Bioregenerative vs Physicochemical Life Support: MELiSSA's Circular Ecosystem for Long-Duration Space Missions
This article provides a comparative analysis of bioregenerative (exemplified by the MELiSSA project) and traditional physicochemical life support systems for long-duration space missions.
Bioregenerative vs Physicochemical Life Support: MELiSSA's Circular Ecosystem for Long-Duration Space Missions
Abstract
This article provides a comparative analysis of bioregenerative (exemplified by the MELiSSA project) and traditional physicochemical life support systems for long-duration space missions. It explores the foundational principles of these systems, their methodological applications in air, water, and waste management, key integration and optimization challenges, and validation through terrestrial analogs and pilot plants. Aimed at researchers and life support engineers, the synthesis highlights the complementary roles of both approaches in achieving sustainable, closed-loop habitats for deep space exploration and terrestrial circular economy applications.
Defining Life Support Paradigms: From Mechanical Recycling to Biological Regeneration
For long-duration human space exploration missions beyond Earth's orbit, the development of advanced life support systems that minimize resupply requirements from Earth is a critical technological challenge. The current paradigm is dominated by Physicochemical Environmental Control and Life Support Systems (ECLSS), which utilize mechanical and chemical processes to maintain cabin atmospheres and recycle water. However, a competing approach—Bioregenerative Life Support Systems (BLSS)—aims to create more sustainable, ecosystem-inspired systems using biological organisms. This comparison guide examines the core philosophies, performance metrics, and experimental evidence for these competing approaches, with particular focus on the European Space Agency's Micro-Ecological Life Support System Alternative (MELiSSA) vision, which represents one of the most advanced BLSS initiatives globally [1] [2] [3].
The fundamental distinction lies in their operational philosophies: ECLSS relies on engineered physical and chemical processes, while BLSS incorporates biological elements to create a more closed-loop, regenerative system. As space agencies plan for long-duration lunar missions and eventual Mars expeditions, the choice between these approaches carries significant implications for mission architecture, resupply logistics, and crew safety [4] [2].
System Architectures and Core Philosophies
Physicochemical ECLSS: Engineering-Centric Life Support
The Environmental Control and Life Support System (ECLSS) aboard the International Space Station represents the current state-of-the-art in regenerative life support technology. This engineered system performs distinct functions through specialized subsystems:
Air Revitalization: The Oxygen Generation System (OGS) electrolyzes water to produce oxygen for crew respiration. Simultaneously, the Carbon Dioxide Removal Assembly (CDRA) scrubs CO₂ from the cabin atmosphere. A Sabatier reactor can subsequently combine waste hydrogen with carbon dioxide to produce water and methane, partially closing the oxygen loop [5].
Water Recovery: The Water Recovery System consists of a Urine Processor Assembly that uses vacuum distillation to recover water from urine, and a Water Processor Assembly that purifies wastewater through filtration and catalytic oxidation to produce potable water [5].
Waste Management: Unlike biological systems, ECLSS largely treats waste as an end product rather than a resource. Concentrated waste streams are stored for disposal rather than being reintegrated into the life support cycle [6].
The ECLSS philosophy prioritizes predictability, controllability, and reliability through engineered systems with well-understood physical and chemical processes. While effective for Low Earth Orbit operations, this approach faces limitations for deeper space missions due to its dependence on resupply of consumables and limited closure of essential element cycles [6] [2].
Bioregenerative BLSS: Ecosystem-Inspired Life Support
Bioregenerative Life Support Systems adopt a fundamentally different philosophy inspired by terrestrial ecosystems. Rather than treating life support as a series of discrete engineering problems, BLSS aims to create balanced, self-regulating systems where biological processes work in concert to regenerate resources [7] [3].
A canonical BLSS incorporates three essential biological components:
- Producers: Typically plants or microalgae that generate food and oxygen via photosynthesis while consuming carbon dioxide.
- Consumers: The crew members who consume oxygen, food, and water while producing waste streams.
- Degraders: Microorganisms that break down waste products into forms usable by the producers [3].
This ecological approach theoretically offers greater closure of element cycles and reduced reliance on external resupply. However, it introduces challenges related to system stability, control complexity, and the unpredictable nature of biological systems [4].
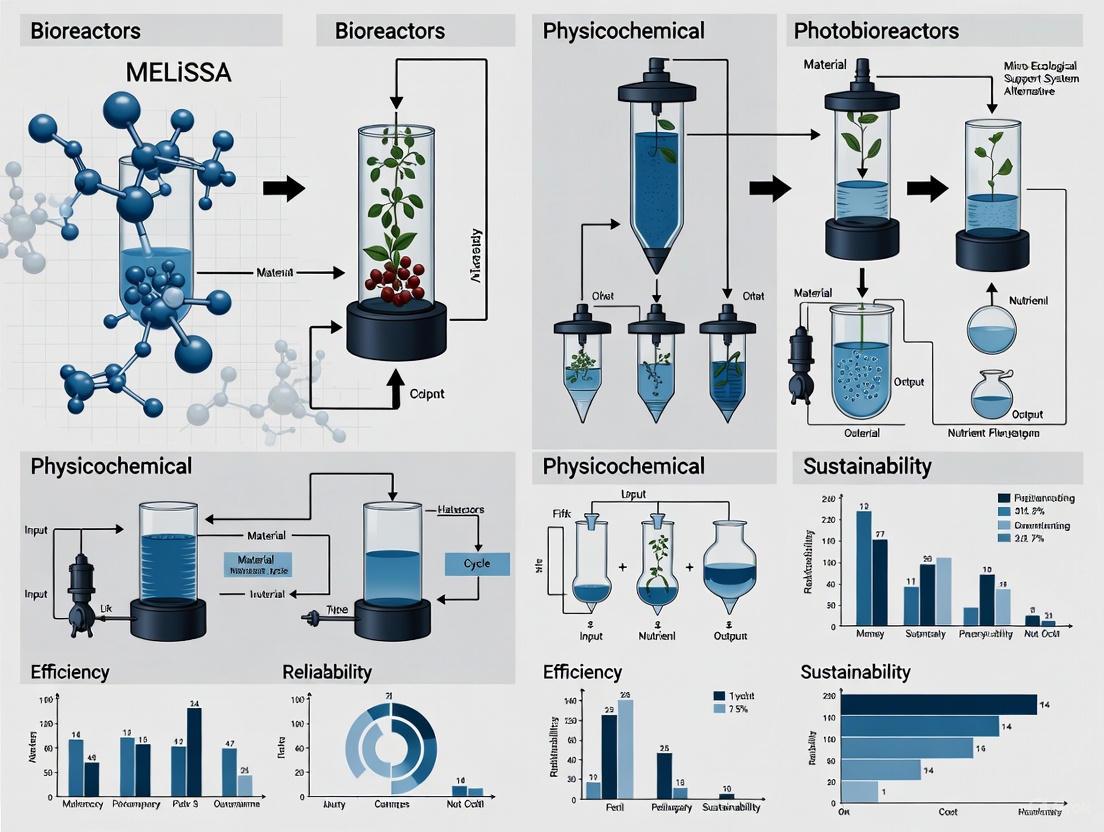
The MELiSSA Vision: Engineering Ecological Principles
The MELiSSA initiative, coordinated by the European Space Agency, represents a unique hybrid philosophy that combines ecological principles with rigorous engineering methodology. Unlike earlier BLSS approaches that attempted to replicate complete ecosystems, MELiSSA adopts a "deterministic approach, to characterize all processes in as much detail as possible as a first step to recreating it" [1].
The MELiSSA loop is architecturally designed as five interconnected compartments, each performing specific functions:
- Compartment I: Waste decomposition using thermophilic anaerobic bacteria
- Compartment II: Photoheterotrophic nitrogen recovery
- Compartment III: Nitrifying bacteria for further nutrient recycling
- Compartment IV: Photoautotrophic organisms (plants and microalgae) for oxygen and food production
- Compartment V: The crew compartment [6] [8]
This compartmentalized architecture allows for precise monitoring and control of each biological process—a key distinction from earlier ecosystem-style BLSS approaches like Biosphere 2 [1]. The MELiSSA philosophy represents a middle path: leveraging biological processes for their regenerative capabilities while maintaining engineering oversight and control.
Figure 1: The MELiSSA Loop Circular Architecture - This simplified diagram illustrates the continuous flow of matter through the five-comparment MELiSSA system, where waste outputs become inputs for other compartments.
Performance Comparison: Quantitative Metrics
Resource Recovery Efficiency
Resource recovery efficiency represents a critical metric for evaluating life support system performance, particularly for long-duration missions where resupply opportunities are limited or nonexistent.
Table 1: Resource Recovery Efficiency Comparison
| Resource | ISS ECLSS | BLSS (Theoretical) | MELiSSA Projection | Notes |
|---|---|---|---|---|
| Oxygen | ~50% closure with Sabatier [5] | Potentially >90% [4] | Target: Near 100% [8] | ECLSS vents methane overboard, losing oxygen |
| Water | 70-85% recovery from urine [5] | Potentially >95% [3] | Target: Near 100% [8] | ECLSS urine processor limited by precipitation issues |
| Food | 0% production (all resupplied) [6] | 100% potential [3] | Target: Significant portion [8] | BLSS produces food via plants/microalgae |
| Nitrogen | Limited recovery [6] | >90% potential via biological processing [6] | Target: Efficient recycling [6] | MELiSSA Compartment III specializes in nitrogen recovery |
The data reveal a fundamental distinction: ECLSS achieves partial closure of atmospheric and water cycles but cannot address food production, while BLSS aims for near-complete closure of all essential element cycles, including food production.
Technology Readiness and System Mass Comparison
Assessing the maturity and practical implementation potential of life support technologies requires consideration of both their development status and equivalent system mass—a metric that accounts for both launch mass and operational resource requirements.
Table 2: Technology Readiness and Mass Considerations
| Parameter | ISS ECLSS | BLSS | MELiSSA |
|---|---|---|---|
| TRL | 9 (Flight Proven) [5] | 3-6 (Ground Demonstrators) [2] | 4-5 (Pilot Plant) [1] [8] |
| Crew Size Supported | 6-7 [5] | Varies by facility (1-4) [2] | Pilot Plant: 3 rats (human crew target) [1] |
| Energy Requirements | Moderate | High (especially lighting) [4] | Target: Optimized with LED technology [4] |
| System Complexity | High but well-characterized | Very high (biological uncertainty) [4] | Compartmentalized for control [1] |
| Mission Class Suitability | LEO, short-term lunar | Long-duration, planetary bases [4] [3] | Long-duration, planetary bases [8] |
The comparison highlights the trade-off between technological maturity and long-term sustainability. While ECLSS represents a proven, operational technology, BLSS approaches like MELiSSA target mission scenarios where resupply is not feasible.
Experimental Evidence and Validation Protocols
ECLSS Validation and Performance Data
The International Space Station's ECLSS has generated extensive performance data through continuous operation:
Water Recovery: The Urine Processor Assembly initially designed for 85% water recovery now operates at approximately 70-75% efficiency due to precipitation issues with calcium sulfate in microgravity. The system processes an average of 1.80L of urine and flush water per crew member daily [5].
Oxygen Generation: The Oxygen Generating System (OGS) produces oxygen via electrolysis of water recovered from various sources. System reliability has been challenged by issues including coolant pump failures and water pH imbalances, requiring contingency oxygen supplies from bottled oxygen or Solid Fuel Oxygen Generation (SFOG) canisters [5].
Carbon Dioxide Management: The Carbon Dioxide Removal Assembly (CDRA) and advanced Sabatier system provide CO₂ removal and partial oxygen recovery. The European Advanced Closed Loop System (ACLS) demonstrates improved efficiency by recovering 50% of CO₂ through Sabatier reaction and subsequent oxygen generation [5].
These systems have undergone iterative improvement based on flight experience, demonstrating the evolutionary engineering approach characteristic of ECLSS development.
MELiSSA Ground Demonstration Experimental Protocol
The MELiSSA Pilot Plant at Universitat Autonoma de Barcelona employs a rigorous experimental protocol to validate compartment integration and system control:
Objective: Demonstrate stable operation of interconnected MELiSSA compartments supporting an animal crew (rats) as a precursor to human testing [1] [8].
Methodology:
- Compartment Characterization: Individual compartments are first operated independently to establish baseline performance metrics under controlled conditions.
- Progressive Integration: Compartments are systematically interconnected, with continuous monitoring of input and output streams between compartments.
- Steady-State Operation: The integrated system operates at predefined "steady states" to evaluate stability and control performance over extended durations (up to six months).
- Disturbance Testing: Intentional set-point changes are introduced to test system robustness and control algorithm performance [1].
Key Performance Metrics:
- Gas exchange rates (O₂ production, CO₂ consumption)
- Contaminant levels in the loop
- Biomass production rates
- System closure efficiency [1] [8]
Recent Results: The Pilot Plant has successfully maintained "crews of three rats alive and comfortable for almost six months at a time" using algae-based oxygen production and CO₂ capture, validating the basic MELiSSA concept [1].
Comparative Food Production Experiments
Controlled environment agriculture experiments provide critical data on BLSS food production capabilities:
LED Lighting Optimization: Recent experiments have quantified the equivalent system mass reductions achievable using advanced LED lighting technologies for plant growth, improving the economic viability of BLSS approaches [4].
Crop Selection Protocols: Methodologies have been developed to select plant species based on multiple criteria including nutritional content, growth cycle duration, resource requirements, and edible biomass ratio [3]. Fast-growing species (lettuce, kale) target short-duration missions, while staple crops (wheat, potato) address long-duration needs.
Nutrient Recycling Efficiency: Experiments with urine recycling as a nutrient source for plant growth have demonstrated nitrogen recovery efficiencies exceeding 90%, a critical capability for reducing external fertilizer requirements [6].
Research Reagents and Essential Materials
The experimental development of advanced life support systems requires specialized reagents and materials tailored to the unique demands of closed-loop operation.
Table 3: Essential Research Reagents for Life Support System Experiments
| Reagent/Material | Function | ECLSS Application | BLSS/MELiSSA Application |
|---|---|---|---|
| Lithium Perchlorate | Oxygen generation via chemical decomposition | SFOG canisters for emergency oxygen [5] | Not typically used |
| Activated Charcoal | Trace contaminant removal | Vozdukh system for atmospheric purification [5] | Water and air purification in closed ecosystems |
| Nitrifying Bacteria Consortia | Biological nitrogen conversion | Not used | MELiSSA Compartment III for urine nitrification [6] |
| Arthrospira platensis | Oxygen production & food source | Not used | MELiSSA Compartment IV as photoautotrophic component [1] |
| Anaerobic Digester Cultures | Waste decomposition | Not used | MELiSSA Compartment I for solid waste processing [6] |
| Hydroponic Nutrient Solutions | Plant growth support | Not used | Higher plant cultivation in BLSS [3] |
| Specific LED Spectra | Optimized plant growth | Limited use | Tailored photosynthetic efficiency in BLSS [4] |
Implementation Roadmaps and Future Directions
ECLSS Development Trajectory
The evolution of physicochemical life support systems continues with incremental improvements focused on reliability and efficiency:
- Advanced Sabatier Systems: Development of more efficient CO₂ reduction technologies to improve oxygen recovery rates beyond the current ~50% closure.
- Water Processor Enhancements: Addressing precipitation and scaling issues in urine processing to restore design efficiency levels.
- Modular Architecture: Creating more compact, fault-tolerant systems for use in constrained spacecraft environments [5].
These evolutionary improvements maintain ECLSS as the near-term solution for lunar orbital missions and initial surface expeditions where resupply remains feasible.
MELiSSA Strategic Development Pathway
The MELiSSA program follows a structured technology development roadmap with both space and terrestrial applications:
Near-Term (2025-2030):
- Completion of integrated ground demonstration with animal crews
- Flight testing of individual compartments on the International Space Station (e.g., ArtemISS project investigating microgravity effects on algae) [1]
- Further optimization of energy and mass parameters using LED technologies and advanced control algorithms [4]
Medium-Term (2030-2035):
- Human-rated integrated system testing in ground facilities
- Demonstration of hybrid BLSS-ECLSS architectures for lunar surface missions
- Validation of food production systems with crew consumption trials [8]
Long-Term (2035+):
- Operational deployment for lunar surface habitats
- Technology maturation for Mars transit and surface missions [2]
The MELiSSA program explicitly positions itself as a 50-year development effort, recognizing the extensive testing and validation required for human-rated biological life support systems [1].
Figure 2: MELiSSA Development Roadmap - This timeline illustrates the progressive technology maturation pathway from current capabilities to operational deployment.
The comparison between ECLSS and BLSS philosophies reveals a fundamental strategic choice for space agencies pursuing long-duration human spaceflight. ECLSS provides immediately available, proven technology with known reliability characteristics but limited closure of essential element cycles. BLSS, particularly the MELiSSA implementation, offers the potential for substantially greater self-sufficiency but requires significant further development to achieve human-rated reliability.
The experimental evidence indicates that hybrid approaches—leveraging ECLSS reliability for critical functions while integrating biological components for food production and enhanced resource recovery—may represent the most viable pathway for near-term lunar missions. As mission durations extend and distances from Earth increase, the balance will likely shift toward increasingly biological solutions.
The MELiSSA vision of a compartmentalized, rigorously engineered biological system offers a promising middle ground, potentially overcoming the stability challenges that hampered earlier ecosystem-style BLSS approaches. Its success will depend on continued systematic development through ground demonstration and incremental flight validation, following the structured roadmap that has characterized the program to date.
For the research community, these competing approaches offer rich opportunities for investigation across multiple disciplines, from fundamental microbiology to advanced control systems engineering. The choice between ECLSS and BLSS is not merely technical but philosophical, reflecting different visions of how humans will ultimately sustain themselves in the hostile environment of space.
For long-duration space missions beyond low Earth orbit, the traditional "store and dump" approach to life support becomes impractical due to the excessive mass and volume requirements for consumables and the impossibility of resupply [9]. This challenge has driven the development of regenerative Life Support Systems (LSS) that recycle wastes into oxygen, water, and food. Two landmark projects in this endeavor are the Russian BIOS-3 and the European MELiSSA (Micro-Ecological Life Support System Alternative) Consortium [9] [10]. This guide traces the historical evolution from the pioneering BIOS-3 experiments to the sophisticated international MELiSSA project, providing a structured comparison of their technologies, performance, and experimental approaches within the broader context of bioregenerative versus traditional physiochemical life support systems.
BIOS-3: The Pioneer of Closed Ecosystems
BIOS-3, located at the Institute of Biophysics in Krasnoyarsk, Russia, was an early and ambitious experimental closed ecosystem. Its construction began in 1965 and was completed in 1972 [11] [12]. This underground steel facility provided 315 cubic meters of volume and was designed to support a crew of up to three people for extended durations [11].
- Primary Objective: To develop and validate a closed ecological system capable of supporting human life through the recycling of air and water, and the partial recycling of food.
- Key Biological Components: The system initially relied on Chlorella algae for air revitalization, with higher plants (wheat and vegetables) cultivated in dedicated "phytotrons" [11].
- Crewed Testing: The facility conducted ten crewed closure experiments, with the longest mission lasting 180 days with a three-person crew from 1972 to 1973 [11] [12].
MELiSSA: The Engineered Ecosystem
The MELiSSA project represents a more recent, consortium-based approach initiated by the European Space Agency (ESA) in 1989 [10] [13]. It was designed from the outset as a highly controlled, compartmentalized, and engineerable system inspired by aquatic ecosystems [10] [13].
- Primary Objective: To achieve near-total closure of the life support loop by processing all mission wastes (inedible biomass, CO₂, feces, urea) to regenerate food, water, and oxygen [14].
- Key Differentiator: Unlike BIOS-3, MELiSSA is structured into distinct, interconnected compartments, each hosting specific microbial or plant communities to perform dedicated waste-processing functions [10].
- Project Scale: MELiSSA is an international effort, involving around 50 organizations across Europe and Canada, making it one of the most advanced initiatives in closed-loop life support [15] [16].
Table 1: Fundamental Characteristics of BIOS-3 and MELiSSA
| Characteristic | BIOS-3 | MELiSSA |
|---|---|---|
| Initiation Date | 1965 (Construction) | 1989 [15] [13] |
| Lead Organization | Institute of Biophysics, Russian Academy of Sciences [11] | European Space Agency (ESA) [8] [10] |
| System Philosophy | Holistic, compartmentalized cultivation | Functional ecology, compartmentalized & engineered [10] [14] |
| Primary Air Revitalization | Chlorella algae & higher plants [11] | Cyanobacteria (Arthrospira platensis) & higher plants [10] |
| Crew Capacity | Up to 3 persons [11] | Target: Human crew support (2020-2025) [8] |
System Architecture and Functional Comparison
BIOS-3 Loop Structure
BIOS-3 was divided into four compartments: one crew compartment and three cultivation compartments. The initial design featured one algal cultivator and two phytotrons, though the algal compartment was later converted into a third phytotron [11]. The system's loop was relatively direct, with algae and plants responsible for the bulk of atmospheric regeneration.
MELiSSA Loop Structure
The MELiSSA loop is a more complex, multi-stage process designed to mimic a terrestrial aquatic ecosystem, with each compartment performing a specific transformation.
- Compartment I (Liquefying): Processes all mission waste (urea, inedible biomass) anaerobically at 55°C to produce volatile fatty acids (VFAs), ammonium, CO₂, and minerals [10].
- Compartment II (Photoheterotrophic): Eliminates the VFAs produced in Compartment I [10].
- Compartment III (Nitrifying): Converts ammonium (NH₄⁺) into nitrates (NO₃⁻), the preferred nitrogen source for plants, using a mix of Nitrosomonas and Nitrobacter bacteria [10].
- Compartment IV (Photoautotrophic): Split into two parts; one uses the cyanobacteria Arthrospira platensis (spirulina), while the other uses higher plants (e.g., wheat, rice, salad ingredients) for final oxygen production and food generation [10].
Performance Data and Experimental Outcomes
Documented Performance Metrics
Both systems have been extensively tested, with their performance quantified in terms of closure rates for air, water, and nutrients.
Table 2: Experimental Performance Metrics
| Parameter | BIOS-3 Achieved Performance | MELiSSA Projected Performance |
|---|---|---|
| Air Recycling (O₂/CO₂) | ~99% balance achieved using 8 m² of Chlorella per person [12] | Target: Near-total closure of O₂/CO₂ loop [14] |
| Water Recycling | 85% efficiency achieved by 1968 [11] [12] | Target: Near-total water recovery from waste and humidity [14] |
| Food Production & Recycling | Approximately 50% of food and nutrients recycled; diet supplemented with imported dried meat [12] | Target: Significant food production from higher plants and spirulina; full waste recycling [10] |
| Waste Processing | Urine and feces were partially recycled, with the remainder dried and stored [11] | All wastes (feces, urea, inedible biomass) are processed in Compartment I [10] |
| Crew Duration | 180 days (3 crew members) [11] [12] | Pilot plant testing with animal "crew"; human testing targeted for 2020-2025 [8] |
Comparison with Physicochemical Systems
A critical trade study compared a hybrid bioregenerative system (like MELiSSA) against a purely physicochemical (P/C) LSS for a 780-day Mars mission for a crew of six. The analysis used Equivalent System Mass (ESM), a metric that converts all system parameters like volume and power into an equivalent mass [9].
This study concluded that the bioregenerative system had an ESM approximately four times higher than the purely physicochemical alternative, though it noted that redundancies had not yet been fully included in the bioregenerative model [9].
Experimental Protocols and Methodologies
BIOS-3 Closure Experiments
The core experimental protocol in BIOS-3 involved long-term isolation of human crews.
- Crew Selection and Preparation: Crews of one to three individuals were selected for closures ranging from days to 180 days [11].
- Atmospheric Monitoring: Oxygen and carbon dioxide levels were continuously monitored. The Chlorella algal cultivators were sized at 8 m² per person to maintain this balance [11] [12].
- Water Management: Water was recycled from humidity condensate and other sources, achieving an 85% recovery rate. Unrecycled water and wastes were stored [12].
- Data Collection: Crew health, plant growth metrics, and system parameters (gas composition, water quality) were regularly recorded [11].
MELiSSA's Structured Development Phases
MELiSSA follows a rigorous, phased development and validation protocol, which is a key differentiator from earlier projects [13].
The operation of the MELiSSA Pilot Plant at Universitat Autònoma de Barcelona is a critical part of Phase 3. Its methodology involves [8] [10]:
- Individual Compartment Validation: Each of the four compartments is developed and optimized independently by consortium partners.
- Progressive Integration: Compartments are interconnected step-by-step to study interactions and stability.
- Control Strategy Validation: The plant is used to test and refine deterministic control strategies for the entire loop, ensuring it can quickly respond to changes in human consumption and waste production.
- Animal "Crew" Testing: The facility uses animals as a proxy for human crew to test gas exchange and loop stability, paving the way for future human testing [8].
The Scientist's Toolkit: Key Research Reagents and Materials
This section details the essential biological and technological components that form the backbone of research in closed ecological systems.
Table 3: Key Research Reagents and Materials in Bioregenerative Life Support
| Item Name | Type | Function in Research |
|---|---|---|
| Chlorella Algae | Biological Agent | In BIOS-3, used as the primary photosynthetic unit for CO₂ absorption and O₂ production via photosynthesis [11] [12]. |
| Arthrospira platensis (Spirulina) | Biological Agent | In MELiSSA, a cyanobacterium used in the photoautotrophic compartment for oxygen production and as a high-protein food source [10]. |
| Nitrosomonas & Nitrobacter | Bacterial Consortia | In MELiSSA Compartment III, these nitrifying bacteria work in sequence to oxidize ammonium to nitrites and then to nitrates for plant nutrition [10]. |
| Higher Plants (Wheat, Salad) | Biological Agent | Used in both systems (BIOS-3 phytotrons, MELiSSA Comp. IVb) for food production, oxygen generation, and water transpiration [11] [10]. |
| Thermophilic Fermenter | Bioreactor | A key unit in MELiSSA Compartment I; operates at 55°C for efficient anaerobic breakdown of solid waste into simpler compounds [10]. |
| Vapor Compression Distillation (VCD) | Physicochemical Unit | A technology considered for water recovery from urine and other waste streams, achieving up to 98% recovery rates in related NASA tests [9] [17]. |
The evolution from BIOS-3 to the MELiSSA Consortium marks a significant paradigm shift in the approach to bioregenerative life support. BIOS-3 demonstrated the fundamental feasibility of sustaining human life in a closed ecosystem for prolonged periods, using a relatively direct and holistic design. Building on this foundation, MELiSSA has introduced a more engineered, compartmentalized, and safety-oriented architecture, backed by a structured, international R&D program.
While current analyses indicate that physiochemical systems may hold a mass advantage for specific missions like a Mars flight, bioregenerative systems offer the unique potential for greater self-sufficiency and food production, which could be decisive for permanent bases on the Moon or Mars [9]. The research conducted within the MELiSSA framework continues to refine these technologies, pushing the boundaries of what is possible in closed-loop life support and driving innovations that also have applications in sustainable terrestrial processes.
For long-duration human space missions, the provision of essential life support resources—oxygen, water, and food—presents a monumental challenge. The current paradigm relies primarily on physicochemical (PC) systems, which effectively recycle air and water but lack the capability to produce food, thus requiring extensive resupply from Earth [10]. In contrast, regenerative biological systems aim to create a circular economy where waste is continuously recycled into resources. Among the most advanced concepts is the Micro-Ecological Life Support System Alternative (MELiSSA), an artificial ecosystem conceived by the European Space Agency [10]. This guide provides a detailed comparison between the MELiSSA loop architecture and traditional PC systems, framing the analysis within the broader context of life support system research for space exploration. We objectively evaluate their performance through experimental data, methodological protocols, and system-level analysis to inform researchers, scientists, and development professionals.
MELiSSA Loop Architecture: A Compartmentalized Biological System
The MELiSSA loop is engineered as an assembly of interconnected unit processes, or compartments, each with a specialized biological function. This compartmentalization simplifies the behavior of a complex ecosystem and enables a deterministic engineering approach [18]. The system is structured around five key elements, with the human crew at its center.
Table 1: The Five Compartments of the MELiSSA Loop Architecture
| Compartment | Key Function | Biological Agents | Primary Inputs | Primary Outputs |
|---|---|---|---|---|
| CI: Liquefying Compartment | Organic waste degradation & solubilisation | Thermophilic anoxygenic bacteria | Crew waste, non-edible plant material [18] | CO₂, volatile fatty acids, ammonia, minerals [18] [10] |
| CII: Photoheterotrophic Compartment | Removal of volatile fatty acids | Photoheterotrophic bacteria | Volatile fatty acids from CI [18] | Inorganic carbon source [18] |
| CIII: Nitrifying Compartment | Conversion of ammonia to nitrates | Nitrosomonas europaea, Nitrobacter winogradskyi [18] | Ammonia from CI [18] | Nitrates (suitable nitrogen for plants) [18] |
| CIVa: Photoautotrophic (Algae) Compartment | Food and oxygen production | Cyanobacteria (Arthrospira platensis) [18] | CO₂ from crew and CI, nitrates from CIII [18] | Edible biomass, oxygen, water [18] |
| CIVb: Photoautotrophic (Higher Plants) Compartment | Food, oxygen, and water production | Higher plants (e.g., wheat, rice, salad crops) [18] | CO₂ from crew and CI, nitrates from CIII [18] | Edible food, oxygen, water [18] |
| CV: Crew | Consumption and waste production | Humans | Food, water, oxygen from CIVa & CIVb [18] | CO₂, urine, faeces, other waste [10] |
The intelligence of the MELiSSA system is based on the adequacy of models representing each unit operation and their interrelations, forming the foundation for a hierarchical control strategy [19] [20]. A critical feature of this architecture is biosafety. By separating processes into distinct compartments, the system mitigates risk; for instance, the first compartment operates at 55°C (thermophilic conditions), which aids efficiency and acts as a microbial barrier [10].
The following diagram illustrates the mass flow and logical relationships between these compartments within the closed-loop system.
MELiSSA vs. Physicochemical Systems: A Comparative Performance Analysis
The fundamental distinction between MELiSSA and traditional PC systems lies in their core operational principles: biology versus chemistry/physics. PC systems typically employ discrete mechanical units—such as CO₂ scrubbers, Sabatier reactors, and water purification systems—to perform individual life support functions [10]. While highly efficient for specific tasks like air revitalization, they form an open-loop system incapable of food production. MELiSSA, conversely, integrates biological processes to create a closed-loop ecosystem that mimics Earth's natural cycles, aiming for near-complete recycling of the major elements: Carbon, Hydrogen, Oxygen, Nitrogen, Sulphur, and Phosphorus (CHONSP) [10].
Table 2: Performance Comparison: MELiSSA vs. Traditional Physicochemical Systems
| Performance Metric | MELiSSA (Biological) | Traditional Physicochemical Systems | Experimental Data & Key Findings |
|---|---|---|---|
| Food Production | Fully Integrated (via higher plants & cyanobacteria) [18] | Not Available (requires resupply) [10] | 32 crop species are currently considered for cultivation in CIVb [18]. Arthrospira platensis provides a high-protein food source [10]. |
| Oxygen Regeneration | Biological (via photosynthesis in CIVa & CIVb) [18] | Physicochemical (e.g., water electrolysis) [10] | Pilot plant demonstrates integration of oxygen production from photoautotrophic compartments [10]. |
| Waste Processing | Biological Valorization (waste is a resource for other compartments) [18] | Storage/Partial Processing (often a terminal burden) [10] | Compartment I successfully transforms waste into ammonium, CO₂, volatile fatty acids, and minerals [10]. |
| Water Recovery | Integrated Biological & Physical (water is a byproduct of photosynthesis and transpiration) [18] | High-Efficiency Physical (e.g., filtration, distillation) [10] | The higher plant compartment (CIVb) contributes to water recovery [18]. |
| System Closure | High (aims for near 100% recycling of key elements) [10] | Low to Medium (open-loop for food, requires regular resupply) [10] | The loop is designed to autonomously recycle wastes with only energy input [10]. Dynamic control is required to respond to human behavior changes [10]. |
| Energy Requirements | Lower Temperature/Pressure (ambient conditions using sunlight/light) [10] | High Energy (often requires high temperature, pressure, or electricity) [10] | Photosynthesis operates at ambient temperatures and pressures, unlike, for example, the Sabatier reaction [10]. |
| Technology Readiness Level (TRL) | Advancing (under continuous development and integration since 1989) [10] | High (operationally proven on ISS) [10] | A Pilot Plant at Universitat Autònoma de Barcelona tests integrated loop feasibility [10]. |
A critical operational difference is the energy profile. PC processes like the Sabatier reaction achieve high efficiencies but demand significant energy in the form of elevated temperature and pressure [10]. In contrast, MELiSSA leverages photosynthesis, which operates at ambient conditions, converting light energy into chemical energy [10]. While the base energy source is potentially more efficient, the conversion efficiencies of biological systems are typically lower than targeted physicochemical processes.
Experimental Protocols and Research Methodologies
Ground-Based Testing in Sealed Mini-Ecosystems
Recent research into closed ecological systems has adopted smaller-scale models to elucidate fundamental principles. One rigorous experimental protocol involved creating sealed "Ecosphere" systems to investigate plant survivability and life support dynamics using simulated planetary soils [21].
Objective: To identify essential ecosystem components for non-physicochemical life support systems and understand the challenges of sustaining life in a closed environment, with a focus on plant growth and the role of microorganisms [21].
Methodology:
- System Design: Compact, sealed glass containers ("Ecosphere 1" and "Ecosphere 2") were constructed with enhanced airtightness, sealed with melted rubber or silicone [21].
- Experimental Variables:
- Ecosphere 1: Contained nutrient-rich natural soil (with inherent microbes) and clover seeds. Placed outdoors to observe natural cycles over 4 years [21].
- Ecosphere 2: Featured a critical design improvement: an expansive underground aquifer (groundwater layer) to maintain consistent soil moisture [21].
- Soil Types: Experiments included simulated extraterrestrial soils (Luna and Ryugu asteroid regolith) to quantify microbial communities and their role in plant growth [21].
- Data Collection: Researchers conducted metagenomic analysis to quantify and characterize microbial communities. They also monitored plant growth rates, survival, and overall ecosystem stability [21].
Key Results:
- The presence of a groundwater layer was found to be critical, acting as a stable water supply and thermal buffer, which drastically improved plant survival chances [21].
- In closed systems with a properly designed groundwater layer, plants grew in total length equal to or greater than those in open systems, though leaf elongation was suppressed, suggesting unique closed-system factors like humidity or CO₂ concentration [21].
- Microbial communities, including Cyanobacteria, played a crucial symbiotic role, contributing to oxygen production and ecosystem stability [21].
The experimental workflow for this type of research is summarized below.
The MELiSSA Pilot Plant and Control Strategy
The MELiSSA project employs a large-scale, integrated testing facility to validate the loop concept.
Objective: To demonstrate, evaluate, and improve the feasibility of the MELiSSA loop concept in ground conditions, guiding development for space applications [10].
Methodology:
- Pilot Plant Operation: The plant, located at Universitat Autònoma de Barcelona, integrates research results into a functioning physical loop of interconnected compartments [10].
- Hierarchical Control Strategy: The system uses a non-linear predictive model-based control strategy. Each compartment has a local control system, while an upper-level control determines setpoints for each compartment based on the global state and a desired functioning point [20].
- Modeling Approach: The control strategy is based on first-principles models of each compartment (physicochemical equations, stoichiometries, kinetic rates) [20]. Recent research advocates for a system-level approach (OSCAR - Optimal System-in-system Control & Architecture) to move from modeling independent processes to a network system design, improving global simulation and control [19].
The Scientist's Toolkit: Key Research Reagents and Materials
Research into closed-loop life support systems relies on a specific set of biological and technological components.
Table 3: Essential Research Materials for Artificial Ecosystem Development
| Item | Function/Application |
|---|---|
| Arthrospira platensis (Cyanobacteria) | Key organism in Compartment IVa for oxygen production, CO₂ removal, and as a high-protein food source [18] [10]. |
| Nitrosomonas europaea & Nitrobacter winogradskyi | Nitrifying bacteria used in Compartment III to convert toxic ammonia into nitrates, the preferred nitrogen source for plants [18]. |
| Higher Plant Species (e.g., wheat, rice, salad crops) | Grown in Compartment IVb for a balanced diet, oxygen production, and water recovery. A total of 32 crops are under consideration [18]. |
| Thermophilic Anoxygenic Bacteria | Used in Compartment I for the initial anaerobic degradation of solid organic wastes into volatile fatty acids and ammonia [18] [10]. |
| Simulated Lunar & Asteroid Regolith | Artificial soils used in experiments to test plant cultivation and microbial community dynamics in simulated extraterrestrial environments [21]. |
| Fixed-Bed Bioreactors | Reactor design used for Compartment III, ideal for hosting slow-growing bacterial biofilms with high conversion activity [18]. |
The comparative analysis reveals a clear trade-off: traditional physicochemical systems offer high reliability and technological maturity for air and water revival, while the MELiSSA loop presents a path toward full system closure, including food production. The choice between them is not merely technical but strategic, depending on mission duration, distance from Earth, and overarching goals for human space exploration.
Future research will focus on integrating the complexity of the system, advancing the hierarchical control strategy with tools like digital twins and artificial intelligence [22] [19], and further closing the loop in ground-based demonstrators. Upcoming forums, such as the 2025 MELiSSA Conference in Granada, will continue to be a platform for sharing the latest findings on these challenges, fostering collaboration between public and private organizations to advance the development of robust, sustainable life-support systems for both space and terrestrial applications [22].
The design of life support systems for space exploration is governed by a critical trinity of constraints: mass closure, crew time, and mission duration [2]. These fundamental drivers determine the choice between predominantly physicochemical systems and more biologically-based regenerative approaches. As mission duration extends beyond Earth orbit, the requirement for increasingly closed-loop systems becomes paramount, directly impacting the mass of consumables that must be launched and the crew time required for system maintenance [23] [2].
Traditional physicochemical life support systems have historically dominated short-duration missions from Apollo to the International Space Station (ISS), focusing primarily on air and water recycling while relying on food resupply [23]. In contrast, bioregenerative systems, exemplified by the European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) project, aim to achieve higher degrees of autonomy by using biological components to produce food, recycle water and oxygen, and process wastes [15]. This comparison guide objectively evaluates these competing approaches through the lens of our three fundamental drivers, providing researchers with a structured analysis of their relative performance and technological readiness.
Comparative System Architectures and Methodologies
Physicochemical Life Support Systems
Traditional life support systems employ physical and chemical processes to maintain crew survival. The functional architecture typically includes separate subsystems for oxygen supply, carbon dioxide removal, and water purification [23]. For short-duration missions aboard vehicles like the Crew Exploration Vehicle (CEV) and Lunar Lander, NASA has implemented open-loop systems that directly supply all atmosphere, water, and food without recycling [23]. For longer missions such as the Lunar Outpost, regenerative systems recycle oxygen and water but continue to supply food from Earth, representing an intermediate level of closure [23].
Table 1: Key Subsystems in Physicochemical Life Support Architectures
| Subsystem | Primary Function | Common Technologies | Closure Level |
|---|---|---|---|
| Oxygen Supply | Provide breathable O₂ | Oxygen tanks, electrolysis of water | Partial |
| CO₂ Removal | Remove exhaled carbon dioxide | Lithium hydroxide, molecular sieves | None |
| Condensate Purification | Recover water from humidity | Filtration, catalytic oxidation | High |
| Urine Purification | Recover water from urine | Vapor compression distillation | Moderate |
| Waste Management | Process solid wastes | Compaction, storage | Minimal |
MELiSSA Bioregenerative Systems
The MELiSSA project aims to develop a fully closed, ecosystem-inspired life support system using interconnected compartments of microorganisms, higher plants, and human crew [15]. Unlike traditional systems, MELiSSA seeks to simplify the functional architecture by using biological processes to simultaneously address multiple life support functions, potentially reducing system complexity while improving closure rates [22] [15]. The system is conceived as a micro-ecological loop where waste streams become resources for other compartments, theoretically enabling higher mass closure than purely physicochemical approaches [15].
Table 2: MELiSSA's Compartment-Based Biological Architecture
| Compartment | Primary Function | Biological Components | Inputs Processed |
|---|---|---|---|
| I | Waste breakdown | Thermophilic anaerobes | Solid waste, inedible biomass |
| II & III | Air and water revitalization | Photoheterotrophic bacteria | CO₂, waste minerals |
| IV | Food production | Higher plants, algae | CO₂, recovered nutrients |
| Crew | System driver | Human metabolism | O₂, water, food |
Comparative Performance Analysis
Mass Closure and Resupply Requirements
Mass closure represents the percentage of life support consumables that can be regenerated within the system rather than requiring resupply from Earth. The relationship between mission duration and the preferred system architecture is fundamentally driven by mass considerations.
Table 3: Mass Closure Comparison Between System Types
| Mission Parameter | Open-Loop Physicochemical | Regenerative Physicochemical | MELiSSA Bioregenerative |
|---|---|---|---|
| Oxygen Closure | None | High (~95%) | Target: Near-complete |
| Water Closure | None | High (~95%) | Target: Near-complete |
| Food Closure | None | None | Target: Significant portion |
| Resupply Mass | Linear increase with duration | Reduced increase | Minimal increase after maturity |
| Crossover Point | Preferred for <~180 days | Intermediate duration | Preferred for >~500-1000 days |
For short-duration missions (under approximately 180 days), open-loop physicochemical systems maintain a mass advantage due to their technological simplicity and lower initial mass [23]. As mission duration extends, the resupply mass penalty of open-loop systems becomes prohibitive, making regenerative physicochemical approaches more mass-efficient [23]. The MELiSSA system, with its target of significant food production in addition to air and water recycling, aims for the highest degree of autonomy, theoretically becoming mass-advantageous for the longest duration missions (likely exceeding 500-1000 days) once the biological components achieve stable operation [15] [2].
Crew Time and Operational Complexity
Crew time requirements for system operation and maintenance represent a critical resource often overlooked in initial system design. The competing systems demonstrate markedly different profiles in this dimension.
The diagram above illustrates how crew time allocation differs significantly between system types. Traditional physicochemical systems demand highly proceduralized maintenance of mechanical and chemical components [23] [24]. In contrast, MELiSSA's biological approach requires different skill sets focused on agricultural tasks and monitoring living systems, which may offer psychological benefits but presents challenges in troubleshooting complex ecological interactions [22].
Computer-simulated life support system studies have shown that crew members develop different adjustment patterns in response to variations in workload, with task management strategies evolving over mission duration [24]. This suggests that the optimal system design must consider not just total crew time requirements, but also the cognitive load and variability of tasks.
Technology Readiness and Mission Duration Fit
The appropriate life support architecture depends heavily on mission duration, with technological readiness varying significantly across approaches.
The decision framework above illustrates how mission duration directly drives architectural selection. While China's CNSA has demonstrated significant progress in bioregenerative systems through its Lunar Palace program, sustaining a crew of four analog taikonauts for a full year [2], MELiSSA and similar Western efforts remain at a lower technology readiness level for fully integrated operation.
Current NASA approaches continue to rely on resupply of food and some consumables paired with physicochemical recycling of air and water, representing a strategically conservative position that accepts ongoing logistics costs in exchange for higher reliability [2]. The comparative risk assessment between known physicochemical systems and developing bioregenerative approaches remains a central consideration for mission architects.
Experimental Protocols and Research Methodologies
Ground Analogue Testing Protocols
Research in life support systems relies heavily on ground-based analogue facilities that simulate space mission constraints. The BIO-Plex (Bioregenerative Planetary Life Support Systems Test Complex) at NASA Johnson Space Center represented a peak in integrated testing of bioregenerative systems, though the facility was eventually decommissioned [2]. Current MELiSSA research utilizes the Pilot Plant located at the Engineering School of the Universitat Autònoma de Barcelona (UAB), which serves as a terrestrial demonstration platform for the closed-loop ecosystem concept [15].
The standard experimental protocol for system-level validation involves:
- Habitat sealing and baseline measurement of all input and output masses
- Introduction of crew analogues (human subjects or metabolic simulators)
- Continuous monitoring of atmospheric gases (O₂, CO₂, trace contaminants), water quality parameters, and biomass production
- Assessment of closure rates by measuring the percentage of oxygen, water, and food that can be regenerated within the system
- Evaluation of system stability over designated test periods (typically 30 days to over a year)
The Chinese Lunar Palace programme has established particularly rigorous testing protocols, having demonstrated closed-system operations supporting a crew of four for a full year [2]. This represents the current benchmark for integrated bioregenerative system validation.
Computer-Simulated Performance Testing
Computer simulations provide complementary methodology to physical analogues, allowing for extended-duration testing of system dynamics and crew interaction. The Cabin Air Management System (CAMS) represents a validated computer-based simulation of a generic life support system used to measure performance parameters including primary and secondary task performance, and system control activities [24].
Standard simulation protocols involve:
- 30-minute performance tasks administered daily during mission simulations
- Measurement of multiple task management variables under different workload conditions
- Assessment of prospective memory (remembering to perform future tasks) as a sensitive indicator of workload impacts
- Subjective state measurements to correlate performance with psychological factors
Studies using these methodologies have revealed that prospective memory tasks show greater sensitivity to workload increases than simple reaction time measurements, providing important insights for system interface design [24].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Materials for Life Support System Investigation
| Reagent/Material | Primary Function | Research Application | System Relevance |
|---|---|---|---|
| Lithium hydroxide | CO₂ absorption | Atmosphere management benchmarking | Physicochemical systems |
| Nitrogen-fixing bacteria | Convert atmospheric N₂ | Soil replacement in plant growth | MELiSSA Compartments I & IV |
| Spirulina algae | Oxygen production, food source | Photosynthetic efficiency studies | MELiSSA Compartment IV |
| Ion-specific resins | Trace contaminant removal | Water purification optimization | Both system types |
| Lithium chloride | Humidity control | Cabin atmosphere testing | Physicochemical systems |
| Specific plant cultivars | Food production, O₂ generation | Space-adapted agriculture research | MELiSSA Compartment IV |
| Molecular sieves | CO₂ concentration | Air revitalization systems | Physicochemical systems |
| Whey-based media | Bacterial growth substrate | Waste processing optimization | MELiSSA Compartments I-III |
The comparative analysis of life support systems through the fundamental drivers of mass closure, crew time, and mission duration reveals a complex trade space without universal solutions. Traditional physicochemical systems maintain advantages in technological maturity and reliability for short to medium duration missions, while MELiSSA-inspired bioregenerative approaches offer the potential for superior mass closure and crew well-being benefits on longer-duration missions.
Current evidence suggests that strategic investment gaps in Western bioregenerative research, contrasted with China's advancing Lunar Palace programme, create potential vulnerabilities for future leadership in long-duration human space exploration [2]. A balanced portfolio approach appears most prudent, advancing regenerative physicochemical systems for nearer-term lunar missions while sustaining research and development into biological systems for future Mars missions and long-duration lunar habitation.
The optimal path forward likely involves hybrid architectures that selectively integrate biological components where they provide clear advantages, while retaining robust physicochemical systems for critical life support functions. This balanced approach mitigates risk while progressively advancing closure rates and autonomy in preparation for humanity's future as a multi-planetary species.
Operational Technologies: Side-by-Side Comparison of System Functions
For long-duration human space missions, achieving a closed-loop life support system is a critical necessity. These systems must reliably capture carbon dioxide (CO₂) and generate breathable oxygen (O₂) to minimize dependence on Earth-based resupply. The European Space Agency's Micro-Ecological Life Support System Alternative (MELiSSA) program aims to create a robust, bioregenerative life support system (BLSS) by emulating a terrestrial ecosystem [3]. This guide objectively compares two fundamental approaches for air revitalization: the established chemical Sabatier process and biological algal/plant photosynthesis, framing the analysis within the ongoing research between MELiSSA-style BLSS and traditional physicochemical systems [25] [3].
The Sabatier process and biological photosynthesis represent fundamentally different pathways for air revitalization, each with distinct performance characteristics, advantages, and challenges.
Chemical Sabatier Process
The Sabatier process is a thermochemical reaction that hydrogenates CO₂ to produce methane (CH₄) and water (H₂O). On the International Space Station (ISS), this water is then electrolyzed to provide oxygen, completing a semi-closed loop [26]. The process is highly exothermic and requires efficient thermal management to maintain optimal catalyst performance and prevent deactivation [27].
Core Reaction: CO₂ + 4H₂ → CH₄ + 2H₂O (ΔH = -164.9 kJ/mol) [27]
Biological Photosynthesis (Algal/Plant)
Biological systems use photosynthesis, where light energy powers the conversion of CO₂ and water into carbohydrates and oxygen. This approach is championed by BLSS initiatives like MELiSSA, which uses interconnected compartments of microorganisms and plants to recycle wastes and produce oxygen, water, and food [3]. In algae and cyanobacteria, this process is driven by massive protein complexes like Photosystem II (PSII), which splits water to produce oxygen [28].
Core Reaction: 6CO₂ + 6H₂O → C₆H₁₂O₆ + 6O₂
Performance Data Comparison
The table below summarizes key performance metrics and characteristics of the two technologies, highlighting their different operational paradigms.
Table 1: Performance and Characteristics Comparison of Sabatier and Photosynthetic Systems
| Parameter | Chemical Sabatier Process | Algal/Plant Photosynthesis |
|---|---|---|
| Primary Products | Methane (CH₄), Water (H₂O) [26] | Oxygen (O₂), Biomass (Food) [3] |
| O₂ Production | Indirect (via H₂O electrolysis) [26] | Direct [3] |
| CO₂ Conversion Efficiency | >90% CH₄ yield demonstrated in optimized reactors [27] | Limited by natural pathway inefficiency (~1% of theoretical carbon bound in biomass) [29] |
| Operational Challenges | Thermal management, catalyst deactivation, H₂ stoichiometry [27] [26] | System stability, nutrient management, light requirements, algal die-offs causing O₂ depletion [30] [3] |
| Technology Readiness | High (operational on the ISS) [26] | Varies (component testing on ISS; ground demonstrations like Lunar Palace 1) [25] [3] |
| System Complexity | Requires separate O₂ generation (electrolyser) and deals with H₂ logistics [26] | Integrated but complex biological system requiring control of growth environment [3] |
| Additional Outputs | Potentially renewable natural gas [27] | Food production, water purification, psychological benefits [3] |
Experimental Protocols and Methodologies
Research and development in air revitalization rely on sophisticated experimental setups to test, optimize, and validate system performance.
Sabatier Reactor Testing and Analysis
Research on advanced Sabatier reactors focuses on overcoming thermal management challenges to maintain high conversion efficiency.
Table 2: Key Research Reagents and Materials for Sabatier Reaction
| Item | Function in Experiment |
|---|---|
| Nickel-based Catalyst | The most widely used catalyst to accelerate the methanation reaction; offers high selectivity and low cost [27] [26]. |
| Molten Salt Coolant | A heat transfer fluid (e.g., molten salt) for efficient, isothermal heat removal from the exothermic reaction, preventing hotspot formation and catalyst sintering [27]. |
| Packed Bed Reactor | A vessel filled with catalyst pellets where the CO₂ and H₂ gases react; its design is crucial for heat and mass transfer [27]. |
Detailed Workflow:
- Reactor Setup: A heat-exchanger type packed bed reactor is constructed, with internal tubes for coolant (e.g., molten salt) flow and the packed nickel catalyst [27].
- Parameter Control: The reactor is operated at elevated temperatures (300–400 °C) and pressures (~30 bar). Critical parameters like feed gas temperature, space velocity (e.g., up to 10,000 h⁻¹), and coolant flow rate are meticulously controlled [27].
- Performance Monitoring: The composition of the outlet gas stream is analyzed using gas chromatography to determine CO₂ conversion and CH₄ yield. Temperature sensors along the reactor bed track the formation of any "hot spots" [27].
- Data Analysis: A pseudo-homogeneous mathematical model is used to simulate reactor performance, optimizing operating conditions to achieve >90% conversion while managing heat release [27].
Figure 1: Experimental workflow for testing and analyzing a Sabatier reactor, showing the integration of feed gases, reactor core, cooling system, and performance monitoring.
Photosynthetic System Characterization
For biological systems, research aims to understand and improve the fundamental efficiency of photosynthesis, both at the molecular and system level.
Table 3: Key Research Reagents and Materials for Photosynthesis Research
| Item | Function in Experiment |
|---|---|
| Cyanobacteria / Algae | Model photosynthetic organisms (e.g., for MELiSSA); easily genetically manipulated and grown rapidly in bioreactors [29] [3]. |
| Higher Plants (e.g., Lettuce, Wheat) | Food-producing crops for BLSS; selected for nutritional value, resource requirements, and edible biomass ratio [3]. |
| Spectroscopy Systems | Advanced tools (e.g., 2D electronic-vibrational spectroscopy) to track energy flow and dynamics within light-harvesting complexes like PSII [28]. |
Detailed Workflow:
- Organism Cultivation: Cyanobacteria or plants are grown in controlled environment chambers (e.g., MELiSSA's PaCMan unit) that regulate light intensity, temperature, CO₂, and nutrient delivery [3].
- Molecular Analysis: For fundamental studies, Photosystem II supercomplexes are isolated. Advanced spectroscopy techniques like two-dimensional electronic-vibrational spectroscopy are employed. This method provides high-resolution insights into energy transfer pathways among chlorophyll molecules, overcoming the challenge of analyzing a "sea of identical molecules" [28].
- System Modeling: Data from spectroscopic studies is used to build simulations of PSII's energy landscape. These models help explain design principles, such as how a "flat" layout allows energy to roam randomly before being used, providing both efficiency and built-in photoprotection [28].
- Performance Metrics: At the system level, key metrics are monitored: O₂ production rate, CO₂ uptake, biomass growth, and the impact of environmental factors (e.g., microgravity, radiation). For algal ponds, stability is critical to avoid die-offs that consume O₂ [30].
Figure 2: The dual-level experimental approach for characterizing photosynthetic systems, combining molecular-level analysis of light-energy conversion with system-level monitoring of gas exchange and growth.
The MELiSSA Framework and System Integration
The MELiSSA project exemplifies the effort to integrate biological and physicochemical components into a reliable, closed-loop ecosystem for space [3]. Its goal is to recycle organic and inorganic wastes into oxygen, potable water, and fresh food through a loop of interconnected compartments, each performing a specific function (e.g., photoautotrophic oxygen production by cyanobacteria, waste degradation by fermentative bacteria, food production by higher plants) [22] [3].
In this context, the choice between Sabatier and photosynthesis is not necessarily mutually exclusive but is strategic. Current US approaches for lunar exploration largely rely on physicochemical systems with resupply, whereas China's CNSA, having embraced and advanced the bioregenerative programs NASA previously discontinued, has demonstrated a fully integrated, closed-loop BLSS in the Beijing Lunar Palace, sustaining a crew of four for a full year [25]. This represents a significant divergence in strategy and capability.
The MELiSSA conference highlights the ongoing research into integrating these systems, with sessions covering everything from "Air Revitalization" using biological and chemical methods to "Plant Characterization" and "Waste Valorisation" [22]. The ultimate aim is a system that is not only technologically feasible but also autonomous and robust enough for long-duration missions, where resupply is not an option [3].
Figure 3: A simplified schematic of the MELiSSA loop concept, showing the integration of different biological compartments to create a closed ecosystem where waste from one compartment becomes a resource for another.
The comparison between the chemical Sabatier process and algal/plant photosynthesis reveals a trade-off between high-technology readiness and direct closure of multiple loops. The Sabatier process offers a high-efficiency, compact solution for specific CO₂ reduction and water recovery needs, making it suitable for near-term missions where system simplicity and proven reliability are paramount, as on the ISS.
In contrast, biological photosynthesis, as developed within programs like MELiSSA and by the CNSA, offers a broader, bioregenerative approach. It directly produces oxygen and food from CO₂ and waste, contributing to a more self-sustainable and psychologically supportive habitat. While challenges in system stability, scaling, and understanding space-environment effects remain, the potential for a fully closed, Earth-like ecosystem makes it a compelling solution for the future of long-duration "endurance-class" missions to the Moon and Mars [25] [3]. The path forward will likely involve continued refinement of both approaches, with the optimal architecture potentially being a hybrid that leverages the strengths of both chemical and biological systems.
The pursuit of sustainable, closed-loop life support systems is a critical challenge for long-duration human space exploration. Effective water recovery from waste streams is a cornerstone of this effort, with two principal technological paradigms emerging: physicochemical filtration and biological processing. The European Space Agency's Micro-Ecological Life Support System Alternative (MELiSSA) initiative exemplifies the biological approach, aiming to create a closed-loop life support system with near-100% efficiency through a consortium of microorganisms [8]. In contrast, traditional systems often rely on sequential physicochemical filtration units. This guide objectively compares the performance, applications, and experimental data for these two approaches within the context of advanced life support research, providing scientists and engineers with a structured analysis to inform technology selection and development.
Fundamental Principles and System Architectures
The two approaches are founded on distinct operational principles, which dictate their system architectures and integration logic.
Physicochemical Filtration
Physicochemical systems rely on physical separation and chemical transformation processes to purify water. A typical integrated system for complex waste streams, such as laundry wastewater, employs a sequential, multi-barrier architecture [31]. The process usually begins with a pre-filtration stage to remove large particulates, followed by a low-pressure membrane process like ultrafiltration (UF) for clarification and removal of finer solids and microbes. A high-pressure membrane process, such as nanofiltration (NF) or reverse osmosis (RO), often serves as the final polishing stage to remove dissolved ions, small organic molecules, and surfactants. Alternatively, an adsorption process using activated carbon can be used to remove trace organic pollutants and odors [31].
Biological Processing (MELiSSA)
The MELiSSA framework is a bioregenerative life support system that mimics a terrestrial ecosystem. Its core principle is using defined consortia of microorganisms in interconnected bioreactors to convert waste streams, including carbon dioxide and human metabolic waste, into oxygen, clean water, and food [8] [32]. The system is compartmentalized into five main processes, each hosting specific microbial communities and functions, which work in concert to close the ecological loop [8]. This approach aims not merely to clean water but to reintegrate it into a broader ecological cycle.
The logical workflow of each system highlights their fundamental differences in approaching loop-closure, as summarized below.
Performance Comparison and Experimental Data
The following tables summarize quantitative performance data and key characteristics of both approaches, synthesized from terrestrial and space-oriented research.
Table 1: Treatment Performance of Physicochemical Systems for Specific Waste Streams
| Waste Stream | Technology | Key Performance Metrics | Source |
|---|---|---|---|
| Swimming Pool Filter Wash Water | Sedimentation, Granular Filtration, UF, ClO₂ Disinfection | Turbidity: 23.1 NTU → 0.25 NTU\nCOD_Mn: 10.4 → 1.6 mg O₂/L\nMicrobial Count: 1.6×10⁴ → 30 CFU/mL [33] | |
| Laundry Wastewater (Domestic) | Coagulation/Flocculation (pH 6, 2.5 g/L dose) | Anionic Surfactant Removal: 92%\nCOD Removal: 83%\nTurbidity Removal: 85% [31] | |
| Laundry Wastewater (Industrial) | Coagulation/Flocculation + Microfiltration | Anionic Surfactant Removal: 72%\nCOD Removal: 69%\nTurbidity Removal: 99% [31] | |
| Laundry Wastewater | Ultrafiltration (UF) as a Unit Process | COD Removal: ~88%\nTurbidity Removal: ~98.4% [31] | |
| Laundry Wastewater | Nanofiltration (NF) as a Unit Process | COD Removal: ~97%\nTurbidity Removal: ~98% [31] |
Table 2: Comparative Analysis of System Characteristics
| Parameter | Physicochemical Filtration | Biological Processing (MELiSSA) |
|---|---|---|
| Core Principle | Physical separation & chemical transformation [31] | Microbial ecosystems & natural cycles [8] [32] |
| System Architecture | Sequential, multi-barrier units [31] | Interconnected, compartmentalized bioreactors [8] |
| Primary Output | Clean water [31] | Water, oxygen, and food (bioregenerative) [32] |
| Technology Readiness | High (Terrestrial); Established on ISS [32] | Medium (Under development; Pilot Plant phase) [8] |
| Loop-Closure | Limited to water recovery | High potential for full air, water, and nutrient cycling [32] |
| Key Challenge | Membrane fouling; waste brine/sludge management [31] | System stability & control of complex biological communities [8] |
Experimental Protocols
To ensure reproducibility and provide a clear framework for comparative research, detailed methodologies for key experiments are outlined below.
Protocol 1: Integrated Physicochemical Treatment of Laundry Wastewater
This protocol is adapted from studies on laundry wastewater treatment and reflects a multi-barrier, full-scale approach [31].
- 1. Sample Collection & Characterization: Collect laundry wastewater from the first rinse cycle. Characterize the raw wastewater by measuring pH, turbidity (NTU), Chemical Oxygen Demand (COD), and surfactant concentration.
- 2. Coagulation-Flocculation Pretreatment:
- Use Jar test apparatus.
- Adjust wastewater sample pH to 6.0 using 1M H₂SO₄ or 1M NaOH.
- Add a coagulant (e.g., alum or ferric chloride) at a dose of 2.5 g/L.
- Operate at 100 rpm for 2 minutes (rapid mix), then 30 rpm for 15 minutes (slow mix).
- Allow solids to settle for 30 minutes.
- Analyze supernatant for COD, turbidity, and surfactant removal.
- 3. Low-Pressure Membrane Filtration (Ultrafiltration - UF):
- Pass the pretreated supernatant through a flat-sheet or hollow-fiber UF membrane with a specified molecular weight cutoff (e.g., 10-100 kDa).
- Operate in dead-end or cross-flow filtration mode at a transmembrane pressure of 1-3 bar.
- Collect the permeate and analyze for turbidity and COD.
- 4. High-Pressure Membrane Polishing (Nanofiltration - NF):
- Feed the UF permeate into a spiral-wound NF membrane.
- Operate at a transmembrane pressure of 5-15 bar.
- Collect the final permeate and analyze for conductivity, hardness, and trace surfactant concentration.
- 5. Data Analysis: Calculate removal efficiencies for all key parameters at each stage to determine the contribution of each unit process.
Protocol 2: Biological Water Reclamation in a Compartmentalized Bioreactor
This protocol outlines the general principles for operating a bioreactor system inspired by the MELiSSA loop, focusing on the degradation of organic pollutants in wastewater [8] [32].
- 1. Bioreactor Inoculation and Conditioning:
- Compartment I (Liquefaction): Inoculate a stirred-tank anaerobic bioreactor with a mixed culture of fermentative bacteria (e.g., from anaerobic sludge). Feed it with a synthetic or real wastewater stream containing complex organic matter (e.g., from human waste or food residues).
- Compartment II (Photoheterotrophs): Inoculate a photobioreactor with a defined strain of photosynthetic bacteria (e.g., Rhodospirillum rubrum). Feed it with the effluent from Compartment I, which contains volatile fatty acids.
- 2. System Operation and Monitoring:
- Operate the system in continuous or batch-fed mode, maintaining optimal temperature, pH, and nutrient levels for each compartment.
- For the photobioreactor, provide continuous illumination and monitor optical density to track microbial growth.
- Continuously monitor the system's effluent for Total Organic Carbon (TOC), ammonium (NH₄⁺), nitrate (NO₃⁻), and pH.
- 3. Process Integration and Stability Testing:
- Once individual compartments are stable, connect them to establish a continuous flow from Compartment I to Compartment II.
- Operate the integrated system for an extended period (e.g., several weeks) to assess stability, control strategies, and overall removal efficiency of organic carbon.
- 4. Data Analysis: Correlate microbial growth (optical density, cell count) in each compartment with the reduction of TOC and the transformation of nitrogen species in the effluent.
The Scientist's Toolkit: Key Research Reagents and Materials
Successfully implementing and researching these water recovery technologies requires a suite of specialized reagents and materials.
Table 3: Essential Research Reagents and Materials
| Item | Function/Application | Relevance |
|---|---|---|
| Ultrafiltration (UF) & Nanofiltration (NF) Membranes | Removal of colloids, macromolecules, viruses (UF), and divalent ions/small organics (NF) [31]. | Core component of physicochemical systems; used for molecular separation and polishing. |
| Activated Carbon | Adsorption of dissolved organic compounds, fragrances, and trace contaminants via a high surface area [31]. | A polishing step in integrated filtration trains for improving water quality and odor control. |
| Coagulants (e.g., Alum, FeCl₃) | Destabilization of colloidal particles and emulsions in wastewater to form settleable flocs [31]. | Critical pretreatment step to reduce fouling load on downstream membrane units. |
| Defined Microbial Strains (e.g., Anabaena sp., Rhodospirillum rubrum) | Serve as the catalytic core of biological systems for functions like carbon fixation, nitrogen cycling, and waste degradation [8] [32]. | Essential for establishing and studying controlled, predictable bioregenerative processes. |
| Specialized Bioreactors (Photobioreactor, Stirred-Tank) | Provide a controlled environment (light, temperature, gas exchange, mixing) for cultivating microorganisms [8]. | Enables the study and optimization of biological processes under defined conditions. |
| Chemical Oxygen Demand (COD) Test Kits | Standardized method for quantifying the amount of oxidizable organic matter in a water sample. | A universal metric for evaluating and comparing the performance of both physicochemical and biological systems. |
This comparison guide illustrates that the choice between physicochemical filtration and biological processing is not a simple matter of superiority, but one of strategic alignment with mission requirements. Physicochemical systems offer high-technology readiness and robust, predictable performance for dedicated water recovery, making them suitable for near-term missions where reliability is paramount. In contrast, the MELiSSA-inspired biological approach represents a paradigm shift towards a truly bioregenerative, closed-loop ecosystem capable of producing multiple resources—air, water, and food—from waste [32]. While presenting significant challenges in system stability and control, its potential to enable extreme long-duration space exploration is unmatched. Future research should focus on hybrid systems that leverage the immediate efficiency of physicochemical methods with the long-term sustainability of biological processes, ultimately creating the resilient life support systems necessary for humanity to become a multi-planetary species.
The quest for sustainable human presence in space, particularly for long-duration missions to the Moon and Mars, has intensified the focus on advanced life support systems. Central to this endeavor is the effective management of organic waste, a challenge that mirrors terrestrial needs for circular economies. Within this context, the Micro-Ecological Life Support System Alternative (MELiSSA) project, driven by the European Space Agency, aims to create a robust, bioregenerative life support system [4]. This system is designed to recycle waste into oxygen, water, and food through interconnected biological processes. A critical function within MELiSSA, and its terrestrial analogs, is the choice of waste processing technology. This guide provides an objective comparison between two principal valorization pathways: incineration/oxidation and anaerobic digestion (AD), with a specific focus on their performance metrics, experimental data, and relevance to closed-loop systems.
Incineration/Oxidation is a physicochemical process that involves the combustion of organic waste at high temperatures (typically >850°C) in an oxygen-rich environment. The primary goals are volume reduction, destruction of pathogens and toxic compounds, and energy recovery in the form of heat [34]. In a life support context, this heat can be utilized for power generation or thermal control, while the resulting flue gases require extensive cleaning before being vented or recycled.
Anaerobic Digestion (AD) is a biological process where a consortium of microorganisms breaks down biodegradable material in the absence of oxygen. The process yields biogas (a mixture of methane and carbon dioxide) and a nutrient-rich effluent called digestate [35]. The biogas can be used as a renewable fuel for electricity, heat, or, as demonstrated in recent research, further reformed into syngas for broader applications [36]. The digestate can be applied as a biofertilizer, closing the nutrient loop—a feature of paramount importance for food production in bioregenerative life support systems like MELiSSA [37].
The following diagram illustrates the logical decision-making process for selecting a waste valorization technology within a life support system context, highlighting the divergent pathways and outputs of AD and incineration.
Performance Comparison: Quantitative Data
The choice between AD and incineration involves trade-offs between energy efficiency, environmental impact, and resource recovery. The following tables summarize key performance metrics based on recent experimental studies.
Table 1: Energy Recovery and Process Efficiency
| Parameter | Anaerobic Digestion | Incineration/Oxidation | Experimental Context |
|---|---|---|---|
| Primary Energy Product | Biogas (CH~4~, CO~2~) | Thermal Energy | [36] [34] |
| Typical Methane Yield | 302 - 526 mL CH~4~/g VS | Not Applicable | BMP tests on municipal/organic waste [38] |
| Net Energy Recovery | Positive (energy generation > consumption) | Highly dependent on waste composition & water content; can be net-negative for high-moisture waste like sewage sludge [34] | Comparison for sewage sludge treatment [34] |
| Carbon Conversion | Up to 80% increase in methane yield via co-digestion vs mono-digestion [35] | Near-total conversion to CO~2~ | Review of AD process enhancements [35] |
Table 2: Environmental Impact and Nutrient Management
| Parameter | Anaerobic Digestion | Incineration/Oxidation | Experimental Context |
|---|---|---|---|
| Greenhouse Gas Impact | Avoided emissions; reduces landfill methane [34] [37] | Direct CO~2~ emissions; potential for air pollutants (SO~x~, NO~x~) without advanced flue gas treatment [34] | Life-cycle comparison of waste treatment methods [34] |
| Nutrient Recovery | High (Digestate is a source of N, P, K for fertilizing) [37] | Limited (Nutrients largely lost; P and K may be concentrated in ash, but often in less bioavailable forms) [34] | Review of AcoD for poultry manure and food waste [37] |
| Residue Management | Digestate (can be used as fertilizer) | Ash (requires disposal as hazardous waste) [34] | Comparison of sewage sludge treatment [34] |
Experimental Protocols and Methodologies
Protocol for Anaerobic Digestion with Biochar Enhancement
The following workflow details a modern AD experiment designed to enhance methane production using co-pyrolysis biochar, a methodology relevant for increasing the resilience and efficiency of a BLSS [39].
Detailed Methodology [39] [38]:
- Feedstock and Inoculum Preparation: Food waste (FW) is collected, homogenized, and characterized for total solids (TS), volatile solids (VS), and elemental composition (C, N, H). Anaerobic sludge from a functioning digester is used as the inoculum. The inoculum-to-substrate (I:S) ratio is typically set to 2.0 on a VS basis to ensure adequate microbial activity.
- Biochar Synthesis and Characterization: Biochar is produced via co-pyrolysis of digestate and rice straw (e.g., at 500°C under limited oxygen). The resulting biochar (DRB) is characterized using:
- Scanning Electron Microscopy (SEM): To analyze surface morphology and porous structure.
- X-ray Photoelectron Spectroscopy (XPS) and FTIR: To determine surface chemical composition and functional groups (e.g., C-O, C=O).
- Surface Area and Porosity Analysis (S_BET): To measure specific surface area.
- Electrical Conductivity (EC): To assess potential for direct interspecies electron transfer (DIET).
- Biochemical Methane Potential (BMP) Assay: Experiments are conducted in batch reactors (e.g., 500 mL to 2 L working volume) maintained at mesophilic temperature (35-37°C). The experimental setup includes a control (no biochar) and treatments with different biochars (e.g., DB, RB, DRB) added at a defined dosage (e.g., 5-10 g/L).
- Process Monitoring: Methane production is measured daily, typically by water displacement or automated gas metering systems. Liquid samples are periodically taken to monitor pH, volatile fatty acids (VFA) concentration, and ammonia-nitrogen (NH~3~-N) to assess process stability.
- Kinetic Modeling and Statistical Analysis: Cumulative methane production data is fitted to kinetic models (e.g., Modified Gompertz, First-order) to determine the methane production potential (B~0~), maximum production rate (R~max~), and lag phase (λ). Statistical analyses confirm the significance of observed differences between treatments.
Protocol for Incineration Process Evaluation
While the provided search results focus more on the outcomes of incineration, a standard experimental protocol involves [34]:
- Feedstock Preparation: Waste is dried to reduce moisture content, which is critical for achieving autothermal combustion (where the energy from combustion is sufficient to sustain the process without auxiliary fuel).
- Combustion Process: The waste is fed into a furnace and combusted at temperatures exceeding 850°C with excess air to ensure complete oxidation and destruction of organic pollutants.
- Emission and Residue Analysis:
- Flue Gas Analysis: Concentrations of O~2~, CO~2~, CO, SO~x~, NO~x~, and particulate matter are continuously monitored.
- Ash Analysis: The resulting bottom ash and fly ash are collected and analyzed for heavy metal content and leachability to determine hazardousness and disposal requirements.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Advanced Anaerobic Digestion Research
| Reagent/Material | Function in Research | Specific Example |
|---|---|---|
| Co-pyrolysis Biochar (DRB) | Enhances microbial DIET, buffers pH, adsorbs inhibitors (e.g., ammonia), increases methane yield [39]. | Biochar from digestate and rice straw (75:25 ratio), pyrolyzed at 500°C [39]. |
| Ni-based Catalyst | Catalytic reforming of biogas into syngas (H~2~ + CO), expanding product utility [36]. | Rh-doped Ni-catalyst on MgAl mixed oxides for dry reforming of methane (DRM) [36]. |
| Anaerobic Sludge Inoculum | Source of a diverse, mixed microbial community essential for the four stages of AD (hydrolysis, acidogenesis, acetogenesis, methanogenesis) [38]. | Granular sludge from a municipal wastewater treatment plant digestor. |
| Modified Gompertz Model | Kinetic model used to predict methane production profile, including lag phase and maximum production rate [38]. | Standard equation for non-linear regression analysis of cumulative BMP data. |
| Sorption Bed for H~2~S Removal | Pre-treatment step to clean biogas by removing hydrogen sulfide, which is toxic to catalysts and microbes [36]. | Iron hydroxide-based sorbent material [36]. |
The integration of waste valorization technologies is a critical subsystem for the success of long-duration space missions and terrestrial circular economies. The experimental data and comparisons presented herein demonstrate that the choice between incineration/oxidation and anaerobic digestion is not a simple binary.
- Incineration/Oxidation serves a vital role in volume reduction and pathogen destruction, and can be an effective waste-to-energy solution for drier, more combustible waste streams. However, its limitations in nutrient recycling and potential for net-negative energy balance with wet wastes are significant drawbacks for a closed-loop system.
- Anaerobic Digestion aligns more closely with the bioregenerative philosophy of MELiSSA. It demonstrates a clear superiority in nutrient recovery and recycling, producing a valuable biofertilizer essential for sustainable food production. Furthermore, the biogas it produces is a versatile renewable energy carrier. Recent advancements, such as the use of biochar to enhance microbial stability and methane yield, and the catalytic reforming of biogas to syngas, significantly increase the technology's resilience and product flexibility [36] [39].
For a future where life support systems must be largely self-sustaining, Anaerobic Digestion presents a more synergistic and multi-output pathway. It effectively transforms the challenge of waste management into an opportunity for producing energy, closing nutrient cycles, and contributing to the overall stability of a controlled ecological life support system.
The viability of long-duration space missions, such as a human presence on the Moon or a crewed mission to Mars, hinges on the development of robust and sustainable life support systems. These systems must reliably provide astronauts with essential consumables—food, oxygen, and water—while managing waste in a environment where resupply from Earth is prohibitively expensive or impossible. Two primary technological paths have emerged: the well-established Physical-Chemical (PC) systems, which rely on stored consumables and mechanical recycling, and the advanced Bioregenerative Life Support Systems (BLSS), which use biological processes to create a regenerative environment [2]. The European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) project is a leading BLSS research initiative, aiming to create a robust, circular ecosystem for space [40]. At the core of this paradigm shift is the method of food production. This guide provides a comparative analysis of the incumbent technology, Stored Rations, and the bioregenerative alternative, In-Situ Cultivation, framing the discussion within the broader context of MELiSSA versus traditional PC systems research.
Stored Rations (The Physical-Chemical Approach)
The stored rations approach is an extension of the open-loop life support system model. All food required for a mission is launched from Earth and stored on the vehicle or habitat. This method is coupled with PC systems for air and water revitalization, such as the Oxygen Generation System and the Water Recovery System used on the International Space Station (ISS). These systems rely on energy-intensive mechanical and chemical processes (e.g., Sabatier reactors, molecular sieves) to recycle water and oxygen, but they cannot produce food [17]. The food itself is pre-packaged, stabilized for long-term storage, and represents a fixed mass that is consumed linearly throughout the mission.
In-Situ Cultivation (The MELiSSA-Inspired BLSS Approach)
In-situ cultivation is the cornerstone of a closed-loop, or circular, life support system. Within a BLSS, biological components—specifically higher plants and microorganisms like cyanobacteria—are cultivated to regenerate the atmosphere, purify water, and produce food. The MELiSSA project is a prominent example, conceptualized as a closed-loop ecosystem with multiple compartments, each performing a specific metabolic function [40]. In this framework, cyanobacteria are particularly versatile; they can perform bioweathering of regolith (lunar or Martian soil) to free up nutrients, produce oxygen through photosynthesis, fix carbon dioxide, and generate nutritious biomass for direct human consumption or as a feedstock for other biological processes [17]. Higher plants, cultivated in controlled environment agriculture (CEA) systems, provide a more diverse and palatable food source while contributing to water and air purification.
Table 1: High-Level System Comparison between Stored Rations and In-Situ Cultivation
| Parameter | Stored Rations (PC-Based) | In-Situ Cultivation (BLSS/MELiSSA) |
|---|---|---|
| System Open/Closed Loop | Open loop (food); Partially closed (air, water) | Fully closed or semi-closed loop |
| Primary Food Source | Pre-packaged, processed food from Earth | Fresh biomass from plants & cyanobacteria |
| Oxygen Production | Electrolysis of water, chemical oxygen generators | Photosynthesis (plants, cyanobacteria) |
| Carbon Dioxide Processing | Mechanical (e.g., molecular sieves), Sabatier reaction | Photosynthetic fixation |
| Waste Recycling | Limited processing (e.g., water recovery from urine) | Integration of organic waste into nutrient loops |
| Initial Mass to Orbit | Very High (all consumables launched) | Lower (requires only initial seeds/starter cultures) |
| Technology Readiness Level | High (flight-proven on ISS) | Low to Medium (under active R&D, e.g., Lunar Palace) |
| System Complexity | High complexity in PC hardware | High complexity in biological control & integration |
Table 2: Quantitative Performance Metrics for a 4-Person Crew
| Metric | Stored Rations | In-Situ Cultivation | Notes & Sources |
|---|---|---|---|
| Daily Food Mass (dry) | 3.2 kg/day | Produced on-demand | Based on 0.80 kg/astronaut [17] |
| Daily Oxygen Requirement | 3.56 kg/day | 3.56 kg/day | Must be produced by the system [17] |
| Daily CO₂ Production | 4.32 kg/day | 4.32 kg/day | Must be processed by the system [17] |
| Mission Mass Penalty (1 yr) | ~1,168 kg (food only) | Minimal (after initial setup) | A major driver for BLSS research [2] |
| Caloric Output | Fixed at launch | Theoretically unlimited with system stability | Dependent on cultivation area and efficiency |
| Nutritional Quality | Stable, can degrade over time | Fresh, high in vitamins & antioxidants | Cyanobacteria are rich in protein & nutrients [17] |
Experimental Protocols and Methodologies
Protocol for Analyzing Stored Ration System Efficiency
The efficiency of a PC-based life support system is typically validated through integrated tests in closed-loop habitat analogs.
- Objective: To determine the mass closure, energy efficiency, and reliability of a PC life support system over a defined mission duration.
- Test Setup: A closed-chamber facility, such as those used in the Lunar-Mars Life Support Test Project (LMLSTP), equipped with a full suite of PC systems: CO₂ reduction systems (e.g., Sabatier reactors), Oxygen Generation Assemblies (OGA), and Water Recovery Systems (WRS) [17].
- Procedure:
- Baseline Measurement: The total initial mass of all stored consumables (food, water, oxygen canisters, chemical CO₂ scrubbers) is recorded.
- Crew Habitation: A crew resides in the closed chamber for the test duration (e.g., 60 days as in LMLSTP Phase IIa).
- System Monitoring: PC systems are operated to maintain oxygen levels (20.3-21.4%) and CO₂ levels (0.30-0.55%). Water is recycled from all sources (shower, urine, condensate) using Vapor Compression Distillation (VCD) and Ultrafiltration/Reverse Osmosis (UF/RO) [17].
- Data Collection: Daily tracking of (i) mass of food consumed, (ii) water recycled vs. lost, (iii) oxygen consumed, (iv) CO₂ scrubbed, and (v) total energy consumption of all PC hardware.
- Analysis: Calculation of key metrics: total mass closure, water recovery rates, oxygen resupply mass, and power consumption per kg of consumable supported.
Protocol for Evaluating Cyanobacteria-Based In-Situ Cultivation
This protocol assesses the viability of cyanobacteria as a core component of a BLSS for resource generation.
- Objective: To measure the oxygen production, biomass yield, and nutrient output of cyanobacteria in a simulated space-grade photobioreactor (PBR).
- Strain Selection & Inoculation: Select extremophile cyanobacterial strains (e.g., Anabaena, Spirulina) known for high photosynthetic efficiency and resilience. Cultures are inoculated into a liquid growth medium within a sealed PBR [17].
- Growth Conditions: The PBR environment is strictly controlled:
- Light: Constant photon flux provided by LED arrays at specific wavelengths optimal for photosynthesis.
- Atmosphere: The PBR is fed with a simulated Martian atmosphere (95% CO₂) or cabin air to assess carbon fixation rates.
- Nutrients: In some experiments, crushed lunar or Martian regolith simulant is added to the medium to test bioweathering and in-situ nutrient acquisition (Stage 1 of a proposed three-stage system) [17].
- Data Collection & Harvesting:
- Gas Exchange: Continuous monitoring of O₂ production and CO₂ consumption rates via gas analyzers.
- Biomass Yield: Regular harvesting and measurement of biomass. The biomass is then analyzed for nutritional content (proteins, carbohydrates, lipids, vitamins).
- Downstream Processing: A portion of the biomass can be diverted to a third-stage bioreactor for the production of biofuels like methane, demonstrating system versatility [17].
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Materials for BLSS and PC System Experimentation
| Reagent / Material | Function in Research | Example Application |
|---|---|---|
| Lunar/Martian Regolith Simulant | Geochemical analog for in-situ resource utilization (ISRU) experiments. | Testing cyanobacteria-mediated bioweathering to liberate nutrients [17]. |
| Cyanobacteria Starter Cultures | Model photosynthetic organisms for O₂ production and biomass. | Cultivation in photobioreactors to study gas exchange and nutritional output [17]. |
| Controlled Environment Agriculture (CEA) Chambers | Precisely managed growth environments for higher plants. | Studying plant growth, yield, and resource use efficiency in closed-loop habitats. |
| Gas Chromatography-Mass Spectrometry (GC-MS) | Analytical instrument for identifying and measuring trace gases and volatile organic compounds. | Monitoring air quality and metabolic outputs in closed-system tests [17]. |
| Sabatier Reactor | Physical-chemical system for converting CO₂ and hydrogen into methane and water. | Used as a baseline technology for CO₂ reduction in PC system comparisons [17]. |
| Vapor Compression Distillation (VCD) Unit | Hardware for recovering potable water from wastewater (e.g., urine). | Key component for water recycling in PC systems; used to establish baseline recovery rates (~98%) [17]. |
| Model Predictive Control (MPC) Software | Advanced algorithm for managing complex, interconnected systems. | Supervisory control of the entire MELiSSA loop to optimize recycling and ensure crew safety [40]. |
System Control and Integration: The Role of Model Predictive Control
Operating a regenerative BLSS like MELiSSA is a complex challenge involving the coordination of heterogeneous biological and physicochemical processes. A single failure can cascade through the system, threatening crew survival. To address this, research is focusing on supervisory Model Predictive Control (MPC) [40].
MPC is an advanced control strategy that uses a dynamic mathematical model of the entire life support system (e.g., all MELiSSA compartments) to predict future system states. It then computes optimal control actions to keep the system within safe operating bounds while minimizing resource consumption and energy use. For example, an MPC controller can anticipate increased CO₂ levels from crew activity and proactively adjust the photon flux to cyanobacteria PBRs to increase photosynthetic rate, thereby maintaining atmospheric balance. This approach is crucial for managing the inherent instability of biological systems and is a key differentiator from the more static control paradigms of traditional PC systems [40].
The choice between stored rations and in-situ cultivation is not merely a choice of a food source but a fundamental decision regarding the architecture and long-term sustainability of human presence in space. Stored rations, coupled with PC systems, offer high reliability and technological maturity, making them the only option for near-term missions. However, they impose a significant and linear mass penalty and lack true closure of material loops.
In-situ cultivation, as the core of a MELiSSA-inspired BLSS, presents a transformative alternative. It promises a regenerative, mass-efficient environment capable of producing fresh food, revitalizing the atmosphere, and recycling waste. While significant challenges in system control, stability, and scalability remain, the ongoing research—from the Beijing Lunar Palace to the ESA's MELiSSA project—demonstrates a clear path forward. For endurance-class missions beyond low-Earth orbit, the development of a robust, cybernetically controlled BLSS is not just an optimization strategy; it is an imperative for sustainable human exploration.
The quest for sustained human presence in deep space and on other celestial bodies hinges on the development of robust Environmental Control and Life Support Systems (ECLSS). These systems are responsible for providing astronauts with breathable air, potable water, and nutritious food, while managing waste in a closed-loop environment. Current research is framed by a pivotal comparison: the established, highly automated Physicochemical Life Support Systems (PCLSS) against the emerging, complex approach of Bioregenerative Life Support Systems (BLSS). The European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) project exemplifies the latter, aiming to create a closed ecosystem inspired by Earth's own biosphere [4] [41] [42].
This guide objectively compares the monitoring and control paradigms of these two approaches. Automated PCLSS relies on precise, engineered hardware for rapid processing. In contrast, dynamic biological process management, as pursued by MELiSSA, leverages living organisms—plants, algae, and microbes—to regenerate resources, offering long-term sustainability at the cost of increased biological complexity [4] [43]. The integration of BLSS with traditional ECLSS is a key facet of plans for long-term habitats on the Moon and Mars, making this comparison critical for future mission planning [4].
Performance Comparison: Key Metrics and Experimental Data
Direct comparison of these systems requires evaluating their performance across critical life support functions. The data below summarizes their respective capabilities based on current research and technology.
Table 1: Performance Comparison of PCLSS and BLSS across Core Life Support Functions
| Life Support Function | Automated PCLSS (e.g., ISS-like) | Dynamic Biological Process Management (BLSS/MELiSSA) |
|---|---|---|
| Oxygen Generation | Electrolysis of water; rapid, predictable output [43]. | Plant/algal photosynthesis; slower, light-dependent, produces food as a co-product [4] [43]. |
| Carbon Dioxide Removal | Adsorption by zeolite beds; highly efficient and controllable [43]. | Fixation by plants/algae during photosynthesis; integrated with food production [43]. |
| Water Recovery | Physical filtration & chemical treatment (e.g., distillation); high efficiency, multi-step process [43]. | Biological filtration & plant transpiration; can utilize waste as fertilizer, but slower [43]. |
| Waste Management | Solid waste compacted & stored; liquid waste processed via Water Recovery System [43]. | Composting & anaerobic digestion; waste is broken down into resources for plant growth [43]. |
| Food Production | Pre-packaged meals with long shelf-life; no in-situ production [43]. | Grown in-situ (hydroponics/aeroponics); renewable but requires significant space, energy, and time [4] [43]. |
| System Response Time | Fast (seconds to hours); inputs/outputs can be turned on/off [4]. | Slow (days to months); biological processes cannot be switched off [4]. |
| Closure & Sustainability | Limited by consumables (filters, chemicals); requires resupply [43]. | Potential for high closure and self-sufficiency over the long term [41]. |
| Key Monitoring Parameters | Pressure, gas concentration, flow rates, electrical conductivity [4]. | Biomass growth rates, gas exchange rates, nutrient solution chemistry, microbial activity [4]. |
Quantitative analysis is essential for system-level evaluation. The Equivalent System Mass (ESM) metric, which considers the total cost of launching and operating a system, is a key tool for comparison. Recent studies with updated LED lighting technologies have improved the ESM estimates for BLSS food production, calculating a potential return on investment (ROI) time for bioregenerative food production compared to shipping all food from Earth [4]. Furthermore, the degree of material closure—the percentage of resources recycled within the system—is a critical measure of sustainability for which CELSS aims to achieve near-total closure [41].
Table 2: Quantitative System-Level Metrics for Life Support Systems
| Performance Metric | Automated PCLSS | Dynamic BLSS/CELSS |
|---|---|---|
| Equivalent System Mass (ESM) for Food Production | Lower for short missions (< ~2-3 years) | Improves for long-duration missions; enhanced by LED lighting [4]. |
| Return on Investment (ROI) Time for Food | Not applicable (no production) | Estimated for various crop types; determines break-even point vs. shipped food [4]. |
| Material Closure Rate | Partial (air, water); no food closure [41]. | Aims for near-total closure of air, water, and food [41]. |
| Energy Demand per kg of Food | N/A | High; primarily for plant-growth lighting [4]. |
| Cascade Structure of Processes | Linear or single-loop recycling [43]. | Complex, multi-loop functional cascades (see Diagram 1) [44]. |
Experimental Protocols: Methodologies for System Validation
Research in this field relies on both ground-based prototypes and computational models to predict system behavior and validate control strategies.
Integrated System Testing Protocol
Objective: To evaluate the stability and closure of a BLSS when integrated with PCLSS components over an extended period [4] [41].
- Chamber Setup: A hermetically sealed ground-based facility (e.g., inspired by BIOS-3, Lunar Palace-1) is established, containing higher plant growth compartments, algae photobioreactors, and waste processing bioreactors [41].
- Biological Component Inoculation: Selected crops (e.g., wheat, potato, lettuce), algae (e.g., Chlorella, Spirulina), and microbial strains (e.g., for nitrification, anaerobic digestion) are introduced into their respective subsystems [41].
- System Closure: The crew (human or robotic) enters the chamber. All material exchanges are monitored. Oxygen and food are supplied by the biological systems; water is recovered from humidity, urine, and wastewater; solid waste is processed by composters or bioreactors.
- Data Collection & Monitoring:
- Atmospheric: Continuous logging of O₂, CO₂, and trace gas concentrations (e.g., ethylene) [4].
- Aqueous: Regular analysis of nutrient solution chemistry (N, P, K) and contaminants in the water recovery loop [4].
- Biological: Periodic measurement of plant biomass growth, algal density, and microbial community composition via DNA sequencing.
- Control Intervention: PCLSS systems (e.g., CO₂ scrubbers, oxygen tanks) are activated based on pre-defined thresholds to maintain atmospheric balance, simulating a hybrid control approach [4].
Dynamic Model Calibration and Validation Protocol
Objective: To create a predictive model of a BLSS for monitoring and control purposes, accounting for its dynamic biological processes [4] [45].
- Problem Formulation: Define the research question, such as predicting O₂ production fluctuations in response to a simulated crew activity cycle [45].
- Model Construction: Use a process-based design methodology.
- Define Entities: Model constituents (e.g., crew, wheat plants, Spirulina algae, nitrifying bacteria).
- Define Processes: Formulate interactions (e.g., photosynthesis, respiration, nitrification) as mathematical equations (e.g., using Michaelis-Menten kinetics) [46] [45].
- State Variables & Parameters: Set dynamic variables (e.g., biomass, O₂ level) and fixed parameters (e.g., maximum photosynthetic rate, microbial growth rate) [46].
- Parameter Estimation: Use optimization algorithms (e.g., least squares minimization, genetic algorithms) to fit model parameters to experimental data collected from the integrated test protocol [46] [45].
- Model Validation & Refinement: Simulate the system under conditions not used for parameter estimation. Compare model predictions (e.g., of CO₂ levels) with new experimental data. Refine the model structure and parameters based on discrepancies [46] [45].
- Sensitivity Analysis: Perform local or global sensitivity analysis (e.g., using Sobol indices) to identify which parameters (e.g., plant growth rate, microbial degradation rate) most strongly influence critical system outputs like oxygen concentration [46].
Visualization: Signaling Pathways and Workflow Diagrams
Understanding the flow of information and control in these complex systems is aided by visual representations. The following diagrams illustrate the core control architectures.
BLSS Functional Cascade Network
The dynamic management of a BLSS involves tracking the sequential activation of biological functions over time, a concept known as a causal process model [44]. This is crucial for understanding phase transitions and system state changes.
BLSS Functional Cascade
Hybrid ECLSS Control Architecture
Future habitats will likely employ a hybrid architecture, integrating the reliability of PCLSS with the sustainability of BLSS. The control system must manage both physicochemical and biological components.
Hybrid ECLSS Control Architecture
The Scientist's Toolkit: Key Research Reagents and Materials
Research and development in BLSS and PCLSS rely on a suite of specialized reagents, materials, and software tools.
Table 3: Essential Research Reagents and Materials for Life Support System Research
| Item | Function/Application | Relevance to System Type |
|---|---|---|
| Zeolite Adsorbents | Porous minerals used for physicochemical adsorption of CO₂ from cabin air [43]. | PCLSS |
| Hydroponic Nutrient Solutions | Pre-mixed aqueous solutions of essential mineral nutrients (N, P, K, Ca, Mg, trace elements) for soilless plant cultivation [41]. | BLSS / CELSS |
| Selected Microbial Consortia | Defined mixtures of bacteria and fungi used in bioreactors to break down solid waste and recover nutrients [41] [43]. | BLSS / CELSS |
| Algal Cultures (e.g., Chlorella) | Unicellular photosynthetic organisms for rapid oxygen production and biomass generation in photobioreactors [41]. | BLSS / CELSS |
| LED Grow Light Systems | Energy-efficient lighting providing specific light spectra (wavelengths) to optimize plant photosynthesis and morphology in closed environments [4]. | BLSS / CELSS |
| Gas Chromatography-Mass Spectrometry (GC-MS) | Analytical instrument for identifying and quantifying trace volatile organic compounds (VOCs) in the cabin atmosphere, crucial for crew health [4]. | Both (especially BLSS) |
| Process-Based Modeling Software (e.g., COPASI) | Software tool for constructing, simulating, and analyzing dynamic models of biochemical networks, enabling predictive control [46] [45]. | Both (especially BLSS) |
| Sensors for pH, Dissolved O₂, EC | In-line probes for continuous monitoring of the root zone environment in hydroponic systems or aquatic bioreactors [4]. | BLSS / CELSS |
Critical Challenges and Pathways to System Integration and Reliability
Biosafety and Microbial Contamination Control in Confined BLSS
The success of long-duration human space exploration hinges on the development of robust Bioregenerative Life Support Systems (BLSS). These systems are designed to sustain human crews by regenerating air, water, and producing food through biological processes, thereby reducing dependence on Earth-based resupply [3]. A central challenge in their operation, particularly within the confined environments of spacecraft or planetary habitats, is ensuring effective biosafety and microbial contamination control. Uncontrolled microbial growth can threaten system functionality by disrupting delicate ecological balances, compromising the health of plant or microbial compartments, and posing direct risks to crew health [3] [47].
This guide objectively compares the approaches to microbial contamination control between two foundational frameworks: the MELiSSA (Micro-Ecological Life Support System Alternative) project, a leading BLSS concept, and Traditional Physicochemical Life Support Systems. The performance, operational protocols, and safety implications of each system are evaluated based on existing ground-based demonstrators and research, providing a critical resource for researchers and scientists in the field [3].
MELiSSA (Micro-Ecological Life Support System Alternative)
MELiSSA is an advanced BLSS concept that mimics a terrestrial ecosystem. Its goal is to achieve a high degree of resource loop closure by interconnecting biological compartments (photoautotrophic bacteria, higher plants, and microbial communities) with the crew (consumers) [3]. The system is designed to recycle organic waste and carbon dioxide into oxygen, water, and food.
- Contamination Control Philosophy: The MELiSSA loop relies on a controlled, compartmentalized ecology. Each compartment is designed to host specific, defined microbial consortia or plant species that perform dedicated functions, such as nitrification or carbon fixation [3]. The primary contamination control strategy is prevention through system design, which includes isolating compartments and selecting for desired organisms through controlled operational parameters (e.g., pH, temperature, substrate). The closed-loop nature means that a contamination event could potentially propagate through multiple compartments, making monitoring and control paramount [3].
Traditional Physicochemical Life Support Systems
Traditional Physicochemical Systems, such as those used on the International Space Station, rely on mechanical and chemical processes to regenerate resources. These include catalysts, filters, and processors for air revitalization (e.g., removing CO2, generating O2) and water recovery (e.g., recycling humidity and urine) [3].
- Contamination Control Philosophy: The core philosophy is direct physical removal or chemical inactivation of microbial contaminants. These systems employ technologies like HEPA filtration, UV irradiation, chemical biocides, and thermal sterilization (e.g., autoclaves) to eliminate microorganisms from air and water streams [48] [49]. The approach is inherently robust against biological disruption but does not contribute to food production and can be resource-intensive in terms of mass, power, and the need for spare parts or consumable chemicals.
Comparative Performance Analysis
The following tables summarize the key performance characteristics and biosafety considerations of the two systems based on current research and ground-based testing.
Table 1: System Performance and Contamination Control Comparison
| Performance Metric | MELiSSA (BLSS) | Traditional Physicochemical |
|---|---|---|
| Core Contamination Control Principle | Ecological management & selective pressure [3] | Physical/Chemical eradication [48] [49] |
| Food Production Capability | Direct production of fresh food [3] | None |
| Resilience to Microbial Disruption | Potentially vulnerable to ecological imbalance [3] | Highly resilient; components can be sterilized |
| Waste Processing Method | Biological degradation (mineralization) [3] | Physicochemical processing (oxidation, distillation) |
| Primary Biosafety Risk | Systemic collapse due to pathogen or pest outbreak [3] | Mechanical failure leading to exposure or ineffective treatment |
| Typical Crew Role in Maintenance | Active gardening and system monitoring [3] | Mechanical repair and component replacement |
Table 2: Operational and Safety Protocol Requirements
| Operational Aspect | MELiSSA (BLSS) | Traditional Physicochemical |
|---|---|---|
| Required Monitoring | Microbial community sequencing, plant health diagnostics, water/air chemistry [3] | Pressure differentials, flow rates, biocide levels, microbial counts in water [49] |
| Key Safety Equipment | Sealed compartmentalized reactors, biological safety cabinets for sample handling [3] | HEPA filters, UV lamps, chemical injectors, incinerators, autoclaves [48] [49] |
| Crew Training Focus | Microbiology, horticulture, ecological system management [3] | Mechanical engineering, fluid systems, hazardous chemical handling |
| Decontamination Protocol | System-specific biocides, compartment isolation and sterilization [3] | Standardized chemical decontamination, component replacement and sterilization (autoclaving) [48] [50] |
| Inherent Biosafety Level (BSL) Analogy | BSL-2 for crew-facing compartments (moderate hazard) [51] | BSL-1 to BSL-2, depending on waste stream handling [51] |
Experimental Protocols for Contamination Control
MELiSSA: Protocol for Simulating a Microbial Contamination Event
This protocol is designed to test the resilience of a MELiSSA-like compartment to an introduced contaminant and to validate decontamination procedures.
- System Baseline Establishment: Operate the target compartment (e.g., a bioreactor) at a steady state, monitoring key parameters like pH, dissolved oxygen, and microbial density (via optical density) for a defined period to establish a normal operational baseline [3].
- Contaminant Introduction: Introduce a known quantity of a non-pathogenic but easily traceable "challenge" microorganism (e.g., a specific strain of Bacillus subtilis engineered with a fluorescent marker) into the compartment feed line.
- Impact Monitoring: Intensively monitor the compartment for 72-96 hours.
- Culture-Based Methods: Plate samples on selective media to enumerate the challenge organism.
- Molecular Methods: Extract DNA and use quantitative PCR (qPCR) with primers specific to the challenge organism to track its population dynamics [3].
- Functional Assessment: Measure the compartment's key performance metrics (e.g., nitrification rate, carbon conversion efficiency) to assess functional impact [3].
- Decontamination & Recovery: If the contaminant persists and impacts function, initiate a decontamination protocol. This may involve:
- Circulating a system-compatible biocide (e.g., low-concentration hydrogen peroxide) through the compartment for a set contact time.
- Flushing the system with sterile medium.
- Re-inoculating with the original, defined microbial culture from a protected master stock [3].
- Data Analysis: Compare the time taken for the compartment's function to return to the pre-established baseline. The success of decontamination is confirmed by the absence of the challenge organism in post-recovery molecular assays.
Traditional System: Protocol for Validating Sterilization Efficacy
This protocol tests the effectiveness of a physicochemical system's microbial elimination components, such as a waste water processor's sterilization unit.
- Test Solution Preparation: Create a challenge solution containing a high concentration (e.g., 10^8 CFU/mL) of a standardized microbial surrogate. A common surrogate is Geobacillus stearothermophilus spores for thermal systems or Bacillus atrophaeus for chemical/radiation systems, due to their high resistance.
- System Challenge: Introduce the test solution into the inlet of the sterilization unit (e.g., UV reactor, catalytic oxidizer, or waste line leading to an autoclave) at the system's normal operational flow rate.
- Sample Collection: Collect effluent (output) samples from the unit at regular intervals during the test run.
- Viability Assay: Filter the effluent samples through a membrane filter and place the filter on nutrient agar. Incubate the plates at the surrogate's optimal growth temperature for 48 hours.
- Data Analysis: Count the number of colony-forming units (CFUs) on the plates. The sterilization efficacy is calculated using the log reduction value (LRV): LRV = log10 (Influent CFU/mL / Effluent CFU/mL). Regulatory standards often require a minimum 6-log (99.9999%) reduction for potable water systems [49].
System Workflow and Safety Logic
The diagrams below illustrate the fundamental workflows and safety decision trees for contamination control in each system.
MELiSSA Loop Compartmentalized Workflow
Diagram 1: MELiSSA Simplified Workflow
Contamination Response Decision Logic
Diagram 2: Contamination Response Logic
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Materials for BLSS Contamination Control Research
| Item | Function in Research |
|---|---|
| Selective Culture Media | To isolate and enumerate specific microbial populations from a complex consortium, helping to detect contaminants [3]. |
| DNA Extraction Kits & qPCR Reagents | For molecular quantification of total and specific microbial biomass, allowing for tracking of community dynamics and pathogen detection without culturing [3]. |
| Fluorescent Viability Stains (e.g., PMA) | To distinguish between live and dead cells in a sample, critical for validating decontamination efficacy [3]. |
| HEPA Filters & Laminar Flow Cabinets | To provide a sterile workspace for handling plant samples or microbial inoculants, preventing external contamination during experiments [48] [51]. |
| Autoclave | To sterilize growth media, surgical tools, and other equipment, ensuring aseptic techniques in ground-based BLSS research [48] [49]. |
| Biosafety Cabinets (Class II) | To provide personnel, product, and environmental protection when handling potentially hazardous biological samples from BLSS compartments [51] [49]. |
| Defined Microbial Stock Cultures | Master stocks of the specific strains used to inoculate MELiSSA compartments, ensuring system consistency and allowing for recovery after a contamination event [3]. |
Managing System Dynamics and Response Time in Biological Systems
The pursuit of long-duration human space exploration necessitates the development of advanced Life Support Systems (LSS) capable of reliably recycling vital resources. The management of system dynamics and response time presents a fundamental engineering challenge in this domain. Two paradigmatic approaches have emerged: bioregenerative systems, which utilize biological organisms to create ecological cycles, and physicochemical (P/C) systems, which rely on engineered processes. This guide provides a systematic comparison of the European Space Agency's Micro-Ecological Life Support System Alternative (MELiSSA), a leading bioregenerative system, against traditional P/C LSS, with a focus on dynamic behavior, control strategies, and performance metrics [9] [52].
The core challenge for any closed-loop LSS is intrinsic instability; biological systems are inherently dynamic and subject to complex, non-linear interactions [53]. MELiSSA addresses this through a hierarchical control strategy based on first-principles models of each compartment in its ecosystem. This approach uses mathematical deterministic modeling and simulation as the "brain-level" of the system to ensure robust performance [53] [52]. In contrast, traditional P/C systems are typically less complex dynamically but offer less potential for complete resource regeneration.
MELiSSA: A Bioregenerative Ecosystem
MELiSSA is conceived as an artificial ecosystem of interconnected micro-organisms designed to recover oxygen and edible biomass from astronaut waste. Its operation is based on compartmentalizing the different metabolic processes into a structured chain, transforming organic waste step-by-step into usable products [53] [52]. Due to the inherent instability of complex biological systems and the stringent safety requirements of manned space missions, a sophisticated control architecture is paramount. This control strategy is hierarchical: each compartment has a local control system, while an upper-level controller determines setpoints for each compartment based on their collective states and a global desired functioning point [52].
Traditional Physicochemical (P/C) Systems
Traditional P/C LSS, such as the one designed for the NASA Design Reference Mission, rely on mechanical and chemical processes (e.g., filters, chemical reactors, vapor compression distillation) to revitalize air and recycle water. Food is typically supplied from stored sources rather than produced in-situ. These systems are generally considered more mature and predictable but offer limited closure of the carbon and food loops [9].
Table 1: Fundamental Architectural Comparison
| Feature | MELiSSA (Bioregenerative) | Traditional P/C System |
|---|---|---|
| Core Principle | Artificial ecosystem of microorganisms [53] [52] | Engineered mechanical & chemical processes [9] |
| Food Production | In-situ via photoautotrophic compartments [53] | Not applicable (relies on stored food) [9] |
| Waste Processing | Biological degradation and conversion [53] [52] | Physicochemical oxidation (e.g., incineration) [9] |
| Control Approach | Hierarchical, non-linear predictive model based [52] | Typically, conventional process control |
| System Dynamics | High, non-linear, multi-scale [53] | Lower, more linear and predictable [9] |
Quantitative Performance Comparison
The Equivalent System Mass (ESM) metric is a standard methodology in the field for comparing different LSS technologies. ESM transforms all required system parameters, including non-mass parameters like volume and power demand, into an equivalent mass, allowing for a holistic comparison [9].
A dedicated trade study scaled both bioregenerative and P/C systems for a 780-day Mars mission supporting a crew of six. The analysis revealed a significant performance trade-off: the MELiSSA-based bioregenerative system had a calculated ESM of 18,088 kg, which was about four times higher than that of a pure P/C LSS, designed in an earlier study, which had an ESM of 4,830 kg [9]. This difference underscores the substantial mass, volume, and power overheads associated with maintaining a stable biological ecosystem compared to operating more compact, though less regenerative, engineered systems.
Table 2: Equivalent System Mass (ESM) Breakdown for a Mars Mission
| System Component | MELiSSA-based Hybrid LSS | Traditional P/C LSS |
|---|---|---|
| Total ESM | 18,088 kg [9] | 4,830 kg [9] |
| Food Supply | Higher ESM (Bioreactor, Algae, Higher Plant Compartments) [9] | Lower ESM (Stored food only) [9] |
| Solid Waste Processing | Higher ESM (Biological processing) [9] | Lower ESM (Incineration, physico-chemical) [9] |
| Atmosphere Revitalization | Integrated with food supply (Photoautotrophic compartments) [53] | Separate, dedicated subsystems [9] |
| System Redundancy | Not fully included in base ESM calculation [9] | Double redundancy included [9] |
Methodologies for Modeling and Control
The management of system dynamics and response times relies heavily on advanced computational modeling. The approaches differ significantly between the complex biology of MELiSSA and the more straightforward engineering of P/C systems.
MELiSSA's Mechanistic Dynamic Modeling
MELiSSA's control strategy is fundamentally based on mechanistic dynamic models. These are mathematical representations based on first principles (e.g., physico-chemical equations, stoichiometries, kinetic rates) that explain the mechanisms by which a phenomenon takes place [54] [52]. For MELiSSA, this involves:
- Intensive characterization of all processes at the level of main chemical elements (C, H, N, O, S, P...) [53].
- Developing mechanistic models for each compartment, often formulated as ordinary differential equations (ODEs), to simulate the time course of the system [54].
- Implementing a non-linear predictive model-based control strategy, which uses these models to predict future system behavior and optimize control actions [52].
A key application has been the validation of the ARTEMISS flight experiment, which successfully demonstrated the accurate prediction of CO₂ to oxygen conversion on board the International Space Station [53].
Modeling Approaches for Traditional P/C Systems
Modeling for P/C systems typically involves more conventional engineering models, such as:
- Mass flow models and energy balance equations to track the movement and transformation of resources [9].
- Process control models that are often linear or simpler in structure, focusing on maintaining setpoints for temperature, pressure, and concentration.
The following diagram illustrates the contrasting control and modeling architectures for the two systems.
Diagram 1: Control architectures for MELiSSA (yellow) and P/C (blue) systems.
Experimental Protocols for System Validation
Ground Demonstration of MELiSSA Loops
The validation of MELiSSA's dynamic control involves progressive integration within the MELiSSA Pilot Plant. The experimental protocol typically follows these stages [53]:
- Start-up with High-Dynamics Processes: The demonstration begins with gas recycling (CO₂ to Oxygen), which is the most dynamic process.
- Progressive Integration: MELiSSA processes are integrated upwards in a continuous mode over extended periods (months).
- Data Collection and Model Validation: Key metrics like controlled oxygen concentration in the connected habitats are logged and compared against model predictions. For example, the control system has successfully maintained oxygen concentration in a rat habitat at different set points (e.g., 21, 19, 20, 21%) [53].
Integration Site (IS) Analysis for Safety Assessment
A critical experimental protocol for the safety of bioregenerative systems using viral vectors (e.g., for genetic modification of organisms) is Integration Site (IS) analysis. The MELISSA (ModELing IS for Safety Analysis) statistical framework is a regression-based method developed for this purpose [55].
- Objective: To assess the risk of insertional mutagenesis by estimating gene-specific integration rates and their impact on clone fitness.
- Inputs: IS tables (in .bed file format) with clone size estimates, a design matrix with sample-specific covariates, and genome annotation files.
- Methodology: It employs two complementary statistical models:
- IS targeting rate analysis: Uses logistic regression to estimate if specific genomic regions are preferentially targeted by IS events.
- Clone fitness analysis: Evaluates whether an IS within a gene affects the expansion dynamics of clones over time, using logistic regression for binomial count data.
- Validation: The framework's performance is evaluated through comprehensive Monte Carlo simulation studies to assess Positive Predictive Value (PPV) and detection rates [55].
The workflow for this safety analysis is detailed below.
Diagram 2: Workflow for the MELISSA safety analysis framework.
The Scientist's Toolkit: Key Research Reagents and Materials
The development and analysis of advanced biological systems like MELiSSA rely on a suite of specialized computational and biological tools.
Table 3: Essential Research Reagents and Tools
| Tool/Reagent | Function/Application | Relevance to System Dynamics |
|---|---|---|
| Mechanistic Dynamic Models (ODEs) | Describe system dynamics using differential equations based on biochemical first principles [54]. | Core to MELiSSA's predictive control strategy; simulates time-course behavior of compartments [52]. |
| Systems Biology Markup Language (SBML) | A standard format for storing and exchanging computational models of biological processes [56]. | Ensures model interoperability and reuse, critical for collaborative projects like MELiSSA. |
| MELISSA R Package | A specialized statistical framework for analyzing Integration Site (IS) data from gene therapies [55]. | Quantifies safety risks (insertional mutagenesis) by modeling gene targeting rates and clonal fitness. |
| PhiSystem Modeling Tool | A tool-based methodology for design and evaluation of complex systems, derived from MELiSSA systemic approach [53]. | Used for control design in terrestrial applications (e.g., vehicle energy systems) and circular resource management. |
| Photobioreactors | Controlled environment systems for cultivating phototrophic microorganisms (e.g., cyanobacteria, algae) [52]. | Key compartment in MELiSSA for oxygen production and biomass generation; tightly coupled with light transfer models. |
| Stoichiometric Models | Define the quantitative relationships between reactants and products in metabolic networks. | Used for intensive characterization of MELiSSA processes at the level of chemical elements (C, H, N, O, etc.) [53]. |
The choice between a bioregenerative system like MELiSSA and a traditional P/C LSS involves a fundamental trade-off between system closure and equivalent system mass. MELiSSA offers the potential for greater sustainability and in-situ resource production but at the cost of significantly higher mass/volume overheads and vastly more complex, non-linear dynamics requiring advanced model-predictive control [9]. The maturity and predictability of P/C systems make them a lower-risk choice for nearer-term missions, while the continued development of MELiSSA's biological and control components is crucial for long-term human presence beyond Earth. Future advancements in mechanistic modeling, uncertainty quantification, and the integration of machine learning with first-principles models will be key to managing the dynamics and improving the response times of these complex biological systems [54] [57].
The pursuit of long-duration human space exploration beyond Low Earth Orbit, particularly to the Moon and Mars, is fundamentally constrained by the logistics of supplying essential life support resources. The current physico-chemical (PC) life support systems used aboard the International Space Station (ISS), while reliable, are limited in their ability to close the loop on key resources like food and waste [58]. For missions where resupply from Earth is impractical, a higher degree of system closure and resource regeneration is required. This has catalyzed the development of Bioregenerative Life Support Systems (BLSS), which leverage biological processes to regenerate air, water, and food from crew waste.
Among the most advanced BLSS initiatives is the European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) project. Active for over 30 years, MELiSSA aims to create a closed-loop, self-sustaining ecosystem with near-100% efficiency [8] [59]. This guide provides a objective comparison between the emerging MELiSSA paradigm and traditional PC systems, focusing on the critical mass and energy balance trade-offs that dictate their operational efficiency and suitability for future deep-space missions.
Performance Comparison: MELiSSA vs. Traditional Physico-Chemical Systems
The core difference between these systems lies in their approach to resource recovery. PC systems primarily rely on mechanical and chemical engineering processes, whereas MELiSSA integrates biological components to create a synergistic, bio-regenerative loop. The following tables summarize the key performance metrics and characteristics of each system.
Table 1: Quantitative Performance Metrics for Life Support Systems
| Performance Parameter | Traditional Physico-Chemical (ISS ECLSS) | MELiSSA (BLSS) |
|---|---|---|
| Water Recovery Rate | ~85% (via UPA & PWPA assemblies) [58] | Target: Near 100% [8] |
| Oxygen Production | From water electrolysis; <50% of input CO2 converted via Sabatier system [58] | From photosynthesis (microalgae, plants) [58] |
| Food Production | None; entirely reliant on resupply | Target: Significant portion from microalgae (e.g., Spirulina, Chlorella) and higher plants [59] [58] |
| Waste Processing | Fecal and food waste incinerated on resupply vehicle re-entry [58] | Organic waste recycled by microbial bioreactors as nutrient source [8] [58] |
| Edible Biomass Productivity | Not Applicable | Microalgae: High (e.g., Spirulina: 3000 dry tons/year terrestrially) [58] |
| Key Inputs Requiring Resupply | Food, some water, spare parts | Initial resources; system designed for minimal resupply |
Table 2: System Characteristics and Qualitative Trade-offs
| Characteristic | Traditional Physico-Chemical | MELiSSA |
|---|---|---|
| Core Philosophy | Engineering-driven, physico-chemical processes | Bio-engineering driven, ecological simulation |
| System Architecture | Largely linear (with some recycling) | Circular, compartmentalized loop [8] |
| Primary Energy Consumer | Electrical systems for pumps, filters, electrolysis, etc. | Lighting for photosynthetic components [58] |
| Mass Penalty (Upfront vs. Long-term) | Lower upfront mass, but continuous resupply mass | High upfront mass for infrastructure, lower long-term resupply mass |
| Technology Readiness Level (TRL) | High (flight-proven on ISS) | Medium (ground demonstration, e.g., Pilot Plant in Barcelona) [8] |
| Resilience & Stability | Well-understood, high mechanical reliability | Complex, requires sophisticated control of biological dynamics [60] |
| By-products / Losses | CH4 vented from Sabatier system [58] | Aims to minimize losses through continuous recycling |
Experimental Protocols for System Validation
Rigorous ground-based testing is essential to quantify the mass and energy flows of life support systems. The following protocols describe the standard methodologies used for evaluating BLSS and PC components.
Plant Characterization Unit (PCU) Protocol for BLSS
The PCU, developed for the MELiSSA program, is a state-of-the-art ground facility designed to collect high-quality data on plant growth and gas exchange in a tightly controlled environment [59].
- Objective: To quantify oxygen production, CO2 consumption, water transpiration, and biomass yield of plant candidates for BLSS.
- Methodology:
- Chamber Setup: Plants are cultivated in a sealed growth chamber with minimized leakage, precisely controlling temperature, relative humidity, and airflow [59].
- Nutrient Delivery: An advanced system doses acid, base, and up to eight stock nutrient solutions via peristaltic pumps, with precise control over solution composition and pH [59].
- Lighting: LED lights provide specific photoperiods and light intensities (e.g., 200-600 μmol photons m⁻² s⁻¹) to optimize photosynthesis [61].
- Data Acquisition: Integrated sensors continuously monitor O2 and CO2 concentrations, water uptake, and environmental parameters. Biomass is harvested and measured at the end of the growth cycle.
- Application: This protocol has demonstrated the capability of systems to produce oxygen through photosynthesis and recycle water, key for reducing stored consumables on long missions [59].
Physico-Chemical System Performance Protocol
Testing of PC systems, like the ISS's Environmental Control and Life Support System (ECLSS), focuses on the efficiency of individual mechanical processes.
- Objective: To determine the recovery efficiency of water and oxygen from waste streams.
- Methodology:
- Water Recovery: Wastewater (urine, cabin condensate, hygiene water) is processed in a closed-loop test stand. The Urine Processing Assembly (UPA) uses distillation, while the Water Processing Assembly (WPA) uses filtration and catalytic oxidation. Performance is measured by the ratio of purified water output to wastewater input [58].
- Air Revitalization: A simulated cabin atmosphere is maintained. CO2 is removed via adsorption systems (e.g., CDRA). A subset of this CO2 is processed in a Sabatier Reactor with H2 from water electrolysis to produce water and methane. Efficiency is measured by the percentage of CO2 converted and the rate of O2 production from electrolysis [58].
System Architecture and Workflow
The fundamental difference between the two approaches is visualized in the following diagrams, which map the flow of mass and energy.
MELiSSA Circular Ecosystem Architecture
The MELiSSA system is designed as a closed loop of interconnected compartments, each with a specific biological function, mimicking a simplified ecosystem.
Diagram 1: MELiSSA Circular Ecosystem Architecture. This diagram illustrates the flow of matter through the five core compartments of the MELiSSA loop, transforming waste back into vital resources.
Traditional Physico-Chemical Linear Workflow
In contrast, traditional PC systems operate on a more linear principle with limited recycling and significant inputs and waste outputs.
Diagram 2: Traditional Physico-Chemical Linear Workflow. This diagram shows the primarily linear flow of resources in PC-based systems, highlighting dependence on resupply and the venting or disposal of waste products.
The Scientist's Toolkit: Key Research Reagents and Materials
Research and development in BLSS and PC systems rely on a suite of specialized biological and engineering components.
Table 3: Essential Research Reagents and Materials for Life Support System Research
| Reagent / Material | Function / Role in Research | Category |
|---|---|---|
| Spirulina platensis / Chlorella vulgaris | Fast-growing cyanobacterium/microalga; studied for O2 production, CO2 sequestration, and as a protein-rich food source [58]. | Biological Component |
| Aquatic Bryophytes (e.g., Java Moss) | Non-vascular plants investigated for their biofiltration capabilities, including removal of nitrogen compounds and heavy metals from water [61]. | Biological Component |
| Nitrosomonas / Nitrobacter spp. | Nitrifying bacteria used in biofilters to convert toxic ammonia from waste into nitrate, a valuable plant nutrient [58]. | Biological Component |
| Liquid Sorbents | Emerging physico-chemical technology for capturing CO2 and humidity in microgravity; aim for higher efficiency and reliability [62]. | Physico-Chemical Material |
| Controlled Environment Chamber | Sealed growth chamber (e.g., PCU) with precise control of temperature, light, humidity, and gas composition for plant/microorganism studies [59]. | Research Infrastructure |
| Pressure-Sensitive Paint (PSP) | Advanced experimental technique used in wind tunnel testing to optimize the design of spacecraft and re-entry capsules, impacting overall mission efficiency [62]. | Engineering Tool |
| Stoichiometric Models | Mathematical models used to calculate mass balances of elements (C, O, H, N) through the entire system, ensuring closure and identifying bottlenecks [60]. | Modeling & Simulation Tool |
Crop Optimization and Characterization for Space Environments
The renewal of global lunar exploration ambitions has brought the critical challenge of life support for long-duration missions to the forefront. Two fundamentally different technological pathways have emerged: bioregenerative life support systems that utilize biological processes, exemplified by the European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) initiative, and traditional physicochemical systems that rely on mechanical and chemical processes [2] [63]. The core distinction lies in their approach to closure—the degree to which resources are recycled within the system. Bioregenerative systems aim to create an ecosystem where plants, microorganisms, and humans coexist in a balanced cycle, regenerating air, water, and food [15]. Physicochemical systems primarily purify air and water while depending on resupply from Earth for nutrition and other consumables [2].
Current geopolitical dynamics underscore the urgency of this comparison. NASA's historical bioregenerative programs, including the Bioregenerative Planetary Life Support Systems Test Complex (BIO-PLEX), were discontinued after 2004, while China's CNSA has aggressively advanced these technologies, demonstrating a fully-integrated closed-system supporting crew for one year in the Beijing Lunar Palace (Yuegong-1) [2]. This analysis objectively compares the performance of these competing approaches through the lens of crop optimization and characterization, providing researchers with experimental data and methodologies critical for next-generation life support development.
Technology Comparison: MELiSSA versus Physicochemical Systems
Fundamental Architecture and Operating Principles
The MELiSSA project, operational since 1989, is an ambitious European effort to achieve the highest degree of crew autonomy by producing food, water, and oxygen from mission wastes [15]. Its architecture mimics a terrestrial ecosystem, consisting of interconnected compartments where waste is progressively broken down by microorganisms, and the resulting nutrients are used to cultivate higher plants for food and air revitalization [15]. This bioregenerative approach is fundamentally circular and biological, with crop plants serving multiple simultaneous functions: food production, oxygen generation, carbon dioxide consumption, and water purification [22].
In contrast, traditional Environmental Control and Life Support Systems (ECLSS) rely on physicochemical processes [63]. These systems utilize mechanical components like fans, filters, and chemical reactors to maintain habitability. The International Space Station's ECLSS exemplifies this approach, with subsystems for oxygen generation (typically through water electrolysis), carbon dioxide removal (using materials like lithium hydroxide), and water recovery (through filtration and distillation) [63]. Crops, if present, are supplementary rather than integral to core life support functions.
Table 1: Fundamental Characteristics of MELiSSA and Physicochemical Life Support Systems
| Characteristic | MELiSSA (Bioregenerative) | Traditional Physicochemical |
|---|---|---|
| Core Principle | Ecological simulation with biological components [15] | Mechanical and chemical engineering processes [63] |
| Primary Oxygen Source | Plant photosynthesis [22] | Water electrolysis (e.g., OGA on ISS) [63] |
| Carbon Dioxide Management | Plant consumption [22] | Chemical absorption (e.g., LiOH, CDRA) [63] |
| Food Production | Integral; provided by cultivated crops [2] [22] | Not applicable; reliant on resupply or prepackaged food |
| Water Recovery | Biological transpiration & physicochemical treatment [63] [22] | Multistage filtration & distillation (e.g., UPA on ISS) [63] |
| Waste Processing | Microbial digestion & nutrient recycling [22] [15] | Drying, compaction, and storage for disposal [63] |
| System Closure Potential | Theoretical full closure of air, water, and food cycles [2] | Partial closure of air and water only [2] |
Quantitative Performance Metrics
Data from ground demonstrations reveal significant differences in operational capabilities. The Chinese Yuegong-1 (Lunar Palace) facility, a MELiSSA-inspired system, achieved 100% oxygen regeneration and 100% water recycling during a 370-day crewed experiment [63]. The crew produced more than half of their food internally, cultivating cereals, vegetables, and strawberries, with inedible plant waste used to raise protein-rich mealworms [63]. This demonstrates the multi-loop functionality of bioregenerative systems.
Physicochemical systems demonstrate high efficiency in specific domains. For example, the HESTIA facility's electrolyzer efficiently produces oxygen, and its Air Revitalization System controls air quality through a condensing heat exchanger, Trace Contaminant Removal System, and Reactive Plastic Lithium Hydroxie unit [63]. However, these systems do not address food production, creating a fundamental limit to mission duration without resupply.
Table 2: Documented Performance Metrics from Testbed Facilities
| Performance Metric | MELiSSA-Type Systems (Yuegong-1) | Physicochemical Systems (HESTIA/ISS-type) |
|---|---|---|
| Atmospheric Closure (O₂) | 100% regeneration achieved [63] | High-efficiency electrolysis from stored water [63] |
| Water Recovery Rate | 100% recycling demonstrated [63] | ~93-95% recovery typical (ISS data) [63] |
| Food Self-Sufficiency | >50% of crew diet produced internally [63] | 0% (complete reliance on resupply) |
| Waste Valorization | Organic waste processed as fertilizer/insect feed [63] | Solid waste stored; urine vented or processed [63] |
| Mission Duration Support | Theoretical indefinite operation with closure [2] | Limited by food resupply capacity [2] |
| Crew Time Requirement | Higher (for plant cultivation and maintenance) [2] | Lower (primarily system monitoring) [2] |
Crop Characterization and Optimization Methodologies
Experimental Protocols for Plant Characterization in Space Environments
Characterizing plant responses to space conditions is a foundational research activity for bioregenerative systems. The following integrated protocol is synthesized from current MELiSSA research priorities and previous BIOS-3 and Yuegong-1 experiments [63] [22].
Objective: To comprehensively evaluate candidate crop species for performance in closed-loop life support systems under simulated space environmental stressors.
Materials and Reagents:
- Plant Growth Chambers: Equipped with precise environmental control (temperature, humidity, photoperiod, CO₂) [63] [22].
- LED Lighting Systems: Tunable spectra and intensity to optimize photosynthesis and morphology [64] [22].
- Hydroponic or Nutrient Film Technique (NFT) Systems: For precise delivery of water and nutrients [63] [64].
- Gas Chromatography-Mass Spectrometry (GC-MS): For monitoring volatile organic compounds and gas exchange rates (O₂, CO₂) [22].
- Nutrient Solution Analysis Kits: For monitoring pH, EC, and specific macronutrients [63].
- PCR and RNA-Seq Equipment: For genomic and transcriptomic analysis of plant stress responses [22].
Procedure:
- Species Selection: Preselect candidate species based on high yield, nutritional value, and low resource requirements (e.g., wheat, potato, lettuce, soy) [63] [22].
- Environmental Stress Application: Expose plants to defined space-relevant stressors in controlled chambers:
- Radiation: Apply controlled doses of ionizing radiation (e.g., gamma-rays, heavy ions) simulating space radiation spectra [22].
- Altered Gravity: Use clinostats to simulate reduced gravity or conduct experiments on parabolic flights or the ISS [22].
- Atmospheric Composition: Cultivate plants under reduced pressure and elevated oxygen scenarios, as planned for habitats [63] [22].
- Phenotypic Data Collection: Monitor and record:
- Nutritional and Biochemical Analysis: Post-harvest, analyze:
- Macronutrients: Protein, carbohydrate, and lipid content.
- Micronutrients: Vitamin and mineral content.
- Anti-nutrients: Compounds that may interfere with nutrient absorption.
- Molecular Characterization: Perform transcriptomic (RNA-Seq) and microbiome analysis to understand genetic and microbial responses to space stressors [22].
The Scientist's Toolkit: Key Research Reagents and Solutions
Table 3: Essential Research Materials for Space Crop Optimization
| Item/Category | Function in Research | Example Application |
|---|---|---|
| Tunable LED Arrays | Provide specific light wavelengths (red, blue, far-red) to control plant growth, photosynthesis, and morphology [64]. | Optimizing light recipes for lettuce and wheat in Veggie-like systems [64]. |
| Hydroponic Nutrient Solutions | Deliver precise mineral nutrition (N, P, K, Ca, Mg, micronutrients) directly to plant roots in soliless culture [63] [64]. | Formulating solutions for specific crops in closed-loop water systems [63]. |
| Ionizing Radiation Sources | Simulate the space radiation environment to study DNA damage, oxidative stress, and mutagenic effects in plants [22]. | Screening for radiation-tolerant crop cultivars. |
| Controlled Environment Chambers | Isolate and manipulate environmental variables (CO₂, temperature, humidity, pressure) to simulate habitat conditions [63] [22]. | Testing plant growth under reduced pressure for lunar surface habitats. |
| GC-MS & HPLC Systems | Identify and quantify volatile organic compounds, hormones, and nutritional components in plant tissues and the atmosphere [22]. | Monitoring air quality in closed systems and analyzing food nutritional content. |
| DNA/RNA Extraction Kits | Isolate high-quality nucleic acids for genomic, transcriptomic, and microbiome analysis [22]. | Studying gene expression changes in plants exposed to microgravity or radiation. |
Visualization of System Architectures and Workflows
MELiSSA's Hierarchical Control Structure for Crop Management
The operation of a complex bioregenerative system requires sophisticated control. The following diagram illustrates a hierarchical control architecture, as proposed in recent research, for managing oxygen concentration—a key output of crop photosynthesis—in a life support system [65].
This control structure ensures that the biological oxygen production from crops is dynamically matched to crew consumption. The Regulatory Control layer maintains real-time O₂ levels in the crew cabin. The Optimizing Control layer coordinates gas flows between different biological compartments (e.g., crop chambers, algal reactors) to maximize efficiency. The Supervisory Control layer oversees long-term system performance and makes strategic adjustments, such as scheduling crop planting and harvest to ensure a continuous oxygen supply [65].
The MELiSSA Loop Ecosystem Workflow
The MELiSSA concept is based on a continuous loop that breaks down organic waste to support food production. The following diagram maps this ecosystem workflow, which is central to its bioregenerative function [15].
This workflow illustrates the stepwise conversion of waste into resources. The process begins with crew waste, which is broken down by thermophilic (heat-loving) bacteria. The resulting compounds feed other specialized microorganisms that further process the material into nitrates, which serve as fertilizer for the higher plant chamber [15]. The crops, in turn, produce food and oxygen for the crew, thereby closing the loop.
The comparative analysis reveals a clear trade-off: physicochemical systems offer proven reliability and lower operational complexity for near-term missions, while MELiSSA-type bioregenerative systems present a path to superior long-term sustainability and self-sufficiency [2] [63]. The experimental data from Yuegong-1 confirm that near-complete closure of air and water loops is achievable, with food self-sufficiency being the ongoing challenge [63].
For researchers and drug development professionals, the methodologies and controls outlined provide a framework for advancing crop optimization. The immediate research priorities should focus on radiation-tolerant crop cultivars, automated cultivation systems to reduce crew time, and genomic studies to understand plant adaptation to space environments [22]. The future of human presence in deep space hinges on the successful integration of the biological robustness of MELiSSA with the engineering reliability of physicochemical systems, creating hybrid life support architectures that are greater than the sum of their parts.
The Role of AI and Digital Twins in Predictive System Management
The integration of Artificial Intelligence (AI) and Digital Twins represents a transformative approach to predictive system management across diverse fields, from healthcare to environmental science. These technologies enable a fundamental shift from reactive to proactive management through dynamic, data-driven simulations and forecasts. AI algorithms excel at identifying complex patterns within vast datasets, while digital twins serve as virtual replicas of physical entities, updating and evolving in real-time to mirror changes in their real-world counterparts. This powerful combination facilitates unprecedented capabilities in forecasting system behaviors, predicting failures, and optimizing performance, thereby enhancing reliability, safety, and efficiency in critical systems [66] [67].
The core advantage of this paradigm lies in its capacity for real-time monitoring and predictive analytics. Unlike traditional static models or scheduled maintenance protocols, AI-driven digital twins create a closed-loop feedback system between the virtual and physical worlds. This enables continuous system assessment and the early detection of anomalies that may signal impending failures or performance degradation. By leveraging historical data, real-time sensor inputs, and advanced simulation capabilities, these technologies provide decision-support tools that can anticipate problems before they manifest, ultimately reducing downtime, extending equipment lifespan, and improving overall system resilience [68] [67].
Technological Foundations: AI and Digital Twins Explained
What are Digital Twins?
Digital twins are dynamic, real-time virtual simulations of physical entities, processes, or systems. They transcend simple digital models by creating an active, bidirectional link with their physical counterparts, continuously updating based on real-time data feeds. This enables them not only to mirror the current state of a system but also to simulate future scenarios and outcomes. In healthcare, for instance, digital twins can replicate individual patient physiology, hospital equipment, or even entire emergency care ecosystems, allowing for sophisticated modeling and prediction [66] [67].
The architecture of a digital twin typically integrates multiple data sources, including historical records, real-time sensor data (from Internet of Things (IoT) devices and wearables), and environmental factors. Advanced analytics and machine learning algorithms process this information to create a living model that evolves alongside the physical entity. This allows for a comprehensive view of the system's health, performance, and potential failure points, providing a powerful platform for testing interventions and predicting future states without risking the actual system [66].
The Role of AI in Powering Predictive Capabilities
Artificial Intelligence, particularly machine learning (ML), serves as the analytical engine of predictive systems. AI algorithms excel at finding complex, non-linear patterns within multidimensional datasets that are often imperceptible to human analysts or traditional statistical methods. In the context of predictive management, AI models are trained on historical performance data and real-time inputs to identify early indicators of system stress, component wear, or process inefficiency [68] [69].
Various AI methodologies are employed in these systems. Ensemble methods like Random Forest have demonstrated 99.6% accuracy in predicting defibrillator performance, while Support Vector Machines (SVM) have achieved 96.1% accuracy in fault prediction for critical medical equipment [68]. For forecasting complex dynamic systems like hurricanes, AI models can process decades of historical data—45 years of storm data in DeepMind's case—to predict intensity changes with significantly higher accuracy than traditional models [69]. These capabilities make AI indispensable for transforming raw data into actionable predictive insights within digital twin frameworks.
Performance Comparison: AI-Driven Approaches vs. Traditional Methods
Predictive Accuracy and Operational Efficiency
The performance advantages of AI-enhanced predictive systems are evident across multiple domains. The following table summarizes key comparative findings from recent implementations:
Table 1: Performance Comparison of AI-Driven vs. Traditional Predictive Methods
| Application Domain | AI/Digital Twin Approach | Traditional Approach | Key Performance Metrics | Result Summary |
|---|---|---|---|---|
| Medical Equipment Maintenance [68] | AI-based Predictive Maintenance (PdM) using ensemble classifiers (Random Forest, SVM) | Preventive maintenance schedules & reactive repairs | Accuracy: Up to 99.6%Cost Savings: ~25%Downtime: Significant reduction | AI models predict failures proactively, optimizing maintenance resources and reducing unexpected equipment failures. |
| Hurricane Intensity Forecasting [69] | DeepMind AI model trained on 45 years of cyclone data | Traditional numerical simulation models (ECMWF, NOAA HAFS-A) | Prediction Window: 5-day intensity forecastError Rate: Significantly lower than traditional models | AI model outperformed traditional systems in predicting rapid intensification, generating forecasts in minutes versus hours. |
| ICU Performance Monitoring [70] | Risk-adjusted statistical process control charts (p-charts) | Standardized Mortality Ratio (SMR) monitoring | Sensitivity for 50% Mortality Increase: 88% for p-charts vs. 59% for SMR | P-charts demonstrated superior sensitivity in detecting simulated increases in mortality, offering simpler implementation than CUSUM. |
| Healthcare Resource Allocation [67] | Digital twin simulations of patient flow and resource needs | Simplified decision-making models & historical patterns | Response Time: Reduced through optimized allocationResource Use: Improved efficiency | Digital twins incorporate real-time data (traffic, population density) for nuanced and effective resource deployment. |
Implementation and Practical Considerations
Beyond raw performance metrics, the implementation of AI and digital twin systems presents distinct advantages and challenges compared to traditional methods. AI-driven predictive maintenance in healthcare, for instance, requires significant upfront investment in sensor infrastructure, data integration platforms, and specialized expertise. However, this initial cost is frequently offset by long-term benefits, including lower maintenance costs, improved equipment visibility, and enhanced patient safety through reduced device downtime [68] [71].
A critical challenge identified in healthcare settings is the potential for a "digital divide." A 2023 study of U.S. hospitals found that while approximately 65% use AI-assisted predictive tools, fewer than half evaluated these tools for potential bias. Well-resourced hospitals often develop and rigorously evaluate their own models, while under-resourced hospitals tend to purchase "off-the-shelf" solutions that may not reflect their local patient population, potentially exacerbating health disparities [72]. This highlights the importance of not only technical performance but also equitable implementation and ongoing bias assessment in predictive systems.
Experimental Protocols and Methodologies
Protocol for AI-Based Predictive Maintenance in Healthcare
Implementing an AI-driven predictive maintenance framework for medical equipment involves a structured, multi-stage process. The following workflow outlines the key phases from data collection to actionable insights:
Diagram 1: Predictive Maintenance Workflow
The methodology begins with comprehensive data collection from equipment sensors, historical maintenance records, and usage logs [68]. This data is fused and preprocessed to handle missing values and normalize signals. Subsequently, feature engineering identifies critical parameters indicative of impending failure, such as vibration patterns, temperature deviations, or operational inconsistencies [68] [71].
The core analytical phase involves model training using algorithms like Random Forest (RF) and Support Vector Machines (SVM), which have proven effective in healthcare contexts [68]. These models are trained to recognize patterns preceding equipment failures. The final stage generates actionable outputs, sending maintenance alerts to technicians, scheduling resources, and notifying stakeholders, thereby enabling proactive interventions before critical failures occur [68] [67].
Protocol for Digital Twin-Enabled Emergency Care Optimization
Digital twins create a virtual replica of emergency care systems to optimize processes from pre-hospital care to recovery. The implementation involves:
Table 2: Research Reagent Solutions for Digital Twin Systems
| Component | Function | Implementation Example |
|---|---|---|
| Wearable Biosensors | Continuously monitors patient vital signs (heart rate, blood pressure, oxygen saturation) in real-time. | Creates dynamic health profiles for risk stratification in pre-hospital care [67]. |
| IoT Devices & Medical Equipment Sensors | Tracks equipment status, usage patterns, and performance metrics for Medical Device Twins (MDTs). | Enables predictive maintenance of ventilators and defibrillators; forecasts equipment demand surges [67]. |
| Urban Data Layers | Integrates historical emergency call patterns, real-time traffic, and population density data. | Simulates emergency scenarios to optimize ambulance deployment and reduce response times [67]. |
| Predictive Analytics Algorithms | Applies machine learning to patient data for disease progression forecasting and relapse prediction. | Generates early warnings for complications during patient recovery, enabling timely intervention [66] [67]. |
Diagram 2: Digital Twin System Architecture
The methodology for creating an emergency care digital twin involves developing a data integration layer that consolidates information from electronic health records (EHRs), wearable devices, environmental sensors, and equipment monitors [66] [67]. This integrated data feeds into an analytics and AI engine where predictive models simulate patient trajectories, disease progression, and system demands. These simulations enable the generation of actionable insights, such as personalized treatment plans, proactive maintenance alerts for equipment, and optimized resource allocation. A critical feature is the closed-loop feedback, where actions taken in the physical world are monitored and fed back into the digital twin, continuously refining its accuracy and predictive capabilities [67].
The evidence demonstrates that AI and digital twins fundamentally enhance predictive system management by offering superior accuracy, efficiency, and proactive capabilities compared to traditional methods. The integration of real-time data with advanced analytics enables a shift from reactive problem-solving to anticipatory management, whether applied to medical equipment maintenance, clinical decision-making, or environmental forecasting. However, successful implementation requires addressing significant challenges, including data quality, system integration, and ethical considerations regarding bias and equity [68] [72] [67].
As these technologies evolve, future developments will likely focus on improving model transparency, facilitating integration across diverse platforms, and establishing robust ethical frameworks for their use. The convergence of AI and digital twin technology represents a paradigm shift in how we manage complex systems, offering the potential to not only predict outcomes but also to actively shape them through data-driven intervention, ultimately creating more resilient, efficient, and responsive systems across healthcare and beyond.
Performance Metrics and Terrestrial Applications: Validating the Future
For long-duration space missions beyond Earth's orbit, such as established bases on the Moon or Mars, achieving crew autonomy is paramount. Life Support Systems (LSS) must reliably provide astronauts with essential necessities—food, water, and breathable air—while effectively managing waste. Two primary technological pathways have emerged: the well-established Physicochemical (PC) systems and the biologically-driven Bioregenerative Life Support Systems (BLSS). The European Space Agency's MELiSSA (Micro-Ecological Life Support System Alternative) project exemplifies a sophisticated BLSS approach, aiming to create a robust, artificial ecosystem for space [73]. This guide provides an objective comparison of these systems based on key performance indicators (KPIs)—Closure Rates, Reliability, and Crew Time—to inform researchers and scientists in the field.
KPI Comparison: MELiSSA vs. Physicochemical Systems
The comparative performance of BLSS, like MELiSSA, and traditional PC systems can be quantified across several KPIs. The data in the table below synthesizes information from experimental platforms, including MELiSSA and Lunar Palace 1 (LP1), a ground-based BLSS prototype in China [74] [75].
Table 1: Key Performance Indicator Comparison for Life Support Systems
| Key Performance Indicator (KPI) | MELiSSA (BLSS) | Traditional Physicochemical Systems | Data Source / Experimental Basis |
|---|---|---|---|
| Air Revitalization Closure | High (Theoretically closed via photosynthesis) | Partial (O2 generated from water electrolysis; CO2 processed separately) | MELiSSA Pilot Plant (MPP) & ARTEMISS flight experiment [75] |
| Water Recovery Closure | High (Theoretically closed via biological processing) | High (Mechanical filtration & chemical processing) | MELiSSA Grey Water Recycling in Concordia Station [75] |
| Food Production Closure | Targeted (Integrated production from plants & microbes) | None (All food is stowed) | MELiSSA objectives; LP1 370-day experiment [73] [74] |
| Waste Processing Closure | High (Organic waste is a resource for the loop) | Low (Waste is stored or partially processed) | MELiSSA compartment functions [75] |
| System Reliability (Mean Lifetime) | 52.4 years (Estimated for a BLSS under normal maintenance) | Data Gaps (Relies on component redundancy) | LP1 370-day experiment & Monte Carlo simulation [74] |
| Crew Time Demand | Potentially High (System monitoring, plant cultivation) | Lower (Maintenance of mechanical systems) | LP1 operational experience; MELiSSA's ALiSSE criteria include "crew time" [74] [73] |
Analysis of Comparative KPIs
- Closure Rates: MELiSSA's core strength is its pursuit of high closure rates across multiple cycles (air, water, waste) by design, creating an integrated, circular economy [73] [75]. In contrast, PC systems excel in water recovery but cannot close the loop on food production and have limited capacity for converting organic waste back into useful resources.
- Reliability: Quantitative estimation for a BLSS, derived from the long-duration LP1 experiment, suggests a potential mean lifetime of over 52 years under normal operation and maintenance [74]. This data provides a crucial benchmark; however, direct, long-term reliability data for full-scale PC systems in comparable closed environments is less publicly available, making a definitive comparison challenging.
- Crew Time: This KPI represents a significant challenge for BLSS. The LP1 experiment identified several units (e.g., temperature and humidity control, water treatment) with higher failure probabilities, requiring maintenance [74]. MELiSSA explicitly includes "crew time" as a critical trade-off factor in its design criteria (ALiSSE), acknowledging the operational burden of managing a complex biological system [73]. PC systems, while still requiring maintenance, generally involve less labor-intensive daily care.
Experimental Protocols and Methodologies
Robust experimental data is vital for validating the performance claims of life support systems. The following protocols detail how critical KPIs are tested.
Protocol for Reliability and Lifetime Estimation (BLSS)
The methodology used in the LP1 study provides a framework for quantifying BLSS reliability [74].
- Objective: To accurately estimate the reliability and mean lifetime of a BLSS based on empirical failure data.
- Experimental Setup: A 370-day closed human experiment was conducted within the Lunar Palace 1 facility, which comprises interconnected units (e.g., water treatment, atmosphere management, plant cabins).
- Data Collection: The number and precise time of failure for each unit in the system were meticulously recorded throughout the experiment.
- Data Analysis:
- Parameter Estimation: The failure rate (λ) for each unit was calculated using maximum likelihood estimation.
- Probability Distribution: A composite failure probability distribution function for the entire system was formulated based on the series and parallel connections of the units.
- Monte Carlo Simulation: Thousands of system lifetimes were simulated by generating pseudo-random numbers that obeyed the overall failure distribution. This allowed for statistical estimation of the mean lifetime and its confidence interval.
- Outcome: The study reported a mean lifetime of 19,112.37 days (~52.4 years) for the BLSS under normal maintenance [74].
Protocol for Integrated Loop Demonstration (MELiSSA)
The MELiSSA Pilot Plant (MPP) employs a structured, multi-phase approach to validate its ecosystem [75].
- Objective: To demonstrate the feasibility, reliability, and efficiency of the integrated MELiSSA loop.
- Experimental Setup: The MPP is a ground-based facility containing distinct compartments (bioreactors) for specific functions: waste degradation, nitrification, air revitalization with micro-algae, and food production with higher plants. A mock crew of rats provides the metabolic inputs.
- Methodology:
- Compartment-Level Development: Each individual bioreactor is first developed and optimized independently under its specific control laws.
- Loop Integration: Once stable, the compartments are physically connected via gas, liquid, and solid streams to close the loop.
- Modeling and Control: The development of accurate mathematical models is a critical component for predicting system behavior and implementing control strategies.
- Synergy with Flight Experiments: Knowledge gained from the MPP informs and supports smaller-scale flight experiments (e.g., ARTEMISS, URINIS), which test specific processes in the space environment [75].
System Architecture and Testing Workflow
The following diagrams illustrate the logical structure of a BLSS and the process for empirically validating its reliability.
MELiSSA Loop Functional Architecture
The MELiSSA system is designed as a closed-loop of interconnected biological and physicochemical processes, mimicking a simplified ecosystem.
BLSS Reliability Estimation Workflow
The reliability of a complex system like a BLSS is estimated through long-term experiments and statistical modeling, as demonstrated by the Lunar Palace 1 study.
The Scientist's Toolkit: Key Research Reagents and Materials
Research and development in BLSS rely on a combination of biological agents and specialized technological components.
Table 2: Essential Research Materials for BLSS Experiments
| Material / Reagent | Function in BLSS Research | Example Use Case |
|---|---|---|
| Arthrospira platensis (Spirulina) | Photosynthetic micro-algae for oxygen production and as a protein-rich food supplement. | MELiSSA's air revitalization compartment [73] [75]. |
| Higher Plants (e.g., wheat, tomato, potato) | Primary food production, water transpiration, and contribution to air revitalization. | Lunar Palace 1 plant cabins; MELiSSA food production [74] [73]. |
| Nitrifying Bacteria Consortia | Convert ammonia from waste streams into nitrates, a key plant nutrient. | MELiSSA nitrification compartment for recycling nitrogen [75]. |
| Waste Degrading Bacteria | Break down solid organic wastes (inedible biomass, human feces) into simpler compounds. | First step in the MELiSSA loop for waste mineralization [75]. |
| LED Light Source Units | Provide specific light wavelengths for optimized photosynthesis in plants and algae. | Lunar Palace 1 identified this as a critical unit [74]. |
| Specialized Water Treatment Units | Recover potable water from humidity, urine, and greywater through filtration and biological processing. | Critical unit in LP1; MELiSSA Grey Water Recycling in Concordia [74] [75]. |
The development of life support systems for long-duration space missions requires extensive ground-based testing to validate concepts, assess performance, and refine designs under controlled conditions that simulate space mission constraints. Ground demonstrators serve as essential testbeds for bridging the gap between theoretical concepts and practical implementation for both bioregenerative and physicochemical systems [25]. These facilities allow researchers to evaluate closed-loop operations, system integration challenges, and long-term reliability without the risks and costs associated with space-based testing. This comparison guide objectively examines the capabilities and experimental approaches of major life support system demonstrators, with particular focus on the European Space Agency's MELiSSA Pilot Plant alongside other significant international testbeds.
The table below provides a systematic comparison of key ground demonstrators for life support systems, highlighting their distinct approaches, technological readiness, and primary research focus areas.
Table 1: Comparison of Major Life Support System Ground Demonstrators
| Demonstrator Name | Lead Organization/Country | System Type | Key Research Focus Areas | Notable Achievements/Duration |
|---|---|---|---|---|
| MELiSSA Pilot Plant (MPP) | European Space Agency (ESA) | Bioregenerative (BLSS) | Air revitalization, water recovery, food production, waste recycling via interconnected biological compartments [22] [3] | Continuous operation with rodent models; focused on loop closure and control strategies [3] |
| BIO-Plex | NASA (Cancelled) | Bioregenerative (BLSS) | Integrated testing of regenerative life support systems for Mars missions [25] | Program developed plans but was discontinued and physically demolished after 2004 [25] |
| Lunar Palace 1 | China National Space Administration (CNSA) | Bioregenerative (BLSS) | Closed-system operations for atmosphere, water, and nutrition recovery [25] | Sustained crew of four analog taikonauts for a full year [25] |
| Lunar-Mars Life Support Test Project (LMLSTP) | NASA | Hybrid (Physicochemical & Biological) | Air revitalization, water recycling, environmental sensing technologies with human crews [76] | 30-day human stay in closed-loop environment; first NASA recycling of water for potable use [76] |
| International Space Station ECLSS | NASA (with international partners) | Physicochemical | Oxygen generation, water recovery, atmospheric management [77] | Operational system providing >96.5% water recovery in actual space environment [77] |
Experimental Protocols and Methodologies
MELiSSA Pilot Plant Compartmentalized Architecture
The MELiSSA Pilot Plant employs a five-compartment bioengineered system designed to mimic a lake ecosystem [77]. Each compartment represents a specific trophic level in the ecological cycle, with the following experimental configuration:
- Compartment I: Waste digestion using thermophilic anoxygenic bacteria (e.g., Hydrogenophilaceae) to break down organic solid waste and convert it to volatile fatty acids, carbon dioxide, and ammonium [77]
- Compartment II: Photoheterotrophic bacteria (e.g., Rhodospirillum rubrum) further break down volatile fatty acids from Compartment I while producing microbial biomass [77]
- Compartment III: Nitrifying bioreactor for nitrogen recovery, converting ammonium to nitrate using a co-culture of Nitrosomonas europaea and Nitrobacter winogradskyi [77]
- Compartment IV: Photoautotrophic organisms (e.g., Arthrospira platensis) for oxygen production, carbon dioxide assimilation, and edible biomass production [78]
- Compartment V: Higher plant cultivation (e.g., lettuce, wheat) for additional food production, air revitalization, and water purification [3]
The experimental protocol for integrated system testing involves continuous monitoring of mass balances (O₂, CO₂, H₂O, nutrients), microbial community stability, and system controllability under varying load conditions [65].
Physicochemical System Testing Protocol (ISS ECLSS)
The International Space Station's Environmental Control and Life Support System (ECLSS) represents the current state-of-the-art in physicochemical life support, with the following experimental validation approach for its subsystems:
Water Recovery System (WRS): Combines Urine Processor Assembly (UPA) and Water Processor Assembly (WPA)
- UPA protocol: Uses vacuum distillation with pretreated urine (stabilized with H₃PO₄ and Cr⁶+ solution) followed by catalytic removal of volatile organics [77]
- WPA protocol: Multi-filtration beds (particulate filter, carbon, ion exchange) followed by catalytic oxidation reactor at 135°C for contaminant destruction [77]
- Performance metric: 75-85% water recovery from urine [77]
Oxygen Generation System (OGS):
Nitrogen Recovery Experimental Comparisons
Nitrogen recycling represents a critical differentiator between bioregenerative and physicochemical systems. The experimental approaches differ significantly:
Table 2: Comparison of Nitrogen Recovery Methodologies
| Parameter | MELiSSA (Biological Approach) | Traditional Physicochemical (ISS) |
|---|---|---|
| Nitrogen Source | Urine (85% of recoverable N) and other liquid wastes [77] | Urine processed for water recovery only |
| Processing Method | Sequential nitrification (NH₄⁺ → NO₂⁻ → NO₃⁻) using immobilized nitrifying bacteria [77] | Acidic stabilization (H₃PO₄) with Cr⁶+ to prevent scaling [77] |
| End Product | Nitrate fertilizer for plant compartments [77] | Waste product (vented or stored) |
| Nitrogen Recovery Efficiency | Target: >90% recovery for food production [77] | Minimal recovery (nitrogen largely lost) |
| Crew Time Requirement | Higher (system monitoring, potential maintenance) | Lower (largely automated) |
System Architecture and Operational Workflows
The following diagram illustrates the comparative architectures and material flows of bioregenerative versus physicochemical life support systems:
Life Support System Architecture Comparison
Quantitative Performance Metrics
The table below summarizes key performance metrics for life support system functions, highlighting the comparative capabilities of different approaches:
Table 3: Performance Metrics for Life Support System Functions
| System Function | MELiSSA (BLSS) | ISS ECLSS (Physicochemical) | Lunar Palace 1 (CNSA BLSS) |
|---|---|---|---|
| Oxygen Production | Biological (Arthrospira, plants): 100% from crew CO₂ [3] | Water electrolysis: 100% of crew needs [77] | Biological: Demonstrated for 4 crew for 1 year [25] |
| Water Recovery Rate | Target: >95% (including transpiration water) [3] | Current: 75-85% from urine, ~96.5% overall system [77] | Demonstrated closed-loop operation [25] |
| Food Production | In development (Arthrospira, higher plants) [3] | None (all food supplied from Earth) | Included crop production [25] |
| Nitrogen Recovery | Target: >90% via nitrification [77] | Minimal (nitrogen lost as waste) | Demonstrated nutrient recycling [25] |
| Crew Time Requirement | Higher (system maintenance) [3] | Lower (highly automated) | Not specified |
| Resupply Mass Requirements | Potentially lower for long missions | 25,287 kg for 4-person/3-year mission [77] | Significantly reduced via closure |
The Scientist's Toolkit: Key Research Reagents and Materials
Table 4: Essential Research Reagents and Materials for Life Support System Experiments
| Reagent/Material | Function/Application | Example Use Cases |
|---|---|---|
| Nitrifying Bacteria (Nitrosomonas europaea, Nitrobacter winogradskyi) | Biological nitrogen conversion (NH₄⁺ → NO₂⁻ → NO₃⁻) in BLSS [77] | MELiSSA Compartment III; nitrogen recovery from urine [77] |
| Photoautotrophic Organisms (Arthrospira platensis) | Oxygen production, CO₂ assimilation, edible biomass production [78] | MELiSSA Compartment IV; air revitalization and nutrition [3] |
| Higher Plant Species (Lettuce, wheat, potato) | Food production, air revitalization, water purification, psychological benefits [3] | MELiSSA Compartment V; "salad machine" concept [3] |
| Acidic Stabilization Solution (H₃PO₄ with Cr⁶+) | Urine stabilization to prevent scaling in physicochemical systems [77] | ISS Urine Processor Assembly; originally used H₂SO₄ [77] |
| Sabatier Catalyst (Typically ruthenium or nickel-based) | CO₂ reduction with H₂ to produce CH₄ and H₂O [77] | ISS Carbon Reduction System; closes oxygen loop [77] |
| Polymer Electrolyte Membrane (PEM) | Oxygen generation via water electrolysis [77] | ISS Oxygen Generation System; primary O₂ source [77] |
Ground demonstrators play an indispensable role in advancing life support system technologies for long-duration space missions. The MELiSSA Pilot Plant represents a sophisticated approach to bioregenerative life support with its compartmentalized ecosystem architecture, while traditional physicochemical systems like the ISS ECLSS offer high reliability and operational maturity but limited closure of material cycles. The experimental data from these testbeds reveals a fundamental trade-off: bioregenerative systems offer greater long-term sustainability and resource closure, while physicochemical systems provide more immediate reliability and lower operational complexity [25] [77].
The continuing development of these ground demonstrators addresses critical knowledge gaps in system integration, control strategies, and long-term stability. As research progresses, hybrid approaches that combine the strengths of both biological and physicochemical systems may offer the most practical pathway for near-term mission applications while advancing toward the ultimate goal of fully sustainable life support for enduring human presence beyond Earth.
Suitability Analysis for Different Mission Profiles (LEO, Moon, Mars)
The choice of Environmental Control and Life Support Systems (ECLSS) is a fundamental determinant of feasibility, cost, and safety for long-duration human space missions. As space agencies and commercial entities plan missions beyond Low Earth Orbit (LEO), the debate between primarily physicochemical (PC) systems and bioregenerative approaches like the Micro Ecological Life Support System Alternative (MELiSSA) becomes increasingly critical [63] [25]. This guide provides an objective comparison of these technologies, analyzing their suitability across three distinct mission profiles: LEO, Lunar, and Martian missions. The analysis is grounded in experimental data and the operational parameters of existing systems, providing a framework for researchers and mission planners.
Physicochemical (PC) Life Support Systems
Traditional PC systems rely on engineered processes to maintain crew life. These are characterized by discrete, mechanical subsystems for specific functions: carbon dioxide scrubbing (e.g., with lithium hydroxide), oxygen generation (e.g., through water electrolysis), water purification (via filtration and chemical treatment), and waste management (encompassing storage, processing, and disposal) [63]. The International Space Station (ISS) represents the most advanced implementation of this technology, achieving high recovery rates for water and oxygen, but requiring regular resupply from Earth and unable to produce significant amounts of food [63].
Bioregenerative Life Support Systems (BLSS)
Bioregenerative systems incorporate biological components—such as microalgae, higher plants, and microorganisms—to create a more closed-loop ecosystem. These systems aim to regenerate air and water and produce food by recycling waste streams biologically [75] [79].
The MELiSSA project, led by the European Space Agency, is a prominent BLSS example. Its design is inspired by aquatic ecosystems and is structured into several interconnected compartments, each with a specific microbial or plant culture that processes waste and regenerates resources [75] [13]. The ultimate goal is to achieve a high degree of circularity, thereby reducing dependency on Earth-based resupply [75].
Table 1: Fundamental Characteristics of PC and BLSS Approaches
| Feature | Physicochemical (PC) Systems | Bioregenerative (MELiSSA) |
|---|---|---|
| Core Principle | Mechanical & chemical processes [63] | Simulated ecosystem with compartmentalized microbes & plants [75] [13] |
| Air Revitalization | CO₂ scrubbing, O₂ generation (e.g., electrolysis) [63] | Photosynthesis (micro-algae, higher plants) [75] |
| Water Recovery | Filtration, distillation, chemical treatment [63] | Biological processing, membrane technologies [75] [63] |
| Waste Processing | Storage, compaction, partial physico-chemical processing [63] | Use of wastes (CO₂, organic wastes) as feedstock [75] [79] |
| Food Production | None; entirely reliant on resupply | Integrated food production (e.g., cereals, vegetables) [63] [79] |
| Technology Readiness | High (flight-proven on ISS) [63] | Low to Medium (ground demonstrators, ongoing research) [75] [25] |
Mission Profile Suitability Analysis
The optimal life support architecture varies significantly with mission distance, duration, and logistical constraints.
Low Earth Orbit (LEO) Missions
Characteristics: Proximity to Earth enables frequent resupply and crew evacuation. Volume, mass, and power constraints are significant.
Analysis: For LEO, the high Technology Readiness Level and reliability of PC systems make them the dominant choice. The ISS demonstrates that with regular resupply missions, PC systems can effectively support a crew indefinitely. The primary challenge is the high cost of launching water and food. While BLSS technologies like MELiSSA are tested in ground-based pilot plants for LEO-relevant research, their higher complexity, larger volume/mass footprint, and longer development cycles currently preclude them from being the primary system for LEO stations [75] [63].
Lunar Missions
Characteristics: A few days' travel from Earth, allowing for limited resupply but at a much higher cost and latency than LEO. Missions may range from short-term stays to long-duration habitation at a lunar base.
Analysis: Lunar missions represent a transition zone. For short-duration missions, compact and reliable PC systems retain an advantage. However, for a sustained human presence, the logistical and economic benefits of BLSS become compelling. The Chinese Yuegong-1 (Lunar Palace) program, which successfully demonstrated a closed-loop system sustaining a crew for a full year, provides strong evidence for the viability of BLSS in this context [63] [25]. The system achieved full oxygen regeneration and 100% water recycling, with over half of the food produced internally [63]. This significantly reduces the mass of consumables that need to be launched from Earth, making a permanent lunar base more feasible.
Mars Missions
Characteristics: Long duration (3+ years), extreme distance from Earth making resupply impossible or prohibitively expensive, and communication delays. This mission profile demands the highest level of self-sufficiency.
Analysis: For a mission to Mars, a bioregenerative life support system is considered essential for long-term crew survival and mission sustainability [75] [25]. The closed-loop capabilities of MELiSSA, which aims to produce food, recover water, and regenerate the atmosphere from waste streams, directly address the core challenge of Mars missions: the inability to resupply [75]. While PC systems could potentially handle air and water revitalization for the duration of a transit, they cannot address food production. The integrated approach of a BLSS is the only known technology that can close all major loops (atmosphere, water, and food) for multi-year missions without massive initial mass penalties [79] [25].
Table 2: Suitability Analysis Across Mission Profiles
| Mission Parameter | LEO | Moon (Outpost) | Mars |
|---|---|---|---|
| Recommended Primary System | Physicochemical | Hybrid / BLSS | Bioregenerative (BLSS) |
| Resupply Feasibility | High | Medium (Costly) | Very Low / Impossible |
| Key System Driver | Reliability, Compactness | Logistical Sustainability | Total Self-Sufficiency |
| Food Source | Resupplied | Partially Resupplied, Partially Grown (≈50%) [63] | Primarily BLSS-Produced |
| Water Closure | High Recovery (PC) | Full Recycling (BLSS demonstrated) [63] | Full Recycling (BLSS required) |
| Atmosphere Closure | High Recovery (PC) | Full O₂ Regeneration (BLSS demonstrated) [63] | Full O₂ Regeneration (BLSS required) |
| Notable Projects | ISS ECLSS | Yuegong-1 [63], future MELiSSA implementation | MELiSSA Goal [75], BIO-Plex Concept [25] |
Experimental Protocols & Data
MELiSSA Pilot Plant Ground Demonstration
The MELiSSA Pilot Plant (MPP) at the Universitat Autònoma de Barcelona serves as a key ground-based experimental platform for validating the BLSS concept [75].
- Objective: To demonstrate the feasibility, reliability, and efficiency of a closed-loop life support system by connecting different biological compartments (waste degradation, nitrification, air revitalization with algae, food production with plants) in the gas, liquid, and solid phases [75].
- Methodology: The loop is operated with a mock-up crew of rats. Their waste (CO₂ and organic matter) is fed into the first compartments, which consist of specific bacteria for breakdown and nitrification. The resulting nutrients are then used to cultivate cultures of the cyanobacterium Arthrospira platensis (for air revitalization and potential biomass) and higher plants in dedicated chambers for food production [75]. The process is continuously monitored, and accurate mathematical models are developed to predict and control the system's behavior.
- Key Metrics: System closure rates, stability of each compartment, nutrient flow efficiency, and oxygen production/consumption balance.
Yuegong-1 Year-Long Experiment
The Chinese Yuegong-1 (Lunar Palace 1) experiment provides critical data on long-term human habitation within a BLSS.
- Objective: To demonstrate the viability of a closed artificial ecosystem with humans, animals, plants, and microorganisms for extended durations [63].
- Methodology: A crew of three lived inside the Yuegong-1 facility for 370 days. The system comprised a vegetation compartment for growing cereals and vegetables and a compartment for recycling organic waste using microorganisms. The primary food source was internally cultivated wheat, which also served as the main oxygen producer. Mealworms were raised on vegetative waste as a protein source. Human and organic waste was treated microbiologically and partly used as fertilizer [63].
- Key Results: The experiment achieved 100% regeneration of oxygen and 100% recycling of water within the system. The crew produced more than half of their food internally, a critical milestone for BLSS viability [63].
System Architecture and Workflows
The functional logic of a BLSS like MELiSSA can be visualized as a series of interconnected processes that mirror a natural ecosystem. The following diagram illustrates the core workflow for converting waste into vital resources.
Diagram 1: Simplified workflow of the MELiSSA loop, showing the conversion of crew waste into oxygen, food, and clean water through a series of interconnected biological compartments.
The Scientist's Toolkit: Key Research Reagents & Materials
Research and development in BLSS rely on a specific set of biological and technological components. The following table details essential elements used in experiments like those conducted in the MELiSSA Pilot Plant and Yuegong-1.
Table 3: Essential Research Materials for BLSS Experimentation
| Research Material / Solution | Function in BLSS Research |
|---|---|
| Arthrospira platensis (Spirulina) | A cyanobacterium used in the "air revitalization" compartment for efficient CO₂ capture and O₂ production via photosynthesis [75]. |
| Higher Plant Chambers (Phytotrons) | Controlled environment chambers for studying food production (e.g., wheat, vegetables), O₂ regeneration, and water transpiration from plants [75] [63]. |
| Nitrogen & Phosphorus Recovery Systems | Technologies for recovering essential plant nutrients, particularly from human urine, to create fertilizing solutions for hydroponic plant growth [79]. |
| Hydroponic Nutrient Solutions | Aqueous solutions containing essential mineral nutrients required for plant growth in soilless (hydroponic or aeroponic) cultivation systems [79]. |
| Mathematical Models & Control Laws | Computational models that simulate the dynamics of the entire ecosystem. These are crucial for predicting behavior, optimizing performance, and developing automated control strategies [75]. |
| Biofilm Carriers | Physical substrates used in continuous-flow reactors (e.g., for nitrification) to support the growth of beneficial bacterial biofilms, enhancing process stability and efficiency [75]. |
| Membrane Bioreactors | Integrated systems using biological treatment and membrane filtration (e.g., ultrafiltration, reverse osmosis) for advanced wastewater and greywater recycling [75] [63]. |
The suitability of life support technologies is inherently tied to mission profile. Physicochemical systems provide a proven, reliable solution for LEO, where resupply is viable. In contrast, bioregenerative systems like MELiSSA are not merely alternatives but are enabling technologies for long-duration lunar outposts and especially for Mars missions, where total self-sufficiency is the ultimate requirement. The ground demonstrations by MELiSSA and the year-long closed-loop experiment in Yuegong-1 provide compelling data that BLSS is transitioning from theoretical concept to a practical solution for humanity's sustainable future in deep space. Future research must focus on integrating these systems, optimizing their mass and volume, and increasing their Technology Readiness Level for flight.
The pursuit of long-duration human spaceflight has catalyzed the development of advanced regenerative life support systems, with the Micro-Ecological Life Support System Alternative (MELiSSA) project standing as a prominent European endeavor [15]. Established in 1989, MELiSSA aims to achieve the highest degree of crew autonomy by producing food, water, and oxygen from mission wastes, embodying a perfect circular economy in a closed environment [15]. This research provides a critical foundation for addressing sustainability challenges on Earth, particularly in transforming linear agricultural processes into circular systems.
Simultaneously, vertical farming has emerged as a revolutionary method for cultivating crops in vertically stacked layers within controlled environments [80]. This approach is increasingly crucial as urbanization accelerates and arable land diminishes. The core premise of this analysis is that the principles underpinning MELiSSA and other controlled ecological life support systems (CELSS) can be directly applied to enhance the sustainability and efficiency of terrestrial vertical farming [65]. This article frames the comparison within a broader thesis on ecological (MELiSSA-type) versus traditional physicochemical life support systems, evaluating their respective philosophies and their applicability to modern agriculture. Where traditional physicochemical systems rely on physical and chemical processes for air and water revitalization, ecological systems like MELiSSA incorporate biological components—plants and microorganisms—to create a more synergistic, regenerative loop [63] [65].
Comparative Framework: Ecological vs. Physicochemical Systems
The fundamental difference between ecological and physicochemical life support systems lies in their approach to closure and regeneration. The following table outlines their core characteristics, which form the conceptual basis for their terrestrial spin-offs.
Table 1: Fundamental Comparison of Life Support System Paradigms
| Feature | Ecological Systems (MELiSSA-like) | Traditional Physicochemical Systems |
|---|---|---|
| Core Principle | Biological regeneration using interconnected compartments of microbes and plants [15] | physico-chemical processing of wastes [63] |
| Waste Processing | Mineralization of waste by microorganisms to produce nutrients for plants [15] | Filtration, distillation, and chemical oxidation (e.g., TCRS, RP-LiOH) [63] |
| Gas Revitalization | Plant photosynthesis for O2 production and CO2 consumption [63] | Oxygen supplied from stored tanks or water electrolysis; CO2 removed by chemical scrubbers [63] |
| Water Recovery | Biological processes (e.g., plant transpiration) combined with physico-chemical steps [63] | Primarily physico-chemical methods (e.g., reverse osmosis, vapor compression distillation) [63] |
| System Character | Network of biological reactors with complex, adaptive control [65] | Series of engineering units with linear, set-point control |
| Terrestrial Analog | Circular vertical farming integrated with urban waste streams [81] [82] | Linear vertical farming with imported resources and exported waste [82] |
This dichotomy extends to vertical farming. Linear vertical farms, akin to physicochemical systems, depend on external inputs of resources and export waste, while circular vertical farms mimic ecological systems by creating closed-loop flows of energy, water, and nutrients [82].
Experimental Investigation of Circular Vertical Farming
Aims and Rationale
A recent 2024 study directly addressed the integration of circular economy principles into vertical farming, aiming to evaluate the efficiency of different methods and introduce a zero-waste circular economy model [81]. The study was motivated by knowledge gaps regarding the practical viability of such systems and the lack of a clear understanding of the flow of value-adding components [81]. Its primary goal was to provide a blueprint for agriculturalists and decision-makers to adopt more sustainable and efficient farming practices that reduce waste and generate profits [81].
Detailed Experimental Protocol
1. System Design and Nutrient Source Preparation: The experiment proposed and implemented a circular economy model utilizing waste streams as nutrients for six distinct halophyte plants in a secondary farming setup [81]. The four nutrient sources tested were:
- Hydroponic waste: Spent nutrient solution from a primary hydroponic farm.
- Aquaponic waste: Effluent from a primary aquaponic system combining fish cultivation and plant growth.
- Vermicompost tea: A liquid fertilizer derived from vermicompost (worm castings).
- Aerobic compost tea/quick compost: A liquid extract from actively aerated compost.
2. Plant Growth and Monitoring: Halophyte plants were cultivated using the above nutrient sources in a controlled secondary farming setup. The growth of these plants was meticulously monitored and compared over a period of six weeks [81]. Key growth metrics were recorded to assess the performance of each nutrient source.
3. Data Analysis and Simulation Model Development: Using the results from the experimental observation, a computer-based simulation model was developed [81]. The model's accuracy was validated by comparing its outcomes with the actual experimental data. Subsequently, the validated model was used to optimize resource allocation and balance capacities at each stage of the vertical farming process within the circular framework [81].
Table 2: Comparison of Experimental Nutrient Sources for Circular Vertical Farming
| Nutrient Source | Origin in Circular System | Experimental Performance | Key Advantage |
|---|---|---|---|
| Hydroponic Waste | Spent solution from primary hydroponic farm | Superior plant growth | Efficient reuse of dissolved minerals, high nutrient availability [81] |
| Aquaponic Waste | Effluent from integrated fish cultivation system | Superior plant growth | Symbiotic system producing nutrient-rich effluent, adds protein source [81] |
| Vermicompost Tea | Processing of organic urban waste | Inferior growth compared to hydroponic/aquaponic waste | Valorizes solid urban organic waste, improves soil health [82] |
| Aerobic Compost Tea | Processing of organic urban waste | Inferior growth compared to hydroponic/aquaponic waste | Rapid production, valorizes solid urban organic waste [81] |
System Workflow and Logical Relationships
The following diagram illustrates the integrated, closed-loop workflow of a circular vertical farming system, synthesizing concepts from the MELiSSA philosophy and the described experiment.
Figure 1: Closed-loop workflow of a circular vertical farming system, showing material and nutrient flows from waste inputs to food outputs.
Results and Comparative Analysis
Quantitative Performance of Circular Models
The experimental data and systems analysis allow for a direct comparison between linear and circular vertical farming models, highlighting the tangible benefits of adopting a MELiSSA-inspired approach.
Table 3: Performance Comparison of Linear vs. Circular Vertical Farms
| Aspect | Linear Vertical Farm | Circular Vertical Farm | Supporting Experimental Data |
|---|---|---|---|
| Energy Source | Grid-dependent, often fossil fuel-based [82] | Integrated renewables, waste heat recovery [82] | N/A (System-level comparison) |
| Water Management | Recirculation within the farm only [82] | Closed-loop with urban wastewater capture [82] | Water use reduced by up to 95% vs. conventional agriculture [80] |
| Nutrient Sourcing | Virgin mineral fertilizers [82] | Valorized urban organic waste, nutrient recovery from wastewater [81] [82] | Hydroponic & aquaponic waste showed superior growth as nutrients [81] |
| Waste Management | Landfill disposal of organic waste [82] | On-site composting, conversion to biofuel/animal feed [82] | 100% of water recycled internally in systems like Yuegong-1 [63] |
| Economic Outcome | High operational costs, potential for social inequity [82] | Lower input costs, potential for new revenue streams (e.g., waste valorization) [81] | Model optimized for "high profitability" and "minimum cost" [81] |
Hierarchical Control and System Stability
A key challenge in managing complex, multi-compartment biological systems like MELiSSA or an advanced circular farm is control. Research has led to the development of a hierarchical control structure for ecological life support systems [65]. This architecture uses Model Predictive Control (MPC) at a high level to coordinate a network of biological compartments with varying generation, consumption, and storage capabilities, ensuring the balance of key compounds like O2 despite fluctuating demand [65]. This sophisticated control philosophy, essential for stable space missions, translates directly to the reliable and efficient operation of large-scale, integrated circular vertical farms on Earth.
Figure 2: Hierarchical control structure for managing a complex, closed-loop ecological system, ensuring stability against demand fluctuations.
The Scientist's Toolkit: Research Reagent Solutions
Translating these concepts from theoretical models to practical applications requires a specific suite of research tools and reagents. The following table details essential materials for developing and experimenting with circular vertical farming systems.
Table 4: Essential Research Reagents and Materials for Circular Vertical Farming Experiments
| Reagent / Material | Function in Experimental Protocol |
|---|---|
| Halophyte Plant Seeds | Model organisms for testing nutrient recovery in secondary farming setups; tolerant of varying nutrient conditions [81]. |
| Hydroponic & Aquaponic Effluent | Waste stream used as experimental nutrient source; contains dissolved minerals and organic compounds from primary production loops [81]. |
| Vermicompost | Processed organic waste used to produce vermicompost tea, a liquid fertilizer for testing valorization of solid urban wastes [81]. |
| Viral Vectors (e.g., Lentiviral) | Note: Not used in agriculture. This is a key differentiator. Tools for gene therapy and cell therapy in medical research, unrelated to vertical farming systems [55]. |
| MELISSA Statistical Framework (R Package) | Note: A naming conflict. This is a different "MELISSA". A regression-based tool for analyzing Integration Site (IS) data to assess insertional mutagenesis risk in gene therapy [55]. |
| Sensor Array (pH, DO, EC, Light) | Critical for real-time monitoring of controlled environment agriculture (CEA); provides data for IoT and AI-driven optimization [80]. |
| Defined Microbial Consortia | Selected microorganisms for waste bioremediation and mineralization, mirroring the functional compartments of the MELiSSA loop [15]. |
| Computer Simulation Software | For developing system dynamics models to simulate and optimize resource flows, capacities, and economic viability before physical implementation [81]. |
This comparison elucidates a clear trajectory for the future of sustainable agriculture. The experimental evidence demonstrates that hydroponic and aquaponic waste streams are superior nutrient sources within a circular framework, validating the technical feasibility of closing nutrient loops [81]. This aligns with the core MELiSSA principle of waste-as-a-resource.
The hierarchical control strategies developed for complex ecological life support systems provide a robust blueprint for managing the dynamic interactions within a circular vertical farm, ensuring system stability and efficiency [65]. While traditional physicochemical systems offer reliability, their terrestrial analogs in agriculture perpetuate a linear, resource-intensive model. The fusion of vertical farming with circular economy principles, guided by the wisdom of space-life-support research, transforms vertical farms from mere food producers into integral nodes of a resilient urban metabolism [82]. This ascent, however, is contingent upon continued research into system integration, energy-efficient renewable power, and supportive policy frameworks that encourage the transition from linear to circular systems.
Future Research Directions for Achieving Full System Integration
For long-duration human space missions beyond Earth orbit, life support systems transition from being mere life-sustaining utilities to critical mission-enabling infrastructure. The fundamental challenge lies in achieving full system integration—seamlessly combining biological and physicochemical processes into a reliable, self-sustaining ecosystem that can operate with minimal resupply from Earth. Current life support technologies exist on a spectrum from purely physicochemical systems (exemplified by the International Space Station's ECLSS) to bioregenerative approaches (as demonstrated in the MELiSSA project and China's Lunar Palace), each with distinct advantages and limitations in terms of closure, resilience, and operational complexity [2] [63].
The pursuit of integrated systems represents more than a technical optimization problem; it constitutes a fundamental reimagining of how humans will sustainably inhabit the space environment. As space agencies plan for enduring presence on the Moon and eventual missions to Mars, the strategic necessity of advanced life support has become increasingly apparent. Historical decisions to discontinue programs like NASA's BIO-PLEX and CELSS have created significant capability gaps that now require urgent attention, particularly as other spacefaring nations have advanced their own bioregenerative technologies [2]. This comparison guide examines the current state of life support technologies, analyzes integration challenges through experimental data, and outlines the critical research directions needed to achieve fully functional integrated life support systems.
Comparative Analysis of Life Support System Architectures
Fundamental Design Philosophies and Historical Context
Physicochemical Life Support Systems (PCLSS) operate on a principle of mechanical recycling through engineered processes. These systems typically employ discrete subsystems for specific functions: carbon dioxide removal (using absorbents like lithium hydroxide), oxygen generation (through water electrolysis), and water recovery (via filtration and distillation technologies) [63] [83]. The architecture is characterized by high reliability through redundancy and precise controllability, but suffers from limited closure rates—the International Space Station's system recovers approximately 90% of water and 40-50% of oxygen, with food remaining entirely Earth-dependent [83].
In contrast, Bioregenerative Life Support Systems (BLSS) embrace an ecological approach where biological components perform multiple life support functions simultaneously. The MELiSSA (Micro-Ecological Life Support System Alternative) project, initiated by the European Space Agency in 1989, exemplifies this approach with its compartmentalized ecosystem designed to recycle waste into food, water, and oxygen [75] [13]. Similarly, China's Yuegong-1 (Lunar Palace 1) has demonstrated closed-system operations supporting crews for extended durations—including a notable 370-day experiment where the system regenerated all oxygen, recycled all water, and produced over half of the food consumed [2] [63].
Table 1: Comparative Overview of Life Support System Architectures
| System Characteristic | Physicochemical Systems (ISS) | Bioregenerative Systems (MELiSSA) | Hybrid Systems (BIO-PLEX Concept) |
|---|---|---|---|
| Oxygen Regeneration | Water electrolysis (OGA) ~40-50% recovery with Sabatier [83] | Photosynthesis (algae & plants) Near-complete regeneration [75] | Combined biological & physicochemical approaches |
| Water Recovery | Multistage filtration & distillation ~90% recovery [63] | Biological processing + membranes 100% demonstrated in Yuegong-1 [63] | Advanced membranes with biological pretreatment |
| Food Production | None (all supplied) | Up to 50-80% in demonstration systems [63] | Balanced supplement to stored food |
| Waste Processing | CO₂ vented after Sabatier, solids stored | Complete recycling of organic wastes [79] | Partial recycling with waste stabilization |
| System Closure | Partial (~50% O₂, ~90% H₂O, 0% food) | High (approaching full closure in tests) [63] | Moderate to high (dependent on design) |
| Technology Readiness | TRL 9 (flight proven) | TRL 4-6 (ground demonstration) [75] | TRL 3-5 (conceptual studies) |
| Energy Requirements | High (electrolysis, compressors) | Moderate (primarily for lighting) [63] | High (both biological and mechanical needs) |
Performance Metrics and Experimental Data
Quantitative performance data from various testbeds and operational systems reveals critical differences in efficiency, resource requirements, and closure capabilities. These metrics are essential for evaluating the maturity and potential of each approach for future space missions.
Table 2: Experimental Performance Data from Life Support System Testing
| System/Testbed | Duration & Scale | Closure Achievements | Key Limitations Identified |
|---|---|---|---|
| ISS ECLSS | Continuous (operational) | 90% water recovery, 40-50% O₂ with Sabatier [83] | Methane venting, food entirely resupplied, limited closure |
| Yuegong-1 (China) | 370-day crewed test [63] | 100% O₂, 100% water, >50% food [63] | System complexity, control challenges, nitrogen balance |
| MELiSSA Pilot Plant | Ongoing (rat crews) [75] | Compartment-level validation, food production [75] | Human-rated testing pending, sodium accumulation [79] |
| Bios-3 (Russia) | Multiple experiments (1970s) | 93% food, full air & water regeneration [63] | High energy requirements, limited dietary variety |
| NASA BIO-PLEX | Concept (canceled) | Designed for ~97% closure [2] | Program discontinued before implementation |
| NGLS Project | Technology development | Targeting 100% oxygen recovery [83] | Focused on subsystems, not integrated testing |
The data demonstrates that while physicochemical systems offer proven reliability, they face fundamental limitations in closure rates. Bioregenerative systems show potential for near-complete closure but require further development to address stability, control complexity, and integration challenges. The resource efficiency of biological systems is particularly notable—in the Yuegong-1 system, wheat served not only as a primary food source but also as the main producer of oxygen, demonstrating the multifunctionality that characterizes bioregenerative approaches [63].
Critical Integration Challenges and Research Frontiers
Nutrient Balance and Metabolic Integration
One of the most significant challenges in full system integration lies in achieving precise nutrient balance across the combined biological and physicochemical processes. In the MELiSSA loop, research has highlighted the critical need to efficiently remove sodium and chloride from urine and other organic wastes to prevent the spread of these elements throughout the system [79]. Similarly, maintaining appropriate nitrogen balance at the habitat level presents a complex optimization problem—sufficient N₂ must remain to maintain atmospheric pressure while adequate mineral nitrogen must be available for plant nutrition [79].
The MELiSSA loop's compartmentalized approach represents a structured methodology for addressing these integration challenges. Each compartment contains specific bacteria or higher plants optimized for particular functions: waste degradation, nitrification, air revitalization through micro-algae photosynthesis, and food production with higher plants [75]. This compartmentalization enables precise control and troubleshooting but introduces interface challenges between biological and physicochemical components.
Diagram 1: MELiSSA Loop Compartment Integration
Control Systems and Operational Stability
Achieving operational stability in integrated life support systems requires advanced control strategies that can accommodate the different timescales of biological and physicochemical processes. Physicochemical systems typically respond within minutes to hours, while biological components may require days or weeks to reach steady-state operations. The development of accurate mathematical models has been identified as particularly relevant for predicting system behavior and designing effective control laws [75].
Research from the MELiSSA Pilot Plant emphasizes a progressive approach to control strategy validation, beginning with process characterization and mathematical modeling before advancing to integrated demonstration [75]. This methodical development process highlights the complexity of managing biological systems, which exhibit nonlinear responses to environmental conditions and require sophisticated monitoring of parameters that cannot be easily measured in real-time, such as nutrient solution composition and microbial community health.
Experimental Protocols for System Integration Research
Integrated Loop Closure Testing Methodology
Objective: To evaluate the stability and closure capabilities of hybrid life support systems during extended-duration operation with continuous recycling of atmospheric, water, and nutrient streams.
Experimental Setup: The test facility must incorporate both biological and physicochemical components in a closed configuration. The MELiSSA Pilot Plant exemplifies this approach with interconnected compartments for waste degradation, nitrification, air revitalization (using micro-algae), food production (using higher plants), and a mock crew compartment (currently using rats as human analogs) [75].
Key Parameters Monitored:
- Atmospheric Balance: O₂ and CO₂ concentrations, trace gas accumulation
- Water Quality: Recycling efficiency, contaminant accumulation, pH and conductivity
- Nutrient Flux: Macronutrient (N, P, K) and micronutrient cycling efficiency
- Biological Performance: Plant growth rates, algal productivity, microbial community stability
- System Resources: Energy consumption, maintenance requirements, operational complexity
Protocol Duration: Experiments should span multiple recycling cycles to identify accumulation effects. The 370-day Yuegong-1 mission demonstrates the value of extended testing for observing seasonal variations and long-term trends [63].
Comparative Performance Assessment Protocol
Objective: To quantitatively compare the efficiency, reliability, and resource requirements of integrated systems against conventional physicochemical systems.
Experimental Design: Parallel operation of different system architectures under identical metabolic loading conditions, with careful measurement of all inputs and outputs.
Table 3: Essential Research Reagent Solutions for Life Support Integration Studies
| Reagent Category | Specific Examples | Research Function | Integration Challenge Addressed |
|---|---|---|---|
| Nutrient Solutions | Hoagland's solution, MeliSSA nutrient medium [79] | Plant growth optimization | Balancing nutrient availability with recycling streams |
| Microbial Consortia | Nitrifying bacteria, Arthrospira platensis, specific waste-degrading strains [75] | Waste processing and air revitalization | Maintaining functional stability in mixed cultures |
| Water Quality Markers | Ionic tracers (Na⁺, Cl⁻), fluorescent dyes, stable isotopes | Tracking contaminant pathways | Identifying accumulation points in closed loops |
| Gas Standards | CO₂ in air mixtures, trace gas standards (ethylene, methane) | Atmospheric monitoring calibration | Controlling trace contaminant buildup |
| Analytical References | Internal standards for HPLC, GC-MS, ICP-MS | Quantitative performance verification | Ensuring measurement accuracy across disciplines |
Measurement Methodology:
- Closure Metrics: Direct measurement of recycling rates for water, oxygen, and food
- Resource Intensity: Comprehensive accounting of mass, volume, and power requirements per person-day of support
- Stability Assessment: Response to simulated failure scenarios and variable loading conditions
- Crew Time Requirements: Measurement of operational and maintenance demands
Strategic Research Priorities for Full Integration
Near-Term Research Imperatives (1-5 Years)
The achievement of fully integrated life support systems requires addressing several critical research gaps through targeted investigations:
Nutrient Management and Contaminant Control: Research must focus on developing efficient processes for sodium and chloride removal from urine and other waste streams to prevent the spread of these elements throughout the system [79]. Simultaneously, techniques for maintaining nutrient solution balance in hydroponic systems using waste-derived nutrients require refinement to support diverse crop production.
System Control and Modeling: Advanced control strategies must be developed to manage the different timescales and responses of biological and physicochemical components. This includes creating dynamic mathematical models that can predict system behavior under varying conditions and support the development of fault detection and isolation protocols [75].
Interface Standardization: Research should establish standardized interfaces between biological and physicochemical subsystems to enable modular development and testing. This includes defining common protocols for gas, liquid, and solid exchanges between system components.
Long-Term Research Vision (5-15 Years)
Multi-System Hybridization: Future research should explore optimal combinations of biological and physicochemical technologies rather than treating them as competing alternatives. The Next Generation Life Support (NGLS) project's approach of developing multiple technology options for specific functions provides a model for this work [83].
In-Situ Resource Utilization Integration: For planetary surface missions, integrated life support must incorporate local resource utilization. Research is needed to connect life support systems with technologies that extract water from Martian soil or lunar regolith, as analyzed in site candidate studies for Mars habitats [84].
Autonomous Operation Capabilities: As missions extend farther from Earth, life support systems must operate with increasing autonomy. This requires research into self-diagnostic capabilities, predictive maintenance, and adaptive control systems that can respond to changing conditions without ground intervention.
The pursuit of fully integrated life support systems represents one of the most complex interdisciplinary challenges in space exploration, requiring convergence of biology, engineering, materials science, and informatics. While physicochemical systems provide immediate, proven capabilities for near-term missions, bioregenerative approaches offer the only credible path to long-term sustainability for extended-duration missions beyond Earth orbit.
The experimental evidence from testbeds worldwide demonstrates that no single approach currently provides an optimal solution across all metrics of performance, reliability, and sustainability. The strategic imperative is therefore to advance both technological pathways while placing increased emphasis on integration methodologies that leverage the complementary strengths of biological and physicochemical processes. This will require sustained investment in ground demonstration projects, international collaboration to leverage diverse expertise, and a systematic approach to technology maturation that bridges the gap between subsystem innovation and full-system validation.
As noted in historical analyses of life support development, decisions made today regarding research investment and international partnership will determine space exploration capabilities for decades to come [2]. By prioritizing integrated system development and learning from both past successes and discontinuations, the space community can create the life support infrastructure needed to sustain human presence beyond Earth orbit.
Conclusion
The journey toward sustainable long-duration space habitation does not present a binary choice between bioregenerative and physicochemical systems, but rather a strategic integration of both. MELiSSA represents the pinnacle of bioregenerative research, offering unparalleled potential for food production and complex waste recycling, thereby enhancing crew well-being and mission sustainability. However, its current challenges in reliability and dynamic response are effectively complemented by the robust, high-efficiency nature of traditional ECLSS. The future lies in hybrid systems that leverage the strengths of both paradigms. The ongoing research, facilitated by advanced ground demonstrators and a growing international consortium, is steadily paving the way for the closed-loop life support systems essential for humanity's future on Mars and beyond, with simultaneous profound implications for circular economy solutions on Earth.
References
- 1. ESA's MELiSSA life-support programme wins academic ... [https://www.esa.int/Enabling_Support/Space_Engineering_Technology/ESA_s_MELiSSA_life-support_programme_wins_academic_recognition]
- 2. Critical investments in bioregenerative life support systems ... [https://www.nature.com/articles/s41526-025-00518-4]
- 3. Plant and microbial science and technology as ... [https://www.nature.com/articles/s41526-023-00317-9]
- 4. Importance and challenges of integrating BLSS into ECLSS [https://www.sciencedirect.com/science/article/abs/pii/S0094576524002157]
- 5. ISS ECLSS [https://en.wikipedia.org/wiki/ISS_ECLSS]
- 6. Development of Nitrogen Recycling Strategies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8548772/]
- 7. BLSS: a contribution to future life support [https://pubmed.ncbi.nlm.nih.gov/11537782/]
- 8. ESA - MELiSSA life support project, an innovation network ... [https://www.esa.int/Enabling_Support/Space_Engineering_Technology/MELiSSA_life_support_project_an_innovation_network_in_support_to_space_exploration]
- 9. The conceptual design of a hybrid life support system ... [https://www.sciencedirect.com/science/article/abs/pii/S0273117705007933]
- 10. MELiSSA [https://en.wikipedia.org/wiki/MELiSSA]
- 11. BIOS-3 [https://en.wikipedia.org/wiki/BIOS-3]
- 12. BIOS-3 – BIOlogical closed life support System [https://www.ibp.ru/science/bios3.php]
- 13. Melissa: The European project of a closed life support system [https://ui.adsabs.harvard.edu/abs/2008cosp...37.1706L/abstract]
- 14. MELiSSA SPACE RESEARCH PROGRAM [https://www.melissafoundation.org/page/melissa-project]
- 15. Melissa Foundation [https://www.melissafoundation.org/]
- 16. ESA - Organisation [https://www.esa.int/Enabling_Support/Space_Engineering_Technology/Melissa/Organisation]
- 17. Biologically-Based and Physiochemical Life Support and In ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8398003/]
- 18. ESA - Closed Loop Compartments [https://www.esa.int/Enabling_Support/Space_Engineering_Technology/Melissa/Closed_Loop_Compartments]
- 19. Architecture: a system level representation of the MELiSSA ... [http://ui.adsabs.harvard.edu/abs/2022cosp...44.2833D/abstract]
- 20. MELISSA: global control strategy of the artificial ecosystem ... [https://pubmed.ncbi.nlm.nih.gov/11542550/]
- 21. Survivability and life support in sealed mini-ecosystems ... [https://www.nature.com/articles/s41598-024-75328-x]
- 22. 2025 MELiSSA Conference [https://2025melissaconference.org/]
- 23. Exploration life support technology challenges for the Crew ... [https://www.sciencedirect.com/science/article/abs/pii/S0273117709006814]
- 24. Multiple-task performance on a computer-simulated life ... [https://pubmed.ncbi.nlm.nih.gov/11541761/]
- 25. Critical investments in bioregenerative life support systems for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12357894/]
- 26. Sabatier reaction [https://en.wikipedia.org/wiki/Sabatier_reaction]
- 27. Thermal management of a Sabatier reactor for CO ... [https://www.sciencedirect.com/science/article/abs/pii/S2212982016303614]
- 28. New Insights Into Nature's Oxygen-Making Machinery [https://newscenter.lbl.gov/2025/07/08/how-plants-manage-light-new-insights-into-natures-oxygen-making-machinery/]
- 29. Boosting photosynthesis for carbon capture [https://www.mpg.de/24879409/carbon-capture-photosynthesys-booster]
- 30. If algae produce oxygen in a pond, how can having too ... [https://freshwater-aquaculture.extension.org/if-algae-produce-oxygen-in-a-pond-how-can-having-too-much-algae-cause-an-oxygen-depletion/]
- 31. Water Recovery from Laundry Wastewater by Integrated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12029547/]
- 32. Toward sustainable space exploration: a roadmap for ... [https://www.nature.com/articles/s41467-023-37070-2]
- 33. Studies on the Recovery of Wash Water from Swimming ... [https://www.mdpi.com/2073-4441/17/19/2854]
- 34. Anaerobic Digestion Vs Incineration: A Comparison [https://blog.anaerobic-digestion.com/anaerobic-digestion-vs-incineration/]
- 35. Reviewing the Anaerobic Digestion of Food Waste [https://www.mdpi.com/2076-3417/8/10/1804]
- 36. Valorization of food waste into renewable fuels via ... [https://www.sciencedirect.com/science/article/pii/S0378382025001729]
- 37. A critical review of food waste and poultry manure ... [https://www.frontiersin.org/journals/sustainable-food-systems/articles/10.3389/fsufs.2025.1695945/full]
- 38. Impacts of Municipal and Industrial Organic Waste ... [https://link.springer.com/article/10.1007/s12649-025-02951-8]
- 39. Enhancing Methane Production in Anaerobic Digestion of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12029908/]
- 40. Towards supervisory Model Predictive Control for circular ... [https://www.sciencedirect.com/science/article/pii/S2214552425000707]
- 41. Progress and prospect of research on controlled ecological ... [https://www.sciencedirect.com/science/article/abs/pii/S2352309317300044]
- 42. 2025 MELiSSA Conference [https://space4water.org/events/2025-melissa-conference]
- 43. Comparing Life Support Solutions: PCLSS vs BLSS vs ... [https://thespringinstitute.com/comparing-life-support-solutions-pclss-vs-blss-vs-celss/]
- 44. Tracing dynamic biological processes during phase transition [https://pmc.ncbi.nlm.nih.gov/articles/PMC3403121/]
- 45. Process-based design of dynamical biological systems [https://www.nature.com/articles/srep34107]
- 46. 9.7 Dynamic modeling of biological systems - Bioinformatics [https://fiveable.me/bioinformatics/unit-9/dynamic-modeling-biological-systems/study-guide/JPPa2d8urlsOuXBK]
- 47. Insights In Biosafety & Biosecurity 2024/2025: Novel ... [https://www.frontiersin.org/research-topics/66300/insights-in-biosafety-biosecurity-20242025-novel-developments-current-challenges-and-future-perspectivesundefined]
- 48. Biosafety Levels 1, 2, 3 & 4: What's the Difference? [https://consteril.com/biosafety-levels-difference/]
- 49. Biosafety Levels & Lab Safety Guidelines [https://aspr.hhs.gov/S3/Pages/Biosafety-Level-Requirements.aspx]
- 50. BSL-3 Regulations: Critical Updates for 2025 - QUALIA [https://qualia-bio.com/blog/bsl-3-regulations-critical-updates-for-2025/]
- 51. CDC LC Quick Learn: Recognize the four Biosafety Levels [https://www.cdc.gov/training/quicklearns/biosafety/]
- 52. MELISSA: Global control strategy of the artificial ecosystem ... [https://www.sciencedirect.com/science/article/abs/pii/S0273117799004901]
- 53. Modelling & Control [https://www.melissafoundation.org/page/modelling-control]
- 54. Mechanistic dynamic modelling of biological systems [https://www.sciencedirect.com/science/article/abs/pii/S2452310025000137]
- 55. Modeling integration site data for safety assessment with ... [https://www.nature.com/articles/s41467-025-63017-w]
- 56. Modelling biological systems [https://en.wikipedia.org/wiki/Modelling_biological_systems]
- 57. Perspectives on computational modeling of biological systems ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11213628/]
- 58. Microalgae in bioregenerative life support systems for ... [https://www.sciencedirect.com/science/article/abs/pii/S221192642300365X]
- 59. LIFE SUPPORT SYSTEMS in the Space Industry [https://www.enginsoft.com/newsroom/eng/life-support-systems-in-the-space-industry.html]
- 60. Modeling and Simulation Tools for Smart Local Energy ... [https://www.mdpi.com/2076-3417/15/16/9219]
- 61. Aquatic bryophytes as biofilters and resource regenerators ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1667463/full]
- 62. 2025 Summer Seminar Series [https://www.nasa.gov/2025-nasa-summer-series/]
- 63. Life Support Systems - an overview [https://www.sciencedirect.com/topics/physics-and-astronomy/life-support-systems]
- 64. 3 Space-Based Technologies That Are Changing How We ... [https://www.spacefoundation.org/2022/10/11/space-agriculture-3-space-based-technologies-that-are-changing-how-we-grow-food/]
- 65. Hierarchically controlled ecological life support systems [https://www.sciencedirect.com/science/article/pii/S0098135421004038]
- 66. Digital twin for healthcare systems - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10513171/]
- 67. Advancing Emergency Care With Digital Twins - JMIR Aging [https://aging.jmir.org/2025/1/e71777]
- 68. AI-Driven Decision Support Framework for Preventing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12499571/]
- 69. DeepMind Identifies Melissa Five Days in Advance from a ... [https://eu.36kr.com/en/p/3542382439313280]
- 70. Comparison of risk-adjusted cumulative quality control ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11870913/]
- 71. Predictive Maintenance in Healthcare with AI | by Lena Tyson [https://medium.com/@lenaztyson/predictive-maintenance-in-healthcare-with-ai-1e1fffe05ebc]
- 72. New study analyzes hospitals' use of AI-assisted predictive ... [https://www.sph.umn.edu/news/new-study-analyzes-hospitals-use-of-ai-assisted-predictive-tools-for-accuracy-and-biases/]
- 73. FAQ [https://www.melissafoundation.org/page/faq]
- 74. Reliability and lifetime estimation of bioregenerative life ... [https://www.sciencedirect.com/science/article/abs/pii/S0094576522006294]
- 75. MELiSSA Pilot Plant [https://www.melissafoundation.org/page/melissa-pilot-plant]
- 76. Biologically-Based and Physiochemical Life Support and In ... [https://www.mdpi.com/2075-1729/11/8/844]
- 77. Development of Nitrogen Recycling Strategies for ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2021.700810/full]
- 78. ESA - Life support [https://www.esa.int/Science_Exploration/Human_and_Robotic_Exploration/Research/Life_support]
- 79. Recycling nutrients from organic waste for growing higher ... [https://www.sciencedirect.com/science/article/pii/S2214552423000639]
- 80. Vertical Farming Integration: The Future of Sustainable Agriculture [https://gothicarchgreenhouses.com/vertical-farming-integration-the-future-of-sustainable-agriculture]
- 81. A circular economy integration approach into vertical ... [https://www.sciencedirect.com/science/article/pii/S0959652624027057]
- 82. Circular Economy Principles Applied to Vertical Agriculture ... [https://prism.sustainability-directory.com/scenario/circular-economy-principles-applied-to-vertical-agriculture-systems/]
- 83. Next Generation Life Support (NGLS) [https://www.nasa.gov/directorates/stmd/game-changing-development-program/next-generation-life-support-ngls/]
- 84. Analysis of suitable site candidates for Mars human habitat ... [https://www.sciencedirect.com/science/article/abs/pii/S2214552425000215]