Computational Modeling of Turing Patterns in Plants: From Morphogenesis to Biomedical Insights
This article synthesizes current research and computational methodologies for modeling Turing pattern formation in plant systems.
Computational Modeling of Turing Patterns in Plants: From Morphogenesis to Biomedical Insights
Abstract
This article synthesizes current research and computational methodologies for modeling Turing pattern formation in plant systems. It explores the foundational theory of reaction-diffusion systems and their application across biological scales, from intracellular ROP protein patterning to whole-plant phyllotaxis and vegetation landscapes. For a research audience, it details practical modeling frameworks, addresses common troubleshooting and parameterization challenges, and provides rigorous validation and comparative analysis against alternative patterning mechanisms. The review highlights how plant models serve as accessible paradigms for understanding self-organization, with direct implications for pattern dysregulation in biomedical contexts such as tissue morphogenesis and regenerative medicine.
The Turing Principle in Botany: Revisiting Reaction-Diffusion Theory and Its Plant-Based Origins
In his seminal 1952 paper, "The Chemical Basis of Morphogenesis," English mathematician Alan Turing proposed a revolutionary mechanism for biological pattern formation [1]. He demonstrated how two diffusible substances, termed morphogens, could interact to spontaneously generate periodic spatial patterns from an initially homogeneous state—a process he called "diffusion-driven instability" (DDI) [2]. This was profoundly counter-intuitive, as diffusion is typically understood as a homogenizing process, yet Turing showed that when two chemicals with different diffusion rates interact—one as a self-activating activator and the other as an inhibitory inhibitor—diffusion can destabilize a uniform equilibrium and drive the emergence of structured patterns [1] [3]. This theoretical framework provides a potential explanation for a vast array of patterns observed in nature, from the stripes of a zebrafish to the spacing of leaves on a plant stem.
Core Theoretical Principles
Mathematical Foundations of Diffusion-Driven Instability
Turing's analysis considers a system of two reacting and diffusing morphogens, with concentrations (u) and (v). The system is described by partial differential equations of the form [2]: [ \frac{\partial u}{\partial t} = Du \nabla^2 u + f(u,v) ] [ \frac{\partial v}{\partial t} = Dv \nabla^2 v + g(u,v) ] where (Du) and (Dv) are diffusion coefficients, and (f) and (g) specify the reaction kinetics. The stability of the homogeneous steady state ((u0, v0)) is analyzed by considering perturbations. For pattern formation via DDI, the following conditions must be met [2]:
- Stability without diffusion: The homogeneous steady state is stable to small, spatially homogeneous perturbations.
- Instability with diffusion: The same steady state becomes unstable when diffusion is introduced, with certain spatial wavelengths growing exponentially.
This requires that the inhibitor diffuses significantly faster than the activator ((Dv > Du)) [1] [2]. The resulting patterns depend on domain size, boundary conditions, and the specific parameters of the reaction kinetics.
The Local Activation-Lateral Inhibition Framework
A popular biological interpretation of Turing's model is the Local Activation-Lateral Inhibition (LALI) scheme formalized by Gierer and Meinhardt [1]. In this framework:
- A short-range self-activating activator promotes its own production and that of a long-range inhibitor.
- A long-range inhibitor suppresses the activator.
- The differential diffusivity ensures that activation remains local, while inhibition acts over a longer range, preventing activator expansion everywhere and leading to the formation of isolated peaks or stripes [1] [4].
Table 1: Classic Turing-Type Reaction Kinetics
| Kinetic Model | Reaction Functions | Key Characteristics |
|---|---|---|
| Schnakenberg [2] | ( f(u,v) = a - u + u^2v ) ( g(u,v) = b - u^2v ) | Based on a simple tri-molecular reaction; often used as a canonical example. |
| Gierer-Meinhardt [2] | ( f(u,v) = a - bu + \frac{u^2}{v(1 + ku^2)} ) ( g(u,v) = u^2 - v ) | Explicitly models activator-inhibitor dynamics; widely applied in biological modeling. |
| Thomas [2] | ( f(u,v) = a - u - \frac{\rho uv}{1+u+Ku^2} ) ( g(u,v) = \alpha(b - v) - \frac{\rho uv}{1+u+Ku^2} ) | Derived from a specific enzymatic reaction system. |
Diagram 1: Core Turing (LALI) principle.
Computational Modeling of Turing Systems
Implementation and Analysis Protocols
Computational models are essential for studying Turing systems, allowing researchers to integrate processes across spatial and temporal scales to investigate developmental outcomes [5].
Protocol 1: Simulating a Two-Component Turing System
- Define the Model Geometry: Specify a 1D, 2D, or 3D domain. For plant tissue modeling, a 2D rectangular or circular domain is often appropriate.
- Set Reaction Kinetics and Parameters: Choose a kinetic model (e.g., from Table 1) and initial parameters. The Schnakenberg model is a common starting point for its simplicity.
- Specify Initial and Boundary Conditions:
- Initial Condition: Set the homogeneous steady state ((u0, v0)) with a small random perturbation applied to every grid point to break symmetry.
- Boundary Conditions: Use zero-flux (Neumann) boundary conditions to simulate an isolated tissue, or periodic boundaries for a repetitive field.
- Numerical Solution: Discretize the domain and solve the coupled reaction-diffusion equations using a finite difference method. An explicit Euler method is simple to implement but may require small time steps for stability.
- Analysis: Run the simulation until the pattern stabilizes. Analyze the final pattern's wavelength and morphology (e.g., spots, stripes). Perform a linear stability analysis on the chosen parameters to predict the expected wavelength and compare it with the simulation output.
Accounting for Biological Complexity: Multi-Component and Mechanistic Models
While classical two-component models are insightful, real biological systems are more complex. Modern computational approaches extend the theory in several ways [6]:
- Multi-Component Systems: Turing instability can emerge from networks with three or more components, even without a single, classical self-activating component. Positive feedback loops between multiple components can generate patterns [4].
- Incorporating Cellular Structures: In plants, the polar transport of the hormone auxin via PIN-FORMED (PIN) proteins can be modeled as an advection-diffusion process. This mechanism can generate periodic organ initiation (phyllotaxis) and has been compared to a Turing system where PIN polarization creates an inhibitory field [4].
- Integrating Tissue Growth: Domain growth is a critical factor for robust patterning. Logistic growth, for instance, can reliably lead to pattern doubling (e.g., new stripes forming between old ones as a fish grows) and can buffer against variations in final domain size [2].
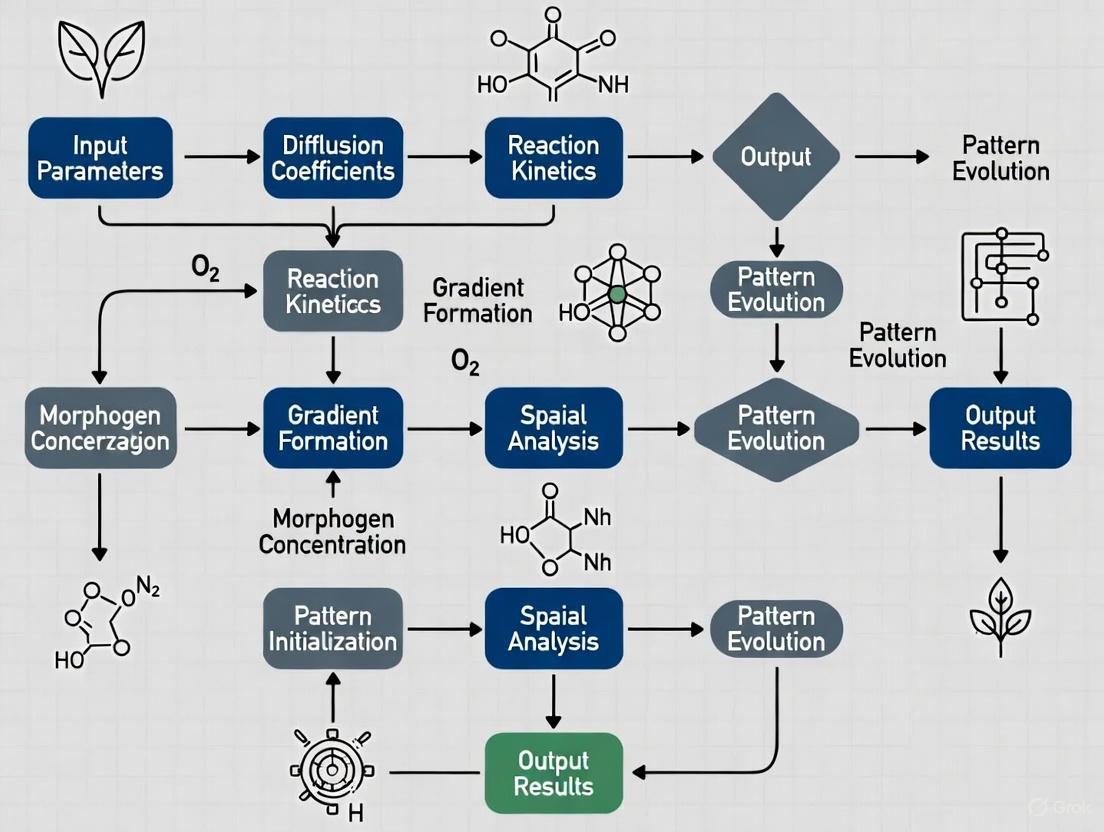
Diagram 2: Computational modeling workflow.
Experimental Validation in Model Systems
Protocol: Validating Turing Patterning in Chia Seedlings
Recent work has provided direct experimental validation of Turing's model in a living plant system using chia seeds [7]. This protocol outlines the key steps.
Table 2: Research Reagent Solutions for Chia Patterning Experiment
| Item | Function/Description | Role in Turing Mechanism |
|---|---|---|
| Chia Seeds (Salvia hispanica) | Model plant organism; forms a near-uniform 2D lawn of seedlings. | Provides a homogeneous biological "domain" for pattern emergence. |
| Hydration Trays | Shallow, flat trays to grow seeds in a controlled, 2D geometry. | Defines the physical boundary and scale of the reaction-diffusion system. |
| Water/Moisture | Critical resource for plant growth and survival. | Acts as the depleted substrate; competition for water drives the instability [4] [7]. |
| Controlled Environment | Growth chamber to regulate light, temperature, and humidity. | Isolates the Turing mechanism (resource competition) from confounding environmental noise. |
Procedure:
- Preparation: Fill multiple shallow trays with a standard, uniform growth medium.
- Sowing: Sow chia seeds evenly and densely across the surface of each tray to create a homogeneous initial state—a uniform lawn of seedlings.
- Parameter Manipulation: This is the core of the experiment. Adjust the "moisture availability" parameter across different trays. This directly corresponds to tweaking a key parameter in the underlying Turing equations that models the competition for a critical resource [7].
- Incubation and Monitoring: Place trays in a controlled growth environment. Monitor the emergence of spatial patterns in plant density and vitality over time.
- Data Collection and Comparison:
- Photograph the patterns in each tray at regular intervals.
- Quantify the pattern wavelength (average distance between dense vegetation patches) and morphology (gaps, labyrinths, spots).
- Compare the experimental results directly with computer simulations of the Turing model run with parameters corresponding to the different moisture levels.
Expected Outcome: Under appropriate moisture stress, the initially homogeneous lawn of chia seedlings will self-organize into intricate, regular patterns of vegetation and bare ground that closely resemble the patterns produced by computer simulations of the Turing model [7]. This experiment demonstrates that the principles of diffusion-driven instability can underlie pattern formation in living vegetation.
Diagram 3: Chia experiment validation workflow.
Applications in Plant Developmental Biology
Turing's theory provides a powerful framework for understanding a wide range of periodic patterns in plant development, operating across different spatial scales.
Case Study 1: Epidermal Patterning in Arabidopsis Roots
The patterning of non-hair cells in the Arabidopsis root epidermis follows a striped, hair-hair-nonhair sequence, which can be explained by a Turing mechanism.
Key Components:
- Activator: A transcription factor complex (e.g., WEREWOLF, GLABRA3) promotes non-hair cell fate in a cell-autonomous manner.
- Inhibitor: The small peptide SIGNAL (or a related molecule) moves laterally through the cell walls, inhibiting non-hair fate in neighboring cells.
- Differential Diffusivity: The peptide inhibitor diffuses more readily than the transcription factor complex, creating the short-range activation and long-range inhibition required for a striped pattern.
Case Study 2: ROP Protein Patterning within Single Cells
Inside single plant cells, Rho-of-Plants (ROP) GTPases can form Turing patterns to determine cell shape [4].
Mechanism:
- ROPs exist in an active (membrane-bound) state and an inactive (cytosolic) state.
- Active ROPs promote their own activation (self-amplification) and recruit inhibitors.
- The inhibitor (e.g., a GAP) has a higher effective diffusion rate in the cytosol than the membrane-bound active ROP.
- This system can generate multiple stable clusters of active ROP, which drive the formation of lobes in puzzle-shaped leaf epidermal cells or the spaced secondary cell wall thickenings in xylem cells [4].
Table 3: Turing Patterns Across Scales in Plants
| Scale | System | Putative Activator/Initiator | Putative Inhibitor/Substrate | Resulting Pattern |
|---|---|---|---|---|
| Intracellular | ROP GTPases [4] | Membrane-bound active ROP (slow diffusion) | Cytosolic ROP inhibitor (fast diffusion) | Multiple ROP clusters; puzzle-shaped cells, xylem wall thickenings. |
| Multicellular | Root Epidermis | Transcription factor complex (cell-autonomous) | Mobile peptide signal (lateral diffusion) | Striped pattern of hair and non-hair cell files. |
| Tissue/Organ | Phyllotaxis (Organ spacing) [4] | Auxin accumulation in incipient primordia | Depletion of auxin from surrounding area via PIN transporters | Regular spacing of leaves, branches, and flowers. |
| Organism/ Population | Dryland Vegetation [4] [7] | Vegetation patch (improves local water infiltration) | Water (depleted substrate) consumed by plants | Regular patterns of vegetation patches (spots, labyrinths, stripes). |
Advanced Concepts and Current Research Frontiers
Robustness and the Impact of Noise and Growth
A significant challenge for Turing models in biology is ensuring robustness—the reliable reproduction of patterns despite stochastic fluctuations and domain growth [2].
- Stochastic Effects: Intrinsic noise from low molecular copy numbers can excite pattern modes outside the deterministic Turing space. Analysis using the Chemical Master Equation and Fokker-Planck equations shows that the stochastically excited modes can align with deterministic predictions, but noise can also lead to pattern variability [2].
- Domain Growth: As noted in the protocols, growth is not an obstacle but can be a key factor for robustness. Certain growth laws (e.g., logistic growth) can reliably select a specific pattern mode and facilitate pattern doubling, ensuring a consistent pattern element size even as the organism grows [2].
Beyond Classic Morphogens: Mechanical Patterning
Emerging research highlights that mechanical stresses within tissues can also propagate signals and participate in pattern formation, sometimes independently of classical diffusing morphogens [4] [3]. For example, the regular patterning of feather and hair-follicle precursors in animal skins may arise through the self-aggregation of mesenchymal cells, dependent on their mechanical properties and the stiffness of the extracellular environment [1]. This suggests that mechanical cues can work in tandem with, or independently from, reaction-diffusion systems to generate biological patterns.
The concept of short-range activation and long-range inhibition forms the cornerstone of Alan Turing's reaction-diffusion theory of morphogenesis, first proposed in 1952 [8]. This mechanism explains how seemingly identical cells can self-organize into complex, periodic patterns without pre-existing positional information. In biological systems, this theoretical framework manifests through molecular interactions where a slowly diffusing activator promotes its own production while simultaneously activating a rapidly diffusing inhibitor that suppresses the activator [8] [9]. The spatial difference in diffusion rates creates instability in the homogeneous state, leading to the spontaneous emergence of patterns. The critical wavelength of these patterns depends on the diffusion coefficients and kinetic parameters of the interacting molecules, scaling with the square root of the product of the diffusion coefficients [8]. In plants, this mechanism underpins diverse developmental processes from root hair patterning to organ spacing and epidermal differentiation.
Quantitative Parameters for Turing Systems
Table 1: Key parameters governing Turing pattern formation
| Parameter | Mathematical Relation | Biological Interpretation | Typical Values/Examples |
|---|---|---|---|
| Diffusion Coefficient Ratio | ( D{\text{inhibitor}} / D{\text{activator}} ) | Ratio of diffusion rates between inhibitor and activator | >1 (Often 10-100x difference) [8] |
| Critical Wavelength (( l_c )) | ( lc \sim \sqrt{D1\cdot D2/(c1D1+c2D_2)} ) [8] | Characteristic spacing between pattern elements | Cell-to-tissue scale (µm to mm) [8] |
| Activation Range | Local (1-few cell diameters) | Spatial extent of self-activating signal | Determined by slow diffusion [8] [9] |
| Inhibition Range | Long (multiple cell diameters) | Spatial extent of inhibitory signal | Determined by fast diffusion or directed transport [8] |
Experimental Protocols for Investigating Turing Patterns in Plants
Protocol: Computational Identification of Turing Parameters
Objective: To identify parameter regions capable of generating Turing patterns in a reaction-diffusion system.
Materials:
- Software: PDE simulation environment (e.g., MATLAB, Python with FENICS)
- Model Definition: Reaction-diffusion equations with appropriate boundary conditions
- Parameter Space: Biologically plausible ranges for kinetic parameters and diffusion coefficients
Methodology:
- Define the reaction-diffusion system using partial differential equations: ( \frac{\partial a}{\partial t} = F(a,h) + Da\nabla^2a ) ( \frac{\partial h}{\partial t} = G(a,h) + Dh\nabla^2h ) where ( a ) and ( h ) represent activator and inhibitor concentrations, ( F ) and ( G ) their reaction kinetics, and ( Da ), ( Dh ) their diffusion coefficients [8] [9].
Establish homogeneous steady state by solving ( F(a0,h0) = 0 ) and ( G(a0,h0) = 0 ).
Perform linear stability analysis by calculating the Jacobian matrix of the reaction terms at the homogeneous steady state.
Identify Turing space by searching for parameters where:
- The homogeneous state is stable without diffusion (all eigenvalues have negative real parts)
- The homogeneous state becomes unstable with diffusion (at least one eigenvalue has positive real part for some wavenumber k ≠ 0) [9]
Determine critical wavelength from the wavenumber exhibiting maximum growth rate using dispersion relation analysis.
Validate with numerical simulation of the full nonlinear system on a 2D domain to observe emergent patterns.
Expected Outcomes: Identification of parameter regions supporting spot, stripe, or labyrinthine patterns; determination of pattern wavelength as a function of kinetic parameters and domain size.
Figure 1: Computational workflow for identifying Turing parameters
Protocol: Experimental Validation in Root Development
Objective: To experimentally verify Turing patterning mechanisms in plant root development.
Materials:
- Plant Materials: Arabidopsis thaliana wild-type and mutant lines
- Molecular Biology Reagents: GFP reporter constructs, hormone biosynthesis inhibitors
- Imaging Equipment: Confocal laser scanning microscope with time-lapse capability
- Image Analysis Software: Fiji/ImageJ with custom macros for pattern quantification
Methodology:
- Generate transcriptional reporters for candidate activator and inhibitor molecules by fusing promoters to fluorescent proteins (e.g., pActivator:GFP, pInhibitor:RFP).
Establish observation system using vertical plate growth with controlled environmental conditions for live imaging of root development.
Perform time-lapse imaging of reporter expression patterns at 6-12 hour intervals over 3-5 days of root development.
Quantify expression patterns by measuring:
- Spatial correlation between putative activator and inhibitor expression domains
- Expression domain periodicity using Fast Fourier Transform (FFT) analysis
- Expression dynamics following laser ablation of specific cell files
Test perturbation responses through:
- Hormone/signaling inhibitor applications at varying concentrations
- Genetic perturbations (mutants, overexpression lines)
- Physical constraints on root growth to alter domain size
Compare with model predictions by quantifying whether experimental observations match critical wavelength predictions and parameter sensitivities of the computational model.
Expected Outcomes: Spatial correlation between predicted and observed expression patterns; altered patterning under perturbation consistent with model predictions; quantitative match between observed and simulated pattern wavelengths.
Research Reagent Solutions
Table 2: Essential research reagents for investigating Turing patterns
| Reagent Category | Specific Examples | Research Application | Key Function |
|---|---|---|---|
| Fluorescent Reporters | GFP, RFP, YFP transcriptional fusions | Live imaging of pattern dynamics | Visualize spatial distribution of putative morphogens [5] |
| Pharmacological Inhibitors | Auxin transport inhibitors (NPA), biosynthesis inhibitors | Perturb reaction-diffusion systems | Test model predictions by disrupting specific interactions [5] |
| Genetic Tools | CRISPR/Cas9 mutants, inducible overexpression lines | Manipulate specific network components | Alter kinetic parameters of putative activator/inhibitor systems [9] |
| Computational Frameworks | Reaction-diffusion PDE solvers, custom MATLAB/Python code | Simulate and test patterning models | Quantitative prediction of pattern formation under various parameters [5] [9] |
Advanced Concepts and Alternative Mechanisms
While the classical activator-inhibitor model remains influential, recent research has revealed that Turing patterns can emerge from surprisingly simple biochemical systems. Regulated degradation pathways can generate patterning without imposed feedback loops, with the simplest pattern-enabling reaction requiring only trimer formation via sequential binding and altered degradation rate constants of monomers upon binding [9]. This expansion beyond classical models is significant as it suggests that thousands of proteins and RNAs with appropriate binding and degradation characteristics could potentially form Turing patterns [9].
In plants, the PIN/AUXIN module represents a specialized implementation where polar auxin transport creates inhibitory fields around primordia, effectively functioning as a long-range inhibitor despite not fitting the classical diffusion-based model [8]. This mechanism demonstrates how directed transport can substitute for differential diffusion in establishing the necessary short-range activation and long-range inhibition.
Figure 2: Core Turing patterning mechanism
The critical wavelength emerging from these systems demonstrates how pattern periodicity depends on both molecular and physical parameters. In addition to diffusion coefficients, parameters without inherent length dimensions can significantly impact pattern wavelength, particularly when the homogeneous equilibrium approaches instability against spatially homogeneous perturbations [8]. This theoretical insight explains how biological systems can generate patterns at multiple spatial scales using similar molecular machinery.
Application Note: Turing-Wardlaw Collaboration on Phyllotaxis
Intellectual Exchange and Historical Significance
The collaboration between Alan Turing and botanist C.W. Wardlaw represents a foundational intersection of mathematical biology and plant sciences during the early 1950s. Following the publication of his seminal 1952 paper "The Chemical Basis of Morphogenesis," Turing actively sought biological applications for his reaction-diffusion theory and found a receptive colleague in Wardlaw [4] [8]. Their dialogue specifically addressed the long-standing puzzle of phyllotaxis—the regular arrangement of lateral organs around a central axis in plants [10]. This exchange was tragically cut short by Turing's death in 1954, leaving much of his subsequent work on phyllotaxis as unpublished fragments [11] [12]. Wardlaw subsequently published commentaries that helped disseminate and interpret Turing's ideas for the botanical community, notably in his 1953 article "A Commentary on Turing's Diffusion-Reaction Theory of Morphogenesis" [12].
Core Theoretical Framework and Modern Validation
Turing's fundamental insight was that diffusion, traditionally understood as a homogenizing process, could spontaneously generate regular patterns through a reaction-diffusion system [4] [8]. This mechanism requires an activator-inhibitor pair with significantly different diffusion coefficients, creating the principle of short-range facilitation and long-range inhibition [4] [8]. While Turing's initial models provided a conceptual breakthrough, contemporary research has revealed that phyllotaxis involves more complex mechanisms than simple reaction-diffusion, incorporating directed transport of the plant hormone auxin via dynamically positioned PIN proteins [4] [13] [8]. Modern computational models confirm that Turing-like instabilities, when integrated with auxin transport dynamics, can indeed generate the characteristic spiral patterns with Fibonacci number relationships observed in nature [13].
Table 1: Key Historical Developments in Turing-Inspired Phyllotaxis Research
| Year | Development | Key Contributors | Significance |
|---|---|---|---|
| 1952 | "The Chemical Basis of Morphogenesis" published | Alan Turing | Introduced reaction-diffusion theory of pattern formation [4] [8] |
| 1953 | Dialogue and correspondence on phyllotaxis | Turing and Wardlaw | Application of Turing's theory to plant morphology [4] [12] |
| 1953 | "A Commentary on Turing's Diffusion-Reaction Theory" published | C.W. Wardlaw | Interpreted and promoted Turing's ideas for botanical audience [12] |
| 2006 | Computational model of auxin-based phyllotaxis | Jönsson et al., Smith et al. | Provided plausible molecular mechanism for phyllotactic patterning [13] |
Experimental Protocols
Protocol 1: Computational Modeling of Turing-Type Phyllotaxis
Objective and Principle
To implement a simplified computational model demonstrating how a Turing-type reaction-diffusion system can generate phyllotaxis-like patterns on a cylindrical domain, representing a plant stem [12]. The protocol is based on Turing's unpublished work on the "Daisy" model and cylindrical lattices [11] [10].
Materials and Software Requirements
- Software Platform: MATLAB, Python with NumPy/SciPy, or equivalent numerical computing environment
- Spatial Discretization: 2D cylindrical coordinate grid (θ, z)
- Reaction-Diffusion Solver: Finite difference method with periodic boundary conditions in θ-direction
- Visualization Tools: Surface plotting and animation capabilities
Procedure
System Definition:
- Define two morphogen fields: activator (a) and inhibitor (h)
- Set up reaction kinetics (e.g., Gierer-Meinhardt model):
- Activator reaction: ρa = k1 - k2a + k3(a^2/h)
- Inhibitor reaction: ρh = k4a^2 - k_5h
- Establish diffusion coefficients: Da << Dh (typically Dh/Da ≈ 10-100) [4]
Parameter Initialization:
- Set diffusion constants: Da = 0.0002, Dh = 0.01 (non-dimensional units)
- Set reaction rates: k1 = 0.02, k2 = 0.04, k3 = 0.1, k4 = 0.05, k_5 = 0.04
- Initialize morphogen concentrations with small random fluctuations around homogeneous steady state
Numerical Integration:
- Use explicit Euler method with sufficiently small time step (Δt = 0.001) for stability
- Implement discrete Laplacian operator for diffusion on cylindrical grid
- Apply periodic boundary conditions along angular coordinate
- Run simulation for sufficient iterations (typically 10^5-10^6 steps) to reach pattern stabilization
Pattern Analysis:
- Identify peaks in activator concentration as primordia positions
- Measure divergence angles between successive primordia
- Calculate parastichy numbers (spiral counts) from spatial arrangement
Table 2: Key Parameters for Turing-Type Phyllotaxis Simulation
| Parameter | Symbol | Typical Value Range | Biological Interpretation |
|---|---|---|---|
| Activator diffusion coefficient | D_a | 0.0001-0.001 | Limited mobility of activating morphogen [4] |
| Inhibitor diffusion coefficient | D_h | 0.01-0.1 | High mobility of inhibitory signal [4] |
| Diffusion ratio | Dh/Da | 10-100 | Critical for pattern instability [4] |
| Domain size (circumference) | L_θ | 2π | Angular coordinate on cylindrical surface |
| Domain size (height) | L_z | 5-20 | Axial extent of patterning region |
Protocol 2: Experimental Validation of Auxin-Based Phyllotaxis
Objective and Principle
To experimentally investigate the role of auxin transport in phyllotaxis using Arabidopsis shoot apical meristems, based on the model proposed by Reinhardt et al. (2003) and subsequent computational validation [13]. This protocol tests the hypothesis that polar auxin transport through PIN1 proteins establishes auxin maxima that determine primordia initiation sites.
Materials and Reagents
- Plant Material: 3-4 week old Arabidopsis thaliana plants (wild-type and pin1 mutants)
- Reporter Lines: DR5::GFP or DR5::GUS auxin response reporters
- Chemical Reagents:
- N-1-naphthylphthalamic acid (NPA, 100 μM in DMSO) - auxin transport inhibitor
- Synthetic auxin (e.g., 1-Naphthaleneacetic acid, NAA)
- Fixation solution: 4% formaldehyde in PBS
- GUS staining solution if using DR5::GUS lines
Experimental Workflow
Meristem Observation:
- Image live DR5::GFP expression in shoot apical meristems using confocal microscopy
- Document spatial pattern of auxin response maxima relative to existing primordia
Auxin Transport Inhibition:
- Apply NPA solution to shoot apices using soft brush application
- Monitor changes in DR5::GFP expression pattern over 24-72 hours
- Document disruption of phyllotactic patterning
Auxin Application:
- Locally apply NAA solution (10 μM) to pin-formed meristems (after NPA treatment)
- Observe induction of new primordia at application sites
- Verify organ initiation through histological analysis
PIN1 Localization:
- Perform immunohistochemistry for PIN1 protein in shoot apices
- Correlate PIN1 polarization patterns with auxin maxima locations
Computational Integration:
- Quantify divergence angles between successive primordia in control and treated meristems
- Compare experimental patterns with computational model predictions
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Tools for Phyllotaxis and Pattern Formation Studies
| Reagent/Resource | Function/Application | Example Use in Phyllotaxis Research |
|---|---|---|
| DR5::GFP/GUS Reporter | Visualizes auxin response maxima in tissues | Identifying sites of incipient primordia formation in shoot apices [13] |
| NPA (N-1-naphthylphthalamic acid) | Inhibitor of polar auxin transport | Testing necessity of auxin transport for phyllotaxis; induces pin-formed meristems [13] |
| PIN1 Antibodies | Immunodetection of PIN1 auxin efflux carrier | Mapping polarization patterns during primordia initiation [13] |
| pin1 Mutants | Genetic disruption of auxin transport | Establishing necessity of PIN1 for pattern formation [13] |
| Computational Modeling Frameworks | Simulating reaction-diffusion and auxin transport dynamics | Testing mechanistic hypotheses and parameter relationships [4] [13] |
Signaling Pathway Diagram
The 1952 seminal work by Alan Turing proposed that diffusion-driven instability could lead to the spontaneous formation of regular patterns, a concept now recognized as a fundamental principle in biological pattern formation [8]. In plant biology, Turing mechanisms, often implemented via reaction-diffusion models, have moved beyond the classical interpretation of spots and stripes to explain a remarkable diversity of complex structures. This Application Note explores the expanding role of Turing-like mechanisms in plant science, detailing how these models recreate patterns ranging from microscopic epidermal puzzles to three-dimensional vascular networks. We provide a structured synthesis of quantitative parameters, standardized protocols for computational modeling, and essential reagent solutions to equip researchers with the tools to investigate these patterning phenomena in silico and in vivo.
Turing's revolutionary insight was that diffusion, typically a homogenizing process, could destabilize a uniform equilibrium and lead to spontaneous pattern formation through a reaction-diffusion system [8]. The core principle involves at least two components: a slowly-diffusing activator (or self-activating component) that promotes its own production and the production of an inhibitor, and a rapidly-diffusing inhibitor that suppresses the activator. This "short-range activation and long-range inhibition" is the cornerstone of Turing patterning [8]. In plants, the manifestation of this principle is incredibly diverse, governing patterns across different spatial scales and tissue types. Modern interpretations extend beyond pure reaction-diffusion to include advection, mechanical stresses, and multi-component feedback loops, enabling the modeling of complex three-dimensional structures like vascular bundles and root development architectures [14] [15] [16].
Application Notes: Turing Patterning Across Scales and Structures
The following application notes summarize key experimental findings and the quantitative parameters that define Turing-like patterning in specific plant contexts.
Vascular Bundle Patterning in Stems and Roots
Supplemental Vascular Bundles (SVBs) in succulent species provide a classic example of three-dimensional patterning. A stochastic reaction-diffusion model successfully recreates their formation, suggesting a mechanism for their recurrent evolution.
Table 1: Key Parameters for SVB Patterning Model [15]
| Parameter/Variable | Description | Role in Model | Biological Correlate |
|---|---|---|---|
| H | Regulatory molecule concentration | Initiates vascular development | Putative morphogen (e.g., auxin) |
| B | Substrate molecule concentration | Consumed by H for pattern initiation | Developmental competence factor |
| M | Inhibitory molecule concentration | Suppresses H activity; diffuses from cortex | Inhibitory signal (e.g., cytokinin) |
| dH, dB | Diffusion coefficients of H, B | Determines spatial spread of activator | Mobility of signaling molecules |
| Parenchyma Width | Spatial context parameter | SVBs form only above a threshold width | Evolutionary character loss/gain |
Key Insights:
- The model consists of four to five molecules and successfully recreates empirical SVB patterns observed in over 38 plant families [15].
- A critical prediction is that SVBs fail to develop below a threshold width of parenchymatous tissue, providing a mechanistic explanation for evolutionary character loss due to changes in organ size [15].
- Altered diffusion rates of the activator (H) and substrate (B) molecules directly affect the number and size of the simulated SVBs [15].
Leaf Epidermal Patterning (Jigsaw Puzzle)
The interdigitated "jigsaw puzzle" pattern of leaf epidermal cells is regulated by opposing activities of ROP GTPases, a system that can be mapped to a Turing-like interface model.
Table 2: Key Parameters for Leaf Epidermal Patterning Model [17]
| Parameter/Component | Description | Role in Patterning |
|---|---|---|
| ROP2 | A Rho-like GTPase | Localizes to cell wall protrusions; promotes outgrowth via F-actin and targeted exocytosis. |
| ROP6 | A Rho-like GTPase | Localizes to concave regions; promotes cortical microtubule accumulation and cell wall reinforcement. |
| Auxin | Plant hormone | Regulates ROP activity; high concentration promotes ROP6, low concentration promotes ROP2. |
| Cell Wall Remodeling | Physical process | The interface between ROP2 (activator) and ROP6 (inhibitor) domains drives interdigitation. |
Key Insights:
- The model is mathematically equivalent to the mechanism governing human skull suture interdigitation, highlighting a universal patterning logic [17].
- It successfully reproduces the maintenance of cell wall thickness alongside the formation of interdigitations, explaining counter-intuitive experimental results [17].
General Turing System Requirements
For a two-component reaction-diffusion system to produce Turing patterns, specific conditions must be met regarding the reaction kinetics and diffusion.
Table 3: Conditions for Turing Instability in a Two-Component System [8]
| Condition | Mathematical Expression | Biological Interpretation |
|---|---|---|
| Stable Homogeneous State | a11 + a22 < 0 and a11*a22 - a12*a21 > 0 |
The non-spatial system (without diffusion) must be in a stable equilibrium. |
| Activator-Inhibitor | a11 > 0, a22 < 0, a12 < 0, a21 > 0 |
The activator (u) is self-enhancing and activates the inhibitor (v); the inhibitor suppresses the activator. |
| Differential Diffusion | D_v >> D_u |
The inhibitor must diffuse significantly faster than the activator to create long-range inhibition. |
Experimental Protocols
Protocol: Computational Modeling of 3D Vascular Patterning
This protocol outlines the procedure for developing a stochastic reaction-diffusion model to simulate Supplemental Vascular Bundle (SVB) formation, based on the work of Hearn (2019) [15].
I. Research Reagent Solutions
- Modeling Environment: Python (with NumPy/SciPy) or MATLAB for numerical computation.
- Spatial Discretization: A 50x50 (or larger) 2D grid to represent a cross-section of the stem/root.
- Numerical Solver: Euler integration method for solving partial differential equations (PDEs).
- Stochastic Engine: A random number generator for introducing stochastic noise into the reaction terms.
II. Methodology
- Define the Reaction-Diffusion System:
- Formulate the core PDEs. For a model with activator
Hand inhibitorM:∂H/∂t = F(H, B, M) + D_H * ∇²H∂M/∂t = G(H, M) + D_M * ∇²M + δ * ∇M FandGare functions defining the reaction kinetics.D_HandD_Mare diffusion coefficients.δis an advection constant for directional flow (e.g., tide, internal flow) [14] [15].
- Formulate the core PDEs. For a model with activator
Set Initial and Boundary Conditions:
- Initial State: Set initial concentrations of
H,B, andMto a homogeneous steady state, with a small random perturbation. - Boundary Conditions: Implement no-flux (Neumann) or periodic boundaries to confine the system.
- Initial State: Set initial concentrations of
Parameterize the Model:
- Use parameters derived from the literature (see Table 1) as a starting point. Key parameters include production, degradation, and diffusion rates.
Implement the Numerical Simulation:
- Discretize the Laplacian (
∇²) using a finite difference method (e.g., five-point stencil in 2D). - For the advection term, apply an upwinding scheme to maintain numerical stability [14].
- Set a timestep (
Δt = 0.05) and space stepsize (Δh = 1) [14]. Iterate the simulation until a stable pattern emerges.
- Discretize the Laplacian (
Analyze Pattern Output:
- Quantify the number, size, and spatial distribution of simulated SVB foci (high
Hregions). - Perform sensitivity analysis by varying key parameters (e.g.,
D_H, parenchyma width) to test model predictions.
- Quantify the number, size, and spatial distribution of simulated SVB foci (high
Protocol: Validating Turing Mechanisms Empirically
Computational predictions must be tested with biological experiments. This protocol describes the steps for validating a hypothesized Turing mechanism.
I. Research Reagent Solutions
- Molecular Biology Reagents: Antibodies for immunolocalization of putative activator/inhibitor molecules (e.g., ROPs, PINs, hormones).
- Live-Imaging Setup: Confocal laser scanning microscope (e.g., Olympus FV300), chamber slides, and fluorescent markers (e.g., GFP-PIP2a for plasma membrane) [17].
- Genetic Tools: Mutants or transgenic lines with altered expression or activity of the candidate patterning molecules.
II. Methodology
- Spatio-Temporal Correlation:
- Use live-imaging and immunolocalization to track the distribution of candidate molecules (activator/inhibitor) during pattern initiation and progression.
- Compare the observed patterns (e.g., wavelength, spacing) with those predicted by the model.
Perturbation Analysis:
- Chemical Perturbation: Apply inhibitors of diffusion (e.g., gap junction blockers), cytoskeletal dynamics (e.g., cellulase for cell walls), or specific signaling pathways [17].
- Genetic Perturbation: Analyze mutants where the diffusion or reaction kinetics of the system are altered (e.g., overexpressors, knockouts). The model should predict the qualitative changes in the pattern (e.g., spots to stripes, change in number/size of elements) [15].
Quantitative Morphometrics:
- Extract quantitative features from the observed biological patterns (e.g., SVB count and diameter, epidermal lobe number and depth).
- Statistically compare these metrics with the outputs of the computational model under corresponding conditions.
The Scientist's Toolkit
Table 4: Research Reagent Solutions for Investigating Plant Turing Patterns
| Reagent / Tool | Function / Application | Example Use Case |
|---|---|---|
| Reaction-Diffusion Modeling (PDE Solver) | In silico simulation of pattern formation using partial differential equations. | Recreating 3D vascular patterning via a stochastic Turing model [15]. |
| Finite Difference Method | Numerical discretization of continuous space for computational simulation. | Solving the Laplacian operator in spatial models on a defined grid [14]. |
| Live-Cell Fluorescent Markers (e.g., GFP-PIP2a) | Visualizing cell contours and dynamics in real-time. | Time-lapse imaging of leaf epidermal cell interdigitation [17]. |
| ROP GTPase Mutants/Analysis | Perturbing and observing the core regulators of cell shape. | Testing the roles of ROP2 (activator) and ROP6 (inhibitor) in jigsaw puzzle formation [17]. |
| Advection-Diffusion Modeling | Incorporating directional flow into pattern formation models. | Simulating the effect of tidal current on plant-wrack patterns [14]. |
The application of Turing's principles in plant biology has evolved to explain a stunning array of complex patterns far beyond simple spots and stripes. By integrating computational modeling with empirical validation, as detailed in these application notes and protocols, researchers can continue to decode the self-organizing principles that shape the plant body. The provided frameworks for modeling vascular patterning, epidermal puzzles, and other structures, alongside the essential toolkit of reagents and methods, offer a concrete pathway for advancing this frontier in evolutionary-developmental biology.
ROP Protein Patterning in Single Plant Cells
Application Notes
Rho-of-Plants (ROP) proteins constitute a key intracellular patterning system governing membrane domain specification in plant cells. This system operates via a Turing-type reaction-diffusion mechanism based on a substrate-depletion model, where active (membrane-bound) and inactive (cytosolic) ROP states diffuse at different rates, creating the necessary conditions for spontaneous pattern formation [4]. This mechanism enables a single cell to self-organize multiple domains, which is crucial for generating complex cellular morphologies.
The ROP patterning system is evolutionarily conserved and responsible for specifying both single domains (e.g., in cell polarity and tip growth) and multiple domains (e.g., in puzzle-shaped pavement cells and xylem secondary wall patterns) [4]. The stable coexistence of multiple ROP clusters is mathematically possible and biologically essential for forming numerous lobes on leaf epidermal pavement cells and regularly spaced secondary cell wall reinforcements in xylem cells [4].
Experimental Protocol
Objective: To observe and quantify ROP-mediated pattern formation within single plant cells.
Materials:
- Arabidopsis thaliana lines expressing fluorescently tagged ROP proteins (e.g., ROP4-GFP)
- Confocal laser scanning microscope
- Image analysis software (e.g., MorphoGraphX, FIJI/ImageJ)
- Pharmacological inhibitors (e.g., cytoskeletal disruptors)
Procedure:
- Sample Preparation: Grow Arabidopsis plants expressing ROP4-GFP under controlled conditions. For pavement cell analysis, use young expanding leaves. For xylem analysis, use differentiating vascular tissues [4].
- Live Imaging: Mount tissue samples and image using confocal microscopy with appropriate settings for GFP detection. Capture time-series images to track pattern dynamics.
- Perturbation Experiments: Apply cytoskeletal inhibitors (e.g., Latrunculin B for actin disruption) to test mechanism robustness.
- Quantitative Analysis: Use image analysis software to quantify:
- Number of ROP clusters per cell
- Cluster size and intensity
- Spatial distribution of clusters
- Model Validation: Compare experimental results with computational predictions from ROP Turing models that incorporate cluster-level bookkeeping [4].
Expected Outcomes: The protocol should reveal whether ROP patterning follows Turing dynamics, characterized by spontaneous pattern formation from near-homogeneous conditions and the stable coexistence of multiple activator clusters in sufficiently large domains.
Table 1: Key Parameters for ROP Patterning in Different Cell Types
| Cell Type | Number of ROP Clusters | Cluster Stability | Biological Function |
|---|---|---|---|
| Pavement Cells | Increases during cell growth [4] | Transient to stable | Lobes and indentations |
| Xylem Cells | Regularly spaced | Stable | Secondary wall reinforcements |
| Tip-growing Cells | Single domain | Stable | Polarized growth |
Epidermal Patterning in Arabidopsis Root and Leaf
Application Notes
Epidermal patterning in Arabidopsis provides compelling examples of Turing-inspired mechanisms operating across multiple cells. In the root epidermis, a gene regulatory network (GRN) coupled with protein diffusion generates an interspersed pattern of hair (trichoblast) and non-hair (atrichoblast) cells [18]. This system employs lateral inhibition dynamics where the transcription activation complex (MBW) promotes production of diffusible inhibitors (CPC, GL3/EGL3), preventing adjacent cells from adopting the same fate [18].
In leaf and sepal epidermis, a common genetic pathway controls cell size patterning, leading to non-random distributions of giant cells [19] [20]. These giant cells form when ATML1 concentration surpasses a threshold during G2 phase, triggering early endoreduplication [20]. The spatial pattern emerges from stochastic giant cell initiation combined with divisions of surrounding cells, creating clustered patterns in mature tissues [19] [20].
Experimental Protocol
Objective: To analyze epidermal pattern formation in Arabidopsis root and leaf/sepal tissues.
Materials:
- Arabidopsis wild-type and mutant lines (e.g., lgo-2, ATML1-OX, cpc, wer)
- Confocal microscope
- Molecular biology reagents for genotyping
- Fixation and staining solutions
Procedure:
Root Epidermis Patterning:
- Sample Preparation: Grow Arabidopsis seedlings vertically on agar plates for 5-7 days.
- Imaging: Image root epidermis using confocal microscopy, focusing on the differentiation zone.
- Pattern Quantification: Calculate the percentage of hair cells in H and N positions using image analysis software.
- Mutant Analysis: Compare patterning in mutants (e.g., cpc, wer) against wild-type.
- GRN Modeling: Implement a meta-GRN model incorporating CPC and GL3/EGL3 diffusion on a 24×24 cell grid with random initial conditions [18].
Leaf/Sepal Giant Cell Patterning:
- Sample Preparation: Grow Arabidopsis plants and collect stage 14 sepals or mature leaves.
- Imaging: Capture epidermal images using microscopy and create cell area heat maps.
- Pattern Analysis: Compare giant cell distributions with computationally randomized tissues to assess clustering [20].
- Genetic Analysis: Examine giant cell formation in mutants (acr4, atml1, lgo) and overexpression lines (ATML1-OX, LGO-OX) [20].
Expected Outcomes: The root epidermis should show characteristic alternating columns of hair and non-hair cells, while leaf/sepal epidermis should exhibit non-random clustering of giant cells. Mutant phenotypes should align with predictions from Turing-type models.
Table 2: Epidermal Patterning Mutants and Phenotypes
| Genotype | Giant Cell Number | Root Hair Pattern | Spatial Organization |
|---|---|---|---|
| Wild Type | Normal [20] | Regular columns [18] | Alternating cell fates [18] |
| lgo-2 | Reduced [20] | N/A | N/A |
| ATML1-OX | Ectopic formation [20] | N/A | N/A |
| cpc | N/A | Disrupted | Ectopic hair cells [18] |
| wer | N/A | Disrupted | Ectopic non-hair cells [18] |
Dryland Vegetation Patterning
Application Notes
Dryland ecosystems exhibit striking large-scale vegetation patterns—including gaps, spots, stripes, and labyrinths—that represent classic examples of Turing systems in ecology [21] [22]. These patterns emerge from feedback between biomass and water availability, where vegetation acts as a local activator by improving water infiltration, while water scarcity serves as a long-range inhibitor [21]. The Klausmeier model and its extensions provide mathematical frameworks for understanding these patterns, incorporating factors like finite soil resources, inertial effects, and grazing pressures [21] [22].
Vegetation patterning represents an ecosystem-scale adaptation to arid conditions, enabling systems to maximize water utilization and resist desertification [22]. Pattern transitions (e.g., from spots to gaps) can signal impending ecosystem collapse, making them important indicators for land management [22].
Experimental Protocol
Objective: To document and analyze vegetation pattern formation in dryland ecosystems.
Materials:
- Aerial or satellite imagery of dryland regions
- Climate data (precipitation, evaporation rates)
- Soil moisture sensors
- Mathematical modeling software (e.g., MATLAB, Python)
Procedure:
- Field Observation: Select study sites in dryland ecosystems. Capture high-resolution aerial imagery of vegetation patterns across rainfall gradients.
- Pattern Classification: Categorize observed patterns as gaps, spots, stripes, or labyrinths.
- Environmental Data Collection: Record precipitation, evaporation rates, and soil characteristics.
- Model Implementation: Implement an extended Klausmeier model incorporating hyperbolic reaction-transport, finite soil resources, and inertial effects [21]:
Model Equations:
- Biomass equation: ∂B/∂t = f(B,W) - k·g(B) + D_B·∇²B
- Water equation: ∂W/∂t = p - e·W - h(B,W) + D_W·∇²W
Where B is biomass, W is water, p is precipitation, e is evaporation, k is grazing pressure, and DB, DW are diffusion coefficients [21] [22].
- Linear Stability Analysis: Perform linear stability analysis to identify Turing instability conditions.
- Numerical Simulation: Simulate pattern evolution under different climate scenarios and grazing pressures [22].
Expected Outcomes: The protocol should reveal how specific environmental parameters (precipitation, evaporation, grazing) drive transitions between different vegetation patterns and identify potential tipping points for ecosystem collapse.
Table 3: Vegetation Pattern Types and Environmental Drivers
| Pattern Type | Precipitation Range (mm/year) | Grazing Impact | Ecosystem Function |
|---|---|---|---|
| Gaps | Lowest [22] | High sensitivity | Survival in aridity |
| Spots | Low to medium [22] | Moderate sensitivity | Water harvesting |
| Stripes | Medium [22] | Low to moderate sensitivity | Directional water flow |
| Labyrinths | Medium to high | Low sensitivity | Maximum coverage |
The Scientist's Toolkit
Table 4: Essential Research Reagents and Resources
| Reagent/Resource | Application | Function |
|---|---|---|
| Fluorescently tagged ROP lines [4] | ROP patterning | Visualize protein localization and dynamics |
| Arabidopsis epidermal mutants (cpc, wer, lgo) [18] [20] | Epidermal patterning | Disrupt specific patterning components |
| Confocal microscopy | All imaging | High-resolution spatial data collection |
| MorphoGraphX software [20] | Image analysis | 3D segmentation and quantitative analysis |
| Extended Klausmeier model [21] | Vegetation patterning | Simulate pattern formation under climate change |
Signaling Pathway and Mechanism Diagrams
ROP Patterning Mechanism
Root Epidermis Patterning Network
Dryland Vegetation Pattern Formation
Computational Frameworks for Simulating Plant Patterning: From Single Cells to Ecosystems
Rho-of-Plant (ROP) GTPases represent a plant-specific subfamily of the Rho family of small GTPases that serve as central molecular switches in establishing cell polarity and regulating intracellular patterning [23]. As the sole representatives of the Ras superfamily in plants, ROPs uniquely fulfill functions attributed to both Rho and Ras GTPases in animals, integrating signaling pathways that control cytoskeletal organization, vesicular trafficking, and response to extracellular cues [24]. These proteins cycle between active GTP-bound and inactive GDP-bound states, and their precise spatiotemporal organization within the plasma membrane generates diverse patterning outcomes essential for plant development—from single clusters in tip-growing cells to multiple clusters in leaf pavement cells and striped patterns in xylem cell walls [25].
The patterning capability of ROPs stems from their ability to form self-organizing membrane domains of varying scales. According to established nomenclature, ROP microdomains exceed 1µm in size and are hallmarks of cell polarity, while ROP nanodomains fall below this limit and are critical for signaling specificity and amplification [23]. This hierarchical domain organization creates emergent properties that determine fundamental cellular processes including polarized growth, division orientation, and cell wall patterning [23] [26]. The formation of these patterns is governed by reaction-diffusion principles, wherein local self-activation coupled with long-range inhibition generates stable spatial patterns from initial homogeneity [25] [27].
Theoretical Framework: Reaction-Diffusion Principles
Turing-Type Patterning Mechanisms
The theoretical foundation for ROP patterning lies in Alan Turing's reaction-diffusion theory, which demonstrates how the interaction between an activator and inhibitor species with different diffusion rates can spontaneously generate periodic spatial patterns [28] [27]. In the context of ROP signaling, this translates to local self-activation of ROP GTPases combined with global inhibition, creating what is often described as a "local excitation, global inhibition" system [28].
Mathematical modeling reveals that the core ROP activation-inactivation cycle inherently contains Turing-type pattern-forming capabilities [27]. The standard model incorporates two key states of ROP GTPases: the active membrane-bound form and the inactive cytosolic form. The critical patterning principle emerges from the differential diffusion rates between these states—active ROP diffuses slowly in the membrane (Du ≈ 0.01-0.1 µm²/s), while inactive ROP diffuses rapidly in the cytosol (Dv ≈ 10-100 µm²/s) due to guanine nucleotide dissociation inhibitor (GDI)-mediated cycling [25]. This diffusion disparity, combined with nonlinear positive feedback in ROP activation, creates the necessary conditions for spontaneous pattern formation.
Core Mathematical Formulation
The fundamental reaction-diffusion equations governing ROP patterning can be expressed as a mass-conserved system [25]:
Where u represents active membrane-bound ROP, v represents inactive cytosolic ROP, Du and Dv are their respective diffusion coefficients (with Dv ≫ Du), and f(u,v) describes the biochemical interconversion between states. A typical conversion function incorporates baseline activation, positive feedback, and saturation:
Here, b represents the basal activation rate, γ the maximum feedback-enhanced activation rate, K the concentration for half-maximal feedback, and δ the inactivation rate [25]. This formulation generates bistable behavior essential for pattern formation, allowing coexistence of low background activity and high activity within ROP domains.
Table 1: Key Parameters in ROP Patterning Models
| Parameter | Biological Significance | Typical Range | Effect on Patterning |
|---|---|---|---|
| Du | Diffusion of active ROP in membrane | 0.01-0.1 µm²/s | Smaller values promote finer patterns |
| Dv | Diffusion of inactive ROP in cytosol | 10-100 µm²/s | Larger values promote pattern regularity |
| γ | Strength of positive feedback | Variable | Higher values promote pattern emergence |
| K | Feedback saturation constant | Variable | Affects cluster density and size |
| δ | Inactivation rate | Variable | Higher values suppress patterning |
Molecular Components of the ROP Patterning System
Core Regulatory Machinery
The ROP patterning system comprises a minimal set of molecular components that govern the GTPase cycle and spatial organization. These include the ROP GTPases themselves, their regulatory proteins, and downstream effectors [23] [24].
ROP GTPases are approximately 20 kDa proteins consisting primarily of a G domain with short N- and C-terminal extensions. The hypervariable C-terminal region undergoes lipid modification (geranylgeranylation or palmitoylation), facilitating membrane association and determining specific membrane dynamics [23]. Plants possess two distinct types of ROPs with different molecular properties, which influence their patterning capabilities [25].
Guanine nucleotide exchange factors (GEFs) activate ROPs by catalyzing GDP to GTP exchange. Plants possess both conserved DHR2-type GEFs and plant-specific RopGEFs [24]. RopGEFs can form dimers that interact with activated ROP, creating positive feedback loops essential for pattern formation [27].
GTPase-activating proteins (GAPs) inactivate ROPs by stimulating GTP hydrolysis. Some plant GAPs contain CRIB motifs that enable them to bind active ROP, potentially creating negative feedback loops [24].
Guanine nucleotide dissociation inhibitors (GDIs) sequester inactive ROP in the cytosol, facilitating rapid diffusion and preventing membrane association [23]. This function is critical for establishing the differential diffusion rates necessary for pattern formation.
Effector Proteins and Downstream Signaling
Active ROP GTPases interact with various effector proteins to translate patterns into cellular responses. The primary plant-specific ROP effectors are ROP INTERACTIVE CRIB MOTIF-CONTAINING PROTEINS (RICs) [24]. Different RICs perform counteracting functions—for example, in pollen tubes, RIC4 promotes actin assembly while RIC3 promotes actin disassembly through calcium-mediated pathways, enabling precise control of cytoskeletal dynamics [24].
Other effectors include ICRs/RIPs that scaffold the exocyst complex, RBKs/RKKs receptor-like cytoplasmic kinases, and Rboh NADPH oxidases involved in reactive oxygen species production [24]. The specific complement of effectors expressed in a cell type determines how ROP patterns manifest morphologically.
Figure 1: ROP GTPase Signaling Pathway. This diagram illustrates the core regulatory cycle of ROP GTPases and their interaction with regulators, effectors, and downstream cellular processes. Positive feedback loops critical for pattern formation are indicated with dashed lines.
Experimental Protocols for Investigating ROP Patterning
Genetic Manipulation of ROP Signaling Components
Objective: To determine the role of specific regulators in ROP domain formation and patterning density.
Materials:
- Arabidopsis thaliana wild-type and mutant lines (ropgef4-1, ropgef7-1, ropgap3-1, ropgap4-2)
- Constructs for complementation (pROPGEF4:GFP-ROPGEF4, pROPGEF7:YFP-ROPGEF7)
- Gateway cloning system for vector construction
- Agrobacterium tumefaciens strain GV3101 for plant transformation
Methodology:
- Identify T-DNA insertion mutants or EMS mutants for ROP regulators from available collections [27].
- Generate double mutants through genetic crossing and confirm genotypes by PCR.
- For complementation assays, clone genomic sequences including native promoters fused to fluorescent tags into binary vectors.
- Transform mutants via floral dip method and select transformants on appropriate antibiotics.
- Analyze metaxylem vessels in 7-day-old light-grown seedlings by staining with propidium iodide or basic fuchsin to visualize secondary cell walls [27].
- Quantify pit density (number/µm²), pit area, and distribution patterns using image analysis software (e.g., ImageJ).
- Assess pattern regularity by measuring nearest-neighbor distances and calculating coefficient of variation.
Expected Results: ropgef4 ropgef7 double mutants show reduced pit density (≈60-70% of wild-type) and irregular spacing, while ropgap3 ropgap4 double mutants exhibit larger pits with normal density but altered distribution [27].
Live Imaging of ROP Dynamics in Tip-Growing Cells
Objective: To visualize ROP domain formation and dynamics in real-time during polarized growth.
Materials:
- Arabidopsis lines expressing ROP2/4/6-GFP under native promoters
- Confocal laser scanning microscope with high-sensitivity detectors
- Microfluidic chambers for root immobilization
- Image analysis software with particle tracking capabilities
Methodology:
- Grow seedlings vertically on half-strength MS medium for 5 days.
- Mount seedlings in microfluidic chambers with liquid medium for stability during imaging.
- For root hair initiation studies, focus on trichoblasts in the differentiation zone.
- Acquire time-lapse images every 2-5 seconds using high-resolution confocal microscopy (e.g., 512×512 pixels, 2× zoom).
- Use low laser power (1-5%) to minimize phototoxicity during extended time-lapse imaging.
- Analyze fluorescence intensity distribution, domain size, and lifetime using specialized tracking algorithms.
- Correlate ROP dynamics with growth rates by simultaneous bright-field imaging.
Expected Results: ROP2/4/6 form stable microdomains (5-10µm) at future root hair initiation sites, with two-phase recruitment: initial GEF3-dependent initiation followed by GEF4-dependent maintenance [23].
Mathematical Model Implementation and Simulation
Objective: To computationally test patterning mechanisms and predict outcomes of genetic perturbations.
Materials:
- MATLAB, Python, or similar computational environment
- Partial differential equation solver (e.g., COMSOL, FEniCS, or custom finite difference code)
- High-performance computing resources for parameter screening
Methodology:
- Implement the core reaction-diffusion equations using finite difference or finite element methods.
- Define a 1D or 2D spatial domain representing the plasma membrane with no-flux boundary conditions.
- Set initial conditions as homogeneous active ROP levels with small random perturbations.
- Parameterize the model using experimental data where available (see Table 2).
- Simulate pattern formation over relevant timescales (typically minutes to hours).
- Perform bifurcation analysis to identify parameter regions supporting different pattern types (single cluster, multiple clusters, stripes).
- Validate model predictions by comparing with experimental observations of mutant phenotypes.
Expected Results: The model should recapitulate wild-type patterning and predict altered patterning in genetic mutants, revealing whether specific regulators primarily affect activation, inhibition, or feedback strength [25] [27].
Table 2: Experimentally Derived Parameters for ROP Patterning Models
| Parameter | Experimental System | Measurement Technique | Value Range |
|---|---|---|---|
| ROP diffusion coefficient | Pollen tubes | FRAP | 0.05-0.1 µm²/s |
| GEF membrane residence | Metaxylem | Fluorescence recovery | >60 seconds |
| ROP activation rate | Root hairs | Kinetic modeling | 0.1-1.0 s⁻¹ |
| ROP inactivation rate | Wound response | GTPase activity assays | 0.5-2.0 s⁻¹ |
| Domain size | Metaxylem pits | Super-resolution microscopy | 1-5 µm |
| Domain spacing | Metaxylem pits | Nearest-neighbor analysis | 2.5-7.5 µm |
Case Studies in ROP Patterning
Metaxylem Vessel Pitting Pattern
The metaxylem vessel system provides a compelling example of ROP-based reaction-diffusion patterning. In this system, ROP11 forms activated domains that prefigure secondary cell wall pits through interaction with cortical microtubules [27]. Genetic evidence demonstrates that ROPGEF4 and ROPGEF7 positively regulate pit density and size, while ROPGAP3 and ROPGAP4 negatively regulate pit size but positively influence density [27]. The competing actions of these activators and inhibitors create a Turing-type system that spontaneously generates regularly spaced pits.
Mathematical modeling of this system revealed that positive feedback through ROP11-ROPGEF4 interaction and slow diffusion of the ROP11-ROPGEF4 complex are essential for pattern formation [27]. The model successfully predicted that ROPGEF4 forms stable dimers on the plasma membrane that interact with activated ROP11, creating the necessary nonlinear feedback for domain formation. This case study exemplifies how integrating genetics, live imaging, and mathematical modeling can unravel complex patterning mechanisms.
Pavement Cell Interdigitation
The jigsaw-puzzle shape of leaf epidermal pavement cells represents another prominent example of ROP-dependent patterning. In this system, ROP2 and ROP4 form alternating activation domains that correspond to lobe and neck regions, respectively [23] [24]. These patterns emerge from counteracting pathways downstream of ROP activation: RIC4 promotes actin assembly in lobes, facilitating local expansion, while RIC1 promotes microtubule stabilization in necks, restricting expansion [24].
This system demonstrates how the same ROP isoforms can generate either single-cluster or multi-cluster patterns depending on contextual factors and regulatory tuning [25]. The coexistence of multiple stable ROP clusters requires specific model formulations that avoid winner-takes-all competition, often implemented through saturation of self-activation or inclusion of additional stabilizing factors [25].
Figure 2: Integrated Experimental-Computational Workflow. This diagram outlines the synergistic approach combining genetic manipulation, live imaging, and mathematical modeling to investigate ROP patterning mechanisms. Dashed lines indicate iterative feedback between experimental and computational approaches.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Investigating ROP Patterning
| Reagent/Category | Specific Examples | Function/Application | Key References |
|---|---|---|---|
| Mutant Lines | ropgef4-1, ropgef7-1, ropgap3-1, ropgap4-2 | Genetic dissection of regulator functions | [27] |
| Fluorescent Reporters | ROP2/4/6-GFP, ROP11-YFP | Live imaging of ROP dynamics and domain formation | [23] [27] |
| Expression Constructs | pROPGEF4:GFP-ROPGEF4, pROPGEF7:YFP-ROPGEF7 | Complementation tests and protein localization | [27] |
| Mathematical Models | Mass-conserved reaction-diffusion framework | Theoretical analysis of patterning mechanisms | [25] [28] |
| Cell Type Markers | Trichoblast-specific promoters, xylem vessel markers | Cell-type specific manipulation and observation | [23] [27] |
| Pharmacological Agents | Latrunculin B (actin disruptor), Orobol (GEF inhibitor) | Acute perturbation of cytoskeleton or signaling | [24] |
The study of ROP GTPase patterning exemplifies how integrating experimental biology with mathematical modeling reveals fundamental principles of cellular organization. The reaction-diffusion framework provides a powerful explanatory paradigm for diverse patterning phenomena, from the regularly spaced pits in metaxylem vessels to the intricate interdigitation of pavement cells. Key insights emerging from this field include the importance of lipid modifications in ROP membrane dynamics, the role of plant-specific regulators like RopGEFs, and the critical contribution of differential diffusion rates between active and inactive ROP pools.
Future research directions will likely focus on several frontiers. First, understanding how ROP patterns are integrated with tissue-level cues and mechanical forces represents a significant challenge. Second, the development of higher-resolution imaging techniques will reveal nanoscale organization within ROP domains and its functional implications. Third, extending modeling approaches to incorporate crosstalk between multiple ROP isoforms and their effectors will provide more comprehensive understanding of pattern specificity. Finally, exploring conservation and divergence of ROP patterning mechanisms across plant species may reveal fundamental design principles of eukaryotic cell polarity systems.
The protocols and frameworks presented here provide a foundation for continued investigation into how simple molecular interactions give rise to complex cellular patterns through self-organization—a question at the heart of developmental biology and biophysics alike.
The regular arrangement of leaves, known as phyllotaxis, has long fascinated biologists and mathematicians alike. This phenomenon represents a classic example of biological pattern formation. Contemporary research has established that the plant hormone auxin serves as a primary activator for leaf primordium development, with its spatiotemporal distribution largely governed by the polar subcellular localization of the PIN-FORMED1 (PIN1) efflux transporter [29] [30]. Two principal mechanisms for PIN1 polarization have been proposed: the "up-the-gradient" (UTG) model, where PIN1 localizes towards cells with higher auxin concentration, and the "with-the-flux" (WTF) model, where PIN1 polarization aligns with the direction of auxin flow [29] [31]. This application note examines the integration of these models within the broader theoretical framework of Turing pattern formation, providing detailed protocols for key computational and experimental approaches.
Theoretical Background: From Turing Patterning to Auxin Transport
Alan Turing's 1952 theory demonstrated that the interaction between a short-range activator and a long-range inhibitor could spontaneously break symmetry to generate regular patterns [4] [8]. While the auxin/PIN system in phyllotaxis is more complex than a canonical reaction-diffusion system, it operates on similar principles of local self-enhancement and long-range inhibition [4].
In modern phyllotaxis models, auxin acts as the activator, with its accumulation initiating primordia. The polarization of PIN1 transporters toward emerging primordia creates auxin depletion in surrounding areas, establishing an inhibitory field that prevents new primordia from forming too close to existing ones [4] [8]. This feedback loop between auxin distribution and PIN1 localization generates the highly regular patterns observed in nature.
Table 1: Core Concepts in Phyllotaxis Modeling
| Concept | Description | Theoretical Basis |
|---|---|---|
| Up-the-Gradient (UTG) | PIN1 polarizes toward neighboring cells with higher auxin concentration [29]. | Forms auxin convergence points for primordium initiation [30]. |
| With-the-Flux (WTF) | PIN1 polarization strengthens in the direction of high auxin flux [29]. | Underlies canalization during midvein formation [29]. |
| Dual Polarization | UTG and WTF mechanisms operate concurrently within the same tissue [29] [32]. | Explains simultaneous high auxin concentration and flux in veins [29]. |
| Turing Patterning | Short-range activation coupled with long-range inhibition generates periodic patterns [4] [8]. | Provides a conceptual framework for understanding phyllotactic regularity. |
Integrated Signaling and Polarization Pathways
The following diagram illustrates the core signaling logic integrating auxin transport, PIN polarization, and the formation of phyllotactic patterns.
Diagram 1: Signaling Logic in Phyllotaxis. This diagram illustrates the core feedback loops between auxin transport and PIN1 polarization. The system generates both convergence points for organ initiation and canalized strands for vascular development through the integrated operation of UTG and WTF mechanisms.
The Dual Polarization Model: A Unified Framework
A significant challenge in phyllotaxis research has been reconciling the UTG model for primordia positioning with the WTF model for vein formation, as these processes intersect during midvein initiation [29]. The dual polarization model addresses this by proposing that both UTG and WTF mechanisms operate simultaneously within the same cells [29] [32].
Computer simulations of this integrated model successfully reproduce the observed dynamics of PIN1 localization during leaf initiation and midvein formation [29]. Crucially, these simulations demonstrate the appearance of high auxin concentration throughout midvein initiation, resolving a long-standing criticism of the canalization hypothesis by explaining how both high flux and high concentration can coexist in emerging veins [29] [31].
Table 2: Characteristics of PIN1 Polarization Mechanisms
| Feature | Up-the-Gradient (UTG) | With-the-Flux (WTF) | Dual Polarization |
|---|---|---|---|
| Primary Role | Primordium positioning in the L1 layer [29]. | Vascular strand formation in inner tissues [29]. | Integrates both patterning processes [29]. |
| Spatial Context | Operates predominantly in the epidermal L1 layer [30]. | Functions in subepidermal tissues during venation [29]. | Functions across tissue layers [32]. |
| Auxin Signal | Responds to local auxin concentration gradients [30]. | Responds to directional auxin flux [29]. | Responds to both concentration and flux [29]. |
| Turing Analogy | Contributes to local self-enhancement (activation) [4]. | Establishes long-range inhibition [4]. | Embodies the full activator-inhibitor system. |
Experimental Protocols
Protocol: Computational Modeling of Dual Polarization
This protocol outlines steps for creating a 3D cellular model to simulate auxin transport and PIN1 dynamics, based on methods from [29] and [32].
Research Reagents & Computational Tools
- Template Geometry: 3D representation of a tomato or Arabidopsis shoot apical meristem (SAM)
- Auxin Transport Model: Parameters from the chemiosmotic hypothesis (e.g., membrane permeabilities, pH values) [30]
- PIN1 Cycling Model: Equations describing PIN1 endocytosis/exocytosis dynamics
- Solver Software: Platform for solving partial differential equations (e.g., CellMod, VirtualLeaf)
Procedure
- Template Acquisition: Obtain or generate a 3D cellular template of the SAM. Tomato meristems are often preferred for their larger size and accessibility for live imaging [29].
- Parameter Initialization:
- Set initial auxin concentration to a near-homogeneous state across cells
- Initialize PIN1 concentrations uniformly at membrane compartments
- Model Implementation:
- Implement auxin transport equations incorporating both passive diffusion and active PIN1-mediated transport
- Implement the dual polarization rule:
PIN1_localization = f(auxin_concentration) + g(auxin_flux)
- Simulation Execution:
- Run simulations with a time step sufficiently small for numerical stability
- Continue simulation until the system reaches a dynamic steady state or completes the desired number of iterations
- Data Collection:
- Record auxin concentrations and PIN1 polarities for each cell at each time step
- Document the emergence of auxin convergence points and their development into canalized strands
Validation: Compare simulation outputs with experimental confocal microscopy images of PIN1::GFP and DR5rev::GFP auxin response markers [29].
Protocol: Empirical Analysis of PIN1 Polarization Dynamics
This protocol describes an experimental approach for visualizing and quantifying PIN1 polarization during phyllotaxis, adapted from [29].
Research Reagents & Materials
- Plant Material: Tomato (Solanum lycopersicum) seedlings or Arabidopsis thaliana PIN1::GFP reporter lines
- Fixation Solution: 4% formaldehyde in phosphate buffer
- Primary Antibody: Rabbit anti-tomato PIN1 polyclonal antibody
- Secondary Antibody: Goat anti-rabbit IgG conjugated to Alexa Fluor 488
- Imaging Equipment: Confocal laser scanning microscope
Procedure
- Sample Preparation:
- Dissect shoot apices from 3-week-old tomato plants or use Arabidopsis PIN1::GFP seedlings
- Fix tissues in 4% formaldehyde for 2 hours at room temperature
- Immunolocalization (for tomato):
- Permeabilize cell walls with 2% cellulase for 30 minutes
- Incubate with primary anti-PIN1 antibody (1:500 dilution) overnight at 4°C
- Wash and incubate with secondary antibody (1:1000 dilution) for 2 hours
- Confocal Microscopy:
- Mount samples in antifading solution
- Image using a confocal microscope with appropriate excitation/emission settings for GFP or Alexa Fluor 488
- Capture z-stacks through multiple cell layers of the SAM
- Image Analysis:
- Quantify PIN1 signal intensity at different membrane domains using image analysis software (e.g., ImageJ)
- Calculate polarization indices by comparing signal intensity at membrane facets
- Correlate PIN1 polarity patterns with predicted sites of primordium initiation
Expected Outcomes: PIN1 should localize toward incipient primordia in the L1 layer (UTG pattern), while in subepidermal cells, it should align in continuous files toward the center of the meristem (WTF pattern) [29].
The Scientist's Toolkit
Table 3: Essential Research Reagents for Phyllotaxis Studies
| Reagent / Tool | Function / Application | Example Use |
|---|---|---|
| PIN1::GFP Reporter Line | Visualizes PIN1 expression and localization in live tissue [29]. | Real-time tracking of PIN1 polarization dynamics during primordium initiation. |
| DR5rev::GFP Reporter | Serves as a synthetic auxin response reporter [29]. | Marks sites of auxin accumulation and maxima. |
| Anti-PIN1 Antibody | Immunodetection of PIN1 protein in fixed tissues [29]. | PIN1 localization in species without reliable transgenic reporters. |
| NPA (N-1-naphthylphthalamic acid) | Chemical inhibitor of polar auxin transport [33]. | Testing the role of auxin transport in pattern formation. |
| Computational Frameworks (CellMod, etc.) | Provides a platform for implementing and testing mathematical models [32]. | Simulating the interplay between UTG and WTF polarization mechanisms. |
The integration of auxin transport models with PIN protein polarization dynamics represents a significant advance in understanding phyllotaxis. The dual polarization model successfully unifies previously disparate mechanisms, demonstrating how UTG and WTF patterning can operate concurrently within the same tissue. This integrated framework, grounded in Turing's principles of pattern formation, provides a powerful paradigm for investigating how molecular-scale interactions give rise to macroscopic biological patterns. Future research will benefit from continued refinement of 3D models and experimental validation across diverse plant species.
Reaction-Diffusion-Advection Systems for Large-Scale Vegetation Patterning
Reaction-diffusion-advection systems provide a powerful mathematical framework for explaining the emergence of large-scale vegetation patterns in water-limited ecosystems. These patterns, observed as periodic arrangements of vegetation bands, spots, or labyrinths, arise from the interplay between biological processes and environmental constraints. The theoretical foundation traces back to Alan Turing's seminal work on morphogenesis, which demonstrated how diffusion could drive the spontaneous formation of regular patterns from an initially homogeneous state [8] [4]. This mechanism, now known as Turing patterning, represents a paradigm shift in understanding how simple local interactions can generate complex global order through a process of diffusion-driven instability [34].
In the context of dryland vegetation, the Turing mechanism manifests through scale-dependent feedbacks involving essential resources, primarily water. The vegetation patterns form at the transition between homogeneous states—dense vegetation and barren land—with specific environmental conditions determining the particular pattern morphology [35]. The reaction-diffusion framework has been extended to include advection to account for the unidirectional flow of water down slopes, which is essential for modeling the anisotropic stripe patterns characteristic of hillslope environments [35] [8]. This integration of advection distinguishes vegetation patterning models from classical Turing systems and enables more realistic simulation of ecosystem-scale phenomena.
The core mechanism follows the principle of short-range facilitation and long-range competition [8] [4]. Vegetation acts as a self-activating component (activator) that improves local water infiltration and promotes further growth, while water serves as a depleted substrate that is consumed by vegetation and transported across the landscape. The differential mobility of these components—with water flowing much farther than vegetation spreads—creates the necessary conditions for pattern formation, analogous to the differential diffusion in classical Turing systems [35]. This biophysical feedback system generates predictable patterns whose characteristics serve as indicators of ecosystem health and resilience to environmental change [35].
Quantitative System Parameters and Characteristics
Table 1: Core Components and Their Roles in Vegetation Patterning Systems
| Component | Mathematical Role | Biological/Physical Role | Typical Spatial Scale |
|---|---|---|---|
| Vegetation Density | Activator (slowly diffusing) | Self-enhancing through improved water infiltration; exhibits logistic growth | Local (meters) |
| Soil Water Content | Inhibitor/Depleted Substrate (fast-diffusing/advecting) | Limited resource consumed by vegetation; transported via diffusion and overland flow | Landscape (10-100 meters) |
| Terrain Slope | Advection coefficient | Drives directional water flow; induces anisotropy | Hillslope (varies) |
| Infiltration Capacity | Nonlinear feedback term | Enhanced under vegetation patches; creates positive feedback | Patch-scale (1-10 meters) |
Table 2: Pattern Morphology and Environmental Correlations
| Pattern Type | Environmental Context | Characteristic Wavelength | Ecosystem Indicators |
|---|---|---|---|
| Isotropic Spots | Flat terrain, low water availability | 10-100 meters | Transition to desertification |
| Labyrinthine | Flat terrain, intermediate water availability | 20-50 meters | Critical transition state |
| Anisotropic Stripes | Sloped terrain, unidirectional water flow | 3-40 meters (depending on slope) | Ecosystem resilience |
The mathematical formulation of vegetation patterning models incorporates specific parameters that determine both the qualitative pattern morphology and quantitative characteristics such as wavelength and amplitude. The critical wavelength against which the homogeneous state first becomes unstable depends on the diffusion coefficients and other parameters, scaling with the square root of the product of the diffusion coefficients divided by a weighted sum [8]. For isotropic patterns on flat terrain, the characteristic wavelength typically ranges from 10 to 100 meters, while anisotropic patterns on hillslopes exhibit wavelengths generally between 3 and 40 meters, depending on the slope gradient and water availability [35].
Natural vegetation patterns systematically differ from idealized model outputs by exhibiting greater irregularity in patch size and spacing [35]. This variation arises from exogenous spatial heterogeneities such as microtopography, spatially varying soil properties, and stochastic dispersal processes. The incorporation of such environmental noise produces model-generated patterns that more closely resemble natural systems, moving beyond the highly regular, periodic patterns generated in idealized homogeneous environments [35]. This recognition has led to the interpretation of regular vegetation patterns as random fields generated by stochastic processes where spatial noise is filtered through scale-dependent biophysical feedbacks.
Computational Protocols and Implementation
Core Mathematical Framework
The reaction-diffusion-advection system for vegetation patterning is built upon the Klausmeier-type model and its extensions, which capture the essential coupling between vegetation biomass and water dynamics. The fundamental structure consists of two coupled partial differential equations representing the temporal evolution of plant biomass and water resources [35] [36]. The vegetation equation incorporates diffusion representing local plant dispersal, a nonlinear growth term dependent on water availability, and a mortality term. The water equation includes diffusion representing soil water movement, advection accounting for downhill flow, a constant input representing rainfall, a nonlinear loss term due to vegetation consumption, and an evaporation term.
The numerical implementation requires spatial discretization of the domain, typically using finite difference methods for their simplicity and efficiency in handling reaction-diffusion systems [36]. For two-dimensional domains representing real landscapes, the spatial discretization must adequately resolve the characteristic pattern wavelength, generally requiring grid resolutions of 0.5-1 meter for patterns with 10-100 meter wavelengths. Temporal discretization generally employs explicit or semi-implicit methods, with care taken to satisfy stability conditions, particularly when handling the nonlinear reaction terms [36]. The advection term in the water equation requires specialized treatment, such as upwind differencing, to maintain numerical stability while minimizing artificial diffusion.
Advanced Computational Implementation
For large-scale simulations encompassing landscape-level domains, high-performance computing approaches are essential. The Julia programming language with CUDA.jl extensions provides an effective framework for GPU-accelerated solutions of these systems, combining high-level programming paradigms with efficient parallel execution [36]. This approach is particularly valuable for parameter studies and long-time simulations needed to observe steady-state pattern formation.
The implementation involves structuring the computation to maximize data locality and parallel execution. Memory management should minimize transfers between CPU and GPU, with entire simulation steps executed on the GPU where possible [36]. Kernel configuration should optimize thread block sizes and grid dimensions based on the specific GPU architecture and problem size. For the finite difference stencils used in discretizing diffusion and advection terms, shared memory utilization can significantly improve performance by reducing global memory accesses [36].
Modeling Workflow for Vegetation Pattern Simulation
Boundary conditions play a crucial role in determining pattern formation and localization. While classical models often employ no-flux (Neumann) boundary conditions, these can lead to patterns forming directly adjacent to boundaries, which may not reflect biological reality [37]. Alternative approaches implement mixed boundary conditions that enforce homogeneous conditions near boundaries, effectively isolating patterns within the interior domain [37]. This approach not only produces more biologically plausible patterns but also enhances robustness to initial conditions and reduces sensitivity to parameter variations.
Application Notes and Pattern Interpretation
Environmental Context and Pattern Significance
The morphology of vegetation patterns provides critical insights into ecosystem status and resilience. Isotropic patterns (spots or gaps) typically form on flat terrain, with spot patterns indicating lower water availability than gap patterns [35]. As environmental conditions change, particularly water availability, pattern morphology transitions between these states, providing visual indicators of ecosystem trajectory. Labyrinthine patterns represent intermediate states between spots and gaps and often signal critical transitions in ecosystem state [35].
Anisotropic stripe patterns form on sloped terrain, with stripes aligned parallel to elevation contours [35]. The spacing between stripes correlates with water availability and slope gradient, making them quantifiable indicators of ecosystem function. As aridity increases, stripe patterns typically move uphill, with changing stripe spacing and integrity providing early warning signals of desertification [35]. This directional response to environmental change makes striped patterns particularly valuable for monitoring ecosystem resilience.
Field validation requires mapping pattern morphology and quantifying characteristic wavelengths across environmental gradients. Remote sensing platforms provide efficient pattern mapping at landscape scales, while ground truthing establishes correlations with soil moisture, vegetation health, and infiltration capacity [35]. The statistical analysis of natural patterns reveals systematic deviations from idealized models, particularly in the distribution of patch sizes and spacing, which reflect the influence of environmental heterogeneities [35].
Model Extensions and Heterogeneity Incorporation
Realistic pattern simulation requires moving beyond idealized homogeneous environments to incorporate spatial heterogeneities. These heterogeneities include microtopography, spatially varying soil properties, stochastic seed dispersal, and localized disturbance [35]. Implementation involves introducing spatially variable parameters rather than homogeneous fields, with correlation structures reflecting measured environmental variation.
The peridynamic framework offers an alternative approach for handling heterogeneous media and evolving interfaces [38]. This nonlocal continuum theory replaces spatial derivatives with integral operators, naturally accommodating discontinuities and long-range interactions that challenge classical partial differential equation approaches [38]. While computationally more intensive, peridynamic models provide enhanced capability for simulating pattern evolution in complex, heterogeneous landscapes with evolving material interfaces.
Table 3: Research Reagent Solutions for Vegetation Pattern Modeling
| Tool/Category | Specific Implementation | Function/Purpose |
|---|---|---|
| Programming Languages | Julia with CUDA.jl [36] | High-performance computing with GPU acceleration |
| Spatial Discretization | Finite Difference Methods [36] | Numerical solution of partial differential equations |
| Spectral Analysis | Physics-Informed Neural Networks (PINNs) [39] | Stability analysis and eigenvalue computation |
| Nonlocal Modeling | Peridynamic Framework [38] | Handling heterogeneous media and evolving interfaces |
| Parameter Estimation | Amplitude-Based Inverse Methods [34] | Recovery of system parameters from observed patterns |
| Boundary Conditions | Mixed/Robin Conditions [37] | Pattern isolation away from domain boundaries |
Protocol: Implementing a Vegetation Patterning Simulation
System Setup and Parameterization
Begin by defining the spatial domain representing the landscape of interest. For initial investigations, a 500m × 500m domain with a grid resolution of 1m provides a reasonable balance between computational expense and pattern resolution. Specify the topographic characteristics, starting with a flat domain for isotropic patterns or incorporating a uniform slope (2-5% grade) for anisotropic stripe patterns. Set initial conditions as the homogeneous vegetated state with small random perturbations (1-5% amplitude) to seed pattern formation.
Parameter assignment should follow established values from the literature, with adjustments based on specific ecosystem characteristics. Critical parameters include vegetation growth rate (0.1-1.0 yr⁻¹), mortality rate (0.2-0.8 yr⁻¹), water diffusion coefficient (0.1-1.0 m²/yr), plant dispersal coefficient (0.01-0.1 m²/yr), and rainfall rate (100-800 mm/yr) [35]. Implement these parameters with a table-driven approach to facilitate parameter studies and sensitivity analysis. The computational implementation should log all parameter values alongside simulation outputs to ensure reproducibility.
Numerical Solution and Analysis
Implement the finite difference discretization with a grid spacing Δx = 1m and time step Δt determined by stability conditions. For explicit methods, the time step must satisfy Δt < Δx²/(2D) where D is the largest diffusion coefficient in the system. Solve the system iteratively, monitoring for convergence to steady-state patterns. Most vegetation patterning systems require simulation of 100-1000 years of model time to reach steady state, depending on parameters and initial conditions.
Analyze the resulting patterns by computing spatial power spectra to identify dominant wavelengths [35]. Compare pattern morphology with known classifications (spots, labyrinths, stripes) and quantify patch size distributions and spatial correlations. For anisotropic patterns, compute orientation order parameters to quantify directional alignment. Validate patterns against known analytical results for idealized cases before proceeding to heterogeneous scenarios. Incorporate environmental heterogeneity by introducing spatially varying rainfall or soil properties and observe how this modifies pattern regularity and morphology [35].
Biophysical Feedback Loops in Vegetation Patterning
Concluding Remarks and Future Directions
Reaction-diffusion-advection systems provide a mechanistic framework for understanding large-scale vegetation patterning, linking local biotic-abiotic interactions to emergent landscape-scale organization. The strength of this approach lies in its ability to generate testable predictions about ecosystem responses to environmental change and its capacity to identify critical transitions in ecosystem state. The integration of computational modeling with empirical observation continues to refine our understanding of these fascinating patterns.
Future developments in this field will likely focus on enhanced incorporation of environmental heterogeneity, multiscale interactions, and improved parameter estimation techniques. Approaches leveraging physics-informed neural networks show promise for spectral analysis and parameter recovery [39] [34], while peridynamic frameworks offer new capabilities for modeling complex media and evolving interfaces [38]. As computational power increases through exascale computing and GPU acceleration, high-resolution simulations spanning broader environmental gradients will become feasible, further strengthening the bridge between theoretical models and empirical observations of ecosystem patterning.
The classic activator-inhibitor framework, characterized by a short-range activating molecule and a long-range inhibiting molecule, has long been the dominant model for explaining self-organized pattern formation in biology via Turing mechanisms [9] [8]. However, the scarcity of experimentally verified molecular systems conforming to this classic intuition starkly contrasts with the ubiquity of periodic patterns in nature, suggesting our understanding of pattern-enabling networks is incomplete [9].
Recent research reveals that the capacity for spontaneous pattern formation is far more widespread than previously assumed. A paradigm shift is underway, moving beyond the requirement for imposed feedback loops and pre-assigned activator/inhibitor roles. Systematic analyses show that elementary biochemical networks, describing common post-translational interactions like sequential protein binding and complex formation, can generate robust Turing patterns without being designed with the classic intuition in mind [9]. These findings significantly expand the universe of possible pattern-forming circuits and provide new lenses through which to study morphogenesis in plants and other organisms.
Quantitative Analysis of Emerging Motifs
A systematic computational investigation of 23 distinct biochemical reaction networks, based on mass-action kinetics and without assigned activator/inhibitor identities, revealed that nearly half are capable of producing Turing patterns [9]. The study focused on networks leading to the formation of 11 characteristic molecular complexes (e.g., dimers, trimers, hetero-complexes) with up to four subunits.
Table 1: Pattern-Forming Capability by Characteristic Complex Topology
| Complex Topology | Total Reaction Paths | Turing-Capable Paths | Key Pattern-Enabling Feature |
|---|---|---|---|
| Homodimer (AA) | 1 | 1 | Regulated degradation |
| Heterodimer (AB) | 1 | 1 | Regulated degradation |
| Homotrimer (AAA) | 2 | 2 | Sequential binding with modified stability |
| Heterotrimer (AAB) | 4 | 2 | Asymmetric binding and degradation |
| Two-Heterodimer (AABB) | 3 | 1 | Multi-stage complex formation |
| Heterotetramer (ABCD) | 12 | 3 | High-order interaction complexity |
The unifying motif identified across many of these diverse, pattern-enabling networks is regulated degradation. In these systems, the altered degradation rate of monomers upon forming a complex—a common biochemical process—is a critical driver of instability, replacing the need for a dedicated, pre-imposed inhibitory feedback loop [9]. The simplest pattern-enabling reaction discovered requires only the formation of a trimer via sequential binding, coupled with flexibility in the diffusion rates of individual molecules [9].
Table 2: Key Parameters for Turing Patterning in a Model Trimer System
| Parameter | Symbol | Typical Range for Patterning | Biological Interpretation |
|---|---|---|---|
| Monomer Synthesis Rate | ( k_{sA} ) | 0.1 - 10 µM·s⁻¹ | Constitutive gene expression/production |
| Dimer Binding Rate | ( k_1 ) | 10² - 10⁴ M⁻¹·s⁻¹ | Affinity of monomer-monomer interaction |
| Trimer Binding Rate | ( k_2 ) | 10² - 10⁴ M⁻¹·s⁻¹ | Affinity of dimer-monomer interaction |
| Monomer Diffusion Coefficient | ( D_A ) | 1 - 10 µm²·s⁻¹ | Effective mobility in cytoplasm |
| Trimer Diffusion Coefficient | ( D{A3} ) | 0.1 - 1 µm²·s⁻¹ | Significantly slower due to larger size |
| Degradation Rate (Bound vs. Unbound) | ( \delta{A3} << \delta_A ) | Factor of 10-100 difference | Stabilization upon complex formation |
Application Notes & Experimental Protocols
The discovery of these widespread motifs provides new experimental avenues for probing pattern formation in plant developmental biology, from root hair patterning to venation and phyllotaxis.
Protocol: Computational Screening for Pattern-Forming Potential
This protocol outlines how to computationally assess if a plant biochemical network of interest has inherent pattern-forming capabilities [9].
- Network Abstraction: Map the biological system (e.g., a post-translational modification cascade or a small signaling network) into a set of biochemical reactions involving production, degradation, binding, unbinding, and diffusion. Use mass-action kinetics for all reactions.
- Model Formulation: Convert the reaction network into a system of partial differential equations (PDEs) describing the concentration change of each species over time and space:
∂C_i/∂t = D_i ∇²C_i + R_i(C₁, C₂, ...)whereC_iis concentration,D_iis diffusion coefficient, andR_iis the reaction term for speciesi. - Parameter Sampling: Perform a broad parameter scan. Sample rate constants and synthesis/degradation parameters from biologically plausible ranges (e.g., spanning two orders of magnitude). Diffusion coefficients should be sampled to allow for significant differences between monomers and larger complexes.
- Linear Stability Analysis (LSA): For each parameter set, calculate the homogeneous steady state. Perform LSA on the PDE system to identify parameters where the steady state is stable in the absence of diffusion but becomes unstable when diffusion is added—a hallmark of a Turing instability.
- Numerical Simulation: For parameter sets passing LSA, run 2D or 3D numerical simulations of the full PDE system with a finite-element or finite-volume method to confirm the emergence of stable, stationary patterns from random initial conditions.
Diagram 1: Computational screening workflow for identifying Turing-capable networks.
Protocol: Validating a Regulated Degradation MotifIn Planta
This protocol describes a wet-lab approach to test a predicted pattern-forming circuit, such as a trimer-based system, in a plant model system like Arabidopsis thaliana.
Construct Design:
- Clone genes for proteins A, B, and C (putative complex components) under a constitutive promoter.
- Fuse each gene to sequences encoding fluorescent proteins (e.g., GFP, RFP, mCherry) for visualization.
- Engineer mutant versions of protein A that disrupt key binding domains, designed to act as non-complexing controls.
Plant Transformation and Selection:
- Transform constructs into appropriate plant lines (e.g., Arabidopsis wild-type or relevant mutant backgrounds) using Agrobacterium-mediated floral dip.
- Select stable transformants on antibiotic plates and confirm transgene integration via PCR and expression via confocal microscopy.
Patterning Assay:
- Image the expression and localization of fluorescently tagged proteins in the root tip or leaf primordia using confocal microscopy.
- Quantify the emergence of periodic patterns (stripes, spots) in the fluorescence channels.
Perturbation Analysis:
- Treat seedlings with drugs that disrupt the cytoskeleton or membrane trafficking to indirectly alter effective diffusion rates of the proteins.
- Use tissue-specific inducible promoters to transiently overexpress a dominant-negative version of one component and observe the disruption of the pattern.
Biophysical Validation:
- Perform Fluorescence Recovery After Photobleaching (FRAP) to experimentally measure the in vivo diffusion coefficients of the monomeric and complexed proteins.
- Use Co-Immunoprecipitation (Co-IP) followed by mass spectrometry to confirm the physical interaction and formation of the predicted multi-protein complex.
Diagram 2: Experimental validation workflow for a putative patterning motif in plants.
The Scientist's Toolkit
Table 3: Essential Research Reagents and Computational Tools
| Category / Item | Function / Description | Application in Protocol |
|---|---|---|
| Biological Reagents | ||
| Fluorescent Protein Fusions | Visualizing protein localization and dynamics in vivo | Protocol 3.2, Step 1 & 3 |
| Dominant-Negative Mutants | Disrupting specific protein-protein interactions | Protocol 3.2, Step 4 |
| Inducible Promoter Systems | Spatiotemporally controlled gene expression | Protocol 3.2, Step 4 |
| Analytical Tools | ||
| FRAP (Fluorescence Recovery After Photobleaching) | Measuring protein mobility and effective diffusion coefficients in vivo | Protocol 3.2, Step 5 |
| Co-Immunoprecipitation (Co-IP) | Confirming physical interactions within protein complexes | Protocol 3.2, Step 5 |
| Computational Resources | ||
| PDE Solvers (e.g., FEniCS, COMSOL) | Numerical simulation of reaction-diffusion models | Protocol 3.1, Step 5 |
| Parameter Continuation Software (e.g., AUTO, MATCONT) | Tracking steady states and bifurcations in ODE/PDE systems | Protocol 3.1, Step 4 |
| Plant Models | ||
| Arabidopsis thaliana | Primary model organism for plant developmental genetics | Protocol 3.2 |
The discovery that Turing patterns can arise from widespread, simple biochemical circuits like regulated degradation pathways represents a significant expansion of the theoretical basis for morphogenesis [9]. This shift away from the necessity of pre-defined activator-inhibitor pairs opens up new avenues for interpreting experimental data in plant development. Many observed genetic interactions and protein complexes, previously not considered in the context of self-organization, may contribute to patterning through these more generic and ubiquitous network motifs.
For the plant biology community, this means that the search for patterning mechanisms should be broadened. The focus can now legitimately include systematic surveys of post-translational interaction networks and protein complex dynamics, in addition to the traditional search for dedicated activator-inhibitor pairs. Integrating these broader theoretical principles with computational models that respect plant-specific contexts—such as cell wall constraints, auxin transport, and tissue mechanics—will be essential for fully unraveling the computational power of the biochemical networks that build plants [40] [8].
Leveraging Mass-Action Kinetics for Post-Translational and Post-Transcriptional Regulation Models
The application of mass-action kinetics to model post-translational modification (PTM) and post-transcriptional regulatory networks provides a fundamental framework for understanding the dynamic biochemical processes that underlie pattern formation in living systems. Within plant computational biology, these models are essential for bridging the gap between molecular mechanisms and emergent phenomena, such as Turing pattern formation, which governs the development of regular structures like leaf phyllotaxis and root hair spacing [8]. The principle of mass-action kinetics, which describes reaction rates as proportional to the product of reactant concentrations, enables the construction of ordinary differential equation (ODE) systems that accurately capture the dynamics of complex biochemical networks [41].
This protocol details the application of mass-action kinetics to model two critical regulatory systems: multisite phosphorylation networks as a canonical example of PTM and small RNA (sRNA)-mediated regulation as a key post-transcriptional mechanism. The rational parameterization theorem for multisite PTM systems demonstrates that steady-state modform concentrations can be represented as rational functions of enzyme concentrations, enabling a dramatic reduction in system complexity from exponentially many equations to a tractable algebraic system [42]. Similarly, for post-transcriptional regulation, kinetic modeling of sRNA-mRNA interactions reveals how co-transcriptional and post-transcriptional regulation shape gene expression dynamics, with implications for understanding regulatory hierarchies within stress response networks [43].
Theoretical Foundation
Mass-Action Kinetics for Biochemical Networks
The fundamental principle of mass-action kinetics states that the rate of an elementary chemical reaction is proportional to the product of the concentrations of the reactants. For a generalized reaction:
[ \text{aA + bB} \xrightarrow{k} \text{cC + dD} ]
the reaction rate ( r ) is given by ( r = k [A]^a [B]^b ), where ( k ) is the rate constant, and ( [A] ), ( [B] ) represent concentrations. In the context of post-translational modification systems, this principle applies to enzyme-substrate interactions, such as phosphorylation and dephosphorylation cycles [41].
For a basic phosphorylation/dephosphorylation mechanism:
[ S0 + K \underset{k2}{\stackrel{k1}{\rightleftharpoons}} S0K \xrightarrow{k3} S1 + K \quad \text{and} \quad S1 + F \underset{k5}{\stackrel{k4}{\rightleftharpoons}} S1F \xrightarrow{k6} S0 + F ]
the corresponding ODE system under mass-action kinetics is:
[ \begin{align} \frac{d[S_0]}{dt} &= -k_1[S_0][K] + k_2[S_0K] + k_6[S_1F] \ \frac{d[K]}{dt} &= -k_1[S_0][K] + k_2[S_0K] + k_3[S_0K] \ \frac{d[S_0K]}{dt} &= k_1[S_0][K] - (k_2 + k_3)[S_0K] \ \frac{d[S_1]}{dt} &= k_3[S_0K] - k_4[S_1][F] + k_5[S_1F] \ \frac{d[F]}{dt} &= -k_4[S_1][F] + k_5[S_1F] + k_6[S_1F] \ \frac{d[S_1F]}{dt} &= k_4[S_1][F] - (k_5 + k_6)[S_1F] \end{align} ]
These equations form the foundation for modeling the dynamics of PTM networks, with conservation laws for total substrate and enzyme concentrations providing additional constraints [41].
Connection to Turing Pattern Formation
Turing patterns arise from reaction-diffusion systems where a short-range activator and long-range inhibitor interact to generate spontaneous pattern formation [8]. The classic Turing system requires:
- Reaction Kinetics: Nonlinear interactions between morphogens, typically modeled using mass-action or Michaelis-Menten kinetics
- Differential Diffusion: The inhibitor diffuses significantly faster than the activator
In plant systems, PTM networks and post-transcriptional regulation can provide the necessary nonlinear kinetics for Turing pattern formation. For example, phosphorylation cascades in MAPK signaling can exhibit bistability and ultrasensitivity, creating the nonlinear responses essential for pattern generation [41]. Similarly, sRNA-mediated regulatory networks can create sharp threshold responses through coupled degradation mechanisms [43].
The integration of mass-action-based regulatory networks with diffusion processes enables the modeling of plant developmental patterns, from ROP protein patterning in single cells to phyllotaxis and leaf venation [8].
Application Note 1: Modeling Multisite Phosphorylation Systems
Background and Significance
Multisite phosphorylation represents a ubiquitous PTM mechanism in eukaryotic cells, enabling sophisticated signal processing and encoding capabilities beyond simple binary switches. Proteins with multiple modification sites can exist in an exponential number of modification states (modforms), creating combinatorial complexity that challenges conventional modeling approaches [42]. For a protein with ( n ) sites, there are ( 2^n ) possible modforms, creating ( 2^n + P + L ) state variables in a full mass-action model (where ( P ) represents enzyme-substrate complexes and ( L ) represents enzymes) [42].
Protocol: Rational Parameterization Approach
Objective: To determine steady-state concentrations in a multisite phosphorylation system without numerical integration of the full ODE system.
Theoretical Basis: The Rational Parameterization Theorem states that steady-state modform concentrations in a multisite PTM system can be expressed as rational functions of the free enzyme concentrations, with coefficients that are rational functions of the rate constants [42]. This enables reduction of the system from ( L + N + P ) differential equations to ( L ) algebraic equations, where ( L \ll N, P ).
Procedure:
System Definition:
- Define all modification sites and their possible states (e.g., phosphorylated/unphosphorylated)
- Identify all forward and reverse enzymes and their specificities
- Specify all elementary reactions with their mass-action rate constants
Intermediate Complex Elimination:
- Express enzyme-substrate complex concentrations in terms of free enzyme and substrate concentrations using the Matrix-Tree theorem [42]
- For each enzyme ( Ei ), the concentration of complex ( Cj ) is given by: [ [Cj] = \frac{\sum{\text{trees } T} \kappa(T) [Ei] [Sk]}{\sum_{\text{trees } T'} \kappa(T')} ] where ( \kappa(T) ) represents the tree constant (product of rate constants) for directed spanning tree ( T )
Steady-State Parameterization:
- Express each modform concentration as a rational function of free enzyme concentrations
- For a dual-site phosphorylation system with processive phosphorylation and distributive dephosphorylation (Equation 3 from [41]), the parameterization yields: [ [S2] = \frac{k3 k4 [K]^2 [S0]}{\Phi(K, F, k)} ] where ( \Phi ) is a polynomial function of enzyme concentrations and rate constants
Conservation Law Application:
- Apply conservation laws for total substrate and enzymes: [ \begin{align} K_{\text{tot}} &= [K] + [S_0K] + [S_1K] \ F_{\text{tot}} &= [F] + [S_2F] + [S_1F] \ S_{\text{tot}} &= [S_0] + [S_1] + [S_2] + [S_0K] + [S_1K] + [S_2F] + [S_1F] \end{align} ]
System Solution:
- Solve the ( L ) algebraic equations for free enzyme concentrations using numerical methods
- Back-substitute to obtain all modform concentrations at steady state
Validation:
- Verify that the reduced system exhibits the same steady states as the full ODE system
- Confirm that the parameterization satisfies all original steady-state equations
- Check conservation laws are maintained throughout the computation
Table 1: Key Parameters for Multisite Phosphorylation Models
| Parameter | Description | Estimation Method | Typical Range |
|---|---|---|---|
| ( k_{\text{cat}} ) | Catalytic rate constant | Enzyme assays | 0.1-100 s⁻¹ |
| ( K_M ) | Michaelis constant | Steady-state kinetics | 1-100 μM |
| ( k_{\text{on}} ) | Binding rate constant | Surface plasmon resonance | 10⁵-10⁷ M⁻¹s⁻¹ |
| ( k_{\text{off}} ) | Dissociation rate constant | Surface plasmon resonance | 0.1-100 s⁻¹ |
| ( [E]_{\text{tot}} ) | Total enzyme concentration | Quantitative Western blot | 0.01-1 μM |
| ( [S]_{\text{tot}} ) | Total substrate concentration | Quantitative proteomics | 0.1-10 μM |
Workflow Visualization
Application Note 2: Post-Transcriptional Regulation by Small RNAs
Background and Significance
Small non-coding RNAs (sRNAs) mediate post-transcriptional regulation in both prokaryotes and eukaryotes, affecting mRNA stability and translation efficiency. In bacteria, sRNAs such as SgrS and RyhB in E. coli regulate critical stress response pathways through base-pairing interactions with target mRNAs [43]. Recent studies reveal that sRNA regulation can occur co-transcriptionally, as soon as the sRNA binding site is transcribed, leading to Rho-dependent termination and premature transcription termination [43].
Protocol: Kinetic Modeling of sRNA Regulation
Objective: To develop a mass-action kinetic model for sRNA-mediated post-transcriptional regulation that captures both co-transcriptional and post-transcriptional mechanisms.
Theoretical Basis: The model incorporates transcription, translation, degradation processes, and sRNA-mRNA interactions through mass-action kinetics. Key reactions include sRNA-mRNA binding, translation inhibition, and active/passive degradation mechanisms [43] [44].
Procedure:
Reaction Scheme Definition:
- Transcription: ( \varnothing \xrightarrow{\alpha_m} mRNA )
- Translation: ( mRNA \xrightarrow{k_x} mRNA + Protein )
- Endogenous mRNA degradation: ( mRNA \xrightarrow{\beta_m} \varnothing )
- Protein degradation: ( Protein \xrightarrow{\beta_p} \varnothing )
- sRNA transcription: ( \varnothing \xrightarrow{\alpha_s} sRNA )
- sRNA degradation: ( sRNA \xrightarrow{\beta_s} \varnothing )
- sRNA-mRNA binding: ( sRNA + mRNA \underset{k{\text{off}}}{\stackrel{k{\text{on}}}{\rightleftharpoons}} Complex )
- Translation of bound mRNA: ( Complex \xrightarrow{k_{xs}} Complex + Protein )
- Active degradation: ( Complex \xrightarrow{\beta_e} \varnothing )
- Translation-coupled degradation: ( Complex \xrightarrow{\beta_{ms}} \varnothing )
ODE System Construction: [ \begin{align} \frac{d[mRNA]}{dt} &= \alpha_m - k_{\text{on}}[sRNA][mRNA] + k_{\text{off}}[Complex] - \beta_m[mRNA] \ \frac{d[sRNA]}{dt} &= \alpha_s - k_{\text{on}}[sRNA][mRNA] + k_{\text{off}}[Complex] - \beta_s[sRNA] \ \frac{d[Complex]}{dt} &= k_{\text{on}}[sRNA][mRNA] - k_{\text{off}}[Complex] - \beta_e[Complex] - \beta_{ms}[Complex] \ \frac{d[Protein]}{dt} &= k_x[mRNA] + k_{xs}[Complex] - \beta_p[Protein] \end{align} ]
Co-Transcriptional Regulation Extension:
- Incorporate nascent mRNA species: ( mRNA_{\text{nascent}} )
- Add sRNA binding to nascent transcripts: ( sRNA + mRNA{\text{nascent}} \rightleftharpoons Complex{\text{nascent}} )
- Include Rho-dependent termination: ( Complex_{\text{nascent}} \xrightarrow{\gamma} \varnothing )
Parameter Estimation:
Model Analysis:
- Identify steady states through nullcline analysis
- Perform sensitivity analysis to determine critical parameters
- Analyze regulatory hierarchy within sRNA regulons
Validation:
- Compare model predictions with single-cell imaging data
- Verify hierarchical regulation patterns across multiple targets
- Test predictions using sRNA overexpression and knockout strains
Table 2: Experimentally Determined Kinetic Parameters for SgrS Regulation
| Parameter | Description | Target: ptsG | Target: manXYZ | Units |
|---|---|---|---|---|
| ( k_{\text{on}} ) | Binding rate constant | 0.024 | 0.015 | min⁻¹molecule⁻¹ |
| ( k_{\text{off}} ) | Dissociation rate constant | 0.12 | 0.08 | min⁻¹ |
| ( \beta_{\text{ms}} ) | Translation-coupled degradation | 0.35 | 0.28 | min⁻¹ |
| ( \beta_e ) | Active degradation | 0.15 | 0.09 | min⁻¹ |
| ( k_x ) | Basal translation rate | 0.45 | 0.38 | min⁻¹ |
| ( k_{xs} ) | Translation rate when bound | 0.05 | 0.07 | min⁻¹ |
Workflow Visualization
Integration with Turing Pattern Formation
Bridging Molecular Regulation and Macroscopic Patterning
The integration of mass-action kinetic models for PTM and post-transcriptional regulation with reaction-diffusion systems provides a mechanistic foundation for understanding Turing pattern formation in plants. At the cellular level, ROP protein patterning exemplifies how GTPase cycles regulated by GEFs and GAPs (employing PTM mechanisms) can generate spontaneous symmetry breaking through reaction-diffusion dynamics [8].
Protocol: Incorporating Regulatory Networks into Turing Systems
Identify Network Motifs:
- Map PTM networks (phosphorylation cascades) or post-transcriptional circuits (sRNA-mRNA interactions) that exhibit necessary nonlinearities
- Test for bistability, ultrasensitivity, or oscillatory capabilities using mass-action models
Formulate Reaction-Diffusion System: [ \begin{align} \frac{\partial A}{\partial t} &= F(A, I) + D_A \nabla^2 A \ \frac{\partial I}{\partial t} &= G(A, I) + D_I \nabla^2 I \end{align} ] where ( F(A, I) ) and ( G(A, I) ) represent the reaction kinetics derived from mass-action models
Parameter Space Exploration:
- Use the rational parameterization approach to efficiently explore parameter space for multisite PTM systems [42]
- Identify regions supporting Turing instability through linear stability analysis
Pattern Selection Analysis:
- Determine critical wavelength from diffusion coefficients and kinetic parameters
- Compare predicted patterns with experimental observations in plant systems
Case Study: Phyllotaxis Patterning Modern models of phyllotaxis integrate auxin transport with PIN protein polarization, where phosphorylation dynamics of PIN proteins represent a critical PTM system [8]. The mass-action kinetics of PIN phosphorylation and dephosphorylation cycles influence the effective diffusion coefficients in the resulting reaction-diffusion system, ultimately controlling spiral and whorled patterns in plant meristems.
The Scientist's Toolkit
Table 3: Essential Research Reagents and Computational Tools
| Category | Item | Specification/Function | Example Applications |
|---|---|---|---|
| Experimental Systems | Nicotiana benthamiana | Plant chassis for transient expression | Reconstruction of biosynthetic pathways [45] |
| E. coli SgrS/RyhB systems | Bacterial sRNA regulation models | Kinetic parameter estimation [43] | |
| Molecular Biology | CRISPR/Cas9 systems | Genome editing for pathway engineering | Gene knockout/activation in metabolic pathways [45] |
| Fluorescent reporter fusions | Protein/mRNA quantification | Single-cell imaging of expression dynamics [43] | |
| Analytical Techniques | Single-molecule FISH | RNA quantification at single-cell level | sRNA and mRNA copy number determination [43] |
| LC-MS/GC-MS | Metabolite profiling | Validation of metabolic pathway outputs [45] | |
| Computational Tools | MATLAB/Python ODE solvers | Numerical integration of mass-action systems | Dynamic simulation of regulatory networks [41] |
| Graphviz | Network visualization | Pathway diagram generation (this protocol) | |
| Parameter estimation algorithms | Linear/nonlinear optimization | Kinetic parameter inference from experimental data [44] |
The application of mass-action kinetics to model post-translational and post-transcriptional regulation provides a powerful framework for understanding the biochemical basis of pattern formation in plant systems. The rational parameterization theorem for multisite PTM systems enables researchers to overcome the combinatorial explosion of modification states, while kinetic modeling of sRNA regulation reveals how hierarchical control emerges from molecular interactions. Integration of these detailed regulatory models with reaction-diffusion frameworks bridges molecular mechanism and macroscopic pattern, offering insights into Turing pattern formation in developing plant systems.
The protocols presented here provide practical methodologies for implementing these approaches, with specific applications to multisite phosphorylation networks and sRNA-mediated regulation. By combining computational modeling with experimental validation, researchers can elucidate how complex biological patterns emerge from simple biochemical rules governed by mass-action kinetics.
Overcoming Modeling Challenges: Parameter Identification, Stability, and Model Selection
The accurate identification of parameters in mathematical models is a fundamental challenge in computational biology, particularly in the study of Turing pattern formation in plants. Parameter identification constitutes an inverse problem where model parameters must be estimated from observed experimental data rather than predicting outcomes from known parameters (the forward problem). In developmental plant biology, these parameters typically govern reaction rates, diffusion coefficients, and interaction strengths between morphogens that collectively produce patterned outcomes such as root hair spacing, phyllotaxis, and vascular arrangement [40] [8].
The significance of robust parameter identification methods has grown alongside the increasing integration of computational modeling with experimental plant biology. Models have evolved from purely theoretical constructs to frameworks that genuinely guide experimental design, demanding higher standards of parameter accuracy and model credibility [40] [46]. This document outlines established and emerging methodologies for addressing parameter identification challenges, with particular emphasis on amplitude-based recovery techniques suitable for Turing pattern systems in plant research.
Computational Framework for Turing Pattern Formation
Theoretical Foundations of Turing Patterning
Turing patterns emerge from reaction-diffusion systems where two or more chemical species (morphogens), with different diffusion rates, interact in ways that destabilize homogeneous equilibrium, leading to spontaneous pattern formation. The core mechanism involves short-range activation coupled with long-range inhibition [8] [34]. In plant systems, these patterns manifest at multiple biological scales: from ROP protein patterning within single cells to epidermal patterning across cell layers and even vegetation patterns at ecosystem levels [8].
The standard mathematical formulation for Turing systems involves partial differential equations of the form:
where n and c represent morphogen concentrations, d_n and d_c their diffusion coefficients, χ the chemotactic sensitivity, and f and g kinetic interaction terms [34]. Parameter identification in these systems is particularly challenging due to the nonlinear interactions between parameters and their collective influence on emergent patterns.
Challenges in Parameter Identification for Plant Systems
Parameter identification in biological models presents several distinctive challenges:
- Multi-scale dynamics: Plant developmental processes integrate events across temporal scales from milliseconds (signaling) to weeks (organogenesis) and spatial scales from cellular to tissue levels [40].
- Feedback interactions: Regulatory interactions occur across all biological levels, with tissue-level mechanics feeding back on molecular signaling patterns [40].
- Environmental plasticity: Plant development exhibits exceptional sensitivity to environmental conditions, introducing additional variability in parameter values [40].
- Practical limitations: Experimental measurements are often restricted to boundary observations or limited sampling of internal states, resulting in mathematically ill-posed inverse problems where solutions may be non-unique or unstable [47].
Amplitude-Based Recovery Framework
Theoretical Basis
Traditional inverse problems in pattern formation have predominantly relied on non-biological data sources such as boundary measurements. However, a novel approach directly leverages physical observables from nature—specifically, the spatial amplitude profile of Turing patterns—to achieve complete parameter identification [34]. This amplitude-based framework utilizes the fact that the amplitude of periodic patterns encodes essential information about the underlying system parameters.
The methodology employs the spatial amplitude profile of a single pattern to simultaneously recover all system parameters, including wavelength, diffusion constants, and the full nonlinear forms of chemotactic and kinetic coefficient functions [34]. This represents a significant advancement over traditional approaches that require multiple pattern observations under different conditions.
Mathematical Formulation
For a generalized reaction-diffusion-advection system modeling population dynamics of motile microorganisms in response to chemotactic agents:
where n denotes microorganism density and c represents chemotactic agent concentration, the amplitude-based recovery method enables identification of:
- Diffusion coefficients
d_nandd_c - Chemotactic sensitivity function
χ(n,c) - Kinetic functions
f(n,c)andg(n,c)[34]
This approach has been successfully demonstrated on models of chemotactic bacteria, establishing a biologically grounded, mathematically rigorous paradigm for reverse-engineering pattern formation mechanisms [34].
Experimental Protocols and Methodologies
Protocol: Amplitude-Based Parameter Recovery for Root Patterning Systems
Objective: To identify parameters in a Turing patterning system from observed amplitude profiles of root epidermal patterning.
Materials and Equipment:
- Confocal microscopy system for live imaging of root development
- Fluorescent protein tags for morphogen visualization
- Image analysis software (e.g., ImageJ, MATLAB)
- Computational environment for numerical optimization (e.g., Python, MATLAB)
Procedure:
Pattern Imaging and Preprocessing
- Grow Arabidopsis seedlings under controlled conditions for 5-7 days
- Image root epidermal cells using confocal microscopy at 4-hour intervals over 48 hours
- Apply Gaussian filtering to reduce noise while preserving pattern structures
- Generate spatial amplitude profiles using Hilbert transform or similar techniques
Model Selection and Discretization
- Select appropriate reaction-diffusion model based on biological knowledge
- Discretize the computational domain to match imaging resolution
- Implement finite difference or finite element approximation of model equations
Parameter Recovery Optimization
- Define objective function quantifying difference between observed and simulated patterns
- Implement hybrid regularization combining monotonicity principles with truncated singular value decomposition (TSVD) [47]
- Apply ensemble Kalman inversion (EKI) for derivative-free optimization [48]
- Execute parameter search with biologically plausible bounds
Validation and Sensitivity Analysis
- Validate recovered parameters against held-out pattern observations
- Perform local sensitivity analysis to identify most influential parameters
- Conduct robustness tests with bootstrapped resampling of imaging data
Troubleshooting Tips:
- If pattern amplitude is weak, consider longer imaging intervals or enhanced fluorescence tags
- If optimization fails to converge, review parameter bounds based on biological constraints
- For unstable solutions, increase regularization strength or implement entropy conditions [49]
Protocol: Support Identification for Parameter Variations in PDE Systems
Objective: To recover spatial support of parameter variations in plant tissue models from boundary measurements.
Materials and Equipment:
- Experimental apparatus for boundary condition control
- Sensors for boundary measurement acquisition
- Computational resources for PDE-constrained optimization
Procedure:
Boundary Data Acquisition
- Apply controlled Neumann boundary conditions to tissue samples
- Measure Dirichlet boundary responses using appropriate sensors
- Repeat for multiple boundary condition configurations
Inverse Problem Formulation
- Model tissue as PDE system with unknown parameter distribution
- Define Neumann-to-Dirichlet operator mapping applied fluxes to measured responses
Support Reconstruction
Reconstruction Validation
- Compare reconstructed supports with experimental histology
- Quantify reconstruction accuracy using similarity metrics
- Assess robustness against measurement noise through multiple trials
Research Reagent Solutions
Table 1: Essential Research Reagents and Computational Tools for Turing Pattern Analysis in Plants
| Item | Function | Example Applications |
|---|---|---|
| Fluorescent protein tags (e.g., GFP, RFP) | Visualizing morphogen distribution and dynamics | Live imaging of auxin response factors; PIN protein localization [40] |
| Mass spectrometry proteomics | Quantifying protein abundance and modifications | Identifying pattern-forming morphogens; verifying model predictions [50] |
| CRISPR/Cas9 gene editing | Precise manipulation of genetic components | Testing model predictions by perturbing putative morphogens [50] |
| Physics-Informed Neural Networks (PINNs) | Solving forward and inverse problems for PDEs | Parameter estimation for reaction-diffusion models [49] |
| Weak-Form PINNs (WF-PINNs) | Handling solutions with steep gradients | Shock wave capture in Burgers equation; improving training stability [49] |
| Ensemble Kalman Inversion (EKI) | Derivative-free parameter optimization | Recovering elastic constants; solving ill-posed inverse problems [48] |
Data Presentation and Analysis
Table 2: Quantitative Parameters in Turing Pattern Models of Root Development
| Parameter | Biological Role | Typical Range | Identification Method | Sensitivity Ranking |
|---|---|---|---|---|
| Diffusion coefficient (D) | Morphogen mobility through tissue | 0.1–10.0 µm²/s [40] | Fluorescence recovery after photobleaching (FRAP) | High |
| Reaction rate (k) | Biochemical interaction strength | 10⁻³–10⁻¹ s⁻¹ [9] | Kinetic assays with purified components | High |
| Degradation rate (δ) | Morphogen turnover | 10⁻⁴–10⁻² s⁻¹ [9] | Cycloheximide chase experiments | Medium |
| Production rate (α) | Morphogen synthesis | 10³–10⁵ molecules/cell/h [9] | Metabolic labeling with quantitative MS | Medium |
| Cross-reaction coefficient | Activator-inhibitor interaction | 0.1–5.0 dimensionless [34] | Amplitude-based recovery from patterns | High |
Visualization Framework
Workflow Diagram: Amplitude-Based Parameter Recovery
Amplitude-Based Parameter Recovery Workflow: This diagram illustrates the iterative process of recovering model parameters from observed pattern amplitudes, combining experimental data with computational optimization through ensemble Kalman inversion (EKI).
Regulatory Network Diagram: Turing Pattern Formation
Turing Pattern Regulatory Network: This diagram shows the core regulatory interactions in Turing pattern formation, highlighting the activator-inhibitor system with trimer complex formation that enables regulated degradation pathways identified as a unifying motif for pattern formation [9].
Implementation Considerations for Plant Research
Model Selection and Simplification
Effective parameter identification requires careful balance between model complexity and identifiability. For plant Turing systems, consider these principles:
- Appropriate Simplification: When mRNA and protein expression domains significantly differ due to prolonged protein stability and movement, distinct equations for each are necessary. Otherwise, collapsing transcription and translation into a single process is justified [40].
- Feedback Incorporation: If intermediary steps in regulatory cascades contain consequential feedbacks or delays, these should be explicitly modeled rather than simplified [40].
- Minimal Essential Processes: Models should incorporate sufficient processes to allow patterns to dynamically unfold without pre-patterning key components [40].
Robustness and Sensitivity Analysis
Parameter identification should always be accompanied by rigorous sensitivity analysis:
- Local Sensitivity: Evaluate how small perturbations in each parameter affect pattern outcomes using partial derivatives or Monte Carlo sampling.
- Global Sensitivity: Assess parameter influences across their entire feasible ranges using methods like Sobol indices or Morris screening.
- Identifiability Analysis: Determine whether parameters can be uniquely identified from available data using structural or practical identifiability measures.
Model robustness should be demonstrated by showing that qualitatively similar behavior arises for moderate changes in parameter values [40].
Future Directions and Integration with Emerging Technologies
The field of parameter identification in plant pattern formation is rapidly evolving with several promising directions:
- Integration of Machine Learning: Combining mechanistic models with machine learning approaches can address data gaps and improve individual-level predictions [46] [51].
- Multi-omics Integration: Proteomic and transcriptomic data provide additional constraints for parameter identification, reducing solution space dimensionality [50].
- Language Models for Genomics: Emerging large language models applied to genomic sequences may help identify novel pattern-forming genes and regulatory elements [51].
- Community Standards: Development of shared benchmarks, standardized protocols, and FAIR data principles will enhance reproducibility and model credibility [46].
As these methodologies mature, parameter identification will transition from a specialized technical challenge to an integral component of the plant biology research cycle, enabling more predictive models of plant development and ultimately supporting applications in crop improvement and sustainable agriculture.
The computational modeling of Turing pattern formation has become an indispensable tool for understanding the mechanisms of self-organization in plant development, from epidermal patterning to organ spacing. A model's predictive power and biological relevance hinge critically on the plausible constraint of its kinetic parameters and diffusion coefficients. This Application Note provides a structured framework for establishing these constraints, integrating current research findings and practical methodologies to guide researchers in developing biologically credible models of Turing pattern formation in plant systems.
Quantitative Constraints for Biological Plausibility
Fundamental Diffusion Coefficient Relationships
The core requirement for Turing instability is a sufficient disparity in the diffusion rates of the interacting morphogens. Table 1 summarizes key quantitative relationships identified in theoretical and experimental studies.
Table 1: Key Quantitative Constraints for Turing Pattern Formation
| Parameter / Relationship | Quantitative Constraint / Typical Range | Biological System / Model Context |
|---|---|---|
| Diffusion Coefficient Ratio | Inhibitor diffusion > Activator diffusion [4] | Gierer-Meinhardt "rule of thumb" [4] |
| Specific Ratio Example | D~Lefty~ / D~Nodal~ ≈ 29 [52] | Nodal-Lefty synthetic mammalian circuit [52] |
| Enabling Motif | Regulated degradation with flexible diffusion [9] | 10 elementary biochemical reaction networks [9] |
| Alternative Mechanism | Complex formation with immobile component [4] | Effective diffusion reduction (e.g., ROP patterning) [4] |
| Critical Wavelength (λ~c~) | λ~c~ ∝ √(D~slow~ * D~fast~) [4] | General reaction-diffusion system [4] |
Rate Constant Ranges from Elementary Networks
A systematic study of 23 elementary biochemical reaction networks with mass-action kinetics reveals that Turing patterns can emerge from widespread, simple biochemical reactions, such as trimer formation with altered monomer degradation rates, without imposed feedback loops [9]. The parameter search in this study utilized biologically plausible ranges covering two orders of magnitude for reaction rate constants [9], providing a practical baseline for model constraint.
Experimental Protocols for Parameter Determination
Protocol: Establishing Diffusion Coefficients via Fluorescence Recovery After Photobleaching (FRAP)
Application: Quantifying the effective diffusion coefficients of fluorescently tagged proteins (e.g., CPC, GL3/EGL3 in Arabidopsis root epidermis) or other morphogens in plant tissues [18].
Workflow Diagram:
Procedure:
- Sample Preparation: Express a functional fusion of the protein of interest (e.g., CPC) with a fluorescent protein (e.g., GFP) in the transgenic plant. Mount living plant tissue (e.g., root tips) for confocal microscopy.
- Photobleaching: Define a small, precise Region of Interest (ROI) within a cell or tissue region. Apply a high-intensity laser pulse to fully bleach the fluorescence within the ROI.
- Recovery Imaging: Immediately after bleaching, capture a time-series of low-intensity laser images to monitor the fluorescence recovery within the bleached ROI due to the influx of unbleached molecules from the surrounding area.
- Data Analysis: Plot the fluorescence intensity recovery over time. Fit the recovery curve to an appropriate diffusion model to calculate the effective diffusion coefficient (D). This experimentally derived
Dprovides a direct constraint for computational models [18].
Protocol: Computational Pipeline for Parameter Space Exploration
Application: Identifying combinations of kinetic rate constants and diffusion coefficients that support Turing pattern formation in a proposed reaction network [9].
Workflow Diagram:
Procedure:
- Define ODE Model: Formulate the ordinary differential equations (ODEs) for the proposed reaction network using mass-action kinetics, excluding diffusion terms [9].
- Bifurcation Analysis: Systematically sample kinetic parameters (e.g., production, degradation, and binding rates) from biologically plausible ranges (e.g., spanning two orders of magnitude). Use numerical continuation tools to identify parameter sets that produce Hopf bifurcations, which can indicate a potential for pattern formation [9].
- Incorporate Diffusion: Extend the ODE model to a system of partial differential equations (PDEs) by adding Fickian diffusion terms for all mobile species. Sample diffusion coefficients, ensuring the critical ratio between components is satisfied [9] [52].
- Pattern Validation: Analyze the linear stability of the homogeneous steady state by calculating the dispersion relation. Finally, perform numerical simulations of the full PDE system over a 2D domain to confirm the emergence of stable, stationary Turing patterns [9] [52].
Case Study: The Arabidopsis Root Epidermis Patterning System
The Arabidopsis thaliana root epidermis is a premier model for studying Turing-pattern-like cellular organization, featuring an interspersed pattern of hair and non-hair cells [18]. A meta-Gene Regulatory Network (meta-GRN) model successfully recovers 28 mutant phenotypes and highlights the critical role of protein diffusion.
Signaling Pathway Diagram:
Key Constraints and Insights:
- Core Logic: The system operates on lateral inhibition. Cells in the N-position initially promote the formation of the MBW transcription activation complex, which promotes non-hair fate (via GL2) and also activates the production of the mobile inhibitor CPC [18].
- Critical Diffusion: CPC protein diffuses into neighboring H-position cells. There, it competes with WER to form an inhibitory complex (IC), which suppresses the MBW complex and promotes hair cell fate [18].
- Dynamic Feedback: The model explicitly simulates the diffusion of CPC and GL3/EGL3, creating a dynamic feedback loop with the GRN. This coupling is essential for the emergence of the correct spatial pattern and generates a 2D phenotypic landscape dependent on diffusion levels [18].
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Reagents for Experimental Validation
| Reagent / Resource | Function and Application in Patterning Research |
|---|---|
| FRAP-Compatible FPs (e.g., GFP, YFP) | Tagging proteins like CPC or Lefty to visualize and quantify their mobility and diffusion coefficients in vivo [18]. |
| Synthetic Gene Circuits | Engineered systems (e.g., Nodal-Lefty) in model organisms to test Turing network topologies and parameter constraints under controlled conditions [52]. |
| SBML (Systems Biology Markup Language) | Standardized, machine-readable format for encoding computational models, ensuring reproducibility and interoperability [53]. |
| MIRIAM Guidelines | Minimum Information Requested in the Annotation of Models; a standard for annotating models with critical metadata to enhance credibility and reuse [53]. |
| CURE Principles | A framework for developing Credible, Understandable, Reproducible, and Extensible computational models in biology [54]. |
| Weakly Nonlinear Analysis (WNL) | Mathematical method used to determine the nature (supercritical vs. subcritical) of a Turing bifurcation, crucial for understanding pattern selection [52]. |
The credible computational modeling of Turing patterns in plants depends on rigorously constraining parameters using biological evidence. Adhering to quantified diffusion ratios, employing structured computational pipelines, utilizing standard model annotation, and integrating experimental data are all critical for developing models that are not only mathematically sound but also biologically plausible. The protocols and constraints outlined here provide a actionable path for researchers to enhance the predictive power and reliability of their models in developmental biology.
Distinguishing True Turing Patterns from Other Patterning Processes
The concept of Turing patterns, proposed by Alan Turing in 1952, revolutionised our understanding of how simple physical processes can generate complex biological patterns through a mechanism known as reaction-diffusion [8]. In plant biology, this framework has been applied to explain a diverse range of phenomena, from the spots and stripes on petals to the spacing of leaves and root hairs [5] [8]. However, the mere presence of a regular, repetitive pattern does not confirm that a Turing mechanism is at work. Several other processes, including mechanical stresses, directed transport, and genetic pre-patterning, can create similarly regular structures [8]. Misidentification of the underlying mechanism can lead to flawed experimental interpretations and an inability to effectively manipulate developmental outcomes. This Application Note provides a structured framework, combining computational and experimental approaches, to robustly distinguish true Turing patterns from other patterning processes in plant systems.
Core Principles of Turing Patterning
A Turing system arises from the interaction between at least two morphogens (or active molecular species): a self-activating, slowly-diffusing activator and a rapidly-diffusing inhibitor. The key is that diffusion, typically a homogenising force, destabilises a uniform steady state, leading to spontaneous pattern formation [8]. The classical "Gierer-Meinhardt" model encapsulates this as a requirement for short-range activation and long-range inhibition [8].
Contemporary research has expanded this classical view. It is now established that:
- Turing patterns can emerge from a wider family of biochemical networks than previously assumed, including those without an explicitly designated activator or inhibitor [9].
- Simple post-translational reactions, such as regulated degradation and trimer formation, are sufficient to generate robust patterns, making this highly relevant to protein and RNA-based patterning in plants [9].
- An optimal network size exists for the most robust Turing patterns, typically involving only a handful of molecular species, which balances stability and pattern-forming capability [55].
A Diagnostic Framework for Pattern Identification
The following workflow provides a step-by-step protocol for determining whether an observed biological pattern is likely generated by a Turing mechanism. This involves a combination of phenomenological observation, genetic perturbation, and computational validation.
Experimental Observation and Perturbation
Objective: To collect qualitative and quantitative data on pattern dynamics that can discriminate between mechanisms.
Protocol:
- Spatio-temporal Imaging: Perform live imaging of the patterning process from initial homogeneity to final patterned state. Use appropriate fluorescent reporters or in-situ hybridisation to track the spatial distributions of putative morphogens and their downstream targets.
- Domain Size Alteration: Measure the pattern (e.g., number of stripes or spots) in wild-type tissues or embryos of different sizes. A key prediction of a Turing system is that the number of pattern elements increases with the size of the field [8].
- Genetic/Perturbation Assays:
- Test for Regeneration: After surgical ablation or laser-mediated destruction of a pattern element (e.g., one spot), monitor whether the system regenerates the correct pattern. Turing systems often exhibit regeneration capabilities.
- Inhibit Diffusion: Genetically or chemically perturb putative diffusible signals (e.g., by altering cell-to-cell connectivity via plasmodesmata). A Turing pattern should be highly sensitive to changes in effective diffusion rates.
- Modify Reaction Kinetics: Use inducible overexpression or knockdown of putative activator or inhibitor components. A Turing pattern should shift its wavelength or disappear entirely when the reaction parameters are altered.
Computational and Mathematical Validation
Objective: To build a mathematical model that can quantitatively recapitulate the observed patterning dynamics.
Protocol:
- Model Formulation: Construct a reaction-diffusion model based on the hypothesized molecular network. Use mass-action kinetics or other appropriate formalisms to define the reaction terms [9].
- Parameter Estimation: Use quantitative data from imaging and perturbation experiments to inform model parameters. Employ parameter sampling or optimisation algorithms to find parameter sets that produce patterns consistent with wild-type observations.
- Bifurcation Analysis: Use computational tools to analyse the model's Jacobian matrix and identify the parameter regions where Turing instability occurs [9] [55]. This step mathematically confirms the system's capability to form patterns via diffusion-driven instability.
- Predictive Validation: The most critical test is to use the calibrated model to predict the outcome of a novel genetic perturbation or environmental change not used in model fitting. A successful prediction strongly supports the Turing hypothesis.
The following diagram illustrates the core logic of this diagnostic framework.
Key Discriminatory Criteria in a Comparative Table
The table below summarizes the key characteristics that can help distinguish Turing patterns from other common patterning mechanisms.
Table 1: Diagnostic Criteria for Different Patterning Mechanisms
| Criterion | Turing Pattern | Instructive Patterning (e.g., Gradient) | Genetic Oscillator (e.g., Clock-and-Wavefront) | Mechanical Patterning |
|---|---|---|---|---|
| Initial State | Homogeneous, isotropic field | Pre-existing asymmetry or organiser | Homogeneous field with oscillatory capability | Homogeneous, mechanically coupled tissue |
| Role of Diffusion | Instigator: Destabilises homogeneity | Communicator: Transmits positional signal | Synchroniser: Coordinates phases | Not the primary driver |
| Response to Domain Size | Yes: Number of elements increases with size | No: Pattern scales with size | Possible: Number of segments may change | Variable: Can depend on mechanical constraints |
| Response to Tissue Cutting | Regeneration: Pattern re-forms correctly | Truncation: Partial pattern forms | Resets: Oscillations may re-synchronise | Altered: Stress fields are reorganized |
| Key Molecular Signature | Short-range activation, long-range inhibition [8] | Morphogen gradient with threshold-dependent responses | Genetic network with sustained oscillations | Force-sensing proteins (e.g., microtubules, auxin transporters) |
Case Study: Epidermal Patterning in Plant Roots
The regular spacing of root hair and non-hair cells in the Arabidopsis root epidermis is a classic example where a Turing mechanism has been proposed.
Background: A network of transcription factors, including WEREWOLF (WER), CAPRICE (CPC), and GLABRA2 (GL2), dictates cell fate. WER promotes non-hair fate and also activates its own inhibitor, CPC, which moves to neighbouring cells [5].
Application of the Diagnostic Framework:
- Observation: The pattern of hair and non-hair cells is regular and spaced. Live imaging shows cell fate commitment in a positioned manner.
- Domain Size Test: While difficult to test in the root due to fixed geometry, computational models of the WER/CPC network show that it can produce spaced patterns from homogeneity.
- Perturbation: Mutations that affect the mobility of CPC (the inhibitor) disrupt the pattern, consistent with a Turing mechanism where differential diffusion is critical [8].
- Computational Modeling: Mathematical models treating the WER/CPC/GL2 network as a reaction-diffusion system successfully recapitulate the wild-type pattern and predict mutant phenotypes, providing strong support for the Turing hypothesis in this context [5].
The signaling pathway for this system can be visualized as follows:
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Reagents for Studying Turing Patterns in Plant Systems
| Reagent/Material | Function/Application | Example Use in Patterning Research |
|---|---|---|
| Fluorescent Protein Reporters (e.g., GFP, RFP) | Live imaging of putative morphogen localization and dynamics. | Tagging transcription factors like WER or CPC to monitor their spatial expression and mobility in real-time [5]. |
| Inducible Promoter Systems (e.g., dexamethasone-inducible) | Spatio-temporally controlled gene expression. | Overexpressing an activator or inhibitor component at a specific time to test model predictions and observe pattern shifts. |
| Spatial Transcriptomics | Genome-wide profiling of gene expression while retaining spatial information. | Identifying novel candidate morphogens by mapping entire transcriptomes to pattern domains in developing organs. |
| Computational Modeling Software (e.g., MATLAB, Python with SciPy, COPASI) | Simulating reaction-diffusion equations and performing bifurcation analysis. | Building and analyzing mathematical models to test if a hypothesized network can produce a Turing instability [9] [55]. |
| Molecular Tools for Altered Mobility (e.g., size-variant GFP fusions, plasmodesmata mutants) | Experimentally manipulating diffusion rates of candidate molecules. | Testing the Turing requirement for differential diffusion by restricting mobility of a putative inhibitor. |
Distinguishing true Turing patterns is a non-trivial task that requires more than a superficial analysis of the final pattern. It demands a rigorous, iterative cycle of experimental observation and computational modeling. By applying the diagnostic framework outlined here—paying close attention to the system's response to domain size changes and perturbations, and by building predictive mathematical models—researchers can confidently identify Turing mechanisms. This clarity is fundamental for advancing our understanding of how plants build their complex and beautiful forms from simple, self-organizing principles.
The understanding of complex biological systems requires integrating processes across multiple spatial and temporal scales, from molecules to cellular functions, and from collections of cells to entire organisms. A fundamental challenge in biology is unraveling how the complex interactions between singular elements give rise to "emergent" properties at the system level [56]. Computational modeling has become an indispensable tool for integrating these different processes and spatio-temporal scales to investigate how their interplay determines developmental outcomes [5]. This integration is particularly crucial in plant biology, where developmental processes arise from the interplay between gene expression, cell-cell signaling, cell growth and division, and tissue mechanics across wide temporal and spatial ranges [5].
Multi-scale computational modeling in biology addresses the coupling of increasingly complex models at all levels of biology, producing integrated models across multiple spatial scales and physical processes [57]. These approaches are uniquely suited to bridge the gap between diverse processes such as gene expression, cell-cell signaling, and tissue mechanics, which unfold over different scales [5]. In plant systems, this multi-scale perspective has revealed how Turing mechanisms—reaction-diffusion systems first proposed by Alan Turing in 1952—can generate regular patterns across different biological scales, from intracellular protein patterning to organ spacing and whole vegetation patterns [8].
Theoretical Foundations: Turing Patterns in Biological Systems
Core Principles of Turing Pattern Formation
Alan Turing's seminal 1952 paper proposed that diffusion, typically considered an equalizing process, could spontaneously generate regular patterns when coupled with chemical reactions [8]. This reaction-diffusion theory requires at least two components with different diffusion rates: a slowly diffusing self-activating component (activator) and a rapidly diffusing inhibitory component (inhibitor) [8]. The fundamental principle involves short-range facilitation and long-range inhibition, creating a symmetry-breaking instability that transforms a homogeneous steady state into a patterned state [8].
The critical wavelength (𝑙𝑐) of emerging patterns depends on the diffusion coefficients, scaling with the square root of their product divided by a weighted sum [8]. Turing himself envisioned applications to plant science, discussing with botanist C.W. Wardlaw how these mechanisms might explain phenomena such as phyllotaxis (leaf arrangement) [8]. Modern implementations often extend beyond simple two-component systems to include more complex regulatory networks and transport mechanisms.
Recent Theoretical Advances
Recent research has revealed that Turing patterns can emerge from more widespread biochemical systems than previously recognized. A 2024 systematic study of 23 elementary biochemical networks found that ten simple reaction networks could generate Turing patterns without the imposed feedback loops traditionally considered essential [9]. These networks function through mass-action kinetics describing post-synthesis interactions applicable to most proteins and RNAs in multicellular organisms.
The study identified a unifying network motif enabling Turing patterns via regulated degradation pathways with flexible diffusion rate constants of individual molecules [9]. This represents a significant expansion beyond the classical activator-inhibitor framework and suggests that pattern-forming capabilities may be inherent in many basic biochemical systems, not just specialized regulatory circuits. The simplest pattern-enabling reaction requires only the formation of a trimer molecule via sequential binding, with altered degradation rate constants of monomers upon binding [9].
Multi-Scale Computational Modeling Approaches
Modeling Frameworks and Their Applications
Computational models for multi-scale biological integration employ diverse approaches tailored to specific scales and phenomena. The following table summarizes key modeling frameworks and their representative applications in plant biology:
Table 1: Computational Modeling Frameworks for Multi-Scale Plant Biology
| Modeling Framework | Spatial Scale | Temporal Scale | Biological Applications | Key Features |
|---|---|---|---|---|
| Reaction-Diffusion PDEs [58] | μm×10 to mm×10⁻¹ | s×10 to min | Morphogen field dynamics, chondrogenesis in limb buds [58] | Continuous concentration fields, pattern formation |
| Cellular Potts Model (CPM) [58] | μm | min | Cell dynamics, migration, shape changes [58] | Discrete cell representation, energy minimization |
| Gene Regulatory Networks (ODEs) [58] | μm×10 | min | Cell differentiation, signaling pathways [58] | Biochemical kinetics, state transitions |
| 3D Biomodelling Environment [58] | Subcellular to organ | s to days | Vertebrate limb development [58] | Hybrid approach, modular components |
| Root Development Models [5] | Molecular to tissue | Minutes to days | Plant root development, patterning [5] | Integrates processes across scales |
Protocol: Implementing a Multi-Scale Model for Developmental Patterning
Objective: To simulate pattern formation in a developing plant organ using a hybrid modeling approach combining continuous and discrete elements.
Materials and Computational Tools:
- CompuCell3D or similar multi-scale modeling environment [58]
- Numerical solver for partial differential equations (PDEs)
- Visualization software for 3D structures
- High-performance computing resources for parameter exploration
Methodology:
Model Specification:
- Define the spatial domain representing the developing organ (e.g., 3D rectangular domain for limb bud or plant primordium)
- Specify submodel components: molecular-scale reaction-diffusion, cellular dynamics, tissue mechanics
- Establish interfaces between interacting submodels with appropriate scale-bridging
Reaction-Diffusion Submodel Implementation:
- Formulate PDEs for morphogen dynamics: ∂[A]/∂t = Dₐ∇²[A] + Rₐ([A],[I]); ∂[I]/∂t = Dᵢ∇²[I] + Rᵢ([A],[I])
- Set diffusion coefficients (Dₐ, Dᵢ) with Dᵢ > Dₐ (typically 2-10x difference)
- Define reaction kinetics Rₐ, Rᵢ using mass-action or Michaelis-Menten formulations
Cellular Dynamics Implementation:
- Configure Cellular Potts Model with Hamiltonian: H = ∑₍ᵢ,ⱼ₎ J₍τ₍σ₍ᵢ₎₎,τ₍σ₍ⱼ₎₎₎(1-δ₍σ₍ᵢ₎,σ₍ⱼ₎₎) + λ(vσ - Vₜ)²
- Incorporate cell responses to morphogen concentrations via effective fields
- Implement cell division and differentiation rules based on local conditions
Simulation Protocol:
- Initialize system with small random perturbations around homogeneous steady state
- Execute Monte Carlo steps for CPM coupled with finite difference method for PDEs
- Implement dynamic time-stepping with error control
- Run multiple realizations to assess stochastic effects
Parameterization and Validation:
- Conduct parameter sensitivity analysis using Latin Hypercube sampling
- Compare simulation outputs with experimental pattern metrics (wavelength, element number, spatial arrangement)
- Validate model predictions through collaboration with experimental biologists
Troubleshooting:
- If patterns fail to emerge, verify that Turing instability conditions are satisfied for reaction-diffusion parameters
- For unstable simulations, reduce time step size and check boundary conditions
- If cellular patterns lack biological fidelity, adjust adhesion energies and target volumes in CPM
Experimental and Computational Protocols
Protocol: Analyzing 3D Plant Phenomics with Deep Learning
Objective: To extract quantitative phenotypic traits from 3D plant architecture using deep learning approaches.
Materials:
- 3D digitizer, LiDAR scanner, or multi-view stereo imaging system [59] [60]
- High-performance computing workstation with GPU acceleration
- Deep learning frameworks (PyTorch, TensorFlow)
- Plant seedlings or specific organs of interest
Methodology:
3D Data Acquisition:
- Capture multi-view images using controlled imaging system or UAV-based platform
- For indoor imaging: Use rotary stage with multiple camera angles under consistent lighting
- For field applications: Implement UAV cross-circling oblique imaging at 10-20m altitude [60]
- Apply Structure-from-Motion and Multi-View Stereo (SfM-MVS) algorithms for point cloud generation [60]
Point Cloud Preprocessing:
- Remove noise and outliers using statistical filtering (e.g., radius-based outlier removal)
- Apply Euclidean clustering for plant organ separation
- Implement color filtering and region-growing algorithms for stem and leaf segmentation [60]
- Use mesh subdivision and planar parameterization for area-preserving 2D model generation [60]
Deep Learning Model Implementation:
- Select appropriate network architecture based on task (classification, segmentation, detection)
- For organ segmentation: Implement PointNet++ or similar 3D convolutional networks
- Configure multi-scale feedforward networks (MSFN) to capture complex data features [61]
- Incorporate attention mechanisms (ECA, CA) to enhance feature discrimination [61]
Phenotypic Trait Extraction:
Validation and Analysis:
- Compare automated measurements with manual ground truth annotations
- Perform statistical analysis of phenotypic variations across genotypes or conditions
- Integrate extracted traits with growth models for predictive analysis
Table 2: Key Research Reagent Solutions for Multi-Scale Plant Modeling
| Category | Specific Tools/Reagents | Function/Application | Key Features |
|---|---|---|---|
| Computational Frameworks | CompuCell3D [58] | Multi-scale modeling environment | Object-oriented, modular, CPM-based |
| V-cell, BioSym [58] | Subcellular process modeling | Detailed biochemical networks | |
| Plant-specific root models [5] | Root development simulation | Incorporates mechanical and chemical signaling | |
| Imaging & Phenotyping | 3D digitizers, LiDAR [60] | Plant architecture capture | High-resolution spatial data |
| UAV-based hyperspectral imaging [60] | Canopy trait monitoring | Spectral + structural information | |
| Multi-view stereo systems [60] | 3D reconstruction | Cost-effective compared to LiDAR | |
| Data Analysis | Deep learning models (YOLO variants) [60] [61] | Plant organ detection | Real-time processing capabilities |
| Dual attention mechanisms [61] | Early plant classification | Enhanced feature discrimination | |
| Multi-scale modules [61] | Complex feature capture | Handles variable scales and resolutions | |
| Theoretical Foundations | Turing pattern analysis [8] [9] | Pattern formation prediction | Identifies minimal requirements for self-organization |
| Mass-action reaction networks [9] | Biochemical system modeling | No presupposed activator/inhibitor roles |
Case Studies: Multi-Scale Integration in Plant Systems
Turing Patterning at Multiple Biological Scales in Plants
Turing mechanisms operate across diverse scales in plant systems, as illustrated in these representative examples:
Table 3: Turing Patterns Across Scales in Plant Biology
| Biological Scale | System | Molecular Players | Pattern Type | Regulatory Features |
|---|---|---|---|---|
| Intracellular | ROP protein patterning [8] | Rho-of-Plants GTPases | Spots, stripes | Membrane association feedback |
| Tissue Level | Epidermal patterning [8] | Transcription factors, peptides | Hair spacing, stomatal spacing | Cell-to-cell signaling |
| Organ Level | Phyllotaxis (leaf arrangement) [8] | Auxin, PIN proteins | Spiral, whorled patterns | Polar transport with feedback |
| Organism Level | Vegetation patterns [8] | Water, nutrients | Bands, gaps | Resource-based feedback |
| Biochemical | Generic protein/RNA networks [9] | Various binding partners | All Turing pattern types | Regulated degradation pathways |
Case Study: Limb Bud Chondrogenesis Patterning
The development of cartilage patterns in embryonic vertebrate limbs provides an exemplary model of multi-scale integration. A simplified simulation implementation includes these components:
- Molecular Scale: Reaction-diffusion system for TGF-β and inhibitor dynamics
- Cellular Scale: CPM for cell adhesion, migration, and shape changes
- Tissue Scale: Accumulation of extracellular matrix (fibronectin)
- Organ Scale: Growth and shaping of the limb bud domain
This integrated model successfully reproduces the proximal-distal sequence of element formation: stylopod (humerus/femur), zeugopod (radius-ulna/tibia-fibula), and autopod (carpals/digits) [58]. The simulation demonstrates how generic physical mechanisms interacting with genetic regulation can generate complex biological structures.
Visualization of Multi-Scale Integration Concepts
Turing Pattern Formation Mechanism
Diagram 1: Turing Pattern Formation Process
Multi-Scale Modeling Workflow
Diagram 2: Multi-Scale Modeling Integration Workflow
Future Perspectives and Challenges
As multi-scale modeling continues to evolve, several key challenges and opportunities emerge. First, there is a growing need to develop new methods for coupling across the interface between stochastic and deterministic processes [57]. Second, efficient computation of increasingly complex models will require leveraging massively parallel computers and developing specialized algorithms [57]. Third, the integration of deep learning with multiscale modeling presents promising avenues for enhancing both predictive capability and computational efficiency [5].
In plant biology specifically, future work should focus on several critical areas: (1) constructing benchmark datasets using synthetic data and generative artificial intelligence; (2) developing accurate and efficient 3D point cloud analysis through multitask learning and self-supervised approaches; and (3) exploring model interpretability, extensibility, and multimodal data utilization in deep learning for 3D plant phenomics [59]. The ongoing convergence of experimental advances, computational power, and theoretical insights promises to unlock new dimensions in understanding plant development and physiology across scales.
The exploration of widespread biochemical reaction networks capable of Turing pattern formation suggests that many biological systems may have inherent pattern-forming capabilities that have been overlooked due to rigid adherence to the classical activator-inhibitor paradigm [9]. This expanded perspective opens new avenues for identifying patterning mechanisms in plant development and for engineering synthetic patterning systems in biotechnology applications.
The quest to understand Turing pattern formation in plants, from the intricate spots on a petal to the regular spacing of leaves (phyllotaxis), relies heavily on computational modeling. These models simulate the interplay of genetic regulation, hormone signaling, and mechanical forces across vast spatial and temporal scales. However, this complexity often results in mathematical models of such high dimension that they become computationally prohibitive for simulation, analysis, or parameter exploration. Model Order Reduction (MOR) provides a suite of techniques designed to lower the computational complexity of these high-fidelity models by creating accurate, low-dimensional approximations, known as reduced-order models. Their application is crucial for making feasible the multi-scale, feedback-driven simulations central to modern plant developmental biology [40] [62].
Core Model Reduction Techniques: A Comparative Analysis
Model reduction techniques can be broadly classified into several categories, each with distinct strengths for different aspects of Turing pattern research. The table below summarizes the primary methods.
Table 1: Key Model Order Reduction Techniques for Computational Biology
| Method Category | Key Principle | Primary Advantage | Typical Application in Pattern Formation |
|---|---|---|---|
| Projection-Based Methods (POD, Reduced Basis) [62] | Projects the high-dimensional system onto a lower-dimensional subspace spanned by an optimal basis (e.g., from simulation snapshots). | Strong theoretical foundations; well-suited for parameterized systems. | Simulating pattern formation under varying morphogen diffusion rates. |
| Balancing Methods [62] | Reduces models while preserving the input-output behavior most critical to the system's dynamics. | Maintains system stability and key properties; good for control analysis. | Studying the robustness of patterning to stochastic fluctuations in gene expression. |
| Simplified Physics [62] | Uses physical/biological insight to derive a less complex model via assumptions and simplifications. | Highly interpretable; can lead to mechanistic understanding. | Initial, conceptual models of activator-inhibitor dynamics. |
| Nonlinear & Manifold Learning (Autoencoders) [63] | Uses neural networks to learn a non-linear mapping between high-dimensional states and a low-dimensional latent space. | Can capture complex, non-linear dynamics more efficiently than linear methods. | Learning latent dynamics of complex reaction-diffusion systems from data. |
For plant patterning, the choice of method depends on the research question. While simplified physics models have been instrumental in building intuition—for instance, conceptualizing the "short-range activation, long-range inhibition" principle of Turing systems [8]—modern research increasingly leverages data-driven methods like Proper Orthogonal Decomposition (POD) and Deep Learning-based autoencoders to handle the non-linearities and large scales of realistic models [63].
Protocol for Implementing Reduced-Order Modeling of Turing Systems
This protocol outlines the steps for applying projection-based MOR to a reaction-diffusion model of plant Turing pattern formation.
Protocol: Proper Orthogonal Decomposition for a Reaction-Diffusion Patterning Model
Application Note: This methodology is ideal for reducing the complexity of a pre-defined, high-fidelity model of a Turing system (e.g., simulating epidermal patterning) to enable rapid parameter exploration [40] [62].
Materials and Software Requirements
- High-Fidelity Solver: A computational solver for your full reaction-diffusion system (e.g., a custom finite element method implementation in MATLAB, Python, or C++).
- Computational Environment: Software with linear algebra capabilities (e.g., Python with NumPy/SciPy, MATLAB).
- MOR Library: (Optional) Libraries such as pyMOR [62] can streamline the reduction process.
Procedure
- Full-Order Model (FOM) Definition and Snapshot Generation:
- Define your high-fidelity model, including the governing equations (e.g., reaction-diffusion PDEs for activator and inhibitor concentrations), parameters, domain, and boundary conditions.
- Run the FOM for a representative set of training parameters (e.g., different diffusion coefficients, domain sizes, reaction kinetics) that span the expected operating conditions of your study.
- Collect snapshots, which are numerical solutions of the FOM (e.g., concentration fields at discrete time steps) for each parameter configuration.
Basis Construction via Singular Value Decomposition (SVD):
- Assemble all snapshots into a matrix ( S ), where each column is a single snapshot.
- Perform the SVD on ( S ): ( S = U \Sigma V^T ).
- The columns of matrix ( U ) are the Proper Orthogonal Decomposition (POD) modes, which form an optimal basis for representing the snapshot data.
- Select the first ( r ) POD modes to form a reduced basis ( U_r ), truncating modes associated with singular values below a desired tolerance. This defines the dimension of your reduced model.
Projection to Form Reduced-Order Model (ROM):
- Let ( u ) represent the state vector of the FOM (e.g., concentrations at all grid points). Approximate the full state using the reduced basis: ( u \approx Ur ur ), where ( u_r ) is the reduced state vector of dimension ( r ).
- Project the FOM equations onto the reduced subspace ( Ur ). This is typically done via a Galerkin projection, which involves substituting the approximation into the FOM and ensuring the residual is orthogonal to the reduced basis. This process yields a system of ( r ) equations governing the evolution of ( ur ).
ROM Execution and Analysis:
- Solve the small ( r )-dimensional ROM for new parameter values or initial conditions. This is computationally much faster than solving the FOM.
- To reconstruct the full, high-dimensional solution for visualization, use the approximation ( u \approx Ur ur ).
Troubleshooting
- Poor ROM Accuracy: The training parameter space may be insufficient. Generate more snapshots across a wider range of parameters or consider a non-linear reduction method (e.g., autoencoders) if the system dynamics are highly non-linear [63].
- ROM Instability: The projection may not preserve the stability of the FOM. Consider using stability-preserving methods like Balanced Truncation [62].
Advanced Technique: Deep Learning-Based Non-Linear Reduction
For highly non-linear patterning systems where linear subspaces are insufficient, non-linear reduction using Deep Learning has emerged as a powerful tool. The core component is an autoencoder network, which learns to compress a high-dimensional system state into a low-dimensional latent code and then decode it back with minimal error [63].
Table 2: Components of a Deep Learning-Based Reduced Order Model
| Network Component | Function | Role in Patterning Simulation |
|---|---|---|
| Encoder (( f_e )) | Maps the full state ( \mathbf{s}t \in \mathbb{R}^{n^3} ) to a latent code ( \mathbf{c}t \in \mathbb{R}^{m} ), where ( m \ll n^3 ). | Compresses a complex concentration field (e.g., of ROP proteins [8]) into essential features. |
| Decoder (( f_d )) | Maps the latent code ( \mathbf{c}t ) back to an approximation of the full state ( \mathbf{s}{t+1} ). | Reconstructs the full patterning landscape from the latent representation for analysis. |
| Predictor (( f_p )) | A recurrent network (e.g., LSTM) that evolves the latent code in time: ( \mathbf{c}{t+1} = fp(\mathbf{c}_t) ). | Learns and predicts the dynamics of the pattern formation process directly in the efficient latent space. |
The learning objective for an end-to-end trained model is: [ \text{arg min}{\thetae,\thetap,\thetad} \| fd( fp( fe( \mathbf{s}{t} ;\thetae) ;\thetap) ;\thetad) - \mathbf{s}{t+1} \|_2^2 ] This approach allows the model to learn both a highly efficient non-linear representation of the state and a complex time-evolution law, moving beyond the limitations of linear Koopman operators [63].
Diagram 1: Deep Learning ROM Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Software and Libraries for Model Reduction
| Tool / Library | Language/Platform | Primary Function | Application Note |
|---|---|---|---|
| pyMOR [62] | Python | Building model order reduction applications, with a focus on parameterized PDEs. | Highly suitable for integrating with existing Python-based finite element solvers; ideal for structured MOR studies. |
| RBmatlab [62] | MATLAB | Reduced basis methods for linear and non-linear, parameterized evolution problems. | Comprehensive library for the reduced basis method; good for prototyping in a MATLAB environment. |
| Pressio [62] | C++/Python | Projection-based model reduction for large-scale codes using generic programming. | Designed for high-performance computing (HPC) environments; minimizes intrusiveness into existing C++ codes. |
| libROM [62] | C++ | Scalable and parallel methods for POD, dynamic mode decomposition, and hyper-reduction. | Excellent for large-scale, parallel snapshot generation and reduction of complex 3D models. |
| Custom Autoencoder Code [63] | Python/TensorFlow/PyTorch | Learning non-linear latent spaces and dynamics for complex systems. | Essential for applying the deep learning-based ROM approach outlined in Section 4. |
Visualizing Turing Pattern Formation Logic
The following diagram illustrates the core logic of a two-component Turing system, which underpins many models in plant pattern formation, and how reduced-order modeling interacts with this process.
Diagram 2: Turing Logic and Model Reduction
Validating and Contrasting Patterning Mechanisms: Turing vs. Alternative Hypotheses
The integration of computational modeling, particularly of Turing pattern formation, with robust experimental validation frameworks is fundamental to advancing modern plant research. Computational models that simulate pattern formation mechanisms, such as the reaction-diffusion systems proposed by Alan Turing, provide testable hypotheses for developmental processes like phyllotaxis and epidermal patterning [40] [8]. However, the inherent complexity of plant systems—spanning gene regulation, hormone signaling, and tissue mechanics—requires that these in silico predictions be rigorously tested in planta. This document details established and emerging protocols for validating developmental models, from initial phenotypic screening in mutants to the precise pathway engineering enabled by synthetic biology. The presented methodologies provide a critical bridge between theoretical pattern formation and empirical discovery, enabling researchers to decode the principles of plant development and engineer novel traits [40] [45].
Application Notes & Experimental Protocols
High-Throughput Phenotypic Screening of Mutants
Phenotype-based chemical screening offers a powerful, unbiased method to elucidate gene function and biological mechanisms by creating temporary, reversible perturbations, an advantage over conventional genetic knockout studies [64]. The following protocol describes a high-throughput screen designed to identify small molecules that cause differential growth in a DNA repair mutant (mus81) compared to the wild-type Arabidopsis thaliana [64].
Key Applications:
- Target Identification: Uncover genes and pathways involved in specific biological processes, such as DNA damage repair [64].
- Chemical Genetics: Discover genotype-specific small molecule probes that can conditionally alter plant growth or development [64].
- Model Validation: Generate empirical data on mutant phenotypes to constrain and refine computational models of plant development [40] [64].
Protocol: Differential Growth Chemical Screen
1. Experimental Design & Preparation
- Biological Materials: Arabidopsis thaliana wild-type (e.g., Col-0) and mutant seeds (e.g., mus81). Surface-sterilize seeds using standard ethanol and bleach protocols [64].
- Chemical Library: Utilize a curated library of small molecules, such as the off-patent Prestwick chemical library. Prepare stock solutions in DMSO and store at -20°C [64].
- Controls: Include internal controls on every plate:
- Negative Control: DMSO-only treatment (healthy seedling phenotype).
- Positive Control: Mitomycin C (MMC) treatment for the mus81 mutant (altered growth phenotype) [64].
- Plate Format: Use 24-well microtiter plates. A 96-well format is not recommended as it restricts seedling growth and complicates image acquisition [64].
2. Plant Growth & Chemical Treatment
- Sowing: Suspend sterilized seeds in sterile, liquid plant growth medium. Dispense into 24-well plates, ensuring three seeds per well to provide biological replicates [64].
- Stratification: Seal plates and place them at 4°C in the dark for 2-3 days to synchronize germination.
- Treatment & Growth: Add small molecules from the library to achieve a desired final concentration (e.g., 10 µM). Incubate plates under standard growth chamber conditions (e.g., 16h light/8h dark photoperiod at 22°C) for 10 days [64].
3. Image Acquisition & Machine Learning-Based Analysis
- Image Capture: Capture high-resolution images of each well using a light macroscope after the growth period [64].
- Image Classification with CNN:
- Tool: Train a Convolutional Neural Network (CNN), such as a Residual Neural Network (ResNet), to classify seedling images as "normal growth" or "altered growth" [64].
- Training Set: Use a dataset of several hundred images of wild-type and mutant seedlings treated with DMSO (normal) or MMC (altered). Allocate 80% for training, 10% for validation, and 10% for testing [64].
- Output: The model provides a probability score for each image, allowing for automated, high-accuracy phenotypic categorization [64].
- Image Segmentation for Quantification:
4. Hit Identification & Validation
- Data Analysis: Compare the growth (via CNN classification scores or segmented area measurements) of wild-type versus mutant seedlings for each chemical.
- Hit Criteria: Identify "hits" as molecules that induce a significant growth defect specifically in the mutant background while having little to no effect on the wild-type [64].
- Validation: Re-test initial hits in a secondary, dose-response assay to confirm genotype-specific activity [64].
The experimental workflow for this high-throughput screening method is summarized in the diagram below:
Integrating Omics and Genome Editing for Pathway Validation
Computational models of pattern formation often predict the involvement of specific genes and metabolic pathways. An integrative approach combining multi-omics data with CRISPR/Cas-based genome editing provides a powerful strategy for the functional validation of these model predictions and the engineering of traits [45] [65].
Key Applications:
- Pathway Discovery: Identify candidate genes within complex biosynthetic pathways for plant natural products (PNPs) [45] [65].
- Functional Validation: Test the in planta role of genes predicted by models or omics data through targeted knockout, activation, or fine-tuning [45].
- Metabolic Engineering: Reprogram plant hosts to enhance the production of valuable compounds or introduce novel biosynthetic capabilities, providing a direct test of pathway understanding [66] [45].
Protocol: CRISPR/Cas-Mediated Pathway Engineering
1. Omics-Driven Candidate Gene Identification
- Genomics/Transcriptomics: Sequence the genome and/or transcriptome of the target plant species under conditions that induce the pathway of interest. Perform co-expression analysis to identify gene clusters [45] [65].
- Metabolomics: Profile secondary metabolites (e.g., using LC-MS or GC-MS) to correlate metabolite accumulation with gene expression patterns [45].
- Bioinformatics: Reconstruct putative biosynthetic pathways using systems biology and bioinformatics tools [45] [65].
2. Vector Design and Assembly
- gRNA Design: Design and clone 20-nt guide RNA (gRNA) sequences specific to the target gene(s) into a CRISPR/Cas9 binary vector. For multiplexed editing, assemble multiple gRNA expression cassettes in a single vector [45].
- Cas9 Selection: Use a plant-codon-optimized Cas9 nuclease driven by a constitutive promoter (e.g., CaMV 35S) [45].
- Transformation: Mobilize the final construct into Agrobacterium tumefaciens strain GV3101 for plant transformation [45].
3. Plant Transformation and Regeneration
- Host Selection: Use a tractable plant system. Nicotiana benthamiana is ideal for transient expression, while stable transformation can be performed in Arabidopsis, rice, or tomato [45] [65].
- Transformation: For stable transformation, use Agrobacterium-mediated transformation of leaf explants or floral dip (for Arabidopsis). For transient assays, infiltrate young N. benthamiana leaves with the Agrobacterium culture [45] [65].
- Regeneration and Selection: Transfer transformed explants to selection media containing appropriate antibiotics (e.g., kanamycin) to regenerate transgenic plantlets. Confirm gene editing events via DNA sequencing of the target locus [45].
4. Phenotypic and Metabolomic Validation
- Genotype Analysis: Sequence the target genomic region in T0 or T1 plants to identify insertion/deletion (indel) mutations and calculate editing efficiency [45].
- Phenotypic Screening: Assess mutant plants for expected developmental or physiological phenotypes.
- Metabolite Quantification: For metabolic engineering, use LC-MS/MS to quantify the target metabolite in transgenic lines. For example, editing glutamate decarboxylase genes in tomato increased GABA accumulation by 7- to 15-fold [45] [65].
The workflow for this integrative approach is detailed in the diagram below.
The Scientist's Toolkit: Research Reagent Solutions
The following table catalogues essential reagents, tools, and platforms critical for executing the protocols described in this document.
Table 1: Key Research Reagent Solutions for Plant Experimental Validation
| Item | Function & Application | Example Use Cases |
|---|---|---|
| Prestwick Chemical Library | A library of off-patent, bioavailable small molecules for phenotype-based screening [64]. | Identifying genotype-specific chemical regulators of plant growth in a high-throughput differential screen [64]. |
| CRISPR/Cas9 Systems | RNA-guided genome editing technology for targeted gene knockout, activation, or fine-tuning [66] [45]. | Validating gene function in metabolic pathways (e.g., editing GAD genes in tomato to increase GABA) [45] [65]. |
| Nicotiana benthamiana | A model plant host for transient gene expression due to high transformation efficiency and rapid biomass production [45] [65]. | Rapid reconstruction and validation of biosynthetic pathways for plant natural products via agroinfiltration [45] [65]. |
| Convolutional Neural Networks (CNNs) | Deep learning algorithms for automated, high-accuracy image classification and segmentation [64]. | Quantifying seedling growth and classifying phenotypes in high-throughput chemical screens [64]. |
| Protocol Databases (e.g., Bio-protocol, Cold Spring Harbor Protocols) | Repositories of peer-reviewed, detailed life science protocols [67]. | Providing standardized, reproducible methodologies for plant transformation, imaging, and molecular analysis [67]. |
Data Presentation and Quantitative Analysis
Robust data presentation is key to interpreting validation experiments. Below are templates for summarizing quantitative results from phenotypic and metabolomic analyses.
Table 2: Quantitative Analysis of Seedling Growth in a Differential Chemical Screen [64]
| Genotype | Treatment | CNN Classification (% Altered Growth) | Segmented Leaf Area (px², Mean ± SD) | Segmented Root Area (px², Mean ± SD) |
|---|---|---|---|---|
| Wild-Type | DMSO (Control) | 2% | 15,500 ± 1,200 | 8,300 ± 950 |
| Wild-Type | Chemical A | 5% | 14,800 ± 1,050 | 7,950 ± 880 |
| mus81 mutant | DMSO (Control) | 3% | 15,200 ± 1,350 | 8,100 ± 1,100 |
| mus81 mutant | Chemical A | 95% | 5,100 ± 600 | 2,300 ± 450 |
| mus81 mutant | MMC (Positive Control) | 100% | 4,800 ± 550 | 2,100 ± 400 |
Table 3: Metabolite Yields from Engineered Pathways in Plant Chassis [45] [65]
| Target Compound | Plant Chassis | Engineering Strategy | Yield | Analytical Method |
|---|---|---|---|---|
| Diosmin | N. benthamiana (transient) | Coordinated expression of 5-6 flavonoid pathway enzymes | 37.7 µg/g Fresh Weight (FW) | LC-MS [65] |
| QS-7 Saponin | N. benthamiana (transient) | Co-expression of 19 pathway genes (P450s, glycosyltransferases) | 7.9 µg/g Dry Weight (DW) | LC-MS [65] |
| GABA | Tomato (stable) | CRISPR/Cas9 knockout of SlGAD2 & SlGAD3 genes | 7- to 15-fold increase | LC-MS [45] |
By employing these detailed protocols and analytical frameworks, researchers can effectively ground computational models of plant development in empirical evidence, driving a cycle of prediction, validation, and refined understanding.
The quest to understand how biological patterns emerge from initially homogeneous tissues is a central theme in developmental biology. Within plant sciences, this quest is increasingly supported by computational modeling, which integrates processes across vast spatio-temporal scales—from gene expression and cell signaling to tissue mechanics [5] [16]. Three major theoretical frameworks have been pivotal in explaining these self-organized patterning events: the reaction-diffusion (Turing) model, the positional information (French Flag) model, and the more recent mechanochemical patterning models. This article provides a comparative analysis of these frameworks, focusing on their application in plant systems. We detail specific protocols for their computational and experimental investigation and provide a toolkit for researchers aiming to dissect the mechanisms of plant morphogenesis.
Theoretical Framework and Key Differentiators
The core principles of the three patterning mechanisms are distinct, yet they can operate in a complementary fashion within a developing organism [68] [1].
Turing Patterns (Reaction-Diffusion): Proposed by Alan Turing in 1952, this model posits that two diffusible morphogens—an activator and a inhibitor—can spontaneously generate periodic patterns from a homogeneous state through a diffusion-driven instability [4] [1]. The essential principle is short-range activation coupled with long-range inhibition. The activator promotes its own production and that of the inhibitor, while the inhibitor suppresses the activator. For instability to occur, the inhibitor must diffuse significantly faster than the activator [4] [69]. This mechanism is exemplified in plants by the patterning of ROP (Rho-of-Plants) proteins within single cells, which define domains for localized growth, such as in the jigsaw-puzzle shapes of leaf epidermal cells or the secondary cell wall thickenings in xylem [4].
Positional Information (French Flag Model): Pioneered by Lewis Wolpert, this model suggests that cells acquire positional value based on the concentration of a morphogen gradient that is established across a tissue [5] [16]. Cells then interpret this concentration threshold to adopt different fates, much like the three bands of a French flag. This model relies on a pre-patterned gradient and is less about de novo pattern generation and more about the interpretation of a pre-existing spatial cue.
Mechanochemical Models: These models challenge the purely chemical perspective by incorporating tissue mechanics as an active driver of patterning. They propose feedback loops where chemical signals influence mechanical properties (e.g., cell contraction or growth), and mechanical cues (e.g., stress, strain, or compression), in turn, influence gene expression and morphogen distribution [70] [69]. In this framework, mechanics can provide the necessary "long-range inhibition," potentially overcoming limitations of pure diffusion-based models, such as the requirement for implausibly high diffusion rates for inhibitors on a tissue scale [70] [69].
Table 1: Comparative Analysis of Patterning Models in Plant Development
| Feature | Turing (Reaction-Diffusion) | Mechanochemical | Positional Information |
|---|---|---|---|
| Core Principle | Short-range activation, long-range inhibition via differential diffusion [4] [1] | Feedback between chemical signaling and tissue mechanics (stress, strain) [70] [69] | Interpretation of a pre-established morphogen gradient [5] |
| Key Components | Activator & Inhibitor morphogens with different diffusivities [68] | Morphogens, cytoskeleton, cell walls, adhesion molecules [70] | Morphogen, source/sink regions, threshold-sensitive genes [5] |
| Pattern Initiation | Spontaneous (de novo) from near-homogeneity [1] | Spontaneous (de novo) or from mechanical pre-patterning [69] | Reliant on a pre-patterned morphogen source [5] |
| Role of Mechanics | Often considered a passive by-product of chemical patterns [70] | Active, integral driver of pattern formation [70] [69] | Mechanics is typically passive or secondary to the chemical gradient |
| Robustness | Can be sensitive to parameter changes; robustness is a key research area [68] | Highly robust to changes in diffusion rates; patterns scale with tissue size [69] | Robustness depends on the stability of the morphogen gradient |
| Example in Plants | ROP protein patterning in epidermal cells; Phyllotaxis (with auxin/PIN) [4] | Phyllotaxis; Root development; Embryogenesis [70] [5] | Root development; Vascular tissue patterning [5] [16] |
Experimental and Computational Protocols
Protocol 1: Simulating a Turing Pattern in a Plant Epidermal Cell
This protocol outlines the steps to computationally model the formation of ROP protein domains, a canonical example of a Turing system in a single plant cell [4].
1. Model Selection and Formulation:
- Objective: Simulate the formation of multiple, stable clusters of active ROP on the cell membrane.
- Model Type: Employ a two-component reaction-diffusion system based on the "active ROP" and "inactive ROP" states. The system uses a substrate-depletion mechanism where active ROP (membrane-bound, slow diffusion) depletes a pool of inactive ROP (cytosolic, fast diffusion) [4].
- Governing Equations:
- ( \frac{\partial A}{\partial t} = DA \nabla^2 A + \gamma (A^2 / I + \rho) - \kappa A ) (Active ROP, A)
- ( \frac{\partial I}{\partial t} = DI \nabla^2 I - \gamma (A^2 / I + \rho) + \kappa A + \sigma ) (Inactive ROP, I)
- Where (DA) and (DI) are diffusion coefficients ((DI >> DA)), (\gamma) is the activation rate, (\kappa) is the deactivation rate, (\rho) is a basal production, and (\sigma) is a constant source.
2. Parameterization and Discretization:
- Parameters: Use literature-based values as a starting point [4]. For instance, set (DA = 0.01 \ \mu m^2/s), (DI = 1.0 \ \mu m^2/s), and tune the reaction rates ((\gamma, \kappa)) to achieve Turing instability.
- Spatial Grid: Discretize a 2D plane representing the cell membrane into a grid of at least 100x100 points.
- Initial Conditions: Set a homogeneous steady state with a small random perturbation ((<1\%)) to break symmetry.
3. Numerical Simulation and Analysis:
- Solver: Use a finite difference method with a no-flux (Neumann) boundary condition.
- Time Integration: Implement an explicit Euler method with a small time step to ensure numerical stability.
- Output Analysis: Monitor the emergence of spots over time. The final pattern is assessed by the number, size, and stability of the ROP clusters.
Protocol 2: Investigating a Mechanochemical Feedback Loop in a Tissue
This protocol describes how to model and test a mechanochemical patterning mechanism, where tissue deformation and a morphogen influence each other [70] [69].
1. System Coupling:
- Objective: Simulate how feedback between a morphogen and tissue strain leads to de novo pattern formation.
- Model Components:
- Chemical Model: A reaction-diffusion equation for a morphogen concentration (c).
- Mechanical Model: A 3D finite element model of the tissue, treated as a viscoelastic material.
- Feedback Loop: Define the coupling. For example: "Morphogen (c) promotes local apical constriction. Constriction induces tissue strain. Strain, in turn, inhibits morphogen production" [69].
2. Implementation:
- Geometry: Create a 3D mesh of the tissue (e.g., a spherical blastula or a root tip segment). A 2D simplification can be used but may yield unrealistic mechanical behavior [70].
- Simulation Platform: Use a coupled solver, such as COMSOL Multiphysics or a custom-built finite element code, that can iteratively solve the chemical and mechanical equations.
3. Experimental Validation:
- Perturbation Tests: Simulate the effect of inhibiting morphogen diffusion or altering tissue mechanical properties (e.g., via drug treatments that disrupt the cytoskeleton).
- Prediction: Unlike a pure Turing system, the pattern wavelength in this mechanochemical model is expected to be relatively insensitive to changes in morphogen diffusion rate but should scale with tissue thickness [69].
Protocol 3: Distinguishing Turing vs. Positional Information in Root Development
The plant root is a system where both repetitive patterning (potentially Turing-like) and positional information gradients operate [5] [16].
1. Experimental Design:
- System: Use the Arabidopsis thaliana root tip, with its highly structured organization of cell files.
- Hypothesis: Test whether the pattern of a specific cell type (e.g., xylem strands) forms via a Turing mechanism or is specified by a positional information gradient.
2. Perturbation and Imaging:
- Surgical Perturbation: Use laser ablation to remove the putative source of a positional information morphogen (e.g., the quiescent center). If the pattern re-forms correctly, it suggests a self-organizing (Turing) mechanism is at play.
- Chemical Perturbation: Apply drugs that disrupt cytoskeletal transport (e.g., Actin inhibitor Latrunculin B) to interfere with polar auxin transport, a key player in both models.
- Live Imaging: Use confocal microscopy of live reporters for candidate morphogens (e.g., auxin, cytokinin) and cell fate markers over time.
3. Data Analysis and Model Fitting:
- Expected Outcomes for Turing: Pattern regeneration after perturbation should occur with a characteristic wavelength, potentially leading to a change in the number of patterned elements (e.g., xylem strands) [4].
- Expected Outcomes for Positional Information: Pattern regeneration would be dependent on the re-establishment of the morphogen gradient from the source.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Tools for Investigating Patterning Mechanisms
| Reagent / Tool | Function / Target | Application in Patterning Research |
|---|---|---|
| Latrunculin B | Actin cytoskeleton disruptor | Inhibits active transport processes (e.g., PIN polarization), allowing dissection of diffusion-vs-transport in pattern formation [4]. |
| Auxin Biosensors (e.g., DII-VENUS) | Report auxin distribution and signaling | Live imaging of morphogen gradients in positional information and phyllotaxis (a Turing-like process) [4] [5]. |
| ROP GTPase Mutants (e.g., constitutively active/dominant negative) | Perturb ROP signaling pathways | Test the necessity of ROP activity for intracellular Turing patterns in epidermal cells [4]. |
| OSCILLATOR: A 3D Finite Element Solver | Computational modeling of tissue mechanics | Simulate the mechanical part of mechanochemical models; essential for capturing realistic 3D tissue behavior [70]. |
| VirtualLeaf Framework | Agent-based modeling platform | Model tissue patterning integrating cell division, growth, and chemical signaling to test interplay between models [5]. |
The paradigms of Turing patterning, positional information, and mechanochemical feedback are not mutually exclusive but are increasingly understood as complementary and often intertwined forces in plant development. Computational modeling has been instrumental in demonstrating that purely chemical Turing models, while powerful, face challenges in biological realism that can be resolved by incorporating mechanics [70] [69]. Conversely, mechanical patterns often require chemical inputs for their initiation. The future of understanding plant morphogenesis lies in integrated models that combine reaction-diffusion dynamics, pre-patterned positional cues, and the active physical forces exerted by and on growing tissues. The protocols and tools provided here offer a starting point for researchers to design critical experiments that can distinguish the contribution of each mechanism to the beautiful and complex patterns observed in the plant kingdom.
Phyllotaxis, the regular arrangement of organs like leaves and flowers around a plant stem, represents a quintessential problem in developmental biology. The most prevalent pattern, spiral phyllotaxis, exhibits a remarkable consistency with a divergence angle of approximately 137.5° between successive organs, often following the Fibonacci sequence [71]. For decades, the seminal work of Alan Turing has provided a theoretical framework for understanding such repetitive patterning. Turing's reaction-diffusion model posits that the interplay between a slowly diffusing, short-range activator and a rapidly diffusing, long-range inhibitor can spontaneously break symmetry and generate regular patterns from an initially homogeneous state [4]. However, modern plant biology has uncovered a complex, transport-driven system centered on the plant hormone auxin. This case study examines the evidence for both mechanisms, evaluating whether phyllotaxis is a classic Turing instability or a process fundamentally driven by polar auxin transport.
Theoretical Frameworks: A Comparative Analysis
The table below summarizes the core components and dynamics of the two competing models for phyllotaxis.
Table 1: Core Components of Turing vs. Transport-Driven Models of Phyllotaxis
| Feature | Turing Reaction-Diffusion Model | Auxin Transport-Driven Model |
|---|---|---|
| Primary Patterning Mechanism | Spontaneous symmetry breaking via feedback loops [4] | Canalization of auxin flux to sites of organ initiation [40] |
| Key Molecules | Hypothetical activator and inhibitor molecules | Auxin (Indole-3-acetic acid) and PIN-FORMED (PIN) efflux carriers [71] |
| Role of Auxin | Could be mapped to either the activator or inhibitor | Morphogen that accumulates at incipient primordia; its maximum defines organ initiation sites [71] |
| Spatial Coordination | Differential diffusion coefficients (D~inhibitor~ >> D~activator~) [4] | Polar, active transport of auxin via dynamically localized PIN proteins [4] |
| Inhibitory Field | Generated by the long-range inhibitor | Generated by auxin depletion from the meristem center and surrounding areas due to canalization into primordia [71] |
Computational and Experimental Evidence
Evidence Supporting a Turing-Type Mechanism
At a conceptual level, the phyllotactic pattern exhibits hallmarks of a Turing system. The emerging primordia act as self-organizing peaks, with the existing primordia creating an "inhibitory field" that prevents new organs from forming too close, effectively fulfilling the role of long-range inhibition [4]. Furthermore, well-established Turing systems operate at other scales in plant biology, demonstrating that the core principle is viable. A key example is the patterning of Rho-of-Plants (ROP) proteins within single cells, which governs the formation of lobed epidermal cells and xylem secondary wall patterns. This system relies on an active, membrane-bound ROP state (slow diffusion) and an inactive, cytosolic state (fast diffusion), perfectly embodying the substrate-depletion version of a Turing mechanism [4].
Evidence for a Transport-Driven Process
Molecular genetic studies in Arabidopsis thaliana have identified a detailed, transport-based mechanism. The following diagram illustrates the core auxin transport-driven patterning module.
Diagram 1: The core auxin transport-driven phyllotaxis module.
The process can be broken down into a detailed experimental workflow, as shown below.
Diagram 2: Detailed workflow of transport-driven phyllotactic patterning.
Beyond this core module, recent research on Cyanella alba has revealed an even deeper connection, showing that the inherent left-right asymmetry established by the phyllotactic spiral can determine the handedness of mirror-image flowers, linking organ positioning to macroscopic floral morphology [72]. Furthermore, regulatory complexes fine-tune this process. As detailed in the diagram below, a complex involving REM34/35 and ARF7/19 transcription factors integrates auxin signaling with cell cycle control to stabilize the pattern by modulating meristem size [71].
Diagram 3: The REM-ARF complex integrates auxin response with meristem size control.
Critical Synthesis: A Liberal Turing Instability?
The prevailing view leans towards phyllotaxis being a transport-driven process, but one that can be abstractly considered a "liberal" Turing system [4]. The core logic of short-range facilitation (auxin peak reinforcement via PIN polarization) and long-range inhibition (auxin depletion creating an inhibitory field) is preserved. However, the mechanism is not a passive reaction-diffusion but an active, transport-based system. As one review notes, modern models for phyllotaxis "include directed transport of the plant hormone auxin via dynamically positioned, polarly localised PIN proteins and often more," making it far more complicated than a simple Turing system [4]. Therefore, phyllotaxis is best described as a transport-driven process whose emergent, self-organizing behavior is analogous to a Turing instability, rather than being a direct embodiment of one.
Experimental Protocols for Key Phyllotaxis Studies
Protocol: Quantifying Phyllotactic Patterns and Permutations
Application: Phenotypic analysis of phyllotaxis in wild-type and mutant plants (e.g., Arabidopsis thaliana) [71].
Materials:
- Fixed inflorescence samples from wild-type and mutant plants (e.g., rem34 rem35, arf7 arf19).
- Scanning Electron Microscope (SEM) or high-resolution digital microscope.
- Image analysis software (e.g., Fiji/ImageJ).
Procedure:
- Sample Preparation: Collect and fix inflorescence meristems in FAA (Formalin-Acetic Acid-Alcohol) or glutaraldehyde for SEM.
- Imaging: Capture high-resolution top-down images of the inflorescence meristem, ensuring all floral primordia (P1, P2, P3, ... I1, I2) are visible.
- Divergence Angle Measurement:
- In the image software, set the meristem center as the origin.
- Draw vectors from the center to the center of successive primordia (e.g., from P1 to P2, P2 to P3).
- Measure the angle between successive vectors. This is the divergence angle.
- Data Collection & Analysis: Record at least 20 successive divergence angles per meristem across multiple biological replicates. Calculate the average divergence angle and standard deviation. Note any "permutation events," where the angle deviates significantly from the canonical ~137.5°.
Protocol: Live Imaging of Auxin Response and PIN Polarity
Application: Visualizing the spatiotemporal dynamics of auxin signaling and transport during primordia formation [71].
Materials:
- Transgenic plant lines expressing auxin response reporters (e.g., DR5rev:GFP) and PIN protein fusion markers (e.g., PIN1:PIN1-GFP).
- Confocal Laser Scanning Microscope (CLSM) with time-lapse capability.
- Custom chamber for maintaining live plant specimens under the microscope.
Procedure:
- Plant Preparation: Mount a live, intact seedling or inflorescence in the microscopy chamber with appropriate medium to prevent desiccation.
- Microscope Setup: Use appropriate laser lines and filters for the fluorescent proteins (e.g., 488 nm laser for GFP).
- Time-Lapse Imaging:
- Focus on the shoot apical meristem or inflorescence meristem.
- Capture Z-stacks at regular intervals (e.g., every 2-4 hours) over 24-72 hours to track the entire initiation process of one or more primordia.
- Image Analysis:
- Auxin Maxima: Identify emerging bright spots of DR5:GFP signal at the meristem periphery, marking incipient primordia.
- PIN Polarity: Analyze the subcellular localization of PIN1-GFP. In cells at the summit of a new auxin maximum, PIN1 should be polarized towards the center of the maximum.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents and Solutions for Phyllotaxis Research
| Reagent / Material | Function / Application | Example Use Case |
|---|---|---|
| DR5rev:GFP / DR5:GUS | Synthetic reporter for auxin response maxima [71] | Visualizing sites of organ initiation in live (GFP) or fixed (GUS) tissue. |
| PIN:PIN-GFP Transgenic Lines | Visualizing the polar localization of auxin efflux carriers [4] | Live imaging of PIN protein dynamics during pattern formation. |
| auxin transport inhibitors (e.g., NPA, TIBA) | Chemical inhibition of polar auxin transport | Experimental perturbation of phyllotaxis to test the transport-driven model. |
| CRISPR/Cas9 Mutagenesis System | Generating targeted knockouts of redundant genes [71] | Creating multiple mutants (e.g., rem34 rem35) to study gene function. |
| Fixed Meristem Samples | Structural preservation for high-resolution imaging | Scanning Electron Microscopy (SEM) to analyze meristem architecture and primordia positioning. |
The formation of repetitive patterns in biology, from the stripes of a zebra to the ridges of a fingerprint, has long been a subject of intense scientific inquiry. Alan Turing's seminal 1952 theory of reaction-diffusion systems provided a foundational framework for understanding how simple interactions between molecular components can spontaneously generate complex spatial patterns [4]. This mechanism, now known as Turing patterning, relies on the interplay between a short-range activator and a long-range inhibitor with different diffusion coefficients [8].
Contemporary research has revealed that Turing patterns operate across biological kingdoms and organizational scales, from vegetation patterns in ecosystems to subcellular protein distributions [4]. This application note examines the conserved principles and divergent implementations of Turing pattern formation by comparing two mammalian systems—animal skin spotting and human fingerprint development—to extract valuable insights for computational modeling of plant systems.
Theoretical Framework of Turing Patterning
Core Principles
Turing patterning arises from a reaction-diffusion mechanism wherein two chemical morphogens interact to spontaneously break spatial symmetry and generate periodic patterns [4]. The fundamental requirements include:
- Short-range activation: A self-enhancing activator that promotes its own production and that of an inhibitor
- Long-range inhibition: A rapidly diffusing inhibitor that suppresses activator formation
- Differential diffusion: The inhibitor must diffuse significantly faster than the activator to create instability in the homogeneous state [8]
Mathematically, this system can be described by partial differential equations of the form:
Where A and I represent activator and inhibitor concentrations, DA and DI their diffusion coefficients, and f and g nonlinear functions describing their interactions.
Recent Theoretical Advances
Recent research has revealed that Turing patterns can emerge from biochemical networks beyond the classical activator-inhibitor framework. Systematic analysis of elementary biochemical reactions has identified ten simple reaction networks capable of generating Turing patterns without imposed feedback loops [9]. These networks often involve regulated degradation pathways and can produce patterns with flexible diffusion rate constants of individual molecules.
Table 1: Key Parameters in Turing Pattern Formation Across Biological Systems
| Parameter | Classical Turing Model | Animal Skin Patterning | Fingerprint Formation | Plant Root Development |
|---|---|---|---|---|
| Activator Examples | Theoretical morphogens | EDAR, WNT signaling | WNT, EDAR pathways | Auxin, ROP proteins |
| Inhibitor Examples | Theoretical morphogens | BMP signaling | BMP pathways | Unknown inhibitors |
| Diffusion Ratio (DI/DA) | >1 required | Enhanced via diffusiopherosis | Not specified | Combined diffusion-advection |
| Pattern Wavelength | Determined by parameters and domain size | Cell size-dependent imperfections | Initiation site-dependent | Scale-dependent on tissue |
| Additional Mechanisms | Pure reaction-diffusion | Diffusiophoresis, cell size effects | Epithelial buckling, volar pads | Mechanical stresses, polar transport |
Mammalian Patterning Systems as Model Processes
Animal Skin Spot Formation
The intricate spots and stripes on animal pelts represent a classic manifestation of Turing patterning in nature. Recent research on ornate boxfish patterning has refined the classical Turing model by incorporating additional physical mechanisms that enhance biological realism [73].
Key Molecular Components:
- EDAR Pathway: Serves as a primary activator signal promoting pigment cell aggregation
- WNT Signaling: Functions cooperatively with EDAR as an activation mechanism
- BMP Proteins: Act as long-range inhibitors that create zones of inhibition between spots
Pattern Refinement Mechanism: The classical Turing model often produces patterns with blurry boundaries that lack the sharpness observed in biological systems. The incorporation of "diffusiophoresis"—a process where diffusing particles pull other particles along with them—generates patterns with significantly sharper outlines [73]. This mechanism operates similarly to soap dragging dirt out of laundry during diffusion.
Furthermore, the integration of individual cell size parameters explains natural imperfections in animal patterns. When models account for finite cell size and movement through tissue, they spontaneously generate the variations in spot size, distribution, and pattern interruptions observed in actual animal coats, moving beyond the mathematically perfect patterns of early simulations [73].
Human Fingerprint Ridge Patterning
Fingerprint formation provides a remarkable example of Turing patterning in human development, revealing how conserved principles generate unique individual variations [74]. The process begins during the third month of fetal development and results in permanent, individualized ridge patterns.
Developmental and Molecular Basis: Fingerprint ridges are epithelial structures that undergo a truncated hair follicle developmental program, recruiting molecular components shared with other skin appendages but failing to recruit mesenchymal cells [74]. The core signaling system involves:
- WNT and EDAR: Function as activators promoting ridge formation
- BMP Pathways: Serve as inhibitors creating spacing between ridges
- Initiation Sites: Pattern formation begins at three specific locations on the fingertip—near the nail, central fingertip, and first knuckle crease
The interaction of these components establishes a Turing reaction-diffusion system that resolves epithelial growth into bands of focalized proliferation under a precociously differentiated suprabasal layer [74]. Ridge formation occurs as waves spreading from variable initiation sites, with the propagation and meeting of these waves determining the ultimate pattern type (arch, loop, or whorl).
Genetic Control and Pleiotropy: Large-scale genomic studies have identified at least 43 genomic regions influencing fingerprint formation, with the EVI1 gene (ecotropic viral integration site 1) showing particularly strong association [75]. This gene plays a central role in embryonic limb development, demonstrating pleiotropy where the same genes influence both limb patterning and dermatoglyphic features. Reduced EVI1 expression in mouse models results in abnormal digital ridge formations, confirming its functional role [75].
Computational Modeling Approaches
Model Formulation and Implementation
Computational modeling of Turing systems typically employs reaction-diffusion frameworks, with recent advances incorporating additional biological realism. The general formulation for two-component systems follows:
Where n represents cell density, c morphogen concentration, dn and dc diffusion coefficients, χ the chemotactic sensitivity, and f and g kinetic terms [34].
Specific Model Variants:
- Density-Dependent Chemotaxis: χ(n,c) = χ_0n
- Ratio-Dependent Chemotaxis: χ(n,c) = χ_0n/c
- Extended Systems: Multi-component models incorporating mechanical stresses or advection
Thermodynamically Consistent Modeling
Recent work has focused on developing thermodynamically consistent models that obey physical laws while retaining pattern-forming capabilities. The variational Gray-Scott model incorporates all reverse reactions and introduces virtual species to transform classical open systems into closed subsystems of larger systems [76]. This approach reveals that stationary patterns can persist as transient states, with pattern persistence times scaling as O(ϵ^(-1)), where ϵ represents the reaction rate of reverse reactions.
Experimental Protocols for Pattern Formation Analysis
Protocol 1: Computational Simulation of Turing Systems
Purpose: To implement and analyze a minimal Turing patterning system using computational methods.
Materials and Software:
- Mathematical computing environment (MATLAB, Python with NumPy/SciPy)
- Partial differential equation solver with visualization capabilities
- High-performance computing resources for parameter screening
Procedure:
- System Definition: Implement the Schnakenberg kinetics as a canonical Turing system: Where u is the activator, v is the inhibitor, and Du < Dv
Parameter Selection: Establish baseline parameters a=0.1, b=0.9, Du=0.01, Dv=1.0
Domain Setup: Create a 2D spatial domain with zero-flux boundary conditions
Numerical Simulation:
- Discretize the system using finite difference methods
- Implement time integration using Euler or Runge-Kutta methods
- Introduce random perturbations to break initial symmetry
Pattern Analysis:
- Quantify pattern wavelength as a function of diffusion coefficients
- Perform sensitivity analysis on kinetic parameters
- Map parameter regions supporting spots versus stripes
Troubleshooting:
- If patterns fail to emerge, verify that the diffusion ratio Dv/Du > 1
- For numerical instability, reduce time step or implement more stable integration schemes
Protocol 2: Identification of Pattern-Enabling Biochemical Networks
Purpose: To systematically identify elementary biochemical reactions capable of supporting Turing pattern formation.
Materials:
- Computational pipeline for bifurcation analysis
- Parameter sampling algorithms
- Reaction network enumeration tools
Procedure:
- Network Enumeration: Generate all topologically distinct biochemical complexes with up to four subunits and their associated reaction paths [9]
Model Construction: For each reaction network, build a mathematical model with:
- Mass-action kinetics for all reactions
- Constant synthesis of unbound molecules
- Degradation of all molecular species
- Diffusion terms for all components
Bifurcation Analysis:
- Use computational continuation to identify Hopf bifurcations
- Screen parameter spaces for Turing instabilities
- Verify pattern formation through numerical simulation of PDEs
Motif Identification: Identify unifying network motifs across pattern-enabling systems
Applications: This protocol has revealed that ten simple reaction networks, including trimer formation with regulated degradation, can generate Turing patterns without classical activator-inhibitor feedback [9].
Research Reagent Solutions for Pattern Formation Studies
Table 2: Essential Research Reagents for Turing Pattern Investigation
| Reagent/Category | Function in Patterning Studies | Example Applications | Technical Notes |
|---|---|---|---|
| Morphogen Pathway Modulators | Activate or inhibit key Turing system components | Small molecule WNT agonists/antagonists; BMP pathway modulators | Critical for experimental perturbation of patterning systems |
| Live Imaging Reagents | Real-time visualization of pattern dynamics | Fluorescent biosensors for calcium, cAMP; vital dyes for cell boundaries | Enable correlation of molecular patterns with cellular behaviors |
| Genetic Model Systems | In vivo analysis of pattern formation | Transgenic mice with tuned EDAR expression; zebrafish pigment models | Provide whole-organism context for computational predictions |
| Single-Cell RNA Sequencing | Identification of gene expression patterns in patterning tissues | 10x Genomics platform; droplet-based sequencing | Reveals heterogeneity in cellular responses to morphogen gradients |
| Customizable Computational Frameworks | Simulation and parameter screening for Turing systems | VisualPDE; FEniCS; custom MATLAB/Python scripts | Essential for bridging theoretical and experimental approaches |
Applications to Plant Developmental Biology
The insights gained from animal skin and fingerprint patterning systems provide valuable perspectives for computational modeling of plant development. Plant root development, for instance, involves processes spanning multiple spatial and temporal scales, from gene expression and cell signaling to tissue mechanics and organ patterning [40].
Key Transferable Concepts:
- Multi-scale Integration: Effective models must integrate processes across scales, from molecular interactions to tissue-level mechanics
- Simplification Principles: Model building should incorporate the minimal components necessary to capture phenomena of interest, removing unnecessary complexity while retaining essential dynamics [40]
- Robustness Analysis: Models should generate consistent behaviors across parameter variations rather than depending on finely-tuned values
Plant-Specific Adaptations: While the core principles of Turing patterning apply across kingdoms, plant systems introduce additional complexities including cell walls, polar transport mechanisms (e.g., PIN proteins for auxin transport), and continuous growth throughout development [4]. The Rho-of-Plants (ROP) proteins represent a plant-specific Turing system operating at the subcellular level to generate patterns in epidermal cells and xylem tissues [8].
Cross-kingdom analysis of Turing pattern formation reveals both conserved principles and system-specific adaptations. The comparison of animal skin patterning and human fingerprint development highlights how a core reaction-diffusion mechanism can be implemented through different molecular components and physical processes to generate diverse biological patterns.
For plant computational biology, these insights suggest several promising research directions:
- Development of multi-scale models that integrate subcellular Turing systems with tissue-level mechanics
- Exploration of how growth and cell division feedback on pattern formation dynamics
- Identification of plant-specific molecular implementations of Turing systems beyond the classical activator-inhibitor framework
The continued integration of theoretical modeling with experimental validation across biological systems will further elucidate how simple physical principles generate breathtaking biological complexity through the ubiquitous process of Turing pattern formation.
Benchmarking Model Predictions Against High-Resolution Spatiotemporal Data
In computational biology, the accurate prediction of complex biological patterns depends on the rigorous benchmarking of theoretical models against high-resolution empirical data. This is particularly true in the study of Turing pattern formation in plants, where models describing the spontaneous emergence of order from homogeneity must be validated against observations across multiple spatial and temporal scales [4]. Alan Turing's revolutionary theory proposed that diffusion, typically a homogenizing process, could instead destabilize a stable equilibrium and lead to the spontaneous formation of regular patterns through reaction-diffusion systems [4] [3]. This framework has profound implications for understanding plant morphogenesis, from the microscopic patterning of Rho-of-Plants (ROP) proteins within single cells to the macroscopic organization of vegetation in dryland ecosystems [4]. This article provides detailed application notes and experimental protocols for the acquisition of high-resolution spatiotemporal data and its use in benchmarking computational models of Turing pattern formation in plant systems.
Turing Pattern Formation in Plant Systems: Application Scenarios
The theory of Turing pattern formation finds several compelling applications in plant biology, each occurring at a distinct spatial scale and requiring specialized measurement techniques for model validation.
- Intracellular Patterning (ROP Proteins): Inside single plant cells, ROP proteins self-organize to define domains for processes like tip growth and the development of puzzle-shaped epidermal cells or patterned secondary cell walls in xylem [4]. This system operates via a substrate-depletion mechanism where the active, membrane-bound ROP (slow-diffusing activator) interacts with the inactive, cytosolic ROP (fast-diffusing substrate or inhibitor) [4]. Benchmarking models at this scale requires high-resolution, time-resolved imaging of membrane domains.
- Tissue-Level Patterning (Epidermis): The regular arrangement of structures on the plant epidermis, such as stomata or trichomes, is a classic candidate for Turing patterning. The Gierer-Meinhardt model of short-range activation and long-range inhibition provides an intuitive conceptual framework for these phenomena [4]. Data for benchmarking includes microscopic imagery tracking the emergence and positioning of these structures over time.
- Macroscopic Patterning (Dryland Vegetation): At the landscape level, periodic vegetation patterns in arid regions can be explained by Turing mechanisms. Here, water acts as the depleted substrate (or its inverse, lack-of-water, as the inhibitor), with the differential transport rates necessary for pattern formation being achieved through a combination of root uptake and surface/subsurface flow [4]. Remote sensing and aerial imagery provide the spatiotemporal data for benchmarking models at this scale.
Quantitative Benchmarking Framework
A robust benchmarking pipeline requires quantitative metrics to compare model predictions against experimental data, moving beyond qualitative assessments of "spots and stripes."
Table 1: Key Metrics for Benchmarking Spatiotemporal Predictions
| Metric Category | Specific Metric | Description | Application in Plant Turing Systems |
|---|---|---|---|
| Spatial Accuracy | Pattern Wavelength | The distance between repeating pattern elements (e.g., spacing between ROP clusters or vegetation bands). | Critical for validating the model's predicted critical wavelength, which depends on kinetic parameters and effective diffusion coefficients [4]. |
| Pattern Symmetry & Type | Classification of pattern morphology (e.g., spots, stripes, labyrinths). | Determines if the model can reproduce the correct pattern topology observed in nature. | |
| Anomaly Correlation Coefficient (ACC) | Measures the spatial correlation between predicted and observed patterns after removing the mean. | Useful for large-scale patterns (e.g., vegetation); used in benchmarking AI weather models [77]. | |
| Temporal Accuracy | Pattern Onset Time | The time from initial homogeneous conditions to the first visible instability. | Tests the model's prediction of the timing of symmetry breaking. |
| Pattern Evolution Rate | The rate at which the pattern amplitude grows and stabilizes. | Assesses if the model captures the correct dynamics of pattern establishment. | |
| Root Mean Square Error (RMSE) | Measures the average magnitude of difference between predicted and observed states over time. | A standard metric for temporal fidelity; used in atmospheric river forecasting benchmarks [77]. | |
| Model Performance | Pearson Correlation Coefficient (PCC) | Measures the linear correlation between temporal changes in predictions and data. | Evaluates the model's ability to capture the dynamics of pattern refinement [77]. |
| Computational Efficiency | Time and resources required to simulate the pattern formation process. | Important for practical application and parameter exploration, especially for complex, multi-component models. |
| Data Type | Measurement Technology | Spatial Resolution | Temporal Resolution | Example Plant Application |
|---|---|---|---|---|
| Subcellular | Confocal Microscopy, 2-Photon Calcium Imaging [78] | Sub-micrometer | Seconds to minutes | Imaging of ROP protein dynamics and intracellular calcium waves [4]. |
| Cellular/Tissue | Light Sheet Microscopy, Image Mueller Polarimetry [79] | Micrometer | Minutes to hours | Tracking epidermal cell shape changes [4]; detecting early disease-induced structural changes via depolarization metrics [79]. |
| Organism/Canopy | UAV (Drone) Imaging, Spectral Imaging | Centimeter to Meter | Days to weeks | Monitoring leaf development and phyllotaxis over time. |
| Ecosystem | Satellite Remote Sensing (e.g., Sentinel-5P [80]) | Meters to Kilometers | Days | Analyzing large-scale vegetation patterns in drylands [4]. |
Experimental Protocols for Data Acquisition
This section outlines detailed protocols for acquiring high-quality spatiotemporal data relevant to plant Turing patterns.
Protocol 4.1: Intracellular ROP Patterning Analysis using 2-Photon Calcium Imaging
Objective: To characterize the spatiotemporal dynamics of ROP protein activity in living plant cells with high resolution. Key Resources: Genetically encoded biosensors (e.g., ROP FRET sensors), 2-photon laser scanning microscope, Thy1-GCaMP6s transgenic lines [78].
- Sample Preparation: Grow seedlings of model plants (e.g., Arabidopsis thaliana) expressing ROP activity biosensors on appropriate solid media.
- Mounting: Gently mount intact seedlings or excised organs in a custom imaging chamber with liquid media, ensuring minimal mechanical stress.
- Image Acquisition: Using a 2-photon microscope, define a region of interest (ROI) over the target cells (e.g., leaf epidermal pavement cells). Acquire time-series images at high spatial and temporal resolution (e.g., 512x512 pixels, 1-5 second intervals) for a sufficient duration to capture multiple patterning events (e.g., 30-60 minutes).
- Data Pre-processing: Extract fluorescence traces from individual membrane domains. Apply motion correction and background subtraction algorithms. De-randomize and average traces across repeated experimental trials to improve signal-to-noise ratio [78].
- Quantitative Analysis: Calculate metrics such as cluster lifetime, size, and separation distance. Use these to benchmark predictions from ROP Turing models [4].
Protocol 4.2: Enhanced Visualization of Tissue-Level Patterns using Polarimetric Imaging
Objective: To detect and quantify subtle, early-stage pattern changes in plant tissues (e.g., from pathogen infection) that are invisible to standard imaging. Key Resources: Complete image Mueller polarimeter, samples with known and suspected patterns [79].
- Sample Preparation: Collect healthy and diseased plant leaves. Gently mount them flat in the sample holder of the polarimeter. For transmission measurements, ensure leaves are thin enough for light penetration.
- System Calibration: Calibrate the Mueller polarimeter using standard samples (e.g., air for reference) to ensure accurate measurement of all 16 elements of the Mueller matrix.
- Image Acquisition: Illuminate the sample with polarized light at a chosen wavelength (e.g., 625 nm for deeper penetration [79]). Measure the full Mueller matrix for each pixel of the sample in either scattering or transmission configuration.
- Data Processing: For each pixel, calculate depolarization metrics from the Mueller matrix:
- Benchmarking: The resulting maps of IPPs and CPs provide a high-contrast, quantitative dataset against which to benchmark models predicting tissue-level pattern changes, such as those arising during infection.
Protocol 4.3: Spatial Transcriptomics with Optimized Colorization (Spaco)
Objective: To accurately visualize and analyze the spatial distribution of cell types or gene expression, which is crucial for understanding the molecular underpinnings of patterns. Key Resources: Spatially resolved transcriptomics dataset (e.g., from 10X Visium, MERFISH), Spaco software package (Python or R version) [81].
- Data Input Preparation: Process your spatial transcriptomics data to obtain cell or spot coordinates and corresponding cluster annotations or gene expression labels using standard bioinformatics tools (e.g., Seurat, Squidpy).
- Install Spaco Package: Install the Spaco package in your chosen programming environment.
- Python:
pip install spaco-release - R:
devtools::install_github("https://github.com/BrainStOrmics/SpacoR")[81]
- Python:
- Calculate Cluster Interlacement: Use Spaco to compute a spatial interlacement matrix, which quantifies the degree of neighborhood relationships (DOI metric) between different cell clusters [81].
- Generate & Align Color Palette: Spaco generates a color palette with high perceptual contrast between colors. The algorithm then optimizes the assignment of colors to clusters by aligning the cluster interlacement matrix with the color contrast matrix.
- Visualization and Analysis: Plot the spatial data using the optimized color map. This ensures that spatially adjacent categories are assigned highly contrastive colors, enabling unbiased visual perception of tissue domains and pattern organization for more accurate benchmarking [81].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Tools for Turing Pattern Research
| Item | Function/Benefit | Example Use Case |
|---|---|---|
| GCaMP6s Transgenic Lines | Genetically encoded calcium indicator for monitoring intracellular Ca²⁺ dynamics, a common player in signaling networks. | Imaging calcium fluxes associated with ROP GTPase activity during pavement cell morphogenesis [78]. |
| Mueller Polarimeter | Measures the complete polarization response of a sample, providing metrics (IPPs, CPs) sensitive to microstructural changes. | Non-destructive detection of early plant disease symptoms before they are visually apparent [79]. |
| Spataco Software Package | A spatially-aware colorization algorithm that optimizes color assignments for categorical data in spatial visualizations. | Eliminating perceptual ambiguity in spatial transcriptomics plots, leading to clearer interpretation of spatial patterns [81]. |
| Low-Cost Sensor Networks | Enables dense deployment for high-resolution spatiotemporal monitoring of environmental variables. | Capturing microclimate data (humidity, temperature) at the scale of plant canopies for ecological pattern models [82]. |
| Multi-Task Graph-XGBoost (MTGXGB) | A machine learning model effective for spatiotemporal gap-filling in sparse sensor data. | Estimating street-level air pollution at 30-200m resolution, a technique adaptable for modeling soil moisture or VOC gradients in plant environments [80]. |
Experimental and Computational Workflow Diagrams
Diagram 1: Turing Pattern Benchmarking Workflow
Diagram 2: Key Turing Mechanism (Activator-Inhibitor)
Conclusion
Computational modeling solidifies Turing's reaction-diffusion framework as a powerful, unifying principle for explaining the self-organized regularity observed across plant biology, from subcellular structures to ecosystem-scale patterns. The key takeaways are the versatility of Turing systems beyond classic two-component models, the critical importance of rigorous model validation against biological data, and the utility of plants as ideal testbeds for exploring general principles of morphogenesis. Future directions involve tighter integration of mechanistic models with omics data, exploration of Turing principles in synthetic biological circuits, and translating insights from plant pattern formation to understand dysregulated patterning in human development and disease, particularly in processes like tissue branching, digit formation, and skin patterning. The continued dialogue between computational theory and plant biology promises to unlock further fundamental mechanisms of life's inherent order.
References
- 1. Turing pattern [https://en.wikipedia.org/wiki/Turing_pattern]
- 2. Turing's model for biological pattern formation and the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3363041/]
- 3. A Modern View on Turing's Theory of Pattern Formation [https://royalsociety.org/blog/2021/11/turing-theory-pattern-formation/]
- 4. The Turing heritage for plant biology: all spots and stripes? [https://pmc.ncbi.nlm.nih.gov/articles/PMC11811860/]
- 5. Computational modeling of plant root development [https://pubmed.ncbi.nlm.nih.gov/40269551/]
- 6. Pattern formation mechanisms of self-organizing reaction- ... [https://www.sciencedirect.com/science/article/pii/S001216061930377X]
- 7. Chia seedlings verify Alan Turing's ideas about patterns in ... [https://www.sciencenews.org/article/seeds-alan-turing-patterns-nature-math]
- 8. The Turing heritage for plant biology: all spots and stripes? [https://www.cambridge.org/core/journals/quantitative-plant-biology/article/turing-heritage-for-plant-biology-all-spots-and-stripes/27ACF2553C11B9F8E5A0D4218E0CE283]
- 9. Widespread biochemical reaction networks enable Turing ... [https://www.nature.com/articles/s41467-024-52591-0]
- 10. Watching the Daisies Grow: Turing and Fibonacci Phyllotaxis [https://link.springer.com/chapter/10.1007/978-3-662-05642-4_20]
- 11. After 1952: The later development of Alan Turing's ideas on ... [https://www.sciencedirect.com/science/article/pii/S0315086015000397]
- 12. Morphotexts — Alan Turing's Manchester [https://www.manturing.net/morphotexts]
- 13. A plausible model of phyllotaxis - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC1345713/]
- 14. Pattern formation in a spatial plant-wrack model with tide ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2825308/]
- 15. Turing-like mechanism in a stochastic reaction-diffusion model ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0219055]
- 16. Computational modeling of plant root development: the art ... [https://research-portal.uu.nl/en/publications/computational-modeling-of-plant-root-development-the-art-and-the-/]
- 17. A Theoretical Model of Jigsaw-Puzzle Pattern Formation by ... [https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1004833]
- 18. Cellular patterns in Arabidopsis root epidermis emerge ... [https://www.nature.com/articles/s41540-025-00551-9]
- 19. A common pathway controls cell size in the sepal and leaf ... [https://pubmed.ncbi.nlm.nih.gov/41183025/]
- 20. A common pathway controls cell size in the sepal and leaf ... [https://journals.plos.org/plosbiology/article?id=10.1371/journal.pbio.3003469]
- 21. Vegetation pattern formation and transition in dryland ... [https://www.sciencedirect.com/science/article/pii/S0167278925000806]
- 22. Differential impacts of grazing on vegetation patterning in ... [https://pubmed.ncbi.nlm.nih.gov/41258803/]
- 23. Function of membrane domains in rho-of-plant signaling - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8133555/]
- 24. RHO GTPase in plants: Conservation and invention of ... - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3116591/]
- 25. Rho of Plants patterning: linking mathematical models and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10901209/]
- 26. RHO GTPase of plants regulates polarized cell growth and ... [https://www.sciencedirect.com/science/article/pii/S0960982223007662]
- 27. A Rho-based reaction-diffusion system governs cell wall ... [https://www.nature.com/articles/s41598-018-29543-y]
- 28. A mathematical model of GTPase pattern formation during ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4992738/]
- 29. Integration of transport-based models for phyllotaxis and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2648550/]
- 30. An auxin-driven polarized transport model for phyllotaxis - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC1326488/]
- 31. Integration of transport-based models for phyllotaxis and ... [https://genesdev.cshlp.org/content/23/3/373]
- 32. Toward a 3D model of phyllotaxis based on a biochemically ... [https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1006896]
- 33. The role of auxin transport through plasmodesmata in leaf ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1621815/full]
- 34. Unveiling Biological Models Through Turing Patterns [https://arxiv.org/html/2509.07458v1]
- 35. Formation of spatial vegetation patterns in heterogeneous ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0324181]
- 36. Accelerated Numerical Simulations of a Reaction-Diffusion [https://www.mdpi.com/2227-7390/13/9/1488]
- 37. Isolating Patterns in Open Reaction–Diffusion Systems [https://link.springer.com/article/10.1007/s11538-021-00913-4]
- 38. Advection-reaction-diffusion coupled with viscous flow in a ... [https://www.sciencedirect.com/science/article/pii/S0045782525005833?dgcid=rss_sd_all]
- 39. Spectral analysis of reaction-diffusion systems via physics ... [https://www.aimspress.com/article/doi/10.3934/math.2025809?viewType=HTML]
- 40. Computational modeling of plant root development [https://pmc.ncbi.nlm.nih.gov/articles/PMC12095987/]
- 41. Dynamics of Posttranslational Modification Systems [https://pmc.ncbi.nlm.nih.gov/articles/PMC5985040/]
- 42. The rational parameterisation theorem for multisite post- ... [https://www.sciencedirect.com/science/article/abs/pii/S0022519309004238]
- 43. Kinetic modeling reveals additional regulation at co ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8634553/]
- 44. Modeling post-transcriptional regulation activity of small non ... [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/1471-2105-10-S4-S6]
- 45. Plant synthetic biology: from knowledge to biomolecules [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1562216/full]
- 46. Practical Perspectives on Enhancing Predictive Modeling ... [https://link.springer.com/article/10.1007/s40495-025-00426-x]
- 47. Support identification for parameter variations in a PDE ... [https://www.aimsciences.org/article/doi/10.3934/cac.2025023]
- 48. Recovery of transversely-isotropic elastic material parameters ... [https://mathematicsinindustry.springeropen.com/articles/10.1186/s13362-025-00179-8]
- 49. WF-PINNs: solving forward and inverse problems of ... [https://www.nature.com/articles/s41598-025-24427-4]
- 50. The plant proteome delivers from discovery to innovation [https://www.sciencedirect.com/science/article/abs/pii/S1360138525000639]
- 51. Artificial intelligence-driven plant bio-genomics research [https://www.maxapress.com/article/doi/10.48130/tp-0025-0008]
- 52. Turing patterns in a morphogenetic model with single ... [https://www.sciencedirect.com/science/article/abs/pii/S0025556425001622]
- 53. Adapting modeling and simulation credibility standards to ... [https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-023-04290-5]
- 54. From FAIR to CURE: Guidelines for Computational Models ... [https://ui.adsabs.harvard.edu/abs/2025arXiv250215597S/abstract]
- 55. Optimal network sizes for most robust Turing patterns [https://ui.adsabs.harvard.edu/abs/2025NatSR..15.2948S/abstract]
- 56. Multiscale integration in Biological Systems [https://training.institut-curie.org/courses/multiscale-integration-in-biological-systems-2025]
- 57. Review Multi-scale computational modelling in biology and ... [https://www.sciencedirect.com/science/article/pii/S0079610707000673]
- 58. On multiscale approaches to three-dimensional modelling of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1629079/]
- 59. Deep learning for three-dimensional (3D) plant phenomics [https://www.sciencedirect.com/science/article/pii/S264365152500113X]
- 60. Editorial: Leveraging phenotyping and crop modeling in ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1626622/full]
- 61. Early Plant Classification Model Based on Dual Attention ... [https://www.mdpi.com/2624-7402/7/3/66]
- 62. Model order reduction [https://en.wikipedia.org/wiki/Model_order_reduction]
- 63. Model Reduction and Time Series [https://physicsbaseddeeplearning.org/others-timeseries.html]
- 64. A high-throughput differential chemical genetic screen ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12032938/]
- 65. Plant synthetic biology: from knowledge to biomolecules - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12602438/]
- 66. Synthetic biology and metabolic engineering strategies in ... [https://www.sciencedirect.com/science/article/pii/S0926669025006065]
- 67. Plant Sciences: Protocols & Methods - Research Guides [https://guides.library.ucdavis.edu/plant-sciences/protocols-and-methods]
- 68. Turing pattern design principles and their robustness - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8580431/]
- 69. Beyond Turing: mechanochemical pattern formation in ... [https://biologydirect.biomedcentral.com/articles/10.1186/s13062-016-0124-7]
- 70. Post-Turing tissue pattern formation - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC6047832/]
- 71. Behind phyllotaxis, within the meristem: a REM‐ARF complex ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11873677/]
- 72. Spiral phyllotaxis predicts left-right asymmetric growth and ... [https://www.nature.com/articles/s41467-025-58803-5]
- 73. How animals get their spots, and why they are beautifully ... [https://www.colorado.edu/today/2025/10/27/how-animals-get-their-spots-and-why-they-are-beautifully-imperfect]
- 74. The developmental basis of fingerprint pattern formation ... [https://www.sciencedirect.com/science/article/pii/S0092867423000454]
- 75. Genetic Insights Into Arch Fingerprint Patterns [https://www.technologynetworks.com/genomics/news/fingerprint-patterns-influenced-by-the-same-genes-as-limb-development-357292]
- 76. On pattern formation in the thermodynamically-consistent ... [https://www.sciencedirect.com/science/article/abs/pii/S0025556425000793]
- 77. Global performance benchmarking of artificial intelligence ... [https://www.nature.com/articles/s43247-025-02823-y]
- 78. Diversity of spatiotemporal coding reveals specialized ... [https://www.nature.com/articles/s41467-022-29656-z]
- 79. Polarimetric observables for the enhanced visualization of ... [https://www.nature.com/articles/s41598-022-19088-6]
- 80. High-resolution spatio-temporal estimation of street-level ... [https://www.sciencedirect.com/science/article/abs/pii/S0301479725006188]
- 81. Protocol for enhancing visualization clarity for categorical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11103473/]
- 82. PM 2.5 Concentration Prediction: Ultrahigh Spatiotemporal ... [https://pubmed.ncbi.nlm.nih.gov/40942956/]