Carbon Nanotubes vs. Graphene: The Race to Develop Advanced Plant H2O2 Sensors
This article provides a comparative analysis of carbon nanotubes (CNTs) and graphene as foundational materials for electrochemical biosensors targeting hydrogen peroxide (H2O2) in plants.
Carbon Nanotubes vs. Graphene: The Race to Develop Advanced Plant H2O2 Sensors
Abstract
This article provides a comparative analysis of carbon nanotubes (CNTs) and graphene as foundational materials for electrochemical biosensors targeting hydrogen peroxide (H2O2) in plants. Aimed at researchers and scientists, it explores the fundamental properties and sensing mechanisms of these nanomaterials, details cutting-edge methodological applications for real-time plant health monitoring, addresses key challenges in sensor optimization, and presents a direct performance comparison. By synthesizing the latest research, this review serves as a guide for selecting and developing nanomaterial-based sensing platforms to decode plant stress signaling, with significant implications for precision agriculture and crop development.
Understanding the Building Blocks: The Intrinsic Properties of CNTs and Graphene for H2O2 Sensing
The development of advanced sensing platforms for detecting hydrogen peroxide (H₂O₂) in plant systems represents a critical frontier in agricultural and botanical research. As a key signaling molecule and stress indicator in plants, H₂O₂ requires precise monitoring to understand plant physiology and stress responses. Two carbon allotropes have emerged as particularly transformative materials for electrochemical sensing: one-dimensional carbon nanotubes (CNTs) and two-dimensional graphene. These nanomaterials provide the fundamental building blocks for a new generation of sensors, each leveraging distinct structural and electronic characteristics that dictate their performance in H₂O₂ detection applications. This guide provides an objective comparison of these materials, supported by experimental data and detailed methodologies, to inform researchers and scientists developing next-generation plant biosensors.
Fundamental Structural and Electronic Properties
The dimensional character of carbon nanomaterials—whether 1D or 2D—imparts distinct electronic and structural properties that directly influence their sensing capabilities.
Carbon Nanotubes (CNTs) are cylindrical nanostructures composed of rolled graphene sheets with sp²-hybridized carbon atoms in a hexagonal lattice. They are categorized as single-walled (SWCNTs), consisting of a single graphene cylinder with diameters of 0.4-2 nm, or multi-walled (MWCNTs), composed of multiple concentric cylinders with diameters of 2-100 nm [1]. Their quasi-1D structure creates a high aspect ratio, providing exceptional electron transport pathways along their longitudinal axis. CNTs exhibit either metallic or semiconducting behavior depending on their chirality and diameter, with bandgaps varying from zero to approximately 2 eV [2]. This tunable electronic structure, combined with remarkable tensile strength (~100 times stronger than steel) and high thermal conductivity (3000-3500 W/mK for SWCNTs), makes them particularly suitable for sensor applications where direct electron transfer is critical [1].
Graphene is a single atomic layer of graphite arranged in a two-dimensional honeycomb lattice with a carbon-carbon distance of 0.142 nm [3]. As a 2D material, it provides an extensive planar surface area (theoretically ~2630 m²/g) for analyte interaction [1]. Pristine graphene is a zero-bandgap semiconductor with exceptional charge carrier mobility, high electrical conductivity, and substantial mechanical flexibility [2] [3]. However, for sensing applications, graphene derivatives are often employed, including graphene oxide (GO) and reduced graphene oxide (rGO), which introduce functional groups that enhance analyte binding and dispersion capabilities [3].
Table 1: Fundamental Properties of CNTs and Graphene for Sensing Applications
| Property | Carbon Nanotubes (CNTs) | Graphene |
|---|---|---|
| Dimensionality | 1D (Quasi-one-dimensional) | 2D (Two-dimensional) |
| Electrical Conductivity | 10²-10⁵ S/m [1] | ~10⁴ S/m [1] |
| Specific Surface Area | >1000 m²/g [1] | ~2630 m²/g [1] |
| Mechanical Strength | Young's modulus ~1 TPa [1] | Young's modulus ~1 TPa [1] |
| Bandgap Characteristics | Metallic or semiconducting based on chirality (0-2 eV) [2] | Zero bandgap (semimetal) [2] |
| Functionalization Capability | Excellent (covalent and non-covalent) [1] | Excellent [1] |
Performance Comparison in H₂O₂ Sensing
Experimental data from recent studies demonstrates how these fundamental properties translate to practical H₂O₂ sensing performance, with direct implications for plant research where monitoring oxidative stress is crucial.
CNT-Based H₂O₂ Sensors leverage the material's high aspect ratio and efficient electron transfer pathways. A notable approach integrated multi-walled carbon nanotubes with hemin-polyethyleneimine (hemin-PEI) composites on screen-printed graphene electrodes for pseudo-peroxidase non-enzymatic H₂O₂ monitoring. This configuration demonstrated a sensitivity of 18.09 ± 0.89 A·M⁻¹·cm⁻² with a low onset potential of +0.2 V for H₂O₂ reduction, making it suitable for detecting biologically relevant H₂O₂ concentrations in complex matrices like exhaled breath condensate [4]. The hemin provided peroxidase-mimicking activity, while MWCNTs enhanced electrode conductivity and electron transfer efficiency, addressing the stability issues associated with natural enzymes [4].
Graphene-Based H₂O₂ Sensors exploit the material's extensive planar surface for functionalization and analyte interaction. A recent study developed a sensor using electrodeposited silver nanoparticle/reduced graphene oxide (AgNPs/rGO) nanocomposites on glassy carbon electrodes. This configuration demonstrated a linear response to H₂O₂ in the range of 5 μM to 620 μM with a sensitivity of 49 μA·mM⁻¹·cm⁻² and a detection limit of 3.19 μM [5]. The synergistic effect between the catalytic properties of AgNPs and the large surface area and excellent conductivity of rGO enabled non-enzymatic H₂O₂ detection with good stability over time [5].
Hybrid Approaches combining both materials have shown exceptional performance by leveraging their complementary characteristics. A 3D porous Au/CuO/Pt hybrid framework demonstrated an ultra-high sensitivity of 25,836 μA·mM⁻¹·cm⁻² with a very low limit of detection of 9.8 nM for H₂O₂ [6]. While not exclusively carbon-based, this architecture exemplifies how multidimensional carbon structures can enhance sensor performance through increased electrochemical surface area and improved electron transfer pathways.
Table 2: Experimental H₂O₂ Sensing Performance of Carbon Nanomaterials
| Material Platform | Sensitivity | Linear Range | Limit of Detection (LOD) | Key Advantages |
|---|---|---|---|---|
| Hemin-PEI/MWCNT/SPGE [4] | 18.09 ± 0.89 A·M⁻¹·cm⁻² | Not specified | Low μM range (suitable for biological applications) | Low onset potential (+0.2 V), high selectivity, avoids enzyme instability |
| AgNPs/rGO/GCE [5] | 49 μA·mM⁻¹·cm⁻² | 5 μM to 620 μM | 3.19 μM | Facile fabrication, good stability, eco-friendly preparation |
| 3D Porous Au/CuO/Pt [6] | 25,836 μA·mM⁻¹·cm⁻² | Not specified | 9.8 nM | Ultra-high sensitivity, abundant active sites, improved electron transfer |
| Prussian Blue-Based Sensors [7] | Varies by design | 4 μM to 1064 μM (example) | 0.226 μM (example) | "Artificial peroxidase," operates at low voltages minimizing interference |
Experimental Protocols and Methodologies
CNT-Based Sensor Fabrication (Hemin-PEI/MWCNT)
The development of hemin-PEI/MWCNT modified screen-printed graphene electrodes involves a multi-step process that leverages the unique properties of each component [4]:
- MWCNT Modification: Multi-walled carbon nanotubes are dispersed in appropriate solvents and integrated with the screen-printed graphene electrode substrate through drop-casting or electrochemical deposition.
- Hemin-PEI Composite Formation: Hemin (chloroprotoporphyrin IX iron(III)) is entrapped in a polyethyleneimine (PEI) matrix at a specific ratio (e.g., 1:5 hemin:PEI) to prevent hemin dimerization and maintain its catalytic activity.
- Electrode Modification: The hemin-PEI composite is applied to the MWCNT-modified electrode surface, creating a pseudo-peroxidase sensing interface.
- Characterization: The modified electrode is characterized using scanning electron microscopy (SEM) to confirm morphology and electrochemical impedance spectroscopy (EIS) to verify enhanced electron transfer properties.
This configuration capitalizes on the peroxidase-mimicking activity of hemin while utilizing MWCNTs to enhance conductivity and electron transfer efficiency between the electrode and the catalytic sites [4].
Graphene-Based Sensor Fabrication (AgNPs/rGO)
The construction of silver nanoparticle/reduced graphene oxide sensors employs a combination of hydrothermal synthesis and electrochemical deposition [5]:
- AgNPs/GO Composite Synthesis: Graphene oxide solution is mixed with silver nitrate (AgNO₃) and sodium citrate as a reducing agent. The mixture is heated at 60°C with stirring for several hours, resulting in the growth of silver nanoparticles on graphene oxide sheets.
- Electrode Preparation: Glassy carbon electrodes are polished with alumina powders (1.0, 0.3, and 0.05 µm) and thoroughly cleaned through sonication in ethanol and deionized water.
- Electrodeposition: The polished electrodes are placed in the AgNPs/GO composite solution and electrodeposited at -1.3 V for 600 seconds to reduce GO to rGO and simultaneously deposit the nanocomposite on the electrode surface.
- Characterization: The successful formation of AgNPs on graphene sheets is verified using SEM, UV-Vis spectroscopy, and X-ray diffraction (XRD).
This method provides a green, one-step approach for sensor fabrication that eliminates the need for harsh reducing agents typically used in graphene oxide reduction [5].
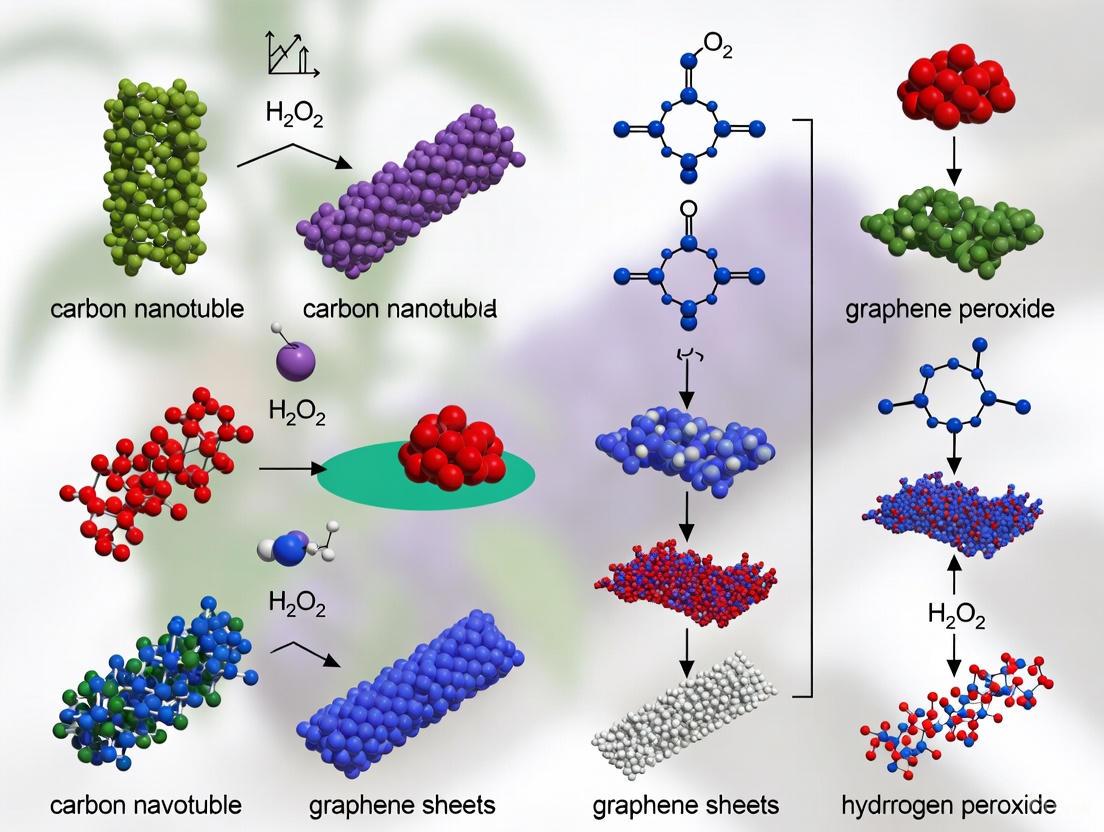
Material Synthesis and Functionalization Pathways
The synthesis and functionalization of carbon nanomaterials significantly influence their properties and performance in sensing applications. The following diagram illustrates the key pathways for developing functionalized CNT and graphene platforms for H₂O₂ sensing.
Carbon Nanomaterial Synthesis Pathways for H₂O₂ Sensing
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful development of CNT and graphene-based H₂O₂ sensors requires specific materials and reagents tailored to exploit the unique properties of each nanomaterial.
Table 3: Essential Research Materials for Carbon Nanomaterial-Based H₂O₂ Sensors
| Material/Reagent | Function in Research | Examples in CNT/Graphene Sensors |
|---|---|---|
| Carbon Nanotubes | Primary sensing element providing conductivity and scaffold | SWCNTs for high sensitivity; MWCNTs for robust platforms [1] |
| Graphene Oxide (GO) | Precursor for rGO-based sensors; provides functional groups for modification | Starting material for AgNPs/rGO composites [5] |
| Metal Nanoparticles | Enhance catalytic activity and electron transfer | AgNPs, AuNPs, PtNPs for H₂O₂ electrocatalysis [5] [6] |
| Hemin | Peroxidase-mimicking cofactor for enzyme-free sensing | Hemin-PEI complexes on MWCNT electrodes [4] |
| Polyethyleneimine (PEI) | Cationic polymer matrix for stabilizing hemin | Prevents hemin aggregation in CNT-based sensors [4] |
| Electrode Substrates | Platform for sensor construction | Screen-printed graphene electrodes, glassy carbon electrodes [4] [5] |
| Functionalization Agents | Modify surface properties for enhanced selectivity | Covalent (acid treatment) and non-covalent (polymer wrapping) approaches [8] |
The selection between 1D CNTs and 2D graphene for plant H₂O₂ sensing applications depends critically on the specific research requirements. CNTs offer advantages in electron transfer efficiency along their longitudinal axis and tunable semiconducting properties, making them ideal for applications requiring direct electron transfer and high sensitivity. Graphene provides an extensive 2D surface for functionalization and interaction with analytes, beneficial for sensors requiring high functional group density and planar architecture. Emerging hybrid approaches that combine these materials in three-dimensional architectures demonstrate particularly promising performance by leveraging the complementary advantages of both dimensionalities. For plant research specifically, where H₂O₂ functions as a key signaling molecule in stress responses and developmental processes, CNT-based sensors may offer superior sensitivity for detecting subtle concentration changes, while graphene-based platforms may provide better stability for prolonged monitoring applications. The continuing advancement of functionalization strategies and hybrid material systems promises to further enhance the capabilities of both platforms for precise H₂O₂ monitoring in plant systems.
The Critical Role of H2O2 as a Plant Stress Signaling Molecule
Hydrogen peroxide (H₂O₂) is a reactive oxygen species (ROS) that has evolved from being considered merely a cytotoxic byproduct of metabolism to a crucial signaling molecule regulating numerous aspects of plant growth, development, and stress adaptation [9] [10] [11]. At elevated concentrations, H₂O₂ can cause oxidative damage to cellular components; however, at low nanomolar levels, it serves as a key mediator in signal transduction pathways that enable plants to respond to environmental challenges [11]. The dual nature of H₂O₂ necessitates precise spatial and temporal control of its concentration within plant tissues, maintained through a balance between production systems (e.g., NADPH oxidases, electron transport chains) and scavenging mechanisms (e.g., catalases, peroxidases, ascorbate peroxidase) [10].
In the context of plant stress responses, H₂O₂ functions as a secondary messenger that can modulate gene expression, hormone signaling, and physiological adaptations [11]. Its relatively longer half-life compared to other ROS and its ability to diffuse across membranes make it an ideal signaling molecule [9]. This review explores the critical role of H₂O₂ in plant stress signaling, with particular emphasis on emerging applications of carbon nanomaterials, specifically carbon nanotubes and graphene, in detecting and quantifying H₂O₂ for plant research.
H₂O₂ Signaling Mechanisms in Plant Stress Responses
Production and Homeostasis of H₂O₂ in Plant Cells
Hydrogen peroxide is continually generated in plant cells through both enzymatic and non-enzymatic pathways, with its concentration varying significantly across different cellular compartments and in response to environmental conditions [10]. The major sites of H₂O₂ production include chloroplasts, where it results from photosynthetic electron transport; mitochondria, where it is generated by electron leakage from the respiratory chain; and peroxisomes, where it is produced during photorespiration [10]. Additionally, dedicated enzymes such as NADPH oxidases (also known as Respiratory Burst Oxidase Homologs or RBOHs) actively generate H₂O₂ at the plasma membrane in response to specific stimuli [10] [11].
Table 1: Primary Sources and Scavengers of H₂O₂ in Plant Cells
| Category | Component | Location | Function |
|---|---|---|---|
| Production Sources | NADPH Oxidases | Plasma Membrane | Enzymatic production of superoxide which is converted to H₂O₂ |
| Photosynthetic Electron Transport | Chloroplasts | Generates H₂O₂ through reduction of oxygen | |
| Photorespiration | Peroxisomes | H₂O₂ synthesis associated with glycolate oxidation | |
| Electron Transport Chain | Mitochondria | Produces H₂O₂ via superoxide formation at complexes I and III | |
| Scavenging Systems | Catalase (CAT) | Peroxisomes | Direct decomposition of H₂O₂ to water and oxygen |
| Ascorbate Peroxidase (APX) | Chloroplasts, Cytosol, Mitochondria | Uses ascorbate to reduce H₂O₂ to water | |
| Peroxidase (POX) | Cell Wall, Cytosol | Reduces H₂O₂ while oxidizing various substrates | |
| Glutathione Reductase (GR) | Various compartments | Maintains glutathione pool for H₂O₂ detoxification | |
| Non-enzymatic Antioxidants | Throughout cell | Ascorbate and glutathione directly scavenge H₂O₂ |
The homeostasis of H₂O₂ is maintained by an intricate antioxidant system comprising both enzymatic and non-enzymatic components [10]. Enzymes such as catalases (CAT), ascorbate peroxidases (APX), and various peroxidases (POX) work in concert with low-molecular-weight antioxidants like ascorbate and glutathione to regulate H₂O₂ levels, allowing it to function as a signal without causing oxidative damage [10]. This precise control enables H₂O₂ to participate in diverse signaling cascades while minimizing cellular harm.
H₂O₂-Mediated Signal Transduction Pathways
Hydrogen peroxide influences plant stress responses through multiple interconnected signaling pathways. One key mechanism involves the oxidative post-translational modification of cysteine residues in proteins, which can alter their activity, stability, or interactions [11]. For instance, H₂O₂ can oxidize cysteine thiol groups (-SH) to sulfenic (-SOH), sulfinic (-SO₂H), or sulfonic (-SO₃H) acids, effectively acting as a redox switch that controls protein function [11].
Another significant pathway involves the activation of mitogen-activated protein kinase (MAPK) cascades, which transduce extracellular signals to intracellular responses [11]. H₂O₂ can activate specific MAPKs that subsequently phosphorylate transcription factors, leading to changes in gene expression that enhance stress tolerance [11]. Moreover, H₂O₂ influences the expression of various transcription factors that regulate stress-responsive genes, including those involved in antioxidant defense, osmolyte synthesis, and detoxification processes [11].
Diagram 1: H₂O₂-Mediated Stress Signaling Pathways in Plants. Hydrogen peroxide acts as a central signaling molecule that transduces stress signals through multiple interconnected pathways to elicit adaptive responses.
Cross-Talk with Other Signaling Molecules
H₂O₂ does not function in isolation but engages in extensive cross-talk with other signaling molecules, including calcium (Ca²⁺), nitric oxide (NO), and various phytohormones [10] [11]. This integrative signaling network allows plants to fine-tune their responses to simultaneous or sequential environmental challenges.
The interplay between H₂O₂ and calcium signaling is particularly important for stress adaptation. H₂O₂ can activate calcium channels in the plasma membrane and organelles, leading to increases in cytosolic Ca²⁺ that subsequently activate calcium-dependent protein kinases and other downstream effectors [10]. Similarly, the cross-talk between H₂O₂ and nitric oxide regulates key processes such as stomatal closure, programmed cell death, and gene expression in response to abiotic and biotic stresses [10]. Furthermore, H₂O₂ interacts with multiple plant growth regulators, including auxin, abscisic acid (ABA), salicylic acid (SA), and jasmonic acid (JA), often in a synergistic or antagonistic manner to modulate stress responses [9] [11].
Table 2: H₂O₂ Cross-Talk with Other Signaling Molecules in Stress Responses
| Signaling Molecule | Nature of Cross-Talk with H₂O₂ | Functional Outcome |
|---|---|---|
| Calcium (Ca²⁺) | H₂O₂ activates Ca²⁺ channels; Ca²⁺ regulates NADPH oxidases | Amplification of stress signals; Regulation of stomatal closure |
| Nitric Oxide (NO) | Mutual enhancement or suppression depending on context | Modulation of defense gene expression; Programmed cell death |
| Auxin | H₂O₂ regulates auxin gradients and signaling | Root architecture remodeling under stress |
| Abscisic Acid (ABA) | H₂O₂ acts as downstream signaling component in ABA responses | Stomatal closure; Activation of antioxidant defenses |
| Salicylic Acid (SA) | Synergistic interaction in pathogen responses | Systemic acquired resistance; Pathogen defense |
| Jasmonic Acid (JA) | Complex, context-dependent interactions | Defense against herbivores and necrotrophic pathogens |
Carbon Nanotubes vs. Graphene for H₂O₂ Sensing in Plant Research
Properties and Applications in Sensing Platforms
The detection and quantification of H₂O₂ in plant tissues present significant technical challenges due to its low concentration, transient nature, and the complexity of the plant matrix. Carbon nanomaterials have emerged as promising platforms for H₂O₂ sensing owing to their exceptional electrical, thermal, and mechanical properties [12] [4]. Among these, carbon nanotubes (CNTs) and graphene have received particular attention for electrochemical sensing applications.
Carbon nanotubes exist as single-walled (SWCNTs) or multi-walled (MWCNTs) structures, with diameters ranging from 4 to 30 nm and lengths up to 1 μm [12]. Their high surface area, excellent conductivity, and ability to be functionalized make them attractive for sensor design [12]. Graphene, a two-dimensional material consisting of single-layer carbon atoms arranged in a honeycomb lattice, offers high electrical conductivity, large surface area, and exceptional electrocatalytic activity [12]. Both materials can be integrated into various electrode configurations, including screen-printed electrodes, which provide a disposable and reproducible platform for scalable sensor fabrication [4].
A critical consideration in the selection of sensing materials is their environmental impact and biodegradability. Studies indicate that graphene and carbon dots are more readily biodegradable compared to CNTs, which can form persistent colloidal suspensions in the environment [12]. This distinction may influence material selection for certain applications, particularly those involving direct plant exposure or field deployment.
Comparative Performance in H₂O₂ Detection
Direct comparison of CNT-based and graphene-based sensors reveals distinct advantages for each material depending on the specific application requirements. Recent research has developed innovative sensor designs utilizing both material types with impressive performance characteristics for H₂O₂ detection.
A notable CNT-based sensor incorporating hemin-polyethyleneimine/MWCNT modified screen-printed graphene electrodes demonstrated high sensitivity (18.09 ± 0.89 A M⁻¹ cm⁻²) for H₂O₂ detection with a low onset potential for H₂O₂ reduction at approximately +0.2 V [4]. This sensor configuration leveraged the peroxidase-mimicking activity of hemin while utilizing MWCNTs to enhance electrode conductivity and electron transfer efficiency [4].
Table 3: Performance Comparison of Carbon Nanomaterial-Based H₂O₂ Sensors
| Sensor Type | Detection Limit | Sensitivity | Linear Range | Key Advantages |
|---|---|---|---|---|
| Hemin-PEI/MWCNT/SPGE [4] | Not specified | 18.09 ± 0.89 A M⁻¹ cm⁻² | 0.05-1000 μM | Low onset potential; High sensitivity; Good selectivity |
| Prussian Blue-modified SPCE [4] | 5 μM | Not specified | Not specified | Simplicity; Compatibility with wearable formats |
| HRP-modified Gold Electrodes [4] | 10 nM | 1.4-1.5 A M⁻¹ cm⁻² | Not specified | High sensitivity; Fast electron transfer |
| Sugar Beet Hb-Modified Graphite [4] | 10 μM | 87 mA M⁻¹ cm⁻² | Not specified | Biocompatibility; Moderate sensitivity |
Graphene-based electrodes benefit from the material's high conductivity and large surface area, which promote efficient electron transfer and high loading capacity for catalytic materials [12] [4]. The integration of graphene into screen-printed electrodes (SPGEs) offers additional advantages for practical applications, including disposability, reproducibility, and cost-effectiveness for mass production [4]. While direct performance comparisons between CNT and graphene sensors for plant H₂O₂ detection are limited in the current literature, both platforms show promise for different aspects of plant research, with selection dependent on the specific requirements for sensitivity, selectivity, cost, and environmental considerations.
Experimental Approaches for H₂O₂ Analysis in Plant Systems
DAB Staining for In Situ H₂O₂ Detection
The 3,3'-diaminobenzidine (DAB) staining method is widely used for the in situ detection of H₂O₂ in plant tissues, providing spatial information about H₂O₂ accumulation at the organ, tissue, or cellular level [13]. This method exploits the principle that DAB is oxidized by H₂O₂ in the presence of peroxidases, generating a dark brown precipitate that can be visualized microscopically [13].
The standard DAB staining protocol involves the following key steps [13]:
- Preparation of DAB staining solution: Dissolve DAB powder in sterile water (1 mg/mL) and adjust pH to 3.0 using HCl. Add Tween 20 (0.05% v/v) and Na₂HPO₄ to achieve a 10 mM phosphate concentration.
- Sample collection and infiltration: Excise plant tissues and immerse in DAB solution. Apply gentle vacuum infiltration for 5 minutes to ensure solution penetration.
- Incubation: Shake samples in the dark for 4-8 hours to allow stain development.
- Destaining: Replace DAB solution with bleaching solution (ethanol:acetic acid:glycerol = 3:1:1) and incubate in a boiling water bath for ~15 minutes to remove chlorophyll.
- Visualization and documentation: Observe stained tissues against a white background under uniform lighting.
Diagram 2: DAB Staining Workflow for In Situ H₂O₂ Detection in Plant Tissues. This histochemical method enables spatial localization of H₂O₂ accumulation in plant samples.
Electrochemical Sensing with Carbon Nanomaterials
Electrochemical sensors based on carbon nanomaterials offer complementary advantages for H₂O₂ detection, including high sensitivity, rapid response, minimal sample volume requirements, and potential for in vivo monitoring [4]. The general fabrication process for these sensors involves several key steps [12] [4]:
- Electrode preparation: Clean and activate the base electrode (typically glassy carbon electrode) to ensure proper adhesion of the nanomaterial.
- Nanomaterial functionalization: Modify CNTs or graphene with oxygen-containing functional groups (-COOH, -OH) to enhance dispersibility and provide tethering points for catalytic nanoparticles.
- Composite formation: Decorate functionalized nanomaterials with catalytic nanoparticles (e.g., metals, metal oxides, hemin) to enhance sensitivity and selectivity for H₂O₂ detection.
- Electrode modification: Deposit the nanomaterial composite onto the electrode surface, often using a binder such as Nafion to ensure stability.
- Electrochemical measurement: Employ techniques such as cyclic voltammetry, chronoamperometry, or differential pulse voltammetry to quantify H₂O₂ based on its reduction or oxidation current.
A particularly effective approach involves the use of hemin-based composites, where hemin (an iron protoporphyrin complex that serves as the catalytic center of peroxidases) is entrapped in a polymer matrix such as polyethyleneimine (PEI) and combined with MWCNTs on screen-printed graphene electrodes [4]. This design mimics the catalytic activity of natural peroxidases while offering the stability and conductivity advantages of carbon nanomaterials.
Applications in Plant Stress Phenotyping
Measurement of H₂O₂ concentrations in plant tissues has practical applications beyond basic research, including its use as an indicator of abiotic stress in ecological and agricultural contexts [14]. A recent study demonstrated that foliar H₂O₂ concentration can serve as a sensitive biomarker for determining species-specific distribution zones of riparian vegetation along elevation gradients relative to water level [14].
Researchers found that different plant species maintained characteristic H₂O₂ concentrations corresponding to their preferred soil moisture zones [14]. For example, Salix species growing in high soil moisture conditions showed decreasing H₂O₂ concentrations with increasing soil moisture, while other species exhibited different response patterns [14]. Importantly, all species showed spatial distributions limited to elevations where their foliar H₂O₂ concentrations remained below approximately 40 μmol/g fresh weight, suggesting this threshold represents a physiological limit for sustainable growth [14]. This application demonstrates how H₂O₂ measurements can provide valuable insights for vegetation management and restoration ecology.
The Scientist's Toolkit: Essential Reagents and Materials
Table 4: Key Research Reagents for Studying H₂O₂ in Plant Systems
| Reagent/Material | Function/Application | Examples/Specific Uses |
|---|---|---|
| DAB (3,3'-Diaminobenzidine) | Histochemical detection of H₂O₂ | In situ localization of H₂O₂ in plant tissues; Qualitative assessment |
| Carbon Nanotubes (CNTs) | Electrode modification for sensing | Enhance conductivity and surface area; Support for catalytic nanoparticles |
| Graphene | Electrode material for sensing | High conductivity platform; Base material for screen-printed electrodes |
| Hemin | Peroxidase mimic for catalysis | Electrocatalytic reduction of H₂O₂; Alternative to enzyme-based detection |
| Polyethyleneimine (PEI) | Cationic polymer matrix | Stabilizes hemin; Prevents aggregation; Enhances catalytic performance |
| Screen-Printed Electrodes | Disposable sensor platforms | Reproducible, mass-producible sensors; Point-of-care applications |
| Catalytic Nanoparticles | Signal enhancement | Prussian blue, metal oxides; Improve sensitivity and selectivity |
| Antioxidant Enzymes | Specificity controls | Catalase, peroxidase; Confirm H₂O₂ identity in complex samples |
Hydrogen peroxide serves as a central signaling molecule in plant stress responses, integrating information from various environmental cues and coordinating adaptive processes through complex networks involving calcium, nitric oxide, and phytohormones [10] [11]. The detection and quantification of H₂O₂ in plant systems present both challenges and opportunities for methodological innovation.
Carbon nanotubes and graphene offer complementary advantages for H₂O₂ sensing applications in plant research [12] [4]. CNT-based sensors, particularly when functionalized with catalytic materials such as hemin, provide high sensitivity and efficient electron transfer [4]. Graphene-based platforms offer superior conductivity, ease of functionalization, and advantages for scalable production through screen-printing technologies [12] [4]. The choice between these nanomaterials depends on specific research requirements, including sensitivity needs, sample type, measurement environment, and considerations of environmental impact.
Future developments in this field will likely focus on enhancing the specificity and temporal resolution of H₂O₂ measurements, enabling researchers to capture the dynamic spatial and temporal patterns of H₂O₂ signaling in living plants. The integration of these sensing technologies with other omics approaches will provide unprecedented insights into the role of H₂O₂ in plant stress adaptation and potentially contribute to the development of stress-resilient crops for sustainable agriculture.
In plant physiology, hydrogen peroxide (H₂O₂) is a crucial signaling molecule that mediates responses to environmental stresses such as UV radiation, pathogen attack, and gravity changes [15]. The precise monitoring of H₂O₂ homeostasis is essential for understanding redox signaling in plants, a process intrinsically linked to cytosolic calcium levels [15]. Traditional enzymatic biosensors, while effective, often suffer from limitations such as enzyme degradation over time, prompting the development of robust, enzyme-free alternatives [7].
Nanomaterials, particularly carbon nanotubes (CNTs) and graphene, have emerged as premier transducers for electrochemical H₂O₂ sensing due to their exceptional properties, including high electrical conductivity, large surface area, and excellent electrocatalytic capabilities [16] [7]. This guide provides an objective comparison of CNT and graphene-based sensing platforms, focusing on their inherent sensing mechanisms—charge transfer, adsorption, and electrostatic gating—within the context of plant H₂O₂ research, supported by experimental data and detailed methodologies.
Fundamental Sensing Mechanisms at the Nanoscale
The operational principles of CNT and graphene-based electrochemical sensors revolve around three core mechanisms that occur when the target analyte, H₂O₂, interacts with the nanomaterial surface. The following diagram illustrates the logical relationships and workflows involved in these fundamental sensing mechanisms.
Charge Transfer
This mechanism involves the direct exchange of electrons between H₂O₂ molecules and the carbon lattice of the nanomaterial. H₂O₂ can act as an electron donor or acceptor, modifying the carrier concentration and shifting the Fermi level of the semiconductor, which leads to a measurable change in electrical conductance [17].
Adsorption
The physical or chemical adsorption of H₂O₂ molecules onto the nanomaterial's surface can alter the local surface potential and act as a doping agent. This is particularly effective in graphene field-effect transistors (GFETs), where adsorbed molecules can induce charge carrier scattering or doping effects, modulating the device's conductivity [18] [17].
Electrostatic Gating
In a Field-Effect Transistor (FET) configuration, adsorbed H₂O₂ molecules can create an electrostatic gating effect. The molecules function similarly to a gate voltage, electrostatically modulating the charge carrier density in the channel (composed of CNTs or graphene), thereby changing the source-drain current without direct charge transfer [17].
Performance Comparison: Carbon Nanotubes vs. Graphene for H₂O₂ Sensing
The table below summarizes key performance metrics from recent studies for a direct comparison of CNT and graphene-based H₂O₂ sensors.
Table 1: Performance Comparison of CNT and Graphene-Based H₂O₂ Sensors
| Material Platform | Sensitivity (μA mM⁻¹ cm⁻²) | Linear Range (mM) | Detection Limit (μM) | Working Potential (V vs. Ag/AgCl) | Key Mechanism |
|---|---|---|---|---|---|
| CNT-based Sensor (RGO/CNTs-Pt/GCE) [19] | 347 ± 5 | 0.0003 - 0.018 & 0.01 - 4.0 | 0.31 | -0.2 V | Charge Transfer & Catalytic Reduction |
| Graphene-based Sensor (3D rGO–Ti₃C₂–MWCNTs) [20] | 235.2 (1-60 μM) & 103.8 (0.06-9.77 mM) | 0.001 - 9.77 | 0.3 | -0.25 V | Adsorption & Charge Transfer |
| Prussian Blue/Graphene (PB-MWCNTs with Ionic Liquid) [7] | 0.436 | 0.005 - 1.645 | 0.35 | ~0.0 V (Low potential) | Electrostatic Gating & Charge Transfer |
Analysis of Comparative Data
- Sensitivity: The CNT-based composite demonstrates superior sensitivity, attributed to the synergistic effect between the carbon nanocomposite and platinum nanoparticles, which enhances the electrocatalytic reduction of H₂O₂ [19].
- Detection Limit: Both CNT and graphene platforms achieve sub-micromolar detection limits, which is crucial for tracking the subtle, dynamic changes in H₂O₂ concentration found in plant signaling pathways [19] [20].
- Operational Potential: Sensors operating at low or near-zero potentials, like the Prussian Blue hybrid, are highly advantageous for complex biological samples. This minimizes interference from other electroactive species commonly found in plant tissue extracts, such as ascorbic acid or dopamine [7].
Experimental Protocols for Key Setups
To ensure reproducibility, this section outlines detailed methodologies for fabricating and operating the two primary types of sensors discussed.
This protocol details the creation of a 3D nanocomposite sensor for enzyme-free H₂O₂ detection.
1. Electrode Modification:
- Surface Preparation: Begin by polishing a Glassy Carbon Electrode (GCE) with alumina slurry (e.g., 0.05 μm). Clean sequentially with distilled water, acetone, and water in an ultrasonic bath, then dry under a gentle air or nitrogen stream.
- Nanocomposite Preparation: Disperse a mixture of Carbon Nanotubes (CNTs) and Graphene Oxide (GO) in a suitable solvent via prolonged ultrasonication. Add a precursor salt for platinum nanoparticles (e.g., H₂PtCl₆).
- Modification and Reduction: Drop-cast the suspension onto the cleaned GCE surface. The reduction of Graphene Oxide (GO) to RGO and the formation of Platinum Nanoparticles (Pt NPs) can be achieved in-situ via electrochemical or chemical methods (e.g., cyclic voltammetry in a suitable potential window or using a reducing agent like NaBH₄).
2. Material Characterization:
- Use Scanning Electron Microscopy (SEM) to confirm the formation of a 3D porous structure.
- Perform Energy-Dispersive X-ray Spectroscopy (EDS) to verify the uniform distribution of Carbon (C), Oxygen (O), and Platinum (Pt) within the composite.
3. Electrochemical Measurement:
- Technique: Use Chronoamperometry (i-t) at a constant working potential of -0.2 V vs. Ag/AgCl in a stirred solution.
- Calibration: Record the steady-state current response upon successive additions of standard H₂O₂ solution into the electrochemical cell. Plot the current vs. concentration to establish a calibration curve.
This protocol describes a one-pot hydrothermal synthesis for a 3D hydrogel-based sensor, suitable for real-time detection.
1. Hydrogel Electrode Fabrication:
- Suspension Preparation: Create a homogeneous mixture of Graphene Oxide (GO, 0.4 mg mL⁻¹), MXene (Ti₃C₂, 0.4 mg mL⁻¹), and Multi-Walled Carbon Nanotubes (MWCNTs, 0.4 mg mL⁻¹) in deionized water via ultrasonication for 2 hours.
- Hydrothermal Reaction: Transfer the suspension into a Teflon-lined autoclave. Insert a copper wire (diameter 0.2 mm) to act as a substrate. React at 180°C for 4 hours to form a 3D reduced Graphene Oxide (rGO) hydrogel film modified on the copper wire.
- Sensor Assembly: Coat the side of the electrode cylinder with insulating wax to create a defined disk electrode surface.
2. Structural and Chemical Characterization:
- SEM is used to visualize the 3D porous network.
- X-ray Photoelectron Spectroscopy (XPS) confirms the successful reduction of GO and identifies elemental compositions.
3. Electrochemical Detection:
- System Setup: Use a standard three-electrode system with the modified electrode as the working electrode, a Pt wire as the counter electrode, and an Ag/AgCl reference electrode.
- Measurement: Apply Amperometry at -0.25 V while stirring. Add H₂O₂ standards and plot the calibration curve from the resulting current responses.
The Scientist's Toolkit: Essential Research Reagents and Materials
The table below lists key materials and their functions for researchers developing these nanomaterial-based sensors.
Table 2: Essential Research Reagents and Materials for H₂O₂ Sensor Development
| Material / Reagent | Function in Sensor Development | Example Application |
|---|---|---|
| Carbon Nanotubes (CNTs) | High-conductivity transducer; forms 3D scaffold for catalyst support [19] [17]. | Creates conductive network in RGO/CNTs-Pt composite [19]. |
| Graphene Oxide (GO) / Reduced GO (rGO) | 2D backbone with high surface area; can be formed into 3D hydrogels [20] [7]. | Primary matrix in 3D rGO–Ti₃C₂–MWCNTs hydrogel [20]. |
| Platinum Nanoparticles (Pt NPs) | Provides electrocatalytic sites for H₂O₂ reduction, enhancing sensitivity [19] [7]. | Catalyst in RGO/CNTs-Pt/GCE for H₂O₂ reduction [19]. |
| MXene (Ti₃C₂Tx) | 2D conductive additive; surface functional groups boost redox activity [20]. | Enhances conductivity and surface area in 3D composite [20]. |
| Prussian Blue (PB) | "Artificial peroxidase"; catalyzes H₂O₂ reduction at very low potentials [7]. | Active material for selective H₂O₂ detection at ~0.0 V [7]. |
| Ionic Liquids (IL) | High-conductivity electrolyte modifier; improves stability and electron transfer [7]. | Dopant in PB-MWCNTs composite to enhance performance [7]. |
| Chitosan | Biocompatible polymer for enzyme immobilization; forms stable films [21]. | Membrane matrix in HRP-based biosensor construction [21]. |
The choice between CNT and graphene-based platforms for plant H₂O₂ sensing depends on the specific research requirements. CNT-based composites, particularly when hybridized with metallic nanoparticles, currently offer superior sensitivity and are ideal for detecting very low concentrations. In contrast, graphene-based platforms, especially 3D hydrogels and FET designs, provide excellent versatility, a wide dynamic range, and the ability to exploit electrostatic gating mechanisms.
Future research will likely focus on the intelligent integration of these nanomaterials to create synergistic composites that maximize sensitivity, selectivity, and stability. Furthermore, the application of these sensors in plant research will deepen our understanding of H₂O₂'s role in calcium-mediated signaling [15], providing unprecedented real-time insights into plant stress responses and adaptation mechanisms.
The selection of appropriate carbon nanomaterials is a critical determinant of success in the development of advanced biosensors for plant hydrogen peroxide (H₂O₂) research. Carbon nanotubes (CNTs) and graphene represent two of the most prominent carbon allotropes with exceptional properties that make them well-suited for electrochemical sensing applications. This comparison guide provides an objective analysis of their fundamental material properties—conductivity, surface area, and functionalization capabilities—within the specific context of plant H₂O₂ sensing. Understanding their comparative advantages and limitations enables researchers to make informed material selections for specific experimental requirements, ultimately enhancing sensor sensitivity, selectivity, and reliability in complex plant physiological studies.
Fundamental Property Comparison
The table below summarizes the key quantitative properties of carbon nanotubes and graphene relevant to H₂O₂ sensing applications.
Table 1: Comparative Properties of Carbon Nanotubes and Graphene
| Property | Carbon Nanotubes (CNTs) | Graphene |
|---|---|---|
| Electrical Conductivity | ~10⁶ S/m for SWCNTs; excellent electron transfer in 1D structure [22] | Up to 10⁸ S/m; superior 2D electron transport [23] |
| Specific Surface Area | 100-1000 m²/g (high but limited by bundling) [24] | Theoretical: 2630 m²/g; Practical (3D porous): up to 1500 m²/g [23] |
| Common Functionalization Methods | Covalent (sidewall oxidation), Non-covalent (π-π stacking) [25] | Covalent (GO/rGO chemistry), Non-covalent (surfactants) [25] [23] |
| Enzyme Immobilization Efficiency | High surface curvature can affect enzyme conformation/activity [24] | Large 2D surface promotes stable enzyme binding; better retention of activity [26] [27] |
| Material Integration in Composites | Tendency to aggregate; difficult to disperse evenly [26] | Superior dispersion and integration with polymers [26] |
| Typical H₂O₂ Sensing Mechanism | Direct electron transfer from enzyme (e.g., Catalase) to CNT [24] [22] | Enhanced electron transport across 2D plane; catalyst support [28] |
Experimental Approaches for H₂O₂ Sensing
Enzyme Immobilization Protocols
The immobilization of enzymes like catalase (CAT) or glucose oxidase (GOx) is fundamental to creating effective H₂O₂ biosensors. The following protocols detail established methodologies for both CNT and graphene substrates.
Protocol A: Adsorptive Immobilization on Carbon Nanotubes
This method, adapted from studies on catalase adsorption, relies on physical adsorption and has been shown to significantly affect enzyme secondary structure and function [24].
- CNT Pretreatment: Suspend 10 mg of multi-walled or single-walled CNTs in 20 mL of a 3:1 (v/v) mixture of H₂SO₄ and HNO₃. Sonicate for 30 minutes at 40°C to oxidize and introduce carboxylic acid groups, then wash thoroughly with deionized water until neutral pH [24].
- Enzyme Incubation: Prepare a 0.1 mg/mL solution of catalase (or other peroxidase) in phosphate buffer saline (PBS, 0.1 M, pH 7.2). Mix the enzyme solution with the pretreated CNT suspension at a 1:1 volume ratio.
- Adsorption Process: Incubate the mixture with gentle shaking for 12 hours at 4°C to allow enzyme adsorption. Key factors influencing the outcome are the surface O-functionalities of the CNTs and the resulting hydrophobic or π-π stacking interactions, which can induce conformational changes in the enzyme [24].
- Washing and Storage: Centrifuge at 12,000 rpm for 15 minutes to separate the CNT-enzyme complex. Wash the pellet twice with PBS to remove unbound enzyme. The final biocomposite can be re-suspended in buffer for immediate use or lyophilized for storage.
Protocol B: Covalent Binding on 3D Porous Graphene Oxide
This protocol leverages the high surface area and functional groups of 3D graphene structures, which can overcome enzyme leaching problems and enhance direct electron transfer [22].
- Synthesis of 3D Porous Graphene: Follow a modified breath-figure method or use a template-free in-situ reduction of graphene oxide (GO) to create a macroporous 3D structure with pore sizes typically ranging from 1 to 4 μm [23].
- Surface Activation: Treat the 3D graphene structure with a glutaraldehyde solution (2.5% v/v) for 2 hours. Glutaraldehyde acts as a crosslinker, its aldehyde groups reacting with the oxygen functionalities on graphene and the amino groups on the enzyme [22].
- Enzyme Conjugation: Immerse the activated 3D graphene scaffold in a solution of glucose oxidase (GOx, 10 mg/mL in PBS) for 24 hours at 4°C, ensuring full infiltration of the porous structure.
- Stabilization and Washing: Remove the scaffold and wash extensively with PBS to remove any physically adsorbed enzyme. The resulting bioelectrode demonstrates high enzyme loading and stability, maintaining functionality for multiple use cycles [22].
Plant Growth and Nanomaterial Exposure Studies
Investigating the role of H₂O₂ in plant signaling requires models where its production is modulated. Studies exposing plants to carbon nanomaterials provide such models, as summarized below.
Table 2: Experimental Models of Plant Response to Carbon Nanomaterials
| Plant Species | Nanomaterial & Concentration | Experimental Setup | Key Findings Relevant to H₂O₂ |
|---|---|---|---|
| Catharanthus roseus (Periwinkle) | MWCNTs; 50, 100, 150 mg/L [29] | In vitro culture on hormone-free MS medium for 60 days [29] | ~2x increase in catalase (CAT) and peroxidase (POD) activities; 1.7-fold increase in alkaloids [29] |
| Vigna angularis (Adzuki bean) | Graphene; 0.01 to 100 mg/L [30] | Soil culture, watered with graphene weekly for 45 days [30] | 199.3% increase in root CAT activity at 1 mg/L; ~60% increase in leaf H₂O₂; up-regulated photosynthesis genes [30] |
| Artemisia annua (Sweet wormwood) | Graphene; 10, 20 mg/L [28] | Cultivation with graphene in growth medium [28] | ~60% increase in H₂O₂; 9-fold increase in CAT activity in vitro; inhibited miR828 biogenesis [28] |
Signaling Pathways and Experimental Workflows
The molecular response of plants to carbon nanomaterials involves specific signaling pathways that modulate hydrogen peroxide levels and antioxidant defenses. The following diagram illustrates the key regulatory mechanism triggered by graphene exposure in Artemisia annua.
The experimental workflow for evaluating the effects of carbon nanomaterials on plant systems and for developing H₂O₂ sensors is structured as follows.
The Scientist's Toolkit: Essential Research Reagents
The table below lists key reagents, materials, and instruments essential for conducting research on carbon nanotubes and graphene for H₂O₂ sensing in plant biology.
Table 3: Essential Reagents and Materials for H₂O₂ Sensing Research
| Item | Function/Application | Examples from Experimental Protocols |
|---|---|---|
| Single/Multi-walled CNTs | Core sensing material; electron conduit [24] [22] | Oxidized MWCNTs for catalase adsorption [24] |
| Graphene Oxide (GO) & Reduced GO (rGO) | Versatile 2D platform for composite fabrication [23] | Building block for 3D porous scaffolds [23] [22] |
| Catalase (CAT) & Glucose Oxidase (GOx) | Model enzymes for H₂O₂ recognition and sensing [24] [22] | Catalase for activity studies on CNTs [24]; GOx for biofuel cell anodes [22] |
| Glutaraldehyde | Common crosslinker for covalent enzyme immobilization [22] | Used to functionalize 3D graphene for GOx binding [22] |
| Phosphate Buffer Saline (PBS) | Standard physiological buffer for bio-conjugation [24] [22] | Medium for enzyme incubation and washing steps [24] |
| Murashige and Skoog (MS) Medium | Standard plant tissue culture medium [29] | Base medium for in vitro plant nanomaterial exposure [29] |
| FTIR & Raman Spectroscopes | Characterize functional groups and material quality [22] | Confirm CNT oxidation, graphene reduction [22] |
| Electrochemical Workstation | Validate sensor performance (DET, sensitivity) [22] | Test bioanode performance in enzymatic biofuel cells [22] |
From Lab to Leaf: Sensor Architectures and Real-World Applications in Plant Science
Hydrogen peroxide (H₂O₂) is a crucial signaling molecule in plant metabolic processes, acting as a key mediator in oxidative stress pathways and cellular signaling networks. Its detection is vital for understanding plant stress responses, defense mechanisms, and developmental regulation. Carbon nanotube (CNT)-based sensors have emerged as powerful tools for monitoring H₂O₂ due to their exceptional electrical properties, high surface-to-volume ratio, and versatile functionalization capabilities. These sensors offer significant advantages for plant research, enabling real-time, non-invasive monitoring of H₂O₂ fluctuations in various plant tissues and cellular compartments. This guide provides a comprehensive comparison of CNT-based electrochemical biosensors and optical nanosensors, framing their development within the broader context of carbon nanomaterial applications alongside graphene-based alternatives for plant science research.
Performance Comparison: CNT vs. Graphene-Based H₂O₂ Sensors
The following tables summarize the performance characteristics of various CNT and graphene-based sensors for H₂O₂ detection, highlighting their respective advantages in plant research applications.
Table 1: Performance Metrics of CNT-Based H₂O₂ Sensors
| Sensor Design | Detection Mechanism | Linear Range | Sensitivity | Limit of Detection (LOD) | Key Advantages |
|---|---|---|---|---|---|
| Hemin-PEI/MWCNT on SPGE [4] | Electrocatalytic reduction | Not specified | 18.09 ± 0.89 A M⁻¹ cm⁻² | Not specified | Low onset potential (+0.2 V), avoids interference, non-enzymatic |
| 3DGH/NiO25 Nanocomposite [31] | Electrocatalytic oxidation | 10 µM – 33.58 mM | 117.26 µA mM⁻¹ cm⁻² | 5.3 µM | Wide linear range, good selectivity, non-enzymatic |
| PB-MWCNT with Ionic Liquid [7] | Electrocatalytic reduction | 5 – 1645 µM | 0.436 μA·mM⁻¹·cm⁻² | 0.35 μM | Low working potential, high selectivity |
| CNT Field-Effect Transistor (FET) [17] | Charge transfer modulation | Varies with functionalization | High (semiconducting SWCNTs) | Can reach nM-pM | Label-free, real-time, miniaturizable |
Table 2: Comparison with Graphene-Based H₂O₂ Sensors
| Sensor Design | Detection Mechanism | Linear Range | Sensitivity | Limit of Detection (LOD) | Key Advantages |
|---|---|---|---|---|---|
| Ag-SiO₂-Ag Graphene Biosensor [32] | Plasmonic / Optical | Not specified | 1785 nm/RIU | Not specified | High optical sensitivity, machine learning optimization |
| NiO/3D Graphene Hydrogel [31] | Electrocatalytic oxidation | 10 µM – 33.58 mM | 117.26 µA mM⁻¹ cm⁻² | 5.3 µM | Prevents graphene restacking, large surface area |
| Laser-Induced Graphene (LIG) [33] | Electrochemical / Resistive | Varies with design | High (e.g., GF >20) | Can be low (e.g., 0.05% strain) | Facile, direct-write fabrication, flexible substrates |
Table 3: Suitability for Plant H₂O₂ Research Applications
| Application Scenario | Recommended Sensor Type | Rationale | Key Considerations |
|---|---|---|---|
| In vivo apoplastic H₂O₂ flux | Hemin-PEI/MWCNT Electrochemical [4] | Low operating potential minimizes interference from phenolics & ascorbate. | Microelectrode design, spatial resolution. |
| Leaf surface & stomatal monitoring | Flexible LIG/MXene Sensor [33] | Conforms to irregular plant surfaces; detects micro-strains from H₂O₂-induced movements. | Integration with plant epidermis, humidity effects. |
| Cellular & sub-cellular sensing | Functionalized SWCNT-FET [17] | Nanoscale size; label-free, real-time detection in micro-environments. | Biocompatibility, delivery into cells. |
| High-sensitivity lab-based analysis | 3D Graphene Hydrogel/NiO [31] | Wide linear range encompasses typical plant [H₂O₂]; high sensitivity. | Requires extracted sap or tissue homogenate. |
Experimental Protocols for Key Sensor Designs
Objective: To fabricate a pseudo-peroxidase non-enzymatic sensor for sensitive H₂O₂ monitoring in biological matrices like plant apoplastic fluid.
Synthesis and Modification Workflow:
- MWCNT Dispersion: Disperse functionalized multi-walled carbon nanotubes (MWCNTs) in a suitable solvent (e.g., DMF) using prolonged ultrasonication to create a homogeneous suspension.
- Hemin-PEI Complex Preparation: Prepare an aqueous solution of polyethyleneimine (PEI). Add hemin to the PEI solution to form a stable hemin-PEI complex via electrostatic interactions and coordination, preventing hemin dimerization.
- Electrode Modification: Drop-cast the MWCNT suspension onto the working electrode of a screen-printed graphene electrode (SPGE) and allow to dry. Subsequently, drop-cast the hemin-PEI complex onto the MWCNT/SPGE surface.
- Sensor Characterization: Use scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDS) to confirm the morphology and elemental composition of the modified electrode. Employ cyclic voltammetry (CV) in a buffer solution with and without H₂O₂ to validate the electrocatalytic activity.
Figure 1: Hemin-PEI/MWCNT Sensor Fabrication Workflow
Objective: To develop a highly sensitive and stable non-enzymatic H₂O₂ sensor with a wide linear range for applications in complex media.
Synthesis Workflow:
- NiO Octahedron Synthesis: Use mesoporous silica SBA-15 as a hard template. Impregnate the template with a solution of nickel nitrate hexahydrate in ethanol. After drying, calcinate the material at 550°C for 3 hours. Finally, remove the silica template by washing with 2 M NaOH.
- Graphene Oxide (GO) Preparation: Synthesize graphene oxide from graphite powder using a modified Hummers' method.
- 3DGH/NiO Self-Assembly: Mix the as-prepared NiO octahedrons with the GO dispersion. Subject the mixture to a hydrothermal treatment (e.g., at 180°C for 12 hours) in a Teflon-lined autoclave. This process simultaneously reduces GO and assembles it into a 3D hydrogel structure embedded with NiO octahedrons.
- Electrode Preparation and Testing: Apply the 3DGH/NiO nanocomposite onto a glassy carbon electrode (GCE). Use chronoamperometry and cyclic voltammetry in PBS (pH 7.4) with successive additions of H₂O₂ to evaluate sensitivity, linear range, and detection limit.
Sensing Mechanisms and Signaling Pathways
Electrochemical Sensing Mechanism in CNT-Based Sensors
The exceptional electrocatalytic properties of CNT-based sensors stem from their unique electronic structure and functionalization.
Figure 2: CNT Electrochemical Sensing Mechanism
- Charge Transfer Modulation: When H₂O₂ molecules adsorb onto the CNT surface, particularly at defect sites or functional groups, they act as electron donors or acceptors. This charge transfer shifts the Fermi level of the CNT, thereby modulating its electrical conductivity, which is measured as a change in current or resistance in resistivitive or FET-based sensors [17].
- Electrocatalytic Reduction/Oxidation: For sensors functionalized with catalytic materials like hemin or metal oxides (NiO), the H₂O₂ undergoes a direct electrocatalytic reaction at the electrode interface. For instance, hemin, acting as a peroxidase mimic, catalyzes the reduction of H₂O₂. The CNT backbone serves as a highly conductive pathway, facilitating rapid electron transfer from the electrode surface to the catalytic sites and then to H₂O₂, significantly amplifying the electrochemical response [4] [31] [7].
CNT vs. Graphene: A Comparative Sensing Pathway
The fundamental structural differences between CNTs and graphene dictate their respective sensing pathways and performance.
Figure 3: Comparative Sensing Pathways: CNT vs. Graphene
- CNT Advantages: The 1D structure of CNTs, particularly single-walled carbon nanotubes (SWCNTs), promotes ballistic electron transport and offers a confined nanoscale environment that can enhance interaction with target molecules. This often results in faster electron transfer kinetics and higher sensitivity in electrochemical detection compared to 2D graphene platforms [17].
- Graphene Advantages: Graphene's 2D planar structure provides an ultra-large surface area for biomarker binding and functionalization. This makes it exceptionally suitable for optical sensing platforms like surface plasmon resonance (SPR), where its planar geometry efficiently interacts with light, leading to high sensitivity in wavelength shifts, as demonstrated by the Ag-SiO₂-Ag graphene biosensor [32].
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 4: Essential Materials for CNT-Based H₂O₂ Sensor Development
| Category | Item | Function in Sensor Development | Example Use Case |
|---|---|---|---|
| Carbon Nanomaterials | Multi-Walled Carbon Nanotubes (MWCNTs) | High-surface-area scaffold; enhances electron transfer and catalyst loading. | Hemin-PEI/MWCNT/SPGE sensor [4] |
| Single-Walled Carbon Nanotubes (SWCNTs) | Semiconducting channel for FET sensors; high sensitivity to surface charges. | CNT-FET based chemical sensors [17] | |
| Catalysts & Functionalizers | Hemin | Porphyrin complex providing intrinsic peroxidase-mimicking activity. | Non-enzymatic, catalytic H₂O₂ reduction [4] |
| Polyethyleneimine (PEI) | Cationic polymer matrix to stabilize hemin and prevent aggregation. | Hemin-PEI complex formation [4] | |
| Nickel Oxide (NiO) Nanostructures | Transition metal oxide with excellent electrocatalytic activity for H₂O₂ oxidation. | 3DGH/NiO octahedron sensor [31] | |
| Prussian Blue (PB) | "Artificial peroxidase" for electrocatalytic H₂O₂ reduction at low potentials. | PB-MWCNT composite sensor [7] | |
| Substrates & Electrodes | Screen-Printed Graphene Electrodes (SPGE) | Disposable, reproducible, and mass-producible transducer platform. | Point-of-care compatible sensor design [4] |
| Glassy Carbon Electrode (GCE) | Polished, stable baseline electrode for drop-casting nanomaterials. | Testing 3DGH/NiO composite [31] | |
| Key Reagents | Phosphate Buffered Saline (PBS) | Standard physiological pH electrolyte for electrochemical testing. | Most reported electrochemical detections [31] [7] |
| Nafton | Perfluorosulfonated ionomer; used as a binder and protective membrane. | Stabilizing casted films on electrode surfaces. |
The evolution of biosensing technologies increasingly relies on advanced nanomaterials, with carbon nanotubes (CNTs) and graphene standing at the forefront of research. This guide objectively compares the performance of emerging graphene-based platforms, specifically Laser-Induced Graphene (LIG) biochips and hybrid electrodes, against traditional CNT-based alternatives. The comparison is framed within a broader thesis on their application for hydrogen peroxide (H2O2) sensing in plants, a critical research area due to H2O2's role as a key signaling molecule in plant physiology and stress responses. For researchers and scientists engaged in drug development and agricultural biotechnology, understanding the performance characteristics, fabrication complexities, and practical limitations of these platforms is essential for selecting appropriate technologies for precision agriculture, environmental monitoring, and pharmaceutical applications.
Table 1: Core Characteristics of Carbon Nanomaterial Platforms
| Platform Characteristic | LIG Biochips | Graphene Hybrid Electrodes | CNT-Based Platforms |
|---|---|---|---|
| Primary Fabrication Method | Direct laser writing on polymers/biomass [34] [35] | Chemical synthesis & composite formation [36] | Chemical vapor deposition or solution processing [37] |
| Electrical Conductivity | High (tunable via laser parameters) [34] | Enhanced via material hybridization [36] | High (dependent on tube alignment/chirality) [37] |
| Active Surface Area | Hierarchical porous structure [38] | High (rGO provides extensive surface) [36] | High (tubular nanostructure) [37] |
| Mechanical Flexibility | Excellent (suitable for wearables) [34] [39] | Moderate (depends on substrate/binder) [36] | Good (flexible but prone to deformation) [37] |
| Biocompatibility | Good (functionalization enhances) [34] | Variable (depends on composite materials) [36] | Good (but concerns about nanotube persistence) [37] |
Platform Architectures and Fabrication Methods
Laser-Induced Graphene (LIG) Biochips
LIG fabrication represents a mask-free, single-step approach for creating graphene electrodes directly on flexible substrates. The process utilizes focused laser beams (typically CO2 lasers) to induce localized pyrolysis of carbon-rich precursors, converting sp3-hybridized carbon into sp2-hybridized graphene through rapid heating at extreme temperatures (2500-3000K) [34] [35]. The mechanism involves three sequential stages: carbonization (250-500°C releasing heteroatoms as gaseous byproducts), graphitization (∼3000K reorganizing amorphous carbon into graphitic structures), and exfoliation (separating graphene layers through thermal expansion) [34]. Precise control over laser parameters (power, speed, wavelength) enables tailoring of microstructure, crystallinity, and surface chemistry [34]. Sustainable precursors include polyimide, wood, paper, and food-based materials, making LIG particularly suitable for disposable, low-cost sensors for plant monitoring [34] [39] [35].
Graphene-Based Hybrid Electrodes
Hybrid electrodes combine graphene with other nanomaterials to create synergistic effects that enhance electrochemical performance. Common architectures include graphene-pseudocapacitive material composites (e.g., α-MnO2/rGO) and graphene-battery type material systems (e.g., po-nSi/rGO) [36]. These configurations leverage graphene's high conductivity and surface area while incorporating additional charge storage mechanisms from pseudocapacitive or battery-type components. Fabrication typically involves multi-step chemical processes including reduction of graphene oxide, nanoparticle synthesis, and composite formation [36]. The resulting electrodes benefit from combined electric double-layer capacitance (from graphene) and faradaic redox reactions (from pseudocapacitive materials), enabling higher specific capacitances exceeding 600 F/g compared to single-component systems [36].
Carbon Nanotube (CNT) Platforms
CNT-based electrochemical platforms typically utilize multi-walled carbon nanotubes (MWCNTs) in paste or composite electrodes. The MWCNT paste electrode (PMWCNT) preparation involves chemical activation through successive nitric and sulfuric acid treatment to introduce surface functional groups, followed by mixing with mineral oil (70/30 w/w ratio) to form a paste [37]. This paste is then packed onto glassy carbon electrodes or other substrates. The tubular nanostructure of CNTs provides high surface area and favorable electron transfer kinetics, while their inherent functionalization potential enables effective biomolecule immobilization for biosensing applications [37].
Figure 1: LIG biochip fabrication involves sequential carbonization, graphitization, and functionalization of precursor materials under laser irradiation.
Performance Comparison for H2O2 Sensing
Electrochemical detection of hydrogen peroxide serves as a critical benchmark for evaluating biosensor performance, particularly in plant research where H2O2 functions as a key signaling molecule in stress responses and physiological processes.
Table 2: H2O2 Sensing Performance Metrics Comparison
| Performance Metric | LIG Biochips | Graphene Hybrid Electrodes | CNT-Based Platforms | Experimental Conditions |
|---|---|---|---|---|
| Detection Limit | ~0.1 µM (metallized) [40] | Sub-µM range [36] | 0.43 µM (PMWCNT/ChOx) [37] | Phosphate buffer (pH 7.4) |
| Linear Range | 0.5-100 µM [39] | Not specified | 0.4-4.0 mM [37] | Various buffer systems |
| Sensitivity | Tunable via metallization [40] | 26.15 µA/mM (composite) [37] | 21x enhancement with ChOx [37] | Amperometric detection |
| Response Time | Fast (seconds) [34] | Fast (seconds) [36] | Fast (seconds) [37] | Room temperature |
| Stability | Good (flexible substrates) [39] | ≥85-90% retention [36] | Good (enzyme-dependent) [37] | Multiple cycles |
Sensing Mechanisms and Selectivity
The electrochemical detection of H2O2 occurs primarily through reduction or oxidation reactions at the electrode surface. LIG platforms benefit from porous structures that facilitate mass transport and electron transfer, with performance进一步增强 through heteroatom doping (N, S, P, B) or metallization (e.g., Pt electrodeposition) [40] [38]. CNT-based biosensors achieve high selectivity through enzyme immobilization (e.g., cholesterol oxidase), where the enzymatic reaction generates H2O2 as a measurable product [37]. In silico studies confirm spontaneous binding between cholesterol oxidase and H2O2, validating the molecular recognition mechanism in CNT-based biosensors [37]. For plant H2O2 sensing, selectivity against interfering compounds (phenols, other reactive oxygen species) remains challenging for non-enzymatic approaches, though appropriate electrode modification and potential tuning can mitigate these effects.
Experimental Protocols and Methodologies
LIG Biochip Fabrication for Plant Sensing
Materials: Polyimide film (0.005″ thick) or paper substrates; CO2 laser system; Ethanol (70%); Conductive polyester metal tape; Potentiostat with smartphone connectivity [40] [39].
Procedure:
- LIG Patterning: Design electrode geometry in CAD software. Irradiate polyimide/paper substrate using CO2 laser (40% power, 75% speed, 1000 PPI, 5.8 cm lens distance) under ambient conditions [40].
- Post-processing: Rinse LIG electrodes with 70% ethanol (3×) followed by DI water. Air dry or use nitrogen stream [40].
- Metallization (Optional): Perform Pt electrodeposition via galvanostatic or frequency-modulated methods in chloroplatinic acid solution to enhance electrochemical performance [40].
- Device Assembly: Passivate connection areas with acrylic lacquer. Attach bonding pads using conductive tape. Integrate with portable potentiostat and smartphone for field deployment [39].
- H2O2 Detection: Apply square wave voltammetry from -0.80 V to 0.20 V in plant extracts using portable potentiostat. Correlate current response with H2O2 concentration [39].
CNT-Based Biosensor for H2O2 Detection
Materials: MWCNTs (6-13 nm diameter, >98% purity); Mineral oil; Cholesterol oxidase (ChOx) lyophilized powder; Sodium phosphate buffer (0.050 M, pH 7.4); Nitric acid (1 M); Sulfuric acid (1 M) [37].
Procedure:
- MWCNT Activation: Sonicate MWCNTs in 1 M nitric acid (30 min), filter, then sonicate in 1 M sulfuric acid (30 min). Repeat twice. Wash extensively with ethanol and acetone until neutral pH [37].
- Paste Preparation: Mix activated MWCNTs with mineral oil (70:30 w/w ratio) to form homogeneous paste [37].
- Electrode Assembly: Pack PMWCNT onto polished glassy carbon electrode (5 mm diameter). For biosensor, drop-cast 10 μL ChOx solution (20 U/mL) onto PMWCNT surface. Dry for 10 minutes at room temperature [37].
- Electrochemical Characterization: Perform cyclic voltammetry from -0.80 V to 0.20 V at 0.10 V/s in phosphate buffer. Use electrochemical impedance spectroscopy to verify enzyme immobilization [37].
- H2O2 Quantification: Conduct amperometry at constant potential in plant samples spiked with H2O2. Measure reduction current and correlate with concentration using calibration curve [37].
Figure 2: H2O2 sensing occurs through direct electron transfer at electrode surfaces or via enzymatic recognition, generating measurable current proportional to concentration.
Critical Analysis: Research Challenges and Opportunities
Reproducibility and Scalability Considerations
A significant challenge in LIG technology is batch-to-batch variation, with studies reporting approximately 5% variability for bare LIG electrodes but up to 30% variation after metallization processes [40]. This variability stems from substrate deformation during graphitization, focal plane inconsistencies, and non-uniform metallization. For CNT-based platforms, reproducibility challenges include maintaining dispersion stability and consistent enzyme immobilization. Hybrid electrodes face composite homogeneity issues that can affect performance reliability [36] [40] [37]. Machine learning approaches are being developed to predict LIG formation and optimize processing parameters, potentially reducing variability in future applications [35].
Application-Specific Performance Trade-offs
For plant H2O2 sensing requiring field deployment, LIG biochars on paper substrates offer advantages including mechanical flexibility, direct patterning capability, and compatibility with portable electronics [39]. However, CNT-based platforms currently provide superior selectivity through enzymatic recognition, crucial for complex plant matrices [37]. Hybrid electrodes demonstrate enhanced specific capacitance and energy density but involve more complex fabrication processes [36]. Researchers must balance sensitivity requirements with scalability needs when selecting appropriate platforms for specific applications.
Table 3: Research Reagent Solutions for Platform Development
| Reagent/Material | Function | Example Application | Key Considerations |
|---|---|---|---|
| Polyimide Film | LIG substrate | Flexible biochips [40] | Thickness (0.005″), thermal stability |
| Cholesterol Oxidase | Biological recognition element | H2O2 biosensing [37] | Thermal stability, activity retention |
| Chloroplatinic Acid | Metallization precursor | Electrode performance enhancement [40] | Deposition method affects uniformity |
| Multi-Walled CNTs | Conductivity enhancement | Paste electrodes [37] | Activation pre-treatment required |
| Morpholinoethanesulfonic Acid (MES) | Buffer component | Electrochemical characterization [40] | pH stability, compatibility |
| Nail Polish | Hydrophobic coating | Paper-based LIG protection [39] | Creates synergistic porous structure |
LIG biochips excel in flexibility, rapid fabrication, and field deployment potential for plant H2O2 sensing, while CNT-based platforms currently offer superior selectivity through enzymatic recognition. Hybrid electrodes provide enhanced electrochemical performance but at the cost of fabrication complexity. The choice between platforms depends on specific research priorities: LIG for disposable, on-site monitoring; CNT for precise, laboratory-based measurements; and hybrid systems for applications demanding highest sensitivity. Future research directions include addressing reproducibility challenges through machine learning-guided fabrication [35], developing sustainable substrates from wood and biomass [34] [35], and integrating these platforms with artificial intelligence for intelligent agricultural monitoring systems. As these technologies mature, graphene-based platforms are poised to significantly advance plant science research through enhanced spatiotemporal monitoring of H2O2 signaling in response to environmental stresses.
The accurate, real-time monitoring of hydrogen peroxide (H₂O₂) in plants is crucial for understanding early stress signaling and developing climate-resilient crops. This guide compares the performance of sensing platforms based on two leading carbon nanomaterials—carbon nanotubes (CNTs) and graphene—for in-planta H₂O₂ deployment. While CNTs currently dominate in fundamental research and multiplexed sensing within living plants, graphene-based systems show significant promise in cost-effective, scalable electrochemical sensors. The choice between them hinges on the specific application requirements: CNTs for high-fidelity, real-time dynamic sensing in living tissue, and graphene for robust, disposable point-of-care form factors.
Performance Comparison: Carbon Nanotubes vs. Graphene for H₂O₂ Sensing
The following table summarizes key performance metrics and characteristics of CNT and graphene-based platforms, highlighting their suitability for different deployment scenarios.
Table 1: Performance Comparison of Carbon Nanomaterials in Plant H₂O₂ Sensing
| Feature | Carbon Nanotubes (CNTs) | Graphene |
|---|---|---|
| Primary Sensing Modality | Optical (nIR fluorescence) [41] | Electrochemical (amperometry) [4] |
| Typical Limit of Detection (LOD) | Nanomolar (nM) range in living plants [41] | Low micromolar (μM) to nanomolar (nM) range in vitro [4] |
| Key Advantage | Capability for non-destructive, multiplexed sensing in vivo; minimal interference from plant autofluorescence [41] | High sensitivity and compatibility with low-cost, scalable screen-printed electrodes (SPEs) [4] |
| In-Planta Deployment Format | Infused into leaf mesophyll for continuous monitoring; integrated into microneedle patches [42] [41] | Primarily used in electrode modifications; potential for integration into wearable patches [4] [43] |
| Multiplexing Capability | High (e.g., simultaneous monitoring of H₂O₂ and salicylic acid) [41] | Moderate (typically requires multiple electrode arrays) |
| Scalability & Cost | Complex sensor synthesis; higher cost [44] | Highly scalable production; lower cost per device [4] [44] |
Experimental Protocols and Deployment Workflows
Protocol 1: CNT-Based Optical Nanosensors for Living Plants
This protocol details the method for creating and using CNT-based nanosensors to monitor H₂O₂ dynamics in living plants, as exemplified by state-of-the-art research [41].
- Sensor Synthesis: Single-walled carbon nanotubes (SWCNTs) are non-covalently functionalized with a specific single-stranded DNA oligomer, (GT)₁₅. This DNA corona forms around the SWCNT, creating a recognition site that selectively binds to H₂O₂, modulating the nanotube's near-infrared (nIR) fluorescence [41].
- Plant Incorporation: A suspension of the (GT)₁₅-SWCNT nanosensors is introduced into the leaf mesophyll (the inner tissue of the leaf) of a living plant, such as Brassica rapa (pak choi), via infiltration using a needleless syringe [41].
- Stimulation and Imaging: The plant is subjected to a controlled stressor (e.g., light, heat, pathogen, mechanical wounding). The nIR fluorescence of the incorporated nanosensors is monitored in real-time using a custom-built nIR imaging system. A decrease in fluorescence intensity correlates with an increase in local H₂O₂ concentration [41].
- Data Analysis: The temporal waveforms of H₂O₂ generation are analyzed. Research shows that different stress types produce distinct, characteristic H₂O₂ signatures within hours of treatment, enabling early stress identification [41].
Protocol 2: Graphene-Based Electrochemical Sensor with Microneedle Fluid Extraction
This protocol combines a graphene-based electrochemical sensor with a minimally invasive microneedle array for fluid extraction, representing an integrated approach to plant health monitoring [43].
- Sensor Fabrication: A screen-printed graphene electrode (SPGE) serves as the transducer. To enhance its performance, it is modified with a nanocomposite of multi-walled carbon nanotubes (MWCNTs) and hemin (an iron-containing porphyrin) entrapped in polyethylenimine (PEI). This "artificial peroxidase" platform electrocatalytically reduces H₂O₂ at low operating potentials [4] [43].
- Microneedle Array Fabrication: A hollow microneedle array (HMA) is fabricated using low-cost stereolithography (SLA) 3D printing, achieving tip diameters of ~26 μm for minimal plant damage [43].
- Fluid Extraction and Sensing: The HMA is pierced into a plant leaf to extract apoplastic fluid (the fluid in spaces between cells). The extracted fluid is wicked onto a paper-based sampling pad that also functions as an electrochemical cell. The SPGE is then interfaced with this pad to perform amperometric detection of H₂O₂ [43].
- Measurement: The hemin-PEI/MWCNT/SPGE system detects H₂O₂ via electrocatalytic reduction at a low applied potential. The resulting current is proportional to the H₂O₂ concentration in the extracted fluid [4].
System Workflow and Signaling Pathways
The following diagram illustrates the logical workflow and signaling pathways involved in deploying these sensing systems to decode early plant stress signals.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful deployment of in-planta H₂O₂ sensing systems requires a specific set of materials and reagents. The table below details key components for both CNT and graphene-based approaches.
Table 2: Key Research Reagents and Materials for In-Planta H₂O₂ Sensing
| Item | Function/Description | Relevant Platform |
|---|---|---|
| Single-Walled Carbon Nanotubes (SWCNTs) | The core transducer; nIR fluorescence is modulated by binding to H₂O₂. [41] | CNT Optical Nanosensor |
| (GT)₁₅ DNA Oligomer | A specific single-stranded DNA sequence that wraps around SWCNTs to create a selective corona phase for H₂O₂ recognition. [41] | CNT Optical Nanosensor |
| Screen-Printed Graphene Electrode (SPGE) | A low-cost, disposable, and mass-producible electrochemical transducer. [4] | Graphene Electrochemical Sensor |
| Hemin-PEI/MWCNT Composite | An "artificial peroxidase" catalyst. Hemin mimics enzyme activity, PEI stabilizes it, and MWCNTs enhance electron transfer and surface area. [4] | Graphene Electrochemical Sensor |
| 3D-Printed Hollow Microneedle Array (HMA) | A minimally invasive platform for extracting apoplastic fluid from plant leaves for ex-situ analysis. [43] | Microneedle Deployment |
| Hydrogel Coating (Chitosan/Graphene Oxide) | A biocompatible coating for microneedles that enhances fluid uptake and can be functionalized with enzymes (e.g., HRP) for sensing. [42] | Microneedle Deployment |
The landscape of in-planta H₂O₂ sensing is defined by a trade-off between the high-fidelity, dynamic multiplexing offered by carbon nanotube-based optical platforms and the practical, scalable advantages of graphene-based electrochemical systems. For researchers aiming to decode the complex temporal dynamics of plant stress signaling in real-time, CNT nanosensors are the more advanced and capable tool. Conversely, for applications demanding cost-effective, field-deployable sensors for point-of-care stress detection, graphene-based electrodes, especially when integrated with microneedles, present a highly promising path forward. The ongoing development of both platforms will be instrumental in creating more resilient crops and precise agricultural interventions.
In plant systems, hydrogen peroxide (H₂O₂) functions as a crucial reactive oxygen species (ROS) and signaling molecule during stress responses. Under stress conditions such as pathogen infection, light intensity, heat, and mechanical damage, plants experience oxidative stress that leads to excessive H₂O₂ production [45] [46]. While chemically poorly reactive, H₂O₂ can become highly dangerous when it converts into reactive hydroxyl radicals upon exposure to UV radiation or when interacting with transition metal ions [45]. This dual nature as both signaling molecule and potential cellular damage agent makes accurate H₂O₂ monitoring essential for understanding plant stress physiology.
Electrochemical sensing has emerged as a superior methodology for H₂O₂ detection in plant systems due to its simplicity, rapid response, excellent sensitivity, and affordability compared to traditional methods like colorimetry, chemiluminescence, or spectrophotometry [45] [47] [46]. Classical enzymatic biosensors face limitations due to enzyme degradation over time, complex immobilization procedures, and instability under environmental fluctuations [47] [7]. This has driven the development of non-enzymatic sensors leveraging carbon nanomaterials, particularly carbon nanotubes (CNTs) and graphene, which offer exceptional electrocatalytic properties, large specific surface areas, and tunable electronic characteristics [45] [44] [3].
This analysis provides a comprehensive comparison of carbon nanotube and graphene-based electrochemical sensors for detecting H₂O₂ in plant stress applications, supported by experimental case studies and performance data.
Carbon Nanomaterial Properties for H₂O₂ Sensing
Fundamental Characteristics of Carbon Nanotubes and Graphene
Carbon nanotubes (CNTs) and graphene represent distinct structural forms of carbon nanomaterials with unique properties for electrochemical sensing applications. CNTs are one-dimensional (1D) cylindrical structures composed of rolled graphene sheets, classified as single-walled (SWCNTs) or multi-walled (MWCNTs) based on their layer number [3]. Their fibrous, high-aspect-ratio structure provides exceptional electrical conductivity along their length and large surface areas for catalyst immobilization [45] [46]. MWCNTs demonstrate significant structural diversity and stable physical and chemical properties, standing out among nanomaterials with enzyme-like electrocatalytic activity [46].
Graphene is a two-dimensional (2D) single layer of sp²-hybridized carbon atoms arranged in a hexagonal honeycomb lattice with a carbon-carbon distance of 0.142 nm [3]. This structure provides exceptional electrical conductivity, high intrinsic charge carrier mobility, optical transparency, substantial specific surface area, and remarkable mechanical flexibility [3]. Graphene-based nanomaterials are recognized for their superior biocompatibility compared with many other types of nanomaterials, enabling their application in self-organizing functional two-dimensional membranes and three-dimensional composites [3].
Table 1: Fundamental Properties of Carbon Nanomaterials for H₂O₂ Sensing
| Property | Carbon Nanotubes (CNTs) | Graphene |
|---|---|---|
| Dimensionality | 1D (fibrous, high aspect ratio) | 2D (planar sheet) |
| Electrical Conductivity | Excellent along tube axis | Exceptional in-plane |
| Surface Area | High (external and internal surfaces) | Very high (theoretical ~2630 m²/g) |
| Structural Characteristics | Tubular morphology, porous networks | Planar honeycomb lattice, flexible |
| Functionalization Potential | High (sidewall and tip functionalization) | Extensive (basal plane and edges) |
| Biocompatibility | Good | Superior to many nanomaterials |
Sensing Mechanisms for H₂O₂ Detection
Both CNT and graphene-based sensors facilitate H₂O₂ detection through electrocatalytic reactions, primarily the reduction of H₂O₂ at specific applied potentials. The mechanism involves the transfer of electrons during the redox reaction, generating a measurable current proportional to H₂O₂ concentration [7]. Nanomaterial enhancements improve sensitivity by increasing the electroactive surface area, facilitating electron transfer kinetics, and reducing the overpotential required for H₂O₂ electrocatalysis [45] [47].
Carbon nanomaterials can be further functionalized with metal nanoparticles (Pd, Au, Pt) or metal oxides to enhance their catalytic performance. For instance, palladium nanoparticles exhibit excellent electrocatalytic performance for hydrogen peroxide, enabling enhanced electron transfer and reduced overpotential properties [46]. The integration of these materials creates synergistic effects that significantly improve sensor performance compared to individual components [46].
Diagram 1: Plant Stress Signaling and H₂O₂ Electrochemical Detection Pathway
Case Study: CNT-Based Sensor for Salt Stress Monitoring in Arabidopsis
Experimental Protocol and Sensor Fabrication
A comprehensive study demonstrated the application of MWCNT-based sensors for monitoring salt stress in Arabidopsis thaliana by detecting H₂O₂ release from leaves [46]. The experimental methodology encompassed sophisticated nanomaterial synthesis, electrode modification, and plant stress analysis:
Nanocomposite Synthesis: Researchers developed a MWCNT-Ti₃C₂Tₓ-Pd nanocomposite by combining multi-walled carbon nanotubes with MXene (Ti₃C₂Tₓ) and palladium nanoparticles. The MWCNTs were first activated through sequential sonication in 1 M nitric acid solution for 30 minutes, followed by filtration and treatment in 1 M sulfuric acid with sonication for another 30 minutes. This procedure was repeated twice to ensure proper activation, followed by extensive washing with ethanol and acetone until neutral pH was achieved [46].
Material Integration: The MWCNT-Ti₃C₂Tₓ nanocomposite was formed through π-π interactions, with Pd nanoparticles adsorbed on the Ti₃C₂Tₓ surface via redox reaction of PdCl₂. Energy dispersive X-ray spectroscopy confirmed the successful synthesis by verifying the presence of both Pd and Ti elements in the final nanocomposite [46].
Electrode Modification: The nanocomposite was dispersed using optimized dispersants (PVP, PDDA, or DMF) to enhance conductivity, then applied to electrode surfaces. Cyclic voltammetry characterization in 10 mM K₃Fe(CN)₆ solution with 0.1 M KCl demonstrated that the MWCNT-Ti₃C₂Tₓ-Pd modified electrode exhibited fast electron transfer speed and the largest closed area of the CV curve, indicating an expanded active surface area and significantly improved sensor sensitivity [46].
Plant Stress Application: Arabidopsis plants were subjected to salt stress conditions, and the sensor was utilized to measure H₂O₂ released from leaves at different time intervals. The direct measurement approach enabled real-time dynamic detection of H₂O₂ release without requiring leaf lysis or fixation, thereby maintaining the living and original forms of biological samples [46].
Table 2: Key Research Reagents for CNT-Based H₂O₂ Plant Stress Sensing
| Reagent/Material | Function/Application | Experimental Role |
|---|---|---|
| MWCNTs (Multi-walled Carbon Nanotubes) | Primary sensing substrate | Provides high surface area, electrocatalytic activity, and electron transfer pathways |
| Ti₃C₂Tₓ MXene | Two-dimensional conductive support | Enhances conductivity and catalytic activity, provides attachment sites for Pd nanoparticles |
| Palladium Nanoparticles | Electrocatalytic enhancer | Improves H₂O₂ reduction through efficient electron transfer and reduced overpotential |
| Nitric Acid & Sulfuric Acid | MWCNT activation | Purifies and functionalizes MWCNT surfaces for improved composite formation |
| Polyvinylpyrrolidone (PVP) | Dispersing agent | Enhances nanomaterial dispersion and stability for consistent sensor performance |
| Arabidopsis thaliana | Model plant system | Provides standardized biological platform for salt stress response studies |
Performance Data and Validation
The MWCNT-Ti₃C₂Tₓ-Pd based sensor demonstrated exceptional performance characteristics for H₂O₂ detection in plant stress monitoring [46]:
- Linear Detection Range: 0.05–18 mM H₂O₂
- Detection Limit: 3.83 μM (S/N = 3)
- Sensitivity Enhancement: Superior electron transfer kinetics compared to individual components
- Selectivity: Effective measurement in complex plant matrix without interference
- Real-time Monitoring Capability: Continuous measurement of H₂O₂ release from Arabidopsis leaves at different salt stress timepoints
Validation studies confirmed strong correlation between sensor measurements and conventional staining methods (diaminobenzidine tetrahydrochloride), while offering the significant advantage of non-destructive, real-time monitoring without causing irreversible damage to plant tissues [46].
Comparative Analysis: CNTs vs. Graphene for Plant H₂O₂ Sensing
Direct Performance Comparison
While the search results provide substantial evidence of CNT-based sensors in direct plant stress monitoring applications, current data for graphene-based sensors in specific plant stress case studies is more limited. However, general performance comparisons can be drawn from electrochemical sensing applications:
Carbon Nanotube Advantages: The fibrous, high-aspect-ratio structure of CNTs creates porous, three-dimensional networks that facilitate efficient H₂O₂ diffusion and contact with electroactive sites [46]. Their demonstrated success in plant stress monitoring applications highlights practical applicability for real-time, in situ detection. The case study on Arabidopsis salt stress monitoring validated CNT-based sensors for non-destructive, continuous measurement of H₂O₂ release from living plant tissues [46].
Graphene Advantages: Graphene offers exceptional electrical conductivity, high intrinsic charge carrier mobility, and enormous specific surface area (theoretical value ~2630 m²/g) [3]. These properties position graphene as a strong candidate for high-sensitivity detection applications. Functionalized graphene derivatives, including graphene oxide (GO) and reduced graphene oxide (rGO), provide tunable surface chemistry for enhanced biocompatibility and catalyst integration [3].
Table 3: Carbon Nanomaterial Sensor Performance Comparison for H₂O₂ Detection
| Performance Parameter | CNT-Based Sensors | Graphene-Based Sensors |
|---|---|---|
| Reported Sensitivity | 551.02 μA mM⁻¹ cm⁻² (Au-Co@C-CNT/CC) [47] | Data limited for direct plant applications |
| Linear Range | 10–27,080 μM (Au-Co@C-CNT/CC) [47] | 0.05–18 mM (MWCNT composite) [46] |
| Detection Limit | 0.013 μM (Au-Co@C-CNT/CC) [47] | 3.83 μM (MWCNT composite) [46] |
| Response Time | Fast response demonstrated [47] | Theoretical fast electron transfer |
| Stability | Good repeatability and stability [47] [46] | High mechanical and chemical stability |
| Plant Application Evidence | Direct evidence (Arabidopsis salt stress) [46] | Limited direct plant stress evidence |
Material Integration and Functionalization Strategies
Both CNT and graphene platforms benefit significantly from composite formation and functionalization with additional nanomaterials:
CNT Composite Strategies: The successful integration of MWCNTs with MXene (Ti₃C₂Tₓ) and palladium nanoparticles demonstrates the synergistic approach to enhancing sensor performance [46]. The MWCNTs provide structural framework and conduction pathways, MXene offers high conductivity and catalytic sites, while Pd nanoparticles significantly enhance the electrocatalytic reduction of H₂O₂.
Graphene Composite Strategies: While direct plant stress monitoring evidence is limited, graphene-based composites show promise through integration with metal-organic frameworks (MOFs), metal nanoparticles, and polymers [47] [3]. These combinations aim to leverage graphene's exceptional surface area and conductivity while addressing potential limitations such as restacking of sheets or insufficient catalytic sites.
Diagram 2: Experimental Workflow Comparison: CNT vs. Graphene-Based H₂O₂ Sensors
The comparative analysis of carbon nanotubes and graphene for plant H₂O₂ sensing reveals that CNT-based sensors currently provide stronger experimental validation for direct plant stress monitoring applications. The documented case study of MWCNT-Ti₃C₂Tₓ-Pd nanocomposites for detecting salt stress in Arabidopsis demonstrates practical implementation with validated performance characteristics including wide linear range (0.05–18 mM), low detection limit (3.83 μM), and effective real-time monitoring capability [46].
While graphene offers exceptional theoretical properties including high electrical conductivity, charge carrier mobility, and specific surface area, direct evidence of its application in plant stress monitoring remains limited in the current literature. The extensive characterization of graphene's fundamental properties suggests significant potential, but practical implementation for plant H₂O₂ sensing requires further development and validation [3].
Future research directions should focus on expanding graphene-based sensor applications to direct plant stress monitoring, developing standardized comparative protocols for CNT and graphene platforms, exploring hybrid nanomaterial systems that leverage the advantages of both carbon allotropes, and advancing flexible, wearable sensor designs for non-invasive plant monitoring. These developments will enhance our understanding of plant stress physiology and support advancements in precision agriculture and plant phenotyping.
Overcoming Practical Hurdles: Strategies to Enhance Sensitivity, Selectivity, and Stability
The development of high-performance electrochemical sensors for hydrogen peroxide (H₂O₂) detection represents a critical focus in plant physiology research, environmental monitoring, and biomedical diagnostics. Within this domain, carbon nanotubes (CNTs) and graphene have emerged as particularly promising transducer materials due to their exceptional electrical conductivity, high surface-to-volume ratio, and versatile chemistry for biomolecular functionalization [48] [17]. However, the intrinsic properties of pristine carbon nanomaterials often prove insufficient for achieving the requisite selectivity toward specific analytes in complex matrices like plant extracts or biological fluids. This limitation has driven extensive research into deliberate functionalization strategies designed to engineer selectivity into these materials.
Functionalization—the process of attaching specific chemical groups or molecules to carbon nanomaterial surfaces—serves dual purposes in sensor development. First, it enables the immobilization of recognition elements (enzymes, antibodies, artificial peroxidases) that confer molecular specificity. Second, it often improves the dispersibility and interfacial compatibility of nanomaterials in aqueous or biological environments [48] [17]. These strategies are broadly categorized into covalent and non-covalent approaches, each with distinct mechanisms, advantages, and trade-offs that significantly influence sensor performance metrics including sensitivity, selectivity, stability, and reproducibility.
Within plant research, monitoring H₂O₂ dynamics is particularly valuable as this molecule functions as a key signaling compound in stress responses, growth regulation, and cellular metabolism [45] [7]. The ability to detect H₂O₂ selectively in the presence of structurally similar interferents like ascorbic acid, uric acid, and glucose is therefore paramount. This guide systematically compares covalent and non-covalent functionalization approaches for CNTs and graphene, providing researchers with experimental protocols, performance data, and practical insights to inform sensor design for plant H₂O₂ sensing applications.
Covalent Functionalization: Mechanisms and Methodologies
Fundamental Principles and Chemical Approaches
Covalent functionalization involves the formation of strong, irreversible chemical bonds between functional groups on carbon nanomaterials and modifier molecules. This approach typically begins with an oxidative pretreatment that introduces oxygen-containing functional groups (such as carboxyl, hydroxyl, or epoxy groups) onto CNT sidewalls or graphene basal planes, creating anchoring sites for subsequent chemical reactions [48]. The most common covalent strategy employs carbodiimide chemistry (typically using EDC/NHS reagents) to activate carboxyl groups on oxidized carbon nanomaterials for conjugation with primary amine groups present in biomolecules or other functional modifiers [48].
This covalent grafting approach creates stable, permanent attachments that resist desorption under changing environmental conditions such as pH shifts or ionic strength variations. The resulting functionalized materials typically exhibit enhanced aqueous dispersibility and provide precisely controlled surface chemistries for biomolecule immobilization. However, the formation of covalent bonds necessarily disrupts the extended π-conjugation system of graphene and CNTs, potentially diminishing their electrical conductivity and electron transfer kinetics—a significant consideration for electrochemical sensing applications [48] [49].
Representative Experimental Protocol: Enzyme Immobilization on CNTs
A well-established protocol for covalent functionalization involves immobilizing horseradish peroxidase (HRP) onto CNTs for enzymatic H₂O₂ sensing [48]:
- Step 1: CNT Oxidation – Pristine MWCNTs (50 mg) are refluxed in concentrated HNO₃ or H₂SO₄:HNO₃ (3:1 v/v) at 60°C for 4-6 hours to generate carboxyl groups. The resulting carboxylated CNTs are collected by vacuum filtration through a 0.22 μm membrane and washed thoroughly with deionized water until neutral pH.
- Step 2: Carboxyl Activation – The oxidized CNTs (10 mg) are dispersed in 10 mL of MES buffer (pH 6.0) containing 20 mM EDC and 10 mM NHS. The activation proceeds with gentle shaking for 30-60 minutes at room temperature to form amine-reactive NHS esters.
- Step 3: Enzyme Conjugation – The activated CNTs are separated by centrifugation and redispersed in 10 mL of phosphate buffer (pH 7.4) containing HRP (2 mg/mL). The reaction proceeds for 2 hours at room temperature with continuous mixing.
- Step 4: Washing and Storage – The HRP-CNT conjugates are collected by centrifugation and washed repeatedly with phosphate buffer to remove physically adsorbed enzyme. The final product is resuspended in storage buffer (typically pH 7.4 PBS) at 4°C.
This covalent approach typically achieves high enzyme loading densities with stable attachment, but may partially compromise the intrinsic electrochemical properties of the CNTs and requires careful control of reaction conditions to prevent enzyme denaturation.
Non-Covalent Functionalization: Mechanisms and Methodologies
Fundamental Principles and Assembly Approaches
Non-covalent functionalization relies on supramolecular interactions—including π-π stacking, van der Waals forces, electrostatic interactions, and hydrophobic effects—to adsorb functional molecules onto carbon nanomaterial surfaces without forming permanent chemical bonds [48] [49]. This approach preserves the pristine sp² carbon network of CNTs and graphene, maintaining their exceptional electrical conductivity and structural integrity while still imparting desired surface functionalities.
Aromatic organic molecules (e.g., pyrene derivatives), surfactants (e.g., SDS), polymers (e.g., polyethyleneimine, polystyrene sulfonate), and biomolecules (e.g., single-stranded DNA) can all adsorb strongly to carbon nanomaterial surfaces through these non-covalent interactions [48] [49] [4]. The resulting assemblies are thermodynamically stable under appropriate conditions but may be susceptible to displacement or desorption under changing solvent conditions or in the presence of competing molecules.
Representative Experimental Protocol: Hemin-PEI/MWCNT Modified Electrodes
A sophisticated non-covalent functionalization approach for H₂O₂ sensing involves modifying screen-printed graphene electrodes with hemin-polyethyleneimine (PEI) and MWCNTs [4]:
- Step 1: MWCNT Dispersion – MWCNTs (1 mg/mL) are dispersed in DMF by ultrasonication for 30-60 minutes to create a stable suspension.
- Step 2: Hemin-PEI Complex Formation – Hemin (2 mg/mL) is dissolved in DMSO and mixed with an equal volume of aqueous PEI solution (1% w/v) under vigorous stirring. The mixture is incubated for 1 hour to allow complex formation between hemin and the cationic polymer.
- Step 3: Electrode Modification – The MWCNT suspension (5 μL) is drop-cast onto the screen-printed graphene electrode and dried at room temperature. Subsequently, the hemin-PEI complex (5 μL) is deposited onto the MWCNT-modified electrode and dried.
- Step 4: Electrode Conditioning – The modified electrode is rinsed with phosphate buffer (pH 7.0) to remove loosely bound materials and stabilized by cyclic voltammetry scanning between -0.5 and 0.5 V until a stable response is obtained.
This design leverages π-π interactions between MWCNTs and graphene, electrostatic interactions between hemin and PEI, and the peroxidase-mimicking activity of hemin to create a highly sensitive H₂O₂ sensor without covalent modification of the carbon nanostructures [4].
Comparative Performance Analysis: Covalent vs. Non-Covalent Approaches
Direct Comparison of Functionalization Strategies
The table below summarizes the key characteristics of covalent versus non-covalent functionalization strategies for carbon nanomaterials in H₂O₂ sensing applications:
Table 1: Comparison of Covalent and Non-Covalent Functionalization Strategies
| Parameter | Covalent Functionalization | Non-Covalent Functionalization |
|---|---|---|
| Bond Type | Strong covalent bonds | Weak non-covalent interactions (π-π stacking, electrostatic, van der Waals) |
| Impact on Carbon Nanostructure | Disrupts π-conjugation, creates defect sites | Preserves intrinsic sp² carbon network |
| Stability | Excellent (irreversible attachment) | Moderate (potential desorption under stress conditions) |
| Electrical Properties | May reduce conductivity | Maintains high conductivity |
| Biomolecule Activity | Risk of denaturation due to harsh chemistry | Milder conditions preserve bioactivity |
| Reproducibility | High (controlled reaction stoichiometry) | Variable (dependent on assembly conditions) |
| Typical Applications | Enzyme electrodes, immunosensors [48] | Polymer-nanotube composites, artificial peroxidase mimics [49] [4] |
Analytical Performance in H₂O₂ Sensing
The table below compares the performance of selected covalently and non-covalently functionalized carbon nanomaterial-based sensors for H₂O₂ detection:
Table 2: Performance Comparison of Functionalized Carbon Nanomaterial-Based H₂O₂ Sensors
| Sensor Design | Functionalization Strategy | Linear Range (μM) | Detection Limit (μM) | Sensitivity | Reference |
|---|---|---|---|---|---|
| HRP-CNT/Chitosan | Covalent (EDC/NHS) | 5–5,130 | 1.7 | Not specified | [48] |
| AuNPs/BDMT/Graphene | Non-covalent (π-π) | Not specified | 1.8 | Not specified | [49] |
| Hemin-PEI/MWCNT | Non-covalent (electrostatic) | 0.5–200 | 0.15 | 18.09 A M⁻¹ cm⁻² | [4] |
| Prussian Blue-MWCNT-IL | Hybrid approach | 5–1,645 | 0.35 | 0.436 μA mM⁻¹ cm⁻² | [7] |
The data reveals that non-covalent approaches frequently achieve superior detection limits and sensitivities, attributable to better preservation of the carbon nanomaterials' electrical properties. The hemin-PEI/MWCNT system demonstrates particularly impressive performance with sub-micromolar detection limits relevant to plant H₂O₂ sensing applications [4].
CNTs vs. Graphene for H₂O₂ Sensing Applications
Material-Specific Considerations for Functionalization
While both CNTs and graphene offer exceptional properties for sensor development, each presents distinct advantages and challenges concerning functionalization strategies and resulting sensor performance:
Table 3: Comparison of CNTs and Graphene for H₂O₂ Sensing Applications
| Parameter | Carbon Nanotubes (CNTs) | Graphene |
|---|---|---|
| Dimensionality | One-dimensional (high aspect ratio) | Two-dimensional (planar geometry) |
| Surface Area | High external surface area | Extremely high theoretical surface area |
| Electron Transfer | Fast electron transfer kinetics | Excellent in-plane conductivity |
| Functionalization Sites | Ends and sidewall defect sites | Basal plane and edges |
| Typical H₂O₂ Detection Limit | ~0.15 μM (hemin-PEI/MWCNT) [4] | ~1.8 μM (AuNPs/BDMT/graphene) [49] |
| Dispersion Stability | Moderate (tendency to bundle) | Variable (restacking issues) |
| Mechanical Properties | High strength and flexibility | Exceptional stiffness and strength |
Molecular dynamics simulations and experimental studies suggest that graphene may provide more efficient load transfer to polymer matrices compared to CNTs, potentially explaining its superior performance in some composite configurations [50]. However, CNTs often demonstrate advantages in creating three-dimensional conductive networks that facilitate electron transfer to embedded catalytic sites.
Visualizing Functionalization Strategies and Sensing Mechanisms
The following diagram illustrates the key functionalization strategies and their relationship to sensing mechanisms for H₂O₂ detection:
Carbon Nanomaterial Functionalization for H₂O₂ Sensing
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of functionalization strategies requires specific reagents and materials optimized for carbon nanomaterial processing:
Table 4: Essential Research Reagents for Carbon Nanomaterial Functionalization
| Reagent/Material | Function/Purpose | Example Applications |
|---|---|---|
| N-Ethyl-N'-(3-dimethylaminopropyl) carbodiimide (EDC) | Carboxyl group activation for amide bond formation | Covalent conjugation of enzymes to oxidized CNTs/graphene [48] |
| N-Hydroxysuccinimide (NHS) | Stabilization of activated esters improves coupling efficiency | Enhanced yield in biomolecule immobilization [48] |
| Polyethyleneimine (PEI) | Cationic polymer for electrostatic functionalization | Hemin entrapment and dispersion stabilization [4] |
| 1,4-Benzenedimethanethiol (BDMT) | π-π stacking linker with thiol termini | Bridge between graphene and gold nanoparticles [49] |
| Hemin | Iron protoporphyrin with peroxidase-mimicking activity | Non-enzymatic H₂O₂ sensing catalyst [4] |
| Prussian Blue (PB) | Artificial peroxidase catalyst | H₂O₂ reduction at low overpotentials [7] |
| N,N-Dimethylformamide (DMF) | High-quality dispersion of carbon nanomaterials | Solvent for MWCNT suspension preparation [4] |
The strategic selection between covalent and non-covalent functionalization approaches for carbon nanomaterials involves critical trade-offs between stability, selectivity, and electron transfer efficiency in H₂O₂ sensor design. Covalent methods provide robust, permanent biomolecule attachment ideal for applications requiring long-term operational stability, while non-covalent approaches preserve the exceptional electrochemical properties of carbon nanomaterials, enabling superior sensitivity for detecting physiologically relevant H₂O₂ concentrations in plant systems.
Future developments will likely focus on hybrid strategies that combine the advantages of both approaches—for instance, using non-covalent π-π stacking to attach molecular anchors that subsequently permit oriented covalent immobilization of recognition elements. Additionally, the growing availability of high-purity, defect-controlled CNTs and graphene [44] [51] will enable more reproducible functionalization outcomes. As the fundamental understanding of nanomaterial-biointerfaces advances, rational design of functionalized carbon nanomaterials will further enhance the selectivity, sensitivity, and practical utility of H₂O₂ sensors in plant science research and beyond.
The exceptional properties of carbon nanotubes (CNTs) and graphene make them premier materials for advanced sensing applications, including the precise detection of hydrogen peroxide (H₂O₂) in plant research. However, their translational potential from laboratory demonstrations to reliable, commercially viable sensors is severely hampered by significant reproducibility challenges. For CNTs, batch-to-batch variations in synthesis yield inconsistent electronic and physical properties, while for graphene, the transfer process from growth substrates to application-ready devices introduces defects and contamination that cripple performance consistency [44] [1]. This guide provides an objective, data-driven comparison of these challenges, offering researchers a clear framework for selecting and implementing these nanomaterials with a focus on achieving reliable and reproducible results in H₂O₂ sensing.
Material Comparison: CNTs vs. Graphene for H₂O₂ Sensing
The choice between CNTs and graphene involves a fundamental trade-off between the intrinsic reproducibility of the material itself and the complexity of integrating it into a functional device. The table below summarizes the core challenges and performance implications for each material.
Table 1: Core Reproducibility Challenges in CNT Synthesis and Graphene Transfer
| Parameter | Carbon Nanotubes (CNTs) | Graphene |
|---|---|---|
| Primary Challenge | Chirality & Metallic/Semiconducting Ratio Control [17] [1] | Defect-Free Transfer & Substrate Contamination [44] |
| Impact on H₂O₂ Sensing | Inconsistent sensor baseline resistance & sensitivity; semiconducting CNTs required for FET-based sensors [17] | Unpredictable electron transfer kinetics & electrocatalytic activity [44] [52] |
| Key Reproducibility Metrics | Semiconducting purity (>99.9999% for electronics), Diameter (1.3 nm ± 0.12 nm) [44] | Device yield (up to 97%), Contamination levels (<10¹² atoms/cm²) [44] |
| Industrial Scaling Status | CVD growth scaled to 500-1000 tons/year; prices declining 15-25% in 2024 [44] | NanoXplore capacity at 4,000 tons/year; costs at $100-1,000/kg [44] |
Quantitative Performance Data for H₂O₂ Sensing
To objectively compare the real-world sensing potential of these materials, their performance must be evaluated against key metrics. The following table compiles experimental data from recent studies on CNT and graphene-based sensors configured for H₂O₂ detection.
Table 2: Performance Comparison of CNT and Graphene-Based H₂O₂ Sensors
| Sensor Material | Detection Limit | Linear Range | Sensitivity | Response Time | Reference |
|---|---|---|---|---|---|
| Fe₃O₄/Graphene/CC | 4.79 µM | 10–110 µM | 0.037 µA µM⁻¹ cm⁻² | Not Specified | [52] |
| Fe₃O₄/3D Graphene | ~78 nM | Not Specified | 274.15 mA M⁻¹ cm⁻² | 2.8 s | [52] |
| Carbon Dots/Fe₃O₄ | 1.0 nM | 10 nM – 1 mM | Not Specified | Not Specified | [52] |
| Au@Pt Nanorods/GC* | 189 nM | 500 nM – 50 µM | High (Nearly 2x smoother NRs) | <5 s | [53] |
| CNT-based (General) | ppt to ppq levels (for gases) [54] | Varies by functionalization | Very High (down to ppb/ppt) [1] | 5 – 1000 s [54] | [54] [1] |
Note: Au@Pt Nanorods represent a high-performance noble-metal sensor shown for benchmark comparison, demonstrating the performance level achievable with optimal catalysis. GC = Glassy Carbon.
Experimental Protocols for Reproducible Fabrication
Protocol for CNT-Based H₂O₂ Sensor Fabrication
Achieving reproducible CNT sensors hinges on precise control over the network formation and functionalization of the nanotubes.
- CNT Dispersion Preparation: Begin with high-purity semiconducting Single-Walled CNTs (SWCNTs). Disperse the CNTs in a suitable solvent (e.g., N-Methyl-2-pyrrolidone or aqueous surfactant solutions like sodium dodecyl sulfate) using probe ultrasonication for 30-60 minutes. This step is critical to break up aggregates and create a stable, homogeneous suspension [17] [1].
- Surface Functionalization (Non-Covalent): To enhance selectivity towards H₂O₂ and reduce interference, non-covalent functionalization is preferred to preserve the CNT's π-conjugated network and electrical properties. Introduce functional molecules or polymers via π-π stacking. For instance, immobilize metalloporphyrins or specific metal oxides (e.g., Fe₃O₄) that catalyze H₂O₂ redox reactions [17] [45].
- Electrode Modification & Network Formation: Deposit the functionalized CNT dispersion onto the target electrode (e.g., glassy carbon or flexible carbon cloth) via drop-casting, spin-coating, or vacuum filtration. Control the density of the CNT network precisely, as the junction resistance between nanotubes is a key factor in the sensor's chemiresistive response [52] [17]. Anneal the device at a mild temperature (e.g., 80-120°C) to remove residual solvent and stabilize the film.
Protocol for Graphene-Based H₂O₂ Sensor Fabrication
The primary challenge for graphene sensors lies in the transfer and interface engineering of the graphene layer.
- Graphene Growth & Preparation: Utilize large-area, high-quality graphene grown via Chemical Vapor Deposition (CVD) on a copper foil substrate. Alternatively, employ commercially available Laser-Induced Graphene (LIG) patterned directly onto a polyimide substrate, which avoids transfer issues [55].
- Etch-Free Dry Transfer: For CVD graphene, avoid wet-etching transfer methods that leave polymer residues (e.g., PMMA). Instead, use an etch-free dry transfer technique. This involves laminating a thermal release tape to the graphene/copper stack and electrochemically delaminating the graphene by bubbling hydrogen at the interface. The graphene is then directly bonded to the target substrate (e.g., SiO₂/Si or a sensor electrode) upon heating [44].
- Functionalization & Electrode Integration: Decorate the transferred graphene surface with catalytic nanoparticles to boost H₂O₂ sensitivity. A common method is the direct application of a metal oxide suspension, such as CuO, by spray-coating [55]. Alternatively, electrodeposit Fe₃O₄ nanoparticles to form a Fe₃O₄/Graphene nanocomposite on a flexible carbon cloth electrode, creating a robust three-dimensional catalytic interface [52].
Visualization of Workflows and Challenges
The following diagrams illustrate the critical pathways and workflows discussed, highlighting the points where reproducibility is most at risk.
Diagram 1: Graphene transfer challenge. The transfer process is a critical failure point where contamination and defects are introduced [44].
Diagram 2: CNT synthesis and processing challenges. Multiple steps, from synthesis to network formation, contribute to performance variability [17] [1].
The Scientist's Toolkit: Essential Research Reagents & Materials
Selecting the appropriate starting materials and reagents is fundamental to overcoming reproducibility hurdles.
Table 3: Essential Reagents for CNT and Graphene H₂O₂ Sensor Research
| Material / Reagent | Function & Rationale | Key Considerations for Reproducibility |
|---|---|---|
| Semiconducting SWCNTs | The sensing channel in FET or resistive sensors; high carrier mobility enhances sensitivity [17] [1]. | Specify semiconducting purity (e.g., >99.9%) and source. Verify diameter distribution via absorption spectroscopy. |
| CVD Graphene on Cu Foil | Provides a high-quality, continuous monolayer sheet for transparent or flexible electrodes [44]. | Inquire about grain size and the availability of etch-free dry transfer services to minimize contamination. |
| Laser-Induced Graphene (LIG) | A transfer-free alternative; a 3D porous graphene network formed by lasing polyimide [55]. | Consistency depends on laser power, scan speed, and substrate material. Optimal for custom, in-situ fabrication. |
| Metal Oxide Nanoparticles (e.g., CuO, Fe₃O₄) | Catalytic elements that enhance sensitivity and selectivity for H₂O₂ redox reaction [55] [52]. | Control nanoparticle size, crystallinity, and dispersion quality on the carbon surface. |
| Functionalization Agents | Molecules (e.g., polymers, porphyrins) for non-covalent CNT/graphene modification to improve selectivity [17]. | Preserve the sp² carbon network to maintain electrical properties while introducing specific binding sites. |
| Flexible Carbon Cloth (CC) | A robust, porous, and flexible support for working electrodes in wearable or plant-root sensors [52]. | Ensure consistent porosity and surface pretreatment to guarantee uniform nanomaterial loading. |
The effective interfacing of biological components with electronic transducers is a central challenge in developing advanced biosensors. For the specific application of plant hydrogen peroxide (H₂O₂) sensing—a key signaling molecule in plant physiology and stress responses—the choice of electrode material profoundly influences sensitivity, selectivity, and stability. Among the various nanomaterials available, carbon nanotubes (CNTs) and graphene have emerged as front-runners due to their exceptional electrical, chemical, and physical properties [56]. This guide provides a objective comparison between CNTs and graphene, focusing on their performance in configuring bio-interfaces for enzymatic H₂O₂ sensing, with a particular emphasis on strategies to achieve efficient Direct Electron Transfer (DET).
DET is a highly sought-after mechanism in bioelectrochemistry as it allows electrons to shuttle directly between the enzyme's active site and the electrode surface without requiring secondary mediators [57] [58]. This simplifies sensor design, minimizes potential interference, and can improve stability. However, a significant challenge is that the redox cofactors of many enzymes are deeply embedded within a protein matrix, making direct electron tunneling difficult over distances typically greater than 10-20 Å [57] [58]. Both CNTs and graphene offer nanostructured surfaces that can help overcome this barrier by facilitating closer contact and more efficient wiring of enzymes to the electrode.
Material Properties and Functionalization Strategies
The intrinsic properties of carbon nanomaterials directly impact their effectiveness in bio-interfaces. The table below summarizes and compares the key characteristics of CNTs and graphene.
Table 1: Comparative Properties of Carbon Nanotubes and Graphene for Biosensing
| Property | Carbon Nanotubes (CNTs) | Graphene |
|---|---|---|
| Dimensionality | 1D (cylindrical nanostructure) [17] | 2D (planar sheet) [3] |
| Electrical Conductivity | Very high, can be metallic or semiconducting [56] [17] | Very high, semi-metallic [3] |
| Surface Area | High, but aggregation can reduce accessibility [17] | Extremely high theoretical surface area (2630 m²/g) [3] |
| Functionalization | Covalent (sidewall/end oxidation) and non-covalent (π-π stacking, polymer wrapping) [56] [17] | Covalent (e.g., graphene oxide) and non-covalent (π-π stacking) [3] |
| Dispersion | Poor without functionalization; tends to bundle [56] [17] | Sheets can restack via van der Waals forces, reducing surface area [59] [3] |
| Enzyme Immobilization | Nanotube diameter can allow penetration into enzyme grooves [57] [58] | Planar surface for enzyme adsorption; composite forms can create 3D scaffolds [60] [59] |
Functionalization is often essential to render these nanomaterials processable and bioactive. Both materials can be modified via:
- Covalent functionalization: Introduces chemical groups (e.g., carboxyl) onto the carbon framework, improving solubility and providing anchor points for enzyme attachment [56] [17].
- Non-covalent functionalization: Uses aromatic molecules or polymers that adsorb onto the material surface via π-π interactions, preserving the intrinsic electrical properties of the carbon lattice while enabling biomolecule immobilization [56] [17].
Performance Comparison for H₂O₂ Sensing
The ultimate test of a sensing platform is its analytical performance. The following table compiles experimental data from recent studies on CNT- and graphene-based electrochemical H₂O₂ sensors.
Table 2: Experimental Performance of CNT- and Graphene-Based H₂O₂ Sensors
| Electrode Material | Linear Range (μM) | Sensitivity (μA mM⁻¹ cm⁻²) | Detection Limit (μM) | Key Findings & Reference |
|---|---|---|---|---|
| Fe₃O₄/Graphene/Carbon Cloth | 10 – 110 | 0.037 | 4.79 | Flexible sensor; highly reproducible and stable [52]. |
| 3D rGO–Ti₃C₂–MWCNTs | 1 – 60 / 60 – 9770 | 235.2 / 103.8 | 0.3 | Used for real-time H₂O₂ detection in cancer cells and tissue; excellent interference immunity [59]. |
| Fe₃O₄/3D Graphene | Not specified | 274.15 | 0.078 | In situ detection of H₂O₂ from living cells; fast response time (2.8 s) [52]. |
| rGO–MXene–MWCNTs (with Pd) | 50 – 18000 | Not specified | Not specified | Demonstrated a wide linear range for H₂O₂ detection [59]. |
| Debundled SWNTs with FAD-GDH | Glucose-dependent current response observed | N/A | N/A | Debundled SWNTs enabled DET, while aggregated SWNTs/MWNTs did not [57] [58]. |
Analysis of Comparative Data
- Sensitivity and Detection Limit: Graphene-based composites, particularly three-dimensional (3D) architectures like 3D rGO–MXene–MWCNTs and Fe₃O₄/3D Graphene, demonstrate superior sensitivity and lower detection limits. This is attributed to their large electroactive surface area and synergistic effects that enhance electron transfer [52] [59].
- Role of Composites: The highest performance is consistently achieved not by pristine CNTs or graphene, but by their composites with other nanomaterials (e.g., metals, metal oxides, MXenes) or with each other. For instance, a composite of single-walled carbon nanotubes (SWNTs) with graphene showed a 4.61-fold and 2.45-fold higher DET efficiency for Glucose Oxidase (GOx) compared to SWNTs or graphene alone, respectively [60]. This hybrid approach mitigates the weaknesses of individual materials, such as CNT bundling or graphene restacking [59].
Experimental Protocols for Key Performers
To enable researchers to replicate and build upon these findings, this section details the experimental methodologies for fabricating two high-performing sensor architectures from the comparison table.
This protocol describes a one-step hydrothermal method to create a 3D porous hydrogel electrode ideal for sensitive H₂O₂ detection.
- Solution Preparation: Prepare an aqueous mixture of Graphene Oxide (GO, 0.4 mg mL⁻¹), MXene (Ti₃C₂, 0.4 mg mL⁻¹), and multi-walled carbon nanotubes (MWCNTs, 0.4 mg mL⁻¹).
- Dispersion: Subject the mixture to ultrasonication for 2 hours to achieve a homogeneously dispersed suspension.
- Hydrothermal Reaction: Transfer 2 mL of the suspension into a 5 mL Teflon-lined autoclave, suspending a copper wire (diameter 0.2 mm) inside. Maintain the autoclave at 180 °C for 4 hours. This process simultaneously reduces GO to rGO and self-assembles the components into a 3D hydrogel film on the copper wire.
- Drying and Insulation: Dry the modified electrode at room temperature. Finally, coat the side of the electrode cylinder with insulating wax to form a disk electrode with a defined surface area.
This protocol focuses on preparing debundled SWNTs to minimize the electron tunneling distance to the FAD cofactor of FAD-dependent glucose dehydrogenase (FAD-GDH), thereby enabling DET.
- CNT Debundling: Disperse SWNT powder in distilled water with a surfactant (e.g., Tween 20).
- Sonication: Bath sonicate the mixture for 30 minutes to break down large aggregates, followed by tip ultrasonication (e.g., 20 kW for 1 hour in an ice bath) to exfoliate individual nanotubes.
- Purification: Centrifuge the sonicated solution at 10,000× g for 20 minutes at 4 °C to remove any remaining aggregates. Collect the supernatant containing the debundled SWNTs.
- Enzyme Integration: Mix the dispersed, debundled SWNT solution with the FAD-GDH enzyme solution. The key is that the small diameter (~1.2 nm) of the debundled SWNTs allows them to plug into the grooves of the enzyme, bringing the electrode surface within tunneling distance of the FAD cofactor.
Visualizing Direct Electron Transfer Pathways
The following diagram illustrates the core concepts of DET and the superior configuration achieved by composite materials, which is critical for optimizing the bio-interface.
Diagram 1: Contrasting DET pathways shows that composites create shorter, more efficient electron transfer.
The Scientist's Toolkit: Essential Research Reagents
This table lists key materials and their functions for researchers developing CNT- or graphene-based enzymatic biosensors for H₂O₂.
Table 3: Essential Reagents for Enzyme-Based H₂O₂ Sensor Development
| Reagent / Material | Function in Experiment | Key Consideration |
|---|---|---|
| Single-Walled Carbon Nanotubes (SWNTs) | 1D conductive nanomaterial; can facilitate DET when debundled [57] [58]. | Chirality affects electrical properties; requires dispersion for use [60] [17]. |
| Graphene Oxide (GO) / Reduced GO (rGO) | 2D platform for enzyme immobilization; rGO offers good conductivity [59] [3]. | rGO has more defects than pristine graphene but is easier to process [3]. |
| FAD-Dependent Glucose Dehydrogenase (FAD-GDH) | Oxygen-insensitive biocatalyst for glucose oxidation; preferred over GOx for DET studies [57] [58]. | Avoids side reactions with O₂, improving coulombic efficiency in fuel cells/sensors. |
| MXene (e.g., Ti₃C₂) | 2D conductive transition metal carbide; used to form 3D porous composites with rGO [59]. | Provides abundant surface functional groups for redox reactions; prevents restacking [59]. |
| Surfactant (e.g., Tween 20) | Aids in the dispersion and debundling of CNTs in aqueous solutions [57] [58]. | Critical for achieving a homogeneous suspension and exposing more surface area. |
| Phosphate Buffered Saline (PBS) | Standard electrolyte for maintaining stable pH and ionic strength during electrochemical tests [60]. | Essential for ensuring enzyme stability and reproducible electrochemical measurements. |
For plant H₂O₂ sensing, the choice between carbon nanotubes and graphene is not a simple binary one. Pristine graphene tends to offer advantages in creating high-surface-area scaffolds, while debundled SWCNTs show unique capability in plugging into enzyme grooves for shorter DET pathways. However, the most significant performance enhancements are realized through the formation of synergistic nanocomposites that combine CNTs, graphene, and other nanomaterials like MXenes [60] [59].
Future research directions will likely focus on the rational design of these 3D hierarchical structures and the use of protein engineering to tailor enzymes for more efficient coupling with nanocarbon surfaces [61]. For the plant science researcher, this translates to selecting a composite material strategy to develop highly sensitive, stable, and reliable H₂O₂ biosensors for probing plant stress signaling and physiology.
The deployment of advanced nanomaterials like carbon nanotubes (CNTs) and graphene in electrochemical sensing, particularly for the detection of plant hydrogen peroxide (H₂O₂), represents a significant technological frontier. H₂O₂ is a crucial signaling molecule in plant physiology, involved in stress responses, growth regulation, and systemic signaling. Accurate, stable measurement in complex plant matrices is essential for understanding these processes. While both CNTs and graphene offer exceptional electrical conductivity, high surface-to-volume ratios, and promising electrocatalytic properties, their translation from controlled laboratory settings to real-world agricultural and research applications is hampered by two persistent challenges: environmental interference and signal drift.
Environmental interference arises when non-target species (e.g., other electroactive chemicals in plant sap, variations in pH, or ionic strength) affect the sensor's signal. Concurrently, signal drift—a gradual change in the sensor's baseline response over time—can be caused by factors such as material fouling, hydration changes, or electrode degradation. These issues compromise the long-term reliability and accuracy of measurements. This guide provides a objective comparison of CNT and graphene-based sensors for plant H₂O₂ detection, focusing on strategies to overcome these critical limitations. It synthesizes recent experimental data and detailed methodologies to equip researchers with the knowledge to select and optimize the most suitable nanomaterial platform for their specific needs.
Material Properties and Sensing Performance Comparison
The intrinsic properties of carbon nanotubes and graphene directly influence their performance and vulnerability to environmental interference in H₂O₂ sensing.
Carbon Nanotubes (CNTs) are cylindrical nanostructures with a quasi-one-dimensional morphology. This structure provides an exceptionally high aspect ratio and a surface area that often exceeds 1000 m²/g, creating abundant active sites for H₂O₂ interaction [1]. Their electronic properties can be semiconducting or metallic, depending on their chirality, and they exhibit excellent electrical conductivity (10²–10⁵ S/m) [1]. A key advantage is their excellent functionalization capability, allowing for the covalent or non-covalent attachment of groups that enhance selectivity toward H₂O₂ and mitigate fouling [1]. However, a significant drawback is their tendency toward batch-to-batch variability during synthesis, which can impact the reproducibility of sensor performance [1]. Furthermore, controlling their dispersion and alignment on sensor substrates remains challenging, potentially leading to inconsistent responses.
Graphene is a single layer of sp²-hybridized carbon atoms arranged in a two-dimensional honeycomb lattice. It boasts an even higher theoretical surface area (~2630 m²/g) and outstanding electron mobility, which can exceed 200,000 cm²/V·s in pristine forms [62] [63]. This facilitates rapid and sensitive signal transduction. However, in practice, graphene sheets, especially in the form of graphene oxide (GO) or reduced graphene oxide (rGO), are prone to restacking due to strong π-π interactions and van der Waals forces [31]. This agglomeration can significantly reduce the accessible surface area and hinder analyte diffusion, thereby diminishing sensor performance over time and contributing to signal drift.
Table 1: Comparative Analysis of CNT and Graphene for H₂O₂ Sensing
| Property | Carbon Nanotubes (CNTs) | Graphene |
|---|---|---|
| Dimensionality | Quasi-1D (Cylindrical) [1] | 2D (Planar Sheet) [63] |
| Specific Surface Area | >1000 m²/g [1] | ~2630 m²/g (theoretical) [63] |
| Electrical Conductivity | 10² – 10⁵ S/m [1] | ~10⁴ S/m (rGO films) [1] |
| Key Advantage for H₂O₂ Sensing | High functionalization capability; efficient electron transfer through 1D structure [1] | Very high intrinsic electron mobility; large basal plane for adsorption [62] |
| Primary Vulnerability to Interference/Drift | Batch-to-batch variability; uncontrolled agglomeration [1] | Restacking of sheets; fouling of basal plane [31] |
Experimental Data and Performance Metrics
Direct comparisons and data from specific sensor configurations highlight how these material properties translate into sensing performance. Hybrid structures often demonstrate superior performance by leveraging synergistic effects.
CNT-Based Sensor Performance: CNTs integrated into screen-printed electrodes (SPEs) have demonstrated strong performance. One study developed a disposable SPE modified with a composite of multi-walled carbon nanotubes (MWCNTs) and graphene, which achieved a sensitivity of 0.0027 µA µM⁻¹ and a limit of detection (LOD) of 7.1 µM for H₂O₂ [64]. This composite approach mitigates some limitations of individual materials by creating a more robust conductive network.
Graphene-Based Sensor Performance: Three-dimensional (3D) graphene structures have been engineered to overcome the restacking problem. A recent study created a 3D graphene hydrogel (3DGH) decorated with NiO octahedrons for enzymeless H₂O₂ detection [31]. This 3D porous network provides a large accessible surface area and efficient mass transport. The optimized sensor exhibited a high sensitivity of 117.26 µA mM⁻¹ cm⁻², a wide linear range (10 µM–33.58 mM), and a LOD of 5.3 µM [31].
Hybrid Material Performance: Hybridizing metal oxides with nanocarbons can drastically enhance sensing capabilities. Research on zinc oxide (ZnO) nanowire networks hybridized with CNTs showed that the CNT concentration is critical. An optimal CNT content of 2.0 wt% increased the sensor's response to an external stimulus (used as a proxy for enhanced sensitivity) by about 50 times compared to the pristine ZnO network [65]. This synergy is often attributed to the CNTs improving electrical conductivity and providing additional catalytic sites, which enhances charge transfer and stabilizes the signal.
Table 2: Experimental H₂O₂ Sensing Performance of Different Nanocarbon Platforms
| Sensor Platform | Sensitivity | Linear Range | Limit of Detection (LOD) | Key Findings |
|---|---|---|---|---|
| MWCNT/Graphene/SPE [64] | 0.0027 µA µM⁻¹ | Not Specified | 7.1 µM | Composite electrode shows feasibility for disposable, low-cost H₂O₂ sensing. |
| 3D Graphene Hydrogel/NiO [31] | 117.26 µA mM⁻¹ cm⁻² | 10 µM – 33.58 mM | 5.3 µM | 3D structure prevents restacking, provides high sensitivity and wide dynamic range. |
| ZnO-CNT Network (2.0 wt% CNT) [65] | Response increased ~50x | Not Specified | Not Specified | Demonstrates the critical impact of optimal nanocarbon hybridization for enhancing response. |
Mitigation Strategies: Protocols for Enhanced Stability
To combat environmental interference and signal drift, specific material engineering and experimental strategies are employed.
Strategy 1: Surface Functionalization
Objective: To improve selectivity for H₂O₂ and reduce non-specific binding from other molecules in plant extracts (e.g., ascorbic acid, uric acid, sugars). Detailed Protocol:
- Covalent Functionalization: Pristine CNTs or graphene can be treated with strong acids to generate carboxyl (-COOH) groups on their surface. These groups can then be activated using carbodiimide chemistry (e.g., EDC/NHS) to covalently link specific recognition elements, such as enzymes or catalysts. For instance, functionalizing with metal nanoparticles or porphyrin complexes can enhance the electrocatalytic reduction or oxidation of H₂O₂ [1] [63].
- Non-Covalent Functionalization: This involves coating the nanomaterial with polymers (e.g., Nafion) or surfactants. A common method is to disperse the CNTs or graphene in a solution of the polymer (e.g., 0.5% Nafion in ethanol) via sonication. The polymer coats the carbon nanomaterial through hydrophobic or π-π interactions, creating a size-exclusion or charge-selective barrier that repels common interferents found in plant tissues, thereby reducing interference and fouling [1].
Strategy 2: Constructing 3D Hybrid Networks
Objective: To prevent agglomeration (for graphene) or uncontrolled bundling (for CNTs), thereby increasing active site accessibility and reducing signal drift caused by active site occlusion. Detailed Protocol:
- 3D Graphene Hydrogel Synthesis: This is typically achieved via a one-step hydrothermal method [31].
- Begin by synthesizing graphene oxide (GO) from graphite powder using a modified Hummers method.
- Prepare a homogeneous aqueous dispersion of GO (e.g., 2 mg/mL) and seal it in a Teflon-lined autoclave.
- Heat the autoclave to 180°C for 12 hours. Under this high-temperature and high-pressure environment, the GO sheets are partially reduced and self-assemble into a 3D porous hydrogel via π-π stacking and hydrophobic interactions.
- The resulting 3DGH can be freeze-dried to preserve its porous structure. This 3D network offers a high surface area, minimizes restacking, and facilitates the decoration of other nanomaterials (e.g., NiO) for enhanced H₂O₂ catalysis [31].
Strategy 3: Incorporating Catalytic Nanoparticles
Objective: To lower the overpotential for H₂O₂ oxidation/reduction, which minimizes the impact of other electroactive species that oxidize at higher potentials, thus improving selectivity and signal-to-noise ratio. Detailed Protocol:
- Decoration with Nickel Oxide (NiO):
- Synthesize NiO octahedrons using a hard template method with mesoporous silica (SBA-15) and nickel nitrate hexahydrate [31].
- Mix the as-prepared NiO particles with the GO dispersion prior to the hydrothermal process described in Strategy 2.
- During the hydrothermal reaction, the NiO particles become embedded within the forming 3D graphene network, creating a stable hybrid nanocomposite [31]. The NiO acts as an excellent catalyst for H₂O₂, allowing for sensitive "enzymeless" detection, which avoids the instability associated with enzyme-based biosensors.
The following diagram illustrates the logical relationship between the core challenges and the strategies designed to mitigate them.
The Scientist's Toolkit: Essential Research Reagents
The following table lists key materials and their functions for developing and testing CNT or graphene-based H₂O₂ sensors.
Table 3: Essential Reagents for Sensor Development
| Research Reagent / Material | Function in Sensor Development |
|---|---|
| Graphite Powder / Commercial CNTs | The starting material for synthesizing graphene oxide (GO) or for preparing CNT dispersions as the core sensing element [31] [64]. |
| Nickel Nitrate Hexahydrate (Ni(NO₃)₂·6H₂O) | A common precursor for synthesizing nickel oxide (NiO) nanocatalysts, which are used to enhance the electrocatalytic detection of H₂O₂ [31]. |
| Nafion | A perfluorosulfonated ionomer used as a dispersing agent for nanocarbons and as a permselective membrane to coat the electrode surface, reducing fouling and anion interference [1]. |
| EDC / NHS Crosslinkers | Carbodiimide crosslinkers used in covalent functionalization protocols to immobilize biomolecules or specific catalysts onto the carboxyl groups of oxidized CNTs or graphene [1]. |
| Phosphate Buffered Saline (PBS) | The standard electrolyte solution (e.g., 0.1 M, pH 7.4) for electrochemical testing and calibration, providing a stable ionic environment [31]. |
The choice between carbon nanotubes and graphene for plant H₂O₂ sensing is not a matter of declaring one universally superior. Instead, it hinges on the specific application requirements and the implementation of effective mitigation strategies against interference and drift. Carbon nanotubes excel with their high functionalization potential, which is key to engineering selectivity. In contrast, graphene, particularly in engineered 3D forms, offers immense surface area and superior electron transport, leading to very high sensitivity.
The experimental data indicates that hybrid approaches—such as combining 3D graphene with metal oxides or forming CNT-composite electrodes—consistently outperform either nanomaterial alone. These hybrids leverage synergistic effects to deliver enhanced sensitivity, stability, and selectivity. For researchers, the path forward involves a careful consideration of these material properties and a commitment to employing strategic functionalization and structural engineering to develop robust, reliable sensors capable of operating in the complex and dynamic environment of plant systems.
Head-to-Head Performance: Validating and Comparing CNT and Graphene Sensor Metrics
The detection of hydrogen peroxide (H2O2) is crucial in plant physiology research, where it acts as a key signaling molecule in stress responses, defense mechanisms, and cellular redox regulation. Electrochemical biosensors utilizing carbon nanomaterials have emerged as powerful tools for monitoring H2O2 due to their high sensitivity, rapid response, and compatibility with complex biological systems. Among these materials, carbon nanotubes (CNTs) and graphene represent two of the most prominent candidates, each offering distinct advantages and limitations for plant science applications [45] [66] [67].
This guide provides an objective performance comparison between CNT and graphene-based sensors, focusing on the key analytical metrics of sensitivity, detection limit, response time, and linear range. The comparative data and experimental protocols presented herein are designed to assist researchers in selecting the optimal nanomaterial platform for their specific plant H2O2 sensing requirements, framed within the broader thesis of evaluating the relative merits of these carbon allotropes for biological sensing applications.
Performance Metric Comparison
The table below summarizes the performance of various carbon nanomaterial-based sensors reported in recent literature, providing a direct comparison of key analytical metrics for H2O2 detection.
| Material Platform | Sensitivity (μA mM⁻¹ cm⁻²) | Detection Limit (μM) | Linear Range (mM) | Response Time (s) | Citation |
|---|---|---|---|---|---|
| CNT-based Sensors | |||||
| • Amine-functionalized CNTs | Not Specified | ~0.1 - 1 (for neurochemicals) | Not Specified | Not Specified | [45] |
| • CNT Forest Microelectrodes | Not Specified | 0.017 (for Dopamine) | Not Specified | Not Specified | [66] |
| Graphene-based Sensors | |||||
| • 3DGH/NiO25 Nanocomposite | 117.26 | 5.3 | 0.01 - 33.58 | Not Specified | [31] |
| • NiO Octahedron/3D Graphene | Demonstrated high | Low | Wide | Not Specified | [31] |
| Other Nanomaterial Sensors | |||||
| • Au@Pt Hairy Nanorods (Smooth) | Not Specified | 0.37 | 0.001 - 0.05 | <5 | [53] |
| • Au@Pt Hairy Nanorods (Hairy) | Nearly 2x Smooth NRs | 0.189 | 0.0005 - 0.05 | <5 | [53] |
- Key Findings: Graphene-based composites, particularly 3D structures combined with metal oxides like NiO, demonstrate superior sensitivity and a very wide linear range, making them suitable for detecting H2O2 across a broad concentration spectrum, from trace levels to millimolar amounts [31]. Core-shell nanorod sensors, while not pure carbon, achieve exceptional low-end detection limits and rapid response times (under 5 seconds), which is critical for capturing transient concentration changes in dynamic plant systems [53]. CNT-based sensors are well-established for biological sensing, showing excellent low detection limits for various analytes, though specific H2O2 performance data in the provided literature was less explicit [45] [66].
Experimental Protocols for Key Sensor Platforms
3D Graphene Hydrogel/NiO Octahedron Nanocomposite
Objective: To fabricate a highly sensitive, non-enzymatic H2O2 sensor with a wide linear detection range for applications in complex media [31].
Synthesis Protocol:
- NiO Octahedron Synthesis: Dissolve 10 mg of mesoporous silica (SBA-15) in 100 ml of ethanol containing 10 mg of nickel nitrate hexahydrate. Stir for 24 hours at room temperature. Dry the mixture at 80°C for 48 hours, grind the powder, and repeat the rinsing process. Calcinate the final product at 550°C for 3 hours (heating rate: 2°C min⁻¹). Remove the silica template by treating with 2 M NaOH at 60°C, followed by repeated washing with ethanol and water [31].
- 3DGH/NiO Nanocomposite Self-Assembly: Disperse 48 mg of graphene oxide (GO) in 32 mL of deionized water with a specific amount of NiO octahedrons (e.g., 12 mg for a 25% composite). Sonicate the mixture for 2 hours (bath) followed by 1.5 hours (probe). Transfer the solution to a 45 mL Teflon-lined autoclave and maintain at 180°C for 12 hours. After cooling, wash the resulting 3D graphene hydrogel (3DGH) and freeze-dry it [31].
- Electrode Modification and Measurement: Modify a glassy carbon electrode (GCE) with the 3DGH/NiO nanocomposite. Perform electrochemical detection of H2O2 using cyclic voltammetry (CV) and chronoamperometry (CA) in a standard three-electrode system with 0.1 M phosphate buffer solution (PBS, pH 7.4) as the electrolyte [31].
Au@Pt Core-Shell Nanorod Modified Electrode
Objective: To develop a sensor for the rapid and sensitive detection of low-concentration H2O2, simulating physiological conditions in plants [53].
Fabrication Protocol:
- Nanoparticle Synthesis: Fabricate Au@Pt core-shell nanorods (~40 nm in length) using a seed-mediated growth method with cetyltrimethylammonium bromide (CTAB) as a stabilizing agent. Precisely control the morphology to produce "Smooth" and "Hairy" (appendaged-surface) nanorods [53].
- Electrode Modification: Polish a 3 mm diameter glassy carbon working electrode with a diamond suspension. Drop-cast 5 μL of the Au@Pt nanorod solution onto the polished electrode surface and allow it to dry for 2 hours in the dark at room temperature [53].
- Electrochemical Detection: Conduct CV and CA measurements in a three-electrode setup with Ag/AgCl reference and Pt wire counter electrodes. Use a phosphate buffered saline (PBS, pH 7.4) solution to simulate physiological conditions. Evaluate sensor performance, including oxygen interference and stability in biologically relevant environments [53].
Sensing Mechanisms and Workflow
The following diagram illustrates the general experimental workflow for developing and evaluating nanomaterial-based H2O2 sensors, from material synthesis to performance validation.
Diagram 1: General workflow for the development and validation of a carbon nanomaterial-based H₂O₂ sensor.
The enhanced performance of CNT and graphene sensors stems from their distinct but complementary transduction mechanisms, depicted below.
Diagram 2: Fundamental sensing mechanisms of CNT and graphene platforms for H₂O₂ detection.
The Scientist's Toolkit: Essential Research Reagents
This table details key reagents and materials required for fabricating and testing carbon nanomaterial-based H₂O₂ sensors, as referenced in the experimental protocols.
| Reagent/Material | Function in Experiment | Example from Protocol |
|---|---|---|
| Graphene Oxide (GO) | Precursor for forming 3D conductive hydrogel scaffolds. | Starting material for 3DGH/NiO composite [31]. |
| Nickel Nitrate Hexahydrate | Metal oxide precursor for creating electrocatalytic NiO nanostructures. | Synthesis of NiO octahedrons [31]. |
| Mesoporous Silica (SBA-15) | "Hard template" for controlling the morphology of synthesized metal oxides. | Creating the octahedral structure of NiO [31]. |
| Gold(III) Chloride Trihydrate | Source of gold for forming the core of catalytic nanorods. | Synthesis of Au@Pt core-shell nanorods [53]. |
| Potassium Tetrachloroplatinate(II) | Source of platinum for forming the catalytically active shell on nanorods. | Synthesis of Au@Pt core-shell nanorods [53]. |
| Cetyltrimethylammonium Bromide (CTAB) | Surfactant and stabilizing agent to control nanoparticle growth and prevent aggregation. | Used in the fabrication of Au@Pt nanorods [53]. |
| Phosphate Buffered Saline (PBS) | Electrolyte solution that mimics physiological pH and ionic strength. | Standard medium for electrochemical testing (pH 7.4) [53] [31]. |
| Glassy Carbon Electrode (GCE) | Common, well-defined substrate/support for modifying with nanomaterials. | Working electrode platform for drop-casting nanomaterials [53] [31]. |
The benchmarking data indicates that graphene-based composites excel in applications demanding high sensitivity and a wide working range, such as monitoring prolonged oxidative stress in plants. In contrast, CNT-based platforms and advanced nanostructures like core-shell nanorods are advantageous for capturing rapid, low-concentration H2O2 fluctuations due to their fast response times and low detection limits [53] [31].
Future research should focus on enhancing the specificity of these sensors in the complex chemical environment of plant tissues and on integrating them into minimally invasive or implantable formats for real-time, in planta monitoring. The ongoing development and cost reduction of both CNT and graphene production will be pivotal in translating these sophisticated sensing platforms from laboratory research to broader applications in plant physiology and agricultural science [68] [69] [70].
The accurate detection of hydrogen peroxide (H₂O₂) in plant systems represents a critical capability in modern plant science, as this reactive oxygen species serves as a crucial signaling molecule in physiological processes and stress responses [45]. The emergence of nanotechnology has provided powerful new tools for probing plant signaling pathways, with carbon nanotubes (CNTs) and graphene standing out as two of the most promising carbon-based nanomaterials for advanced sensing applications [71] [72]. These materials offer unique advantages for monitoring plant metabolites, hormones, and signaling molecules with exceptional sensitivity [72]. This analysis provides a systematic comparison of carbon nanotubes and graphene specifically for plant H₂O₂ sensing research, examining their respective strengths, weaknesses, opportunities, and threats to guide researchers in selecting appropriate materials for their investigative needs. The evaluation encompasses fundamental material properties, sensing performance characteristics, practical implementation considerations, and future research directions that collectively inform their application in plant science studies.
Comparative Performance Analysis
Material Properties and Sensing Performance
Table 1: Comparative analysis of fundamental properties between carbon nanotubes and graphene for plant H₂O₂ sensing
| Property | Carbon Nanotubes (CNTs) | Graphene | Impact on Plant H₂O₂ Sensing |
|---|---|---|---|
| Structural Dimensionality | One-dimensional (1D) tubular structure [1] | Two-dimensional (2D) planar sheet [73] | CNTs provide confined charge transport pathways; graphene offers uniform surface adsorption |
| Electrical Conductivity | 10²–10⁵ S/m [1] | ~10⁴ S/m [1] | Both enable sensitive electrochemical detection; CNTs show advantage in electron transfer kinetics |
| Specific Surface Area | >1000 m²/g [1] | ~2630 m²/g [1] | Graphene provides superior analyte adsorption capacity; CNTs still offer ample active sites |
| Mechanical Strength | Exceptional (Young's modulus ~1 TPA) [1] | High (Young's modulus ~1 TPA) [1] | Both suitable for flexible sensor platforms; CNTs maintain structural integrity under stress |
| Functionalization Capability | Excellent (covalent and non-covalent) [17] [1] | Excellent [1] | Both enable selective H₂O₂ detection through surface modification with enzymes or recognition elements |
Table 2: Experimental sensing performance metrics for H₂O₂ detection
| Performance Metric | Carbon Nanotubes (CNTs) | Graphene | Experimental Context |
|---|---|---|---|
| Detection Limit | 0.43 µM (MWCNT/ChOx platform) [37] | Information Missing | Amperometric detection in buffer solution [37] |
| Sensitivity | 26.15 µA/mM (MWCNT/ChOx) [37] | Information Missing | Linear range from 0.4 to 4.0 mM H₂O₂ [37] |
| Linear Range | 0.4-4.0 mM [37] | Information Missing | Enzymatic platform with cholesterol oxidase [37] |
| Response Time | Fast (seconds to minutes) [45] | Fast (seconds to minutes) [45] | Electrochemical detection in nanomaterial-based sensors [45] |
| Selectivity Enhancement | Functionalization with enzymes (e.g., ChOx) [37] | Graphene oxide supporting material [45] | Enzyme integration improves specificity; GO enhances catalytic properties [45] [37] |
Experimental Protocols for Plant H₂O₂ Sensing
MWCNT-Cholesterol Oxidase Biosensing Platform
A highly effective biosensing platform for H₂O₂ detection has been demonstrated using multi-walled carbon nanotubes (MWCNTs) functionalized with cholesterol oxidase (ChOx) [37]. The experimental protocol involves several critical steps:
MWCNT Activation: MWCNTs (outer diameter: 6–13 nm, length: 2.5–20 μm, purity >98%) are activated through sequential treatment with 1 M nitric acid and 1 M sulfuric acid, each with 30-minute sonication cycles. This process is repeated twice to ensure proper functionalization, followed by extensive washing with ethanol and acetone until neutral pH is achieved [37].
Paste Electrode Preparation: Activated MWCNTs are mixed with mineral oil in a 70/30 w/w ratio to form a paste electrode (PMWCNT). The glassy carbon electrode surface is polished with 1 µm and 0.5 µm alumina slurry, rinsed with deionized water, sonicated, and dried with nitrogen gas before applying the PMWCNT [37].
Enzyme Immobilization: The biosensing platform is completed by drop-casting 10 μL of cholesterol oxidase solution (20 U/mL) onto the PMWCNT surface and allowing it to dry for 10 minutes at room temperature [37].
Electrochemical Characterization: The platform is characterized using cyclic voltammetry (−0.80 V to 0.20 V at 0.10 V/s scan rate) and electrochemical impedance spectroscopy in phosphate buffer (0.050 M, pH 7.4). H₂O₂ quantification is performed via amperometry in the range of 0.4–4.0 mM H₂O₂ [37].
This platform demonstrated a sensitivity of 26.15 µA/mM, with the presence of ChOx enhancing sensitivity by 21 times compared to non-enzymatic detection, highlighting the synergistic effect between CNTs and biological recognition elements [37].
Nanomaterial-Based Electrochemical Sensing Mechanisms
Both CNTs and graphene enable effective H₂O₂ detection through electrochemical mechanisms that can be adapted for plant studies [45]. The general experimental approach involves:
Electrode Modification: Nanomaterials are deposited on electrode surfaces (glassy carbon, gold, or screen-printed electrodes) to create a large surface area for H₂O₂ interaction and electron transfer.
Electrochemical Detection: Techniques include amperometry (applying constant potential and measuring current), cyclic voltammetry (scanning potential and measuring current), and electrochemical impedance spectroscopy (measuring impedance changes).
Signal Transduction: H₂O₂ oxidation or reduction at the nanomaterial-modified electrode produces measurable electrical signals proportional to concentration [45].
These mechanisms have been successfully employed for H₂O₂ detection in biological contexts, with potential adaptation for plant systems through appropriate functionalization to enhance selectivity in complex plant matrices [45].
SWOT Analysis
Carbon Nanotubes (CNTs) for Plant H₂O₂ Sensing
Table 3: SWOT Analysis - Carbon Nanotubes for Plant H₂O₂ Sensing
| Strengths | Weaknesses |
|---|---|
| • High aspect ratio and quantum effects enhance electron transfer for sensitive detection [1]• Proven enzymatic integration with oxidoreductases like cholesterol oxidase for specific H₂O₂ detection [37]• Ballistic charge transport enables rapid response times [17]• Large surface-to-volume ratio provides ample active sites [17] | • Potential cytotoxicity concerns in plant systems; SWCNTs can induce oxidative stress and H₂O₂ production in lettuce [74]• Poor dispersibility and aggregation tendencies require functionalization [17]• Batch-to-batch variability in synthesis affects reproducibility [1]• Non-specific responses to multiple analytes without functionalization [17] |
| Opportunities | Threats |
| • Functionalization strategies (covalent/non-covalent) to enhance selectivity for plant-specific applications [17]• Integration with AI technologies for intelligent sensing systems [73]• Wearable plant sensors for continuous monitoring [1]• Multi-walled CNT composites with polymers to control tunneling conduction [17] | • Regulatory concerns regarding nanomaterial persistence in food crops [74]• Competition from emerging materials like graphene quantum dots and MOFs [71]• Complexity in plant integration requiring specialized methodologies [72]• Signal interference from other plant metabolites [17] |
Graphene for Plant H₂O₂ Sensing
Table 4: SWOT Analysis - Graphene for Plant H₂O₂ Sensing
| Strengths | Weaknesses |
|---|---|
| • Exceptional electrical conductivity and high surface area (~2630 m²/g) for sensitive detection [1]• Tunable bandgap through functionalization for optimized sensing [44]• Flexible, transparent properties enable novel sensor form factors [73]• Graphene oxide serves as excellent supporting material with high conductivity [45] | • Zero-bandgap limitation in pristine graphene requires modification for semiconductor applications [44]• Potential plant toxicity though less documented than CNTs [74]• Complex transfer processes to appropriate substrates [44]• Limited enzyme integration studies specifically for H₂O₂ detection compared to CNTs [37] |
| Opportunities | Threats |
| • Doping with heteroatoms (N, S) to tune electronic properties for plant sensing [71]• Composite structures with CNTs or metals for synergistic effects [71]• Sustainable production from biomass sources [71]• Integration with flexible electronics for plant wearables [73] | • High production costs for quality-controlled material [72]• Technical complexity in large-scale, defect-free synthesis [44]• Competition from established CNT platforms with proven track records [37]• Limited standardization in plant sensing applications [72] |
Visualization of Sensing Mechanisms and Experimental Workflows
CNT-Based Electrochemical Sensing Mechanism
Experimental Workflow for Plant H₂O₂ Sensor Development
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 5: Essential research reagents and materials for CNT and graphene-based plant H₂O₂ sensing
| Material/Reagent | Function in Research | Example Specifications | Application Notes |
|---|---|---|---|
| Multi-walled Carbon Nanotubes (MWCNTs) | Primary sensing material; electron transfer mediator | Outer diameter: 6–13 nm; Length: 2.5–20 μm; Purity >98% [37] | Require acid activation for functionalization |
| Single-walled Carbon Nanotubes (SWCNTs) | High-sensitivity sensing; FET-based detection | Semiconductor purity >99.9%; Specific chirality for optical sensing [17] | Potential phytotoxicity concerns [74] |
| Graphene Oxide (GO) | Supporting material; enhances catalytic properties | High conductivity; large surface area [45] | Can be functionalized with metal nanoparticles |
| Cholesterol Oxidase (ChOx) | Biological recognition element for H₂O₂ | Microbial source; lyophilized powder; 20 U/mL working concentration [37] | Provides specificity through enzymatic recognition |
| Phosphate Buffer (PB) | Electrochemical cell electrolyte | 0.050 M concentration; pH 7.4 [37] | Maintains physiological pH for biological components |
| Mineral Oil | Binder for paste electrodes | Analytical grade; forms 70/30 w/w mixture with MWCNTs [37] | Creates stable paste electrode matrix |
| Nitric Acid & Sulfuric Acid | MWCNT activation and functionalization | 1 M concentration for activation [37] | Creates functional groups for biomolecule attachment |
| Electrochemical Cell Components | Signal measurement and transduction | Three-electrode system: working, reference, counter electrodes [37] | Standard setup for electrochemical detection |
The systematic comparison of carbon nanotubes and graphene for plant H₂O₂ sensing reveals a complex landscape where material selection depends heavily on specific research requirements. Carbon nanotubes demonstrate particular strengths in enzymatic integration and established sensing protocols, with proven performance in H₂O₂ detection systems [37]. Their one-dimensional structure facilitates efficient electron transfer, while functionalization strategies enable enhanced selectivity [17] [1]. However, concerns regarding potential phytotoxicity and batch-to-batch reproducibility present significant challenges for plant research applications [74].
Graphene and its derivatives offer exceptional surface properties and tunable electronic characteristics through doping and functionalization [71]. The two-dimensional structure provides uniform analyte interaction, while flexible transparent properties enable innovative sensor designs [73]. Current limitations include less extensive validation with enzymatic systems specifically for H₂O₂ detection and higher production costs for quality-controlled material [72].
Future research directions should focus on developing standardized protocols for plant integration, addressing nanomaterial toxicity concerns, and exploring hybrid approaches that leverage the complementary advantages of both materials [71] [72]. The integration of artificial intelligence with nanomaterial-based sensors presents a promising avenue for advanced data analysis and intelligent sensing systems in plant science research [73]. As both technologies continue to mature, they offer powerful tools for unraveling the complex roles of H₂O₂ in plant signaling pathways and stress responses, potentially transforming our understanding of plant physiology.
The accurate detection of hydrogen peroxide (H₂O₂) within plant systems represents a critical challenge for plant physiologists and stress biologists. As a key signaling molecule and stress indicator, H₂O₂ participates in numerous physiological processes, yet its quantification is complicated by the complex chemical environments of plant matrices such as apoplastic fluid and xylem sap. These fluids contain diverse metabolites, proteins, and ions that can interfere with conventional detection methods. The emergence of carbon-based nanomaterials has opened new possibilities for overcoming these limitations, with carbon nanotubes (CNTs) and graphene-based sensors leading this innovation. This review provides a systematic comparison of CNT and graphene-based electrochemical sensors for H₂O₂ detection in plant-relevant matrices, evaluating their performance against stringent criteria required for plant science research.
Performance Comparison: Carbon Nanotubes vs. Graphene-Based Sensors
The following tables summarize the comparative performance characteristics of CNT and graphene-based sensors, with emphasis on their applicability for plant matrix analysis.
Table 1: Overall Performance Metrics for H₂O₂ Sensing in Complex Matrices
| Performance Parameter | Carbon Nanotube-Based Sensors | Graphene-Based Sensors |
|---|---|---|
| Typical Linear Range (μM) | 5–6000 [75] | 0.8–500 (Prussian Blue composite) [7] |
| Limit of Detection (μM) | 1.4 [75] | 0.25 [7] |
| Sensitivity | High (synergistic enhancement with metal NPs) [75] | Moderate to High (dependent on functionalization) [7] |
| Selectivity in Complex Matrices | Excellent (core-shell structures reduce interference) [75] | Good (can be impaired by non-specific adsorption) [7] |
| Stability in Aqueous Environments | Good to Excellent [75] [17] | Moderate (can vary with substrate) [44] |
| Electrical Conductivity | Excellent (ballistic charge transport) [17] | Very Good (high carrier mobility) [44] |
| Ease of Functionalization | High (versatile covalent and non-covalent approaches) [17] | Moderate (depends on graphene derivative) [44] |
Table 2: Suitability for Plant Matrix Applications
| Characteristic | Carbon Nanotube-Based Sensors | Graphene-Based Sensors |
|---|---|---|
| Tolerance to Plant Metabolites | High (with proper electrode modification) [75] | Moderate (can be affected by phenolic compounds) [7] |
| Performance in Low-pH Apoplast | Good (maintains function at pH 5.2-7.3) [7] | Variable (pH-dependent electron transfer) [7] |
| Compatibility with Extraction Methods | Excellent (works with small sample volumes) [76] [75] | Good (requires optimization for ionic strength) [76] |
| Reproducibility | Good to Excellent (with controlled fabrication) [75] [17] | Moderate (batch-to-batch variation in graphene quality) [44] |
| Potential for Miniaturization | High (compatible with microelectrode designs) [17] | High (2D structure suits planar designs) [44] |
Experimental Protocols for Plant Matrix Validation
Apoplastic Fluid Extraction Methodology
The infiltration-centrifugation technique represents the most widely adopted approach for extracting apoplastic washing fluid (AWF) from plant tissues suitable for sensor validation [76] [77]. The following protocol has been optimized for compatibility with electrochemical sensing:
Plant Material Preparation: Collect fully expanded leaves from tobacco (Nicotiana tabacum), Arabidopsis (Arabidopsis thaliana), or poplar (Populus spp.) plants grown under controlled conditions [76] [77].
Infiltration Process:
- Cut leaf discs (typically 1 cm diameter) avoiding major veins
- Submerge discs in infiltration solution (distilled deionized water or 20% methanol)
- Apply vacuum (0.5-1 bar) for 2-5 minutes until tissues appear water-soaked
- Release vacuum slowly and gently blot excess surface liquid [76]
Centrifugation Extraction:
Contamination Assessment:
- Monitor malate dehydrogenase activity as cytosolic marker
- Measure ion leakage to assess membrane integrity
- Analyze phosphorylated hexoses as symplastic contamination indicators [76]
This method typically yields 5-50 μL AWF per 100 mg leaf tissue, with protein concentrations of 0.1-0.5 μg/μL, sufficient for sensor validation studies [76] [77].
Sensor Fabrication and Validation Protocols
Table 3: Core-Shell CNT Sensor Fabrication [75]
| Step | Procedure | Purpose |
|---|---|---|
| 1. CNT Preparation | Functionalize MWCNTs with acid treatment | Introduce surface groups for subsequent binding |
| 2. Core-Shell Synthesis | Precipitate Au@TiO₂ onto CNT surfaces via SDS-capping and calcination (500°C, 4h) | Create synergistic nanostructure with enhanced electrocatalytic properties |
| 3. Electrode Modification | Deposit CNT nanocomposite onto GCE surface (drop-casting or electrodeposition) | Create uniform sensing interface with high surface area |
| 4. Sensor Characterization | CV, EIS, and DPV in standard solutions | Verify electrode performance and establish baseline metrics |
Table 4: Plant Matrix Validation Workflow
| Validation Step | Procedure | Acceptance Criteria |
|---|---|---|
| 1. Standard Curve | DPV measurements in synthetic buffer (pH 5.5) | R² > 0.995 across 1-1000 μM H₂O₂ range |
| 2. Spike Recovery | Add known H₂O₂ concentrations to AWF samples | 85-115% recovery with RSD < 10% |
| 3. Interference Testing | Expose to common apoplastic compounds (sugars, organic acids, ions) | Signal change < 5% for relevant concentrations |
| 4. Long-term Stability | Repeated measurements in AWF over 8h | Sensitivity retention > 90% |
Sensing Mechanisms and Material Properties
Fundamental Sensing Mechanisms
The superior performance of carbon nanomaterial-based sensors stems from their distinct structural and electronic properties. Carbon nanotubes (CNTs) exhibit a unique one-dimensional hollow structure that functions as an excellent charge transporter, while graphene provides a two-dimensional surface with exceptional surface area for catalytic interactions [78]. In CNT-based sensors, the sensing mechanism primarily occurs through charge transfer processes where H₂O₂ molecules interact with the CNT surface or with catalytic nanoparticles (such as Au@TiO₂) deposited on the CNT surface [75]. This interaction modulates the electrical conductance of the CNT network, which can be precisely measured electrochemically.
Graphene-based sensors operate on similar principles but leverage the two-dimensional planar structure which provides abundant active sites for H₂O₂ electrocatalysis [44]. When functionalized with Prussian blue (an "artificial peroxidase"), graphene sensors can catalyze H₂O₂ reduction at low voltages (close to 0 V), minimizing interference from other electroactive species commonly found in plant matrices [7]. The large specific surface area of graphene nanoplatelets enhances signal amplification while the sp² hybridized carbon network facilitates rapid electron transfer kinetics [79].
Nanomaterial Synergies in Hybrid Approaches
Recent advances demonstrate that hybrid approaches combining CNTs and graphene can leverage the complementary advantages of both materials [78] [79]. In these architectures, CNTs create bridging networks between graphene sheets, enhancing charge transport while preventing restacking of graphene layers. This synergistic interaction creates hierarchical networks with enhanced electrical conductivity and more abundant catalytic sites [78]. Experimental studies confirm that CNT-graphene hybrids exhibit lower percolation thresholds and higher electrical conductivities compared to single-component systems [78]. For plant H₂O₂ sensing, this translates to improved sensitivity and lower detection limits in complex apoplastic fluids.
CNT vs Graphene Sensing Mechanisms
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 5: Essential Materials for Plant H₂O₂ Sensor Development
| Material/Reagent | Function/Purpose | Example Specifications |
|---|---|---|
| Multi-Walled Carbon Nanotubes (MWCNTs) | Conductive backbone for electrode modification; provides high surface area and electron transfer pathways [75] | Purity >95%, length 1-5 μm, diameter 9.5 nm [75] |
| Gold Chloride (HAuCl₄·H₂O) | Precursor for gold nanoparticle synthesis; enhances catalytic activity toward H₂O₂ reduction [75] | ≥99.9% trace metals basis [75] |
| Titanium(IV) Tetraisopropoxide (TTIP) | Titanium source for TiO₂ shell formation; creates core-shell structures with improved stability [75] | 98% purity [75] |
| Prussian Blue (Fe₄[Fe(CN)₆]₃) | "Artificial peroxidase" catalyst; enables H₂O₂ reduction at low potentials minimizing interference [7] | Electrochemical grade [7] |
| Glassy Carbon Electrodes (GCE) | Conductive substrate for sensor fabrication; provides defined surface for nanomaterial deposition [7] [75] | 3 mm diameter, polished to mirror finish [75] |
| Sodium Dodecyl Sulfate (SDS) | Surfactant for nanomaterial dispersion; prevents aggregation during sensor fabrication [75] | ≥99.0% purity [75] |
| Phosphate Buffered Saline (PBS) | Electrolyte solution for electrochemical measurements; provides controlled ionic environment [75] | 0.1 M, pH 7.4 [75] |
| Infiltration Solution | Medium for apoplastic fluid extraction; must not interfere with subsequent H₂O₂ detection [76] | Distilled deionized water or 20% methanol [76] |
Plant H₂O₂ Sensor Validation Workflow
The validation of H₂O₂ sensors in complex plant matrices represents a significant advancement in plant stress physiology research. Based on current evidence, carbon nanotube-based sensors, particularly those employing core-shell nanostructures, demonstrate superior performance in apoplastic fluids and plant sap due to their enhanced selectivity, stability, and tolerance to plant metabolites. Graphene-based sensors offer complementary advantages in specific applications requiring ultra-low detection limits. The emerging trend toward hybrid CNT-graphene architectures promises to further enhance sensor capabilities by leveraging the synergistic effects between these remarkable carbon nanomaterials. As these technologies mature, they will undoubtedly provide plant scientists with unprecedented insights into the spatial and temporal dynamics of H₂O₂ signaling in plant stress responses and developmental processes.
Conclusion
The development of nanomaterial-based sensors is revolutionizing our ability to decipher plant stress signaling in real time. Both carbon nanotubes and graphene offer distinct advantages; CNTs excel in applications requiring high-aspect-ratio and versatile functionalization for optical multiplexing, while graphene, particularly in its laser-induced form, provides exceptional platforms for miniaturized, enzymatic electrochemical biosensors with direct electron transfer. Future directions point toward the creation of hybrid nanomaterial systems that leverage the strengths of both allotropes, the expansion of multiplexed sensor arrays to decode complex stress hormone crosstalk, and the integration of these sensors into automated, wireless platforms for scalable precision agriculture. Overcoming challenges in reproducibility and selectivity will be paramount for translating these powerful laboratory tools into reliable field-deployable diagnostics, ultimately enabling early stress intervention and the development of more resilient crops.
References
- 1. Unlocking the Future: Carbon Nanotubes as Pioneers in ... [https://www.mdpi.com/2227-9040/13/7/225]
- 2. Carbon nanotubes and graphene - properties, applications ... [https://www.graphene-info.com/carbon-nanotubes]
- 3. Innovations in graphene-based electrochemical biosensors in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11982133/]
- 4. Detection of Hydrogen Peroxide Using Hemin-PEI/MWCNT ... [https://www.sciencedirect.com/science/article/abs/pii/S0013468625020997]
- 5. Hydrogen Peroxide and Dopamine Sensors Based on ... [https://www.mdpi.com/1424-8220/24/2/355]
- 6. Ultra-sensitive H2O2 sensing with 3-D porous Au/CuO/Pt ... [https://www.sciencedirect.com/science/article/pii/S0925400523012273?dgcid=rss_sd_all]
- 7. Nanostructures in Hydrogen Peroxide Sensing [https://www.mdpi.com/1424-8220/21/6/2204]
- 8. Carbon nanotubes and nanofibers as building blocks for ... [https://www.sciencedirect.com/science/article/pii/S0921510725006464]
- 9. Hydrogen Peroxide Signaling in the Maintenance of Plant ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11118955/]
- 10. Hydrogen Peroxide Signaling in Plant Development and ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2016.00230/full]
- 11. Hydrogen peroxide as a signalling molecule in plants and ... [https://www.sciencedirect.com/science/article/abs/pii/S0045653520306792]
- 12. Carbon Nanotubes, Graphene, and Carbon Dots as ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8587008/]
- 13. Detection of Hydrogen Peroxide by DAB Staining in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4932902/]
- 14. Measurement of foliar H2O2 concentration can be an ... [https://www.nature.com/articles/s41598-022-17658-2]
- 15. Hydrogen peroxide homeostasis: Activation of plant catalase ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC122654/]
- 16. Advancements in nanomaterials for nanosensors [https://pubs.rsc.org/en/content/articlehtml/2024/na/d4na00214h]
- 17. Carbon Nanotube-Based Chemical Sensors: Sensing ... [https://www.mdpi.com/2227-9040/13/10/367]
- 18. An Elucidation of Substrate Effects in Graphene-Based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12481550/]
- 19. An Amperometric Hydrogen Peroxide Sensor Based on ... [https://www.sciencedirect.com/science/article/pii/S145239812302117X]
- 20. Highly Sensitive Detection of Hydrogen Peroxide in Cancer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11201644/]
- 21. A Biosensor Based on Immobilization of Horseradish ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3933900/]
- 22. A high-power hybrid carbon nanotube/three-dimensional ... [https://www.sciencedirect.com/science/article/abs/pii/S001346862401291X]
- 23. Nanostructured porous graphene and its composites for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5661008/]
- 24. Activity of catalase adsorbed to carbon nanotubes [https://www.sciencedirect.com/science/article/abs/pii/S0039914013001768]
- 25. A comparison study between CNT and graphene [https://www.sciencedirect.com/science/article/abs/pii/S0079670016301204]
- 26. Difference between carbon nanotubes and graphene [https://www.layeronematerials.com/insights/difference-between-nanotubes-and-graphene]
- 27. Enzyme Immobilization on Nanomaterials and Their ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12430663/]
- 28. Graphene enhances artemisinin production in the ... [https://www.sciencedirect.com/science/article/pii/S2590346223002882]
- 29. Multi-walled carbon nanotubes improved growth, anatomy ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6800878/]
- 30. Graphene promotes the growth of Vigna angularis by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10919591/]
- 31. Enzymeless electrochemical detection of hydrogen ... [https://www.nature.com/articles/s41598-025-10472-6]
- 32. Enhancement and optimization of a graphene-based ... [https://www.nature.com/articles/s41598-025-20570-0]
- 33. MXene-Enhanced Laser-Induced Graphene Flexible Sensor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12114579/]
- 34. Laser-induced graphene for biomedical applications [https://pubs.rsc.org/en/content/articlehtml/2025/nh/d5nh00377f?page=search]
- 35. Atomistic and data-driven modeling of laser-induced ... [https://www.nature.com/articles/s41598-025-15945-2]
- 36. Designing high-performance asymmetric and hybrid ... [https://www.nature.com/articles/s41598-024-79622-6]
- 37. Experimental and In Silico Studies on the Development of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12109943/]
- 38. A Review on Laser-Induced Graphene-Based Electrocatalysts ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12299054/]
- 39. New graphene-based wearable plant sensor can be ... [https://www.graphene-info.com/new-graphene-based-wearable-plant-sensor-can-be-fabricated-paper]
- 40. Batch-to-Batch Variation in Laser-Inscribed Graphene (LIG) ... [https://www.mdpi.com/2072-666X/15/7/874]
- 41. Decoding early stress signaling waves in living plants ... [https://www.nature.com/articles/s41467-024-47082-1]
- 42. A microneedle patch senses plant stress - C&EN [https://cen.acs.org/analytical-chemistry/chemical-sensing/microneedle-patch-senses-plant-stress/103/web/2025/04]
- 43. A 3D-printed hollow microneedle-based electrochemical ... [https://www.sciencedirect.com/science/article/abs/pii/S0956566324001362]
- 44. The State Of Carbon Science In 2025: Graphene ... [https://briandcolwell.com/the-state-of-carbon-science-in-2025-graphene-nanotubes-quantum-dots-and-more/]
- 45. Recent progress in nanomaterial-based electrochemical ... [https://www.sciencedirect.com/science/article/abs/pii/S0039914024018290]
- 46. Assessment of Salt Stress to Arabidopsis Based on the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9604455/]
- 47. A novel flexible electrochemical sensor with multi-level ... [https://www.sciencedirect.com/science/article/abs/pii/S0026265X24006088]
- 48. Non-Covalent Functionalization of Carbon Nanotubes for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6358788/]
- 49. Non-covalent functionalization of CVD-grown graphene ... [https://link.springer.com/article/10.1007/s40097-016-0201-6]
- 50. A Comparative Study of Carbon Nanotubes and Graphene ... [https://www.sciencedirect.com/science/article/abs/pii/S0042207X25004191]
- 51. Carbon Nanotubes 2025-2035: Market, Technology & ... [https://www.idtechex.com/en/research-report/carbon-nanotubes-2025/1099]
- 52. Electrochemical Sensing of H2O2 by Employing a Flexible ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10096334/]
- 53. Various Configurations of Au@Pt Nanostructures on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12322878/]
- 54. Advances in carbon nanotube-based gas sensors [https://www.sciencedirect.com/science/article/abs/pii/S0010854524003953]
- 55. CuO-embedded laser-induced graphene microfluidic ... [https://www.sciencedirect.com/science/article/abs/pii/S0009250925006621]
- 56. An Overview of Carbon and Nanotubes for Biosensing... Graphene [https://link.springer.com/article/10.1007/s40820-017-0128-6]
- 57. Recent Advances in the Direct Electron Transfer-Enabled ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7902792/]
- 58. Frontiers | Recent Advances in the Direct -Enabled... Electron Transfer [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2020.620153/full]
- 59. Highly Sensitive Detection of Hydrogen Peroxide in Cancer ... [https://www.mdpi.com/2079-6374/14/6/261]
- 60. Investigation of Direct of Glucose Oxidase on... Electron Transfer [https://pmc.ncbi.nlm.nih.gov/articles/PMC11243339/]
- 61. Advanced strategies for enzyme–electrode interfacing in ... [https://www.sciencedirect.com/science/article/pii/S0167779924003445]
- 62. and Carbon towards soft electronics - PMC nanotubes graphene [https://pmc.ncbi.nlm.nih.gov/articles/PMC5591626/]
- 63. Next-Generation Chemical Sensors: The Convergence of ... [https://www.preprints.org/manuscript/202508.0582/v1]
- 64. A high- performance , disposable screen-printed carbon electrode... [https://link.springer.com/article/10.1007/s10800-018-1268-1]
- 65. of pure and hybridized Sensing ... performances carbon [https://www.nature.com/articles/s41598-017-14544-0?error=cookies_not_supported&code=c9e0fabf-934c-444c-8d7c-83fec75e003b]
- 66. Recent trends in carbon nanomaterial-based electrochemical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4557208/]
- 67. Solid State Sensors for Hydrogen Peroxide Detection [https://www.mdpi.com/2079-6374/11/1/9]
- 68. The Growing Graphene Market and the Shift from Carbon ... [https://www.idtechex.com/en/research-article/the-growing-graphene-market-and-the-shift-from-carbon-nanotubes/33066]
- 69. Carbon Nanotubes Market Size & Share Analysis [https://www.mordorintelligence.com/industry-reports/carbon-nanotubes-market]
- 70. Carbon Nanotubes Market Outlook 2025 to 2035 [https://www.futuremarketinsights.com/reports/global-carbon-nanotubes-market]
- 71. New carbon materials offer eco-friendly solutions for water ... [https://scientificinquirer.com/2025/11/24/new-carbon-materials-offer-eco-friendly-solutions-for-water-pollution-energy-storage-and-green-chemistry/]
- 72. Nanosensor Applications in Plant Science - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9496508/]
- 73. Recent progress in low dimensional carbon nanomaterials ... [https://www.aimspress.com/article/doi/10.3934/matersci.2025048?viewType=HTML]
- 74. Stress impacts of different types of micro- and nanomaterials ... [https://pubs.rsc.org/en/content/articlehtml/2025/en/d5en00237k]
- 75. A Core–Shell Au@TiO2 and Multi-Walled Carbon ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9599508/]
- 76. Toward the apoplast metabolome: Establishing a leaf ... [https://www.sciencedirect.com/science/article/abs/pii/S0981942824007484]
- 77. Apoplast proteome reveals that extracellular matrix contributes ... [https://bmcgenomics.biomedcentral.com/articles/10.1186/1471-2164-11-674]
- 78. Numerical investigation of synergistic enhancement ... [https://www.sciencedirect.com/science/article/abs/pii/S0266353825003835]
- 79. Hybrid strategy of graphene/carbon nanotube hierarchical ... [https://www.nature.com/articles/s41598-021-00307-5]