Sustaining Life in the Void: The Critical Roles of Producers, Consumers, and Decomposers in Bioregenerative Life Support Systems (BLSS)
This article provides a comprehensive analysis of the functional roles and integration of producers, consumers, and decomposers within Bioregenerative Life Support Systems (BLSS).
Sustaining Life in the Void: The Critical Roles of Producers, Consumers, and Decomposers in Bioregenerative Life Support Systems (BLSS)
Abstract
This article provides a comprehensive analysis of the functional roles and integration of producers, consumers, and decomposers within Bioregenerative Life Support Systems (BLSS). Tailored for researchers, scientists, and drug development professionals, it explores the foundational ecology of these systems, methodologies for their application, strategies for troubleshooting microbial contamination and system imbalances, and comparative validation from historical and current BLSS experiments. The synthesis offers critical insights for advancing closed-loop ecosystem research and its potential implications for biomedical applications in isolated environments.
The Ecological Engine: Understanding the Core Principles of BLSS Trophic Dynamics
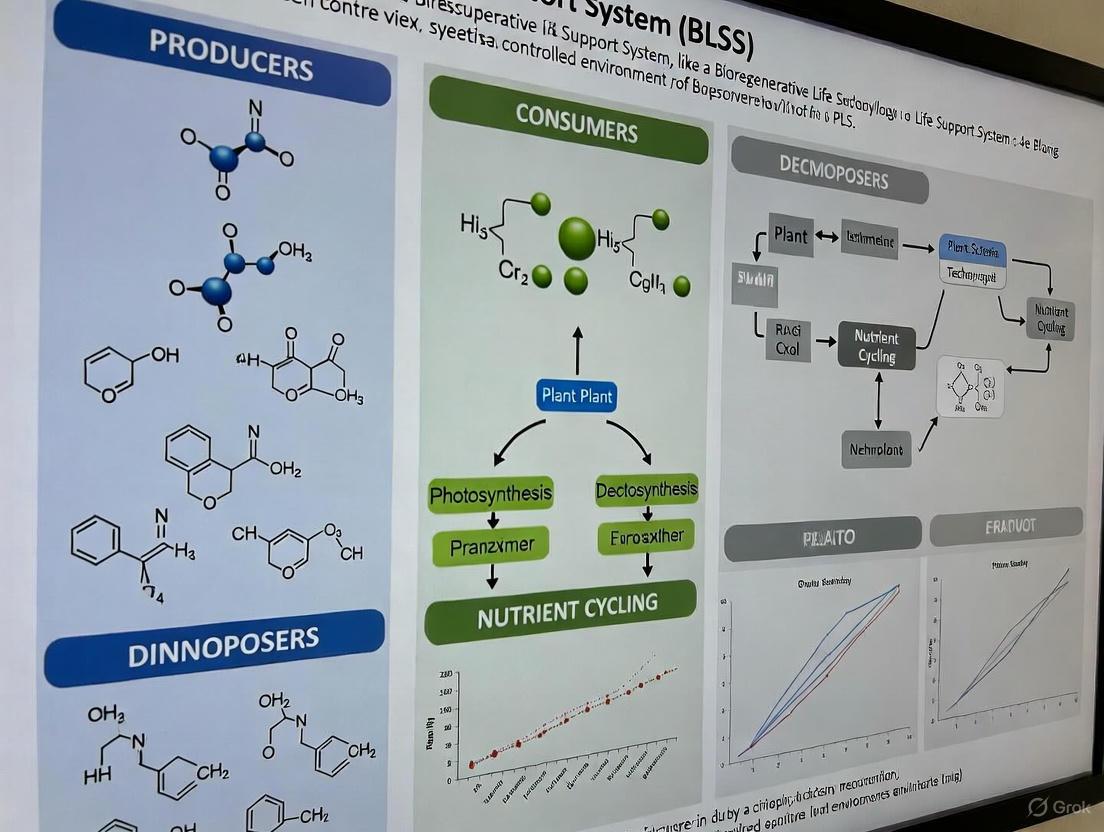
The development of self-sustaining environments for long-duration space exploration represents one of the most formidable challenges in aerospace science. Bioregenerative Life Support Systems (BLSS) are engineered ecosystems designed to regenerate air, water, and food for crewed missions through the integration of biological processes [1]. The fundamental operational principle of any BLSS hinges on the precise coordination and optimization of three core biological components: producers, consumers, and decomposers. These elements form a functional "trinity" that enables the continuous cycling of matter and flow of energy within a closed system, mirroring the processes found in Earth's biosphere [2] [3].
The objective of a BLSS is to achieve a high degree of resource circularity, minimizing the need for external resupply. This is accomplished by orchestrating the metabolic functions of various organisms to create a synergistic cycle: producers (typically plants and algae) generate food and oxygen through photosynthesis, consumers (astronauts and potentially animals) utilize these products and produce waste, and decomposers (bacteria and fungi) break down waste into simple nutrients that producers can reuse [1] [3]. The successful integration of these three groups is therefore not merely an academic exercise but a critical engineering requirement for humanity's future beyond Earth.
Theoretical Foundations of Ecosystem Trinity
Fundamental Roles and Interactions
In ecological terms, producers, consumers, and decomposers constitute the hierarchical trophic levels that govern energy transfer and nutrient cycling in all biological systems [3]. Their defined roles and interactions form the bedrock upon which BLSS architecture is built.
Producers (Autotrophs): These organisms form the foundation of the ecosystem by converting light energy into chemical energy via photosynthesis. They are the primary source of organic matter and oxygen within a BLSS. The simplified photosynthetic equation is:
6CO₂ + 6H₂O + Light Energy → C₆H₁₂O₆ + 6O₂[3]. Examples relevant to BLSS include higher plants (e.g., wheat, lettuce, potatoes), algae (e.g., Chlorella, Spirulina), and cyanobacteria [1] [3].Consumers (Heterotrophs): These organisms cannot produce their own food and must consume other organisms to obtain energy and nutrients. In the context of a BLSS, the primary consumers are the human crew. Their role is to consume the biomass and oxygen produced by the autotrophs, and in turn, they release carbon dioxide and metabolic waste, which serve as inputs for other system components [3].
Decomposers (Saprotrophs): Primarily bacteria and fungi, decomposers are the "recyclers" of the ecosystem. They catalyze the breakdown of complex dead organic matter (e.g., inedible plant biomass, human waste) into simple inorganic molecules (e.g., ammonium, phosphate, CO₂) [3]. This process is vital for returning essential nutrients to the production cycle, thereby closing the elemental loops (carbon, nitrogen, phosphorus) and maintaining soil (or substrate) fertility within the BLSS [3].
The interaction between these three components creates a circular flow of matter, while energy flows unidirectionally, entering as light and exiting as heat. This relationship is fundamental to achieving the self-sustainability goals of a BLSS [2].
The "Ecosystem Core" Hypothesis in BLSS Context
The "Ecosystem Core" hypothesis provides a valuable theoretical model for understanding BLSS dynamics. This hypothesis posits that the abiotic component of an ecosystem—the "Resource Core"—provides the essential matter and energy that serve as the nucleus for biological activity [2]. The biotic components (producers, consumers, decomposers) are organized around this core, similar to electrons surrounding an atomic nucleus.
In a BLSS, the Resource Core consists of the initial and ongoing inputs of:
- Material Reservoirs: Water, nutrient salts, and atmospheric gases (O₂, CO₂).
- Energy Inputs: Light (typically artificial) and, in some cases, chemical energy [2].
When artificial energy and controlled matter inputs are introduced into the system, they alter the state of the Resource Core. This perturbation forces the biological community to re-organize, effectively driving ecosystem evolution from a natural state to an engineered, intelligent state tailored for human life support [2]. The stability of the entire BLSS depends on managing these inputs and the interactions between the living components that depend on them.
Quantitative Analysis of Trophic Components in BLSS
The design and modeling of a BLSS require a quantitative understanding of the biomass, energy, and nutrient flows between the trophic components. System resilience is heavily dependent on the size and buffering capacity of material reservoirs that interface with the biological processors [4].
Table 1: Key Quantitative Parameters for Trophic Components in BLSS Design
| Trophic Component | Primary Function | Key Input Metrics | Key Output Metrics | Efficiency Considerations |
|---|---|---|---|---|
| Producers (Plants, Algae) | O₂ production, food biomass, water purification | Light intensity (PPFD), CO₂ concentration, nutrient load | Edible biomass yield, O₂ evolution rate, transpiration rate | Light use efficiency, harvest index, CO₂ fixation rate |
| Consumers (Crew) | CO₂ production, waste regeneration, system maintenance | Caloric intake, O₂ consumption, water intake | CO₂ excretion, metabolic waste (urine, feces), greywater | Respiration quotient, specific waste production rates |
| Decomposers (Bacteria, Fungi) | Nutrient recycling, waste processing | Organic waste load, C:N ratio of feedstock | Mineralized N/P/K, CO₂ production, biomass | Degradation rate constants, nutrient recovery efficiency |
Energy transfer between these trophic levels is inherently inefficient. In natural ecosystems, only about 10% of energy is transferred from one trophic level to the next, with the remainder lost as heat during metabolic processes [3]. This principle necessitates that a BLSS be designed with a large producer base relative to consumer mass to meet energy demands.
Table 2: Material Flow Budget in a Conceptual BLSS (per crew member per day)
| Material | Consumer Output (Waste) | Decomposer Processing | Producer Input Requirement |
|---|---|---|---|
| Carbon | ~1.0 kg CO₂ | CO₂ released to atmosphere | ~1.0 kg CO₂ fixed via photosynthesis |
| Water | ~1.5 L urine, ~3.0 L greywater | Nitrification, filtration | ~4.5 L for transpiration/nutrient delivery |
| Nitrogen | ~15-20 g in urine & solids | Mineralization to Ammonium/Nitrate | ~15-20 g for plant biomass synthesis |
| Inedible Biomass | ~0.3 kg plant waste | Solid-state decomposition | Nutrients recycled for new growth |
BLSS Implementation: Integration and Case Studies
Historical and Current BLSS Platforms
The translation of trophic theory into functional BLSS platforms has been demonstrated in several ground-based experimental facilities. These systems serve as testbeds for integrating the trinity of producers, consumers, and decomposers.
- MELiSSA (Micro-Ecological Life Support System Alternative): Developed by the European Space Agency, MELiSSA is a comprehensive benchmark for BLSS research. It is a closed-loop system with multiple compartments that simulate the functions of producers (photoautotrophic bacteria, higher plants), consumers (crew), and decomposers (anaerobic and aerobic microbial reactors) in a highly controlled manner. Its pilot plant facility allows for the study of coordination and control strategies between these compartments [1].
- The CEBAS (Closed Equilibrated Biological Aquatic System): This German-developed system is an aquatic BLSS that has successfully maintained a closed equilibrium between aquatic plants (producers), snails (consumers), and microbial communities (decomposers) for extended periods. It demonstrates the viability of aquatic modules for bioregenerative life support [1].
- Aquarack: An early closed-system bioreactor that successfully sustained the photosynthetic flagellate Euglena gracilis (a producer) using real-time monitoring, highlighting the importance of process control for maintaining the stability of a core biological component [1].
Methodological Framework for BLSS Experimentation
Research into the integration of trophic components follows a systematic workflow. The diagram below outlines a generalized experimental protocol for developing and testing a BLSS module.
BLSS Experimental Workflow
Detailed Experimental Protocol:
Selection of Biological Species:
- Producers: Choose candidate species based on high harvest index, short life cycle, and high edibility (e.g., wheat, potato, lettuce, Spirulina). Criteria include photosynthetic efficiency, O₂ yield, and resistance to closed-environment stressors [1].
- Decomposers: Select microbial consortia from waste water treatment or composting research. Isolation and screening are performed for efficient degradation of target waste streams (human waste, inedible biomass) and tolerance to system conditions [1].
Design of Physical Architecture:
- Develop a modular hardware design where each trophic processor (plant growth chamber, waste bioreactor, crew habitat) interfaces primarily with shared material storage reservoirs (for water, O₂, CO₂, nutrients). This uses the reservoirs as system-wide buffers to coordinate the asynchronous dynamics of the biological components [4].
Establishment of Control Parameters:
- Define and calibrate setpoints for critical environmental variables: light intensity (PPFD: 200-600 µmol m⁻² s⁻¹), photoperiod (16:8 light:dark), atmospheric CO₂ (1000-5000 ppm), temperature (22-25°C), and nutrient solution composition (e.g., Hoagland's solution) [1].
System Integration, Closure, and Monitoring:
- Seal the system and initiate material flows. Monitor in real-time: gas concentrations (O₂, CO₂), pressure, temperature, humidity, and microbial load. Track biomass accumulation, waste processing rates, and nutrient levels in the hydroponic solution [1].
Failure Mode Simulation and Buffer Analysis:
- Conduct stress tests by inducing partial or complete failures of system components (e.g., plant chamber light failure, waste processor halt). Monitor the transient responses and the effectiveness of material reservoirs as buffers, determining the minimum required buffer sizes for mission scenarios [4].
The Scientist's Toolkit: Essential Research Reagents and Materials
The study and development of BLSS components rely on a suite of specialized reagents, instruments, and materials.
Table 3: Essential Research Tools for BLSS Experimentation
| Category / Item | Specification / Example | Primary Function in BLSS Research |
|---|---|---|
| Producer Growth Media | Hoagland's Nutrient Solution, BG-11 for Cyanobacteria | Provides essential macro and micronutrients (N, P, K, Ca, S, Mg, Fe, Mn, Zn, etc.) for autotrophic growth. |
| Decomposer Inoculum | Activated Sludge, Thermophilic Compost Extract | Introduces a diverse microbial community capable of degrading complex organic wastes into mineral nutrients. |
| Gas Analysis Instrument | Infrared CO₂ Analyzer, Paramagnetic O₂ Sensor | Precisely monitors the gas exchange ratios (CO₂ consumption, O₂ production) to gauge system metabolic balance. |
| Water Quality Probes | pH & EC (Electrical Conductivity) Meters, Ion-Selective Electrodes | Monitors the chemical environment of hydroponic solutions and waste streams to maintain optimal conditions. |
| Hydroponic Systems | Nutrient Film Technique (NFT), Deep Water Culture (DWC) | Provides the physical substrate and delivery mechanism for water and nutrients to plant roots in a soil-free environment. |
| Sterilization Agents | 70% Ethanol, Sodium Hypochlorite Solution | Maintains microbiological control within the system and prevents contaminant overgrowth during initial setup. |
The trinity of producers, consumers, and decomposers forms an indivisible functional unit at the heart of Bioregenerative Life Support Systems. The theoretical understanding of their interactions, guided by principles such as the Ecosystem Core hypothesis, provides the foundation for engineering these complex closed ecosystems. Quantitative modeling and experimental platforms like MELiSSA have demonstrated the feasibility of integrating these components to achieve a degree of resource circularity.
However, significant challenges remain on the path to a fully operational BLSS. Future research must focus on the precise integration of diverse systems for synchronous recycling, achieving source-sink balance for waste streams, and developing robust control strategies to manage the inherent dynamics of biological systems [1]. Furthermore, the translation of mature, centralized bioprocesses into decentralized, self-sustaining ecosystems will require advances in automation, monitoring, and the selection of even more efficient biological species. The continued study of producers, consumers, and decomposers—the fundamental trinity of life—will be critical in enabling humanity to create self-sustaining habitats wherever we venture.
In Bioregenerative Life Support Systems (BLSS), producers form the foundational trophic level, tasked with converting external energy into chemical energy to sustain human and heterotrophic life. These systems, essential for long-duration space missions, rely on a closed-loop ecology where "producers" (plants, microalgae, photosynthetic bacteria), "consumers" (crew), and "degraders" (microorganisms) are interconnected, with the waste of one compartment serving as the vital resource for another [5]. The autonomy of future lunar or Martian outposts hinges on the efficient performance of these biological producers, which provide three critical functions: food production, air regeneration (CO₂ consumption and O₂ production), and water purification [5]. This whitepaper examines the molecular physiology, experimental performance data, and practical integration of photosynthetic and chemosynthetic primary producers within the BLSS context.
Photosynthetic Producers: Harnessing Light Energy
Higher Plants in BLSS
Higher plants are central to BLSS concepts, as they directly produce food, regenerate air, and recycle water. Their cultivation in space must be optimized for different mission scenarios, ranging from short-duration "salad machines" providing fresh, nutritious supplements to long-duration missions requiring staple crops like wheat and potato for caloric needs [5].
Light Quality and Photoreceptor Signaling The effects of light quality on plant physiology are mediated by specialized photoreceptors. Cryptochromes (CRYs) and Phototropins (PHOTs) are the primary blue light (400–500 nm) receptors, regulating processes from phototropism to stress responses [6]. CRYs are flavin-binding proteins located in the nucleus, cytoplasm, chloroplasts, and mitochondria, influencing growth, development, and ROS generation [6]. PHOTs are plasma membrane-associated receptors with LOV (Light-Oxygen-Voltage) domains that undergo autophosphorylation upon blue light exposure; they mediate chloroplast movement, stomatal opening, and oxidative stress adaptation [6]. The signaling pathways initiated by these receptors are summarized below.
Figure 1: Blue Light Perception and Signaling in Plants. Blue light activates cryptochrome and phototropin photoreceptors, triggering downstream signaling cascades that regulate key physiological processes relevant to BLSS performance.
Experimental Evidence: Light Quality Effects on Plant Physiology Controlled studies demonstrate the significant impact of light spectra on plant growth and function. The following table synthesizes key quantitative findings from research on sweet pepper and saffron, highlighting the synergistic effects of combined red and blue light.
Table 1: Effects of Light Quality on Growth and Photosynthetic Parameters in Plants
| Plant Species | Light Treatment | Key Growth Findings | Key Photosynthetic Findings | Source |
|---|---|---|---|---|
| Sweet Pepper (Capsicum annuum) | Red (R), Blue (B), Mixed (RB) | Highest shoot dry weight under RB. Thickest leaves & mesophyll under RB. Lowest biomass under R. | Highest Pn(max), AQE, ETR under RB. Highest Rubisco & Calvin cycle enzyme activity under RB. | [7] |
| Saffron (Crocus sativus) | Monochromatic B (100%B) to R (0%B) | Heaviest daughter corms (key yield) under high B (75-100%B). Biomass directed to corms/flowers under B light. | Best photosynthetic functionality (OJIP), efficient e⁻ transport, lower energy dissipation under B light. | [8] |
Microalgae: Efficient Single-Celled Phototrophs
Microalgae, such as Chlorella vulgaris, represent a highly efficient photosynthetic compartment for BLSS, offering rapid growth and high nutritional value.
Optimizing Microalgae Cultivation with LED Light Research on Chlorella vulgaris has identified optimal cultivation parameters using blue LED light. The following experimental protocol summarizes the methodology for establishing and assessing microalgae growth under controlled light conditions.
Figure 2: Experimental Workflow for Microalgae Cultivation Optimization. This protocol outlines the key steps for determining the optimal light conditions for Chlorella vulgaris growth and lipid production, critical parameters for BLSS integration.
The systematic optimization of light parameters yields clear results. Under optimal conditions (blue LED light at 200 μmol m⁻² s⁻¹ with a 12:12 hour light-dark cycle), Chlorella vulgaris achieves a high specific growth rate of 1.20 per day and a significantly increased cell dry weight, alongside enhanced lipid content essential for biofuel and nutritional applications [9]. This demonstrates the potential for tuning microalgae cultivation to maximize productivity in a BLSS.
Chemosynthetic Producers: Energy from Inorganic Molecules
Chemosynthesis presents an alternative primary production strategy for environments with limited light, such as within specific modules of a BLSS or as a ground-based analogy for cave ecosystems on other planets.
Aerotrophy: Primary Production from Atmospheric Trace Gases
Recent metagenomic and biogeochemical profiling of aerated cave ecosystems has revealed a novel and widespread form of chemosynthesis termed 'aerotrophy,' which could inform BLSS design [10].
Metagenomic Findings from Cave Ecosystems Analysis of 1,458 metagenome-assembled genomes (MAGs) from limestone and basalt caves showed that over half of the microbial cells encode enzymes to use atmospheric trace gases as energy and carbon sources [10]. The most abundant primary producers were:
- Candidatus Methylocavales (a gammaproteobacterial methanotrophic order): Uses atmospheric methane (CH₄).
- Uncultivated actinobacterial genera: Predicted to grow on atmospheric hydrogen (H₂), carbon dioxide (CO₂), and carbon monoxide (CO).
Biogeochemical measurements confirmed rapid consumption of these gases, sustaining a substantial fraction of the community and driving primary production through the Calvin-Benson-Bassham cycle [10]. This process operates continuously, independent of light, making it a compelling model for stable BLSS processes.
Table 2: Key Enzymes and Substrates for Atmospheric Trace Gas Consumption in Cave Microbiomes
| Energy Substrate | Key Enzyme | Phyla Encoding Enzyme | Approx. % of Community (Reads) | Role in Primary Production |
|---|---|---|---|---|
| Carbon Monoxide (CO) | Form I CO Dehydrogenase | 11 different phyla | 74.3% | Energy liberation & CO₂ fixation |
| Hydrogen (H₂) | Group 1/2 [NiFe]-Hydrogenase | 14 different phyla | 43.0% | High-affinity H₂ oxidation for energy |
| Methane (CH₄) | Particulate Methane Monooxygenase | - | 5.5% | Dual energy & carbon source |
The Scientist's Toolkit: Research Reagent Solutions
The following table details essential reagents, materials, and instruments used in the experimental research cited in this whitepaper, providing a resource for protocol replication and further investigation.
Table 3: Key Research Reagents and Materials for BLSS Producer Studies
| Reagent / Material / Instrument | Function / Application | Example Use Case |
|---|---|---|
| Hoagland & Arnon Nutrient Solution | Standardized plant nutrition in hydroponic systems. | Fertigation of saffron corms in perlite medium [8]. |
| Bold's Basal Medium (BBM) | Cultivation of fresh water microalgae. | Axenic culture of Chlorella vulgaris [9]. |
| Blue & Red LED Light Modules | Precisely controlled light quality for plant physiology studies. | Testing effects of R:B ratios on pepper and saffron [8] [7]. |
| Chlorophyll Fluorometer (e.g., FluorPen) | Measures photosynthetic efficiency (PSII) via OJIP transient. | Assessing photosynthetic functionality in saffron leaves [8]. |
| Clark-type Oxygen Microsensor | Measures O₂ concentration & gross photosynthesis at micro-scales. | Profiling O₂ gradients in photosynthetic microbial mats [11]. |
| Isotope Ratio Mass Spectrometry (IRMS) | Quantifies ¹³C incorporation into biomass to measure net primary productivity. | Tracking ¹³C-HCO₃⁻ fixation in microbial mat layers [11]. |
The integration of producers within a BLSS is a complex, multi-faceted challenge requiring a deep understanding of organismal physiology, system ecology, and bioprocess engineering. Photosynthetic producers, primarily higher plants and microalgae, remain the most advanced option, providing essential ecosystem services alongside nutritional and psychological benefits. The optimization of their growth through light quality control is a mature field with direct applications. Meanwhile, chemosynthetic producers, especially aerotrophic microbes utilizing atmospheric trace gases, represent a frontier in BLSS research with the potential to enhance system robustness and matter recycling. Future research must focus on the integration of these diverse producer groups into a stable, resilient, and efficient closed ecosystem capable of supporting human life beyond Earth.
Bioregenerative Life Support Systems (BLSS) are engineered ecosystems designed to sustain human life in isolated environments, such as space habitats, by regenerating essential resources through biological processes. The core principle of a BLSS is the synergistic integration of producers, consumers, and decomposers to create a closed-loop system for the exchange of materials and energy [1]. Within this framework, the "consumer niche" encompasses all organisms that are unable to produce their own food and must source energy and carbon from other organisms. This niche ranges from human crewmembers (macro-consumers) to various microfauna (micro-consumers), each playing a distinct and vital role in the system's stability and efficiency [1]. Understanding the trophic dynamics and material transfer between these consumer levels is paramount for designing resilient and self-sustaining BLSS. This whitepaper provides an in-depth technical guide to the consumer niche, detailing quantitative relationships, experimental methodologies for their study, and essential research tools.
Quantitative Analysis of Consumer-Driven Material and Energy Flows
The efficient functioning of a BLSS depends on quantifying the flows of key elements like carbon and nitrogen through its consumer tiers. The following tables synthesize data on consumption rates, metabolic outputs, and elemental turnover for major consumer groups within a hypothetical BLSS.
Table 1: Mass and Energy Balance for Key Consumer Groups in a BLSS.
| Consumer Group | Avg. Biomass (kg) | Daily O₂ Consumption (L/person/day) | Daily CO₂ Production (kg/person/day) | Daily Water Consumption (L/person/day) | Daily Caloric Intake (kCal) | Solid Waste Production (kg/person/day) |
|---|---|---|---|---|---|---|
| Human Crewmember | 70 | 600 [1] | 1.0 [1] | 3.0 | 2800 | 0.2 |
| Aquatic Food Web (Fish) | Variable | - | - | - | - | - |
| Microfauna (in soil/substrate) | Variable | - | - | - | - | - |
Table 2: Trophic Niche Metrics for Aquatic and Terrestrial Consumers in a BLSS.
| Consumer Species/Group | Trophic Level | Primary Carbon Source(s) | Contribution to Biomass (%) | Nitrogen Excretion Rate (mg/kg/day) | Trophic Niche Width (δ¹³C Range ‰) |
|---|---|---|---|---|---|
| Omnivorous Fish Species | 2.5 - 3.5 | Phytoplankton, Epiphytes, Detritus [12] | 25-40% | 350-500 | 4.5 - 6.5 |
| Detritivorous Invertebrates | 2.0 | Mangrove/Plant Detritus, Microbes [12] | 10-20% | 150-300 | 3.0 - 5.0 |
| Microfauna (e.g., Nematodes) | 2.0 - 3.0 | Bacteria, Fungal Biomass | <5% | 50-150 | 2.0 - 4.0 |
Experimental Protocols for Analyzing Consumer Trophic Dynamics
A rigorous, data-driven approach is essential for mapping the consumer niche. The following protocols outline standardized methodologies for investigating these complex relationships.
Stable Isotope Analysis for Trophic Positioning and Carbon Flow
Objective: To determine the trophic level of consumers and identify the primary sources of carbon assimilated into their biomass [12].
Materials:
- Freeze-drier
- Analytical balance (precision ±0.001 mg)
- Ball mill or mortar and pestle
- Elemental analyzer coupled to an isotope ratio mass spectrometer (EA-IRMS)
- Tin capsules for solid samples
Procedure:
- Sample Collection: Collect tissue samples (e.g., muscle tissue from fish, whole body for macroinvertebrates) from target consumers and potential basal resources (plant matter, algae, detritus, suspended particulate organic matter).
- Preparation: Rinse samples with deionized water to remove contaminants. Freeze-dry samples to constant weight to remove all moisture.
- Homogenization: Grind dried samples to a fine, homogeneous powder using a ball mill or mortar and pestle.
- Weighing: Precisely weigh 0.5 to 1.0 mg of powdered sample into tin capsules.
- Isotope Analysis: Introduce the encapsulated samples into the EA-IRMS. The instrument combusts the sample, separates the resulting gases (N₂, CO₂), and measures the ratio of heavy to light isotopes (¹³C/¹²C and ¹⁵N/¹⁴N).
- Data Processing: Express isotope ratios in delta (δ) notation in parts per thousand (‰) relative to international standards. Calculate trophic position using established formulas incorporating δ¹⁵N baseline values. Use Bayesian mixing models (e.g., MixSIAR, SIAR) to estimate the proportional contributions of different basal resources to the consumer's diet [12].
Trophic Niche Width Assessment using Bayesian Ellipses
Objective: To quantify and compare the trophic niche space and functional diversity of different consumer assemblages within the BLSS.
Materials:
- Stable isotope data (δ¹³C and δ¹⁵N) for all consumer individuals
- Statistical computing environment (e.g., R)
Procedure:
- Data Compilation: Compile a dataset of δ¹³C and δ¹⁵N values for all sampled individuals from each consumer group or study site.
- Model Calculation: In R, use the
SIBER(Stable Isotope Bayesian Ellipses in R) package. The model calculates the standard ellipse area (SEA) for each group, which contains approximately 40% of the data and provides a measure of the core trophic niche. - Bayesian Estimation: Run a Bayesian iteration (e.g., 10,000 posterior draws) to estimate the SEA for each group (SEA(_B)). This accounts for uncertainty based on sample size.
- Comparison: Calculate the percentage of overlap between the posterior distributions of SEA(_B) for different consumer groups. A high overlap suggests similar trophic niche utilization, while low overlap indicates niche segregation [12].
Respiration and Metabolic Rate Measurement
Objective: To quantify the oxygen consumption and carbon dioxide production rates of non-human consumers, critical for BLSS gas exchange calculations.
Materials:
- Respirometry chambers of appropriate size
- Dissolved oxygen (DO) probe or optical oxygen sensor
- CO₂ probe or gas chromatograph
- Data-logging system
- Temperature-controlled water bath (for aquatic specimens)
Procedure:
- Acclimation: Acclimate the test organisms to the experimental conditions (temperature, light) for 24 hours.
- Baseline Measurement: Fill the respirometry chamber with water/air from the acclimation environment and record the initial DO and CO₂ concentrations.
- Experimental Run: Introduce a known biomass of the test organism(s) into the chamber. Seal the chamber to prevent gas exchange with the outside environment.
- Monitoring: Log the changes in DO and CO₂ concentrations over time (typically 30-120 minutes). Ensure the duration is within the organism's tolerance and before oxygen levels become critically low.
- Calculation: The metabolic rate is determined from the slope of the linear regression of oxygen concentration against time, corrected for chamber volume and organism biomass.
Workflow Visualization of Trophic Analysis
The following diagram illustrates the integrated experimental workflow for analyzing the consumer niche, from sample collection to data interpretation.
Experimental Workflow for Trophic Analysis
Research Reagent Solutions and Essential Materials
A well-equipped toolkit is fundamental for conducting research on consumer niches in BLSS. The following table details key reagents and materials, along with their specific functions in the experimental protocols.
Table 3: Research Reagent Solutions and Essential Materials for Trophic Ecology Studies.
| Item Name | Function/Application | Technical Specification |
|---|---|---|
| Tin Capsules | Encapsulation of solid samples for introduction into the Elemental Analyzer. | High-purity tin, 4x6 mm or 5x9 mm size, pre-cleaned. |
| International Isotope Standards | Calibration of the Isotope Ratio Mass Spectrometer (IRMS) to ensure data accuracy and inter-laboratory comparability. | Certified reference materials (e.g., USGS40, IAEA-N-2) for δ¹³C and δ¹⁵N. |
| Ultra-Pure Helium Gas | Carrier gas for the Elemental Analyzer and Isotope Ratio Mass Spectrometer. | 99.999% purity, with integrated oxygen/moisture traps. |
| Laboratory Gases | Creation of calibration curves and instrument tuning. | Certified mixtures of CO₂ in He and N₂ in He. |
| Freeze-Dryer (Lyophilizer) | Removal of water from biological samples without altering their isotopic composition. | Capability to reach -50°C or lower, with a vacuum of <0.01 mBar. |
| Optical Oxygen Sensor Spots | Non-consumptive measurement of dissolved oxygen in respirometry experiments. | Pre-calibrated sensor spots compatible with fiber-optic readout devices. |
| R Statistical Software with Packages | Data analysis, including calculation of trophic metrics and running Bayesian mixing models. | Installation of core R and packages such as SIBER, MixSIAR, and siar. |
In the context of Bioregenerative Life Support Systems (BLSS), which aim to create sustainable environments for long-duration space missions, the efficient cycling of matter and energy is paramount. These systems rely on the synergistic integration of producers (plants), consumers (animals, humans), and decomposers (microbes and invertebrates) to regenerate air, water, and food from waste materials [13]. While producers and consumers have been extensively studied, decomposers often represent the most underutilized yet critical link in closing the loop within a BLSS. This whitepaper provides an in-depth technical examination of decomposers as nature's ultimate recyclers, framing their function within the constraints and requirements of BLSS research. We detail their mechanistic roles in nutrient cycling, experimental methodologies for quantifying their activity, and the visualization of key processes essential for integrating these biological agents into robust, sustainable engineered ecosystems.
The Fundamental Role of Decomposers in Ecosystems
Decomposers, primarily consisting of fungi, bacteria, and invertebrates (collectively known as the "FBI"), are a guild of organisms that catalyze the breakdown of dead organic matter into simpler inorganic substances [14] [15]. This process, known as decomposition, is the cornerstone of nutrient cycling, making essential elements like carbon, nitrogen, and phosphorus available for uptake by primary producers [16] [17].
- Nutrient Cycling: Decomposers transform dead material into essential nutrients such as nitrogen, phosphorus, and potassium, which are then absorbed by plant roots [14]. This process maintains soil fertility, which in turn supports plant growth and the entire food web [14]. In a BLSS, this translates to the continuous regeneration of plant growth substrates from inedible biomass and human waste.
- Soil Health and Structure: As decomposers break down organic material, they enrich the soil with humus, a dark organic component that improves soil structure, water retention, and fertility [14]. Furthermore, soil fauna like earthworms improve soil aeration and aggregation, creating an ideal environment for microbial activity and root growth [17].
- Carbon Cycling: Decomposers play a significant role in the global carbon cycle by breaking down organic carbon in dead material and releasing carbon dioxide (CO₂) back into the atmosphere through cellular respiration [13] [14]. In the closed atmosphere of a BLSS, understanding and managing this CO₂ flux is critical for maintaining optimal levels for plant photosynthesis.
- Waste Processing and Disease Control: By breaking down dead organisms and organic waste, decomposers prevent the accumulation of material that could harbor pathogens, thereby helping to control the spread of disease within an ecosystem [14]. This function is of utmost importance in a closed-loop BLSS to maintain crew health and system hygiene.
The following table summarizes the primary groups of decomposers and their specific functions.
Table 1: Key Decomposer Groups and Their Functional Roles in Ecosystems
| Organism Group | Examples | Specific Functions |
|---|---|---|
| Fungi | Mushrooms, Molds | Break down complex organic polymers like cellulose and lignin; essential in forest ecosystems for decomposing wood [14]. |
| Bacteria | Various species (e.g., Bacillus, Pseudomonas) | Microscopic decomposers crucial for nutrient cycling, especially in transforming nitrogen into plant-available forms [14] [17]. |
| Earthworms | Lumbricus terrestris | Detritivores that consume soil and organic matter, improving aeration and structure through burrowing; release nutrient-rich castings [14]. |
| Detritivorous Insects | Beetles, Ants, Millipedes | Fragment large pieces of organic material (e.g., leaf litter, carcasses), increasing surface area for microbial decomposition [16] [14]. |
Decomposers in the BLSS Context: Closing the Loop
A BLSS mimics Earth's natural ecosystems by creating a web of processes where the waste from one group of organisms becomes a resource for another. In this framework, decomposers are the critical link that closes the nutrient loop, converting waste streams into fertilizers.
The fundamental principle of energy flow, governed by the First Law of Thermodynamics, underpins this process. Energy cannot be created or destroyed, only transformed [13]. In a BLSS, solar energy is captured by producers (plants) via photosynthesis and converted into chemical energy stored in biomass. This energy then flows through consumers and is finally processed by decomposers, which release it as heat back into the system while transforming the matter into reusable forms [13].
Table 2: Role of Biological Components in a Simplified BLSS
| Biological Component | Primary Function in BLSS | Inputs | Outputs |
|---|---|---|---|
| Producers (Plants, Algae) | Photosynthesis: Convert light energy to chemical energy; produce food and O₂, consume CO₂ [13]. | CO₂, Water, Light, Nutrients | Food, O₂, Inedible Biomass |
| Consumers (Humans, Animals) | Cellular Respiration: Consume food and O₂ to produce energy; release CO₂ and waste [13]. | Food, O₂ | CO₂, Urine, Feces, Metabolic Waste |
| Decomposers (Fungi, Bacteria) | Decomposition & Respiration: Mineralize waste and inedible biomass into inorganic nutrients [16] [14]. | CO₂, Inedible Biomass, Solid & Liquid Waste | Inorganic Nutrients (N, P, K), CO₂ |
The diagram below illustrates the continuous flow of energy and matter between these components in an idealized BLSS.
Quantitative Analysis of Decomposition and Nutrient Release
The efficacy of decomposers in a BLSS must be quantified to model and manage nutrient fluxes. Key metrics include decomposition rates and the elemental composition of processed materials. These rates are influenced by factors such as temperature, moisture, the carbon-to-nitrogen (C:N) ratio of the waste, and the decomposer community structure [17].
For instance, studies in terrestrial ecosystems have shown that decomposition rates can accelerate with increased soil temperature, leading to higher concentrations of ammonium and organic carbon in the environment [17]. This principle is directly applicable to optimizing bioreactor conditions in a BLSS.
The table below provides a compositional analysis of cassava processing residues, representative of the agricultural waste that could be generated from a BLSS crop and valorized through decomposition.
Table 3: Composition of Cassava Processing Residues (g/100 g dry matter) [18]
| Residue Type | Crude Protein | Crude Fiber | Lipids | Ash | Moisture |
|---|---|---|---|---|---|
| Bagasse | 1.12 | 19.3 | 2.37 | 2.84 | 84.2 |
| Cassava Wastewater | 2.46 | - | - | 1.88 | 96.7 |
| Peels | 4.20 | 29.6 | 3.26 | 7.47 | 82.1 |
| Effluent | 2.92 | 6.69 | 1.75 | 3.16 | 91.4 |
Experimental Protocols for Monitoring Decomposition
Integrating decomposers into a BLSS requires rigorous experimental protocols to select, cultivate, and monitor decomposer communities and their functions. The following methodologies are adapted from both ecological research and biotechnology development for application in controlled environments.
Protocol: Measuring Microbial Decomposition Rates
Objective: To quantify the rate of organic matter breakdown and CO₂ respiration by a microbial consortium.
- Sample Preparation:
- Substrate: Prepare 10g (dry weight equivalent) of a standardized organic substrate (e.g., inedible plant biomass, sterilized human waste simulant) with a known C:N ratio.
- Inoculum: Source a defined microbial consortium (e.g., from commercial compost) or a specific mixture of fungi (Aspergillus niger) and bacteria (Bacillus subtilis).
- Bioreactor Setup:
- Use sealed, aerated bioreactors to maintain controlled conditions.
- Mix the substrate and inoculum in a 1:1 ratio (w/w) with moisture content adjusted to 60-70% water-holding capacity.
- Include control reactors with sterilized inoculum to account for abiotic degradation.
- Incubation and Monitoring:
- Incubate reactors at a constant temperature (e.g., 28°C).
- Continuously monitor CO₂ evolution using an infrared gas analyzer (IRGA). The cumulative CO₂ release is a direct indicator of microbial metabolic activity and decomposition rate [17].
- Endpoint Analysis:
- After a predetermined period (e.g., 30, 60, 90 days), destructively sample the material.
- Analyze the remaining material for mass loss, changes in C:N ratio, and concentrations of key plant-available nutrients (NH₄⁺, NO₃⁻, PO₄³⁻) via standardized soil extraction and colorimetric analysis (e.g., using an automated discrete analyzer).
Protocol: Vermicomposting for Solid Waste Processing
Objective: To utilize earthworms for the stable conversion of solid organic waste into high-quality fertilizer (vermicompost) for BLSS plant production units.
- Worm Farm Establishment:
- Species Selection: Use red wiggler worms (Eisenia fetida), known for high consumption and reproduction rates in organic waste [15].
- Bedding: Create a bedding layer of moistened, shredded cardboard or coconut coir in a ventilated container.
- Waste Introduction:
- Introduce a pre-composted mixture of inedible plant biomass and simulated human solid waste as feed. Pre-composting for ~1 week initiates microbial breakdown, making the material more palatable for worms.
- Bury the feed in the bedding, rotating feeding locations to prevent pile-up.
- System Maintenance:
- Maintain temperature at 15-25°C and moisture at 70-80%.
- Harvest vermicompost every 60-90 days using a migration method, where fresh feed is placed on one side to lure worms away from finished compost.
- Product Analysis:
- Analyze the harvested vermicompost for nutrient content, pH, electrical conductivity, and stability.
- Conduct plant growth trials (e.g., with lettuce) to compare the efficacy of vermicompost against synthetic nutrient solutions.
The workflow for establishing and monitoring a decomposer-based waste processing system is outlined below.
The Scientist's Toolkit: Research Reagent Solutions
Research into decomposer processes for BLSS applications requires specific biological and chemical reagents. The following table details key materials and their functions.
Table 4: Essential Research Reagents for Decomposer Studies
| Reagent / Material | Function and Application in Research |
|---|---|
| Defined Microbial Consortia (e.g., Bacillus subtilis, Pseudomonas aeruginosa, Aspergillus niger) | Used as model decomposers in controlled experiments to study the breakdown of specific waste streams (e.g., cassava residue, lignocellulose) and the production of target molecules like biosurfactants or enzymes [18]. |
| Earthworms (Eisenia fetida) | Key detritivores for vermicomposting protocols; used to process solid organic waste into stabilized, nutrient-rich humus for plant growth studies [15]. |
| Enzyme Assay Kits (Cellulase, Laccase, Protease) | Quantify extracellular enzyme activity in decomposing samples, providing a direct measure of the functional capacity of the microbial community to degrade key polymers (cellulose, lignin, proteins) [17]. |
| Nutrient Analysis Kits/Reagents (for NH₄⁺, NO₃⁻, PO₄³⁻) | Colorimetric reagents or test strips for rapid quantification of plant-available nutrients in compost leachates or extracted samples, enabling tracking of nutrient mineralization rates [18] [17]. |
| Selective Growth Media (e.g., Potato Dextrose Agar, Luria-Bertani Agar) | For the isolation, cultivation, and enumeration of specific decomposer groups (fungi vs. bacteria) from complex environmental samples like compost or soil [18]. |
Decomposers are not merely passive participants in ecosystems; they are dynamic engineers that drive the biogeochemical cycles essential for life. Their deliberate integration into Bioregenerative Life Support Systems is a prerequisite for achieving long-term sustainability and operational closure. By understanding their biological functions, quantifying their processing rates, and developing robust experimental protocols for their management, researchers can harness the power of these natural recyclers. The successful implementation of decomposer-based processes will ensure the efficient conversion of waste to resources, thereby supporting plant production, maintaining crew health, and ultimately enabling humanity's prolonged presence in space. Future research must focus on optimizing multi-species decomposer communities, integrating their processes with other BLSS subsystems, and validating their performance in long-duration, closed-system trials.
The Unidirectional Flow of Energy and the 10% Rule in BLSS Efficiency
The functioning of both natural ecosystems and engineered Bioregenerative Life Support Systems (BLSS) is governed by fundamental thermodynamic principles, chief among them being the unidirectional flow of energy through biological components. Unlike nutrients that can be recycled, energy flows in a single direction—entering the system primarily as solar radiation and exiting as waste heat, while being transformed and transferred through feeding relationships [19]. This energy transfer is characterized by significant inefficiencies, commonly generalized as the 10% rule, whereby only approximately 10% of the energy from one trophic level is transferred to the next [20] [21]. Understanding these principles is paramount for designing efficient BLSS that can sustainably support human life in extraterrestrial environments or in terrestrial applications focused on circular bioeconomy [1].
BLSS are artificial ecosystems that integrate producers, consumers, and decomposers to replicate Earth's biosphere principles, with the primary objective of providing sustainable food, oxygen, and water while recycling waste [22] [1]. These systems operate with open energy flow and closed material flow, making them critical technologies for long-duration space missions and resilient terrestrial communities [22] [1]. The efficiency of these systems is directly constrained by the unidirectional energy flow and the significant energy losses at each trophic transfer, which this review examines in detail.
Theoretical Foundations of Energy Flow
The Unidirectional Energy Pathway
In ecological terms, energy flow is the passage of energy through the different trophic levels of a biological community [19]. This process begins when primary producers (autotrophs) capture solar energy and convert it into chemical energy via photosynthesis [19] [21]. The generalized equation for photosynthesis is:
[ 6\text{CO}2 + 6\text{H}2\text{O} + \text{light energy} \rightarrow \text{C}6\text{H}{12}\text{O}6 + 6\text{O}2 ] [21]
This chemical energy, stored as biomass, then passes through the ecosystem via consumption. Primary consumers (herbivores) feed on producers, secondary consumers (carnivores) feed on herbivores, and so forth up the food chain [19] [20]. At each step, organisms utilize the consumed energy for metabolic processes such as growth, respiration, and reproduction [20].
A critical distinction exists between the flow of energy and nutrients. While nutrients cycle within the ecosystem through the action of decomposers that break down dead organic matter and waste, energy does not cycle [19]. Instead, it flows unidirectionally, with a substantial portion lost as heat at each transfer due to the second law of thermodynamics [19] [20]. This fundamental difference necessitates a constant input of energy (typically sunlight) to sustain the ecosystem.
The 10% Rule and Ecological Efficiency
The 10% rule is a ecological guideline stating that only about 10% of the energy from one trophic level is transferred and incorporated into the biomass of the next trophic level [20] [21]. The remaining 90% is lost primarily through:
- Undigested biomass that is excreted as waste [20]
- Energy expended as heat during respiration and other metabolic processes [19] [20]
- Organisms that die without being consumed by the next trophic level [20]
This energy loss can be mathematically modeled to understand the constraints it imposes on ecosystem structure. If primary producers capture 10,000 kJ of energy, the energy available to subsequent trophic levels would be:
| Trophic Level | Energy Available (kJ) | Calculation |
|---|---|---|
| Primary Producers | 10,000 | - |
| Primary Consumers | 1,000 | ( 10,000 \times 0.10 ) |
| Secondary Consumers | 100 | ( 1,000 \times 0.10 ) |
| Tertiary Consumers | 10 | ( 100 \times 0.10 ) |
Table 1: Mathematical modeling of energy transfer between trophic levels based on the 10% rule [21].
This exponential decline in available energy explains why food chains are typically limited to four or five trophic levels, as there is insufficient energy to support viable populations at higher levels [19] [20]. The 10% figure is a generalization; actual ecological efficiency varies based on organism types, metabolic rates, and environmental conditions [21]. In some marine ecosystems, for instance, energy transfer efficiency can exceed 10% [21].
Energy Flow in BLSS: Components and Interactions
Trophic Components of BLSS
BLSS are designed with three fundamental functional groups that facilitate energy flow and material recycling:
Producers (Autotrophs): These organisms form the foundation of BLSS by capturing light energy and converting it into chemical energy. Examples include higher plants (e.g., potatoes, Arabidopsis, cotton, rape), microalgae (e.g., Chlorella), and cyanobacteria [22] [1]. They are responsible for primary productivity, generating food for consumers, producing oxygen through photosynthesis, and consuming carbon dioxide [22].
Consumers (Heterotrophs): In BLSS contexts, humans are the primary consumers, though other consumer organisms may be included for dietary variety or ecological functions. Consumers ingest the chemical energy stored in producer biomass (or other consumer biomass) to sustain their metabolic processes [19] [1]. They return carbon dioxide to the system through respiration and produce organic waste [1].
Decomposers: Primarily bacteria and fungi, decomposers break down dead organic matter and consumer waste into inorganic nutrients [19] [1]. While they release some energy as heat during decomposition, their crucial role is in nutrient recycling, making elements like nitrogen and phosphorus available again for uptake by producers [19] [1].
Integration and Energy Flow Dynamics
In a successfully integrated BLSS, these components function synergistically in a closed-loop system [1]. The energy flow begins with producers capturing solar or artificial light energy. This energy is then transferred to consumers when they consume producer biomass. Throughout these transfers, the unidirectional energy flow principle applies, with significant energy losses at each step. Decomposers do not recapture the energy lost as heat but are essential for closing the nutrient cycle, allowing producers to continue capturing new energy [19].
Figure 1: Unidirectional Energy Flow in a BLSS. Energy (yellow arrows) enters as sunlight, is converted by producers, and flows inefficiently (~10% transfer) to consumers, with most lost as waste heat. Nutrients (gray dashed arrow) are recycled by decomposers to support producers.
The configuration of these components is strategically designed for the effective flow of materials and energy, with each species functioning in synergy with others to fulfil the objectives of the BLSS [1]. The primary functions within the ecosystem include energy capture and transformation, mineral retention and cycling, and cycle regulation and control [1].
Quantitative Analysis of Energy Transfer Efficiency
Energy Pyramids and Productivity Metrics
The flow of energy through a BLSS can be quantitatively represented using an energy pyramid, which visually depicts the rapid decrease in available energy at successive trophic levels [20] [21]. These pyramids are always upright due to the mandatory energy losses at each transfer [21].
The base of the energy pyramid is determined by the system's primary productivity:
- Gross Primary Productivity (GPP): The total rate at which producers capture and convert solar energy into chemical energy [19].
- Net Primary Productivity (NPP): The energy remaining after producers have used a portion of the GPP for their own respiration (GPP - Producer Respiration) [19]. NPP represents the actual energy available to primary consumers and forms the foundation of the energy budget for the entire BLSS [19].
NPP is influenced by various environmental factors, including light availability and quality, nutrient availability (often nitrogen and phosphorus), water availability, temperature, and carbon dioxide concentration [19] [22].
Factors Influencing Energy Transfer Efficiency
The overall ecological efficiency between trophic levels is a product of three component efficiencies [19]:
- Consumption Efficiency: The percentage of energy (or biomass) in a trophic level that is actually consumed by the next trophic level. This varies greatly between ecosystems and organism types.
- Assimilation Efficiency: The percentage of consumed energy that is assimilated (absorbed into the bloodstream) rather than egested as waste. Carnivores generally have higher assimilation efficiencies than herbivores because animal tissues are more digestible than plant tissues containing complex carbohydrates like cellulose.
- Production Efficiency: The percentage of assimilated energy that is converted into new biomass (growth and reproduction) versus being used for respiration. Ectotherms ("cold-blooded" animals) typically have higher production efficiencies than endotherms ("warm-blooded" animals) because they do not expend energy to maintain a constant body temperature.
These efficiencies combine to determine the overall ecological efficiency, which averages around 10% but can vary significantly [19].
| Factor | Impact on Energy Transfer Efficiency | BLSS Design Implication |
|---|---|---|
| Organism Metabolic Type | Endotherms (e.g., mammals) have lower production efficiency than ectotherms (e.g., fish, insects) due to higher metabolic heat loss [21]. | Consider incorporating ectothermic animals (e.g., fish in aquaponics) for protein production to improve overall system efficiency [1]. |
| Diet Quality | Animal-based biomass (for carnivores) is generally more digestible than plant-based biomass (for herbivores), leading to higher assimilation efficiency [19]. | While human diets will be primarily plant-based, the inclusion of some animal protein may improve energy and nutrient assimilation. |
| Food Web Complexity | Simple, linear food chains can be more efficient for targeting specific outputs, but complex webs may be more stable [19]. | Design for controlled simplicity, minimizing unnecessary trophic links that compound energy losses before reaching human consumers. |
| Environmental Conditions | Temperature, light intensity/spectrum, and stress factors affect metabolic rates and growth efficiency [19] [22]. | Precise environmental control is needed to optimize producer and consumer growth conditions and maximize productivity. |
Table 2: Factors affecting energy transfer efficiency and their implications for BLSS design.
Experimental Approaches and Methodologies
Ground-Based and Space-Based BLSS Experiments
Several pioneering experiments have demonstrated the feasibility of BLSS and provided critical data on energy flow and efficiency:
- BIOS-3 (Krasnoyarsk, Russia): A facility that incorporated humans into a closed material cycle, achieving a 91% closure of inner material, thereby demonstrating the viability of BLSS [22].
- Biosphere 2 (Arizona, USA): Contained multiple biomes (rainforest, ocean, savanna, desert, agricultural systems) and human habitats, conducting the most extended BLSS mission to date (730 days) [22].
- Lunar Palace 1 (China): A ground-based BLSS facility that achieved high rates of system closure, with successful integration of plant cultivation, food processing, and waste treatment [22] [1].
- Chang'e 4 Biological Experiment Payload (BEP): The first biological experiment on the lunar surface, which tested a micro-ecosystem containing producers (cotton, potato, Arabidopsis, and rape seeds), consumers (fruit fly eggs), and decomposers (yeast) under lunar microgravity and radiation conditions [22]. This experiment confirmed the critical need for thermal control, radiation shielding, and reliable energy input for extraterrestrial BLSS operation.
Protocol: Testing Producer Performance in Altered Gravity
Objective: To evaluate the germination, growth, and primary productivity of candidate plant species under lunar or Martian gravity conditions.
Materials:
- European Modular Cultivation System (EMCS) or equivalent facility on a space platform, allowing control of gravity levels (e.g., microgravity, 1/6 g, 1/2 g) [22].
- Sealed growth chambers with controlled atmosphere, temperature, and humidity.
- Seed varieties selected for BLSS (e.g., cotton, potato, Arabidopsis, rape) [22].
- Automated imaging system (CCD cameras) for monitoring growth and development [22].
- Nutrient delivery system (e.g., agar-based substrate or vermiculite with nutrient solution) [22].
- Light-emitting diodes (LEDs) to provide specific light wavelengths optimized for photosynthesis [22].
- Gas exchange analysis system to measure O₂ production and CO₂ consumption rates.
Methodology:
- Sterilization and Planting: Surface-sterilize seeds and aseptically plant them in the growth substrate within the sealed chamber.
- Environmental Control: Set and maintain temperature (~22-25°C), relative humidity (~70%), and CO₂ concentration (~400-1000 ppm). Initiate a controlled light/dark cycle (e.g., 16h/8h) using LEDs.
- Gravity Exposure: Activate the centrifuge to expose the plants to the target gravity level (e.g., 1/6 g for Moon). Include a 1 g control group.
- Watering and Nutrient Initiation: At landing/experiment start, release water into the growth medium to initiate germination [22].
- Data Collection:
- Continuous Imaging: Use CCD cameras to capture images at regular intervals to monitor germination rate, leaf expansion, and overall growth morphology [22].
- Gas Exchange Measurements: Periodically measure O₂ and CO₂ concentrations within the chamber to calculate net photosynthetic rate.
- Biomass Harvest: At the end of the experiment, harvest plant material, separate roots and shoots, and measure fresh and dry biomass to determine Net Primary Productivity (NPP).
- Data Analysis: Compare germination rates, growth rates, gas exchange parameters, and final biomass between different gravity treatments and the 1 g control.
Figure 2: Experimental workflow for testing plant growth in altered gravity.
The Scientist's Toolkit: Key Research Reagents and Materials
| Reagent/Material | Function in BLSS Energy Research |
|---|---|
| Candidate Plant Seeds (e.g., Potato, Cotton, Arabidopsis, Rape) | Act as primary producers to test viability, growth rates, and productivity (NPP) under controlled and space conditions [22]. |
| Microalgal Cultures (e.g., Chlorella, Euglena gracilis) | Serve as highly efficient primary producers in aquatic modules for O₂ production, CO₂ sequestration, and potential food source [22] [1]. |
| Inert Growth Substrate (e.g., Vermiculite, Agar) | Provides physical support for plant roots while allowing for controlled water and nutrient delivery in microgravity [22]. |
| Defined Nutrient Solutions | Deliver essential minerals (N, P, K, etc.) for plant growth, enabling precise study of nutrient cycling and its impact on primary productivity [22]. |
| Heterotrophic Microorganisms (e.g., Yeast, Specific Bacteria) | Function as decomposers to break down organic waste, facilitating nutrient recycling and closing the material loop [22] [1]. |
| Gas Analysis System | Measures O₂, CO₂, and other gas concentrations to quantify photosynthetic and respiratory rates, key indicators of energy flow efficiency [22]. |
Table 3: Essential research materials for studying energy flow in BLSS.
Implications for BLSS Design and Optimization
The principles of unidirectional energy flow and the 10% rule have profound implications for the design and management of BLSS:
Minimizing Trophic Levels: To reduce cumulative energy losses, human diets in BLSS should be primarily based on the lowest possible trophic level—primary producers (plants and algae) [19] [20]. Relying heavily on animal protein, which represents a secondary or tertiary trophic level, drastically reduces the overall energetic efficiency of the system.
Maximizing Primary Productivity: The entire energy budget of a BLSS is set by its NPP [19]. Design efforts must therefore focus on optimizing environmental conditions (light, CO₂, temperature, nutrients) to maximize the conversion of light energy into plant biomass [19] [22]. This includes selecting high-yield, fast-growing crop species and using efficient lighting such as LEDs with specific wavelength spectra.
Integrating Waste Recycling: While decomposers do not recapture lost energy, their role in nutrient recycling is essential for sustaining primary productivity without external inputs [1]. Effective waste management subsystems that convert inedible biomass and human waste into mineral nutrients are non-negotiable for long-term system closure [1].
System Monitoring and Control: Precise monitoring of energy flow (e.g., through gas exchange, biomass tracking) is critical for detecting imbalances and ensuring system stability [22] [1]. Automated controls are needed to adjust resource allocation (e.g., light, nutrients) to different components in response to real-time performance data.
In conclusion, the unidirectional flow of energy and the constraints of the 10% rule are not merely academic concepts but fundamental design parameters for Bioregenerative Life Support Systems. A deep understanding of ecological energetics allows researchers to configure systems of producers, consumers, and decomposers that maximize sustainable output for human life support, whether in space or in terrestrial applications inspired by circular economy principles.
Engineering the Cycle: Methodologies for Integrating Trophic Levels in BLSS
The producer compartment is a cornerstone of Bioregenerative Life Support Systems (BLSS), tasked with performing the essential functions of oxygen generation, food production, and carbon dioxide sequestration through photosynthesis. Within the closed-loop framework of a BLSS, which integrates producers, consumers (crew), and decomposers, the performance of the producer compartment directly determines the system's autonomy and resilience [5]. This guide provides a technical overview for researchers on the selection, cultivation, and integration of higher plants and microalgae, focusing on current advancements in photosynthetic efficiency and their application to long-duration space missions.
Scientific Foundation of Photosynthesis in BLSS
The core function of the producer compartment relies on the efficient operation of photosynthesis. Recent research has yielded significant insights into this process, particularly concerning its key enzyme and light-harvesting structures.
2.1 The Rubisco Bottleneck and Enhancement Strategies The enzyme Ribulose-1,5-bisphosphate carboxylase/oxygenase (Rubisco) is central to carbon fixation but is notoriously inefficient. It is slow, catalyzing only one to ten reactions per second, and has poor specificity for carbon dioxide over oxygen, leading to a wasteful process called photorespiration [23] [24]. In many crops, this inefficiency forces plants to dedicate up to 40% of their soluble leaf protein to Rubisco to compensate, representing a massive investment of nitrogen and energy [25] [24].
Several advanced strategies are being pursued to overcome this limitation:
- Directed Evolution of Rubisco: Researchers at MIT have used a continuous directed evolution technique (MutaT7) on a bacterial Rubisco, introducing mutations that improve its resistance to oxygen and boost its catalytic efficiency by up to 25% [23].
- Engineering CO₂-Concentrating Mechanisms (CCMs): Algae possess a specialized structure called the pyrenoid, which aggregates Rubisco and surrounds it with a high concentration of CO₂, effectively turbocharging the enzyme and nearly eliminating photorespiration [24]. The pyrenoid matrix is formed by a flexible linker protein, EPYC1, which binds multiple Rubisco molecules through a phase-separation mechanism [24]. Researchers have successfully engineered a "proto-pyrenoid" in the model plant Arabidopsis by introducing a hybrid plant-algal Rubisco and the EPYC1 linker protein, a major step toward creating a functional CCM in crops [24].
- Modular Nanocompartments: An alternative to complex natural CCMs is the use of encapsulins—simple, single-gene protein cages that can be reprogrammed to house different types of Rubisco. This modular system functions like a nanoscale office for the enzyme, simplifying the engineering process compared to installing natural carboxysomes [25].
2.2 Advanced Insights into Photosystem II The photosystem II (PSII) supercomplex, which performs the water-splitting reaction in oxygenic photosynthesis, exhibits a highly sophisticated design. Contrary to a simple funnel model, PSII in plants uses a "flat, sprawling energy landscape" that allows light energy to explore multiple routes before being locked into the photosynthetic process [26]. This design enables a dynamic balance between efficient photon harvesting and built-in photoprotection, preventing damage from excess light. Research from Lawrence Berkeley National Laboratory has revealed that energy within PSII undergoes a two-phase process: an initial "wandering phase" driven by entropy, followed by more targeted movement toward the reaction centers [26]. Understanding these principles is crucial for designing highly efficient producer compartments and could inform the engineering of crops with faster recovery from light stress, potentially boosting yields [26].
Selection Criteria for Producers in BLSS
The selection of species for the producer compartment is governed by the mission scenario, resource constraints, and the required functional output. The criteria differ significantly between higher plants and microalgae.
Table 1: Selection Criteria for Higher Plants in BLSS
| Criterion | Short-Duration Missions (e.g., LEO) | Long-Duration/Planetary Outposts (e.g., Mars) |
|---|---|---|
| Primary Role | Dietary complement, psychological benefits | Staple food supply, major resource recycling |
| Species Types | Leafy greens (e.g., lettuce, kale), microgreens, dwarf cultivars (e.g., tomato) [5] | Staple crops (e.g., wheat, potato, rice, soy), longer-cycle vegetables/fruits (e.g., peppers, beans, berries) [5] |
| Key Traits | Fast growth, minimal volume, high nutritive/antioxidant value [5] | High nutritional value, high edible biomass ratio, efficient resource use (water, nutrients) [5] |
| Resource Contribution | Minimal contribution to recycling; requires upload of seeds [5] | Substantial contribution to O₂ production, CO₂ removal, and water purification [5] |
Table 2: Selection Criteria and Potential of Microalgae in BLSS
| Criterion | Considerations & Examples |
|---|---|
| Cultivation Mode | Phototrophic (light as energy source), Mixotrophic (utilizes both light and organic carbon), or Heterotrophic (organic carbon only) [27]. Mixotrophic cultivation can enhance biomass and lipid yields [28]. |
| Species & Value-Added Products | Species like Galdieria sulphuraria are studied for pigment production under mixotrophic conditions [28]. Algae are promising for producing high-value lipids (e.g., for biofuels or nutraceuticals like EPA/DHA), proteins, and carotenoids [27]. |
| Alternative Nutrient Sources | Microalgae can be cultivated in nutrient-rich hydrolysates derived from pre-treated food waste, valorizing waste streams within the BLSS [27]. |
| Cultivation System | Closed photobioreactors (e.g., bubble columns, airlift reactors) are typically used to maintain sterile, controlled conditions [28]. |
Experimental Protocols for Producer Compartment R&D
Protocol: Quantifying Oxygen Production and Consumption in Mixotrophic Algae
This protocol is essential for monitoring and optimizing the balance of photosynthesis and respiration in algal cultures, which is critical for managing oxygen levels in a BLSS [28].
1. Principle: The double gassing-out method is used to measure the net oxygen production rate (OPRₙₑₜ) and oxygen uptake rate (OUR) in bioreactors mixed by aeration (e.g., bubble columns), where stopping the gas supply for a standard dynamic measurement is not feasible [28].
2. Equipment & Reagents:
- Photobioreactor (PSM): A sterilizable bubble column with a working volume of 700 mL, temperature control, and internal illumination [28].
- Mass Flow Controller: To precisely regulate gas flow rates [28].
- Optical Dissolved Oxygen Sensor: e.g., Visiferm DO 225, for continuous DO monitoring [28].
- Gases: Pressurized air and pure gaseous nitrogen (N₂) [28].
- Spherical Light Sensor: A submersible sensor to measure photon flux density within the culture [28].
3. Procedure:
a. Culture Preparation: Inoculate the photobioreactor with the algal strain (e.g., Galdieria sulphuraria) and operate under mixotrophic conditions with a defined organic carbon source (e.g., glycerol) [28].
b. System Calibration: Determine the oxygen displacement rate by nitrogen (ODR_N₂) under abiotic conditions by sparging with N₂ and measuring the rate of oxygen concentration decline [28].
c. Measurement under Illumination: During active mixotrophic growth, switch the gas supply from air to pure N₂. Record the slope of the descending DO curve (dCO2L/dt) [28].
d. Measurement in Darkness: After restoring air and reaching equilibrium, turn off the illumination. Repeat the N₂ gassing-out procedure and record the new slope of the DO curve (dCO2D/dt), which represents the respiration rate [28].
e. Calculation: Calculate the volumetric OPRₙₑₜ using the formula: OPRₙₑₜ = dCO2D/dt - dCO2L/dt. Specific rates (oprₙₑₜ, our) are obtained by dividing the volumetric rates by the biomass concentration (X) [28].
Diagram 1: Workflow for oxygen rate measurement.
Protocol: Engineering a Synthetic Pyrenoid into a Higher Plant
This protocol outlines the key steps for creating a proto-pyrenoid in a plant chloroplast to enhance photosynthetic efficiency.
1. Principle: Introduce and co-express two key algal components in a plant: a Rubisco enzyme engineered to bind the linker protein and the EPYC1 linker protein itself, which induces phase separation to form a liquid-like condensate—the proto-pyrenoid [24].
2. Equipment & Reagents:
- Plant Material: Model plant (e.g., Arabidopsis thaliana).
- Genetic Engineering Tools: Agrobacterium tumefaciens strain for plant transformation or a gene gun for biolistics.
- Molecular Biology Reagents: Vectors for plant expression, genes coding for the engineered hybrid Rubisco (with algal peptide tags) and EPYC1, including a chloroplast-targeting signal peptide for the latter [24].
- Microscopy: Confocal microscopy to visualize the formation of the proto-pyrenoid via phase separation.
3. Procedure: a. Gene Construct Design: Clone the gene for an algal-compatible Rubisco (e.g., from Chlamydomonas reinhardtii) and the gene for EPYC1 into plant expression vectors. Fuse the EPYC1 gene to a chloroplast transit peptide to ensure localization to the chloroplast [24]. b. Plant Transformation: Introduce the genetic constructs into the model plant using standard transformation techniques (e.g., floral dip for Arabidopsis). c. Selection and Screening: Select transgenic plants on antibiotic-containing media and screen for successful expression of both the hybrid Rubisco and EPYC1 protein. d. Phenotypic Confirmation: Use microscopy to identify plants where the EPYC1 linker protein has successfully aggregated the Rubisco into distinct proto-pyrenoid bodies within the chloroplast [24].
Table 3: Research Reagent Solutions for Producer Compartment Experiments
| Reagent / Material | Function / Application | Example / Specification |
|---|---|---|
| Optical DO Sensor | Real-time monitoring of dissolved oxygen in bioreactors [28]. | Hamilton Visiferm DO 225 [28]. |
| Mass Flow Controller | Precise regulation of gas flow rates (air, N₂, CO₂) for cultivation and experimentation [28]. | Bronkhorst High-Tech B.V. mass flow controller [28]. |
| Linker Protein EPYC1 | Key molecular component for inducing Rubisco phase separation to form pyrenoids [24]. | EPYC1 from Chlamydomonas reinhardtii, expressed in plants with a chloroplast targetting signal [24]. |
| Encapsulin System | A modular, single-gene protein cage system for creating synthetic nanocompartments to house and enhance Rubisco [25]. | Bacterial encapsulins, reprogrammed with a 14-amino-acid "address tag" to load Rubisco [25]. |
| Food Waste Hydrolysate | Sustainable, nutrient-rich alternative culture medium for microalgae cultivation [27]. | Pre-treated (e.g., enzymatically hydrolyzed) food waste from household or industrial sources [27]. |
System Integration and Future Perspectives
Integrating the producer compartment with the consumer (crew) and decomposer (waste processing) units is critical for closing the loop in a BLSS. Higher plants and microalgae can be synergistically combined. For instance, plant-derived inedible biomass and human food waste can be pre-treated (e.g., via enzymatic hydrolysis) and used as a nutrient source for heterotrophic or mixotrophic microalgae cultivation [27]. The resulting algal biomass, rich in proteins and lipids, can then supplement the crew's diet or be processed into other valuable products, creating an internal recycling stream.
Future research should focus on closing the loop by integrating decomposers to recycle inedible plant biomass and human waste, a critical link for long-term mission sustainability [5] [29]. Furthermore, while plants dominate BLSS research, there is a significant underrepresentation of animal and insect components, which could provide additional protein and nutrient recycling services [29]. Testing producer species and their integrated systems under simulated space conditions (microgravity, radiation) is also a necessary next step toward operational readiness [5].
In the context of Bioregenerative Life Support Systems (BLSS), the role of consumers extends beyond mere crew sustenance. This technical guide posits the human crew as the central heterotrophic driver, a functional unit whose metabolic and interactive processes are critical for system stability and efficiency. Framed within a broader thesis on producers, consumers, and decomposers in BLSS, this article synthesizes current ecological theory and microbial ecology to provide a novel framework for modeling, monitoring, and managing crew-driven ecosystem functions. We provide structured quantitative data, detailed experimental protocols for simulating human impacts, and essential diagnostic tools to advance BLSS research and development.
In BLSS research, the conventional view of the crew as end-users of system outputs is insufficient. A paradigm shift recognizes the crew as integral, active biotic components of the ecosystem. Humans are the dominant consumers, or central heterotrophs, whose biological processes—respiration, nutrient assimilation, waste production, and microbial interactions—directly govern the system's metabolic equilibrium [30]. This role mirrors that of dominant heterotrophs in natural ecosystems, such as marine environments where heterotrophic bacteria drive carbon cycling and determine the fate of organic matter [31]. The crew's interactions with producers (plants, algae) and decomposers (bacteria, archaea) create a complex web of feedback loops. Understanding the crew through this ecological lens is paramount for designing resilient, self-sustaining life support systems for long-duration space missions.
Theoretical Foundation: Ecological Principles for BLSS
The design and management of a BLSS can be significantly advanced by applying established macrosystems ecology principles. This framework examines ecological processes at large scales and high levels of biological organization, focusing on the interplay among patterns, processes, and perturbations [32].
- Macrosystems Ecology in BLSS: A BLSS is a quintessential macrosystem—a complex, spatially extensive ecosystem shaped by the interplay of self-organization and anthropogenic regulation [32]. Viewing it as such allows researchers to model the entire system as a network of interacting biological units (crew, plants, microbes) and abiotic factors (water, atmosphere, nutrient solutions).
- Metabolic Theory and Stoichiometry: The metabolic rates of all organisms, including humans, are influenced by body size and temperature. This theory can predict crew respiration, nutrient requirements, and heat production. Furthermore, ecological stoichiometry examines the balance of elements (e.g., C:N:P) in crew inputs (food) and outputs (waste), which is critical for managing nutrient cycles between producers and decomposers.
- Network Stability and Trophic Dynamics: The crew occupies the apex consumer position in the BLSS food web. Their consumption rate directly influences producer biomass and indirectly affects decomposer activity through waste production. Incorporating these trophic dynamics into system models is vital for predicting stability and preventing cascading failures.
Quantitative Profiling of the Central Heterotroph
Effective BLSS design requires quantitative models of human metabolic function. The following data, synthesized from ecological studies of heterotrophs, provides a basis for parameterizing the crew's role. These values are illustrative; individualized profiling is necessary for mission-specific planning.
Table 1: Metabolic and Respiratory Parameters of a Central Heterotroph (Crew Member)
| Parameter | Estimated Value (per person, per day) | Notes and Context |
|---|---|---|
| Oxygen Consumption | 0.84 kg | Based on an average respiratory quotient and caloric intake. |
| Carbon Dioxide Production | 1.00 kg | Directly linked to respiration and metabolic rate [33]. |
| Caloric Requirement | 2000-3000 kcal | Drives the required biomass production from autotrophs. |
| Water Intake | 2-4 L | Includes water from food and beverages for hydration and system functions. |
| Dry Solid Waste Production | 0.02-0.04 kg | Source of organic carbon and nutrients for decomposer subsystems [31]. |
| Liquid Waste (Urine) Production | 1-2 L | Critical source of nitrogen (urea), phosphorus, and potassium for recycling. |
Projections under changing conditions, such as increased temperature, are critical for modeling BLSS resilience. Based on models of marine heterotrophs, which show biomass declines but respiration increases with warming, we can project potential crew impacts [34].
Table 2: Projected Metabolic Changes of Heterotrophs Under Warming
| Heterotroph Type | Projected Biomass Change per °C | Projected Respiration Change per °C | Implications for BLSS |
|---|---|---|---|
| Marine Prokaryotes [34] | -1.5% | +3.5% | Suggests increased microbial activity and carbon cycling in BLSS. |
| Marine Zooplankton/Fish [34] | -4.5% to -5.0% | Data not provided | Highlights greater sensitivity of larger organisms; crew may show complex responses. |
| Crew (Inferred) | To be determined | To be determined | Emphasizes the need for empirical data on human metabolic shifts in closed systems. |
Experimental Protocols: Simulating and Measuring Crew Impacts
A critical research need is the experimental dissection of the crew's influence on BLSS biogeochemistry. The following protocols provide a methodology for this inquiry.
Protocol: Metabolic Preference Assay for Crew-Associated Microbiomes
This protocol, adapted from methodologies used to characterize marine heterotrophs, is designed to map the metabolic capabilities of the human microbiome in response to different dietary inputs [31].
1. Objective: To characterize the carbon source utilization preferences of microbial communities associated with crew waste, and to identify key enzymatic pathways involved.
2. Experimental Workflow:
3. Methodology:
- Sample Collection: Collect fecal or waste processing system samples under anaerobic conditions.
- Media Formulation: Refactor complex organic matter (e.g., inedible plant biomass, synthetic waste) into defined media, each containing a single category of carbon source: peptides, amino acids, neutral sugars, acidic sugars, organic acids, and lipids [31]. Ensure all media are carbon-mass balanced.
- Inoculation and Incubation: Inoculate each media type in triplicate with a standardized inoculum of the sample community. Incubate under controlled temperature and atmospheric conditions relevant to the BLSS.
- Phenotypic Measurement: Monitor microbial growth kinetically by measuring optical density (OD) at 600 nm. The maximum OD (maxOD) is a proxy for the community's productivity on a given carbon source.
- Data Analysis: Create a "phenotypic fingerprint" by plotting maxOD values across all carbon sources. This fingerprint reveals the preferred carbon sources for the community.
- Genomic Correlation: Perform metagenomic sequencing of the initial inoculum. Corregate growth phenotypes with the abundance of key metabolic genes and pathways (e.g., glyoxylate shunt, amino acid degradation pathways) to identify genetic determinants of metabolic preferences [31].
Protocol: Quantifying System-Level Net Ecosystem Production
This protocol measures the balance between primary production and community respiration, determining if the BLSS is a net carbon source or sink, with the crew as the primary respiratory component.
1. Objective: To determine the Net Ecosystem Production (NEP) of a BLSS test module by simultaneously measuring gross primary productivity (GPP) and ecosystem respiration (ER).
2. Experimental Workflow:
3. Methodology:
- System Isolation: Seal the BLSS test module from the external environment for a defined measurement period.
- Dark Respiration (ER): Measure the rate of CO2 accumulation in the sealed module during a dark period when primary production ceases. This represents the total ecosystem respiration (ER) from the crew and all decomposers.
- Net Primary Production (NPP): Under illuminated conditions, measure the net change in CO2 concentration. This value, NPP, represents the balance of photosynthesis and concurrent respiration.
- Calculations:
- Gross Primary Production (GPP) = NPP + ER. This represents the total carbon fixed by producers.
- Net Ecosystem Production (NEP) = GPP - ER.
- Interpretation: An NEP > 0 indicates a net autotrophic system that is sequestering carbon. An NEP < 0 indicates a net heterotrophic system that is releasing carbon, potentially unsustainable without external input [33]. The crew's respiratory burden is a major factor driving the system towards heterotrophy.
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Research Reagents for BLSS Heterotroph Studies
| Reagent / Material | Function in Research | Example Use Case |
|---|---|---|
| Defined Carbon Source Media | To isolate and quantify metabolic preferences of crew-associated microbiomes for specific organic compounds. | Formulating minimal media with peptides, sugars, or organic acids as the sole carbon source to create phenotypic fingerprints [31]. |
| Gas Chromatography System | To accurately measure concentrations of O2, CO2, and trace gases (e.g., CH4, N2O) in the system atmosphere. | Quantifying real-time ecosystem respiration (ER) and net primary production (NPP) for NEP calculations [33]. |
| Flow Cytometer / Spectrophotometer | To enumerate microbial cells and measure microbial growth density (OD) in liquid cultures. | Tracking the growth kinetics of microbial consortia from crew waste in different media during metabolic preference assays [31]. |
| Metagenomic Sequencing Kits | To profile the taxonomic and functional gene composition of microbial communities. | Correlating observed metabolic phenotypes of the crew microbiome with the presence of specific enzymatic pathways [31]. |
| Dissolved Oxygen/CO2 Probes | For in-situ monitoring of metabolic activity in aquatic subsystems (e.g., hydroponics, bioreactors). | Measuring planktonic metabolic rates in nutrient recycling wastewater processing systems. |
Integrating the human crew as the central heterotroph within BLSS models marks a critical evolution in life support strategy. This approach moves beyond viewing the crew as a passive consumer to recognizing them as a dynamic, interactive driver of system ecology. By applying macrosystems principles, obtaining precise quantitative profiles, employing robust experimental protocols, and utilizing advanced research tools, scientists can better predict, manage, and optimize the complex interactions that underpin a sustainable, bioregenerative habitat for the future of human space exploration.
Microbial reactors represent a cornerstone technology for advancing Bioregenerative Life Support Systems (BLSS) by functionally integrating the roles of producers, consumers, and decomposers. These systems leverage consortia of bacteria, fungi, and archaea to degrade organic waste, simultaneously converting it into valuable resources such as biofertilizers, nutrients for food production, and reclaimed water. This technical guide elucidates the core mechanisms—including nitrification, anaerobic digestion, and enzymatic breakdown—underpinning microbial waste transformation. It provides detailed protocols for reactor setup and monitoring, visualizes critical pathways, and catalogues essential reagents. By closing the nutrient loop, engineered microbial reactors enhance system resilience and sustainability, directly supporting the long-term viability of closed-loop human habitats.
In a Bioregenerative Life Support System (BLSS), the fundamental ecological functions of producers (plants), consumers (humans/animals), and decomposers (microbes) are engineered to create a sustainable, closed-loop environment. Microbial reactors are the technological embodiment of the decomposer role, serving as the metabolic engine of the system. They are responsible for the critical tasks of waste degradation and nutrient recycling, converting solid and liquid wastes from consumers and inedible biomass from producers into readily available plant nutrients [35]. This process is indispensable for sustaining plant growth and, consequently, supporting human life in isolated environments such as space habitats.
The imperative for such technology is underscored by the severe inefficiencies in current global food systems, where a significant proportion of produced food is wasted, leading to environmental harm and resource loss [35]. Microbial bioreactors address this by leveraging the rapid doubling times of microorganisms (e.g., 20-30 minutes for Escherichia coli) and their high metabolic diversity to process waste streams with far greater efficiency and lower environmental footprint than conventional methods [35]. By integrating these reactors into a BLSS, waste is reframed not as a disposal problem, but as a resource for continuous nutrient production, mirroring natural biogeochemical cycles and advancing the principles of a circular bioeconomy.
Fundamental Microbial Pathways for Waste Valorization
Microbial consortia within reactors facilitate waste valorization through a series of interconnected metabolic pathways. The effectiveness of these pathways hinges on the specific microbial species present and the controlled environmental conditions within the reactor.
Key Degradation and Synthesis Pathways
The core pathways involved in the breakdown of waste and synthesis of new products are summarized in the table below.
Table 1: Core Microbial Pathways in Waste Valorization
| Pathway Name | Primary Microbes | Substrate | Key Enzymes | End Products |
|---|---|---|---|---|
| Nitrification | Nitrosomonas, Nitrospira (comammox) | Ammonia (NH₃) | Ammonia monooxygenase, Nitrite oxidoreductase | Nitrate (NO₃⁻) [36] |
| Anaerobic Digestion | Methanogenic archaea, Hydrolytic bacteria | Complex organics (carbohydrates, proteins, lipids) | Hydrolases, Polymerases | Biogas (CH₄, CO₂), Organic acids [35] |
| Lignocellulose Degradation | Fungi (e.g., Trichoderma), Actinobacteria | Lignin, Cellulose, Hemicellulose | Lignin peroxidase, Cellulase, Xylanase | Simple sugars, Organic acids [35] |
| Protein & Lipid Catabolism | Diverse bacteria and fungi | Proteins, Lipids | Proteases, Lipases | Amino acids, Fatty acids, Ammonia [35] |
A critical revision in the understanding of the nitrogen cycle is the discovery of comammox Nitrospira, bacteria capable of complete ammonia oxidation to nitrate in a single organism [36]. This pathway enhances nitrogen cycling efficiency in recirculating systems, a key consideration for BLSS.
The following diagram illustrates the logical workflow and key transformations of organic waste as it passes through a generalized microbial reactor system.
Quantitative Performance Metrics
The efficiency of these pathways can be measured using key performance indicators. The following table compiles quantitative data from relevant studies to provide benchmarks for reactor performance.
Table 2: Quantitative Metrics for Microbial Waste Valorization Pathways
| Process/Parameter | Metric | Reported Value/ Range | Context/Source |
|---|---|---|---|
| Microbial Doubling Time | Time | 20-30 min (E. coli, B. subtilis) | Rapid biomass production [35] |
| Total Ammonia Nitrogen (TAN) in Aquaponics | Concentration | 0.350 - 8.989 mg/L | Must be managed below harmful thresholds [36] |
| Nutrient Content in Fish Sludge | % of Input Feed | N: 6%, P: 18%, K: 6% | Potential for nutrient recovery [36] |
| Microbial Protein Content | % of Dry Cell Weight | Up to 70% | Nutritional quality of microbial biomass [35] |
Experimental Protocols for Reactor Setup and Analysis
This section provides a detailed methodology for establishing a laboratory-scale microbial reactor for organic waste degradation and for analyzing its metabolic output.
Protocol: Establishing an Anaerobic Digestion Reactor
Objective: To set up a batch-process anaerobic microbial reactor for the conversion of organic food waste into biogas and nutrient-rich effluent.
Materials:
- Reactor Vessel: 5-L glass jar with airtight lid, fitted with ports for gas outlet and sampling.
- Inoculum: Anaerobic sludge from a wastewater treatment plant (500 mL).
- Substrate: Synthetic or real food waste (e.g., blended vegetables, fruits) [35].
- Media: Anaerobic nutrient medium (e.g., containing macro and micronutrients, bicarbonate buffer).
- Equipment: pH meter, heating mantle, gas collection bag (Tedlar bag), tubing.
Methodology:
- Substrate Preparation: Homogenize 1 kg of food waste and determine its initial moisture content and total solids.
- Reactor Inoculation: Mix the substrate with 2 L of anaerobic nutrient medium and the 500 mL inoculum inside the reactor vessel.
- Conditioning: Purge the headspace of the reactor with nitrogen gas (N₂) for 5 minutes to ensure anoxic conditions.
- Incubation: Seal the reactor and place it in a temperature-controlled environment (e.g., 35°C mesophilic condition). Connect the gas outlet to the gas collection bag.
- Monitoring: Monitor the system daily for:
- Gas Production: Measure the volume and composition (via GC-TCD for CH₄ and CO₂) of the collected biogas.
- pH: Maintain pH near neutral (6.8-7.5) using sodium bicarbonate if necessary.
- Chemical Oxygen Demand (COD): Periodically sample the liquid effluent to track organic matter removal.
Duration: The batch process typically runs for 30-40 days, until biogas production ceases.
Protocol: Colorimetric Detection of Microbial Urease Activity
Objective: To rapidly detect and semi-quantify specific bacterial contamination or metabolic activity (e.g., urease producers) in a reactor sample using a colorimetric assay.
Principle: This method falls under the "external pH change-induced pH indicator reactions" category [37]. The enzyme urease, produced by certain bacteria, catalyzes the hydrolysis of urea into ammonium and carbon dioxide. The release of ammonium increases the pH of the local environment, causing a visible color change in a pH indicator.
Materials:
- Reagent Solution: Urea substrate (10 mM) prepared in a sterile buffer.
- pH Indicator: Phenol red (0.01% w/v) or Bromothymol Blue (BTB).
- Sample: Liquid sample from the microbial reactor, centrifuged to remove large debris.
- Positive Control: Solution with known urease-producing bacteria (e.g., Proteus mirabilis).
- Negative Control: Sterile buffer only.
- Equipment: 96-well microplate or clear test tubes, micropipettes, spectrophotometer (optional).
Methodology:
- Reagent Preparation: Mix the urea substrate solution with the pH indicator in a 1:1 ratio. The initial color should be yellow (for phenol red at acidic pH).
- Sample Loading: In a microplate well, add 100 µL of the sample. Add 100 µL of the urea-indicator reagent.
- Incubation and Observation: Incubate the plate at room temperature and observe color change visually for up to 60 minutes. A positive result is indicated by a color change from yellow to pink/red (for phenol red).
- Quantification (Optional): Measure the absorbance at 560 nm (for phenol red) at time zero and at 10-minute intervals. The rate of absorbance change is proportional to the urease activity.
The experimental workflow for this detection protocol is outlined below.
The Scientist's Toolkit: Essential Research Reagents
Successful research and monitoring of microbial reactors require a suite of specific reagents and materials. The following table details key items and their functions.
Table 3: Essential Research Reagents for Microbial Reactor Studies
| Reagent/Material | Function/Application | Example Use Case |
|---|---|---|
| Chromogenic Substrates | Compounds that produce a color change when cleaved by specific intracellular enzymes (e.g., β-galactosidase) [37]. | Detecting and enumerating specific bacterial populations in a mixed consortium. |
| Gold Nanoparticles (AuNPs) | Nanomaterials whose color changes from red to blue upon aggregation, used as colorimetric probes [37]. | Developing sensitive, instrument-free biosensors for detecting toxins or specific metabolites. |
| pH Indicators (e.g., Phenol Red, BTB) | Dyes that change color with pH shift, visualizing metabolic activity [37]. | Monitoring urease activity or fermentation processes in real-time. |
| Struvite Precipitation Chemicals (MgCl₂, etc.) | Chemicals that facilitate the crystallization of phosphorus and nitrogen as struvite (MgNH₄PO₄·6H₂O) [36]. | Recovering and recycling valuable nutrients (P, N) from reactor effluent. |
| Selective Media & Antibiotics | Culture media designed to enrich for or suppress specific microbial groups. | Isulating and maintaining pure cultures of key degradative microbes from a complex consortium. |
| DNA/RNA Extraction Kits | For extracting high-quality nucleic acids from complex environmental samples. | Metagenomic and transcriptomic analysis of the reactor's microbial community [35]. |
The integration of engineered microbial reactors is fundamental to realizing robust and self-sustaining Bioregenerative Life Support Systems. By expertly performing the decomposer function, these systems transform waste from a liability into a critical resource, thereby closing the loop between producers and consumers. The pathways, protocols, and tools detailed in this guide provide a foundation for advancing this technology. Future research must focus on optimizing microbial consortia for specific waste streams, enhancing process control through advanced sensing, and scaling up these systems for operational reliability in long-duration space missions and terrestrial applications. Mastering these microbial recycling units is a decisive step toward sustainable, closed-loop habitation.
Long-term human space exploration missions, such as those to the Moon and Mars, require a paradigm shift from resupply-dependent systems to fully self-sufficient and sustainable ecosystems. Unlike current physicochemical Environmental Control and Life Support Systems (ECLSS) on the International Space Station, which require regular resupply from Earth, Bioregenerative Life Support Systems (BLSS) aim to provide in situ regeneration of vital resources through biological processes [38] [5]. These systems combine biological and physicochemical processes to enable the production of food, water, and oxygen through highly efficient recovery of minerals from waste streams, thereby minimizing the need for external consumables [39].
The MELiSSA (Micro-Ecological Life Support System Alternative) project, initiated by the European Space Agency (ESA), represents one of the most advanced and long-running BLSS initiatives [39]. Developed over more than 30 years, MELiSSA is a bioengineered system designed as a closed-loop ecosystem that provides atmospheric control, water purification, waste management, and food production [5] [40]. This case study examines the MELiSSA project's multi-compartment microbial loop, with particular focus on its operational framework within the context of producer, consumer, and decomposer trophic dynamics essential for BLSS research.
The MELiSSA Loop Framework
Systemic Design and Compartmentalization
The MELiSSA loop has been engineered as a five-compartment bioengineered system inspired by the terrestrial ecosystem of a lake [39] [40]. Each compartment represents a specific trophic level and performs distinct functions, with the overall system aiming to sustain the metabolic needs of the crew by recovering nutrients from waste streams [39]. The system is operated in continuous mode under controlled conditions, with compartments connected through gas, liquid, and solid interfaces [40].
The following diagram illustrates the interconnected flow of materials and the specific functions of each compartment within the MELiSSA loop:
Trophic Level Integration
Within the MELiSSA framework, the traditional ecological roles of producers, consumers, and decomposers are clearly delineated across compartments:
Decomposers (Compartments I, II, IIIa, IIIb): These compartments host microbial communities specialized in breaking down complex organic waste from crew metabolism into simpler inorganic compounds [39]. Compartment I performs initial waste degradation, while compartments II, IIIa, and IIIb are responsible for nitrification processes that convert ammonium into nitrate, a usable nitrogen form for producers [39].
Producers (Compartments IVa, IVb, V): These compartments consist of photosynthetic organisms that utilize light energy to convert carbon dioxide and nutrients into biomass and oxygen [5] [40]. Compartment IVa contains the cyanobacterium Limnospira indica, which provides oxygen and edible biomass, while compartments IVb and V focus on higher plant cultivation for additional food production and air revitalization [5] [40].
Consumers (Crew): The human crew occupies the consumer role within this ecosystem, consuming oxygen, water, and food produced by the system while generating carbon dioxide and waste products that feed back into the decomposer compartments [5].
This explicit trophic separation with controlled interfaces enables precise monitoring and management of the material and energy flows essential for maintaining system stability and functionality.
Compartment-Specific Functions and Methodologies
Waste Degradation and Nitrification (Compartments I-III)
The initial waste processing compartments function as the system's decomposers, breaking down crew waste and recovering essential nutrients, particularly nitrogen.
Compartment I receives liquid and solid waste from the crew, with urine being a particularly valuable resource as it accounts for 85% of the total potentially recoverable nitrogen in the system, mostly in the form of urea [39]. The compartment employs thermophilic anaerobic bacteria to break down complex organic molecules into simpler compounds, including volatile fatty acids and ammonium [39].
Compartments II, IIIa, and IIIb are responsible for the nitrification process, which converts toxic ammonium into nitrate that can be utilized by photosynthetic organisms. The specific methodology involves:
Compartment II: Uses fixed-bed reactors with microbial communities that perform photoheterotrophic oxidation of volatile fatty acids and partial nitrification under the influence of UV light [39].
Compartment IIIa: Employs Rhodospirillum rubrum in a packed-bed reactor to perform photoheterotrophic oxidation of remaining organic compounds while converting ammonium to nitrate [39].
Compartment IIIb: Utilizes nitrifying bacteria such as Nitrosomonas europaea and Nitrobacter winogradskyi in a separate packed-bed reactor to complete the nitrification process, ensuring full conversion of ammonium to nitrate [39].
The resulting nitrate-rich effluent from these compartments serves as a fertilizer for the photosynthetic compartments, completing the nutrient recovery loop.
Photosynthetic Production (Compartments IVa, IVb, V)
The producer compartments utilize the recovered nutrients and crew-respired CO2 to generate oxygen and edible biomass through photosynthesis.
Compartment IVa employs an 83L external loop air-lift photobioreactor (PBR) where the cyanobacterium Limnospira indica is cultivated in continuous operation mode [40]. The methodological approach for this compartment includes:
Continuous Culture Operation: The PBR operates at various dilution rates (typically 0.015-0.025 h⁻¹) and photon flux densities (PFD) ranging from 150-930 μmol m⁻² s⁻¹ to optimize productivity [40].
Gas Exchange Monitoring: Real-time monitoring of O2 production and CO2 consumption rates to maintain atmospheric balance for the crew [40].
Morphological Analysis: Confocal microscopy is used to monitor trichome width and size distribution as indicators of cellular health and adaptation to culture conditions [40].
The performance of this compartment is critically dependent on the interaction between light intensity and cell density, quantified as the specific photon flux density (qPFD), which ranges from 6.1 to 19.2 μmol g⁻¹ s⁻¹ in operational conditions [40].
Compartments IVb and V focus on higher plant cultivation using various growth systems:
Hydroponic Systems: Such as the Veggie system used on the ISS, which has successfully grown lettuce, Chinese cabbage, mizuna mustard, and other leafy greens [38] [5].
Soil-Based Systems: Utilizing lunar or Martian regolith supplemented with nitrogen-fixing bacteria to improve soil fertility [38].
Advanced Plant Habitats: Controlled environment chambers like NASA's Advanced Plant Habitat (APH) that automate the monitoring and control of environmental conditions [5].
Plant species are selected based on mission duration and objectives, with fast-growing leafy greens for short-duration missions and staple crops (wheat, potato, rice) for long-duration missions and planetary outposts [5].
Quantitative System Performance
Photobioreactor Operational Data
The performance of Compartment IVa's photobioreactor has been systematically characterized under different operational conditions. The following table summarizes key performance metrics obtained from continuous culture experiments:
Table 1: Performance Metrics of Limnospira indica in an Air-Lift Photobioreactor [40]
| Dilution Rate (h⁻¹) | Photon Flux Density (μmol m⁻² s⁻¹) | Oxygen Productivity (mmol l⁻¹ h⁻¹) | Biomass Composition Changes | Specific Photon Flux Density (μmol g⁻¹ s⁻¹) |
|---|---|---|---|---|
| 0.015 | 600 | 0.75 | - | 6.1 |
| 0.020 | 600 | 0.95 | - | 7.8 |
| 0.025 | 600 | 1.10 | Phycobiliproteins: -62.5%Chlorophyll: -47.8% | 19.2 |
| 0.025 | 930 | 1.35 | - | - |
| 0.030 | 600 | 1.20 | - | - |
The data demonstrates that the highest oxygen productivity (1.35 mmol l⁻¹ h⁻¹) is achieved at a dilution rate of 0.025 h⁻¹ and PFD of 930 μmol m⁻² s⁻¹ [40]. Furthermore, increased specific photon flux density directly impacts cellular composition, causing significant decreases in phycobiliproteins and chlorophyll content while increasing carbohydrate reserves [40].
Resource Recovery and Production Estimates
The overall MELiSSA system targets specific production goals to support human crew requirements:
Table 2: MELiSSA System Production Targets and Resource Recovery [38] [39] [40]
| Parameter | Target Value | Notes |
|---|---|---|
| Oxygen Production | 1.35 mmol l⁻¹ h⁻¹ (max) | From PBR compartment [40] |
| Food Production | 20-40% of crew requirements | Primary from cyanobacteria, supplemented by plants [40] |
| Nitrogen Recovery | 7-16 g N per crew member daily | Primarily from urine (85% of recoverable N) [39] |
| Water Recovery | 1.80 L urine + flush water per crew member daily | Processed through nitrification compartments [39] |
The system aims to substantially reduce resupply needs from Earth, with estimates suggesting that a 3-year Mars mission with a crew of four would require over 25,000 kg of food and water if relying solely on resupply [39].
Experimental Protocols and Research Methodologies
Continuous Photobioreactor Cultivation
The experimental workflow for operating and monitoring the Compartment IVa photobioreactor involves a systematic approach to maintain continuous culture and measure key performance indicators:
Detailed Methodology:
Photobioreactor Startup and Inoculation: The 83L external loop air-lift photobioreactor is axenically inoculated with Limnospira indica (formerly Arthrospira sp. PC8005). The air-lift design is selected to minimize shear stress on cyanobacterial cells while providing efficient gas-liquid mass transfer [40].
Parameter Setting and Steady-State Achievement: Operational parameters including dilution rate (D) and photon flux density (PFD) are set according to experimental requirements. The system is allowed to reach steady-state at each condition, typically requiring 3-4 residence times [40].
Continuous Operation with Gas and Liquid Exchange: The PBR operates in continuous mode with constant inflow of nitrate-rich medium from compartment III and outflow of biomass suspension. The gas exchange (O2 production and CO2 consumption) is continuously monitored using off-gas analysis [40].
Process Monitoring and Data Collection:
Biological Characterization:
- Morphological Analysis: Confocal microscopy is used to examine trichome width and spiral structure. Size distribution parameters are quantified to assess culture heterogeneity [40].
- Composition Analysis: Pigment content (phycobiliproteins, chlorophyll), protein, and carbohydrate concentrations are determined using spectrophotometric and chromatographic methods [40].
Data Integration and Model Validation: Experimental results are integrated into mathematical models that support the design, monitoring, and predictive control of the MELiSSA loop [40].
Nitrogen Recovery and Processing
The experimental approach for nitrogen recovery from urine waste streams involves a multi-stage bioreactor system:
Urine Collection and Stabilization: Urine and urine flush water (approximately 1.80L per crew member daily) is collected in a Wastewater Storage Tank Assembly. The urine is chemically stabilized with phosphoric acid to prevent scaling and convert volatile ammonia to non-volatile ammonium [39].
Nitrification Reactor Operation: Packed-bed reactors containing immobilized nitrifying bacteria are operated under controlled conditions to convert ammonium to nitrate through a two-step process:
- Nitrosomonas europaea oxidizes ammonium to nitrite
- Nitrobacter winogradskyi oxidizes nitrite to nitrate [39]
Process Monitoring and Optimization: Nitrogen conversion efficiency is monitored through regular sampling and analysis of nitrogen species. Reactor performance is optimized by controlling hydraulic retention time, dissolved oxygen concentration, and pH [39].
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key research reagents, biological materials, and equipment essential for conducting MELiSSA-related experiments and system operations:
Table 3: Essential Research Reagents and Materials for MELiSSA Loop Experiments
| Reagent/Material | Function/Application | Specific Examples/Notes |
|---|---|---|
| Limnospira indica | Photosynthetic O2 production, edible biomass | Cyanobacterium; formerly Arthrospira sp. PC8005 [40] |
| Nitrosomonas europaea | Ammonia oxidation in nitrification | Converts NH4+ to NO2- in Compartment III [39] |
| Nitrobacter winogradskyi | Nitrite oxidation in nitrification | Converts NO2- to NO3- in Compartment III [39] |
| Rhodospirillum rubrum | Photoheterotrophic nitrification | Used in Compartment IIIa for organic oxidation [39] |
| Simulated Urine Solution | Waste stream for nitrogen recovery | Contains urea, salts, 7-16gN/L/day per crew member [39] |
| Nitrate-Rich Medium | Cyanobacterial cultivation | Contains NO3- from nitrification compartments as nitrogen source [40] |
| Air-Lift Photobioreactor | Cyanobacterial cultivation | 83L capacity, external loop design for minimal shear stress [40] |
| Confocal Microscopy | Morphological analysis | Monitors trichome width, spiral structure, size distribution [40] |
The MELiSSA project's multi-compartment microbial loop represents a sophisticated implementation of ecological principles in a controlled engineered system. By explicitly separating the functional roles of producers, consumers, and decomposers across specialized compartments, the system achieves efficient recycling of resources essential for long-duration space missions. The continuous operation of the interconnected compartments, supported by rigorous monitoring and mathematical modeling, demonstrates the feasibility of bioregenerative life support systems for future space exploration.
Ground-based demonstration facilities, such as the MELiSSA Pilot Plant, continue to refine the operational parameters and integration strategies necessary for implementing these systems in space habitats. The knowledge gained from these efforts not only advances space exploration capabilities but also contributes to sustainable resource management technologies with potential terrestrial applications. As space agencies prepare for crewed missions to the Moon and Mars, the MELiSSA framework provides a viable pathway toward the self-sufficiency and sustainability required for humanity to become a multi-planetary species.
In the context of Bioregenerative Life Support Systems (BLSS) research, understanding the roles of producers, consumers, and decomposers is fundamental for designing systems capable of sustaining human life during long-duration space missions. A BLSS is essentially a closed ecological system where several levels of trophic connections guarantee biomass cycling in food webs [5]. These systems comprise three main types of compartments: biological producers (e.g., plants, microalgae, photosynthetic bacteria), consumers (i.e., crew), and waste degraders and recyclers (e.g., fermentative and nitrifying bacteria) [5]. Effective monitoring and balancing of the biomass and nutrient flows between these compartments is what enables resource recovery, food production, and waste treatment solutions essential for self-sustainable missions to the Moon and Mars [5].
Fundamental Concepts: Producers, Consumers, and Decomposers in BLSS
In any ecosystem, including an artificial BLSS, matter and energy are transferred among organisms. Producers (primarily plants) produce matter with stored energy that other organisms consume. Consumers are animals that eat plants or other animals, while decomposers (such as fungi and bacteria) break down dead organisms into nutrients that can be reused by producers [41]. This creates an interlocking system of energy and matter transfer—a food web—that is fundamental to ecosystem stability [41].
In a BLSS, this concept is engineered into a controlled environment where the "food of almost any kind of animal can be traced back to plants" [41], and decomposers are integral for "restoring (recycling) some materials back to the soil" [41], or in the case of BLSS, back to the plant growth substrate. The proper functioning of this loop is critical for mission success, as it regenerates oxygen and water while producing food and recycling waste [5].
Quantitative Metrics for Biomass and Nutrient Tracking
Tracking the flows of biomass and nutrients requires the measurement of specific quantitative metrics across the different compartments of the BLSS. The following parameters are essential for maintaining system balance.
Table 1: Key Quantitative Metrics for BLSS Monitoring
| Parameter | Producer Compartment (Plants) | Consumer Compartment (Crew) | Decomposer Compartment (Microbes) |
|---|---|---|---|
| Biomass Flow | Total biomass yield (g/m²/day), Harvest Index (edible/total biomass ratio) [5] | Total caloric intake (kcal/crew/day), Oxygen consumption (L/crew/day) [5] | Volatile Solids destruction rate (%), Biomass conversion efficiency |
| Gas Exchange | Photosynthetic rate (net O₂ production, CO₂ consumption) [5] | Respiration rate (O₂ consumption, CO₂ production) [5] | Respiration rate (O₂ consumption, CO₂ production) |
| Water Flow | Transpiration rate (L/m²/day), Nutrient solution uptake (L/day) [5] | Potable water consumption (L/crew/day), Gray water production (L/crew/day) [5] | Water recycling efficiency (%), Process water demand (L/day) |
| Nutrient Flow | Macronutrient uptake (N, P, K in mg/L), Micronutrient uptake (Fe, Mn, Zn in µg/L) | Metabolic waste production (N, P, K in g/crew/day) | Nutrient mineralization rate (N, P, K mg/L/day) |
Experimental Protocols for System Monitoring
Protocol for Plant Growth and Biomass Production Tracking
This protocol outlines the methodology for monitoring the producer compartment within a BLSS, crucial for providing food, oxygen, and water regeneration [5].
Materials:
- Controlled Environment Agriculture (CEA) chamber with programmable lighting, temperature, and CO₂ control.
- Seedlings of selected species (e.g., lettuce, wheat, potato).
- Hydroponic or aeroponic nutrient delivery system.
- Precision scales (0.01g accuracy).
- Gas Analyzer (for O₂ and CO₂).
- Water quality test kits (for pH, EC, NPK).
Procedure:
- System Initialization: Calibrate all sensors. Program the environmental controls to maintain optimal growth conditions (e.g., photoperiod, temperature, CO₂ at 1000 ppm).
- Plant Cultivation: Introduce a predetermined number of plant seedlings into the growth chamber. For short-duration mission analogs, use fast-growing species like leafy greens; for long-duration analogs, use staple crops like wheat or potato [5].
- Data Acquisition:
- Daily: Record fresh weight biomass of any harvested material. Measure water transpiration by monitoring the level in the nutrient reservoir. Analyze input and output gas concentrations using the gas analyzer.
- Weekly: Destructively harvest a subset of plants (if applicable) to determine dry weight and calculate Harvest Index. Collect water samples to analyze nutrient concentration changes.
- Data Analysis: Calculate daily growth rates, total edible biomass yield, oxygen production, and carbon dioxide consumption. Correlate these outputs with resource inputs (water, light, nutrients).
Protocol for Waste Degradation and Nutrient Recycling Efficiency
This protocol describes how to track the performance of the decomposer compartment in recycling consumer waste into usable plant nutrients.
Materials:
- Bioreactor with temperature and aeration control.
- Heterotrophic microbial inoculant (e.g., Nitrosomonas, Nitrobacter).
- Simulated or real metabolic waste (urea, inedible plant biomass).
- Spectrophotometer or Ion Chromatograph for nutrient analysis.
Procedure:
- Bioreactor Inoculation: Introduce the microbial inoculant into the bioreactor containing a defined mineral medium.
- Waste Introduction: Add a quantified amount of waste substrate (e.g., 1 g/L of urea and 10 g/L of ground inedible plant biomass) to the reactor.
- Process Monitoring:
- Continuous: Monitor and log temperature and dissolved oxygen.
- Daily: Take liquid samples and analyze for ammonium (NH₄⁺), nitrite (NO₂⁻), and nitrate (NO₃⁻) concentrations using the spectrophotometer.
- Endpoint: Measure the total reduction in volatile solids of the solid waste.
- Data Analysis: Calculate the rates of nitrification (conversion of NH₄⁺ to NO₃⁻) and the overall efficiency of nutrient recovery from waste streams.
Visualization of Biomass and Nutrient Flows in a BLSS
The following diagram illustrates the interconnected flows of biomass and nutrients between the key compartments of a BLSS, highlighting the monitoring points essential for system balance.
Figure 1: Material and Energy Flows in a BLSS. This diagram shows the continuous cycling of matter (solid lines) and the input and conversion of energy (dashed line) that sustains the closed-loop system. Critical monitoring points include food biomass to consumers, oxygen and carbon dioxide exchange, and the conversion of waste into plant-available nutrients.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful experimentation in BLSS research requires a suite of specialized reagents and materials. The following table details key items and their functions.
Table 2: Key Research Reagents and Materials for BLSS Experimentation
| Item Name | Function/Application | Technical Specification |
|---|---|---|
| Controlled Environment Chamber | Provides precise control over plant growth conditions (light, temperature, humidity, CO₂) to simulate space habitat settings [5]. | Programmable LED lighting (PAR 200-800 µmol/m²/s), CO₂ control (300-2000 ppm), temperature control (±0.5°C). |
| Hydroponic Nutrient Solution | Delivers essential macro and micronutrients to plants in a soil-less cultivation system, allowing for precise nutrient tracking [5]. | Balanced solution containing N, P, K, Ca, Mg, S, and micronutrients (Fe, Mn, B, Zn, Cu, Mo) at defined concentrations (e.g., Hoagland's solution). |
| Gas Chromatograph / Mass Spectrometer (GC-MS) | Analyzes trace volatile organic compounds (VOCs) in the cabin air and measures O₂ and CO₂ exchange rates for mass balance calculations. | Capable of detecting gases at ppm or ppb levels. |
| Ion Chromatograph (IC) | Quantifies ion concentrations (e.g., NH₄⁺, NO₂⁻, NO₃⁻, PO₄³⁻, Cl⁻) in water samples from hydroponic systems and waste recyclers. | Analytical precision of ±5% or better for major ions. |
| Microbial Culture Media | Used to isolate, identify, and maintain specific strains of nitrifying and waste-degrading bacteria for the decomposer compartment. | Sterile, defined media such as R2A agar for heterotrophs or ammonium oxalate media for nitrifiers. |
| Liquid Scintillation Counter | Tracks the fate of specific nutrients by using radioisotope tracers (e.g., ¹⁴C, ³²P) in closed-loop studies, enabling precise nutrient flow modeling. | Requires appropriate radioisotope licensing and safety protocols. |
The meticulous monitoring and balancing of biomass and nutrient flows are not merely supportive functions but are foundational to the viability of Bioregenerative Life Support Systems. By treating the BLSS as an integrated ecological network of producers, consumers, and decomposers, and by implementing robust quantitative tracking and experimental protocols, researchers can advance these systems from ground-based demonstrators to the technological cornerstones of long-term human presence in space. The protocols and metrics outlined here provide a framework for achieving the closed-loop resource recovery necessary for the future of human space exploration.
Balancing the Biosphere: Troubleshooting Contamination and Instability in BLSS
Identifying and Controlling Harmful Microbial Contamination
In the context of Bioregenerative Life Support Systems (BLSS), the control of harmful microbial contamination is not merely a matter of safety but a fundamental requirement for system stability and sustainability. BLSS are engineered ecosystems designed to operate on the principle of resource regeneration, mirroring the Earth's biosphere by integrating producers, consumers, and decomposers into a closed-loop system [1]. The primary objective of these systems is to provide a sustainable life-support system for effective functioning in extreme environments by constantly recirculating water, nutrients, and oxygen without external replenishment [1]. Within this delicate balance, microorganisms play dual roles: essential decomposers are crucial for nutrient recycling, while opportunistic pathogens pose a threat to system components and crew health. This guide provides an in-depth technical framework for identifying and controlling harmful microbial contamination, specifically framed within the producer-consumer-decomposer paradigm of BLSS research.
BLSS Ecosystem Framework and Microbial Niches
A self-sustaining bioregenerative ecosystem (SSBE) comprises primary producers (photoautotrophs), consumers (humans, animals), and decomposers (microbes), representing a complete food web based on renewable resources [1]. The strategic configuration of this ecosystem is designed for the effective flow of materials and energy, where each functional group has a defined role [1].
- Producers (Plants, Cyanobacteria): These organisms capture light energy and perform carbon fixation, producing biomass and oxygen. Their rhizosphere and phyllosphere provide specific niches for microbial communities. Contamination here can disrupt gas exchange and food production.
- Consumers (Humans): As the central beneficiaries of the system, humans are also a primary source of contamination, shedding microorganisms through skin, respiration, and metabolic waste [42].
- Decomposers (Bacteria, Fungi): This group is responsible for mineralizing waste biomass and recycling nutrients back to producers. While indispensable, an imbalance, such as the overgrowth of a single species or the invasion of a pathogen, can disrupt the entire closed-loop system [1].
Understanding these interactions is critical, as microbial contamination can disrupt the energy capture and transformation, mineral retention and cycling, and cycle regulation and control—the three primary functions within the ecosystem [1]. The following diagram illustrates the material flow and potential contamination points within a simplified BLSS.
Diagram 1: Material flow and contamination risks in a BLSS.
Advanced Detection Technologies for Microbial Identification
Rapid and accurate identification of microbial contaminants is the first step toward control. Moving beyond traditional, time-consuming culture methods, modern technologies offer high-throughput and precise solutions essential for a BLSS [43] [44].
Nucleic Acid-Based Methods
These methods detect specific DNA or RNA sequences of target pathogens, offering high specificity and sensitivity [43].
Polymerase Chain Reaction (PCR): Amplifies a specific target DNA sequence through a cyclic three-step process (denaturation, annealing, and extension). It allows for the detection of a single bacterial pathogen in a sample [43].
- Simple PCR: Targets a single gene sequence.
- Multiplex PCR (mPCR): Simultaneously amplifies multiple gene targets using several sets of specific primers in a single reaction, allowing for the detection of numerous pathogens rapidly [43].
- Real-Time Quantitative PCR (qPCR): Provides quantitative data by measuring the amplification of DNA in real-time, reducing false-negative risks. For example, qPCR sensitivity can reach 10 CFU/g for Listeria detection in dairy products [44].
Loop-Mediated Isothermal Amplification (LAMP): An isothermal amplification technique that does not require a thermal cycler, making it suitable for resource-limited settings. It operates at a constant temperature and uses multiple primers for high specificity [43].
Next-Generation Sequencing (NGS):
- 16S rRNA Amplicon Sequencing: Profiles bacterial communities by sequencing the hypervariable regions of the 16S rRNA gene, allowing for taxonomic identification [45].
- Shotgun Metagenomics: Sequences all accessible DNA in a sample, enabling the reconstruction of entire microbial communities and their functional potential, including viruses and fungi [45] [46]. This is powerful for identifying unknown pathogens and conducting traceability analyses [44].
Biosensor and Immunological-Based Methods
- Biosensors (Optical, Electrochemical): These devices use biological recognition elements (e.g., antibodies, DNA probes) coupled with a transducer to detect target analytes. They are known for portability, speed, and accuracy, enabling on-site rapid detection [44].
- Enzyme-Linked Immunosorbent Assay (ELISA): A plate-based technique that uses antibodies to detect and quantify the presence of a specific antigen, such as a microbial toxin [43].
The workflow for a comprehensive microbial analysis using these technologies is detailed below.
Diagram 2: Microbial detection and analysis workflow.
Taxonomic Profiling from Metagenomic Data
For shotgun metagenomic data, the process of taxonomic profiling involves comparing DNA sequences from the sample to reference databases. The main approaches are [46]:
- Genome-Based Approach: Reads are aligned to reference genomes to measure genome abundance with high detection accuracy.
- k-mer-Based Approach: Databases and sample DNA are broken into strings of length k for comparison. Tools like Kraken use this method, which has a low compute cost but lower detection accuracy and no gene detection capability [46].
- Marker-Based Approach: Tools like MetaPhlAn search for marker genes in reads, which is quick but can introduce bias [46].
Table 1: Comparison of Key Microbial Detection Technologies
| Technology | Principle | Detection Time | Key Advantage | Primary Limitation |
|---|---|---|---|---|
| Culture-Based | Growth on selective media | 2-7 days | Gold standard, viable cells | Time-consuming, labor-intensive [43] |
| qPCR | Target DNA amplification | 2-4 hours | High sensitivity & specificity (~10 CFU/g) [44] | Detects DNA, not necessarily viability |
| LAMP | Isothermal DNA amplification | 1-2 hours | Simplicity, no thermal cycler needed [43] | Primer design complexity |
| MALDI-TOF MS | Protein fingerprint matching | Minutes | Rapid identification (>95% accuracy) [44] | Requires pure cultures, database dependent |
| Shotgun Metagenomics | Sequencing all DNA in a sample | 1-3 days | Comprehensive, detects unknowns, functional insight [45] [44] | High cost, complex data analysis [44] |
Quantitative Data on Microbial Transfer and Cross-Contamination
In a closed-system BLSS—and analogous terrestrial environments like domestic kitchens—understanding and quantifying microbial transfer is critical for exposure assessment and risk characterization [47]. Cross-contamination refers to the transfer of foodborne pathogens from a contaminated source via vehicles (e.g., hands, surfaces) to other surfaces or foods, influencing the infection risk to consumers [47].
Quantitative data on transfer rates (the proportion of microorganisms transferred from one surface to another upon contact) are essential for building robust Quantitative Microbial Risk Assessment (QMRA) models [47]. These models help predict risks and implement targeted interventions.
Table 2: Bacterial Transfer Rates Between Common Kitchen Vehicles and Foods [47]
| Source | Sink | Median Transfer Rate | Notes |
|---|---|---|---|
| Stainless Steel | Cucumber | 32% | Transfer is influenced by surface hydrophobicity and topography [47]. |
| Plastic Cutting Board | Lettuce | 11% | |
| Hands | Lettuce | 17% | Highlights the critical role of hand hygiene. |
| Lettuce | Hands | 22% | Demonstrates bidirectional transfer. |
| Gloves | Pork Jerky | 14% | Transfer can occur even through single-use gloves [47]. |
These data underscore the importance of material selection and hygiene protocol design in a BLSS habitat. Surfaces with lower transfer rates (e.g., certain plastics over stainless steel in some cases) and strict handwashing procedures are crucial to interrupt contamination pathways.
Experimental Protocols for Contamination Control
Protocol for Assessing Surface Cross-Contamination
Objective: To quantify the transfer rate of a surrogate organism (e.g., E. coli K-12) from a contaminated surface to a food product via a handler's hands.
Materials:
- Non-pathogenic surrogate organism (E. coli K-12)
- Sterile buffered peptone water
- Sterile gloves
- Pre-sterilized stainless steel coupons (5x5 cm)
- Lettuce leaves
- Neutralizing agar plates
- Vortex mixer
- Pipettes and sterile tips
Method:
- Inoculation: Spot-inoculate the stainless steel coupon with 100 µL of a bacterial suspension (~10^8 CFU/mL). Air-dry in a biosafety cabinet for 60 minutes.
- Contact Transfer (Source to Hand): A subject wearing sterile gloves touches the inoculated coupon with fingertips for 10 seconds.
- Sampling (Hand): The glove fingertips are immediately rinsed in 10 mL of sterile peptone water and vortexed for 30 seconds to recover microorganisms.
- Contact Transfer (Hand to Sink): The same glove fingertips touch a sterile lettuce leaf for 10 seconds.
- Sampling (Sink): The contacted area of the lettuce leaf is massaged in 10 mL of sterile peptone water for 30 seconds.
- Enumeration: Perform serial dilutions of both peptone water samples and spread-plate on neutralizing agar. Incubate plates at 37°C for 24-48 hours.
- Calculation:
- Transfer Rate (%) = (CFU recovered from Sink / CFU recovered from Hand) x 100
Protocol for Air Sampling in a Controlled Environment
Objective: To monitor the concentration and diversity of airborne fungi in a plant growth module.
Materials:
- Microbial air sampler (e.g., impaction onto solid surfaces)
- Potato Dextrose Agar (PDA) plates with antibiotics (e.g., chloramphenicol) to inhibit bacterial growth
- Flow meter (if required by sampler)
- Incubator
Method:
- Sampling: Place the air sampler at a designated height (e.g., canopy level) within the module. According to the manufacturer's instructions, turn on the sampler and draw a known volume of air (e.g., 100-500 L) over the PDA plate.
- Incubation: Seal the plates and incubate at 25-30°C for 5-7 days to allow for fungal growth.
- Analysis: Count the number of colony-forming units per cubic meter of air (CFU/m³). Identify dominant morphotypes microscopically or through sequencing (ITS region) for a more precise identification [48] [42].
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Materials for Microbial Contamination Research
| Reagent/Material | Function | Example Application |
|---|---|---|
| Neutralizing Broth/Agar | Inactivates disinfectants and antimicrobials in a sample, ensuring accurate microbial enumeration. | Recovery of microorganisms from sanitized surfaces. |
| DNA/RNA Shield | Protects nucleic acids from degradation during sample storage and transport, stabilizing the sample's molecular profile. | Preserving samples for later metagenomic or PCR analysis. |
| PCR Master Mix | A pre-mixed solution containing Taq polymerase, dNTPs, buffers, and MgCl₂ required for PCR amplification. | Standardized setup of qPCR or mPCR for pathogen detection. |
| Shotgun Metagenomic Library Prep Kit | A suite of reagents for fragmenting DNA, attaching sequencing adapters, and amplifying the library for NGS. | Preparing environmental DNA samples for sequencing on platforms like Illumina. |
| Bioaerosol Sampler | Actively draws a known volume of air and impacts airborne particles onto a solid or liquid collection medium. | Quantifying and identifying microbial loads in the air of a BLSS habitat [42]. |
| MALDI-TOF MS Matrix | A chemical (e.g., sinapinic acid) that co-crystallizes with the sample, enabling desorption/ionization for mass spectrometry. | Rapid identification of bacterial or fungal isolates from surfaces. |
The identification and control of harmful microbial contamination within a BLSS is a dynamic challenge that requires a holistic understanding of ecological principles coupled with cutting-edge analytical technologies. By integrating robust detection methods like NGS and qPCR, quantitative transfer data, and stringent protocols modeled after QMRA frameworks, researchers can design more resilient and stable life-support systems. The producer-consumer-decomposer model provides the essential framework for anticipating contamination pathways and implementing targeted, effective control measures to ensure the long-term viability of closed ecological systems.
The Biosphere 2 project, conceived in the late 20th century, remains the largest closed ecological system ever created. This massive, 3.14-acre enclosed structure in Oracle, Arizona was designed as an artificial, materially closed ecological system to experiment with the viability of self-sustaining life support systems for space colonization and to study Earth's ecological complexities [49]. Its mission involved a two-year closure experiment with an eight-person crew living in isolation, sustained by interconnected ecosystems including a tropical rainforest, ocean, mangrove wetlands, savannah grassland, fog desert, and agricultural area [49].
Within the context of Bioregenerative Life Support Systems (BLSS) research, Biosphere 2 served as an unprecedented testbed for understanding the intricate balances between producers (plants, algae), consumers (humans, animals), and decomposers (microbes, fungi) in a closed environment. The facility's airtight design, with an exceptionally low leak rate of less than 10% per year, made it a powerful instrument for studying ecological processes and gas exchange dynamics [50] [51]. The oxygen imbalance that emerged during its first manned mission provides critical insights into the complex interactions governing closed ecological systems and the challenges of maintaining atmospheric homeostasis.
The Biosphere 2 System: Design and Operation
Engineering and Closure Systems
The physical structure of Biosphere 2 consisted of steel tubing and high-performance glass and steel frames, specifically designed to achieve unprecedented airtightness. The patented sealing methods achieved a remarkably low leak rate of less than 10% per year, enabling researchers to track subtle atmospheric changes over time [49] [50]. This level of closure was crucial for observing slow changes in air composition that would otherwise be masked by atmospheric exchange.
A critical engineering feature was the inclusion of two large variable volume structures called "lungs." These diaphragms accommodated thermal expansion and contraction of the internal atmosphere, preventing dangerous pressure differentials that could compromise structural integrity. Without these lungs, normal variations in temperature and humidity would have forced significant leakage, rapidly diluting the internal atmosphere [50] [51]. The system maintained pressure differentials between inside and outside atmospheres within a narrow range of ±8 Pa [50].
Table 1: Key Engineering Specifications of Biosphere 2
| Parameter | Specification | Functional Significance |
|---|---|---|
| Total Volume | 200,000 m³ [50] | Scale relevant for ecological experimentation |
| Airtight Footprint | 1.27 hectares (3.14 acres) [49] | Area for biome development |
| Leak Rate | <10% per year [50] | Enabled detection of subtle atmospheric changes |
| Pressure Differential | ±8 Pa [50] | Minimal pressure variance maintained by lungs |
| Number of Biomes | 7 distinct ecosystems [49] | Biodiversity and functional redundancy |
Ecological Components and Initial Conditions
Biosphere 2 contained seven biome areas representing different Earth ecosystems, each stocked with numerous species to create functional ecological communities. The design employed a "species-packing" strategy, anticipating that some species would not survive as the biomes reached equilibrium [49]. This approach aimed to ensure that food webs and ecological functions could be maintained even with expected species loss.
The agricultural system was designed to provide 100% of the nutritional needs for the eight crew members, incorporating crops such as bananas, papayas, sweet potatoes, beets, peanuts, beans, rice, and wheat [49]. The agricultural area represented a significant anthropogenic biome within the larger ecological context. Soils throughout Biosphere 2 were intentionally rich and young to fuel rapid growth of vegetation, a decision that would later prove critical to understanding the oxygen imbalance [52].
Table 2: Ecological Components of Biosphere 2
| Biome Type | Area | Key Species Introduced | Ecological Function |
|---|---|---|---|
| Tropical Rainforest | 1,900 m² [49] | 400+ plant species from tropical regions [53] | Primary production, oxygen generation |
| Ocean with Coral Reef | 850 m² [49] | Corals, fish, marine invertebrates [53] | Carbon sequestration, biodiversity |
| Mangrove Wetlands | 450 m² [49] | 542 mangroves, aquatic species [53] | Water filtration, habitat provision |
| Savannah Grassland | 1,300 m² [49] | Acacia species, grasses [53] | Hydrological transition, carbon storage |
| Fog Desert | 1,400 m² [49] | Arid-adapted plants [53] | Low-water ecosystem function |
| Agricultural System | 2,500 m² [49] | Food crops, domesticated animals [49] | Human life support, food production |
The Oxygen Imbalance: Manifestation and Crisis
Timeline and Measurements
During the first manned mission from September 1991 to September 1993, Biosphere 2 experienced a steady, unexpected decline in atmospheric oxygen. From early in the 2-year closure, oxygen concentration diminished from the normal terrestrial level of 20.9% to approximately 14.4% over 475 days [50] [52]. This represented an average decline of about 140 parts per million per day during the first 16 months of the mission [50].
The oxygen depletion created conditions equivalent to an altitude of 3,350 meters (11,000 feet) above sea level [52]. Crew members experienced symptoms consistent with altitude sickness, including fatigue, weakness, and sleep disturbances, making their daily farming and maintenance work increasingly arduous [52]. By the 16-month mark, the situation necessitated the injection of supplemental oxygen from external sources to ensure crew safety and continue the mission [49] [52].
Experimental Protocols and Measurement Methodologies
The detection and analysis of the oxygen imbalance relied on sophisticated monitoring systems and experimental protocols:
Atmospheric Monitoring: Hundreds of sensors throughout Biosphere 2 continuously collected data on atmospheric gas concentrations, including oxygen and carbon dioxide levels [51]. This "nerve system" distributed among many terminals provided real-time data on system dynamics.
Leak Rate Quantification: Researchers employed two independent methods to measure atmospheric leakage. The first involved measuring the rate of deflation of the lungs while holding a constant elevated pressure differential. The second method tracked the progressive dilution of trace gases intentionally introduced into the atmosphere [50]. Both methods confirmed the leak rate was less than 10% per year.
Carbon Pathway Tracking: Material closure enabled precise tracking of chemical pathways. The investigation into the "missing oxygen" involved quantitative analysis of carbon storage pools, including measurement of organic carbon in biomass, soil, and water, as well as inorganic carbon in concrete and dissolved in ocean water [51].
Root Cause Analysis: The Biochemical Mechanisms
Microbial Respiration and Carbon Cycle Imbalance
The primary mechanism driving oxygen loss was identified as respiratory consumption by aerobic microorganisms in the soils. The rich, young soils introduced to support rapid plant growth contained abundant organic matter that fueled substantial microbial populations [52]. These microbes, like all aerobic organisms, consume oxygen and release carbon dioxide during respiration.
The imbalance occurred because the photosynthetic oxygen production by the relatively young plants and trees was insufficient to compensate for the respiratory consumption by the soil microbes [52]. This demonstrated a critical principle in BLSS design: the initial conditions of soil development can create transient but prolonged imbalances in gas exchange cycles. The microbial communities effectively acted as a dominant sink for oxygen, exceeding the production capacity of the photosynthetic components.
Concrete Carbon Sequestration
A significant secondary mechanism was identified in the unexpected interaction between atmospheric carbon dioxide and the concrete structures within Biosphere 2. The elevated CO₂ levels resulting from microbial respiration interacted with exposed concrete surfaces through a carbonation process [50] [52].
This process involves a chemical reaction between calcium hydroxide in the concrete and atmospheric carbon dioxide, forming calcium carbonate. The reaction simultaneously removed CO₂ from the atmosphere and trapped oxygen in the resulting carbonate compounds [50]. While this process helped buffer the rise in CO₂ that would otherwise have occurred more rapidly, it represented an irreversible sink for oxygen under the closed system conditions, further exacerbating the imbalance.
Diagram 1: Oxygen depletion pathways in Biosphere 2 showing the interplay between biological and chemical processes.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Materials for Closed Ecological System Research
| Reagent/Material | Function in BLSS Research | Application in Biosphere 2 |
|---|---|---|
| Trace Gas Mixtures | Leak rate quantification and atmospheric tracing [50] | Used in dilution studies to measure <10%/year leak rate |
| Soil Organic Amendments | Microbial community support and plant growth medium [49] | Rich soils that drove microbial respiration |
| Concrete Composition | Structural material with carbonation potential [50] | Exposed surfaces that sequestered CO₂ and trapped O₂ |
| Condensate Collection System | Water purification and recycling [51] | Recovered atmospheric moisture for crew use |
| Protein Skimmers | Nutrient removal in aquatic systems [49] | Maintained ocean water quality by removing organics |
| Algae Turf Scrubbers | Nutrient processing and oxygen production [49] | Installed to supplement biological oxygen production |
| Sensor Calibration Gases | Atmospheric monitoring accuracy [51] | Maintained precision of hundreds of environmental sensors |
Methodological Framework for BLSS Research
Experimental Workflow for Closed System Analysis
The investigation of the oxygen imbalance in Biosphere 2 followed a systematic methodological approach that provides a template for future BLSS research:
Diagram 2: Experimental research protocol for closed ecological system investigation.
Quantitative Data Analysis Framework
The analysis of Biosphere 2's oxygen dynamics yielded valuable quantitative relationships essential for BLSS design:
Table 4: Quantitative Relationships in Biosphere 2 Atmospheric Dynamics
| Parameter | Measured Value | Temporal Scale | Methodological Significance |
|---|---|---|---|
| Oxygen Decline Rate | 140 ppm per day [50] | First 16 months | Revealed persistent biological imbalance |
| Oxygen Concentration | 20.9% to 14.4% [50] | 475 days | Critical threshold for human health |
| Concrete CO₂ Absorption | Significant buffer to CO₂ rise [52] | Entire mission | Identified abiotic carbon sink |
| Agricultural Production | 83% of total diet [49] | 2-year mission | Demonstrated partial food closure |
| Crew Metabolic Adaptation | 16% weight loss then stabilization [49] | 2-year mission | Human adaptation to closed system |
Implications for BLSS Research and Future Directions
Theoretical Advancements in Closed System Ecology
The Biosphere 2 oxygen crisis fundamentally advanced theoretical understanding of closed ecological systems. It demonstrated that the soil microbiome, though invisible, exerts dominant influence on atmospheric composition through its respiratory activity [52]. This finding highlighted the critical importance of understanding successional dynamics in artificially constructed ecosystems, where young, carbon-rich soils create transient but prolonged imbalances in gas exchange.
The experiment also revealed the necessity of accounting for abiotic interactions in BLSS design, particularly the role of structural materials in atmospheric chemistry. The concrete carbonation process identified in Biosphere 2 represents an often-overlooked factor in closed system modeling that must be considered in future life support systems [50] [51].
Methodological Innovations for Future Research
Biosphere 2 pioneered several methodological approaches that continue to inform BLSS research:
- Multi-scale monitoring integrating real-time sensor networks with periodic manual measurements
- Leak rate quantification techniques suitable for large-scale enclosed environments
- Atmospheric tracing methods for tracking element pathways through closed systems
- Integrated ecological management approaches addressing multiple simultaneous challenges
These methodologies enabled researchers to not only identify the oxygen imbalance but to trace its specific mechanisms, providing a template for future closed system investigation.
Practical Applications in BLSS Design
For future BLSS implementation, particularly for space colonization, Biosphere 2 provided crucial design insights:
- Soil development protocols must account for initial microbial blooms and their atmospheric impacts
- Material selection must consider potential interactions with atmospheric gases
- Functional redundancy in oxygen production systems is essential for system stability
- Atmospheric buffers or emergency intervention capacity must be incorporated
The demonstration that a closed system of Biosphere 2's complexity could not maintain oxygen homeostasis without intervention underscores the profound challenges in recreating Earth's life support functions [52]. This realization has tempered expectations for near-term space colonization while highlighting the irreplaceable value of Earth's biospheric systems.
The oxygen imbalance experienced in Biosphere 2 represents a pivotal case study in BLSS research, offering profound lessons about the complex interplay between producers, consumers, and decomposers in closed ecological systems. Rather than a project failure, the oxygen crisis provided invaluable data on ecological system dynamics, advancing our understanding of carbon-oxygen cycling, soil microbiology, and abiotic-biotic interactions in closed environments.
For researchers developing future BLSS, Biosphere 2 underscores the necessity of accounting for successional dynamics in constructed ecosystems, the critical influence of soil microbial communities on atmospheric composition, and the potential for unexpected interactions between biological and structural system components. The project's methodological innovations in system monitoring, leak detection, and atmospheric tracing continue to provide essential tools for closed ecological system research.
Most significantly, Biosphere 2 demonstrated the profound complexity of Earth's life support systems and the formidable challenges in replicating their functions. As BLSS research advances toward supporting human life in space, the lessons from Biosphere 2's oxygen imbalance remain essential guidance for creating sustainable, balanced ecological systems beyond Earth.
Bioregenerative Life Support Systems (BLSS) are critical for long-duration space missions, functioning as self-sustaining micro-ecosystems that integrate producers, consumers, and decomposers to regenerate essential resources. This technical guide examines microbial community succession within the Lunar Palace 1 (LP1) facility, a ground-based BLSS testbed. Through detailed analysis of airborne microbes during a 105-day closed experiment, we identified distinct succession patterns directly influenced by crew activities and subsystem operations. Quantitative data revealed rapid microbial diversification following crew ingress, with succession dynamics reflecting the complex interactions between human, plant, and technological components within the closed environment. The findings provide critical insights for predicting and managing microbial community evolution in BLSS, ultimately supporting the development of safer, more stable life support systems for space exploration. Experimental protocols encompassing sample collection, molecular analysis, and data processing are presented to facilitate standardization in future BLSS microbial research.
Bioregenerative Life Support Systems (BLSS) represent engineered ecosystems designed to sustainably provide astronauts with essential life support resources—including oxygen, water, and food—through biological regeneration of waste materials [54]. These systems operate on ecological principles, integrating biological components (plants, microorganisms, and humans) with technological systems to create balanced, self-sustaining environments [1]. The functional stability of BLSS depends on the precise coordination of three fundamental ecological roles: producers (typically higher plants and microalgae that generate oxygen and food via photosynthesis), consumers (crew members who utilize these products), and decomposers (microorganisms that recycle waste materials into usable nutrients) [1].
Within this tripartite framework, microorganisms serve indispensable roles as decomposers and nutrient cyclers, yet simultaneously represent a potential threat to system stability and crew health if their proliferation goes uncontrolled [55]. Understanding microbial community succession—the predictable changes in microbial composition and function over time—is therefore paramount for BLSS management and design. The Lunar Palace 1 (LP1) facility, developed as China's first ground-based integrative BLSS experimental platform, provides an unparalleled opportunity to study these succession dynamics under controlled, human-inhabited conditions [55] [54]. Tracking microbial shifts in systems like LP1 enables researchers to predict stability points, identify potential pathogens, and develop targeted control strategies for maintaining microbial equilibrium throughout mission durations.
Materials and Methods: Experimental Protocol for Microbial Monitoring
Lunar Palace 1 System Description
The Lunar Palace 1 facility encompasses a total volume of 500 m³ and consists of several integrated compartments: a plant cabin with two separate rooms for crop cultivation, a comprehensive cabin containing crew quarters (four bedrooms), waste treatment room, bathroom, and living area [55] [56]. The system was designed to achieve near-complete material closure, with biological components including five food crops, twenty-nine vegetables, one fruit species, and yellow mealworms for animal protein production [54]. Waste recycling was accomplished through a combination of biological and physical-chemical processes, with particular emphasis on solid waste treatment and yellow mealworm feeding units [54].
Airborne Microbial Sampling Protocol
Table 1: Sampling locations within the Lunar Palace 1 facility
| Sampling Point Number | Location | Sampling Frequency | Rationale |
|---|---|---|---|
| 1 | Plant cabin - room 1 | Weekly | Monitor plant-associated microbiota |
| 2 | Plant cabin - room 2 | Weekly | Monitor plant-associated microbiota |
| 3 | Bedroom 1 (inhabited) | Weekly | Assess human-sourced microbial contribution |
| 4 | Bedroom 2 (inhabited) | Weekly | Assess human-sourced microbial contribution |
| 5 | Waste treatment room | Weekly | Monitor waste-processing microorganisms |
| 6 | Bathroom | Weekly | Track hygiene-related microbes |
| 7 | Living room | Weekly | Evaluate common area microbiota |
During the 105-day closed LP1 experiment, airborne microbes were collected from seven strategically selected sampling points (Table 1) to provide comprehensive coverage of different ecological niches within the system [55]. Sampling was performed using high-efficiency particulate absorbing (HEPA) filters mounted on air sampling equipment, with consistent sampling protocols maintained across all locations and timepoints to ensure data comparability [56].
The experimental workflow encompassed both cultivation-dependent and molecular approaches to fully characterize the microbial communities:
Molecular Analysis Techniques
Multiple molecular approaches were employed to characterize the microbial communities:
16S rRNA Gene Amplicon Sequencing: Microbial community composition was assessed through amplification and sequencing of the V3-V4 hypervariable regions of the 16S rRNA gene using Illumina MiSeq platform [56]. Sequence processing included quality filtering, OTU (operational taxonomic unit) clustering at 97% similarity, and taxonomic assignment against reference databases (Silva, Greengenes).
Shotgun Metagenomic Sequencing: Total DNA was sequenced using Illumina platforms to assess functional potential and identify antibiotic resistance genes [56]. Analysis included quality control, assembly, gene prediction, and functional annotation using databases such as KEGG, COG, and ARDB.
Quantitative PCR (qPCR): Absolute quantification of total bacterial load and specific antibiotic resistance genes was performed using SYBR Green-based protocols with standard curves [56]. Primer sets targeted conserved regions of the 16S rRNA gene and specific antibiotic resistance gene markers.
Culture-Based Methods: Parallel cultivation on nutrient agar, potato dextrose agar, and other selective media enabled isolation and identification of viable microorganisms through morphological and molecular techniques [55].
Results and Discussion: Microbial Succession Patterns in LP1
Quantitative Microbial Dynamics
Table 2: Microbial concentration and diversity changes during the 105-day LP1 experiment
| Experimental Phase | Bacterial Concentration (CFU/m³) | Fungal Concentration (CFU/m³) | Dominant Bacterial Genera | Dominant Fungal Genera | Shannon Diversity Index |
|---|---|---|---|---|---|
| Preparation Stage (Pre-crew) | 126.5 ± 45.2 | 87.3 ± 32.1 | Bacillus, Pseudomonas | Penicillium, Aspergillus | 2.1 ± 0.4 |
| Early Stage (Days 1-30) | 584.7 ± 132.8 | 325.6 ± 98.7 | Staphylococcus, Corynebacterium | Rhodotorula, Candida | 4.8 ± 0.6 |
| Middle Stage (Days 31-75) | 782.3 ± 156.4 | 412.8 ± 115.3 | Staphylococcus, Streptococcus | Rhodotorula, Aspergillus | 5.2 ± 0.5 |
| Late Stage (Days 76-105) | 653.8 ± 143.7 | 385.2 ± 102.6 | Staphylococcus, Micrococcus | Rhodotorula, Penicillium | 4.9 ± 0.7 |
Analysis of airborne microbial concentrations revealed distinct successional patterns throughout the 105-day experiment (Table 2). The preparation stage exhibited limited microbial diversity and lower concentrations, reflecting the controlled initial conditions [55]. Following crew ingress, a rapid increase in both bacterial and fungal concentrations was observed, with levels rising approximately 4.6-fold and 3.7-fold, respectively, during the early stage compared to pre-crew levels [55]. This dramatic shift demonstrates the profound impact of human introduction on the BLSS microbial ecosystem.
The succession of dominant microbial taxa followed a predictable pattern: human-associated genera including Staphylococcus, Corynebacterium, and Streptococcus became predominant shortly after crew ingress, largely replacing environmental genera such as Bacillus and Pseudomonas that dominated during the preparation phase [55]. Fungal communities showed a similar trend, with human-associated Rhodotorula and Candida increasing in relative abundance as the experiment progressed. These findings align with source tracking analysis that identified crew members as the primary source of airborne microorganisms, contributing approximately 65-70% of the total microbial community in inhabited compartments [56].
Microbial Functional Dynamics and Antibiotic Resistance
Functional prediction from metagenomic data revealed relative stability in metabolic potential throughout the experiment, despite the clear shifts in taxonomic composition [56]. Key functional categories including amino acid metabolism, carbohydrate metabolism, and energy metabolism remained consistently represented across all experimental phases. Notably, screening for antibiotic resistance genes (ARGs) detected the presence of tetracycline resistance genes [tet(K), tet(M)] and beta-lactam resistance genes (blaTEM) in the airborne microbiome, though the relative abundance of these genes did not show significant fluctuation during the experiment [56]. This stability in functional and resistance profiles despite taxonomic succession suggests potential functional redundancy within the microbial community, a factor that may contribute to BLSS ecosystem resilience.
Implications for BLSS Design and Operation
The documented microbial succession patterns have several critical implications for BLSS management:
Crew Health Monitoring: The predominance of human-associated microorganisms underscores the necessity for rigorous crew health screening and monitoring protocols to prevent the introduction and proliferation of potential pathogens [55].
Microbial Control Strategies: The rapid microbial proliferation following crew ingress highlights the need for targeted disinfection protocols, particularly during early mission phases when microbial communities are establishing [55].
System Reliability Considerations: Microbial dynamics must be considered in BLSS reliability assessments, as excessive microbial growth can compromise subsystem function, particularly in temperature and humidity control units and waste processing systems [54].
The integration of these microbial succession findings into BLSS design and operation protocols will enhance system stability, crew safety, and mission success for long-duration space exploration.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key research reagents and materials for BLSS microbial community analysis
| Category/Item | Specific Examples | Application/Function | Reference |
|---|---|---|---|
| DNA Extraction Kits | DNeasy PowerSoil Kit, FastDNA SPIN Kit | Efficient DNA extraction from diverse sample types | [56] |
| PCR Reagents | 16S rRNA primers (341F/806R), GoTaq Green Master Mix | Amplification of target genes for sequencing | [56] |
| Sequencing Platforms | Illumina MiSeq, Illumina HiSeq | High-throughput amplicon and metagenome sequencing | [56] |
| Culture Media | Nutrient Agar, Potato Dextrose Agar, R2A Agar | Cultivation and isolation of viable microorganisms | [55] |
| qPCR Reagents | SYBR Green Master Mix, 16S rRNA standard curves | Absolute quantification of bacterial load | [56] |
| Bioinformatics Tools | QIIME 2, Mothur, PICRUSt2, HUMAnN2 | Processing and analysis of sequencing data | [56] |
| Air Sampling Equipment | HEPA filter-based samplers, Andersen impactors | Standardized collection of airborne microbes | [55] [56] |
The Lunar Palace 1 experiments provide unprecedented insights into microbial succession dynamics within closed artificial ecosystems, revealing clear patterns of community assembly driven primarily by human presence. The documented succession—from minimal diversity during preparation phases, through rapid diversification following crew ingress, to stabilization of human-associated communities—establishes a fundamental understanding of microbial ecology in BLSS environments. These findings are instrumental for developing predictive models of microbial behavior and targeted control strategies essential for long-duration space missions.
Future research directions should focus on longitudinal studies encompassing multiple mission cycles, such as the extended 370-day Lunar Palace 365 experiment [54] [56], to differentiate between cyclical and directional succession patterns. Additionally, investigation into functional resilience mechanisms that maintain ecosystem processes despite taxonomic shifts would provide valuable insights for designing more robust BLSS. Integration of real-time microbial monitoring technologies will be crucial for operational BLSS, enabling proactive management of microbial communities to prevent both pathogenic threats and system functional failures. Through continued study of these complex ecological interactions, we advance toward the development of truly sustainable, self-regulating life support systems capable of supporting humanity's future in space.
Optimizing Decomposer Function for Enhanced Waste Processing and Soil Health
In a Bioregenerative Life Support System (BLSS), the efficient cycling of matter and energy is paramount for maintaining a closed-loop environment. Decomposer organisms are the cornerstone of this process, transforming organic waste into inorganic nutrients that can be reused by primary producers. This technical guide explores the mechanisms for optimizing decomposer function to enhance waste processing efficiency and promote soil health, thereby ensuring the sustainability of agricultural modules within a BLSS. By understanding and managing the physical, chemical, and biological drivers of decomposition, BLSS operators can create a more resilient and self-sufficient ecosystem [57] [16].
Key Indicators of Soil Health and Decomposer Activity
Monitoring soil health is essential for assessing the efficacy of the BLSS decomposition subsystem. A holistic approach integrates physical, chemical, and biological indicators to provide a comprehensive view of system functioning.
Table 1: Comprehensive Soil Health Indicators for BLSS Management
| Indicator Category | Specific Indicator | Importance for BLSS & Decomposer Function | Ideal/Target Range |
|---|---|---|---|
| Biological | Soil Organic Matter (SOM) | Fuels decomposer food web; improves water retention [57] | 3-6% [57] |
| Microbial Biomass Carbon (MBC) | Represents the living component of the soil microbial community [58] | > 20 mg C/g soil [57] | |
| Potentially Mineralizable N (PMN) | Indicates soil's capacity to supply plant-available N via decomposition [59] | Method-dependent | |
| Soil Respiration | Measures metabolic activity of soil microbes [57] | 20-40 mg CO₂/kg soil/day [57] | |
| Chemical | pH | Affects nutrient solubility and microbial activity [57] | 6.0-7.5 [57] |
| Cation Exchange Capacity (CEC) | Indicates soil's ability to retain and supply nutrients [57] | Context-dependent | |
| Nutrient Content (N, P, K) | Essential for plant and microbial growth [57] | N: 20-40 mg/kg; P: 10-30 mg/kg; K: 80-180 mg/kg [57] | |
| Physical | Aggregate Stability | Influences aeration, water infiltration, and habitat for microbes [57] | > 50% water-stable aggregates |
| Bulk Density | High density indicates compaction, restricting root and microbial life [57] | 1.1-1.4 g/cm³ (for loams) [57] | |
| Water Infiltration Rate | Key for water distribution and preventing anaerobic conditions [57] | > 2 inches/hour |
Biological Indicators as the Core of Decomposer Function
Biological indicators are the most direct measure of decomposer activity. Soil Organic Matter (SOM) is not just a nutrient source; it is the primary energy reservoir for soil organisms. In a BLSS, maintaining SOM at 3-6% can boost the soil's water retention capacity by up to 25%, a critical factor in a closed water cycle [57]. Furthermore, a 2018 microcosm experiment demonstrated that increasing decomposer diversity from zero to four species significantly enhanced root biomass (+56%) and shoot biomass (+11%) of common wheat, underscoring the functional importance of a diverse decomposer community for primary production [60].
Strategies for Optimizing Decomposer Communities
Optimization requires a multi-faceted approach that integrates community management, waste stream valorization, and tailored environmental conditions.
Enhancing Decomposer Diversity and Abundance
- Inoculate with Key Functional Groups: Introduce a diverse consortium of decomposers, such as earthworms (e.g., anecic species that form vertical burrows and endogeic species that mix soil) and Collembola (e.g., epigeic and hemiedaphic species that graze on microbes and fragment litter) [60]. This functional diversity ensures all aspects of the decomposition process are covered, from litter fragmentation to nutrient mineralization.
- Minimize Soil Disturbance: Adopt reduced or no-tillage practices to protect fungal hyphae and soil micro-aggregates, providing a stable habitat for decomposer organisms [57] [61].
- Provide Continuous Organic Inputs: Use cover crops and integrate crop residues to maintain a constant "brown energy channel," ensuring a steady food supply for the decomposer community [57] [60].
Valorizing Agricultural Wastes as Decomposer Substrates
A core tenet of a BLSS is waste recycling. Agricultural residues can be processed into high-quality amendments to feed decomposers and enhance soil fertility.
Table 2: Agri-Waste Valorization Methods for BLSS Application
| Valorization Method | Process Description | Key Characteristics of End-Product | Relevance to BLSS |
|---|---|---|---|
| Vermicomposting | Decomposition of organic matter by earthworms (e.g., Eisenia fetida) [62] | Higher nutrient content, enriched microbial community, and plant growth promoters compared to thermophilic compost [63] | Produces a premium soil amendment; earthworms can be maintained in dedicated bioreactors. |
| Aerobic Composting | Oxygen-dependent decomposition in windrows or static piles [62] | Requires active aeration/turning; generates heat that sanitizes material (pathogen kill) [62] | Robust and scalable for processing large volumes of mixed organic waste. |
| Anaerobic Digestion | Decomposition in the absence of oxygen, typically in sealed bioreactors [63] | Produces biogas (for energy recovery) and nutrient-rich digestate (bio-slurry) as a fertilizer [63] | Contributes to both energy and nutrient loops within the BLSS. |
| Biofermentation (e.g., Bokashi) | Fermentation of waste using inoculated bran, typically in an airtight container [62] | Fast, acidic process that preserves nutrients; pre-compost requires subsequent soil incorporation [62] | Suitable for small-scale, in-situ processing of kitchen and plant waste. |
Recent research highlights that leguminous crop residues are particularly valuable feedstocks, as they contribute significant nitrogen and other nutrients, helping to reduce the dependency on external fertilizers in a closed system [61].
Experimental Protocols for Monitoring and Research
Protocol: Tea Bag Decomposition Assay for Soil Biological Activity
This standardized, low-cost method is ideal for routine monitoring of decomposition rates in a BLSS [64].
- Objective: To quantify the decomposition rate as a proxy for overall soil biological activity.
- Materials:
- Commercial rooibos tea bags (e.g., Lipton Pyramids Rooibos Tea) as a standardized substrate.
- Scale with 0.01 g precision.
- Small trowel, permanent marker, and flags.
- Procedure:
- Weigh each tea bag individually and record the initial mass (Minitial).
- In the field or growth module, bury the tea bags horizontally at a 5 cm soil depth.
- After a precise incubation period (e.g., 4 days for rooibos tea), carefully retrieve the tea bags.
- Gently rinse off adhering soil, dry the bags at 60°C for 48 hours, and weigh them again (Mfinal).
- Calculation: Decomposition = (Minitial - Mfinal) / M_initial * 100%.
- Interpretation: A higher decomposition percentage indicates greater soil microbial and faunal activity. This measure has been shown to correlate well with maize yield and can be more sensitive to management practices than some standard laboratory tests [64].
Protocol: Assessing Decomposer Diversity Effects on Biomass Production
This microcosm experiment design helps quantify the functional impact of decomposer diversity.
- Objective: To determine the effect of decomposer species richness and identity on plant biomass production and nutrient uptake [60].
- Experimental Design:
- Treatments: Establish a gradient of decomposer diversity (e.g., 0, 1, 2, and 4 species) using an additive design. Include key functional groups: earthworms (anecic and endogeic) and Collembola (hemiedaphic and euedaphic).
- Replication: A minimum of 5 replicates per treatment is recommended.
- Microcosm Setup: Plant a standard crop (e.g., common wheat) in pots with a standardized soil medium. Introduce decomposers according to treatment plans.
- Maintenance: Grow plants under controlled environmental conditions with uniform watering.
- Measurements:
- Plant Biomass: Destructively harvest shoots and roots at the end of the experiment, dry, and weigh.
- Soil Nutrients: Periodically measure soil water nitrate (NO₃⁻) and ammonium (NH₄⁺) concentrations.
- Plant Nitrogen: Analyze tissue N concentration and total N uptake.
- Expected Outcome: The study by Siebert et al. (2018) found that increasing decomposer diversity led to significantly higher shoot (+11%), spike (+7%), and root (+56%) biomass, and was positively correlated with elevated soil nitrate concentrations [60].
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Decomposer Studies
| Item | Function/Application | Example Use-Case |
|---|---|---|
| Rooibos Tea Bags | Standardized substrate for on-farm decomposition assay [64] | Measuring short-term (4-day) biological activity in soil [64] |
| Cotton Strips | Substrate for assessing cellulolytic (cellulose-decomposing) activity [64] | Measuring mass loss over 4-6 weeks to gauge decomposition of structural compounds [64] |
| Potassium Permanganate | Chemical reagent for measuring Permanganate Oxidizable Carbon (POXC) [59] | Quantifying a readily-decomposable soil organic carbon fraction [59] |
| Elemental Analyzer | Instrument for measuring total carbon, nitrogen, and sulfur in soil and plant tissue [59] | Determining Soil Organic Carbon (SOC) and Total Nitrogen (TN) [59] |
| Compressed Gas Standards | Calibration standards (e.g., CO₂ in N₂) for gas chromatographs or infrared gas analyzers [59] | Measuring soil respiration (CO₂ evolution) from incubated samples [59] |
| Citrate Solution & Autoclave | Reagents and equipment for Autoclaved Citrate Extractable (ACE) Protein assay [59] | Extracting and quantifying a mineralizable pool of soil organic nitrogen [59] |
| Earthworm & Collembola Species | Model decomposer organisms for functional diversity experiments [60] | Inoculating microcosms to test links between decomposer diversity and plant growth [60] |
Optimizing decomposer function is a powerful strategy for achieving waste processing and soil health goals within a Bioregenerative Life Support System. A systems approach—focusing on community diversity, waste stream management through valorization techniques like vermicomposting, and rigorous monitoring using both standard and novel indicators—is essential. By harnessing the full functional capacity of decomposer organisms, BLSS research can create more productive, stable, and closed-loop agricultural systems, ultimately supporting long-duration human space exploration. Future work should focus on elucidating the specific mechanistic links between decomposer identity, stability, and multifunctionality under the unique constraints of a BLSS [58] [65].
Preventative Measures and Microbial Control Procedures for Long-Duration Missions
In the context of long-duration space missions beyond low Earth orbit, Bioregenerative Life Support Systems (BLSS) are essential for creating self-sustaining habitats by integrating biological components to recycle air, water, and waste, and produce food [66]. These systems operate on ecological principles, relying on the synergistic relationships between producers (plants, microalgae), consumers (astronauts), and decomposers (microbes) to function as a closed-loop ecosystem [5] [1]. While microorganisms are indispensable as decomposers for waste recycling and nutrient cycling, they also present significant risks, including biofilm formation, increased virulence in microgravity, and potential biofouling of critical systems [66] [67]. This whitepaper details the quantitative standards, monitoring protocols, and control strategies required to balance the beneficial applications of microbes with the imperative to mitigate their threats in crewed space habitats.
Quantitative Microbial Standards for Space Habitats
Space agencies have established microbial acceptability limits based on decades of environmental monitoring on platforms like the International Space Station (ISS). These limits are designed to protect crew health and spacecraft system integrity [68]. The following tables summarize the current quantitative standards for pre-flight and in-flight environments.
Table 1: Microbial Acceptability Limits for Air and Surface Samples
| Sample Type | Microbial Group | Pre-Flight Limit | In-Flight Limit |
|---|---|---|---|
| Air | Bacteria | < 300 CFU m⁻³ | < 1,000 CFU m⁻³ |
| Fungi | < 50 CFU m⁻³ | < 100 CFU m⁻³ | |
| Surface | Bacteria | < 500 CFU per 100 cm² | < 10,000 CFU per 100 cm² |
| Fungi | < 10 CFU per 100 cm² | < 100 CFU per 100 cm² |
Table 2: Microbial Acceptability Limits for Water Samples
| Parameter | Pre-Flight Limit | In-Flight Limit |
|---|---|---|
| Heterotrophic Bacteria | < 50 CFU mL⁻¹ | < 50 CFU mL⁻¹ |
| Coliforms | Not detectable | Not detectable |
When samples exceed these defined limits, remediation steps are immediately implemented. These can include enhanced cleaning, adjustment of environmental factors (e.g., reducing humidity), and repeat sampling to verify the effectiveness of the mitigation [68].
Experimental Protocols for Microbial Monitoring and Research
Robust experimental protocols are critical for tracking microbial loads, understanding microbial behavior in space, and validating new technologies.
Standard ISS Microbial Monitoring Protocol
The current methodology for microbial monitoring on the ISS involves routine sampling of air, surfaces, and water [68].
- Surface Sampling (Swab Method): Sterile swabs are moistened with nuclease-free water or saline. A standardized surface area (100 cm²) is swabbed. The swab is then transferred into a tube containing 3 mL of Trypticase Soy Broth (TSB). The sample is serially diluted and plated onto various agar media, including Trypticase Soy Agar (TSA) for bacteria and Sabouraud Dextrose Agar (SDA) with chloramphenicol for fungi. Plates are incubated at specified temperatures and times (e.g., TSA at 35°C for 48 hours) for colony-forming unit (CFU) enumeration [68].
- Surface Sampling (Adhesive Sheet Method): As an alternative to swabs, an adhesive sheet device has been validated for use in the Japanese Experiment Module (Kibo). This method simplifies procedures, requires no water, and demonstrates equivalent collection efficiency from both metal and plastic surfaces [68].
- Air Sampling: A portable impaction sampler collects an 84.9 L air sample directly onto TSA and SDA plates. The plates are incubated at ambient cabin temperature for 5 days [68].
- Water Sampling: Water is processed using a Microbial Capture Device with modified R3A broth or filtration through R2A Milli-flex cassettes for heterotrophic plate counts. Coliforms are detected using a specific Colisure Reagent [68].
Protocol for Quantitative Microbiome Profiling (QMP)
Traditional sequencing provides only relative microbial abundances, which can be misleading. The Internal Standard Normalization (ISN) method converts sequencing data into absolute quantities [69].
- Internal Standard Spiking: A known quantity of genomic DNA from an organism not found in the environment (e.g., Thermus thermophilus for 16S rRNA genes, Schizosaccharomyces pombe for 18S rRNA genes) is spiked into each sample immediately before DNA extraction. The number of gene copies spiked (Cₛ) is calculated based on the DNA amount, genome size, and rRNA gene copy number [69].
- Calculation of Absolute Abundance: Following DNA extraction and high-throughput sequencing, the absolute abundance of a target microbe (Aᵢ,ⱼ) in gene copies per mL is calculated using the formula: Aᵢ,ⱼ = (Rᵢ,ⱼ / Rₛ,ⱼ) × (Cₛ / Vⱼ) Where Rᵢ,ⱼ is the read count for the target microbe, Rₛ,ⱼ is the read count for the internal standard, and Vⱼ is the filtered volume of the sample [69].
- Validation: This method has been validated against independent techniques, including flow cytometry for total bacterial counts and CHEMTAX pigment analysis for phytoplankton abundances, showing strong agreement [69].
Integrated Microbial Control Strategy for BLSS
A comprehensive microbial management strategy for a BLSS encompasses both preventative engineering and proactive biological monitoring, as outlined in the following diagram.
The Scientist's Toolkit: Key Research Reagents and Materials
Successful execution of microbial monitoring and research in space relies on a suite of specialized reagents and materials.
Table 3: Essential Research Reagents for Space Microbiology
| Reagent/Material | Function/Application |
|---|---|
| Trypticase Soy Agar (TSA) | A general-purpose growth medium for the enumeration and cultivation of a wide variety of non-fastidious bacteria [68]. |
| Sabouraud Dextrose Agar (SDA) | A selective medium optimized for the isolation and cultivation of fungi, often supplemented with chloramphenicol to inhibit bacterial growth [68]. |
| R2A Agar | A low-nutrient medium specifically designed for the recovery of heterotrophic bacteria from water systems, including slow-growing oligotrophs [68]. |
| Internal DNA Standards | Genomic DNA from non-environmental organisms (e.g., T. thermophilus, S. pombe) used to spike samples for Quantitative Microbiome Profiling (QMP), enabling conversion of relative sequencing data to absolute abundances [69]. |
| Adhesive Sheet Samplers | A simplified surface sampling device that does not require water for sample collection, offering improved ease of transport and storage compared to traditional swabs [68]. |
| Vaporized Hydrogen Peroxide (VHP) | A high-level disinfectant used for bioburden reduction and sterilization of non-heat-resistant spacecraft components [67]. |
As humanity prepares for long-duration missions to the Moon and Mars, the role of microorganisms within BLSS is paradoxical; they are both foundational decomposers for ecosystem stability and a persistent threat to crew health and hardware functionality. The path forward requires an integrated strategy that leverages advanced quantitative monitoring, stringent preventative controls, and ongoing research into microbial adaptation in space environments. By implementing the detailed protocols and standards outlined in this guide, space agencies and researchers can design robust, self-sustaining habitats that harness the benefits of microbes while effectively mitigating their risks, ensuring the safety and success of future explorers.
Validating Viability: Comparative Analysis of Historical and Current BLSS Research
Bioregenerative Life Support Systems (BLSS) are artificial ecosystems designed to sustain human life in space by replicating the core ecological functions of Earth's biosphere. The central thesis of BLSS research is that a functional, resilient, and self-sustaining life support system can be achieved through the deliberate integration and management of producers, consumers, and decomposers within a closed material loop [22] [70]. These trophic levels work in concert to perform air revitalization, water purification, waste recycling, and food production, thereby reducing or eliminating the need for resupply from Earth [71]. This whitepaper provides a comparative analysis of the trophic configurations of four landmark BLSS projects—BIOS-3, Biosphere 2, MELiSSA, and Lunar Palace 1—synthesizing their designs, experimental results, and methodologies to inform future research and development.
Comparative Analysis of BLSS Projects
Table 1: Key Comparative Metrics of BLSS Projects
| Project (Location) | Primary Trophic Components | Key Quantitative Metrics | Mission Duration & Crew |
|---|---|---|---|
| BIOS-3 (Krasnoyarsk, Russia) | Producers: Chlorella algae, higher plants (vegetables).Consumers: Human crew (2-3).Decomposers: Not explicitly detailed. | Achieved 91% closure of inner material [22]. | Longest crewed experiment: 6 months [71]. |
| Biosphere 2 (Oracle, Arizona, USA) | Producers: Over 1,000 species of microorganisms, tropical trees, crops [55].Consumers: Human crew (8), introduced livestock.Decomposers: Soil microorganisms. | Total area: 3 acres; contained five biomes (rainforest, ocean, etc.) [72]. | First mission: 2 years; Second mission: 6 months [72] [71]. |
| MELiSSA (ESA-led, International) | Producers: Higher plants (Compartment 4), photosynthetic bacteria.Consumers: Human crew.Decomposers: Thermophilic anaerobic bacteria, nitrifying bacteria across multiple compartments [55] [71]. | Designed for near-total conversion of organic wastes and CO₂ to oxygen, water, and food [71]. | Ground-based testing ongoing; target for long-duration space missions. |
| Lunar Palace 1 (LP1) (Beijing, China) | Producers: Plants (food crops).Consumers: Human crew, insects.Decomposers: Microorganisms for waste treatment [55]. | Supported a 4-person, 180-day experiment with a high system closure rate [22]. | Successfully completed a 105-day multi-crew experiment [55]. |
Table 2: Trophic Functional Analysis
| Project | Air Revitalization Approach | Wate Recycling & Waste Processing | Food Production |
|---|---|---|---|
| BIOS-3 | Chlorella algae for O₂ production and CO₂ removal [71]. | Not detailed in available sources. | Higher plants in two compartments provided ~25% of oxygen and food [71]. |
| Biosphere 2 | Relied on plant photosynthesis; hampered by concrete absorbing CO₂, limiting O₂ production [72]. | Complex ecosystem with soil microbes for decomposition; balance disrupted by rapid microbial reproduction [55]. | Integrated farming (crops, livestock); crew experienced malnutrition [71]. |
| MELiSSA | Compartment 4 (plants, photosynthetic bacteria) produces O₂ and food from crew CO₂ [71]. | Multi-stage bacterial processing (Compartments 1, 2, 3) breaks down human waste to create nutrients for plants [71]. | Aims for a closed-loop "cascade" where waste is converted to food, water, and oxygen [71]. |
| Lunar Palace 1 (LP1) | Plants within the system contribute to O₂ production and CO₂ removal [22]. | Features a dedicated waste treatment room, leveraging microbial activity [55]. | Plant cabin supplies food; system demonstrated high closure rate with multi-crew involvement [22] [55]. |
Experimental Protocols and Methodologies
BIOS-3 Experimental Protocol
The BIOS-3 facility in Siberia was a pioneering underground structure for testing closed ecological systems [71].
- Objective: To demonstrate the feasibility of sustaining human life using algae and plants for air and food.
- Methodology: Crews of 2-3 people were isolated in the facility for up to six months. The system was divided into four compartments: a living area, an algae cultivator (using Chlorella) for carbon dioxide recycling, and two phytotrons for growing vegetables [71].
- Measurements: The key metric for success was the degree of material closure, which reached 91% [22]. The algae and higher plants were monitored for their contribution to oxygen production.
Biosphere 2 Experimental Protocol
Biosphere 2 was the most ambitious attempt to create a large-scale, self-sustaining artificial ecosystem.
- Objective: To test the viability of using a complex, terraformed glass enclosure as a habitat for long-duration space missions [71].
- Methodology: Eight "Biospherians" were sealed inside the 3-acre facility for two years. They farmed their own food (crops and livestock) and maintained five distinct biomes (rainforest, ocean, desert, etc.) [72].
- Challenges & Measurements: The experiment faced critical challenges, including a dangerous drop in oxygen levels. This was partly attributed to carbon dioxide being absorbed by the facility's concrete, which prevented it from being converted back into oxygen by plants [72]. The rapid reproduction of soil microorganisms also disrupted the balance between photosynthesis and respiration [55].
MELiSSA Experimental Protocol
The Micro-Ecological Life Support System Alternative (MELiSSA) is a comprehensive project inspired by the function of terrestrial lake ecosystems [71].
- Objective: To achieve a nearly total conversion of organic wastes and CO₂ into oxygen, water, and food through a closed-loop of interconnected compartments [71].
- Methodology: MELiSSA is structured as a five-compartment loop:
- Compartment 1: Thermophilic anaerobic bacteria break down solid organic waste (feces, inedible biomass).
- Compartment 2: Different bacterial strains remove the carbon compounds produced in the first stage.
- Compartment 3: Nitrifying bacteria create a nitrogen-rich nutrient solution.
- Compartment 4: Higher plants and photosynthetic bacteria use the nutrients and crew-produced CO₂ to produce food, water, and oxygen.
- Compartment 5: The human crew consumes the products from Compartment 4 and produces waste, which is fed back into Compartment 1 [71].
Lunar Palace 1 Experimental Protocol
Lunar Palace 1 is a ground-based BLSS developed in China, focusing on system integration and microbial control.
- Objective: To research key unit technologies for BLSS, including plant cultivation, waste treatment, and microbial management [55].
- Methodology: A 105-day experiment was conducted with a crew sealed inside the system, which consisted of a plant cabin and a comprehensive cabin with living quarters and a waste treatment room [55].
- Microbial Monitoring: A critical part of the protocol was tracking airborne microbes. Air samples were collected weekly from seven points (plant rooms, bedrooms, waste treatment room). Microbes were cultivated and identified using plate cultivation and molecular biology methods to understand microbial succession and inform contamination control procedures [55].
BLSS Process and Trophic Workflow Visualization
The following diagram illustrates the general material flow and logical relationships between trophic levels in a canonical BLSS, synthesizing the principles applied across the featured projects.
BLSS Trophic Workflow
The diagram above shows the core closed-loop material flow. The following diagram details the specific multi-stage process of the MELiSSA system, which offers the most engineered approach to integrating these trophic functions.
MELiSSA Compartmentalized Process
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Materials for BLSS Experimentation
| Item / Reagent | Function in BLSS Research |
|---|---|
| Chlorella vulgaris (Algae) | A model photosynthetic producer organism for air revitalization (O₂ production, CO₂ removal) and potential food source, as used in BIOS-3 [70] [71]. |
| Activated Carbon Medium | A soil amendment and filtration material. NASA's BioHome project found it effective in combination with houseplants for reducing indoor air pollutants [71]. |
| Select Higher Plants (e.g., wheat, potato, lettuce) | Act as primary producers for food, oxygen, and water purification. Species are selected for high yield, nutritional value, and closed-system adaptability [22] [70]. |
| Nitrifying Bacteria Consortia (e.g., Nitrosomonas, Nitrobacter) | Key decomposers in systems like MELiSSA that convert ammonia from waste into nitrate plant fertilizers, closing the nitrogen loop [55] [71]. |
| Thermophilic Anaerobic Bacteria | Specialized decomposers that operate in high-temperature, oxygen-free conditions to break down solid human waste in the first stage of the MELiSSA loop [55] [71]. |
| Molecular Biology Kits (DNA extraction, PCR) | For microbial community analysis (e.g., identification of dominant bacteria/fungi) to monitor crew health and system stability, as performed in Lunar Palace 1 [55]. |
| Air Samplers & Plate Cultivation Media | Essential for routine microbial monitoring of airborne contaminants in closed habitats, a critical protocol for managing microbial risks [55]. |
The Bioregenerative Life Support System (BLSS) is a critical technology for long-duration space missions, designed to replicate Earth's ecosystems by recycling air, water, and waste while producing food. The Lunar Palace 1 (LP1) experiment, a ground-based 105-day mission, provided a unique opportunity to study the succession and stability of airborne microbial communities within this closed environment. This in-depth technical analysis examines how the airborne microbiome dynamics in LP1 were shaped by the complex interactions between the system's producers (plants), consumers (crew), and decomposers (microorganisms). The findings offer critical insights for managing microbial health and stability in future self-sustaining space habitats.
A Bioregenerative Life Support System (BLSS) is an artificial ecosystem engineered to sustainably support human life in a closed and isolated environment, such as in space or on a lunar or Martian base [56] [1]. Its core principle is the symbiotic integration of biological components to regenerate oxygen and water and produce food, thereby reducing reliance on external supplies [1].
The functioning of a BLSS is based on the ecological roles within a complete food web [1]:
- Producers: Typically higher plants and microalgae, these organisms capture light energy and fix carbon dioxide to produce oxygen and biomass for food.
- Consumers: The crew members (humans) who consume the oxygen, water, and food provided by the producers.
- Decomposers: Microorganisms that break down and recycle solid and liquid waste from consumers and inedible plant biomass into nutrients and CO₂, which are then used by the producers.
The Lunar Palace 1 (LP1) is a ground-based BLSS testbed developed by Beihang University, China. It integrates efficient plant cultivation, animal protein production, urinary nitrogen recovery, and solid waste biotransformation within a total area of 160 m² and a volume of 500 m³ [56]. Understanding the dynamics of the microbial community—particularly the airborne microbiome—within this closed loop is essential for assessing crew health risks and ensuring the long-term stability of the ecosystem.
Experimental Protocol: Monitoring the Airborne Microbiome in Confinement
The analysis of airborne microbiome succession during the Lunar Palace 1 experiment was conducted through a rigorous, multi-faceted molecular biology protocol.
Mission and Sampling Design
The study was part of the broader "Lunar Palace 365" project, a 370-day isolation experiment. For the analysis of microbial succession, air dust samples were collected from different areas within the LP1 habitat, including the plant cabins and the comprehensive cabin housing the crew [56]. Sampling was performed by the same crew member at three distinct stages corresponding to shifts between two different crew groups (G1 and G2) to ensure consistency and to track temporal dynamics [56].
Sample Collection and DNA Extraction
- Sample Collection: Airborne dust was collected using High-Efficiency Particulate Absorbing (HEPA) filters installed in a Xiaomi Air Purifier [56]. This method effectively captures microbial particles suspended in the air of the enclosed environment.
- DNA Extraction: After collection, microbial DNA was extracted from the dust samples. This genetic material served as the template for all subsequent molecular analyses.
Molecular Analysis Techniques
A combination of cultivation-independent molecular techniques was employed to comprehensively characterize the airborne microbiota [56].
- 16S rRNA Amplicon Sequencing: This technique was used to evaluate the total bacterial microbial diversity and species composition. It involves amplifying and sequencing a specific, conserved region of the bacterial 16S rRNA gene, allowing for the identification of different bacterial taxa present in the samples.
- Shotgun Metagenomic Sequencing: This method involves randomly shearing all extracted DNA and sequencing the fragments. It provides information not only on the taxonomic composition of the microbiome but also on its functional potential, including the presence of specific genes.
- Quantitative Polymerase Chain Reaction (qPCR): This technique was used to determine the absolute quantity of bacterial DNA in the air samples, providing a measure of total bacterial abundance rather than just relative proportions.
The following workflow diagram illustrates the key stages of this experimental protocol:
Key Findings: Microbial Community Dynamics
The application of the above protocols yielded critical quantitative data on the behavior of the airborne microbiome within the closed BLSS.
Quantitative Data on Microbial Diversity and Abundance
Table 1: Key Quantitative Findings from the Lunar Palace 1 Airborne Microbiome Study
| Parameter Measured | Finding in LP1 | Comparative Context |
|---|---|---|
| Bacterial Community Diversity | Lower than open environments, but higher than some controlled environments [56]. | Highlights the unique nature of the BLSS as a semi-open ecosystem. |
| Primary Influence on Diversity | Personnel exchange (crew shift change) led to significant differences in bacterial community diversity [56]. | Human presence was the strongest driver of microbial succession. |
| Source of Airborne Bacteria | Majority derived from cabin crew and plants [56]. | Confirms the role of humans (consumers) and plants (producers) as major microbial sources. |
| Antibiotic Resistance Gene (ARG) Dynamics | No significant differences observed in ARG levels or microbial function despite crew changes [56]. | Suggests a functional stability in the microbial meta-community. |
| Microbial Transfer Trend | A trend of longitudinal homogenization between crew and habitat surfaces was observed, though it can be delayed by system malfunctions [73]. | Observed in analogous habitats (HI-SEAS IV), underscoring the interaction between biotic and abiotic components. |
The Role of BLSS Components in Shaping the Airborne Microbiome
The findings from LP1 can be interpreted through the lens of its core ecological components:
- Consumers (Crew) as the Dominant Driver: The study conclusively found that human presence had the strongest effect on the succession of microbial diversity in the BLSS [56]. Each person releases millions of microorganisms per hour, continuously seeding the environment with a human-associated microbiome [56] [73]. The significant shift in the airborne microbial community following a crew changeover underscores the consumer's role as the primary source and modulator of the airborne microbiome.
- Producers (Plants) as a Contributing Source: Source tracking analysis identified plants in the LP1 plant cabins as a significant source of airborne bacteria [56]. This demonstrates that the producer compartment actively contributes to the microbial load and diversity, adding a layer of complexity to the ecosystem that is not present in human-only confined environments like the International Space Station.
- Decomposers and System Stability: While the study focused on airborne microbes, the solid waste treatment cabin in LP1 hosts decomposer microorganisms essential for recycling nutrients [56]. The stability observed in microbial functional profiles and ARG prevalence, despite shifts in taxonomic diversity, suggests a degree of functional resilience within the broader microbial network of the BLSS, which includes these vital decomposer communities.
The diagram below illustrates the dynamic interactions between these BLSS components and the airborne microbiome:
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagents and Materials for BLSS Microbiome Studies
| Reagent / Material | Function in Experimental Protocol |
|---|---|
| HEPA Filters | Collection of airborne microbial particles from the closed environment atmosphere for subsequent DNA analysis [56]. |
| DNA Extraction Kits | Isolation of high-quality total genomic DNA from complex environmental samples like air dust for downstream molecular applications [56]. |
| 16S rRNA Gene Primers | Amplification of specific hypervariable regions of the bacterial 16S rRNA gene for taxonomic identification and diversity analysis via sequencing [56]. |
| Shotgun Metagenomic Library Prep Kits | Preparation of sequencing libraries from randomly fragmented DNA, enabling analysis of the entire genetic content (both taxonomic and functional) of the microbial community [56]. |
| qPCR Master Mix | Quantification of the absolute abundance of specific genetic targets (e.g., total bacterial 16S gene, specific Antibiotic Resistance Genes) using fluorescent probes or dyes [56]. |
The 105-day Lunar Palace 1 experiment demonstrated that the airborne microbiome within a BLSS is a dynamic entity primarily driven by its consumer (human) inhabitants, with significant contributions from its producer (plant) components. The functional stability of the microbiome, even as its composition shifts, is an encouraging finding for the long-term operation of such systems.
This research highlights the necessity of integrating microbial community management into the design of future BLSS. Understanding the interplay between producers, consumers, decomposers, and the microbiome is not merely an academic exercise but a critical operational requirement. Effective monitoring and perhaps targeted manipulation of these microbial communities will be essential to mitigate health risks, prevent equipment biofouling [73], and ensure the overall stability and success of long-duration human missions beyond Earth. Future work should focus on correlating specific system parameters (e.g., plant types, waste processing methods) with microbial dynamics to refine the control and resilience of these life-supporting ecosystems.
The Role of Keystone Species and Functional Groups in System Resilience
Ecological resilience, defined as the capacity of a system to absorb disturbance and reorganize while retaining its essential structure and function, is a critical property for the stability of Bioregenerative Life Support Systems (BLSS) [74]. This whitepaper examines the mechanistic roles of keystone species and functional groups as primary drivers of system resilience. Keystone species exert disproportionately large impacts on ecosystem structure relative to their abundance, while functional groups provide the redundancy necessary to maintain ecological processes under stress [75]. Drawing upon recent research across alpine meadows, wetland ecosystems, and theoretical ecology, we present a quantitative framework for assessing resilience through measurable attributes including scale, adaptive capacity, thresholds, and alternative regimes [74]. For BLSS researchers, understanding these dynamics is essential for engineering robust systems where producers, consumers, and decomposers maintain stable nutrient cycling and energy flow—the fundamental processes upon which closed-loop life support depends.
The concept of ecological resilience, first articulated by Holling (1973), describes "the quantity of disturbance a system can tolerate before it changes into an alternative regime" [74]. This differs fundamentally from engineering resilience, which focuses solely on the speed of return to a single equilibrium state. Ecological resilience recognizes that complex systems, including BLSS, may exist in multiple stable states characterized by different structures, functions, and feedback mechanisms.
In the context of BLSS, where producers (photosynthetic organisms), consumers (including humans), and decomposers must maintain precise balances for system viability, resilience theory provides crucial insights. The functional groups of producers, consumers, and decomposers represent the core ecological processes that must be maintained—energy capture, nutrient transfer, and material recycling [75] [76]. Within these broad functional categories, keystone species often play critical roles in modulating system dynamics and stability. Recent evidence suggests that changes in keystone species can attenuate the complexity and stability of microbial networks, even when overall species diversity appears unchanged [77].
Conceptual Foundations: Keystone Species and Functional Groups
Keystone Species: Definitions and Mechanisms
Keystone species are defined by their disproportionate ecological impact relative to their biomass or abundance [75]. These species exert their influence through various mechanisms:
- Ecosystem engineers physically modify habitats, creating new niches for other species
- Predatory regulators control dominant competitor populations, allowing for greater species diversity
- Mutualistic facilitators provide essential services that support multiple species within the community
The profound impact of keystone species is exemplified by cultural keystone species in social-ecological systems. Research on Japanese wetlands demonstrates how specific plant species essential for traditional thatching (Rank A species like I. aristatum var. glaucum) maintain both ecological integrity and cultural practices [78]. When these species decline, the entire social-ecological system undergoes significant transformation, potentially crossing thresholds to alternative states.
Functional Groups: Redundancy and Response Diversity
Functional groups comprise species that perform similar ecological roles, contributing to functional redundancy within ecosystems [74]. This redundancy is a critical component of resilience, as it ensures that ecological processes persist even when individual species are lost or diminished.
The resilience of ecological systems derives from the redundancy of species with similar functional traits within and across the scales present in a system [74]. Different species within a functional group may respond differently to disturbances—a property known as response diversity—further enhancing system capacity to absorb shocks while maintaining essential functions.
Table 1: Classification of Keystone Species by Functional Mechanism
| Mechanism Type | Ecological Function | Impact on Resilience | Example Organisms |
|---|---|---|---|
| Predatory Regulator | Controls dominant competitor populations | Prevents competitive exclusion, maintains diversity | Wolves, sea otters |
| Ecosystem Engineer | Modifies physical habitat | Creates new niches, alters resource availability | Beavers, prairie dogs |
| Mutualistic Facilitator | Provides essential services to multiple species | Supports reproduction/survival of dependent species | Pollinators, mycorrhizal fungi |
| Cultural Keystone Species | Supports socio-ecological systems | Maintains traditional practices, ecological knowledge | Japanese thatch grasses [78] |
Quantitative Framework for Assessing Resilience
Ecological resilience, as an emergent ecosystem phenomenon, can be decomposed into complementary attributes that embrace the complexity inherent to ecosystems [74]. A quantitative framework for assessment should simultaneously measure four key attributes:
Scale and Hierarchical Organization
Ecosystem structure is compartmentalized by distinct spatial and temporal scales [74]. Resilience assessments must account for this hierarchical organization, as the redundancy of functional traits operates within and across these scales. Statistical tools can objectively detect scales at which functional redundancy provides resilience buffers.
Adaptive Capacity
Adaptive capacity reflects a system's ability to prepare for stresses, adjust to changes, and respond to effects caused by stresses [74]. In ecological systems, this capacity is related to genetic and biological diversity, which provide ecosystems with the ability to maintain critical functions during changing environmental conditions.
Thresholds and Alternative Regimes
Thresholds represent critical levels of disturbance beyond which ecosystems undergo non-linear change or shift between alternative regimes [74]. When a system crosses a threshold, its capacity to adapt is exhausted, and it abruptly reorganizes into a new state with different structures, functions, and processes.
Table 2: Quantitative Measures for Resilience Attributes
| Resilience Attribute | Measurement Approaches | Key Indicators | Application in BLSS |
|---|---|---|---|
| Scale | Spatial and temporal pattern analysis, multivariate statistics | Cross-scale redundancy, functional trait distribution | Monitoring producer-decomposer interactions across temporal scales |
| Adaptive Capacity | Functional diversity indices, response trait assessment | Genetic diversity, response diversity, phenotypic plasticity | Measuring functional redundancy in microbial processing communities |
| Thresholds | State transition models, bifurcation analysis, early warning signals | Critical slowing down, increased autocorrelation, regime shift indicators | Identifying critical points in gas exchange balance |
| Alternative Regimes | Multivariate analysis, stable state identification | Distinct species assemblages, different process rates | Characterizing different stable configurations of producer-consumer-decomposer loops |
Experimental Protocols for Resilience Assessment
Monitoring Keystone Species Impacts
Protocol Objective: Quantify the impact of keystone species on ecosystem structure and function.
Methodology:
- Identify potential keystone species through preliminary observations or historical data
- Establish monitoring plots with and without the candidate keystone species
- Measure key ecosystem processes (productivity, decomposition, nutrient cycling) in all plots
- Implement removal experiments where ethically and ecologically feasible
- Monitor system response through before-after-control-impact (BACI) design
Parameters to Measure:
- Species richness and composition
- Trophic structure and interaction networks
- Process rates (primary production, decomposition)
- Nutrient cycling efficiency
This approach mirrors methods used in alpine meadow studies, where changes in keystone species were shown to attenuate the complexity and stability of soil microbial networks [77].
Assessing Functional Redundancy
Protocol Objective: Evaluate the redundancy within functional groups and its relationship to ecosystem resilience.
Methodology:
- Classify all species into functional groups based on trait analysis
- Quantify functional diversity using indices such as Functional Richness, Evenness, and Divergence
- Measure response diversity by exposing systems to perturbations and tracking trait-specific responses
- Analyze the relationship between functional redundancy and ecosystem stability across environmental gradients
Applications in BLSS: For BLSS research, this protocol can be adapted to assess the redundancy in microbial decomposer communities or the diversity of primary producers with complementary resource use strategies.
Research Reagent Solutions for Resilience Studies
Table 3: Essential Research Materials for Keystone Species and Resilience Research
| Reagent/Equipment | Function in Research | Application Examples |
|---|---|---|
| Stable Isotope Tracers (¹⁵N, ¹³C) | Tracking nutrient flow through food webs | Quantifying keystone species effects on nutrient cycling |
| Environmental DNA (eDNA) Sampling Kits | Comprehensive biodiversity assessment | Monitoring community composition changes without destructive sampling |
| Automated Soil Respiration Sensors | Measuring decomposition rates | Assessing functional group performance under disturbance |
| Microcosm/Mesocosm Setup | Controlled manipulation experiments | Testing threshold responses in replicated systems |
| Functional Trait Databases | Classifying species into functional groups | Estimating functional redundancy and response diversity |
| Molecular Analysis Tools (PCR, Sequencing) | Genetic analysis of microbial communities | Characterizing decomposer diversity and functional potential |
| Data Loggers for Abiotic Factors | Monitoring environmental conditions | Correlating system responses with environmental fluctuations |
Application to Bioregenerative Life Support Systems (BLSS)
In BLSS, where producers, consumers, and decomposers must maintain precise balances, the principles of keystone species and functional groups take on critical importance. These systems represent simplified ecological networks where the energy flow from producers to consumers to decomposers must be maintained within narrow parameters [75] [76].
Keystone Species in BLSS
Certain microbial or invertebrate species in decomposition subsystems may function as keystone organisms by:
- Enabling critical transformation steps in nutrient cycling pathways
- Producing essential compounds that support other organisms
- Regulating populations of potentially problematic species
The loss of such keystones could trigger cascading effects that compromise the entire system's functionality, similar to degradation patterns observed in alpine meadow soil networks [77].
Functional Groups in BLSS
BLSS design should incorporate functional redundancy across all major subsystems:
- Producers: Multiple plant species with complementary photosynthetic pathways and resource requirements
- Consumers: Diverse microbial and invertebrate communities ensuring efficient energy transfer
- Decomposers: Redundant pathways for breakdown of different organic compounds
This approach ensures that if one species fails under specific conditions, others can maintain essential processes—a fundamental principle of ecological resilience [74].
Visualization of Resilience Concepts
Diagram 1: Keystone species and functional groups mediate disturbance impacts to maintain system resilience and stability.
Diagram 2: System transition between high and low resilience states when disturbance exceeds critical thresholds.
Understanding the role of keystone species and functional groups is fundamental to designing and managing resilient BLSS. These elements determine a system's capacity to maintain functional balance among producers, consumers, and decomposers—the core components enabling long-term life support. By applying the quantitative framework and experimental protocols outlined in this whitepaper, researchers can measure, monitor, and enhance the resilience of BLSS, ensuring their stability under the fluctuating conditions inherent to closed ecological systems. Future research should focus on identifying potential keystone organisms in BLSS contexts and designing functional group redundancies that maximize adaptive capacity while minimizing resource requirements.
A Bioregenerative Life Support System (BLSS) is an advanced ecosystem engineered for space exploration, designed to sustain human life by regenerating vital resources through biological processes. The core principle of a BLSS mirrors the function of natural ecosystems on Earth, which are built upon the interconnected roles of producers, consumers, and decomposers [79]. In the closed, isolated environment of a space habitat, these components form a circular economy: producers (typically plants and algae) generate oxygen and food via photosynthesis; consumers (astronauts) consume these products and exhale carbon dioxide; and decomposers (microorganisms) break down waste into nutrients that can again be used by the producers [80]. The ultimate goal is to achieve a high degree of closure efficiency, minimizing the need for external resupply from Earth.
The stability of this man-made ecosystem is paramount for mission success. As such, quantifying its performance through specific, robust metrics is a critical area of research in bioastronautics and space bioprocess engineering [80]. This guide provides an in-depth technical framework for assessing BLSS stability and closure efficiency, providing researchers with the methodologies and tools needed to evaluate and advance these complex systems.
Core Stability Metrics for BLSS
The stability of a BLSS refers to its ability to maintain steady-state functionality for all key life support parameters despite internal fluctuations or external perturbations. The following table summarizes the primary quantitative metrics used to assess this stability.
Table 1: Key Quantitative Metrics for BLSS Stability
| Metric Category | Specific Metric | Definition & Formula | Target Value/Range |
|---|---|---|---|
| Gas Exchange Stability | Oxygen Production Rate (OPR) | Mass of O₂ produced per unit area/volume of producers per time (e.g., g O₂/m²/day) | Matches crew consumption (~0.84 kg/person/day) |
| Carbon dioxide Uptake Rate (CUR) | Mass of CO₂ consumed by producers per unit area/volume per time (e.g., g CO₂/m²/day) | Matches crew production (~1.00 kg/person/day) | |
| Biomass Stability | Producer Biomass Productivity | Net biomass (dry weight) produced per unit area per time (e.g., g/m²/day) | Sustains caloric & nutritional needs of crew |
| Crew Biomass Balance | (Caloric & Nutritional Intake) / (Crew Recommended Daily Allowance) | ≥ 1.0 | |
| Water Cycle Stability | Water Closure Efficiency (WCE) | (1 - (Fresh Water Resupplied / Total Water Used)) × 100% | > 95% |
| Water Recovery Rate | Volume of water recycled per unit time (e.g., L/day) via systems (e.g., condensate, urine processing) | Matches or exceeds total crew demand | |
| System Robustness | Resistance | Ability to maintain function amid perturbation (measured as % deviation from baseline after disturbance) | Minimal deviation (<5%) |
| Resilience | Time taken to return to baseline equilibrium after a perturbation (e.g., hours/days) | Short recovery time |
These metrics must be monitored in real-time to ensure that the flows of energy and nutrients remain in balance, supporting all trophic levels within the closed system [80].
Quantifying Closure Efficiency
Closure efficiency measures the BLSS's self-sufficiency. It evaluates how effectively the system recycles its mass and energy, thereby reducing reliance on external resources. The following diagram illustrates the interconnected cycles and the points where efficiency is measured.
Figure 1: BLSS Mass and Energy Flow Diagram. The diagram shows the critical cycles where closure efficiency is calculated, highlighting the roles of producers, consumers, and decomposers.
The overall and component-specific closure efficiencies can be quantified as shown in the table below.
Table 2: BLSS Closure Efficiency Metrics
| Efficiency Metric | Calculation Formula | Measurement Protocol |
|---|---|---|
| Overall Mass Closure | (1 - (Total Resupply Mass / Total System Mass Throughput)) × 100% | Track all mass inputs (resupply) and outputs (waste) over a defined mission period. System throughput includes all consumables (food, water, O₂). |
| Atmospheric Closure (O₂) | (O₂ Produced by Biology / O₂ Consumed by Crew) × 100% | Measure O₂ production via gas analysis in plant growth chambers; monitor crew O₂ consumption with respirometers. |
| Atmospheric Closure (CO₂) | (CO₂ Consumed by Biology / CO₂ Produced by Crew) × 100% | Monitor CO₂ levels in crew and plant chambers with NDIR sensors; calculate consumption/production rates. |
| Water Closure | (1 - (Potable Water Resupplied / Total Water Used by Crew)) × 100% | Meter all water inputs (resupply, from waste processors) and outputs (consumption, latent loss). |
| Food Closure | (Caloric & Nutritional Intake from BLSS / Total Crew Requirement) × 100% | Weigh and analyze nutritional content of all food harvested from BLSS (plants, fungi, etc.). Compare to crew dietary requirements. |
| Waste Recycling Efficiency | (Mass of Waste Processed into Usable Products / Total Waste Generated) × 100% | Quantify solid and liquid waste streams before and after processing by decomposers or physicochemical systems. |
The Beijing Lunar Palace, a leading terrestrial analog, has successfully demonstrated a high level of integration, sustaining a crew of four for a full year by effectively closing the loops on atmosphere, water, and a significant portion of nutrition [81].
Analytical Methods and Experimental Protocols
Validating BLSS performance requires rigorous, repeatable experimental protocols. The methodologies below are foundational for collecting data to compute the stability and efficiency metrics outlined in previous sections.
Gas Exchange Measurement Protocol
Objective: To quantify the net photosynthetic oxygen production and carbon dioxide consumption rates of the producer compartment (e.g., a specific crop).
- Materials: Sealed plant growth chamber, CO₂ and O₂ gas sensors (e.g., NDIR and zirconia-based, respectively), data logging system, environmental control system (for light, temperature, humidity).
- Procedure:
- Calibration: Calibrate gas sensors using standard gases before the experiment.
- Baseline: With the growth chamber sealed and no plants, monitor O₂ and CO₂ levels to establish instrument baseline drift.
- Acclimation: Introduce the test plants into the chamber and allow the system to stabilize under defined light, temperature, and CO₂ conditions for a set period (e.g., 1 hour).
- Measurement: Seal the chamber and initiate continuous monitoring of O₂ and CO₂ concentrations over a period of several hours.
- Data Analysis: Plot the concentration of each gas over time. The slope of the linear portion of the O₂ curve is the net O₂ production rate, and the slope of the CO₂ curve is the net CO₂ consumption rate. Convert these concentration rates to mass rates using the known volume of the chamber.
Biomass and Nutrient Recycling Protocol
Objective: To determine the growth efficiency of producers and the nutrient recovery efficiency from waste processed by decomposers.
- Materials: Plant growth facilities, harvested plant material (edible and inedible), solid waste, bioreactors containing microbial consortia, analytical equipment (e.g., HPLC for nutrient analysis, elemental analyzer, calorimeter).
- Procedure:
- Producer Biomass: Harvest edible and inedible plant biomass separately. Dry samples to constant weight at 60-70°C to determine dry biomass productivity (g/m²/day). Analyze nutritional content (proteins, carbohydrates, lipids, vitamins).
- Waste Processing: Combine inedible biomass with human solid waste and introduce it into a controlled bioreactor containing specific decomposer microbes (e.g., Bacillus spp., Trichoderma spp.).
- Monitoring: Monitor the decomposition process by tracking a reduction in volatile solids and the production of carbon dioxide and heat.
- Product Analysis: After processing, analyze the resulting compost or nutrient solution for levels of key plant nutrients (Nitrogen, Phosphorus, Potassium). The nutrient recycling efficiency is the mass of a specific nutrient recovered divided by the mass of that nutrient in the original waste stream.
The Scientist's Toolkit: Essential Research Reagents and Materials
Success in BLSS research relies on a suite of specialized reagents, biologicals, and equipment.
Table 3: Essential Research Reagents and Materials for BLSS Experiments
| Item | Function/Application |
|---|---|
| Gas Analyzers (NDIR for CO₂, Paramagnetic/Optical for O₂) | Continuous, real-time monitoring of atmospheric composition within crew and plant modules to calculate gas exchange rates [80]. |
| Controlled Environment Chambers | Precisely regulate light intensity, photoperiod, temperature, humidity, and CO₂ levels for plant growth experiments, ensuring reproducible conditions [80]. |
| Selected Plant Cultivars (e.g., dwarf wheat, potato, lettuce, soy) | Fast-growing, high-yield, and dwarf varieties optimized for space and controlled environments to maximize food production per unit area [80]. |
| Defined Microbial Consortia | Tailored communities of decomposer bacteria and fungi to efficiently break down solid waste and inedible biomass into plant-available nutrients [80]. |
| Nutrient Film Technique (NFT) / Aeroponics Systems | Soil-less cultivation methods that deliver water and nutrients directly to plant roots, maximizing water and nutrient use efficiency and minimizing system mass [80]. |
| Liquid Chromatography-Mass Spectrometry | Used to identify and quantify a wide range of biochemicals, from plant nutrients in recycled fertilizer to potential mycotoxins (e.g., Aflatoxin B1) that could contaminate the food supply [82]. |
| Sensors for Water Quality (pH, EC, DO) | Monitor the health of hydroponic solutions and recycled water streams to prevent system failure and ensure plant health. |
The path to long-duration human space exploration—whether to the Moon, Mars, or beyond—is inextricably linked to the development of robust Bioregenerative Life Support Systems. As reviewed in this guide, quantifying the stability and closure efficiency of these systems requires a multifaceted approach, integrating metrics from gas exchange, biomass production, water recovery, and waste processing. The analytical protocols and research tools detailed herein provide a foundation for standardized testing and comparison.
Future work must focus on integrating these individual component metrics into a holistic system-level model that can predict the emergent behavior and stability of the entire BLSS. The pioneering work in facilities like the Beijing Lunar Palace demonstrates that high levels of closure are achievable [81]. For the US and its partners to maintain competitiveness and achieve the goal of enduring, biosustainable deep space habitation, it is imperative to reinvest in and accelerate BLSS research and development, using these quantitative metrics as a guide for success.
Gaps in Knowledge and Research Challenges for Future BLSS Development
Bioregenerative Life Support Systems (BLSS) are fundamental for long-duration human space exploration, aiming to create a sustainable, closed-loop environment by mimicking Earth's ecosystems. The core functional units of a BLSS are the producers (photoautotrophs like plants and algae), consumers (astronauts), and decomposers (microorganisms like bacteria and fungi), which work in concert to regenerate air, water, and food from waste [83]. While the conceptual framework is well-established, the practical integration of these components into a stable, efficient, and resilient system presents multifaceted research challenges. This whitepaper synthesizes current knowledge and identifies critical gaps in BLSS development, with a specific focus on the interactions and synergies between producers, consumers, and decomposers. Drawing on recent advances in related terrestrial symbiosis systems, we outline a path forward for overcoming these hurdles and achieving the technological maturity required for operational space missions.
Critical Knowledge Gaps in BLSS Core Subsystems
The integration of biological components into a reliable engineering system reveals significant knowledge gaps across several domains.
Gaps in Producer-Decomposer Synergistic Integration
The algal-bacterial symbiosis, a model for the producer-decomposer loop, shows great promise for simultaneous air revitalization, water purification, and biomass production. However, its application in BLSS is hindered by an incomplete understanding of the complex interactions at play.
- Unstable Community Dynamics: A primary challenge is the inability to predict and control the long-term stability of the algal-bacterial community. Symbiotic relationships can shift between mutualism, commensalism, and parasitism based on environmental conditions [84]. For instance, certain bacteria can lyse algal cells, collapsing the system [84]. Current models cannot reliably forecast these trophic shifts in the closed, resource-limited environments of a BLSS.
- Poor Biomass Settleability and Harvesting: Free-cell suspensions of microalgae and bacteria complicate in-situ biomass harvesting for nutrient recycling or food processing [85]. The fundamental mechanisms controlling aggregate formation and the secretion of extracellular polymeric substances (EPS) are poorly understood [85].
- Inefficient Metabolic Coupling: While the exchange of oxygen and carbon dioxide is the cornerstone of this symbiosis, the exchange of other critical metabolites, such as vitamins (B12) and growth hormones (e.g., indole-3-acetic acid), is not well optimized [83]. Disrupting the exchange of vitamin B12 can reduce algal growth to just 15% of normal levels, highlighting the fragility of this partnership [83].
Gaps in System Closure and Mass Balance
Achieving a high degree of mass closure for water, carbon, and nutrients is the ultimate goal of a BLSS. Current system efficiencies remain below the thresholds needed for multi-year missions.
- Incomplete Nutrient Recycling: While nitrification and denitrification are relatively well-characterized, the recycling of other essential elements like phosphorus and potassium is less efficient. The release dynamics of these elements from inedible plant biomass and human waste back into forms usable by producers are a critical research area [86].
- Trace Contaminant Accumulation and Management: BLSS will inevitably experience the buildup of volatile organic compounds and heavy metals from biological metabolism, equipment off-gassing, and hygiene activities. The capacity of algal-bacterial systems to remediate these contaminants, and the thresholds at which they become toxic to the ecosystem, are not defined [84]. The system's performance is highly sensitive to environmental conditions, leading to uncertain treatment outcomes across different "seasons" or operational phases [84].
Gaps in Monitoring, Control, and Automation
A BLSS is a dynamic, non-linear system requiring sophisticated control strategies.
- Lack of Real-Time Biosensors: There is a pressing need for robust, miniaturized sensors for real-time monitoring of key microbial population dynamics, quorum-sensing molecules, and metabolic intermediates. Without this data, preemptive correction of system imbalances is impossible.
- Unvalidated Integrated System Models: While numerous subsystem models exist, integrated C-N-O dynamic models that accurately predict the behavior of the coupled producer-consumer-decomposer system under perturbation are lacking. The model developed for an inversed fluidized bed bioreactor, which quantified microbial contributions to oxygen and carbon cycling, is a step in this direction but requires validation for BLSS applications [87].
Table 1: Quantitative Performance Data from Terrestrial Algal-Bacterial Symbiosis Systems Relevant to BLSS
| Performance Parameter | Reported Value | Relevance to BLSS | Source |
|---|---|---|---|
| Nitrogen Removal Efficiency | >95% in a non-aerated system | Demonstrates potential for reduced energy consumption in water recycling. | [87] |
| Oxygen Production Rate | 81.82 mg/L·d by microalgae | Key data for sizing the producer component for crew oxygen demands. | [87] |
| Organic Carbon Release | 148.66 mg/L·d from biomass decay | Highlights the decomposer's role in providing carbon for denitrification. | [87] |
| Biomass Enhancement | 2.3x higher algal growth rate in symbiosis vs. mono-culture | Shows the synergistic benefit of integrated producer-decomposer systems. | [83] |
| Heavy Metal Removal (Cd) | 91.3% removal efficiency | Informs design of bioremediation loops for trace contaminant control. | [83] |
Detailed Experimental Protocols for Key BLSS Research
To address the knowledge gaps, standardized and rigorous experimental protocols are essential.
Protocol for Optimizing Algal-Bacterial Aggregation
Objective: To determine the optimal conditions and material carriers that promote stable aggregation and settleability of algal-bacterial biomass.
Materials:
- Strains: Axenic cultures of Chlorella vulgaris and a known beneficial bacterium (e.g., Gordonia sp.).
- Media: Standard nutrient medium mimicking BLSS wastewater.
- Carriers: Test materials like Magnesium-Enriched Biochar (MBC) [85], plain biochar, and inert glass beads as a control.
- Bioreactors: Set of identical photobioreactors with controlled temperature, light, and mixing.
Methodology:
- Inoculation: Co-inoculate each bioreactor with a standard ratio of algae and bacteria.
- Carrier Amendment: Add different concentrations (e.g., 0.1 g/L, 0.4 g/L, 0.8 g/L) of the test carriers to the reactors.
- Monitoring: Operate the reactors in continuous or batch mode for multiple weeks.
- Analysis:
- Aggregation Metrics: Daily measurements of mean floc diameter using image analysis and sludge volume index.
- Settleability: Measure biomass settling velocity in a graduated cylinder.
- EPS Characterization: Quantify the protein (PN) and polysaccharide (PS) content of extracellular polymeric substances and calculate the PN/PS ratio [85].
- Community Stability: Use 16S/18S rRNA sequencing at the start and end to track shifts in microbial diversity.
Protocol for Quantifying C-N-O Mass Balance
Objective: To construct a dynamic model of carbon, nitrogen, and oxygen fluxes within a closed algal-bacterial system.
Materials:
- Experimental System: A fully instrumented, gas-tight inversed fluidized bed bioreactor (IFBBR) [87].
- Sensors: In-line probes for dissolved O2, pH, and CO2.
- Analytical Equipment: TOC analyzer, ion chromatograph for NO2-, NO3-, NH4+.
Methodology:
- System Setup: Establish a stable algal-bacterial symbiosis in the IFBBR using a defined synthetic wastewater feed.
- Data Collection: Over several operational cycles, continuously log data from all in-line sensors.
- Sampling and Analysis: Take periodic samples from the influent, effluent, and biomass for analysis of TOC, TN, and nitrogen species.
- Model Construction: Use the collected data to calculate and validate the fluxes of each element. The model should account for:
- Photoautotrophic O2 production and CO2 assimilation by algae.
- Heterotrophic respiration and CO2 production by bacteria.
- Nitrification (NH4+ → NO3-) consuming O2.
- Denitrification (NO3- → N2) using organic carbon from biomass decay [87].
Diagram 1: Material flows in a BLSS. The diagram illustrates the core cycles connecting producers, consumers, and decomposers, highlighting the exchange of oxygen, carbon dioxide, and nutrients that sustain the closed-loop environment.
The Scientist's Toolkit: Key Research Reagent Solutions
Advancing BLSS research requires a suite of specialized reagents and materials to probe and manipulate the biological subsystems.
Table 2: Essential Research Reagents and Materials for BLSS Experimentation
| Reagent/Material | Function in BLSS Research | Specific Example |
|---|---|---|
| Functionalized Biochar | Serves as a multifunctional carrier to enhance microbial attachment, stabilize community structure, and buffer pH fluctuations. | Magnesium-enriched biochar (MBC) from corncob pyrolysis, shown to increase mean floc diameter and chlorophyll-a content [85]. |
| Quorum Sensing Molecules | Used to investigate and manipulate inter-kingdom signaling between producers and decomposers, potentially enhancing aggregation and system function. | N-acyl-homoserine lactones (AHLs) to stimulate algal morphogenesis; Autoinducing Peptides (AIP) to up-regulate chlorophyll biosynthesis [83]. |
| Stable Isotope Tracers | Critical for elucidating the pathways and rates of element cycling (e.g., C, N, O) within the closed system, enabling accurate mass balance models. | 13C-labeled CO2 to track carbon fixation by algae and subsequent transfer to bacteria; 15N-labeled ammonium to trace nitrogen transformation pathways. |
| Synthetic Wastewater Formulations | Provides a standardized and reproducible medium for testing BLSS subsystems, free from the variability of real waste streams in early R&D. | Defined media with specific C/N/P ratios, alkalinity sources (e.g., KHCO3), and controlled trace metal content to simulate crew waste [87]. |
Visualization of Key Signaling Pathways
Cross-kingdom communication is a crucial, yet not fully harnessed, process in BLSS. The following diagram details the known signaling pathways between bacteria and microalgae.
Diagram 2: Algal-Bacterial signal pathways. This map visualizes how bacterial signaling molecules (red-derived) directly influence key algal physiological processes (green), governing the health and output of the producer subsystem [83].
The development of a robust BLSS is a monumental interdisciplinary challenge. The gaps in our knowledge—particularly concerning the stable integration of decomposers with producers, the efficient closure of mass loops, and the advanced monitoring and control of the biological system—are significant but not insurmountable. Future research must prioritize long-duration, integrated system tests that move beyond studying components in isolation. A focused effort on harnessing microbial communication, developing smart functional materials, and validating system-level models is essential. By addressing these challenges, we can transform the BLSS from a compelling concept into a dependable technology that sustains human life on the final frontier.
Conclusion
The successful operation of a Bioregenerative Life Support System is fundamentally dependent on the precise understanding and meticulous integration of producers, consumers, and decomposers. As evidenced by historical experiments and ongoing research, these trophic levels form an interdependent web where the performance of one directly impacts the others. Key takeaways include the non-negotiable role of decomposers in nutrient recycling, the constant challenge of managing microbial communities to prevent contamination, and the critical need for real-time monitoring and dynamic balancing of the entire ecosystem. Future directions must focus on enhancing the resilience and self-regulation of these closed systems, automating control processes, and translating the principles of stable, closed-loop ecosystems to terrestrial applications in advanced biotechnology and controlled environmental agriculture, paving the way for sustainable long-duration space missions and novel biomedical isolation technologies.
References
- 1. Symbiotic integration of bioprocesses to design a self- ... [https://www.sciencedirect.com/science/article/abs/pii/S096085242100050X]
- 2. The creation of “Ecosystem Core” hypothesis to explain ... [https://bmcecol.biomedcentral.com/articles/10.1186/s12898-019-0251-y]
- 3. Revision Notes - Producers, Consumers, and Decomposers [https://www.sparkl.me/learn/ib-myp-1-3/science/producers-consumers-and-decomposers/revision-notes/5762]
- 4. A modular BLSS simulation model [https://pubmed.ncbi.nlm.nih.gov/11537271/]
- 5. Plant and microbial science and technology as ... [https://www.nature.com/articles/s41526-023-00317-9]
- 6. The role of blue light in plant stress responses - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12122757/]
- 7. Effects of red and blue light on leaf anatomy, CO2 assimilation ... [https://bmcplantbiol.biomedcentral.com/articles/10.1186/s12870-020-02523-z]
- 8. Blue Light Improves Photosynthetic Performance and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8392054/]
- 9. Chlorella vulgaris - Intensity of blue LED light [https://www.sciencedirect.com/science/article/abs/pii/S096085241301420X]
- 10. Microbial aerotrophy enables continuous primary ... [https://www.nature.com/articles/s41467-025-65209-w]
- 11. Trade-offs between microbiome diversity and productivity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5270574/]
- 12. Assessment of aquatic food web and trophic niche as a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10252823/]
- 13. ESS Subtopic 2.2: Energy and Biomass - AMAZING WORLD OF SCIENCE WITH MR. GREEN [https://www.mrgscience.com/ess-subtopic-22-energy-and-biomass.html]
- 14. The Role of Decomposers: Nature's Recyclers [https://ecoservantsproject.org/the-role-of-decomposers-natures-recyclers/]
- 15. Decomposers [https://www.scienceworld.ca/resource/decomposers/]
- 16. Decomposers - National Geographic Education [https://education.nationalgeographic.org/resource/decomposers/]
- 17. Decomposers [https://www.dnr.sc.gov/marine/mrri/acechar/biological/decomposers.html]
- 18. Biotechnology in Agro-Industry: Valorization of Agricultural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12388710/]
- 19. Energy Flow in Ecosystems → Term [https://energy.sustainability-directory.com/term/energy-flow-in-ecosystems/]
- 20. Energy Flow and the 10 Percent Rule [https://education.nationalgeographic.org/resource/energy-flow-and-10-percent-rule/]
- 21. Revision Notes - Energy efficiency and ecological pyramids [https://www.sparkl.me/learn/ib/biology-hl/energy-efficiency-and-ecological-pyramids/revision-notes/1289]
- 22. The first biological experiment on lunar surface for ... [https://www.sciencedirect.com/science/article/abs/pii/S0094576523005301]
- 23. MIT chemists boost the efficiency of a key enzyme in ... [https://news.mit.edu/2025/mit-chemists-boost-efficiency-key-enzyme-photosynthesis-0707]
- 24. Blog - Tiny but mighty: Algae hold the secret to turbo-charging ... [https://biology.ed.ac.uk/plant-sciences/outreach/blogs/algae-hold-secret-to-turbo-charging-photosynthesis]
- 25. Synthetic biology to supercharge photosynthesis in crops [https://www.sydney.edu.au/news-opinion/news/2025/11/04/synthetic-biology-encapsulins-photosynthesis-szyszka.html]
- 26. New Insights Into Nature's Oxygen-Making Machinery [https://newscenter.lbl.gov/2025/07/08/how-plants-manage-light-new-insights-into-natures-oxygen-making-machinery/]
- 27. Upcycling food waste for microalgae cultivation toward lipid ... [https://biotechnologyforbiofuels.biomedcentral.com/articles/10.1186/s13068-025-02679-6]
- 28. Quantification of oxygen production and respiration rates in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6999335/]
- 29. Insects in bioregenerative life support systems - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12457449/]
- 30. Life-support system | Environmental Benefits & Uses [https://www.britannica.com/technology/life-support-system-environmental]
- 31. Metabolic Phenotyping of Marine Heterotrophs on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9426600/]
- 32. Macrosystems ecology: A new engine and frontier in ... [https://www.sciencedirect.com/science/article/pii/S2666683925000732]
- 33. Net heterotrophy and low carbon dioxide emissions from ... [https://www.sciencedirect.com/science/article/abs/pii/S0043135419312345]
- 34. The global distribution and climate resilience of marine ... [https://www.nature.com/articles/s41467-024-50635-z]
- 35. microbial pathways for sustainable food production [https://pubs.rsc.org/en/content/articlehtml/2025/fb/d5fb00287g]
- 36. Current technologies for nutrient recovery in aquaponic ... [https://www.frontiersin.org/journals/sustainable-food-systems/articles/10.3389/fsufs.2025.1681638/full]
- 37. Colorimetric Systems for the Detection of Bacterial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9313054/]
- 38. Microbial applications for sustainable space exploration ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10284894/]
- 39. Development of Nitrogen Recycling Strategies for ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2021.700810/full]
- 40. Dynamics of long‐term continuous culture of Limnospira ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8913870/]
- 41. Producers, Consumers, and Decomposers [https://curriculum.eleducation.org/curriculum/ls/grade-5/module-2/unit-1/lesson-6]
- 42. A comprehensive review of microbial contamination in the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10701447/]
- 43. Rapid methods for the detection of foodborne bacterial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4290631/]
- 44. Advancing microbial risk assessment: perspectives from ... [https://www.nature.com/articles/s41538-025-00527-3]
- 45. Connect the dots: sketching out microbiome interactions ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10696587/]
- 46. Taxonomic Profiling and Visualization of Metagenomic Data [https://training.galaxyproject.org/training-material/topics/microbiome/tutorials/taxonomic-profiling/tutorial.html]
- 47. Quantitative Data and Models for Bacterial Cross ... [https://www.sciencedirect.com/science/article/abs/pii/S2352352225000167]
- 48. F. Environmental Sampling | Infection Control [https://www.cdc.gov/infection-control/hcp/environmental-control/environmental-sampling.html]
- 49. Biosphere 2 [https://en.wikipedia.org/wiki/Biosphere_2]
- 50. (PDF) Oxygen loss in biosphere 2 [https://www.academia.edu/54407169/Oxygen_loss_in_biosphere_2]
- 51. Biosphere 2 engineering design [https://www.sciencedirect.com/science/article/abs/pii/S0925857498000901]
- 52. How the Biosphere 2 experiment changed our ... [https://www.bbc.com/future/article/20250703-how-the-biosphere-2-experiment-changed-our-understanding-of-the-earth]
- 53. Under the Glass Systems [https://biosphere2.org/research/under-glass-systems]
- 54. Reliability and lifetime estimation of bioregenerative life ... [https://www.sciencedirect.com/science/article/abs/pii/S0094576522006294]
- 55. Microbial community structure and succession of airborne ... [https://www.sciencedirect.com/science/article/abs/pii/S0925857415303268]
- 56. Microbiomes of air dust collected during the ground-based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8793263/]
- 57. Comprehensive Soil Health Indicators for Farmers 2025 [https://farmonaut.com/precision-farming/comprehensive-soil-health-indicators-for-farmers-2025]
- 58. Decoupling Between Functional Diversity and Stability of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12449671/]
- 59. An Overview of Common Soil Health Indicators [https://cropsandsoils.extension.wisc.edu/articles/an-overview-of-common-soil-health-indicators/]
- 60. Decomposer diversity increases biomass production and ... [https://www.nature.com/articles/s41598-018-36294-3]
- 61. Valorization of Agri-Food Waste to Promote Sustainable ... [https://www.mdpi.com/2073-4395/15/10/2254]
- 62. Composting for Farms: Turning Waste into Black Gold [https://www.farmraise.com/blog/composting-for-farms-turning-waste-into-black-gold]
- 63. Valorizing agricultural wastes through composting ... [https://link.springer.com/article/10.1007/s43621-025-01769-7]
- 64. Assessing biological soil health through decomposition of ... [https://www.sciencedirect.com/science/article/abs/pii/S0929139321002213]
- 65. SOIL - Benchmarking soil multifunctionality - Copernicus.org [https://soil.copernicus.org/articles/11/609/2025/]
- 66. Microbial applications for sustainable space exploration ... [https://www.nature.com/articles/s41526-023-00285-0]
- 67. The impact of the space environment on microbial growth and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10995381/]
- 68. Microbial Monitoring of Crewed Habitats in Space— ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4159036/]
- 69. Towards Quantitative Microbiome Community Profiling Using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6384117/]
- 70. Use of Photobioreactors in Regenerative Life Support ... - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8281973/]
- 71. A 21st Century 'Operating Manual for Spaceship Earth' [https://www.vice.com/en/article/the-decades-long-quest-to-recreate-earth-in-miniature/]
- 72. Inside the evolution of Biosphere 2, from '90s punchline to ... [https://www.kpbs.org/news/science-technology/2025/07/07/inside-the-evolution-of-biosphere-2-from-90s-punchline-to-scientific-playground]
- 73. Microbiome dynamics during the HI-SEAS IV mission, and ... [https://microbiomejournal.biomedcentral.com/articles/10.1186/s40168-020-00959-x]
- 74. A quantitative framework for assessing ecological resilience [https://pmc.ncbi.nlm.nih.gov/articles/PMC5759782/]
- 75. Food Web Dynamics → Term [https://energy.sustainability-directory.com/term/food-web-dynamics/]
- 76. Food chains and food webs | Research Starters [https://www.ebsco.com/research-starters/power-and-energy/food-chains-and-food-webs]
- 77. Changes in keystone species attenuate the complexity and ... [https://www.sciencedirect.com/science/article/pii/S1470160X25012245]
- 78. Vegetation change threatens the sustainability of cultural ... [https://www.nature.com/articles/s41598-025-24015-6]
- 79. Food Web - National Geographic Education [https://education.nationalgeographic.org/resource/food-web/]
- 80. Space biological and human survival: Investigations into ... [https://www.sciencedirect.com/science/article/abs/pii/S2214552424000968]
- 81. Critical investments in bioregenerative life support systems for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12357894/]
- 82. Development of a UHPLC-MS/MS Method for Quantitative ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12474191/]
- 83. Harnessing an Algae–Bacteria Symbiosis System [https://www.mdpi.com/2071-1050/17/15/7104]
- 84. Algal-bacterial symbiotic system for treatment of heavy ... [https://www.nature.com/articles/s41545-025-00475-1]
- 85. unlocking productivity and stability in the algal-bacterial ... [https://www.sciencedirect.com/science/article/abs/pii/S1385894725101216]
- 86. Global forest gaps reduce litterfall but increase litter carbon ... [https://www.nature.com/articles/s43247-024-01453-0]
- 87. Microalgae-bacteria symbiosis enhanced nitrogen removal ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2025.1591974/full]