FRET Biosensors for Plant Analytics: A Comparative Guide to Principles, Applications, and Advanced Methodologies
This article provides a comprehensive comparison of Fluorescence Resonance Energy Transfer (FRET)-based biosensors for monitoring diverse plant analytes, including hormones, ions, and metabolites.
FRET Biosensors for Plant Analytics: A Comparative Guide to Principles, Applications, and Advanced Methodologies
Abstract
This article provides a comprehensive comparison of Fluorescence Resonance Energy Transfer (FRET)-based biosensors for monitoring diverse plant analytes, including hormones, ions, and metabolites. Tailored for researchers and scientists, it covers the foundational principles of FRET sensor design, detailed methodologies for real-time analysis of targets like abscisic acid (ABA), phosphate, and ATP, strategies for troubleshooting and optimizing sensor performance, and a critical validation of current sensor technologies. By synthesizing the latest advances, this review serves as an essential resource for selecting, developing, and applying FRET sensors to illuminate dynamic physiological processes in plants with high spatiotemporal resolution.
Understanding FRET Biosensors: Core Principles and the Plant Analyte Landscape
Förster Resonance Energy Transfer (FRET) is a powerful physical phenomenon that allows scientists to measure distances at the molecular scale, functioning as a "spectroscopic ruler." [1] This capability is fundamental to its application in studying biological processes, including the analysis of various analytes in plant research. The mechanism is based on a distance-dependent energy transfer from an excited donor fluorophore to an acceptor fluorophore, providing a window into molecular interactions, conformational changes, and cellular dynamics that are otherwise invisible to conventional microscopy. [2] [3]
The Core Principle of the FRET Ruler
The operation of FRET as a molecular ruler hinges on a single, critical relationship: the efficiency of energy transfer (E) is inversely proportional to the sixth power of the distance (r) between the donor and acceptor fluorophores. [4]
The relationship is quantitatively described by the equation: E = 1 / [1 + (r/R₀)⁶]
Here, R₀ is the Förster radius, a characteristic distance for each specific donor-acceptor pair at which the energy transfer efficiency is 50%. [5] [4] This sharp distance dependence makes FRET exquisitely sensitive to changes in the nanometer range.
Essential Conditions for FRET
For this molecular ruler to work, three primary conditions must be met simultaneously [5] [1]:
- Close Proximity: The donor and acceptor must be within 1–10 nanometers of each other, a scale comparable to the size of biological macromolecules. [5]
- Spectral Overlap: The emission spectrum of the donor must significantly overlap with the absorption (excitation) spectrum of the acceptor. This overlap is quantified by the spectral overlap integral, J(λ). [5] [4]
- Favorable Dipole Orientation: The transition dipole moments of the donor and acceptor must be approximately parallel. The degree of alignment is quantified by the orientation factor, κ², which can range from 0 (perpendicular) to 4 (parallel). [1] [4]
The Förster radius (R₀) consolidates these factors into a single value, calculated as: R₀⁶ ∝ (κ² * QD * J) / n⁴
Where:
- κ² is the orientation factor (often assumed to be 2/3 for dynamically rotating fluorophores). [4]
- QD is the quantum yield of the donor in the absence of the acceptor. [4]
- J is the spectral overlap integral. [4]
- n is the refractive index of the medium. [4]
Quantitative Comparison of Common FRET Pairs
The choice of donor-acceptor pair is critical, as its inherent R₀ value determines the effective distance range over which the FRET ruler can reliably measure. The table below compares the key parameters of commonly used fluorophore pairs.
Table 1: Characteristics of Common Donor-Acceptor FRET Pairs
| Donor | Acceptor | Förster Radius (R₀) in Å | Effective Distance Range | Key Applications & Notes |
|---|---|---|---|---|
| Fluorescein | Tetramethylrhodamine | 55 [5] | ~3-8 nm | Classic organic dye pair; used in immunoassays and nucleic acid detection. [5] |
| IAEDANS | Fluorescein | 46 [5] | ~2.5-7 nm | Used in protein structure and conformation studies. [5] |
| EDANS | Dabcyl | 33 [5] | ~2-5 nm | Common pair for molecular beacons and protease assays (quencher acceptor). [5] |
| BODIPY FL | BODIPY FL | 57 [5] | ~3-8.5 nm | Homo-FRET pair; useful for fluorescence depolarization studies. [5] |
| CFP (e.g., mCerulean3) | YFP (e.g., Venus) | ~49-52 [6] | ~3-7 nm | Genetically encoded; widely used in live-cell biosensors (e.g., Cameleon for calcium). [7] [6] |
| GFP (e.g., Clover) | RFP (e.g., mRuby2) | ~54-59 [6] | ~3-8.5 nm | Genetically encoded; improved photostability and brightness for live-cell imaging. [7] [6] |
Experimental Measurement of FRET Efficiency
To read the molecular ruler, several well-established methods can be employed to quantify the FRET efficiency, each with its own strengths and limitations. [4] [6]
Table 2: Comparison of Key Methods for Measuring FRET Efficiency
| Method | Principle | Suitable for Live Cells? | Temporal Resolution | Key Advantage |
|---|---|---|---|---|
| Sensitized Emission (seFRET) [6] | Measures the increase in acceptor emission upon donor excitation. | Yes [6] | Millisecond [6] | Fast, suitable for kinetic studies and high-throughput screening. [6] |
| Acceptor Photobleaching (apFRET) [4] | Measures the increase in donor fluorescence after permanently bleaching the acceptor. | No [6] | Minutes (time of photobleaching) | Conceptually simple and can be performed on standard microscopes. [4] |
| Fluorescence Lifetime Imaging (FLIM-FRET) [4] [6] | Measures the decrease in the donor's fluorescence lifetime in the presence of the acceptor. | Yes [6] | Second to minute [6] | Highly quantitative and independent of fluorophore concentration. [4] |
| Spectral Imaging FRET (siFRET) [6] | Records full emission spectra of donor and acceptor to calculate energy transfer. | Yes [6] | Second [6] | Provides detailed spectral information for robust calculation. [6] |
FRET Sensor Design for Plant Analyte Research
The fundamental ruler mechanism is harnessed by embedding donor-acceptor pairs into biosensors that respond to specific biological changes. In plant research, two primary sensor designs are employed:
Detailed Experimental Protocol: Monitoring Phosphate Dynamics in Plants
The following protocol, adapted from a 2025 study, exemplifies how a FRET-based biosensor is used in practice to measure analyte levels in plant cells with high spatial and temporal resolution. [8]
Objective: To quantify intracellular phosphate (Pi) levels in the cytosol and plastids of Brachypodium distachyon roots during arbuscular mycorrhizal symbiosis using the FRET biosensor cpFLIPPi-5.3m. [8]
Key Research Reagent Solutions:
| Reagent | Function in the Experiment |
|---|---|
| cpFLIPPi-5.3m biosensor | The intramolecular FRET sensor; binding of Pi induces a conformational change that alters FRET efficiency. [8] |
| cpFLIPPi-Null control sensor | A mutated version that does not bind Pi; controls for non-Pi specific changes in the cellular environment. [8] |
| eCFP & cpVenus plasmids | Controls for quantifying spectral bleed-through and cross-excitation during image analysis. [8] |
| Constitutive (ZmUb1) & Inducible (BdPT7) Promoters | Drive sensor expression in all cell types or specifically in arbuscule-containing cells, respectively. [8] |
| Arbuscular Mycorrhizal Fungi (e.g., Diversispora epigaea) | Establish symbiosis to study Pi transfer from fungus to plant. [8] |
Methodology:
- Plant Material and Transformation: Generate transgenic Brachypodium distachyon lines expressing the cpFLIPPi-5.3m sensor and its controls (cpFLIPPi-Null, eCFP, cpVenus) targeted to the cytosol or plastids using either the constitutive ZmUb1 promoter or the arbuscule-specific BdPT7 promoter. [8]
- Growth and Inoculation: Grow transgenic plants in association with AM fungi (e.g., Diversispora epigaea) to induce the formation of arbuscules, the site of nutrient exchange. [8]
- Image Acquisition: Image live roots using a confocal microscope. For the cpFLIPPi-5.3m sensor (CFP-YFP pair), excite with a 458 nm laser line and collect emission signals for CFP (465–500 nm) and FRET (520–555 nm) channels. [8]
- Sensitized FRET Analysis: Process the acquired images using a semi-automated ImageJ macro. The macro corrects for background noise, spectral bleed-through (donor emission detected in the acceptor channel), and cross-excitation (direct excitation of the acceptor by the donor's laser line). [8]
- Data Quantification: Calculate the sensitized FRET ratio, which is proportional to the intracellular Pi concentration. The use of the control sensor (cpFLIPPi-Null) is critical to confirm that observed FRET ratio shifts are due to genuine Pi fluctuations and not other ionic or environmental changes. [8]
Comparative Performance in Plant Analyte Sensing
The versatility of the FRET ruler is demonstrated by its adaptation to monitor a wide range of biologically critical analytes in plants. The table below compares the performance of different FRET-based sensors.
Table 3: Comparison of FRET-based Biosensors for Plant Analytes
| Target Analyte | Example Sensor Name | Donor-Acceptor Pair | Research Context | Key Findings & Performance |
|---|---|---|---|---|
| Phosphate (Pi) | cpFLIPPi-5.3m [8] | CFP-Venus (YFP variant) [8] | Pi dynamics in arbuscular mycorrhizal symbiosis in Brachypodium. [8] | Successfully revealed variation in Pi levels in cortical cells with arbuscules at different developmental stages. [8] |
| Adenosine Triphosphate (ATP) | ATeam1.03-nD/nA [9] | mseCFP-cp173-mVenus [9] | ATP dynamics in Arabidopsis thaliana during low-oxygen stress (submergence). [9] | Detected a gradual decrease in cytosolic ATP levels under oxygen limitation; ratio was reversible upon re-oxygenation. [9] |
| Calcium (Ca²⁺) | Cameleon [3] [6] | CFP-YFP [3] [6] | General calcium signaling in live cells. [3] | One of the first FP-based FRET biosensors; improved versions using ECFP and EYFP yielded higher signals. [3] |
| pH | pHluorins [3] | - (Single FP, dual excitation) [3] | Acidity of intracellular compartments. [3] | Exhibits a shift in excitation peak from 470 nm to 410 nm as pH decreases. [3] |
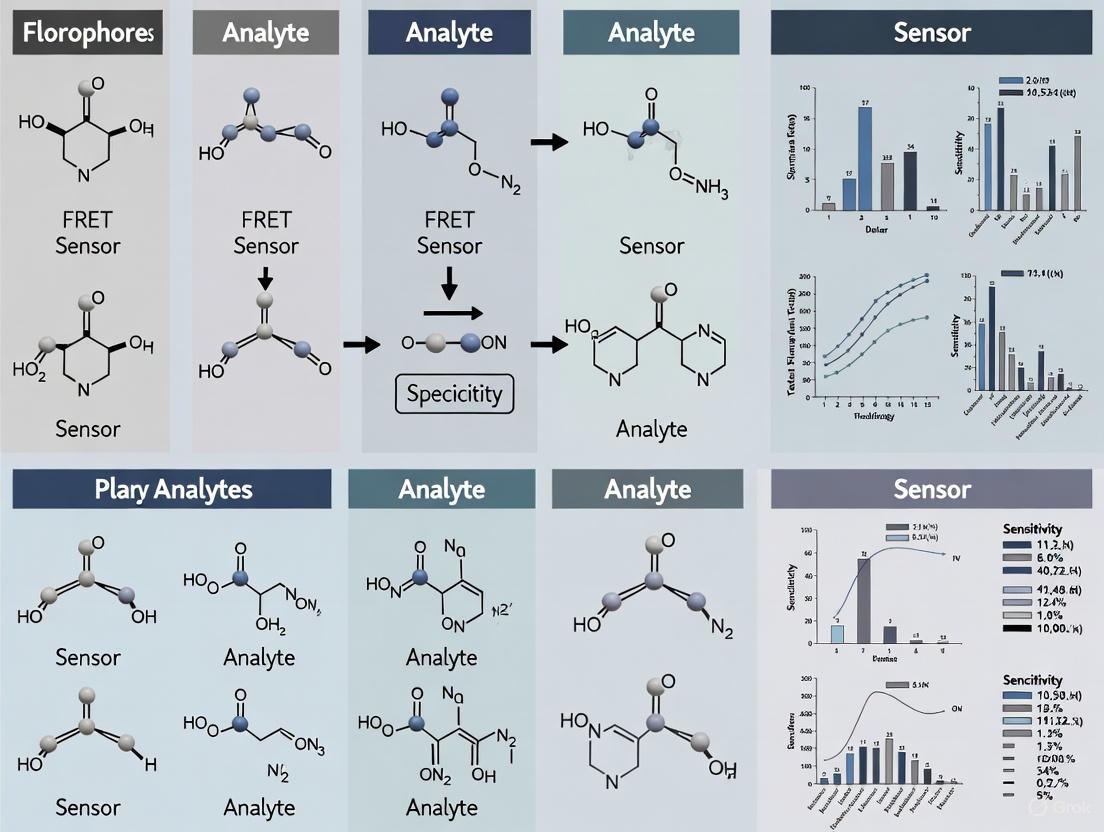
Förster Resonance Energy Transfer (FRET)-based biosensors are powerful tools for visualizing and quantifying biochemical events in live cells with high spatiotemporal resolution. These genetically encoded sensors function as molecular spies, reporting on dynamic cellular processes by converting molecular recognition into a measurable fluorescence signal [1]. The fundamental architecture of a FRET biosensor is modular, comprising three core components: a sensory domain that interacts with the target analyte, a pair of fluorophores that act as the donor and acceptor in the FRET process, and a linker that connects these elements and transduces conformational changes [6] [10]. The performance of a biosensor—its sensitivity, dynamic range, and specificity—is critically dependent on the careful selection and engineering of each of these components. This guide provides a comparative analysis of these key design elements, underpinned by experimental data, to inform their rational selection and optimization for specific research applications, particularly in the context of plant analyte research [11].
Comparative Analysis of Fluorophore Pairs
The choice of fluorophore pair is paramount, as it directly determines the FRET efficiency and the signal-to-noise ratio of the biosensor. The efficiency of energy transfer is highly sensitive to the distance between the donor and acceptor (typically 1-10 nm) and requires substantial spectral overlap between the donor's emission and the acceptor's excitation spectra [6] [2] [3].
Table: Comparison of Common Fluorescent Protein (FP) FRET Pairs
Table 1: Key characteristics of commonly used FRET pairs based on Fluorescent Proteins (FPs). The Förster radius (R₀) is the distance at which FRET efficiency is 50%.
| FRET Pair (Donor-Acceptor) | Förster Radius (R₀) | Key Advantages | Key Limitations | Example Applications |
|---|---|---|---|---|
| CFP/YFP (e.g., Cerulean/Citrine) | ~50 Å [10] | Historically well-characterized; widely used in early biosensors [3]. | Relatively low dynamic range; spectral cross-talk can be an issue [6] [10]. | Cameleon calcium sensors [3]. |
| mseCFP/mVenus (in ATeam sensors) | Not explicitly stated | Engineered for high quantum yield and photostability [12]. | Affinity of the sensory domain (ε subunit) can be modulated by attached FPs [12]. | Intracellular ATP sensing (ATeam) [12]. |
| EGFP/mCherry | Not explicitly stated | Red-shifted pair; can be used with CFP/YFP for multiplexing; compatible with conventional confocal microscopy [13]. | Requires validation for each new sensor construct [13]. | Actinin tension sensors [13]. |
Beyond traditional FPs, quantum dots (QDs) and organic dyes are also employed. QDs offer high brightness and photostability and are often used in FRET-based nano-biosensors. For instance, cadmium telluride (CdTe) QDs paired with rhodamine dyes have been used to detect the Citrus tristeza virus in plants [11]. Organic dyes like Cy3, Cy5, and the BHQ quencher series provide high FRET efficiency but generally require chemical conjugation, unlike genetically encodable FPs [2] [14].
Sensory Domains and Sensor Architecture
The sensory domain is the biorecognition element that confers specificity to the biosensor. Its interaction with the target analyte must induce a robust conformational change that can be mechanically transmitted to the fluorophore pair via the linkers.
Sensor Topologies and Operational Mechanisms
FRET biosensors are primarily categorized into two operational types:
- Intermolecular FRET Sensors: The donor and acceptor fluorophores are fused to two different proteins. FRET occurs when these two proteins interact and come into close proximity, making this design ideal for monitoring protein-protein interactions [1] [6].
- Intramolecular FRET Sensors: Both the donor and acceptor fluorophores are conjoined to the same sensory molecule. A conformational change in the sensory domain, induced by analyte binding, alters the distance and/or orientation between the fluorophores, thereby changing the FRET efficiency. This design is used for sensing ions, metabolites, and enzyme activities [6] [8].
A key design strategy for intramolecular sensors involves engineering mutually exclusive domain interactions. In this approach, the donor and acceptor FPs are designed to interact with each other in one state of the sensor (e.g., analyte-free), leading to high FRET. Analyte binding then triggers a conformational change that disrupts this FP interaction, leading to a large decrease in FRET and a high dynamic range. This principle has been successfully applied in sensors for Zn²⁺ (CALWY sensors) and bile acids [10].
Table: Sensory Domains and Their Applications
Table 2: Examples of sensory domains used in FRET biosensors for various analytes.
| Sensory Domain / Principle | Target Analyte / Process | Induced Conformational Change | Documented Dynamic Range |
|---|---|---|---|
| Calmodulin & M13 peptide [3] | Ca²⁺ | Ca²⁺-dependent binding alters FP proximity. | ~1.6-fold in early cameleons [3]. |
| ATP synthase ε subunit [12] | ATP | ATP binding induces conformational change. | Affinity tuned via mutagenesis (e.g., R103A/R115A) [12]. |
| Ligand-binding domain (LBD) of FXR [10] | Bile Acids | Bile acid binding recruits LXXLL motif, disrupting FP interaction. | 2-fold decrease in emission ratio in vitro [10]. |
| Actinin [13] | Cellular Tension | Mechanical stretch alters the spacing between integrated FPs. | FRET strain sensitivity of -0.64 in aorta [13]. |
| cpFLIPPi-5.3m [8] | Phosphate (Pi) | Pi binding induces a conformational shift. | Measured as a change in FRET ratio via confocal microscopy [8]. |
The Critical Role of Linker Design
Linkers are the mechanical bridges that translate a chemical or physical event in the sensory domain into a change in FRET efficiency. Their length, flexibility, and composition are critical for achieving a high dynamic range [10].
- Length and Flexibility: Optimal linker length is context-dependent. Excessively long, flexible linkers can attenuate the transmission of conformational changes, while very short, rigid linkers may impede the proper folding of the domains or the necessary structural transition. Systematic variation of linker length is a common strategy to optimize sensor performance [10] [12].
- Rational Engineering: The "sticky" FP approach, which uses FPs engineered to weakly dimerize (e.g., with S208F/V224L mutations), creates a competition between FP interaction and the ligand-induced conformational change. This strategy can dramatically increase the dynamic range, as demonstrated by the 6-fold improvement in the CALWY Zn²⁺ sensor [10].
- Tethering in Extracellular Sensors: For cell-surface displayed sensors, the linker connecting the biosensor to the membrane anchor is crucial. Optimizing this tether length was a key factor in developing the high-affinity ECATS2 extracellular ATP sensor [12].
Experimental Protocol: Measuring FRET to Evaluate Sensor Performance
This section outlines a standard protocol for quantifying FRET efficiency in living cells using a confocal microscope, which is essential for characterizing any newly developed or implemented FRET biosensor.
Workflow: Sensitized Emission FRET Measurement
Step 1: Sample Preparation. Introduce the FRET biosensor into your model system. For plant research, this typically involves generating stable transgenic lines (e.g., Brachypodium distachyon as described for phosphate sensing) or transient transformation methods [8]. For cell culture, use transfection or viral transduction (e.g., Adenovirus for the ECATS2 ATP sensor) [12].
Step 2: Image Acquisition. Acquire images using a confocal or fluorescence microscope configured for the specific FRET pair. For a CFP/YFP-based sensor, this involves [8] [12]:
- Donor channel: Excite with a 438/29 nm laser line and collect emission at 470/24 nm.
- Acceptor channel: Excite with a 510/10 nm laser line and collect emission at 540/30 nm.
- FRET (sensitized emission) channel: Excite with the donor laser line (438/29 nm) and collect emission using the acceptor filter (540/30 nm).
Step 3: Image Correction. The FRET channel contains signal from direct donor emission (spectral bleed-through, SBT) and direct acceptor excitation. This must be corrected using control cells expressing only the donor or acceptor fluorophore to establish correction factors [8] [13].
Step 4: Calculate the FRET Ratio. After correction, the sensitized FRET emission is calculated. The most common quantitative readout is the emission ratio, calculated as the background-subtracted intensity of the corrected FRET channel (acceptor emission) divided by the intensity of the donor channel [8] [12]. This ratiometric measurement is independent of the sensor's concentration and laser intensity, allowing for robust comparisons.
Step 5: Analyze Dynamics. Monitor the FRET ratio over time in response to experimental stimuli. For example, change in FRET ratio can be correlated with applied mechanical strain to calculate FRET strain sensitivity (%FRET per %strain) [13], or with changes in analyte concentration to determine the sensor's affinity and dynamic range [12].
Research Reagent Solutions
The following table lists key reagents and materials essential for developing and implementing FRET biosensors, as cited in the literature.
Table 3: Essential research reagents and materials for FRET biosensor work.
| Reagent / Material | Function / Description | Example Use Case |
|---|---|---|
| Transgenic Brachypodium distachyon lines [8] | Biological material expressing FRET biosensor from constitutive or tissue-specific promoters. | Monitoring intracellular phosphate dynamics during arbuscular mycorrhizal symbiosis [8]. |
| FRET Biosensor Constructs (e.g., cpFLIPPi-5.3m, ECATS2, actinin tension sensor) | The genetically encoded sensor itself, targeting specific analytes or forces. | Detection of phosphate (Pi), extracellular ATP, and cellular tension, respectively [8] [13] [12]. |
| AM Fungal Spores (Diversispora epigaea, Rhizophagus irregularis) [8] | To establish symbiotic conditions in plant studies. | Used in the experimental protocol for studying phosphate transfer in mycorrhizal roots [8]. |
| Chemical Stimulants (e.g., Calyculin A, Y27632) [13] | Pharmacological agents to modulate cellular activity and test sensor response. | Calyculin A increases cellular tension (decreases FRET), while Y27632 decreases tension (increases FRET) in actinin sensors [13]. |
| Adenovirus Expression System [12] | For efficient delivery and expression of biosensor constructs in mammalian cells, including primary cultures. | Transduction of cortical astrocytes with the ECATS2 ATP sensor [12]. |
| Custom Isotonic Imaging Solution [12] | A defined buffer for maintaining cell viability during live-cell imaging experiments. | Used for equilibration and perfusion during ATP sensing and hypoosmotic stress experiments [12]. |
Plants rely on a complex language of small molecules—including hormones, ions, and metabolites—to coordinate growth, development, and stress responses. Understanding this chemical language requires tools that can track these dynamic changes in real-time within living plants. Among the most powerful tools developed for this purpose are Förster Resonance Energy Transfer (FRET)-based biosensors. These genetically encoded sensors translate the concentration of a specific analyte into a measurable change in fluorescence, allowing researchers to visualize analyte dynamics with high spatiotemporal resolution directly in plant cells and tissues [15] [16].
This guide provides a comparative analysis of FRET biosensors for key plant analytes, detailing their performance characteristics, experimental protocols, and practical applications to inform research and development in plant science.
Fundamental Operating Principle
FRET biosensors are engineered fusion proteins that undergo a conformational change upon binding a target analyte. This change alters the efficiency of energy transfer from a donor fluorescent protein to an acceptor fluorescent protein.
- Sensor Architecture: A typical FRET biosensor consists of a sensory domain specific to the target analyte, flanked by two fluorescent proteins that form a FRET pair (e.g., a cyan/yellow FP pair) [16].
- Ligand-Induced Conformational Change: Analyte binding induces a shift in the sensory domain's structure, changing the distance and/or orientation between the donor and acceptor FPs.
- Ratiometric Readout: The resulting change in FRET efficiency is measured as a ratio of acceptor-to-donor fluorescence. This ratiometric output is internally controlled, minimizing artifacts from variations in sensor concentration or excitation intensity [12].
Comparative Advantages of FRET Biosensors
- High Specificity: The sensory domain can be engineered for high specificity, as demonstrated by the nitrate sensor NiMet3.0, which shows no response to other anions or nitrogen forms like ammonium [17].
- Quantitative Capability: Sensors can be calibrated to determine dissociation constants (Kd), enabling quantitative concentration measurements. For instance, the NitraMeter3.0 has a Kd of ~90 µM for nitrate [17].
- Non-Invasiveness and Real-Time Monitoring: They enable live-cell imaging over extended periods, allowing observation of rapid analyte dynamics, such as the ultrafast (<1 second) response of the RHSY@N-GQDs probe to salicylic acid [18].
Figure 1: FRET Biosensor Operating Principle. Analyte binding induces a conformational change in the sensory domain, altering the distance/orientation between donor and acceptor fluorescent proteins (FPs) and modulating FRET efficiency.
Comparative Performance Analysis of Plant FRET Biosensors
The following tables summarize the key performance metrics of FRET biosensors for major plant analytes, including hormones, ions, and metabolites.
Table 1: FRET Biosensors for Plant Hormones
| Analyte | Biosensor Name | Sensory Domain / Mechanism | Detection Range / Affinity (Kd) | Key Applications & Findings | Notable Features |
|---|---|---|---|---|---|
| Abscisic Acid (ABA) | ABAleon [16] | ABA receptor PYR/PYL & PP2C | Not specified | ABA distribution and changes in Arabidopsis; transport into tissues [16]. | Among the first FRET biosensors for any plant hormone; based on receptor-coactivatior interaction. |
| Abscisic Acid (ABA) | ABACUS [16] | ABA receptor PYR/PYL & PP2C | Multiple variants with Kd from 100 nM to 1.1 µM | ABA dynamics in roots; correlation with stomatal closure [16]. | Series of sensors with varying affinities to monitor different ABA concentration ranges. |
| Salicylic Acid (SA) | RHSY@N-GQDs [18] | Rhodamine 6G derivative (RHSY) & N-doped GQDs | Response < 1 second | Dynamic monitoring of SA transport; induced stomatal closure (aperture decreased from ~9.1 µm to ~3.7 µm) [18]. | Dual-channel ratiometric probe; overcomes aggregation-caused quenching (ACQ). |
| Cytokinin (CK) | TCSn/TCSv2 [15] | Synthetic promoter with ARR-binding sites | N/A (Transcriptional Reporter) | Tracking transcriptional cytokinin responses [15]. | An indirect transcriptional reporter; included for context alongside direct FRET sensors. |
Table 2: FRET Biosensors for Ions and Metabolites
| Analyte | Biosensor Name | Sensory Domain / Mechanism | Detection Range / Affinity (Kd) | Key Applications & Findings | Notable Features |
|---|---|---|---|---|---|
| Nitrate (NO₃⁻) | NitraMeter3.0 (NiMet3.0) [17] | NasR protein (NIT domain) from bacteria | Kd ~90 µM; max >1 mM [17]. | Spatiotemporal distribution along root axis; disruptions in transport/assimilation mutants [17]. | First genetically encoded biosensor for quantitative NO₃⁻ visualization at cellular level. |
| Extracellular ATP | ecATeam3.10 [12] | ATP synthase ε subunit from Bacillus PS3 | Micromolar range (optimal 10-100 µM) [12]. | Detection of ATP release upon hypoosmotic stress in cultured astrocytes [12]. | Ratiometric readout normalizes for expression level. |
| Extracellular ATP | ECATS2 [12] | Mutated (R103A/R115A) ε subunit | >3x higher affinity vs. ecATeam3.10; Kd ~0.2 µM (purified) [12]. | Detection of low, physiologically relevant (nM-µM) ATP levels [12]. | Second-generation sensor with enhanced affinity via binding site mutagenesis. |
| Calcium (Ca²⁺) | Cameleon [16] | Calmodulin & M13 peptide | N/A | N/A | Historical context; pioneered the conceptual framework for many subsequent FRET biosensors. |
Experimental Protocols for Key Biosensor Applications
Protocol: Visualizing Nitrate Dynamics with NitraMeter3.0 in Arabidopsis Roots
This protocol is adapted from the development and use of the NitraMeter3.0 FRET biosensor to monitor nitrate distribution in plant roots [17].
- Plant Material and Growth: Generate stable transgenic Arabidopsis thaliana lines expressing NitraMeter3.0 under the control of a constitutive promoter like CaMV35S. Germinate and grow seedlings for 6 days under sterile conditions on nitrogen-free agar medium.
- Microscopy Setup: Use a confocal or epifluorescence microscope capable of FRET imaging. Configure lasers and filters for the CFP/YFP FRET pair (e.g., excite with a 438 nm laser and collect emissions at 470-500 nm for CFP and 520-550 nm for YFP).
- Image Acquisition and Ratio Calculation:
- Mount seedlings in a custom imaging chamber with the root immersed in a low-salt buffer.
- Acquire baseline images of the CFP and FRET (YFP emission upon CFP excitation) channels.
- Perfuse the chamber with a solution containing a pulse of exogenous nitrate (e.g., KNO₃).
- Acquire time-lapse image series post-stimulation.
- Generate ratiometric images by calculating the FRET/CFP emission ratio on a pixel-by-pixel basis using image analysis software like ImageJ/FIJI.
- Controls: Always image under identical conditions. Include transgenic lines expressing a non-responsive control sensor (e.g., NiMet3.0-R176A, which has a mutated nitrate-binding pocket) to confirm the specificity of the observed ratio changes [17].
Protocol: Monitoring Salicylic Acid with a Ratiometric Chemical Probe
This protocol outlines the use of the RHSY@N-GQDs nanoprobe for detecting salicylic acid (SA) in plant tissues [18].
- Probe Preparation: Synthesize the RHSY@N-GQDs probe by coupling nitrogen-doped graphene quantum dots (N-GQDs) with the SA-responsive rhodamine 6G derivative (RHSY) via a simple coupling reaction. Characterize the probe using TEM, XPS, and fluorescence spectroscopy [18].
- Plant Treatment and Imaging:
- Apply the dispersed RHSY@N-GQDs probe to plant leaves or roots, allowing it to enter through the surface.
- Use a fluorescence microscope with appropriate filter sets for green (N-GQDs reference signal) and yellow (RHSY SA-responsive signal) channels.
- Before SA application, the probe exhibits green emission. Upon SA addition, monitor the rapid (<1 second) fluorescence shift from green to yellow, indicating SA-induced aggregation and FRET.
- Quantitative Analysis: Calculate the ratiometric signal (yellow/green). Correlate the signal intensity and spatial distribution with physiological responses, such as the decrease in stomatal aperture (e.g., from ~9.11 µm to ~3.71 µm), to validate SA activity [18].
Figure 2: General Workflow for FRET Biosensor Experiments in Plants. The process involves sample preparation, baseline imaging, stimulus application, and ratiometric analysis to track analyte dynamics.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for FRET Biosensor Studies
| Reagent / Material | Function and Role in Experimentation |
|---|---|
| Gateway Cloning System | A highly efficient, site-specific recombination-based method used for constructing FRET biosensor expression vectors (e.g., used for NitraMeter3.0) [17]. |
| Genetically Encoded Biosensors (e.g., ABAleon, NiMet3.0) | The core reagent; a fusion protein that transduces analyte concentration into a fluorescent readout when expressed in transgenic plants [17] [16]. |
| Fluorescent Chemical Probe (e.g., RHSY@N-GQDs) | A synthetic nanosensor applied exogenously to detect specific analytes like salicylic acid, useful for studies in non-transgenic systems [18]. |
| Non-Responsive Mutant Sensor (e.g., NiMet3.0-R176A) | A critical control sensor with a mutated binding pocket; it accounts for non-specific environmental effects, validating that signal changes are due to specific analyte binding [17]. |
| Adenovirus Expression Vectors | Used for high-efficiency delivery and transduction of biosensor genes into difficult-to-transfect cell types, such as primary astrocytes [12]. |
| ImageJ/FIJI Software | Open-source image analysis platform essential for background subtraction, cell masking, ratio image calculation, and quantification of FRET data [12]. |
The expanding toolkit of FRET biosensors, encompassing both genetically encoded and chemical probes, has fundamentally transformed the plant sciences. These tools allow researchers to move beyond static, destructive measurements to dynamic, spatially resolved visualization of hormonal fluxes, nutrient distributions, and metabolic signaling in living plants.
Current trends point toward continued refinement—including higher-affinity sensors like ECATS2, the use of brighter fluorophores to improve the signal-to-noise ratio, and the development of robust sensors for a wider array of metabolites [12]. The ultimate goal is a comprehensive "sensor suite" capable of simultaneously monitoring multiple analytes, providing an integrated view of the complex chemical networks that govern plant life. This progress will be instrumental in addressing grand challenges in agriculture and biotechnology, from engineering stress-resilient crops to optimizing plant productivity.
Genetically encoded biosensors based on Förster resonance energy transfer (FRET) have revolutionized the study of plant biology by enabling real-time, non-invasive monitoring of analytes with high spatiotemporal resolution. These sophisticated molecular tools function as "spectroscopic rulers," exploiting distance-dependent energy transfer between donor and acceptor fluorophores to report on dynamic cellular processes [1]. The evolution from first-generation to next-generation FRET biosensors represents a remarkable journey of scientific innovation, addressing critical challenges in affinity, specificity, and signal-to-noise ratio while expanding the detectable analyte repertoire.
In plant systems, where traditional analytical methods provide limited spatial resolution and require destructive sampling, FRET biosensors have emerged as indispensable tools for deciphering the complex signaling networks that coordinate growth, development, and stress responses [15]. This comparison guide examines the performance characteristics of successive biosensor generations, focusing on key technological breakthroughs that have transformed our ability to visualize plant hormones, signaling molecules, and metabolites in living cells.
Fundamental Principles of FRET Biosensor Design
Core Mechanism and Molecular Architecture
FRET biosensors operate on the principle of non-radiative energy transfer between two fluorophores—a donor and an acceptor—when they are in close proximity (typically 1-10 nm). The efficiency of this transfer depends on several factors: the distance between fluorophores, the spectral overlap of donor emission and acceptor absorption, and their relative orientation [1]. This physical relationship is quantified by the Förster distance (R₀), which represents the separation at which energy transfer is 50% efficient [1].
The basic architecture of a FRET biosensor consists of:
- A sensing domain that specifically binds the target analyte
- Donor and acceptor fluorescent proteins (e.g., CFP/YFP variants)
- Linker regions that connect these elements and transduce binding-induced conformational changes
When the sensing domain binds its target analyte, it undergoes a conformational shift that alters the spatial relationship between the fluorophores, thereby changing FRET efficiency and producing a measurable signal change [19] [1].
Biosensor Engineering Workflow
The development of FRET biosensors follows an iterative engineering cycle that combines structural biology, molecular modeling, and empirical optimization. The following diagram illustrates this complex workflow:
Figure 1. Biosensor Engineering and Optimization Workflow. The development process involves iterative cycles of design, testing, and optimization to achieve desired biosensor characteristics. Key optimization targets include affinity, dynamic range, and orthogonality.
Evolution of FRET Biosensor Generations: Performance Comparison
First-Generation Biosensors: Foundational Technologies
First-generation FRET biosensors established the fundamental design principles and demonstrated the feasibility of real-time metabolite monitoring in living cells. These early sensors typically utilized naturally occurring binding proteins from bacterial periplasmic binding protein (PBP) superfamily members, which undergo significant conformational changes upon ligand binding [20]. For example, initial sensors for maltose, ribose, and glucose were developed by fusing sugar-binding PBPs with GFP variant pairs [20].
While revolutionary, first-generation biosensors faced significant limitations. Their dynamic range was often limited (saturating ratio change typically <0.3), making detection of subtle changes challenging [20]. Additionally, these sensors were susceptible to environmental interference, particularly from pH and halides, with yellow fluorescent protein proving especially vulnerable [20]. Perhaps most importantly, their affinity frequently mismatched physiological concentrations, with dissociation constants (K_D) typically in the micromolar range, insufficient for detecting many plant hormones that function at nanomolar concentrations [21].
Next-Generation Biosensors: Enhanced Performance and Specificity
Next-generation FRET biosensors represent significant advances through rational design and molecular engineering. Key improvements include:
- Greatly enhanced affinities through binding site mutagenesis
- Improved dynamic range via linker optimization
- Reduced interference with endogenous signaling pathways (orthogonality)
- Expanded analyte repertoire beyond naturally occurring binding proteins
These advances are exemplified by the ABACUS series of ABA biosensors. Through systematic engineering of the PYL1 sensory domain, including "latch" mutations (E141D, R143S) and linker optimization, researchers developed ABACUS2-100n with a K_D of 98 nM for ABA—an order of magnitude improvement over previous versions [21]. Simultaneously, the dynamic range increased dramatically, with ABACUS2-400n exhibiting a +71% emission ratio change in vitro [21].
Quantitative Performance Comparison
Table 1. Performance Comparison of Representative FRET Biosensors Across Generations
| Biosensor | Analyte | Generation | Affinity (K_D) | Dynamic Range | Key Innovations |
|---|---|---|---|---|---|
| ABACUS1-2µ [21] | ABA | First | ~1.1-1.8 µM | Moderate | Initial PYL1 H87P-based design |
| ABACUS2-100n [21] | ABA | Next | 98 nM | +67% ratio change | PYL1 E141D mutation, optimized linkers |
| ABACUS2-400n [21] | ABA | Next | 445 nM | +71% ratio change | PYL1 R143S mutation, high ratio change |
| ecATeam3.10 [12] | ATP | First | Micromolar | Limited | Original extracellular ATP sensor |
| ECATS2 [12] | ATP | Next | ~3x higher affinity | Maintained | R103A/R115A mutations, optimized tethering |
| ATeam1.03 [9] | ATP | First | Micromolar | Moderate | Initial intracellular ATP sensor |
Table 2. Comparison of Biosensor Optimization Strategies Across Targets
| Optimization Parameter | First-Generation Approach | Next-Generation Approach | Impact on Performance |
|---|---|---|---|
| Affinity Enhancement | Natural binding domains | Binding site mutagenesis (e.g., R103A/R115A for ATP [12], A190V for ABA [21]) | 3-4x higher affinity, detection of physiological concentrations |
| Dynamic Range Improvement | Limited rational design | Linker optimization, fluorophore truncation [21] | Up to 71% ratio change vs. <30% in early sensors |
| Orthogonality | Unmodified native domains | Domain engineering to reduce crosstalk (e.g., PYL1 S112A [21]) | Minimal perturbation of endogenous signaling |
| Cellular Targeting | Cytosolic expression | Signal peptides, membrane anchors [12] | Subcellular compartment-specific monitoring |
Experimental Protocols for Biosensor Characterization
In Vitro Affinity and Dynamic Range Assessment
Purpose: To quantitatively characterize biosensor affinity (K_D) and dynamic range before cellular implementation.
Methodology:
- Protein Purification: Express and purify biosensor protein using appropriate expression system (E. coli, yeast) [21]
- Fluorometric Titration: Measure fluorescence emission spectra across analyte concentration series
- Data Analysis:
- For FRET biosensors, calculate emission ratios (acceptor emission/donor emission)
- Plot ratio values against analyte concentration
- Fit binding curve to determine K_D using appropriate model (e.g., Hill equation) [21]
- Dynamic Range Calculation: Determine maximum ratio change: (Rmax - Rmin)/R_min × 100% [21]
Key Considerations:
- Perform measurements under physiologically relevant conditions (pH, temperature, ionic strength)
- Include control measurements to account for environmental sensitivity (e.g., pH, halides) [20]
- Validate specificity against structurally similar compounds
Cellular Validation and Imaging Protocols
Purpose: To verify biosensor functionality in living plant systems and establish imaging parameters.
Methodology:
- Plant Transformation:
- Microscopy Setup:
- Calibration:
- Perform in situ calibration using analyte clamping methods where possible
- Establish baseline and saturated sensor responses [20]
Troubleshooting:
- Autofluorescence can be addressed using spectral unmixing or two-photon microscopy [22]
- Sensor-induced phenotypes may require inducible expression systems [20]
- Subcellular localization should be confirmed via co-localization markers
Advanced Applications and Multiplexing Strategies
Multiplexed FRET Imaging
A significant advancement in next-generation biosensors is the ability to monitor multiple analytes simultaneously. This multiplexing approach provides insights into complex signaling networks and metabolic cross-talk. Implementation strategies include:
- Spectral Separation: Using FRET pairs with non-overlapping spectral characteristics (e.g., blue-green and orange-red pairs) [1]
- Temporal Separation: Exploiting differences in response kinetics through computational unmixing
- Spatial Separation: Targeting biosensors to distinct subcellular compartments
However, multiplexed imaging presents technical challenges, including spectral cross-talk, increased phototoxicity, and data deconvolution complexity [1]. Next-generation biosensors address these limitations through improved orthogonality and optimized fluorophore properties.
The Scientist's Toolkit: Essential Research Reagents
Table 3. Key Research Reagent Solutions for FRET Biosensor Implementation
| Reagent/Category | Specific Examples | Function/Application | Key Characteristics |
|---|---|---|---|
| FRET Biosensors | ABACUS2 series [21], ECATS2 [12], ATeam [9] | Target analyte detection | Genetically encoded, ratiometric readout |
| Fluorescent Proteins | mseCFP, mVenus, edCitrine, edCerulean [12] [21] | FRET donor/acceptor pairs | Brightness, photostability, reduced environmental sensitivity |
| Expression Systems | Adenovirus vectors [12], Agrobacterium [22] | Biosensor delivery | Efficient transduction, cell-type specificity |
| Imaging Platforms | CLARIOstar microplate reader [9], Confocal microscopy | Signal detection | Sensitivity, temporal resolution, environmental control |
| Reference Standards | H2B-mApple [12] | Expression normalization | Spectrally distinct, stable expression |
Signaling Pathways and Biological Insights
The application of next-generation FRET biosensors has illuminated previously opaque aspects of plant signaling pathways. The following diagram illustrates a representative signaling network that has been elucidated through biosensor deployment:
Figure 2. ABA Signaling Pathway Elucidated by FRET Biosensors. Next-generation biosensors like ABACUS2 have revealed cellular ABA dynamics driving root growth responses to foliar humidity stress, demonstrating systemic coordination between shoot and root systems [21].
The evolution from first-generation to next-generation FRET biosensors represents a paradigm shift in plant biology research, transforming our ability to monitor cellular processes with unprecedented resolution and precision. Through strategic engineering of sensing domains, optimization of linkers, and refinement of fluorescent protein pairs, next-generation biosensors offer dramatically improved affinity, dynamic range, and orthogonality.
These technological advances have enabled groundbreaking discoveries in plant signaling, such as the role of cellular ABA dynamics in coordinating root growth responses to foliar humidity stress [21] and the subcellular ATP dynamics during oxygen deprivation [9]. The continued expansion of the biosensor toolkit—including the recent development of translocation-based biosensors for MAPK signaling [23]—promises to further illuminate the complex signaling networks that govern plant life.
As biosensor technology continues to evolve, future developments will likely focus on further expanding the analyte repertoire, enhancing multiplexing capabilities, and improving compatibility with advanced imaging modalities. These innovations will undoubtedly yield new insights into plant biology and accelerate both basic research and applied biotechnology.
Sensor Deployment in Action: Methodologies and Real-World Plant Applications
Genetically Encoded Sensors for Real-Time Hormone Imaging (e.g., ABACUS for ABA, ATeam for ATP)
Genetically encoded sensors represent a revolutionary toolset in molecular and cellular biology, enabling the real-time visualization of biomolecules in living systems. Among these, Förster Resonance Energy Transfer (FRET)-based sensors have become indispensable for monitoring the dynamics of hormones, metabolites, and signaling molecules with high spatiotemporal resolution. FRET is a distance-dependent quantum mechanical phenomenon where energy transfers from an excited donor fluorophore to a nearby acceptor fluorophore without photon emission, typically occurring within 1-10 nm distances [2] [1]. This principle has been ingeniously harnessed in sensor design, where molecular recognition events trigger conformational changes that alter the distance or orientation between donor and acceptor fluorophores, resulting in measurable changes in fluorescence emission ratios [2] [1].
The field has evolved to encompass sensors for diverse analytes, with ABACUS (Abscisic Acid Concentration and Uptake Sensors) for the plant hormone abscisic acid (ABA) and ATeam for adenosine triphosphate (ATP) representing significant technological advances. These tools have transcended the limitations of traditional biochemical methods such as mass spectrometry and chromatography, which require tissue extraction and provide limited spatial and temporal resolution [24] [25]. Similarly, while transcriptional reporters and degradation-based sensors offer indirect monitoring capabilities, FRET-based sensors provide direct, quantitative measurements of analyte concentrations with minimal perturbation to native biological processes [15] [25]. This comparison guide examines the performance characteristics, experimental applications, and practical considerations of these sensor platforms to inform selection for specific research applications.
Sensor Working Principles and Design Strategies
Fundamental FRET Mechanism in Biosensing
FRET-based biosensors operate on well-established photophysical principles where energy transfer efficiency depends critically on (1) the distance between donor and acceptor fluorophores (typically within 1-10 nm), (2) the spectral overlap between donor emission and acceptor absorption spectra, and (3) the relative orientation of donor and acceptor transition dipoles [2] [1]. The efficiency of FRET (E) varies inversely with the sixth power of the distance (r) between fluorophores, described by the equation E = 1/[1 + (r/R₀)⁶], where R₀ is the Förster distance at which 50% energy transfer occurs [1]. This extreme distance sensitivity makes FRET exceptionally suitable for detecting molecular interactions and conformational changes.
In practice, intramolecular FRET sensors incorporate both donor and acceptor fluorophores within a single polypeptide chain connected by a sensory domain that undergoes conformational changes upon analyte binding [1]. The ABACUS and ATeam sensors exemplify this design philosophy, though they employ distinct strategies for molecular recognition and signal transduction as detailed in the following sections.
Comparative Schematic Designs of Major Sensor Platforms
The diagram below illustrates the fundamental structural and operational differences between ABACUS and ATeam FRET sensors:
Comparative Performance Analysis of FRET Sensors
Performance Characteristics Across Sensor Variants
Table 1: Performance characteristics of major FRET sensor families for ABA and ATP detection
| Sensor Name | Target Analyte | Affinity (Kd) | Dynamic Range | Signal Direction | Key Applications |
|---|---|---|---|---|---|
| ABACUS2-100n [21] | ABA | 98 nM | +67% emission ratio change | Positive ratio change | High-resolution mapping of cellular ABA dynamics |
| ABACUS2-400n [21] | ABA | 445 nM | +71% emission ratio change | Positive ratio change | In planta ABA studies with optimal sensitivity |
| ABACUS1-2µ [24] [25] | ABA | ~2 µM | +60% emission ratio change | Positive ratio change | ABA uptake and translocation studies |
| ABACUS1-80µ [24] [25] | ABA | ~80 µM | +160% emission ratio change | Positive ratio change | Monitoring high ABA concentrations |
| ABAleon2.1 [25] [26] | ABA | ~79 nM | -8.98% emission ratio change | Negative ratio change | ABA distribution and transport studies |
| ABAleon2.15 [25] [26] | ABA | ~600 nM | -10.09% emission ratio change | Negative ratio change | ABA distribution and transport studies |
| ATeam [27] [28] | ATP | ~3 mM (estimated) | ~1.9 dF/F (iATPSnFR1.1) | FRET change | Mitochondrial ATP monitoring in mammalian cells |
| iATPSnFR1.0 [28] | ATP | ~120 µM | ~2.4 dF/F | Intensity increase | Extracellular and cytosolic ATP imaging |
| iATPSnFR1.1 [28] | ATP | ~50 µM | ~1.9 dF/F | Intensity increase | Higher sensitivity ATP detection |
Engineering and Orthogonality Considerations
Recent advances in sensor engineering have focused on improving affinity, signal-to-noise ratio, and orthogonality (minimal interference with endogenous signaling) [21] [15]. The development of next-generation ABACUS2 sensors exemplifies this progress, where structure-guided mutagenesis of key residues in the ABA binding pocket (PYL1 E141D and R143S) combined with optimized linkers between sensory domains and fluorescent proteins yielded sensors with enhanced performance characteristics [21]. Similarly, engineering of the ATeam platform led to iATPSnFR variants, which employ circularly permuted superfolder GFP (cpSFGFP) inserted into the epsilon subunit of F₀F₁-ATPase to create single-wavelength sensors with improved trafficking and fluorescence properties [28].
A critical consideration in sensor selection is orthogonality – the degree to which the sensor interacts with endogenous signaling components. Early ABA sensors like ABAleons exhibited strong ABA hyposensitivity phenotypes, while ABACUS1 sensors showed minor ABA hypersensitivity [21]. Next-generation sensors address these limitations through strategic mutations that disrupt interaction with native signaling partners while maintaining sensor function [21].
Experimental Protocols for Sensor Validation and Application
Standard Characterization Workflow for FRET Sensors
The experimental validation of FRET sensors follows a systematic workflow to establish performance characteristics and functionality in biological systems. The diagram below outlines the key stages in this process:
Detailed Methodologies for Key Applications
ABACUS Sensor Protocol for Root ABA Dynamics:
- Plant Material: Generate Arabidopsis thaliana lines expressing ABACUS sensors (e.g., ABACUS1-2µ or ABACUS2-400n) under tissue-specific or ubiquitous promoters [21].
- Stress Treatments: Apply abiotic stress conditions including low humidity (30-40% RH), NaCl (100-150 mM), or osmotic stress (200-300 mM mannitol) to trigger ABA responses [21] [26].
- Imaging Setup: Use confocal or two-photon microscopy with appropriate filter sets for FRET imaging (excitation: 405-445 nm for Cerulean, emission: 475-525 nm for Cerulean and 525-575 nm for Citrine) [21] [24].
- Ratio Imaging: Acquire time-lapse sequences of donor and acceptor channels and compute emission ratio values (acceptor emission/donor emission) [21] [26].
- Calibration: Perform in situ calibration using known ABA concentrations where possible, or express sensor response as normalized ratio changes (ΔR/R₀) [24].
ATeam Protocol for Mitochondrial ATP Imaging:
- Sensor Targeting: Express ATeam sensors with mitochondrial targeting sequences (e.g., cytochrome c oxidase subunit VIII presequence) in mammalian cells [27].
- Metabolic Manipulation: Apply metabolic inhibitors (oligomycin for ATP depletion, FCCP for mitochondrial uncoupling) or substrates (glucose, pyruvate) to modulate ATP levels [27] [28].
- FRET Imaging: Use widefield or confocal microscopy with CFP excitation (430-450 nm) and simultaneous collection of CFP (470-500 nm) and YFP (525-550 nm) emissions [27].
- Data Analysis: Calculate FRET ratio (YFP/CFP emission) and normalize to baseline values. Compare with calibration curves generated using controlled ATP conditions [27] [28].
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key research reagents and their applications in FRET sensor studies
| Reagent/Category | Specific Examples | Function/Application | Experimental Considerations |
|---|---|---|---|
| Sensor Constructs | ABACUS2-100n, ABACUS2-400n, ABAleon2.1, ATeam, iATPSnFR | Core sensing element; determines affinity and specificity | Select based on affinity matching expected analyte concentrations |
| Expression Systems | Plant binary vectors, mammalian expression vectors, viral delivery systems | Sensor delivery to target cells and tissues | Consider cell-type specificity and expression level control |
| Reference Sensors | Non-responsive FP variants, substrate-binding mutants | Control for non-specific fluorescence changes | Essential for distinguishing specific responses from artifacts |
| Calibration Reagents | Pure hormone/analyte standards, metabolic inhibitors/inducers | Sensor calibration and quantitative interpretation | Permeable analogs needed for intracellular calibration |
| Microscopy Systems | Confocal, two-photon, FRET-optimized filter sets | Signal detection and quantification | Consider temporal resolution versus phototoxicity tradeoffs |
| Analysis Software | ImageJ/Fiji with FRET plugins, custom MATLAB/Python scripts | Data processing and ratio calculation | Automated analysis essential for high-throughput applications |
Applications and Biological Insights
Key Discoveries Enabled by FRET Sensors
The implementation of ABACUS and ATeam sensors has yielded fundamental insights into cellular signaling dynamics. ABACUS sensors have revealed that root cells accumulate ABA in the elongation zone in response to low foliar humidity, identifying this region as a site for phloem-transported ABA unloading [21]. This discovery established that ABA coordinates root responses to distant shoot stresses, enabling plants to maintain soil water foraging capabilities during aerial drought conditions [21]. Similarly, ABACUS sensors demonstrated that external ABA application triggers rapid induction of ABA degradation, modification, or compartmentation mechanisms, revealing previously uncharacterized homeostatic regulation [24].
In mammalian systems, ATeam sensors targeted to mitochondrial matrices have enabled the correlation of ATP dynamics with metabolic perturbations, providing unprecedented resolution of bioenergetic changes in living cells [27]. The subsequent development of iATPSnFR sensors extended these capabilities to monitoring ATP in various subcellular compartments and at the cell surface, revealing spatial heterogeneity in ATP concentrations during signaling events [28].
Advantages Over Conventional Detection Methods
FRET-based sensors provide distinct advantages compared to traditional analytical approaches. Unlike mass spectrometry methods that require tissue extraction and provide limited spatial resolution, FRET sensors enable non-invasive monitoring of analyte dynamics in specific cell types and subcellular compartments with second-to-minute temporal resolution [24] [25]. Similarly, while transcriptional reporters (e.g., DR5 for auxin, ABRE for ABA) provide valuable information about hormone response, they report indirectly on hormone presence through downstream signaling events and have slower response times due to the requirement for transcription and translation [15] [25].
The direct detection capability of FRET sensors was convincingly demonstrated in studies comparing ABACUS responses with ABA-induced gene expression, where rapid ABA accumulation preceded transcriptional activation by significant time margins [21] [26]. This temporal precision enables researchers to distinguish early signaling events from secondary responses, providing clearer insight into causal relationships in signaling networks.
The continuing evolution of genetically encoded FRET sensors represents a paradigm shift in our ability to visualize cellular signaling dynamics. The ABACUS and ATeam platforms exemplify how strategic protein engineering can yield tools with tailored affinities, optimized dynamic range, and minimal interference with endogenous processes. While each sensor platform has distinct strengths—with ABACUS variants providing exceptional resolution of plant hormone dynamics and ATeam/iATPSnFR sensors enabling comprehensive monitoring of cellular energy status—their shared foundation in FRET technology enables similar experimental approaches and data analysis strategies.
Future directions in the field include the development of multi-analyte detection systems using spectral variants of FRET pairs, enhanced sensors with reduced pH sensitivity, and platforms compatible with advanced imaging techniques such as super-resolution microscopy [28] [1]. Additionally, the integration of biosensor data with computational modeling promises to provide more comprehensive understanding of signaling networks. As these tools become increasingly sophisticated and accessible, they will undoubtedly continue to transform our understanding of cellular communication and metabolic regulation across diverse biological systems.
The arbuscular mycorrhizal (AM) symbiosis is a fundamental mutualistic relationship between soil fungi and the roots of most terrestrial plants, crucial for nutrient exchange, particularly phosphorus [8] [29]. In this symbiosis, AM fungi form intricate structures called arbuscules within root cortical cells, which serve as the primary sites for the exchange of plant-derived carbon for fungal-acquired phosphate (Pi) [30] [29]. Understanding the dynamics of phosphate flux at the cellular and subcellular levels is essential for unraveling the complex mechanisms underlying this symbiotic relationship.
Traditional biochemical methods for measuring phosphate, such as colorimetric assays, provide valuable data but are often insufficient for studying transient molecular events in living tissues, as they typically focus on endpoint measurements and lack spatial resolution [1] [8]. These limitations have driven the development of advanced sensing technologies that enable real-time, non-invasive monitoring of analyte dynamics within living systems.
Among these technologies, Fӧrster Resonance Energy Transfer (FRET)-based biosensors have emerged as powerful tools for probing biological processes in living organisms [1]. FRET is a distance-dependent energy transfer process between two fluorescent molecules – a donor and an acceptor – that occurs when they are in close proximity (typically 1-10 nm) [1] [2]. Genetically encoded FRET biosensors can be targeted to specific cellular compartments, allowing researchers to monitor analyte concentrations and molecular interactions with exceptional temporal and spatial resolution in real time [1] [8] [2]. This review focuses on the application of FRET-based biosensors, particularly for monitoring cellular phosphate levels during AM symbiosis, and provides a comparative analysis of their performance against alternative sensing methodologies.
FRET-Based Phosphate Biosensors: Principle and Design
Fundamental Principles of FRET Technology
FRET is a non-radiative energy transfer process that occurs when an excited donor fluorophore transfers energy to a nearby acceptor fluorophore through long-range dipole-dipole interactions [1] [2]. For FRET to occur efficiently, several conditions must be met: (1) the emission spectrum of the donor must overlap significantly with the absorption spectrum of the acceptor; (2) the donor and acceptor must be in close proximity (typically within 1-10 nm); and (3) the transition dipoles of the donor and acceptor must be favorably oriented [1]. The efficiency of FRET (E) is highly sensitive to the distance between the donor and acceptor, varying with the inverse sixth power of the distance, making it an exquisite "spectroscopic ruler" for measuring molecular-scale distances [1].
In biosensor design, FRET-based sensors typically consist of a ligand-binding domain flanked by two fluorescent proteins – most commonly cyan (CFP) and yellow (YFP) fluorescent protein variants [8] [29]. When the target analyte (in this case, phosphate) binds to the sensor, it induces a conformational change that alters the distance and/or orientation between the donor and acceptor fluorophores, thereby changing the FRET efficiency [8]. This change is quantified as a shift in the emission ratio of the two fluorescent proteins, providing a ratiometric measurement that is largely independent of sensor concentration and excitation intensity [8] [29].
The cpFLIPPi-5.3m Phosphate Sensor
For monitoring phosphate dynamics in AM symbiosis, the cpFLIPPi-5.3m biosensor has been specifically employed [30] [8] [29]. This sensor is a genetically encoded FRET-based biosensor that has been optimized for monitoring intracellular Pi concentrations in plants. The sensor incorporates a phosphate-binding protein domain between CFP (donor) and YFP (acceptor) variants. When phosphate binds, it triggers a conformational change that alters the FRET efficiency between the two fluorophores [8].
A critical control sensor, cpFLIPPi-Null, which contains a mutation that prevents Pi binding, is used alongside the active sensor to account for non-Pi-specific changes in the FRET signal, such as those caused by intracellular ionic shifts or changes in pH [8] [29]. This careful experimental design ensures that observed FRET ratio shifts genuinely reflect intracellular Pi fluctuations rather than artifactual changes in the cellular environment.
Table 1: Key Characteristics of the cpFLIPPi-5.3m FRET Biosensor
| Characteristic | Description |
|---|---|
| Sensor Type | Genetically encoded FRET-based biosensor |
| Donor Fluorophore | Cyan Fluorescent Protein (CFP) variant |
| Acceptor Fluorophore | Yellow Fluorescent Protein (YFP) variant |
| Analyte Specificity | Inorganic phosphate (Pi) |
| Key Control Sensor | cpFLIPPi-Null (Pi-binding deficient mutant) |
| Targeting Capability | Can be targeted to cytosol or specific organelles |
| Primary Readout | FRET ratio (Acceptor emission / Donor emission) |
Comparative Analysis of Phosphate Sensing Methodologies
FRET-Based Biosensors vs. Traditional Phosphate Assays
The development of FRET-based phosphate biosensors represents a significant advancement over traditional phosphate measurement techniques. While methods such as colorimetric assays (e.g., malachite green method) and chromatography provide valuable quantitative data, they typically require tissue destruction, preventing real-time monitoring in living systems [8] [31]. These conventional approaches also lack the spatial resolution to detect phosphate gradients at the cellular or subcellular level, which is particularly important in complex structures like arbusculated cells where phosphate flux varies significantly across different cellular compartments [8] [29].
FRET-based biosensors offer several distinct advantages for studying phosphate dynamics in AM symbiosis: (1) they enable non-invasive, real-time monitoring of phosphate levels in living tissues; (2) they provide exceptional spatial resolution at the cellular and subcellular levels; (3) they allow quantification of phosphate dynamics over time in response to physiological changes; and (4) they can be targeted to specific cellular compartments to investigate subcellular phosphate partitioning [30] [8].
Table 2: Comparison of Phosphate Detection Methodologies
| Methodology | Spatial Resolution | Temporal Resolution | In Vivo Application | Key Limitations |
|---|---|---|---|---|
| FRET Biosensors (e.g., cpFLIPPi-5.3m) | Cellular and subcellular | Real-time (seconds to minutes) | Yes | Requires genetic transformation; complex calibration |
| Colorimetric Assays | Bulk tissue analysis | Endpoint measurements | No | Destructive; no spatial or temporal dynamics |
| Chromatography | Bulk tissue analysis | Minutes to hours | No | Requires tissue extraction; complex sample preparation |
| Fluorescent Phosphors (e.g., Eu(cpboda)) | N/A | Real-time | No (in vitro) | Poor water solubility; photodegradation issues [31] |
Comparison with Alternative FRET Biosensors
The cpFLIPPi-5.3m sensor is part of a broader family of FRET-based biosensors developed for various analytes. For instance, the yAT1.03 sensor was engineered for ATP monitoring in yeast, with specific modifications to make it pH-insensitive – an important consideration given that pH fluctuations often coincide with metabolic changes [32]. Similarly, FRET-based biosensors have been developed for monitoring calcium ions, pH, hormones (e.g., ABA, auxin), and other signaling molecules in plants [8] [29].
What distinguishes the cpFLIPPi-5.3m sensor is its specific optimization for phosphate detection in plant systems, with demonstrated efficacy in monocot species like Brachypodium distachyon [30] [8]. The sensor's performance has been validated in both cytosolic and plastidic compartments, revealing distinct phosphate dynamics in these different subcellular locations during AM symbiosis [8].
Experimental Protocol for Monitoring Phosphate Flux in AM Symbiosis
Biological Materials and Growth Conditions
The protocol for monitoring phosphate flux in AM symbiosis utilizes Brachypodium distachyon (line Bd21-3) transgenic lines expressing the cpFLIPPi-5.3m sensor and its controls [8] [29]. These include:
- For cytosolic Pi quantification: Transgenic lines expressing the sensor and controls (cpFLIPPi-Null, eCFP, cpVenus) under either a mycorrhiza-inducible, cell-type-specific promoter (BdPT7) or a constitutive promoter (ZmUb1).
- For plastidic Pi quantification: Transgenic lines expressing plastid-targeted versions of the sensor and controls under the BdPT7 promoter.
- AM fungal inoculation: The protocol utilizes Diversispora epigaea or Rhizophagus irregularis spores for establishing symbiosis [8] [29].
Plants are grown in an optimized growth system that allows tracing of Pi transfer between AM fungi and host roots, typically employing a two-compartment system that separates root and fungal compartments [30] [8].
Image Acquisition and FRET Analysis
Image acquisition is performed using confocal microscopy with specific excitation and emission settings for CFP and YFP [8] [29]. The protocol employs a semi-automated ImageJ macro for sensitized FRET analysis, which involves several critical steps:
- Background subtraction: Using wild-type roots as negative controls to remove potential background fluorescence.
- Spectral unmixing: Accounting for donor spectral bleed-through and acceptor cross-excitation.
- FRET ratio calculation: Determining the ratio of acceptor emission to donor emission after appropriate corrections.
- Data normalization: Expressing FRET ratios relative to control conditions or baseline measurements.
The cpFLIPPi-Null control sensor is essential for distinguishing Pi-specific FRET changes from non-specific effects caused by factors such as ionic changes, pH fluctuations, or alterations in the cellular microenvironment [8] [29].
Key Research Reagent Solutions
Successful implementation of FRET-based phosphate monitoring in AM symbiosis requires specific biological materials and reagents. The following table outlines essential research solutions and their functions in the experimental protocol.
Table 3: Essential Research Reagents for FRET-Based Phosphate Monitoring
| Research Reagent | Function/Application | Specific Examples |
|---|---|---|
| Transgenic Plant Lines | Express FRET biosensors in specific cell types and compartments | Brachypodium distachyon lines with BdPT7 or ZmUb1 promoters driving cpFLIPPi-5.3m expression [8] |
| Control Sensors | Distinguish Pi-specific changes from non-specific effects | cpFLIPPi-Null (Pi-binding deficient), eCFP (donor only), cpVenus (acceptor only) [8] [29] |
| AM Fungal Inoculum | Establish symbiotic relationship for studying Pi transfer | Diversispora epigaea, Rhizophagus irregularis spores [8] [29] |
| Specialized Growth System | Enable tracing of Pi transfer between fungi and roots | Two-compartment system separating root and fungal compartments [30] |
| Image Analysis Software | Quantify FRET efficiency and calculate Pi concentrations | Semi-automated ImageJ macro for sensitized FRET analysis [8] [29] |
Advanced Technical Considerations
Sensor Targeting and Compartment-Specific Analysis
A significant advantage of genetically encoded FRET biosensors is the ability to target them to specific subcellular compartments, enabling researchers to investigate compartment-specific phosphate dynamics [8]. In the study of AM symbiosis, this capability has revealed distinct phosphate patterns in the cytosol versus plastids of arbusculated cells [8]. The plastid-targeted version of the cpFLIPPi-5.3m sensor has been particularly valuable in investigating the relationship between plastid morphology (stromulation) and phosphate status in colonized cells [8] [29].
Addressing Technical Challenges in FRET Imaging
Several technical challenges must be addressed when implementing FRET-based phosphate monitoring:
- Photostability: Extended imaging can lead to photobleaching, potentially affecting FRET ratios.
- Sensor expression levels: High expression may cause buffering of the target analyte or cellular toxicity.
- Calibration: Converting FRET ratios to absolute phosphate concentrations requires careful in vivo or in vitro calibration.
- Environmental controls: Maintaining stable temperature and imaging conditions is essential for reproducible results.
The use of control sensors (cpFLIPPi-Null, eCFP, and cpVenus) in parallel experiments helps address these challenges by accounting for non-specific changes in the FRET signal [8] [29].
Broader Context: FRET Biosensors in Plant Research
The application of FRET-based phosphate biosensors in AM symbiosis research represents just one example of how this technology is advancing plant science. Similar FRET-based approaches have been developed for monitoring other critical analytes, including:
- Calcium ions: Using sensors such as Cameleon for monitoring signaling events [8]
- pH: Using pH-sensitive FRET sensors to investigate cellular homeostasis [8]
- Hormones: Including ABA and auxin biosensors for phytohormone signaling studies [8]
- Nitrate: Using sensors like nrt1.1-based reporters for nutrient signaling [29]
These diverse applications highlight the versatility of FRET-based biosensing platforms and their growing importance in understanding plant physiology, signaling networks, and stress responses at unprecedented spatial and temporal resolution.
FRET-based biosensors represent a transformative technology for studying nutrient dynamics in plant-microbe interactions, with the cpFLIPPi-5.3m sensor providing unprecedented insights into phosphate flux during arbuscular mycorrhizal symbiosis. Compared to traditional phosphate detection methods, these biosensors offer unique capabilities for real-time, non-invasive monitoring of analyte dynamics at cellular and subcellular resolutions.
The application of this technology has revealed compartment-specific phosphate dynamics in arbusculated cells, enhanced our understanding of the distinct phosphate uptake pathways in mycorrhizal versus non-mycorrhizal roots, and provided new insights into the relationship between plastid morphology and phosphate status during symbiosis. As FRET biosensor technology continues to evolve, with improvements in fluorophore properties, targeting specificity, and data analysis methods, these tools will undoubtedly play an increasingly important role in advancing our understanding of plant nutrition, symbiotic relationships, and environmental adaptation.
The integration of FRET biosensing with other emerging technologies, such as automated imaging systems, advanced computational analysis, and multi-omics approaches, promises to further enhance our ability to decipher the complex molecular dialogues that underlie plant-fungal interactions and their implications for sustainable agriculture and ecosystem functioning.
Cellular energy homeostasis, particularly under hypoxic stress, is a cornerstone of physiological and pathophysiological research across diverse biological systems. The ability to monitor adenosine triphosphate (ATP) dynamics in living cells provides critical insights into metabolic adaptation, stress response pathways, and cellular viability. Förster Resonance Energy Transfer (FRET)-based genetically encoded biosensors have revolutionized this field by enabling real-time, non-invasive quantification of ATP levels with subcellular resolution in their native environments [1]. This guide objectively compares the performance of leading FRET-based ATP biosensors, detailing their operational mechanisms, experimental applications, and performance characteristics under controlled hypoxic conditions, with a specific focus on plant biology applications.
FRET Biosensor Technology: Design and Mechanism
Fundamental Principles of FRET
FRET is a distance-dependent physical process where energy is transferred non-radiatively from an excited donor fluorophore to an acceptor fluorophore through long-range dipole-dipole interactions [33] [1]. The efficiency of this energy transfer (E) is inversely proportional to the sixth power of the distance between the fluorophores (typically effective within 1-10 nm) and depends on several factors as defined by the Förster equation:
- Spectral Overlap: Significant overlap between the donor's emission spectrum and the acceptor's absorption spectrum [33]
- Orientation Factor (κ²): Proper relative orientation of the donor and acceptor transition dipoles [1]
- Förster Distance (R₀): The characteristic distance at which FRET efficiency is 50% [33]
Table 1: Common FRET Pairs Used in ATP Biosensors
| Donor | Acceptor | Förster Radius (R₀) in nm | Dynamic Range (nm) |
|---|---|---|---|
| mseCFP | cp173-mVenus | Not specified | Not specified |
| ECFP | EYFP | 4.9 | 2.5–7.3 |
| mCerulean | Venus | 5.4 | 2.7–8.1 |
| mTurquoise | mVenus | 5.7 | 2.9–8.6 |
Biosensor Engineering and ATP Recognition
Genetically encoded FRET biosensors for ATP typically employ a modular design where the ATP-binding protein domain is sandwiched between donor and acceptor fluorescent proteins (FPs) [33]. The ATeam sensor family uses the ε-subunit of the ATP synthase from Bacillus sp. PS3 as its recognition element [34] [35]. Upon ATP binding, this subunit undergoes a conformational change that alters the distance and/or orientation between the flanking FPs, thereby modifying FRET efficiency [35]. This molecular design translates biochemical binding events into measurable fluorescence signals, creating a quantitative relationship between ATP concentration and the emission ratio of the acceptor to donor channels.
Figure 1: Mechanism of FRET-based ATP Biosensors. ATP binding induces a conformational change in the sensor domain, altering the distance/orientation between donor and acceptor FPs and modulating FRET efficiency.
Comparative Performance Analysis of ATP Biosensors
The ATeam Sensor Family
The ATeam (ATP indicator based on ε-subunit for analytical measurements) sensors represent a well-characterized class of ratiometric ATP biosensors. The ATeam1.03-nD/nA variant has been successfully deployed in plant systems, particularly Arabidopsis thaliana, for monitoring cytosolic, plastid, and mitochondrial ATP dynamics [9] [35].
Performance Characteristics:
- Affinity Range: Optimized for the high micromolar to low millimolar range appropriate for physiological ATP concentrations in plant cells [35]
- Specificity: Highly specific for the biologically active MgATP²⁻ complex over other nucleotide forms [35]
- pH Stability: Demonstrates minimal pH sensitivity within physiological ranges, unlike some alternative sensors like Perceval [35]
- Dynamic Range: Exhibits a well-defined ratiometric shift with an isosbestic point at 512 nm, allowing for robust quantification [35]
The ECATS Series for Extracellular ATP
The ECATS (Extracellular ATP Sensor) series represents specialized biosensors engineered for detecting ATP released into the extracellular space, which functions as an important purinergic signaling molecule [34]. The recently developed ECATS2 variant incorporates R103A/R115A double mutations in the ATP binding site, resulting in a greater than three-fold higher affinity compared to the original ecATeam3.10 sensor [34].
Key Applications:
- Detection of extracellular ATP release during hypoosmotic stress in cultured astrocytes [34]
- Monitoring purinergic signaling dynamics in real-time [34]
- Studies of ATP release during injury and immune responses [34]
Alternative Sensor Architectures
While ATeam and ECATS utilize the ε-subunit of ATP synthase, other sensor designs employ different recognition strategies:
- Perceval/PercevalHR: Utilizes the bacterial regulatory protein GlnK1 and provides a ratiometric readout of the ATP:ADP ratio rather than absolute ATP concentration [35]. However, it exhibits significant pH sensitivity, requiring careful pH monitoring and correction for accurate interpretation [35].
Table 2: Performance Comparison of ATP Biosensors Under Hypoxic Conditions
| Sensor Name | Recognition Element | Target Analyte | Affinity/Dynamic Range | Key Advantages | Limitations |
|---|---|---|---|---|---|
| ATeam1.03-nD/nA | ε-subunit (Bacillus sp. PS3) | MgATP²⁻ | High μM to low mM [35] | Low pH sensitivity; successfully deployed in plants [35] | Requires dual-channel imaging |
| ECATS2 | Mutated ε-subunit | Extracellular ATP | >3-fold higher affinity than ecATeam3.10 [34] | Optimized for extracellular ATP signaling; high affinity [34] | Specialized for extracellular applications |
| PercevalHR | GlnK1 (M. jannaschii) | ATP:ADP ratio | Dynamic range ~4 [35] | Reports on energy charge (ATP:ADP ratio) [35] | Strong pH sensitivity requires correction [35] |
Experimental Protocols for ATP Assessment Under Hypoxia
Establishing Hypoxic Conditions in Plant Systems
For studying ATP dynamics in plants under low oxygen stress, researchers have developed specialized protocols using Arabidopsis thaliana seedlings expressing ATeam1.03-nD/nA in the cytosol [9]. The experimental workflow involves:
Plant Material Preparation:
Oxygen Control:
Data Acquisition:
Figure 2: Experimental Workflow for Monitoring ATP Dynamics in Plants Under Hypoxic Stress
Data Interpretation and Normalization
The ratiometric nature of FRET-based ATP biosensors provides intrinsic normalization for variations in sensor expression level and sample thickness [34] [35]. Data analysis involves:
- Ratio Calculation: Compute Venus/CFP emission ratios for each time point [9] [35]
- Antiparallel Signal Validation: Confirm bona fide FRET changes by verifying antiparallel behavior of donor and acceptor emission intensities [9]
- Temporal Dynamics: Monitor ratio changes over time, where decreasing Venus/CFP ratios indicate declining ATP levels [9]
In hypoxia experiments using Arabidopsis seedlings, researchers observed gradually decreasing Venus/CFP ratios in the cytosol under oxygen restriction, while ratios remained stable in normoxic controls [9]. This response was reversible upon reoxygenation, demonstrating the dynamic monitoring capability of these sensors [9].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for FRET-based ATP Imaging
| Reagent/Tool | Function/Application | Example Use Case |
|---|---|---|
| ATeam1.03-nD/nA | Genetically encoded ratiometric ATP biosensor | Monitoring cytosolic ATP dynamics in Arabidopsis [35] |
| ECATS2 | High-affinity extracellular ATP biosensor | Detecting ATP release during hypoosmotic stress [34] |
| Paper-based Cell Culture Scaffolds | 3D culture platform with controlled oxygen microenvironments | Maintaining physiologically relevant oxygen tensions in hepatocyte studies [36] |
| Hypoxia Chambers/Workstations | Controlled atmosphere for hypoxic experimentation | Maintaining precise oxygen tensions (e.g., 2% O₂) during live-cell imaging [37] |
| CLARIOstar Microplate Reader | Fluorescence detection with atmospheric control | Kinetic monitoring of FRET ratios in multi-well formats [9] |
The objective comparison of FRET-based ATP biosensors reveals a sophisticated toolbox for investigating cellular energetics under hypoxic stress. The ATeam sensor family, particularly ATeam1.03-nD/nA, offers robust performance for intracellular ATP monitoring in plant systems with minimal pH sensitivity and reliable ratiometric readouts. For specialized applications focusing on purinergic signaling, the ECATS series provides optimized extracellular ATP detection with enhanced affinity. The experimental protocols established for plant hypoxia research demonstrate the practical application of these sensors, enabling researchers to quantify spatial and temporal ATP dynamics with high resolution. As the field advances, the integration of these molecular tools with more physiologically relevant culture systems and controlled microenvironments will further enhance our understanding of cellular energy management under metabolic challenge.
Förster Resonance Energy Transfer (FRET) is a powerful spectroscopic technique used to study molecular interactions and conformational changes at the nanoscale level [2]. When applied through advanced microscopy platforms, it enables researchers to investigate protein-protein interactions, monitor cellular signaling events, and quantify analyte concentrations in living cells with high spatiotemporal resolution [38] [39]. The efficiency of FRET is highly dependent on the distance between donor and acceptor fluorophores, typically occurring within 1-10 nanometers, making it an exceptionally sensitive "molecular ruler" for biological research [2] [40]. This comparison guide examines three principal imaging platforms—laser scanning confocal microscopy, Fluorescence Lifetime Imaging Microscopy FRET (FLIM-FRET), and single-molecule FRET (smFRET)—for their performance characteristics, applications, and suitability for plant analyte research.
Technology Comparison and Performance Metrics
The selection of an appropriate FRET imaging platform involves careful consideration of performance specifications relative to research goals. Each technology offers distinct advantages and limitations in resolution, throughput, and implementation complexity.
Table 1: Performance Comparison of Advanced FRET Imaging Platforms
| Technology | Spatial Resolution | Temporal Resolution | Key Strengths | Primary Limitations | Optimal Use Cases |
|---|---|---|---|---|---|
| Confocal FRET | ~250 nm lateral~500-800 nm axial | Seconds to minutes | Accessibility, ease of use, live-cell compatibility | Concentration-dependent signals, photobleaching issues, spectral bleed-through | High-throughput screening, live-cell dynamic studies with high expression levels |
| FLIM-FRET | ~250 nm lateral~500-800 nm axial | Seconds to minutes (wide-field)Minutes (scanning) | Concentration independence, quantitative accuracy, reduced artifacts | Lower acquisition speed, complex data analysis, higher instrumentation costs | Quantitative interaction studies, environments with variable expression levels, autofluorescence-rich samples |
| smFRET | ~1-10 nm molecular scale~250 nm optical | Millisecond to second | Molecular heterogeneity detection, absolute distance measurements, transient state observation | Very low throughput, specialized sample preparation, extreme sensitivity to noise | Molecular mechanics, conformational dynamics, protein folding, nucleic acid studies |
Table 2: Quantitative Performance Specifications for FRET Platforms
| Parameter | Confocal FRET | FLIM-FRET | smFRET |
|---|---|---|---|
| Distance Range | 1-10 nm | 1-10 nm | 1-10 nm |
| FRET Efficiency Range | 5-95% | 1-99% | 5-95% |
| Throughput | High (ensemble) | Medium (ensemble) | Low (single molecules) |
| Photobleaching Resistance | Low | Medium | Low |
| Live-Cell Compatibility | Excellent | Good | Limited |
| Quantitative Accuracy | Moderate | High | Very High |
| Implementation Complexity | Low | High | Very High |
Technology-Specific Methodologies and Protocols
Confocal FRET Imaging
Confocal microscopy represents the most accessible platform for FRET measurements, typically employing intensity-based detection methods. The core principle involves exciting the donor fluorophore and detecting emission from both donor and acceptor channels, followed by computational correction for spectral bleed-through [2] [40].
Experimental Protocol: Sensitized Emission FRET
- Sample Preparation: Express FRET biosensors (e.g., ABACUS for ABA quantification) in plant cells via transfection [21]. Ensure optimal expression levels to avoid artifacts from overexpression.
- Microscope Setup: Configure laser lines for donor excitation (e.g., 458 nm for CFP, 515 nm for YFP). Establish detection channels with appropriate bandpass filters: donor emission (470-500 nm for CFP) and acceptor emission (530-570 nm for YFP) [41].
- Acquisition Parameters: Set pinhole to 1 Airy unit for optimal sectioning. Use low laser power (1-5 μW) to minimize photobleaching. Acquire sequential images: donor channel with donor excitation, acceptor channel with donor excitation, and acceptor channel with acceptor excitation [41].
- FRET Calculation: Apply correction algorithms for spectral bleed-through using reference samples expressing donor-only and acceptor-only constructs. Calculate corrected FRET efficiency using established algorithms [40].
Figure 1: Confocal FRET Workflow
FLIM-FRET Methodology
FLIM-FRET measures the reduction in donor fluorescence lifetime resulting from energy transfer to an acceptor, providing concentration-independent quantification of FRET efficiency [40] [41]. This method is particularly valuable for plant research where expression levels may vary significantly between cells and tissues.
Experimental Protocol: Time-Domain FLIM-FRET
- System Configuration: Employ time-correlated single photon counting (TCSPC) with pulsed laser sources (e.g., 485 nm, 531 nm pulsed diodes at 20-80 MHz) [42] [41]. Use high-sensitivity detectors (SPADs, PMTs) with picosecond timing resolution.
- Lifetime Calibration: Acquire donor-only reference sample to determine unquenched donor lifetime (τD). For mTurquoise2, expect ~3.9 ns [41].
- Sample Imaging: Collect photon arrival times relative to excitation pulses. Build fluorescence decay histograms for each image pixel. Typical acquisition times range from 30-300 seconds depending on signal brightness [41] [43].
- Lifetime Analysis: Fit decay curves to single or multi-exponential models using maximum likelihood estimation. Calculate FRET efficiency using: E = 1 - (τDA/τD), where τDA is donor lifetime in the presence of acceptor [40] [41].
- Data Validation: Implement phasor analysis for model-free lifetime representation [41]. For complex decays, use global fitting approaches with shared lifetime components across pixels.
Advanced Implementation: Multi-Beam FLIM For enhanced temporal resolution, parallelized excitation with multiple beamlets (e.g., 64-beam system) significantly improves acquisition speed. This approach enables time-lapse FLIM at up to 0.5 frames per second, capturing rapid cellular dynamics while maintaining low peak excitation powers (~1-2 μW per beamlet) to minimize photodamage [41].
Figure 2: FLIM-FRET Methodology
Single-Molecule FRET (smFRET) Protocols
smFRET enables the observation of individual biomolecules, revealing heterogeneity and transient states that are obscured in ensemble measurements [39] [42]. This technique is particularly valuable for studying plant receptor conformational changes and enzyme dynamics at unprecedented resolution.
Experimental Protocol: smFRET with PIE-FRET
- Sample Preparation: Label biomolecules with donor-acceptor pairs (e.g., Cy3-Cy5, ATTO550-ATTO647N) via cysteine mutations or hybridization. For plant applications, purify receptors or enzymes and immobilize on functionalized surfaces at low density (~100 molecules/field of view) [42].
- Microscope Configuration: Implement Pulsed Interleaved Excitation (PIE) using alternating 531 nm and 636 nm laser pulses on nanosecond timescales [42]. Use objective-type total internal reflection (TIRF) illumination to reduce background.
- Data Acquisition: Collect donor and acceptor emissions simultaneously using two detectors with precise time-stamping (TCSPC). Acquire data until single molecules photobleach (typically 1-10 seconds per molecule) [42] [44].
- FRET Analysis: Identify single-molecule bursts and calculate apparent FRET efficiency using: E = IA/(ID + IA), where ID and IA are donor and acceptor intensities after appropriate correction [42]. Apply correction factors for direct acceptor excitation, donor leakage into acceptor channel, and differential quantum yields.
- Advanced Implementation: smFLIM-FRET: Combine smFRET with FLIM for robust lifetime-based FRET efficiency measurements independent of intensity variations. This approach is particularly valuable for molecules with dynamic heterogeneity [42] [44].
Wide-Field smFLIM Implementation Recent advances in gated SPAD cameras enable wide-field single-molecule FLIM, allowing parallelized lifetime measurements of thousands of individual molecules simultaneously. This approach uses a rapid lifetime determination scheme with two gate positions, achieving ~300x throughput improvement compared to sequential point scanning [44].
Research Reagent Solutions
Successful implementation of FRET imaging requires careful selection of biosensors, fluorophores, and experimental reagents optimized for each platform and application.
Table 3: Essential Research Reagents for FRET Imaging
| Reagent Category | Specific Examples | Key Features | Optimal Application |
|---|---|---|---|
| Genetically Encoded Biosensors | ABACUS2-100n, ABACUS2-400n [21] | High affinity (KD: 98-445 nM), large emission ratio change (+67-71%) | Plant hormone (ABA) quantification in live cells |
| FRET Biosensors | Epac-based cAMP sensors [41] | mTurquoise2 donor, tddVenus acceptor, optimized for FLIM-FRET | Second messenger dynamics in signaling studies |
| Fluorescent Proteins | mTurquoise2 [41], Turquoise2 [43], Venus [43] | High quantum yield, appropriate maturation, monoexponential decay | Live-cell FRET and FLIM-FRET applications |
| Synthetic Dyes | Cy3B, ATTO550, ATTO647N [42] [44] | High photostability, brightness, well-characterized lifetimes | smFRET and single-molecule studies |
| Immobilization Systems | Biotin-streptavidin, PEG-passivated surfaces [42] | Low nonspecific binding, maintained protein functionality | smFRET and single-molecule imaging |
Applications in Plant Analyte Research
Advanced FRET imaging platforms have enabled groundbreaking research in plant biology, particularly in quantifying hormone dynamics and signaling events at cellular resolution.
The ABACUS biosensors exemplify the power of FRET technology for plant research. Next-generation ABACUS2-100n and ABACUS2-400n variants exhibit significantly improved affinity (KD = 98 nM and 445 nM, respectively) and emission ratio changes (+67% and +71%) compared to earlier versions [21]. These biosensors have revealed cellular ABA accumulation patterns in Arabidopsis root elongation zones in response to low aerial humidity, demonstrating how plants systemically coordinate root growth responses to foliar stress [21].
FLIM-FRET applications in plant research benefit from the technology's concentration independence, which is particularly valuable when studying tissues with variable biosensor expression levels. The quantitative nature of FLIM-FRET enables precise measurement of interaction stoichiometries and fraction of interacting molecules in complex cellular environments [41] [43].
For fundamental studies of plant protein mechanics and conformational changes, smFRET provides unparalleled resolution. This platform can reveal the dynamics of individual receptor proteins, enzyme conformational states, and nucleic acid regulatory elements that would be averaged out in ensemble measurements [39] [42].
Confocal FRET, FLIM-FRET, and smFRET represent complementary technologies in the plant researcher's toolkit, each with distinct advantages for specific experimental questions. Confocal FRET offers accessibility and high throughput for dynamic live-cell imaging, while FLIM-FRET provides quantitative accuracy in complex cellular environments. smFRET delivers the ultimate resolution for molecular-scale mechanistic studies. The continuing development of optimized biosensors like the ABACUS series, coupled with advancements in detector technology and analysis algorithms, promises to further expand the capabilities of these platforms for plant analyte research. Selection among these technologies should be guided by specific research goals, considering the trade-offs between resolution, throughput, and implementation complexity.
Overcoming Technical Hurdles: Optimization and Troubleshooting of FRET Sensors
Förster Resonance Energy Transfer (FRET) biosensors have revolutionized the study of cellular processes by enabling real-time visualization of molecular interactions with high spatiotemporal resolution. These genetically encoded tools function as molecular spies, converting conformational changes into measurable fluorescence signals that report on dynamic cellular events. The performance of these sophisticated tools hinges on two critical engineering elements: the sensory domain responsible for analyte recognition and the linker regions that control spatial orientation and flexibility. Strategic enhancement of these components directly addresses the universal biosensor challenges of achieving sufficient affinity for physiological analyte concentrations while maintaining strict specificity against interfering compounds.
The fundamental working principle of FRET biosensors relies on non-radiative energy transfer between two fluorophores—a donor and an acceptor—when positioned within 1-10 nanometers of each other. Energy transfer efficiency depends critically on the distance and relative orientation between these fluorophores, quantified by the Förster radius (R0), where efficiency drops significantly as separation increases beyond this critical distance [39]. This physical principle makes FRET biosensors exquisitely sensitive to molecular-scale movements, but also imposes stringent design requirements for optimal performance. As this comparison guide will demonstrate through experimental data from recent studies, rational engineering of both sensory domains and linkers provides a powerful approach to tailoring biosensor characteristics for specific research applications across plant biology and drug development.
Sensory Domain Engineering: Strategic Enhancements for Improved Performance
The sensory domain serves as the molecular recognition element of FRET biosensors, determining both affinity and specificity for target analytes. Engineering efforts in this component have focused on structure-guided mutagenesis and utilizing diverse natural protein scaffolds to optimize binding characteristics.
Affinity Enhancement Through Binding Site Mutagenesis
Strategic mutagenesis of key residues in the analyte binding pocket has proven highly effective for enhancing biosensor affinity. A notable example comes from the development of ECATS2, an improved extracellular ATP biosensor, where researchers introduced R103A and R115A double mutations in the ATP synthase ε subunit from Bacillus sp. PS3. This rational design, informed by previous reports, resulted in a greater than three-fold increase in ATP affinity compared to the original ecATeam3.10 biosensor [12]. The enhanced affinity enables detection of lower ATP concentrations, making the biosensor more suitable for monitoring physiologically relevant extracellular ATP fluctuations that typically occur at nanomolar to low micromolar levels [12].
Similar affinity engineering approaches have been successfully applied to biosensors for other analytes. The FRET JH Indicator Agent (FREJIA), developed for monitoring juvenile hormone in insects, was optimized through iterative mutagenesis to achieve nanomolar sensitivity for JH I, II, and III isoforms as well as the JH analog methoprene [45]. Structural insights from both apo and JH-bound states of the Bombyx mori juvenile hormone-binding protein informed the engineering process, highlighting the value of structural data in guiding mutagenesis strategies [45].
Natural Protein Scaffolds as Sensory Domain Foundations
Diverse natural protein scaffolds provide excellent starting points for sensory domain engineering, with different protein classes offering distinct advantages for various applications:
- Periplasmic binding proteins (PBPs) and solute binding proteins (SBPs) undergo large conformational changes upon analyte binding and yield soluble biosensors that can be targeted to various subcellular compartments [19].
- G-protein-coupled receptors (GPCRs) offer membrane-integral sensing capabilities with natural affinity for neurotransmitters and neuromodulators [19].
- Voltage sensing domains (VSDs) enable monitoring of membrane potential changes [19].
- Cyclic nucleotide binding domains (CNBDs) provide specificity for cAMP or cGMP monitoring, with recent iterations like cAMPFIRE offering improved performance characteristics [19].
The selection of an appropriate natural scaffold depends on the target analyte and desired biosensor localization, with previous structural and functional data on conformational changes greatly facilitating the engineering process [19].
Table 1: Comparison of Sensory Domain Engineering Strategies
| Engineering Approach | Target Analyte | Key Mutations/Modifications | Affinity Improvement | Specificity Profile |
|---|---|---|---|---|
| Binding site mutagenesis | ATP | R103A/R115A in ε subunit | >3-fold increase | Maintained specificity for ATP over other nucleotides |
| Binding site mutagenesis | Juvenile Hormone | Optimized insertion of mTFP1 into JHBP | Nanomolar EC50 for JH I, II, III | Cross-reactivity with methoprene but not pyriproxyfen |
| Natural scaffold utilization | Abscisic Acid | PYR/PYL/RCAR receptors with PP2C phosphatases | 100-600 nM range | High specificity for bioactive ABA isomers |
| Natural scaffold utilization | Calcium | Calmodulin-M13 affinity clamp | Various affinities achieved | Highly specific for Ca²⁺ over other cations |
Linker Engineering: Optimizing Structural Transduction
Linker regions serve as critical structural components that transduce binding-induced conformational changes into altered FRET efficiency. These seemingly simple connection segments significantly impact biosensor performance by controlling flexibility, distance, and orientation between domains.
Length Optimization for Cell-Surface Display
The tether length between FRET biosensors and their membrane anchors has emerged as a surprisingly critical factor in biosensor performance. In developing the ECATS2 ATP biosensor, researchers discovered that tether length between the biosensor and cell surface anchor significantly impacted optimization [12]. While the exact optimal length varies between biosensor architectures, systematic testing of different linker lengths has become an essential step in biosensor development, particularly for surface-displayed applications where steric hindrance from the membrane can limit accessibility or conformational freedom.
Advanced Linker Designs for Enhanced Dynamic Range
Beyond simple length optimization, sophisticated linker engineering strategies have been developed to improve biosensor dynamic range. Introduction of ER/K linkers (repeating sequences of glutamic acid and arginine or lysine residues) has successfully addressed the limited dynamic range often observed in fluorescent protein-based FRET biosensors caused by restricted conformational changes [39]. These alpha-helical linkers with complementary charges promote specific coiled-coil interactions that maintain optimal spacing and orientation between sensory and fluorescent domains, resulting in more robust FRET changes upon analyte binding [39].
Similar optimization approaches have proven valuable in plant research applications. When working with Arabidopsis protoplasts, systematic optimization of linker regions in the D3cpv calcium biosensor significantly enhanced FRET imaging precision by reducing background interference and improving signal-to-noise ratios [46]. These modifications help mitigate challenges posed by plant tissue autofluorescence, particularly from chlorophyll which emits strongly at 670nm and overlaps with the spectra of many fluorescent proteins [46].
Table 2: Linker Engineering Strategies and Their Impacts on Biosensor Performance
| Linker Strategy | Biosensor Platform | Key Findings | Performance Impact |
|---|---|---|---|
| Tether length optimization | ECATS2 ATP biosensor | Critical for cell-surface display optimization | Improved accessibility to extracellular analytes |
| ER/K linkers | General FP-based FRET biosensors | Promote specific coiled-coil interactions | Enhanced dynamic range through controlled flexibility |
| Protease-resistant linkers | Biosensors for harsh environments | Increased stability in complex media | Extended functional lifetime for pathogen detection |
| Modular cloning systems | Various FRET biosensors | Enable rapid testing of multiple linker variants | Accelerated optimization workflow |
Experimental Protocols: Methodologies for Engineering and Validation
Robust experimental protocols are essential for reliably engineering and validating enhanced FRET biosensors. The following section details key methodologies cited in recent literature.
Site-Directed Mutagenesis and Molecular Cloning
Molecular biology techniques form the foundation of biosensor engineering. For the ECATS2 development, researchers introduced mutations using the NEB Q5 Site-Directed Mutagenesis kit, with additional cloning performed via NEB Gibson/HiFi Assembly or restriction enzyme cloning [12]. Similar approaches were employed for FREJIA development, where site-directed mutagenesis utilized DpnI digestion to eliminate methylated template DNA followed by self-ligation with the GeneArt Seamless Cloning and Assembly Enzyme Mix [45]. These methods enable precise manipulation of both sensory domains and linker regions.
Protein Purification and In Vitro Characterization
For biosensors like FREJIA, recombinant proteins were expressed in E. coli BL21(DE3) cells through induction with 1 mM IPTG at 16°C for 16 hours in the dark to minimize photobleaching [45]. Purification employed Ni-NTA affinity chromatography (HisTrap HP) followed by additional purification using HiLoad 26/60 Superdex 200 prep-grade size exclusion chromatography [45]. Protein purity was assessed by SDS-PAGE with Coomassie Brilliant Blue staining, and concentrations determined via UV-visible spectrophotometry [45].
FRET Efficiency Measurements and Dose-Response Analysis
Quantitative FRET measurements typically utilize purified sensors at concentrations of 2-5 μM in appropriate buffer systems [45]. For FREJIA characterization, fluorescence spectra were acquired at 25°C using a fluorescence spectrophotometer, with FRET efficiency determined ratiometrically as the emission intensity ratio of mVenus to mTFP1 [45]. Excitation/emission parameters included: donor fluorophore (mTFP1: excitation 450 nm, emission 480 nm), acceptor fluorophore (mVenus: excitation 500 nm, emission 530 nm), and FRET channel (mTFP1 excitation at 450 nm, mVenus emission at 530 nm) [45]. Dose-response curves generated from these measurements enable calculation of apparent dissociation constants and dynamic range.
Live-Cell Imaging and Validation
Functional validation in live cells typically involves transfection (using Effectene, calcium phosphate, or PEI Max methods) or viral transduction followed by fluorescence microscopy [12] [45]. For the ECATS2 biosensor, live-cell microscopy employed an Olympus IX83 inverted fluorescence microscope with Andor iQ3 software, 20X/0.75 NA objective, Lumencor SpectraX light engine, and Andor Zyla 4.2 sCMOS camera [12]. Specific filter sets included: cyan (438/29 nm excitation, 470/24 nm emission), yellow (510/10 nm excitation, 540/30 nm emission), sensitized FRET emission (438/29 nm excitation, 540/30 nm emission) [12]. Image analysis utilized ImageJ/FIJI with background subtraction and careful region of interest selection around cell membranes to exclude intracellular background [12].
Diagram 1: Engineering Strategies for Enhanced FRET Biosensors. This workflow illustrates the relationship between sensory domain engineering (yellow), linker optimization (green), performance metrics (red), and application areas (blue).
Comparative Performance Analysis: Engineered Biosensors Versus Alternatives
Systematic comparison of engineered FRET biosensors reveals how specific modifications translate to improved performance characteristics across different application scenarios.
Affinity-Optimized vs. First-Generation Biosensors
The development of ECATS2 exemplifies the substantial improvements achievable through rational design. Compared to the original ecATeam3.10, the engineered ECATS2 exhibits over three-fold higher affinity for extracellular ATP, enabling detection of lower concentration ranges more relevant to physiological purinergic signaling [12]. This enhanced performance directly results from the combined R103A/R115A mutations in the sensory domain coupled with linker optimization for cell-surface display [12]. Similarly, ABAleons—FRET-based abscisic acid reporters—were engineered with affinities ranging from 100-600 nM to match physiological hormone concentration ranges in plants, allowing direct visualization of ABA distribution and dynamics in response to environmental stresses like low humidity and NaCl exposure [26].
Specificity Profiles Across Sensor Classes
Different sensory domain strategies yield distinct specificity profiles critical for various applications. FREJIA demonstrates broad specificity across JH I, II, and III isoforms plus methoprene, but not pyriproxyfen, reflecting the binding characteristics of its Bombyx mori JHBP foundation [45]. In contrast, ABAleons exhibit high specificity for bioactive ABA isomers, enabled by the PYR/PYL/RCAR receptor domains that naturally confer hormone specificity in plant systems [26]. Calcium biosensors based on calmodulin-M13 affinity clamps maintain exceptional specificity for Ca²⁺ over other cations, a characteristic preserved through numerous generations of optimization [19].
Table 3: Performance Comparison of Engineered FRET Biosensors
| Biosensor | Target Analyte | Engineering Strategy | Affinity (Kd or EC50) | Dynamic Range (ΔR/R%) | Key Applications |
|---|---|---|---|---|---|
| ECATS2 | ATP | Binding site mutagenesis + linker optimization | >3x improvement over wildtype | Not specified | Detection of hypoosmotic stress-induced ATP release in astrocytes |
| FREJIA | Juvenile Hormone | Insertional mutagenesis in JHBP | Nanomolar range | Significant ratio change | Real-time JH monitoring in insect systems |
| ABAleon | Abscisic Acid | PYR/PYL/RCAR-PP2C interaction | 100-600 nM | >1.5-fold FRET ratio change | ABA distribution mapping in response to stress |
| YC3.60 | Calcium | cpVenus implementation | Tunable affinities | High dynamic range | [Ca²⁺]cyt dynamics in Arabidopsis roots |
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of FRET biosensor engineering requires specific reagents and materials optimized for various aspects of development and application.
Table 4: Essential Research Reagents for FRET Biosensor Engineering
| Reagent/Material | Specific Examples | Function in Biosensor Engineering |
|---|---|---|
| Mutagenesis Kits | NEB Q5 Site-Directed Mutagenesis Kit | Introduction of specific mutations in sensory domains |
| Cloning Systems | NEB Gibson/HiFi Assembly, Gateway cloning | Modular construction of biosensor variants |
| Expression Vectors | pRSET-A, pAd/CMV/V5-DEST, pcDNA3.1 | Recombinant protein expression in bacterial and mammalian systems |
| Fluorescent Proteins | mseCFP, mVenus, mTFP1, cpVenus | FRET donor-acceptor pairs with optimized spectral properties |
| Cell Culture Reagents | Effectene, calcium phosphate, PEI Max | Transfection for biosensor expression in mammalian cells |
| Chromatography Media | Ni-NTA affinity columns (HisTrap HP) | Purification of recombinant biosensor proteins |
| Microscopy Systems | Olympus IX83 with Andor iQ3 software | Live-cell FRET imaging with appropriate filter sets |
| Image Analysis Software | ImageJ/FIJI with custom plugins | Quantitative ratio imaging and data processing |
Engineering of sensory domains and linkers represents a powerful approach for enhancing FRET biosensor performance, with different strategies offering distinct advantages for specific research scenarios. Affinity optimization through binding site mutagenesis proves most valuable for detecting low-abundance analytes or rapid transient signals, while natural scaffold utilization provides robust starting points for new biosensor development. Linker engineering, particularly through length optimization and advanced designs like ER/K linkers, significantly impacts dynamic range and cellular targeting.
For researchers selecting FRET biosensors, key considerations should include matching biosensor affinity to expected analyte concentrations, verifying specificity against potential interferents, and ensuring appropriate subcellular targeting through strategic linker design. The continued advancement of engineering strategies promises even more sensitive and specific tools for probing cellular signaling and metabolism across diverse biological systems, from plant hormone research to drug discovery applications. As these technologies evolve, they will undoubtedly provide unprecedented insights into the dynamic molecular events underlying biological function and dysfunction.
Förster Resonance Energy Transfer (FRET)-based biosensors are powerful tools for monitoring biochemical activities in live cells and organisms, enabling real-time observation of plant analytes, hormones, and signaling events with high spatial and temporal resolution. A biosensor's effectiveness hinges on its dynamic range—the magnitude of signal change between unbound and bound states—and its signal-to-noise ratio (SNR), which determines measurement precision. For plant researchers, optimizing these parameters is crucial for detecting subtle changes in analytes like abscisic acid (ABA) or phosphate within complex tissue environments. This guide compares FRET sensor engineering strategies and performance data, providing a framework for selecting and optimizing sensors for plant analyte research.
Sensor Engineering Strategies for Enhanced Performance
The dynamic range and SNR of FRET biosensors can be improved through strategic engineering of their molecular components. The table below compares three key engineering approaches, with examples from recent plant science research.
Table 1: Performance Comparison of FRET Biosensor Engineering Strategies
| Engineering Strategy | Sensor Example & Target | Key Modification | Reported Dynamic Range (ΔR/R₀ %) | Affinity (K_D) | Impact on SNR |
|---|---|---|---|---|---|
| Fluorophore Pair Optimization | REKAR67/76 (ERK activity) [47] | Red-shifted FPs (miRFP670nano3/miRFP720) for multiplexing | REKAR67: HigherREKAR76: Lower Variance [47] | Consistent with CFP/YFP sensors [47] | Enables multiplexing with CFP/YFP sensors, reducing crosstalk noise [47] |
| Sensory Domain & Linker Engineering | ABACUS2 (Abscisic Acid) [21] | "Latch" mutations (PYL1 R143S) and rigid linkers | ABACUS2-400n: +71% [21] | ABACUS2-400n: 445 nM [21] | High signal change improves SNR; orthogonality minimizes host interference [21] |
| Conformational Design | cpFLIPPi-5.3m (Phosphate) [8] | Circularly permuted FPs with a phosphate-binding protein | Quantified via sensitized FRET ratio [8] | Defined by binding affinity of the protein [8] | High specificity reduces off-target noise; controlled by a non-responsive Null sensor [8] |
The relationships between these design strategies and their resulting sensor properties can be visualized as an optimization workflow.
Quantitative Comparison of FRET Biosensor Performance
Direct comparison of biosensors for different analytes reveals how design choices translate to measurable performance. The following table summarizes quantitative data from featured sensors, providing a reference for expected performance metrics.
Table 2: Quantitative Performance Metrics of Featured FRET Biosensors
| Biosensor Name | Target Analyte | FRET Pair | Dynamic Range (ΔR/R₀ %) | Affinity (K_D) | Optimal Use Case |
|---|---|---|---|---|---|
| ABACUS2-400n [21] | Abscisic Acid (ABA) | edCitrine / edCerulean | +71% (in vitro) | 445 nM | In planta studies of cellular ABA dynamics |
| ABACUS2-100n [21] | Abscisic Acid (ABA) | edCitrine / edCerulean | +67% (in vitro) | 98 nM | High-affinity detection of low ABA concentrations |
| REKAR67 [47] | ERK Kinase Activity | miRFP670nano3 / miRFP720 | Higher than REKAR76 | Consistent with CFP/YFP EKAR | Multiplexed experiments requiring high dynamic range |
| REKAR76 [47] | ERK Kinase Activity | miRFP720 / miRFP670nano3 | Lower than REKAR67 | Consistent with CFP/YFP EKAR | Multiplexed experiments requiring low signal variance |
| cpFLIPPi-5.3m [8] | Phosphate (Pi) | eCFP / cpVenus | Quantified via FRET ratio | Defined by binding protein | Subcellular Pi monitoring in plant roots |
Experimental Protocols for Validation and Calibration
Protocol 1: Sensitized FRET Imaging and Analysis for Plant Tissues
This protocol, adapted from studies on phosphate dynamics in Brachypodium distachyon, details how to quantify FRET efficiency in plant tissues using confocal microscopy [8].
- Sample Preparation: Generate transgenic plants expressing the FRET biosensor (e.g., cpFLIPPi-5.3m) and essential controls: a non-responsive mutant (cpFLIPPi-Null), donor-only (eCFP), and acceptor-only (cpVenus) lines. Include wild-type plants for background subtraction.
- Image Acquisition: Use a confocal microscope with suitable lasers and filters. For a CFP/YFP pair, collect three images:
- FRET Channel: Excitate CFP (e.g., 458 nm laser) and collect emission in the YFP range (e.g., 535/30 nm bandpass filter).
- Donor Channel: Excitate CFP and collect its emission (e.g., 480/30 nm bandpass filter).
- Acceptor Channel: Excitate YFP (e.g., 514 nm laser) and collect its emission (e.g., 535/30 nm bandpass filter).
- Sensitized FRET Calculation: Process images using a semi-automated ImageJ macro or similar software. The calculation corrects for spectral bleed-through (SBT) and cross-excitation:
Corrected FRET = FRET Image - (A * Donor Image) - (B * Acceptor Image)- Factors A and B are determined from donor-only and acceptor-only control samples, respectively.
- Ratioing and Quantification: Create a sensitized FRET ratio image by dividing the Corrected FRET image by the Donor image. This ratio is proportional to the analyte concentration.
Protocol 2: Multiplexed Biosensor Barcoding for Robust Calibration
Imaging fluctuations and photobleaching can distort FRET ratios. This calibration protocol uses internal standards to generate quantitative, reproducible data [48].
- Standard Preparation: Create a set of calibration standard constructs: a high-FRET standard ("FRET-ON"), a low-FRET standard ("FRET-OFF"), and donor-only and acceptor-only constructs.
- Cotransformation: Introduce the biosensor of interest and the calibration standards into subsets of cells within the same experiment.
- Image Acquisition: Acquire images of all cell populations under identical imaging parameters.
- Signal Normalization: Use the fluorescence signals from the internal barcodes to normalize the FRET ratios from the experimental biosensor. This corrects for variations in laser power, detector sensitivity, and path length.
The experimental workflow for FRET biosensor development and application, from design to calibrated measurement, follows a structured path.
The Scientist's Toolkit: Key Research Reagent Solutions
Successful implementation of FRET imaging requires specific reagents and genetic tools. This table catalogs essential items for developing and applying FRET biosensors in plant research.
Table 3: Essential Research Reagents for FRET Biosensor Development
| Reagent / Material | Function in FRET Experiments | Example Instances |
|---|---|---|
| Genetically Encoded Biosensors | Core molecular tool that changes fluorescence upon analyte binding. | ABACUS2s (ABA) [21], cpFLIPPi-5.3m (Phosphate) [8], EKAR/REKAR (ERK activity) [47] |
| Control Sensor Constructs | Critical for distinguishing specific sensor response from artifactual signals. | cpFLIPPi-Null (phosphate-insensitive control) [8], Donor-only (eCFP), Acceptor-only (cpVenus) constructs [8] |
| Calibration Standard Constructs | Internal controls for normalizing FRET ratios and correcting for imaging artifacts. | "FRET-ON" and "FRET-OFF" standard proteins [48] |
| Fluorescent Protein Variants | FRET donor-acceptor pairs with optimized brightness, pH stability, and spectral profiles. | CFP/YFP variants, red/Far-Red pairs (miRFP670nano3/miRFP720) [47] |
| Modular Cloning Systems | For efficient vector assembly and testing of different sensor architectures (linkers, FPs). | Systems like Gibson Assembly were used for constructing REKAR biosensors [47] |
Maximizing the dynamic range and signal-to-noise ratio of FRET biosensors is achievable through a multi-faceted approach. As demonstrated by the compared sensors, strategic engineering of fluorophore pairs, sensory domains, and linkers directly translates to enhanced performance. Furthermore, rigorous experimental protocols and internal calibration standards are not merely best practices but are essential for transforming qualitative observations into robust, quantitative data. By leveraging these strategies and reagents, researchers can develop and deploy highly sensitive biosensors to illuminate the complex dynamics of plant analytes, from hormone signaling in response to abiotic stress to nutrient distribution within root systems.
The deployment of Förster Resonance Energy Transfer (FRET)-based biosensors in plant research represents a paradigm shift in our ability to monitor cellular processes with spatiotemporal precision. FRET functions as a "spectroscopic ruler" that is exquisitely sensitive to distance changes in the 1-10 nanometer range, making it ideal for tracking molecular interactions and conformational changes in live cells [1] [49]. These biosensors typically consist of donor and acceptor fluorescent proteins flanking a sensing domain that undergoes conformational changes in response to specific analytes or cellular conditions [50]. When applied to plant systems, researchers encounter unique challenges stemming from plant-specific physiology, including autofluorescence, cell wall barriers, complex compartmentalization, and the simultaneous need to monitor multiple signaling pathways.
This guide objectively compares FRET sensor technologies through the critical lens of sensor orthogonality—the ability to operate multiple sensors without interference—and physiological interference—the impact of sensor expression on plant health and function. We present experimental data and methodologies that address these challenges directly, providing researchers with a framework for selecting and optimizing FRET sensors for specific in-plant applications, from fundamental phytobiology to agricultural biotechnology.
Fundamental Principles and Design Strategies for Robust Plant FRET Sensors
Core Mechanism of FRET and Design Considerations
FRET relies on non-radiative energy transfer through dipole-dipole coupling between a donor fluorophore and an acceptor fluorophore. The efficiency of this transfer (E) depends on the inverse sixth power of the distance (r) between them (E = R₀⁶/(R₀⁶ + r⁶)), where R₀ is the Förster distance at which efficiency is 50% [1] [50]. This distance dependency establishes the fundamental working range of FRET biosensors, confining their effective detection to molecular events occurring within 1-10 nanometers [49] [51].
Genetically encoded FRET sensors for plant applications typically adopt either intramolecular or intermolecular designs:
- Intramolecular sensors incorporate donor and acceptor fluorescent proteins within a single polypeptide chain, along with a sensing domain that modulates their proximity upon analyte binding [1]. This design offers consistent expression stoichiometry but may present challenges with proper folding in plant cells.
- Intermolecular sensors rely on interactions between separately expressed proteins tagged with different fluorophores [1]. While potentially offering greater flexibility, these sensors face challenges with expression level matching in plant tissues, which can complicate quantitative interpretation.
The most established FRET pairs for biological sensing utilize Cyan and Yellow Fluorescent Proteins (CFP-YFP), though newer pairs with red-shifted spectra are emerging to overcome plant autofluorescence [51].
Table: Comparison of Major FRET Fluorophore Classes for Plant Applications
| Fluorophore Class | Key Representatives | Advantages | Limitations for Plant Applications | Orthogonality Potential |
|---|---|---|---|---|
| Fluorescent Proteins (FPs) | CFP, YFP, RFP, GFP variants | Genetically encodable; subcellular targetable; minimal disruption to cells [50] [51] | Sensitive to pH and temperature; can form aggregates; large size may sterically interfere with target function [51] | Moderate (spectral overlap can limit multiplexing) |
| Organic Fluorescent Dyes | Fluorescein, Rhodamine, Acridine | Brightness; photostability; small size [51] | Require delivery mechanisms or conjugation chemistry; difficult to target to specific subcellular compartments in plants | Low (limited to extracellular applications or microinjected cells) |
| Quantum Dots (QDs) | CdTe, CdSe–ZnS core–shell | Extreme brightness; photostability; narrow emission peaks [11] | Potential cytotoxicity; size-dependent delivery challenges; environmental concerns | High (narrow emission facilitates multiplexing) |
| Lanthanides | Europium, Terbium complexes | Long lifetime enables time-gating to avoid autofluorescence [49] | Not genetically encodable; require complex conjugation | High (time-resolved detection avoids background) |
Critical Analysis of Sensor Orthogonality: Strategies and Experimental Validation
Spectral and Temporal Separation Approaches
Sensor orthogonality enables researchers to monitor multiple analytes simultaneously within the same plant cell, providing systems-level insights into signaling networks. The fundamental challenge lies in the limited spectral space and the potential for cross-talk between different FRET pairs. Spectral orthogonality is achieved by selecting fluorophores with non-overlapping excitation and emission spectra. While the traditional CFP-YFP pair works well for single-analyte detection, multiplexed imaging requires careful selection of FP pairs with distinct spectral profiles, such as blue-red or green-red combinations [52].
Recent advances address this challenge through barcoding strategies and computational unmixing. One innovative approach cultures cells expressing different biosensors separately, then labels them with distinct combinations of blue or red fluorescent proteins targeted to different subcellular locations. These "barcoded" cells are mixed before imaging, and a machine learning model identifies each biosensor based on its barcode pattern, enabling simultaneous imaging of multiple FRET sensors despite spectral overlap [52]. This method effectively expands the usable spectral space without requiring entirely new fluorophore development.
Temporal orthogonality represents another strategic approach, particularly through fluorescence lifetime imaging (FLIM-FRET). This technique measures the donor fluorescence lifetime, which is independent of sensor concentration and excitation intensity, making it robust for multiplexed experiments [49]. When combined with time-gated detection of lanthanide-based probes, this approach can effectively separate FRET signals from background autofluorescence—a significant advantage in plant tissues [49].
Experimental Validation of Orthogonality: A Protocol
Objective: To validate the orthogonality of two FRET biosensors (e.g., for Ca²⁺ and ATP) in plant cells.
Materials:
- Plant lines expressing individual FRET biosensors
- Plant line co-expressing both FRET biosensors
- Confocal microscope with spectral imaging capabilities
- Image analysis software (e.g., ImageJ with FRET analysis plugins)
Methodology:
- Express sensors in controlled systems: Generate plant lines expressing each biosensor individually under the same promoter, plus a line co-expressing both biosensors [8].
- Acquire reference spectra: For each individual biosensor line, collect emission spectra with donor excitation to establish reference spectral profiles [52] [8].
- Image co-expressing lines: Capture images of cells co-expressing both biosensors using multiple excitation wavelengths and emission channels.
- Linear unmixing: Apply linear unmixing algorithms to separate the contributions of each biosensor based on the reference spectra [52].
- Stimulus application: Apply specific stimuli for each analyte (e.g., osmotically induced Ca²⁺ waves and metabolic modulators for ATP) to confirm independent sensor responses in co-expressing cells.
- Quantify cross-talk: Calculate the percentage of signal bleed-through between channels and the false activation rate of one biosensor when stimulating the other analyte.
Expected Outcomes: Successful orthogonal sensing demonstrates less than 5% cross-talk and maintains dynamic range comparable to singly-expressed sensors. The positive control (cpFLIPPi-Null sensor) should show no FRET ratio shift, confirming that observed changes are analyte-specific [8].
Figure 1: Experimental workflow for validating FRET sensor orthogonality in plant systems.
Quantitative Assessment of Physiological Interference
Systematic Evaluation of Sensor Impact on Plant Health
The expression of foreign genetic material, including FRET biosensors, inevitably raises concerns about physiological interference. These effects can manifest as altered growth patterns, changes in metabolic activity, or aberrant stress responses. A comprehensive assessment of physiological interference should include both phenotypic and molecular analyses.
Growth and Development Metrics:
- Root elongation rates: Compare primary root growth between sensor-expressing and wild-type plants over 7-14 days under controlled conditions.
- Biomass accumulation: Measure fresh and dry weight of shoots and roots at specific developmental stages.
- Repductive fitness: Quantify flowering time, seed set, and germination rates in subsequent generations.
Molecular and Physiological Assays:
- Chlorophyll fluorescence: Use pulse-amplitude modulation (PAM) fluorometry to assess photosynthetic efficiency (Fv/Fm ratio) as an indicator of plant stress.
- Transcriptomic analysis: Perform RNA sequencing on sensor-expressing plants to identify differentially expressed genes, particularly those involved in stress responses, immune pathways, and metabolism.
- Metabolite profiling: Analyze primary metabolites to detect potential perturbations in central metabolic pathways.
The cpFLIPPi series of phosphate sensors exemplifies proper control implementation by including a cpFLIPPi-Null variant with a mutation that prevents phosphate binding. This control accounts for non-specific effects of sensor expression, ensuring that observed FRET ratio shifts genuinely reflect phosphate fluctuations rather than artifacts [8].
Table: Physiological Interference Assessment of Documented Plant FRET Sensors
| Sensor Name | Target Analyte | Expression System | Documented Physiological Effects | Control Strategies Employed |
|---|---|---|---|---|
| cpFLIPPi-5.3m | Phosphate | Brachypodium distachyon (cytosol/plastid) | Minimal impact on plant growth and mycorrhizal colonization when expressed at moderate levels [8] | Use of non-binding cpFLIPPi-Null control; tissue-specific promoters (BdPT7) to limit expression to relevant cells [8] |
| Cameleon | Ca²⁺ | Various plant species | Buffer Ca²⁺ transients when overexpressed; can alter Ca²⁺-dependent processes if not properly calibrated [50] | Expression level optimization; use of weaker promoters; comparison with dye-based measurements |
| Cys-His | Zn²⁺ | Arabidopsis thaliana | No reported growth defects at moderate expression levels | Targeted to specific subcellular compartments to minimize disruption to zinc homeostasis |
| GCaMP | Ca²⁺ | Various plant species | Potential buffering of native Ca²⁺ signals at high concentrations | Codon optimization for plant expression; inducible expression systems |
Experimental Protocols for Minimizing Sensor-Induced Artifacts
Optimizing Expression and Targeting Strategies
Controlling biosensor expression is paramount for minimizing physiological interference while maintaining sufficient signal for detection. The following protocol outlines a systematic approach for optimizing FRET sensor expression in plant systems:
Promoter Selection and Engineering:
- Test multiple promoter strengths: Compare sensor performance under constitutive promoters of varying strengths (e.g., 35S, UBQ10) and cell-type-specific promoters (e.g., BdPT7 for arbuscule-containing cells) [8].
- Implement inducible systems: Use chemically-inducible or stress-responsive promoters to limit sensor expression to specific experimental windows.
- Employ genetic circuits: Develop synthetic circuits that auto-regulate sensor expression to maintain optimal levels.
Subcellular Targeting Verification:
- Confirm localization: Validate subcellular targeting using co-localization with established organellar markers.
- Assess targeting efficiency: Quantify the percentage of cells showing correct sensor localization.
- Evaluate compartment effects: Test sensor function in different subcellular environments, as performance can vary between compartments.
Sensor Concentration Titration:
- Generate expression series: Create multiple transgenic lines with varying expression levels.
- Determine optimal range: Identify the minimum expression level that provides sufficient signal-to-noise ratio without causing phenotypic abnormalities.
- Correlate expression with function: Establish the relationship between sensor abundance and dynamic range.
The successful implementation of the cpFLIPPi-5.3m sensor in Brachypodium distachyon exemplifies this approach, where both constitutive (ZmUb1) and cell-type-specific (BdPT7) promoters were employed, with expression carefully monitored to avoid disruption of arbuscular mycorrhizal symbiosis [8].
Advanced Calibration and Signal Processing Techniques
Accurate interpretation of FRET signals requires robust calibration methods that account for plant-specific challenges, including high autofluorescence, light scattering, and variable sensor expression.
FRET Calibration Using Reference Standards:
- Incorporate calibration standards: Co-culture or co-express "FRET-ON" and "FRET-OFF" reference standards alongside experimental sensors [52] [53].
- Normalize signals: Use reference standards to normalize FRET ratios, compensating for variations in excitation intensity, detector sensitivity, and photobleaching [52].
- Calculate actual FRET efficiency: Include donor-only and acceptor-only controls to determine crosstalk correction factors and compute true FRET efficiency rather than relying solely on ratio measurements [52].
Sensitized FRET Imaging Protocol for Plant Tissues:
- Image acquisition:
- Collect donor channel images with donor excitation
- Collect FRET channel images with donor excitation
- Collect acceptor channel images with acceptor excitation
- Collect autofluorescence images from wild-type tissues under identical settings
Background subtraction:
- Subtract autofluorescence signals using the wild-type reference images
- Apply uniform thresholding to eliminate low-signal regions
Crosstalk correction:
- Determine spectral bleed-through coefficients using donor-only and acceptor-only specimens
- Apply correction algorithms to calculate net FRET signals
Ratio calculation and calibration:
- Compute FRET ratio (acceptor emission/donor emission)
- Normalize using reference standards imaged under identical conditions [8]
This comprehensive calibration approach restores the expected reciprocal relationship between donor and acceptor signals that is often obscured by imaging artifacts in plant tissues, enabling more reliable quantitative comparisons across experiments and extended time-lapse studies [52] [53].
Figure 2: Identifying and mitigating sources of physiological interference in plant FRET biosensing.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of FRET biosensing in plant systems requires carefully selected reagents and methodologies. The following table compiles key solutions for addressing the dual challenges of orthogonality and physiological interference.
Table: Research Reagent Solutions for Plant FRET Biosensing
| Reagent/Method | Specific Examples | Primary Function | Considerations for Plant Applications |
|---|---|---|---|
| Orthogonal FRET Pairs | CFP-YFP; GFP-RFP; BFP-mCherry [51] | Enable multiplexed imaging of multiple analytes | Select pairs with minimal spectral overlap; verify performance in plant tissue context |
| Reference Standards | FRET-ON and FRET-OFF constructs [52] [53] | Calibrate signals across experiments and imaging sessions | Express in same plant background; confirm stable expression over time |
| Control Sensors | cpFLIPPi-Null (binding-deficient mutant) [8] | Distinguish specific responses from artifacts | Develop for each sensor type; validate lack of analyte response |
| Tissue-Specific Promoters | BdPT7 (arbuscule-containing cells) [8] | Limit sensor expression to relevant cell types | Verify specificity in target species; monitor for leaky expression |
| Subcellular Targeting Sequences | Plastid, nuclear, plasma membrane localization signals [8] | Direct sensors to specific compartments | Confirm proper targeting; test sensor function in each compartment |
| Spectral Unmixing Algorithms | Linear unmixing; machine learning classification [52] | Resolve overlapping signals in multiplexed experiments | Validate with control samples; account for plant autofluorescence |
| FRET Calibration Protocols | Sensitized emission with crosstalk correction [8] | Quantify accurate FRET efficiency | Adapt for specific microscope systems; establish plant-specific correction factors |
The comparative analysis presented in this guide demonstrates that addressing sensor orthogonality and physiological interference requires integrated strategies spanning sensor design, expression control, and signal processing. The most successful implementations combine thoughtful sensor selection with rigorous validation using appropriate controls and calibration standards.
Key findings indicate that:
- Orthogonality is best achieved through combined spectral and computational approaches, particularly using barcoding strategies and advanced unmixing algorithms that overcome the limitations of traditional CFP-YFP pairs.
- Physiological interference can be minimized through targeted expression systems and careful titration of sensor levels, with the cpFLIPPi system providing an exemplary model with its use of binding-deficient controls and cell-type-specific promoters.
- Quantitative reliability depends on robust calibration methods that account for plant-specific challenges, with recent advances in reference standards enabling more accurate cross-experiment comparisons.
As FRET biosensing continues to evolve in plant research, emerging technologies including single-molecule FRET (smFRET) and nanomaterials-based sensors offer promising avenues for further reducing interference while enhancing detection specificity. By applying the systematic comparison and validation frameworks outlined here, researchers can select and implement FRET sensors that deliver maximal insight with minimal perturbation, advancing our understanding of plant function from molecular to organismal scales.
Förster Resonance Energy Transfer (FRET)-based biosensors are powerful tools for monitoring the spatiotemporal dynamics of biochemical activities in live cells. Their operation hinges on non-radiative energy transfer between a donor fluorophore and an acceptor fluorophore, which alters the emitted fluorescence ratio upon changes in the target analyte. However, the commonly used acceptor-to-donor signal ratio (FRET ratio) is highly sensitive to imaging parameters such as laser intensity and detector sensitivity, complicating data interpretation and comparison across experiments. Consequently, robust calibration and control strategies are paramount for generating reliable, quantitative biological data. This guide compares the performance of different calibration methodologies, providing researchers with a framework for selecting and implementing optimal practices for their specific applications in plant analyte research and beyond.
Table: Fundamental Challenges in FRET Ratiometric Measurement
| Challenge | Impact on Measurement | Common Consequence |
|---|---|---|
| Variable Imaging Conditions (laser power, detector sensitivity) | Alters raw FRET ratio independent of biological signal | Inability to compare data across sessions or labs [52] |
| Spectral Crosstalk | Acceptor directly excited by donor excitation wavelength; donor emission bleeds into acceptor channel | Over/underestimation of true FRET efficiency [54] |
| Photobleaching | Non-uniform fluorescence loss in donor and acceptor over time | Obscures reciprocal donor/acceptor signal trends and creates false kinetics [52] |
| Dynamic Range Limitations | Small conformational changes in biosensor yield small ratio changes | Reduced sensitivity and lower signal-to-noise ratio [39] |
Core Calibration Methodologies and Comparative Performance
A range of calibration methods has been developed to overcome the inherent vulnerabilities of ratiometric FRET measurements. These approaches vary in their complexity, hardware requirements, and the level of quantitative rigor they provide. The selection of a method often involves a trade-off between experimental simplicity and quantitative accuracy.
Figure 1: Logical workflow outlining major FRET measurement challenges and the primary calibration methodologies used to address them.
Donor and Acceptor-Only Controls for Crosstalk Correction
This foundational method involves imaging separate control samples expressing only the donor or the acceptor fluorophore under the same conditions used for the FRET biosensor. The signals from these controls are used to calculate and subtract the spectral bleed-through (SBT) and direct excitation coefficients, which are major sources of error in sensitized emission FRET measurements. The corrected FRET efficiency can then be calculated using established formulas. While this method is essential for determining absolute FRET efficiency, the process is time-consuming, and the multiple imaging steps can introduce additional variability.
FRET Standard Samples for Signal Normalization
To compensate for daily fluctuations in microscope performance, the use of stable calibration standards is highly effective. This approach involves engineering reference constructs locked in "FRET-ON" (high FRET efficiency) and "FRET-OFF" (low FRET efficiency) conformations using the same donor-acceptor pair as the biosensor. Theoretical and experimental studies have confirmed that by normalizing the biosensor's FRET ratio against these standards, the calibrated output becomes independent of variable imaging settings such as excitation intensity. This method is particularly powerful for long-term and cross-experimental studies, as it directly addresses the problem of instrument-dependent signal drift [52]. Furthermore, in multiplexed imaging, these standards can be introduced into subsets of cells via a barcoding system, streamlining calibrated measurements of multiple analytes simultaneously [52].
Advanced Ratiometric Analysis: Lux-FRET
For researchers requiring the highest quantitative accuracy, the Lux-FRET method provides a comprehensive spectral analysis. This technique uses three measured fluorescence signals: donor and acceptor emission during short-wavelength (donor) excitation, and acceptor emission during long-wavelength (acceptor) excitation. By calculating ratios from these three signals, Lux-FRET can determine not only the FRET efficiency but also the total abundance of donor and acceptor fluorophores. While this method provides a more complete picture, the signal-to-noise ratio (SNR) for these derived quantities is lower than that of a plain emission ratio due to error propagation. However, when calculating final ligand concentration, the SNR is comparable to that achieved by calibrating the standard emission ratio [55].
Table: Comparison of FRET Calibration and Control Methods
| Method | Key Procedure | Primary Advantage | Key Limitation | Best Suited For |
|---|---|---|---|---|
| Donor/Acceptor-Only Controls | Image separate donor-only and acceptor-only samples to calculate crosstalk coefficients [52]. | Corrects spectral bleed-through and direct excitation for absolute FRET efficiency. | Time-consuming; multiple steps can introduce variability [52]. | Determining quantitative FRET efficiency (E) in single-sensor experiments. |
| FRET Standard Samples | Normalize biosensor ratio against "FRET-ON" and "FRET-OFF" constructs in barcoded cells [52]. | Compensates for instrument drift; enables cross-experiment comparison [52]. | Requires generation and validation of additional stable cell lines/constructs. | Long-term kinetics studies and multiplexed imaging with multiple biosensors [52]. |
| Lux-FRET Analysis | Collect three fluorescence signals to calculate ratios for FRET efficiency and fluorophore concentration [55]. | Provides robust quantification of FRET efficiency and fluorophore stoichiometry [55]. | Lower signal-to-noise ratio for derived parameters due to error propagation [55]. | High-precision studies where concentration and efficiency are both critical. |
| In Situ Titration | Expose sensor to buffers with known analyte concentrations (e.g., Zn²⁺) to generate a calibration curve [56]. | Directly correlates FRET ratio to absolute analyte concentration in the cellular context. | Technically challenging; not feasible for all analytes (e.g., specific kinase activities). | Quantifying absolute concentrations of ions and metabolites (e.g., Pi, Zn²⁺, ATP) [8] [56]. |
Experimental Protocols for Robust Calibration
Protocol: In Situ Calibration for Quantitative Ion Measurement
This protocol is adapted from methods used to measure labile cytosolic Zn²⁺ and phosphate (Pi) and allows for the conversion of a FRET ratio into an absolute analyte concentration [56] [8].
- Sensor Expression: Transfert or transduce cells with the FRET biosensor targeted to the desired compartment (e.g., cytosol, plastids). Include control sensors (e.g., a non-binding "Null" variant) to account for non-specific ratio changes [8].
- Solution Preparation: Prepare a series of calibration buffers that mimic the intracellular milieu (ionic strength, pH) but contain a chelator (e.g., EDTA) to deplete the native analyte. For the high-concentration point, add a known quantity of the analyte (e.g., Zn²⁺, Pi) to saturate the sensor.
- Image Acquisition: For each cell, take a baseline measurement in normal growth media. Then, perfuse the cells sequentially with the calibration buffers, finishing with the saturated solution. Acquire donor and acceptor channel images at each step using constant imaging parameters.
- Data Analysis: For each cell, plot the FRET ratio (or corrected FRET efficiency) against the known free analyte concentration in the buffers. Fit the data with a binding isotherm (e.g., Hill equation). This calibration curve quantitatively links the observed ratio to the intracellular analyte concentration.
Protocol: Using FRET Standards for Normalization
This protocol leverages FRET-ON and FRET-OFF standards to normalize data against imaging fluctuations [52].
- Standard Preparation: Generate cell lines or constructs expressing stable FRET-ON and FRET-OFF standards. These can be co-cultured with biosensor-expressing cells or identified via a barcoding system using spectrally distinct fluorescent proteins targeted to different subcellular locations [52].
- Simultaneous Imaging: Image the biosensor-expressing cells and the standard-expressing cells in the same session or same field of view. Acquire donor and acceptor channel images for all samples.
- Ratio Calculation and Normalization: Calculate the raw FRET ratio (acceptor emission / donor emission) for both the biosensor (Rbiosensor) and the standards (RON, ROFF). The calibrated FRET ratio (Rcal) can be computed as: R_cal = (R_biosensor - R_OFF) / (R_ON - R_OFF) This normalized ratio is largely independent of the specific laser power or detector gain used [52].
The Scientist's Toolkit: Key Research Reagent Solutions
Successful implementation of FRET calibration requires specific reagents and tools. The following table details essential components for setting up controlled experiments.
Table: Essential Reagents and Tools for FRET Biosensor Calibration
| Reagent / Tool | Function in Calibration | Exemplars & Notes |
|---|---|---|
| FRET Standard Constructs | Provide internal reference for normalizing instrument-dependent signal variation. | "FRET-ON" (high-E) and "FRET-OFF" (low-E) constructs using the same FP pair as the biosensor [52]. |
| Control Sensor Variants | Distinguish specific sensor response from non-specific environmental effects. | "cpFLIPPi-Null" (a Pi-binding site mutant); "REKAR-T/A" (phospho-acceptor site mutant) [8] [47]. |
| Optimized FP Pairs | Maximize FRET efficiency and dynamic range while minimizing spectral crosstalk. | Cyan/Yellow: mTurquoise2/mVenus (bright, photostable, well-separated spectra) [54]. Red/Far-Red: miRFP670nano3/miRFP720 (enables multiplexing with CFP/YFP sensors) [47]. |
| Ion Buffers & Chelators | For in situ titration to generate a calibration curve for absolute quantification. | Two-ion buffer system (e.g., Zn²⁺/EDTA) for precise control of free analyte concentration [56]. |
| Image Analysis Software | Essential for calculating ratios, applying crosstalk corrections, and analyzing kinetics. | FIJI/ImageJ with semi-automated macros for sensitized FRET analysis [8]. Custom MATLAB scripts for advanced analysis like Lux-FRET [55]. |
Accurate ratiometric measurement with FRET biosensors is not achievable through a one-size-fits-all approach but relies on a systematic strategy of calibration and control. The choice between methods depends on the experimental goal: using FRET standards is ideal for normalizing against instrumental noise in long-term or multiplexed imaging of relative dynamics, while in situ titration is necessary for determining absolute analyte concentrations. For the highest level of quantification, particularly in plant analyte research, combining these methods—using controls for crosstalk correction, standards for normalization, and optimized FP pairs for a strong signal—provides the most robust framework for generating reliable, comparable, and quantitatively meaningful data.
Critical Evaluation and Comparison: Validating FRET Sensor Performance
The development and application of Förster Resonance Energy Transfer (FRET)-based biosensors have revolutionized the study of biological processes by enabling real-time monitoring of analytes and cellular activities in living systems. For researchers investigating plant analytes, selecting the appropriate FRET biosensor requires a critical understanding of three fundamental performance parameters: affinity (quantified by the dissociation constant, Kd), dynamic range (the maximum change in signal output), and kinetics (the response time to analyte binding and dissociation). The affinity (Kd) of a biosensor determines the concentration range over which it can effectively measure an analyte, with lower Kd values indicating higher affinity and suitability for detecting lower analyte concentrations [34]. The dynamic range, often reported as the maximum change in emission ratio (ΔR/R), reflects the sensor's signal-to-noise ratio and its ability to detect subtle changes in analyte concentration [47]. Meanwhile, kinetic parameters govern the temporal resolution of the biosensor, determining how quickly it can respond to rapid changes in analyte levels, which is particularly crucial for capturing transient signaling events in plant systems [1].
The optimization of these parameters involves sophisticated protein engineering approaches, including mutagenesis of binding pockets, optimization of linker sequences between domains, and selection of appropriate fluorescent protein pairs [34] [57]. For plant research specifically, additional challenges such as tissue autofluorescence, subcellular compartmentalization, and the unique chemical environment of plant cells must be considered when selecting and optimizing FRET biosensors [22] [8]. This guide provides a comprehensive comparison of FRET biosensor performance metrics based on recent experimental data, with a particular emphasis on applications relevant to plant analyte research.
Performance Comparison of FRET Biosensors
Table 1: Performance Metrics of Featured FRET Biosensors for Plant Analytes
| Sensor Name | Target Analyte | Affinity (Kd) | Dynamic Range (ΔR/R) | Key Applications | Organism/System |
|---|---|---|---|---|---|
| ECATS2 [34] | Extracellular ATP | >3x higher affinity than ecATeam3.10 (original) | Ratiometric readout | Detection of ATP release upon hypoosmotic stress | Cultured astrocytes |
| cpFLIPPi-5.3m [8] | Phosphate (Pi) | Not specified | Sensitized FRET emission | Monitoring intracellular Pi dynamics during AM symbiosis | Brachypodium distachyon |
| CaMPARI-nano [57] | Ca²⁺ | 17-25 nM | Comparable to parental CaMP2_F391W | Detection of low nanomolar Ca²⁺ dynamics | Multiple cell types |
| BGECO-nano [57] | Ca²⁺ | ~3x lower Kd than B-GECO1 template | Not specified | Ultrahigh-affinity Ca²⁺ imaging | Multiple cell types |
| RCaMP-nano [57] | Ca²⁺ | ~3x lower Kd than R-CaMP1.01 template | Not specified | Ultrahigh-affinity Ca²⁺ imaging | Multiple cell types |
| REKAR67/REKAR76 [47] | ERK activity | Not specified | REKAR67: Higher dynamic range; REKAR76: Lower variance | Multiplexed imaging with other pathways | MCF-10A cells |
Table 2: Advanced FRET Biosensor Engineering Strategies and Outcomes
| Engineering Strategy | Sensor Example | Effect on Affinity | Effect on Dynamic Range | Effect on Kinetics |
|---|---|---|---|---|
| Binding site mutagenesis [34] | ECATS2 (R103A/R115A) | >3-fold increase | Maintained ratiometric properties | Not specified |
| Linker length optimization [57] | CaMPARI-nano | Kd = 19 nM (from 121 nM) | Preserved dynamic range and brightness | Not specified |
| Topology mutation [57] | cp-CaMP2_F391W | Kd = 54 nM (from 121 nM) | Comparable to original | Not specified |
| Combined approaches [57] | cp-CaMP2F391Wlinker12+3mut | Kd = 19 nM (ultrahigh-affinity) | Maintained optical properties | Not specified |
| Fluorophore position swapping [47] | REKAR67 vs REKAR76 | Consistent ERK reporting | Differential dynamic range vs variance | Comparable to EKAREN4 |
Experimental Protocols for FRET Biosensor Characterization
Affinity Determination (Kd Measurement)
The protocol for determining biosensor affinity involves generating a dose-response curve by measuring FRET signals across a range of analyte concentrations. For extracellular ATP sensors like ECATS2, researchers apply ATP concentrations ranging from near the detection limit (e.g., 3 µM) to saturation levels (e.g., 300 µM) while continuously monitoring FRET ratio changes [34]. The FRET ratio is calculated from background-subtracted images, with cell masks created using thresholds of at least two times the mean background intensity. For intracellular sensors, purified proteins may be used in solution-based assays to determine apparent dissociation constants without interference from cellular environment factors [34]. Data are typically fitted using appropriate binding models in scientific analysis software such as OriginPro to derive the Kd value. For plant applications, this protocol must be adapted to account for cell wall permeability and potential compartmentalization of analytes.
Dynamic Range Quantification
The dynamic range of FRET biosensors is typically quantified as the maximum change in emission ratio between the analyte-free and analyte-saturated states. For ratiometric FRET biosensors like those used for phosphate detection in plants, the dynamic range is determined by measuring sensitized FRET emission before and after analyte application [8]. The emission ratio is calculated from background-corrected images collected through specific filter sets: cyan (438/29 nm excitation, 470/24 nm emission) and yellow (510/10 nm excitation, 540/30 nm emission) channels [34]. In plant systems, control sensors with mutated analyte-binding domains (e.g., cpFLIPPi-Null) are essential to account for non-specific FRET ratio changes caused by intracellular ionic shifts or environmental factors [8].
Kinetic Parameter Assessment
The kinetic characterization of FRET biosensors involves measuring the time course of FRET ratio changes in response to rapid changes in analyte concentration. This is typically performed using live-cell microscopy with continuous perfusion systems to enable rapid solution exchange. The association rate (kₒₙ) and dissociation rate (kₒff) constants can be determined by fitting the time-dependent FRET ratio changes to appropriate kinetic models. For plant systems, specialized imaging chambers may be required to maintain tissue viability during prolonged experiments, and perfusion rates may need optimization to ensure adequate analyte delivery through plant cell walls [22].
Signaling Pathways and Experimental Workflows
The following diagrams illustrate key signaling pathways and experimental workflows relevant to FRET biosensor applications in plant research.
Diagram 1: FRET biosensor signaling pathway for analyte detection.
Diagram 2: Experimental workflow for plant FRET biosensor application.
Research Reagent Solutions for FRET Biosensing
Table 3: Essential Research Reagents for FRET Biosensor Experiments
| Reagent/Category | Specific Examples | Function/Application | Relevance to Plant Research |
|---|---|---|---|
| Fluorescent Protein Pairs | mseCFP/mVenus [34], miRFP670nano3/miRFP720 [47], CFP/YFP [58] | FRET donor-acceptor pairs | Spectral compatibility with plant autofluorescence |
| Molecular Biology Kits | NEB Q5 Site-Directed Mutagenesis kit, NEB Gibson/HiFi Assembly [34] | Biosensor engineering and construction | Optimization for plant-specific codon usage |
| Cell Culture Reagents | DMEM, cosmic calf serum, penicillin-streptomycin [34] | Maintenance of cell lines | Adaptation for plant protoplast culture |
| Imaging Systems | Olympus IX83 microscope, Andor Zyla camera, Lumencor SpectraX [34] | Live-cell FRET imaging | Specialized setups for plant tissue imaging |
| Image Analysis Software | ImageJ/FIJI [34], NIS-Elements [47], MATLAB [47] | FRET ratio calculation and analysis | Custom macros for plant cell segmentation |
| Viral Delivery Systems | Adeno-associated viral vectors (AAVs) [58] | In vivo sensor delivery | Optimization for plant transformation |
| Specialized Linkers | E/RK linkers [34], Flexible linkers (GGTGGS)ₙ [57] | Sensor domain connection | Optimization for plant-specific applications |
The systematic benchmarking of FRET biosensor performance parameters—affinity, dynamic range, and kinetics—provides critical guidance for researchers selecting appropriate tools for plant analyte research. Recent engineering advances, including binding site mutagenesis, linker optimization, and topological restructuring, have significantly enhanced biosensor performance, enabling detection of analytes across wider concentration ranges with improved sensitivity. The experimental protocols and reagent solutions outlined in this guide offer practical frameworks for implementing these advanced biosensors in plant systems. As FRET biosensor technology continues to evolve, incorporating red-shifted fluorescent pairs and addressing challenges such as recombination during viral delivery [58] will further expand their utility in plant research, opening new avenues for understanding plant signaling pathways and physiological responses at the molecular level.
Comparative Analysis of Leading Hormone Biosensors (e.g., ABAleons vs. ABACUS2)
The direct visualization and quantification of phytohormones in living plant tissues represent a significant challenge in plant biology. Among these hormones, abscisic acid (ABA) plays a central role in mediating plant responses to abiotic stresses such as drought, salinity, and extreme temperatures, while also influencing growth and developmental processes [59]. Understanding the spatiotemporal dynamics of ABA is essential for deciphering how plants perceive environmental cues and translate them into adaptive responses. Traditional methods for hormone measurement, including immunoassays and mass spectrometry, require tissue destruction and lack the spatial and temporal resolution needed to capture rapid, cell-type-specific hormone dynamics.
The development of Förster Resonance Energy Transfer (FRET)-based biosensors has revolutionized the field by enabling non-destructive, real-time monitoring of hormone concentrations in living cells with high resolution [21] [60]. These genetically encoded biosensors typically consist of a ligand-binding domain flanked by two fluorescent proteins that function as a FRET pair. Upon ligand binding, a conformational change in the sensory domain alters the distance or orientation between the fluorophores, resulting in a measurable change in FRET efficiency [14]. This review provides a comprehensive comparative analysis of two leading ABA biosensor families: the pioneering ABAleons and the next-generation ABACUS2 sensors, examining their engineering principles, performance characteristics, and experimental applications to guide researchers in selecting the most appropriate tool for their specific investigations.
Technical Specifications and Performance Metrics
Comparative Engineering and Design Principles
Table 1: Design and Engineering Features of ABA FRET Biosensors
| Feature | ABAleons | ABACUS1 | ABACUS2-400n | ABACUS2-100n |
|---|---|---|---|---|
| Basic Architecture | PYL1-ABA-ABI1 complex connected to FRET pair [60] | PYL1 (H87P)-ABI1aid connected to FRET pair with attB linkers [21] | Evolved from ABACUS1 with modified linkers and latch [21] | Evolved from ABACUS1 with modified linkers and latch [21] |
| Sensory Domain | PYL1 ABA receptor & PP2C phosphatase (ABI1) [60] | Mutated PYL1 (H87P) receptor & truncated ABI1 (ABI1aid) [21] | PYL1 with R143S latch mutation & ABI1aid [21] | PYL1 with E141D latch mutation & ABI1aid [21] |
| FRET Pair | N/A (Original publication doesn't specify FPs) | edCitrine (acceptor) & edCerulean (donor) [21] | edCitrine (acceptor) & edCerulean (donor) [21] | edCitrine (acceptor) & edCerulean (donor) [21] |
| Key Mutations | N/A | PYL1 H87P [21] | PYL1 R143S ("latch" mutation), shorter proline linkers [21] | PYL1 E141D ("latch" mutation), shorter proline linkers [21] |
| Linker Structure | Not specified | Flexible attB1/attB2 linkers [21] | Shorter, less flexible proline linkers [21] | Shorter, less flexible proline linkers [21] |
Quantitative Performance Comparison
Table 2: Performance Characteristics of ABA FRET Biosensors
| Parameter | ABAleons | ABACUS1-2μ | ABACUS2-400n | ABACUS2-100n |
|---|---|---|---|---|
| ABA Affinity (KD) | ~100-600 nM [60] | ~1.1-1.8 μM [21] | 445 nM [21] | 98 nM [21] |
| Emission Ratio Change | Low (Poor signal-to-noise ratio) [21] | High positive ratio change [21] | +71% (Highest among ABACUS sensors) [21] | +67% [21] |
| Signal-to-Noise Ratio | Poor [21] | High [21] | High [21] | High [21] |
| Orthogonality | Strong ABA hyposensitivity phenotypes [21] | Minor ABA hypersensitivity phenotypes [21] | Improved orthogonality [21] | Improved orthogonality [21] |
| Dynamic Range | Suitable for endogenous ABA [21] | Poor sensitivity for endogenous ABA [21] | Optimized for in planta studies [21] | High affinity for low ABA levels [21] |
Experimental Protocols and Methodologies
Biosensor Validation and Characterization Workflows
The development and validation of both ABAleons and ABACUS biosensors involved rigorous in vitro and in planta testing to characterize their performance parameters. The following workflow outlines the standard protocol for biosensor validation:
Diagram Title: Biosensor Validation Workflow
For in vitro characterization, biosensor proteins are typically expressed in E. coli and purified using affinity chromatography (e.g., Ni-NTA columns for His-tagged proteins) [21] [45]. Purified proteins are exposed to a concentration series of ABA in a buffer system, and fluorescence emissions are measured for both donor and acceptor channels using a fluorescence spectrophotometer or microplate reader. The FRET ratio is calculated as the emission intensity of the acceptor divided by the emission intensity of the donor when exciting the donor fluorophore [45]. Titration curves are generated by plotting the FRET ratio against ABA concentration, and the dissociation constant (KD) is determined through nonlinear regression fitting.
In plant experiments, transgenic Arabidopsis lines expressing the biosensors are generated, and FRET imaging is typically performed using rationetric fluorescence microscopy or confocal microscopy with appropriate filter sets for the specific fluorescent protein pair [21] [60]. For quantitative imaging, protocols must account for background autofluorescence, which can be a significant challenge in plant tissues [22]. The development of protoplast-based FRET imaging offers a rapid alternative for functional screening, though it may not fully recapitulate the complexity of intact tissues [22].
Experimental Applications in Stress Response Research
Table 3: Documented Experimental Applications of ABA Biosensors
| Experimental Context | ABAleons Applications | ABACUS2 Applications |
|---|---|---|
| Low Humidity Stress | Detected ABA increases in guard cells [60] | Mapped root ABA accumulation in elongation zone; revealed phloem-transported ABA unloading [21] |
| Salt Stress (NaCl) | Detected ABA concentration increases in guard cells and roots [60] | Revealed cellular basis for systemic ABA functions in roots [21] |
| Osmotic Stress | Detected ABA increases in roots [60] | Confirmed essential role of phloem ABA and root signaling in maintaining root growth [21] |
| ABA Transport Studies | Visualized long-distance transport from hypocotyl to shoot and root [60] | High-resolution mapping of stress-induced ABA dynamics [21] |
Critical Analysis of Signaling Pathways and Mechanisms
The molecular basis for ABA sensing involves a core signaling pathway that both ABAleons and ABACUS biosensors exploit, albeit with different engineering optimizations. The following diagram illustrates the fundamental ABA signaling pathway and how biosensors incorporate its components:
Diagram Title: ABA Pathway and Biosensor Engineering
Both biosensor families utilize the core ABA perception mechanism involving PYR/PYL/RCAR receptors and PP2C phosphatases [60]. In the natural signaling pathway, ABA binding induces conformational changes in the receptor, particularly in the 'Pro-cap/gate' and 'Leu-lock/latch' loops, creating a surface that facilitates interaction with PP2Cs [60]. This receptor-phosphatase interaction is the fundamental event that both ABAleons and ABACUS sensors capture and translate into a FRET signal.
The ABACUS2 sensors incorporate several key improvements based on structural insights. The introduction of latch mutations (E141D and R143S) at the PYL1-ABI1 interaction interface was crucial for enhancing both affinity and FRET response [21]. These residues face out of the ABA binding pocket but influence the accessibility and kinetics of ABA binding and the stability of the receptor-phosphatase complex [21]. Additionally, replacing the flexible attB linkers with shorter, more rigid proline linkers improved the transduction of the ligand-binding event into a measurable FRET change by reducing flexibility and potentially enhancing allosteric coupling between the sensory domain and fluorescent proteins [21].
A critical difference between the systems lies in their orthogonality—the degree to which the biosensor interacts with endogenous signaling components. ABAleons exhibit strong ABA hyposensitivity phenotypes, suggesting they sequester ABA or signaling components, thereby interfering with normal physiological responses [21]. In contrast, the ABACUS2 sensors demonstrate improved orthogonality, likely due to mutations that disrupt interaction with endogenous PP2Cs while maintaining the intramolecular interaction within the biosensor itself [21].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagents for FRET-Based ABA Biosensing
| Reagent/Material | Function/Application | Examples/Specifications |
|---|---|---|
| FRET Biosensor Plasmids | Genetic encoding of biosensor for plant transformation | ABAleon variants, ABACUS2-400n, ABACUS2-100n [21] [60] |
| Fluorescent Protein Pairs | FRET donor and acceptor for rationetric imaging | edCitrine/edCerulean (ABACUS), mTFP1/mVenus (other FRET sensors) [21] [45] |
| Plant Transformation Vectors | Stable integration of biosensor genes into plant genome | Binary vectors for Agrobacterium-mediated transformation [21] |
| Protoplast Isolation System | Transient expression and rapid screening | Enzymatic cell wall digestion, PEG-mediated transfection [22] |
| Rationetric Microscope | Quantitative FRET imaging | Fluorescence microscope with appropriate filter sets, confocal microscope [21] [22] |
| Fluorescence Spectrophotometer | In vitro biosensor characterization | Measurement of emission spectra, determination of KD values [45] |
| ABA Standards | Calibration and control experiments | (±)-ABA, various concentrations for titration curves [21] [45] |
The evolution from ABAleons to ABACUS2 biosensors represents significant progress in the quest for optimal tools to visualize ABA dynamics in plants. While ABAleons pioneered the direct visualization of ABA and demonstrated utility in mapping hormone distributions in response to various stresses [60], their limitations in signal-to-noise ratio and orthogonality constrained their application. The ABACUS2 series, particularly ABACUS2-400n and ABACUS2-100n, addressed these limitations through rational engineering of latch residues and linkers, resulting in biosensors with improved affinity, dynamic range, and reduced interference with endogenous signaling [21].
The choice between these biosensor families depends on specific research requirements. For studies requiring high temporal resolution and minimal perturbation of native physiology, the ABACUS2-400n sensor offers an optimal balance of affinity and large emission ratio change [21]. For investigating low ABA concentrations or subtle changes in hormone levels, the higher-affinity ABACUS2-100n may be preferable despite its slightly lower ratio change [21]. Researchers interested in historical comparisons or working with established ABAleon lines should account for their hyposensitivity phenotypes in experimental interpretations [21].
Future directions in ABA biosensor development will likely focus on further enhancing orthogonality, expanding the dynamic range, and creating multiplexed systems capable of simultaneously monitoring ABA and other signaling components. The integration of these biosensors with emerging imaging technologies and computational analysis will continue to illuminate the complex spatiotemporal dynamics of ABA signaling, ultimately advancing our understanding of plant adaptation to environmental challenges.
The validation of novel biosensing technologies against established benchmarks is a critical step in method development, ensuring data reliability and biological relevance. Förster Resonance Energy Transfer (FRET) biosensors have emerged as powerful tools for monitoring analytes and molecular interactions in live cells with high spatiotemporal resolution [1] [39]. However, their quantitative accuracy must be rigorously assessed against traditional analytical methods. This guide objectively compares FRET biosensors with two cornerstone techniques—mass spectrometry (MS) and transcriptional reporters—focusing on their application in plant analyte research. We summarize comparative performance data, provide detailed experimental protocols for cross-validation, and visualize the conceptual relationships between these methodologies to assist researchers in selecting appropriate validation strategies for their specific applications.
Each technique compared here offers distinct advantages and limitations based on its fundamental operating principles. The table below summarizes their core characteristics.
Table 1: Fundamental Characteristics of the Three Analytical Methods
| Method | Core Principle | Key Strengths | Inherent Limitations |
|---|---|---|---|
| FRET Biosensors | Conformational change alters non-radiative energy transfer between fluorophores [1] | Real-time, subcellular resolution in live cells; minimal disruption to cellular processes [8] | Requires genetic engineering; limited by photobleaching; signal calibration challenges [52] |
| Mass Spectrometry (MS) | Measurement of mass-to-charge ratio of ionized molecules [61] [62] | Highly multiplexed, absolute quantification; untargeted discovery capability [62] | Requires cell lysis; complex sample preparation; cannot monitor real-time dynamics in living cells |
| Transcriptional Reporters | Regulatory element activity drives reporter gene expression (e.g., GFP) [63] | Measures functional biological outcome; high sensitivity through signal amplification | Slow response (hours-days); indirect measurement; no subcellular resolution |
Quantitative Cross-Validation Data
Direct comparative studies provide the most robust evidence for methodological validation. The following table synthesizes quantitative performance data from studies where FRET biosensors were cross-validated against traditional methods.
Table 2: Experimental Cross-Validation Data Between Analytical Methods
| Analyte / Process | FRET Biosensor Performance | Traditional Method Performance | Correlation & Key Findings | Reference Context |
|---|---|---|---|---|
| Phosphate (Pi) | cpFLIPPi-5.3m sensor detected cytosolic Pi dynamics at cellular resolution [8] | Mass spectrometry (method unspecified) | FRET sensor revealed spatial heterogeneity in mycorrhizal roots masked by bulk MS measurements | [8] |
| Kinase Activity | FRET ratio as proxy for activity; subject to imaging condition variability [52] | Enzymatic activity assays | Excellent correlation after FRET ratio calibration using engineered "FRET-ON" and "FRET-OFF" standards [52] | [52] |
| Covalent Inhibitors | Not specifically applied | SPR (Surface Plasmon Resonance): kinact/KI results | SPR exhibited excellent correlation with intact protein MS and time-dependent enzymatic assays [61] | [61] |
| Cis-Regulatory Elements | Not the primary method | lentiMPRA: 41.7% of ~680,000 sequences showed regulatory activity [63] | lentiMPRA provides "in-genome" readout highly correlated with ENCODE annotations and sequence-based models [63] | [63] |
Detailed Experimental Protocols for Cross-Validation
Protocol: FRET-Based Phosphate Measurement in Plant Roots
This protocol, adapted from research on arbuscular mycorrhizal symbiosis, details the procedure for monitoring intracellular phosphate dynamics using FRET biosensors, with controls to ensure specificity [8].
- Biological Materials: Transgenic Brachypodium distachyon lines expressing:
- Experimental Sensor: cpFLIPPi-5.3m (cytosolic or plastid-targeted)
- Control Sensors: cpFLIPPi-Null (Pi-binding deficient mutant), eCFP (donor-only), cpVenus (acceptor-only)
- Promoters: Constitutive (ZmUb1) or mycorrhiza-inducible, cell-type-specific (BdPT7)
- Growth System: Plants inoculated with AM fungi (Diversispora epigaea or Rhizophagus irregularis) in an optimized system for tracing Pi transfer.
- Imaging Procedure:
- Image Acquisition: Capture confocal microscopy images using appropriate laser lines and emission filters for CFP (donor) and YFP (acceptor) channels.
- Sensitized FRET Analysis: Use a semi-automated ImageJ macro to process images. The workflow corrects for:
- Background fluorescence (using wild-type tissue images)
- Spectral bleed-through (SBT)
- Acceptor direct excitation
- FRET Ratio Calculation: Compute the background- and crosstalk-corrected acceptor-to-donor signal ratio (FRET ratio) for individual cells or organelles.
- Validation: The control sensor (cpFLIPPi-Null) confirms that FRET ratio shifts in the experimental sensor are Pi-specific and not caused by intracellular ionic changes or environmental factors.
Protocol: Calibrating FRET Biosensor Signals Using Standards
This protocol addresses a major challenge in FRET quantification—the variability of the FRET ratio due to imaging parameters—by implementing a calibration strategy with internal standards [52].
- Calibration Standards:
- High-FRET Standard: Engineered construct locked in "FRET-ON" conformation.
- Low-FRET Standard: Engineered construct locked in "FRET-OFF" conformation.
- Donor-only and Acceptor-only Controls: For determining crosstalk parameters and actual FRET efficiency.
- Experimental Workflow:
- Cell Preparation: Co-culture or mix cells expressing the biosensor of interest with barcoded cells expressing the calibration standards.
- Multiplexed Imaging: Acquire fluorescence signals from all cell types under identical imaging conditions.
- Signal Normalization: Use the fluorescence signals from the calibration standards to normalize the FRET ratio of the biosensor, compensating for laser intensity and detector sensitivity fluctuations.
- Efficiency Calculation: With calibrated ratios and control cells, compute actual FRET efficiency, restoring expected reciprocal donor and acceptor trends often obscured by imaging drifts.
- Application: This calibrated approach enables robust cross-experimental and long-term studies of FRET biosensors, facilitating direct comparison with endpoint measurements like MS.
Conceptual Framework for Method Selection and Cross-Validation
The following diagram illustrates the decision-making workflow for selecting and cross-validating these methods in plant analyte research.
Method Selection and Cross-Validation Workflow
Essential Research Reagent Solutions
The table below details key reagents and their functions for implementing the described protocols.
Table 3: Essential Research Reagents for Cross-Validation Experiments
| Reagent / Material | Function / Application | Specific Examples / Notes |
|---|---|---|
| Genetically Encoded FRET Biosensors | Target-specific molecular sensing in live cells | cpFLIPPi-5.3m for phosphate; null mutant controls (cpFLIPPi-Null) essential for specificity validation [8] |
| Fluorescent Proteins | Donor/Acceptor FRET pairs; reporter gene readouts | CFP-YFP FRET pairs; EGFP for transcriptional reporters [1] [63] |
| Calibration Standards | Normalization of FRET signals across experiments | Engineered "FRET-ON" and "FRET-OFF" constructs for quantitative cross-experiment comparison [52] |
| Cross-linking Reagents | Stabilizing protein complexes for MS structural analysis | Enrichable, MS-cleavable cross-linkers (e.g., DSBSO, PhoX) for capturing weak/transient interactions in vivo [62] |
| Reporter Assay Libraries | High-throughput testing of regulatory elements | lentiMPRA libraries for analyzing thousands of candidate cis-regulatory elements in parallel [63] |
| Specialized Growth Systems | Maintaining biological relationships during imaging | Optimized plant-fungal co-culture systems for studying nutrient exchange in symbiosis [8] |
Integrated Analysis and Future Directions
Cross-validation studies consistently demonstrate that FRET biosensors, mass spectrometry, and transcriptional reporters provide complementary rather than redundant data. FRET excels at revealing spatiotemporal heterogeneity within cellular populations—a dimension lost in bulk MS analyses or slow transcriptional reporter outputs [8]. Conversely, MS provides unbiased, absolute quantification of analyte concentrations and can validate the specificity of FRET biosensor responses by confirming the absence of interference from structurally similar molecules [62]. Transcriptional reporters, while indirect and slow-responding, measure the functional biochemical outcome of analyte fluctuations or signaling events, providing critical context for interpreting FRET-based activity measurements [63].
The emerging trend is toward integrative approaches that combine these methodologies. For instance, cross-linking mass spectrometry (XL-MS) is increasingly used to study protein conformational dynamics, providing spatial restraints that can validate or inform the design of FRET biosensors [62]. Furthermore, the development of calibration standards for FRET imaging, as previously described, enables more reliable quantitative comparisons with endpoint MS measurements [52]. Future advancements will likely focus on standardizing these cross-validation workflows and developing new computational tools to integrate multi-modal data, thereby providing a more comprehensive understanding of plant analyte dynamics and signaling networks.
Förster Resonance Energy Transfer (FRET)-based biosensors have revolutionized the field of molecular sensing by enabling researchers to monitor biochemical events with exceptional spatiotemporal resolution in living systems. These sensors function as spectroscopic rulers that can resolve nanometer-scale distances between biomolecules, making them indispensable for studying protein-protein interactions, conformational changes, and analyte concentrations within intact cellular environments [3] [64]. The fundamental principle governing FRET involves non-radiative energy transfer from an excited donor fluorophore to a nearby acceptor fluorophore through long-range dipole-dipole interactions, with efficiency dramatically decreasing as the distance between fluorophores increases beyond 1-10 nanometers [1] [2]. This distance dependency provides the mechanistic foundation for FRET-based detection, as molecular binding events or conformational changes that alter the proximity between donor and acceptor fluorophores produce measurable changes in fluorescence output [39].
The application of FRET biosensors has particularly transformed plant science research by enabling real-time monitoring of analytes such as phosphate (Pi) during arbuscular mycorrhizal symbiosis—a mutualistic relationship between plants and fungi that enhances nutrient uptake [8]. Unlike traditional biochemical methods that require tissue destruction and provide static snapshots, FRET biosensors allow non-invasive observation of dynamic molecular processes in living plants with cellular and subcellular resolution [8] [11]. This capability has opened new avenues for investigating plant physiology, stress responses, and symbiotic relationships without disrupting the native cellular environment. As the technology continues to evolve, understanding both the capabilities and constraints of current FRET sensing platforms becomes essential for maximizing their research potential and guiding future innovations in the field.
Fundamental Principles and Design Strategies
The Physical Mechanism of FRET
The theoretical framework for FRET was first established by Theodor Förster in the 1940s, who described it as a distance-dependent energy transfer process between fluorophores via long-range dipole-dipole coupling [1]. This physical phenomenon occurs without photon emission and requires several critical conditions to proceed efficiently. First, the emission spectrum of the donor fluorophore must substantially overlap with the absorption spectrum of the acceptor fluorophore, typically by at least 30% [65]. Second, the donor and acceptor must be in close spatial proximity, generally within 1-10 nanometers, which corresponds well with the scale of most biomolecular interactions [1] [2]. Third, the relative orientation of the donor and acceptor transition dipoles, numerically defined by the orientation factor (κ²), must be favorable for energy transfer [1].
The efficiency of FRET (E) follows an inverse sixth-power relationship with the distance (r) between donor and acceptor fluorophores, as described by the equation E = 1/(1 + r⁶/R₀⁶), where R₀ represents the Förster distance at which energy transfer efficiency is 50% [39] [65]. This steep distance dependence makes FRET exquisitely sensitive to molecular-scale proximity changes, enabling detection of subtle conformational shifts or binding events that would be invisible to conventional microscopy. The Förster distance R₀ is calculated based on the spectral properties of the fluorophore pair and their environment, incorporating factors such as the donor's quantum yield, the spectral overlap integral, and the refractive index of the medium [1].
Figure 1: FRET Energy Transfer Mechanism. This diagram illustrates the fundamental process of Förster Resonance Energy Transfer, showing the non-radiative energy transfer from an excited donor fluorophore to a nearby acceptor fluorophore.
Biosensor Architecture and Configuration
FRET-based biosensors employ two primary architectural configurations: intramolecular and intermolecular designs [1] [65]. Intramolecular biosensors incorporate both donor and acceptor fluorophores within a single molecular construct, typically connected by a ligand-binding domain that undergoes conformational changes upon analyte recognition [1]. This design strategy is particularly effective for monitoring ion concentrations, metabolite levels, and enzyme activities, as the binding event directly alters the distance or orientation between the fluorophores, thereby modulating FRET efficiency [8]. For example, the cpFLIPPi-5.3m sensor used for monitoring phosphate levels in plants employs this intramolecular architecture, with conformational changes in the phosphate-binding domain altering the proximity between cyan (CFP) and yellow (YFP) fluorescent protein variants [8].
Intermolecular FRET biosensors feature donor and acceptor fluorophores fused to separate molecules that interact upon specific biological events, such as protein-protein interactions or complex assembly [1]. This approach enables detection of association and dissociation events between binding partners but introduces additional experimental variables, including variable stoichiometric ratios between separately expressed components and potential non-specific interactions [3]. While intermolecular biosensors can provide valuable insights into dynamic protein interactions within living cells, their implementation requires careful optimization and validation to ensure reliable FRET measurements [3].
More recently, multiplexed and multi-step FRET systems have been developed to enable simultaneous monitoring of multiple analytes within the same biological sample [1]. These advanced configurations provide insights into complex signaling networks and pathway crosstalk but face challenges related to spectral overlap between multiple fluorophores and potential cross-reactivity between different sensing domains [1]. Successful implementation requires meticulous selection of FRET pairs with well-separated emission spectra and minimal spectral crosstalk, often incorporating novel fluorescent proteins or nanomaterials to expand the available color palette [39].
Critical Assessment of Technological Strengths
Unparalleled Spatiotemporal Resolution
FRET biosensors provide researchers with the unique capability to monitor biochemical events at single-cell and subcellular resolution with temporal precision sufficient to track ultrafast molecular processes [1]. This high-resolution imaging is possible because FRET efficiency changes occur on timescales faster than most biological processes, allowing real-time observation of dynamic cellular events [8]. In plant research, this capability has enabled scientists to monitor phosphate fluctuations within individual cortical cells during arbuscular mycorrhizal symbiosis, revealing previously inaccessible information about nutrient exchange at cellular and subcellular levels [8]. The spatial precision of FRET measurements extends to the nanoscale range of 1-10 nanometers, providing molecular-scale insights that would require electron microscopy if attempted with structural techniques [3].
The non-invasive nature of FRET imaging allows prolonged observation of living specimens without disrupting native physiological processes, enabling researchers to track the same cells or tissues over extended periods [8]. This longitudinal monitoring capability is particularly valuable for studying developmental processes, signaling cascades, and cellular responses to environmental stimuli in real time. For plant research specifically, FRET biosensors have been successfully deployed to monitor cellular tension in various tissues using actinin tension sensors expressed in reporter mouse lines, demonstrating the technology's versatility across biological systems [13]. The combination of high spatial and temporal resolution positions FRET biosensors as powerful tools for investigating dynamic molecular events within their native biological contexts.
Versatility and Molecular Specificity
The modular architecture of FRET biosensors enables remarkable design flexibility, allowing researchers to tailor sensing platforms for diverse applications ranging from intracellular ion monitoring to protein-protein interaction detection [1] [39]. This adaptability extends to the choice of fluorophores, with options including fluorescent proteins for genetic encoding and live-cell compatibility, organic dyes for superior brightness and photostability, and quantum dots for exceptional resistance to photobleaching [2] [65]. Each fluorophore class offers distinct advantages that can be matched to specific experimental requirements, with fluorescent proteins particularly valued for their genetic encodability and subcellular targeting capabilities [65].
The molecular specificity of FRET biosensors represents another significant strength, as sensing domains can be engineered to recognize specific analytes with high selectivity amidst complex cellular environments [8]. In plant pathology, for example, FRET-based nanobiosensors have been developed for specific detection of viral pathogens such as Citrus tristeza virus, demonstrating the technology's application in agricultural diagnostics [11]. This targeting specificity extends to subcellular compartments, with signal peptides enabling localization of biosensors to specific organelles or cellular regions, thereby providing compartment-resolved measurements of analyte concentrations or enzymatic activities [8]. The combination of molecular specificity and subcellular targeting allows researchers to investigate biochemical gradients and microdomain-specific signaling events that would be obscured in bulk measurements.
Table 1: Comparison of Major Fluorophore Classes for FRET Biosensing
| Fluorophore Class | Key Advantages | Common Examples | Ideal Applications |
|---|---|---|---|
| Fluorescent Proteins | Genetically encodable; Live-cell compatible; Subcellular targeting | CFP/YFP, GFP/mCherry | Long-term live-cell imaging; Transgenic organisms |
| Organic Dyes | High brightness; Small size; Superior photostability | Cy3/Cy5, Alexa Fluor dyes | In vitro assays; Fixed cell imaging |
| Quantum Dots | Extreme photostability; Broad excitation; Narrow emission | CdTe, CdSe-ZnS core-shell | Long-term tracking; Multicolor experiments |
Analysis of Current Limitations and Challenges
Technical Constraints in Measurement and Implementation
Despite their considerable strengths, FRET biosensors face several significant technical limitations that can constrain their experimental application. A primary challenge is the inherently low signal-to-noise ratio (SNR) characteristic of FRET measurements, which arises from the energy loss associated with the FRET process itself and the contribution of two fluorescent molecules to the measured signal [66]. This limited SNR often necessitates long exposure times (>1-2 seconds) that can reduce temporal resolution and increase the risk of photodamage in live specimens [66]. The problem is particularly pronounced for weakly expressed biosensors or when monitoring small FRET efficiency changes, potentially obscuring biologically relevant signals.
The limited dynamic range of many FRET biosensors, especially those utilizing fluorescent proteins, presents another significant constraint [39]. This restricted range between minimum and maximum FRET efficiency values can compromise detection sensitivity for subtle biochemical changes and is often exacerbated by suboptimal orientation factors between donor and acceptor fluorophores [1]. While some improvement has been achieved through linker optimization strategies such as ER/K linkers, the dynamic range of fluorescent protein-based FRET biosensors generally remains inferior to alternative sensing approaches [39]. Additionally, most FRET biosensors operate effectively only within a narrow distance window of approximately 1-10 nanometers, making them unsuitable for monitoring larger-scale conformational changes or more distant molecular interactions [1].
Figure 2: FRET Experimental Workflow. This diagram outlines the key steps in a FRET-based experiment, highlighting the complex workflow from sensor design to biological interpretation.
Biological and Environmental Vulnerabilities
FRET biosensors exhibit significant vulnerability to environmental factors that can confound experimental interpretations, as the fluorescence properties of both donor and acceptor fluorophores are sensitive to changes in local microenvironmental conditions [66]. Fluctuations in pH, ionic concentrations, oxidation state, temperature, and refractive index can all influence fluorescence output independently of FRET efficiency changes, potentially generating artifactual signals or masking genuine biological responses [66]. This sensitivity is particularly problematic when studying cellular processes that inherently alter microenvironmental conditions, such as metabolic shifts or stress responses.
The performance of FRET biosensors is further compromised by photobleaching of fluorophores during extended imaging sessions, which progressively diminishes signal intensity and can introduce quantification errors [39] [66]. This limitation becomes especially constraining for long-term time-lapse experiments or when monitoring slow biological processes. Additionally, the molecular size of fluorescent protein-based biosensors may sterically interfere with normal biological function, particularly for sensors targeting structurally sensitive proteins or compact molecular complexes [65]. Finally, the expression variability of genetically encoded FRET biosensors in cell populations or tissues introduces heterogeneity that can complicate data interpretation and require more sophisticated analytical approaches [3]. These combined vulnerabilities necessitate careful experimental design and appropriate control conditions to ensure reliable FRET measurements.
Table 2: Primary Limitations of FRET Biosensing Technologies
| Limitation Category | Specific Challenges | Impact on Research |
|---|---|---|
| Technical Constraints | Low signal-to-noise ratio; Limited dynamic range; Narrow distance window | Reduced sensitivity; Missed biological events; Restricted applications |
| Environmental Vulnerabilities | pH sensitivity; Temperature dependence; Oxidation susceptibility | Artifactual signals; Measurement inaccuracy; Need for extensive controls |
| Biological Challenges | Photobleaching; Molecular size effects; Expression variability | Limited experiment duration; Potential functional disruption; Data heterogeneity |
Experimental Approaches and Research Reagent Solutions
Key Methodologies for FRET Quantification
Researchers employ several established methodologies to quantify FRET efficiency, each with distinct advantages and limitations. Sensitized emission FRET represents the most practical approach for dynamic measurements, offering simple implementation and fast imaging times suitable for tracking rapid molecular events [65]. This method typically calculates a ratiometric value between acceptor and donor emission intensities following donor excitation, providing a relative measure of FRET changes without requiring complex instrumentation [8]. However, ratiometric measurements require careful interpretation, as the relationship between ratio changes and actual FRET efficiency is nonlinear and influenced by instrument-specific factors such as filter transmission spectra and camera sensitivity [65].
Acceptor photobleaching FRET provides a more direct measurement of FRET efficiency by comparing donor fluorescence intensity before and after selective photodestruction of the acceptor fluorophore [65]. The resulting increase in donor fluorescence (dequenching) enables calculation of FRET efficiency using the formula E = 1 - (Ipre/Ipost), where Ipre and Ipost represent donor intensity before and after bleaching, respectively [65]. While this method offers straightforward interpretation and does not require specialized equipment beyond standard laser scanning capabilities, it is inherently endpoint in nature and cannot be used for time-lapse imaging of the same sample [65].
Fluorescence Lifetime Imaging FRET (FLIM-FRET) measures the nanosecond-scale decay kinetics of donor fluorescence, which decreases when FRET occurs due to the additional energy transfer pathway [65]. The FRET efficiency is calculated as E = 1 - (τDA/τD), where τDA and τD represent the donor fluorescence lifetime in the presence and absence of the acceptor, respectively [65]. FLIM-FRET offers superior quantification accuracy and is less susceptible to artifacts related to fluorophore concentration, excitation intensity, or photobleaching, but requires specialized instrumentation and expertise [13] [65]. Recent advancements include the development of semi-automated ImageJ macros for sensitized FRET analysis, which streamline data processing and improve reproducibility for researchers without specialized training [8].
Essential Research Reagent Solutions
The successful implementation of FRET biosensing relies on a curated collection of research reagents and methodologies. The following table summarizes key solutions that form the foundation of robust FRET experiments in plant research and beyond.
Table 3: Essential Research Reagent Solutions for FRET Biosensing
| Reagent Category | Specific Examples | Function and Application |
|---|---|---|
| Genetically Encoded Biosensors | cpFLIPPi-5.3m (phosphate); Cameleon (calcium); Actinin tension sensors | Target-specific molecular detection; Live-cell imaging; Transgenic expression |
| Fluorescent Protein Pairs | CFP/YFP; GFP/mCherry; mTFP1/Venus | Donor-acceptor combinations; Spectral compatibility; Genetic encoding |
| Control Constructs | cpFLIPPi-Null (phosphate-insensitive); Donor-only; Acceptor-only | Specificity validation; Signal correction; Background determination |
| Imaging Reagents | Calyculin A; Y27632; Osmotic stressors | Cellular manipulation; Functional validation; Response dynamics |
| Analysis Tools | Semi-automated ImageJ macros; MATLAB GUI; FLIM analysis software | Data processing; FRET efficiency calculation; Statistical validation |
Future Perspectives and Concluding Remarks
The evolving landscape of FRET biosensing technology points toward several promising directions that may address current limitations and expand application possibilities. The ongoing development of improved fluorescent labels with higher quantum efficiencies, enhanced photostability, and reduced environmental sensitivity represents a primary focus for innovation [66]. These advancements, coupled with the emergence of novel nanomaterials such as up-converting nanoparticles and conjugated polymers, promise to significantly boost FRET biosensor performance by increasing brightness, extending imaging duration, and improving signal-to-noise ratios [39]. Additionally, the integration of artificial intelligence and Internet of Things (IoT) technologies with FRET sensing platforms may enable automated analysis, remote monitoring, and sophisticated pattern recognition capabilities beyond current manual approaches [2].
The growing adoption of single-molecule FRET (smFRET) techniques offers particular promise for elucidating molecular heterogeneities and asynchronous processes that are obscured in ensemble measurements [39]. This approach provides unprecedented insights into protein folding, conformational dynamics, and molecular interaction mechanisms at the individual molecule level, potentially revealing biological complexities inaccessible through conventional bulk measurements [39]. Furthermore, the continued refinement of multiplexed FRET imaging systems capable of simultaneously monitoring multiple analytes will enhance our understanding of signaling networks and pathway interconnections in complex biological systems [1].
In conclusion, while FRET-based biosensors face significant technological boundaries related to their dynamic range, environmental vulnerabilities, and measurement constraints, they remain indispensable tools for molecular sensing in plant research and numerous other biological disciplines. Their unique capacity for non-invasive, real-time monitoring of molecular events with high spatiotemporal resolution continues to provide insights unattainable through alternative methodologies. As technological innovations address current limitations and expand capability frontiers, FRET biosensors will likely maintain their central position in the molecular imaging toolkit, increasingly enabling researchers to decipher the intricate biochemical language of living systems.
Conclusion
FRET-based biosensors have unequivocally transformed our ability to visualize and quantify analytes in living plants with unprecedented spatial and temporal resolution. This comparison highlights a clear trajectory from foundational sensors to sophisticated next-generation tools like the high-affinity ABACUS2, which offer enhanced performance for studying complex physiological responses to environmental stresses. Key takeaways include the critical importance of sensor affinity and orthogonality for accurate in-planta measurement, the successful application of these tools in decoding systemic signaling, and the value of advanced microscopy in data acquisition. Future directions must focus on expanding the repertoire of analytes, developing robust multiplexing strategies for simultaneous monitoring, and engineering biosensors with improved stability and reduced phototoxicity for long-term studies. These advancements will not only deepen fundamental plant science research but also accelerate applications in crop improvement and biomedical discovery by providing precise tools for metabolic engineering and diagnostic development.
References
- 1. Exploring the landscape of FRET-based molecular sensors [https://www.sciencedirect.com/science/article/abs/pii/S0734975024001605]
- 2. FRET Based Biosensor: Principle Applications Recent ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10136898/]
- 3. Basics of FRET Microscopy [https://www.microscopyu.com/applications/fret/basics-of-fret-microscopy]
- 4. Förster resonance energy transfer [https://en.wikipedia.org/wiki/F%C3%B6rster_resonance_energy_transfer]
- 5. Fluorescence Resonance Energy Transfer FRET—Note 1.2 [https://www.thermofisher.com/us/en/home/references/molecular-probes-the-handbook/technical-notes-and-product-highlights/fluorescence-resonance-energy-transfer-fret.html]
- 6. A Guide to Fluorescent Protein FRET Pairs - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5038762/]
- 7. Over the rainbow: A practical guide for fluorescent protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6898725/]
- 8. Utilizing FRET-based Biosensors to Measure Cellular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11769715/]
- 9. ATP in plants using FRET sensor [https://www.bmglabtech.com/en/application-notes/fret-sensor-assesses-atp-levels-in-living-plants-experiencing-low-oxygen-conditions/]
- 10. Rational design of FRET sensor proteins based on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3822149/]
- 11. Nanomaterial-based biosensors: a new frontier in plant ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1570318/full]
- 12. An Improved Ratiometric FRET Biosensor with Higher ... [https://www.mdpi.com/1424-8220/25/18/5903]
- 13. In situ FRET measurement of cellular tension using ... [https://www.nature.com/articles/s41598-023-50142-z]
- 14. Recent Advances in Fluorescence Resonance Energy ... [https://www.mdpi.com/1467-3045/47/4/235]
- 15. Fluorescent biosensors illuminating plant hormone research [https://pmc.ncbi.nlm.nih.gov/articles/PMC8491072/]
- 16. Plant biologists FRET over stress - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3985516/]
- 17. In vivo visualization of nitrate dynamics using a genetically ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9581486/]
- 18. An ultrafast dual-channel ratiometric fluorescence probe ... [https://www.sciencedirect.com/science/article/abs/pii/S1385894725037581]
- 19. Next-generation Genetically Encoded Fluorescent Biosensors ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11786609/]
- 20. Genetically Encoded FRET Sensors for Visualizing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1150369/]
- 21. Next-generation ABACUS biosensors reveal cellular ABA ... [https://www.nature.com/articles/s41477-023-01447-4]
- 22. Optimization of FRET imaging in Arabidopsis protoplasts [https://www.sciencedirect.com/science/article/pii/S1016847825000044]
- 23. Expanding the Toolkit of Fluorescent Biosensors for ... [https://www.mdpi.com/1422-0067/21/15/5350]
- 24. Abscisic acid dynamics in roots detected with genetically ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3985517/]
- 25. Sensors for the quantification, localization and analysis of the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7898640/]
- 26. FRET-based reporters for the direct visualization of ... [https://elifesciences.org/articles/01739]
- 27. Application of FRET-Based Biosensor "ATeam" for ... [https://pubmed.ncbi.nlm.nih.gov/28276022/]
- 28. A genetically encoded single-wavelength sensor for ... [https://www.nature.com/articles/s41467-019-08441-5]
- 29. Utilizing FRET-based Biosensors to Measure Cellular ... [https://bio-protocol.org/en/bpdetail?id=5158&type=0]
- 30. Utilizing FRET-based Biosensors to Measure Cellular ... [https://pubmed.ncbi.nlm.nih.gov/39872724/]
- 31. Fluorescence-Based Aqueous Phosphate Sensing Using ... [https://www.mdpi.com/2304-6732/11/3/250]
- 32. An Improved ATP FRET Sensor For Yeast Shows ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7106129/]
- 33. Fluorescent Proteins as Genetically Encoded FRET ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4634415/]
- 34. An Improved Ratiometric FRET Biosensor with Higher Affinity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12473246/]
- 35. ATP sensing in living plant cells reveals tissue gradients ... [https://elifesciences.org/articles/26770]
- 36. Evaluating the Impact of Physiological Oxygen Tensions on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10249672/]
- 37. A Genetically Encoded FRET Sensor for Hypoxia and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5026574/]
- 38. Progress and Prospects in FRET for the Investigation of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12467375/]
- 39. Expanding Horizons in Advancements of FRET Biosensing ... [https://www.mdpi.com/2079-6374/15/7/452]
- 40. What is FRET with FLIM (FLIM-FRET)? | Learn & Share [https://www.leica-microsystems.com/science-lab/life-science/what-is-fret-with-flim/]
- 41. Quantitative real-time imaging of intracellular FRET ... [https://www.nature.com/articles/s41598-020-61478-1]
- 42. Quantitative single-molecule FLIM and PIE-FRET imaging of ... [https://bmcmethods.biomedcentral.com/articles/10.1186/s44330-025-00048-1]
- 43. FRET-analysis in living cells by fluorescence lifetime im... [https://www.degruyterbrill.com/document/doi/10.1515/mim-2024-0027/html?lang=en&srsltid=AfmBOopwyov0YUZHi4Lm3VcxMgCvbL_CteWNBzl7pJ9xUW0FWzXBTOLw]
- 44. Wide-field fluorescence lifetime imaging of single ... [https://www.nature.com/articles/s41377-025-01901-2]
- 45. Genetically Encoded FRET-Based Nanosensor for Insect ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12612880/]
- 46. Optimization of FRET imaging in Arabidopsis protoplasts [https://pmc.ncbi.nlm.nih.gov/articles/PMC11879690/]
- 47. Two novel red-FRET ERK bosensors in the 670-720 nm range [https://pmc.ncbi.nlm.nih.gov/articles/PMC12616953/]
- 48. Robust calibration and quantification of FRET signals using ... [https://pubmed.ncbi.nlm.nih.gov/41210971/]
- 49. Progress and Prospects in FRET for the Investigation of ... [https://www.mdpi.com/2079-6374/15/9/624]
- 50. Engineering Genetically Encoded FRET Sensors - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4168480/]
- 51. Review of FRET biosensing and its application in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10006758/]
- 52. Robust calibration and quantification of FRET signals using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12595091/]
- 53. Robust calibration and quantification of FRET signals using ... [https://www.sciencedirect.com/science/article/pii/S2589004225020048]
- 54. A practical method for monitoring FRET-based biosensors in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4725439/]
- 55. Signal/Noise Analysis of FRET-Based Sensors [https://www.sciencedirect.com/science/article/pii/S0006349510009252]
- 56. Protocol for measuring labile cytosolic Zn2+ using an in ... [https://www.sciencedirect.com/science/article/pii/S2666166724002958]
- 57. High-affinity tuning of single fluorescent protein-type ... [https://www.nature.com/articles/s42003-024-06394-0]
- 58. Abundance-biased codon diversification prevents ... [https://www.nature.com/articles/s42003-025-08677-6]
- 59. Molecular Insights into ABA-Mediated Regulation of Stress ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12387064/]
- 60. FRET-based reporters for the direct visualization of abscisic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3985518/]
- 61. Application and Cross-Validation of a High-Throughput ... [https://pubmed.ncbi.nlm.nih.gov/40687013/]
- 62. In vivo cross-linking mass spectrometry: Advances and ... [https://www.sciencedirect.com/science/article/abs/pii/S1367593125000626]
- 63. Massively parallel characterization of transcriptional ... [https://www.nature.com/articles/s41586-024-08430-9]
- 64. Key Steps to Follow in a FRET Experiment [https://www.spectroscopyonline.com/view/key-steps-to-follow-in-a-fret-experiment]
- 65. A Guide to Fluorescent Protein FRET Pairs [https://www.mdpi.com/1424-8220/16/9/1488]
- 66. Overcoming Limitations of FRET Measurements - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5835960/]