Building Robust Multimodal Plant Datasets: A Comprehensive Guide to Data Preprocessing Pipelines for Agricultural AI and Drug Discovery
This article provides a comprehensive guide for researchers and scientists on constructing effective data preprocessing pipelines for multimodal plant datasets.
Building Robust Multimodal Plant Datasets: A Comprehensive Guide to Data Preprocessing Pipelines for Agricultural AI and Drug Discovery
Abstract
This article provides a comprehensive guide for researchers and scientists on constructing effective data preprocessing pipelines for multimodal plant datasets. It explores the foundational principles of plant multimodality, detailing methodological steps for integrating diverse data types such as images of different plant organs, textual descriptions, and environmental data. The content addresses critical challenges including data heterogeneity, missing modalities, and label noise, offering practical troubleshooting and optimization strategies. Furthermore, it outlines robust validation and comparative analysis frameworks to benchmark pipeline performance, emphasizing the pipeline's pivotal role in enhancing the accuracy and reliability of downstream applications in plant phenotyping, disease diagnosis, and drug discovery.
The Why and What: Understanding the Imperative for Multimodal Plant Data
Frequently Asked Questions (FAQs)
Q1: Why is analyzing multiple plant organs (multimodal) better than just using leaves for classification? Relying on a single organ, like a leaf, is often biologically insufficient for accurate classification. The same plant species can show different appearances, while different species can share similar features in a single organ. Using images from multiple organs—such as flowers, leaves, fruits, and stems—provides complementary biological data, leading to a more comprehensive and accurate representation of the plant species [1].
Q2: What is a key technical challenge when working with multimodal plant data, and how can it be addressed? A primary challenge is determining the optimal strategy for fusing data from different modalities (organs). Simple methods like late fusion (averaging predictions from single-organ models) can be suboptimal. An automated fusion approach using a Multimodal Fusion Architecture Search (MFAS) can discover more effective fusion strategies, leading to significantly higher accuracy compared to manual methods [1].
Q3: How can I make my multimodal model robust to missing data, for example, if fruit images are not available for a particular sample? You can incorporate multimodal dropout techniques during training. This approach teaches the model to perform classification effectively even when one or more input modalities (e.g., fruits or stems) are missing, making it more practical for real-world applications where data for all plant organs may not be available [1].
Q4: My dataset was designed for single-organ analysis. How can I adapt it for multimodal research? You can create a multimodal dataset through a dedicated preprocessing pipeline. This involves restructuring an existing unimodal dataset. For instance, the Multimodal-PlantCLEF dataset was created from PlantCLEF2015 by grouping images of different organs (flowers, leaves, fruits, stems) from the same plant species into a single, multi-input sample [1].
Q5: What does single-cell analysis reveal that bulk tissue analysis cannot? Single-cell multi-omics can uncover that the biosynthesis of complex plant compounds (like the anti-cancer alkaloids vinblastine and vincristine) is organized across distinct, rare cell types. Bulk analysis dilutes these specific signals. Single-cell analysis allows researchers to discover new biosynthetic genes and understand that pathway intermediates accumulate at very high concentrations in specific, specialized cells [2].
Troubleshooting Guides
Issue 1: Poor Model Performance on Multimodal Plant Data
Problem: Your model's classification accuracy is low, potentially underperforming simpler, single-organ models.
Diagnosis & Solutions:
| Possible Cause | Diagnostic Check | Recommended Solution |
|---|---|---|
| Suboptimal Fusion Strategy | Check if you are using a simplistic fusion method (e.g., late fusion). | Implement an automated fusion search (e.g., Multimodal Fusion Architecture Search) to find a more effective integration method [1]. |
| Misalignment of Features | Examine feature vectors from different organ models for scale and semantic misalignment. | Introduce a feature alignment layer in your pipeline before fusion to project features into a common space [3]. |
| Missing Modalities | Evaluate if your model fails when an organ image is missing. | Use multimodal dropout during training to improve model robustness to incomplete data [1]. |
Issue 2: Challenges in Preprocessing and Pipelines for Heterogeneous Data
Problem: Managing and processing different data types (e.g., 3D organ images, text annotations, single-cell data) is complex and inefficient.
Diagnosis & Solutions:
| Possible Cause | Diagnostic Check | Recommended Solution |
|---|---|---|
| Non-Standardized Workflow | Check if processing steps for each data type are manual and disjointed. | Implement a graph-based, modular preprocessing pipeline (e.g., using a framework like Pliers) to standardize and chain operations [3]. |
| Difficulty in Cell-Type Identification | For single-cell analysis, check if you rely on manual annotation or transgenic markers. | Use a computational tool like 3DCellAtlas, which leverages the intrinsic geometric properties of cells for accurate, automated identification without needing reference atlases [4]. |
Experimental Protocols
Protocol 1: Constructing a Multimodal Plant Classification Model with Automated Fusion
Objective: To build a high-accuracy plant classification model that automatically fuses data from images of four plant organs (flower, leaf, fruit, stem).
Methodology:
- Dataset Preparation: Use a multimodal dataset like Multimodal-PlantCLEF. Ensure each data sample consists of image sets (flower, leaf, fruit, stem) for a single plant species [1].
- Unimodal Model Training: Train a separate Convolutional Neural Network (CNN), such as a pre-trained MobileNetV3Small, on each individual organ modality [1].
- Automated Fusion: Apply a Multimodal Fusion Architecture Search (MFAS) algorithm. This algorithm will automatically discover the optimal points and methods to combine the features extracted from the four unimodal models [1].
- Robustness Training: Incorporate multimodal dropout during the training of the fused model to enhance its performance when some organ images are missing [1].
Expected Outcome: A multimodal model that achieves higher classification accuracy (e.g., 82.61% on 979 classes) compared to a late-fusion baseline, with robustness to missing data [1].
Protocol 2: Single-Cell Multi-Omics Analysis for Plant Metabolic Pathway Elucidation
Objective: To map the cell-type-specific biosynthesis of a target plant natural product (e.g., vinblastine in Catharanthus roseus).
Methodology:
- Sample Preparation & Imaging: Prepare thin sections of the plant organ of interest (e.g., leaf). Use confocal microscopy to acquire 3D image z-stacks of the tissue [4].
- 3D Cell Segmentation & Identification: Process the 3D images with software like MorphoGraphX. Use the 3DCellAtlas pipeline to segment individual cells and identify distinct cell types (e.g., epidermis, idioblasts) based on their intrinsic 3D geometry [4].
- Single-Cell Omics: Isolate or profile the contents of the identified individual cell types. Perform single-cell RNA sequencing (scRNA-seq) to measure gene expression and metabolomics to measure metabolite levels in each cell type [2].
- Data Integration: Correlate the expression of biosynthetic genes with the accumulation of pathway intermediates across the different cell types. This integration reveals the complete, spatially organized metabolic pathway [2].
Expected Outcome: Identification of which specific cell types express different steps of a biosynthetic pathway and where key intermediates accumulate, leading to the discovery of new pathway genes [2].
Table 1: Performance Comparison of Plant Classification Models
| Model Type | Fusion Strategy | Number of Classes | Top-1 Accuracy | Key Advantage |
|---|---|---|---|---|
| Multimodal (4 organs) | Automated Fusion Search | 979 | 82.61% [1] | Optimal architecture discovery |
| Multimodal (4 organs) | Late Fusion (Averaging) | 979 | 72.28% [1] | Simplicity |
| Unimodal (Single organ) | N/A | 979 | (Lower than multimodal) | Reduces need for multiple images |
Table 2: Cell-Type-Specific Accumulation in Catharanthus roseus Alkaloid Pathway
| Cell Type | Role in Vinblastine Biosynthesis | Key Observation |
|---|---|---|
| IPAP cells (Specialized vascular) | Express the first stage of the pathway [2] | Confines initial steps to specific cells. |
| Epidermis | Express the second stage of the pathway [2] | Middle steps occur in a separate tissue layer. |
| Idioblasts (Rare leaf cells) | Express the final stages; site of precursor accumulation (catharanthine & vindoline) [2] | Precursors concentrated 1000x higher than in whole-leaf extract [2]. |
Research Reagent Solutions
Table 3: Essential Tools and Reagents for Advanced Plant Analysis
| Item | Function in Research | Application Context |
|---|---|---|
| 3DCellAtlas | A computational pipeline for semiautomated identification of cell types and quantification of 3D cellular anisotropy from 3D image data [4]. | Single-cell analysis of radially symmetric plant organs (roots, hypocotyls). |
| Multimodal Preprocessing Pipeline (e.g., Pliers) | A structured workflow to extract, transform, and align features from heterogeneous data (video, audio, images, text) into a standardized format [3]. | Building unified datasets from multiple sources for multimodal machine learning. |
| Confocal Microscopy Z-Stacks | High-resolution 3D imaging of plant tissues and cellular structures [4]. | Essential for accurate 3D segmentation and analysis of cell shape and size. |
| Single-cell RNA Sequencing (scRNA-seq) | Profiling the complete set of RNA transcripts in individual cells [2]. | Identifying gene expression patterns specific to rare or specialized cell types. |
Workflow and Pathway Visualizations
Multimodal Plant Analysis Pipeline
Cell-Type-Specific Alkaloid Biosynthesis
Frequently Asked Questions
Q1: What exactly is considered a "modality" in plant science research? A modality refers to a distinct type or source of data that provides unique information about a plant. In multimodal learning, these diverse data sources are integrated to provide a comprehensive representation, leveraging their complementary nature [1]. Common modalities in plant science include:
- Images: RGB images of plant organs (leaves, flowers, fruits, stems) [1], as well as data from multispectral, hyperspectral, and thermal sensors [5] [6].
- Climate & Weather Data: Historical and temporal data on temperature, precipitation, humidity, and solar radiation [5].
- Phenotypic & Tabular Data: Manually or automatically measured traits, such as plant density, height, date to anthesis, and parental line information [5].
- Molecular Data: Genomic sequences and genetic data, which can be analyzed with advanced tools like large language models (e.g., Agronomic Nucleotide Transformer or AgroNT) to uncover regulatory patterns [6].
- Text: Scientific literature, taxonomic descriptions, and curated knowledge from public databases [7].
Q2: Why should I use a multimodal approach instead of relying on a single data type? A single data source, such as an image of a leaf, is often insufficient for accurate classification or prediction as it cannot capture the full biological diversity of a plant species [1]. Multimodal deep learning models integrate complementary information from different sources, leading to significantly improved predictive power and explanatory capabilities compared to single-modality models [5]. For example, fusing images with weather data allows a model to understand the impact of meteorological events on maize growth, which images alone cannot capture [5].
Q3: What are the main strategies for fusing different data modalities? The main fusion strategies are early, intermediate (or feature-level), late (decision-level), and hybrid fusion [1]. The choice of fusion strategy is a critical challenge, and the optimal point for modality fusion can even be discovered automatically using algorithms like the multimodal fusion architecture search (MFAS) [1].
Q4: I have a unimodal dataset. Can I adapt it for multimodal research? Yes. One pioneering approach involves creating a data preprocessing pipeline to transform an existing unimodal dataset into a multimodal one. For instance, the PlantCLEF2015 dataset was restructured into the "Multimodal-PlantCLEF" dataset by grouping images of multiple plant organs (flowers, leaves, fruits, stems) for the same species [1].
Q5: What is a common pitfall when building a multimodal data pipeline, and how can it be avoided? A common pitfall is creating an inefficient pipeline that results in idle GPUs due to excessive data padding, especially with text sequences [8]. A solution is to implement smarter batching strategies, such as "knapsack packing," which groups samples of similar lengths together to minimize padding and maximize GPU utilization [8].
Troubleshooting Guides
Problem: My Multimodal Model is Overfitting
| Step | Action | Expected Outcome |
|---|---|---|
| 1 | Apply Data Augmentation | Increased effective dataset size and improved model generalization. |
| 2 | Incorporate Multimodal Dropout | A more robust model that maintains performance even if a modality is missing at test time [1]. |
| 3 | Leverage Transfer Learning | Faster training and better performance, especially when labeled multimodal data is limited [6]. |
| 4 | Validate on Diverse Environments | A model that generalizes better across different growing conditions and is less biased [5]. |
Problem: My Data Modalities Are Misaligned or Incompatible
| Step | Action | Expected Outcome |
|---|---|---|
| 1 | Implement a Standardized Preprocessing Pipeline | Clean, uniformly structured data for each modality, ready for integration. |
| 2 | Adopt a Modular, Graph-Based Pipeline | Simplified management of heterogeneous data and seamless inter-modal conversion (e.g., extracting text from audio) [3]. |
| 3 | Use a Unified Output Format | Simplified merging and joint analysis of features from different modalities [3]. |
| 4 | Address Spatial/Temporal Biases | A more reliable model with predictions that are not skewed by data collection biases [7]. |
Experimental Protocols for Multimodal Integration
Protocol 1: Creating a Multimodal Dataset from Unimodal Sources This protocol is based on the methodology used to create the Multimodal-PlantCLEF dataset [1].
- Dataset Selection: Identify a public unimodal dataset with a wide array of plant species and images, such as PlantCLEF2015.
- Organ-Based Grouping: Restructure the dataset by grouping images of different organs (flower, leaf, fruit, stem) belonging to the same plant species and individual.
- Data Cleaning: Filter out species that do not have a sufficient number of images for each of the required organ types to ensure data completeness.
- Standardization: Resize and preprocess all images to a consistent resolution and format to facilitate model training.
Protocol 2: An Intermediate Fusion Workflow for Image and Weather Data This protocol summarizes the approach used for early prediction of maize yield [5].
- Data Collection:
- Imagery: Capture RGB images of crops at regular intervals using UAVs or ground platforms.
- Weather Data: Obtain historical weather data (temperature, humidity, solar radiation) for the field location from sources like NASA Power [5].
- Phenotypic Data: Record tabular data such as plant density, hybrid parental line, and date to anthesis.
- Feature Extraction:
- Images: Use a pre-trained Convolutional Neural Network (CNN) to extract deep feature representations from the images.
- Tabular Data: Normalize phenotypic and weather data.
- Temporal Alignment: Align the image-derived features and weather data temporally with key growth stages (e.g., anthesis, silking).
- Feature Fusion: Concatenate the extracted image features with the normalized tabular data vectors at an intermediate layer of a deep neural network.
- Model Training & Interpretation: Train a multimodal DNN for regression (yield prediction) and use explainability tools like SHAP to identify the most influential features from each modality.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function |
|---|---|
| Public Data Platforms (e.g., G2F, Plant Village) | Provide large-scale, annotated plant image and phenotypic datasets for training and benchmarking models [5] [6]. |
| Darwin Core Standards | A standardized framework for sharing biodiversity data, crucial for achieving interoperability across different datasets and platforms [7]. |
| Pre-trained Models (e.g., MobileNetV3) | Provide a robust foundation for feature extraction, especially for image-based modalities, reducing the need for large, private datasets [1] [6]. |
| Neural Architecture Search (NAS) | Automates the design of optimal neural network architectures, which can be applied to find the best fusion strategy for a given multimodal problem [1]. |
| Modular Preprocessing Frameworks (e.g., Pliers) | Support the construction of structured workflows to extract, transform, and align features from heterogeneous data sources (video, audio, images, text) [3]. |
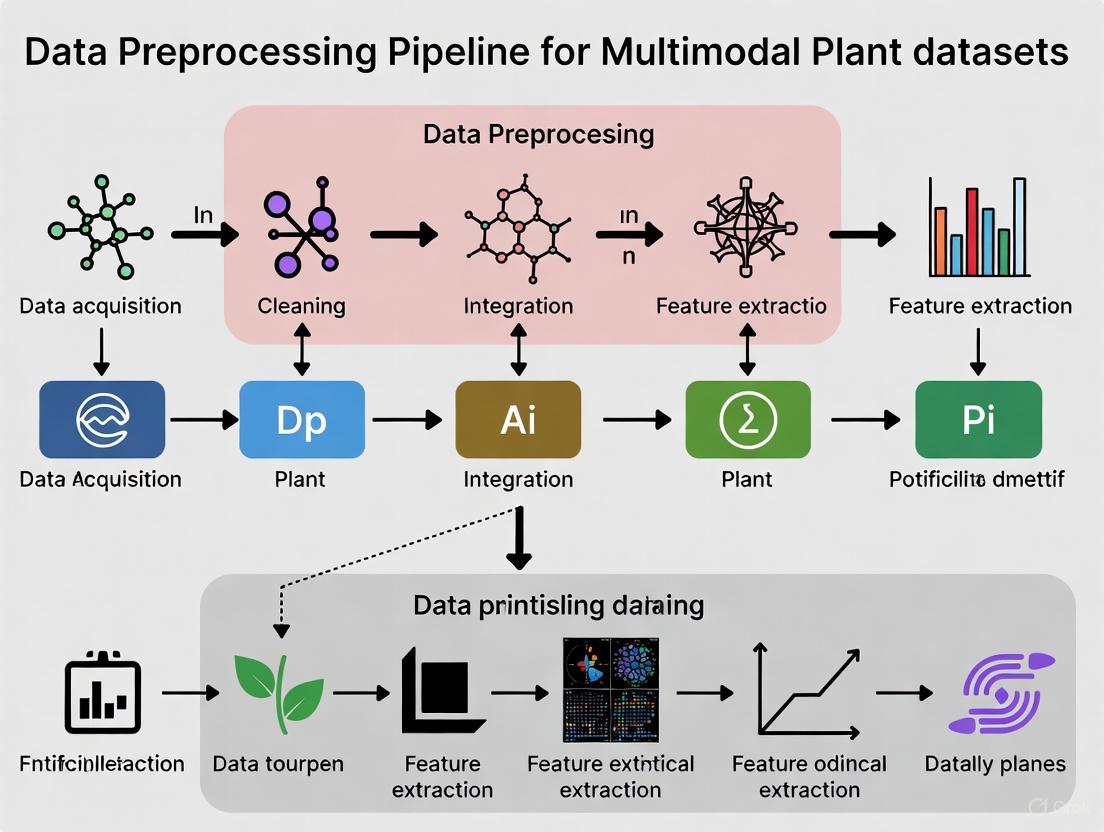
Multimodal Data Preprocessing Pipeline Workflow
The following diagram illustrates a generalized, modular workflow for preprocessing multimodal plant data, from raw data ingestion to model-ready features.
Generalized Multimodal Preprocessing Pipeline
Key Quantitative Guidelines for Dataset Curation
The table below summarizes recommended dataset sizes for different machine learning tasks in plant image analysis, which is a critical component of multimodal studies.
| Task Complexity | Recommended Minimum Dataset Size | Key Considerations |
|---|---|---|
| Binary Classification | 1,000 - 2,000 images per class [6] | A balanced dataset with roughly equal samples for each class is ideal. |
| Multi-class Classification | 500 - 1,000 images per class [6] | Requirements increase with the number of classes. Data augmentation is highly recommended. |
| Object Detection | Up to 5,000 images per object [6] | Requires bounding box annotations, which are labor-intensive to create. |
| Deep Learning Models (CNNs) | 10,000 - 50,000+ images total [6] | Larger models require more data. Transfer learning can reduce this requirement significantly. |
| Using Transfer Learning | As few as 100 - 200 images per class [6] | Effective for small datasets by leveraging features from a model pre-trained on a large, general dataset. |
Overcoming the Limitations of Unimodal Datasets in Complex Real-World Conditions
Multimodal Data Fusion Troubleshooting FAQs
FAQ 1: What are the most significant performance gaps between laboratory and real-world conditions for plant disease detection, and how can multimodal data help?
Laboratory conditions often achieve 95-99% accuracy in controlled settings, while real-world field deployment typically yields only 70-85% accuracy [9]. This significant performance gap stems from environmental variability, lighting changes, background complexity, and diverse growth stages that unimodal systems struggle to handle.
Multimodal data directly addresses these limitations by combining complementary information sources. For instance, integrating RGB imaging (for visible symptoms) with hyperspectral data (for pre-symptomatic physiological changes) provides more robust detection capabilities [9]. Research demonstrates that transformer-based architectures like SWIN achieve 88% accuracy on real-world datasets compared to just 53% for traditional CNNs, highlighting the importance of advanced fusion techniques [9].
Table: Performance Comparison Across Imaging Modalities and Environments
| Modality | Laboratory Accuracy | Field Accuracy | Key Strengths | Deployment Cost |
|---|---|---|---|---|
| RGB Imaging | 95-99% | 70-85% | Visible symptom detection, accessibility | $500-$2,000 USD |
| Hyperspectral Imaging | N/A reported | N/A reported | Pre-symptomatic detection, physiological analysis | $20,000-$50,000 USD |
| Multimodal Fusion (RGB+HSI) | N/A reported | 88% (SWIN transformers) | Combined strengths, robust to environmental variability | Cost-prohibitive for widespread use |
FAQ 2: How can I resolve synchronization issues between multiple data streams in field deployment?
Synchronization problems represent one of the most common technical challenges in multimodal research. These issues typically manifest as temporal misalignment between data streams, leading to inaccurate correlations and analysis errors [10].
Step-by-Step Resolution Protocol:
- Implement Hardware Synchronization: Use a master clock source with Precision Time Protocol (PTP) to maintain temporal alignment across all sensors [10]
- Address Clock Drift: Establish periodic re-synchronization routines, as internal device clocks can diverge by parts per million, causing significant misalignment over long recordings [10]
- Utilize Shared Event Markers: Incorporate visual or auditory markers (flashes, beeps) simultaneously recorded by all sensors for post-hoc alignment verification [10]
- Apply Post-Processing Correction: Deploy drift correction algorithms that estimate and compensate for temporal discrepancies based on timing signals [10]
FAQ 3: What data preprocessing pipeline effectively handles multimodal dataset inconsistencies?
Effective preprocessing must address the "heterogeneous hardware landscape" where sensors from various manufacturers use proprietary formats and protocols [10]. A robust pipeline should systematically resolve format inconsistencies, sampling rate mismatches, and data quality issues.
Comprehensive Preprocessing Protocol:
Data Cleaning Phase:
Format Standardization:
Temporal Alignment:
Quality Validation:
Table: Multimodal Preprocessing Solutions for Common Data Issues
| Data Issue | Detection Method | Resolution Techniques | Quality Metrics |
|---|---|---|---|
| Missing Values | Descriptive statistics, data profiling | Imputation (mean/median/mode), deletion, indicator variables | Percentage of completeness, pattern analysis |
| Noisy Data | Range validation, domain rules | Filtering, smoothing algorithms, format standardization | Signal-to-noise ratio, validation against constraints |
| Format Inconsistency | Data type checking, pattern matching | Type conversion, standardization protocols | Format compliance rate, parsing success rate |
| Sampling Rate Mismatch | Temporal analysis, frequency detection | Resampling, interpolation, alignment algorithms | Temporal alignment precision, data point correlation |
| Outliers | Statistical methods (IQR, Z-score), visualization | Winsorizing, transformation, domain-expert validation | Distribution analysis, impact assessment on models |
The Scientist's Toolkit: Essential Research Reagents & Solutions
Table: Critical Resources for Multimodal Plant Data Research
| Resource Category | Specific Tool/Solution | Function/Purpose | Implementation Considerations |
|---|---|---|---|
| Imaging Hardware | RGB Cameras | Capture visible spectrum symptoms | Cost-effective ($500-$2,000); suitable for initial deployment [9] |
| Imaging Hardware | Hyperspectral Sensors | Detect pre-symptomatic physiological changes | High cost ($20,000-$50,000); requires specialized expertise [9] |
| Synchronization | Lab Streaming Layer (LSL) | Resolve hardware compatibility and synchronization | Abstracts hardware-specific details; enables cross-platform data collection [10] |
| Data Management | TileDB Carrara | Multimodal data organization and governance | Manages data lake to warehouse transition; addresses governance challenges [14] |
| Fusion Algorithms | PlantIF Framework | Graph-based multimodal feature fusion | Achieves 96.95% accuracy on plant disease datasets; handles phenotype-text heterogeneity [15] |
| Annotation Software | Mangold INTERACT | Behavioral timeline creation and event annotation | Enables qualitative observation structuring; supports inter-rater reliability assessment [10] |
FAQ 4: How can I address the challenge of limited annotated datasets for multimodal plant pathology research?
The development of accurate plant disease detection models relies heavily on well-annotated datasets, which remain difficult to obtain at scale due to the need for expert plant pathologists to verify classifications [9]. This expert dependency creates bottlenecks in dataset expansion and diversification.
Experimental Protocol for Data Scarcity Mitigation:
Leverage Transfer Learning:
- Utilize pre-trained models on large-scale datasets (e.g., ImageNet) as feature extractors [9]
- Fine-tune final layers on limited domain-specific data
- Implement progressive unfreezing of layers during training
Apply Data Augmentation:
- Generate synthetic data using Generative Adversarial Networks (GANs) [9]
- Employ traditional augmentation techniques (rotation, flipping, color adjustment)
- Ensure augmentation reflects real-world environmental variations
Implement Few-Shot Learning:
- Utilize models like PlantIF that demonstrate strong performance with limited examples [15]
- Apply metric learning approaches to learn robust feature representations
- Use prototypical networks for classification with limited samples
Cross-Geographic Generalization:
- Incorporate data from multiple geographic regions during training [9]
- Apply domain adaptation techniques to address regional biases
- Implement test-time adaptation for new environments
FAQ 5: What fusion strategies work best for integrating heterogeneous data modalities in agricultural applications?
Effective multimodal fusion must address the fundamental challenge of heterogeneity between plant phenotypes and other modalities, such as textual descriptions or spectral data [15]. The optimal approach depends on the specific modalities involved and the agricultural application context.
Multimodal Fusion Experimental Protocol:
Early Fusion Strategy:
- Combine raw data from multiple modalities before feature extraction
- Suitable for highly correlated modalities with similar sampling rates
- Implement using concatenation or cross-modal attention mechanisms
Intermediate Fusion Approach:
Late Fusion Methodology:
- Process each modality through separate models to generate predictions
- Combine predictions using weighted averaging or meta-learning
- Effective for modalities with significant heterogeneity
Graph-Based Fusion Implementation:
The PlantIF framework demonstrates the effectiveness of graph-based fusion, achieving 96.95% accuracy on multimodal plant disease diagnosis—1.49% higher than existing models—by processing and fusing different modal semantic information through specialized attention mechanisms [15].
Troubleshooting Guides and FAQs
Data Complementarity
Q1: My multimodal plant dataset has missing organ images for many samples. How can I maintain data complementarity? A: Implement a robustness strategy directly within your deep learning model. Research on automatic fused multimodal deep learning for plant identification successfully addresses this by using multimodal dropout during training. This technique artificially drops certain modalities (e.g., flower or leaf images), forcing the model to learn robust representations and maintain performance even when some plant organ data is missing [1].
Q2: Are there specific plant organs that provide more complementary information than others? A: From a biological standpoint, a single organ is insufficient for accurate classification [1]. The most significant complementarity often comes from organs with distinct biological functions. For instance, integrating images of flowers, leaves, fruits, and stems provides a comprehensive representation of plant characteristics, as each organ encapsulates a unique set of biological features [1]. The optimal combination can be dataset-specific.
Data Alignment
Q3: What is the most common cause of temporal misalignment in continuously captured multimodal data, and how can it be corrected? A: The most pervasive cause is clock drift, where the internal clocks of different data collection devices gradually diverge over time [10]. This drift can accumulate over long recording sessions. Correction requires periodic re-synchronization using a master clock (e.g., via the Precision Time Protocol) or the use of post-hoc algorithms that estimate and correct for drift based on shared timing signals [10].
Q4: My data streams have different sampling rates (e.g., high-frequency sensors and lower-frequency images). How should I align them? A: This is a classic sampling rate mismatch challenge [10]. You have two main strategies:
- Downsampling: Reduce the higher-frequency data to match the lower frequency.
- Upsampling: Increase the lower-frequency data using interpolation techniques. The choice depends on your analysis goals. Downsampling is computationally simpler but may lose temporal precision, whereas upsampling can introduce artifacts if not carefully validated [10].
Data Heterogeneity
Q5: I need to integrate data from different sensor manufacturers, each with a proprietary output format. What is the best approach? A: This issue of data format inconsistency is common [10]. The recommended strategy is to use a middleware solution or custom data conversion scripts to transform all data into a standardized, common format (e.g., HDF5) before integration [10]. Careful selection of components with open standards or well-documented APIs during the experimental design phase can significantly reduce this problem.
Q6: How can I monitor data quality and heterogeneity in a multimodal pipeline that includes both structured metadata and unstructured image data? A: Adopt a split and monitor strategy. Independently monitor different data types and combine the results on a unified dashboard [16]:
- Structured Metadata: Use descriptive statistics, check for missing values, and monitor distribution drift.
- Unstructured Images: Use embedding monitoring or generate structured descriptors from the images (e.g., average brightness, texture metrics) and analyze them alongside your other structured data [16].
The table below summarizes key quantitative metrics and thresholds related to the core principles, derived from experimental protocols and system specifications.
Table 1: Quantitative Metrics for Multimodal Data Principles
| Principle | Metric | Reported Value / Threshold | Context / Rationale |
|---|---|---|---|
| Complementarity | Classification Accuracy | 82.61% | Achieved on 979 plant classes using a fused multimodal (flower, leaf, fruit, stem) model [1]. |
| Complementarity | Performance Gain over Unimodal | +10.33% | Accuracy increase over a late fusion baseline, highlighting the value of complementary data [1]. |
| Alignment | Synchronization Tolerance | <1 ms (typical target) | Required precision for temporal alignment to avoid erroneous conclusions in behavioral or physiological analysis [10]. |
| Alignment | Common Sampling Rates | EEG: 1000 Hz, Eye-tracker: 240 Hz, Video: 60 fps | Example rates leading to sampling rate mismatch [10]. |
| Heterogeneity | Data Format Variety | CSV, EDF, HDF5, proprietary binary | Common formats causing data format inconsistency [10]. |
| Heterogeneity | Network Bandwidth Requirement | Gigabit/10-Gigabit Ethernet | Recommended infrastructure to prevent data loss from bandwidth limitations during collection [10]. |
Experimental Protocols
Detailed Methodology: Automatic Fused Multimodal Deep Learning for Plant Identification
This protocol outlines the process for building a plant classification model using images from multiple plant organs [1].
1. Dataset Preprocessing and Curation:
- Input: A unimodal dataset (e.g., PlantCLEF2015) containing images labeled by plant species and organ type.
- Transformation: Restructure the dataset into a multimodal format, Multimodal-PlantCLEF, where each data sample consists of a set of images corresponding to different organs (flowers, leaves, fruits, stems) from the same plant species [1].
- Handling Missing Modalities: During training, apply multimodal dropout to simulate missing organ images and ensure model robustness [1].
2. Unimodal Model Training:
- For each modality (plant organ), train a separate deep learning model (e.g., MobileNetV3Small) using transfer learning from a pre-trained model [1].
3. Automated Multimodal Fusion:
- Fusion Algorithm: Apply a Multimodal Fusion Architecture Search (MFAS). This algorithm automatically discovers the optimal way to combine the features extracted from the unimodal models, rather than relying on a fixed, human-defined fusion strategy like late fusion [1].
- Output: A single, compact multimodal model that effectively integrates information from all available plant organs.
4. Model Evaluation:
- Metrics: Use standard performance metrics such as classification accuracy.
- Baseline Comparison: Validate the model against established benchmarks (e.g., late fusion with averaging) and use statistical tests like McNemar's test to confirm superiority [1].
Diagram: Multimodal Plant Data Preprocessing Workflow
Multimodal Dataset Creation and Model Training Pipeline
Diagram: Data Synchronization and Alignment Challenge
Common Challenges in Multimodal Data Alignment
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Tools for Multimodal Plant Research
| Item / Solution | Function / Application |
|---|---|
| Standardized Datasets (e.g., Multimodal-PlantCLEF) | Provides a curated, preprocessed benchmark for developing and evaluating multimodal plant classification models, ensuring reproducibility [1]. |
| Middleware (e.g., Lab Streaming Layer - LSL) | A software solution that abstracts away hardware-specific details, enabling the synchronization of data streams from different sensors and resolving compatibility issues [10]. |
| Multimodal Fusion Architecture Search (MFAS) | An algorithmic tool that automates the discovery of optimal neural network architectures for combining data from different modalities, outperforming manual fusion strategies [1]. |
| High-Performance Network Switch (Gigabit/10-Gigabit) | Critical hardware infrastructure to handle the enormous data volumes generated during multimodal collection, preventing data loss from bandwidth limitations [10]. |
| Graph Neural Networks (GNNs) | A class of deep learning models particularly effective for integrating and analyzing heterogeneous, network-structured data, such as biological interaction networks in drug discovery [17] [18]. |
Frequently Asked Questions (FAQs)
Q1: What are the key differences between the Multimodal-PlantCLEF and Augmented PlantVillage benchmarks?
Table 1: Core Characteristics of Featured Multimodal Benchmarks
| Feature | Multimodal-PlantCLEF | Augmented PlantVillage |
|---|---|---|
| Primary Task | Plant species identification [1] | Crop disease detection and diagnosis [19] [20] |
| Core Modalities | Images of multiple plant organs (flowers, leaves, fruits, stems) [1] | Plant disease images + Textual symptom descriptions & metadata [19] |
| Data Source | Restructured from PlantCLEF2015 [1] | Augmented from the original PlantVillage collection [19] |
| Key Innovation | Automatic modality fusion strategy for robust classification [1] | Expert-curated text prompts for vision-language model training [19] |
| Typical Model | Multimodal Deep Learning with fusion architecture search [1] | Vision-Language Models (e.g., CLIP, BLIP), Multimodal LLMs [19] [20] |
Q2: Why is a data preprocessing pipeline critical for building a multimodal plant dataset from unimodal sources? A robust preprocessing pipeline is essential to address the modality gap—the inherent differences in data structure and representation between various data types. Without careful processing, models cannot effectively learn the complementary relationships between modalities, such as how the visual features of a leaf correspond to its textual disease description [1] [19]. The creation of Multimodal-PlantCLEF from PlantCLEF2015 demonstrates a pipeline that reorganizes single-organ images into a structured, multi-organ (multimodal) dataset where each sample combines specific views of the same species [1].
Q3: What is modality dropout and how is it used to improve model robustness? Modality dropout is a training technique where one or more input modalities (e.g., a fruit image) are randomly omitted during training. This forces the model to learn to make accurate predictions even with incomplete data, mimicking real-world scenarios where certain data might be missing. Research on Multimodal-PlantCLEF has shown that this technique significantly enhances model robustness [1].
Q4: My multimodal model performs well on lab data but fails in the field. What could be wrong? This is a common challenge due to the simplicity gap between controlled lab images and complex field conditions [9]. Field images contain variable lighting, complex backgrounds, and different plant growth stages. To mitigate this:
- Utilize Augmented PlantVillage's contextual metadata (e.g., soil conditions, climate data) to make your model aware of environmental factors [19].
- Incorporate data augmentation during training to simulate field conditions.
- Fine-tune models on real-field datasets if available, as models trained solely on lab images like the original PlantVillage see significant accuracy drops (from 95-99% to 70-85%) when deployed in real environments [9].
Troubleshooting Guides
Issue: Poor Model Performance Due to Missing Modalities
Problem: Your trained multimodal model encounters samples during testing where one or more modalities (e.g., stem image) are missing, leading to unreliable or failed predictions.
Solution: Implement robustness strategies during training and inference.
- Step 1: Apply Modality Dropout. During the training phase, intentionally and randomly drop modalities. This teaches the model not to become over-reliant on any single data source and to leverage cross-modal relationships [1].
- Step 2: Design a Robust Inference Pipeline. Structure your inference code to handle missing inputs gracefully. The model, trained with modality dropout, will be capable of producing a prediction even with an incomplete sample [1].
- Step 3 (Advanced): Explore Generative Imputation. For critical applications, investigate using generative models to create plausible synthetic data for the missing modality based on the available ones, though this adds complexity.
Issue: Data Scarcity and Class Imbalance for Rare Species/Diseases
Problem: Certain plant species or diseases have very few examples in your dataset, leading to a model that is biased toward common classes.
Solution: Leverage Few-Shot Learning (FSL) techniques and data augmentation strategies.
- Step 1: Employ Contrastive Pre-training. Use a contrastive learning objective (e.g., using a Siamese network) on all available data. This helps the model learn a powerful and generalized feature representation that is effective even with few examples per class [21].
- Step 2: Adopt a Few-Shot Learning Framework. Utilize prototype-based networks, which compute a prototypical representation for each class from its few support examples. Query samples are classified based on their distance to these prototypes [21].
- Step 3: Augment with Synthetic Data. Use Large Language Models (LLMs) to generate additional textual descriptions for rare diseases, as demonstrated in multimodal few-shot learning research [21]. For images, consider Generative Adversarial Networks (GANs) to create synthetic samples.
Issue: Ineffective Fusion of Multimodal Features
Problem: Simply combining image and text features (e.g., by concatenation) does not lead to performance improvement, indicating poor fusion strategy.
Solution: Systematically explore and search for an optimal fusion architecture rather than relying on a fixed, manual design.
- Step 1: Benchmark Standard Fusion Techniques. Start by implementing and comparing early (feature-level), late (decision-level), and hybrid fusion to establish a baseline [1].
- Step 2: Implement Automated Fusion Search. Move beyond manual design by using a Multimodal Fusion Architecture Search (MFAS). This algorithm automatically discovers the most effective way to combine features from different encoders, which has been shown to significantly outperform simple late fusion (e.g., by over 10% accuracy) [1].
- Step 3: Analyze Cross-Modal Attention. If using transformer-based models, analyze the attention maps between image patches and text tokens. This can reveal if the model is truly learning meaningful cross-modal interactions or ignoring one modality.
Table 2: Comparison of Multimodal Fusion Strategies
| Fusion Strategy | Description | Advantages | Disadvantages |
|---|---|---|---|
| Early Fusion | Raw data from modalities is combined before feature extraction. | Allows modeling of low-level interactions. | Highly susceptible to noise and misalignment; requires synchronized data [1]. |
| Late Fusion | Decisions from unimodal models are combined (e.g., by averaging). | Simple, flexible, and modalities can be processed independently [1]. | Cannot capture complex cross-modal relationships at the feature level [1]. |
| Intermediate (Hybrid) Fusion | Features from unimodal encoders are merged within the model. | Balances flexibility with the capacity for rich interaction. | The fusion point and method are critical and non-trivial to design manually [1]. |
| Automated Fusion (MFAS) | Uses neural architecture search to find the optimal fusion structure. | Data-driven, can discover highly effective and non-intuitive architectures [1]. | Computationally more expensive during the search phase. |
Experimental Protocols & Methodologies
Protocol: Replicating the Multimodal-PlantCLEF Preprocessing Pipeline
This protocol outlines the steps to create a multimodal dataset from a unimodal source, based on the methodology used to create Multimodal-PlantCLEF from PlantCLEF2015 [1].
Objective: To transform a collection of single-organ plant images into a structured multimodal dataset where each data point consists of multiple organ views for a single plant species.
Materials: Source dataset (e.g., PlantCLEF2015), computing environment with storage.
Procedure:
- Data Annotation Audit: Review the source dataset's annotations to identify labels specifying the plant organ depicted in each image (e.g., "leaf," "flower," "fruit," "stem").
- Species-Organ Grouping: Group all images by their species label. Within each species group, further subgroup the images by their organ type.
- Multimodal Sample Construction:
- For each species, create a new data sample.
- For each required modality (e.g., leaf, flower), select one image at random from the corresponding organ subgroup.
- If an organ type is not available for a species, mark that modality as
nullor missing. This prepares the dataset for robustness techniques like modality dropout.
- Dataset Splitting: Perform a stratified split on the newly constructed multimodal samples (not the original images) to create training, validation, and test sets, ensuring all species are represented in each split.
Protocol: Fine-tuning a Vision-Language Model on Augmented PlantVillage
This protocol describes how to adapt a general-purpose Vision-Language Model (VLM) for the specialized task of plant disease diagnosis using a dataset like the Augmented PlantVillage [19] [20] [22].
Objective: To specialize a pre-trained VLM (e.g., CLIP, LLaVA, Qwen-VL) to accurately diagnose plant diseases from images and textual prompts.
Materials: Augmented PlantVillage dataset (images and text), access to a GPU cluster, pre-trained VLM weights.
Procedure:
- Data Preparation: Load the dataset, which includes image files and paired JSON annotation files containing text prompts, disease classes, and contextual metadata [19].
- Model Selection: Choose a base VLM. Models like Qwen-VL have shown success in this domain after fine-tuning [22].
- Fine-tuning Strategy:
- Full Fine-tuning: For maximum performance, fine-tune the entire model (visual encoder, connector, and language model) on the target dataset. This can be computationally intensive.
- Parameter-Efficient Fine-Tuning (PEFT): Use methods like Low-Rank Adaptation (LoRA). This technique fine-tunes the visual encoder, adapter, and language model simultaneously by injecting trainable rank decomposition matrices into the layers of the pre-trained model, dramatically reducing the number of trainable parameters and computational cost while maintaining high performance [22].
- Training & Evaluation: Train the model using standard vision-language objectives (e.g., contrastive loss, captioning loss). Evaluate on a held-out test set, reporting metrics like classification accuracy, F1-score, and qualitative analysis of generated diagnoses.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Resources for Multimodal Plant Data Research
| Resource Name / Type | Function in Research | Example / Source |
|---|---|---|
| Pre-trained Vision Models | Serves as a feature extractor for image modalities, providing a strong starting point and transfer learning. | MobileNetV3, EfficientNetB0, ResNet-50 [1] [20] [23] |
| Vision-Language Models (VLMs) | Base architecture for building systems that jointly understand plant images and textual descriptions. | CLIP, BLIP, LLaVA, Qwen-VL [19] [20] [24] |
| Neural Architecture Search (NAS) | Automates the design of optimal neural network architectures, including multimodal fusion layers. | Multimodal Fusion Architecture Search (MFAS) [1] |
| Parameter-Efficient Fine-Tuning (PEFT) | Enables effective adaptation of large models to new tasks with minimal computational overhead. | Low-Rank Adaptation (LoRA) [22] |
| Explainable AI (XAI) Tools | Provides post-hoc interpretations of model predictions, building trust and providing biological insights. | LIME (for images), SHAP (for tabular/weather data) [23] |
| Contrastive Learning Framework | Used for pre-training to learn high-quality, generalized feature representations, beneficial for few-shot learning. | Siamese Networks, Prototypical Networks [21] |
The How: A Step-by-Step Pipeline for Multimodal Data Curation and Fusion
For researchers building preprocessing pipelines for multimodal plant datasets, the acquisition and sourcing of high-quality, diverse data is a critical first step. This process often involves integrating disparate sources, including citizen science platforms, structured field studies, and public data repositories. Each source presents unique advantages and specific challenges that can impact data quality and usability. This technical support center provides targeted troubleshooting guides and FAQs to help you navigate common issues, mitigate data biases, and implement robust experimental protocols for effective multimodal data integration.
FAQs and Troubleshooting Guides
Citizen Science Data
Q1: How can we address spatial and taxonomic biases in citizen science data? Citizen science platforms, such as iNaturalist, are among the largest sources of plant occurrence data but are prone to spatial biases (e.g., oversampling in easily accessible areas) and taxonomic biases (e.g., under-sampling of cryptic or non-charismatic species) [25]. To mitigate this:
- Leverage Multispecies Deep Learning Models: Employ Deep Neural Networks (DNNs) designed for multispecies distribution modeling. These models are comparably robust to spatial sampling bias because they learn from the relative observation probabilities across many species simultaneously, reducing the effect of uneven sampling intensity [25].
- Implement Strategic Sampling Guidance: Proactively guide citizen scientists to explore identified plant diversity "darkspots"—regions with many undescribed or unrecorded species. Provide them with guidance on when to search and which diagnostic plant characters to photograph [26].
Q2: What are the best practices for discovering new species or rare phenotypes using citizen science? The discovery of novel species is often dependent on expert engagement with citizen science platforms [26].
- For Experts: Routinely review observations in your taxonomic group of expertise. Build collaboration networks with the community by regularly identifying records and interacting with other users. This cultivates a culture where users will actively notify you of potentially significant records [26].
- For All Researchers: Prioritize the collection of data for ephemeral species or those with diagnostic characters that are better captured by photographs than by preserved specimens, such as flower color or corolla shape, which can be lost in herbarium samples [26].
Field Studies and Sensor-Based Data Acquisition
Q3: What is a systematic method for troubleshooting a field-based data acquisition system that is not recording data? A structured approach to troubleshooting is crucial for resuming data collection quickly [27].
- Start with the Basics: Verify the data logger has power and is turned on [27].
- Test Your Assumptions: Systematically ask, "Is my assumption correct that...?" for each component. For every "yes" answer, support it with evidence from a simple test (e.g., "How do I know the data logger has power? Because my voltmeter reads 13.4 V") [27].
- Isolate the Problem: Understand the measurement chain: environmental parameter → sensor → electrical signal → data logger → stored data → computer. Test each link independently. For a faulty temperature reading, independently verify the sensor by placing it in ice water (0°C) to see if the reading changes as expected [27].
Q4: What are the common problems with data loggers, and how can they be solved? Data loggers, while useful, have several limitations that can be mitigated by moving towards real-time data acquisition systems [28].
Table 1: Common Data Logger Problems and Solutions
| Problem | Impact | Solution |
|---|---|---|
| Gaps Between Measurements [28] | Missed events that occur between logging intervals, jeopardizing sample integrity. | Use a real-time data acquisition system that can trigger high-frequency measurement and alarms immediately when a parameter is breached. |
| Missed Alarms During Network Failure [28] | No timely alert for out-of-spec conditions, leading to potential data or sample loss. | Implement a system with 4G failover connectivity and unlimited data buffering to ensure alarm delivery even during network issues. |
| Battery Power Limitations [28] | Requires manual replacement, risking invalid sensor calibration and data loss. | Use a professionally installed system with battery backup only for power outages, not as primary power. |
| Risk of Human Error in Setup [28] | Portable loggers can be moved or misconfigured, invalidating calibration and data. | Opt for a professionally installed system where sensors and recording units are integrated to ensure correct setup. |
Public Repositories and Multimodal Integration
Q5: How can we effectively integrate multimodal data from different sources (e.g., images and environmental data) for plant disease diagnosis? Integrating diverse data types addresses the limitations of single-modality systems [23].
- Adopt a Multimodal Deep Learning Framework: Implement a model that uses separate streams for different data types (e.g., EfficientNetB0 for image classification and a Recurrent Neural Network for environmental time-series data) and fuses the predictions [23].
- Implement Explainable AI (XAI) Techniques: Use tools like LIME (for image-based predictions) and SHAP (for weather-based predictions) to interpret the model's decisions. This improves transparency and trust in the diagnostic outcomes, which is critical for real-world applications [23].
Q6: What are the key challenges in using public image repositories for AI model training, and how can they be overcome? A major barrier in agricultural AI is the lack of large, well-labeled, and curated image sets that account for the high variability in real-world conditions [29].
- Challenge: A stop sign looks the same everywhere, but a pea plant's appearance varies with genetics, weather, and growth stage. Models trained on limited datasets fail to generalize [29].
- Solution: Utilize large-scale, open-source repositories like the Ag Image Repository (AgIR). These provide high-quality images of plants across different growth stages, varieties, and environmental conditions, which are essential for training robust computer vision models [29].
Experimental Protocols for Data Sourcing
Protocol 1: Building a Multimodal Plant Image Dataset
This methodology is derived from the development of the Ag Image Repository and related research [29] [30].
- Image Acquisition: Use automated systems like wheel-mounted "Benchbots" with high-resolution cameras programmed to capture time-series images of plants in pots arranged in rows. This ensures consistent, high-quality data collection over the plant's lifecycle [29].
- Data Curation and Annotation: Develop software tools to streamline the creation of "cut-outs" (plants removed from their background) and to annotate images with detailed metadata (species, growth stage, health status). This step is critical for creating a dataset usable for supervised learning [29].
- Multimodal Structuring: For plant identification tasks, restructure the dataset to include images of multiple plant organs (flowers, leaves, fruits, stems) per species, forming a multimodal dataset like Multimodal-PlantCLEF [30].
- Repository Deployment: Share the final, curated dataset on a publicly accessible platform or high-performance computing cluster (e.g., USDA SCINet) to accelerate AI research in agriculture [29].
Protocol 2: Modeling Plant Distributions from Citizen Science Data
This protocol uses multispecies Deep Neural Networks (DNNs) to handle biases in opportunistic observations [25].
- Data Compilation: Gather and quality-filter citizen science occurrence records (e.g., from platforms like InfoFlora). Compile high-resolution environmental predictors (e.g., climate, topography) and seasonal predictors (sine-cosine transforms of the day of the year) for the study region [25].
- Model Training: Train an ensemble of DNNs using cost functions like Cross-Entropy Loss (CEL) or Normalized Discounted Cumulative Gain (NDCG). The multispecies, joint modeling approach makes the model more robust to spatial sampling bias [25].
- Validation and Analysis:
- Performance Validation: Validate the model against left-out citizen science data and independently collected, systematically distributed plant community inventories [25].
- Ecological Insight Extraction: Use the trained model to map fine-grained species distributions, investigate spatial variations in flowering phenology (by analyzing the timing of peak observation probability), and project future changes under climate scenarios [25].
Workflow Visualization
Data Sourcing and Integration Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Tools for Data Acquisition and Analysis
| Item | Function | Application Context |
|---|---|---|
| Digital Multimeter [27] | Provides independent verification of voltages and checks electrical continuity. | Troubleshooting field data acquisition systems and sensors. |
| iNaturalist Platform [26] | A citizen science platform for recording and identifying biodiversity observations. | Sourcing large volumes of plant occurrence data and facilitating species discovery. |
| Ag Image Repository (AgIR) [29] | A public repository of high-quality, curated plant images with metadata. | Training and benchmarking robust computer vision and deep learning models for agriculture. |
| Deep Neural Networks (DNNs) [25] | Machine learning models for joint, multispecies distribution modeling. | Predicting species distributions and community composition from biased citizen science data. |
| Explainable AI (XAI) Tools (LIME & SHAP) [23] | Provides post-hoc explanations for predictions made by complex AI models. | Interpreting and validating diagnoses from multimodal plant disease models. |
| Benchbots / Automated Imaging Rigs [29] | Robotic systems for automated, high-throughput plant imaging. | Generating consistent, time-series image data for phenotyping and dataset creation. |
| Species Distribution Models (SDMs) [7] | Algorithms to characterize habitat suitability and species' environmental niches. | Modeling potential species ranges based on environmental variables. |
| Darwin Core Standards [7] | A standardized framework for publishing biodiversity data. | Ensuring interoperability and integration of biodiversity data from different sources. |
Troubleshooting Guide: Image Standardization for Plant Phenotyping
Q: What are the primary challenges in aligning images from different camera technologies for plant phenotyping, and how can they be addressed?
A: The main challenges are parallax effects and occlusion effects inherent in plant canopy imaging. A effective solution is to integrate 3D information from a depth camera (e.g., a time-of-flight camera) into the registration process. This depth data helps mitigate parallax, facilitating more accurate pixel alignment. Furthermore, implementing an automated mechanism to identify and filter out various types of occlusions can minimize registration errors. This method is robust across different plant types and is not reliant on detecting plant-specific image features [31].
Q: How do I standardize a dataset containing plant images from multiple organs for a multimodal classification model?
A: Standardizing multi-organ images involves creating a cohesive dataset and processing pipeline. You can transform an existing unimodal dataset into a multimodal one by implementing a data preprocessing pipeline that groups images by plant organ (e.g., flowers, leaves, fruits, stems). Each organ, treated as a distinct modality, should be processed through a dedicated feature extractor (e.g., a pre-trained CNN like MobileNetV3). The fusion of these features can then be optimized automatically using algorithms like Multimodal Fusion Architecture Search (MFAS) to determine the most effective integration point, significantly boosting classification performance [1].
Experimental Protocol: 3D Multimodal Plant Image Registration This protocol is adapted from a novel registration algorithm for plant phenotyping [31]:
- Data Acquisition: Set up a multimodal monitoring system with a time-of-flight depth camera and other arbitrary camera technologies (e.g., RGB, multispectral).
- Depth Data Capture: Capture 3D information of the plant canopy using the depth camera.
- Ray Casting for Registration: Leverage the depth data and a ray casting technique to align pixels from the different camera modalities accurately.
- Occlusion Filtering: Run the integrated automated detection algorithm to identify and filter out pixels affected by occlusion.
- Output: Generate the final registered images and 3D point clouds of the plants for downstream analysis.
Troubleshooting Guide: Text Tokenization for Agricultural Literature Mining
Q: What are the essential steps for preprocessing text data, such as research abstracts or field notes, for summarization or classification tasks in an agricultural context?
A: A standard preprocessing pipeline for textual data involves several key steps [32]:
- Data Cleaning: Remove irrelevant characters, correct typos, and handle encoding issues.
- Tokenization: Split the raw text into smaller units (tokens), which can be words, subwords, or characters.
- Normalization: Convert text to a standard form, such as converting all characters to lowercase.
- Truncation and Padding: Ensure all text sequences are of uniform length by truncating long sequences or padding short ones to a fixed length.
- Attention Masks: For models like Transformers, create attention masks to indicate to the model which tokens are actual data and which are padding.
Experimental Protocol: Text Preprocessing for Model Training This protocol outlines the steps for preparing a text dataset (e.g., the CNN/Daily Mail dataset) for training a summarization model [32]:
- Data Loading: Load the dataset, typically divided into training, validation, and test CSV files.
- Cleaning & Preprocessing: Apply the techniques listed above: tokenization, normalization, truncation, and the creation of attention masks.
- Exploratory Data Analysis (EDA): Perform EDA to understand data insights, such as the distribution of article and summary lengths.
- Model Input Preparation: Feed the preprocessed tokens and attention masks into deep learning models (e.g., T5, BART, PEGASUS) for training.
- Evaluation: Use metrics like ROUGE and BLUE scores to assess the model's summarization performance.
Troubleshooting Guide: Environmental Data Normalization
Q: How do I identify and handle outliers in my environmental dataset, such as sensor readings for temperature or soil moisture?
A: Outliers can be identified using several statistical methods [33]:
- Interquartile Range (IQR) method: Any data point below
Q1 – 1.5 * IQRor aboveQ3 + 1.5 * IQRis considered an outlier. - Z-score method: Data points that fall more than 3 standard deviations from the mean are flagged.
- Tukey's fences: A more stringent variant using
Q1 – 3 * IQRandQ3 + 3 * IQR. - Model-based methods: Algorithms like Local Outlier Factor (LOF) use the local density of neighboring data points to identify outliers. It is critical to combine these methods with an understanding of the data collection process and domain knowledge to decide whether to correct or remove outliers [33].
Q: My environmental data (e.g., nutrient concentrations, pollutant levels) is highly skewed. Which normalization method should I use and why?
A: For skewed environmental data, logarithmic transformation is often the most appropriate method. The goal of normalization is to change the values to a common scale without distorting value ranges, and to make the data's distribution more Gaussian (bell-curved) for further statistical analysis. The Shapiro-Wilk test can confirm if data is normally distributed. A p-value < 0.05 indicates a non-normal distribution. Log transformation compresses the scale for large values, effectively reducing positive skewness and making the data more suitable for parametric statistics and regression analysis [34].
Experimental Protocol: Normalizing Skewed Environmental Data This protocol is based on standard practices for handling non-Gaussian environmental data [34]:
- Assess Distribution: Visually inspect the data distribution using histograms or kernel density plots. Perform the Shapiro-Wilk test for normality. A p-value < 0.05 confirms the data is not normally distributed.
- Apply Log Transformation: Transform the data using a logarithmic function (
log(original_value)). - Re-assess Distribution: Run the Shapiro-Wilk test again on the log-transformed data. The p-value should now be > 0.05, confirming a normal distribution. Visually compare box plots and density plots before and after transformation to confirm the decrease in skewness.
- Proceed with Analysis: The normalized data is now ready for further analysis, such as multivariate linear regression.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 1: Key computational tools and data solutions for multimodal plant research.
| Item Name | Function/Brief Explanation | Relevant Context |
|---|---|---|
| Time-of-Flight (ToF) Depth Camera | Captures 3D information to mitigate parallax effects in image registration. [31] | Plant phenotyping, 3D reconstruction. |
| Multimodal Fusion Architecture Search (MFAS) | Automates the discovery of optimal fusion points for combining data from multiple modalities. [1] | Integrating image, environmental, and genomic data. |
| Pre-trained Deep Learning Models (T5, BART, PEGASUS) | Provides a foundation for natural language processing (NLP) tasks like text summarization. [32] | Mining agricultural literature and reports. |
| Explainable AI (XAI) Libraries (LIME, SHAP) | Provides post-hoc explanations for model predictions, enhancing interpretability and trust. [23] | Diagnosing plant disease and validating model decisions. |
| Python Libraries (e.g., Scikit-learn, PyOD) | Offers comprehensive algorithms for data preprocessing, outlier detection, and machine learning. [33] | General-purpose data cleaning and analysis. |
Workflow Visualization: Multimodal Preprocessing Pipeline
The diagram below illustrates a logical workflow for preprocessing the three data modalities discussed, preparing them for a fusion-based model.
Diagram 1: A logical workflow for preprocessing multimodal data.
FAQs on Data Annotation and Labeling
Q1: What are the primary strategies to create labeled datasets when annotated data is scarce? A combination of expert curation and weak supervision is highly effective. Expert curation provides high-quality labels but is resource-intensive. Weak supervision uses lower-cost, noisier sources to generate labels programmatically. For example, multiple noisy labeling functions—such as heuristics, knowledge bases, or predictions from other models—can be aggregated to create a probabilistic training set [35]. In species-level trait imputation, models can be trained on existing data to predict missing traits for related species.
Q2: How can weak supervision be applied to complex, non-categorical data like plant trait rankings? Traditional weak supervision focuses on classification, but it can be universalized. For rankings, the label model can be reoriented to minimize a specific distance metric, such as the Kendall Tau distance, which measures the number of adjacent swaps needed to match two permutations [35]. This framework allows weak supervision to be applied to regression, graphs, and other complex structures where simple categorization isn't sufficient.
Q3: Can large language models (LLMs) be used for weak supervision in specialized domains like plant science? Yes, LLMs can be prompted to generate weak labels (pseudo-labels) for training smaller, more efficient downstream models. To enhance performance in a specialized domain, the LLM can first be fine-tuned on a small set of expert-annotated data. The fine-tuned LLM then generates weak labels for a much larger unlabeled dataset, which are used to train a compact model like BERT. This strategy minimizes the need for domain knowledge to create labeling functions and avoids the computational expense of deploying large LLMs in production [36].
Q4: What is the key challenge in building multimodal plant classification models, and how can it be addressed? A major challenge is modality fusion—determining the optimal strategy to combine information from different data sources (e.g., images of flowers, leaves, fruits, and stems) [1] [37]. Manually designed fusion architectures can be suboptimal. This can be addressed by using a Multimodal Fusion Architecture Search (MFAS), which automates the discovery of the best fusion strategy, leading to more accurate and robust models compared to common practices like late fusion [1] [37].
Q5: How can we ensure our model is robust when some data modalities are missing? Incorporating multimodal dropout during training is a key technique. It randomly drops subsets of modalities, forcing the model to learn robust representations that do not over-rely on any single data type. This results in a model that maintains higher performance even when, for example, only leaf images are available instead of the full set of organ images [1] [37].
Troubleshooting Guides
Problem: Labels generated through weak supervision are noisy, leading to poor model performance.
- Potential Cause: The labeling functions (LFs) are low-quality, conflicting, or have broad coverage errors.
- Solution:
- Analyze LF Conflicts: Use the data programming framework to compute a covariance matrix between your LFs. This helps identify groups of LFs that frequently disagree.
- Refine LFs: Based on the analysis, rewrite heuristics or adjust knowledge-based LFs to reduce conflict.
- Leverage the Label Model: Use the label model (e.g., in Snorkel AI) to de-noise the weak labels by estimating their accuracies and correlations, rather than simply taking a majority vote [35].
Problem: My multimodal model performs worse than a unimodal one.
- Potential Cause: Suboptimal fusion strategy or poor alignment between modalities.
- Solution:
- Audit Unimodal Backbones: First, ensure each unimodal model (e.g., a CNN for images) is competently trained on its specific task.
- Automate Fusion Search: Replace manual fusion (e.g., simple concatenation or averaging) with a Neural Architecture Search (NAS) method tailored for multimodal problems, such as MFAS, to find a better fusion structure [1] [37].
- Check Data Alignment: Verify that samples from different modalities (e.g., a leaf image and a corresponding soil measurement) are correctly paired and aligned in your dataset.
Problem: High computational cost of using LLMs for weak labeling on a large dataset.
- Potential Cause: Performing inference with a full-scale LLM (e.g., Llama2-13B) on thousands of samples is computationally expensive [36].
- Solution:
- Use a Two-Stage Pipeline: Fine-tune an LLM on a small gold-standard dataset, then use it to generate pseudo-labels for the entire unlabeled corpus.
- Distill Knowledge: Use the generated pseudo-labels to train a much smaller, task-specific model (e.g., BERT). This smaller model is then used for production inference, drastically reducing computational costs [36].
Experimental Protocols and Data
Table 1: Performance Comparison of Multimodal Fusion Strategies on Plant Identification
| Fusion Strategy | Description | Accuracy on Multimodal-PlantCLEF | Key Advantage |
|---|---|---|---|
| Late Fusion (Baseline) | Averages predictions from unimodal models [1] [37]. | ~72.28% | Simple to implement |
| Automatic Fusion (MFAS) | Uses architecture search to find optimal fusion points [1] [37]. | 82.61% | Superior performance |
| With Multimodal Dropout | MFAS model trained with randomly dropped modalities [1] [37]. | High robustness | Handles missing data |
Table 2: Weak Supervision Pipeline Performance with Limited Gold-Standard Data
| Method | 3 Gold Standard Notes (F1) | 10 Gold Standard Notes (F1) | Key Insight |
|---|---|---|---|
| BERT (Fine-tuned) | 0.5953 (Events) / 0.2753 (Time) | N/A | Struggles with very low data |
| LLM (Fine-tuned) | 0.7418 (Events) / 0.6045 (Time) | N/A | Better, but computationally heavy |
| LLM-WS-BERT | 0.7765 (Events) / 0.7538 (Time) | 0.8466 (Events) / 0.8448 (Time) | Dominant strategy: Combines weak supervision and efficient final model [36] |
Workflow Visualization
Weak Supervision and Imputation Workflow
Automated Multimodal Fusion with MFAS
The Scientist's Toolkit
Table 3: Essential Research Reagents and Resources
| Item | Function in the Pipeline |
|---|---|
| Multimodal-PlantCLEF Dataset | A restructured version of PlantCLEF2015 providing aligned images of flowers, leaves, fruits, and stems for the same plant specimen, enabling multimodal model development [1] [37]. |
| Pre-trained Models (e.g., MobileNetV3) | Provide a strong feature extraction backbone for image-based modalities, enabling effective transfer learning, especially when training data is limited [1] [37]. |
| Multimodal Fusion Architecture Search (MFAS) | An algorithm that automates the discovery of the optimal neural architecture for fusing information from different data modalities, replacing error-prone manual design [1] [37]. |
| Weak Supervision Framework (e.g., Snorkel) | Provides a programming model for defining labeling functions and a label model that aggregates their noisy signals to create a probabilistic training set without manual labeling [35]. |
| Large Language Model (e.g., Llama2) | Can be fine-tuned and used as a source of weak labels for textual or structured data, minimizing the need for hand-crafted rules and domain-specific ontologies [36]. |
Frequently Asked Questions
Q1: What is the core difference between early, intermediate, and late fusion? The core difference lies at which stage in the model pipeline the data from different modalities is combined [38] [39].
- Early Fusion: Integration happens at the input level, combining raw or low-level data before feature extraction.
- Intermediate Fusion: Integration happens at the feature level, combining latent representations extracted from each modality.
- Late Fusion: Integration happens at the decision level, combining the outputs or predictions of modality-specific models.
Q2: How do I choose the right fusion strategy for my plant dataset? The choice depends on your data characteristics and research goal [38] [40] [39].
- Use Early Fusion if your modalities are tightly synchronized and you want to model low-level interactions, but be wary of its sensitivity to noise and misalignment.
- Use Intermediate Fusion to capture rich, non-linear interactions between modalities; this is the most widely used strategy but requires all modalities to be present.
- Use Late Fusion if your data modalities are asynchronous, might be missing at inference, or are best processed by specialized models; it is simpler but may miss cross-modal interactions.
Q3: What are the common data alignment issues in multimodal plant studies? Challenges include temporal misalignment (e.g., RGB images and hyperspectral scans taken at different times) and spatial misalignment (e.g., different resolutions or fields of view). Furthermore, data from various sensors may have different sampling rates, requiring synchronization [41] [39].
Q4: How can I handle missing modalities in my dataset during training? A technique called Modality Dropout can be used. During training, one or more modalities are randomly dropped or obscured in each iteration. This forces the model to adapt and learn robust representations, enabling it to make reasonable predictions even when some data is missing at inference time [38].
Q5: Why does my multimodal model perform well in the lab but poorly in the field? This is a common issue often due to the domain gap between controlled lab conditions and variable field environments. Field data introduces new challenges like complex backgrounds, varying illumination, and occlusions. Techniques such as data augmentation, domain adaptation, and using more robust architectures (e.g., Transformers) can help bridge this gap [9] [42].
Troubleshooting Guides
Issue 1: Model Performance is Poor with Early Fusion
- Potential Cause: High-dimensional and noisy input space due to concatenating raw data [40] [39].
- Solution:
- Apply rigorous preprocessing to each modality to reduce noise (e.g., artifact removal, filtering) [41].
- Implement dimensionality reduction techniques (e.g., PCA) on the raw features before fusion.
- Consider switching to an intermediate fusion strategy, which is often more robust for learning from heterogeneous data [38].
Issue 2: Inconsistent Results When One Sensor Data is Missing
- Potential Cause: The model was trained on a complete dataset and cannot handle missing inputs [38].
- Solution:
Issue 3: Difficulty Combining Image and Numerical Sensor Data
- Potential Cause: The features from different modalities exist in incompatible scales and representations [3].
- Solution:
- Normalize all features into a common numerical range. For image data, this means pixel normalization; for sensor data, use Z-score or Min-Max scaling [43].
- Process each modality into a common embedding space (e.g., convert both images and sensor readings into 128-length vectors) before fusion, which is the core of intermediate fusion [38].
Issue 4: Low Accuracy in Real-World Field Deployment
- Potential Cause: The model has overfitted to clean, lab-condition data and fails to generalize [9] [42].
- Solution:
- Use datasets that contain field-environment variations (e.g., PlantVillage, PlantDoc).
- Employ data augmentation techniques during training to simulate field conditions (e.g., background substitution, lighting changes, occlusions).
- Utilize more robust model architectures like Vision Transformers (ViTs) or hybrid ViT-CNN models, which have shown better performance in field conditions compared to traditional CNNs [9].
Fusion Strategy Comparison
The table below summarizes the key characteristics of the three primary fusion strategies to guide your selection.
| Feature | Early Fusion | Intermediate Fusion | Late Fusion |
|---|---|---|---|
| Integration Stage | Input / Data Level [38] [40] | Feature Level [38] [39] | Decision / Output Level [38] [40] |
| Information Captured | Low-level, raw interactions [40] | High-level, complex modal interactions [38] | High-level decisions from each modality [39] |
| Handling Missing Data | Poor [39] | Difficult [39] | Good [38] [39] |
| Computational Complexity | Can be complex due to high-dimensional input [40] [39] | High due to joint representation learning [38] | Lower, as models can be trained in parallel [40] |
| Best For | Tightly synchronized, homogeneous modalities [38] | Learning complementary features between modalities [38] | Asynchronous data or when modularity is key [38] [40] |
Experimental Protocol: Implementing a Multimodal Fusion Pipeline for Plant Disease Detection
This protocol outlines a methodology for benchmarking fusion strategies using RGB and hyperspectral images.
1. Hypothesis: Intermediate fusion will yield superior accuracy for early plant disease detection by effectively combining visible symptoms from RGB images with pre-symptomatic physiological changes from hyperspectral data.
2. Data Acquisition & Preprocessing:
- Modality 1 (RGB Images): Capture high-resolution images of plant leaves under controlled lighting. Apply preprocessing including background removal, resizing, and normalization [42].
- Modality 2 (Hyperspectral Data): Collect hyperspectral cubes. Key preprocessing steps include:
- Artifact Removal & Outlier Detection: Use Median Absolute Deviation (MAD) to identify and remove outliers [41].
- Filtering: Apply a Moving Average (MA) filter to reduce noise [41].
- Interpolation: Use Piecewise Cubic Hermite Interpolating Polynomial (PCHIP) to handle missing data points [41].
- Normalization: Normalize the mean pupil diameter (or analogous spectral signal) using linear regression [41].
3. Feature Extraction:
- RGB: Use a pre-trained CNN (e.g., ResNet50) to extract a feature vector from the final layer before classification [9].
- Hyperspectral: Use a custom CNN or pre-trained feature extractor to process the spectral bands and generate a feature vector [9].
4. Fusion Implementation:
- Early Fusion: Concatenate the raw hyperspectral data (flattened) with the RGB pixel data (flattened) to create a single, high-dimensional input vector. Train a classifier on this combined vector.
- Intermediate Fusion: Concatenate the 512-length feature vector from the RGB model with the 256-length feature vector from the hyperspectral model. Feed this combined 768-length vector into a final classification layer.
- Late Fusion: Train two separate classifiers: one on the RGB features and one on the hyperspectral features. Combine their final prediction scores (e.g., softmax probabilities) using a weighted average or a meta-classifier.
5. Evaluation: Evaluate all models on a held-out test set using metrics appropriate for imbalanced data [42]:
- Primary Metric: F1-Score (macro-average).
- Secondary Metrics: Precision, Recall, and Balanced Accuracy.
Data Preprocessing Workflow
The following diagram illustrates a standardized preprocessing pipeline for transforming raw, multimodal data into a fusion-ready format.
Multimodal Fusion Architectures
This diagram visualizes the core architectural differences between early, intermediate, and late fusion strategies.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Multimodal Research |
|---|---|
| iMotions Platform | A multimodal platform that facilitates synchronized data collection from various sensors, such as eye trackers and facial expression analysis software, which is crucial for acquiring aligned datasets [41]. |
| Standardized Preprocessing Pipeline (e.g., SurvBench) | An open-source pipeline (like SurvBench for EHR data) that transforms raw data from multiple sources into standardized, model-ready tensors, ensuring reproducibility and fair model comparison [43]. |
| Modality Dropout | A regularization technique used during model training where one or more input modalities are randomly omitted. This enhances model robustness, allowing it to perform reasonably even when some data is missing at inference time [38]. |
| Graph-Based API (e.g., Pliers) | A framework that allows for the construction of complex, multi-step preprocessing workflows as directed acyclic graphs (DAGs). This simplifies the management of feature extraction and transformation across different modalities [3]. |
| Explicit Missingness Masks | A data structure that accompanies the main dataset, explicitly indicating which values were originally missing and subsequently imputed. This provides the model with crucial information about data quality and reliability [43]. |
Frequently Asked Questions & Troubleshooting Guides
This section addresses common challenges encountered when building a data preprocessing pipeline for multimodal plant organ classification, based on a thesis research context.
FAQ 1: Why does my model perform poorly despite using images of multiple plant organs?
- Problem: The model's classification accuracy is low, even though you are providing images of different organs (e.g., leaves, flowers, stems).
- Diagnosis: This is often due to a suboptimal fusion strategy for combining the features from the different organ modalities. Simply combining decisions at the end (late fusion) may not leverage the complementary relationships between organs effectively [37].
- Solution: Implement an automated fusion strategy. Instead of manually choosing a fusion point (early, intermediate, or late), use a Neural Architecture Search (NAS) method tailored for multimodal problems, such as a modified Multimodal Fusion Architecture Search (MFAS). This automatically discovers the most effective way to integrate features from different plant organs, which has been shown to significantly boost accuracy [37].
FAQ 2: How can I create a multimodal dataset from existing public plant image data?
- Problem: A lack of dedicated multimodal datasets hinders model development, as most public resources contain only one image per plant instance [37].
- Diagnosis: Researchers need a method to restructure unimodal datasets for multimodal tasks.
- Solution: Implement a data preprocessing pipeline to transform a unimodal dataset. The following methodology was successfully applied to create the Multimodal-PlantCLEF dataset from PlantCLEF2015 [37]:
- Data Collation: Gather all available images for each unique plant specimen in the original dataset.
- Organ Categorization: Manually annotate or filter these images based on the plant organ depicted (e.g., flower, leaf, fruit, stem).
- Specimen-Level Grouping: For each plant specimen, create a data entry that links to its corresponding set of organ-specific images. Specimens missing one or more organ types can still be used with techniques like multimodal dropout to improve model robustness [37].
FAQ 3: What is the recommended dataset size for training a deep learning model in this domain?
- Problem: Uncertainty about how many images are needed to train a robust and generalizable model.
- Diagnosis: Data requirements vary based on task complexity and model architecture.
- Solution: Refer to the following quantitative guidelines for dataset sizing and expansion [6]:
| Task Complexity | Minimum Recommended Images per Class | Notes |
|---|---|---|
| Binary Classification | 1,000 - 2,000 | Covers two classes (e.g., healthy vs. diseased) [6] |
| Multi-class Classification | 500 - 1,000 | Required number may increase with the total number of classes [6] |
| Object Detection | Up to 5,000 per object | More complex tasks require larger datasets [6] |
| Deep Learning (CNNs) | 10,000 - 50,000 (total) | Very large models may require 100,000+ images [6] |
| Transfer Learning | 100 - 200 per class | Effective for smaller datasets [6] |
To effectively expand your dataset, employ data augmentation techniques. This can multiply your usable dataset size by 2 to 5 times. Recommended augmentations for plant images include random rotation, flipping, contrast adjustment, and scaling to improve model adaptability and prevent overfitting [6].
FAQ 4: How do I achieve accurate alignment when using multiple different camera sensors?
- Problem: Pixel-precise alignment (registration) of images from different cameras (e.g., RGB, multispectral, depth) is challenging due to parallax and occlusion effects in plant canopies [31].
- Diagnosis: Traditional 2D registration methods that rely on plant-specific image features are often insufficient.
- Solution: Implement a 3D multimodal image registration algorithm.
- Data Acquisition: Use a monitoring system that includes a Time-of-Flight (ToF) or other depth camera to capture 3D information of the plant canopy [31].
- 3D Integration: Leverage the depth data in the registration algorithm to mitigate parallax effects, facilitating accurate pixel alignment across different camera modalities [31].
- Occlusion Handling: Integrate an automated mechanism to identify and filter out various types of occlusions, thereby minimizing registration errors [31]. This method is not reliant on specific plant features, making it suitable for a wide range of species [31].
Experimental Protocols & Methodologies
This section provides detailed methodologies for key experiments and procedures cited in the case study.
Protocol 1: Automatic Fused Multimodal Deep Learning for Plant Identification
This protocol outlines the method for building a plant classification model that automatically fuses data from multiple plant organs [37].
- Objective: To classify plant species by automatically finding the optimal fusion strategy for images of flowers, leaves, fruits, and stems.
- Materials:
- Dataset: A multimodal dataset (e.g., Multimodal-PlantCLEF) containing images categorized by plant organ [37].
- Software Framework: A deep learning library such as TensorFlow or PyTorch.
- Pre-trained Models: MobileNetV3Large or similar architectures as a feature extraction backbone.
- Procedure:
- Unimodal Model Training: First, train a separate convolutional neural network (CNN) model for each plant organ modality (flower, leaf, etc.) using a pre-trained model. Fine-tune each model on its specific organ class.
- Automatic Fusion: Apply a modified Multimodal Fusion Architecture Search (MFAS) algorithm. This algorithm will automatically search for and construct the optimal connections between the pre-trained unimodal models to create a single, fused multimodal architecture.
- Evaluation: Benchmark the performance of the automatically fused model against a standard late-fusion baseline (e.g., averaging predictions from individual organ models). Use standard metrics (accuracy, F1-score) and McNemar's statistical test for comparison [37].
Protocol 2: Two-Stage 3D Plant Organ Instance Segmentation
This protocol describes a generalized method for segmenting individual leaves and stems from 3D plant point clouds, applicable to both monocot and dicot species [44].
- Objective: To perform instance segmentation of plant organs (leaves, stems) from 3D point cloud data.
- Materials:
- 3D Plant Point Clouds: Data can be acquired via 3D scanners or LiDAR from plants like sugarcane, maize, and tomato [44].
- Computing Environment: A machine with a capable GPU for deep learning model training.
- Procedure:
- Semantic Segmentation: Train an improved PointNeXt model on the 3D point clouds to classify each point into semantic categories:
stem,leaf, orbackground. This step identifies what each point is, but not which specific leaf it belongs to [44]. - Leaf Instance Segmentation: Apply the Quickshift++ algorithm to the points classified as "leaf." This algorithm encodes the global spatial structure and local connections of the plant to cluster leaf points into individual leaf instances [44].
- Validation: Evaluate the results using metrics like mean Precision (mPrec), mean Recall (mRec), mean F1-score (mF1), and mean Intersection over Union (mIoU) against manually annotated ground truth data [44].
- Semantic Segmentation: Train an improved PointNeXt model on the 3D point clouds to classify each point into semantic categories:
Research Reagent Solutions
The following table details key computational tools and data resources essential for building a fused plant organ classification pipeline.
| Item Name | Type | Function / Application |
|---|---|---|
| Convolutional Neural Network (CNN) [6] | Algorithm | A deep learning model that automatically extracts hierarchical features from plant images, eliminating the need for manual feature engineering. Essential for tasks like species identification and disease detection [6]. |
| Multimodal-PlantCLEF [37] | Dataset | A restructured dataset for multimodal learning, comprising images from multiple plant organs (flowers, leaves, fruits, stems). It supports the development of models requiring specific organ inputs [37]. |
| Plant Village Dataset [6] | Dataset | A widely used public resource containing plant images, primarily for disease diagnosis research. Serves as a valuable benchmark and training resource [6]. |
| PointNeXt Model [44] | Algorithm | A deep learning model designed for 3D point cloud data. It can be trained to perform semantic segmentation of plant organs (stems, leaves) from 3D scans [44]. |
| Quickshift++ Algorithm [44] | Algorithm | A clustering algorithm used for instance segmentation. It is applied to semantically segmented 3D point clouds to group points into individual organ instances (e.g., separate each leaf) [44]. |
| MobileNetV3 [37] | Pre-trained Model | A lightweight, efficient CNN architecture. Often used as a pre-trained backbone for feature extraction, especially beneficial for deployment on resource-limited devices like smartphones [37]. |
| Neural Architecture Search (NAS) [37] | Methodology | A technique that automates the design of neural network architectures. It can be tailored for multimodal problems to find the optimal fusion strategy, surpassing manually designed models [37]. |
| Time-of-Flight (ToF) Camera [31] | Hardware | A depth-sensing camera that captures 3D spatial information. It is integrated into multimodal systems to provide depth data for robust 3D image registration, mitigating parallax errors [31]. |
Experimental Workflow Visualization
The following diagram illustrates the logical sequence and core components of the automated fused multimodal deep learning pipeline for plant identification.
Automated Multimodal Plant Classification Pipeline
The following diagram outlines the two-stage methodology for 3D plant organ instance segmentation.
3D Plant Organ Instance Segmentation Workflow
Navigating Pitfalls: Strategies for Handling Noise, Missing Data, and Integration Challenges
Frequently Asked Questions
What is the practical difference between data imputation and architectural robustness techniques like multimodal dropout? Data imputation is a preprocessing step that fills in missing values before the data is fed to a model. Techniques include mean/median imputation or advanced methods like MICE and missForest [45]. In contrast, architectural robustness techniques like multimodal dropout are built into the model itself, allowing it to make predictions even when some input modalities are missing, without requiring any data filling [1]. Imputation creates a complete dataset, while multimodal dropout creates a flexible model.
Which data imputation method should I start with for heterogeneous plant omics data? For researchers new to imputation, k-Nearest Neighbors (kNN) and Multiple Imputation by Chained Equations (MICE) are strong starting points for heterogeneous data [45]. kNN is intuitive and model-free, making it suitable for diverse data types common in plant studies. MICE is particularly powerful for complex, mixed-type data (continuous clinical measures, categorical traits, etc.) as it models each variable separately according to its type [45].
How does multimodal dropout work, and why is it useful for plant classification? Multimodal dropout is a training technique where random subsets of modalities are temporarily "dropped" or set to zero during model training [1]. This forces the model to not become overly reliant on any single data type and learn robust representations from any available combination of inputs. In plant classification, this is particularly useful when images of specific organs (e.g., fruits or stems) are unavailable for some samples, as the model can still make accurate predictions using the available organs [1].
My model performs well in lab conditions but fails in the field. Could missing modality robustness help? Yes, this is a classic scenario where robustness techniques are valuable. Field conditions often mean certain data types (e.g., specific sensor data, high-quality leaf images) are missing or corrupted. Employing multimodal dropout during training simulates these real-world scenarios, preventing the model from developing a dependency on ideal, lab-only data and significantly improving field performance [42].
Troubleshooting Guides
Problem: Model Performance Degrades with Missing Data
Symptoms
- High accuracy on complete test sets but significant performance drop when any modality is missing
- Model cannot produce predictions if certain data types are unavailable
- Inconsistent performance across different field deployment scenarios
Diagnosis Steps
- Audit Your Training Data: Check if your training set only contains samples with all modalities present, which creates an unrealistic expectation for deployment.
- Test with Ablated Data: Systematically remove each modality from your test set and measure performance degradation.
- Check Model Dependency: Analyze feature importance scores to see if the model is overly reliant on specific modalities.
Solutions
- Implement Multimodal Dropout: During training, randomly omit entire modalities with a specified probability (e.g., 0.2 per modality) to force robust feature learning [1].
- Adopt Hybrid Fusion: Instead of only late fusion, implement intermediate fusion strategies that can handle partial inputs through learned compensation mechanisms.
- Use Advanced Imputation: For planned deployments with known missing data patterns, implement MICE or missForest imputation tailored to your specific data types [45].
Problem: Choosing the Wrong Imputation Method
Symptoms
- Introduced biases in downstream analysis
- Reduced statistical power in predictive models
- Inconsistent feature importance across different imputation methods
Diagnosis Steps
- Identify Missingness Mechanism: Determine if data is Missing Completely at Random (MCAR), Missing at Random (MAR), or Missing Not at Random (MNAR) through pattern analysis [45].
- Evaluate Impact: Train the same model on datasets imputed with different methods and compare performance metrics.
- Check Variable Types: Ensure your imputation method matches your data types (continuous, categorical, mixed).
Solutions
- For MCAR/MAR Data: Use MICE or missForest for high-dimensional multimodal data [45].
- For Small Datasets: Start with kNN imputation as it requires fewer parameters and computational resources.
- Validate Rigorously: Always evaluate multiple imputation methods using a test set with complete data only, and use statistical tests like McNemar's test to verify significant performance differences [45].
Experimental Protocols & Methodologies
Protocol: Implementing Multimodal Dropout for Plant Organ Classification
Research Context: This protocol details the methodology adapted from an automatic multimodal fusion approach for plant classification using images from multiple organs [1].
Materials Needed
- Multimodal plant dataset (e.g., Multimodal-PlantCLEF with flower, leaf, fruit, stem images)
- Deep learning framework (PyTorch/TensorFlow)
- Pre-trained models for each modality (e.g., MobileNetV3 for images)
Methodology
- Individual Modality Training: First, train separate models for each plant organ modality using standard supervised learning.
- Fusion Architecture Search: Implement multimodal fusion architecture search (MFAS) to automatically find optimal fusion points [1].
- Multimodal Dropout Integration: During fusion model training, implement modality-wise dropout where random subsets of modalities are zeroed out during each training iteration.
- Progressive Training: Start with lower dropout probabilities (0.1-0.3) and gradually increase to higher probabilities (0.5-0.7) as training progresses.
- Validation: Evaluate on a validation set with complete and incomplete modality combinations.
Expected Outcomes: The resulting model should maintain >80% of original accuracy even with up to 50% of modalities missing, significantly outperforming standard fusion approaches [1].
Protocol: Comparative Evaluation of Imputation Methods for Plant Phenotyping Data
Research Context: Systematic evaluation of imputation methods for handling missing values in multimodal plant data, adapted from methodologies used in clinical neuroscience [45] and multi-omics studies [46].
Materials Needed
- Multimodal plant dataset with intentional missing values
- Python with scikit-learn, missingpy, and fancyimpute libraries
- Computational resources for multiple imputation methods
Methodology
- Data Preparation: Start with a complete dataset and artificially introduce missing values (10-30%) under MCAR and MAR mechanisms.
- Method Implementation: Apply five core imputation methods:
- Mean/Median: Basic statistical imputation
- k-Nearest Neighbors (kNN): Distance-based imputation
- Multiple Imputation by Chained Equations (MICE): Regression-based
- missForest: Random forest-based
- Evaluation Framework: Train identical models (Random Forest, SVM) on each imputed dataset and compare performance on a held-out complete test set.
- Statistical Validation: Use McNemar's test to assess significant differences in classifier performance across imputation methods.
Expected Outcomes: MICE and missForest typically outperform simpler methods, with MICE achieving 5-15% higher accuracy for classification tasks on multimodal data [45].
Comparative Analysis Tables
Performance Comparison of Imputation Methods
Table 1: Comparative performance of different imputation methods on multimodal classification tasks
| Imputation Method | Accuracy Range | Best Classifier Pairing | Computational Complexity | Data Type Suitability |
|---|---|---|---|---|
| Mean/Median | 70-76% | SVM | Low | Continuous numerical data |
| k-Nearest Neighbors | 72-79% | Random Forest | Medium | Mixed data types |
| MICE | 76-81% | Logistic Regression | High | Complex mixed-type data |
| missForest | 74-80% | Random Forest | High | Mixed data types |
Data synthesized from comparative studies on multimodal biological data [45]
Multimodal Robustness Techniques Comparison
Table 2: Comparison of architectural approaches for handling missing modalities
| Technique | Handles Unseen Missing Patterns | No Retraining Required | Accuracy Preservation | Implementation Complexity |
|---|---|---|---|---|
| Data Imputation | Limited to trained patterns | Once trained | Variable (70-81%) | Medium |
| Multimodal Dropout | Generalizes to new patterns | >82% with 50% modalities missing [1] | High | |
| Late Fusion | Requires all modalities | Poor with missing data | Low | |
| Early Fusion | Cannot handle partial inputs | Fails with missing data | Medium |
Research Reagent Solutions
Table 3: Essential computational tools and methods for multimodal robustness research
| Reagent/Method | Function | Implementation Example |
|---|---|---|
| Multimodal Dropout | Prevents overreliance on specific modalities during training | Custom layer that randomly zeros full modalities during training [1] |
| MICE Imputation | Handles mixed data types through iterative regression | IterativeImputer in scikit-learn with different estimators per variable type [45] |
| missForest | Non-parametric imputation for complex data distributions | MissForest implementation from missingpy Python package [45] |
| Multimodal Fusion Search | Automatically finds optimal fusion architecture | Modified MFAS algorithm for plant organ modalities [1] |
| SHAP/LIME Explainers | Model interpretability with missing modalities | SHAP for weather data, LIME for image data in multimodal models [23] |
Mitigating Feature and Label Noise in Weakly Supervised Citizen Science Data
Troubleshooting Guides
Common Problems and Solutions
Problem: Model performance is poor due to label noise from non-expert annotators.
- Symptoms: High training accuracy but low validation/test accuracy, inconsistent model predictions on similar samples, model fails to generalize.
- Possible Causes: Label noise from non-expert citizen scientists, ambiguous samples where even experts might disagree, feature-dependent noise where certain data patterns are consistently mislabeled.
- Solutions:
- Implement a two-stage weakly supervised learning framework that distinguishes between informative hard samples and detrimental noisy samples [47].
- Use loss dynamics analysis: track how loss values for each sample change over training epochs rather than relying on single-epoch loss values [47].
- Apply sample selection methods with small-loss criteria, where samples with consistently small losses are treated as clean [47].
- For citizen science platforms, incorporate an "I don't know" option to allow volunteers to abstain from difficult classifications, reducing noise [48].
Problem: Model activates only on most discriminative features rather than full objects.
- Symptoms: Incomplete segmentation masks, model focuses on small parts of objects rather than entire regions of interest.
- Possible Causes: Class Activation Maps (CAM) in CNN architectures tend to activate only the most discriminative regions, insufficient global context in model architecture.
- Solutions:
- Use transformer-based architectures (e.g., Vision Transformer, Conformer) that capture global features through self-attention mechanisms [49].
- Implement background noise reduction techniques for attention maps, such as inputting CAM enhanced with attention maps into the loss function [49].
- Apply uncertainty estimation via response scaling (URN method) to identify and mitigate noisy pixels in segmentation masks [50].
Problem: Multimodal data fusion yields suboptimal results.
- Symptoms: Model performance with multiple data modalities is worse than using single modalities, modalities contradict each other.
- Possible Causes: Suboptimal fusion strategy, missing modalities in some samples, unequal contribution of different modalities.
- Solutions:
- Use automatic multimodal fusion architecture search (MFAS) to find optimal fusion points rather than relying on manual architecture design [1] [37].
- Implement multimodal dropout to maintain robustness when some modalities are missing [1] [37].
- For plant identification, fuse multiple plant organ images (flowers, leaves, fruits, stems) to capture complementary biological features [1].
Problem: Noisy pixels in pseudo-masks degrade segmentation performance.
- Symptoms: Segmentation boundaries are irregular, obvious mislabeling of pixels, performance plateaus despite training.
- Possible Causes: Noisy pseudo-labels from weakly supervised methods, error propagation from initial seeds, high-confidence errors that are difficult to correct.
- Solutions:
- Implement uncertainty-weight transform modules that assign different weights to pixels based on their estimated uncertainty [51].
- Use frequency-based methods to estimate pixel uncertainty rather than predefined thresholds [51].
- Apply loss reweighting strategies that reduce the influence of high-uncertainty pixels during training [51].
Performance Comparison of Noise Mitigation Techniques
Table 1: Quantitative performance of different noise mitigation methods on benchmark datasets
| Method | Dataset | Performance Metric | Result | Key Advantage |
|---|---|---|---|---|
| Two-Stage WSL Framework [47] | CEPRI 36-bus System | Dominant Instability Mode Identification Accuracy | Significant improvement over baseline | Distinguishes hard samples from noisy samples |
| Two-Stage WSL Framework [47] | Northeast China Power System (2131 buses) | Dominant Instability Mode Identification Accuracy | Significant improvement over baseline | Works on real-world large-scale systems |
| Background Noise Reduction for Attention Maps [49] | PASCAL VOC 2012 | Segmentation Accuracy (mIoU) | 70.5% (val), 71.1% (test) | Reduces background noise in attention weights |
| Background Noise Reduction for Attention Maps [49] | MS COCO 2014 | Segmentation Accuracy (mIoU) | 45.9% | Effective on complex datasets |
| Uncertainty-Weight Transform Module [51] | PASCAL VOC 2012 | Segmentation Accuracy (mIoU) | 69.3% | Dynamically transforms pixel uncertainty into loss weights |
| Uncertainty-Weight Transform Module [51] | MS COCO 2014 | Segmentation Accuracy (mIoU) | 39.3% | Adaptable to different datasets |
| Automatic Fused Multimodal DL [1] | Multimodal-PlantCLEF (979 classes) | Plant Identification Accuracy | 82.61% | Automatically finds optimal fusion strategy |
| Label Noise-Resistant Mean Teaching (LNMT) [52] | Fake News Detection | Detection Performance | Superior performance | Resistant to noise in weak labels |
Frequently Asked Questions (FAQs)
Q1: What is the fundamental difference between feature noise and label noise in citizen science data?
Label noise refers to incorrect annotations in the training data, where samples are assigned wrong categories. In citizen science, this occurs when non-expert volunteers misclassify samples [47] [48]. Feature noise refers to issues with the input data itself, such as image artifacts, ambiguous samples where classes overlap, or missing modalities in multimodal datasets [53] [1]. Both types of noise are prevalent in citizen science data and require different mitigation strategies.
Q2: How can we distinguish between "hard samples" and "noisy samples" since both may exhibit large losses during training?
Hard samples are correctly labeled examples that are difficult to learn due to complexity or ambiguity, while noisy samples have incorrect labels. The key differentiator is their loss dynamics throughout training: hard samples tend to show decreasing but fluctuating losses over epochs, while noisy samples maintain consistently high losses [47]. Advanced methods analyze the entire training loss trajectory rather than single-epoch values, and use auxiliary machine learning models to classify samples based on these dynamics [47].
Q3: What is the advantage of providing an "I don't know" option to citizen scientists?
The "I don't know" option enhances data quality by allowing volunteers to abstain from classifying ambiguous cases rather than guessing [48]. This provides valuable information about task difficulty and helps identify samples that need expert attention. Studies show this approach improves overall accuracy, particularly for true negative rates, and the abstentions themselves provide useful information entropy for dynamic task allocation [48].
Q4: How does multimodal learning help mitigate noise in plant identification tasks?
Multimodal learning integrates multiple data sources (e.g., images of different plant organs - flowers, leaves, fruits, stems), providing complementary information that reduces dependence on potentially noisy features from any single modality [1] [37]. If one modality is ambiguous or noisy, other modalities can compensate. Automatic fusion methods can optimally combine these modalities without manual design bias [1].
Q5: What are the main approaches for handling noisy pixels in weakly supervised semantic segmentation?
Table 2: Approaches for handling noisy pixels in weakly supervised semantic segmentation
| Approach | Methodology | Advantages | Limitations |
|---|---|---|---|
| Uncertainty Estimation via Response Scaling (URN) [50] | Scales prediction maps multiple times to estimate uncertainty, uses uncertainty to weight segmentation loss | Effectively mitigates high-confidence noisy pixels, state-of-the-art results | Predefined threshold may not generalize across datasets |
| Uncertainty-Weight Transform Module [51] | Frequency-based uncertainty estimation, dynamic weight assignment without fixed thresholds | Adaptable to different datasets, no need for predefined thresholds | Complex implementation, computationally expensive |
| Background Noise Reduction for Attention Maps [49] | Reduces background noise in attention weights by incorporating enhanced CAM into loss function | Addresses specific issue of background contamination in transformer-based methods | Specifically designed for attention-based architectures |
| Loss Reweighting [51] | Modifies loss function weights based on estimated noise level | Directly addresses the core problem, flexible implementation | Requires accurate uncertainty estimation |
Experimental Protocols and Methodologies
Two-Stage Weakly Supervised Learning Framework
Purpose: To mitigate label noise in dominant instability mode identification for power systems [47].
Workflow:
- Stage I - Noise Detection:
- Train base deep learning models on the noisy dataset
- Extract entire training loss dynamics (sequence of loss values over epochs) for each sample
- Train an auxiliary machine learning model to classify large-loss samples as either "hard" or "noisy" based on loss patterns
- Divide dataset into simple, hard, and noisy subsets
- Stage II - Semi-Supervised Learning:
- Use labeled simple and hard samples for supervised learning
- Exploit noisy samples as unlabeled data through virtual adversarial training (VAT)
- Learn a smooth model where similar inputs have similar outputs
Key Implementation Details:
- Use different patterns in loss dynamics: noisy samples maintain high losses, hard samples show decreasing but fluctuating losses
- Virtual Adversarial Training encourages smoothness by adding perturbations to inputs and maintaining consistent outputs
Diagram 1: Two-stage weakly supervised learning framework
Automatic Multimodal Fusion for Plant Identification
Purpose: To optimally fuse multiple plant organ images for robust identification [1] [37].
Workflow:
- Dataset Preparation:
- Transform unimodal dataset (PlantCLEF2015) into multimodal dataset (Multimodal-PlantCLEF)
- Organize images by plant organs: flowers, leaves, fruits, stems
- Ensure each sample has corresponding organ images where available
Unimodal Model Training:
- Train separate models for each modality using MobileNetV3Small pre-trained on ImageNet
- Extract features from each unimodal model
Multimodal Fusion Architecture Search:
- Apply modified Multimodal Fusion Architecture Search (MFAS) algorithm
- Automatically discover optimal fusion points and strategies
- Implement multimodal dropout for robustness to missing modalities
Evaluation:
- Compare against baseline late fusion with averaging strategy
- Use standard performance metrics and McNemar's statistical test
Key Implementation Details:
- MFAS explores different fusion strategies: early, intermediate, late, hybrid
- Multimodal dropout randomly masks modalities during training to improve robustness
Uncertainty-Weight Transform for Noisy Pixels
Purpose: To mitigate the impact of noisy pixels in weakly supervised semantic segmentation [51].
Workflow:
- Uncertainty Estimation:
- Generate multiple predictions for each image using response scaling
- Calculate frequency of each pixel being labeled as different classes
- Select class with maximum probability as correct classification
- Record maximum probability as confidence level
Threshold Determination:
- Perform statistical analysis on confidence levels of all pixels
- Select confidence level value with highest frequency as threshold
Weight Assignment:
- Transform uncertainty into loss weights through designed functions
- Assign higher weights to confidence levels above threshold
- Assign smaller weights to confidence levels below threshold
Model Training:
- Use weighted loss function during segmentation model training
- Dynamically update weights based on uncertainty estimates
Key Implementation Details:
- Uses frequency-based approach rather than predefined thresholds
- Adaptable to different datasets through multiple scales
Research Reagent Solutions
Table 3: Essential research reagents and computational tools for noise mitigation experiments
| Tool/Reagent | Type | Function | Example Applications |
|---|---|---|---|
| Conformer Architecture [49] | Neural Network Architecture | Hybrid CNN-Transformer model combining local and global features | Weakly supervised semantic segmentation, background noise reduction |
| MobileNetV3Small [1] | Pre-trained Model | Feature extraction from individual modalities | Multimodal plant identification, transfer learning |
| MFAS Algorithm [1] | Architecture Search | Automatically finds optimal multimodal fusion points | Plant identification with multiple organ images |
| Virtual Adversarial Training [47] | Regularization Technique | Improves model smoothness using unlabeled data | Semi-supervised learning with noisy samples |
| Uncertainty-Weight Transform [51] | Loss Weighting Module | Dynamically assigns weights based on pixel uncertainty | Noisy pixel mitigation in semantic segmentation |
| Response Scaling [50] | Uncertainty Estimation | Generates multiple predictions at different activation scales | Identifying high-confidence noisy pixels |
| Dynamic Task Allocation [48] | Crowdsourcing Strategy | Optimizes volunteer effort allocation based on entropy | Citizen science data collection with "I don't know" option |
| Label Noise-Resistant Mean Teaching [52] | Training Framework | Robust to label noise using teacher-student models | Fake news detection with weak supervision |
Diagram 2: Automatic multimodal fusion for plant identification
Troubleshooting Guides
FAQ: Common Data Integration Challenges
1. What are the most common causes of interoperability failure in multimodal plant datasets? Interoperability failures most frequently stem from a lack of standardized data formats and semantic heterogeneity, where the same terms have different meanings across datasets. Spatial and temporal biases in data collection, and the difficulty in integrating remote sensing data with traditional in-situ observations also pose significant challenges [7]. Achieving harmonization requires consistent use of community standards.
2. How can I handle missing data in my multimodal pipeline without compromising analysis? Rather than relying solely on simple imputation, a robust strategy involves implementing explicit missingness tracking. This means generating binary masks that record whether a value was originally observed or imputed, allowing analytical models to distinguish between true zeros and missing data. For time-series plant phenotyping data, advanced interpolation methods like the Piecewise Cubic Hermite Interpolating Polynomial (PCHIP) can be employed to handle gaps effectively [43] [41].
3. What is the best strategy for integrating data types with fundamentally different structures? A late integration strategy, specifically Ensemble Integration (EI), is highly effective for handling heterogeneous data structures. Instead of forcing all data into a uniform format early on (early integration), EI involves building separate local predictive models for each data modality (e.g., genomics, phenomics, environmental sensors). These models are then aggregated into a final, powerful ensemble model using methods like mean aggregation or stacking, thereby preserving the unique information within each modality [54].
4. How do I prevent data leakage when preparing my dataset for machine learning? Data leakage is a critical issue that invalidates model performance. To prevent it, you must enforce strict patient-level or, in the context of plant research, specimen-level data splitting. This ensures that all data points originating from the same biological individual (e.g., all measurements from the same plant across time) are assigned entirely to either the training, validation, or test set. This prevents the model from artificially learning the identity of the specimen rather than the underlying biological patterns [43].
5. My model performance varies wildly between datasets. How can I improve generalizability? Generalizability is often hampered by inconsistent preprocessing methodologies across studies. Implementing a standardized, configuration-driven preprocessing pipeline is key. Using a tool like SurvBench (adapted for plant data) ensures that every step—from temporal aggregation and feature selection to normalization and splitting—is reproducible and transparent. This allows for a fair comparison of models and a true assessment of their performance [43].
Experimental Protocols for Data Integration
Protocol 1: Implementing a Standardized Preprocessing Pipeline for Multimodal Data
This protocol outlines a method to transform raw, heterogeneous data into standardized, model-ready tensors, adapted from benchmarks in clinical data science for plant research [43].
- Data Source Identification & Ingestion: Identify all data sources (e.g., species occurrence databases, spectral imaging, soil sensor data, trait databases). Use or develop data loaders to ingest raw data directly from its source to ensure transparency.
- Modality-Specific Processing:
- Time-Series Data (e.g., sensor data): Apply temporal aggregation (e.g., hourly or daily binning). Handle missing points via interpolation (e.g., PCHIP) and generate missingness masks.
- Static Data (e.g., species, genotypes): Encode categorical variables.
- Text Data (e.g., scientific literature, notes): Extract and convert into numerical embeddings.
- Data Harmonization: Map all data to a common standard. For biodiversity data, use the Darwin Core standard to unify terms and formats, which is critical for interoperability [7].
- Quality Control & Splitting: Apply data quality controls (e.g., outlier detection using Median Absolute Deviation). Split the entire dataset into training, validation, and test sets at the specimen-level to prevent data leakage.
- Output Standardized Tensors: The final output should be a set of harmonized tensors (for static and time-series data) alongside their missingness masks, ready for input into machine learning models.
Protocol 2: Ensemble Integration for Predictive Modeling from Multimodal Data
This protocol uses a late integration approach to build a robust predictive model from disparate data types [54].
- Local Model Training: For each individual data modality in your plant dataset (e.g., genomic, phenomic, environmental), train multiple local predictive models using a diverse set of algorithms (e.g., Decision Trees, Support Vector Machines, Random Forests). Use default parameters or cross-validate to tune them.
- Base Prediction Generation: Use each trained local model to generate prediction scores on the validation and test sets.
- Ensemble Aggregation: Integrate these base predictions into a final, global model using one of the following heterogeneous ensemble methods:
- Mean Aggregation: Calculate the mean of all base prediction scores.
- Stacking: Use the base predictions as new input features to train a second-level "meta-predictor" (e.g., a logistic regression model).
- Iterative Ensemble Selection: Iteratively add the local model that most improves the ensemble's performance on a validation set.
- Model Interpretation: Apply interpretation methods (e.g., feature importance analysis) to the final ensemble model to identify which features from which modalities were most influential in the predictions, providing biological insights.
Data Presentation
Table 1: Quantitative Comparison of Data Integration Strategies
| Integration Strategy | Description | Advantages | Disadvantages |
|---|---|---|---|
| Early Integration | Combines raw data from all modalities into a single, uniform representation (e.g., a fused network) before modeling [54]. | Simpler model architecture; can capture fine-grained interactions between modalities. | Reinforces consensus, potentially losing exclusive signals; difficult with heterogeneous data structures [54]. |
| Intermediate Integration | Jointly models multiple datasets through a shared, uniform latent representation [54]. | Can extract a powerful, condensed feature set. | May obscure modality-specific (local) information; complex to implement [54]. |
| Late Integration (Ensemble) | Builds separate models on each modality and aggregates their outputs [54]. | Preserves exclusive local information from each modality; highly flexible and often more accurate [54]. | Requires training multiple models; interpretation of the final ensemble can be complex. |
Table 2: Essential Research Reagent Solutions for Data Integration
| Item | Function in the Research Pipeline |
|---|---|
| Darwin Core Standard | A standardized framework of terms and definitions that enables the harmonization and exchange of biodiversity data, crucial for achieving interoperability [7]. |
| Species Distribution Models (SDMs) | Computational tools that use species occurrence data and environmental variables to model and predict the geographic distribution of species [7]. |
| Explicit Missingness Masks | Binary matrices that track which data values were originally observed versus imputed, providing the model with crucial information about data quality [43]. |
| Heterogeneous Ensemble Algorithms | Methods (e.g., Stacking, Mean Aggregation) that combine predictions from different types of models trained on various data modalities into a single, robust prediction [54]. |
| Configuration-Driven Pipeline | A reproducible data processing framework (e.g., defined by YAML files) that ensures every preprocessing decision is documented and can be exactly replicated [43]. |
Workflow Visualization
Data Integration Workflow
Ensemble Integration Architecture
Computational and Storage Optimization for Large-Scale Multimodal Datasets
Frequently Asked Questions (FAQs)
Q1: Our team is experiencing extremely long data loading times during training, creating a major bottleneck. What are the primary strategies to improve data throughput?
A1: Slow data loading is typically caused by insufficient I/O bandwidth or inefficient data formats. To address this:
- Utilize High-Throughput Storage Protocols: For local clusters, implement parallel file systems like Lustre or GPFS designed for concurrent read/write operations. In the cloud, use high-performance object storage such as Tencent Cloud COS or AWS S3 with optimized, S3-compatible APIs that provide gigabytes per second (GB/s) throughput [55].
- Optimize Data Formats: Convert countless small files (e.g., individual images) into larger, consolidated formats like TFRecord or HDF5. This reduces the metadata overhead and number of read operations, significantly accelerating data retrieval [55] [43].
- Implement a Memory Bank: For repetitive data, like user history in sequential models, a memory bank mechanism that incrementally caches and reuses computed multimodal representations can save a huge amount of computations and I/O operations [56].
Q2: How should we structure our storage to handle the diverse data types in a multimodal plant dataset (images, genomic sequences, environmental sensor data)?
A2: A hybrid storage strategy ensures optimal performance for different data types [55].
- Object Storage: Use scalable object storage (e.g., Tencent Cloud COS, AWS S3) for the vast majority of unstructured data, such as raw images, video footage, and audio recordings. This is ideal for scalable, durable storage [55] [57].
- Block Storage: Reserve high-performance block storage (e.g., Tencent Cloud CBS, AWS EBS) for structured data that requires low-latency access, such as database files, annotation files, and frequently accessed model checkpoints [55].
- Unified Metadata Management: Ensure your storage solution has efficient metadata indexing capabilities. A "metadata-rich unstructured data lake" in Amazon S3 or similar, where all data is tagged with consistent metadata (e.g., plant species, treatment, date), is crucial for accelerated data retrieval and cross-referencing [58] [57].
Q3: What are the best practices for handling missing data in multimodal datasets to avoid biasing our machine learning models?
A3: Proper missing data handling is critical for model robustness.
- Explicit Missingness Tracking: Beyond simple imputation (e.g., mean, median), your preprocessing pipeline should generate explicit missingness masks. These are binary matrices that record whether a value was originally observed or imputed, allowing the model to distinguish between true zeros and missing data. Frameworks like SurvBench implement this to prevent models from learning spurious patterns from imputed values [43].
- Rigorous Data Quality Controls: Establish and automate data quality checks at ingestion. This includes validating data ranges, formats, and completeness against predefined schemas. Platforms like Dataloop orchestrate this process, flagging anomalies for review before they enter the training pipeline [57].
Q4: We are concerned about data leakage because some plant images come from the same genetic line. How can we prevent this in our preprocessing pipeline?
A4: Data leakage invalidates model evaluation. It is prevented through careful data splitting.
- Enforce Subject-Level Splitting: Ensure that all data samples originating from the same entity (a patient in healthcare, a specific genetic line or plant specimen in your research) are assigned to the same data split (training, validation, or test). This prevents the model from seeing correlated data from the same source during training and evaluation, ensuring a realistic assessment of generalizability. Standardized pipelines like SurvBench enforce this by default [43].
Troubleshooting Guides
Issue: Slow or Inefficient Multimodal Preprocessing Pipeline
Symptoms: The pipeline takes hours or days to process a dataset; CPU and GPU utilization are low; the workflow does not scale with added data.
Diagnosis and Resolution:
| Step | Action | Technical Details |
|---|---|---|
| 1 | Profile the Pipeline | Use profiling tools to identify the bottleneck. Common culprits are data conversion steps (e.g., video to frames), feature extraction on a single CPU, or slow I/O. |
| 2 | Adopt a Modular, Graph-Based Design | Refactor the pipeline into a Directed Acyclic Graph (DAG). Frameworks like Pliers represent each processing step (extractor, converter) as a node, enabling parallel execution of independent branches and easier debugging [3]. |
| 3 | Parallelize and Orchestrate | Use workflow orchestration tools (e.g., Apache Airflow, Kubeflow) to run independent processing tasks in parallel across multiple workers. For GPU-bound tasks like feature extraction, NVIDIA NIM microservices can be integrated to accelerate inference [57]. |
| 4 | Implement Caching | Cache the results of expensive, idempotent operations (e.g., converting a video to keyframes, extracting embeddings from a static image). This avoids recomputing the same output repeatedly in subsequent pipeline runs [3] [56]. |
Diagram: Optimized Multimodal Preprocessing Workflow
Issue: Storage System Becomes a Bottleneck During Model Training
Symptoms: Training jobs frequently stall waiting for data; high network latency; inability to scale training to more nodes.
Diagnosis and Resolution:
| Step | Action | Technical Details |
|---|---|---|
| 1 | Audit Storage Performance | Check if your storage solution provides the required Input/Output Operations Per Second (IOPS) and throughput (GB/s) for your data load. Cloud dashboards often provide these metrics. |
| 2 | Upgrade Storage Protocol | Move from traditional file protocols (NFS) to those designed for high performance, such as NVMe over Fabrics (NVMe-oF) for low-latency block storage or object storage with S3-compatible APIs optimized for fast metadata operations [55]. |
| 3 | Optimize Data Locality | Co-locate your compute nodes and data storage in the same cloud availability zone or data center rack to minimize network latency. For on-premise HPC clusters, use a parallel file system that is physically connected to the compute nodes with high-speed networking [55]. |
| 4 | Leverage a Memory Bank | For sequential recommendation tasks, a memory bank stores pre-computed historical representations, drastically reducing read operations and transmission bottlenecks during training [56]. |
Diagram: Storage Architecture for Multimodal Data
The Scientist's Toolkit: Research Reagent Solutions
Table: Essential Components for a Multimodal Data Preprocessing Pipeline
| Item/Reagent | Function in the Experimental Pipeline |
|---|---|
| Directed Acyclic Graph (DAG) Orchestrator | Defines and manages the sequence of preprocessing steps, allowing for parallel execution, branching logic, and reproducible workflows [3]. |
| Specialized Processing Agents | Modular software agents (e.g., for classification, conversion, metadata extraction) handle specific data types, enabling targeted processing and easier debugging [58]. |
| High-Performance Object Storage | Provides scalable and durable storage for massive volumes of unstructured data (images, video) with high throughput for concurrent access [55]. |
| Low-Latency Block Storage | Delivers fast, millisecond-level access for structured data and model checkpoints, preventing I/O bottlenecks during training [55]. |
| Explicit Missingness Mask | A binary matrix generated during preprocessing that records which data points were observed vs. imputed, preventing model bias [43]. |
| Memory Bank Mechanism | A caching system that stores and incrementally updates computed multimodal representations, drastically reducing computational and I/O overhead for sequential data [56]. |
| Human-in-the-Loop Interface | A platform for domain experts to validate automatically extracted metadata, correct errors, and provide labeled data for continuous pipeline improvement [58]. |
Ensuring Data Privacy and Governance in Collaborative Research Environments
Frequently Asked Questions
1. What are the core data privacy principles we must follow in research? All research must adhere to the principles outlined in GDPR Article 5, which require that data processing is lawful, fair, and transparent. Key principles include purpose limitation (collecting data for specified purposes), data minimization (only processing data necessary for the purpose), and storage limitation (retaining data only as long as necessary) [59].
2. How can we ensure our multimodal plant dataset is GDPR-compliant? Begin by conducting a Privacy Impact Assessment (PIA) to identify and mitigate privacy risks [60] [61]. Ensure you have a valid legal basis, such as informed consent that explicitly covers data sharing with collaborators. In your data management plan, document how you will implement data minimization, for instance, by pseudonymizing data and only sharing the specific plant organ images required for the research task [59] [62].
3. What technical measures protect data in a collaborative cloud platform? A secure research data platform should use encryption for data both in transit and at rest. Access should be controlled via multi-factor authentication and strict role-based permissions. To prevent data leaks, deploy a Data Loss Prevention (DLP) solution. Where possible, grant access to a secure central infrastructure rather than transferring raw data files [60] [59].
4. Our consortium includes a commercial partner. What agreements are needed? When multiple organizations determine the "why" and "how" of data processing, they are likely joint controllers. You must formalize roles and responsibilities in a joint controllers agreement. This agreement should define each party's data protection duties, who handles data subject requests, and the main contact for data subjects [62].
5. How do we securely transfer data to collaborators outside the EU? Transferring personal data outside the European Economic Area requires extra measures. You must use a secure method like SURF Filesender with encryption and put additional agreements in place to ensure the recipient is GDPR-compliant. Always consult your privacy officer for international transfers [62].
6. What should we do if a data breach occurs? Activate your incident management procedures immediately. Your plan should include steps for breach containment, reporting to authorities, and communication with affected data subjects. Regular tabletop exercises will prepare your team to handle a real incident effectively [61].
Research Reagent Solutions: Data Governance Toolkit
| Tool / Solution | Primary Function in Research | Relevance to Multimodal Plant Data |
|---|---|---|
| Data Processing Agreement | Legally defines roles (controller/processor) and data protection responsibilities [62]. | Governs data sharing between university researchers and commercial AI partners. |
| Privacy Impact Assessment (PIA) | Identifies and reduces privacy risks before project start [61]. | Assesses risks of combining flower, leaf, fruit, and stem images from various sources. |
| Joint Controllers Agreement | Formalizes governance when multiple parties decide on data processing purposes and methods [62]. | Essential for research consortia where different teams manage and analyze the dataset. |
| Pseudonymization | Replaces identifying fields with pseudonyms to reduce linkage risk [59]. | Applied to location and collector data in plant images to enable analysis while protecting sources. |
| Web Application Firewall (WAF) | Protects web-facing data platforms from exploitation and data theft [60]. | Secures the online portal hosting the Multimodal-PlantCLEF dataset from cyber attacks. |
Experimental Protocol: Creating a Compliant Multimodal Dataset
This protocol outlines the methodology for building a multimodal plant dataset, such as Multimodal-PlantCLEF, in a privacy-conscious manner [1].
1. Data Sourcing and Legal Basis
- Source Selection: Identify and use existing datasets (e.g., PlantCLEF2015) that have a known provenance and a clear legal basis for secondary research use [1].
- Legal Grounds Verification: Confirm that the original data collection obtained informed consent that permits the creation of derived datasets and sharing within research consortia. If consent is insufficient, seek expert advice on alternative legal bases like public interest [62].
2. Data Preprocessing and Minimization
- Organize by Modality: Restructure the source data, grouping images by plant organ (e.g., flowers, leaves, fruits, stems). Each organ is treated as a distinct modality [1].
- Apply Data Minimization: Curate the dataset to include only the image types and metadata fields strictly necessary for the plant classification task. Remove any extraneous personal data about data collectors or specific geolocations that are not essential [59].
3. Pseudonymization and Packaging
- Assign Pseudonyms: Replace any direct identifiers (e.g., original image filenames that contain personal names) with a consistent, random code for each plant specimen [59].
- Create Data Packages: Assemble the final dataset into structured packages where each entry contains the pseudonymized identifier and the set of organ images for one plant specimen [1].
4. Documentation and Governance
- Update Privacy Notice: Ensure the dataset's privacy notice clearly states its use in multimodal research, the types of data processed, and the identities of collaborating partners [61].
- Establish Access Controls: Define and implement role-based access rights to the dataset, ensuring collaborators can only access the data necessary for their role [62].
| Regulation / Principle | Core Requirement | Application in Research |
|---|---|---|
| Lawfulness, Fairness, and Transparency (GDPR Art. 5) | Process data lawfully, inform subjects about processing [59]. | Inform subjects about data sharing in collaborative projects in clear, plain language [61]. |
| Purpose Limitation (GDPR Art. 5) | Collect data for specified, explicit, and legitimate purposes [59]. | Only use consented plant data for the predefined goal of multimodal classification research. |
| Data Minimization (GDPR Art. 5) | Data must be adequate, relevant, and limited to what is necessary [59]. | Share only specific organ images (e.g., only leaves and flowers) needed by a collaborator. |
| Storage Limitation (GDPR Art. 5) | Keep data in an identifiable form only as long as necessary [59]. | Define and enforce a data retention schedule, deleting raw data after derived features are created [61]. |
| Accountability (GDPR Art. 5) | The controller is responsible for and must demonstrate compliance [59]. | Maintain records of processing activities and conduct regular data audits to prove compliance [61]. |
Data Collaboration Workflow
Measuring Success: Benchmarking, Explainability, and Comparative Performance Analysis
Frequently Asked Questions (FAQs)
1. What is the fundamental purpose of splitting my plant dataset into training, validation, and test sets?
The core purpose is to develop a model that generalizes well to new, unseen data. The training set is used to fit the model's parameters, the validation set is used to provide an unbiased evaluation for tuning hyperparameters and selecting the best model during training, and the test set is used only once to provide a final, unbiased assessment of the model's performance on truly unseen data. This strict separation prevents information leakage and overly optimistic performance metrics, ensuring your model for plant disease identification or compound efficacy will be reliable in real-world scenarios. [63] [64]
2. My multimodal plant dataset is highly imbalanced (e.g., many healthy leaf images, few diseased). What is the best splitting strategy to use?
For imbalanced datasets, a standard random split is not appropriate as it can create biased splits. You should use stratified dataset splitting. This method preserves the relative proportions of each class (e.g., disease type, treatment outcome) across the training, validation, and test sets. This ensures that your model is trained and evaluated on a representative subset from each class, leading to more robust and reliable performance metrics for all categories in your research. [64]
3. How much data should I allocate to the training, validation, and test sets?
There are no universal fixed rules, but common practices provide a strong starting point. The optimal ratio often depends on your dataset's total size. The table below summarizes common split ratios: [64] [65]
| Dataset Size Scenario | Typical Training % | Typical Validation % | Typical Test % |
|---|---|---|---|
| Standard Starting Point | 70-80% | 10-15% | 10-15% |
| Very Large Dataset (>>1M samples) | ~98% | ~1% | ~1% |
| Smaller Dataset | 80-90% | 5-10% | 5-10% |
4. What is the difference between a simple train-validation-test split and k-fold cross-validation?
A train-validation-test split performs a single, static split of your data. While simple and computationally efficient, the resulting model performance can be highly dependent on that particular random split. [63] [65] K-fold cross-validation is a more robust technique that divides the data into K folds (e.g., 5 or 10). The model is trained K times, each time using a different fold as the validation set and the remaining folds as the training set. The final performance is the average of the K validation scores, providing a more reliable estimate of model performance and reducing the variance associated with a single data split. [63] [64]
5. I've heard of "nested cross-validation." When is it necessary for my research?
Nested cross-validation is the gold standard for performing both model selection and model evaluation in a single, unbiased workflow. It is particularly crucial when working with smaller datasets, as it is more prone to the idiosyncrasies of different splits. It involves two layers of cross-validation: an inner loop for hyperparameter tuning and model selection, and an outer loop for evaluating the selected model's performance. This method provides the most reliable performance estimate but is computationally very expensive. [63] For very large datasets or deep learning models, a single train-validation-test split is often sufficient and more practical. [63]
Troubleshooting Guides
Problem: Data Leakage Between Splits
Symptoms:
- Your model's performance on the validation and test sets is surprisingly and unrealistically high.
- The model performs excellently in validation but fails dramatically on new, real-world plant data.
- There is a significant performance drop between the validation and test sets.
Solutions:
- Ensure Strict Separation: The test set must be locked away and not used for any aspect of model development, including training, hyperparameter tuning, or feature selection. Information from the test set should never influence the model. [64]
- Preprocess Correctly: Always fit your data preprocessing steps (e.g., imputation, scaling) on the training data only. Then, use the parameters learned from the training set (like the mean and standard deviation) to transform the validation and test sets. Never fit a preprocessor on the entire dataset before splitting. [66]
- Shuffle Data Randomly: Before splitting, always shuffle your dataset to ensure splits are representative and not biased by the initial order of the data (e.g., all early samples from one lab or growth condition). [64] [65]
Problem: High Variability in Model Performance Across Different Splits
Symptoms:
- You get a different "best model" every time you run a new train-validation split.
- Model performance metrics (e.g., accuracy, F1-score) fluctuate widely with different random seeds.
Solutions:
- Implement K-Fold Cross-Validation: Instead of a single validation set, use k-fold cross-validation on your training data to tune hyperparameters. This provides a more stable estimate of model performance by averaging results across K different splits. [63] [64]
- Use Nested Cross-Validation: For the most rigorous and unbiased evaluation, especially with smaller multimodal plant datasets, implement nested cross-validation. This will give you a robust understanding of how your chosen model and preprocessing pipeline will generalize. [63]
- Increase Dataset Size: If possible, collect more data. The larger and more representative your dataset, the less impact a single random split will have on your results. [64]
Problem: Model is Overfitting to the Training Data
Symptoms:
- The model's performance on the training set is nearly perfect, but performance on the validation and test sets is significantly worse.
- The training loss continues to decrease, but the validation loss starts to increase after a certain point.
Solutions:
- Use the Validation Set for Early Stopping: Monitor the model's performance on the validation set during training. Halt the training process when validation performance stops improving, preventing the model from over-optimizing to the training data's noise. [64]
- Simplify the Model: Reduce model complexity by using fewer parameters, adding regularization (e.g., L1, L2), or increasing dropout rates in neural networks.
- Apply Data Augmentation: For image-based plant data, artificially expand your training set using techniques like rotation, flipping, and color jittering to teach the model more invariant features. [64]
Experimental Protocols & Methodologies
Protocol 1: Implementing a Standard Train-Validation-Test Split
This protocol is recommended for initial experiments, large datasets, or when computational resources are limited.
Methodology:
- Shuffle: Randomly shuffle the entire multimodal dataset.
- Initial Split: Perform an initial split (e.g., 80%/20%) to create a temporary "hold-out" test set. Set this test aside and do not use it for any model development.
- Secondary Split: Split the remaining data (e.g., the 80%) into training and validation sets (e.g., 85%/15% of the remainder, resulting in a final split of ~68% train, ~12% validation, ~20% test).
- Train & Tune: Train your model on the training set and use the validation set to tune hyperparameters.
- Final Training: Once hyperparameters are selected, retrain the model on the combined training and validation sets.
- Final Evaluation: Evaluate the final model exactly once on the held-out test set to report its generalization performance. [63] [64]
Protocol 2: Implementing K-Fold Cross-Validation
This protocol provides a more robust estimate of model performance and is ideal for model selection and tuning.
Methodology:
- Partition: Divide the entire dataset into K equally sized, random folds (common values for K are 5 or 10).
- Iterate: For each of the K iterations:
- Designate one fold as the validation set.
- Designate the remaining K-1 folds as the training set.
- Train the model on the training set and evaluate it on the validation set. Record the performance metric.
- Average: Calculate the average and standard deviation of the K performance metrics from the validation sets. This average is your cross-validation score, a reliable indicator of model performance. [63] [64]
Protocol 3: Implementing Nested Cross-Validation
This is the most rigorous protocol for obtaining an unbiased performance estimate when also performing model and hyperparameter selection.
Methodology:
- Define Outer Loop: Split the data into K folds (outer folds). These will be used for evaluation.
- Define Inner Loop: For each outer fold:
- Use one outer fold as the test set.
- Use the remaining K-1 outer folds as the development set.
- On this development set, perform an inner K-fold cross-validation (e.g., 5-fold) to tune the model's hyperparameters and select the best model configuration.
- Train a new model with the selected best configuration on the entire development set.
- Evaluate this final model on the held-out outer test fold and record the performance.
- Report: The final model performance is the average and standard deviation of the metrics obtained from the K outer test folds. [63]
Workflow Visualization
Nested Cross-Validation Workflow
The Scientist's Toolkit: Research Reagent Solutions
This table details key computational frameworks and tools essential for implementing robust validation frameworks in your research.
| Tool / Framework | Function | Key Characteristics for Research |
|---|---|---|
| Scikit-learn [67] [68] | Provides simple and efficient tools for data splitting, cross-validation, and implementing traditional ML models. | Excellent for prototyping. Offers train_test_split, KFold, StratifiedKFold, and GridSearchCV for automated hyperparameter tuning with cross-validation. |
| TensorFlow / PyTorch [67] [68] | Open-source libraries for developing and training deep learning models, commonly used for complex multimodal data (e.g., images, sequences). | High flexibility and control. TensorFlow's Keras API offers built-in support for validation splits and callbacks (e.g., Early Stopping). PyTorch requires more manual setup but is highly modular. |
| Weights & Biases (W&B) [63] | Experiment tracking and hyperparameter optimization platform. | Crucial for managing complex experiments. Logs metrics, hyperparameters, and model outputs across hundreds of runs, facilitating comparison and reproducibility. |
| Encord Active [64] | A platform specifically designed for computer vision projects, useful for managing image-based plant datasets. | Helps visualize and curate datasets, filter images based on quality metrics (blur, brightness), and create balanced training, validation, and test splits to reduce bias. |
| Hugging FaceTransformers [67] | A library providing thousands of pre-trained models, primarily for natural language processing (NLP). | If your multimodal data includes textual descriptions or scientific literature, this library allows you to fine-tune state-of-the-art models on your specific domain text. |
FAQs: Core Concepts and Metrics
1. What are the key performance metrics for evaluating a multimodal plant classification system, and why is accuracy alone insufficient? While classification Accuracy is a fundamental metric, a comprehensive evaluation for multimodal systems must also include Robustness and Generalization [69].
- Accuracy measures the correctness of predictions on a specific test set. For example, a multimodal plant ID model might achieve 82.61% accuracy on its target dataset [1] [37].
- Robustness is the model's ability to maintain performance despite variations in input data, such as image noise, missing modalities (e.g., a missing leaf image), or changes in lighting conditions [69].
- Generalizability assesses how well the model performs on new, unseen data from different distributions, such as images from a different geographic location or captured with a different camera [69]. A model can have high accuracy on its original test set but poor generalizability if it overfits to spurious correlations in the training data.
2. My multimodal model performs well in training but fails on new plant datasets. What strategies can improve generalizability? Poor generalization often stems from overfitting and a failure to learn universal features. Key strategies to address this include [69]:
- Data Augmentation: Artificially expand your training data with transformations (rotations, flipping, color adjustments) to simulate real-world variability.
- Regularization Techniques: Apply methods like Dropout and L2 Regularization during training to prevent the model from becoming over-reliant on specific neurons or features.
- Transfer Learning: Initialize your model with features learned from a large, general dataset before fine-tuning it on your specific plant dataset.
- Ensemble Learning: Combine predictions from multiple models to reduce variance and improve overall stability on unseen data.
3. How can I make my multimodal system robust to missing data, such as when images of a specific plant organ are unavailable? Robustness to missing modalities is a critical challenge. A primary solution is the use of multimodal dropout, a technique where modalities are randomly omitted during training. This forces the model to learn to make accurate predictions even when only a subset of its inputs (e.g., only a flower and a stem, but no leaf) is available, thereby enhancing its resilience for real-world deployment [1] [37].
4. What are the common data synchronization challenges in a multimodal plant phenotyping pipeline, and how can they be resolved? Synchronizing data from various sensors (e.g., multiple cameras, environmental sensors) is a common technical hurdle. Key issues and solutions include [10] [70]:
- Challenge: Sampling Rate Mismatch and Clock Drift. Different devices sample data at different rates, and their internal clocks can drift apart over time, leading to misalignment.
- Solution: Lab Streaming Layer (LSL). Utilize open-source frameworks like LSL, which provide a network-based infrastructure for synchronized multimodal data collection with built-in clock drift correction [70].
- Solution: Post-hoc Synchronization. Employ event markers (e.g., a visual flash or audio cue recorded by all systems) to manually align data streams during analysis, though this can be labor-intensive [10].
Troubleshooting Guides
Problem: Your multimodal deep learning model for plant disease diagnosis is showing low accuracy on the test set.
Investigation & Resolution Protocol:
| Step | Action | Diagnostic Cues & Resolution Strategies |
|---|---|---|
| 1. Data Quality Check | Inspect the preprocessing pipeline for label errors and data corruption. | Look for mislabeled plant species or misaligned image-text pairs. Use data validation scripts. |
| 2. Fusion Strategy Audit | Evaluate the method used to combine modalities (e.g., images and environmental data). | Late fusion is common but may be suboptimal [1]. Consider automated neural architecture search (NAS) for fusion, which has been shown to outperform simple late fusion by over 10% [1] [37]. |
| 3. Model Architecture Review | Check if the model capacity is sufficient for the task complexity. | For image modalities, pre-trained feature extractors like EfficientNetB0 have proven effective [23]. For text or sequential environmental data, RNNs or transformers can be used [23]. |
| 4. Hyperparameter Tuning | Systematically optimize learning rate, batch size, and optimizer settings. | Use adaptive optimizers like Adam [69]. Employ cross-validation to find optimal parameters and prevent overfitting. |
Issue: Poor Model Robustness
Problem: The model's performance degrades significantly when faced with noisy images, occluded plant parts, or when one data modality is missing.
Investigation & Resolution Protocol:
| Step | Action | Diagnostic Cues & Resolution Strategies |
|---|---|---|
| 1. Implement Multimodal Dropout | Intentionally drop one or more modalities during the training phase. | This trains the network to not become over-reliant on any single input source, significantly improving robustness to missing data at inference time [1] [37]. |
| 2. Augment Training Data | Introduce realistic noise and variations into your training set. | Apply techniques like noise injection, random erasing, and color space adjustments to mimic field conditions [69]. This improves the model's resilience to imperfect inputs. |
| 3. Adversarial Training | Expose the model to perturbed inputs during training. | This technique helps the model learn to resist small, malicious perturbations that could lead to incorrect predictions, thereby enhancing its stability [69]. |
Issue: Failure to Generalize to New Data
Problem: The model achieves high accuracy on its original test set but performs poorly on new plant datasets from different sources or environments.
Investigation & Resolution Protocol:
| Step | Action | Diagnostic Cues & Resolution Strategies |
|---|---|---|
| 1. Analyze Domain Shift | Characterize the differences between your training data and the new deployment environment. | Check for differences in image background, lighting, plant varieties, or sensor types. This identifies the source of the generalization failure. |
| 2. Apply Domain Adaptation | Use techniques to minimize the discrepancy between the source (training) and target (new) data distributions. | Algorithms can be used to learn features that are invariant across the source and target domains, improving performance on the new data [69]. |
| 3. Utilize Ensemble Methods | Combine predictions from multiple models trained with different initializations or on different data splits. | Techniques like bagging and boosting reduce model variance and can lead to more reliable performance on unseen datasets [69]. |
| 4. Regularization | Ensure sufficient regularization is applied to prevent overfitting. | Increase the strength of L2 regularization or dropout rates to force the model to learn more general, rather than dataset-specific, features [69]. |
Experimental Protocols & Benchmarking
Quantitative Benchmarking of Fusion Strategies
The following table summarizes key results from recent multimodal studies, primarily in plant science, which can serve as benchmarks for your own experiments.
| Model / Study | Task | Modalities | Fusion Strategy | Key Result / Accuracy |
|---|---|---|---|---|
| Automatic Fused Multimodal DL [1] [37] | Plant Identification (979 classes) | Images of flowers, leaves, fruits, stems | Multimodal Fusion Architecture Search (MFAS) | 82.61% (Outperformed late fusion by 10.33%) |
| PlantIF [15] | Plant Disease Diagnosis | Plant phenotype images & textual descriptions | Graph-based Interactive Fusion | 96.95% (1.49% higher than existing models) |
| Interpretable Tomato Diagnosis [23] | Tomato Disease & Severity | Leaf images & environmental data | Late Fusion (EfficientNetB0 + RNN) | Disease: 96.40%, Severity: 99.20% |
| Late Fusion Baseline [1] | Plant Identification | Images of flowers, leaves, fruits, stems | Late Fusion (Averaging) | ~72.28% (Baseline for comparison) |
Protocol: Evaluating Robustness to Missing Modalities
Objective: Quantify the performance drop when one or more input modalities are unavailable.
Methodology:
- Train your multimodal model using multimodal dropout.
- Create Evaluation Sets: From your test set, generate subsets where a specific modality (e.g., 'flower') is systematically set to zero or masked.
- Measure & Compare: Calculate the accuracy on each of these degraded test sets and compare it to the accuracy on the full, unimpaired test set. A robust model will show a minimal decrease in performance.
The Scientist's Toolkit: Research Reagent Solutions
| Item / Technique | Function in Multimodal Pipeline |
|---|---|
| Lab Streaming Layer (LSL) [70] | An open-source platform for synchronized, multimodal data acquisition from various hardware devices, solving issues of jitter and latency. |
| Multimodal Fusion Architecture Search (MFAS) [1] | An automated method for discovering the optimal neural network architecture to fuse different data modalities, replacing manual, suboptimal design. |
| Multimodal Dropout [1] [37] | A training technique that improves model robustness by randomly ablating entire input modalities, simulating real-world missing data scenarios. |
| Explainable AI (XAI) Tools (LIME & SHAP) [23] | Post-hoc interpretation tools (LIME for images, SHAP for tabular/weather data) to explain model predictions, building trust and providing biological insights. |
| Data Augmentation Techniques [69] | A set of transformations (geometric, color, noise) applied to training data to artificially increase dataset size and diversity, improving generalization. |
Workflow Diagrams
Multimodal Plant Data Preprocessing Pipeline
Evaluating Model Robustness and Generalization
In multimodal data analysis, which integrates diverse data types like images from different plant organs, the strategy for fusing these modalities is critical. Traditional Late Fusion and emerging Automated Fusion represent two distinct approaches. Late Fusion involves training separate models on each data type (e.g., leaves, flowers) and combining their final decisions, valued for its simplicity and robustness [1] [71]. Automated Fusion, leveraging techniques like the Multimodal Fusion Architecture Search (MFAS), automatically discovers the optimal method and point for integrating modalities within a model architecture [1]. This analysis compares these strategies within the context of a multimodal plant data preprocessing pipeline, providing a troubleshooting guide for researchers.
Core Concepts and Definitions
Traditional Late Fusion
Late Fusion, or decision-level fusion, entails training unimodal prediction models independently. The final predictions from these models are aggregated using a function, such as averaging or weighted voting, to produce a unified decision [72] [73]. Its modularity makes it adaptable to missing modalities and less prone to overfitting from weak data sources [71].
Automated Fusion
Automated Fusion employs neural architecture search (NAS) techniques to design a fusion strategy optimized for a specific task and dataset. Unlike pre-defined fusion methods (early, intermediate, late), it automatically determines how and where to combine features from different modalities, potentially discovering more complex and effective integration patterns [1].
Comparative Performance Analysis
The table below summarizes a quantitative comparison based on experimental results from plant identification and medical diagnostics research.
Table 1: Quantitative Comparison of Fusion Strategies
| Performance Metric | Traditional Late Fusion | Automated Fusion (MFAS) | Context and Notes |
|---|---|---|---|
| Top-1 Accuracy | Baseline (72.28%) | 82.61% | Plant identification on 979 classes [1] |
| Performance Gain | — | +10.33% over Late Fusion | Plant identification task [1] |
| Concordance Index (C-Index) | Improvement of 0.0143 over best unimodal model | Not Specified | Medical survival prediction; demonstrates Late Fusion robustness [71] |
| Robustness to Weak Modalities | High (maintains performance) | Not Specified | Late Fusion prevents overfitting when adding noisy/weak data [71] |
| Robustness to Missing Modalities | High (models are independent) | High (with multimodal dropout) | Automated approach can be designed for robustness [1] |
| Model Size (Parameter Count) | Typically larger (ensemble of models) | Significantly smaller | Automated search discovers more efficient architectures [1] |
Experimental Protocols and Methodologies
Protocol for Traditional Late Fusion in Plant Identification
This protocol outlines the baseline method for comparing fusion strategies [1].
- Data Preparation and Preprocessing: Restructure a unimodal plant dataset (e.g., PlantCLEF2015) into a multimodal one (Multimodal-PlantCLEF) where each sample comprises images of multiple specific plant organs (flowers, leaves, fruits, stems).
- Unimodal Model Training:
- Action: Train a separate, pre-trained convolutional neural network (CNN) like MobileNetV3Small on each individual plant organ modality.
- Troubleshooting Tip: If performance for a single organ is poor, verify the data quality for that modality and consider fine-tuning the pre-trained model more extensively.
- Decision Aggregation:
- Action: For each test sample, obtain the prediction scores from all unimodal models.
- Action: Fuse these scores by computing a simple average (or a weighted average based on validation performance).
- Troubleshooting Tip: If one modality is consistently unreliable, assign it a lower weight or exclude it from the fusion step.
Protocol for Automated Fusion via MFAS in Plant Identification
This protocol describes the automated method that searches for an optimal fusion strategy [1].
- Base Model Preparation:
- Action: First, train unimodal models for each plant organ type, as in the Late Fusion protocol. These serve as the starting blocks for the architecture search.
- Multimodal Fusion Architecture Search (MFAS):
- Action: Employ a modified MFAS algorithm to automatically explore different ways to connect and fuse the features from the unimodal models. The search space includes various fusion operations and connections.
- Troubleshooting Tip: This process is computationally intensive. Ensure access to sufficient GPU resources. The search can be accelerated and partially parallelized.
- Incorporation of Multimodal Dropout:
- Action: During training, optionally use multimodal dropout, a technique that randomly drops data from entire modalities during training. This forces the model to not become over-reliant on any single input type.
- Troubleshooting Tip: If the model performance drops significantly when a modality is missing during inference, increase the rate of multimodal dropout during training to improve robustness.
Troubleshooting Guides and FAQs
Q1: Our multimodal model's performance is worse than using the best single modality. What could be wrong? A: This is a classic "multimodal disadvantage" [71]. First, verify the quality and predictive power of each modality independently. The issue often lies in the fusion method. Early or intermediate fusion can be negatively impacted by noisy or weak modalities. Solution: Switch to a Late Fusion strategy, which is more robust to weak modalities as it weighs each model's decision based on its individual performance [71]. Alternatively, an Automated Fusion search might discover a architecture that effectively filters out noise.
Q2: How can we handle experiments where data for some modalities is missing for certain samples? A: Late Fusion naturally handles this, as you can simply omit the missing modality's model from the final decision aggregation for that sample [71]. For automated or other fusion models, you must explicitly design for this. Solution: Incorporate multimodal dropout during training, which teaches the model to make accurate predictions even when one or more inputs are absent [1].
Q3: Our data pipeline is slow, and GPUs are often idle, waiting for data. How can we improve efficiency? A: This indicates a bottleneck in your data preprocessing pipeline. A common culprit is naive sequence padding, where all samples are padded to the length of the longest sample in a batch, wasting GPU memory and computation [8]. Solution: Implement a dynamic batching or "knapsack" packing strategy. This algorithm packs sequences of similar length into the same batch, dramatically reducing the amount of padding and improving GPU utilization [8].
Q4: When should we choose Automated Fusion over a traditional method like Late Fusion? A: The choice involves a trade-off. Use Traditional Late Fusion when you need a simple, robust, interpretable baseline that is easy to implement and handles missing data well [1] [71]. Choose Automated Fusion when you have sufficient computational resources and are seeking to maximize performance for a specific, well-defined task, as it can discover non-obvious, optimal fusion patterns that human designers might miss [1].
Workflow Visualization
The following diagram illustrates the core structural differences between the Traditional Late Fusion and Automated Fusion workflows.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Materials and Computational Tools for Multimodal Plant Research
| Item / Solution | Function / Purpose | Example / Note |
|---|---|---|
| Multimodal Plant Dataset | Provides structured data for training and evaluation. | Multimodal-PlantCLEF (flowers, leaves, fruits, stems) [1] |
| Pre-trained CNN Models | Serves as feature extractors or base models for fusion. | MobileNetV3Small [1] |
| Neural Architecture Search (NAS) | Automates the design of high-performing neural networks. | Used to discover optimal fusion architecture [1] |
| Multimodal Dropout | Regularization technique that improves model robustness to missing data. | Randomly drops entire modalities during training [1] |
| Dynamic Batching (Knapsack Packing) | Data pipeline optimization to reduce padding and GPU memory waste. | Packs sequences of similar length into batches [8] |
| Multimodal Preprocessing Pipeline | A structured workflow to extract, transform, and align features from heterogeneous data sources. | Frameworks like Pliers support video, audio, images, and text [3] |
The Role of Explainable AI (XAI) with LIME and SHAP for Pipeline and Model Validation
Frequently Asked Questions
Q1: In a multimodal plant study, should I use LIME or SHAP for validating my image and sensor data pipeline? The choice depends on your validation goal. For rapid, intuitive checks of individual predictions during pipeline development, LIME is advantageous due to its faster explanation time (~400ms for tabular data) and model-agnostic nature, which allows you to debug any model quickly [74]. However, for the final, auditable model validation report that requires high explanation consistency, SHAP is superior. SHAP provides a 98% feature ranking stability, backed by its game-theoretic foundation, which is crucial for scientific reporting and regulatory compliance [74] [75].
Q2: Our explanations for identical inputs change between pipeline runs. Is this normal and how can we fix it? This is a common issue, primarily with LIME, due to its stochastic perturbation process, leading to a consistency score of only 69% [74]. For SHAP, particularly TreeSHAP, consistency is much higher (98%) [74]. To improve stability:
- For LIME: Increase the number of perturbation samples in your configuration. This makes the local approximation more stable at the cost of computational time [74].
- For KernelSHAP: Ensure you are using a sufficient number of iterations and a fixed background dataset to anchor the explanations [74].
- General Practice: Always set a random seed before generating explanations to ensure reproducibility across runs [74].
Q3: We achieved high model accuracy, but the LIME/SHAP explanations don't highlight biologically relevant features. What does this indicate? This is a critical red flag in model validation. High accuracy with nonsensical explanations often indicates that your model has learned spurious correlations from your dataset rather than the true underlying pathology [75]. For instance, it might be basing decisions on background artifacts, image watermarks, or specific lighting conditions rather than actual leaf lesions. You should:
- Audit Your Data: Closely examine the samples your model classifies correctly but for the wrong reasons.
- Refine Preprocessing: Ensure your pipeline effectively removes confounding elements. Standardized preprocessing, as in frameworks like SurvBench, can prevent such issues [43].
- Reconsider Features: Re-evaluate your feature selection and engineering steps to better align with domain knowledge.
Q4: How can we quantitatively evaluate the quality of our XAI explanations for a plant disease model? Beyond visual inspection, you can use these quantitative metrics:
- Fidelity: Measures how well the explanation matches the actual model's behavior. Research shows LIME can achieve a fidelity of 0.81 in medical imaging tasks, while SHAP may score lower at 0.38 in some contexts [75].
- Stability: Measures how consistent an explanation is when the input is slightly perturbed. You can add calibrated noise to your input image and measure the rank correlation (Spearman) of the feature importance scores before and after [75]. SHAP typically demonstrates better stability, with only an 11% degradation in radiology applications under noise, compared to more significant drops for other methods [75].
Q5: What are the key computational trade-offs between LIME and SHAP in a large-scale multimodal pipeline? Your pipeline's scalability will be affected by your choice of XAI method. The following table summarizes the key performance characteristics:
| Metric | LIME | SHAP (TreeSHAP) | SHAP (KernelSHAP) |
|---|---|---|---|
| Explanation Time (Tabular) | ~400 ms | ~1.3 s | ~3.2 s |
| Memory Usage | 50-100 MB | 200-500 MB | ~180 MB |
| Consistency Score | 65-75% | ~98% | ~95% |
| Model Compatibility | Universal (Black-box) | Tree-based models | Universal (Black-box) |
| Batch Processing | Limited | Excellent | Good |
Source: Adapted from enterprise deployment metrics [74]
For large-scale validation of tree-based models, TreeSHAP is highly efficient. For other model types, LIME offers a faster, less resource-intensive option, though SHAP provides greater consistency [74].
Troubleshooting Guides
Problem: Low Explanation Fidelity
Symptoms: The explanation maps appear random and do not align with the model's output when tested systematically.
Investigation and Resolution:
- Verify the Explanation Method: Confirm that you are using an appropriate XAI technique for your model. For example, use TreeSHAP for tree-based models (e.g., Random Forest, XGBoost) for exact and fast explanations, and KernelSHAP or LIME for neural networks [74].
- Check Background Data (for SHAP): The choice of background dataset for SHAP is critical. Using a single baseline (like zeros) can produce misleading explanations. Instead, use a representative sample (e.g., 100-1000 instances) from your training data to create a meaningful baseline for comparison [74].
- Tune LIME Parameters: The quality of LIME's local approximation is highly sensitive to its parameters. Increase the
num_samplesparameter to generate more perturbed samples, which usually leads to a more faithful explanation. Also, ensure thekernel_widthparameter is appropriately set for your data's feature space [74].
Problem: Inconsistent Explanations Across Modalities
Symptoms: In a multimodal pipeline (e.g., combining images and environmental data), explanations for one modality are stable, while others are not.
Investigation and Resolution:
- Isolate the Modality: Run the XAI method on each data modality independently. This helps identify if the instability originates from a specific data type (e.g., structured sensor data vs. images) [23] [76].
- Validate Preprocessing Consistency: Ensure that the preprocessing steps (normalization, handling of missing values, data augmentation) are applied consistently and are appropriate for each modality. A standardized pipeline like SurvBench can enforce this [43].
- Adopt a Unified XAI Framework: Use a hybrid deployment strategy. For instance, employ LIME for fast, initial validation on image data and SHAP for rigorous, final validation on structured tabular data (e.g., weather metrics). Studies on tomato disease diagnosis have successfully used LIME for image modality and SHAP for weather modality within the same framework [23].
Problem: XAI Causes Significant Pipeline Slowdown
Symptoms: Adding explanation generation to your validation pipeline drastically increases its runtime, making iteration slow.
Investigation and Resolution:
- Profile Explanation Time: Determine which part of the XAI process is the bottleneck. Is it the model inference, the explanation algorithm itself, or the post-processing?
- Implement Explanation Caching: If you are validating the same model on a fixed test set, pre-compute the explanations for the test set once and cache the results. This avoids recomputing them in every pipeline run [74].
- Optimize Batch Processing: SHAP, especially TreeSHAP, is highly efficient at generating explanations in batches. Leverage this by explaining large batches of instances at once instead of using a loop for single instances [74].
- Use Approximation Methods: For very large models or datasets, consider using faster, approximate explanation methods during the development and switch to more rigorous methods only for the final validation report.
Experimental Protocols for XAI Validation
Protocol 1: Quantifying Explanation Fidelity using Systematic Occlusion
This protocol measures how faithfully an explanation reflects the model's actual decision-making process [75].
Methodology:
- Select a Test Image: Choose an input image for which you have a LIME or SHAP explanation (a saliency map highlighting important regions).
- Baseline Prediction: Record the model's confidence score for the predicted class on the original, unmodified image.
- Systematic Occlusion: Iteratively occlude (e.g., by replacing with gray) the image regions identified as most important by the XAI method. Use a sliding window or gradually expand the occluded area.
- Measure Impact: After each occlusion, record the model's confidence score for the original class.
- Calculate Correlation: Compute the Pearson correlation coefficient between the XAI-provided importance scores and the observed drop in model confidence. A high positive correlation indicates high fidelity.
Protocol 2: Evaluating Explanation Stability under Noise Perturbation
This protocol tests the robustness of your explanations, which is crucial for reliable validation [75].
Methodology:
- Create Perturbed Instances: Take a set of test images and create multiple perturbed versions by adding calibrated Gaussian noise at increasing levels (e.g., 0%, 5%, 10%, 15% of maximum image intensity).
- Generate Explanations: Run your XAI method (LIME/SHAP) to generate a feature importance ranking for both the original and each perturbed image.
- Measure Consistency: For each original-perturbed pair, calculate the Spearman's rank correlation coefficient between the feature importance rankings.
- Analyze Degradation: Plot the correlation coefficient against the noise level. A robust explanation method will maintain a high correlation as noise increases. Studies show SHAP may have a 53% degradation in ophthalmology data at 10% noise, while LIME can be less stable [75].
The Scientist's Toolkit: Research Reagents & Materials
| Item | Function in XAI Experimentation |
|---|---|
| Standardized Preprocessing Pipelines (e.g., SurvBench) | Transforms raw, multi-modal data into standardized, model-ready tensors. It enforces patient-level (or plant-level) data splitting to prevent leakage and ensures reproducibility, which is foundational for any downstream XAI validation [43]. |
| LIME (Local Interpretable Model-agnostic Explanations) | Generates local, post-hoc explanations by perturbing the input and seeing how the prediction changes. Ideal for quick, intuitive model debugging and for explaining any black-box model during pipeline development [74] [23]. |
| SHAP (SHapley Additive exPlanations) | Provides theoretically grounded feature importance values based on cooperative game theory. Best used for generating consistent, auditable explanations for final model validation reports, especially with tree-based models [74] [23]. |
| Grad-CAM (Gradient-weighted Class Activation Mapping) | A model-specific method for convolutional neural networks that produces coarse visual explanations. It is often used alongside LIME/SHAP to provide an additional perspective on which image regions activated the model's final layers [75] [76]. |
| Vision Transformer (ViT) Attention Maps | For models based on the Transformer architecture, the built-in attention mechanisms can be visualized to show the relationships between different image patches, offering an intrinsic form of explainability [76]. |
Workflow Visualization
Diagram 1: XAI Validation Workflow
This diagram illustrates the logical workflow for integrating XAI into a multimodal model validation pipeline.
Diagram 2: LIME vs. SHAP Comparison
This diagram provides a visual comparison of the core mechanisms behind LIME and SHAP.
Benchmarking Against Established Datasets and State-of-the-Art Models
Frequently Asked Questions
Q1: What are the most relevant multimodal benchmarks for agricultural plant science research? The MIRAGE benchmark is highly relevant, as it is constructed from over 35,000 real user-expert interactions in agriculture and includes both single-turn (MMST) and multi-turn (MMMT) tasks involving images, text, and metadata [77]. Other pertinent benchmarks include AgMMU for agricultural multiple-choice questions and CROP for multi-turn crop science QA, though CROP is text-only [77].
Q2: My model performs well on general benchmarks but fails on my specific plant dataset. Why? This is a common issue of domain specialization and open-world generalization. State-of-the-art models often struggle with rare entities and real-world, underspecified user queries found in specialized domains like agriculture [77]. Benchmark your model on a domain-specific benchmark like MIRAGE, which is designed to expose this generalization gap. For instance, fine-tuned models can show a persistent performance drop of over 14 points when encountering unseen plant species [77].
Q3: How should I preprocess plant imagery for a multimodal pipeline? A standard preprocessing pipeline for plant images involves several key steps to enhance data quality and feature extraction [78]:
- White Balancing: Normalize color to compare color reliably across images [78].
- Rotation/Shift: Align plants to a grid for subsequent analysis, crucial for multi-plant images [78].
- Color Space Conversion: Convert from RGB to a channel with better contrast, like the green-magenta channel in LAB colorspace [78].
- Thresholding and Noise Removal: Create a binary image and remove small speckles to isolate plant material from the background [78].
Q4: What is a clarify-or-respond decision, and why is it important for my multimodal assistant? In a multi-turn conversation, a model must decide whether it has enough information to answer a user's query or if it needs to ask a clarifying question. This is a core capability tested in benchmarks like MIRAGE-MMMT. Even top models currently achieve only about 63% accuracy on this decision, highlighting its difficulty and importance for building effective interactive assistants [77].
Experimental Protocols for Benchmarking
Protocol 1: Evaluating on MIRAGE-MMST (Single-Turn Task)
This protocol assesses a model's ability to answer a single, multimodal question, typical in a consultation scenario.
- Input Construction: For each test instance, provide the model with a user's natural language question, one or more user-submitted images, and any associated metadata (e.g., location, timestamp) [77].
- Task: The model must generate a structured response that includes identification, diagnostic reasoning, and management recommendations [77].
- Evaluation: Use an ensemble of reasoning-capable LLMs as judges to score responses on fine-grained criteria [77]:
- Identification Accuracy: Percentage correctness in identifying biological entities.
- Reasoning Score: Quality of causal justification (e.g., on a scale of 1-4).
- Relevance & Completeness: How pertinent and thorough the response is.
- Diagnostic Parsimony: Appropriateness of the diagnostic reasoning.
Protocol 2: Evaluating on MIRAGE-MMMT (Multi-Turn Task)
This protocol tests a model's decision-making in an ongoing dialogue.
- Input Construction: Provide the model with the history of a multimodal conversation between a user and an expert [77].
- Task: At each turn, the model must first decide whether to clarify (ask a question) or respond (provide an answer). Subsequently, it must generate the appropriate utterance (clarifying question or final response) [77].
- Evaluation:
- Decision Accuracy: Percentage of correct clarify-or-respond decisions.
- Generation Quality: Quality of the generated questions or answers, assessed via LLM judges or human evaluation.
Benchmark Performance of State-of-the-Art Models
The table below summarizes the performance of various models on the MIRAGE benchmark, illustrating the challenge it presents. All quantitative data is sourced from the MIRAGE benchmark paper [77].
| Model | Identification Accuracy (MMST) | Reasoning Score (MMST, out of 4) | Decision Accuracy (MMMT) |
|---|---|---|---|
| GPT-4.1 | 43.9% | Information Not Provided | Information Not Provided |
| Qwen2.5-VL-72B | 29.8% | 2.47 | Information Not Provided |
| Qwen2.5-VL-3B (Fine-tuned) | 28.4% (on seen entities) | Information Not Provided | Information Not Provided |
| Qwen2.5-VL-3B (Fine-tuned) | 14.6% (on unseen entities) | Information Not Provided | Information Not Provided |
The Scientist's Toolkit: Research Reagent Solutions
| Tool / Benchmark | Function in Experiment |
|---|---|
| MIRAGE Benchmark | Provides a high-fidelity benchmark derived from real-world agricultural consultations to evaluate model performance on expert-level reasoning and decision-making [77]. |
| PlantCV | An open-source image analysis package used to build modular, customizable pipelines for processing plant images, including functions for multi-plant separation, color space conversion, and thresholding [78]. |
| Solaris Preprocessing Library | A Python library providing over 60 classes for building complex geospatial image preprocessing pipelines, useful for tasks like calculating vegetation indices (e.g., NDVI) from multispectral imagery [79]. |
Workflow Diagram: Multimodal Benchmarking for Plant Science
This diagram visualizes the end-to-end workflow for preprocessing a plant dataset and benchmarking a multimodal model.
Workflow Diagram: Multi-Plant Image Analysis Pipeline
This diagram details a specific image processing pipeline for segmenting and analyzing multiple plants in a single image, as implemented in tools like PlantCV [78].
Conclusion
A meticulously constructed data preprocessing pipeline is the cornerstone of any successful multimodal AI project in plant science. This synthesis of key intents demonstrates that overcoming foundational data challenges—through strategic acquisition, fusion-ready structuring, and robust noise handling—directly translates to enhanced model performance, as evidenced by significant accuracy improvements in plant identification and disease diagnosis. The future of this field hinges on developing more automated, scalable, and standardized preprocessing workflows. These advancements will not only accelerate precision agriculture but also have profound implications for biomedical research, where insights from plant-based models can inform drug discovery mechanisms and the understanding of complex biological systems. The continued evolution of these pipelines is essential for unlocking the full potential of multimodal data to address global challenges in food security and health.
References
- 1. Automatic fused multimodal deep learning for plant ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1616020/full]
- 2. Analysis of single plant cells provides insights into natural ... [https://www.mpg.de/20303131/comprehensive-analysis-of-single-plant-cells-provides-new-insights-into-natural-product-biosynthesis]
- 3. Multi-modal Preprocessing Pipeline [https://www.emergentmind.com/topics/multi-modal-preprocessing-pipeline]
- 4. Digital Single-Cell Analysis of Plant Organ Development ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4558707/]
- 5. Multimodal Deep Learning Integration of Image, Weather, ... [https://www.mdpi.com/2072-4292/16/21/4043]
- 6. The pipelines of deep learning-based plant image ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12304785/]
- 7. Multimodal data integration to model, predict, and ... [https://www.sciencedirect.com/science/article/pii/S1574954125004947]
- 8. Efficient MultiModal Data Pipeline [https://huggingface.co/blog/mmdp]
- 9. Recent advances in plant disease detection: challenges and ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-025-01450-0]
- 10. Navigating the Complexities of Multimodal Data [https://www.mangold-international.com/en/research/products/observation-labs/post/navigating-multimodal-data]
- 11. Data Cleaning & Data Preprocessing for Machine Learning [https://encord.com/blog/data-cleaning-data-preprocessing/]
- 12. 10 Frequently Encountered Issues in Data Preprocessing - [https://www.analyticsvidhya.com/blog/2022/08/10-frequently-encountered-issues-in-data-preprocessing/]
- 13. Simplifying Multimodal Clinical Research Data Management [https://pmc.ncbi.nlm.nih.gov/articles/PMC10972680/]
- 14. The state of multimodal data in biotech and pharma 2025 [https://www.tiledb.com/blog/state-of-multimodal-data-2025]
- 15. PlantIF: Multimodal Semantic Interactive Fusion via Graph ... [https://www.sciencedirect.com/science/article/pii/S2643651525001384]
- 16. 3.6. Monitoring multimodal datasets [https://learn.evidentlyai.com/ml-observability-course/module-3-ml-monitoring-for-unstructured-data/monitoring-multimodal-datasets]
- 17. Heterogeneous network drug-target interaction prediction ... [https://www.nature.com/articles/s41598-025-16098-y]
- 18. Network-based multi-omics integrative analysis methods in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11954193/]
- 19. Context-Aware Multimodal Augmented PlantVillage Dataset [https://ieee-dataport.org/documents/context-aware-multimodal-augmented-plantvillage-dataset]
- 20. Plant Disease Detection through Multimodal Large ... [https://arxiv.org/abs/2504.20419]
- 21. Multimodal few-shot learning for plant disease detection ... [https://link.springer.com/article/10.1007/s00521-025-11438-5]
- 22. A Multimodal Benchmark Dataset and Model for Crop ... [https://arxiv.org/html/2503.06973v1]
- 23. Interpretable deep multimodal-based tomato disease ... [https://www.nature.com/articles/s41598-025-21611-4]
- 24. Visual information guided multi-modal model for plant ... [https://www.sciencedirect.com/science/article/pii/S2772375524001734]
- 25. Multispecies deep learning using citizen science data ... [https://www.nature.com/articles/s41467-024-48559-9]
- 26. Citizen science records are fuelling exciting discoveries of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12186130/]
- 27. Troubleshooting Best Practices for Data Acquisition Systems [https://www.campbellsci.com/blog/troubleshooting-data-acquisition-systems]
- 28. 7 Common Problems with Data Loggers [https://www.xiltrixusa.com/resources/7-problems-data-loggers-data-acquistion-system]
- 29. Researchers Share Images to Accelerate AI Use in Agriculture [https://cals.ncsu.edu/news/researchers-share-images-to-accelerate-ai-use-in-agriculture/]
- 30. Automatic fused multimodal deep learning for plant ... [https://pubmed.ncbi.nlm.nih.gov/40838081/]
- 31. 3D multimodal image registration for plant phenotyping [https://www.sciencedirect.com/science/article/pii/S0168169925006441]
- 32. Deep learning for text summarization using NLP ... [https://www.nature.com/articles/s41598-025-20224-1]
- 33. Data Preprocessing and Cleaning Techniques for ... - RTEI [https://rtei.net/data-preprocessing-and-cleaning-techniques-for-environmental-data/]
- 34. Importance of Data Normalization in Complex Environmental ... [https://blog.chemistry-matters.com/data-normalization]
- 35. Weak supervision for non-categorical applications ... [https://snorkel.ai/blog/weak-supervision-for-non-categorical-applications-superalignment/]
- 36. Leveraging large language models for knowledge-free ... [https://www.nature.com/articles/s41598-024-68168-2]
- 37. Automatic fused multimodal deep learning for plant identification [https://pmc.ncbi.nlm.nih.gov/articles/PMC12361144/]
- 38. Multimodal Models and Fusion - A Complete Guide [https://medium.com/@raj.pulapakura/multimodal-models-and-fusion-a-complete-guide-225ca91f6861]
- 39. sciencedirect.com/topics/computer-science/ multimodal - data - fusion [https://www.sciencedirect.com/topics/computer-science/multimodal-data-fusion]
- 40. Early Fusion vs. Late Fusion in Multimodal Data Processing [https://www.geeksforgeeks.org/deep-learning/early-fusion-vs-late-fusion-in-multimodal-data-processing/]
- 41. A Preprocessing Pipeline for Pupillometry Signal from ... [https://pubmed.ncbi.nlm.nih.gov/40807902/]
- 42. A Review on the Detection of Plant Disease Using Machine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12565507/]
- 43. A Standardised Preprocessing Pipeline for Multi-Modal ... [https://arxiv.org/html/2511.11935v1]
- 44. Automatic 3D Plant Organ Instance Segmentation Method ... [https://www.sciencedirect.com/science/article/pii/S2643651525000718]
- 45. Bridging the Gap: Missing Data Imputation Methods and ... [https://www.mdpi.com/2076-3425/15/6/639]
- 46. A Review of Integrative Imputation for Multi-Omics Datasets [https://www.frontiersin.org/journals/genetics/articles/10.3389/fgene.2020.570255/full]
- 47. Two-stage weakly supervised learning to mitigate label ... [https://www.sciencedirect.com/science/article/abs/pii/S0306261924000424]
- 48. Enhancing data quality through an “I don't know” option in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6392254/]
- 49. Background Noise Reduction of Attention Map for Weakly ... Supervised [https://arxiv.org/html/2404.03394v1/]
- 50. (PDF) Uncertainty Estimation via Response Scaling for Pseudo-Mask... [https://www.academia.edu/92197076/Uncertainty_Estimation_via_Response_Scaling_for_Pseudo_Mask_Noise_Mitigation_in_Weakly_Supervised_Semantic_Segmentation]
- 51. Addressing Noisy Pixels in Weakly Supervised Semantic ... [https://www.mdpi.com/2227-7390/12/16/2520]
- 52. Label Noise-Resistant Mean Teaching for Weakly ... [https://arxiv.org/abs/2206.12260]
- 53. Insights into Using Citizen Science with Machine Learning ... [https://theoryandpractice.citizenscienceassociation.org/articles/10.5334/cstp.740]
- 54. Integrating multimodal data through interpretable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9347276/]
- 55. What new requirements does large-model multimodal data ... [https://www.tencentcloud.com/techpedia/121757]
- 56. Large scale End-to-end MUltimodal Recommendation [https://arxiv.org/html/2511.10962v2]
- 57. Spotlight: Dataloop Accelerates Multimodal Data ... [https://developer.nvidia.com/blog/spotlight-dataloop-accelerates-multimodal-data-preparation-pipelines-for-llms-with-nvidia-nim/]
- 58. Build a domain‐aware data preprocessing pipeline: A multi ... [https://aws.amazon.com/blogs/machine-learning/build-a-domain%E2%80%90aware-data-preprocessing-pipeline-a-multi%E2%80%90agent-collaboration-approach/]
- 59. Research collaboration data platform ensuring general ... [https://www.nature.com/articles/s41598-024-61912-8]
- 60. Data Privacy: Key Questions, Challenges & Best Practices [https://www.indusface.com/blog/10-important-data-privacy-questions-you-should-be-asking-now/]
- 61. 20 Important Data Privacy Questions You Should be Asking Now [https://www.cipher.com/blog/cipher/20-important-data-privacy-questions-you-should-be-asking-now]
- 62. Sharing data with collaborators | Data Privacy Handbook [https://utrechtuniversity.github.io/dataprivacyhandbook/data-sharing-collaboration.html]
- 63. Train-val-test splits and cross-validation [https://medium.com/@masadeghi6/how-to-split-your-data-for-machine-learning-eae893a8799c]
- 64. Training, Validation, Test Split for Machine Learning Datasets [https://encord.com/blog/train-val-test-split/]
- 65. Cross Validation: Training, Validation and Testing Splits [https://the-examples-book.com/tools/data-modeling-cross-validation-train-valid-test]
- 66. Data Preprocessing Pipeline - by Dua Rizwan [https://medium.com/@dua-rizwan/data-preprocessing-pipeline-994361b7c986]
- 67. Top 10 Machine Learning Frameworks to Use In 2025 [https://www.igmguru.com/blog/machine-learning-frameworks]
- 68. Top 5 Machine Learning Frameworks for 2025 [https://codex.team/blog/top-5-machine-learning-frameworks-every-data-scientist-should-know-in-2025]
- 69. Strategies to Improve the Robustness and Generalizability ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12014193/]
- 70. Two common issues in synchronized multimodal ... [https://www.sciencedirect.com/science/article/pii/S0168010223002201]
- 71. Robust Multimodal Fusion for Survival Prediction in Cancer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12476512/]
- 72. A Comparative Analysis of Three Data Fusion Methods and ... [https://www.mdpi.com/2227-7390/13/8/1218]
- 73. Using convolutional neural networks with late fusion to ... [https://www.nature.com/articles/s41598-025-25100-6]
- 74. SHAP vs LIME: Which XAI Tool Is Right for Your Use Case? [https://ethicalxai.com/blog/shap-vs-lime-xai-tool-comparison-2025.html]
- 75. A Comprehensive Framework for Accountable AI in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12383817/]
- 76. Enhancing green AI through explainable deep learning ... [https://link.springer.com/article/10.1007/s10791-025-09799-5]
- 77. MIRAGE: A Benchmark for Multimodal Information-Seeking ... [https://arxiv.org/html/2506.20100v1]
- 78. Multi plant pipeline - Tutorial - PlantCV - Read the Docs [https://plantcv.readthedocs.io/en/v2.0/multi-plant_tutorial/]
- 79. Solaris Multimodal Preprocessing Library - Read the Docs [https://solaris.readthedocs.io/en/latest/tutorials/notebooks/preprocessing_pipelines.html]