Designing Higher Plant Compartments for Space Habitats: From Bioregenerative Life Support to Clinical Applications
This article provides a comprehensive analysis of higher plant compartment design for space habitats, targeting researchers and scientists in bioastronautics and related fields.
Designing Higher Plant Compartments for Space Habitats: From Bioregenerative Life Support to Clinical Applications
Abstract
This article provides a comprehensive analysis of higher plant compartment design for space habitats, targeting researchers and scientists in bioastronautics and related fields. It explores the foundational role of Bioregenerative Life Support Systems (BLSS) in long-duration missions for resource regeneration, fresh food production, and psychological support. The content details current methodologies from flight-proven systems like NASA's Veggie and Advanced Plant Habitat, alongside emerging aeroponic and hydroponic technologies. It addresses critical troubleshooting for plant growth in microgravity and radiation environments, and validates designs through comparative analysis of international programs and terrestrial analogues. The synthesis offers implications for sustainable life support and controlled environment agriculture research on Earth.
The Critical Role of Plant Compartments in Bioregenerative Life Support Systems (BLSS)
Bioregenerative Life Support Systems (BLSS) are advanced artificial ecosystems designed to sustain human life in space by biologically regenerating essential resources. Unlike purely physicochemical systems, a BLSS uses biological components—primarily higher plants and microorganisms—to simultaneously revitalize atmosphere, purify water, and generate food within a closed-loop system [1] [2]. For long-duration missions beyond low-Earth orbit, such as to the Moon or Mars, where resupply from Earth is logistically challenging and cost-prohibitive, BLSS technology transitions from a "nice-to-have" to a "must-have" capability [2]. These systems are engineered to mimic Earth's ecological networks, creating a cycle where the waste products from one compartment become the resources for another [2]. The development of BLSS represents a critical step toward achieving the autonomy required for enduring human presence in deep space.
The core function of a BLSS extends far beyond mere food production. While providing fresh nutrition is a vital output, the system's integrated biological processes are fundamental for the continuous regeneration of breathable air through photosynthesis and the recycling of water through plant transpiration and microbial activity [1] [2]. This holistic approach significantly reduces the need for external supplies; for instance, current physicochemical systems on the International Space Station (ISS) can reduce water transport payload by up to 96.5%, but they cannot produce food [3]. A fully operational BLSS aims to close the loop further, recovering nutrients from waste streams to create a highly efficient and robust system for long-distance space travel [3].
Core Functions and Quantitative Performance of a BLSS
The performance of a BLSS is measured by its closure of key material loops—oxygen, water, and food—that are essential for human survival. Ground-based demonstrators have successfully validated these functions. The following table summarizes the resource regeneration capabilities demonstrated in a large-scale, 180-day integrated experiment within a Controlled Ecological Life-Support System (CELSS), a key type of BLSS [4].
Table 1: Resource Regeneration Performance in a 180-Day CELSS Experiment [4]
| Resource Category | Regeneration Rate (%) | Key Processes and Technologies Involved |
|---|---|---|
| Food | 55% (Average); 70.8% (High-Efficiency Period) | Cultivation of staple crops (e.g., wheat, potato) and vegetables; Hydroponics. |
| Atmosphere (Oxygen) | 100% | Plant photosynthesis; Physico-chemical backup systems for carbon dioxide concentration control. |
| Water | 100% | Collection and purification of condensate, transpiration water, and urine; Water Recovery System (WRS). |
| Solid Waste | 87.7% | Drying of inedible plant biomass; Fermentation; Composting for soil-like substrate production. |
The 100% closure of the atmospheric and water loops demonstrates the viability of biological and physico-chemical integration. The food regeneration rate, while not fully closed, highlights the significant progress toward sustaining a crew primarily with in-situ food production. Beyond these quantitative metrics, BLSS provides psychological benefits to crew members. The presence of plants and the activity of gardening have been shown to offer psychological support against the isolation and stress of long-duration missions, functioning as a form of horticultural therapy [2].
The Higher Plant Compartment: Design and Protocol
The higher plant compartment is the cornerstone of the BLSS, acting as the primary "producer" unit. It is responsible for food production, carbon dioxide fixation, oxygen generation, and water purification via transpiration [2]. The design of this compartment, including species selection and cultivation protocols, is directly dictated by the mission scenario.
Mission-Based Crop Selection and Cultivation Protocol
Table 2: Plant Cultivation Strategies for Different Mission Durations [2]
| Mission Scenario | Recommended Plant Types | Primary Objectives | Cultivation Protocol Highlights |
|---|---|---|---|
| Short-Duration (e.g., LEO) | Fast-growing leafy greens (e.g., lettuce, kale), microgreens, dwarf cultivars. | Dietary complementation, nutrient fortification, psychological benefits. | Low-energy, small area, short growth cycles. Focus on minimal inputs and basic technological integration. Systems function as a "salad machine." |
| Long-Duration/Planetary Outpost (e.g., Moon, Mars) | Staple crops (wheat, potato, rice, soy), fruits, and longer-cycle vegetables (tomato, peppers). | Provision of macronutrients (carbs, proteins, fats) and substantial contribution to resource recycling. | Large growing area per astronaut. Deep integration with waste management and nutrient recycling systems. Requires advanced, automated cultivation chambers. |
Detailed Experimental Protocol: Plant Cultivation for BLSS
This protocol outlines the methodology for establishing and maintaining a higher plant growth unit within a BLSS, synthesizing practices from ground-based analog tests and flight experiments [2] [5] [4].
1. Objective: To reliably produce edible biomass, contribute to atmospheric revitalization (O₂ production, CO₂ fixation), and enable water recycling through the cultivation of selected plant species in a controlled environment.
2. Materials and Reagents:
- Seeds: Surface-sterilized seeds of selected species (e.g., 'Outredgeous' red romaine lettuce, wheat, potato).
- Growth Substrate: Inert substrate (e.g., arcillite) for root support and moisture management [5].
- Nutrient Solution: Hydroponic solution containing essential macro and micronutrients (e.g., Hoagland's solution), potentially supplemented with minerals recovered from waste streams [3] [4].
- Growth Chamber: Controlled environment chamber (e.g., Advanced Plant Habitat) with programmable systems for light, temperature, and humidity [5].
- Sensors: Porous tubes and moisture sensors to monitor root-zone water content [5].
- Water Recovery System: System for collecting condensate and transpirated water for purification and reuse [4].
3. Procedure:
- Step 1: Germination.
- Place seeds on the pre-moistened growth substrate within the science carrier.
- Increase relative humidity (RH) and reduce light intensity in the chamber to promote high germination rates. Germination domes may not be necessary with precise environmental control [5].
- Step 2: Growth Chamber Environmental Control.
- Maintain a tightly controlled environment throughout the growth cycle.
- Light: Provide a photosynthetic photon flux density (PPFD) of approximately 550 μmol m⁻² s⁻¹ using LED arrays, with a controlled photoperiod (e.g., 16h light/8h dark) [4].
- Temperature and Humidity: Set according to species-specific requirements.
- Atmosphere: Control CO₂ concentration to enhance photosynthetic efficiency [4].
- Step 3: Irrigation Management.
- Step 4: Atmosphere and Water Recycling Integration.
- The plant cabin atmosphere is connected to the crew cabin. Plants photosynthetically consume CO₂ from the crew and produce O₂ [4].
- Condensate from the cabin atmosphere and water vapor from plant transpiration are collected and transferred to the Water Recovery System for purification and return to the irrigation system [4].
- Step 5: Harvest and Post-Harvest Analysis.
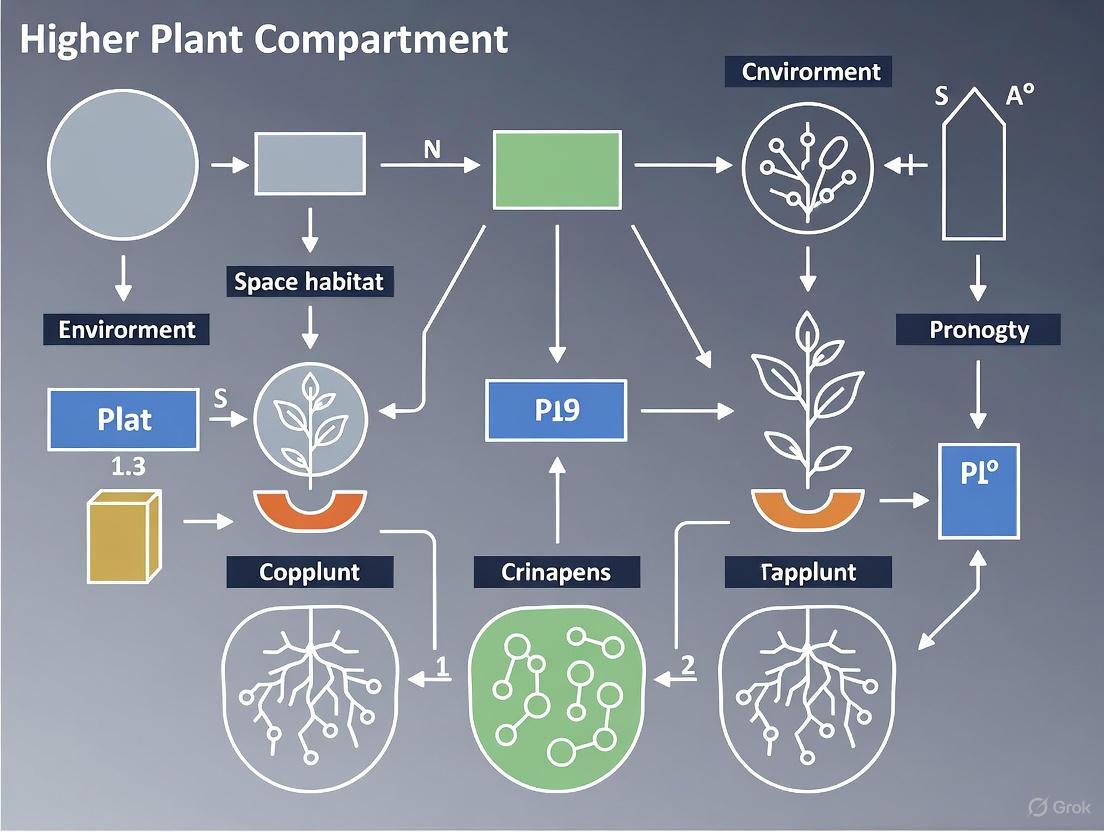
System Workflow and Compartment Integration
A BLSS is a complex network of interconnected compartments that operate synergistically. The following diagram illustrates the flow of mass and energy between the key compartments, highlighting the role of the higher plant unit as the central producer.
Diagram 1: Mass and Energy Flow in a BLSS.
The Scientist's Toolkit: Key Research Reagents and Materials
Research and development of BLSS components require specialized materials and reagents. The following table details essential items for conducting plant-based BLSS experiments, particularly those focused on resource recycling.
Table 3: Essential Research Reagents and Materials for BLSS Plant Experiments
| Reagent / Material | Function and Application in BLSS Research |
|---|---|
| Inert Growth Substrate (e.g., Arcillite) | Provides physical support for plant roots in hydroponic systems. Its inert nature allows for precise control of nutrient delivery via the aqueous solution [5]. |
| Hydroponic Nutrient Solution | Aqueous solution containing all essential mineral nutrients (N, P, K, Ca, S, Mg, and micronutrients) for plant growth. Can be formulated with recovered nutrients from waste streams [3] [4]. |
| Surface Sterilizing Agents (e.g., Ethanol, Dilute Bleach) | Used for seed surface sterilization to ensure a sterile start to the experiment, preventing contamination of the closed system by external microbes [2]. |
| Nitrifying Bacterial Consortia | Specific microbial cultures (e.g., Nitrosomonas, Nitrobacter) used to convert ammonia from waste streams into nitrate, a preferred nitrogen fertilizer for plants [3]. |
| Stabilization Acid (e.g., H₃PO₄) | Used in urine pretreatment to acidify and chemically stabilize the waste, preventing urea hydrolysis and pipeline scaling, thereby facilitating later nutrient recovery [3]. |
| MOPS Buffer | A biological buffer used in microbial compartments (e.g., MELiSSA) to maintain a stable pH for the optimal activity of nitrifying bacteria [3]. |
| LED Lighting Arrays | Provides tunable, energy-efficient light for plant photosynthesis at specific wavelengths and intensities (PPFD), enabling optimal growth in the absence of sunlight [4]. |
| Root Zone Moisture Sensors | Critical for monitoring and controlling water content in the growth substrate in microgravity, where fluid behavior is challenging to manage [5]. |
Bioregenerative Life Support Systems represent a paradigm shift in life support for deep space exploration, moving from direct supply and physicochemical recycling to a biologically sustained, closed-loop ecology. The higher plant compartment is the linchpin of this system, fulfilling a triple function of food producer, atmosphere revitalizer, and water purifier. While significant challenges remain, particularly in scaling and integrating these systems for space and understanding the full impact of the space environment, ground-based demonstrators have proven the fundamental feasibility. The ongoing research and protocols detailed in this document provide a roadmap for advancing this critical technology, which will ultimately enable humanity to become a multi-planetary species.
Application Notes: The Multifunctional Role of Higher Plants in BLSS
Higher plants are fundamental components of Bioregenerative Life Support Systems (BLSS), fulfilling critical roles that extend beyond food production to encompass atmospheric revitalization, water purification, psychological support for crew members, and organic waste recycling [2]. The integration of these functions is paramount for achieving self-sufficiency and sustainability in long-duration space missions, such as those to the Moon and Mars, where resupply from Earth is not feasible [6]. The design of the plant compartment must be tailored to the mission scenario, with short-duration missions prioritizing fast-growing, nutrient-dense crops, and long-duration outposts requiring the inclusion of staple crops to provide a balanced diet [2].
The following table summarizes the key functions, target outcomes, and applicable mission scenarios for a higher plant compartment.
Table 1: Multifunctional Benefits of the Higher Plant Compartment in Space Habitats
| Function | Key Target Outcomes | Relevant Mission Scenarios | Quantitative Metrics / Evidence |
|---|---|---|---|
| Nutritional Supplementation | - Provision of essential vitamins, minerals, and antioxidants [2]- Counteraction of nutrient degradation in stored food [2] | - All mission durations (LEO, Lunar, Martian)- Critical for long-duration missions | - Vitamin C and B1 in stored food degrade to inadequate levels within 3 years [2]- Crop Examples: Leafy greens (e.g., lettuce, kale), microgreens, dwarf tomato, staple crops (wheat, potato, soy) [2] |
| Psychological Support | - Mitigation of isolation and confinement stress [2]- "Horticultural therapy" through gardening activities [2] | - Long-duration missions (e.g., Martian transit, planetary outposts) | - Access to fresh food and gardening provides non-nutritional benefits and acts as an "emotional supporter" [2] |
| Atmospheric Revitalization | - Oxygen production via photosynthesis [2]- Carbon dioxide (CO2) consumption [2] | - Long-duration missions and permanent planetary outposts | - Becomes a "must-have" requirement when resupply from Earth is not feasible [2] |
| Water Recycling | - Purification of water through plant transpiration [2] | - Long-duration missions and permanent planetary outposts | - Contributes to closed-loop water recovery systems [2] |
| Waste Management | - Recycling of organic solid and liquid wastes as nutrients [6] | - Long-duration missions and permanent planetary outposts | - Integration with BLSS waste degradation compartments (e.g., microbes) [6] |
Experimental Protocols
Protocol: Quantifying Nutritional Supplementation Efficacy
Objective: To determine the impact of fresh plant consumption on astronaut nutritional status and health during a simulated long-duration mission.
Materials:
- Controlled environment growth chamber (e.g., Veggie or Advanced Plant Habitat (APH) system analogues) [6]
- Seeds of target crops (e.g., 'Outredgeous' red romaine lettuce, 'Tokyo Bekana' Chinese cabbage, dwarf tomato 'Red Robin')
- Standardized nutrient delivery system (e.g., plant pillow wicks for hydroponics) [6]
- Blood collection and analysis equipment
- High-performance liquid chromatography (HPLC) for phytonutrient analysis
Methodology:
- Pre-Mission Baseline: Collect blood and urine samples from crew members to establish baseline levels of key nutrients (e.g., vitamins C, K, A, and various antioxidants).
- Experimental Design: Implement a randomized crossover study where the crew's diet alternates between periods with and without fresh plant supplementation.
- Crop Cultivation: Grow selected crops in the plant growth system under standardized light (e.g., LED spectrum), temperature, and humidity conditions. Maintain detailed logs of growth parameters and yields.
- Biomass Analysis: At harvest, a sub-sample of edible biomass is analyzed for nutritional content (vitamins, minerals, antioxidants) using HPLC and other standard methods.
- Crew Monitoring: During both supplementation and non-supplementation periods, collect regular biological samples from the crew to monitor changes in nutrient levels.
- Health Metrics: Correlate nutritional status with other health metrics, such as immune function markers and cognitive performance scores.
Data Analysis: Use paired t-tests or ANOVA to compare the mean differences in nutrient levels and health metrics between the supplementation and control periods.
Protocol: Assessing Psychological Benefits of Plant Interaction
Objective: To evaluate the effects of gardening activities and presence of living plants on crew morale and psychological well-being.
Materials:
- Validated psychological questionnaires (e.g., Profile of Mood States (POMS), Perceived Stress Scale (PSS))
- Salivary cortisol sampling kits as a physiological stress marker
- Activity logs and wearable devices to monitor time spent on gardening
Methodology:
- Pre-Study Assessment: Administer psychological questionnaires and collect baseline salivary cortisol samples.
- Intervention Phase: Crew members are scheduled for regular, mandatory interaction with the plant compartment. Activities include planting, monitoring, harvesting, and other maintenance tasks.
- Data Collection:
- Self-Report: Crew complete psychological questionnaires weekly.
- Physiological: Salivary cortisol samples are collected at waking, 30 minutes post-wake, and before bedtime on a designated day each week.
- Behavioral: Time spent voluntarily in the plant compartment outside of mandatory tasks is recorded.
- Control/Comparison: Data from the plant-interaction mission phase can be compared to baseline data or to data from a comparable mission phase without plant interaction.
Data Analysis: Use repeated-measures ANOVA to analyze trends in psychological and cortisol data over time. Correlation analysis can be used to explore the relationship between time spent gardening and well-being scores.
Protocol: Integrating Plant Compartments with Waste Recycling Streams
Objective: To demonstrate the efficient use of recycled nutrients from crew waste for plant growth within a BLSS.
Materials:
- Biological waste processing system (e.g., compost bioreactor, microbial fermentation chamber) [6]
- Nutrient solution mixing and delivery system
- Plant growth chambers with recirculating hydroponics
- Water quality testing equipment (for NH4+, NO3-, NO2-, pH, EC)
Methodology:
- Waste Processing: Solid and liquid crew wastes are introduced into a biological processing system (e.g., inoculated with nitrifying bacteria Nitrosomonas and Nitrobacter) to convert organic nitrogen and other nutrients into plant-available forms [6].
- Nutrient Solution Formulation: The effluent from the waste processor is filtered, sterilized, and supplemented with any required minerals to create a balanced hydroponic nutrient solution.
- Plant Growth Trial: Crops are grown in two parallel systems:
- Experimental Group: Fed with the recycled nutrient solution.
- Control Group: Fed with a standard, chemically defined Hoagland's solution.
- System Monitoring: Monitor and record plant growth parameters (germination rate, biomass accumulation, chlorophyll content) and nutrient solution chemistry daily.
- Biomass and Food Safety Analysis: At harvest, compare the yield and nutritional quality of crops from both groups. Test for potential pathogens to ensure food safety [6].
Data Analysis: Compare the final biomass, growth rates, and nutritional content between the experimental and control groups using independent sample t-tests.
Visualizations
BLSS Functional Integration
Nutritional Study Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Reagents for BLSS Plant Compartment Research
| Item | Function / Application in Research |
|---|---|
| Veggie or APH Growth System | Automated growth chamber for plant cultivation in microgravity or simulated space environments; enables study of plant growth, development, and food safety in space [6]. |
| Plant Pillows | Sealed units containing growth media (e.g., arcillite) and fertilizer, used with the Veggie system to deliver water and nutrients to plant roots in a low-maintenance, contained manner [6]. |
| LED Lighting Arrays | Provides specific light wavelengths (red, blue, green, far-red) optimized for plant photosynthesis, morphology, and nutritional quality in controlled environments [2]. |
| Hydroponic Nutrient Solutions | Standardized mixtures of essential mineral nutrients (e.g., Hoagland's solution) for plant growth; serves as a control against which recycled nutrient solutions from waste processors are tested [6]. |
| Nitrogen-Fixing Bacteria (e.g., Sinorhizobium meliloti) | Inoculant used to enhance soil fertility in regolith studies; fixes atmospheric nitrogen into a plant-usable form, crucial for growing plants in lunar or Martian soil simulants [6]. |
| Nitrifying Bacterial Consortia | A mixture of bacteria (e.g., Nitrosomonas, Nitrobacter) used in waste processing compartments to convert toxic ammonia from liquid waste into nitrate, a preferred nitrogen source for plants [6]. |
| Pathogen Detection Kits | Molecular (PCR) or culture-based tools to monitor for plant pathogens like Fusarium oxysporum in hydroponic systems, ensuring crop health and crew food safety [6]. |
| HPLC System with PDA/MS Detector | Used for the precise identification and quantification of nutritional and anti-nutritional compounds (vitamins, antioxidants, secondary metabolites) in plant and biological samples. |
The success of long-duration human space exploration and the establishment of sustainable habitats beyond Earth depend critically on the reliable cultivation of higher plants. Within the context of designing higher plant compartments for space habitats, plants are envisioned as multifunctional components of Bioregenerative Life Support Systems (BLSS), providing oxygen, fresh food, water purification, and psychological benefits for crew members [2] [7]. However, the space environment presents a unique set of challenges, primarily microgravity and ionizing radiation, which induce complex molecular and physiological responses in plants [7] [8]. Understanding these responses is essential for designing robust life support systems for missions to the Moon and Mars.
This Application Note provides a structured overview of the key abiotic stressors of the space environment on plant biology. It summarizes quantitative data on plant responses, details standard experimental protocols for space-based plant research, and visualizes critical signaling pathways. The information is intended to assist researchers and scientists in designing experiments and developing countermeasures for advanced habitat design.
Quantitative Data on Space Environment Stressors
Gravity and Radiation Parameters Across Environments
The following table compares the primary environmental parameters—gravity and radiation—that plants would encounter in different mission scenarios, from ground research to deep space.
Table 1: Environmental Parameters Across Different Mission and Simulation Scenarios
| Environment / Platform | Gravity Level | Annual Radiation Dose (approx.) | Key Characteristics |
|---|---|---|---|
| Earth Surface | 1 g | ~0.39 mSv [8] | Baseline condition; protected by atmosphere and magnetosphere. |
| International Space Station (ISS) | Microgravity (µg) | 100 - 200 mSv [8] | Primary platform for real spaceflight biology experiments. |
| Simulated Microgravity (RPM, Clinostat) | Simulated µg | N/A (Ground-based) | Machine that randomizes gravity vector to simulate µG conditions on Earth [7]. |
| Lunar Surface | 0.17 g [7] | Higher than ISS [2] | Partial gravity; exposure to galactic cosmic rays and solar particle events. |
| Martian Surface | 0.38 g [7] | Higher than ISS [2] | Partial gravity; thin atmospheric radiation shielding. |
| Deep Space | Microgravity | Hundreds to thousands of mSv [8] | Highest radiation exposure, includes GCR and SPE spectra. |
Documented Plant Phenotypic Responses to Spaceflight
Numerous experiments have documented the phenotypic effects of spaceflight on various plant species. The observed responses are the net result of the combined influence of microgravity and radiation.
Table 2: Documented Plant Responses to Spaceflight Conditions
| Plant Species / System | Observed Phenotypic Response | Experimental Context |
|---|---|---|
| Arabidopsis seedlings | Disruption of meristematic competence; loss of coordinated cell proliferation and growth [7]. | ISS experiments |
| Antarctic Moss (Ceratodon purpureus) | Research aims to detect how combined radiation and microgravity differ from single stressors on growth and physiology [9]. | ARTEMOSS payload (ISS) |
| Lentil roots | Gravity perception threshold estimated to be in the order of 10⁻³ g or lower [7]. | Spaceflight experiments |
| Pea hypocotyls | Growth inhibition correlated with attenuated polar auxin transport [7]. | Spaceflight experiments |
| General crops (e.g., Lettuce, Lentil) | Morpho-physiological alterations reported; yet, plants can complete seed-to-seed life cycle in space [7]. | Early space experiments & ISS |
| Space-grown plants (general) | Accumulation of Reactive Oxygen Species (ROS) is a concern, though plants often achieve Earth-like growth yields [8]. | Multiple ISS cultivation studies |
Key Signaling Pathways and Molecular Responses
Plant adaptation to the space environment involves complex molecular signaling and response pathways. Two of the most critical are the gravitropism signaling pathway and the oxidative stress response pathway.
Gravitropism and Gravity Perception Signaling
In roots, the amyloplasts in the columella cells sediment in the direction of gravity, initiating a signal transduction cascade. The LAZY proteins are key players in relocating the auxin efflux carrier PIN-FORMED (PIN) proteins on the plasma membrane [7]. This leads to a redistribution of the plant hormone auxin, creating an auxin gradient across the root tip. The higher auxin concentration on the lower side of the root inhibits cell elongation, causing the root to curve downward [7]. Under microgravity, this sedimentation and the subsequent polar auxin transport are disrupted, leading to altered root growth patterns.
Oxidative Stress and ROS Defense Pathways
The space environment, particularly ionizing radiation, leads to the generation of Reactive Oxygen Species (ROS) such as superoxide radicals (•O₂⁻), hydrogen peroxide (H₂O₂), and hydroxyl radicals (•OH) [8]. Plants perceive this oxidative stress and activate a sophisticated, multi-layered antioxidant defense system. This includes enzymatic antioxidants like Superoxide Dismutase (SOD), Catalase (CAT), and Ascorbate Peroxidase (APX), which work in concert to detoxify ROS [8]. Non-enzymatic antioxidants like ascorbate and glutathione also play a crucial role in scavenging ROS and protecting cellular components.
Experimental Protocols for Space Plant Biology
Protocol: Ground-Based Simulation of Space Stressors
This protocol outlines a method for pre-screening plant responses to space radiation on Earth before flight experiments.
- Objective: To investigate the molecular and physiological effects of simulated deep space radiation on plant models as a prelude to spaceflight experiments [9].
- Materials:
- Plant samples (e.g., spores of Antarctic moss Ceratodon purpureus)
- NASA Space Radiation Laboratory (NSRL) facility or equivalent
- Sterile Petri plates with growth medium
- Controlled environment chamber
- Procedure:
- Sample Preparation: Aseptically place plant samples on standardized growth medium within Petri plates [9].
- Radiation Exposure: Irradiate sample groups at the NSRL with:
- Group 1: Simulated Galactic Cosmic Rays (GCR)
- Group 2: Simulated Solar Particle Events (SPE)
- Group 3: Non-irradiated control group [9]
- Post-Irradiation Handling: Transport irradiated samples under controlled conditions (e.g., 4°C) to the launch facility [9].
- Data Collection: Image samples and collect tissue for -80°C fixation for subsequent transcriptomic, proteomic, and metabolomic analyses.
Protocol: Spaceflight Experiment Integration and Execution
This protocol describes the workflow for conducting a plant biology experiment on the International Space Station (ISS).
- Objective: To study the combined effects of sequential spaceflight microgravity and pre-administered space radiation on plant growth and molecular responses [9].
- Materials:
- Pre-irradiated and control plant samples integrated into NASA-approved "science bags" or hardware (e.g., Veggie; note: Advanced Plant Habitat (APH) is currently unavailable for new studies [10])
- ISS Cold Stowage (4°C)
- ISS Cabin-temperature environment
- ISS Glacier freezer (-80°C or below)
- Procedure:
- Upload and Stowage: Samples are launched and initially stored in ISS cold stowage to suspend metabolism [9].
- Experiment Initiation: Crew members transfer the science bags from cold stowage to the ISS cabin environment to initiate the growth phase.
- Growth Period: Allow plants to grow for a defined period (e.g., 7 days) under ISS cabin conditions (microgravity, ambient radiation) [9].
- Preservation: After the growth period, crew members transfer the science bags to the ISS Glacier freezer to preserve the plant tissue at -80°C until return to Earth [9].
- Post-Flight Analysis: Conduct multi-omics (genomics, transcriptomics, proteomics) and physiological comparisons between flight samples, ground controls, and radiation-only controls.
The Scientist's Toolkit: Research Reagent Solutions
The following table lists key materials and reagents essential for conducting space plant biology research, from ground simulation to flight experiments.
Table 3: Essential Research Reagents and Materials for Space Plant Biology
| Item / Reagent | Function / Application | Specific Examples / Notes |
|---|---|---|
| Antarctic Moss (Ceratodon purpureus) | Extremophile plant model for studying radiation and microgravity tolerance [9]. | ANT1 isolate used in ARTEMOSS experiment [9]. |
| NASA Space Radiation Lab (NSRL) | Facility for simulating deep space radiation (GCR, SPE) on Earth prior to flight [9]. | Critical for pre-screening radiation effects [9]. |
| Science Bags / Petri Plates | Containment and growth support for plant samples in spaceflight hardware [9]. | Used in the ARTEMOSS payload on ISS [9]. |
| Veggie Plant Growth System | ISS facility for growing plants; provides light and nutrient delivery [11]. | An alternative plant growth system on the ISS. |
| RNA/DNA Stabilization Kits | Preservation of nucleic acids for post-flight transcriptomic and epigenetic analysis. | Essential for measuring gene expression changes in response to spaceflight [7]. |
| Antibodies for Protein Analysis | Detection and quantification of key proteins (e.g., PIN, antioxidant enzymes) via immunohistochemistry or western blot. | Used to study auxin transport and oxidative stress [7] [8]. |
| Fixatives (e.g., Glutaraldehyde) | Preservation of cellular and sub-cellular structures for microscopic analysis post-flight. | Allows for assessment of cytoskeleton and organelle alterations. |
The integration of higher plant compartments into space habitats is not merely a life support consideration but a fundamental pillar for the future of deep space exploration. As mission duration extends, distance from Earth increases, and crew sizes potentially grow, the challenges of resupply, resource recycling, and crew well-being become exponentially more complex. Plant-based systems offer a bioregenerative solution to these challenges, capable of air revitalization, water purification, nutrient production, and psychological support [12]. The design of these compartments is therefore critically driven by the specific mission parameters, moving from technology demonstrations on the International Space Station (ISS) to future self-sustaining habitats on the Moon and Mars.
The interdependence of key mission drivers creates a complex engineering and biological problem space. For instance, long-duration missions to distant destinations inherently preclude rapid resupply, necessitating a high degree of system closure and reliability. Similarly, an increase in crew size directly amplifies the total resource consumption, requiring scalable agricultural output. This document outlines the application notes and experimental protocols essential for advancing the design of higher plant compartments within this multi-faceted context, providing a framework for researchers and scientists engaged in this interdisciplinary field.
Quantitative Analysis of Mission Drivers and System Requirements
The design of plant growth systems must be quantitatively matched to mission profiles. The following tables summarize how core mission drivers influence key design parameters and the subsequent performance requirements for the plant compartment.
Table 1: Impact of Mission Drivers on Plant Compartment Design Parameters
| Mission Driver | Impact on Design Parameters | ISS (Current) | Lunar Habitat (Proposed) | Mars Transit (Proposed) |
|---|---|---|---|---|
| Mission Duration | - Resupply mass - System autonomy - Food closure percentage | Short-term; Low closure | Medium-term; Medium closure | Long-term; High closure |
| Distance from Earth | - Communication latency - Abort/return capability - On-site resource utilization | Low latency; Quick return | Moderate latency; Delayed return | High latency; No return |
| Crew Size | - Volume of food/oxygen - Cultivation area - Labor allocation | Small crew; Limited area | Small-to-medium crew; Expanded area | Medium crew; Significant area |
Table 2: Performance Requirements for Plant Growth Hardware Across Missions
| Performance Metric | Veggie System [12] | Advanced Plant Habitat (APH) [12] | Bioplastic Habitat [13] |
|---|---|---|---|
| Primary Function | Crop production, crew well-being | Fundamental plant research | Closed-loop, sustainable habitat |
| Automation Level | Low (crew-tended) | High (fully automated with remote monitoring) | Conceptual (self-growing) |
| Cultivation Area | ~0.2 m² (6 plant pillows) | Not specified (small-scale research) | Scalable, habitat-scale |
| Key Environmental Controls | LED light (spectrum), root pillows | LED light (full spectrum), automated atmosphere, water, temperature | Pressure containment, UV blocking, light transmission |
| Suitability for Long-Duration/Distant Missions | Limited (supplemental) | High for research, limited for food production | High (potential for self-replication) |
Featured Experimental Protocol: Plant Habitat-07 (PH-07)
The Plant Habitat-07 (PH-07) experiment serves as a critical case study for investigating a key design challenge for long-duration missions: optimizing water and nutrient delivery in microgravity. The following diagram outlines the experimental workflow for this study.
Title: PH-07 Experimental Workflow
Detailed PH-07 Methodology
Objective: To understand the impacts of substrate moisture levels on the growth, nutritional composition, and root microbiome development of 'Outredgeous' red romaine lettuce in the microgravity environment of the International Space Station [5].
Pre-Flight Definition Testing:
- Growth Conditions: Conduct ground tests in strictly controlled environment chambers at Kennedy Space Center to mimic APH settings.
- Duration: Grow plants for 28 days.
- Parameter Definition: Determine the specific substrate moisture conditions required to impose desired growth differentials (e.g., flooded vs. wilt conditions) [5].
- Hardware Setup: Define plant positions within the science carrier quadrants to be between porous tubes and sensors, ensuring post-harvest root recovery is feasible for subsequent microbiome analysis [5].
Verification Testing:
- Science Verification Test (SVT): Execute a 28-day test in the APH ground unit (e.g., Jan 30 - Feb 27, 2024) to refine success criteria and understand moisture control in the flight hardware. Adjust moisture control settings based on observed plant growth [5].
- Experiment Verification Test (EVT): Conduct an extended test (e.g., 35 days from March 20 - April 23, 2024) to investigate any abnormalities in plant growth observed during SVT, potentially requiring sourcing of new growth substrate [5].
Flight Operations:
- Execution: The flight experiment runs on the ISS Advanced Plant Habitat (APH) for three replicate growth cycles of approximately 28 days each.
- In-Flight Procedures: Astronauts conduct the final harvest of the science samples. Minimal crew intervention is required due to the automated nature of the APH [5].
- Sample Preservation: Harvested plant samples are preserved by freezing or chemical fixation for subsequent return to Earth [12] [5].
Post-Flight Analysis: Returned samples are subjected to a multi-faceted analysis:
- Plant Chemistry: Assess nutritional content and potential changes in metabolic profiles.
- Food Safety: Perform cultural microbiology analysis to detect any harmful microbes.
- Microbiome Analysis: Characterize the microbial communities in the root zone.
- Proteomics: Analyze protein expression, often in collaboration with external research centers [5].
- Image Analysis: Quantify growth and morphological changes.
The Scientist's Toolkit: Key Research Reagent Solutions
Successful plant research for space habitats relies on a suite of specialized reagents and materials. The following table details essential items and their functions, drawing from current flight experiments and ground-based protocols.
Table 3: Key Research Reagents and Materials for Space Plant Studies
| Reagent / Material | Function / Application | Example Use-Case |
|---|---|---|
| 'Outredgeous' Red Romaine Lettuce | A well-tested food crop model for spaceflight experiments. | Primary subject in the PH-07 experiment to study moisture stress and microbiome development [5]. |
| Arabidopsis thaliana | Model organism for fundamental plant biology research ("white mouse of the plant research world"). | Used in the Arabidopsis Gravitational Response Omics (Arabidopsis-GRO) study in APH to analyze changes at gene, protein, and metabolite levels [12]. |
| Clay-Based Growth Media / Pillows | Porous substrate to distribute water, nutrients, and air to roots in microgravity, preventing fluid bubbling. | Used in both the Veggie and APH systems to support plant growth [12]. |
| Flag-22 Peptide | A conserved 22-amino acid sequence from bacterial flagella used to experimentally trigger plant immune responses. | Used in BRIC-LED experiments to study the strength of plant defense systems in microgravity without using live pathogens [12]. |
| Polylactic Acid (PLA) Bioplastic | A bioplastic derived from algae that can be 3D-printed into growth chambers; blocks UV radiation while transmitting photosynthetically active light. | Used in habitat experiments to grow algae under Mars-like pressures, demonstrating potential for closed-loop, sustainable systems [13]. |
| Controlled Release Fertilizer | Nutrient source embedded within the growth substrate, providing steady nutrient availability. | Incorporated into the clay substrate of the APH to sustain plant growth throughout the experiment duration [12]. |
Advanced Protocol: Immune Response Interrogation via BRIC-LED
Understanding plant immune function is critical for managing crop health on long-duration missions where resupply of pesticides or replacement plants is impossible. The BRIC-LED protocol provides a method to probe this system safely in space. The signaling pathway involved in this immune response can be visualized as follows:
Title: Plant Immune Signaling Pathway
Detailed BRIC-LED Methodology
Objective: To assess how the microgravity environment alters the gene expression and effectiveness of the plant immune response, using a harmless pathogen mimic [12].
Experimental Workflow:
- Plant Growth: Cultivate Arabidopsis plants within the BRIC-LED hardware for a period of 10 days in the spaceflight environment.
- Immune Elicitation: At day 10, apply a sterile solution of the flag-22 peptide to the plants. This molecule is recognized by plant receptors as a "danger signal," tricking the plant into initiating a full defense response as if it were under pathogen attack [12].
- Response Fixation: Exactly one hour post-elicitation, when the defense response is expected to be at its peak, administer a chemical fixative (e.g., RNAlater or similar) to the plants. This process instantly halts all biological activity, preserving the molecular state of the plant at that precise moment.
- Preservation: Transfer the fixed plant samples to a ultra-low temperature freezer (typically -80°C) on the ISS to maintain biomolecular integrity until return to Earth.
- Ground Analysis: Return samples to Earth for RNA extraction and transcriptomic analysis (e.g., RNA-seq). The resulting gene expression profiles are compared to ground controls to identify how spaceflight alters the plant's investment in and execution of its immune defense [12].
The path to sustainable deep space exploration hinges on the successful integration of bioregenerative life support systems, with higher plant compartments at their core. The design of these compartments is not a one-size-fits-all endeavor but must be meticulously tailored to the triage of mission duration, distance from Earth, and crew size. As demonstrated by the PH-07 and BRIC-LED experiments, this requires a rigorous, iterative approach of ground-based definition testing, flight verification, and post-flight omics analysis.
Future research must focus on closing the loop further, integrating plant compartments with other habitat systems for waste processing, air revitalization, and water recovery. Concepts like the bioplastic habitats being developed at Harvard SEAS point the way toward truly self-sustaining, growable habitats [13]. The protocols and application notes detailed herein provide a foundational framework for researchers to systematically address the key design drivers and advance the capabilities that will one day allow humans to thrive independently of Earth.
The integration of higher plant compartments into Bioregenerative Life Support Systems (BLSS) is a critical requirement for long-duration human space exploration missions [14] [15]. These systems are designed to regenerate resources, produce fresh food, and provide psychological benefits for crew members [14]. However, plant biology in the space environment presents a unique scientific puzzle: studies consistently report significant alterations in gene expression, cell proliferation, and signaling pathways at the cellular level under spaceflight conditions, yet these changes frequently do not manifest as observable phenotypic or developmental abnormalities at the whole-organism level [14]. This apparent paradox represents a fundamental challenge in predicting plant performance and reliability in BLSS, as cellular-level disruptions could potentially compromise system functionality even when plants appear phenotypically normal [14].
Theoretical Framework: Defining the Paradox
Documented Cellular-Level alterations in Space Environments
Spaceflight conditions, particularly microgravity and ionizing radiation, trigger substantial molecular and cellular responses in plants. The following table summarizes key documented alterations across biological organization levels:
Table 1: Documented Plant Responses to Spaceflight Conditions Across Biological Scales
| Biological Scale | Observed Alterations/Responses | Experimental Evidence |
|---|---|---|
| Gene Expression | Reprogramming of gene expression patterns; no specific "gravity genes" identified [14]. | Arabidopsis seedlings in spaceflight and simulated microgravity [14]. |
| Targets include heat shock elements, cell wall remodelling factors, oxidative burst intermediates [14]. | Transcriptomic experiments in simulated and real microgravity [14]. | |
| Cellular Processes | Acceleration of cell cycle progression [14]. | Arabidopsis MM2d cells in Random Positioning Machine (RPM) [14]. |
| Disruption of meristematic competence (coordinated cell proliferation/growth) [14]. | Seedlings onboard ISS and simulation experiments [14]. | |
| Altered auxin and cytokinin distribution and transport [14]. | Studies on Arabidopsis and pea plants in microgravity [14]. | |
| Organism Level | Successful completion of seed-to-seed life cycle [14]. | Plant growth experiments on International Space Station [14]. |
| Production of adult organisms without evident aberrations [14]. | Multiple crop species grown in space habitats [14]. |
Potential Resolution Mechanisms
Several non-exclusive hypotheses may explain the dissociation between cellular alterations and organismic outcomes:
- Developmental Robustness & Genetic Buffering: Plant developmental programs may contain sufficient redundancy to compensate for cellular-level disturbances, ensuring normal phenotypic outcomes despite molecular fluctuations [14].
- Threshold Phenomena: Cellular changes may not reach critical thresholds necessary to disrupt tissue-level functions and overall plant development [14].
- Acclimation vs. Adaptation: Observed cellular changes may represent successful acclimation responses rather than pathological alterations, allowing plants to maintain homeostasis in a novel environment [16].
Experimental Protocols for Paradox Investigation
Protocol: Ground-Based Simulation of Space Factors
Objective: To investigate the effects of simulated microgravity and radiation on plant cellular processes and organismal development.
Materials:
- Plant Materials: Arabidopsis thaliana seeds (wild-type and mutant lines), Solanum lycopersicum 'Microtom' seeds [14] [15].
- Equipment: Random Positioning Machine (RPM) or clinostat, Gamma radiation source, Growth chambers, Sample fixation equipment [14].
Methodology:
- Plant Cultivation:
- Surface-sterilize seeds and sow on appropriate growth media.
- Germinate under controlled conditions (22°C, 16/8h light/dark cycle) [14].
Treatment Application:
Sample Collection and Fixation:
- Harvest samples at multiple time points (e.g., 4h, 12h, 24h, 48h).
- For transcriptomics: Flash-freeze in liquid nitrogen.
- For microscopy: Fix in formaldehyde/glutaraldehyde solution [14].
Parallel Control Experiments:
- Maintain static control plants under identical environmental conditions.
Protocol: Spaceflight Experimentation for BLSS Research
Objective: To validate ground-based findings and assess plant performance under real spaceflight conditions.
Materials:
- Hardware: Vegetable Production System (Veggie), Advanced Plant Habitat (APH), Biological Research in Canisters (BRIC-LED) [12].
- Plant Materials: Selected candidate crops (e.g., dwarf tomatoes, leafy vegetables) [15].
Methodology:
- Pre-flight Preparation:
- Initiate seed germination 10-14 days before launch.
- Pre-install seedlings in spaceflight hardware where applicable.
In-flight Operations:
- Activate growth systems upon reaching orbit.
- Monitor environmental parameters (CO₂, humidity, temperature) continuously.
- Implement manual or automated watering and nutrient delivery.
On-orbit Sampling:
- Conduct crew-assisted sampling where possible.
- Preserve samples using on-board fixation (chemical fixatives) or freezing facilities [12].
Post-flight Analysis:
Essential Research Reagent Solutions
Table 2: Key Research Reagents and Materials for Space Plant Biology Investigations
| Reagent/Material | Function/Application | Specific Examples |
|---|---|---|
| Genetic Tools | Identification of molecular pathways and gene functions | Arabidopsis wild-type and mutant lines (e.g., auxin transport mutants) [14] |
| Fixation Reagents | Preservation of cellular and molecular states for post-flight analysis | Formaldehyde, glutaraldehyde, RNAlater, flash freezing in liquid nitrogen [12] [14] |
| Molecular Biology Kits | Analysis of gene expression and molecular changes | RNA extraction kits, cDNA synthesis kits, qPCR reagents, next-generation sequencing library prep kits [14] |
| Growth Media & Substrates | Support plant growth in confined environments | Clay-based growth "pillows" with controlled-release fertilizer [12] |
| Imaging Reagents | Visualization of cellular structures and processes | Histological stains, fluorescent dyes for cell viability, immunohistochemistry reagents [14] |
Signaling Pathway Visualizations
Diagram 1: Space Stress Perception and Signaling Network
Diagram 2: Experimental Workflow for Paradox Investigation
Quantitative Data Analysis Framework
Key Parameters for BLSS Integration
Table 3: Quantitative Metrics for Assessing Plant Performance in BLSS
| Performance Category | Specific Metrics | Target Values for BLSS | Measurement Techniques |
|---|---|---|---|
| Growth & Development | Time to germination (days) [15] | Species-specific benchmarks | Daily monitoring and imaging |
| Leaf area expansion rate (cm²/day) [15] | Maximize for O₂ production | Image analysis software | |
| Biomass accumulation (g/day) [15] | Optimize for food production | Destructive harvesting | |
| Physiological Function | Photosynthetic rate (μmol CO₂/m²/s) [15] | Maximize efficiency | Gas exchange measurements |
| Transpiration rate (mmol H₂O/m²/s) [15] | Optimize water use | Humidity sensors | |
| Stomatal conductance (mol H₂O/m²/s) [15] | Monitor stress responses | Porometer | |
| Reproductive Success | Time to flowering (days) [14] | Minimize for rapid cycling | Phenological scoring |
| Seed set percentage (%) [14] | >80% of ground control | Harvest and count | |
| Seed viability (%) [14] | >90% germination rate | Germination assays | |
| Molecular Indicators | Stress marker gene expression (fold change) [14] | <2-fold alteration | RNA-seq, qPCR |
| Antioxidant enzyme activity (units/mg protein) [14] | Within 20% of control | Spectrophotometric assays | |
| DNA damage markers (lesions/Mb) [15] | Not significantly elevated | Comet assay, PCR |
Resolving the apparent paradox between cellular-level changes and organismic-level adaptation in plants is fundamental to the reliable design of higher plant compartments for space habitats. The documented success of plants completing full life cycles in space despite molecular alterations suggests inherent robustness mechanisms that ensure developmental stability [14]. Future research should focus on identifying the genetic and physiological basis of this robustness, particularly through comparative studies of species and genotypes with varying sensitivity to space environmental factors [16] [14]. The experimental frameworks and protocols outlined herein provide a systematic approach to investigate this phenomenon, with direct implications for crop selection and BLSS optimization for future missions to the Moon and Mars [14] [15]. A deeper understanding of plant adaptive responses will not only enable safer human space exploration but may also contribute to developing more resilient agricultural systems on Earth.
Implementing Space Agriculture: Hardware, Cultivation Protocols, and Species Selection
The integration of Bioregenerative Life Support Systems (BLSS) is a critical requirement for long-duration human space exploration missions beyond Earth orbit. Within these systems, the higher plant compartment serves a multifunctional role, performing essential functions of resource regeneration, food production, and psychological support for crew members [2]. The design of this compartment must enable plant growth and reproduction in the unique and stressful environment of space, characterized by microgravity and elevated radiation levels [14]. This document details the application notes and experimental protocols for the three flight-proven plant growth systems operating aboard the International Space Station (ISS): the Veggie facility, the Advanced Plant Habitat (APH), and the eXposed Root On-Orbit Test System (XROOTS). Their continued operation provides the foundational research and technological validation necessary for the development of self-sustaining life support for future lunar, Martian, and deep-space missions [11] [2].
Veggie (Vegetable Production System)
Veggie is a modular plant growth unit designed as a "salad machine" to supplement the astronaut diet with fresh, nutritious leafy greens [17] [2]. Its primary design philosophy emphasizes simplicity and crew interaction. The system provides a large, open growth area where plants are cultivated using pillows containing a particulate media-based substrate and fertilizer [18]. Lighting is supplied by an array of light-emitting diodes (LEDs), and environmental control is limited, primarily relying on the circulation of the ISS cabin air [18]. Veggie has successfully grown a variety of crops, including red romaine lettuce, Chinese cabbage, and mizuna mustard, with the produced lettuce being deemed safe for crew consumption [17] [18]. It also serves as a platform for technology demonstrations, such as the XROOTS investigation [19].
Advanced Plant Habitat (APH)
The Advanced Plant Habitat (APH) is the largest and most advanced fully automated, closed-loop environmental control research facility for plant bioscience on the ISS [11] [18]. It occupies the lower half of an EXPRESS Rack and is designed for long-duration experiments of up to 135 days with minimal crew involvement [11]. The APH provides precise control over a wide range of environmental parameters, including light intensity, spectral quality, photoperiod, temperature, CO₂ concentration, and relative humidity [18]. It utilizes a substrate-based, actively watered root module and is equipped with an extensive suite of sensors and three cameras (overhead, sideview, and near-infrared) for continuous, non-destructive monitoring of plant health and gas exchange (canopy photosynthesis and respiration) [18]. Its primary role is to conduct fundamental and applied plant research to understand how plants grow in the spaceflight environment [11].
eXposed Root On-Orbit Test System (XROOTS)
The eXposed Root On-Orbit Test System (XROOTS) is a technology demonstration investigation integrated with the Veggie hardware [19] [17]. It is a middeck locker equivalent (MLE) sized payload that replaces Veggie's baseplate and bellows [19]. XROOTS evaluates soilless cultivation techniques, specifically aeroponic and hydroponic nutrient delivery and recovery systems, in microgravity over a full plant life cycle [19] [20]. The system features multiple independent growth chambers that allow for parallel testing of different configurations and methods [19]. A key feature is the ability to directly observe root zone development and the interaction of fluids with roots through video and still images [17]. The results from XROOTS are critical for developing scalable, gravity-independent plant growth systems for future missions, as particulate media-based systems face mass and sanitation constraints when scaled up [19] [17].
Table 1: Quantitative Comparison of Flight-Proven Plant Growth Systems
| Parameter | Veggie | Advanced Plant Habitat (APH) | XROOTS |
|---|---|---|---|
| System Type | Open, modular production unit | Fully enclosed, automated research chamber | Soilless tech demo (aeroponics/hydroponics) |
| Primary Mission | Food production, crew well-being, tech demo platform | Fundamental & applied plant bioscience | Nutrient delivery system validation |
| Growth Area | ~0.2 m² [18] | ~0.2 m² [18] | Multiple independent chambers (MLE-sized) [19] |
| Environmental Control | Limited (fan-driven ISS air) [18] | Comprehensive (CO₂, humidity, temp, light spectrum) [11] [18] | Dependent on Veggie lighting and ISS environment [19] |
| Nutrient Delivery | Particulate media-based "pillows" (passive watering) [18] | Active watering of substrate-based root modules [18] | Aeroponic and hydroponic techniques [19] |
| Crew Involvement | High (planting, maintenance, harvesting) | Low (water addition, sample collection) [11] | Moderate (system initiation, monitoring) [17] |
| Key Capabilities | Salad machine, psychological benefits | Non-destructive gas exchange, high-intensity LED lighting, imaging | Root zone imaging, nutrient recovery, soilless cultivation [19] [17] |
| Experiment Duration | Varies by crop (e.g., ~30 days for lettuce) | Up to 135 days [11] | 10 to 80 days per test [20] |
Experimental Protocols and Methodologies
Protocol: Plant Cultivation and Gas Exchange Measurement in APH
This protocol outlines the procedure for growing plants and conducting non-destructive gas exchange analysis in the Advanced Plant Habitat, as validated in its hardware demonstration [18].
3.1.1 Research Objective: To cultivate plants from seed to maturity in the APH and measure canopy photosynthetic and respiratory rates using the CO₂ drawdown technique.
3.1.2 Materials and Reagents:
- APH flight unit installed in the ISS Japanese Experiment Module "Kibo"
- Root module pre-loaded with seeds (e.g., Arabidopsis thaliana cv. Col-0, wheat cv. Apogee)
- Sterilized water for system reservoirs
- Seed cassettes and substrate (e.g., clay particles)
3.1.3 Methodology:
- System Initiation and Planting: Crew members install the seed-containing root module into the APH chamber. Water is added to the system's reservoirs. The automated system hydrates the seeds to initiate germination.
- Environmental Profile Execution: A pre-programmed experiment profile is commanded from the Kennedy Space Center (KSC) ground control. This profile schedules:
- Photoperiod: A daily light/dark cycle (e.g., 16h/8h).
- Light Intensity: A photosynthetic photon flux density (PPFD) of up to 1000 μmol m⁻² s⁻¹.
- CO₂ Concentration: Setpoint typically between 400-1200 ppm for controlled drawdown experiments.
- Temperature & Humidity: Maintained at species-optimal levels.
- Routine Monitoring: The three APH cameras (overhead, sideview, NIR) capture predetermined photographic events. Sensor data on chamber environment and root zone moisture are downlinked continuously.
- CO₂ Drawdown Experiment: During the photoperiod, the APH's gas exchange system executes a pre-programmed sequence:
- The CO₂ setpoint is lowered, and the chamber is sealed.
- The rate of CO₂ drawdown by the plant canopy is measured to calculate the canopy photosynthetic rate.
- During the dark period, the rate of CO₂ increase is measured to calculate the canopy respiration rate.
- Data Collection and Analysis: Data on CO₂ concentration, water vapor, and environmental parameters are telemetered to ground stations. Photosynthetic and respiration rates are calculated from the gas exchange data. Image analysis is used to monitor growth and development.
- Sample Harvest (Optional): Upon experiment completion, crew may harvest plant tissue (leaf discs, whole plants) for subsequent return to Earth for omics (genetic, metabolic) analysis [11].
The workflow is summarized in the diagram below.
Protocol: Evaluation of Soilless Nutrient Delivery with XROOTS
This protocol describes the methodology for testing aeroponic and hydroponic techniques in the XROOTS system, which is integrated with the Veggie facility [19] [17] [20].
3.2.1 Research Objective: To evaluate the efficacy of alternative nutrient delivery and recovery systems (aeroponics and hydroponics) for supporting full plant life cycles in microgravity.
3.2.2 Materials and Reagents:
- Veggie flight unit on ISS
- XROOTS module (replaces Veggie baseplate and bellows)
- Seed cartridges
- Pre-mixed nutrient solution
- System reservoir
- Root modules for root observation and fluid capture
3.2.3 Methodology:
- System Integration: Crew members remove the standard Veggie baseplate and bellows. The XROOTS module is mounted to the EXPRESS Rack below the Veggie lighting module [17].
- Initialization:
- Seed cartridges are placed into the designated growth chambers within the XROOTS module.
- A pre-mixed nutrient solution is loaded into the system's reservoir.
- Experiment Initiation: The XROOTS system is powered up and the experiment is initiated. The Veggie unit provides the necessary lighting for plant growth.
- Nutrient Delivery and Recovery:
- Hydroponic Technique: Plant roots are continuously or intermittently submerged in or exposed to a flowing nutrient solution.
- Aeroponic Technique: A fine mist of nutrient solution is sprayed directly onto the exposed root systems.
- The unused nutrient fluid is captured in the root modules and returned to the reservoir for recycling and reuse [17].
- Monitoring and Data Collection:
- Video and Still Imagery: Root zone development and the interaction of the fluid with roots are recorded throughout the growth cycle.
- Crew Observations: Short, periodic crew observations are conducted and recorded.
- Sensor Data: System sensors monitor the performance of the nutrient delivery subsystems.
- Sample Return: At the end of the growth cycle, samples of roots and leaves are harvested, preserved, and returned to Earth for post-flight analysis (e.g., at Kennedy Space Center) to evaluate plant health and system performance [17].
The Scientist's Toolkit: Key Research Reagent Solutions
The following table details essential materials and their functions for conducting plant biology experiments in these flight-proven systems.
Table 2: Essential Research Reagents and Materials for Space-Based Plant Research
| Item | Function & Application | Relevance to System |
|---|---|---|
| Clay-Based Substrate | Serves as a solid growth matrix in particulate media systems; provides support for roots and aids in water and nutrient distribution via capillary action. | Veggie (Pillows), APH (Root Modules) [18] |
| Seed Cassettes/Cartridges | Pre-loaded, standardized containers that simplify the planting process for crew members and ensure correct seed placement. | APH, XROOTS [17] [18] |
| Hydroponic Nutrient Solution | Aqueous solution containing essential mineral nutrients (e.g., N, P, K, Ca, Mg, and micronutrients) required for plant growth in the absence of soil. | XROOTS (Primary), APH (in solution form for root modules) [19] [17] |
| LED Lighting Arrays | Provide the sole source of light for photosynthesis; can be programmed for specific spectral qualities (red, blue, green, white, far-red) and intensities to optimize plant growth and development. | All Systems (Veggie, APH, XROOTS) [11] [19] [18] |
| Root Zone Imaging Systems | Cameras (including near-infrared) and observation windows that allow for non-destructive, in-situ monitoring of root architecture, health, and fluid dynamics. | XROOTS (Primary for roots), APH (Sideview & NIR cameras) [19] [17] [18] |
The operational data and scientific findings from the Veggie, APH, and XROOTS facilities are directly informing the engineering requirements and biological parameters for the higher plant compartment in future BLSS [2]. Veggie demonstrates the viability of fresh-food production and its benefits for crew well-being. APH provides the deep scientific understanding of plant physiological responses to the spaceflight environment required to predict and manage crop performance in closed systems. XROOTS is pioneering the scalable, soilless technologies that will be essential for gravity-independent agriculture on larger scales [19] [17].
The path forward involves scaling these technologies and integrating them into a functional, closed-loop ecosystem. As noted in recent research, "For long-duration missions and the realization of stable planetary outposts, staple crops (e.g., wheat, potato, rice, soy) must be included to provide the carbohydrates, proteins, and fats of the basic diet" [2]. The continued use and evolution of these flight-proven systems on the ISS, and their eventual deployment on lunar and Martian surfaces, will be the cornerstone of achieving the sustainable and self-sufficient human presence in space that is essential for the future of exploration.
The development of robust plant growth systems is a critical component for sustaining long-duration space missions, providing both nutritional supplements and psychological benefits to crew members [12]. In the microgravity environment of space, traditional plant cultivation methods are ineffective due to the absence of gravitational-driven fluid dynamics and sediment settling [21]. This document presents application notes and experimental protocols for three primary cultivation technologies being optimized for space habitats: clay-based substrate pillows, hydroponic systems, and aeroponic systems. These approaches represent the current forefront of research into achieving reliable plant growth despite the unique constraints of spaceflight environments, including microgravity, elevated radiation, and limited resources [22].
Comparative Analysis of Growth Substrates and Systems
The table below summarizes the key characteristics, advantages, and challenges of the three primary plant growth systems investigated for space applications.
Table 1: Quantitative Comparison of Space Plant Growth Systems
| System Parameter | Clay-Based Pillows | Hydroponic Systems | Aeroponic Systems |
|---|---|---|---|
| Growth Substrate | Arcillite (clay-based porous ceramic) [12] [23] | Nutrient solution (soilless) [21] [24] | Mist/aerosol (soilless) [25] |
| Water Delivery | Wick-based capillary action from pre-saturated pillows [12] | Liquid nutrient solution flow [24] | Intermittent misting of nutrient solution [21] |
| Nutrient Delivery | Controlled-release fertilizer embedded in substrate [12] | Direct dissolution in aqueous solution [21] | Direct absorption from aerosolized solution [21] |
| Oxygenation | Passive gas exchange through porous clay [12] | Active/passive aeration of liquid solution [21] | High oxygen availability to exposed roots [25] |
| Implementation Examples | VEGGIE (ISS) [12], VEG-03 MNO [23] | XROOTS (ISS) [25] | XROOTS (ISS) [25] |
| Key Advantage | Simplicity, contained root zone, reduced microbial risk [12] [23] | Efficient nutrient uptake, adaptable to crop variety [21] | Maximum oxygen access to roots, water efficiency [21] [25] |
| Primary Challenge | Limited root volume, single-use design [12] | Managing fluid behavior in microgravity [21] | Nozzle clogging, dependency on precise misting cycles [21] |
Experimental Protocols
Protocol: Plant Cultivation Using Clay-Based Pillows (VEG-03 MNO)
This protocol outlines the procedure for cultivating leafy greens in a Veggie-like unit, based on the VEG-03 MNO experiment performed aboard the International Space Station (ISS) [23].
Research Reagent Solutions & Materials
Table 2: Key Materials for Clay-Based Pillow Experiments
| Item | Function/Description |
|---|---|
| Seed Pillows | Fabric pouches containing a clay-based growth media (e.g., arcillite) and controlled-release fertilizer [12] [23]. |
| Plant Growth Chamber (Veggie Unit) | A compact facility with LED lighting and an environment conducive to plant growth [12]. |
| Seed Library | A selection of sterilized seeds (e.g., 'Dragoon' lettuce, 'Wasabi' mustard, 'Red Russian' kale) [23]. |
| Water Syringe | A tool for manually adding water to the seed pillows to initiate germination and maintain hydration [23]. |
| Data Logger | A device to record temperature, humidity, and other relevant environmental data throughout the growth cycle [23]. |
Methodology
- Preparation and Planting: Insert pre-packaged, fertilizer-containing clay-based substrate pillows into the Veggie facility. Plant surface-sterilized seeds according to a predefined layout within the pillows [12] [23].
- Initiation and Thinning: Use a syringe to hydrate the pillows with a defined volume of water. Seven days after germination, thin seedlings to one plant per pillow to minimize resource competition [23].
- Growth Monitoring: Maintain a bank of light-emitting diodes (LEDs), typically emitting a spectrum rich in red and blue light, to drive photosynthesis [12]. Manually add water as needed. Document plant growth and morphology through regular photographic records [23].
- Harvest and Analysis: After a 28-30 day growth period, harvest the plants. A portion may be consumed by the crew, while samples for scientific analysis must be immediately frozen or chemically fixed for return to Earth. Post-flight analysis can include nutritional profiling and microbial safety testing [12] [23].
Protocol: Hydroponic and Aeroponic Cultivation (XROOTS)
This protocol describes the methodology for the eXposed Root On-Orbit Test System (XROOTS), which investigates soilless cultivation techniques for spaceflight [25].
Research Reagent Solutions & Materials
Table 3: Key Materials for Hydroponic and Aeroponic Experiments
| Item | Function/Description |
|---|---|
| Root Chamber | A module designed to support plants while leaving root systems exposed to the nutrient delivery method [25]. |
| Nutrient Delivery System | For hydroponics: a system to circulate the aqueous nutrient solution. For aeroponics: a high-pressure pump and misting nozzles to create a nutrient-dense aerosol [21] [25]. |
| Nutrient Solution Concentrate | A balanced solution of all essential mineral nutrients required for plant growth, which is diluted with water [24]. |
| Water Recovery System | A system to collect, potentially filter, and recirculate unused nutrient solution and condensate, enabling a closed-loop operation [25]. |
Methodology
- System Priming: Install the root modules and prime the fluidic systems with the nutrient solution. For aeroponics, verify nozzle functionality and misting characteristics [21].
- Seed Germination and Transfer: Germinate seeds in a separate nursery unit. Once seedlings are established, carefully transfer them to the XROOTS system, ensuring roots are properly positioned within the root chamber [25].
- System Operation and Monitoring: Initiate the nutrient delivery cycles. For aeroponics, this involves periodic misting. For hydroponics, a continuous or recirculating flow is maintained. Monitor system parameters including pH, electrical conductivity (nutrient concentration), temperature, and root zone humidity [21] [25]. Use integrated cameras and sensors to monitor root and shoot development non-destructively.
- Sampling and Analysis: At the end of the experiment, harvest plant tissues (roots and shoots). Preserve samples for subsequent omics analyses (e.g., transcriptomics, metabolomics) to understand plant responses to the growth environment and treatment effects [12].
System Workflows and Signaling Pathways
The following diagrams illustrate the logical workflow for operating these systems and a foundational biological pathway relevant to plant growth in space.
Diagram 1: Experimental protocol workflow for space plant growth systems.
Diagram 2: Plant gravity sensing and response pathway.
The design of controlled plant growth compartments is a critical component for long-duration space missions, enabling fresh food production, oxygen regeneration, and water recycling while enhancing crew psychological well-being [12]. These systems must operate with high efficiency and circularity within the extreme constraints of the space environment, including microgravity, cosmic radiation, and limited resource inflow [26]. This document provides application notes and experimental protocols for the precise management of core environmental parameters—LED light spectra, temperature, humidity, and CO₂—to support the advancement of plant growth systems for space habitats.
Quantitative Data Summaries
Optimized LED Light Spectra for Leafy Greens
The following table summarizes the impacts of different supplemental wavelengths added to a white LED base spectrum on the growth of lettuce and basil, as derived from controlled-environment studies [27].
Table 1: Plant Growth Responses Under Different Supplemental LED Spectra
| Light Treatment | Total PPFD (μmol·m⁻²·s⁻¹) | Key Spectral Ratios | Impact on Lettuce (Lactuca sativa) | Impact on Basil (Ocimum basilicum) |
|---|---|---|---|---|
| White (W) [Control] | 122 | Baseline White Light | Lowest values for biomass, leaf number, and area [27]. | Lowest values for biomass, leaf number, and area [27]. |
| White + Deep Red (WDR61) | 122 | Increased DR:B Ratio | Enhanced biomass accumulation compared to W and WFR30 [27]. | Greater fresh and dry weights compared to WFR30 [27]. |
| White + Far-Red (WFR30) | 122 | Decreased DR:FR Ratio | Increased leaf number and canopy expansion compared to W and WDR61 [27]. | Produced larger leaves and more leaves compared to WDR61 [27]. |
| White + DR + FR (WDR61FR30) | 122 | Moderate DR:B & DR:FR | Improved plant performance over single-supplement treatments [27]. | Significantly improved growth metrics over single-supplement treatments [27]. |
| White + DR + FR [High PPFD] (WDR122FR60) | 244 | High DR:B & DR:FR | Highest biomass (76%↑ FW vs. control); highest leaf number, area, and chlorophyll content [27]. | Highest biomass (79%↑ FW vs. control); highest values across all growth parameters [27]. |
Environmental Control Setpoints for Space-Based Plant Growth Systems
The table below synthesizes target parameters for key environmental factors based on operational spaceflight hardware and terrestrial analogues [12] [28] [29].
Table 2: Target Environmental Parameters for Space Habitat Plant Growth Chambers
| Environmental Factor | Target Setpoint / Range | Function and Notes |
|---|---|---|
| Temperature | Controlled to optimum for specific crop (e.g., 22-26°C) [28] [29]. | Regulates metabolic rates; integrated with heating/cooling systems [28]. |
| Relative Humidity | 50-70% for most crops; up to 90% for propagation [30] [31]. | Critical for transpiration and nutrient uptake; prevents plant water stress [28]. |
| CO₂ Concentration | Controlled to specific concentrations [29]. | Enhances photosynthesis; levels are actively managed [29]. |
| Light Intensity (PPFD) | 100-500 μmol·m⁻²·s⁻¹ (adjustable; crop-dependent) [27] [29]. | Drives photosynthesis; red-blue LEDs with supplemental far-red are effective [12] [27]. |
| Light Cycle (Photoperiod) | 16 hours light / 8 hours dark (typical for vegetative growth) [12]. | Regulates plant developmental processes and photosynthesis [12]. |
| Nutrient Delivery | Porous tubes + rooting matrix (e.g., Turface, baked ceramic clay) [12] [29]. | Provides water, nutrients, and oxygen to roots in microgravity [12]. |
| Ethylene Control | < 400 ppb (Non-consumable removal units) [29]. | Prevents accumulation of phytohormone that accelerates senescence and abscission [29]. |
Experimental Protocols
Protocol: Validating Supplemental Far-Red and Deep Red LED Spectra
Objective: To quantify the synergistic effects of supplemental deep red (DR, 660 nm) and far-red (FR, 730 nm) light on biomass and morphological development of leafy greens in a controlled environment.
Materials:
- Plant Material: Seeds of Lactuca sativa cv. 'Batavia-Caipira' and Ocimum basilicum cv. 'Emily' [27].
- Growth Chambers: Multi-chamber setup with independent LED light control.
- LED Light Systems: Banks of white LEDs capable of providing a base PPFD of 122 μmol·m⁻²·s⁻¹, supplemented with independently controllable DR (660 nm) and FR (730 nm) LED arrays [27].
- Data Acquisition: Equipment for measuring fresh and dry weight, leaf area, leaf number, and chlorophyll content [27].
Methodology:
- Chamber Setup: Program five distinct light treatments across growth chambers [27]:
- W: White light only (PPFD 122 μmol·m⁻²·s⁻¹).
- WDR61: White light + DR (61 μmol·m⁻²·s⁻¹), total PPFD 183 μmol·m⁻²·s⁻¹.
- WFR30: White light + FR (30 μmol·m⁻²·s⁻¹), total PPFD 152 μmol·m⁻²·s⁻¹.
- WDR61FR30: White light + DR (61) + FR (30), total PPFD 183 μmol·m⁻²·s⁻¹.
- WDR122FR60: White light + DR (122) + FR (60), total PPFD 244 μmol·m⁻²·s⁻¹.
- Plant Cultivation: Sow seeds in a standardized hydroponic or porous-tube substrate system [12]. Maintain identical temperature, humidity, and CO₂ levels across all chambers.
- Data Collection: After 4-6 weeks of growth (species-dependent) [27]:
- Biomass: Harvest shoots and roots. Record fresh weight immediately. Dry samples in an oven at 70°C for 48 hours to determine dry weight.
- Morphology: Count total leaves. Measure total leaf area using a leaf area meter.
- Physiology: Measure chlorophyll content non-destructively with a SPAD meter or via chemical extraction.
- Data Analysis: Perform statistical analysis (e.g., ANOVA) to compare treatment means for all measured parameters.
Protocol: Integrated Humidity and Temperature Control for Canopy Transpiration
Objective: To maintain optimal relative humidity (RH) and temperature levels in a closed plant growth chamber to ensure efficient plant transpiration and prevent pathogen development.
Materials:
- Sealed Plant Growth Chamber equipped with an air circulation fan.
- Combined Humidity and Temperature Probe (e.g., HC2A-S), calibrated for high accuracy and stability [28].
- Humidification System: High-pressure fogging system with micron-sized nozzles to ensure rapid evaporation [30] [31].
- Dehumidification & Ventilation System: Condensation coils or a system venting to a CO₂ scrubber [29].
- Data Logger connected to the sensor for continuous monitoring.
Methodology:
- System Integration: Connect the humidity/temperature probe to a programmable controller. Integrate the controller with the fogging system and dehumidification/ventilation system.
- Setpoint Programming: Program the controller with a target RH of 60-70% and a temperature setpoint optimal for the crop (e.g., 24°C) [28]. Set a dead band (e.g., ±3%) to prevent constant cycling of equipment.
- Calibration and Sealing: Calibrate all sensors prior to the experiment. Ensure the growth chamber is well-sealed to minimize uncontrolled air exchange.
- Monitoring and Control:
- The controller activates the fogging system when RH falls below the setpoint and activates dehumidification/ventilation when RH exceeds the setpoint [31].
- Monitor the canopy for any signs of water condensation, which indicates poor evaporation and excessive local humidity.
- Validation: Record continuous RH and temperature data throughout the plant growth cycle. Correlate environmental stability with plant health metrics and the absence of fungal pathogens.
System Workflow and Pathway Visualizations
Space Plant Habitat Environmental Control Logic
Light Signaling and Plant Adaptation Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Advanced Plant Growth Environmental Control
| Item Name / System | Function / Application | Specification Notes |
|---|---|---|
| Advanced Plant Habitat (APH) | Fully automated, enclosed plant growth chamber on ISS for research [12]. | Provides control of light (multiple LED colors), temperature, humidity, CO₂, and irrigation. Equipped with 180+ sensors and cameras [12]. |
| Veggie (Vegetable Production System) | Space garden on ISS for plant growth and dietary supplementation [12]. | Uses LED lighting and rooting "pillows" with clay-based media to distribute water and nutrients in microgravity [12]. |
| Porous Tube & Rooting Matrix | Delivers water and nutrients to plant roots in microgravity [29]. | System where water is pushed through a porous tube, drawn into a rooting substrate (e.g., arcillite - baked ceramic clay) [12]. |
| HC2A-S Probe | Accurately measures relative humidity and temperature in greenhouse environments [28]. | Known for high accuracy and stability in humid environments; requires calibration. |
| High-Pressure Fogging System | Humidification and cooling via evaporative misting [30] [31]. | Produces micron-sized water droplets for rapid evaporation; integrated with humidity controllers. |
| White LED Base + Supplemental DR/FR LEDs | Provides precise spectral control for plant growth and morphology studies [27]. | Configurable system with a broad-spectrum white light base, supplemented with 660 nm (DR) and 730 nm (FR) LEDs. |
| Non-Consumable Ethylene Scrubber | Removes ethylene gas from the chamber atmosphere [29]. | Prevents accelerated plant senescence and physiological disorders in closed environments. |
| Biological Research in Canisters (BRIC-LED) | Small, sealed container for studying small organisms and plant seedlings in space [12]. | The LED version supports photosynthesis; used for fixed and frozen sample return. |
The design of the higher plant compartment is a cornerstone of Bioregenerative Life Support Systems (BLSS), which are essential for long-duration human space exploration [2]. These systems aim to achieve a closed-loop regeneration of resources, where plants function as the primary producers of food and oxygen, while also contributing to water purification and waste recycling [2]. The selection of appropriate plant species is therefore not merely an agricultural decision but a critical systems engineering challenge, balancing nutritional output, resource requirements, and cultivation constraints within the extreme environments of space [2]. The overarching goal of a BLSS is to enable mission autonomy by reducing or eliminating the need for resupply from Earth, making crop selection a pivotal factor for mission success [4].
The selection criteria for space crops are inherently mission-dependent [2]. For short-duration missions in Low Earth Orbit (LEO), the focus is on fast-growing leafy greens and dwarf varieties that provide high nutritive value and psychological benefits with minimal volume and resource inputs [2]. In contrast, long-duration missions and planetary outposts (e.g., on the Moon or Mars) require a more comprehensive diet, necessitating the inclusion of staple crops for carbohydrates and proteins, alongside a wider variety of vegetables and fruits to ensure nutritional completeness and crew well-being [2]. This protocol outlines the criteria and methods for selecting and evaluating candidate crops, with a specific focus on the transition from foundational leafy greens to more advanced, nutrient-dense crops like peppers.
Candidate Crop Profiles and Quantitative Comparison
Crop selection for BLSS is guided by a multi-parameter optimization that includes growth cycle duration, edible biomass yield, nutritional density, and resource use efficiency. The following profiles detail key candidate species.
- Leafy Greens (e.g., Lettuce, Kale, Dragoon Lettuce, Wasabi Mustard Greens): These crops are characterized by a short growth cycle and relatively low light and volume requirements, making them ideal for initial food production systems [2]. They are primarily valued for providing fresh vitamins and minerals, complementing a pre-packaged diet [32]. For instance, 'VEG-03' investigations on the International Space Station have included cultivars like Dragoon lettuce and Red Russian kale, demonstrating their feasibility in spaceflight environments [32].
- Dwarf Tomatoes: Determinate and dwarf tomato varieties, such as 'Micro-Tom' and 'Bush Goliath', are selected for their compact growth habit and high fruit yield in confined spaces [33]. These plants typically grow between 0.15 to 1 meter tall, require minimal staking, and are suitable for container and hydroponic cultivation, thus optimizing the use of limited cultivation area [33].
- Peppers (Capsicum annuum L.): Pepper fruits represent a more advanced candidate crop due to their high concentrations of bioactive compounds [34] [35]. They are an excellent source of Vitamin C, carotenoids, and total phenolics [35]. Furthermore, pungent varieties contain capsaicinoids, such as capsaicin and dihydrocapsaicin, which are primarily localized in the fruit's placenta and possess notable antioxidant activity [34]. Studies have shown a positive correlation between the capsaicinoid content and the antioxidant capacity in pepper fruits, suggesting a dual role in nutrition and possible health preservation [34].
Table 1: Nutritional and Growth Profile of Candidate Crops
| Crop Type | Key Bioactive Compounds | Exemplar Cultivars | Growth Habit | Considerations |
|---|---|---|---|---|
| Leafy Greens | Vitamin C, Vitamin K, Fiber | Dragoon Lettuce, Red Russian Kale, Wasabi Mustard Greens [32] | Short cycle, low stature, high harvest index | Fast yield, low energy & mass; limited caloric value [2] |
| Dwarf Tomatoes | Lycopene, Vitamin C, Vitamin A | Micro-Tom, Bush Goliath [33] | Determinate, compact (0.15-1 m), suitable for containers [33] | Provides fruit variety; requires pollination; longer cycle than greens [33] |
| Peppers | Vitamin C, Capsaicinoids, Carotenoids, Phenolics | Alegría riojana, Padrón, Fructus Capsici [34] [35] | Shrub-like; fruit yield at 100+ days for long-cycle varieties [2] | High antioxidant value; pungency varies by cultivar; high Vitamin C content [34] [35] |
Table 2: Antioxidant Profile of Select Pepper Varieties (Capsicum annuum L.)
| Pepper Variety | Pungency Level | Total Phenolic Content | Capsaicinoid Content (e.g., Capsaicin & Dihydrocapsaicin) | Key Antioxidant Compounds |
|---|---|---|---|---|
| Melchor | Sweet (California-type) | Lower | Not Detected [34] | Vitamin C, Carotenoids |
| Piquillo | Slightly Hot | Medium | Low, localized in placenta [34] | Vitamin C, Glutathione, Ascorbate [34] |
| Padrón | Mild Hot | Medium | Medium, localized in placenta [34] | Vitamin C, Glutathione, Ascorbate [34] |
| Alegría riojana | Quite Hot | High | High, localized in placenta [34] | Vitamin C, Glutathione, Ascorbate, High Phenolics [34] |
| Fructus Capsici | Very Hot | Significantly Higher [35] | 1242 μg/g FW [35] | High Phenolics, Vitamin C, Carotenoids [35] |
Experimental Protocols for Crop Evaluation
Protocol: Determination of Capsaicinoids in Pepper Fruits using HPLC-ES/MS
This protocol is critical for quantifying pungency and specific antioxidant compounds in pepper cultivars, a key selection criterion [34].
I. Materials and Reagents
- Research Reagent Solutions:
- Extraction Solvent: Acetonitrile (AcN), HPLC grade.
- Internal Standard Solution: N-[(3,4-dimethoxyphenyl)methyl]-4-methyl-octanamide (DMBMO) at 100 ppm in AcN.
- Mobile Phases: For HPLC, typically acidified water and acetonitrile.
- Plant Material: Fresh pepper fruits; pericarp and placenta should be separated and analyzed independently [34].
II. Sample Preparation
- Homogenization: Freeze plant material (0.5 g) in liquid nitrogen and grind to a fine powder using a laboratory mill [34].
- Extraction: Suspend the powder in 2.0 mL of acetonitrile containing the internal standard (DMBMO, 100 ppm) [34].
- Incubation: Incubate the mixture with sequential shaking for 1 hour at room temperature in the dark, followed by 1 hour at 65°C with intermittent shaking, and a final 1 hour at room temperature [34].
- Clarification: Centrifuge at 16,000× g for 15 minutes. Filter the supernatant through a 0.22 μm PVDF filter prior to HPLC-ES/MS analysis [34].
III. HPLC-ES/MS Analysis
- Chromatography: Separate compounds on a reverse-phase C18 column using a gradient elution with water and acetonitrile, both with 0.1% formic acid.
- Detection: Use electrospray ionization mass spectrometry (ESI/MS) in positive ion mode to detect and quantify capsaicin and dihydrocapsaicin by comparison to the internal standard and authentic chemical standards [34].
Diagram 1: HPLC Capsaicinoid Analysis Workflow
Protocol: Profiling of Antioxidant Enzymatic Systems
This protocol assesses the activity of key antioxidant enzymes, which can indicate plant stress response and metabolic robustness, both crucial for performance in space environments [34].
I. Reagent Preparation
- Enzyme Extraction Buffer: A cold phosphate or HEPES buffer (pH 7.0-7.8) containing polyvinylpyrrolidone (PVP), EDTA, and a protease inhibitor cocktail.
- Assay Buffers: Specific reaction buffers for each enzyme.
- SOD Assay: Buffer containing xanthine, xanthine oxidase, and nitroblue tetrazolium (NBT).
- Catalase Assay: Phosphate buffer with H₂O₂.
- Ascorbate-Glutathione (AGC) Cycle Enzymes: Buffers with specific substrates (e.g., ascorbate for Ascorbate Peroxidase, oxidized glutathione for Glutathione Reductase) and co-factors (e.g., NADPH) [34].
II. Enzyme Extraction
- Homogenize frozen plant tissue in ice-cold extraction buffer.
- Centrifuge the homogenate at high speed (e.g., 15,000× g) for 20-30 minutes at 4°C.
- Collect the supernatant (crude enzyme extract) and keep on ice for immediate assay or flash-freeze for later use.
III. Spectrophotometric Activity Assays
- Superoxide Dismutase (SOD): Monitor the inhibition of NBT reduction by superoxide radicals at 560 nm. One unit of SOD is often defined as the amount that causes 50% inhibition [34].
- Catalase (CAT): Directly monitor the decomposition of H₂O₂ by a decrease in absorbance at 240 nm [34].
- Glutathione Reductase (GR): Monitor the oxidation of NADPH during the reduction of oxidized glutathione (GSSG) at 340 nm [34].
- Ascorbate Peroxidase (APX): Monitor the oxidation of ascorbate at 290 nm [34].
Biochemical Pathways and Research Toolkit
Capsaicinoid Biosynthesis and Antioxidant Interplay
The biosynthesis of capsaicinoids in pepper fruits is a specialized branch of the phenylpropanoid pathway. Understanding this pathway is essential for selecting or engineering varieties with optimal bioactive compound profiles. Research indicates a close interaction between capsaicinoid metabolism and the plant's antioxidant systems, including the ascorbate-glutathione cycle and specific NADP-dehydrogenases, which may help maintain the redox balance required for capsaicinoid production [34].
Diagram 2: Capsaicinoid Biosynthesis Pathway
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for Space Crop Biochemical Profiling
| Reagent / Material | Function / Application | Exemplar Use in Protocol |
|---|---|---|
| HPLC-ES/MS Grade Solvents | High-purity mobile phase and extraction medium for sensitive analyte detection. | Extraction and separation of capsaicinoids [34]. |
| Authentic Chemical Standards | Quantitative calibration and positive identification of target compounds. | Capsaicin, Dihydrocapsaicin for quantification [34]. |
| Stable Isotope-Labeled Internal Standards | Correction for analyte loss during sample preparation and matrix effects in MS. | DMBMO for capsaicinoid analysis [34]. |
| Antibodies for Isozyme Detection | Specific identification and quantification of enzyme isoforms (e.g., Fe-SOD, Mn-SOD). | Differentiating SOD isoenzyme activities [34]. |
| Specific Enzyme Substrates & Cofactors | Spectrophotometric activity assays for antioxidant enzymes. | NADPH for GR activity; H₂O₂ for CAT activity; Ascorbate for APX activity [34]. |
| Controlled Environment Growth Chambers | Simulate space-relevant conditions (light, atmosphere) for ground-based studies. | Pre-flight validation of crop growth and metabolism [2]. |
The integration of advanced automation and monitoring technologies is paramount for the efficient operation of higher plant compartments in space habitats. These systems are designed to sustain plant growth for food, oxygen production, and carbon dioxide recycling while minimizing the demand on precious crew time—a critical limited resource in long-duration space missions [2]. The evolution from direct crew supervision to semi-autonomous and fully remote operation represents a fundamental shift in life support system management, enabling sustainable human presence beyond low Earth orbit [36]. This application note details the current state of sensor integration, camera implementation, and remote operational protocols that collectively enable robust plant growth systems requiring minimal astronaut intervention, directly supporting the advancement of Bioregenerative Life Support Systems (BLSS) for lunar and Martian habitats [2].
System Architectures for Automated Plant Monitoring
Automated plant growth systems deployed in space environments utilize layered architectures that combine physical hardware with intelligent control systems. These architectures enable continuous plant health monitoring and resource management without constant crew presence.
Core Monitoring Subsystems
Table 1: Automated Plant Growth Monitoring Subsystems
| Subsystem | Function | Implementation Examples | Crew Time Reduction |
|---|---|---|---|
| Environmental Sensors | Monitor temperature, humidity, CO₂, O₂ | Advanced Plant Habitat (APH): >180 sensors [12] [37] | Continuous monitoring replaces manual measurements |
| Imaging Systems | Track growth development, root zone dynamics, visual health | Veggie unit: Still and video imagery; XROOTS: Root observation [36] [37] | Reduces need for physical plant inspection |
| Fluidic Management | Deliver water and nutrients via hydroponic/aeroponic systems | XROOTS: Tests nutrient delivery in microgravity [36] | Automates watering and fertilization |
| Lighting Control | Provide programmable spectral quality and photoperiod | Veggie: RGB LED banks; APH: Full-spectrum LEDs with infrared [12] [37] | Eliminates manual lighting adjustment |
| Data Integration | Correlate multiple data streams for system control | APH: Interactive communication with ground teams [12] | Automates response to environmental changes |
The Advanced Plant Habitat (APH) exemplifies this integrated approach, featuring an enclosed, automated chamber with cameras and more than 180 sensors that maintain constant interactive contact with ground teams at Kennedy Space Center [12]. This system autonomously manages water recovery and distribution, atmospheric composition, moisture levels, and temperature, requiring minimal day-to-day crew intervention [12]. Similarly, the eXposed Root On-Orbit Test System (XROOTS) investigates nutrient delivery and recovery techniques using still images and video to observe root development and fluid interaction with roots at different growth stages, providing critical data without crew data collection efforts [36].
Remote Operation Architectures
The implementation of remote operation capabilities follows a tiered approach:
Tier 1: Tele-remote Operation - Operators control systems from a secure environment away from hazardous zones, achieving consistent and precise performance while enhancing operator wellbeing [38]. This approach relocates personnel to controlled environments while maintaining direct control over operations.
Tier 2: Semi-Automation - Systems like AutoNav Lite incorporate autonomous tramming between locations, significantly reducing operator workload while maintaining human oversight for complex decision-making [38].
Tier 3: Multi-Machine Automation - Advanced systems enable a single operator to manage multiple systems on-site through smarter coordination and reduced idle time, dramatically improving personnel efficiency [38].
Tier 4: Fleet Automation - The most advanced level enables operators to remotely oversee entire production areas with intelligent traffic management logic that prevents machine collisions and provides complete operational insight through real-time performance monitoring [38].
Quantitative Data from Deployed Systems
Current space-based plant systems provide substantial data on the performance and efficiency gains achievable through automation.
Table 2: Performance Metrics of Space-Based Plant Growth Systems
| System | Deployment | Automation Features | Crew Time Requirements | Output/Performance |
|---|---|---|---|---|
| Veggie [12] [37] | ISS (2014-present) | Programmable lighting, passive water delivery, manual harvest | Moderate (plant inspection, harvest) | Successfully grown: 3 lettuce types, Chinese cabbage, mizuna mustard, red Russian kale, zinnia flowers |
| APH [12] [37] | ISS (2017-present) | Fully automated environment, 180+ sensors, water recovery, CO₂ control | Minimal (sample collection only) | Research focus: Arabidopsis thaliana, dwarf wheat; Edible crops: radish, Chile peppers |
| XROOTS [36] [37] | ISS (2022-present) | Hydroponic/aeroponic nutrient delivery, root imaging, fluid behavior analysis | Low (system maintenance, cartridge changes) | Testing: 14-60 day growth cycles, multiple crop species to harvest stage |
| Astro Garden (in development) [36] | Ground testing | Large-scale vegetable production, hybrid life support functions | Projected: Minimal daily oversight | Target: Sufficient salad crop production for crew of four |
The data demonstrates a clear evolution toward increasingly autonomous systems. While early systems like Veggie required regular crew attention for basic functions, newer installations like APH operate with remarkable independence. The Advanced Plant Habitat specifically maintains complete automation of its water recovery and distribution systems, atmosphere content, moisture levels, and temperature, requiring intervention only for specific research sample collection or system maintenance [12]. This represents a substantial reduction in crew time commitment compared to earlier plant growth systems.
Experimental Protocols for System Validation
Robust experimental protocols are essential for validating the performance of automated monitoring and control systems for space-based plant growth.
Protocol: Integrated Sensor and Camera System Verification
Objective: Validate the coordinated operation of environmental sensors and imaging systems for autonomous plant health assessment.
Materials:
- Plant growth chamber (Veggie, APH, or equivalent)
- Environmental sensor suite (CO₂, O₂, temperature, humidity, PAR)
- Root and canopy imaging systems
- Data logging system with remote transmission capability
- Reference plant samples (dwarf wheat, red romaine lettuce)
Procedure:
- System Calibration: Calibrate all environmental sensors against reference standards prior to activation. For imaging systems, capture reference images of calibration targets for color and scale verification [12].
- Baseline Establishment: Initiate plant growth with standard nutrient formulations and environmental setpoints. Record initial sensor readings and high-resolution images of planted areas [36].
- Automated Monitoring Cycle: Program the system to execute continuous environmental monitoring with data logged at 15-minute intervals. Schedule canopy imaging daily and root zone imaging every 72 hours for systems with root observation capabilities [36].
- Anomaly Response Testing: Introduce controlled environmental variations (10% CO₂ increase, 15% nutrient concentration decrease) to verify automated system response and alarm generation.
- Data Correlation Analysis: Correlate sensor data with visual observations to establish plant health assessment algorithms. For example, correlate photosynthetic rate changes with visible leaf morphology [39].
- Crew Time Assessment: Document all crew interactions with the system, categorizing by activity type (maintenance, harvest, sampling, system adjustment) to quantify time savings versus conventional plant growth systems.
Protocol: Remote Operation and Control Validation
Objective: Verify system operability from remote locations with minimal crew intervention.
Materials:
- Tele-remote operation workstation
- Video monitoring system with recording capability
- Data transmission system (capable of operating with latency)
- Redundant control pathways
- Performance metric tracking system
Procedure:
- Control Station Setup: Configure remote control station with video monitoring interfaces integrated into control screens, enabling operators to view plant status on the same system used for environmental control [39].
- Communication Latency Testing: Test system responsiveness under simulated communication latencies (0.5-5 second delays) to establish operational parameters for deep space missions [38].
- Automated Event Response: Program the system to automatically respond to common events (low reservoir levels, temperature deviations) and document response efficacy.
- Video Monitoring Integration: Implement continuous recording at the "edge" (where cameras are located) with high-resolution storage and local processing to maintain functionality even during network interruptions [39].
- Failure Mode Testing: Systematically test response to simulated component failures to verify graceful degradation and appropriate alarm generation.
- Crew Time Tracking: Quantify crew time required for system management compared to non-automated baselines, targeting at least 70% reduction in direct oversight requirements [38].
Visualization of System Architecture
The logical relationships and data flow within an automated plant monitoring system can be visualized through the following architecture:
Automated Plant Monitoring Data Flow
This architecture highlights the integrated data flow from sensing to control action, emphasizing minimal crew intervention through edge processing and autonomous control algorithms.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Materials for Automated Plant Monitoring
| Item | Function | Application Example |
|---|---|---|
| Plant Growth Pillows [12] | Clay-based growth media with fertilizer in contained fabric packages | Root support and nutrient delivery in Veggie system; prevents fluid dispersion in microgravity |
| Hydroponic/Aeroponic Assemblies [36] | Soilless nutrient delivery systems for root zone exposure | XROOTS investigation testing fluid dynamics in microgravity |
| Programmable LED Arrays [12] [37] | Specific wavelength control for photosynthesis optimization | Veggie: red, blue, green LEDs; APH: full spectrum plus infrared for imaging |
| BRIC-LED Canisters [12] | Containment system for small plant studies with integrated lighting | Hardware validation for Arabidopsis thaliana experiments |
| Flag-22 Peptide Solution [12] | Elicitor of plant immune response for defense studies | Used in BRIC-LED experiments to simulate pathogen attack in space |
| Chemical Fixatives [12] | Preservation of biological samples for post-flight analysis | RNA preservation for gene expression studies in plant immune response |
| Thermal Imaging Cameras [39] | Non-contact monitoring for stress detection | Leak detection and plant health monitoring in remote systems |
The integration of comprehensive sensor networks, advanced imaging systems, and remote operation capabilities has transformed the management of plant growth systems in space habitats, substantially reducing the crew time requirements for life support operations. Current systems like the Advanced Plant Habitat demonstrate that near-autonomous operation is achievable through extensive sensor integration and ground-based monitoring [12]. The ongoing development of the XROOTS system and next-generation concepts like Astro Garden promise further advances in scalable production systems for long-duration missions [36]. As mission durations extend to Martian scenarios, these automated systems will evolve from supplemental food production to essential components of closed-loop life support, requiring even greater autonomy and reliability [2]. The protocols and architectures presented herein provide a foundation for continued advancement toward fully autonomous plant production systems capable of sustaining human exploration beyond Earth orbit.
Solving Microgravity and Radiation Challenges in Plant Development and Defense
The establishment of robust plant growth systems is fundamental for long-duration space missions, providing food, oxygen, and psychological benefits for crew members [12]. However, the microgravity environment presents significant challenges to plant physiology, primarily through its alteration of fundamental physical processes within the root zone. In the absence of Earth's gravitational pull, fluid behavior, gas exchange, and nutrient uptake are profoundly disrupted, leading to potential constraints on plant growth and development [40]. The root zone, a critical interface for plant resource acquisition, experiences a shift from buoyancy-driven fluid dynamics to a domain dominated by capillary forces [41] [40]. This shift can result in inadequate distribution of water and nutrients, reduced root-zone aeration, and limited oxygen availability to the roots, ultimately impacting overall plant health and yield [40]. This Application Note synthesizes current research and experimental data to provide protocols and design considerations for mitigating these challenges, thereby supporting the advancement of higher plant compartment design for space habitats.
Table 1: Documented Plant Growth and Lignin Content Changes in Microgravity
| Plant Species | Stem Length (Flight, cm) | Stem Length (Control, cm) | Statistical Significance | Lignin Content Change | Citation Source |
|---|---|---|---|---|---|
| Mung Bean (a) | 12.26 | 14.67 | p ≤ 0.001 | Not Specified | [42] |
| Mung Bean (b) | 10.09 ± 2.15 | 12.91 ± 1.62 | p ≤ 0.05 | Not Specified | [42] |
| Oat Seedlings | 14.47 ± 2.44 | 16.23 ± 3.59 | Not Significant | Not Specified | [42] |
| Dwarf Wheat | 14.23 ± 1.25 | 15.13 ± 1.61 | Not Significant | Not Specified | [42] |
| Pine Seedlings | 5.85 ± 0.84 | 6.79 ± 0.80 | p ≤ 0.001 | Not Specified | [42] |
| General Finding | Variable | Variable | -- | Reduced by ~25-100 μg/stem | [42] |
Table 2: Performance of Microgravity Research Platforms
| Platform Type | Microgravity Duration/Capability | g-Level Achieved | Key Advantages | Key Limitations | Best Use Cases |
|---|---|---|---|---|---|
| Random Positioning Machine (RPM) | Unlimited (Ground) | ≤10⁻⁴ g [43] | Cost-effective, unlimited operation time, adjustable gravity [43] | Not real microgravity; introduces mechanical stress [43] | Preliminary studies on gravitropism, mycorrhization [44] [45] |
| Drop Tower | 2.5 – 9.3 seconds | 10⁻³ – 10⁻⁶ g [43] | Highest-quality microgravity; daily access [43] | Very short duration [43] | Fundamental fluid physics, capillary flow validation [41] |
| Parabolic Flight | ~20 seconds per parabola | 10⁻² g [43] | Allows for manned intervention; good for hardware testing [43] | Alternating hyper-gravity phases; limited flight campaigns [43] | Capillary imbibition studies in porous media [41] |
| Orbital Platforms (ISS, Tiangong) | Months to years | ~10⁻⁶ g [43] | Authentic, long-duration microgravity [11] [46] | Extremely high cost; limited access; launch constraints [42] | System-level validation (e.g., APH, Veggie, CRF testing) [11] [46] |
Fundamental Fluid and Root Phenomena in Microgravity
Capillary-Dominated Fluid Dynamics
In microgravity, the absence of gravitational acceleration negates buoyancy-driven convection and sedimentation. This results in fluid behavior being governed primarily by capillary forces, surface tension, and wetting characteristics of the growth substrate [41]. In the context of plant growth, capillary imbibition becomes the principal mechanism for moving water and dissolved nutrients through the porous growth substrate toward the root system [41]. Understanding these dynamics is critical for designing effective watering systems, as improper management can lead to fluid coalescing into bubbles, potentially drowning roots or creating anoxic zones [12].
Altered Root System Architecture and Function
Without a consistent gravity vector, roots lose their primary directional cue (gravitropism). Studies in simulated microgravity show that root growth becomes agravitropic, following multiple directions [45]. To guide growth, plants must rely on other environmental cues, such as hydrotropism (water gradients) and phototropism (light) [44]. Furthermore, the reduced physical demands on plant structural support in microgravity can lead to a significant reduction in the synthesis of lignin, a key structural polymer in plant cell walls, analogous to bone loss in astronauts [42] [12]. This may affect the mechanical strength of plants but could potentially be advantageous for nutrient absorption upon consumption [42].
Experimental Protocols for Root Zone Analysis
Protocol 1: Investigating Capillary Imbibition in Porous Media
This protocol is adapted from spaceflight and parabolic flight experiments to characterize fluid flow in growth substrates under microgravity-like conditions [41].
1. Objectives:
- To quantify the kinetics of capillary-driven fluid front advancement in a porous growth medium under simulated microgravity.
- To validate mathematical models of fluid flow for system design.
2. Materials:
- Porous Medium: Standardized growth substrates (e.g., 80% quartz sand + 20% kaolinite [41], porous ceramic tubes [40], or calcined clay [12]).
- Test Fluid: Water or nutrient solution with optional tracer dye for visualization.
- Apparatus: Random Positioning Machine (RPM) or clinostat [43].
- Monitoring System: High-resolution camera for time-lapse imaging; soil moisture sensors (e.g., TEROS ONE, CS650) for volumetric water content and electrical conductivity [40].
3. Methodology:
- Step 1: Preparation. Pack the porous medium uniformly into a transparent sample column. Ensure initial moisture content is consistent and known (e.g., air-dry).
- Step 2: Mounting. Securely mount the sample column onto the RPM platform. Position the fluid reservoir and wicking interface at the base of the sample.
- Step 3: Experiment Initiation. Start the RPM to achieve simulated microgravity (s‑µg). Activate the fluid supply system to initiate capillary imbibition.
- Step 4: Data Acquisition. Simultaneously record time-lapse video of the fluid front progression and log data from the in-situ moisture sensors at a high frequency (e.g., 1 Hz).
- Step 5: Post-Processing. Analyze video data to measure the distance of the fluid front over time. Correlate visual data with sensor data on moisture content.
4. Data Analysis:
- Plot the position of the fluid front versus the square root of time. A linear relationship is characteristic of capillary-dominated flow (Lucas-Washburn kinetics).
- Compare the imbibition rate and pattern with a 1g control experiment to quantify the microgravity effect.
Protocol 2: Assessing Plant Nutrient Uptake and Mycorrhizal Symbiosis
This protocol outlines methods for evaluating nutrient acquisition efficiency and the role of plant-fungal symbioses in mitigating microgravity-induced nutrient stress [45].
1. Objectives:
- To determine the impact of simulated microgravity on plant phosphate and nitrogen uptake.
- To evaluate the efficacy of arbuscular mycorrhizal (AM) fungi in enhancing nutrient uptake under s‑µg.
2. Materials:
- Plant Models: Petunia hybrida (model for Solanaceae) [45], lettuce (Lactuca sativa) [46].
- Biological Reagent: The AM fungus Rhizophagus irregularis [45].
- Chemical Reagent: The strigolactone mimic rac-GR24 [45].
- Growth System: M1L-type growth chambers or similar, compatible with an RPM [45].
- Analysis Tools: Equipment for measuring plant biomass, elemental analysis (e.g., ICP-MS for phosphate), and gene expression (qPCR for nutrient transporter genes).
3. Methodology:
- Step 1: Plant Germination and Inoculation. Germinate surface-sterilized seeds on agar. For mycorrhizal treatments, inoculate seedlings with R. irregularis spores during transplantation to low-phosphate growth substrates.
- Step 2: Experimental Setup. Transfer plants to M1L chambers. Divide into groups: 1g control, s‑µg (on RPM), and s‑µg with rac-GR24 application.
- Step 3: Growth and Monitoring. Grow plants under controlled light and temperature. Apply rac-GR24 solution to the rhizosphere of designated plants.
- Step 4: Harvest and Analysis. Harvest plants at 30 days post-inoculation.
- Biomass: Measure fresh and dry weight of shoots and roots.
- Mycorrhization: Clear and stain roots, then quantify fungal colonization percentage under a microscope.
- Nutrient Uptake: Analyze shoot and root tissue for phosphate and nitrogen content.
- Gene Expression: Analyze expression of genes related to nutrient deficiency (e.g., LsPHT1 phosphate transporters) [46].
4. Data Analysis:
- Compare biomass and nutrient content between 1g and s‑µg groups using ANOVA.
- Correlate the degree of mycorrhizal colonization with plant nutrient status and growth metrics across treatments.
Signaling Pathways in Microgravity
Diagram Title: Root Response Signaling in Microgravity
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Materials and Reagents
| Reagent / Material | Function / Purpose | Example Application / Note |
|---|---|---|
| Controlled-Release Fertilizer (CRF) | Provides timed-release of nutrients to match plant uptake curves in microgravity, reducing crew intervention [46]. | Bury in middle/lower substrate layers (4:6 ratio) at 3.5 g/L for lettuce in space station tests [46]. |
| Porous Ceramic Tubes | Active irrigation system component that uses capillary action to distribute water in microgravity [40]. | Part of the Utah Reusable Root Module (URRM) and Advanced Plant Habitat (APH) to manage fluid flow [40]. |
| Strigolactone Mimic (rac-GR24) | A synthetic analog of strigolactone phytohormones used to stimulate hyphal branching in mycorrhizal fungi [45]. | Can be applied to challenge the negative effects of simulated microgravity on mycorrhizal symbiosis [45]. |
| Arbuscular Mycorrhizal Fungi | Form symbiotic relationships with plant roots, significantly expanding the functional root surface area for nutrient acquisition [45]. | Rhizophagus irregularis can be inoculated in growth media to enhance phosphate uptake under low-nutrient conditions [45]. |
| Clay-Based Growth Media ("Pillows") | A porous, solid substrate that supports root anchoring and facilitates capillary movement of water and air [12]. | Used in the Veggie system on the ISS to maintain a healthy balance of water, nutrients, and air around roots [12]. |
| Flavonol-Deficient Mutants (e.g., tt4) | Genetic tools to study the role of flavonols in regulating auxin transport and tropic responses [44]. | Used in ground-based studies to elucidate the mechanism of root light avoidance in microgravity [44]. |
Application Notes for Habitat Design
Implement Active, Sensor-Driven Irrigation: Passive watering systems are prone to hypoxia and uneven fluid distribution. Designs must incorporate actively pumped systems with integrated soil moisture sensors (e.g., TEROS ONE) and porous ceramic tubes to ensure precise, gravity-independent water and nutrient delivery directly to the root zone [40]. Automated control loops should maintain moisture within a narrow optimal range (e.g., 15%-25% VWC as in the URRM) to prevent anoxia and salt stress [40].
Optimize Growth Substrate and Fertilizer Formulation: Use low-bulk-density, porous substrates (e.g., calcined clay, peat-moss, processed straw) that promote capillary action and gas exchange [46] [40]. Integrate specialized Controlled-Release Fertilizers (CRFs) with coating thickness and nutrient composition tailored to the altered nutrient uptake curves of plants in microgravity. The application dosage (e.g., 3.5 g/L for lettuce) and placement within the substrate should be optimized for the specific crop [46].
Leverage Biological and Chemical Countermeasures: Inoculate growth systems with mycorrhizal fungi (Rhizophagus irregularis) to extend the root system's reach and improve nutrient foraging in a low-nutrient, confined volume [45]. The application of strigolactone mimics like rac-GR24 can be explored to boost the establishment of this symbiosis under microgravity. Furthermore, selecting or engineering plant varieties with enhanced light avoidance responses (mediated by flavonols and ROS) can help direct root growth appropriately in the absence of gravitropism [44].
Design for Sufficient Root Volume and Aeration: Historical systems like Veggie had limited root volume. Next-generation systems, such as the Utah Reusable Root Module (URRM), double the root growth depth to 33 liters, providing greater resource buffer and root support for multiple harvest cycles [40]. System design must ensure adequate gas exchange within this larger volume to prevent ethylene buildup and root hypoxia.
Gravity is a fundamental environmental factor that has shaped plant evolution and deeply influences all aspects of plant biology [47]. In the context of space exploration, understanding plant gravitational stress responses becomes crucial for designing higher plant compartments in space habitats, as plants will be essential components of Bioregenerative Life Support Systems (BLSS) for generating oxygen, recycling water, and providing fresh food [47] [48]. When plants are subjected to gravitational stress, such as the microgravity conditions of spaceflight or the altered gravity on lunar or Martian surfaces, they initiate complex molecular and biochemical responses that ultimately manifest in changes to growth, development, and structural composition.
The gravitropic response begins with gravity perception by specialized statocytes containing starch-filled statoliths in root columella and shoot endodermis [47]. Recent research has elucidated that in gravity-sensing columella cells, the protein MPK3 phosphorylates LAZY3 and LAZY4 proteins, which associate with amyloplasts [47]. Upon amyloplast sedimentation, these proteins are released and move to the plasma membrane, where they recruit auxin efflux proteins PIN3 and PIN7, establishing an asymmetrical auxin gradient that drives differential cell growth and organ curvature [47]. This fundamental mechanism drives the architectural changes observed in plants exposed to altered gravitational conditions.
Key Experimental Protocols
Gravitropism Stimulation and Tissue Processing for Cell Wall Analysis
Principle: This protocol outlines the methodology for inducing gravitational stress in maize pulvini and analyzing subsequent changes in cell wall composition, providing insights into how plants mechanically adapt to reorientation.
Materials:
- Mature maize plants (60-65 days old)
- RNAlater or similar RNA stabilization solution
- Equipment for mechanical strength testing (puncture probe)
- Reagents for cell wall polysaccharide analysis (for linkage analysis)
- Lignin quantification reagents
- Microarray or RNA-seq equipment for transcriptomic analysis
Procedure:
- Gravistimulation: Place maize plants in a horizontal position to induce gravitropic response. Monitor the recovery to vertical orientation over time [49].
- Tissue Harvesting: Excise pulvini tissues from upper and lower regions at predetermined time intervals after gravistimulation (e.g., 0, 6, 12, 24 hours). Include vertical controls for comparison.
- Tissue Preservation: Immediately stabilize tissues for different analyses:
- For transcriptomics: Preserve in RNAlater at appropriate conditions [50]
- For cell wall analysis: Freeze in liquid nitrogen and store at -80°C
- Cell Wall Composition Analysis:
- Extract non-cellulosic polysaccharides using sequential solvent extraction
- Perform glycosyl linkage analysis to determine polysaccharide structures
- Quantify crystalline cellulose content using Updegraff method or similar
- Determine lignin content using acetyl bromide method or thioglycolic acid assay [49]
- Mechanical Strength Assessment:
- Measure resistance to puncture penetration using a standardized puncture probe
- Compare lower vs. upper pulvini and gravistimulated vs. control plants [49]
- Transcriptomic Profiling:
Spaceflight Transcriptomics Using BRIC Hardware
Principle: This protocol describes the standard approach for conducting plant transcriptomic studies in spaceflight environments using the Biological Research in Canisters (BRIC) hardware, enabling identification of gravity-responsive gene expression patterns.
Materials:
- BRIC hardware with Petri Dish Fixation Units (PDFUs)
- Arabidopsis thaliana seeds (various ecotypes: Col-0, Ler-0)
- Growth medium (agar plates with appropriate nutrients)
- RNAlater fixation system
- -80°C cold stowage when available
Procedure:
- Experimental Design:
- Prepare synchronized Arabidopsis seedlings on agar plates in PDFUs
- Include ground controls with environmental parameters matched to flight conditions
- Spaceflight Implementation:
- In-Flight Fixation:
- Activate RNAlater fixation at experiment termination using actuator gun
- Ensure proper preservation of RNA integrity for subsequent analyses
- Sample Return and Processing:
- Return fixed samples to Earth for analysis
- Extract RNA following standard protocols
- Perform transcriptomic analysis using microarrays or RNA sequencing
- Data Analysis:
- Use bioinformatics methods including RMA (Robust Multi-array Average), MAS5 (Microarray Suite 5.0), or PLIER (Probe Logarithmic Intensity Error Estimation)
- Identify differentially expressed genes with appropriate statistical thresholds
- Perform pathway enrichment analysis for biological interpretation [50]
The Scientist's Toolkit: Research Reagent Solutions
Table 1: Essential Research Reagents for Investigating Gravitational Stress Responses
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| RNA Stabilization | RNAlater | Preserves RNA integrity during spaceflight experiments and tissue processing [51] [50] |
| Cell Wall Analysis | Reagents for glycosyl linkage analysis, acetyl bromide, Updegraff reagent | Quantifies polysaccharide composition, lignin content, and cellulose crystallinity [49] |
| Transcriptomics | Microarray platforms (e.g., Affymetrix), RNA-seq reagents, qRT-PCR kits | Profiles genome-wide gene expression and validates specific targets [49] [50] |
| Hormone Analysis | ELISA kits, LC-MS reagents for auxin, gibberellins, ABA | Measures phytohormone concentrations in responding tissues [49] |
| Polysaccharide Antibodies | Anti-xyloglucan, anti-homogalacturonan, anti-arabinogalactan proteins | Localizes specific cell wall polymers in tissue sections via immunolabeling [50] |
Data Presentation and Quantitative Findings
Cell Wall Composition Changes in Gravistimulated Maize Pulvini
Table 2: Temporal Changes in Cell Wall Components in Lower Pulvini of Gravistimulated Maize Plants [49]
| Time Post-Gravistimulation | Heteroxylan Levels | Lignin Content | Xyloglucan & Heteromannan | Arabinofuranosyl Substitution | Mechanical Strength |
|---|---|---|---|---|---|
| Control (Vertical) | Baseline | Baseline | Baseline | Baseline | Baseline |
| 6 hours | +15-20% | +10-15% | -10-15% | -8-12% | +5-8% |
| 12 hours | +25-35% | +20-25% | -20-25% | -15-20% | +15-20% |
| 24 hours | +40-50% | +30-40% | -30-40% | -25-30% | +25-30% |
| Upper Pulvini (24h) | No significant change | No significant change | No significant change | No significant change | No significant change |
Transcriptomic Changes in Spaceflight-Grown Plants
Table 3: Conserved Transcriptomic Responses Across Multiple Spaceflight Experiments [51] [50]
| Functional Category | Representative Genes | Expression Direction | Biological Significance |
|---|---|---|---|
| Cell Wall Remodeling | XTHs (xyloglucan endotransglucosylases/hydrolases), expansins, cellulose synthases | Mixed (various isoforms) | Modifies cell wall architecture for acclimation to microgravity [50] |
| Stress Responses | Heat shock proteins (HSPs), dehydration-responsive genes, antioxidant enzymes | Upregulated | Counters space environment stressors including radiation and altered gravity [51] [50] |
| Hormone Pathways | Auxin transport (PINs), gibberellin biosynthesis, ABA-responsive genes | Differential regulation | Modulates growth patterns in response to gravitational changes [49] [47] |
| Photosynthesis | Chlorophyll-binding proteins, photosystem components | Often upregulated | Possible compensation mechanism despite being grown in darkness in some experiments [51] |
| DNA Repair | Radiation-responsive genes, DNA ligases, polymerases | Upregulated | Counters cosmic radiation effects in space environment [47] [50] |
Signaling Pathways and Experimental Workflows
Gravitropic Signaling and Cell Wall Remodeling Pathway: This diagram illustrates the molecular pathway from gravity perception to structural adaptation, highlighting how statolith sedimentation triggers auxin redistribution, leading to gene expression changes that modify cell wall composition and enhance mechanical strength through lignin deposition [49] [47].
Spaceflight Plant Experiment Workflow: This workflow outlines the standardized procedure for conducting plant gravitational stress experiments using BRIC hardware, from sample preparation through spaceflight growth to molecular analysis, emphasizing the critical ground control comparisons essential for valid interpretation [51] [50].
Application to Space Habitat Plant Compartment Design
The molecular and structural insights from gravitational stress response studies provide critical design parameters for higher plant compartments in space habitats. Understanding that plants respond to microgravity with specific cell wall remodeling and transcriptional adaptations allows engineers to anticipate and support these biological needs. The consistent findings across multiple spaceflight experiments – including cell wall modification, stress response activation, and altered gene expression patterns – inform the development of targeted environmental controls to optimize plant growth in BLSS [51] [47] [50].
Future space habitat designs can leverage these molecular insights by implementing compensatory environmental stimuli that mitigate stress responses while supporting robust plant growth. This might include specific light regimens that enhance structural strength, atmospheric compositions that optimize cell wall biosynthesis, or mechanical stimulation that substitutes for gravitational cues. The integration of these fundamental biological findings with engineering solutions will be essential for creating sustainable plant growth systems that support long-duration space missions and eventual extraterrestrial settlements.
{# The Application Notes and Protocols}
Mitigating Space Radiation Effects on Plant Physiology, Genetics, and Crop Nutritional Quality
Space radiation constitutes a primary environmental stressor for plant cultivation in Bioregenerative Life Support Systems (BLSS), critical for long-duration exploration missions. The space radiation environment beyond Earth's protective magnetosphere is characterized by a complex field of Galactic Cosmic Rays (GCR) and sporadic Solar Particle Events (SPE), presenting a significant challenge to plant growth, development, and nutritional quality [52] [53]. GCR consists of approximately 85% protons, 14% helium ions, and 1% heavier high-energy ions, while SPEs are predominantly proton-based with lower energy spectra but much higher intensity [53]. This radiation profile induces multifaceted effects on plant systems, from genetic and epigenetic alterations to physiological and nutritional degradation, potentially compromising the sustainability of BLSS for missions to the Moon and Mars [14] [54]. These application notes provide comprehensive protocols for researching and mitigating space radiation effects on plant biology, with specific methodologies for assessing impacts and implementing countermeasures relevant to space habitat design.
Table 1: Space Radiation Environment Components and Biological Significance
| Radiation Type | Composition | Energy Spectrum | Temporal Characteristics | Primary Biological Concerns |
|---|---|---|---|---|
| Galactic Cosmic Rays (GCR) | 85% protons, 14% helium ions, 1% HZE ions [53] | Peaks at 1-2 GeV/nucleon [53] | Constant, isotropic exposure; varies with 11-year solar cycle (higher during solar minimum) [53] | DNA damage, chromosomal aberrations, oxidative stress, chronic effects on growth and development [52] [53] |
| Solar Particle Events (SPE) | Predominantly protons [53] | Peaks ~1 order of magnitude lower than GCR [53] | Sporadic, unpredictable, short duration (hours to days) [53] | Acute radiation syndrome in plants, oxidative burst, membrane damage, potential plant death [52] |
| Secondary Radiation | Neutrons, photons, lighter ions [53] | Varies (depends on primary radiation and shielding) [53] | Continuous inside habitats | Can be more damaging than primary radiation; difficult to shield against [53] |
Quantitative Analysis of Radiation Effects on Crop Nutrition
Research from the International Space Station (ISS) and other platforms has identified significant alterations in the nutritional composition of space-grown crops. The data reveal consistent patterns of nutrient variability that must be addressed through targeted agricultural strategies.
Table 2: Nutritional Composition Changes in Space-Grown Crops (vs. Earth-Grown Controls)
| Nutrient/Metabolite | Crop Example | Change in Space | Recommended Human Daily Intake | Impact of Deficiency on Astronauts |
|---|---|---|---|---|
| Calcium (Ca) | Lettuce (Tiangong II) | Decreased (928 to 642 mg kg⁻¹) [54] | 1000-1300 mg [54] | Bone density loss, osteoporosis risk [54] |
| Magnesium (Mg) | Lettuce (Tiangong II) | Decreased (365 to 274 mg kg⁻¹) [54] | 310-420 mg [54] | Muscle function, neurological issues |
| Iron (Fe) | Lettuce (Tiangong II) | Decreased (9.3 to 6.89 mg kg⁻¹) [54] | Varies (plant sources less bioavailable) [54] | Anemia, fatigue, immune dysfunction [54] |
| Potassium (K) | Lettuce (Tiangong II) | Increased (5280 to 5840 mg kg⁻¹) [54] | ~3500 mg | Generally beneficial for fluid balance, nerve function |
| Total Phenolics | Lettuce (Veggie 2) | Decreased (49.6 to 0.1 mg g⁻¹) [54] | Average intake ~450 mg [54] | Increased disease risk; reduced antioxidant defense [54] |
| Anthocyanins | Lettuce (Veggie) | No significant change (3-5 μg mg⁻¹) [54] | ~12.5 mg [54] | Adequate levels support cardiovascular and visual health [54] |
Experimental Protocols for Assessing Radiation Impacts
Protocol: Gene Expression Analysis in Simulated Space Radiation
Objective: To quantify changes in gene expression patterns in response to simulated space radiation, identifying key pathways involved in stress response and metabolic adaptation.
Materials:
- Plant Material: Arabidopsis thaliana or target crop seeds (e.g., dwarf tomato, lettuce)
- Radiation Source: Ground-based ion accelerator facility capable of delivering proton and HZE ion beams [53]
- RNA Extraction Kit: Column-based with DNase treatment
- qPCR Equipment and SYBR Green master mix
- Microarray or RNA-Seq Platform
Procedure:
- Plant Cultivation: Surface-sterilize seeds and germinate on sterile 0.5x MS medium. Grow seedlings under controlled conditions (22°C, 16/8h light/dark) for 10 days.
- Radiation Exposure: Irradiate experimental groups with:
- Sample Collection: Harvest root and shoot tissues at multiple time points (1h, 6h, 24h, 7d) post-irradiation. Flash-freeze in liquid N₂.
- RNA Extraction: Homogenize 100mg tissue, extract total RNA, quantify purity (A260/280 >1.9).
- Transcriptomic Analysis:
- Perform RNA-seq library preparation and sequencing (Illumina platform).
- Align reads to reference genome (TAIR10 for Arabidopsis).
- Identify differentially expressed genes (DEGs) with threshold of |log2FC|>1 and FDR<0.05.
- Conduct pathway enrichment analysis (GO, KEGG) focusing on oxidative stress, DNA repair, and phenylpropanoid pathways [54].
- Validation: Confirm key DEGs using qPCR with housekeeping genes (ACTIN, UBQ10).
Protocol: Antioxidant and Phytochemical Profiling
Objective: To comprehensively quantify reactive oxygen species (ROS) and antioxidant compounds in space-grown or radiation-exposed plants.
Materials:
- Liquid Nitrogen for sample preservation
- Spectrophotometer or microplate reader
- HPLC-DAD-MS system
- Extraction Solvents: Methanol, acetone, distilled water
- Assay Kits: H₂O₂ content, Lipid peroxidation (MDA), Total antioxidant capacity (ORAC) [54]
- Antioxidant Standards: Ascorbic acid, glutathione, Trolox
Procedure:
- Sample Preparation: Harvest plant tissues, immediately freeze in liquid N₂. Lyophilize and grind to fine powder.
- ROS Quantification:
- Antioxidant Metabolite Analysis:
- Extract 100mg powder with 80% methanol, sonicate, centrifuge.
- Quantify total phenolics using Folin-Ciocalteu method at 765nm, express as gallic acid equivalents [54].
- Measure anthocyanins via pH differential method, absorbance at 520nm and 700nm [54].
- Analyze individual carotenoids (lutein, β-carotene) by HPLC with C30 column, DAD detection at 450nm [54].
- Antioxidant Capacity:
- Perform Oxygen Radical Absorbance Capacity (ORAC) assay using fluorescein probe, AAPH radical generator [54].
- Express results as Trolox equivalents per gram fresh weight.
Protocol: Nutritional Composition Analysis
Objective: To determine mineral content and nutritional value of space-grown crops compared to ground controls.
Materials:
- Inductively Coupled Plasma Mass Spectrometry (ICP-MS) system
- Microwave Digestion System
- Nitric Acid (trace metal grade)
- Multi-element Calibration Standards
Procedure:
- Sample Digestion:
- Weigh 0.5g dried, homogenized plant material into digestion vessels.
- Add 5ml concentrated HNO₃, digest using microwave system (ramp to 180°C, hold 15min).
- Cool, dilute to 25ml with deionized water.
- Mineral Analysis by ICP-MS:
- Calibrate ICP-MS with multi-element standard covering Na, K, Ca, Mg, P, S, Fe, Zn, Cu, Mn.
- Analyze samples using appropriate internal standards (e.g., Sc, Ge, Rh).
- Quantify elements using external calibration, report as mg kg⁻¹ dry weight [54].
- Data Interpretation:
- Compare mineral profiles between space-grown and ground control samples.
- Calculate percentage of recommended daily intake provided by standard serving.
Signaling Pathways and Molecular Responses
The following diagram illustrates the primary molecular signaling pathways activated in plants in response to space radiation, from initial perception to physiological outcomes.
Mitigation Strategies and Countermeasure Protocols
Multi-Layered Radiation Mitigation Framework
A comprehensive approach integrating physical, biological, and nutritional strategies is essential for effective radiation protection in space agriculture. The following diagram outlines this integrated framework.
Protocol: Antioxidant Supplementation for Enhanced Radioresistance
Objective: To apply antioxidant compounds to growth media for mitigating radiation-induced oxidative stress in plants.
Materials:
- Antioxidant Stock Solutions: Ascorbic acid (100mM), Glutathione (50mM), α-Tocopherol (10mM in ethanol)
- Plant Growth Chambers with controlled environment
- Hydroponic or Agar-Based Growth Systems
Procedure:
- Solution Preparation:
- Prepare treatment media by adding antioxidants to sterile growth solution:
- Ascorbate treatment: 1mM final concentration
- Glutathione treatment: 0.5mM final concentration
- Combination treatment: 1mM ascorbate + 0.5mM glutathione
- Filter-sterilize solutions (0.22μm filter) for hydroponic systems or add before agar solidification.
- Prepare treatment media by adding antioxidants to sterile growth solution:
- Plant Treatment:
- Transplant 10-day-old seedlings to treatment media.
- Maintain control groups on antioxidant-free media.
- Apply radiation exposure as described in Protocol 3.1.
- Efficacy Assessment:
- Monitor growth parameters (root length, leaf area, biomass) weekly.
- Quantify ROS levels and lipid peroxidation as in Protocol 3.2.
- Assess antioxidant enzyme activities (SOD, CAT, APX) via commercial assay kits.
Protocol: Crop Biofortification for Nutritional Enhancement
Objective: To develop nutrient-dense crop varieties through traditional breeding and genetic engineering approaches.
Materials:
- Plant Germplasm: Diverse accessions of target crops with varying mineral content
- Molecular Markers for nutrient uptake and transport genes
- Genetic Engineering Tools: CRISPR-Cas9 system, Agrobacterium strains
- Phenotyping Equipment: ICP-MS, HPLC
Procedure:
- Germplasm Screening:
- Grow diverse accessions under controlled radiation conditions.
- Analyze mineral content (Ca, Mg, Fe, Zn) via ICP-MS as in Protocol 3.3.
- Identify high-performing accessions for breeding.
- Gene Identification:
- Analyze expression of nutrient transporter genes (CAX, ZIP, YSL families) under radiation stress.
- Identify candidate genes for enhanced mineral accumulation.
- Genetic Engineering:
- Design CRISPR constructs to modify promoter regions of nutrient transporter genes.
- Transform plant explants via Agrobacterium-mediated transformation.
- Select and regenerate transgenic lines.
- Validation:
- Confirm gene edits by DNA sequencing.
- Evaluate mineral content in T1 and T2 generations under simulated space conditions.
- Assess overall growth and yield parameters.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Materials for Space Plant Radiation Studies
| Reagent/Material | Function/Application | Example Specifications | Research Context |
|---|---|---|---|
| Ion Accelerator Facilities | Simulating space-relevant radiation (protons, HZE ions) [53] | Proton beams: 150-200 MeV/n; HZE: 500-1000 MeV/n [53] | Ground-based simulation of GCR and SPE components for controlled studies |
| Veggie Plant Growth System | Plant cultivation platform on ISS [56] | Low-power chamber holding 6 plants; uses plant "pillows" [56] | Microgravity plant growth experiments; validated for lettuce, Mizuna mustard, tomatoes |
| Advanced Plant Habitat (APH) | Fully automated plant growth facility on ISS [56] | Controlled environment with extensive monitoring and minimal crew attention [56] | Large-scale plant growth studies; enables "seed-to-seed" experiments |
| Random Positioning Machine (RPM) | Ground-based microgravity simulation [14] | 3D rotation to randomize gravity vector [14] | Studying gravity effects independent of radiation; plant cell culture experiments |
| Antioxidant Assay Kits | Quantifying oxidative stress parameters | H₂O₂ content, Lipid peroxidation (MDA), ORAC [54] [55] | Assessing ROS accumulation and antioxidant capacity in space-grown plants |
| RNA-Seq Platforms | Transcriptomic analysis of radiation responses | Illumina sequencing; differential expression analysis [54] | Identifying gene expression changes under space radiation conditions |
| ICP-MS System | Elemental analysis of mineral nutrients | Multi-element detection; low detection limits for Ca, Mg, Fe, Zn [54] | Nutritional profiling of space-grown crops; identifying mineral deficiencies |
| CRISPR-Cas9 Systems | Genetic modification for biofortification | Specific gene edits in nutrient transport pathways [57] | Developing nutrient-enhanced crop varieties for space agriculture |
The protocols and strategies outlined herein provide a comprehensive framework for addressing space radiation effects on plant biology within BLSS. Successful implementation requires an integrated approach combining physical shielding optimization, targeted plant breeding, and nutritional interventions. Researchers should prioritize validation of ground-based radiation simulations with actual space experiments and focus on multi-generational studies to assess long-term adaptation. The development of standardized protocols across the research community will enhance data comparability and accelerate progress toward sustainable plant-based life support for deep space exploration missions.
In the context of designing higher plant compartments for space habitats, maintaining robust plant health is paramount to ensuring food security and life support system reliability. The confined, controlled environments of space agriculture, much like intensive terrestrial cultivation, can predispose crops to pathogen outbreaks. Recent research has heightened scrutiny on the non-target sublethal effects of pesticides, revealing that these compounds can significantly modulate insect immune responses, thereby altering pathogen susceptibility in agroecosystems [58]. This application note examines how both synthetic and biological pesticides can compromise or, in some cases, fortify insect immune defenses, creating unexpected vulnerabilities. We frame these insights within the unique constraints of space-based plant systems, where integrated pest management (IPM) strategies must be precisely controlled, highly reliable, and operate within closed-loop life support systems. The immunotoxicological effects of pesticides, a traditionally overlooked secondary mode of action, now present a critical research frontier for ensuring ecosystem stability in both terrestrial and extraterrestrial agriculture [58].
Data Presentation: Pesticide Effects on Insect Immunity
Table 1: Immunomodulatory Effects of Different Pesticide Classes on Insect Species
| Pesticide Class | Example Compound(s) | Target Insect | Effect on Immune Response | Consequence for Pathogen Susceptibility |
|---|---|---|---|---|
| Neonicotinoids | Various | Model crop pest species | Suppression of immune response [58] | Increased susceptibility to pathogens and parasites [58] |
| Organophosphates | Certain types | Insect species (target & non-target) | Apparent bolstering of immunocompetence under certain circumstances [58] | Potential fitness costs; requires further study [58] |
| Insect Growth Regulators (IGRs) | Certain types | Insect species (target & non-target) | Apparent bolstering of immunocompetence under certain circumstances [58] | Potential fitness costs; requires further study [58] |
| Biological/Bioinsecticides | Bacillus thuringiensis (Bt) | Lepidopteran larvae, mosquitoes, simulids, coleoptera [59] | Gut epithelial cell lysis via δ-endotoxins [59] | Gut paralysis and death; specific mode of action [59] |
Table 2: Entomopathogenic Bacteria as Bioinsecticides for IPM
| Bacterial Species/Group | Key Insecticidal Components | Reported Mode of Action | Primary Target Pests |
|---|---|---|---|
| Bacillus thuringiensis (Bt) | Crystal δ-endotoxins (Cry, Cyt, VIP proteins) [59] | Pore-forming in midgut epithelial cells, disrupting membrane permeability [59] | Lepidopteran larvae, mosquitoes, blackflies, coleoptera [59] |
| Lysinibacillus sphaericus | Binary toxins (BinA, BinB), Mtx proteins [59] | Damage to microvillar epithelial cells in the midgut [59] | Mosquitoes, blackflies, non-biting midges [59] |
| Photorhabdus spp. & Xenorhabdus spp. | Various enzymes and toxins [59] | Proliferation in hemocoel, production of antimicrobials, degradation of tissues [59] | Broad spectrum via symbiotic association with nematodes [59] |
| Brevibacillus laterosporus | Insecticidal Secreted Proteins (ISPs), parasporal bodies [59] | Binary toxin action in midgut, histopathological changes [59] | Coleoptera, Lepidoptera, Diptera, mollusks [59] |
| Serratia spp., Chromobacterium spp. | Various metabolites and toxins [59] | Broad-spectrum insecticidal properties [59] | Various pests (research ongoing) [59] |
Experimental Protocols
Protocol 1: Assessing Sublethal Pesticide Immunotoxicity on Insect Humoral and Cellular Responses
This protocol outlines the methodology for evaluating the effects of sublethal pesticide doses on key insect immune parameters, which is critical for risk assessment in IPM programs.
I. Materials and Reagents
- Test Insects: Rearing cohorts of the target insect species (e.g., crop pests, beneficial pollinators).
- Pesticide Solutions: Prepare serial dilutions of the test pesticide (synthetic or biological) in an appropriate solvent (e.g., acetone, DMSO) and subsequently in a physiological buffer or distilled water. The final concentration of the solvent should be non-toxic to insects (<1%).
- Injection System: Micro-injector (e.g., Nanoject II) or fine glass needles for precise pathogen introduction.
- Immunological Assay Reagents:
- Lysozyme assay kit for lysozyme-like activity measurement.
- Phenoloxidase substrate (L-Dopa) for phenoloxidase (PO) activity.
- Cell culture media and stains (e.g., Giemsa) for hemocyte counting and viability.
- Fine forceps, dissection pins, and microscope slides for encapsulation assay.
- Pathogen Challenge Agents: Bacterial suspensions (e.g., E. coli, Micrococcus luteus), fungal spores (e.g., Beauveria bassiana), or parasitoid eggs.
II. Procedure
- Insect Exposure:
- Expose experimental insect groups to sublethal concentrations of the pesticide via contact, topical application, or tarsal exposure for a defined period (e.g., 24-72 hours). Include a control group exposed to the solvent only.
- Maintain insects under standardized conditions (temperature, humidity, photoperiod) with ad libitum access to food/water.
Hemolymph Collection:
- Anesthetize insects on ice.
- Puncture the insect cuticle (e.g., in a proleg or between abdominal segments) with a fine sterile needle.
- Collect hemolymph using a calibrated microcapillary tube. For enzymatic assays, collect hemolymph directly into a pre-chilled anticoagulant buffer (e.g., sodium citrate, EDTA) on ice to prevent melanization and clotting.
Humoral Immune Assays:
- Phenoloxidase (PO) Activity:
- Dilute a fixed volume of cell-free hemolymph (plasma) in phosphate buffer.
- Add L-Dopa solution and incubate for 30 minutes at 30°C.
- Measure the absorbance at 490 nm spectrophotometrically. The increase in absorbance corresponds to PO activity.
- Lysozyme-like Activity:
- Incubate plasma with a suspension of Micrococcus luteus.
- Measure the decrease in optical density at 450 nm over time, which indicates bacterial cell wall lysis.
- Phenoloxidase (PO) Activity:
Cellular Immune Assays:
- Hemocyte Counts and Viability:
- Dilute a fixed volume of hemolymph in an equal volume of anticoagulant buffer.
- Count total and differential hemocytes using a hemocytometer under a phase-contrast microscope.
- Assess viability using trypan blue exclusion or similar fluorescent dyes.
- Encapsulation Assay:
- Anesthetize the insect and insert a small, sterile nylon monofilament or Sephadex bead into the hemocoel.
- After a standard incubation period (e.g., 2-24 hours), remove the implant.
- Fix the implant and quantify the degree of melanization by measuring absorbance or scoring visually under a microscope.
- Hemocyte Counts and Viability:
In vivo Pathogen Challenge:
- After pesticide exposure, inject a standardized, sublethal dose of a known insect pathogen (e.g., B. bassiana spores) into the hemocoel of the insects.
- Monitor survival and mortality rates daily compared to pesticide-only and pathogen-only control groups.
III. Data Analysis
- Compare mean PO activity, lysozyme activity, hemocyte counts, and encapsulation indices between pesticide-treated and control groups using appropriate statistical tests (e.g., t-test, ANOVA).
- Analyze survival data post-pathogen challenge using Kaplan-Meier survival curves and log-rank tests.
Protocol 2: Molecular Detection of Plant Pathogens for IPM Diagnostics
Robust plant pathology diagnostics are a cornerstone of IPM, especially in closed environments like space habitats where early detection is critical. This protocol adapts modern molecular techniques for this purpose [60].
I. Materials and Reagents
- Sample Material: Plant tissue (leaf, stem, root), soil, or water samples from the growth habitat.
- Nucleic Acid Extraction Kit: Commercial kit for DNA/RNA extraction from the sample matrix.
- PCR Reagents:
- Specific primers for target pathogens (e.g., Pectobacterium, Dickeya, Phytophthora, phytoplasmas) [60].
- PCR master mix (containing Taq polymerase, dNTPs, MgCl₂).
- Nuclease-free water.
- Electrophoresis Equipment: Agarose, TAE buffer, DNA stain, gel documentation system.
- Real-time PCR System: If performing quantitative analysis (qPCR) [60].
II. Procedure
- Sample Collection and Nucleic Acid Extraction:
- Aseptically collect plant tissue showing symptoms or environmental samples.
- Grind the tissue to a fine powder in liquid nitrogen using a sterile mortar and pestle.
- Extract total DNA/RNA using a commercial kit according to the manufacturer's instructions, including appropriate negative controls (no template).
- Quantify and assess the purity of the extracted nucleic acids using a spectrophotometer.
Conventional PCR Amplification:
- Prepare a PCR reaction mix for each sample: 1x PCR buffer, 0.2 mM dNTPs, 0.5 µM each forward and reverse primer, 1 U Taq polymerase, and 50-100 ng of template DNA.
- Run PCR in a thermal cycler with cycling conditions optimized for the primer set (typically: initial denaturation at 95°C for 3 min; 35 cycles of 95°C for 30s, primer-specific annealing temp for 30s, 72°C for 1 min/kb; final extension at 72°C for 5 min).
- Analyze PCR products by agarose gel electrophoresis and visualize under UV light. A band of the expected size indicates the presence of the target pathogen.
Real-Time PCR (qPCR) for Quantification:
- For more sensitive, quantitative detection, use a real-time PCR system with intercalating dyes (SYBR Green) or specific probes (TaqMan, LNA probes) [60].
- Prepare reactions as per the kit protocol and run on the real-time PCR instrument.
- Determine the cycle threshold (Ct) values and quantify the pathogen load against a standard curve of known DNA concentrations.
III. Data Analysis
- In conventional PCR, confirm the identity of the PCR product by sequencing if necessary.
- In qPCR, use the Ct values to quantify the pathogen load. A lower Ct value indicates a higher initial concentration of the target pathogen DNA.
Mandatory Visualization
Diagram 1: Pesticide-Immunity-Pathogen Interaction Network
This diagram illustrates the complex interplay between pesticide exposure, its direct and indirect effects on insect immune pathways, and the resulting impact on pathogen vulnerability.
Diagram 2: IPM Decision Workflow for Closed Habitat Agriculture
This workflow outlines a strategic decision-making process for implementing Integrated Pest Management within the constrained environment of a space habitat plant compartment.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for Investigating Pesticide Immunotoxicity and IPM
| Reagent/Material | Function/Application | Example Use Case |
|---|---|---|
| L-Dopa (L-3,4-dihydroxyphenylalanine) | Substrate for the phenoloxidase (PO) enzyme assay [58] | Quantifying the activation of the key humoral immune pathway in insect hemolymph after pesticide exposure [58]. |
| Lysozyme Activity Assay Kit | Measures lysozyme-like antibacterial activity in insect plasma [58] | Evaluating the effect of a sublethal pesticide dose on the capacity to lyse bacterial cell walls [58]. |
| Pathogen-Specific PCR Primers | Molecular detection and identification of plant pathogens from tissue or environmental samples [60] | Early and accurate diagnosis of bacterial (e.g., Pectobacterium), fungal, or phytoplasma infections in a space habitat plant growth module [60]. |
| Entomopathogenic Bacterial Strains | Biological control agents against specific insect pests [59] | Deployment of Bacillus thuringiensis (Bt) strains for targeted control of lepidopteran larvae without broad-spectrum immunotoxic effects [59]. |
| Nylon Monofilament / Sephadex Beads | Inserts for quantifying the cellular encapsulation response in insects [58] | Assessing the strength of the cellular immune response by measuring melanization on an implanted foreign body after pesticide exposure [58]. |
Application in Space Habitat Plant Compartment Design
The principles outlined in this note directly inform the design of higher plant compartments for space habitats. The recent unavailability of the Advanced Plant Habitat (APH) for new International Space Station studies due to resource constraints underscores the need for highly resilient, self-sustaining agricultural systems [10] [61]. Research must pivot toward IPM strategies that are less reliant on complex, external hardware. Incorporating closed-loop, sustainable habitats using biomaterials, as demonstrated by research into algae-derived bioplastics that can grow under Mars-like pressures, presents a promising avenue [13]. Future space farm designs must integrate on-site molecular diagnostic capabilities [60] and favor biological control agents with specific modes of action [59] over broad-spectrum synthetic pesticides, the sublethal immunotoxicity of which could destabilize the carefully balanced biosphere of a space habitat [58]. This approach minimizes reliance on Earth-resupplied chemicals and hardware, creating a more robust and self-sufficient system for long-duration missions.
Application Notes: The Role of Higher Plants in Bioregenerative Life Support Systems (BLSS)
Core Principles and Rationale
The integration of higher plants into space habitats transcends mere food production; it is a foundational strategy for achieving system resilience through Bioregenerative Life Support Systems (BLSS). A BLSS mimics Earth's ecological networks, where organisms are interconnected through trophic levels, and the waste products from one compartment become the vital resources for another [2]. In this closed-loop paradigm, higher plants function as the primary biological producers, responsible for air revitalization through photosynthesis, water purification via transpiration, food production, and waste recycling through nutrient uptake [2]. This shift from a linear 'take-make-dispose' model to a circular system is the cornerstone of long-term, self-sustainable missions to the Moon and Mars where resupply from Earth is not feasible [62] [2].
The design of the plant compartment is heavily influenced by the mission scenario. For short-duration missions in Low Earth Orbit (LEO), the focus is on fast-growing species that occupy minimal volume and provide high nutritive value, such as leafy greens (e.g., lettuce, kale), microgreens, and dwarf cultivars of tomato [2]. For long-duration planetary outposts, staple crops (e.g., wheat, potato, rice, soy) must be incorporated to provide the carbohydrates, proteins, and fats for a complete diet, substantially contributing to resource recycling [2].
Quantitative Analysis of Space-Grown Crop Nutrition
A critical application note is the observed variance in the nutritional content of plants grown in space compared to Earth-grown controls. Analyses from the Tiangong II space station and the ISS Veggie system have identified specific nutrient deficiencies and shifts, which must be accounted for in dietary planning and soil health management. The following table summarizes key findings from these studies.
Table 1: Nutritional Composition of Space-Grown Lettuce vs. Earth-Grown Controls
| Nutrient/Metabolite | Earth-Grown (Typical Values) | Space-Grown (Typical Values) | Human Daily Recommended Intake | Implication for Astronaut Health |
|---|---|---|---|---|
| Calcium (Ca) | 928 mg kg⁻¹ (Tiangong) [54] | 642 mg kg⁻¹ (Tiangong) [54] | 1000-1300 mg [54] | Compromised bone health [54] |
| 456 mg kg⁻¹ (ISS) [54] | 418 mg kg⁻¹ (ISS) [54] | |||
| Magnesium (Mg) | 365 mg kg⁻¹ (Tiangong) [54] | 274 mg kg⁻¹ (Tiangong) [54] | 310-420 mg [54] | Potential impact on muscle & nerve function |
| Iron (Fe) | 9.3 mg kg⁻¹ (Tiangong) [54] | 6.89 mg kg⁻¹ (Tiangong) [54] | ~8-18 mg [54] | Potential deficiency despite stable levels in some studies; bioavailability is key [54] |
| 10.33 mg kg⁻¹ (ISS) [54] | 11.33 mg kg⁻¹ (ISS) [54] | |||
| Potassium (K) | 5280 mg kg⁻¹ (Tiangong) [54] | 5840 mg kg⁻¹ (Tiangong) [54] | ~2600-3400 mg | Generally stable or increased; beneficial for fluid balance [54] |
| 5295 mg kg⁻¹ (ISS) [54] | 5311 mg kg⁻¹ (ISS) [54] | |||
| Total Phenolics | 49.6 mg g⁻¹ (Veggie 2) [54] | 0.1 mg g⁻¹ (Veggie 2) [54] | ~450 mg (avg. intake) [54] | Increased risk of chronic diseases due to reduced antioxidant intake [54] |
| 54.4 mg g⁻¹ (Veggie 1) [54] | 63.4 mg g⁻¹ (Veggie 1) [54] | Potential stress response in some batches [54] | ||
| Anthocyanins | Variable | 3–5 μg mg⁻¹ (ISS) [54] | ~12.5 mg [54] | Levels in lettuce are stable but insufficient to meet recommended intake [54] |
These nutritional imbalances, particularly in calcium and magnesium, alongside variable antioxidant profiles, may impact astronaut physiology, notably bone health and immune function, especially when coupled with the increased oxidative stress of the space environment [54].
Experimental Protocols for Nutrient Loop Closure
Protocol 1: Hydroponic Cultivation in Microgravity using the Veggie System
Principle: This protocol outlines the operation of the Veggie (Vegetable Production System) on the International Space Station, which uses a porous clay substrate to deliver water, nutrients, and oxygen to plant roots in microgravity, where natural fluid behavior is absent [12].
Applications: Cultivation of leafy greens and dwarf crops for nutritional supplementation and psychological benefits [12] [2].
Materials:
- Veggie Growth Chamber: A light-weight, deployable unit with a bank of LED lights [12].
- Plant Pillows: Pouches containing a calcined clay-based growth media (e.g., Arcillite) and controlled-release fertilizer [12] [21].
- LED Light Bank: Produces a spectrum of light optimized for plant growth, typically emitting a magenta-pink light (combination of red and blue wavelengths) [12].
- Root Mat: A reservoir wicking system for water distribution [12].
- Seeds: Surface-sterilized seeds of approved crops (e.g., lettuce, kale, zinnia) [12].
Procedure:
- Planting: Hydrate the plant pillows with water and install seeds into the clay substrate within the pillows. The pillows are then placed in the Veggie facility [12].
- Initiation: Activate the LED lights and the wicking system to initiate seed germination. The porous clay substrate helps distribute water, nutrients, and air around the roots in the absence of gravity-driven convection [12].
- Growth Monitoring: Plants are monitored daily via downlinked images and direct crew observation. The light cycle is typically set for 16 hours on and 8 hours off [12].
- Harvest: After 28-35 days for leafy greens, plants are harvested. A portion may be consumed fresh, while the rest is preserved for scientific analysis [12].
Notes: The "pillow" system is critical to prevent roots from being engulfed by air bubbles or drowning, as fluids form bubbles in microgravity [12]. The system is semi-automated but requires crew interaction for planting, harvesting, and troubleshooting [12].
Protocol 2: Advanced Automated Cultivation using the Advanced Plant Habitat (APH)
Principle: The Advanced Plant Habitat (APH) is a fully enclosed, sensor-rich, and automated facility for precise plant growth experiments in space. It requires minimal crew intervention for daily operations [12].
Applications: Fundamental plant research to understand plant physiology and genetics in microgravity, enabling future BLSS optimization [12].
Materials:
- APH Chamber: An enclosed and environmentally controlled growth chamber [12].
- Environmental Control System: Manages water recovery and distribution, atmosphere content (CO₂, O₂), moisture levels, and temperature automatically [12].
- Sensors & Cameras: Over 180 sensors and cameras for continuous, interactive monitoring from the ground [12].
- Multi-Spectrum LED Lights: Includes red, green, blue, white, far red, and infrared LEDs for advanced growth and imaging capabilities [12].
Procedure:
- Experiment Loading: The crew loads pre-prepared plant cassettes into the APH [12].
- Automated Growth: The ground team at Kennedy Space Center takes over, using the sensor suite to monitor and control the environment remotely. The system automatically waters the plants and adjusts environmental parameters as per the experimental protocol [12].
- Sample Collection: When plants reach a target growth stage, the crew is instructed to collect plant samples. Samples are either frozen or chemically fixed (e.g., with RNAlater) to preserve the biological state at the moment of fixation [12].
- Sample Return: Preserved samples are stored and eventually returned to Earth for detailed molecular analysis (e.g., RNA sequencing, protein analysis) [12].
Notes: APH's high level of automation and environmental control allows for highly reproducible experiments critical for scientific discovery. The use of chemical fixatives is essential for capturing transient gene expression changes induced by the space environment [12].
System Workflow and Logical Pathway
The following diagram illustrates the integrated logic of a BLSS, highlighting the central role of the higher plant compartment in closing the resource loops for water, nutrients, and air.
BLSS Resource Loop Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
The following table details essential materials and reagents for conducting research on higher plant compartments in space habitats.
Table 2: Essential Research Reagents and Materials for Space Plant Research
| Item Name | Function/Application | Specific Example / Note |
|---|---|---|
| Arcillite | A porous, clay-based substrate used in plant growth "pillows" to anchor roots and distribute water, nutrients, and air in microgravity [12] [21]. | Used in NASA's Veggie system; provides mechanical support and regulates fluid dynamics [12]. |
| Controlled-Release Fertilizer | Provides a steady, long-term supply of essential macro and micronutrients (N, P, K, Ca, Mg, S, etc.) to plants embedded within the growth substrate [12]. | Pre-mixed into arcillite substrate in "plant pillows"; crucial for sustained growth in closed systems [12]. |
| Hydrogels | Polymer matrices that absorb and retain large amounts of water and nutrients, acting as a potential soil-free growth medium or a component thereof [21]. | Proposed for use in substrate-free hydroponic systems to aid the transition to using lunar/Martian regolith [21]. |
| LED Light Arrays | Provides the necessary light spectrum for plant photosynthesis and can be tuned to influence plant growth, morphology, and metabolic output [12]. | Veggie uses red and blue LEDs; APH uses full-spectrum LEDs including white, far-red, and infrared [12]. |
| Chemical Fixatives (e.g., RNAlater) | Preserves the molecular state (e.g., RNA, proteins) of plant tissues at the moment of sampling to enable accurate 'omics analysis post-flight [12]. | Critical for gene expression studies (e.g., assessing plant immune response to spaceflight stressors) [12]. |
| Deep Eutectic Solvents (DES) / Ionic Liquids (IL) | Neoteric solvents used in biomass pre-treatment to break down lignocellulose; can be integrated with nutrient recovery from the biomass [63]. | An emerging technology for recovering mineral nutrients like silicon and potassium during initial processing stages [63]. |
Validating Plant Compartment Designs Through Terrestrial Analogs and International Program Comparisons
Bioregenerative Life Support Systems (BLSS) are fundamental for sustaining long-duration human presence beyond Earth, capable of revitalizing atmosphere, purifying water, producing food, and recycling waste through biological processes. These systems transform exploration missions from logistically dependent ventures into more self-sufficient, sustainable operations. This document details the application notes and experimental protocols derived from three major terrestrial analogues: the Micro-Ecological Life Support System Alternative (MELiSSA), the Bioregenerative Planetary Life Support Systems Test Complex (BIO-PLEX), and the Beijing Lunar Palace (Yuegong-1). The core focus is on the higher plant compartment, a critical subsystem for food production, gas exchange, and water recycling, providing a structured framework for researchers designing plant systems for space habitats.
Historical and Strategic Context
The development of BLSS has been shaped by divergent international strategies. NASA's early pioneering work, through the Controlled Ecological Life Support Systems (CELSS) program and its successor, the BIO-PLEX habitat demonstration program, established the foundational principles of bioregenerative life support [64]. However, following the Exploration Systems Architecture Study (ESAS) in 2004, NASA discontinued and physically demolished the BIO-PLEX facility, opting for a reliance on resupply and physical/chemical-based Environmental Control and Life Support Systems (ECLSS) [64].
In contrast, the China National Space Administration (CNSA) has made substantial investments in BLSS over the last two decades [64]. The CNSA synthesized discontinued NASA research with domestic innovation to develop the Beijing Lunar Palace, an advanced ground-based analog facility. This program successfully demonstrated integrated, closed-system operations, supporting a crew of four analog taikonauts for a full year—a world record that positions China with a demonstrated lead in both the scale and preeminence of BLSS technologies [64]. The European Space Agency's MELiSSA program has pursued a more moderate, yet productive, path focused on the systematic development of individual BLSS component technologies but has not approached fully integrated, crew-tested, closed-loop systems [64].
Table 1: Strategic Posture and Key Characteristics of Major BLSS Programs
| Program | Lead Agency/Country | Primary Focus & Approach | Key Milestone / Status | Notable Strength |
|---|---|---|---|---|
| MELiSSA | European Space Agency (ESA) | Modular, compartmentalized bioregenerative system; robust mechanistic modeling of each compartment. | Continuous component development; no fully integrated human testing to date [64]. | Strong theoretical foundation and detailed compartment modeling. |
| BIO-PLEX | NASA (USA) | Fully integrated, closed-loop habitat demonstration program. | Program discontinued and facility demolished after 2004 [64]. | Pioneered the concept of an integrated bioregenerative habitat. |
| Beijing Lunar Palace | CNSA (China) | Fully integrated, closed-loop architecture for operational lunar outpost. | Crew of 4 sustained for 370 days in a closed system [64]. | Successful long-duration, integrated human testing. |
Quantitative System Performance Comparison
A comparative analysis of key performance metrics reveals the advancements achieved by the Beijing Lunar Palace and highlights the technological gaps resulting from the cancellation of BIO-PLEX. The following table synthesizes available quantitative data from these programs for direct comparison.
Table 2: Comparative Performance Metrics of Terrestrial Closed Ecosystems
| System Parameter | MELiSSA (ESA) | BIO-PLEX (NASA) | Beijing Lunar Palace (CNSA) |
|---|---|---|---|
| Closure Duration (Crewed) | Not achieved [64] | Not achieved (program canceled) [64] | 370 days (crew of 4) [64] |
| Total System Closure | N/A | Targeted >95% mass closure [64] | Demonstrated high closure (specific % not in results) |
| Atmospheric Regeneration | Focus on phototrophic C-fixation and O2 production. | Plant-based O2 production and CO2 sequestration. | Integrated plant compartment for full gas exchange [64]. |
| Water Recovery Rate | Target >95% via biological and physical systems. | Targeted >95% via biological and physical systems [64]. | Demonstrated efficient water recycling from air and waste [64]. |
| Food Production (% of diet) | Component testing (higher plants, spirulina). | Targeted significant contribution [64]. | Successfully supplemented crew diet [64]. |
| Waste Processing Loop | Fully integrated in concept (MELiSSA loop). | Integrated biological waste processing planned [64]. | Fully integrated and demonstrated [64]. |
Core Experimental Protocols for BLSS Research
Protocol: Construction and Monitoring of a Simplified Closed Ecosystem
This foundational protocol, adapted from educational models used by NASA and the National Space Society, allows for the study of basic ecological principles and system stability in a sealed microenvironment [65].
I. Materials (Research Reagent Solutions) Table 3: Essential Materials for Closed Ecosystem Construction
| Item | Function/Justification |
|---|---|
| Large clear glass or polycarbonate bottle (e.g., 2-5 L) | Serves as the primary containment vessel, allowing for light penetration and visual observation [65]. |
| Air-tight sealing mechanism (cap, silicone sealant) | Ensures material closure, preventing exchange of gases and liquids with the external environment [65]. |
| Abiotic Components: Potting soil, water, gravel | Provides the physical substrate, anchor for roots, and reservoir for water and minerals. |
| Biotic Components: Small vascular plants (e.g., Arabidopsis, clover), algae (e.g., from pond water), decomposers (soil microbiota) | Forms the core biological loop: plants produce O2/food, decomposers recycle dead matter, algae can assist in nutrient cycling [65]. |
| Instrumentation: Data logger, pH/CO2/O2 sensors (optional) | Enables quantitative monitoring of internal environmental parameters over time [65]. |
II. Methodology
- Preparation: Clean the bottle thoroughly to remove contaminants. Add a base layer of gravel for drainage, followed by a layer of soil [65].
- Introduction of Biota: Introduce selected plants, ensuring they are securely rooted. Add a small amount of water containing algae or pond scum to introduce microbial and algal diversity [65].
- Sealing: Cap the bottle securely. For enhanced closure, seal the cap-to-bottle interface with melted wax or a silicone-based sealant [65].
- Placement and Energy Input: Place the sealed ecosystem in a location where it will receive at least indirect sunlight. This light is the sole source of energy driving photosynthesis [65].
- Data Collection & Observation:
- Qualitative: At regular intervals, record observations of plant health (color, new growth), algal density, condensation levels, and any visible decay [65].
- Quantitative: If sensors are installed, record data on temperature, light intensity, and gas composition (O2, CO2) to track diurnal and long-term cycles [65].
III. Experimental Design & Hypothesis Testing After establishing a baseline, researchers can use these simple ecosystems for controlled experiments.
- Hypothesis: "System stability, measured by plant survival duration, is positively correlated with initial biodiversity."
- Experimental Variation: Create multiple ecosystems that differ only in their initial biotic complexity (e.g., plants only vs. plants + algae vs. plants + algae + detritus worms) [65].
- Analysis: Monitor all systems until failure (e.g., plant death, anoxic conditions). Compare the mean survival duration and stability of environmental parameters between the different experimental groups [65].
Protocol: Higher Plant Cultivation for Gas Exchange and Food Production
This protocol outlines the key considerations for managing a higher plant compartment, drawing from the integrated successes of the Beijing Lunar Palace and the design parameters of BIO-PLEX.
I. Plant Selection Criteria
- Food Value: High yield, nutritional density, and palatability (e.g., potato, wheat, soybean, leafy greens like lettuce and spinach).
- Growth Characteristics: Short life cycle, dwarf morphology suitable for confined spaces, and high harvest index.
- Environmental Impact: High rates of photosynthesis (O2 production, CO2 consumption) and transpiration (water recycling).
II. Cultivation System Workflow The following diagram outlines the logical workflow and integration points for a higher plant compartment within a BLSS.
III. Key Operational Parameters
- Lighting: Use LED arrays with programmable spectra (Red/Blue for efficiency, full spectrum for plant health). Maintain a Photosynthetic Photon Flux Density (PPFD) of 200-600 µmol/m²/s for most crops with a photoperiod of 16-20 hours.
- Nutrient Delivery: Employ hydroponic (Nutrient Film Technique, Deep Water Culture) or aeroponic systems for precise water and nutrient control. Monitor and adjust Electroconductivity (EC) and pH of the nutrient solution continuously.
- Atmospheric Control: Maintain CO2 levels at 1000-1200 ppm to enhance photosynthesis. Manage relative humidity (50-70%) to optimize transpiration and prevent pathogen growth.
- Pollination: For fruiting crops, implement manual pollination or introduce managed pollinators (e.g., stingless bees or mechanical vibration systems).
The Scientist's Toolkit: Key Research Reagents and Materials
Successful BLSS research relies on a suite of specialized reagents, materials, and instrumentation.
Table 4: Essential Research Reagents and Materials for BLSS Experimentation
| Category / Item | Specific Function in BLSS Research |
|---|---|
| Plant Growth Materials | |
| Hydroponic Nutrient Solutions | Precisely formulated to deliver essential macro (N, P, K, Ca, Mg, S) and micronutrients (Fe, Mn, B, Zn, Cu, Mo) in a readily available form. |
| Seed Stock (Dwarf varieties) | Genetically compact plants with high harvest index are selected for efficient use of confined volume and energy (e.g., dwarf wheat, tomato). |
| Solid Growth Substrates | Inert, porous media (e.g., baked clay pellets, rockwool, oasis foam) for plant support and root zone aeration in hydroponic systems. |
| Environmental Monitoring | |
| CO2 & O2 Gas Sensors | Critical for real-time monitoring of the photosynthetic and respiratory balance within the closed system. |
| Data Logging Systems | For continuous recording of environmental parameters (T, RH, light, gas levels) to correlate with system performance and plant health. |
| Water Quality Test Kits | For monitoring pH, Electroconductivity (EC), and specific nutrient ion concentrations (NO3, NH4, PO4) in hydroponic solutions. |
| Biological System Management | |
| Biological Control Agents (BCAs) | Beneficial insects (e.g., predatory mites) or microorganisms used for integrated pest management, avoiding chemical pesticides. |
| Microbial Inoculants | Selected beneficial bacteria (e.g., Rhizobia, PGPR) and mycorrhizal fungi to enhance plant nutrient uptake and stress resistance. |
| Tissue Culture Reagents | For the sterile, space-efficient propagation and preservation of plant germplasm (Murashige & Skoog medium, agar, plant growth regulators). |
The lessons from MELiSSA, BIO-PLEX, and the Beijing Lunar Palace provide a critical knowledge base for advancing higher plant compartment design. The success of the Beijing Lunar Palace underscores the feasibility of fully integrated, bioregenerative life support and highlights the strategic risk associated with discontinuing development in this field [64]. Key research priorities to mitigate this risk include closing the "last 5%" of mass loops (particularly complex organic waste and trace gases), developing highly automated, diagnostic-driven cultivation systems, and breeding "space-optimized" plant cultivars for controlled environments. Furthermore, comprehensive study of plant-microbe interactions within closed systems and the effects of deep space radiation on plant growth and reproduction are essential for missions beyond Low Earth Orbit [64]. A renewed and collaborative international effort is required to mature these technologies for the deployment of sustainable, endurance-class human space habitats.
Bioregenerative Life Support Systems (BLSS) are advanced closed-loop habitats that use biological processes to recycle air, water, and waste while producing food for crewed space missions. These systems are critical for long-duration exploration beyond Earth orbit where resupply from Earth becomes impractical. This analysis compares the capabilities, investments, and strategic approaches of three major space agencies—NASA (USA), ESA (Europe), and CNSA (China)—in developing BLSS technologies. The content is framed within broader thesis research on higher plant compartment design, providing specific experimental protocols and technical data relevant to scientists and researchers working on biological life support systems.
Agency Capabilities and Strategic Approaches
Table 1: Comparative Overview of Major BLSS Programs
| Agency/Program | Key Facilities & Demonstrators | Primary Focus Areas | Current Status & Achievements | Notable Investments |
|---|---|---|---|---|
| NASA (USA) | BIO-PLEX (demolished), CELSS program historical | Physical/Chemical ECLSS, limited bioregenerative research | Relies on resupply; critical gaps in BLSS after program discontinuations [66] | Budget constrained; shift from historical BLSS investments [66] |
| CNSA (China) | Beijing Lunar Palace 1 | Fully integrated, closed-loop BLSS | 4-crew, 1-year closed-system operation demonstrated [66] | Substantial sustained investment; lead in operational BLSS [66] |
| ESA (Europe) | MELiSSA Pilot Plant (Spain), PaCMan (Italy) | Component technology development, waste recycling, air/water revitalization | Progressive development; no fully integrated human testing yet [2] | Moderate but productive investments; new ACCESS program proposed [67] |
Table 2: Technical Capability Comparison
| BLSS Function | NASA Capability | CNSA Capability | ESA Capability |
|---|---|---|---|
| Air Revitalization | Primarily physicochemical | Biological + physicochemical demonstrated | MELiSA loop component testing |
| Water Recovery | ECLSS system on ISS | Closed-loop biological system demonstrated [66] | Water purification tested at Concordia [2] |
| Food Production | Limited crop research (VEGGIE) | Staple crops & balanced diet production [2] | Plant research (PaCMan), MELiSSA photobioreactors |
| Waste Recycling | Limited biological processing | Integrated waste degradation & recycling [2] | MELiSSA waste processing compartments |
| System Integration | Discontinued after BIO-PLEX | Fully integrated system demonstrated [66] | Component focus, no full human testing [2] |
Experimental Protocols for BLSS Higher Plant Compartment Research
Protocol: Multi-Trophic BLSS Integration Testing
Objective: Evaluate resource exchange efficiency between plant, microbial, and human compartments in a closed-loop environment [2].
Materials:
- Sealed plant growth chamber (≥2m² cultivation area)
- Connected photobioreactor with microalgae (e.g., Spirulina)
- Air and water recycling subsystems
- Gas monitoring system (O₂, CO₂, VOCs)
- Water quality sensors (pH, nutrients, contaminants)
- Data logging system
Procedure:
- Establish baseline operation of individual system components
- Initiate closed-loop operation by connecting air and water streams between compartments
- Introduce human-equivalent metabolic load (CO₂ injection, water vapor, nutrient solution mimicking waste)
- Monitor gas exchange rates every 6 hours for 30 days
- Measure biomass production and resource recovery efficiency
- Analyze system closure stability through mass balance calculations
- Perform contingency testing (component failure simulation)
Data Analysis:
- Calculate daily carbon fixation rate per m² of plant growth area
- Determine oxygen production to consumption ratio (P:C)
- Quantify water transpiration and recovery rates
- Assess nutrient recycling efficiency from waste streams
Protocol: Plant Cultivation Optimization for Different Mission Scenarios
Objective: Determine optimal crop species and cultivation parameters based on mission duration and resource constraints [2].
Materials:
- Controlled environment growth chambers
- Selected candidate species (leafy greens, dwarf crops, staple foods)
- Hydroponic/aeroponic nutrient delivery systems
- LED lighting systems with adjustable spectra
- Environmental monitoring equipment
- Harvest processing and analysis tools
Procedure:
- Short-duration mission simulation (≤6 months):
- Cultivate fast-growing species (lettuce, kale, microgreens)
- Optimize for minimal volume and energy requirements
- Measure growth rate, edible biomass yield, and nutrient density
Long-duration mission simulation (≥12 months):
- Cultivate staple crops (potato, wheat, soy) and longer-cycle vegetables
- Optimize for balanced nutrition and high edible biomass ratio
- Measure resource requirements per kcal produced
Psychological benefit assessment:
- Document crew interaction with plant compartments
- Assess horticultural therapy benefits through standardized questionnaires
Parameters Measured:
- Biomass accumulation rate
- Edible to total biomass ratio
- Water and nutrient use efficiency
- Light energy conversion efficiency
- Nutritional content analysis
BLSS Research Workflow and Signaling Pathways
Diagram 1: BLSS Research and Development Workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagents and Experimental Materials for BLSS Plant Research
| Category/Item | Specification/Purpose | Application in BLSS Research |
|---|---|---|
| Plant Growth Media | Solid: Calcined clay, porous ceramic; Liquid: Hydroponic nutrient solutions | Root support and nutrient delivery in controlled environments [2] |
| LED Lighting Systems | Adjustable spectra (R:B ratio 1-5:1), programmable photoperiods | Optimize photosynthesis and morphology for different crop types [2] |
| Environmental Sensors | CO₂, O₂, ethylene, VOCs, temperature, humidity, PAR | Real-time monitoring of closed-system atmospheric conditions [2] |
| Nutrient Solution Analysers | Ion-selective electrodes, HPLC for phytohormones | Maintain nutrient balance and detect plant stress markers [2] |
| Gas Exchange Equipment | Infrared gas analyzers, portable photosynthesis systems | Quantify photosynthetic and respiratory rates of plants [2] |
| Seed Stock | Space-optimized cultivars (dwarf, high yield, nutrient-dense) | Food production experiments; genetic stability studies [2] |
| Water Recycling Components | Reverse osmosis, catalytic oxidation, biological filters | Process and recycle plant transpiration and crew waste water [2] |
Discussion and Future Research Directions
The comparative analysis reveals significantly divergent paths in BLSS development among major space agencies. CNSA has established leadership in fully integrated, operational BLSS through sustained investment and testing, notably with their Lunar Palace facility supporting crew for extended durations [66]. NASA's historical leadership in this area was compromised by program discontinuations, creating strategic capability gaps that may impact future deep space exploration ambitions [66]. ESA maintains a more focused, incremental approach through the MELiSSA program, advancing component technologies without yet attempting full system integration with human crews [2].
Future research should address critical knowledge gaps in plant and microbial responses to deep space environmental factors, including reduced gravity, increased radiation, and closed-system ecological dynamics. The development of BLSS technologies not only enables long-duration human space exploration but also contributes to sustainable technologies with Earth applications, aligning with United Nations Sustainable Development Goals [68].
The development of higher plant compartments is a critical research frontier for sustaining long-duration human space exploration. Bioregenerative Life Support Systems (BLSS) will require advanced Controlled Environment Agriculture (CEA) technologies to provide astronauts with fresh food, oxygen, water regeneration, and psychological benefits [2]. On Earth, vertical farming has emerged as a robust analog for developing these space-based systems, offering optimized resource efficiency in confined environments. These terrestrial facilities enable researchers to prototype and refine the technologies needed for space habitats, including fully automated, closed-loop agricultural systems [69] [70]. This document outlines application notes and experimental protocols for translating urban agriculture CEA technologies to space habitats, with specific focus on validating systems for integration into BLSS architectures.
Performance Comparison: Terrestrial CEA vs. Space Agriculture Requirements
The table below summarizes key performance metrics for terrestrial vertical farming systems and compares them with the projected requirements for space-based agriculture. This quantitative framework provides benchmarking data for researchers developing space-adapted CEA systems.
Table 1: Performance Metrics for Terrestrial Vertical Farming and Space Agriculture Applications
| Performance Parameter | Terrestrial Vertical Farming (Leafy Greens) | Space Agriculture Requirements & Findings |
|---|---|---|
| Yield (Fresh Weight) | 60–105 kg m⁻² year⁻¹ (cultivated area) [71] | Varies by mission duration; "salad machines" for short-duration, staple crops for long-duration [2] |
| Land Use Efficiency | 350 kg m⁻² year⁻¹ (floor area) [71] | Maximum production in minimal volume is critical [57] |
| Water Use Efficiency | ~140 g FW L⁻¹ H₂O [71] | Closed-loop water recycling is essential; plants contribute to water regeneration [2] |
| Energy Use Efficiency | 0.08–0.13 kg FW kWh⁻¹ [71] | High energy demand is a key challenge; optimization is ongoing [71] |
| Global Warming Potential | ~2.9 kg CO₂ kg⁻¹ FW [71] | System mass and resupply mass reduction are primary goals [2] |
| Growth Cycle (Lettuce) | 4-5 harvests per year possible [72] | Successfully grown on ISS using Veggie system [12] |
Experimental Protocols for Technology Translation and Validation
Protocol: Ground-Based Validation of Plant Growth Systems for Microgravity
This protocol describes the methodology for testing and adapting terrestrial vertical farming hardware for space compatibility using ground-based microgravity simulation platforms.
- Objective: To evaluate the performance of hydroponic and aeroponic growth systems under simulated microgravity conditions and assess plant growth phenotypes.
- Materials:
- Random Positioning Machine (RPM): A 3D clinostat to simulate a randomized gravity vector [43].
- Plant Growth Chambers: Sealed chambers with environmental control (e.g., PaCMan facility) [2].
- Plant Material: Arabidopsis thaliana seeds or dwarf crop varieties (e.g., 'Red Robin' tomato) [2] [57].
- Growth Substrate: Clay-based growth "pillows" or aeroponic systems [12].
- Sensor Arrays: Monitoring for pH, nutrient concentration, dissolved oxygen, and temperature.
- Procedure:
- System Setup: Install the aeroponic or hydroponic growth module on the platform of the RPM.
- Calibration: Calibrate the RPM to maintain a sample gravity threshold of ≤10⁻³ g [43].
- Plant Germination: Germinate seeds under controlled 1g conditions until radicle emergence.
- Experimental Run: Transfer seedlings to the RPM-mounted system. Initiate microgravity simulation for a duration of 10-14 days.
- Environmental Control: Maintain the following conditions throughout the experiment:
- Data Collection:
Protocol: Assessing Plant Immune Function in Closed Environments
Spaceflight may alter plant immune responses. This protocol uses a flagellin peptide assay to probe immune system robustness in plants grown in closed-loop CEA systems.
- Objective: To determine if controlled environment growth affects the ability of plants to mount a defense response against pathogens.
- Principles: Plants possess pattern recognition receptors that detect conserved microbial molecules. The flagellin-derived peptide flg22 is a potent elicitor of plant immunity [12].
- Materials:
- Plant Material: Arabidopsis thaliana (4-week-old) or lettuce plants grown in a controlled environment.
- Reagent: Synthetic flg22 peptide solution.
- Fixative: Chemical fixative (e.g., RNAlater).
- Equipment: Liquid handling system, deep-freeze storage (-80°C).
- Procedure:
- Acclimation: Grow plants in the target CEA system (e.g., vertical farm rack) for four weeks.
- Elicitation: Apply a 1 μM flg22 solution directly to the leaves of test plants. Treat control plants with a water solution.
- Incubation: Allow the immune response to proceed for 60 minutes.
- Preservation: Immediately flash-freeze leaf tissue samples in liquid nitrogen. Alternatively, submerge tissue in a chemical fixative to halt all biological activity [12].
- Analysis:
- Transcriptomics: Extract RNA and perform RNA-Seq or RT-qPCR to analyze expression of defense marker genes (e.g., PR1, FRK1).
- Metabolomics: Analyze production of reactive oxygen species (ROS) and defense-related metabolites.
Visualization of Research Workflows
Technology Translation Pipeline
The following diagram illustrates the logical pathway for translating terrestrial CEA technologies into validated systems for space habitats.
Plant Immune Response Assay Workflow
This diagram outlines the experimental workflow for the immune challenge protocol described in section 3.2.
The Scientist's Toolkit: Key Research Reagents and Materials
The table below details essential materials and their functions for conducting experiments in space-analog vertical farming research.
Table 2: Key Research Reagents and Solutions for CEA-Space Translation Studies
| Reagent/Material | Function/Description | Application Example |
|---|---|---|
| flg22 Peptide | A 22-amino acid peptide derived from bacterial flagellin; a potent elicitor of plant pattern-triggered immunity. | Used to challenge the immune system of plants grown in CEA without introducing live pathogens [12]. |
| Clay-Based Growth Substrate | A porous, inorganic growth media used in "plant pillows" for spaceflight; provides anchor and controls water/air balance. | The substrate used in the ISS Veggie system; ideal for ground-analogue studies of root zone dynamics [12]. |
| LED Light Systems | Provides specific light spectra (e.g., red-blue magenta) for photosynthesis; can include far-red and infrared for research. | Standard for VF and space plant growth (e.g., Veggie, APH). Used to optimize light recipes for space crops [12] [71]. |
| RNAlater / RNA Stabilization Solution | A chemical fixative that rapidly permeates tissues to stabilize and protect cellular RNA. | Preserves the transcriptional state of plant samples at the exact moment of fixation for gene expression studies [12]. |
| Hydroponic Nutrient Solution | A balanced, water-soluble solution of essential macro and micronutrients for plant growth. | The lifeblood of recirculating systems in VF and BLSS; used to study nutrient use efficiency [73] [72]. |
| Random Positioning Machine (RPM) | A ground-based device that randomizes the gravity vector by continuous rotation, simulating microgravity. | Used to study the effects of altered gravity on plant growth, water distribution, and root architecture [43]. |
The design of higher plant compartments for space habitats is a critical cornerstone for enabling long-duration human exploration missions to the Moon and Mars. These systems are not merely food production modules; they are integral components of a Bioregenerative Life Support System (BLSS), where plants contribute to atmospheric regeneration, water purification, and waste recycling [2]. Evaluating their success requires a multifaceted approach, analyzing data on crop yield, resource efficiency, and direct crew health outcomes. Performance data from the International Space Station (ISS) provides the first critical validation of these technologies in a true microgravity environment, offering invaluable insights for future habitat design [2] [74]. This document synthesizes quantitative metrics and protocols from ISS missions to establish a benchmark for researchers and engineers.
Quantitative Performance Data from ISS Missions
Systematic data collection aboard the ISS has yielded key performance indicators for both plant growth systems and crew health. The following tables consolidate this quantitative data for easy comparison and analysis.
Table 1: Crop Production and Resource Recycling Performance on ISS
| Metric Category | Specific Parameter | Reported Value / Performance | Context & Mission |
|---|---|---|---|
| Water Recovery | Water Recycling Rate | ~90% efficiency [75] | ISS Water Recovery System (WRS) recycling urine and cabin moisture. |
| Oxygen Generation | Primary Method | Electrolysis of water [75] | ISS Oxygen Generation System. |
| Food Production | Example Crop | 'Outredgeous' red romaine lettuce [5] | Grown in the Advanced Plant Habitat (APH). |
| Growth Cycle | 28-day growth tests [5] | Standard for PH-07 experiment. | |
| Power Consumption | ISS Power Generation | 75-90 kilowatts [75] [76] | Supplied by solar arrays (2,500 m² area). |
| Crop Health Monitoring | Substrate Moisture | Controlled water stress levels [5] | PH-07 experiment investigating optimal watering strategies. |
Table 2: Standardized Crew Health and Performance Metrics
| Domain | Measurement Tool / Method | Key Findings from ISS Missions |
|---|---|---|
| Cognitive Performance | Cognition Test Battery (CTB) | No systematic decline in overall function, but reductions in processing speed, working memory, and sustained attention during flight [77]. |
| Behavioral Health | Sleep Logs & Actigraphy | Circadian desynchronization and reduced sleep duration are common [77]. |
| Personality (IPIP-NEO) | Astronaut profiles are generally low in neuroticism and high in agreeableness/conscientiousness [77]. | |
| Physical Health | Muscle Performance | Accelerated muscle and bone loss due to microgravity; countered by 2+ hours of daily exercise [78] [76]. |
| Neuro-ocular Structure | Changes in eye structure (lens, retina, optic nerve) are monitored [78]. | |
| Microbiome | Crew & Plant Microbiome Analysis | Studies on how microgravity affects the digestion system and plant-associated microbiomes [78] [5]. |
Detailed Experimental Protocols
To ensure reproducibility and standardized data collection for the research community, the following section outlines critical experimental protocols used in ISS missions.
Protocol: Plant Habitat-07 (PH-07) Experiment
Objective: To study the impacts of substrate moisture on the growth, development, and associated microbiome of 'Outredgeous' red romaine lettuce in microgravity [5].
Materials: Advanced Plant Habitat (APH) facility, science carrier quadrants, 'Outredgeous' lettuce seeds, porous tubes, substrate, moisture sensors.
Workflow:
- Pre-flight Ground Testing:
- Conduct multiple 28-day growth tests in environment chambers mimicking APH settings at Kennedy Space Center.
- Define and verify moisture conditions (e.g., flood, wilt) that impose desired growth differences for flight experiment [5].
- On-orbit Execution:
- Initiate three replicate 28-day growth tests in the APH on the ISS.
- Apply pre-defined substrate moisture treatments to different plant quadrants.
- Monitor plant growth and substrate moisture levels remotely.
- At the end of the growth cycle, astronauts harvest the plants, ensuring root recovery for post-flight analysis [5].
- Post-flight Analysis:
- Return plant samples on a cargo mission (e.g., CRS-32).
- Perform plant chemistry, food safety microbiology, microbiome analysis, and proteomics on returned samples [5].
Protocol: Spaceflight Standard Measures for Crew Health
Objective: To characterize how spaceflight affects human health and performance through a standardized, multidisciplinary set of measures, providing context for other experiments and monitoring human-system risks [77].
Materials: Cognition Test Battery (CTB) software, actiwatches, sleep logs, end-of-day surveys, sample collection kits for biochemical and cellular analysis.
Workflow:
- Pre-flight (Baseline): Data collection at 6 months and 3 months before launch. Includes CTB, personality inventory, medical imaging, and biological sample collection [77].
- In-flight (Monitoring):
- For standard-duration missions (105-240 days), data is collected during the first month, at mid-flight, and during the last month.
- Measures include CTB, actigraphy, sleep logs, end-of-day surveys, and collection of biological samples for cellular, biochemical, and microbiome analysis [77].
- Post-flight (Recovery): Data collection on the day of return, and then at 1 day, 1 week, and 1 month later to track recovery to baseline [77].
The following diagram illustrates the integrated data flow and decision-making process supported by the Spaceflight Standard Measures study.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Materials for Plant Experiments in Space
| Item | Function / Application |
|---|---|
| Advanced Plant Habitat (APH) | A fully automated, closed plant growth facility on the ISS providing controlled environmental conditions (light, temperature, humidity, CO2) for research [5]. |
| 'Outredgeous' Red Romaine Lettuce | A well-tested food crop used in multiple spaceflight experiments (e.g., PH-07) due to its known growth characteristics and as a model for food safety studies [5]. |
| Science Carrier Quadrants | The removable hardware that holds the plant growth substrate and seeds within the APH, allowing for different experimental treatments in a single unit [5]. |
| Porous Tubes & Moisture Sensors | Critical components for the controlled delivery of water to plant roots in microgravity and for monitoring substrate moisture levels in real-time [5]. |
| Substrate | The solid growth medium (e.g., clay particles) used to support plant roots and facilitate water and oxygen delivery in the absence of gravity [5]. |
Integrated System View: The Role of Plants in a BLSS
The ultimate goal of plant compartment research is its seamless integration into a Bioregenerative Life Support System (BLSS). In this closed-loop system, biological components work in concert to sustain human life.
This conceptual model, informed by ground-based demonstrators like MELiSSA and Lunar Palace, shows the interconnectedness of system compartments [2]. The producers (higher plants) are central, consuming crew-respired CO2 and waste-derived nutrients to produce oxygen, fresh food, and clean water. The performance data and protocols outlined in previous sections are essential for optimizing the higher plant compartment to fulfill its role reliably in this delicate balance, which is critical for missions beyond Earth orbit where resupply is not feasible [2].
For long-duration human space exploration missions beyond Earth's orbit, the development of robust Bioregenerative Life Support Systems (BLSS) transitions from a technical advantage to an absolute necessity [2]. Within these closed-loop systems, the higher plant compartment serves multiple critical functions: providing a sustainable food source, regenerating oxygen, purifying water, contributing to waste recycling, and offering psychological benefits for crew members during extended isolation [2] [7]. As mission planners target crewed missions to Mars within the 2040s, the ability to cultivate plants in space environments becomes fundamental to mission success by reducing absolute dependence on resupply from Earth [79] [57]. Current space-based plant systems, while demonstrating proof-of-concept, remain limited in scale and require significant advancement to support life support demands for deep space missions [2]. This document assesses the current Technology Readiness Levels (TRLs) of key plant growth technologies, identifies strategic capability gaps, and provides detailed experimental protocols to address the most pressing research questions in higher plant compartment design for space habitats.
Current Technology Readiness Assessment
The following table summarizes the maturity of various plant growth technologies relevant to deep space missions, based on their demonstrated capabilities in ground-based testing and spaceflight experiments.
Table 1: Technology Readiness Levels for Space-Based Plant Growth Systems
| Technology / System | Current TRL | Key Capabilities Demonstrated | Known Limitations & Gaps |
|---|---|---|---|
| Veggie (VEG) | TRL 8-9(Flight-Proven) | Successful cultivation of leafy greens, Chinese cabbage, and flowers on ISS [12]. Use of root pillows for water/nutrient distribution [12]. | Small scale; limited to leafy greens; significant crew time required; manual system [12]. |
| Advanced Plant Habitat (APH) | TRL 8(Flight-Proven) | Enclosed, automated environment with extensive sensor suite (180+ sensors) and environmental control [12]. | Higher system complexity; not yet scaled for significant food production [12]. |
| XROOTS | TRL 6-7(ISS Demo) | Testing hydroponic and aeroponic techniques for larger-scale production in microgravity [36]. | Technology demonstration ongoing; scaling and long-term reliability data pending [36]. |
| BLSS Ground Demonstrators | TRL 4-5(Ground-Tested) | Partial to full integration of plant compartments with other BLSS elements (e.g., MELiSSA, Lunar Palace) [2]. | Full closed-loop integration with human crews remains a challenge; impacts of space factors not fully tested [2]. |
| Plant Genetic Engineering | TRL 2-4(Concept/Research) | Early research into modifying plants for space (e.g., reduced lignin, enhanced nutrition) [12] [57]. | Regulatory and ethical considerations; unpredictable effects of space environment on engineered traits [57]. |
Strategic Gaps and Targeted Experimental Protocols
To advance the technologies listed in Table 1, targeted research must address specific physiological, technological, and systems-level gaps. The following section outlines key experimental protocols designed to generate critical data for closing these strategic gaps.
Protocol 1: Investigating Microgravity-Induced Hypoxia Stress and Root Zone Aeration
1.0 Objective: To evaluate the efficacy of different fluid delivery and root zone aeration techniques in mitigating microgravity-induced hypoxia stress in the root systems of candidate crop species.
2.0 Background: In microgravity, the absence of buoyancy-driven convection leads to the formation of anaerobic zones around plant roots, as fluid behavior is dominated by surface tension and capillary forces [36] [79]. This hypoxia stress can impair root function and plant health [79].
3.0 Materials:
- Growth Hardware: XROOTS-type platform or equivalent providing capabilities for hydroponic (flow, ebb) and aeroponic (mist, spray) delivery [36].
- Plant Materials: Dwarf tomato (Solanum lycopersicum 'Red Robin') and lettuce (Lactuca sativa 'Outredgeous').
- Sensor Suite: Dissolved oxygen (DO) sensors, pH sensors, and root imaging system integrated into the growth chambers.
- Fixation/Analysis: RNAlater or flash-freezing capability for transcriptomic analysis.
4.0 Methodology:
- Experimental Groups: Plants will be subjected to three different nutrient delivery systems: (a) Aeroponic (intermittent mist), (b) Hydroponic - Flow (continuous nutrient film), and (c) Hydroponic - Ebb & Flow (periodic flooding and draining).
- Culture Conditions: Photoperiod: 16h light/8h dark. Light intensity: 300 µmol m⁻² s⁻¹ PPF (spectrum: 90% red, 10% blue). Air temperature: 22 ± 2°C. Nutrient solution: Half-strength Hoagland's solution.
- Data Collection:
- Continuous: DO and pH levels in the root zone.
- Daily: Canopy imaging for health and growth assessment.
- Terminal (Day 35): Harvest and flash-freeze root and leaf samples for RNA sequencing (RNA-Seq) to analyze hypoxia-responsive gene expression (e.g., ADH, PDC, SUS) [7].
- Post-Harvest: Measure fresh and dry biomass, root architecture analysis.
5.0 Data Analysis: Compare plant growth metrics (biomass, root length), DO levels, and transcriptomic profiles across the three delivery systems. The system that maintains DO > 5 mg/L and shows minimal induction of hypoxia-responsive genes will be identified as the most effective.
The experimental workflow for this protocol is outlined below.
Protocol 2: Assessing Altered Gravity Effects on Plant Immune Function
1.0 Objective: To characterize the molecular and phenotypic response of plant immune pathways to simulated microgravity and Mars gravity, with and without pathogen-associated molecular pattern (PAMP) challenge.
2.0 Background: Spaceflight conditions may compromise plant immune systems. Anecdotal evidence from ISS (e.g., fungal growth on Zinnia) and gene expression studies suggest immune pathways are altered in microgravity [12]. Understanding this phenomenon is critical for plant health in BLSS.
3.0 Materials:
- Gravity Simulation: Random Positioning Machine (RPM) for simulated microgravity (µg); Centrifuge for Moon (0.16 g) and Mars (0.38 g) gravity.
- Plant Material: Arabidopsis thaliana (Col-0) and mutant lines (e.g., fls2, efr).
- Elicitors: Synthetic flg22 peptide solution (1 µM).
- Analysis Tools: Access to RNA-Seq, Microgravity Science Glovebox (MSG) equivalent for sterile handling, chemical fixatives.
4.0 Methodology:
- Growth and Gravitational Exposure:
- Grow Arabidopsis seedlings for 10 days on vertical agar plates under 1g conditions.
- Transfer plates to RPM (µg), Mars-g centrifuge (0.38g), or 1g control.
- Immune Challenge:
- After 24 hours of gravitational acclimation, treat half the seedlings in each group with flg22 solution and the other half with a mock (water) treatment [12].
- Sample Fixation:
- One hour post-elicitation, immerse all seedlings in RNA-later or flash-freeze in liquid nitrogen to "capture" the immune response state [12].
- Downstream Analysis:
- Transcriptomics: RNA-Seq on all samples to map global gene expression changes in response to gravity and immune challenge.
- Phenotypic Analysis: In a separate, non-fixed experiment, monitor ROS burst and callose deposition as quantitative measures of immune activation.
5.0 Data Analysis: Identify differentially expressed genes (DEGs) related to immune pathways (e.g., PRR expression, MAPK signaling, SA/JA phytohormone biosynthesis) across the gravity and treatment conditions. Correlate transcriptional changes with phenotypic outputs.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Research Reagents for Space Plant Biology Investigations
| Reagent / Material | Function / Rationale | Example Application |
|---|---|---|
| Clay-Based Growth Pillows | Provides a porous substrate for anchoring roots and distributing water, nutrients, and air in microgravity [12]. | Primary growth substrate in the Veggie system on ISS [12]. |
| flg22 Peptide | A 22-amino acid peptide derived from bacterial flagellin; a well-characterized PAMP used to artificially and safely activate plant immune responses [12]. | Used in BRIC-LED experiments to study immune system functionality in space [12]. |
| RNAlater / RNA Stabilization Solution | Chemically fixes biological samples, preserving the in vivo RNA expression profile at the moment of fixation without requiring immediate freezing [12]. | Essential for capturing accurate gene expression data in spaceflight experiments where immediate -80°C freezing is not always feasible [12]. |
| Porous Tube (XROOTS-like) Delivery System | A soilless nutrient delivery system that can be configured for hydroponic (liquid flow) or aeroponic (mist) delivery to roots in microgravity [36]. | Being tested in the XROOTS ISS investigation to scale plant production for space habitats [36]. |
| LED Light Arrays (Red-Blue Spectrum) | Provides the primary energy source for photosynthesis. The red-blue spectrum is highly efficient for plant growth, though white, far-red, and infrared are used in APH for advanced research [12]. | Standard for Veggie and APH units; allows for precise control over photoperiod and light quality to optimize plant growth and development [12]. |
Future Investment and Development Roadmap
Bridging the strategic gap from current TRLs to mission-ready systems requires a prioritized investment strategy. The following roadmap outlines critical development areas.
Table 3: Future Investment & Development Roadmap
| Development Area | Short-Term (1-5 years) | Medium-Term (5-10 years) | Long-Term (10+ years) |
|---|---|---|---|
| System Scaling & Automation | Automate APH-like systems for crew-independent operation. Scale XROOTS results to "Astro Garden" prototype [36]. | Integrate multiple scaled plant growth units into a functional subsystem of a habitat. | Fully autonomous BLSS plant compartment for Mars transit and surface habitats. |
| Crop Selection & Optimization | Focus on fast-growing, high-nutrition leafy greens and microgreens for dietary supplement [2]. | Introduce and validate dwarf fruiting crops (tomato, pepper) and carbohydrate sources (dwarf wheat, potato) [2]. | Engineer multi-purpose crops for optimal nutrition, resource recycling, and on-demand biomaterial production [57]. |
| BLSS Integration | Ground-based testing of plant compartment integration with air and water recovery systems [2]. | Human-in-the-loop testing in ground-based demonstrators (e.g., Lunar Palace, MELiSSA) [2]. | In-situ testing of integrated BLSS on Lunar surface as a testbed for Mars missions [2]. |
| Plant Health & Adaptation | Characterize molecular basis of immune suppression and hypoxia stress in microgravity [12] [7]. | Develop countermeasures (e.g., microbial inoculants, optimized growth protocols) to mitigate space-specific plant stresses. | Deploy genetically optimized plants tailored for the Martian and Lunar environments [57]. |
The logical relationship and progression of these development areas is visualized below.
The path to sustainable deep space exploration is inextricably linked to our ability to reliably cultivate higher plants in the space environment. While significant progress has been made, as evidenced by the flight-proven Veggie and APH systems, a strategic gap remains in scaling these technologies, integrating them into regenerative life support loops, and fully understanding plant biology in altered gravity and radiation environments [12] [2]. Closing this gap requires a disciplined, multi-disciplinary approach that combines fundamental plant biology research, focused technology development, and rigorous systems integration testing. The experimental protocols and investment roadmap detailed herein provide a structured path forward. By systematically addressing these priorities, the global research community can deliver the necessary tools and knowledge to make Bioregenerative Life Support Systems a reality, thereby enabling humanity's long-term presence beyond Earth.
Conclusion
The design of higher plant compartments is a critical, cross-disciplinary endeavor essential for the future of long-duration human space exploration. Success hinges on integrating fundamental plant biology research with advanced engineering to create robust, automated BLSS that reliably provide food, regenerate atmosphere and water, and offer psychological benefits. Current flight projects demonstrate feasibility, but strategic investment is urgently needed to close technological gaps and achieve the resilience required for Mars missions. Research into plant responses to combined space stressors must intensify. Furthermore, the technologies and protocols developed—particularly in controlled environment agriculture, resource recycling, and automated monitoring—have profound implications for improving sustainable agriculture and enhancing food security on Earth, creating a synergistic feedback loop between space exploration and terrestrial innovation.
References
- 1. Bioregenerative life-support systems [https://www.sciencedirect.com/science/article/pii/S0002916523185297]
- 2. Plant and microbial science and technology as ... [https://www.nature.com/articles/s41526-023-00317-9]
- 3. Development of Nitrogen Recycling Strategies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8548772/]
- 4. Design and establishment of a large-scale controlled ... [https://www.sciencedirect.com/science/article/abs/pii/S2214552421000602]
- 5. Sierra Space's Contribution to the Plant Habitat-07 ... [https://www.sierraspace.com/blog/celebrating-human-spaceflight-sierra-spaces-contribution-to-the-plant-habitat-07-experiment/]
- 6. Microbial applications for sustainable space exploration ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10284894/]
- 7. Perspectives for plant biology in space and analogue ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10442387/]
- 8. Space-driven ROS in cells: a hidden danger to astronaut ... [https://www.nature.com/articles/s41526-025-00492-x]
- 9. Offworld Plant Biology: ARTEMOSS Experiment Launches to ... [https://astrobiology.com/2025/01/offworld-plant-biology-artemoss-experiment-launches-to-the-international-space-station.html]
- 10. E.9 Space Biology Advanced Plant Habitat No Longer ... [https://science.nasa.gov/researchers/solicitations/roses-2024/amendment-121-e-9-space-biology-advanced-plant-habitat-no-longer-available-for-proposed-iss-studies/]
- 11. Advanced Plant Habitat [https://science.nasa.gov/mission/advanced-plant-habitat/]
- 12. Growing Plants in Space [https://www.nasa.gov/exploration-research-and-technology/growing-plants-in-space/]
- 13. Extraterrestrial Habitats: Bioplastics for Life Beyond Earth [https://seas.harvard.edu/news/2025/07/extraterrestrial-habitats-bioplastics-life-beyond-earth]
- 14. Perspectives for plant biology in space and analogue ... [https://www.nature.com/articles/s41526-023-00315-x]
- 15. Biology and crop production in Space environments [https://www.sciencedirect.com/science/article/abs/pii/S2214552421000183]
- 16. Evolutionary genetics of plant adaptation - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3123387/]
- 17. eXposed Root On-Orbit Test System (XROOTS) [https://www.sierraspace.com/blog/sustainability-in-space-xroots-environmental-system/]
- 18. Hardware Validation of the Advanced Plant Habitat on ISS [https://pmc.ncbi.nlm.nih.gov/articles/PMC7314936/]
- 19. eXposed Root On-Orbit Test System (XROOTS) Tech Demo [https://science.nasa.gov/biological-physical/investigations/xroots/]
- 20. XROOTS ISS Tech Demo of Aeroponics and Hydroponics ... [https://ttu-ir.tdl.org/items/4449167d-d919-4710-87a5-48c02f7239d3]
- 21. Hydroponics for plant cultivation in space – a white paper [https://www.sciencedirect.com/science/article/pii/S221455242400066X]
- 22. The physiology of plants in the context of space exploration [https://pmc.ncbi.nlm.nih.gov/articles/PMC11470014/]
- 23. Mission Report: 19 August 2025 - Space Botany [https://spacebotany.uk/blog/mission-report-19-august-2025/]
- 24. Mission Cosmic Crops: Combat Hunger With Space ... [https://www.sciencefriday.com/educational-resources/crops-combat-hunger/]
- 25. Space farming [https://grokipedia.com/page/Space_farming]
- 26. Space farming: Horticulture systems on spacecraft and ... [https://www.sciencedirect.com/science/article/abs/pii/S0981942822005605]
- 27. Optimizing LED lighting spectra for enhanced growth in ... [https://www.nature.com/articles/s41598-025-15352-7]
- 28. Humidity and temperature control in greenhouses [https://www.processsensing.com/en-us/blog/humidity-temperature-control-green-houses.htm]
- 29. Providing Controlled Environments for Plant Growth in Space [https://pubmed.ncbi.nlm.nih.gov/11541567/]
- 30. Mastering Humidity Control Systems for Greenhouses [https://www.smartfog.com/humidity-control-systems-for-greenhouses-a-complete-guide-to-optimal-plant-growth/]
- 31. A Guide to Greenhouse Climate Control for Growers [https://fogco.com/using-a-humidity-controller-for-greenhouse-climate-control/]
- 32. Space Crops - NASA Science Biological & Physical ... [https://science.nasa.gov/biological-physical/space-crops/]
- 33. Dwarf Tomatoes Save Space | ILRiverHort - Illinois Extension [https://extension.illinois.edu/blogs/ilriverhort/2017-05-12-dwarf-tomatoes-save-space]
- 34. Antioxidant Profile of Pepper (Capsicum annuum L.) Fruits ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7554748/]
- 35. Bioactive characteristics and antioxidant activities of nine ... [https://www.sciencedirect.com/science/article/pii/S1756464612000023]
- 36. A Novel Approach to Growing Gardens in Space [https://science.nasa.gov/science-research/science-enabling-technology/technology-highlights/a-novel-approach-to-growing-gardens-in-space/]
- 37. Plant Growth Systems | Space Station Microgravity & Tech [https://www.sierraspace.com/space-technology/microgravity-environmental-systems/space-station-payloads/]
- 38. AutoNav [https://www.epiroc.com/en-om/products/digital-solutions/agnostic-automation/auto-nav]
- 39. How to Leverage Remote Video Monitoring for Industrial ... [https://blog.isa.org/remote-video-monitoring-industrial-scada-hmi-systems]
- 40. Design and ground testing of a Zero-Discharge plant ... [https://www.sciencedirect.com/science/article/abs/pii/S0168169925011500]
- 41. Capillary driven fluid flows in microgravity [https://www.sciencedirect.com/science/article/abs/pii/S0094576522005872]
- 42. Role and Impact of Microgravity on Plant Growth, Lignin ... [https://nhsjs.com/2025/role-and-impact-of-microgravity-on-plant-growth-lignin-content-and-plant-nutrition/]
- 43. Exploring plant responses to altered gravity for advancing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12177493/]
- 44. Root growth direction in simulated microgravity is modulated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9327515/]
- 45. Simulated microgravity and the antagonistic influence of ... [https://www.nature.com/articles/s41526-018-0054-z]
- 46. Research on a Microgravity-adapted Controlled-Release ... [https://www.sciencedirect.com/science/article/abs/pii/S2214552425001038]
- 47. The physiology of plants in the context of space exploration [https://www.nature.com/articles/s42003-024-06989-7]
- 48. Eustress in Space: Opportunities for Plant Stressors ... [https://www.frontiersin.org/journals/astronomy-and-space-sciences/articles/10.3389/fspas.2022.841211/full]
- 49. Cell Wall Modifications in Maize Pulvini in Response to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3149947/]
- 50. Comparative transcriptomics indicate changes in cell wall ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5821596/]
- 51. Recent transcriptomic studies to elucidate the plant adaptive ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9287483/]
- 52. Space Radiation Protection Countermeasures in Microgravity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8398261/]
- 53. Radiation environment in exploration-class space missions ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2022.1001158/full]
- 54. Feeding the cosmos: tackling personalized space nutrition ... [https://www.nature.com/articles/s41526-025-00490-z]
- 55. Space-driven ROS in cells: a hidden danger to astronaut ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12322194/]
- 56. Station Science 101: Plant Research [https://www.nasa.gov/missions/station/ways-the-international-space-station-helps-us-study-plant-growth-in-space/]
- 57. SpaceHort: redesigning plants to support space ... [https://www.sciencedirect.com/science/article/pii/S0958166921001646]
- 58. Pesticide immunotoxicity on insects – Are agroecosystems ... [https://www.sciencedirect.com/science/article/pii/S0048969724056171]
- 59. Insect Pathogenic Bacteria in Integrated Pest Management [https://pmc.ncbi.nlm.nih.gov/articles/PMC4553484/]
- 60. Plant Pathology: Techniques and Protocols [https://link.springer.com/book/10.1007/978-1-4939-2620-6]
- 61. Space Biology Advanced Plant Habitat No Longer Available ... [https://astrobiology.com/2025/04/space-biology-advanced-plant-habitat-no-longer-available-for-proposed-iss-studies.html]
- 62. Nutrient Loop Closure → Term [https://climate.sustainability-directory.com/term/nutrient-loop-closure/]
- 63. Closing the Nutrient Loop—The New Approaches to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9915181/]
- 64. Critical investments in bioregenerative life support systems for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12357894/]
- 65. Closed Ecosystems [https://nss.org/settlement/nasa/teacher/lessons/bryan/ecosys/]
- 66. Critical investments in bioregenerative life support systems ... [https://www.nature.com/articles/s41526-025-00518-4]
- 67. ESA proposes new programme to streamline support for ... [https://business.esa.int/news/esa-proposes-new-programme-to-streamline-support-for-european-business]
- 68. NASA's lag in BLiSS technology threatens US space ... [https://www.linkedin.com/posts/d-marshall-porterfield-b19a1929_critical-investments-in-bioregenerative-life-activity-7362529980372692992-rYdA]
- 69. and the Future of Space Exploration Vertical Farming [https://green.org/2024/01/30/vertical-farming-and-the-future-of-space-exploration/]
- 70. Controlled Environment Agriculture [https://caes.ucdavis.edu/research/initiative/controlled-environment-agriculture]
- 71. Vertical farming: productivity, environmental impact, and ... [https://link.springer.com/article/10.1007/s13593-025-01055-w]
- 72. 10 Key Benefits of Vertical Farming In 2025 [https://agtech.folio3.com/blogs/10-powerful-benefits-of-vertical-farming/]
- 73. Nitrogen accountancy in space agriculture | npj Microgravity [https://www.nature.com/articles/s41526-024-00428-x]
- 74. Exploring plant responses to altered gravity for advancing ... [https://www.sciencedirect.com/science/article/pii/S2590346225001324]
- 75. Management of Consumable Resources on... | New Space Economy [https://newspaceeconomy.ca/2024/02/01/management-of-consumable-resources-on-the-international-space-station-iss/]
- 76. and Facts (2025) International Space Station Statistics [https://www.sci-tech-today.com/stats/international-space-station-statistics/]
- 77. Spaceflight Standard Measures is a multidisciplinary study ... [https://www.nature.com/articles/s41526-025-00532-6]
- 78. Cutting Edge Medical Studies Look at Crew Fitness and ... [https://www.nasa.gov/blogs/spacestation/2025/09/24/cutting-edge-medical-studies-look-at-crew-fitness-and-vision/]
- 79. How growing plants in space can help us on Earth [https://www.uwa.edu.au/news/article/2025/august/how-growing-plants-in-space-can-help-us-on-earth]