- 41. A hyperspectral plant health monitoring system for space ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10352677/]
- 42. Field Plant Monitoring from Macro to Micro Scale [https://www.mdpi.com/2223-7747/12/22/3851]
- 43. Visual management of medical things with an advanced ... [https://www.nature.com/articles/s41598-021-02501-x]
- 44. Biocompatible, biodegradable, and high-performance ... [https://www.sciencedirect.com/science/article/abs/pii/S2211285525003891]
- 45. AdapTree: Data-Driven Approach to Assessing Stress Through... Plant [https://pmc.ncbi.nlm.nih.gov/articles/PMC12115627/]
- 46. Software improves sensors that check crop health - Futurity [https://www.futurity.org/sensors-leaf-color-plant-health-crops-3197732/]
- 47. New Software Boosts Accuracy of Tech to Measure Crop Health [https://news.ncsu.edu/2024/03/new-software-boosts-accuracy-of-tech-to-measure-crop-health/]
- 48. Mitigating Illumination-, Leaf-, and View-Angle Dependencies ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10959007/]
- 49. Mitigating Illumination-, Leaf-, and View-Angle ... [https://www.sciencedirect.com/science/article/abs/pii/S2643651524002814]
- 50. Unlocking plant health survey data: An approach to quantify ... [https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1012957]
- 51. Recent Advances in Micro- and Nano-Enhanced Intravascular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12349352/]
- 52. Biosensors | Special Issue : Feature Papers in Nano [https://www.mdpi.com/journal/biosensors/special_issues/SRJ8716RX4]
- 53. Scientists unveil leaf-mounted sensors to monitor plant ... [https://www.agtechnavigator.com/Article/2024/12/05/scientists-unveil-leaf-mounted-sensors-to-monitor-plant-health-in-real-time/]
- 54. A new, low-cost ground-based NDVI sensor for manual ... [https://www.sciencedirect.com/science/article/pii/S277237552500125X]
- 55. A True-Color Sensor and Suitable Evaluation Algorithm for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5579884/]
- 56. Low-cost and automated phenotyping system ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10152611/]
- 57. 30 Key Advantages of Wireless Network for Businesses in ... [https://www.techvertu.co.uk/blog/infrastructure-and-networking/30-key-advantages-of-wireless-networks-2025]
- 58. Top 10 predictions for Wi-Fi in 2025 [https://www.iotinsider.com/industries/communications/top-10-predictions-for-wi-fi-in-2025/]
- 59. An Experimental Comparison of Basic Device Localization ... [https://www.mdpi.com/2673-8732/5/2/11]
- 60. New Smart Agriculture Technology for Monitoring Plants [https://www.asiaresearchnews.com/content/new-smart-agriculture-technology-monitoring-plants]
- 61. Evolving landscape of wireless sensor networks: a survey ... [https://link.springer.com/article/10.1007/s42452-025-07070-6]
- 62. Enhancing the effectiveness of wireless sensor networks ... [https://www.nature.com/articles/s41598-025-10813-5]
- 63. Exploration of CIELAB colour system for the colorimetric ... [https://www.sciencedirect.com/science/article/pii/S2211715625007696]
- 64. A Comparative Study of Optical Sensing Methods for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12251617/]
- 65. A Comparative Study of Optical Sensing Methods for ... [https://www.mdpi.com/1424-8220/25/13/3850]
- 66. Paper-based colorimetric sensor for histamine detection ... [https://www.nature.com/articles/s41598-025-15873-1]
- 67. Paper-based colorimetric sensor for easy and simple ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267020303974]
- 68. Visual colorimetric immunosensor for sensitive detection of 4 ... [https://onehealthadv.biomedcentral.com/articles/10.1186/s44280-024-00062-y]
- 69. In Agriculture Market: Hyperspectral Advances... Imaging 2025 [https://farmonaut.com/blogs/hyperspectral-imaging-in-agriculture-market-2025-advances]
- 70. A direct comparison of a next generation hyperspectral ... [https://www.sciencedirect.com/science/article/pii/S1386142524012344]
- 71. Transformative Impact of Hyperspectral on Imaging ... - Specim Plant [https://www.specim.com/case-phenotrait/]
- 72. Frontiers | Hyperspectral segmentation of plants in fabricated... [https://www.frontiersin.org/journals/high-performance-computing/articles/10.3389/fhpcp.2025.1547340/full]
- 73. A Performance Evaluation of Two Hyperspectral Imaging ... [https://www.mdpi.com/1424-8220/24/1/174]
- 74. Multispectral Vs. Hyperspectral Imaging: Differences And ... [https://eos.com/blog/multispectral-vs-hyperspectral-imaging/]
- 75. What is Hyperspectral (HSI) In Agricultural? Uses, How It... Imaging [https://www.linkedin.com/pulse/what-hyperspectral-imaging-hsi-agricultural-uses-how-works-bhiie]
- 76. The pipelines of deep learning-based plant image ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12304785/]
- 77. Deep learning methods for improving the accuracy and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11736776/]
- 78. Tracking plant growth using image sequence analysis [https://www.sciencedirect.com/science/article/pii/S2949798125000407]
- 79. Application of Deep Learning Technology in Monitoring ... [https://www.mdpi.com/2071-1050/17/17/7602]