Nitrogen Cycling in Regenerative Life Support Systems: From Foundational Principles to Biomedical Applications
This article comprehensively examines nitrogen cycling within bioregenerative life support systems (BLSS), a critical technology for sustaining human life during long-duration space missions.
Nitrogen Cycling in Regenerative Life Support Systems: From Foundational Principles to Biomedical Applications
Abstract
This article comprehensively examines nitrogen cycling within bioregenerative life support systems (BLSS), a critical technology for sustaining human life during long-duration space missions. We explore the foundational principles driving the shift from physicochemical to biological systems for in-situ resource recovery, detailing specific methodologies for nitrogen fixation and recycling from waste streams like urine. The analysis covers significant challenges including system stability under space conditions and nutrient use efficiency optimization. Through validation and comparative assessment of different system architectures, we highlight implications for creating closed-loop ecosystems and potential terrestrial applications in sustainable agriculture and resource management.
The Imperative for Closed-Loop Nitrogen Cycling in Life Support
Limitations of Current Physicochemical Life Support Systems (e.g., ISS ECLSS)
Physicochemical Life Support Systems (PCLSS) represent the current technological standard for maintaining human life in the closed environments of spacecraft, such as the International Space Station (ISS). These systems rely on engineered processes to manage atmosphere, purify water, and handle waste [1]. The Environmental Control and Life Support System (ECLSS) on the ISS provides a critical Earth-like environment, regulating atmospheric pressure, oxygen levels, and temperature, while removing carbon dioxide and generating potable water [2].
However, within the broader context of developing fully regenerative life support systems, a fundamental limitation of current PCLSS becomes apparent: its open-loop or semi-closed architecture, particularly concerning nitrogen cycling. In contrast to Bioregenerative Life Support Systems (BLSS) which aim to use biological processes to create a self-sustaining, closed-loop system for air, water, and waste recycling, PCLSS technologies are limited in their ability to fully recover and recycle vital nutrients [1] [3]. This whitepaper details the specific technical limitations of ISS-class PCLSS, with a specific focus on the challenges of nitrogen management, and explores the experimental approaches driving the development of more regenerative solutions.
Core Limitations of PCLSS
The limitations of PCLSS can be categorized into systemic inefficiencies and specific technical challenges, with the management of nitrogen being a central issue.
Systemic and Nutrient Cycling Limitations
The reliance on consumables and limited closure of material loops present significant hurdles for long-duration missions.
- Incomplete Nutrient Recycling and Resupply Dependency: The ISS ECLSS demonstrates high water recovery (up to 85%) and produces oxygen via electrolysis [2]. However, it fails to integrate nitrogen recovery from liquid and solid waste streams into a closed loop. Human urine is the largest source of recoverable nitrogen, contributing 7–16 grams of nitrogen per crew member per day and accounting for 85% of the total potentially recoverable nitrogen in a BLSS [3]. In the current Urine Processor Assembly (UPA), water is recovered, but the concentrated brine containing urea and other nitrogenous compounds is treated as waste [3] [2]. This necessitates the regular resupply of nitrogen buffers (like nitrogen gas) and food from Earth, making the system inherently dependent on terrestrial support [3].
- High Consumable Mass and Cost: The inability to fully recycle nutrients and the reliance on expendable components (e.g., filters, chemical sorbents) result in a significant mass penalty. For a hypothetical three-year mission to Mars with a crew of four, a payload of over 25,000 kg would be required for food and water alone, with launch costs exceeding $10,000 per kg [3]. This makes long-duration missions logistically challenging and cost-prohibitive.
Technical and Operational Challenges
- Limited Food Production Capability: Current PCLSS lack any integrated capacity for in-situ food production. All food is supplied as pre-packaged meals from Earth, which directly contributes to the high resupply mass and does not contribute to closing the nutritional loop [1].
- Vulnerability to System Failures: PCLSS depend on complex mechanical and chemical systems that are prone to specific failure modes. For instance, the ISS's urine processor has experienced issues with scaling and precipitation of minerals, leading to reduced water recovery efficiency and requiring maintenance [2]. The Russian Elektron oxygen generation system has also been plagued by periodic failures, forcing crews to rely on backup oxygen canisters [2].
- Production of Unused Waste Streams: Certain processes within PCLSS generate waste products that are not utilized within the system. The Sabatier reactor, which combines hydrogen and carbon dioxide to produce water and methane, results in methane being vented overboard [2]. This represents a loss of hydrogen, which could otherwise be used to recover more oxygen, and carbon that could be part of a nutrient cycle.
Table 1: Quantitative Comparison of Life Support System Approaches
| Parameter | Current PCLSS (e.g., ISS) | Idealized BLSS/CELSS |
|---|---|---|
| Water Recovery Efficiency | Up to 85% (ISS UPA) [2] | >98% (Theoretical goal) |
| Nitrogen Loop Closure | Very Low (Vented or stored as waste) [3] | High (Targeting near-complete recycling) [3] |
| In-situ Food Production | None [1] | Core function [1] [3] |
| Oxygen Source | Electrolysis of supplied/recovered water [2] | Photosynthesis (plants, algae) [1] |
| Resupply Dependency | High (Food, filters, nitrogen buffer) [3] | Low (Limited to spare parts, seeds) |
| Waste Product Utilization | Low (Methane vented, brines stored) [2] | High (Waste used as fertilizer/feedstock) [1] [3] |
Experimental Approaches for Investigating Nitrogen Cycling
Research into closing the nitrogen loop focuses on biological processes to convert waste nitrogen into forms usable by plants, which can then serve as food.
Protocol: Nitrification-Based Nitrogen Recovery from Urine
This methodology, inspired by systems like the European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative), outlines the process for recovering nitrogen from urine via microbial nitrification [3].
- Urine Collection and Chemical Stabilization: Collect human urine and chemically stabilize it to prevent urea hydrolysis and the volatilization of ammonia. The ISS system uses a phosphoric acid and chromium VI solution for this purpose, though research seeks safer alternatives for integrated biological processing [3].
- Dilution and Introduction to Bioreactor: Dilute the stabilized urine stream with other gray water sources (e.g., condensate) to reduce toxicity and provide a suitable medium for microorganisms.
- Microbial Nitrification: Inoculate the diluted urine in a controlled bioreactor with a defined community of nitrifying bacteria, such as Nitrosomonas and Nitrobacter species.
- Process Conditions: Maintain aerobic conditions with continuous aeration. Control temperature (e.g., 28-30°C), pH (e.g., 7.5-8.0), and hydraulic retention time to optimize bacterial activity.
- Process Biochemistry:
- Ammonia Oxidation: Nitrosomonas spp. oxidize ammonia (NH₃) to nitrite (NO₂⁻).
- Nitrite Oxidation: Nitrobacter spp. oxidize nitrite (NO₂⁻) to nitrate (NO₃⁻).
- Process Monitoring: Regularly monitor the concentrations of total ammonia nitrogen, nitrite, and nitrate in the effluent to confirm complete conversion of ammonia to nitrate.
- Product Integration: The resulting nitrate-rich solution is used as a liquid fertilizer for the hydroponic cultivation of higher plants, thereby closing the nitrogen loop from waste to food.
Protocol: Evaluating Aquatic Bryophytes as Biofilters
Recent investigations explore non-traditional biological components, such as aquatic mosses, for their multifunctional role in BLSS, including water purification and gas exchange [4].
- Species Selection and Cultivation: Select fast-growing aquatic bryophyte species known for phytoremediation, such as Taxiphyllum barbieri (Java moss), Leptodictyum riparium, and Vesicularia montagnei (Christmas moss). Maintain semi-axenic cultures in controlled environments under specific light and temperature conditions (e.g., 24°C at 600 μmol photons m⁻²s⁻¹ and 22°C at 200 μmol photons m⁻²s⁻¹) [4].
- Exposure to Simulated Waste Stream: Expose the moss cultures to a synthetic wastewater solution containing known concentrations of nitrogen compounds (e.g., ammonium, nitrate) and heavy metals (e.g., Zinc).
- Performance Metric Analysis:
- Biofiltration Efficiency: Periodically sample the water and measure the removal rates of total ammonia nitrogen and heavy metals using standardized analytical methods like spectrophotometry.
- Physiological Health: Assess the health and functionality of the mosses by measuring:
- Photosynthetic Efficiency: Using chlorophyll fluorescence (e.g., Fv/Fm ratio).
- Pigment Concentration: Extract and measure chlorophyll a, b, and carotenoids.
- Antioxidant Activity: Analyze antioxidant enzyme levels as a marker of stress response.
- Data Correlation: Correlate the biofiltration performance data with the physiological data to identify the most robust and effective species for integration into a BLSS [4].
The following diagram illustrates the logical workflow and key analysis points for the bryophyte biofiltration experiment.
The Scientist's Toolkit: Key Research Reagents and Materials
Research into regenerative nitrogen cycling relies on specific biological and chemical reagents.
Table 2: Essential Research Materials for Nitrogen Cycling Experiments
| Reagent / Material | Function in Research | Example Application |
|---|---|---|
| Nitrifying Bacteria (Nitrosomonas, Nitrobacter) | Convert toxic ammonia from urine into plant-available nitrate. | Core biocatalyst in MELiSSA-inspired urine recycling [3]. |
| Aquatic Bryophytes (e.g., Taxiphyllum barbieri) | Act as multifunctional biofilters; remove nitrogen compounds and heavy metals from water. | Evaluated for water purification and oxygen production in BLSS [4]. |
| Stabilized Urine Simulant | A chemically defined solution mimicking human urine, used for safe and reproducible testing. | Serves as the primary nitrogenous waste feedstock for process development [3]. |
| Hydroponic Plant Cultures (e.g., lettuce, wheat) | Primary producers that consume recovered nutrients and produce food, oxygen, and water. | The end-user of recovered nitrate fertilizer; key to closing the food loop [1] [3]. |
| Spectrophotometer | Analytical instrument to quantify concentrations of nitrogen species (ammonia, nitrite, nitrate). | Essential for monitoring the efficiency of nitrification processes [4]. |
| Chlorophyll Fluorometer | Measures photosynthetic efficiency (e.g., Fv/Fm) in plants/algae as a health indicator. | Used to assess the physiological impact of space-like stressors on biological components [4]. |
The current Physicochemical Life Support Systems, as exemplified by the ISS ECLSS, provide a proven but fundamentally limited approach to supporting human life in space. Their primary shortcomings lie in the incomplete recycling of nutrients, particularly nitrogen, and a consequent high dependency on resupply from Earth. The analysis of experimental protocols and the defined research toolkit reveals a clear pathway forward. The integration of biological components—from nitrifying bacteria to higher plants and novel biofilters like aquatic mosses—is essential to evolve from open-loop PCLSS to closed-loop Bioregenerative Life Support Systems (BLSS). Mastering nitrogen cycling is not merely an incremental improvement but a fundamental requirement for achieving the long-term goal of sustainable human presence beyond Earth orbit.
Defining Bioregenerative Life Support Systems (BLSS) and Their Core Principles
Bioregenerative Life Support Systems (BLSS) are artificial ecosystems engineered to sustain human life in space by regenerating essential resources through biological processes. These systems are designed to produce food, water, and oxygen, while recycling waste, thereby creating a materially closed loop that minimizes reliance on external resupply from Earth [5] [6]. The core principle of a BLSS is the integration of biological components—typically plants, microorganisms, and sometimes animals—with physicochemical processes to create a controlled, self-sustaining environment that can support human crews during long-duration space missions, such as those to the Moon or Mars [3] [7].
The development of BLSS is driven by the economic and logistical constraints of deep space exploration. It is estimated that a single crew member requires approximately 1.83 kg of food and 2.50 kg of water per day [3]. For a 3-year mission to Mars with a crew of four, this translates to a payload of over 25,000 kg for food and water alone, with launch costs currently exceeding $10,000 per kilogram [3]. BLSS addresses this challenge by enabling in situ resource regeneration, dramatically reducing the initial mass required for missions and enhancing their feasibility [3] [8].
The Shift from Physicochemical to Bioregenerative Systems
Current life support systems aboard the International Space Station (ISS), known as Environmental Control and Life Support Systems (ECLSS), rely predominantly on physicochemical processes [3]. The ECLSS includes a Water Recovery System (WRS) and an Oxygen Generation System (OGS) [3]. The WRS processes urine and cabin condensate, recovering about 85% of the water, while the OGS produces oxygen via water electrolysis [3]. A critical limitation of the ECLSS is its inability to produce food and its requirement for a steady supply of consumables, resulting in the need for frequent and costly resupply missions [3].
BLSS represents a paradigm shift by incorporating biological elements to achieve a higher degree of closure. These systems aim not only to recycle water and oxygen but also to produce food by recovering minerals and carbon from waste streams [3] [9]. This bioregenerative approach is essential for missions where resupply is impractical, making them a cornerstone for sustainable, long-term human presence beyond Low Earth Orbit [5] [6].
Core Principles of BLSS Design
The design and operation of BLSS are governed by several interconnected core principles, which ensure the system's stability, efficiency, and ability to support human life.
Ecological Closure and Material Cycling
The foundational principle of a BLSS is the creation of a materially closed loop that mimics Earth's natural ecosystems [6]. This involves the precise cycling of key elements—Carbon (C), Hydrogen (H), Oxygen (O), and Nitrogen (N)—between the system's compartments [8]. The system's "producers" (plants, microalgae) use light energy to fix CO2 and produce biomass and oxygen. The "consumers" (astronauts) consume the biomass and oxygen, producing CO2 and waste. The "decomposers" (microorganisms) then break down the waste into minerals and CO2, which are reused by the producers, thus closing the loop [5] [6]. Achieving a high degree of material closure, as demonstrated by China's Lunar Palace 365 experiment with a material closure of >98%, is a key indicator of system maturity [6] [7].
The Critical Role of Nitrogen Cycling
Nitrogen is a critical nutrient for human and plant protein synthesis, making its efficient cycling a cornerstone of BLSS operation [9]. With 85% of recoverable nitrogen in a BLSS originating from human urine, the system's ability to convert this nitrogen into forms usable by plants (such as nitrate) is paramount [3] [9]. Key microbial processes for nitrogen recovery include:
- Ureolysis: The conversion of urea in urine into ammonia.
- Nitrification: The aerobic oxidation of ammonia to nitrite and then to nitrate by ammonia-oxidizing bacteria/archaea (AOB/AOA) and nitrite-oxidizing bacteria (NOB).
- Denitrification and Anammox: Processes that can convert nitrogenous compounds back to nitrogen gas, though nitrate is typically the desired product for plant fertilization in a BLSS [9] [10].
Integration and Compartmentalization
BLSS are not monolithic but are composed of discrete, interconnected compartments, each with a specialized metabolic function [3] [8]. This compartmentalization allows for the optimization of environmental conditions (e.g., pH, temperature, O2 levels) for the specific organisms in each compartment, leading to higher overall system efficiency and control. A prominent example is the European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) loop, which is structured as a five-compartment engineered ecosystem [3] [8].
System Resilience and Control
Given the critical nature of life support, BLSS must be highly robust and resilient. Biological systems offer advantages like self-repair and adaptability, but they also introduce complexity and dynamic behavior [8]. Therefore, advanced control and monitoring systems are essential. Mathematical modeling, particularly stoichiometric models that track the flow of elements, is used to predict system behavior, plan for contingencies, maintain gas and nutrient balances, and ensure long-term reliability [8]. The "three key conditions of BLSS gas balance" have been proposed as a fundamental guideline for achieving system stability [6] [7].
Architectural Frameworks and Global Initiatives
Several major BLSS programs have developed distinct architectural frameworks, each contributing to the global knowledge base.
Table 1: Major BLSS Initiatives and Their Characteristics
| Initiative / Program | Lead Agency/Country | Key Features & Focus | Status and Achievements |
|---|---|---|---|
| MELiSSA [3] [8] | European Space Agency (ESA) | - Five-compartment, lake-inspired ecosystem engineering approach- Intensive modeling and control strategy- Focus on nitrogen recovery via nitrification | - Over 30 years of development- Operational pilot plant (MPP) in Spain- Compartment III dedicated to nitrification |
| Lunar Palace [6] [7] | China (CNSA) | - Integrated BLSS with plants, microorganisms, and humans- High material closure goal | - "Lunar Palace 365" experiment: 1-year closed human survival with >98% material closure |
| CELSS / BIO-Plex [11] | USA (NASA) | - Early pioneer in controlled environment agriculture- Focus on higher plant cultivation and system integration | - Program discontinued and facility demolished in 2005- foundational research informs current efforts |
| BIOS / CEEF [6] | USSR/Russia & Japan | - Early ground-based demonstrators (BIOS-1,2,3)- Testing of closed-loop ecosystems with humans | - Provided foundational data on long-term operation and crew psychology |
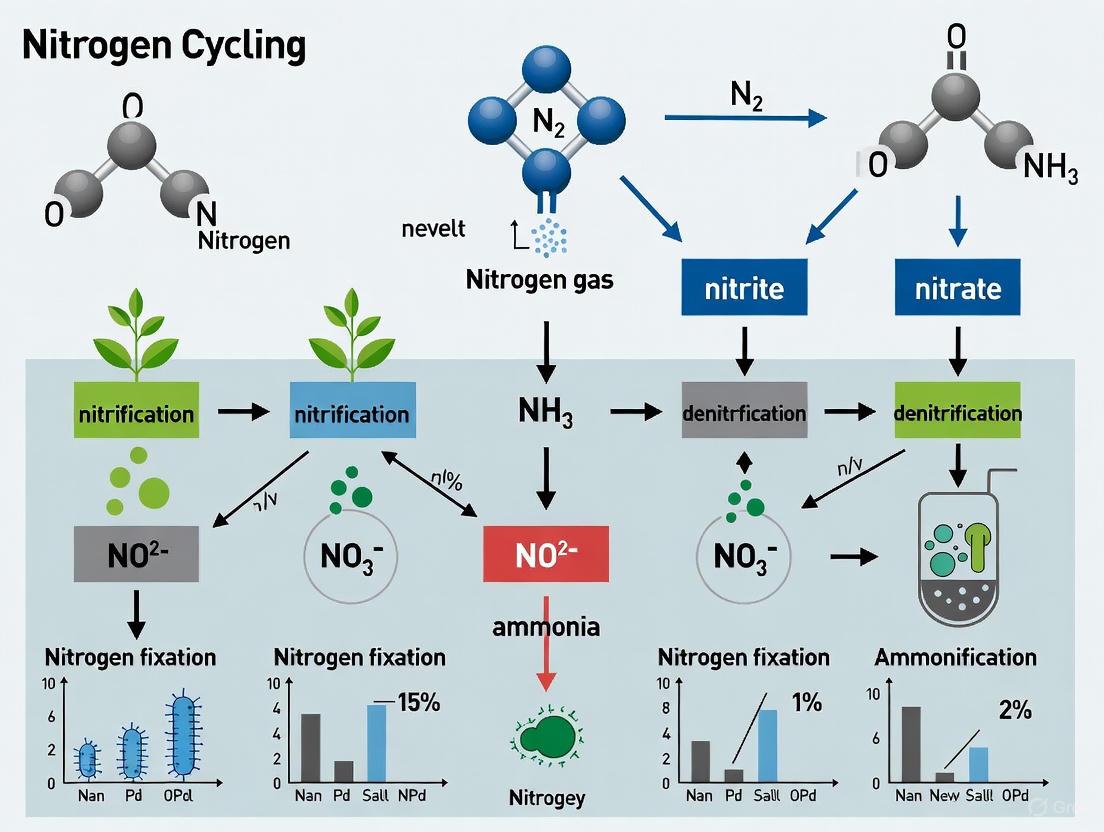
The Nitrogen Cycle: The Core Metabolic Engine
The nitrogen cycle is the metabolic engine that drives nutrient recovery in a BLSS, transforming waste nitrogen into edible biomass.
Nitrogen Flow and Key Processes
The following diagram illustrates the principal pathways of nitrogen transformation in a BLSS, connecting human waste to food production.
Diagram 1: Nitrogen cycling pathways in BLSS.
Methodologies for Studying Nitrogen Cycle Microorganisms in Space
A critical step towards implementing BLSS is verifying that key nitrogen cycle microorganisms remain viable under space conditions. The following workflow outlines a representative experiment that tested this premise.
Diagram 2: Experimental protocol for testing microbial viability after spaceflight.
This experiment demonstrated that a wide range of nitrogen cycle microorganisms, including ureolytic bacteria, ammonia-oxidizing archaea and bacteria, nitrite-oxidizing bacteria, and anammox bacteria, could be reactivated after 44 days in Low Earth Orbit despite exposure to microgravity and elevated radiation (687 ± 170 µGy d⁻¹) [10]. Surprisingly, space exposure generally yielded similar or even higher nitrogen conversion rates compared to terrestrial preservation at a similar temperature, while terrestrial storage at 4°C resulted in the highest rates [10]. This finding is a pivotal enabler for resource recovery in human space exploration.
Reagents and Materials for Nitrogen Cycle Research
Table 2: Key Research Reagents for BLSS Nitrogen Cycle Experiments
| Reagent / Material | Function in Experimental Protocol |
|---|---|
| Defined Microbial Communities (e.g., Nitrosomonas europaea, Nitrobacter winogradskyi) [10] | Axenic or defined co-cultures used to study specific nitrogen transformation pathways (e.g., nitritation, nitratation) under controlled conditions. |
| Reactor Communities (e.g., Nitr-ur, OLAND biofilm) [10] | Complex microbial consortia from operational bioreactors; used to test system resilience and community-level responses to space conditions. |
| LB Agar Plates [10] | Used for heterotrophic plate counts to check for contamination in axenic cultures, crucial for planetary protection. |
| Specific Culture Media [10] | Liquid growth media tailored to the nutritional needs of autotrophic nitrogen cycle microorganisms (e.g., containing NH₄⁺ or NO₂⁻ as energy sources). |
| Illumina Sequencing [10] | A high-throughput DNA sequencing technology used for phylogenetic characterization of reactor communities, assessing diversity and composition. |
Bioregenerative Life Support Systems represent a critical enabling technology for the long-term human exploration of space. Defined by their core principles of ecological closure, integration, and resilience, these systems leverage biological processes, with the nitrogen cycle at their core, to regenerate vital resources from waste. Global initiatives have demonstrated the feasibility of BLSS in ground-based experiments, with research now focused on adapting these systems to the unique challenges of the space environment. The successful reactivation of nitrogen cycle microorganisms after spaceflight confirms the biological viability of this approach and paves the way for the future deployment of BLSS, turning deep space missions from distant dreams into achievable realities.
Nitrogen is a fundamental element for all life forms, serving as a critical building block for amino acids, nucleic acids, and chlorophyll. In the context of regenerative life support systems (RLSS) for long-duration space missions, the efficient management of nitrogen cycles becomes paramount for sustaining human life independent of Earthly resupply. These systems aim to create closed-loop ecosystems where nitrogen is recycled from human metabolic waste back into forms usable for plant nutrition and food production. The interconnection between human nitrogen requirements and plant nitrogen uptake mechanisms represents a core biological process that must be optimized for RLSS to function effectively. This technical guide examines the central role of nitrogen across biological systems, with specific application to the development of robust life support systems for space exploration.
Nitrogen in Human Metabolism
Human Nitrogen Requirements and Protein Metabolism
Human nitrogen requirements are primarily driven by protein metabolism, as nitrogen is an essential component of amino acids, nucleotides, and other vital compounds. Nitrogen balance studies remain the gold standard for determining protein requirements in healthy adults. A recent comprehensive meta-analysis of nitrogen balance studies, incorporating data from 395 individuals, established that the overall mean nitrogen requirement is 104.2 mg N/kg/day [12]. This finding aligns with earlier large-scale analyses that recommended an Estimated Average Requirement (EAR) and Recommended Dietary Allowance (RDA) of 105 and 132 mg N/kg/day (equivalent to 0.65 and 0.83 g good-quality protein/kg/day), respectively [13]. These requirements show no significant differences based on sex, age group, climate, or protein source, though substantial heterogeneity exists between individuals [12].
Table 1: Human Nitrogen Requirement Estimates from Nitrogen Balance Studies
| Parameter | Estimate 1 (2025 Meta-Analysis) | Estimate 2 (Earlier Meta-Analysis) | Units |
|---|---|---|---|
| Mean Nitrogen Requirement | 104.2 | 105 | mg N/kg/day |
| Estimated Average Requirement (EAR) | - | 105 | mg N/kg/day |
| Recommended Dietary Allowance (RDA) | - | 132 | mg N/kg/day |
| Protein EAR | ~0.65 | 0.65 | g protein/kg/day |
| Protein RDA | ~0.83 | 0.83 | g protein/kg/day |
Experimental Protocols for Determining Nitrogen Requirements
The determination of human nitrogen requirements follows rigorous methodological protocols centered around nitrogen balance measurements:
Study Design: Participants are housed in controlled metabolic research units for the study duration. Studies must include at least three different protein intake levels per individual to establish a dose-response relationship [12].
Dietary Control: Subjects receive precisely weighed diets with known nitrogen content. Protein sources may vary (animal, plant, or mixed) but must be carefully characterized.
Sample Collection: All intake and excretion are meticulously collected and measured. This includes complete 24-hour urine collections, fecal samples, and accounting for integumental and miscellaneous losses.
Analysis Period: Each dietary nitrogen level is typically maintained for 1-2 weeks to allow for adaptation, with balance measurements during the final 3-7 days.
Analytical Methods: Nitrogen content in diet, urine, and feces is determined using the Kjeldahl method or more modern combustion analysis techniques.
Calculation: Nitrogen balance is calculated as: N balance = N intake - (Urinary N + Fecal N + Miscellaneous N losses) [12] [13].
Nitrogen in Plant Nutrition
Plant Nitrogen Uptake Mechanisms
Plants primarily absorb nitrogen from the soil solution as two inorganic forms: ammonium (NH₄⁺) and nitrate (NO₃⁻), which collectively account for over 70% of the total anions and cations taken up by plants [14]. The uptake process initiates at the root surface and involves specific plasma membrane transporters that mediate nitrogen translocation across the epidermis and endodermis [15]. These transport systems are tightly regulated by the plant through multiple mechanisms, including transcriptional and post-transcriptional control of nitrogen transporters in root cells [15]. Once inside the root, nitrogen compounds follow diverse metabolic pathways including vacuolar storage, assimilation into organic molecules, or loading into the xylem for translocation to aerial tissues [15].
Table 2: Optimal Ammonium-Nitrate Ratios for Various Plant Species
| Species | Cultivation Method | Nitrogen Supply Concentration | Optimal NH₄⁺:NO₃⁻ Ratio | Reference |
|---|---|---|---|---|
| Maize | Hydroponics | 1 mmol L⁻¹ | 1:3 | [14] |
| Wheat | Hydroponics | 7.14 mmol L⁻¹ | 1:3-1:1 | [14] |
| Rice | Hydroponics | 2.86 mmol L⁻¹ | 1:1 | [14] |
| Tomato | Hydroponics | 5 mmol L⁻¹ | 1:3 | [14] |
| Soybean | Hydroponics | 2.4 mmol L⁻¹ | 1:3 | [14] |
Benefits of Mixed Nitrogen Nutrition
Research demonstrates that a balanced mixture of nitrate and ammonium nitrogen significantly enhances plant growth and nitrogen use efficiency compared to sole application of either form [14]. The synergistic benefits include:
Enhanced Root Development: Mixed nitrogen sources improve root architecture through increased root branching and surface area, thereby expanding the soil exploration capacity.
Improved Nitrogen Uptake Efficiency: Co-application activates complementary transport systems for both ammonium and nitrate, leading to more comprehensive nitrogen acquisition.
Reduced Energy Expenditure: Ammonium assimilation requires less energy than nitrate, which must be reduced before incorporation into amino acids.
pH Regulation: Mixed nitrogen helps maintain favorable rhizosphere pH by balancing anion and cation uptake.
Hormonal Regulation: Balanced nitrogen nutrition influences phytohormone synthesis, particularly auxins, which promote leaf expansion and tillering [14].
Microbial Modulation of Plant Nitrogen Uptake
Beneficial soil microorganisms significantly enhance plant nitrogen acquisition through multiple mechanisms that extend beyond biological nitrogen fixation [15]. Plant growth-promoting microorganisms, including various taxa of bacteria, fungi, microalgae, and yeasts, influence root nitrogen uptake through:
Production of Signaling Molecules: Microbes release phytohormones, volatile organic compounds, and low-molecular-weight organic compounds that affect transporter expression and root architecture [15].
Modification of Root System Architecture: Microbial-derived auxins, particularly indole-3-acetic acid, promote root meristem expansion, rhizodermal cell elongation, and lateral root initiation, resulting in enhanced nutrient foraging capacity [15].
Regulation of Nitrogen Transporters: Microbial signals modulate the expression and activity of key nitrogen transporters, including nitrate transporters and ammonium transporters, through transcriptional and post-transcriptional mechanisms [15].
Nitrogen Cycling in Regenerative Life Support Systems
Current Physicochemical Life Support Systems
The International Space Station currently relies on physicochemical Environmental Control and Life Support Systems that recover oxygen and water but lack complete nitrogen recycling capabilities [16] [3]. The system comprises:
- Oxygen Generation Assembly: Produces oxygen through water electrolysis.
- Carbon Dioxide Removal Assembly: Uses regenerable absorbents to remove CO₂ from cabin air.
- Carbon Dioxide Reduction System: Converts CO₂ to water and methane via the Sabatier reaction.
- Urine Processor Assembly: Recovers water from urine through vacuum distillation [3].
A significant limitation of this system is the venting of methane produced in the Sabatier reaction, resulting in continuous loss of hydrogen and incomplete carbon/nitrogen recovery [3]. This approach requires substantial resupply from Earth and is unsuitable for long-duration missions beyond low Earth orbit.
Bioregenerative Life Support Systems Framework
Bioregenerative Life Support Systems incorporate biological elements to create more closed-loop systems that regenerate air, water, and food from waste streams [16] [3] [5]. The European Space Agency's Micro-Ecological Life Support System Alternative represents one of the most advanced BLSS concepts, designed as a five-compartment bioengineered system mimicking a lake ecosystem [3]. In such systems, nitrogen recovery is particularly crucial, with urine representing the largest recoverable nitrogen source at 7-16g N per crew member daily, accounting for 85% of the total recoverable nitrogen [3].
Urine Recycling and Nitrogen Recovery Technologies
Nitrogen recovery from urine represents a critical process in BLSS, with several technological approaches under development:
Nitrification-Based Systems: These employ nitrifying bacteria to convert ammonium to nitrate through a two-step process (ammonia oxidation followed by nitrite oxidation).
Urine Stabilization: Prevention of urea hydrolysis is essential to avoid ammonia volatilization. Current approaches include acidification with phosphoric acid and chemical oxidation using hexavalent chromium [3].
Direct Fertilizer Production: Processing of urine to produce stabilized fertilizers suitable for plant growth systems.
The MELiSSA loop's Compartment III is specifically dedicated to nitrification and urine recycling, using immobilized nitrifying bacteria to convert ammonia and urea to nitrate, which serves as fertilizer for subsequent plant production compartments [3].
Experimental Protocols for Nitrogen Cycling Research
Protocol for Assessing Plant Nitrogen Uptake Efficiency
Objective: Quantify nitrogen uptake efficiency in plants under different nitrogen formulations and microbial associations.
Materials and Methods:
- Plant Material: Select uniform seeds of target species (e.g., maize, wheat).
- Growth System: Hydroponic systems with controlled nutrient delivery, or soil-based systems with characterized substrate.
- Nitrogen Treatments: Implement at least three nitrogen regimes: 100% nitrate, 100% ammonium, and mixed ratios based on species-specific optima.
- Microbial Inoculation: Include treatments with and without plant growth-promoting rhizobacteria.
- Isotopic Labeling: Use ¹⁵N-labeled nitrogen sources to trace uptake and distribution.
- Harvest and Analysis: Destructive harvesting at multiple time points for measurement of biomass, nitrogen content, and ¹⁵N enrichment.
Measurements:
- Physiological Parameters: Biomass accumulation, root architecture traits, photosynthetic rates.
- Nitrogen Metabolism: Tissue nitrogen concentration, nitrate reductase activity, ammonium transporter expression.
- Microbial Effects: Colonization rates, phytohormone production, gene expression of nitrogen transporters.
Protocol for Nitrogen Balance Studies in Humans
Objective: Determine nitrogen balance and protein requirements in healthy adults.
Materials and Methods:
- Participants: Healthy adults meeting specific inclusion criteria.
- Study Design: Metabolic ward study with controlled dietary interventions.
- Dietary Protocol: Precisely formulated diets with graded levels of nitrogen intake.
- Sample Collection: Complete collection of urine, feces, and record of miscellaneous losses.
- Analysis: Kjeldahl or combustion analysis for nitrogen content in diet and excreta.
Calculations:
- Nitrogen Balance = N intake - (Urinary N + Fecal N + Integumental N)
- Determination of breakpoint intake where nitrogen balance equals zero.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Reagents for Nitrogen Cycling Studies
| Reagent/Material | Function/Application | Technical Specifications |
|---|---|---|
| ¹⁵N-Labeled Compounds | Isotopic tracing of nitrogen pathways in plant and microbial studies | ¹⁵NO₃⁻, ¹⁵NH₄⁺ at 95-99% atom enrichment |
| Nitrogen-Free Nutrient Solutions | Base solutions for hydroponic studies of nitrogen uptake | Contains all essential nutrients except nitrogen |
| Specific Nitrogen Transport Inhibitors | Mechanistic studies of nitrogen uptake pathways | e.g., chlorate for nitrate transport |
| Plant Growth-Promoting Rhizobacteria | Studies of plant-microbe interactions in nitrogen uptake | Specific strains with characterized PGP traits |
| Kjeldahl Digestion Apparatus | Total nitrogen analysis in biological samples | Automated systems with safety controls |
| Elemental Analyzer-Isotope Ratio Mass Spectrometer | Precise measurement of nitrogen content and isotopic composition | Capable of measuring natural abundance and enriched samples |
| Nitrate/Nitrite Test Kits | Rapid quantification of nitrogen species in solution | Colorimetric or ion-selective electrode based |
| Sterile Hydroponic Systems | Axenic plant growth studies | With controlled aeration and nutrient delivery |
The central role of nitrogen in both human metabolism and plant nutrition creates an essential linkage that must be optimized for successful regenerative life support systems. Current research demonstrates that nitrogen use efficiency can be significantly enhanced through balanced nutritional approaches, microbial partnerships, and advanced recycling technologies. The integration of plant science, human physiology, and microbial ecology will be essential for developing the next generation of life support systems that enable long-duration space exploration while simultaneously advancing sustainable nitrogen management practices on Earth. Future research should focus on closing the nitrogen loop completely in ground-based demonstrators, with particular emphasis on optimizing nitrogen recovery from waste streams and improving its bioavailability in food production systems.
Within the context of regenerative life support systems for long-duration space missions, the efficient recycling of nitrogen is a critical prerequisite for sustainable food production. This whitepaper delineates the composition of human urine as a primary nitrogen source and evaluates advanced technological pathways for its recovery and conversion into bioavailable nutrients. With urine contributing approximately 85% of the recoverable nitrogen in a closed-loop habitat, its management is paramount to reducing reliance on external resupply and energy-intensive nitrogen fixation processes. This technical guide provides a comprehensive analysis of urine composition, presents quantitative data on nitrogen content, details state-of-the-art recovery methodologies including membrane distillation and biological processing, and outlines standardized experimental protocols for related research. The integration of these processes into systems such as the Micro-Ecological Life Support System Alternative (MELiSSA) underscores their viability for maintaining nitrogen balance in extraterrestrial environments.
Regenerative Life Support Systems (RLSS) are fundamental for human survival in long-duration space exploration, such as missions to Mars. These systems aim to achieve near-closure of the loops for water, oxygen, and nutrients, thereby minimizing the need for resupply from Earth. Nitrogen is an especially critical element as it is a fundamental component of amino acids, proteins, and nucleic acids, all essential for plant growth and, consequently, food production [17].
The current Environmental Control and Life Support System (ECLSS) on the International Space Station (ISS) relies heavily on physicochemical processes. While it successfully recycles water and oxygen, it does not recover nutrients for food production. The Urine Processor Assembly (UPA) on the ISS stabilizes urine with acids and oxidizers, then uses distillation to recover water; however, the nitrogenous waste is concentrated and stored as a brine, rather than being recycled into a usable fertilizer [3]. This approach is insufficient for missions where resupply is impractical.
Bioregenerative Life Support Systems (BLSS) are being developed as advanced alternatives. These systems integrate biological components, such as microorganisms and plants, to regenerate air, water, and food from waste streams. In such systems, human urine is the most significant source of nitrogen, accounting for 75-87% of the nitrogen load in wastewater while constituting only about 1% of its volume [18] [3]. A crew member excretes an average of 7–16 grams of nitrogen per day, predominantly in the form of urea [CO(NH₂)₂] [3]. Effective recovery and conversion of this nitrogen into plant-available forms, such as ammonium (NH₄⁺) and nitrate (NO₃⁻), is therefore a cornerstone of sustainable RLSS design, drastically reducing the mass and energy penalties associated with transporting nitrogen from Earth or fixing it from atmospheric N₂ [17].
Comprehensive Composition of Human Urine
Human urine is a complex aqueous solution of metabolic wastes and minerals. A detailed understanding of its composition is essential for designing efficient recovery processes.
Physical and Chemical Characteristics
On average, an adult produces 1.4 liters of urine per day, with a typical range of 0.6 to 2.6 liters, depending on hydration, diet, and activity level [19]. Fresh urine has a slightly acidic to neutral pH, typically ranging from 5.5 to 7.0, with an average of 6.2. This pH can be influenced by diet; high-protein diets and alcohol consumption can lower pH (make it more acidic), while diets rich in fruits and vegetables can increase it (make it more alkaline) [19]. The specific gravity of urine ranges from 1.003 to 1.035 [19]. Notably, fresh urine from healthy individuals is not sterile; it contains a diverse community of bacteria, the urinary microbiome, which includes facultative anaerobes like Escherichia coli, Proteus, and Klebsiella [19].
Table 1: Primary Constituents of Human Urine
| Component | Typical Concentration/Range | Details/Significance |
|---|---|---|
| Water | 91 - 96% [19] | The primary solvent; removal is a key step in concentration. |
| Total Solids | ~59 g/day [19] | Varies with diet and exercise. |
| Urea | >50% of total solids [19] | The major nitrogen-containing compound, constituting over 50% of solids. |
| Total Nitrogen | 8,830 mg/L [19] | Represents the bulk of recoverable N. |
| Ammonia/Ammonium-N | 460 mg/L [19] | Product of urea hydrolysis. |
| Creatinine | Present [20] | Organic byproduct of muscle metabolism. |
| Uric Acid | Present [19] | Product of purine metabolism. |
| Chemical Oxygen Demand (COD) | 6,000 mg/L [19] | Indicator of organic content. |
| Chloride (Cl⁻) | 4,970 mg/L [19] | Major anion. |
| Sodium (Na⁺) | 3,450 mg/L [19] | Major cation. |
| Potassium (K⁺) | 2,740 mg/L [19] | Essential plant nutrient. |
| Sulphate (SO₄²⁻) | 1,500 mg/L [19] | |
| Total Phosphorus (P) | 800 - 2,000 mg/L [18] | Essential plant macronutrient. |
| Calcium (Ca²⁺) | 230 mg/L [19] | Can cause scaling (e.g., CaSO₄). |
| Magnesium (Mg²⁺) | 120 mg/L [19] |
Nitrogen Speciation and Quantification
Nitrogen in fresh urine is primarily found in the form of urea, which accounts for over 85% of the nitrogen excreted [17]. Other nitrogenous compounds include ammonium ions, creatinine, uric acid, and trace amounts of amino acids [19] [20]. The urea nitrogen test, which requires a 24-hour urine collection, is a standard clinical method for quantifying urea excretion. Normal results typically range from 12 to 20 grams per 24 hours (or 26 to 43 grams of urea nitrogen per 24 hours), reflecting dietary protein intake and the body's protein catabolic state [21] [22].
Urea is relatively stable in fresh, acidic urine but undergoes rapid enzymatic hydrolysis to ammonium (NH₄⁺) and bicarbonate (HCO₃⁻) in the presence of the enzyme urease, which is produced by some bacteria [17]. This reaction increases the pH of the solution, favoring the formation of volatile ammonia (NH₃), which can be lost to the atmosphere, thereby reducing nitrogen recovery efficiency. Controlling this hydrolysis is a major challenge in urine management.
Nitrogen Recovery Technologies and Methodologies
Several advanced technologies show high potential for recovering nitrogen and other valuable nutrients from source-separated urine.
Membrane Distillation (MD)
Membrane Distillation (MD) is an emerging thermal separation process that uses a hydrophobic microporous membrane. The temperature difference across the membrane creates a vapor pressure gradient, allowing water vapor to pass through while retaining non-volatile constituents, including nutrients like nitrogen and phosphorus [18].
- Principle: A PTFE-polypropylene (PTFE-PP) composite membrane is often used due to its high hydrophobicity, chemical stability, and anti-fouling properties. The feed solution (urine) is warmed, and vapor molecules diffuse through the membrane pores, condensing on the cooler permeate side, producing purified water and concentrating the nutrients in the feed [18].
- Performance: A recent study using a PTFE-PP membrane demonstrated exceptional recovery rates over a 14-day continuous operation, with 95% recovery of nitrogen and 99% recovery of phosphorus. The process also achieved a 95% recovery of dissolved organic carbon and effectively retained urea and larger organic molecules [18].
- Optimization: Key operational parameters include feed temperature and flow rate. Research indicates that at a temperature difference (ΔT) of 40 ± 2 °C and a feed flow rate of 0.7 L/min, membrane flux can reach a maximum of 2.61 kg/(m²·h) [18].
Figure 1: Membrane Distillation Process for Nitrogen Recovery. A thermal gradient drives water vapor through a hydrophobic membrane, concentrating nutrients.
Biological Processing and Nitrification
Biological systems leverage microorganisms to convert urea and other organic nitrogen compounds into plant-available forms. The European Space Agency's MELiSSA loop is a prominent example of this approach [3].
- Ureolysis and Nitrification: The process begins with the enzymatic hydrolysis of urea to ammonium by urease-producing bacteria. The ammonium is then sequentially oxidized by nitrifying bacteria: first by Nitrosomonas spp. to nitrite (NO₂⁻), and then by Nitrobacter spp. to nitrate (NO₃⁻) [17]. Nitrate is the preferred nitrogen source for many crop plants.
- System Integration: In the MELiSSA loop, this nitrification process occurs in a dedicated compartment (Compartment III). The resulting nitrate solution is then used to fertilize higher plants or algae, which produce food and oxygen for the crew, thus closing the loop [3].
Figure 2: Biological Nitrogen Transformation Pathway. Microorganisms convert urea into nitrate, a readily available plant nutrient.
Stabilization and Pre-treatment Strategies
Preventing nitrogen loss via ammonia volatilization is critical. Stabilization strategies often involve acidification of urine to maintain a pH below 7, which shifts the ammonium-ammonia equilibrium towards the non-volatile ammonium ion (NH₄⁺). On the ISS, urine is stabilized with a mixture of phosphoric acid (H₃PO₄) and an oxidizing agent like chromium (VI) to prevent urea hydrolysis and subsequent scaling [3]. Alternative biological pre-treatments, such as anaerobic fermentation, have also been explored. This process can convert urea and other organics into compounds like methylamine, acetate, and propionic acid, which can subsequently be processed in systems like Microbial Electrolysis Cells (MECs) for enhanced energy and resource recovery [20].
Experimental Protocols for Nitrogen Analysis and Recovery
Standardized protocols are essential for the R&D of nitrogen recovery technologies.
Protocol 1: 24-Hour Urine Urea Nitrogen Test
This protocol is used to quantify total urea nitrogen excretion, a key metric for mass balance calculations [21] [22].
- Equipment: Pre-chilled 24-hour urine collection container, ice chest or refrigerator, graduated cylinder.
- Procedure:
- On Day 1, discard the first morning urine sample and note the time.
- For the next 24 hours, collect every subsequent urine sample into the provided container, which must be kept refrigerated at 4°C throughout the collection period.
- On Day 2, void at the same time as the start time, adding this final sample to the container.
- Record the total 24-hour volume.
- Mix the total collection thoroughly and submit a representative aliquot for laboratory analysis.
- Analysis: The urea nitrogen concentration (in mg/dL or mmol/L) is determined, typically via automated clinical chemistry analyzers. The total mass excreted is calculated as: Concentration × Total Volume.
Protocol 2: Membrane Distillation for Nutrient Concentration
This protocol outlines a lab-scale MD process for nutrient recovery from synthetic or real urine [18].
- Equipment: Direct Contact Membrane Distillation (DCMD) unit; PTFE-PP hydrophobic flat-sheet or hollow-fiber membrane; peristaltic pumps; heating and cooling circulators; data acquisition system for flux measurement.
- Feed Preparation: Synthetic urine or real source-separated urine, pre-filtered through a 0.45 µm filter to remove particulates. The urine is stored at 4°C prior to use to inhibit hydrolysis.
- Operational Parameters:
- Set the feed side temperature (e.g., 60-70°C) and the permeate (distillate) side temperature (e.g., 20°C) using circulating water baths.
- Set the cross-flow velocity on the feed side using a peristaltic pump (e.g., 0.7 L/min).
- Run the process in continuous or batch mode for a set duration (e.g., 24 hours to 14 days).
- Monitoring and Analysis:
- Permeate Flux: Measure the mass of the permeate collected over time to calculate flux (kg/m²·h).
- Nutrient Recovery: Periodically sample the feed (concentrate) and permeate streams. Analyze for:
- Total Nitrogen and Ammonium (e.g., via colorimetric methods or ion chromatography).
- Total Phosphorus (e.g., via ascorbic acid method).
- Chemical Oxygen Demand (COD) to track organic matter removal.
- Recovery rates are calculated based on the concentration factor in the feed stream.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 2: Essential Materials for Urine Nitrogen Recovery Research
| Reagent/Material | Function/Application | Example Use Case |
|---|---|---|
| PTFE-PP Membrane | Hydrophobic, microporous separation medium. | Serves as the core component in Membrane Distillation for vapor transport and solute rejection [18]. |
| Urease Enzyme | Catalyzes the hydrolysis of urea to ammonium and carbonate. | Used in studies to simulate or accelerate biological urea breakdown in pretreatment stages [17]. |
| Nitrosomonas & Nitrobacter Cultures | Nitrifying bacteria for bio-conversion of ammonium to nitrate. | Inoculum for bioreactors (e.g., MELiSSA Compartment III) designed for producing plant-ready fertilizer [3] [17]. |
| Ascorbic Acid & Molybdate Solution | Reagents for colorimetric determination of phosphorus. | Essential for quantifying phosphorus recovery efficiency in permeate and concentrate streams [18]. |
| Phosphoric Acid (H₃PO₄) | Acidifying agent for urine stabilization. | Used to lower urine pH, suppressing ammonia volatilization and preventing scaling in collection systems [3]. |
Human urine presents a validated and highly concentrated source of nitrogen, making it an indispensable asset for closing the nutrient loop in regenerative life support systems. Technologies such as Membrane Distillation and controlled biological nitrification have demonstrated high efficiency, with recovery rates exceeding 95% for nitrogen, proving their technical feasibility.
Future research must focus on the integration and optimization of these technologies into a robust, reliable, and automated system for space flight. Key challenges include mitigating membrane fouling in MD systems, managing the microbial community stability in bioreactors under space conditions (e.g., microgravity, radiation), and further minimizing gaseous nitrogen losses throughout the process chain. The success of these endeavors will be a cornerstone for achieving the self-sufficiency required for the long-term exploration of the Moon, Mars, and beyond.
Bioregenerative Life Support Systems (BLSS) are artificial ecosystems designed to sustain human life in space by regenerating vital resources through biological processes. These systems integrate biological components (plants, microorganisms) with physicochemical technologies to create closed-loop cycles for air, water, and food [5]. The core principle involves using producer organisms (e.g., plants, microalgae) to generate oxygen and food, consumer organisms (humans) to consume these resources, and degraders (microorganisms) to recycle waste back into usable forms [5]. This approach addresses the critical limitation of current physicochemical systems on the International Space Station (ISS), which cannot produce food and require frequent, costly resupply missions from Earth [23]. For long-duration missions to the Moon or Mars, BLSS offers the potential for greater self-sufficiency by minimizing payload and enabling sustainable human presence in space [6] [5].
The MELiSSA Initiative
Foundation and Objectives
The Micro-Ecological Life Support System Alternative (MELiSSA) is a European Space Agency (ESA) program, established as one of the most long-standing and engineering-oriented BLSS initiatives [23]. Its design is inspired by a lake ecosystem, structured into five interconnected compartments, each representing a specific function within the closed-loop system [24] [23]. The primary objective of MELiSSA is to achieve a highly efficient, closed-loop recovery of resources, enabling the production of food, oxygen, and water, and the recycling of waste, with minimal consumables [23]. This is pursued through dedicated research infrastructures for studying, characterizing, and mathematically modeling biological processes [24].
System Architecture and Nitrogen Cycling
The MELiSSA loop is engineered to process waste and regenerate resources in a step-wise manner. Compartment III is particularly critical for nitrogen recovery, where nitrifying bacteria convert ammonia and urea from liquid waste (including urine) into nitrate [23]. This nitrate is then supplied as a key fertilizer to the higher plant compartment (Compartment IV), supporting plant growth for food production and air revitalization [23]. The system employs gas-liquid membrane bioreactors to facilitate efficient gas and nutrient exchange, which is vital for the health of both microbial and plant communities [23].
Table: Key Compartments of the MELiSSA Loop
| Compartment | Primary Function | Key Biological Components |
|---|---|---|
| Compartment I | Waste liquefaction and decomposition | Thermophilic anaerobic bacteria |
| Compartment II | Further breakdown of organic matter | Photoheterotrophic bacteria |
| Compartment III | Nitrogen recovery & nitrification | Nitrifying bacteria (e.g., Nitrosomonas, Nitrobacter) |
| Compartment IVa | Food production, O₂ generation, CO₂ consumption | Higher plants (crops) |
| Compartment IVb | O₂ generation, water recycling, CO₂ consumption | Photosynthetic bacteria (e.g., Spirulina) |
Key Experimental Research and Protocols
MELiSSA research utilizes specialized facilities like the Plant Characterization Unit (PaCMAN), a leak-proof plant chamber for detailed studies on crop development, gas exchange, and water transfer between the aerial and root zones [24]. Microgravity research is a core focus, with experiments such as ANTHEMS (studying leaf-level transpiration) and WAPS (investigating whole-plant water transport) conducted on parabolic flights and planned for the International Space Station to understand the impacts of weightlessness on plant physical processes [24].
Diagram: MELiSSA Loop Architecture and Nitrogen Flow
The Lunar Palace 1 Initiative
Foundation and Objectives
Lunar Palace 1 (LP1) is China's first ground-based integrative BLSS experimental facility and represents a significant milestone for being the site of the world's longest closed human BLSS experiment [25] [6]. With a total volume of 500 m³, it was designed as an artificial closed ecosystem integrating four biological loops: higher plants, animals (yellow mealworms), microorganisms, and humans [25]. The project's objectives center on achieving a high degree of material closure and demonstrating the feasibility of a sustainable, multi-species regenerative system to support human survival in deep space exploration and future lunar bases [25] [26].
System Architecture and Nitrogen Cycling
LP1's architecture comprises several integrated units. The Water Treatment Unit (WTU) plays a pivotal role in nitrogen management, using Membrane Biological Activated Carbon Reactoirs (MBARs) to treat various waste streams [26]. These MBARs are crucial for nitrogen recovery: they efficiently hydrolyze urea in urine, converting it to ammonium (NH₄⁺), and achieve nitrification of ammonium to nitrate (NO₃⁻) in domestic wastewater, making nitrogen available for plant uptake [26]. The system also includes dedicated plant cabins and a Mineral Element Supply Unit (MESU) to deliver recovered nutrients to plants. The integration of a Solid Waste Treatment and Yellow Mealworm Feeding Unit (SWT-YMFU) allows for the recycling of inedible plant biomass into animal protein, creating an additional nutrient loop [25].
Table: Key Units in the Lunar Palace 1 System
| Unit Name | Primary Function | Role in Nitrogen Cycle |
|---|---|---|
| Water Treatment Unit (WTU) | Wastewater processing & water reclamation | Nitrification; Urea hydrolysis |
| Mineral Element Supply Unit (MESU) | Nutrient solution preparation | Delivers recovered N (as nitrate) to plants |
| Plant Cabin 1 & 2 | Food production, O₂ generation | Assimilation of N by food crops |
| Atmosphere Management Unit (AMU) | Air revitalization (O₂/CO₂ control) | --- |
| Solid Waste Treatment & Yellow \nMealworm Feeding Unit (SWT-YMFU) | Processes inedible biomass & human waste | Recycles organic N into animal feed |
Key Experimental Research and Protocols
The landmark "Lunar Palace 365" experiment was a 370-day manned mission that provided extensive performance data [25] [26]. The water recycling system demonstrated high efficacy: the MBAR treating condensate wastewater produced water with a CODMn index of 0.74 ± 0.15 mg/L, meeting drinking water standards, while the MBAR for domestic wastewater achieved an 85.7% ± 10.2% removal rate of organic contaminants and stable nitrification [26]. Microbial community analysis via 16S rDNA sequencing identified Meiothermus, Rhodanobacter, and Ochrobactrum as the dominant genera responsible for the stable performance of the MBARs during long-term operation [26]. Reliability analysis based on the 370-day experiment estimated the mean lifetime of a BLSS like LP1 to be approximately 52.4 years under normal operation and maintenance [25].
Diagram: Lunar Palace 1 MBAR Waste Processing
Comparative Framework of BLSS Initiatives
A comparative analysis of major BLSS programs reveals distinct architectural philosophies and shared technological challenges.
Table: Comparative Analysis of Global BLSS Initiatives
| Initiative / \nCharacteristic | MELiSSA (ESA) | Lunar Palace 1 (China) | Historical Precedents \n(BIOS-3, Biosphere 2) |
|---|---|---|---|
| Primary Architecture | Five-compartment, \nengineered bioreactor loop | Multi-unit integrated system \nwith four biological loops | Large-scale, \nsoil-based closed ecology |
| Core Nitrogen \nCycling Strategy | Dedicated nitrification \ncompartment (Comp. III) | MBAR-based nitrification & \nurea hydrolysis | Natural soil \nmicrobial processes |
| Higher Plant Role | Food production, O₂ \ngeneration (Comp. IVa) | Food production, O₂ \ngeneration, feed for animals | Primary food \nproduction & air revival |
| Animal Integration | Limited (proposed insects/fish) | Yes (Yellow mealworms \nfor protein) | Included in \noriginal Biosphere 2 |
| Notable Performance \nData / Closure Rate | --- | >98% material closure; \n370-day human experiment | 100% water & air \nrecycling (BIOS-3) |
The Scientist's Toolkit: Key Research Reagents and Materials
Advancing BLSS research requires specialized reagents and materials for monitoring and maintaining system health.
Table: Essential Research Reagents and Materials for BLSS Experiments
| Reagent / Material | Primary Function | Application Example |
|---|---|---|
| ¹⁵N Isotope Tracers | Quantifying gross N transformation rates & pathways | Tracing nitrogen flux from urine to plant biomass [27] |
| Biological Activated Carbon | Medium for biofilm growth & adsorption of contaminants | Used in LP1's MBARs to support nitrifying microbial communities [26] |
| Specific Nutrient Media | Culturing and maintaining defined microbial consortia | Growing nitrifying bacteria (Nitrosomonas, Nitrobacter) in MELiSSA CIII [23] |
| DNA/RNA Extraction Kits & \n16S rDNA Sequencing Reagents | Analyzing microbial community composition and evolution | Tracking microbial population dynamics in LP1's MBARs over 370 days [26] |
| Hydroponic Nutrient Solutions | Delivering precise mineral nutrition to plants | Formulating solutions with recovered nitrate for plant growth chambers [26] [5] |
Future Research Directions and Open Questions
The future development of BLSS hinges on several critical research frontiers. A primary challenge is transitioning from Earth-based simulations to functional systems in space, where factors like microgravity and space radiation can significantly alter biological processes, including plant physiology and microbial nitrogen cycling [6] [5]. Future research will focus on lunar probe payload experiments to study small, uncrewed ecosystems in space and correct Earth-based design parameters [6]. Integrating discoveries of novel nitrifying microorganisms (e.g., Comammox bacteria that perform complete ammonia oxidation) could enhance the efficiency and robustness of nitrogen recovery subsystems [27]. Finally, developing and applying standardized sustainability assessment frameworks, such as the proposed Terraform Sustainability Assessment Framework, is crucial for quantitatively measuring a BLSS's ability to function indefinitely and comparing the performance of different system designs [28].
Technological Architectures for Nitrogen Recovery and Recycling
In the context of Bioregenerative Life Support Systems (BLSS) for long-duration space missions, the efficient recycling of nitrogen is a critical challenge for maintaining crew survival and system sustainability. With the high cost of resupply from Earth—exceeding $12,000 per kilogram—and the infeasibility of regular missions to distant locations like Mars, closed-loop nutrient recycling becomes paramount for food production [29] [3]. Human crew members excrete 7–16 grams of nitrogen per day, with 85% of this nitrogen originating from urine, primarily in the form of urea [3] [17]. This represents the largest flux of recoverable nitrogen within the habitat, making its efficient conversion into forms usable by plants a cornerstone of BLSS design. Biological nitrification, the microbial-mediated oxidation of ammonia to nitrate, serves as the essential engine for converting this waste-stream nitrogen into a suitable plant fertilizer, thereby closing the nitrogen loop between human consumption and food production [30] [17].
The current Environmental Control and Life Support System (ECLSS) on the International Space Station relies on physicochemical processes that recover water and oxygen but lack nutrient recycling capabilities, resulting in wasted nitrogen that must be resupplied [3]. In contrast, BLSS initiatives like the European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) project aim to integrate biological and physicochemical processes to create a closed-loop system where water, oxygen, and food are produced, and waste is minimized [3] [31]. This article provides an in-depth technical examination of the biological nitrification process, detailing the microbial agents, biochemical pathways, operational parameters, and experimental protocols for its application within regenerative life support systems.
The Science and Microbiology of Nitrification
Biochemical Pathways and Microbial Agents
Nitrification is a two-stage aerobic process performed by specific groups of autotrophic bacteria and archaea. The complete process oxidizes ammonia to nitrate, with nitrite as an intermediate, and can be summarized by the following energy-yielding reactions [30] [32]:
- Ammonia Oxidation: ( 2NH4^+ + 3O2 \rightarrow 2NO2^- + 4H^+ + 2H2O ) (Nitrosomonas)
- Nitrite Oxidation: ( 2NO2^- + O2 \rightarrow 2NO_3^- ) (Nitrobacter, Nitrospina)
The first stage, the oxidation of ammonia ((NH3)) or ammonium ((NH4^+)) to nitrite ((NO_2^-)), is known as nitritation and is typically the rate-limiting step in nitrification [30]. This process is primarily carried out by two distinct groups of microorganisms:
- Ammonia-Oxidizing Bacteria (AOB): These are typically gram-negative bacteria belonging to the Betaproteobacteria and Gammaproteobacteria classes, such as Nitrosomonas and Nitrosococcus. They possess the enzyme ammonia monooxygenase (AMO), which catalyzes the conversion of ammonia to hydroxylamine ((NH_2OH)), a crucial intermediate. Hydroxylamine is then oxidized to nitrite by the enzyme hydroxylamine oxidoreductase [30] [32].
- Ammonia-Oxidizing Archaea (AOA): Since their discovery in 2005, AOA such as Nitrosopumilus maritimus and Nitrososphaera viennensis have been recognized. In many environments, including soils and marine systems, AOA can dominate over AOB and may have a much higher affinity for ammonia, making them potentially significant contributors to nitrification in BLSS [30].
The second stage, the oxidation of nitrite to nitrate ((NO_3^-)), is known as nitratation and is performed by Nitrite-Oxidizing Bacteria (NOB). These bacteria belong to taxa such as Nitrospirota, Nitrospinota, Pseudomonadota, and Chloroflexota, including genera like Nitrospira, Nitrospina, and Nitrobacter [30]. The enzyme nitrite oxidoreductase is responsible for this oxidation step, which is coupled to proton translocation [32].
A revolutionary discovery in the field was the identification of comammox (complete ammonia oxidation) bacteria within the Nitrospira genus. These organisms can perform the complete oxidation of ammonia to nitrate in a single step within one organism, as demonstrated by Nitrospira inopinata [30]. This challenges the traditional two-step paradigm and has significant implications for optimizing nitrification processes in engineered systems.
Table 1: Key Microorganisms in the Biological Nitrification Process
| Functional Group | Representative Genera | Primary Metabolic Function | Key Enzyme |
|---|---|---|---|
| Ammonia-Oxidizing Bacteria (AOB) | Nitrosomonas, Nitrosococcus | Oxidizes (NH3)/(NH4^+) to (NO_2^-) | Ammonia monooxygenase (AMO) |
| Ammonia-Oxidizing Archaea (AOA) | Nitrosopumilus, Nitrososphaera | Oxidizes (NH3)/(NH4^+) to (NO_2^-) | Ammonia monooxygenase (AMO) |
| Nitrite-Oxidizing Bacteria (NOB) | Nitrospira, Nitrobacter, Nitrospina | Oxidizes (NO2^-) to (NO3^-) | Nitrite oxidoreductase |
| Comammox Bacteria | Nitrospira inopinata | Oxidizes (NH3) to (NO3^-) | Both AMO and nitrite oxidoreductase |
Nitrification in the Context of the Broader Nitrogen Cycle
In a BLSS, nitrification is one integral part of a complex regenerative nitrogen cycle [17]. The cycle begins when nitrogen-fixing microbes or industrial processes convert atmospheric dinitrogen gas ((N2)) into ammonia, a form that can be assimilated by organisms. Crew members consume nitrogen primarily as protein in their diet and excrete it mainly as urea in urine. Urea is hydrolyzed by the enzyme urease—produced by many bacteria—to ammonia and carbonic acid [17]. This ammonia then becomes the substrate for nitrification, being converted to nitrate. Plants uptake the nitrate (and some ammonium), assimilating it into proteins to produce food, thus completing the cycle. Losses from the system can occur through volatilization of ammonia under alkaline conditions or the production of gaseous nitrogen forms ((N2), (N_2O)) via denitrification or other microbial pathways, making process control essential [17].
Diagram 1: Simplified Nitrogen Cycle in a BLSS, highlighting the central role of nitrification and potential loss pathways. The cycle must be managed to minimize losses (red arrows) and ensure efficient recycling (blue and green arrows).
Technical Implementation in Life Support Systems
Urine Pretreatment and Processing
A major engineering challenge in BLSS is the initial processing of human urine to make it suitable for biological nitrification. On the ISS, urine is chemically stabilized to prevent microbial growth and scaling. Currently, a mixture of phosphoric acid ((H3PO4)) and chromium trioxide ((CrO_3)) is used. The acid converts volatile ammonia to non-volatile ammonium, while the hexavalent chromium acts as an oxidizing agent to suppress urea hydrolysis, thereby preventing ammonia formation and controlling microbial activity [3]. However, the use of toxic heavy metals like chromium is undesirable in a BLSS where the goal is full nutrient recycling for food production.
For regenerative systems, alternative pretreatment strategies are under investigation. The primary goals are to prevent urea hydrolysis prematurely, which leads to ammonia volatilization, and to manage the high salt content (sodium and chloride) in urine, which could accumulate to toxic levels for plants and nitrifying microbes if continuously recycled [3] [31]. Source separation of urine and controlled hydrolysis in a dedicated bioreactor are key areas of research within projects like MELiSSA [3].
Operational Parameters for Optimal Nitrification
The efficiency of nitrification in a bioreactor is highly dependent on maintaining strict environmental conditions. The following parameters are critical for maximizing conversion rates and minimizing the production of greenhouse gases like nitrous oxide ((N_2O)) [17]:
- pH: The optimal pH for nitrification is generally between 7.5 and 8.0. A pH below 6 can severely inhibit the activity of ammonia-oxidizing microorganisms. Furthermore, maintaining a pH below 7 is crucial in waste streams to minimize the volatilization of nitrogen as ammonia gas ((NH_3)) [17].
- Oxygen: Nitrification is an obligately aerobic process. Continuous aerobic conditions are required to ensure complete conversion to nitrate and to minimize the production of (N_2O) as a byproduct of incomplete nitrification or denitrification [17].
- Temperature: Mesophilic temperatures in the range of 25–35°C are typically optimal for most nitrifying consortia.
- Ammonium Concentration: The process is susceptible to inhibition by high levels of free ammonia ((NH3)) and nitrous acid ((HNO2)), which are dependent on the total ammonium concentration and pH.
Table 2: Key Operational Parameters for Efficient Nitrification in a BLSS Bioreactor
| Parameter | Optimal Range | Effect of Deviation from Optimum |
|---|---|---|
| pH | 7.5 - 8.0 | Low pH (<6) inhibits AOB/AOA; High pH (>8) increases risk of NH3 volatilization. |
| Dissolved Oxygen | > 2 mg/L (Aerobic) | Anoxic conditions halt nitrification, can lead to denitrification and N2O production. |
| Temperature | 25 - 35 °C (Mesophilic) | Lower temperatures slow microbial activity; high temperatures can deactivate enzymes. |
| Ammonium Load | System Dependent | High NH4+ and high pH can cause free ammonia inhibition; high NO2- can cause nitrous acid inhibition. |
| Salinity (NaCl) | As low as possible | Accumulation can inhibit both nitrifying microbes and downstream plant growth. |
Quantitative Nitrogen Mass Balance
For a BLSS to be sustainable, a high degree of nitrogen recovery is required. A crew member's daily nitrogen excretion can be broken down as follows, illustrating the flux that must be managed [3] [17]:
- Total Nitrogen Excreted per Crew Member: ~14 g N/person/day (based on 90-100 g protein intake)
- Nitrogen in Urine: ~12 g N/person/day (85% of total)
- Nitrogen in Feces: ~2 g N/person/day (15% of total)
With efficient recycling, initial estimates indicate that nearly all of this nitrogen can be recovered in forms usable by plants, drastically reducing the need for external nitrogen fixation [17]. The MELiSSA project, for instance, aims to achieve this through a multi-compartment system where one compartment is specifically dedicated to nitrification and nutrient recovery [3] [31].
Experimental Protocols for Nitrification Research
Enrichment and Cultivation of Nitrifying Consortia
Objective: To establish a stable, enriched culture of nitrifying bacteria from an environmental inoculum (e.g., activated sludge, soil) for use in BLSS bioreactor studies.
Materials:
- Inorganic Basal Medium: Prepare a solution containing (per liter): 0.5 g (KH2PO4), 0.5 g (K2HPO4), 0.2 g (MgSO4•7H2O), 0.01 g (CaCl2•2H2O), and 1 mL of trace element solution.
- Ammonium Substrate Stock Solution: 10 g/L ( (NH4)2SO_4 ) in deionized water.
- Trace Element Solution: Contains Fe, Mn, Zn, Cu, Co, Mo, etc.
- Environmental Inoculum: Collected from a wastewater treatment plant nitrification tank.
- Bioreactor or Erlenmeyer Flasks
- pH and dissolved oxygen (DO) probes and controllers
- Air pump with sterile air filter for aeration.
Methodology:
- Inoculation: Mix the inorganic basal medium with the environmental inoculum (e.g., 10% v/v) in the bioreactor.
- Substrate Addition: Add ammonium substrate stock to achieve an initial (NH_4^+)-N concentration of 50-100 mg/L.
- Condition Control: Adjust pH to 7.8 using a sterile sodium carbonate ((Na2CO3)) or sodium bicarbonate ((NaHCO_3)) solution and maintain using an automated controller. Maintain DO above 2 mg/L via aeration and stirring. Incubate at 28°C.
- Monitoring and Sub-culturing: Monitor (NH4^+), (NO2^-), and (NO3^-) concentrations daily via colorimetric assays or ion chromatography. When (NH4^+) is depleted and (NO_3^-) is the dominant nitrogen species, transfer a portion (e.g., 10-20%) of the culture to fresh medium with ammonium substrate. This serial sub-culturing enriches for the desired nitrifiers.
- Confirmation: The success of enrichment is confirmed by a high rate of ammonium removal coupled with stoichiometric accumulation of nitrate, and minimal accumulation of nitrite.
Diagram 2: Experimental Workflow for Enriching Nitrifying Consortia. The process involves sequential sub-culturing under controlled conditions to select for a stable and efficient microbial community.
Protocol for Assessing Nitrification Kinetics
Objective: To quantify the rate of ammonia oxidation and nitrite oxidation in a enriched culture or bioreactor sample.
Materials:
- Enriched nitrifying culture
- Inorganic basal medium (as above)
- Ammonium substrate stock (as above) OR Sodium nitrite ((NaNO_2)) stock for NOB testing
- Thermostated batch reactor vessel with magnetic stirrer
- pH and DO meters
- Equipment for nitrogen species analysis (e.g., spectrophotometer with reagent kits, ion chromatograph).
Methodology:
- Culture Preparation: Harvest cells from the enriched culture by gentle centrifugation and wash them with fresh basal medium to remove residual nitrogen species.
- Reactor Setup: Re-suspend the washed cells in basal medium within the batch reactor. Add either the ammonium or nitrite substrate to achieve a known initial concentration (e.g., 50 mg N/L).
- Kinetic Run: Maintain constant temperature, pH, and DO. Immediately begin taking small, frequent samples (e.g., every 15-30 minutes) over a period of 4-8 hours.
- Analysis and Calculation: Immediately analyze each sample for (NH4^+)-N, (NO2^-)-N, and (NO3^-)-N. Plot the concentration of each species versus time. The ammonia oxidation rate is calculated from the initial, linear slope of (NH4^+) depletion and (NO2^-) production. The nitrite oxidation rate is calculated from the initial, linear slope of (NO2^-) depletion and (NO_3^-) production. Rates are typically expressed as mg N/L/h or mg N/g biomass/h.
The Scientist's Toolkit: Key Reagents and Materials
Table 3: Essential Research Reagents for Nitrification Studies
| Reagent / Material | Function / Application | Technical Notes |
|---|---|---|
| Ammonium Sulfate ((NH₄)₂SO₄) | Standardized substrate for enriching AOB/AOA and for kinetic assays. | Provides a non-volatile source of ammonium. Concentration must be controlled to avoid inhibition. |
| Sodium Nitrite (NaNO₂) | Standardized substrate for enriching NOB and for kinetic assays. | Nitrite can be inhibitory at high concentrations; kinetics assays typically use <100 mg N/L. |
| Inorganic Basal Medium | Provides essential minerals (P, Mg, Ca, K, S) and trace elements for autotrophic growth. | Must be free of organic carbon to selectively enrich for autotrophic nitrifiers over heterotrophs. |
| Sodium Carbonate (Na₂CO₃) | pH buffer and alkalinity source. | Nitrification consumes alkalinity (produces H+). Sodium carbonate is used to maintain optimal pH (~7.8). |
| Specific Inhibitors (e.g., Allylthiourea) | To selectively inhibit AOB and distinguish between AOA and AOB activity, or to separate the two nitrification steps. | Used in mechanistic studies, not in continuous operation of a BLSS bioreactor. |
| Urease Enzyme | To catalyze the hydrolysis of urea in pretreated urine, generating the ammonia substrate for nitrification. | Required in a dedicated urea hydrolysis bioreactor stage prior to the nitrification reactor [3]. |
Current Research Challenges and Future Directions
Despite being a well-understood natural process, engineering robust nitrification for BLSS presents several ongoing challenges. Nitrogen loss remains a primary concern, with potential losses occurring at multiple points: during urine collection and pretreatment as (NH3) volatilization, and within the bioreactor itself through the production of (N2O) or (N_2) via denitrification in anoxic pockets [29] [17]. The accumulation of sodium and chloride from urine and pretreatment chemicals is another critical issue, as these can inhibit microbial activity and are toxic to plants, necessitating the development of efficient removal techniques [31].
The space environment itself introduces unique variables. Microgravity can affect fluid dynamics, gas transfer, and microbial biofilm formation, while increased ionizing radiation may cause microbial mutations or affect community stability [29] [3]. Ground-based demonstration facilities, such as those used in the MELiSSA program, are essential for testing integration and control strategies before space deployment.
Future research will focus on integrating nitrification with other BLSS subsystems, optimizing the use of comammox bacteria for simpler process control, and developing advanced sensors and control algorithms to maintain optimal conditions autonomously. By systematically addressing these challenges, biological nitrification will be a cornerstone technology for achieving the closed-loop nutrient recycling required for humanity's long-term presence in space.
In the context of developing regenerative life support systems (RLSS) for long-duration space missions, the efficient management of nitrogen is paramount. Nitrogen is an essential element for crew nutrition, constituting a major component of proteins and nucleic acids. With the metabolic requirement for a crew member estimated at approximately 1 mole of nitrogen per day, and the majority of this nitrogen excreted as urea in urine, its recovery and recycling become critical for system closure [17]. Physicochemical methods, particularly distillation and membrane separation, offer robust technological pathways for achieving this nitrogen loop closure. These technologies enable the recovery of vital nitrogen compounds from waste streams and the production of breathable air, thereby reducing the need for resupply from Earth and enhancing the sustainability of deep-space habitats [33] [23]. This technical guide provides an in-depth examination of distillation and membrane separation processes, their operational principles, and their integration within the broader nitrogen cycle of a RLSS.
Core Technological Principles
Distillation: Cryogenic Air Separation
Cryogenic distillation is a widely used method for separating nitrogen from air, capable of producing high-purity nitrogen gas [33].
Process Overview: The process begins with the intake of ambient air, which is then compressed. The compressed air is subsequently cooled to approximately 10°C and passed through a series of filters to remove contaminants such as moisture, oil, and water vapour. The cleaned air is then channeled through a heat exchanger into an expansion engine. Here, the rapid expansion of the compressed gas causes its temperature to drop drastically, falling below the condensation point of nitrogen (approximately -195.8 °C at 1 atmosphere of pressure), leading to the liquefaction of air. Finally, the different components of air are separated based on their differing boiling points through fractional distillation in a distillation column, allowing for the collection of high-purity nitrogen [33].
Key Principle: This technique exploits the differences in the volatilities (boiling points) of the constituent gases in air. The primary components, nitrogen and oxygen, have boiling points of -195.8°C and -183°C, respectively, enabling their separation at cryogenic temperatures [33].
Membrane Separation Technologies
Membrane separation techniques leverage the differential permeability of gases through a semi-permeable membrane material.
Process Overview: In nitrogen generation from air, compressed ambient air is introduced into a module containing hundreds of thousands of hollow-fiber membranes. These membranes are permeable to gas molecules, but the rate at which different gases permeate varies significantly. So-called 'fast' gases, such as oxygen (O₂), carbon dioxide (CO₂), and water vapour (H₂O), permeate the fiber walls more rapidly and are vented as waste stream. Nitrogen (N₂), being a 'slow' gas, passes through the membrane much more slowly, resulting in a nitrogen-enriched stream at the outlet [33] [34]. The surface of these membranes is uniquely shaped into hollow fibers to maximize the surface area available for permeation, thereby enhancing the separation efficiency [33].
Key Principle: The separation is driven by a difference in partial pressure across the membrane and is based on the solution-diffusion mechanism, where gases dissolve into the membrane material and then diffuse through it at different rates [35]. The selectivity of the membrane for different gases determines the efficiency of the separation.
Table 1: Comparison of Distillation and Membrane Separation for Nitrogen Production
| Feature | Cryogenic Distillation | Membrane Separation |
|---|---|---|
| Primary Principle | Separation based on differences in boiling points | Separation based on differences in gas permeation rates |
| Process Complexity | High; involves compression, cooling, liquefaction, and distillation | Low; involves compression and single-pass filtration |
| Typical Purity | Very High Purity | Up to 99.5% purity [34] |
| Key Equipment | Heat exchangers, expansion engines, distillation columns | Hollow-fiber membrane modules, filters |
| Footprint & Weight | Large and heavy due to complex equipment | Smaller footprint and lower weight [34] |
Nitrogen Recovery from Waste Streams
In a RLSS, a significant portion of nitrogen is found in liquid waste, particularly urine. Recovering this nitrogen is crucial for fertilizing plant growth compartments, which in turn provide food and oxygen.
Membrane technologies are a cornerstone for nitrogen recovery from waste streams. The research focus has evolved from simple nutrient concentration to the production of marketable products, such as fertilizers, through improved membrane materials and system designs [36]. Among various membrane techniques, Gas Permeable Membrane (GPM) technology has been identified as particularly promising. GPM can be used to recover ammonia from waste streams like urine after hydrolysis. The process involves acidification of the liquid to convert ammonium ions (NH₄⁺) to ammonia gas (NH₃), which then diffuses through a hydrophobic microporous membrane and is trapped in an absorbing solution on the other side, producing a concentrated ammonium salt solution suitable for use as a fertilizer [36].
This approach is more efficient than traditional nitrogen removal processes, such as nitrification/denitrification, which consume large amounts of energy (accounting for 50-60% of a wastewater treatment plant's energy use) and convert valuable reactive nitrogen back into inert atmospheric N₂, thus losing it from the life support loop [36].
Experimental Protocols for Nitrogen Recovery and Separation
Protocol: Nitrogen Production via Membrane Separation
Objective: To generate a high-purity nitrogen stream from compressed air using a hollow-fiber membrane module.
Materials:
- Compressed air supply
- General efficiency coalescing filter
- High efficiency coalescing filter
- Activated carbon filter (if using an oil-lubricated compressor) [37]
- Immersion heater (optional, for maintaining optimal membrane temperature)
- Hollow-fiber membrane nitrogen generator
- Nitrogen purity analyzer
Methodology:
- Air Preparation: Pass the compressed air through the filtration train: coalescing filter → high efficiency coalescing filter → activated carbon filter. This removes particulate matter, liquid aerosols, and oil vapours, protecting the membrane [33] [37].
- Pressure Regulation: Adjust the pressure of the cleaned, compressed air to the manufacturer's specified inlet pressure for the membrane module (typical operating pressures range from 5 to 20 bar).
- Separation: Direct the pressurized feed air into the inlet of the membrane module. The 'fast' gases (O₂, CO₂, H₂O) will permeate through the fiber walls and be vented from the module's permeate port.
- Product Collection: The nitrogen-enriched product gas is collected from the non-permeate (residue) outlet of the module.
- Purity Control: The purity of the nitrogen product stream is controlled by adjusting the feed gas flow rate and pressure. A higher flow rate through the module typically results in lower purity but higher yield, and vice versa.
- Analysis: Use a nitrogen purity analyzer or an oxygen sensor to monitor the purity of the product gas.
Protocol: Nitrogen Recovery from Waste Water using Gas Permeable Membrane
Objective: To recover nitrogen from a synthetic urine stream in the form of ammonium sulfate using gas permeable membrane technology.
Materials:
- Synthetic urine solution (containing urea or hydrolyzed ammonium ions)
- pH meter and adjustment chemicals (e.g., NaOH, H₂SO₄)
- Gas Permeable Membrane (GPM) module (e.g., with hydrophobic microporous fibers)
- Peristaltic or membrane pump
- Acidic trapping solution (e.g., 0.1M H₂SO₄)
- Spectrophotometer or ion-specific electrode for ammonium analysis
Methodology:
- Waste Stream Preparation: Hydrolyze the synthetic urine if it contains urea, using the enzyme urease to convert it to ammonium carbonate. Ensure the pH of the solution is sufficiently high (pH > 9) to shift the equilibrium towards volatile ammonia gas (NH₃) [36].
- System Setup: Circulate the prepared wastewater on one side of the GPM (the "feed side"). Circulate the acidic trapping solution (e.g., H₂SO₄) on the other side of the membrane (the "strip side").
- Ammonia Transfer: NH₃ from the alkaline feed solution volatilizes, diffuses through the membrane pores, and is trapped by the acidic solution on the strip side, where it is converted to non-volatile ammonium sulfate ((NH₄)₂SO₄).
- Process Monitoring: Periodically sample the trapping solution and analyze its ammonium concentration using a spectrophotometric method (e.g., Nessler's method or phenate method) or with an ion-selective electrode.
- Mass Balance: Track the decrease in ammonium concentration in the feed solution and the corresponding increase in the trap solution to calculate the recovery efficiency.
The following workflow diagrams the logical relationship and application of these two core technologies within a regenerative life support system.
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key materials and reagents essential for research and development in distillation and membrane separation for RLSS.
Table 2: Key Research Reagents and Materials
| Item | Function/Application |
|---|---|
| Hollow-Fiber Membrane Modules | The core separation unit for membrane-based N₂ production or NH₃ recovery; consists of thousands of fibers providing high surface area [33] [36]. |
| Carbon Molecular Sieve (CMS) | Adsorbent material used in Pressure Swing Adsorption (PSA) systems; preferentially adsorbs oxygen over nitrogen, allowing for nitrogen production [33] [34]. |
| Coalescing Filters | Essential pre-filtration for removing liquid aerosols and particulate matter from compressed air, protecting sensitive membrane materials from fouling and damage [33] [37]. |
| Activated Carbon Filters | Removes trace oil vapours and other hydrocarbon contaminants from the compressed air stream, which is critical for the long-term stability of both membranes and CMS [33] [37]. |
| Urease Enzyme | Catalyzes the hydrolysis of urea in urine into ammonia and carbon dioxide, a critical first step for subsequent nitrogen recovery via membranes or other processes [17] [3]. |
| Gas Permeable Membrane (GPM) | A specialized hydrophobic microporous membrane that allows the passage of ammonia gas (NH₃) while blocking the liquid phase, enabling nitrogen recovery from waste streams [36]. |
Integration in Regenerative Life Support Systems
The ultimate goal of these physicochemical methods is their seamless integration into a closed-loop ecosystem, such as the European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) loop [23] [5] [3]. In this context, membrane systems and distillation units are not standalone technologies but are interconnected with biological components.
The nitrogen cycle within a BLSS for a Martian outpost can be visualized as follows, showing the integration of physicochemical and biological processes:
Membrane-based nitrogen recovery from urine and other waste streams provides the necessary ammonium and nitrate fertilizers for plant growth in hydroponic systems [31]. The plants, in turn, produce food, oxygen, and clean water for the crew, while also consuming carbon dioxide. This integration drastically reduces the reliance on initial nitrogen stocks brought from Earth and minimizes the energy-intensive need for continuous biological nitrogen fixation, which is particularly challenging given the low concentration of N₂ in the Martian atmosphere [17]. The success of this integration hinges on maintaining high nutrient recovery efficiency while controlling the buildup of sodium and chloride in the system [31].
In the context of Bioregenerative Life Support Systems (BLSS) for long-duration space missions, the efficient recovery and recycling of nitrogen is a critical challenge. The inability of current physicochemical systems on the International Space Station (ISS) to recover nitrogen for food production creates a significant payload burden, with resupply costs exceeding $10,000 per kilogram [3]. Human urine, containing 85% of the recoverable nitrogen excreted by crew members (approximately 7-16g per person daily), represents the most significant untapped nutrient source for closing the nitrogen cycle [3]. Nitrogen cycling is therefore essential for transforming waste streams into fertilizers for food production, enabling mission self-sufficiency [38].
Traditional urine processing, such as the Urine Processor Assembly (UPA) on the ISS, relies on distillation and relies on chemical stabilization to prevent urea hydrolysis and scaling, but does not recover nitrogen for agricultural use [3]. This paper proposes an advanced hybrid methodology integrating immobilized urease technology with reduced pressure distillation to transform urea nitrogen into a valuable, concentrated nutrient solution.
Technical Background
The Nitrogen Cycle in Regenerative Life Support Systems
In a BLSS, the nitrogen cycle must be meticulously managed to sustain both crew nutrition and crop production. The goal is to convert waste nitrogen, primarily from urine, into forms assimilable by plants or edible microorganisms. Urine recycling plays an essential role, providing the nitrogen source for higher plant and microalgae growth, which in turn contribute to crew dietary protein intake [3]. The European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) project, a leading BLSS initiative, is engineered as a five-compartment loop to regenerate water, oxygen, and food from crew waste, with a specific compartment dedicated to nitrification and nitrogen recovery [3].
Limitations of Current Physicochemical Systems
The ISS Environmental Control and Life Support System (ECLSS) exemplifies the physicochemical approach. Its UPA stabilizes urine with acids and oxidizers to prevent urea hydrolysis, then recovers water via distillation, leaving a nitrogen-rich brine that is considered waste rather than a resource [3]. This approach fails to close the nitrogen loop, representing a significant strategic gap for long-duration missions [11].
Enzymatic Urea Hydrolysis via Immobilized Urease
Urease catalyzes the hydrolysis of urea into ammonia and bicarbonate. Employing free urease enzyme is impractical due to stability, reusability, and cost issues [39]. Enzyme immobilization on solid supports addresses these challenges by enhancing enzyme stability, facilitating separation, and enabling reuse across multiple cycles [40]. Recent research demonstrates the effectiveness of urease immobilized on glutaraldehyde-crosslinked chitosan-coated magnetic iron oxide nanoparticles (Urease/GA/CS/MIONPs), which allows for easy magnetic recovery and maintains robust activity across a range of pH and temperature conditions [39].
Hybrid Processing Methodology
The proposed hybrid process synergistically combines biological catalysis and physical separation. The enzymatic step efficiently converts urea to readily separable ammonium, while the distillation step under vacuum concentrates the ammonia solution and mitigates thermal degradation.
Experimental Protocol: Urease Immobilization and Hydrolysis
Materials and Reagents:
- Magnetic Iron-Oxide Nanoparticles (MIONPs): Synthesized via alkaline hydrolysis of ferrous chloride (FeCl₂) [39].
- Chitosan (CS): A biopolymer for coating MIONPs, providing functional groups for cross-linking.
- Glutaraldehyde (GA): A bifunctional cross-linker that covalently binds enzymes to the chitosan-coated support [39].
- Urease (Jack bean, Canavalia ensiformis): The catalyst for urea hydrolysis.
- Synthetic (SUr) and Real Human Urine (HUr): For testing and optimization. SUr composition should mirror real urine, containing urea, uric acid, creatinine, and inorganic salts [39].
Immobilization Procedure [39]:
- Chitosan Coating: Disperse 0.25 g of MIONPs in a surfactant solution (e.g., CTAB). Gradually add a chitosan solution (0.02 g CS in 100 mL of 1% acetic acid) to the nanoparticle suspension.
- Cross-linking and Immobilization: Activate the chitosan-coated MIONPs with a glutaraldehyde solution (2.5% v/v). Introduce the urease enzyme to the activated support for covalent bonding.
- Washing and Storage: Recover the Urease/GA/CS/MIONPs construct magnetically, wash thoroughly to remove unbound enzyme, and store at 4°C.
Hydrolysis and Optimization:
- Urine Dilution: Optimal urea hydrolysis by Urease/GA/CS/MIONPs is achieved at specific dilutions—1/20 for SUr and 1/24 for HUr—to mitigate inhibition from high urea concentrations and ionic strength [39].
- pH and Temperature: The immobilized urease exhibits peak activity at a neutral pH of 7.0 and remains efficient up to at least 45°C [39].
- Kinetic Analysis: The immobilized system demonstrates Michaelis-Menten kinetics, with reported ( Km ) and ( V{max} ) values of 14.81 mM and 18.55 μM min⁻¹ for SUr, and 12.36 mM and 10.10 μM min⁻¹ for HUr, indicating high affinity and activity [39].
Experimental Protocol: Ammonia Concentration via Reduced Pressure Distillation
Principle: Applying reduced pressure lowers the boiling point of the ammonia-containing solution post-hydrolysis. This minimizes the thermal decomposition of other urine constituents and reduces energy input, which is crucial for space applications.
Distillation Setup and Procedure:
- Apparatus Setup: Utilize a standard distillation setup modified for vacuum operation, including a round-bottom flask, a distillation column or condenser, a receiving flask, and a vacuum pump.
- Sample Loading: Transfer the ammonium-rich hydrolysate into the distillation flask.
- Vacuum Application: Engage the vacuum pump to achieve a controlled reduced pressure within the system.
- Distillation: Gently heat the solution. The reduced pressure will allow the ammonia-enriched water vapor to distill over at a lower temperature than at atmospheric pressure.
- Collection: Collect the distillate in the receiving flask. This concentrate contains a higher molarity of ammonium ions, suitable for use as a liquid fertilizer.
Table 1: Key Performance Data for Immobilized Urease [39]
| Parameter | Value for Synthetic Urine (SUr) | Value for Human Urine (HUr) |
|---|---|---|
| Optimal Dilution | 1/20 | 1/24 |
| Optimal pH | 7.0 | 7.0 |
| Operational Temperature | Up to 45°C | Up to 45°C |
| Michaelis Constant ((K_m)) | 14.81 mM | 12.36 mM |
| Maximum Reaction Rate ((V_{max})) | 18.55 μM min⁻¹ | 10.10 μM min⁻¹ |
| Reusability | >10 cycles with >80% activity retention | >10 cycles with >80% activity retention |
Integrated System Workflow
The following diagram illustrates the sequential and integrated workflow of the hybrid processing system, from urine collection to the production of a concentrated nitrogen fertilizer.
Diagram 1: Integrated workflow for hybrid nitrogen recovery from urine.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagents and Materials for Nitrogen Recovery Experiments
| Reagent/Material | Function and Rationale |
|---|---|
| Urease (Jack bean) | Source of the enzyme that catalyzes the hydrolysis of urea into ammonia and bicarbonate [39]. |
| Magnetic Iron Oxide Nanoparticles (MIONPs) | A core support material that allows for easy separation and recovery of the immobilized enzyme using an external magnetic field [39]. |
| Chitosan | A biocompatible polymer used to coat MIONPs, providing functional amino groups for subsequent cross-linking with the enzyme [39]. |
| Glutaraldehyde | A bifunctional cross-linking agent that forms stable covalent bonds between the chitosan support and the enzyme, preventing enzyme leakage [40] [39]. |
| Synthetic Urine | A defined, consistent model fluid containing urea, creatinine, uric acid, and inorganic salts, used for controlled experimental optimization and to avoid the variability of real urine [39]. |
| Vacuum Distillation Apparatus | A system including a vacuum pump, distillation flask, and condenser used to concentrate the ammonium solution at lower temperatures, saving energy and preventing thermal degradation [3]. |
Discussion and Future Perspectives
The data demonstrates that this hybrid process effectively addresses key challenges in nitrogen recovery. The immobilized urease construct offers significant operational stability, retaining over 80% of its activity after ten consecutive reuse cycles, a critical factor for long-duration space missions where resupply is impossible [39]. The use of magnetic separation simplifies the process flow and enhances practicality.
The kinetic parameters confirm the efficacy of the immobilized enzyme in both synthetic and real human urine matrices, with the performance in SUr validating its use as a reliable model for process development [39]. The subsequent distillation step upgrades the dilute ammonium solution into a concentrated product, directly addressing the logistical challenge of transporting large urine volumes for agriculture, a noted obstacle in both terrestrial and space-based recycling [3] [39].
For future integration into a BLSS like MELiSSA, this hybrid processing unit would act as a critical nitrogen refinery stage. The concentrated ammonium fertilizer produced can be directed to hydroponic systems for higher plant cultivation or to photobioreactors for the cultivation of edible microalgae and cyanobacteria, thereby closing the nitrogen loop and contributing to crew food production [3] [38].
Further research should focus on optimizing the integration of these two unit operations, conducting long-term continuous testing, and validating system performance under simulated space conditions, such as microgravity and increased radiation. Success in this endeavor will mark a profound advancement toward the logistical biosustainability required for human endurance-class exploration missions to Mars and beyond [11].
The establishment of sustainable life support systems is a critical prerequisite for long-duration space exploration and extraterrestrial habitation. Within this context, the efficient management of nitrogen (N) is paramount, as it is an essential element for the production of amino acids, proteins, and nucleic acids, supporting both food production and crew nutrition. In a closed-loop habitat, nitrogen undergoes a continuous cycle of consumption and regeneration. Bioregenerative Life Support Systems (BLSS) aim to replicate and miniaturize Earth's natural nutrient cycles within a controlled engineering framework, moving beyond the current Physicochemical Life Support Systems (PLSS) used on the International Space Station (ISS), which do not recover nitrogen for food production [3] [9]. The core challenge lies in configuring two fundamental processes: Nitrogen Fixation (NF), which introduces new, bioavailable nitrogen into the system (e.g., from inert atmospheric N₂), and Nitrogen Recycling (R), which reclaims nitrogen from crew waste streams [41]. The integration of these processes presents a critical systems engineering choice, leading to the conceptualization of two primary architectures: the Separate NFR model and the Combined NFR model [41]. This whitepaper provides an in-depth technical analysis of these two configurations, examining their underlying principles, quantitative performance, and experimental approaches within the broader scope of nitrogen cycling research for regenerative life support.
Core Architectural Principles of NFR Models
The Separate NFR Model
The Separate NFR Model is characterized by a system architecture where nitrogen fixation and recycling operate as distinct, parallel processes. In this configuration, an external N₂ fixation fermenter acts as an independent supplier of new nitrogen to a primarily closed recycling loop [41]. The recycling subsystem internally processes waste streams—such as inedible crop biomass, feces, and urine—using technologies like aerobic digestion (AE) and anaerobic digestion (AN) to recover nutrients. The role of the NF fermenter is specifically to offset nitrogen losses that occur due to inefficiencies in these recycling processes. A key feature of this model is the direct transfer of urea from urine to agriculture after salts removal, which simplifies the system closure [41]. This architecture effectively decouples the challenges of waste remediation from those of new nitrogen production, allowing for specialized optimization of each subsystem.
The Combined NFR Model
In contrast, the Combined NFR Model integrates the functions of recycling and fixation into a single, synergistic process. This model incorporates waste streams, particularly urine, directly into the N₂ fixation fermenter. Here, the fixed nitrogen and carbon sources present in the waste serve as substrates to accelerate the growth and metabolic activity of nitrogen-fixing microorganisms [41]. This approach necessitates the use of specialized microbial strains, such as the photoheterotrophic Rhodopseudomonas palustris nifA* mutant, which has been genetically engineered to constitutively express nitrogenase, the enzyme complex responsible for fixation, even in the presence of fixed nitrogen compounds like ammonium [41]. For organisms without constitutive expression, carefully timed feeding of limited fixed N at the start of fermenter cycles can still boost biomass growth and subsequent N₂ fixation rates. This model creates a more deeply integrated loop, where waste is not merely processed but is actively used to drive the process that replenishes the system's nitrogen capital.
The fundamental architectural difference is illustrated in the following system diagrams, generated from the DOT scripts detailing the logical flow of materials and processes in each model.
Separate NFR Model Architecture
Combined NFR Model Architecture
Quantitative System Comparison and Performance Modeling
The operational performance and resource demands of the Separate and Combined NFR models can be evaluated and compared through mathematical modeling. The fundamental mass balance for a colony of size x crewmembers (CM) at time t is given by [41]: ΔN = xt(Nin + Nrec - Nout) Where ΔN is the net change in system nitrogen, Nin is the mass of fixed N from N₂, Nrec is the mass of recycled N, and Nout is the mass of N lost without recycling.
Table 1: Model Equations and Key Parameters for NFR Systems
| Model Component | Mathematical Formulation | Key Parameters |
|---|---|---|
| General N Balance | ΔN = x*t*(Nin + Nrec - Nout) | x: Crew size; t: Time [41] |
| N Input (Nin) | Nin = rVηBη[x]WCZ | r: Vol. fixation rate (g L⁻¹ d⁻¹); V: Reactor vol. (L CM⁻¹); ηB: N harvest efficiency [41] |
| N Recycling (Nrec) | Nrec = ND[WC(1/ηH-1) + WFηWW + ηU(1-WF)]η[x] | ηH: Harvest efficiency; ηU: Urine N recyclable; WF: Fecal fraction [41] |
| N Output (Nout) | Nout = ND / (ηFηH) | ND: Crew N demand (~14 g CM⁻¹ d⁻¹); ηF: Fertilization efficiency [41] |
| Digestion Efficiency (η[x]) | η[x] = ηAEx + ηAN(1-x) | ηAE, ηAN: Aerobic/Anaerobic digestion efficiencies; x: Flow fraction [41] |
The following table synthesizes critical performance metrics and parameters that dictate the efficiency and feasibility of each NFR configuration, based on modeled scenarios.
Table 2: Comparative Performance Metrics for Separate and Combined NFR Models
| Performance Metric | Separate NFR Model | Combined NFR Model | Impact on System Design |
|---|---|---|---|
| Fixation Fermenter Volume | Demands larger volumes to compensate for all recycling losses (>2500 L CM⁻¹ without recycling) [41]. | Potentially reduced volume due to waste-driven acceleration of microbial growth [41]. | Major impact on system mass, volume, and energy footprint. |
| Loop Complexity | Simplified, decoupled processes. Direct urine-to-soil fertilization is feasible [41]. | Highly integrated and interdependent processes. Requires precise control [41]. | Affects operational robustness, control engineering, and failure management. |
| Microbial Requirement | Standard N₂-fixing organisms suffice. | Requires specialized strains (e.g., R. palustris nifA*) that fix N₂ in presence of fixed N [41]. | Impacts biological stability and genetic reliability of the system. |
| Energy Demand | High energy cost for standalone fixation. Recycling process energy is separate. | Potentially lower specific energy cost for fixation, but integrated system energy must be evaluated. | Critical for total power budget of the habitat. |
| Key Efficiency Lever | Maximizing N recovery (ηrec) from waste streams to minimize Nin demand [41]. | Optimizing the use of waste C and N to maximize the rate (r) of N₂ fixation [41]. | Guides research and development priorities. |
| Sensitivity to Losses | Highly sensitive to N recovery efficiencies (ηAE, ηAN, ηU) and harvest index (ηH) [41]. | Sensitive to the same recycling efficiencies, but accelerated growth may mitigate some demand. | Identifies critical points of failure and required reliability targets. |
Experimental Protocols for NFR Model Validation
Robust experimental validation is required to move from theoretical models to deployable systems. The following protocols outline key methodologies for evaluating components relevant to both NFR models.
Protocol for Quantifying Nitrogen Fixation Rates (NFRP& NFRD)
This protocol, adapted from marine biology for BLSS application, measures fixation in both particulate and dissolved fractions [42].
- Sample Collection and Preparation: Collect samples from the fermenter broth or relevant liquid medium. For vertical profile studies, samples are taken from various depths/zones representing different light regimes [42].
- ¹⁵N₂ Isotope Labeling Incubation:
- Add a precise amount of ¹⁵N₂-enriched gas to the sample vials.
- Conduct parallel incubations under controlled environmental conditions (e.g., light intensity, temperature) relevant to the tested organism (e.g., Trichodesmium, R. palustris).
- Include killed controls (e.g., with formalin) to account for abiotic processes.
- Size-Fractionated Filtration:
- After a defined incubation period (e.g., 6-24 hours), terminate the reaction.
- Sequentially filter the sample through membrane filters (e.g., 10 μm and 0.2 μm pore sizes) to separate large particulate, small particulate, and dissolved fractions.
- Analysis and Calculation:
- Analyze the particulate material on the filters and the dissolved fraction in the filtrate for ¹⁵N enrichment using a Mass Spectrometer or Nanometer-Scale Secondary Ion Mass Spectrometry (NanoSIMS).
- Calculate the Nitrogen Fixation Rate in the Particulate fraction (NFRP) and the Nitrogen Fixation Rate in the Dissolved fraction (NFRD) based on the isotopic enrichment and incubation time. The sum of NFRP and NFRD gives the Total Nitrogen Fixation Rate (TNFR) [42].
Protocol for Light Manipulation Experiments
This protocol tests the critical hypothesis that light intensity regulates DDN release, which is fundamental to optimizing photobioreactor designs [42].
- Experimental Setup: Establish cultures of the target photoautotrophic or photoheterotrophic diazotroph (e.g., Trichodesmium, UCYN-B).
- Light Gradient Exposure: Subject parallel cultures to a gradient of light intensities, simulating conditions from the upper to the lower euphotic zone (e.g., from 145 μmol E m⁻² s⁻¹ down to 0.1% surface PAR) [42].
- Incubation and Sampling: Incubate cultures for a set period and then sample them for analysis.
- Measurement: Apply the ¹⁵N₂ labeling protocol (4.1) to each light treatment to quantify how NFRP and NFRD shift with changing light intensity. This identifies the optimal light regime for total fixation and for the release of bioavailable dissolved nitrogen [42].
The Scientist's Toolkit: Essential Research Reagents and Materials
The experimental study of NFR models relies on a suite of specialized reagents and equipment.
Table 3: Key Research Reagents and Materials for NFR Experimentation
| Item | Function/Application | Relevance to NFR Models |
|---|---|---|
| ¹⁵N₂ Gas (Isotope Label) | Tracer for quantifying nitrogen fixation rates and tracking DDN transfer [42]. | Core to Protocols 4.1 & 4.2; essential for validating model predictions of N influx. |
| Mass Spectrometer | Measures isotopic ratios (e.g., ¹⁵N/¹⁴N) in samples for calculating fixation and assimilation rates [42]. | Required for final analytical measurement in all isotopic tracer studies. |
| Nanometer-Scale SIMS (NanoSIMS) | High-resolution imaging mass spectrometry to visualize and quantify ¹⁵N incorporation into specific cells or tissues [42]. | Enables precise tracking of DDN transfer to specific non-diazotrophic organisms in a community. |
| Aerobic & Anaerobic Bioreactors | Controlled vessels for simulating waste digestion processes (AE/AN) [41] [9]. | Used to determine key efficiency parameters (ηAE, ηAN) for the recycling (R) component. |
| Specialized Microbial Strains | Engineered or selected diazotrophs (e.g., Rhodopseudomonas palustris nifA* mutant) for combined NFR systems [41]. | Critical experimental variable for testing the feasibility of the Combined NFR model. |
| Light-Controlled Incubators | Precisely manipulate light intensity and photoperiod for photo-bioreactor studies [42]. | Essential for Protocol 4.2 and for optimizing growth conditions for phototrophic diazotrophs. |
The choice between the Separate and Combined NFR models represents a fundamental trade-off between the simplicity and specialization of the former and the integration and potential efficiency of the latter. The Separate NFR model, with its decoupled processes, may offer greater initial robustness and easier control, a significant advantage for early missions. The Combined NFR model presents a compelling vision of a more tightly closed loop, where waste is not a burden but a resource to drive essential processes, potentially reducing the mass and volume of dedicated fixation hardware [41]. Future research must focus on closing the knowledge gaps identified in this analysis. Key directions include the quantitative characterization of nitrogen recovery efficiencies (ηAE, ηAN, ηU) across different waste streams and processing technologies [41] [9]; the optimization of crop harvest index (ηH) for closed-loop systems; the long-term stability and performance of constitutive nitrogen-fixing microbes under simulated space conditions; and the integrated system-level testing of both models to validate the mathematical predictions and identify emergent challenges. The development of efficient nitrogen management, combining the best of both fixation and recycling, is not merely a technical obstacle but an enabling technology for humanity's sustainable future beyond Earth.
The integration of waste processing with hydroponic food production represents a cornerstone of Bioregenerative Life Support Systems (BLSS), which are critical for long-term space exploration missions [3]. These systems aim to achieve a high degree of resource closure by recovering water, oxygen, and nutrients from astronaut waste and channeling them into food production systems [31]. Nitrogen, being a fundamental macronutrient for plant growth, is a primary target for recycling, with human urine constituting the most significant source of recoverable nitrogen—accounting for 85% of the total potentially recoverable nitrogen in a BLSS [3]. This technical guide examines the system integration strategies for creating a robust link between waste processing and hydroponics, with a specific focus on nitrogen cycling within the context of regenerative life support research.
The Nitrogen Cycle in Regenerative Life Support Systems
The Centrality of Nitrogen Recovery
In a closed-loop life support system, the efficient recovery and recycling of nitrogen are paramount. It is estimated that a single crew member excretes 7–16 grams of nitrogen per day, predominantly in the form of urea in urine [3]. Shipping this mass of fertilizers from Earth for long-duration missions is logistically and economically prohibitive, with cargo costs exceeding $10,000 per kilogram [3]. Therefore, the in-situ conversion of this waste stream into plant-available nitrogen is a critical technological challenge. The overarching goal of system integration is to transform this waste nitrogen into a safe and stable fertilizer for hydroponic cultivation, thereby supporting crop production and contributing to food self-sufficiency.
Key Nitrogen Transformation Pathways
The transformation of organic nitrogen from waste into forms readily absorbable by plants involves several key biochemical pathways, primarily mediated by microorganisms:
- Ureolysis: The enzyme-mediated hydrolysis of urea ([CO(NH₂)₂]) into ammonium (NH₄⁺) and bicarbonate (HCO₃⁻).
- Nitrification: A two-step aerobic process where ammonium (NH₄⁺) is first oxidized to nitrite (NO₂⁻) by bacteria such as Nitrosomonas, and then to nitrate (NO₃⁻) by bacteria such as Nitrobacter. Nitrate is the preferred nitrogen form for most higher plants in hydroponics [43].
- Assimilation: The uptake and incorporation of nitrate (NO₃⁻) and ammonium (NH₄⁺) by plant roots to synthesize organic compounds.
The following diagram illustrates the integrated flow of nitrogen from waste streams to plant biomass within a BLSS.
System Integration Architectures and Methodologies
System integration involves connecting disparate software and hardware modules into a cohesive infrastructure to enable efficient data and resource sharing [44]. For waste-to-hydroponics systems, this can be realized through several architectural models.
Comparison of Integration Architectures
The choice of integration architecture depends on the system's scale, complexity, and required robustness. The following table compares three prevalent models.
Table 1: System Integration Architectures for BLSS
| Architecture | Core Principle | Pros | Cons | Suitability for BLSS |
|---|---|---|---|---|
| Point-to-Point (P2P) | Direct connection between every system pair [44] | Simple for very small systems (e.g., 2-3 components) [44] | Becomes unmanageable "spaghetti integration" as systems grow; low scalability [44] | Low. Unsuitable for complex, multi-compartment systems like MELiSSA. |
| Hub-and-Spoke | Central hub (message broker) manages all communication [44] | Simplified connections; easier security management; more scalable than P2P [44] | Central hub is a single point of failure; can become a bottleneck [44] | Medium. Could be applied to smaller-scale terrestrial prototypes. |
| Enterprise Service Bus (ESB) | Decentralized "bus" with individual integration engines for each system [44] | High scalability and reliability; systems can be changed with minimal impact [44] | Complex to design and maintain; troubleshooting can be difficult [44] | High. Ideal for complex, modular systems like MELiSSA where compartments must be decoupled and robust [3]. |
The MELiSSA Loop: A Paradigm for Closed-Loop Integration
The European Space Agency's Micro-Ecological Life Support System Alternative (MELiSSA) is a pioneering example of a highly integrated BLSS [3]. It is designed as a five-compartment, bioengineered ecosystem that mimics the function of a lake to recycle waste and produce food, water, and oxygen [3] [31]. Its integration logic is complex and follows a flow of matter and energy between compartments, from waste processing to higher plant cultivation.
In the MELiSSA context, Compartment III is particularly critical for nitrogen cycling. It is responsible for the nitrification process, converting ammonium from processed waste into nitrate, making nitrogen available for the higher plants in Compartment V [3]. This compartmentalized, ESB-like architecture allows for specialized microbial processes and independent control, enhancing overall system stability and efficiency.
Experimental Protocols for Nitrogen Recovery and Management
Protocol 1: Creation of a Nitrified Digestate Fertilizer
This protocol outlines the methodology for converting ammonium-rich anaerobic digestate into a nitrate-based hydroponic nutrient solution, as investigated in recent research [45].
- Objective: To produce a stable, plant-available nutrient solution from organic waste digestate via controlled nitrification.
- Materials:
- Source of anaerobic digestate (e.g., from biogas production).
- Aerobic bioreactor with aeration system and temperature control (25-30°C).
- Inoculum of nitrifying bacteria (e.g., from activated sludge or commercial products).
- pH and Dissolved Oxygen (DO) probes and controllers.
- Chemicals for pH adjustment (e.g., KOH, H₂SO₄).
- Methodology:
- Inoculation and Aeration: Mix the digestate with the nitrifying bacteria inoculum in the bioreactor. Initiate continuous aeration to maintain DO > 2 mg/L, essential for the aerobic nitrification process.
- pH Monitoring and Control: Monitor pH continuously. The nitrification process consumes alkalinity and naturally acidifies the solution (2NH₄⁺ + 3O₂ → 2NO₂⁻ + 4H⁺ + 2H₂O). Maintain a stable pH (e.g., ~7.0) using automated hydroxide (OH⁻) addition.
- Process Completion: The process is complete when ammonium levels are negligible and nitrate levels have plateaued. This can be confirmed with ion-specific electrodes or colorimetric test kits.
- Key Control Strategy: A study demonstrated that a novel pH-based control strategy using hydroxide and ammonium dosing at a specific molar ratio can maintain high plant growth rates at nitrogen concentrations ten times lower than conventional methods, drastically improving Nitrogen Use Efficiency (NUE) [45].
Protocol 2: Formulation and Use of Compost-Based Liquid Extracts
This protocol details the preparation and application of organic liquid fertilizers, such as compost tea or vermicompost tea, for hydroponic use [46].
- Objective: To create a nutrient-rich, organic hydroponic solution from composted materials and evaluate its efficacy.
- Materials:
- High-quality compost or vermicompost (e.g., worm castings, in-vessel compost).
- Dechlorinated water.
- Brewing vessel with aeration (air pump, tubing, air stones).
- Mesh bags (200-400 microns) for containing solid compost.
- Molasses or another microbial food source (optional).
- Methodology:
- Extraction ("Brewing"): Place compost in a mesh bag and suspend it in the brewing vessel filled with water. Aerate vigorously for 24-48 hours. Aeration encourages the growth of beneficial, aerobic microorganisms and prevents anaerobiosis.
- Dilution and Application: After brewing, the concentrated tea is diluted with water. A 2025 study on lettuce in Deep-Water Culture (DWC) systems found optimal dilution ratios varied by compost type: Worm Casting Tea (WCT) at a 2:3 (v/v) ratio and other compost teas (e.g., InVCT, RMCT) at a 1:4 (v/v) ratio produced the highest biomass yields [46].
- System Monitoring: Closely monitor the hydroponic solution's pH, Electrical Conductivity (EC), and ammonium levels. Organic solutions can be unstable, and high ammonium can be phytotoxic.
- Performance Data: The 2025 study concluded that WCT (2:3) yielded ∼20% lower than synthetic fertilizers but had comparable nitrate use efficiency and tissue nutrient profiles, making it a strong candidate as a stand-alone organic nutrient source [46].
The Scientist's Toolkit: Key Reagents and Materials
Table 2: Essential Research Reagents for Nitrogen Cycling Experiments
| Item | Function in Research | Application Context |
|---|---|---|
| Nitrifying Bacteria Inoculum | Converts toxic ammonium (NH₄⁺) into plant-available nitrate (NO₃⁻) [43]. | Core to bioreactors in waste processing compartments (e.g., MELiSSA III) [3]. |
| Ion-Selective Electrodes / Photometric Kits | Precisely measure concentrations of NH₄⁺, NO₂⁻, and NO₃⁻ in solution. | Critical for monitoring nitrification efficiency and nutrient solution composition [45]. |
| pH & EC Controllers | Automatically maintain optimal pH (5.5-6.5) and Electrical Conductivity (nutrient strength) in hydroponic solutions [47]. | Essential for stable plant growth, especially when using variable waste-derived nutrients. |
| Organic Digestate / Compost | Serves as the raw material for nutrient recovery, providing a complex mix of macro/micronutrients [45] [46]. | Feedstock for creating nitrified digestate or compost teas. Worm castings are a high-quality source [46]. |
| Hydroponic System (DWC, NFT) | Provides the platform for plant production in a soil-less, controlled environment, allowing precise nutrient delivery and recycling. | The endpoint for the integrated system; where recovered nutrients are validated for crop production [48]. |
Data Presentation and Analysis
Performance Comparison of Alternative Nutrient Solutions
Evaluating the efficacy of waste-derived nutrient solutions against conventional synthetic fertilizers is a critical step in research.
Table 3: Quantitative Comparison of Nutrient Solutions in Hydroponic Lettuce Production
| Nutrient Solution Type | Key Characteristic | Relative Yield (%) (vs. Control) | Nitrogen Use Efficiency (NUE) | Key Challenges & Notes |
|---|---|---|---|---|
| Synthetic Control (e.g., Hoagland) | Balanced NO₃⁻/NH₄⁺ mix [47] | 100% (Baseline) | Baseline | Standard for comparison; relies on Earth-sourced minerals. |
| Nitrified Digestate | Derived from aerated biogas waste [45] | Data Not Specified | 10x improvement with advanced pH control [45] | High salt content; requires tailored control strategies (e.g., pH-based) [45]. |
| Worm Casting Tea (WCT 2:3) | Liquid extract from vermicompost [46] | ~80% [46] | Comparable to synthetic control [46] | Yield 20% lower, but a promising stand-alone organic option [46]. |
| Other Compost Teas (e.g., InVCT 1:4) | Liquid extract from thermophilic compost [46] | Lower than WCT | Lower than WCT | Often limited by high ammonium and total dissolved solids [46]. |
| Direct Organic Nitrogen (e.g., Blood Meal) | Complex organic molecules [43] | <50% [43] | Very Low | Plants cannot efficiently uptake complex organic nitrogen; causes severe deficiencies [43]. |
The seamless integration of waste processing and hydroponic production is fundamental to achieving sustainable life support for long-duration space missions. The core challenge lies in the efficient bioconversion of organic nitrogen from waste streams into plant-available nitrate, a process that can be optimized through well-designed system architectures like the MELiSSA loop and precise experimental protocols. While solutions like nitrified digestate and high-quality compost teas show significant promise, yielding up to 80% of conventional production with greatly enhanced resource efficiency, challenges remain in the consistent management of nutrient profiles and the removal of excess salts. Future research must focus on developing more robust sensors, adaptive control algorithms, and hardening these integrated biological-physicochemical systems for the unique constraints of the space environment, including microgravity and limited volume. Success in this endeavor will not only enable deep space exploration but also provide transformative solutions for sustainable food production on Earth.
Challenges, Efficiency Analysis, and System Optimization Strategies
Nutrient Balance (NB) and Nutrient Use Efficiency (NUE) as Key Performance Indicators
In the context of regenerative life support systems (RLSS) for long-duration space missions, the management of nitrogen (N) and other nutrients is a critical determinant of system stability and success. These systems aim to achieve a high degree of closure by recycling resources, with a particular focus on recovering nutrients from waste streams to sustain food production [3] [49]. Within this framework, Nutrient Balance (NB) and Nutrient Use Efficiency (NUE) emerge as two indispensable Key Performance Indicators (KPIs). NB provides a mass-balance account of nutrient inputs, internal pools, and outputs, quantifying the closure of nutrient loops. Concurrently, NUE measures the effectiveness with which a system converts available nutrients into biomass, directly impacting the productivity and resupply mass requirements of the system [50]. The integration of these KPIs offers a holistic view of system performance, assessing not just the efficiency of nutrient use but also the overall sustainability and resilience of the nutrient cycle—a non-negotiable requirement for environments isolated from Earth, such as a lunar base or a spacecraft on a Mars transit mission [51] [3].
The imperative for robust NB and NUE metrics is driven by the extreme economic and logistical constraints of space travel. The cost of shipping supplies from Earth is prohibitive, estimated at approximately $10,000 per kilogram to low Earth orbit and $300,000 per kilogram to Mars [51] [3]. Furthermore, a crewed mission to Mars lasting three years would require thousands of kilograms of food and water per crew member if reliant solely on resupply [3]. Bioregenerative Life Support Systems (BLSS) present a viable alternative to purely physicochemical systems by using biological processes to regenerate air, water, and food from waste. A core function of a BLSS is the recycling of nitrogen, a fundamental component of amino acids, proteins, and nucleic acids [52] [3]. Human urine is the most significant waste stream for nitrogen recovery, containing nearly 80-90% of the nitrogen excreted by humans and representing the largest source of recoverable nitrogen in a closed habitat [49]. Therefore, achieving a favorable NB through high-efficiency recycling of urine nitrogen and optimizing the NUE of crop plants are paramount research objectives for the development of self-sustaining RLSS [51] [3] [49].
Defining the Core Key Performance Indicators
Quantitative Definitions and Formulas
For precise monitoring and control, NB and NUE must be defined by quantifiable formulas. These KPIs can be applied at different scales within an RLSS, from a single plant growth chamber to the entire system habitat.
Nutrient Balance (NB) is fundamentally based on the mass conservation principle. It is calculated as the difference between the total nutrient inputs into a defined system and the total nutrient outputs from that system over a specific time period. A simplified representation is:
NB = Total Nutrient Inputs - Total Nutrient Outputs
Where inputs include all external supplies and initially loaded nutrient stocks, while outputs encompass harvested biomass, any waste vented or stored, and other losses. In an ideal, perfectly closed system, the NB would be zero, indicating that all inputs are accounted for in the outputs with no accumulation or unexplained loss. In practice, a positive NB may indicate accumulation within the system (e.g., in soil or biomass), while a negative NB signals a depletion of nutrient stocks [50]. For nitrogen, tracking the N Surplus is a common approach, which is directly derived from the NB concept and is calculated as N input - N output [50].
Nutrient Use Efficiency (NUE) is a ratio of output to input. The generic formula is:
NUE = (Nutrient Output in Harvested Product) / (Nutrient Input)
For nitrogen in plant production systems, this is often expressed as NUE = Yield (kg) / N Applied (kg) [50]. However, this core concept can be broken down into more specific agronomic efficiency indices that provide deeper insights, as detailed in Table 1.
Table 1: Key Agronomic Efficiency Indices for Nitrogen Use Efficiency [53] [54] [50]
| Efficiency Index | Definition | Formula |
|---|---|---|
| Partial Factor Productivity (PFP) | Economic yield obtained per unit of nutrient applied. | PFP = (Yield_fert) / (Nutrient Applied) |
| Agronomic Efficiency (AE) | Economic yield increase per unit of nutrient applied. | AE = (Yieldfert - Yieldunfert) / (Nutrient Applied) |
| Apparent Recovery Efficiency (ARE) | Quantity of nutrient absorbed per unit of nutrient applied. | ARE = (Uptakefert - Uptakeunfert) / (Nutrient Applied) |
| Physiological Efficiency (PE) | Biological yield produced per unit of nutrient absorbed. | PE = (Yieldfert - Yieldunfert) / (Uptakefert - Uptakeunfert) |
Interrelationship of NB and NUE in a Closed System
NB and NUE are deeply interconnected KPIs. In a closed RLSS, a high NUE directly supports a tighter NB. When plants efficiently convert absorbed nutrients into edible biomass, less nutrient mass is required as input for a given output, reducing the pressure on the upstream waste recycling subsystems and minimizing the system's total nutrient inventory [52] [49]. Conversely, a system with a low NUE requires greater nutrient inputs to achieve the same yield, leading to a larger N Surplus. This surplus represents nitrogen that is not converted to food and is instead susceptible to loss through pathways like denitrification or leaching, thereby disrupting the NB and potentially causing pollution within the closed environment [52] [55]. Therefore, the simultaneous optimization of both KPIs is essential for a stable, productive, and sustainable RLSS. The relationship between input, output, and efficiency can be visualized as a functional framework for system management.
Methodologies for Monitoring and Analysis
Accurate measurement of NB and NUE relies on a combination of well-established chemical, molecular, and sensor-based techniques. The following protocols detail the key methodologies.
Protocol for Establishing System-Level Nitrogen Balance
This protocol outlines the procedure for quantifying the NB for nitrogen across an entire RLSS or a major subsystem.
- System Boundary Definition: Precisely define the physical and functional boundaries of the system under study (e.g., the entire habitat, the plant growth module, or a specific soil bioreactor).
- Inventory and Mass Measurement:
- Inputs: Accurately mass all nitrogen inputs entering the boundary during the study period. This includes initial nutrient solutions, recycled fertilizers (e.g., processed urine, compost), nitrogen-fixing inoculants, and any food or biomass imported.
- Internal Pools: Measure the nitrogen mass in key system pools at the beginning and end of the study. This includes soil or growth substrate, plant biomass (both edible and inedible parts), microbial biomass, and liquid reservoirs.
- Outputs: Mass all nitrogen outputs leaving the boundary. This includes harvested edible biomass, removed inedible biomass, any liquid or gaseous waste vented from the system, and leachate.
- Chemical Analysis:
- For solid samples (biomass, soil), use Kjeldahl digestion (or Dumas combustion) to determine total nitrogen content.
- For liquid samples (urine, nutrient solutions, leachate), analyze for ammonium (NH₄⁺), nitrate (NO₃⁻), and urea concentrations using standardized colorimetric methods (e.g., spectrophotometry).
- Data Calculation and Reconciliation:
- Calculate total N input (ΣNinput).
- Calculate total N output (ΣNoutput).
- Calculate the change in internal N pools (ΔNpools).
- Compute the system NB: NB = ΣNinput - ΣNoutput. A more complete mass balance would be: ΣNinput - ΣNoutput = ΔNpools. A significant imbalance indicates unaccounted losses (e.g., gaseous emissions) or accumulation.
Protocol for Quantifying Terrestrial NUE in Plant Growth Systems
This protocol measures the NUE of a crop within an RLSS plant growth unit, based on agronomic standards [53] [54].
- Experimental Design: Establish replicated treatments with defined nitrogen application rates. Include a control treatment with no nitrogen application to establish baseline yield and uptake.
- Plant Cultivation and Harvest: Grow the crop to maturity using standardized cultivation practices. At harvest, separately collect the economic yield (e.g., grains, fruits) and the total above-ground biomass from a defined area.
- Biomass and Yield Measurement:
- Oven-dry the biomass and yield samples at 70°C until constant weight.
- Record the dry weight of the economic yield and total biomass.
- Plant Tissue Nitrogen Analysis:
- Grind the dried plant material to a fine powder.
- Determine the nitrogen concentration in the powder using an elemental analyzer (Dumas combustion method) or via Kjeldahl digestion followed by titration/colorimetry.
- Calculation of NUE Indices:
- Nutrient Uptake (kg/ha) = (Plant N concentration (%) × Dry biomass weight (kg/ha)) / 100
- Use the formulas provided in Table 1 to calculate PFP, AE, ARE, and PE.
Molecular Analysis of the Nitrogen-Cycling Microbiome
The microbial community is the engine of nitrogen transformations in soil-based RLSS. Monitoring its composition and function is crucial for understanding NB. Metagenomic and metatranscriptomic analyses provide deep insight [56] [55] [57].
- Sample Collection: Aseptically collect soil, sediment, or biofilm samples from critical locations (e.g., rhizosphere, urine processing bioreactor). Immediately preserve samples in RNAlater for transcriptomic studies or flash-freeze in liquid N₂ for DNA analysis. Store at -80°C.
- Nucleic Acid Extraction: Extract total genomic DNA and/or RNA using commercial kits optimized for soil/microbial biomass (e.g., PowerSoil DNA Isolation Kit). For RNA, perform DNase treatment to remove genomic DNA contamination.
- Quantitative Analysis (qPCR/ddPCR):
- Shotgun Metagenomic Sequencing:
- Fragment purified DNA and construct sequencing libraries.
- Sequence using an Illumina or similar platform to generate high-throughput short reads.
- Bioinformatically assemble reads and map them to databases of nitrogen-cycling genes to determine the metabolic potential of the community [56].
- Metatranscriptomic Sequencing:
- Enrich mRNA from total RNA, prepare libraries, and sequence.
- Map sequences to a reference genome or metagenome-assembled genomes (MAGs) to identify and quantify actively transcribed genes, providing a real-time view of microbial activity [57].
Table 2: Key Molecular Markers for Monitoring Nitrogen-Cycling Microbes [56] [55] [57]
| Target Gene | Function / Process | Utility as a Biomarker |
|---|---|---|
| nifH | Nitrogenase / Nitrogen Fixation | Measures potential for biological N input. Generally unresponsive to N fertilization [55]. |
| amoA | Ammonia Monooxygenase / Nitrification | Quantifies ammonia-oxidizing archaea (AOA) and bacteria (AOB). AOB abundance increases dramatically (e.g., 313%) with N input [55]. |
| nxr | Nitrite Oxidoreductase / Nitrite Oxidation | Indicates presence of nitrite-oxidizing bacteria (e.g., Nitrospira). |
| nirK, nirS | Nitrite Reductase / Denitrification | Measures potential for NO₂⁻ reduction to NO. Abundance increases (~50%) with N fertilization [55]. |
| nosZ | Nitrous Oxide Reductase / Denitrification | Measures potential for N₂O reduction to N₂. A key indicator for completing denitrification and mitigating greenhouse gas emissions. Abundance increases (~75%) with N fertilization [55]. |
| hzs | Hydrazine Synthase / Anammox | Indicates potential for anaerobic ammonium oxidation, a key N-loss pathway in anoxic niches [57]. |
The workflow for a comprehensive molecular analysis, from sample to insight, is summarized below.
The Scientist's Toolkit: Key Reagents and Materials
Table 3: Essential Research Reagents and Kits for NB/NUE Analysis
| Item | Function / Application | Example Use Case |
|---|---|---|
| PowerSoil DNA/RNA Isolation Kit | Simultaneous or separate extraction of high-quality genomic DNA and total RNA from complex environmental samples. | Extracting nucleic acids from soil or biofilm samples for qPCR and metagenomics [56]. |
| RNAlater Stabilization Solution | Stabilizes and protects cellular RNA in fresh tissue samples immediately upon collection, preventing degradation. | Preserving microbial transcriptomes in field-collected aquifer or soil samples during transport [57]. |
| Droplet Digital PCR (ddPCR) System | Absolute quantification of target DNA or cDNA sequences without the need for a standard curve, offering high precision. | Quantifying the abundance of low-copy nitrogen-cycling genes like hzsB (anammox) or nosZ [57]. |
| Elemental Analyzer | Determines the total nitrogen and carbon content in solid samples (e.g., plant tissue, soil) via Dumas combustion. | Measuring nitrogen concentration in harvested plant biomass for NUE calculations [53]. |
| Spectrophotometer / Flow Analyzer | Measures concentration of specific nitrogen species (NH₄⁺, NO₃⁻, NO₂⁻, Urea) in liquid samples via colorimetric assays. | Analyzing nutrient solutions, urine, and leachate for nitrogen mass balance calculations [51]. |
| gBlock Gene Fragments | Synthetic double-stranded DNA fragments used as positive controls and absolute standards in qPCR/ddPCR assays. | Creating standard curves for functional gene quantification (e.g., amoA, nifH) [57]. |
Application in Life Support: Case Studies and Data
The theoretical frameworks for NB and NUE are being validated through advanced research into BLSS. The following case studies and synthesized data illustrate their practical application.
Case Study: Urine-Fuelled Soil-Based Bioregenerative System
A computational study investigated a closed-loop system where crew urine was used to supply primary (N, P, K) and secondary (S, Ca, Mg) nutrients to wheat and soybean plants [51].
- Methodology: An advanced computational tool (BRTSim) was used to model the biogeochemistry of a soil-based cropping unit in microgravity over a 20-year period. The model tracked the breakdown of urine compounds, nutrient uptake, and the production of byproduct gases.
- Key Findings:
- Human urine could satisfy the demand for at least 3 to 4 out of the 6 essential nutrients for the test crops.
- The resulting offset in soil pH and salinity was within a range tolerable by the plants.
- The system demonstrated the potential for long-term sustainability, reducing the need for external resupply of fertilizers.
- Implication for KPIs: This study directly demonstrates the potential for achieving a tight Nitrogen Balance by closing the nutrient loop between human waste and food production, a core principle for RLSS [51].
Synthesized Data: NUE in Wheat and Microbial Responses
Empirical data from terrestrial agricultural research provides benchmarks and insights relevant to RLSS crop production.
- NUE in Wheat Cultivation: A two-year field study in Nepal determined the optimal nutrient rates for wheat, finding that grain yields were maximized at 125 kg N ha⁻¹ and 50 kg K₂O ha⁻¹, yielding 6.33 and 6.30 t ha⁻¹, respectively [53]. The study concluded that NUE was highest at lower doses of the respective nutrients, and that balanced application (N @ 125 kg ha⁻¹, P₂O₅ @ 25 kg ha⁻¹, K₂O @ 50 kg ha⁻¹) was optimal for efficient nutrient management. This highlights the importance of precision fertilization for maximizing NUE.
- Microbial Community Response to N: A meta-analysis of 47 field studies revealed that nitrogen fertilization significantly alters the abundance of key nitrogen-cycling genes [55]. As shown in Table 4, genes involved in nitrification and denitrification become more abundant, while nitrogen fixation genes remain unaffected. This demonstrates that the system's NB directly shapes its microbial functional potential, with implications for nutrient retention and loss.
Table 4: Meta-Analysis of N Fertilization Impact on N-Cycling Gene Abundance in Agricultural Soils [55]
| Functional Gene | Representative Microbes | Process | Average Change in Abundance |
|---|---|---|---|
| nifH | Nitrogen-fixing bacteria (e.g., Rhizobium) | Nitrogen Fixation | No significant effect |
| amoA (AOA) | Ammonia-Oxidizing Archaea | Nitrification | +31.1% |
| amoA (AOB) | Ammonia-Oxidizing Bacteria | Nitrification | +313% |
| nirK / nirS | Denitrifying bacteria | Denitrification (NO₂⁻ to NO) | +53.3% / +39.7% |
| nosZ | Denitrifying bacteria | Denitrification (N₂O to N₂) | +75.1% |
In regenerative life support systems (RLSS), such as those required for long-duration space missions, the efficient management of sodium chloride (NaCl) and overall salinity is critical for system stability. The inability to effectively remove salts can disrupt core bioprocesses, most notably nitrogen cycling, which is essential for converting waste streams into resources like air, water, and food [3] [29]. In the context of a Bioregenerative Life Support System (BLSS), salinity control is not merely a water purification task; it is a fundamental prerequisite for maintaining the microbial and plant-based processes that enable crew survival. The European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) project, a leading BLSS initiative, highlights the necessity of highly efficient mineral recovery from waste streams to produce nutrients for food cultivation [3]. Failure to manage NaCl and other salts jeopardizes this closed-loop cycle, leading to system toxicity and potential failure. This guide details the technical approaches for mitigating salinity within the specific constraints of RLSS.
NaCl and Salinity Control Technologies
Selecting the appropriate technology for NaCl removal in an RLSS depends on the specific wastewater stream, the desired water recovery rate, and the integration points with nitrogen cycle bioprocesses.
Technology Performance Comparison
The following table summarizes key performance metrics for advanced desalination technologies relevant to RLSS applications.
Table 1: Performance comparison of NaCl removal technologies for high-recovery desalination.
| Technology | Key Mechanism | Max Water Recovery | Specific Energy Consumption | Key Advantages | Integration Considerations for Nitrogen Cycling |
|---|---|---|---|---|---|
| Modified Electrodialysis Metathesis (mEDM) [58] | Electrochemical separation with monovalent-selective ion-exchange membranes. | Up to 95% | 1.1 - 1.7 kWh/m³ | Chemical-free operation; superior scaling prevention; can treat reverse osmosis retentate. | Purified water stream can be recycled; concentrated brine requires downstream processing. |
| Reverse Osmosis (RO) [58] | Pressure-driven separation through semi-permeable membranes. | ~80% (can be extended to 92% with mEDM polish) | Typically higher than mEDM | Established technology; effective salt rejection. | Membrane scaling limits recovery; pretreatment chemicals may affect downstream microbiology. |
| Microbial Desalination Cells | Bioelectrochemical desalination using microbial metabolic energy. | Under research | Potentially lower energy | Simultaneous organic pollutant removal and desalination; energy-producing. | Emerging technology; can be integrated with nitrogen-removing biocatalysts; desalination rate may be low. |
Detailed Experimental Protocol: Modified Electrodialysis Metathesis (mEDM)
The mEDM process represents a significant advance for high-recovery, chemical-free desalination in systems where reliability is paramount [58].
1. Principle: mEDM incorporates monovalent-selective ion exchange membranes (mIEMs) to prevent scaling by strategically separating divalent cations (e.g., Ca²⁺, Mg²⁺) and anions (e.g., SO₄²⁻). This separation prevents their meeting and exceeding solubility products, thereby avoiding scale formation. The process relies on a feedwater with sufficient NaCl, which participates in an ion-recombination mechanism that facilitates the removal of problematic divalent salts [58].
2. Materials and Setup:
- mEDM Stack: Configured with alternating monovalent-selective cation-exchange membranes (MV-CEMs) and anion-exchange membranes (AEMs).
- Feed Solution: NaCl-MgSO₄ solutions in a 4:1 molar ratio, or authentic brackish groundwater, with a total dissolved solids (TDS) of approximately 3-3.5 g/L [58].
- Power Supply: Direct current (DC) power source.
- Analytical Equipment: Conductivity meter, ion chromatography system.
3. Step-by-Step Methodology: 1. System Assembly: Arrange the mEDM stack with mIEMs to create dedicated channels for concentrate (divalent-rich) and diluate (product water). 2. Feed Introduction: Pump the saline feedwater through the diluate cells. 3. Applied Potential: Apply a DC electric field across the stack. This drives Na⁺ and Cl⁻ ions through their respective selective membranes, while the mIEMs block a significant fraction of divalent ions. 4. Ion Recombination: Divalent ions are redirected into separate concentrate streams, where they recombine into non-scaling salt pairs. 5. Performance Monitoring: Track the diluate conductivity to monitor desalination efficiency. Measure specific energy consumption (SEC) and calculate gross water recovery (GWR) using flow rates and salt balances. System performance is validated by operating at recoveries >90% without observable scaling, unlike RO and traditional ED [58].
Salinity Impacts on Nitrogen Cycle Bioprocesses
In a BLSS, nitrogen recovery is a cornerstone function, primarily achieved by a consortium of microorganisms. High salinity, specifically NaCl concentration, imposes osmotic stress and ionic toxicity on these microbes, disrupting enzyme activity and plasma membrane function [59]. This can lead to a critical failure in converting ammonia to nitrate or reclaiming nitrogen from urine.
Protocol: Assessing Salinity Tolerance of Nitrogen-Cycling Microbes
Understanding the resilience of essential microorganisms is vital for system design and contingency planning.
1. Principle: This protocol tests the reactivation potential and metabolic activity of key nitrogen-cycling microorganisms after exposure to preservative conditions, including those simulating spaceflight (microgravity and elevated radiation) [10].
2. Materials:
- Microbial Cultures: Axenic or defined communities of ureolytic bacteria (e.g., Cupriavidus pinatubonensis), ammonia-oxidizing bacteria (AOB, e.g., Nitrosomonas europaea), nitrite-oxidizing bacteria (NOB, e.g., Nitrobacter winogradskyi), and anammox bacteria [10].
- Growth Media: Specific liquid media for each functional group (e.g., containing urea, NH₄⁺, or NO₂⁻ as substrates).
- Storage Conditions: Facilities for refrigerated (4 °C) and ambient (23 °C) ground storage, and access to spaceflight exposure platforms (e.g., FOTON-M4 satellite) [10].
- Analytical: Spectrophotometer, ion chromatography or colorimetric kits for NH₄⁺, NO₂⁻, NO₃⁻ quantification.
3. Step-by-Step Methodology: 1. Preservation: Preserve microbial cultures under different conditions: Ground Control at 4°C (G4), Ground Control at 23°C (G23), and Flight exposure in Low Earth Orbit (F). 2. Reactivation: After the preservation period (e.g., 44 days), inoculate samples into fresh, specific growth media. 3. Activity Measurement: Monitor the depletion of substrates (e.g., urea, NH₄⁺) and production of products (e.g., NO₂⁻, NO₃⁻, N₂) over time. 4. Data Analysis: Calculate volumetric conversion rates (e.g., mg N/L/day) for each functionality (ureolysis, nitritation, nitratation, anammox). Compare rates between F, G23, and G4 conditions to determine the impact of preservation environment [10].
4. Key Findings: A landmark study demonstrated that all major nitrogen cycle functionalities can be reactivated after exposure to space conditions, with rates often similar to or higher than terrestrial preservation at a similar temperature. This confirms the functional resilience of these microbial communities, a crucial insight for designing reliable BLSS [10].
Nitrogen Cycle Pathway in a BLSS
The following diagram illustrates the interconnected microbial processes responsible for nitrogen recycling in a system like MELiSSA, and the critical points where salinity can cause toxicity.
Diagram 1: Nitrogen cycle pathway and salinity stress impact. The diagram shows the key microbial conversion steps (solid lines) in nitrogen recycling and the points where high salinity imposes critical stresses (dashed lines) that can disrupt the entire cycle.
The Scientist's Toolkit
Table 2: Essential research reagents and materials for RLSS salinity and nitrogen cycling research.
| Item | Function/Application |
|---|---|
| Monovalent-Selective Ion Exchange Membranes [58] | Core component in mEDM systems for selective separation of Na⁺ and Cl⁻ from divalent ions, preventing scaling. |
| Defined Microbial Consortia (e.g., Nitrosomonas europaea, Nitrobacter winogradskyi) [10] | For studying the specific effects of salinity on nitrification kinetics and community stability. |
| Salt-Tolerant Halophilic Microbial Strains [59] | Investigated for their potential to perform biological treatment (e.g., organic pollutant removal) in high-salinity brines. |
| Urea & Ammonium Salts (e.g., NH₄Cl, (NH₄)₂SO₄) [10] | Essential substrates for cultivating and testing the activity of ureolytic and ammonia-oxidizing microorganisms. |
| Specific Ion Electrodes/Probes (for NH₄⁺, NO₃⁻, Na⁺, Cl⁻) | Real-time monitoring of nitrogen species and salt ions in bioreactor and desalination experiments. |
| Synthetic Brackish Water/Urine Simulants [58] [3] | Standardized feed solutions for testing desalination technologies and nitrogen recovery processes under controlled conditions. |
Effective control of sodium chloride and salinity is not a peripheral utility but a central pillar for ensuring the stability of nitrogen cycling and overall functionality of regenerative life support systems. The integration of advanced, scaling-resistant physicochemical technologies like mEDM with resilient, well-characterized microbial communities provides a robust strategy for mitigating system toxicity. Future research must focus on the intelligent coupling of these systems—for instance, developing halophilic nitrogen cycle bacteria that can process concentrated brines produced by desalination units. This synergistic approach is essential for achieving the closed-loop resource recovery required to sustain human life in long-duration space exploration and other isolated, critical environments.
The development of robust Bioregenerative Life Support Systems (BLSS) is paramount for long-duration human space exploration, moving beyond the limited capabilities of current physicochemical systems. Effective nitrogen cycle closure is a critical cornerstone of this endeavor, enabling the in-situ production of food and the recycling of water and oxygen. This whitepaper details the principal operational hurdles—scaling bioreactors, managing energy demands, and ensuring process stability—that must be overcome to achieve this goal. We provide a technical analysis of these challenges, supported by quantitative data, detailed experimental protocols for key processes, and visualization of system workflows, offering a roadmap for researchers and engineers in the field.
Current Environmental Control and Life Support Systems (ECLSS) on platforms like the International Space Station rely heavily on physicochemical processes and resupply from Earth. They recover water and oxygen but are unable to produce food or achieve full nutrient recycling, making them unsuitable for long-distance missions [3]. A transition to Bioregenerative Life Support Systems (BLSS) is essential, as these systems aim to combine biological and physicochemical processes to create a closed-loop ecosystem. Within this system, nitrogen is a fundamental element, crucial for the synthesis of dietary protein for the crew and for the health of the photosynthetic components that produce oxygen and food.
Urine is the most significant source of recoverable nitrogen in a BLSS, accounting for approximately 85% of the total nitrogen waste stream, with an average daily excretion of 7–16 grams of Nitrogen per crew member [3]. The inability to efficiently recover and recycle this nitrogen represents a major logistical and mass penalty for missions where every kilogram of payload is cost-prohibitive. Therefore, the development of highly efficient, stable, and scalable reactor systems for nitrogen conversion is not merely an engineering challenge but a prerequisite for self-sufficient human presence in space.
Core Operational Hurdles in Nitrogen Cycle Management
Scaling Bioreactors: From Microcosm to Macro-System
Scaling biological processes from laboratory benchtops to integrated systems capable of supporting a crew presents a fundamental challenge. The goal is to reproduce the performance of a target nitrogen conversion process in a larger, and often differently configured, reactor system.
- The Scaling Problem: The core issue lies in the non-linear relationships between system parameters when size changes. As a reactor's volume increases by the cube of its radius, the heat-transfer area increases only by the square, creating a significant challenge for thermal management, especially for temperature-sensitive biological processes [60]. This can lead to scaling distortions, where a process that is stable at a small scale behaves unpredictably in a larger one due to factors like temperature gradients, inadequate mixing, or altered microbial ecology [61].
- Established Scaling Methodologies: Several methodologies have been developed to address this in complex systems, which can be adapted for BLSS reactor design:
- Power-to-Volume Scaling: A common approach for integral test facilities that aims to maintain a constant power-to-volume ratio between the model and the prototype [61].
- Hierarchical Two-Tiered Scaling (H2TS): This method considers different tiers of system functions, from the mission level down to the component level, ensuring that critical processes are scaled appropriately [61].
- Fractional Scaling Analysis (FSA): Used to analyze the scaling of individual phenomena, helping to identify and quantify potential distortions [61].
Table 1: Key Scaling Parameters for BLSS Nitrogen Recovery Reactors
| Parameter | Laboratory Scale (1L) | Pilot Scale (100L) | Scaling Consideration |
|---|---|---|---|
| Volume | 1 L | 100 L | Linear scale-up factor: 100x |
| Heat Transfer Area | ~0.01 m² | ~1 m² | Scale-up factor: ~100x (Area ∝ r²) |
| Power/Volume (for mixing) | 1 W/L | 1 W/L | Constant ratio required, but power input becomes 100W. |
| Thermal Mass | Low | High | Larger systems resist temperature changes, affecting control. |
| Mixing Time | Seconds | Minutes | Increased mixing time can create substrate gradients. |
| Gravity Effects | Negligible | Potentially Significant | Phase separation (gas/liquid) can be impacted in microgravity. |
Energy Demands and Thermal Management
Biological nitrogen conversion processes are exothermic, and the energy balance of the system is a critical factor in reactor stability and control.
- Process Energy Balance: A comprehensive energy balance must account for the heat generated by the desired biological reactions (e.g., nitrification), the heat removal capacity of the reactor system, and the thermal stability of the reaction mixture. An imbalance can lead to thermal runaway, a situation where heat generation exceeds heat removal, leading to a rapid, uncontrolled temperature increase that can halt biological activity or even damage the system [60].
- Safer Process Design: To mitigate this, reactions should be designed to occur fairly rapidly, and semi-batch processes are recommended for exothermic reactions. In a semi-batch mode, reagents are added controllably, allowing the reaction rate and heat release to be managed, rather than having all chemical energy present at the onset as in a batch process [60]. Furthermore, temperature control alone should not be the only means for limiting the reaction rate for highly exothermic reactions; engineering controls on reagent addition rates are essential.
Process Stability and Control
Ensuring the long-term, predictable operation of nitrogen recycling bioreactors is a multi-faceted challenge involving biological, chemical, and control engineering.
- Feedback Mechanisms and Dynamics: Like nuclear reactors, biological reactors have inherent feedback loops. A key stabilizing mechanism in BLSS reactors is the negative temperature coefficient, where an increase in temperature decreases microbial activity, thus reducing heat generation. However, positive feedback loops, such as those driven by substrate inhibition or product toxicity, can be destabilizing and must be understood and controlled [62].
- Impact of Space Environment: Microgravity and increased ionizing radiation may alter microbial behavior, including the performance and stability of nitrifying bacteria essential for nitrogen recovery [3]. Ground-based simulations and eventual in-situ testing are necessary to characterize these effects and design robust systems that can maintain stability under space conditions.
Experimental Protocols for Key Nitrogen Cycle Processes
To ground the theoretical challenges in practical science, below are detailed protocols for two critical processes in nitrogen cycling.
Protocol: Quantifying Heat of Reaction and Off-Gas Rates for Nitrification
This protocol is essential for understanding the energetics and gas exchange of a key nitrogen conversion process, directly informing reactor scaling and safety.
- 1. Objective: To experimentally determine the heat of reaction and the rate of carbon dioxide (CO₂) off-gassing for the microbial nitrification process (conversion of ammonium, NH₄⁺, to nitrate, NO₃⁻) in a simulated BLSS wastewater stream.
- 2. Materials:
- Reaction Calorimeter (RC)
- Bioreactor vessel with pH, dissolved oxygen (DO), and temperature control
- Nitrifying microbial inoculum (e.g., from Nitrosomonas and Nitrobacter genera)
- Synthetic wastewater feed containing ammonium salts (e.g., (NH₄)₂SO₄)
- In-line Gas Analyzer (for CO₂, O₂)
- Data acquisition system
- 3. Methodology:
- Calibration: Calibrate the reaction calorimeter and all sensors according to manufacturer specifications. Perform a heat capacity calibration of the system using a known electrical heat input.
- Inoculation and Baseline: Charge the bioreactor with a known volume of the nitrifying inoculum. Establish baseline conditions (temperature, pH, agitation) and monitor until a stable thermal baseline is achieved.
- Semi-Batch Reaction: Initiate a continuous or controlled semi-batch addition of the synthetic wastewater feed to the reactor.
- Data Collection:
- Continuously record the heat flow (Q) required to maintain the setpoint temperature. The integral of the heat flow curve over time gives the total heat of reaction (ΔHᵣₓₙ).
- Simultaneously, monitor the off-gas composition and flow rate to calculate the real-time CO₂ evolution rate (CER).
- Track ammonium and nitrate concentrations via periodic sampling and analysis (e.g., ion chromatography) to correlate thermal and gas data with conversion efficiency.
- Worst-Case Scenario: Repeat the experiment at the maximum possible operating temperature to assess the thermal stability of the reaction mixture and identify any adverse exotherms [60].
Protocol: Adiabatic Calorimetry for Thermal Stability Assessment of Waste Streams
This protocol identifies and characterizes potential thermal runaway reactions in stored or processing waste streams.
- 1. Objective: To assess the thermal stability of a BLSS waste stream (e.g., stabilized urine) and characterize the kinetics of any decomposition reactions under adiabatic conditions.
- 2. Materials:
- Advanced Reactive System Screening Tool (ARSST) or Vent Sizing Package 2 (VSP2)
- Sample of chemically stabilized urine (e.g., with H₃PO₄ and Cr⁶⁺ as on the ISS [3])
- Sample holder
- 3. Methodology:
- Sample Preparation: Load a representative sample (~10-50 mL for ARSST) of the test waste stream into the sample holder.
- Test Initiation: Place the sample holder in the adiabatic calorimeter and initiate a heat-wait-search cycle. The instrument maintains adiabatic conditions (no heat loss) while searching for a self-sustaining exothermic reaction.
- Data Collection: Upon detection of an exotherm, the instrument tracks the temperature and pressure rise as a function of time. Key data collected includes:
- Onset temperature of decomposition (Tₒₙₛₑₜ)
- Maximum self-heat rate (dT/dt)ₘₐₓ
- Adiabatic temperature rise (ΔTₐₕ)
- Time-to-maximum rate (TMR)
- Data Application: The generated data is directly scalable and used for:
- Defining safe operating temperature limits.
- Designing emergency relief systems for process vessels using DIERS methodology [60].
- Understanding the energy potential of worst-case scenario reactions.
Visualization: The MELiSSA Loop and Nitrogen Recovery Workflow
The European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) project is a leading example of a BLSS designed to address these hurdles. Its nitrogen recovery compartment can be visualized as follows:
Diagram 1: Nitrogen Recovery in the MELiSSA Loop. This diagram illustrates the closed-loop pathway of nitrogen from crew waste to food production, highlighting the critical role of Compartment III, the nitrifying bioreactor where operational hurdles are most acute.
The logical workflow for the experimental characterization of these reactor systems is outlined below:
Diagram 2: Reactor Characterization and Scaling Workflow. This flowchart details the systematic approach to characterizing a nitrogen conversion process and using the data to inform the scaled reactor design and its safety controls.
The Scientist's Toolkit: Essential Reagents and Materials
Table 2: Key Research Reagent Solutions for Nitrogen Cycling Research
| Item / Reagent | Function in Research | Example Application |
|---|---|---|
| Stable Isotope Tracers (e.g., ¹⁵N-labeled Ammonium/Nitrate) | To trace the pathway and fate of nitrogen through different biological and chemical pools. | Quantifying nitrogen uptake preferences in plants [63] or partitioning N₂O production from different microbial processes in soils [63]. |
| Nitrifying Bacterial Consortia (e.g., Nitrosomonas, Nitrobacter) | The biological agents responsible for the aerobic conversion of ammonia to nitrite and then to nitrate. | Inoculating Compartment III of the MELiSSA loop to recover nitrogen from pretreated urine [3]. |
| Chemical Stabilization Agents (e.g., H₃PO₄, Cr⁶+) | Prevents urea hydrolysis and scaling in urine collection and storage systems, stabilizing the waste stream for processing. | Used in the ISS Urine Processor Assembly to allow for efficient water distillation [3]. |
| Synthetic Wastewater Feed | A defined, reproducible medium simulating human waste for controlled experimentation. | Used in bench-scale reactors to test and optimize nitrogen conversion efficiency without the variability of real waste. |
| Adiabatic Calorimeter (e.g., ARSST, VSP2) | Experimental apparatus to study thermal runaway reactions under no heat loss conditions. | Characterizing the thermal stability of waste streams and providing data for emergency relief system design [60]. |
| Reaction Calorimeter (RC) | Measures heat flow and kinetics of chemical or biological reactions in a controlled reactor vessel. | Quantifying the heat of reaction and heat removal requirements for the nitrification process [60]. |
Overcoming the operational hurdles of scaling reactors, energy demands, and process stability is not an isolated engineering challenge but the central path to achieving closed-loop nitrogen cycling in Bioregenerative Life Support Systems. The integration of rigorous scaling methodologies, comprehensive thermal hazard assessments, and robust experimental protocols—as exemplified by programs like MELiSSA—provides a structured framework for progress. Success in this endeavor will hinge on a multidisciplinary approach that merges deep process chemistry understanding with advanced engineering design and control strategies. The solutions developed will not only enable long-term human exploration of space but also contribute to the advancement of closed-loop resource recovery technologies on Earth.
In the context of Bioregenerative Life Support Systems (BLSS) for long-duration space missions, the efficient recycling of nitrogen is a critical technological challenge. Microbial processes are the engine that drives this recycling, converting waste nitrogen into forms usable for food production. However, the space environment, characterized by microgravity and elevated radiation, poses a unique set of challenges to these essential microorganisms. Understanding their adaptation and survival is not merely an academic exercise but a prerequisite for designing reliable, self-sustaining life support for missions to Mars and beyond. This whitepaper synthesizes current research on how space conditions affect microbial physiology, with a specific focus on implications for nitrogen cycling within BLSS.
Key Experimental Evidence from Spaceflight
Ground-based simulations provide valuable insights, but experiments in real spaceflight conditions are the gold standard for validating microbial survival and functionality. A landmark study exposed a broad spectrum of nitrogen cycle microorganisms to a 44-day Low Earth Orbit (LEO) flight on the FOTON-M4 satellite [10]. The experimental conditions are summarized below.
Table 1: Spaceflight Exposure Conditions for Nitrogen Cycle Microorganisms (FOTON-M4 Mission)
| Parameter | Experimental Condition | Control Condition (Ground) |
|---|---|---|
| Duration | 44 days | 44 days |
| Temperature | 17.5 ± 2 °C (in flight) | 23.1 ± 3 °C (G23) / 4 ± 1 °C (G4) |
| Gravity | 10⁻³ – 10⁻⁴ g | 1 g |
| Radiation Dose | 687 ± 170 µGy day⁻¹ | 2.1 ± 0.3 µGy day⁻¹ |
After return to Earth, the microorganisms were reactivated and their metabolic activities were measured. The results demonstrated that all key nitrogen conversion functionalities could be recovered [10]. Notably, for some reactor communities, ureolytic activity after spaceflight was not significantly different from, or even higher than, ground controls stored at a similar temperature. Furthermore, refrigerated storage at 4°C on Earth generally resulted in the highest metabolic rates, suggesting that temperature control is a more critical factor for preserving microbial viability than radiation exposure at these doses [10].
Table 2: Post-Flight Reactivation of Key Nitrogen Cycle Processes
| Microbial Process | Key Microorganism(s) Tested | Reactivation Success | Noteworthy Findings |
|---|---|---|---|
| Ureolysis | Cupriavidus pinatubonensis, reactor communities | Activity not reduced by LEO; in one community, rates were 600% of ground control [10]. | |
| Nitritation | Nitrosomonas europaea (AOB) | Presence of other bacteria in defined communities positively influenced reactivation [10]. | |
| Nitratation | Nitrobacter winogradskyi (NOB) | Successfully reactivated, confirming the robustness of the second nitrification step [10]. | |
| Anammox | Members of the Brocadiaceae family | First report of anammox survival and reactivation after Space exposure [10]. |
Another study focusing on the resilience of microbes to launch and re-entry forces found that spores of Bacillus subtilis, a bacterium important for human health, could survive extreme acceleration (up to 13 g) and deceleration (up to 30 g) during a suborbital flight unharmed [64]. This research underscores that certain hardy microbes can withstand the mechanical rigors of space travel, which is a separate but equally important stressor.
Methodologies for Space Microbiology Research
Experimental Workflow for Spaceflight Experiments
Conducting microbiological research in space requires meticulous planning and a standardized workflow to ensure meaningful, comparable results. The process integrates flight hardware preparation, ground controls, and post-flight analysis.
Ground-Based Simulation Techniques
Due to the cost and infrequency of spaceflight, ground-based facilities (GBFs) are essential for preliminary research [65]. These devices simulate microgravity by randomizing the gravity vector or creating a low-shear environment.
Table 3: Key Ground-Based Facilities for Microgravity Simulation
| Facility Type | Simulative Effect | Principle of Operation | Suitable Organisms |
|---|---|---|---|
| 2-D Clinostat | Simulated Microgravity (SMG) | Continuous rotation perpendicular to gravity vector, averaging its direction to near zero [65]. | Plant tissues, microbial cultures |
| Random Positioning Machine (RPM) | Simulated Microgravity (SMG) | Uses two independent rotation axes to randomize the sample's orientation relative to gravity in 3D space [65]. | Microbial cultures, small cell cultures |
| Rotating Wall Vessel (RWV) | Low-Shear Modeled Microgravity (LSMMG) | Maintains cells in a constant state of free-fall by rotating a fluid-filled vessel, minimizing shear forces [66]. | Human/animal cells, microorganisms |
Molecular and Metabolic Adaptations
The space environment induces significant changes in microbial physiology at the molecular level. The absence of gravity-driven forces creates a diffusion-limited microenvironment around bacterial cells. This reduces nutrient influx and waste efflux, leading to accumulation of metabolic by-products and triggering oxidative stress [66]. These subtle changes in the extracellular environment are believed to be a primary driver of the observed physiological adaptations [66].
Microbes respond to these stresses through global alterations in gene expression and metabolism. While no universal bacterial response exists, some broad trends have been observed [66]. In primary metabolism, changes often include increases in carbohydrate metabolism and alterations in amino acid metabolism that reflect the increased oxidative stress. Secondary metabolism, responsible for producing compounds like antibiotics, shows highly variable responses; production can increase, decrease, or remain unchanged, sometimes even for different metabolites within the same organism [66].
These molecular adaptations can have operational consequences for BLSS. For instance, studies have reported that microgravity can lead to enhanced biofilm formation [67] [68]. While biofilms can be detrimental by clogging systems, a more robust biofilm in a nitrogen recycling bioreactor could potentially enhance process stability. Furthermore, alterations in virulence and antibiotic resistance have been observed in some pathogens, underscoring the need for careful monitoring of microbial payloads in a closed spacecraft environment [67].
The Scientist's Toolkit: Research Reagents & Materials
Table 4: Essential Research Reagents and Materials for Space Microbiology Studies
| Item | Function/Application | Example Use Case |
|---|---|---|
| Axenic Cultures | Defined, single-strain models to study specific metabolic functions and isolate individual stress responses. | Nitrosomonas europaea for studying ammonia oxidation under microgravity [10]. |
| Defined Microbial Communities | Simplified multi-strain systems to investigate microbial interactions and community resilience. | Co-cultures of N. europaea and Nitrobacter winogradskyi to model nitrification [10]. |
| Reactor Communities | Complex, enriched microbial consortia from operational bioreactors, representing a more applied, robust system. | OLAND (partial nitritation/anammox) biofilm for studying efficient nitrogen removal [10]. |
| Random Positioning Machine (RPM) | Ground-based facility to simulate microgravity conditions by continuous, random reorientation of samples [65]. | Preliminary testing of microbial growth and nitrogen conversion rates before a spaceflight experiment. |
| Illumina MiSeq Platform | High-throughput sequencing technology for phylogenetic characterization of microbial communities post-flight. | Analyzing changes in community structure (e.g., diversity, evenness) in reactor communities after space exposure [10]. |
| LB Agar Plates | Standard growth medium for evaluating heterotrophic contamination in autotrophic cultures post-flight [10]. | Ensuring sample purity and planetary protection compliance before reactivation tests. |
The body of evidence confirms that nitrogen-cycling microorganisms exhibit a remarkable capacity to survive and maintain metabolic function after exposure to space conditions. The successful reactivation of ureolytic, nitrifying, and anammox bacteria after a 44-day LEO mission is a strong positive indicator for their integration into BLSS [10]. Future research must focus on optimizing storage conditions, understanding the long-term functional stability of these microbes in integrated BLSS, and developing countermeasures for challenges like biofilm formation and increased virulence. The research tools and experimental frameworks outlined in this whitepaper provide a roadmap for advancing the microbial component of regenerative life support, a critical step toward sustainable human exploration of the solar system.
Optimizing Fertilization Efficiency and Crop Harvest Index for Maximum N Recovery
Within the framework of regenerative life support systems research, the efficient management of nitrogen (N) is not merely an agronomic goal but a fundamental prerequisite for sustaining human life in isolated environments, such as long-duration space missions [3]. In these closed-loop systems, termed Bioregenerative Life Support Systems (BLSS), an efficient conversion of wasted nitrogenous compounds into food is mandatory, as resupply from Earth is impractical [9]. On Earth, inefficient cropping systems add a substantial amount of unconsumed N fertilizer to the soil profile as residual N, which can contribute to groundwater pollution and other environmental issues [69]. The challenge, both terrestrially and in BLSS, is to synchronize nutrient availability with plant uptake, thereby maximizing the Nitrogen Recovery Efficiency (NRE)—the proportion of applied N that is taken up by the crop [69]. This guide provides a technical overview of strategies to optimize fertilization practices and the crop harvest index to achieve maximum N recovery, with a specific focus on implications for BLSS.
Core Concepts: Nitrogen Use Efficiency and the Harvest Index
Defining Nitrogen Recovery Efficiency
Nitrogen Recovery Efficiency (NRE), also referred to as Nitrogen Use Efficiency (NUE), is a critical metric for evaluating the performance of cropping systems. It is fundamentally defined as the proportion of applied nitrogen fertilizer that is successfully taken up and utilized by the crop [69]. A global meta-analysis has revealed that the N recovery for major cereals like wheat, maize, and rice is often suboptimal, typically ranging between 34% and 45% [69]. This inefficiency means the majority of applied N fertilizer is not utilized by the crop in the season of application; instead, it either accumulates in the soil as residual N or is lost to the environment, causing pollution [69].
The Harvest Index and its Relationship to N Recovery
The Harvest Index (HI) is a measure of the efficiency with which a plant partitions its accumulated biomass (including acquired nutrients) into the economically valuable component—the grain or fruit. A higher HI indicates that a greater proportion of the plant's total nitrogen uptake is directed toward the harvestable yield, rather than being stored in residual biomass like stems and leaves. While not directly quantified in the search results, the concept is intrinsically linked to the N recovery efficiency of the system. Optimizing management practices to enhance the HI ensures that the recovered N is effectively converted into food, a principle of paramount importance for food production in BLSS where system closure and efficiency are critical [9].
Quantitative Data on Nitrogen Dynamics
Legacy Effects and Residual Nitrogen
Long-term field experiments provide critical data on the fate of residual N. One seven-year study with a wheat-maize rotation offers key insights, as shown in Table 1 [69].
Table 1: Legacy Impact of Nitrogen Fertilization on Crop Yield and Nitrogen Recovery
| Parameter | Fertilized Years (Data after 2 years of N application) | Unfertilized Years (Data after 5 years without N application) |
|---|---|---|
| Application Rate | N340 (160 kg N/ha for wheat, 180 for maize) & N500 (220 kg N/ha for wheat, 280 for maize) | No nitrogen fertilizer applied |
| Grain Yield | Significantly higher than control plots | Did not decrease in the first year without fertilization; started to decrease considerably in later years |
| Cumulative N Recovery | 37%–44% | Increased to 74%–80% for the N340 + DCD treatment |
| Nitrate Accumulation in 0-200 cm Soil Profile | 237–489 kg N ha⁻¹ | Soil N accumulation became less than 60 kg ha⁻¹ after four years without fertilization |
Global Potential for Enhancing Nitrogen Recovery
A recent global analysis assessed the potential for improving NRE through optimized management practices. An initial estimate suggested a mean global potential NRE increase of nearly 30% [70]. However, a subsequent addendum that accounted for the pre-existing adoption of some improved practices provided a more refined and realistic projection, as summarized in Table 2 [70].
Table 2: Revised Global Potential for Increasing Nitrogen Recovery Efficiency (NUEr) through Improved Management
| Management Practice | Original Estimated Mean Impact on NUEr | Revised Estimated Mean Impact on NUEr (Considering Existing Adoption) |
|---|---|---|
| Combined Optimal Practices | ~30% increase | ~19% increase |
| Optimal Nutrient Management | ~27% increase | ~17% increase |
| Optimal Soil Management | ~6% increase | ~6% increase |
| Optimal Crop Management | ~1% increase | ~1% increase |
Experimental Protocols for Studying Nitrogen Recovery
Long-Term Field Experiment on Residual Nitrogen
Objective: To investigate the long-term fate of residual N in the soil profile and its contribution to crop yield and nitrogen recovery efficiency after the cessation of fertilization [69].
Methodology:
- Site Description: The experiment was conducted on the Chinese Loess Plateau, a region with deep soil profiles prone to N accumulation. The soil is classified as a Eum-Orthic Anthrosol.
- Experimental Design:
- Duration: Seven years.
- Crop System: Annual rotation of winter wheat and summer maize.
- Phase 1 (First 2 years): N fertilizer was applied at two rates (N340 and N500) with and without the nitrification inhibitor dicyandiamide (DCD). A control treatment received no N fertilizer.
- Phase 2 (Subsequent 5 years): All treatments, including previously fertilized ones, were grown without any N fertilization.
- Data Collection:
- Grain Yield: Measured at harvest for both crops each year.
- Grain N Uptake: Determined to calculate N recovery.
- Soil Nitrate Accumulation: Soil samples were taken annually from depths of 0-200 cm, and at the experiment's conclusion, from 4-8 m depths, to monitor the movement and depletion of residual nitrate.
- Calculation:
- Cumulative Fertilizer N Recovery was calculated for both the fertilized and unfertilized periods.
Protocol for N Recovery in a BLSS Context (Nitrification of Urine)
Objective: To recover nitrogen from human urine in a BLSS by converting urea into nitrate, a readily available plant fertilizer [3] [9].
Methodology:
- Waste Collection: Collect source-separated human urine. Note that on the International Space Station, urine is currently stabilized with chromium trioxide and sulfuric acid to prevent microbial growth, which creates a nitrogen "dead end" [9].
- Ureolysis: The urea in urine is hydrolyzed by microbial urease enzymes, converting it into ammonium and carbonate. This can be achieved in a dedicated bioreactor.
- Nitrification: The ammonium-rich solution is transferred to a second bioreactor where nitrifying bacterial communities perform a two-step oxidation:
- Ammonia Oxidation: Nitrosomonas spp. oxidize ammonium to nitrite.
- Nitrite Oxidation: Nitrobacter spp. oxidize nitrite to nitrate.
- Process Control: Carefully monitor and control parameters such as pH, temperature, and oxygen levels to ensure optimal conditions for the nitrifying bacteria.
- Fertilizer Application: The resulting nitrate solution is supplied to hydroponic or soil-based plant production systems as a key component of the nutrient solution.
Visualization of Nitrogen Recovery Pathways
Workflow for Quantifying Residual Nitrogen in Cropping Systems
The following diagram illustrates the experimental and conceptual workflow for analyzing residual nitrogen, based on the long-term field study [69].
Diagram 1: Workflow for analyzing residual nitrogen in a long-term crop rotation field experiment.
Nitrogen Cycle in a Bioregenerative Life Support System (BLSS)
This diagram outlines the integrated nitrogen cycle within a BLSS, highlighting the refinery and production stages essential for closing the loop [3] [9].
Diagram 2: Simplified nitrogen cycle in a Bioregenerative Life Support System (BLSS).
The Scientist's Toolkit: Key Reagents and Materials
Table 3: Essential Research Reagents and Materials for Nitrogen Recovery Studies
| Reagent/Material | Function/Application | Technical Notes |
|---|---|---|
| Dicyandiamide (DCD) | A nitrification inhibitor that slows the microbial conversion of ammonium to nitrate in soil. | Used to reduce nitrate leaching and N₂O emissions, increasing N availability in the root zone. Shown to increase yield at high N rates [69]. |
| Enhanced Efficiency Fertilizers (EEFs) | A class of fertilizers designed to gradually release nutrients. Includes controlled-release and slow-release types. | Coated fertilizers (e.g., with polymers) retard nutrient availability, ensuring better synchronization with crop uptake [71]. |
| Calcium Ammonium Nitrate (CAN) / Ammonium Nitrate (AN) | Common synthetic nitrogen fertilizers providing nitrogen in the ammonium and nitrate forms. | Nitrate is immediately available, while ammonium is retained on soil particles. AN is effective for crops needing fast nitrogen release [72]. |
| Urea | A high-nitrogen content synthetic fertilizer (≥44% N). | Requires hydrolysis by soil urease to become plant-available. Subject to volatilization losses as ammonia [72]. |
| Nitrifying Bacteria Consortia (Nitrosomonas, Nitrobacter) | Used in BLSS to convert toxic ammonia from waste streams into plant-available nitrate. | Core biological component of the nitrogen recovery loop in systems like MELiSSA. Performance under space conditions must be validated [3] [9]. |
| Ion-Exchange Resins / HPLC Columns | For separation and quantitative analysis of nitrogen species (e.g., NH₄⁺, NO₃⁻, NO₂⁻) in soil, water, and plant extracts. | Critical for accurate measurement of nitrogen pools and fluxes in experimental systems. |
Optimizing fertilization efficiency for maximum nitrogen recovery requires a paradigm shift from single-season management to a multi-annual perspective that accounts for the significant legacy effects of residual soil nitrogen [69]. The strategies outlined—including the use of nitrification inhibitors, enhanced efficiency fertilizers, and precision application—are essential for terrestrial agriculture to reduce environmental impact and improve economic returns. For Bioregenerative Life Support Systems, these principles are not optional but fundamental. The successful closure of the nitrogen cycle through robust biological and physicochemical waste refinery processes is the cornerstone of sustainable long-term space exploration [3] [9]. Future research must focus on integrating these optimized nutrient management practices with advanced crop varieties exhibiting a high Harvest Index, and on validating the performance of these integrated systems under simulated and actual space conditions.
Quantitative Performance, Terrestrial Analogues, and Future Directions
Comparative Analysis of Nitrogen Recovery Efficiencies Across Different BLSS
Nitrogen recovery efficiency (NRE) is a pivotal metric for assessing the sustainability and feasibility of Bioregenerative Life Support Systems (BLSS) in long-duration space missions. Efficient nitrogen cycling is essential for in-situ production of food, regeneration of breathable air, and recycling of water, thereby minimizing reliance on Earth resupply. This whitepaper provides a comparative analysis of NRE across different BLSS subsystems, drawing on data from terrestrial agriculture, current space station systems, and advanced BLSS prototypes like the Micro-Ecological Life Support System Alternative (MELiSSA). The analysis synthesizes quantitative data on NRE, details experimental methodologies for its determination, and outlines the critical research pathways needed to achieve closed-loop nitrogen cycles for future lunar and Martian habitats.
In a BLSS, the nitrogen cycle is a fundamental process that connects crew metabolism to food production systems. The primary source of nitrogen is crew urine, which accounts for 85% of the potentially recoverable nitrogen, predominantly in the form of urea [23] [3]. The overarching goal of a BLSS is to convert this waste nitrogen into forms usable by plants or other food-producing organisms, such as nitrate (NO₃⁻) or ammonium (NH₄⁺), thereby closing the nutrient loop [9]. The effectiveness of this conversion and subsequent uptake is quantified as Nitrogen Recovery Efficiency (NRE), a benchmark indicator reflecting the proportion of added nitrogen that is incorporated into harvestable biomass [73].
The inability of current physicochemical systems on the International Space Station (ISS) to recover nitrogen for food production creates a critical dependency on Earth [23] [3]. In contrast, BLSS initiatives like MELiSSA (European Space Agency), BIOS (Russia), and Lunar Palace (China) aim to integrate biological components—specifically nitrifying bacteria and higher plants—to achieve a high level of resource closure [9] [5]. The success of these systems hinges on optimizing NRE at every stage, from waste processing to crop cultivation.
Quantitative Analysis of Nitrogen Recovery Efficiencies
A comparative analysis of NRE requires examining performance across different subsystems, from terrestrial agriculture to space-based prototypes.
Nitrogen Recovery in Terrestrial Agriculture
Terrestrial agriculture provides a baseline for understanding NRE in controlled systems. A recent global meta-analysis established benchmarks for N use efficiency (NUEr) in croplands and the potential improvements from various management practices.
Table 1: Global Nitrogen Use Efficiency (NUEr) in Croplands and Improvement Potential [74]
| Region / Management Practice | Current Average NUEr | Potential NUEr with Optimal Management | Key Management Practices for Improvement |
|---|---|---|---|
| Global Average | 48% | 78% (30% increase) | Optimal combination of nutrient, crop, and soil management |
| High-Income Regions (e.g., USA, EU) | 66-69% | Increase below global average | Precision farming, crop diversification |
| Middle-Income Regions (e.g., China, India) | 21-35% | Increase above global average | Right fertilizer type, rate, time, and place |
| Low-Income Regions (e.g., sub-Saharan Africa) | >80% | Not applicable (limited by low input) | Increased total nutrient input |
| Nutrient Management | - | +3.6% to +11% | Enhanced efficiency fertilizers, right placement/rate/timing |
| Crop Management | - | +4.4% to +8% | Cover crops, diverse crop rotations, residue retention |
| Soil Management (Reduced Tillage) | - | No significant impact | - |
Nitrogen Recovery in Life Support Systems
The NRE of a system is heavily influenced by its technological composition, ranging from purely physicochemical to fully bioregenerative.
Table 2: Nitrogen Recovery in Different Life Support System Architectures
| System / Compartment | System Type | Key Nitrogen Recovery Process | Reported Efficiency / Performance | Challenges |
|---|---|---|---|---|
| ISS ECLSS | Physicochemical | Water recovery from urine via distillation; N is not recycled [23] [3]. | 0% (Nitrogen ends up in a waste brine) [9]. | System designed for water recovery, not nutrient recycling; uses toxic oxidizers (Cr⁶⁺). |
| MELiSSA Compartment III | Biological (Bacterial) | Nitrification: Conversion of urea/ NH₄⁺ to NO₃⁻ by nitrifying bacteria [23] [3]. | High conversion efficiency reported; quantitative NRE data from pilot plant (MPP) required. | Stability under space conditions (microgravity, radiation); process control. |
| BLSS Higher Plant Compartment | Biological (Plant) | Nutrient Uptake: Assimilation of NO₃⁻/NH₄⁺ from recycled nutrient solutions [31] [5]. | Depends on crop species and solution quality; NRE for entire BLSS not yet quantified. | Balancing nutrient solution; preventing Na⁺/Cl⁻ buildup; variable plant NUE. |
| Integrated BLSS (Ground Demonstrators) | Hybrid (Bio-Physicochemical) | Combination of bacterial nitrification and plant uptake in a closed loop [9] [5]. | Full system closure and N mass balance are primary research goals; definitive NRE values are a key outcome of ongoing research. | Integration of all compartments; nitrogen loss management; system robustness. |
Experimental Protocols for Assessing NRE
Accurate determination of NRE is critical for comparing BLSS technologies. The following protocols are adapted from both agricultural and BLSS-specific research.
Protocol for Determining NRE in Plant Cultivation Systems
This protocol is used to measure the efficiency with which plants incorporate applied nitrogen into their biomass [73].
- Experimental Design: Establish a controlled growth environment (e.g., hydroponic system). Apply treatments that vary the nitrogen source (e.g., synthetic urine effluent, mineral fertilizer) and rate. Include a zero-nitrogen control.
- Biomass Sampling and Analysis:
- At harvest, separate plant tissue into relevant components (e.g., shoots, roots, grain).
- Dry the biomass to a constant weight at 60-70°C and record the dry mass.
- Mill the dried plant material to a fine powder.
- Determine the nitrogen concentration in the powdered tissue using an elemental analyzer (e.g., via Dumas combustion).
- Calculation:
- Calculate crop N uptake for each treatment:
N_uptake (kg ha⁻¹) = Biomass (kg ha⁻¹) × N_concentration (%) - Calculate Nitrogen Recovery Efficiency (NRE) using the difference method:
NRE (%) = [ (N_uptake_fertilized - N_uptake_control) / N_applied ] × 100
- Calculate crop N uptake for each treatment:
Protocol for Determining Nitrification Efficiency in Bacterial Bioreactors
This protocol assesses the performance of the microbial compartment responsible for converting waste nitrogen into plant-available forms [23] [9].
- Bioreactor Operation: Operate a continuous-flow or batch bioreactor inoculated with a nitrifying consortium (e.g., Nitrosomonas and Nitrobacter spp.). The feed solution should contain a synthetic urine analogue or real hydrolyzed urine with known concentrations of ammonium (NH₄⁺).
- Process Monitoring:
- Regularly sample the influent and effluent.
- Analyze samples for key nitrogen species: Total Nitrogen (TN), NH₄⁺-N (using spectrophotometry or ion-selective electrodes), NO₂⁻-N, and NO₃⁻-N (using ion chromatography).
- Monitor and control key environmental parameters: pH (7-8), temperature (25-30°C), and dissolved oxygen.
- Calculation:
- Ureolysis Efficiency:
% Urea hydrolyzed = [ (Urea_in - Urea_out) / Urea_in ] × 100 - Nitrification Efficiency:
% NH₄⁺ converted = [ (NH₄⁺_in - NH₄⁺_out) / NH₄⁺_in ] × 100 - Total Nitrogen Conversion: Ensure mass balance to account for potential N losses (e.g., denitrification, assimilation into bacterial biomass).
- Ureolysis Efficiency:
Visualization of Nitrogen Pathways in a BLSS
The following diagram illustrates the complex pathway of nitrogen through a representative BLSS, such as the MELiSSA loop, highlighting the critical transformation points and integration between compartments.
Diagram: Nitrogen Transformation and Cycling Pathway in a BLSS
The Scientist's Toolkit: Key Research Reagents and Materials
Research and development of BLSS require specific reagents, materials, and technological systems to study and optimize nitrogen recovery.
Table 3: Essential Research Materials and Technologies for BLSS Nitrogen Cycling Studies
| Category | Item / Technology | Function in Nitrogen Recovery Research |
|---|---|---|
| Analytical Tools | Ion Chromatograph | Quantifies specific nitrogen ions (NO₂⁻, NO₃⁻) in waste streams and nutrient solutions [23]. |
| Elemental Analyzer | Measures total nitrogen content in plant and microbial biomass for NRE calculations [73]. | |
| Spectrophotometer | Determines concentrations of ammonium (NH₄⁺) and other nutrients via colorimetric assays. | |
| Biological Components | Nitrifying Consortium (e.g., Nitrosomonas, Nitrobacter) | Performs the critical nitrification process in bioreactors, converting ammonia to nitrate [23] [3]. |
| Selected Crop Cultivars (e.g., lettuce, wheat, potato) | Acts as the primary nitrogen sink; chosen for high NRE, yield, and nutritional value [5]. | |
| Engineering Systems | Hydroponic/Aeroponic Growth System | Provides the platform for soilless plant cultivation using recycled nutrient solutions [31] [5]. |
| Continuous-Flow Bioreactor | Houses nitrifying bacteria; allows for controlled study of waste processing and conversion rates [23] [9]. | |
| Gas Chromatograph / Mass Spectrometer | Monitors gaseous nitrogen species (e.g., N₂, N₂O) to identify and quantify nitrogen losses [9]. |
The comparative analysis underscores a significant gap between the nitrogen recovery capabilities of current operational systems like the ISS ECLSS (0% NRE) and the potential of integrated BLSS. While terrestrial agriculture demonstrates that NRE can exceed 70% with optimal management, translating this efficiency to closed BLSS loops remains a formidable challenge. The variability in NRE across system components—bacterial nitrification versus plant uptake—highlights the need for a systems-level approach to optimization.
Critical research gaps must be addressed to achieve operational BLSS for lunar and Martian missions. Key future directions include:
- Quantifying Integrated System NRE: Ground-based testing in facilities like the MELiSSA Pilot Plant is essential to obtain definitive NRE values for fully integrated systems and close the nitrogen mass balance [9] [5].
- Space Environment Effects: Research must prioritize understanding the impact of microgravity and space radiation on the biological nitrogen cycle, particularly the stability of nitrifying bacterial communities and plant nutrient uptake mechanisms [23] [5].
- Real-Time Monitoring and Control: Developing advanced sensors and modeling tools is crucial for the dynamic management of nutrient solutions, preventing the accumulation of toxic elements like sodium and chloride, and maximizing overall system NRE [31] [73].
Success in these endeavors will not only enable sustainable human exploration deeper into space but also contribute valuable technologies for closing nutrient loops and improving sustainability on Earth.
The Micro-Ecological Life Support System Alternative (MELiSSA) is a European Space Agency (ESA) initiative developing bioregenerative life support technologies for long-term human space missions. Its fundamental objective is to achieve a closed-loop ecosystem that provides atmosphere regeneration, water recycling, food production, and waste treatment through a series of interconnected, controlled biological compartments [75] [76]. The project is inspired by a terrestrial lake ecosystem, engineered for maximum efficiency and reliability within the constraints of spaceflight [77] [78].
Within this context, nitrogen cycling is a critical process. A crew member excretes 7-16 grams of nitrogen per day, primarily in urine, making it the main recoverable nitrogen source [79] [23]. Effective nitrogen recycling is therefore a prerequisite for the self-sufficient production of food, as nitrogen is an essential nutrient for the photosynthetic compartments that generate edible biomass [23]. The MELiSSA loop addresses this challenge through a dedicated nitrification process, which stabilizes waste and converts it into a usable form for plants and cyanobacteria [79].
Compartment Functions and Integration
The MELiSSA loop is conceived as a network of five core compartments, each performing a specific function in the recycling chain. The current integration work at the MELiSSA Pilot Plant (MPP) in Barcelona focuses on connecting three of these key compartments [77] [80].
Table 1: Core Compartments of the MELiSSA Loop
| Compartment | Primary Function | Key Microorganisms/Components | Role in Nitrogen Cycle |
|---|---|---|---|
| Compartment 3 | Nitrification | Co-culture of Nitrosomonas europaea & Nitrobacter winogradsky [77] | Converts toxic ammonium (NH₄⁺) to nitrate (NO₃⁻) [77] |
| Compartment 4a | Photosynthesis, O₂ production, edible biomass production | Cyanobacteria Limnospira indica [77] [78] | Assimilates nitrate (NO₃⁻) from C3 as a nutrient source for growth [77] |
| Compartment 4b | Higher plant food and O₂ production | Lettuce, wheat, red beet [77] [80] | Assimilates nitrogen for plant growth. |
| Compartment 5 | Crew compartment (mock-up) | Laboratory rats [77] [76] | Produces liquid and gaseous waste (e.g., CO₂, urea) as system inputs. |
The integration strategy is progressive. Initial stages involved connecting Compartment 4a (photobioreactor) and Compartment 5 (crew) in a closed gas loop [77]. This was followed by linking Compartment 3 (nitrifier) and Compartment 4a in the liquid phase, enabling the transfer of nitrate from the nitrifier to the photobioreactor [77]. The most advanced integration tests have successfully operated Compartments 3, 4a, and 5 together in continuous operation for several months, demonstrating high system robustness and reliability under both steady-state and transitory conditions [77] [80].
Figure 1: Material flow between key MELiSSA compartments.
Performance Analysis of Key Compartments
Compartment 3: The Nitrifying Bioreactor
Experimental Protocol: The nitrification compartment is a 7-liter cylindrical packed-bed bioreactor [77]. The reactor is packed with polystyrene beads that serve as a physical support for a stable biofilm formed by a co-culture of the nitrifying bacteria Nitrosomonas europaea (ammonia-oxidizing bacteria, AOB) and Nitrobacter winogradsky (nitrite-oxidizing bacteria, NOB) [77]. The system is operated in continuous mode with liquid recirculation and a closed gas-loop to ensure aerobic conditions. Key monitored parameters include pH, temperature, dissolved oxygen (pO₂), and conductivity, measured using sterilizable probes (e.g., Mettler Toledo) [77]. The effluent is filtered to prevent nitrifying bacteria from reaching the downstream photobioreactor [77].
Performance Data: The compartment's performance is measured by its efficiency in converting ammonium to nitrate. The process is highly aerobic, requiring 2 moles of oxygen for the complete oxidation of 1 mole of N-NH₄⁺ [77]. The system has demonstrated long-term stability for over a year of continuous operation in the Pilot Plant [79]. Its performance is guided by predictive mathematical models that allow for precise control, such as managing nitrite concentrations in response to changes in dissolved oxygen levels [79].
Table 2: Compartment 3 Performance and Operational Parameters
| Parameter | Value / Specification | Context / Significance |
|---|---|---|
| Reactor Type | Packed-bed bioreactor [77] | Provides high surface area for biofilm formation. |
| Operational Volume | 7 L [77] | Scale for pilot demonstration. |
| Key Microorganisms | Nitrosomonas europaea (AOB), Nitrobacter winogradsky (NOB) [77] | Axenic co-culture for two-stage nitrification. |
| Oxygen Requirement | 2 mol O₂ per 1 mol N-NH₄⁺ [77] | Indicates the aerobic nature of the process. |
| Primary Function | Transform ammonium (NH₄⁺) to nitrate (NO₃⁻) [77] | Converts a toxic waste product into a plant-usable nutrient. |
Compartment 4a: The Photobioreactor
Experimental Protocol: Compartment 4a is an 83-liter external-loop air-lift photobioreactor (PBR) for the axenic culture of the edible cyanobacteria Limnospira indica [77] [78]. The air-lift design avoids shear stress on the cells and provides efficient gas-liquid transfer for O₂ removal and CO₂ uptake [78]. The system is operated in a continuous mode, and its performance is characterized by analyzing the effect of two key variables: dilution rate (D) and photon flux density (PFD) illumination [78]. A critical operational variable is the specific photon flux density (qPFD), which accounts for both light intensity and cell density, providing a more accurate measure of light availability to the culture [78].
Performance Data: The performance is primarily assessed through oxygen productivity and biomass composition. A maximum oxygen productivity (rO₂) of 1.35 mmol L⁻¹ h⁻¹ was achieved at a dilution rate of 0.025 h⁻¹ and a PFD of 930 µmol m⁻² s⁻¹ [78]. The system exhibits reversible photoinhibition at very high PFD (>1700 µmol m⁻² s⁻¹), demonstrating the cells' adaptability [78]. Furthermore, qPFD directly influences biomass composition; higher qPFD leads to a decrease in phycobiliproteins (up to 62.5%) and chlorophyll (47.8%), while carbohydrate content increases [78]. This multiplicity of steady states depending on previous culture conditions is a key finding for operational control [78].
Table 3: Compartment 4a Performance and Operational Parameters
| Parameter | Value / Significance | Conditions / Notes |
|---|---|---|
| Reactor Type | 83 L External-loop air-lift [77] [78] | Gentle mixing, high gas transfer. |
| Organism | Limnospira indica (edible cyanobacteria) [78] | Source of O₂ and edible biomass. |
| Max Oxygen Productivity | 1.35 mmol L⁻¹ h⁻¹ [78] | At D=0.025 h⁻¹, PFD=930 µmol m⁻² s⁻¹. |
| Photoinhibition | Reversible at PFD >1700 µmol m⁻² s⁻¹ [78] | Demonstrates system and biological robustness. |
| qPFD effect on composition | Phycobiliproteins ↓62.5%, Carbohydrates ↑ [78] | When qPFD increased from 6.1 to 19.2 µmol g⁻¹ s⁻¹. |
Figure 2: Key performance factors for the photobioreactor.
Research Toolkit: Essential Reagents and Materials
The ground-based demonstration of the MELiSSA loop relies on a suite of specific biological, chemical, and engineering components.
Table 4: Essential Research Reagents and Materials for MELiSSA Experiments
| Item / Solution | Function / Application | Specific Example / Note |
|---|---|---|
| Nitrifying Co-culture | Performs core nitrification function in C3 [77] | Axenic co-culture of Nitrosomonas europaea & Nitrobacter winogradsky [77]. |
| Limnospira indica | Photosynthetic producer of O₂ and edible biomass in C4a [78] | Axenic cyanobacteria strain, high protein content [78]. |
| Polystyrene Beads | Biofilm support material in the packed-bed nitrifying reactor [77] | Provides high surface area for bacterial immobilization [77]. |
| Nutrient Solution (N source) | Feeds the photobioreactor with a usable nitrogen form [77] | Nitrate (NO₃⁻) solution supplied from C3 effluent [77]. |
| Process Gases | Provides O₂ for nitrification (C3); supplies CO₂ and removes O₂ in C4a [77] | Mass flow-meters (e.g., Bronkhorst) for precise control of gas loops [77]. |
| Analytical Sensors | On-line monitoring of key bioreactor parameters [77] | pH probes (Mettler Toledo), Clark amperometric pO₂ sensors [77]. |
The MELiSSA Pilot Plant has successfully demonstrated the integrated, long-term operation of its core compartments, proving the robustness of the nitrogen cycling and gas exchange processes under controlled conditions [77] [80]. The project represents a significant advancement in bioregenerative life support, moving from theoretical concept to ground-based demonstration at a pilot scale relevant to human needs.
Future work is focused on further closing the loop, including the integration of compartments for solid waste processing (Compartments 1 and 2) and the transition to using real human urine as a primary nitrogen source [80] [23]. Concurrently, space adaptation studies, such as the BISTRO, NITRIMEL, and upcoming URINIS flight experiments, are crucial for validating the performance of these biological systems under the unique constraints of microgravity and space radiation [79]. The continuous development and testing within the MELiSSA framework are essential steps toward achieving the ultimate goal: enabling long-duration human exploration beyond Earth orbit through sustainable, closed-loop life support.
Integrated Crop-Livestock Management Systems (ICLS) represent a paradigm shift in agricultural science, leveraging ecological principles to optimize nitrogen cycling, enhance soil health, and improve system resilience. This technical guide examines ICLS and regenerative agricultural frameworks through the lens of nitrogen management, with particular relevance to bioregenerative life support systems (BLSS) for long-duration space missions. We synthesize current research on nitrogen flow optimization, present quantitative metrics for system performance, and provide experimental protocols for evaluating nitrogen use efficiency across integrated agricultural systems. The findings demonstrate that strategic recoupling of crop and livestock production can significantly reduce synthetic nitrogen dependency while maintaining productivity—a critical consideration for closed-loop life support systems where external inputs are logistically constrained or cost-prohibitive.
Nitrogen represents both a critical agricultural macronutrient and a significant source of environmental pollution when mismanaged. In terrestrial agriculture, conventional approaches reliant on synthetic nitrogen fertilizers have created linear nutrient flows characterized by significant losses to air and water systems. The integrated crop-livestock management system framework reestablishes circular nutrient economies by using livestock manure as organic fertilizer and crop residues as animal feed, thereby closing nutrient loops and reducing dependence on external inputs [81].
The relevance of these systems extends beyond terrestrial agriculture to bioregenerative life support systems (BLSS) for space exploration, where complete nutrient recycling is essential for mission sustainability. Research initiatives such as the European Space Agency's Micro-Ecological Life Support System Alternative (MELiSSA) program explicitly focus on recovering nutrients from waste streams to support plant growth in closed environments [3] [31]. The lessons from terrestrial ICLS provide valuable insights for optimizing nitrogen cycling in these space-based systems.
Core Principles and Nitrogen Cycling Mechanisms
Fundamental Principles of Integrated Crop-Livestock Systems
ICLS operate on several interconnected principles designed to optimize nutrient cycling:
- Nutrient Recycling: Animal manure and slurry serve as organic fertilizers, reducing dependency on synthetic inputs while returning organic matter to soil ecosystems [81].
- Feed Optimization: Crop residues (straw, stover, green regrowth) and processing by-products (bran, molasses) are utilized as livestock feed, converting waste resources into nutritional inputs [81].
- Strategic Cropping Patterns: Implementation of catch crops, leys (fields with crops rotated with grass for pasture), and cereal-legume intercropping captures potentially leached nutrients and provides high-quality forage [81].
- Spatial Integration: Landscape mosaics that combine crops for human consumption, forage crops, and grassland create synergistic relationships between system components [81].
Nitrogen Cycling Pathways in Integrated Systems
The following diagram illustrates key nitrogen flows and transformation pathways in an integrated crop-livestock system:
Nitrogen Cycling in Integrated Crop-Livestock Systems
This nitrogen cycle demonstrates the critical pathways through which nitrogen flows between system components. Biological fixation converts atmospheric nitrogen into plant-available forms, while careful management of crop residues and animal manure returns nitrogen to the soil pool. The potential loss pathways (leaching and volatilization) represent system inefficiencies that proper management seeks to minimize.
Quantitative Assessment of System Performance
Nitrogen Cycling and Environmental Impact Metrics
Research from multiple agricultural systems demonstrates the measurable benefits of integrated approaches for nitrogen management and environmental impact mitigation.
Table 1: Nitrogen Cycling and Environmental Impact Metrics in Integrated Agricultural Systems
| System Type | Nitrogen Cycling Efficiency | GHG Emission Reduction | Soil Carbon Impact | Primary Productivity | Reference |
|---|---|---|---|---|---|
| Integrated Crop-Livestock (Model) | Increased by 65% | Reduced by 49% | Trade-offs with reduced organic inputs | Slight reduction maintained | [82] |
| Dutch Dairy Farm (Transition) | Improved cycling | Improved climate regulation | 7% increase in SOM surplus | Medium (from High) | [83] |
| Regenerative Agriculture | Building soil N while reducing inputs | N₂O reduction (300× CO₂e potential) | Increased soil organic matter | Initially reduced, then stabilized | [84] [85] |
| Pastoral Systems (SSA) | Inputs dominated by atmospheric deposition (80%) | Hotspots in animal congregation areas | Varies with management | Limited by aridity | [86] |
Nitrogen Balance and Loss Pathways
Different agricultural systems exhibit characteristic nitrogen flow patterns, with significant implications for system efficiency and environmental impact.
Table 2: Nitrogen Balance and Loss Pathways Across Agricultural Systems
| System Component | Nitrogen Input Sources | Nitrogen Output Pathways | Major Loss Mechanisms | Efficiency Considerations | |
|---|---|---|---|---|---|
| Conventional Cropping | Synthetic fertilizer (>70%) | Crop harvest, leaching, volatilization | Leaching (33%), NH₃ volatilization | High inputs, significant losses | [84] [85] |
| Integrated Systems | Manure, BNF, atmospheric deposition | Meat/milk, controlled losses | Leaching (NO₃⁻ > DON), targeted management | Recycling reduces external inputs | [82] [81] |
| BLSS (MELiSSA) | Recycled urine (85% of recoverable N), processed waste | Plant uptake, system losses | N₂ emissions (18%, unquantified), processing inefficiencies | Complete recycling imperative | [3] [31] |
| Pastoral Systems | Atmospheric deposition (~80%), BNF | Animal products, leaching, gaseous losses | Bomas: NH₃ (16% total N), N₂O hotspots | Mobility patterns affect distribution | [86] |
Experimental Framework and Methodologies
Modeling Approaches for System Assessment
Research into integrated agricultural systems employs sophisticated modeling frameworks to evaluate multiple ecosystem services and socioeconomic outcomes simultaneously:
Combined Modeling Framework: The integration of Soil Navigator (SN) and FarmDESIGN (FD) models enables ex-ante redesign and assessment of farming systems. SN evaluates soil functions at field scale, while FD assesses environmental and socioeconomic outcomes at farm scale [83].
Multi-Indicator Assessment: A systems approach quantifies multiple indicators (nitrogen cycling, greenhouse gas emissions, energy balance, soil organic matter) simultaneously at both farm and regional levels, providing a comprehensive understanding of benefits and trade-offs [82].
Nitrogen Flow Analysis: Development of models based on nitrogen flow analysis allows comparison of scenarios with varying levels of manure digestion, synthetic fertilizer application, and feed import restrictions [82].
Experimental Protocol: Modeling ICLS Transitions
Baseline Assessment: Quantify existing nitrogen flows, soil health indicators, and economic metrics for the current farming system.
Scenario Development: Create alternative farm configurations with varying integration levels between crop and livestock components.
Model Parameterization: Input site-specific data on soil attributes, climate conditions, crop rotations, and livestock management practices.
Trade-off Analysis: Evaluate how changes in management practices affect the delivery of multiple ecosystem services, including primary productivity, climate regulation, and farm profitability [83].
Validation: Compare model predictions with empirical data from pilot implementations to refine model parameters and assumptions.
Field-Based Monitoring and Verification
For researchers implementing integrated systems, several tools and MRV (monitoring, reporting, verification) systems enable robust data collection and analysis:
- GLEAM-i: An open-source tool to calculate GHG emissions from livestock production [81].
- EX-Ante Carbon-balance Tool (EX-ACT): Estimates and tracks outcomes of agricultural interventions on GHG emissions [81].
- Collect Earth Online: A free, open-source system for viewing and interpreting high-resolution satellite imagery to collect up-to-date land cover and land use information [81].
- GrassSignal: A decision support tool for sustainable grassland monitoring [81].
Research Reagents and Essential Materials
Table 3: Research Reagents and Essential Materials for Nitrogen Cycling Studies
| Reagent/Material | Function/Application | Technical Specifications | Relevance to Nitrogen Cycling |
|---|---|---|---|
| Soil Sampling Equipment | Collection of representative soil samples | Stainless steel corers, sterile containers, cold chain maintenance | Quantification of soil N pools, microbial biomass |
| Gas Chromatography System | Quantification of N₂O and other greenhouse gases | Electron capture detector, automated sampling systems | Measurement of gaseous N losses from soils and manure |
| Lysimeters | Collection of soil pore water | Ceramic cups, vacuum extraction systems | Monitoring nitrate leaching below root zone |
| Isotopic Tracers (¹⁵N) | Tracing nitrogen pathways through ecosystems | Enriched K¹⁵NO₃, (¹⁵NH₄)₂SO₄, application at field rates | Quantification of N use efficiency, partitioning of N sources |
| Microbial Assay Kits | Assessment of soil microbial community function | DNA extraction kits, PCR reagents, enzyme activity assays | Evaluation of N-cycling microbial populations (nitrifiers, denitrifiers) |
| Soil Moisture Sensors | Continuous monitoring of soil water dynamics | Time-domain reflectometry, capacitance sensors, data loggers | Correlation of N transformation rates with soil environmental conditions |
| Anaerobic Digestion Systems | Processing of manure and crop residues | Bioreactors with temperature and mixing control, biogas collection | Evaluation of nutrient recovery efficiency from waste streams |
Application to Bioregenerative Life Support Systems
The principles and methodologies developed for terrestrial integrated agricultural systems have direct relevance to the development of BLSS for space exploration. Several critical considerations emerge from this interdisciplinary application:
Nutrient Recovery from Waste Streams: Research from the MELiSSA program demonstrates that approximately 85% of potentially recoverable nitrogen in BLSS comes from human urine, predominantly in the form of urea [3]. Efficient recovery and processing of this nitrogen source is essential for maintaining crop production without external inputs.
Nitrogen Balance at Habitat Level: BLSS design must achieve full nitrogen balance, with sufficient N₂ maintained for atmospheric pressure regulation while providing adequate mineral nitrogen for plant biomass production [31].
Sodium and Chloride Management: Efficient removal of sodium and chloride from urine and other organic waste streams is essential to prevent the spread of these elements to other BLSS components [31].
System Integration and Control: Advanced sensors and control systems must be developed to manage plant growth in BLSS, accounting for microgravity conditions, limited volume availability, variations in plant nutrient requirements, and recycling of nutrient solutions [31].
The experimental frameworks and modeling approaches described in Sections 4 and 5 provide methodologies for optimizing these systems before implementation in resource-constrained space environments.
Integrated Crop-Livestock Management Systems and regenerative agricultural frameworks offer sophisticated approaches to nitrogen cycling that balance productivity with environmental sustainability. The quantitative assessments presented in this review demonstrate significant potential for improved nitrogen use efficiency, reduced greenhouse gas emissions, and enhanced soil health through the strategic integration of crop and livestock production components.
The experimental protocols and modeling frameworks developed for terrestrial applications provide valuable methodologies for optimizing bioregenerative life support systems for space exploration, where complete nutrient recycling is essential for mission success. Future research should focus on quantifying nitrogen loss pathways more precisely, especially in underrepresented systems such as pastoral systems in sub-Saharan Africa, and refining nutrient recovery processes from waste streams for BLSS applications.
The transfer of knowledge between terrestrial agricultural systems and space life support research represents a promising avenue for innovation in closed-loop ecosystem management, with potential benefits for both space exploration and sustainable terrestrial agriculture.
Long-duration human space exploration, such as missions to the Moon and Mars, requires advanced Bioregenerative Life Support Systems (BLSS) to sustainably provide essential resources. These systems aim to create closed-loop environments where air, water, and food are regenerated, and waste is recycled, dramatically reducing the need for resupply from Earth [5]. Nitrogen is a crucial element in these systems, constituting a fundamental component of proteins, nucleic acids, and other biological molecules. Its efficient cycling is therefore imperative for sustaining both plant-based food production and crew health [3] [41].
The current Environmental Control and Life Support System (ECLSS) on the International Space Station (ISS) relies primarily on physicochemical processes. It recovers oxygen and water but does not produce food and requires a steady supply of consumables from Earth [3]. In contrast, BLSS incorporates biological components—plants and microorganisms—to create a more self-sufficient ecosystem. These systems face the significant challenge of managing nitrogen loss pathways and efficiently recovering nutrients from waste streams, with urine representing the primary source of recoverable nitrogen (85% of the total) [3]. This whitepaper synthesizes the current validation status and results from key ground and space demonstration missions that are paving the way for mature nitrogen cycling technologies in space.
Key Ground Demonstration Missions and Facilities
Ground-based test facilities are critical for developing and integrating the individual compartments of a BLSS. They allow for the validation of subsystem performance and the study of system-level integration before deployment in space.
The MELiSSA Program
The European Space Agency's Micro-Ecological Life Support System Alternative (MELiSSA) is one of the longest-running and most advanced BLSS initiatives. Designed as a five-compartment bioengineered system, it mimics the ecosystem of a lake to recycle waste and produce water, oxygen, and food [3]. A key objective is the highly efficient recovery of minerals, particularly nitrogen, from waste streams for use as fertilizer [3].
- MELiSSA Pilot Plant (MPP): Located in Spain, this facility is designed for designing and testing the closed-loop connection of multiple MELiSSA compartments [5].
- Plant Characterization Unit (PaCMan): Located in Italy, this facility focuses on fundamental biological experiments on plants in a closed chamber [5].
Beijing Lunar Palace 1
China has emerged as a leader in integrated BLSS testing through its Beijing Lunar Palace 1 program. This facility has successfully demonstrated the viability of a closed-loop habitat by supporting a crew of four analog taikonauts for a full year. The system demonstrated closed-system operations for atmosphere, water, and nutrition, marking a significant milestone in BLSS development [11].
Other Historical Ground Demonstrators
Several other large-scale ground-based demonstrators have contributed to BLSS knowledge, including:
- BIOS-1, 2, 3, and 3 in Russia
- Biosphere 2 in the USA
- The Closed Ecology Experiment Facility (CEEF) in Japan
- The NASA BIO-Plex, which was discontinued and demolished after 2004 [11] [5].
These facilities have been used to test specific technologies for controlled cultivation chambers, food production, and biological waste management, and to evaluate the physiological and psychological impacts of confinement on isolated crews [5].
Space-Based Demonstration Missions
Testing biological systems in the actual space environment is a necessary step to mature BLSS technology. The unique conditions of space, particularly microgravity and increased ionizing radiation, can significantly affect biological processes [3].
Initial Biological Experiments in LEO
To date, bioreactors and plant cultivation components have been tested in Low Earth Orbit (LEO), onboard satellites like FOTON and the International Space Station (ISS). These experiments have served as a proof of concept, confirming that it is feasible to operate a bioreactor and grow plants in LEO [5]. However, these tests have typically been conducted on single biological systems at a small scale (e.g., less than 100 mL or 0.2 m²), with low overall yield, over short durations, and with significant crew involvement [5].
Proba-3 Mission
Launched in December 2024, ESA's Proba-3 mission is a technology demonstrator for precise formation flying, which involves two satellites maintaining a configuration with millimetric precision [87]. While its primary goal is solar observation, the mission demonstrates the high level of autonomous control and system reliability that will be required for the complex, interdependent subsystems of a future BLSS [87].
ISRO SpaDeX Mission
The Indian Space Research Organisation (ISRO) is advancing in-space robotic operations with its Space Docking Experiment (SpaDeX). Launched on December 30, 2024, this mission involves two small satellites (a Chaser and a Target) demonstrating autonomous docking technology [88]. This capability is a critical precursor for future space missions where multiple modules carrying different BLSS components (e.g., a plant growth module and a waste processing module) may need to dock and exchange resources [88].
Table 1: Key Recent and Upcoming Demonstration Missions
| Mission/Facility | Lead Agency/Country | Primary Focus | Key Achievement/Status |
|---|---|---|---|
| Beijing Lunar Palace 1 | CNSA (China) | Integrated BLSS | 1-year crewed mission in a closed-loop system [11] |
| MELiSSA Pilot Plant | ESA (Europe) | Nitrogen recovery & loop closure | Ground testing of interconnected bioregenerative compartments [3] [5] |
| Proba-3 | ESA (Europe) | Formation Flying | Launched 2024; first results released June 2025 [87] |
| SpaDeX | ISRO (India) | Autonomous Rendezvous & Docking | Launched Dec 2024; docking experiment postponed to Jan 9, 2025 for ground validation [88] |
| ISS Plant/Bloreactor Experiments | International Partners | Biological component validation | Proof-of-concept for plant growth & bioreactor operation in microgravity [5] |
Experimental Protocols for Nitrogen Cycle Research
Research into nitrogen cycling for BLSS involves protocols spanning microbial ecology, plant physiology, and system integration.
Protocol for Investigating Nitrification in Simulated Space Conditions
This protocol assesses the impact of space-relevant conditions (e.g., simulated microgravity, radiation) on nitrifying bacteria, which are vital for converting ammonia into nitrate.
- Microbial Cultivation: Enrich nitrifying bacterial cultures (e.g., from Nitrosomonas and Nitrobacter species) in a defined ammonium-containing mineral medium under optimal aerobic conditions (e.g., 28°C, pH 7.5-8.0) [3].
- Application of Simulated Space Conditions:
- Radiation Exposure: Expose cultures to controlled doses of gamma or ionizing radiation from a sealed source, simulating a multi-year space mission dose over a shorter period [3].
- Simulated Microgravity: Use a clinostat or rotating wall vessel bioreactor to create a low-shear, modeled microgravity environment for the bacterial cultures [3].
- Process Monitoring: Regularly monitor culture density (optical density at 600 nm), ammonium (NH₄⁺), nitrite (NO₂⁻), and nitrate (NO₃⁻) concentrations using spectrophotometric or chromatographic methods (e.g., ion chromatography) [3].
- Microbial Community Analysis: At endpoint, analyze the microbial community structure using 16S rRNA gene sequencing to identify shifts in the populations of ammonia-oxidizing and nitrite-oxidizing bacteria [3].
- Data Analysis: Compare nitrification rates (the disappearance of NH₄⁺ and appearance of NO₃⁻) and microbial diversity between treatment groups and ground controls to determine the effects of the simulated space conditions.
Protocol for Optimizing Plant Nitrogen Uptake and Nutritional Value
This protocol determines the optimal cultivation parameters for maximizing nitrogen use efficiency and the production of nutrients, like antioxidants, in BLSS crops.
- Plant Material and Growth: Select candidate crops (e.g., lettuce varieties 'youmaicai' and 'rapid'). Germinate seeds under controlled conditions and transplant seedlings into a hydroponic system [89].
- Experimental Treatment Application: Implement a factorial design with varying levels of:
- Nitrogen Supply: Use different concentrations of nitrogen (e.g., 2.5, 10.5, and 18.5 mmol/L) in the nutrient solution, with a controlled NH₄⁺:NO₃⁻ ratio [89].
- Light Quality: Expose plants to different spectral compositions, specifically varying the proportion of green light (e.g., 10%, 20%, 30%) using LED lighting systems [89].
- Harvest and Analysis: Harvest plant tissues at a mature stage and analyze for:
- Biomass: Record fresh and dry weight of edible and inedible biomass.
- Nitrogen Content: Determine tissue nitrogen concentration using elemental analysis (combustion method).
- Nutrient Quality: Quantify ascorbic acid (Vitamin C) content via high-performance liquid chromatography (HPLC) and measure the activity of key enzymes in the ascorbic acid-glutathione (AsA-GSH) cycle [89].
- Gene Expression: Analyze the expression of genes involved in ascorbic acid biosynthesis (e.g., GGP) using quantitative real-time polymerase chain reaction (qRT-PCR) [89].
- Statistical Modeling: Use analysis of variance (ANOVA) to identify significant main and interaction effects of nitrogen and light on the measured parameters, determining the optimal cultivation conditions for each variety [89].
Diagram 1: Nitrogen cycling research workflow.
Quantitative Data and Validation Status
The validation of BLSS technologies is measured through key performance parameters, including nitrogen recovery efficiency, system closure rates, and crop productivity.
Table 2: Performance Data from Nitrogen Cycling Research
| System / Experiment | Parameter Measured | Result / Performance | Context / Implication |
|---|---|---|---|
| Beijing Lunar Palace 1 | System Closure & Crew Duration | 1-year mission with 4 crew [11] | Demonstrated operational closure for atmosphere, water, and nutrition. |
| ISS ECLSS (UPA) | Water Recovery from Urine | ~85% efficiency [3] | Physicochemical system; nitrogen is a waste product, not recycled. |
| Lettuce Cultivation Optimization | Ascorbic Acid Content | Increased under optimized green light & N [89] | 30% green light & 10.5-18.5 mmol/L N optimal for 'rapid' lettuce. |
| Nitrogen Redistribution Modeling | Global Cereal Production | Potential 12% increase with optimal N use [90] | Ground model highlighting impact of efficient N management. |
| Combined NFR System Modeling | Nitrogen Loss & Fixation Demand | Recycling reduces N2 fixation demand to amount lost via inefficiencies [41] | Model shows recycling is critical to reduce energy-intensive N fixation. |
The Scientist's Toolkit: Key Research Reagents and Materials
Research and development of nitrogen cycling subsystems rely on a suite of specialized reagents, biologicals, and technological components.
Table 3: Essential Research Reagents and Materials for Nitrogen Cycling Experiments
| Item | Function / Application | Example Usage in Protocols |
|---|---|---|
| Defined Mineral Media | Supports growth of specific microorganisms without undefined contaminants. | Cultivating nitrifying bacteria in MELiSSA compartment studies [3]. |
| Ion Chromatography System | Precisely separates and quantifies ions like ammonium (NH₄⁺), nitrite (NO₂⁻), and nitrate (NO₃⁻). | Monitoring nitrification efficiency in bioreactor effluents [3]. |
| LED Photoregimen Systems | Provides tunable light spectra for plant growth experiments. | Optimizing green light proportion for ascorbic acid synthesis in lettuce [89]. |
| Hydroponic Nutrient Solutions | Deliver precise concentrations and forms of nitrogen (e.g., NO₃⁻ vs NH₄⁺) to plants. | Studying plant nitrogen use efficiency under different N regimes [89]. |
| qRT-PCR Assays | Quantifies the expression levels of genes involved in N metabolism and stress responses. | Measuring expression of GGP gene in lettuce under different light qualities [89]. |
| 16S rRNA Sequencing Reagents | Allows for characterization of microbial community structure and dynamics. | Profiling the nitrifying bacterial community after exposure to simulated space conditions [3]. |
| Clinistats / Rotating Wall Vessels | Earth-based simulators for studying biological effects of microgravity. | Investigating the effect of low-shear modeled microgravity on nitrifying bacteria [3]. |
Ground and space demonstration missions have proven the fundamental feasibility of individual BLSS components and small-scale integrated systems. The successful year-long mission in the Beijing Lunar Palace 1 represents a major leap forward, providing invaluable data on system-level integration and stability [11]. Furthermore, ongoing missions like Proba-3 and SpaDeX are validating the precise autonomous control and robotic operations that will be necessary for maintaining future BLSS [88] [87].
However, critical challenges remain before a fully operational, space-based BLSS can be deployed. Key research gaps include:
- Microgravity and Radiation Effects: A deeper understanding of how space conditions affect all biological processes in the nitrogen cycle, from microbial nitrification to plant nutrient uptake, is needed [3] [5].
- System Integration and Control: Robust, autonomous control algorithms must be developed to manage the highly interconnected and dynamically changing BLSS compartments over long durations [5].
- Technology Scaling: Current space-tested biological systems are small. Technologies must be scaled up to support a crew without prohibitive mass, volume, and energy requirements [5].
Addressing these gaps requires a sustained commitment to a structured research pipeline, from ground-based laboratory studies and integrated analog testing to dedicated technology demonstration missions in LEO and on the lunar surface, which will serve as the ultimate testbed for future Mars missions [11] [5].
Diagram 2: Simplified BLSS nitrogen cycling loop.
For long-duration human space exploration missions beyond low Earth orbit, regenerative life support systems (RLSS) become essential for crew survival when resupply from Earth is not feasible or reliable [91] [92]. These systems must reliably recycle essential resources—air, water, and nutrients—through integrated biological and physico-chemical processes. Nitrogen cycling represents a particularly critical and complex component within these closed-loop systems, as it governs key functions from atmospheric management to nutrient recovery for food production [93] [5].
The integration of a nitrogen cycle is a recent advancement even in sophisticated Earth system models, with diverse formulations introducing substantial uncertainty in how nitrogen affects interconnected biogeochemical processes [93]. In RLSS research, this challenge is amplified by the extreme constraints of the space environment and the absolute reliability requirements for human life support. This technical guide examines the critical pathway from reduced-complexity models and laboratory-scale experiments to fully implemented, operational nitrogen cycling systems for space exploration contexts, with specific methodologies and frameworks to accelerate this transition.
Nitrogen Cycle Fundamentals and Modeling Approaches
Core Nitrogen Processes in Closed Systems
In regenerative life support systems, nitrogen undergoes multiple transformations mediated by biological and chemical processes. Understanding these pathways is essential for system design:
Table 1: Key Nitrogen Transformation Processes in RLSS
| Process | Biological/Chemical Basis | System Function | Primary Organisms/Mechanisms |
|---|---|---|---|
| Biological Nitrogen Fixation | Conversion of atmospheric N₂ to ammonia | Introduces new nitrogen into system | Nitrogen-fixing bacteria/cyanobacteria |
| Nitrification | Oxidation of ammonia to nitrite then nitrate | Converts waste ammonia to plant-available forms | Nitrosomonas, Nitrobacter spp. |
| Assimilatory Nitrate Reduction | Nitrate reduction to ammonium for biomass incorporation | Nitrogen uptake for plant/microbial growth | Plants, algae, microorganisms |
| Denitrification | Reduction of nitrates to nitrogen gases | Nitrogen removal from system; can cause nitrogen loss | Denitrifying bacteria |
| Ammonification | Organic nitrogen decomposition to ammonia | Recycles nitrogen from waste streams | Heterotrophic microorganisms |
These processes create an interconnected nitrogen network where the balance between different pathways determines overall system efficiency and stability [94] [5]. In terrestrial environments, the MAGICC carbon-nitrogen cycle model (CNit v1.0) has demonstrated the profound impact of nitrogen availability on carbon cycling, with nitrogen limitation consistently reducing net primary production in Earth system models [93]. This coupling has direct analogs in RLSS, where nitrogen availability will govern both food production and atmospheric regeneration capabilities.
Modeling Frameworks for Nitrogen Cycle Integration
Reduced-complexity models (RCMs) provide essential tools for understanding nitrogen dynamics before implementing costly full-scale systems. The Model for the Assessment of Greenhouse Gas Induced Climate Change (MAGICC) with its CNit v1.0 extension represents a relevant approach, featuring a globally integrated, annually averaged box model that simulates terrestrial carbon and nitrogen dynamics [93].
The CNit model architecture includes carbon and nitrogen pools for plant (P), litter (L), soil (S), and an inorganic mineral (M) nitrogen pool, with atmospheric (A) exchanges. This compartmentalized structure allows tracking of element fluxes through the system while maintaining computational efficiency [93]. For RLSS applications, similar modeling approaches can be adapted to the more constrained environment of space habitats, with compartments representing crew, plants, waste processing microorganisms, and atmospheric systems.
Diagram 1: CNit Model Structure for C-N Cycling
Experimental Methodologies for Nitrogen Cycle Research
Laboratory-Scale Investigation of Nitrogen Processes
Controlled laboratory experiments provide the foundation for understanding specific nitrogen transformation pathways. The following experimental protocol exemplifies approaches for investigating particular aspects of nitrogen cycling, adapted from recent atmospheric chemistry research with relevance to RLSS applications [95]:
Experimental Protocol: Investigating Halide-Enhanced Nitrate Photolysis
Objective: To quantify the enhancement effect of iodide on HONO production during nitrate photolysis under conditions relevant to marine aerosols, with applications to RLSS atmospheric processing.
Materials and Equipment:
- Dynamic chamber with xenon lamp irradiation source
- Time-of-Flight Chemical Ionization Mass Spectrometry (ToF-CIMS)
- Chemiluminescence/photolytic converter NOx analyzer
- Sodium nitrate (NaNO₃) solutions
- Sodium halide salts (NaCl, NaBr, NaI) at varying concentrations
Procedure:
- Prepare control solution of pure sodium nitrate (0.1 M)
- Prepare experimental solutions with nitrate and halides at atmospherically relevant concentrations:
- Chloride: 1.0 M
- Bromide: 1.5 × 10⁻³ M
- Iodide: 3.6 × 10⁻⁴ M
- Introduce solutions into dynamic chamber under controlled temperature and humidity
- Initiate xenon lamp irradiation to simulate solar spectrum
- Quantify gaseous products (HONO and NO₂) using ToF-CIMS and NOx analyzer at regular intervals
- Continue monitoring for minimum of 150 minutes to observe temporal patterns
- Conduct coexistence experiments with multiple halides to identify dominant effects
Data Analysis:
- Calculate HONO/NO₂ production ratios for each condition
- Determine enhancement factors relative to pure nitrate photolysis
- Model interfacial phenomena using molecular dynamics simulations
- Parameterize results for integration into larger system models
This experimental approach demonstrated that iodide enhances HONO production during nitrate photolysis by over tenfold under typical marine conditions, providing insights into how specific catalysts can dramatically accelerate nitrogen cycling rates [95].
Integrated System Testing Approaches
Beyond investigating individual processes, research must advance to testing integrated nitrogen cycles in increasingly complex environments. The supervisory Model Predictive Control (MPC) framework developed for the MELiSSA (Micro-Ecological Life Support System Alternative) project represents a sophisticated methodology for coordinating multiple subsystems in a circular life support architecture [91].
Experimental Protocol: Supervisory MPC for Circular Nitrogen Management
Objective: To implement and validate a nonlinear model predictive control system for coordinating nitrogen flows across multiple compartments of a regenerative life support system.
System Components:
- Photobioreactors for microbial photosynthesis
- Higher plant growth compartments
- Waste processing bioreactors
- Atmosphere revitalization systems
- Water recovery systems
Control Framework:
- Derive prediction model addressing generic regenerative LSS
- Formulate optimization problem with nitrogen balance constraints
- Implement state estimation for key nitrogen pools
- Design constraint handling for critical human safety parameters
- Develop failure scenario response protocols
Validation Methodology:
- Conduct 14-week mission simulation
- Introduce system failure scenarios to test robustness
- Measure closure rates for nitrogen cycles
- Quantify system stability under perturbation
- Evaluate controller performance against predefined requirements [91]
This approach enables researchers to test nitrogen cycle integration under controlled but increasingly realistic conditions, identifying emergent behaviors and control challenges before full-scale implementation.
Implementation Framework: From Models to Operational Systems
Implementation Science Applied to RLSS Development
The transition from validated laboratory models to operational systems represents a critical phase in nitrogen cycle research. Implementation science frameworks, though developed for healthcare contexts, offer valuable methodologies for this technology transfer process [96]. The Consolidated Framework for Implementation Research (CFIR) provides a structured approach to addressing adoption barriers across five domains: intervention characteristics, outer setting, inner setting, individual implementers, and implementation process [96].
For nitrogen cycling systems, key implementation considerations include:
Table 2: Implementation Framework for RLSS Nitrogen Cycling
| CFIR Domain | Application to Nitrogen Cycle Implementation | Assessment Methods |
|---|---|---|
| Intervention Characteristics | Reliability requirements, technical complexity, operational burden | Failure mode analysis, complexity assessment |
| Outer Setting | Mission constraints, resupply limitations, safety requirements | Stakeholder analysis, requirements mapping |
| Inner Setting | Crew training, organizational structure, facility readiness | Readiness assessments, communication analysis |
| Individual Implementers | Crew expertise, motivation, perceived burden | Surveys, competency evaluations, training needs assessment |
| Implementation Process | Integration sequencing, testing protocols, adaptation needs | Process mapping, iterative testing, measure-change-measure cycles |
The "measure-change-measure" approach, analogous to the "study" and "act" components of plan-do-study-act cycles, enables continuous refinement during implementation. This involves regularly collecting data on system uptake and barriers, adapting the implementation strategy based on findings, and remeasuring to assess improvement [96].
Scaling Considerations and Technical Requirements
The transition from laboratory models to full-system implementation requires careful attention to scaling effects and technical integration:
Scaling Challenges in Nitrogen Cycling Systems:
- Mass Transfer Limitations: Gas-liquid transfer phenomena differ significantly under microgravity conditions, directly affecting nitrogen transformation processes in bioreactors [97]
- System Closure Requirements: Target closure rates of 90-98% for air, water, and nutrient cycles are necessary to significantly reduce resupply mass [92]
- Reliability Specifications: Ultra-low failure rates (e.g., less than 1 in 10,000 per major component) must be achieved through redundancy and modular design [92]
- Crew Time Optimization: Automation and integration with electronic management systems reduce operational burden and mitigate staffing turnover challenges [96]
Technical Integration Priorities:
- Gradual System Integration: Implement supervisory control layers to coordinate heterogeneous subsystems involving mechanical, chemical, biological, and energetic processes [91]
- Failure Response Capabilities: Develop contingency protocols for critical component failures, including nitrogen imbalance scenarios
- Multi-scale Modeling: Couple reduced-complexity models with detailed process models to maintain predictive capability across scales
- Human-System Interface Design: Optimize crew interaction requirements through user-centered design and comprehensive training programs
Research Reagent Solutions for Nitrogen Cycling Studies
Table 3: Essential Research Reagents for Nitrogen Cycling Investigations
| Reagent/Category | Function in Nitrogen Cycle Research | Specific Application Examples |
|---|---|---|
| Halide Salts (NaI, NaBr, NaCl) | Catalyze nitrate photolysis processes | Enhancing HONO production from nitrate photolysis under marine-relevant conditions [95] |
| Nitrate Isotopes (¹⁵NO₃⁻) | Tracing nitrogen transformation pathways | Quantifying process rates and identifying dominant pathways in complex systems [94] |
| Molecular Probes for functional genes | Marker detection for nitrogen transformation capabilities | Assessing microbial potential for nitrification, denitrification, and nitrogen fixation [94] |
| Photobioreactor Systems | Controlled cultivation of photosynthetic organisms | Biological air revitalization using microalgae/cyanobacteria for O₂ production and CO₂ removal [97] |
| Ion Chromatography Systems | Quantifying nitrogen species concentrations | Monitoring ammonium, nitrite, and nitrate levels in water recycling systems [94] |
| Time-of-Flight Chemical Ionization Mass Spectrometry | Detection of gaseous nitrogen compounds | Real-time monitoring of HONO and NO₂ production during photolysis experiments [95] |
Bridging the research gap between laboratory models and full-system implementation requires a systematic approach that integrates fundamental process understanding, controlled experimental validation, and deliberate implementation frameworks. Nitrogen cycling represents a particularly critical challenge due to its essential role in multiple life support functions and its complex, interconnected transformation pathways.
The pathway forward involves continued refinement of reduced-complexity models informed by targeted laboratory experiments, progressive testing in increasingly integrated ground-based demonstrators, and the application of implementation science principles to navigate the transition from proven concepts to operational systems. As research advances, nitrogen cycling systems will evolve from supplemental functions to core infrastructure enabling long-duration human presence beyond Earth, ultimately achieving the high closure rates and ultra-reliability required for sustainable space exploration.
Conclusion
Effective nitrogen cycling is the linchpin for achieving sustainable, closed-loop life support, essential for the future of long-duration space exploration. This synthesis demonstrates that while significant progress has been made in developing biological and hybrid methodologies for nitrogen recovery, challenges in system efficiency, stability, and integration remain. Key performance metrics like Nutrient Use Efficiency (NUE) are critical for optimization. Future research must focus on closing the full nitrogen balance at the habitat level, developing advanced sensors and control techniques for dynamic plant growth systems, and validating these technologies in increasingly realistic ground and space-based demonstrations. The principles developed for these advanced systems also offer profound insights for improving nutrient cycling and sustainability in terrestrial agricultural and ecological systems, creating a valuable feedback loop between space technology and Earth-based applications.
References
- 1. Comparing Life Support Solutions: PCLSS vs BLSS vs ... [https://thespringinstitute.com/comparing-life-support-solutions-pclss-vs-blss-vs-celss/]
- 2. ISS ECLSS [https://en.wikipedia.org/wiki/ISS_ECLSS]
- 3. Development of Nitrogen Recycling Strategies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8548772/]
- 4. Aquatic bryophytes as biofilters and resource regenerators ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12502980/]
- 5. Plant and microbial science and technology as ... [https://www.nature.com/articles/s41526-023-00317-9]
- 6. Review of research into bioregenerative life support system ... [https://www.sciencedirect.com/science/article/abs/pii/S2214552421000663]
- 7. Bioregenerative life support system that ensures long-term ... [http://ui.adsabs.harvard.edu/abs/2022cosp...44.2600L/abstract]
- 8. Stoichiometric model of a fully closed bioregenerative life ... [https://www.frontiersin.org/journals/astronomy-and-space-sciences/articles/10.3389/fspas.2023.1198689/full]
- 9. Challenges for waste refinery and food production processes [https://www.sciencedirect.com/science/article/abs/pii/S0376042117300234]
- 10. Nitrogen cycle microorganisms can be reactivated after ... [https://www.nature.com/articles/s41598-018-32055-4]
- 11. Critical investments in bioregenerative life support systems for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12357894/]
- 12. A Systematic Review and Meta-Analysis of Nitrogen ... [https://pubmed.ncbi.nlm.nih.gov/40871643/]
- 13. Meta-analysis of nitrogen balance studies for estimating ... [https://www.sciencedirect.com/science/article/pii/S0002916523055788]
- 14. Enhancing Crop Nitrogen Efficiency: The Role of Mixed Nitrate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12108568/]
- 15. Modulation of Root Nitrogen Uptake Mechanisms Mediated ... [https://www.mdpi.com/2223-7747/14/17/2729]
- 16. Use of Photobioreactors in Regenerative Life Support ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2021.699525/full]
- 17. Optimizing Nitrogen Fixation and Recycling for Food ... [https://www.frontiersin.org/journals/astronomy-and-space-sciences/articles/10.3389/fspas.2021.699688/full]
- 18. Recovery nitrogen and phosphorus from source-separated ... [https://www.sciencedirect.com/science/article/abs/pii/S1001074225002116]
- 19. Urine [https://en.wikipedia.org/wiki/Urine]
- 20. Anaerobic biological fermentation of urine as a strategy to ... [https://www.sciencedirect.com/science/article/pii/S096014811930285X]
- 21. Urea nitrogen urine test [https://www.ucsfhealth.org/medical-tests/urea-nitrogen-urine-test]
- 22. Urine Urea Nitrogen Test: Purpose, Procedure, and Results [https://www.healthline.com/health/urea-nitrogen-urine]
- 23. Development of Nitrogen Recycling Strategies for ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2021.700810/full]
- 24. Higher plant characterization [https://www.melissafoundation.org/page/higher-plant-characterization]
- 25. Reliability and lifetime estimation of bioregenerative life ... [https://www.sciencedirect.com/science/article/abs/pii/S0094576522006294]
- 26. Lunar Palace 1: Treatment performance and microbial ... [https://www.sciencedirect.com/science/article/abs/pii/S0048969721064482]
- 27. Uncovering the soil nitrogen cycle from microbial pathways ... [https://www.maxapress.com/article/doi/10.48130/nc-0025-0005]
- 28. Terraform Sustainability Assessment Framework for ... [https://www.frontiersin.org/journals/astronomy-and-space-sciences/articles/10.3389/fspas.2021.789563/full]
- 29. Challenges for waste refinery and food production processes [https://www.sciencedirect.com/science/article/pii/S0376042117300234]
- 30. Nitrification - Wikipedia [https://en.wikipedia.org/wiki/Nitrification]
- 31. Recycling nutrients from organic waste for growing higher ... [https://www.sciencedirect.com/science/article/pii/S2214552423000639]
- 32. 5.10E: Nitrification - Biology LibreTexts [https://bio.libretexts.org/Bookshelves/Microbiology/Microbiology_(Boundless)/05:_Microbial_Metabolism/5.10:_Chemolithotrophy/5.10E:_Nitrification]
- 33. How to Separate from Air – Nitrogen Extraction... | GENERON Nitrogen [https://www.generon.com/how-separate-nitrogen-air-extraction/]
- 34. How a Nitrogen Generator Works: PSA vs. Membrane ... [https://www.ecscorrosion.com/blog/how-a-nitrogen-generator-works]
- 35. Techno-economic and life cycle assessment of membrane ... [https://www.sciencedirect.com/science/article/abs/pii/S2949908924001973]
- 36. Membrane Technologies for Nitrogen Recovery from Waste ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9864344/]
- 37. How Long Does a Nitrogen Generator Last? [https://adgastech.com/how-long-does-nitrogen-generator-last/]
- 38. Nitrogen cycling in Bioregenerative Life Support Systems [http://ui.adsabs.harvard.edu/abs/2017PrAeS..91...87C/abstract]
- 39. Ammonium release in synthetic and human urine by a ... [https://pubs.rsc.org/en/content/articlehtml/2024/ra/d3ra07606g]
- 40. Enzyme immobilization: an overview on techniques and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3563746/]
- 41. Approaches to nitrogen fixation and recycling in closed life ... [https://www.frontiersin.org/journals/astronomy-and-space-sciences/articles/10.3389/fspas.2023.1176576/full]
- 42. Light-driven integration of diazotroph-derived nitrogen in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11502797/]
- 43. Organic nitrogen in hydroponics, the proven way [https://scienceinhydroponics.com/2021/04/organic-nitrogen-in-hydroponics-the-proven-way.html]
- 44. System Integration: Definition, Types, and Approaches [https://www.altexsoft.com/blog/system-integration/]
- 45. Nitrogen management in nitrification-hydroponic systems ... [https://www.sciencedirect.com/science/article/pii/S2352186422000475]
- 46. Advancing sustainable production in hydroponic systems ... [https://www.sciencedirect.com/science/article/abs/pii/S0959652625018141]
- 47. Function and Management of Nitrogen in Hydroponics [https://hortamericas.com/blog/news/function-and-management-of-nitrogen-in-hydroponics/?srsltid=AfmBOoo51pQgbGElOV8-16QLFosnwRgOkjioJC7eiEmL6tc8NzTtWTtY]
- 48. Advanced Hydroponic Systems 2025: Build Towers & Go ... [https://farmonaut.com/blogs/advanced-hydroponic-systems-2025-build-towers-go-organic]
- 49. Recycled Nitrogen for Regenerative Agriculture: A Review ... [https://www.mdpi.com/2073-4395/15/11/2503]
- 50. Nitrogen use efficiency (NUE): an indicator for the ... [https://research.wur.nl/en/publications/nitrogen-use-efficiency-nue-an-indicator-for-the-utilisation-of-n/]
- 51. A urine-fuelled soil-based bioregenerative life support ... [https://www.sciencedirect.com/science/article/abs/pii/S2214552417300706]
- 52. Enhancing nitrogen use efficiency in agriculture by ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1543714/full]
- 53. Nutrient use efficiency (NUE) of wheat (Triticum aestivum L ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8794114/]
- 54. Enhancing NUE through site specific nutrient management and in... [https://www.slideshare.net/slideshow/enhancing-nue-through-site-specific-nutrient-management-and-in-problematic-soils/238365231]
- 55. Effect of nitrogen fertilization on the abundance ... [https://www.sciencedirect.com/science/article/abs/pii/S0038071718302864]
- 56. Metagenomic analysis of nitrogen‐cycling genes in upper ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6528593/]
- 57. Nitrogen cycling and microbial cooperation in the terrestrial ... [https://www.nature.com/articles/s41396-022-01300-0]
- 58. High-recovery and chemical-free desalination of sodium ... [https://www.sciencedirect.com/science/article/pii/S001191642500195X]
- 59. Recent Advances in Organic Pollutant Removal ... [https://www.mdpi.com/2073-4441/17/16/2494]
- 60. How to Scale-up Chemical Reactions/ Runaway ... [https://www.fauske.com/blog/how-to-scale-up-chemical-reactions-runaway-reactions-in-a-safer-way]
- 61. The scaling technology in nuclear reactor thermal hydraulic [https://www.sciencedirect.com/science/article/abs/pii/S0306454921003169]
- 62. Reactor Stability Analysis → Term [https://energy.sustainability-directory.com/term/reactor-stability-analysis/]
- 63. 利用Elementar的穩定同位素分析儀,開展氮循環對陸地生態 ... [https://www.meritscience.com.tw/portal_b1_page.php?owner_num=b1_56051&button_num=b1&cnt_id=18673]
- 64. Rocket Test Proves Bacteria Survive Space Launch And Re ... [https://astrobiology.com/2025/10/rocket-test-proves-bacteria-survive-space-launch-and-re-entry-unharmed.html]
- 65. Effects of spaceflight and simulated microgravity on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5971428/]
- 66. The Impacts of Microgravity on Bacterial Metabolism - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9225508/]
- 67. Impact of microgravity on space microbes and application ... [https://www.sciencedirect.com/science/article/abs/pii/S0094576525004941]
- 68. Microbial Evolution in Microgravity or Space: Understanding ... [https://publishing.emanresearch.org/Journal/FullText/6186]
- 69. Long-term legacy impacts of nitrogen fertilization on crop ... [https://www.sciencedirect.com/science/article/abs/pii/S1161030125000097]
- 70. Global mean nitrogen recovery efficiency in croplands can ... [https://www.nature.com/articles/s41467-025-62095-0]
- 71. Enhanced efficiency fertilizers: Overview of production ... [https://www.sciencedirect.com/science/article/pii/S2950305124000068]
- 72. Types Of Fertilizers: Different Compositions, Origins, And ... [https://eos.com/blog/types-of-fertilizers/]
- 73. Assessing crop nitrogen status under organic amendments ... [https://link.springer.com/article/10.1007/s10705-025-10431-7]
- 74. Global mean nitrogen recovery efficiency in croplands can ... [https://www.nature.com/articles/s41467-023-41504-2]
- 75. MELiSSA SPACE RESEARCH PROGRAM [https://www.melissafoundation.org/page/melissa-project]
- 76. ESA - MELiSSA life support project, an innovation network ... [https://www.esa.int/Enabling_Support/Space_Engineering_Technology/MELiSSA_life_support_project_an_innovation_network_in_support_to_space_exploration]
- 77. Integration of Nitrifying, Photosynthetic and Animal ... [https://www.frontiersin.org/journals/astronomy-and-space-sciences/articles/10.3389/fspas.2021.750616/full]
- 78. Dynamics of long‐term continuous culture of Limnospira ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8913870/]
- 79. Nitrification [https://www.melissafoundation.org/page/nitrification]
- 80. MELiSSA Pilot Plant Regenerative Life Support System [https://ttu-ir.tdl.org/items/a9cc0b34-c3ce-4d5c-99dd-20c8e1e0efce]
- 81. Implementing integrated crop-livestock management systems [https://foodforwardndcs.panda.org/food-production/implementing-integrated-crop-livestock-management-systems/]
- 82. Environmental effects of improved regional nitrogen cycling ... [https://www.sciencedirect.com/science/article/pii/S0308521X24003949]
- 83. How to make regenerative practices work on the farm [https://www.sciencedirect.com/science/article/pii/S0308521X22000075]
- 84. Regenerative Agriculture can play a key role in combating ... [https://www.oneearth.org/regenerative-agriculture-can-play-a-key-role-in-combating-climate-change/]
- 85. The Nitrogen Problem in Agriculture [https://regenerationinternational.org/2022/07/11/the-nitrogen-problem-in-agriculture/]
- 86. Nitrogen cycling in pastoral livestock systems in Sub‐Saharan ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8459270/]
- 87. Editor's note: This page was last updated on 16 June [https://www.esa.int/Enabling_Support/Space_Engineering_Technology/Proba_Missions/Proba-3_Frequently_Asked_Questions]
- 88. Isro delays SpaDeX docking to Jan 9 for validation by ground ... [https://www.business-standard.com/india-news/isro-delays-spadex-docking-to-jan-9-for-validation-by-ground-simulations-125010600619_1.html]
- 89. Green light and nitrogen: Optimizing antioxidant production ... [https://www.sciencedirect.com/science/article/abs/pii/S2214552425000689]
- 90. Redistribution of nitrogen to feed the people on a safer planet [https://pmc.ncbi.nlm.nih.gov/articles/PMC11093128/]
- 91. Towards supervisory Model Predictive Control for circular ... [https://www.sciencedirect.com/science/article/pii/S2214552425000707]
- 92. Controlled ecological life-support system [https://grokipedia.com/page/Controlled_ecological_life-support_system]
- 93. the MAGICC coupled carbon–nitrogen cycle model v1.0 - GMD [https://gmd.copernicus.org/articles/18/2193/2025/]
- 94. Nitrogen cycling and functional gene diversity of drinking ... [https://www.sciencedirect.com/science/article/pii/S1470160X25002808]
- 95. Aerosol iodide accelerates reactive nitrogen cycling in the ... [https://www.nature.com/articles/s41467-025-63420-3]
- 96. A beginner's guide to implementation science - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12348288/]
- 97. Use of Photobioreactors in Regenerative Life Support Systems for Human Space Exploration - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8281973/]