Bioregenerative Life Support: Plant Cultivation Systems for Closed Ecological Environments in Space Exploration and Biomedical Research
This article provides a comprehensive analysis of Bioregenerative Life Support Systems (BLSS) for closed ecological environments, targeting researchers and scientists in aerospace and biomedical fields.
Bioregenerative Life Support: Plant Cultivation Systems for Closed Ecological Environments in Space Exploration and Biomedical Research
Abstract
This article provides a comprehensive analysis of Bioregenerative Life Support Systems (BLSS) for closed ecological environments, targeting researchers and scientists in aerospace and biomedical fields. It explores the foundational principles of using plants for air revitalization, water recycling, and food production, as demonstrated in terrestrial analogs like Biosphere 2. The scope covers advanced methodological approaches including hydroponics, controlled environment agriculture, and nutrient recycling from waste streams. It details troubleshooting for challenges such as microgravity effects on plant growth and system closure failures. Finally, the article presents validation frameworks and comparative analyses of different plant species and technological systems, highlighting their implications for sustainable long-duration space missions and controlled-environment research on Earth.
The Principles and Necessity of Bioregenerative Life Support
Closed Ecological Life Support Systems (CELSS) and BLSS
Closed Ecological Life Support Systems (CELSS) are scientific and engineering endeavors aimed at creating self-reliant environments that can support and maintain human life by recycling all necessary resources [1]. These systems are designed to be isolated and independent from Earth's ecosystem, meaning no physical matter needs to be added or removed for the system to maintain equilibrium, though energy exchange (e.g., sunlight) is permitted [1] [2]. The core idea is to create a regenerative environment that can provide 100% of life support—including organic food, fresh oxygen-rich air, and clean water—by continuously recycling the waste products generated by its inhabitants and the system itself [1] [3].
Bioregenerative Life Support Systems (BLSS) represent a further development of this concept, incorporating biological elements into what have historically been mostly abiotic life support systems [4]. A BLSS is a balanced ecological system, biotechnical in nature, consisting of a combination of human beings, plants, and microorganisms integrated with mechanical and physico-chemical hardware [5]. The purpose of incorporating biological elements is to enhance resource recovery, enable food production, and improve waste treatment solutions, which becomes increasingly critical for long-duration missions where resupply from Earth is not feasible [4] [5].
Table: Core Concepts and Definitions
| Term | Full Name | Core Objective | Key Characteristics |
|---|---|---|---|
| CELSS | Closed (or Controlled) Ecological Life Support System | Create a self-reliant, regenerative environment for human life [1] [2]. | Material closure; integration of biological and technological components; replication of ecological cycles [1] [6]. |
| BLSS | Bioregenerative Life Support System | Incorporate biological elements to regenerate resources and produce food [4] [5]. | Based on ecological networks with trophic connections; includes producers, consumers, and degraders [4]. |
System Components and Ecological Principles
A fully functional CELSS/BLSS is comprised of interconnected subsystems that manage the core flows of matter and energy. These subsystems are characterized by fundamental human physiological needs and inherently cross traditional boundaries because the natural cycles they manage (e.g., carbon, oxygen, water) are deeply intertwined [1].
Core Subsystems
- Air Subsystem: This subsystem is responsible for storing and maintaining the atmospheric composition, pressure, and trace constituents. Its primary bioregenerative function is the removal of CO₂ and the generation of O₂ through photosynthesis performed by plants, microalgae, or photosynthetic bacteria [1] [2].
- Water Subsystem: This component collects wastewater from all sources (humans, plants, condensate), recovers potable water, and distributes it for consumption and hygiene. Plants contribute to water purification through evapotranspiration, and the system often integrates technological processes for filtration and recycling [1] [4].
- Food Subsystem: This subsystem involves the stabilization, storage, and processing of agricultural products into ready-to-eat food. It receives harvested biomass from the plant compartment and is central to providing nutritional and psychological benefits to the crew [1] [4].
- Waste Management Subsystem: This subsystem processes solid and liquid human waste, as well as inedible plant biomass. It relies on microorganisms (bacteria, fungi) and composting to break down waste into nutrients that can be recycled back to the plant compartment [4] [2].
Trophic Compartments
Ecologically, a BLSS is structured around three main types of compartments that form a web of trophic connections [4]:
- Producers: Organisms such as higher plants, microalgae, and photosynthetic bacteria that use light energy to convert CO₂ and water into food and oxygen.
- Consumers: The crew members who consume the producers for food, water, and oxygen, and in turn produce waste CO₂, urine, and feces.
- Degraders and Recyclers: Microorganisms (e.g., fermentative and nitrifying bacteria) that break down and process consumer and plant wastes into forms that can be reused by the producers.
Figure 1: Material and Energy Flow in a BLSS. This diagram illustrates the fundamental trophic compartments and the cyclical exchange of resources that characterizes a Bioregenerative Life Support System.
Quantitative Data for System Sizing
Designing a CELSS/BLSS requires careful consideration of the physical space needed to support human life, particularly for food production. The required plant growth area is a critical parameter that directly impacts the mass, volume, and energy requirements of a habitat.
Table: Estimated Plant Growth Area Requirements per Person [1]
| Source | Estimated Required Area (m² per person) |
|---|---|
| Gitalson | 14 |
| Bios-3 | 56 |
| Cullingford & Schwatekopf | 20 - 30 |
| Bugsbee & Salisbury | 13 - 50 |
| Oleson & Olson | 56.9 |
| MacElroy & Averner | 8 - 20 |
| Eckhart | 15 - 20 |
| Hoff | 24 |
| Vasilyew | 15 |
The variation in these estimates stems from differences in plant species, growth conditions (e.g., light intensity, CO₂ level), and the extent of closure targeted. For instance, some figures focus primarily on food production, while others also account for oxygen production and air filtration. Research at NASA Ames has demonstrated that all air, water, and food for one person can be produced in a highly engineered 16m x 16m (256 m²) space under optimal conditions with controlled lighting and nutrients [1].
For different mission scenarios, the selection of plant species and the design of the cultivation system must be adapted [4]:
- Short-duration missions (e.g., LEO): Focus on fast-growing, low-volume species with high nutritive value, such as leafy greens (lettuce, kale), microgreens, and dwarf cultivars of tomato. These systems primarily supplement prepackaged food and provide psychological benefits.
- Long-duration missions & planetary outposts: Require staple crops (wheat, potato, rice, soy) to provide carbohydrates, proteins, and fats. Systems must contribute substantially to resource recycling and require larger growing areas.
Experimental Protocols for Closed Ecosystem Research
Protocol: Establishing and Monitoring a Sealed Mini-Ecosystem
This protocol is adapted from ground-based experiments with sealed "Ecosphere" systems, which are designed to study the dynamics of closed ecosystems and serve as testbeds for space life-support technologies [7].
1. Objective: To create a small-scale, sealed ecological system to study plant survival, oxygen production, microbial community dynamics, and overall system stability over an extended period.
2. Materials:
- Sealed Container: A glass container with an airtight seal (e.g., using melted rubber or silicone) [7].
- Growth Medium: Nutrient-rich soil collected from a natural environment to introduce a diverse microbial community. Alternatively, simulated planetary soils (e.g., lunar or asteroid regolith simulants) can be tested [7].
- Water: Dechlorinated water.
- Biological Components:
- Seeds of a leguminous plant (e.g., clover, which can fix nitrogen via root nodules) [7].
- Naturally derived microbes present in the soil.
- Environmental Chamber or Location: A location with stable, adequate light (natural sunlight or LED growth lights) and temperature control [7].
3. Methodology: 1. Assembly: - Add a layer of gravel or similar material to the bottom of the container for drainage, if a groundwater layer is not part of the design. - Introduce the growth medium (soil or regolith simulant) to the container. - Sow the plant seeds evenly on the surface of the growth medium and cover lightly. - Slowly add water to moisten the growth medium without creating waterlogged conditions. - Seal the container airtight. 2. Experimental Groups: - Closed System with Groundwater Layer: A dedicated aquifer layer below the soil to provide a stable moisture source [7]. - Closed System without Groundwater Layer: Relies solely on initial water content [7]. - Open System Control: An identical, unsealed container for comparison. 3. Data Collection and Monitoring: - Plant Growth Metrics: Regularly measure and record plant germination rate, survival rate, height, leaf size, and overall health [7]. - Imaging: Document system appearance periodically to track visual changes, including plant growth and microbial (e.g., Cyanobacteria) colonization [7]. - Gas Composition: If possible, use gas sensors to monitor internal O₂ and CO₂ concentrations non-invasively. - Microbial Analysis: At the end of the experiment, collect samples from the growth medium for metagenomic analysis to characterize the microbial community structure [7].
4. Key Considerations:
- The presence of a groundwater layer has been proven critical for long-term plant survival in closed systems, acting as a buffer and stable water source [7].
- Even with adequate water, plant growth in closed systems may be stunted compared to open controls, potentially due to the accumulation of plant hormones like ethylene or other closed-system effects [7].
- The proliferation of Cyanobacteria and other microbes is a common and important observation, as they contribute to oxygen production and nutrient cycling [7].
Protocol: Hydroponic Cultivation of Salad Crops for BLSS
This protocol outlines the methodology for growing leafy greens in a controlled hydroponic environment, representative of a "salad machine" for short-duration missions [4].
1. Objective: To reliably produce fresh, nutritious leafy greens for crew consumption in a space-efficient hydroponic system, contributing to dietary variety and psychological well-being.
2. Materials:
- Growth Chamber: An enclosed chamber with controlled environmental parameters (temperature, humidity, CO₂) [4].
- Lighting System: LED arrays capable of providing a light intensity of 200-300 µmol m⁻² s⁻¹ PAR with a photoperiod of 16-18 hours light / 6-8 hours dark [4].
- Hydroponic System: A nutrient film technique (NFT) or deep-water culture (DWC) system.
- Nutrient Solution: A standardized, pH-balanced hydroponic nutrient solution containing all essential macro and micronutrients.
- Plant Materials: Seeds of leafy greens (e.g., lettuce Lactuca sativa, kale, Mizuna).
- Data Logging Equipment: Sensors for pH, electrical conductivity (EC) of the nutrient solution, air temperature, and humidity.
3. Methodology: 1. System Preparation: - Sterilize the hydroponic system and growth chamber to prevent algal and microbial contamination. - Prepare the nutrient solution according to the manufacturer's instructions and adjust the pH to 5.8-6.0. - Circulate the nutrient solution in the system to stabilize temperature and pH. 2. Plant Initiation: - Germinate seeds in sterile rockwool or oasis cubes under mild light. - Transfer seedlings to the hydroponic system once the first true leaves have developed. 3. Cultivation Management: - Environmental Control: Maintain air temperature at 22-25°C, relative humidity at 60-70%, and CO₂ concentration at 1000-1200 ppm. - Nutrient Management: Monitor and adjust the pH and EC of the nutrient solution daily. Top up with water and replenish nutrients as needed. Replace the entire solution weekly. - Pollination: For fruiting crops, manual pollination may be required in the absence of insect pollinators. 4. Harvest and Post-Harvest: - Harvest leaves or whole plants at maturity, typically 4-6 weeks after transplanting for leafy greens. - Weigh and record the fresh mass yield. - Perform nutritional analysis (e.g., vitamin C, antioxidants) on a subset of samples.
The Scientist's Toolkit: Key Research Reagent Solutions
Table: Essential Materials for CELSS/BLSS Plant Research
| Reagent/Material | Function in Research | Application Example |
|---|---|---|
| Simulated Planetary Regolith | A terrestrial analog of lunar or Martian soil used to study plant growth and microbial interactions in extraterrestrial substrates [7]. | Testing the viability of using in-situ resources for plant cultivation in lunar or Martian habitats [7]. |
| Hydroponic Nutrient Solutions | A precisely formulated water-based solution containing all essential mineral nutrients required for plant growth, excluding soil [4]. | Cultivating crops in controlled environments where soil is impractical, such as in space stations or planetary habitats [4]. |
| Specific Bacterial and Microbial Consortia | Defined communities of microorganisms (e.g., nitrifying bacteria, Cyanobacteria) used to initiate and stabilize waste processing and nutrient cycling [4] [7]. | Inoculating closed systems to enhance the breakdown of organic waste and to support the oxygen and carbon cycles [4] [7]. |
| LED Growth Lighting Systems | Provides photosynthetically active radiation (PAR) for plant growth. Allows for precise control over light intensity, spectrum, and photoperiod [4]. | Enabling plant cultivation in sun-limited environments like inside spacecraft or habitats, and during the long lunar night [4]. |
| Metagenomic Analysis Kits | Reagents and protocols for extracting, sequencing, and analyzing the collective genetic material of microbes recovered from an environment [7]. | Characterizing the microbial community within a closed ecosystem to understand its role in plant health and system stability [7]. |
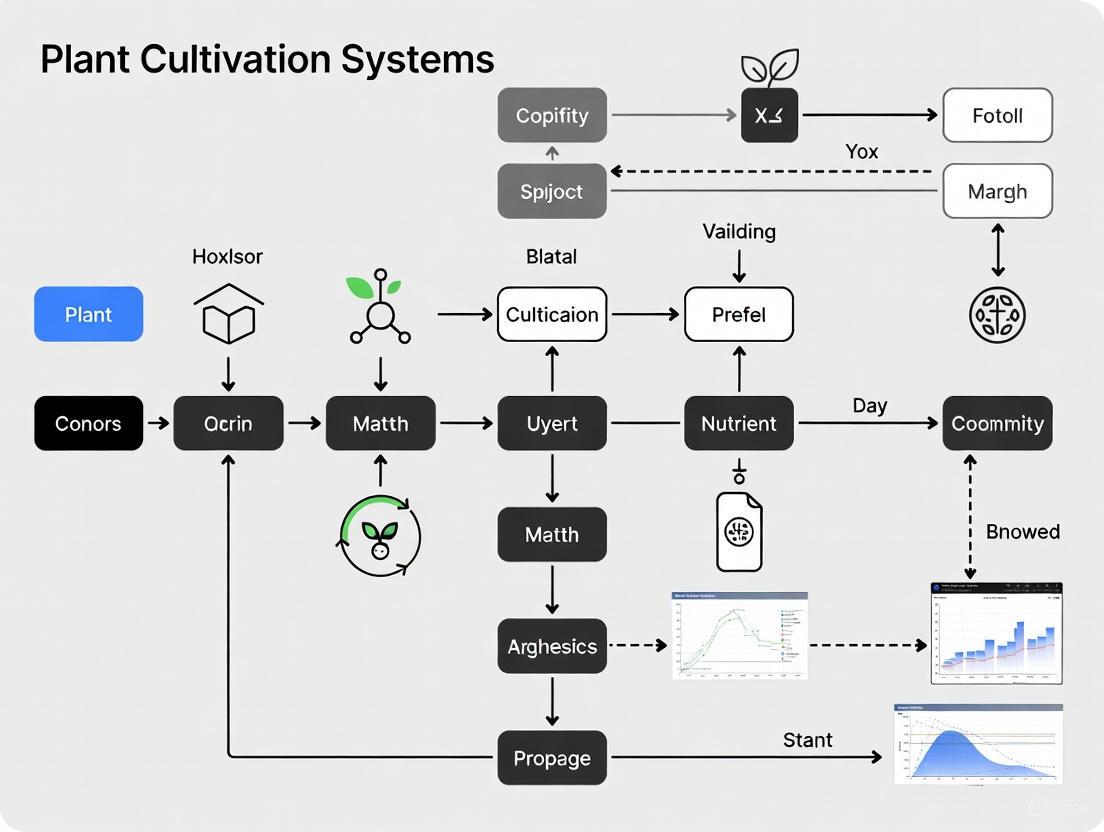
Figure 2: BLSS Research and Development Workflow. This diagram outlines the logical flow and iterative process for developing and testing Bioregenerative Life Support Systems, from initial concept to integrated system testing.
Table 1: Performance Metrics of Biological Subsystems for CELSS
| Subsystem / Component | Oxygen Production Rate | CO2 Sequestration / Fixation Rate | Biomass / Food Production Yield | Water Usage / Recycling Efficiency | Key Metric Source |
|---|---|---|---|---|---|
| Algal Photobioreactor (Chlorella vulgaris) | 4–6 mmol O₂/L culture/h [8] [9] | Implied by O₂ production | Cell density: up to 10⁹ cells/mL [9] | Operates in aqueous medium; water recycled within system | Illumination: Internal fiber optic [9] |
| Higher Plant Hydroponics (Leafy Greens, e.g., Lettuce) | Contributes to air revitalization [4] | Consumes CO₂ for photosynthesis [4] [10] | Yield up to 20x per acre vs. soil [11] | Up to 90% less water than conventional farming [11] | Illumination: ~250 μmol m⁻² s⁻¹ PAR (LED) [10] |
| Staple Crops (e.g., Wheat, Potato) | Primary role in food production, contributes to O₂ [4] | Primary role in food production, consumes CO₂ [4] | Provides carbohydrates, proteins, fats [4] | Water is purified via plant transpiration [4] | Essential for long-duration, autonomous missions [4] |
| Closed-Loop Wastewater System | N/A | N/A | N/A | Can reduce water consumption by up to 80% [12] | Treats and reuses greywater and blackwater [12] |
Experimental Protocols
Protocol 1: Operation of a High-Density Algal Photobioreactor for Oxygen Production
Objective: To achieve efficient, continuous oxygen production and CO₂ sequestration using a controlled photobioreactor system with Chlorella vulgaris.
Materials:
- Organism: Chlorella vulgaris culture.
- Photobioreactor Vessel: Equipped with an internal optical illumination system (e.g., fiber optic light transmission) to ensure uniform light distribution [9].
- Medium Reservoir: Containing standard algal growth medium.
- Ultrafiltration Unit: For continuous medium exchange and cell concentration [9].
- Gas Exchange Module: Designed for gravity-independent operation to remove produced O₂ and introduce supplied CO₂ [9].
- Light Source: External to the reactor to prevent heat generation, providing an intensity suitable for ~450 μmol m⁻² s⁻¹ PAR [10].
- Monitoring Equipment: Dissolved O₂ sensor, pH probe, CO₂ flow meter.
Methodology:
- System Sterilization: Sterilize the photobioreactor vessel and all associated tubing and modules prior to inoculation.
- Inoculation and Batch Growth: Aseptically introduce the Chlorella vulgaris inoculum into the reactor. Initiate batch operation with continuous lighting and CO₂ supplementation (e.g., 0.5-5% in air). Monitor cell density until the late exponential growth phase.
- Transition to Continuous Operation: Once high cell density is approached, activate the ultrafiltration unit to continuously remove spent medium and add fresh medium at a controlled dilution rate. This maintains cells in a steady, high-growth state [9].
- Process Monitoring: Continuously monitor and record the dissolved oxygen concentration, pH, cell density (via optical density), and gas flow rates.
- Data Collection: Calculate the oxygen production rate from the dissolved O₂ data and the flow rates of influent and effluent gases. The system can be operated in this continuous mode for extended periods (e.g., >2 months) [8].
Visualization: Algal Photobioreactor Workflow
Protocol 2: Hydroponic Cultivation of Higher Plants for Food and Resource Regeneration
Objective: To reliably produce edible biomass, contribute to oxygen production, and aid in water purification through a controlled hydroponic system.
Materials:
- Plant Material: Seeds or seedlings of selected crops (e.g., lettuce, tomato, dwarf cultivars).
- Hydroponic System: Nutrient Film Technique (NFT) or Deep Water Culture (DWC) system [11].
- Growth Chamber: Enclosed environment with temperature, humidity, and CO₂ control.
- Lighting System: LED arrays capable of delivering species-specific PAR levels (e.g., 250 μmol m⁻² s⁻¹ for lettuce, 100 μmol m⁻² s⁻¹ for tomatoes) [10].
- Nutrient Solution: Standard Hoagland's solution or equivalent, with pH and Electrical Conductivity (EC) sensors.
- Support Substrate (optional): Inert medium such as rockwool, perlite, or coco peat [11] [10].
Methodology:
- Germination and Seedling Establishment: Germinate seeds in a sterile, inert starter plug. Provide moisture and light until seedlings develop true leaves.
- System Transfer: Transfer seedlings to the hydroponic system, ensuring roots are in contact with the nutrient solution.
- Environmental Control: Maintain a controlled environment:
- Nutrient Management: Continuously circulate the nutrient solution. Monitor and adjust pH (typically 5.5-6.5) and EC regularly to maintain optimal nutrient availability. Top up or replace the solution as needed.
- Harvest and Data Collection: Harvest edible biomass at maturity. Record fresh weight, dry weight, and yield. Collect transpired water from the air handling system for quality analysis and recycling.
Visualization: Hydroponic Cultivation Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CELSS Plant Cultivation Research
| Item | Function / Application in CELSS Research | Reference |
|---|---|---|
| Chlorella vulgaris | A model microalga for high-efficiency oxygen production and CO₂ sequestration in photobioreactors. | [8] [9] |
| Hoagland's Nutrient Solution | A standardized, complete nutrient mix for hydroponic cultivation of higher plants, providing essential macro and micronutrients. | [11] |
| LED Grow Lights | Energy-efficient, wavelength-specific light source for photosynthesis. Allows precise delivery of Photosynthetically Active Radiation (PAR). | [11] [10] |
| Inert Growth Substrates (Rockwool, Perlite) | Provide physical root support in hydroponic systems without introducing pathogens or altering nutrient chemistry. | [11] [10] |
| Ultrafiltration Membranes | Used in photobioreactors and water recycling systems to separate cells from spent medium and purify wastewater for reuse. | [9] |
| CO₂ Gas Cylinders | Used for environmental enrichment in plant growth chambers to boost photosynthetic rates and biomass yield. | [10] |
Closed ecological life support systems are critical for the future of long-duration human space exploration, enabling sustainability by regenerating air, water, and food. Research in this field bridges historical large-scale experiments with advanced current programs, creating a continuum of knowledge essential for developing bioregenerative life support. This application note synthesizes the experimental protocols and findings from foundational projects like Biosphere 2 with contemporary NASA initiatives, providing researchers with standardized methodologies and comparative data to advance the development of sustainable cultivation systems for space and terrestrial applications.
Historical Benchmark: The Biosphere 2 Experiment
Biosphere 2, constructed between 1987 and 1991, remains the largest closed ecological system ever created, covering 3.14 acres under a sealed glass and spaceframe structure [13]. This pioneering facility was designed to explore the viability of closed ecological systems to support and maintain human life in outer space as a substitute for Earth's biosphere ("Biosphere 1") [13].
Table 1: Biosphere 2 Technical Specifications and Biome Distribution
| Parameter | Specification | Notes |
|---|---|---|
| Total Area | 3.14 acres (12,700 m²) | Largest closed ecological system ever built [13] |
| Rainforest Biome | 1,900 m² | Simulated tropical ecosystem [13] |
| Ocean with Coral Reef | 850 m² | Included wave action and coral systems [13] |
| Mangrove Wetlands | 450 m² | Analogous to Florida Everglades [13] |
| Savanna Grassland | 1,300 m² | Seasonal biomass management required [13] |
| Fog Desert | 1,400 m² | Evolved into chaparral habitat [13] |
| Agricultural System | 2,500 m² | Provided food for human crew [13] |
| Human Habitat | Incorporated living spaces, labs, workshops | Supported crew of 8 [13] |
| Airtightness | Leak rate <10% per year | Patented sealing methods [13] |
The engineering infrastructure included variable volume structures called "lungs" to accommodate air expansion and contraction, while an independent energy center provided electricity and thermal control [13]. The life systems were designed to function with minimal external inputs, testing the principles of ecological self-organization and closed-loop material cycling.
Agricultural Protocol and Performance
The agricultural system within Biosphere 2 demonstrated remarkable productivity despite initial challenges. During the first closure mission (1991-1993), the system produced 83% of the total diet for the eight-person crew through intensive cultivation methods [13].
Table 2: Biosphere 2 Agricultural Production and Outcomes
| Parameter | Result | Significance |
|---|---|---|
| Food Self-Sufficiency | 83% of total diet | Demonstrated feasibility of closed-system agriculture [13] |
| Caloric Content | Low-calorie, nutrient-dense diet | Based on Roy Walford's longevity research [13] |
| Crops Cultivated | Bananas, papayas, sweet potatoes, beets, peanuts, beans, rice, wheat | Diverse nutritional sources [13] |
| Productivity Rate | 5x higher than efficient agrarian communities | Exceeded yields of Indonesian, Chinese, and Bangladeshi farming [13] |
| Crew Health Impact | 16% average weight loss initially, then stabilization | Improved health markers (cholesterol, blood pressure) [13] |
| Animal Systems | Pygmy goats, hens, Ossabaw dwarf pigs, tilapia | Integrated protein sources [13] |
| Soil Fertility | Maintained through recycling | Biofiltration of air simultaneously achieved [14] |
The agricultural protocol employed a "species-packing" strategy, introducing multiple species to ensure ecological function even if some species failed to thrive [13]. This approach acknowledged the inevitable learning curve in establishing new ecosystems and provided redundancy for critical functions.
Atmospheric Management Protocol
The Biosphere 2 experiment revealed critical challenges in atmospheric management that informed subsequent closed ecological system designs. During the first mission, oxygen levels declined at a rate of approximately 0.25% per month, eventually requiring injection of oxygen to maintain crew safety [13]. Research traced this oxygen loss to unexpected microbial respiration in the soils, which was consuming oxygen faster than the photosynthetic systems could regenerate it, while simultaneously the concrete structural materials were absorbing carbon dioxide, creating an imbalanced carbon cycle [13].
The second closure experiment in 1994 achieved significantly improved performance, reaching total food sufficiency and not requiring oxygen injection before the experiment concluded early for management reasons [13]. This improvement demonstrated the learning curve in operating closed ecological systems and the importance of system maturity.
Diagram 1: Atmospheric Management Challenge in Biosphere 2
Contemporary Space Agency Programs
NASA's Vegetable Production System (Veggie)
The Vegetable Production System (Veggie) represents NASA's current approach to space-based plant cultivation, residing aboard the International Space Station as a platform for studying plant growth in microgravity while supplementing astronaut nutrition and psychological well-being [15].
Veggie Protocol for Space-Based Cultivation:
- System Setup: The Veggie unit is approximately the size of carry-on luggage and typically accommodates six plants using a "pillow" system filled with clay-based growth media and fertilizer [15]
- Planting Method: Seeds are mounted on water-soluble strips that dissolve after planting, facilitating easier handling in microgravity [16]
- Watering Procedure: Crew members manually inject water into plant pillows using syringes, carefully monitoring to prevent both underwatering and overwatering [16]
- Lighting Regime: Banks of light emitting diodes (LEDs) produce a spectrum optimized for plant growth, typically appearing magenta pink due to emphasis on red and blue wavelengths that plants utilize most efficiently [15]
- Growth Monitoring: Regular observation and photographic documentation of plant development, with attention to morphological changes and potential issues like fungal growth [15]
- Harvest Procedure: Mature plants are harvested, with portions consumed by crew and samples returned to Earth for analysis of nutritional content and potential microbial contamination [15]
The Veggie system has successfully grown multiple crops including three types of lettuce, Chinese cabbage, mizuna mustard, red Russian kale, and zinnia flowers [15]. The psychological benefits of gardening in space have been consistently observed, with astronauts reporting the activity as a enjoyable reprieve from typical station duties [16].
Advanced Plant Habitat (APH) Protocol
The Advanced Plant Habitat provides a more sophisticated, automated growth chamber for plant research with extensive monitoring capabilities and environmental control [15].
APH Experimental Protocol:
- System Initialization: The enclosed, automated system self-checks cameras and over 180 sensors that maintain constant communication with ground control teams at Kennedy Space Center [15]
- Environmental Control: Automated systems manage water recovery and distribution, atmospheric content, moisture levels, and temperature without requiring daily crew intervention [15]
- Advanced Lighting: Expanded LED spectrum including red, green, blue, white, far red, and infrared to support diverse experimental needs and enable nighttime imaging [15]
- Sample Collection: Crew members collect plant samples at specified developmental stages, preserving them by freezing or chemical fixation for post-mission analysis [15]
- Gene Expression Analysis: Returned samples undergo RNA extraction and analysis to understand space effects on plant growth at genetic, protein, and metabolite levels [15]
A key research focus in APH experiments has been investigating the relationship between microgravity and plant lignin content. Researchers are examining whether plants genetically engineered for reduced lignin can survive and function normally in space, potentially improving nutrient absorption for human consumption and simplifying composting of plant waste [15].
Plant Health Monitoring Protocol
Understanding and maintaining plant health in closed systems requires specialized monitoring approaches, as exemplified by the Biological Research in Canisters (BRIC) system and related investigations [15].
Plant Immune Response Assessment Protocol:
- Plant Material Preparation: Arabidopsis thaliana specimens are grown for 10 days in the BRIC-LED system or similar growth apparatus [15]
- Immune Challenge Application: Researchers apply a harmless solution containing "flag-22" (a conserved 22-amino acid peptide from bacterial flagella) to trick plants into activating defense systems as if under pathogen attack [15]
- Response Fixation: One hour post-elicitation, when defense responses are fully active, plants are treated with chemical fixative to halt all biological processes while preserving response state [15]
- Cryopreservation: Fixed samples are transferred to deep freeze conditions to maintain molecular integrity until analysis [15]
- Transcriptomic Analysis: RNA is extracted from returned samples and analyzed for expression patterns of defense-related genes to evaluate immune system functionality in space conditions [15]
This protocol has revealed that space-grown plants exhibit patterns suggesting increased oxidative stress and altered immune responses, potentially compromising their ability to fight infections [15]. These findings were corroborated by anecdotal evidence from zinnia plants in the Veggie system that developed fungal growth when conditions were suboptimal [15].
Phytoremediation Applications for Air Revitalization
NASA Biohome and Phytoremediation Protocol
Research conducted in NASA's Biohome facility demonstrated the capacity of plant-soil systems to remove volatile organic compounds (VOCs) from enclosed atmospheres, establishing the foundation for phytoremediation applications in closed ecological life support systems [14].
Phytoremediation Assessment Protocol:
- Chamber Setup: Establish sealed test chambers containing synthetic materials known to off-gas target VOCs (e.g., formaldehyde from urea-formaldehyde resins) [14]
- Plant Introduction: Introduce test plant species (e.g., lady palm - Rhapis excelsa) into experimental chambers while maintaining control chambers without plants [14]
- Continuous Monitoring: Use mass spectrometer/gas chromatograph systems to track VOC concentrations over time under various temperature conditions [14]
- Mechanism Isolation: Compare removal efficiency between plants with exposed potting soil versus those with soil covered by sterilized sand to distinguish leaf versus root microbe contributions [14]
- Adaptation Assessment: Evaluate changes in removal efficiency over extended exposure periods to measure metabolic adaptation of plant-microbe systems [14]
This research demonstrated that approximately 50-65% of VOC removal could be attributed to root and soil microbes, with plants increasingly effective at chemical removal as exposure time increased due to microbial adaptation [14]. Plants with higher transpiration rates generally showed greater purification capacity due to enhanced atmospheric gas transport to root zones [14].
Diagram 2: Phytoremediation Pathway for VOC Removal
Comparative Performance of Growth Substrates
Research has evaluated different growth substrates for their phytoremediation effectiveness in closed systems, with significant implications for system design in space applications.
Table 3: Growth Substrate Efficacy for Phytoremediation
| Growth Substrate | Removal Efficiency | Advantages | Disadvantages |
|---|---|---|---|
| Commercial Potting Soil | Baseline efficiency | Natural microbial communities; established protocols | Potential for mold; maintenance intensive [14] |
| Activated Carbon Mixture | Enhanced efficiency | Improved chemical adsorption; stable structure | Requires specialized formulation; higher cost [14] |
| Hydroculture Systems | 30-50% more effective than potting soil | Reduced mold risk; minimal maintenance; water efficient | Different nutrient management; system adaptation period [14] |
| Sterilized Sand Cover | 35-50% reduction vs. exposed soil | Limits microbial contribution; useful for mechanism studies | Not suitable for production systems [14] |
The integration of plant-based air revitalization with food production systems represents a promising multi-functional approach for closed ecological life support systems, potentially reducing reliance on energy-intensive mechanical filtration systems while providing nutritional and psychological benefits [14].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagents and Materials for Closed System Plant Research
| Reagent/Material | Function/Application | Research Context |
|---|---|---|
| Plant Growth Pillows | Clay-based growth media with controlled-release fertilizer contained in fabric modules | NASA Veggie system; provides root support and nutrient delivery in microgravity [15] |
| Rhapis excelsa (Lady Palm) | Model species for phytoremediation studies; effective at formaldehyde removal | NASA Biohome research; demonstrates plant VOC removal capabilities [14] |
| Flag-22 Peptide | Conserved 22-amino acid flagellar peptide used to elicit plant immune responses | BRIC-LED experiments; assesses plant defense system functionality in space [15] |
| Arabidopsis thaliana | Model plant organism for genetic and physiological studies | Advanced Plant Habitat; understanding space effects on plant gene expression [15] [17] |
| Clay-Based Growth Media | Inorganic substrate for plant growth; minimizes microbial complications | Veggie system; provides stable root zone environment in microgravity [15] |
| LED Lighting Systems | Specific wavelength delivery optimized for plant photosynthesis and research | Veggie and APH systems; energy-efficient growth lighting with spectral control [15] |
| Chemical Fixatives | Preservation of biological samples for post-mission analysis | APH and BRIC experiments; maintains molecular state for Earth analysis [15] |
The historical precedents established by Biosphere 2, combined with contemporary space agency research programs, provide a robust foundation for developing closed ecological life support systems. The experimental protocols and quantitative data synthesized in this application note demonstrate the progressive refinement of controlled environment agriculture and atmospheric management techniques essential for long-duration space missions. As research continues through platforms like the Advanced Plant Habitat and Veggie system, the integration of plant cultivation into life support architectures continues to evolve, offering sustainable solutions for human exploration beyond Earth while providing applications for terrestrial sustainability challenges.
Closed Ecological Life Support Systems (CELSS) are engineered biological systems designed to sustain human life in space by replicating Earth's natural recycling processes for air, water, and food [18]. These systems represent a critical technological frontier for extended-duration missions to Mars and beyond, where resupply from Earth becomes impractical. The core innovation of CELSS lies in creating a materially closed loop where waste products are continuously regenerated into life-supporting resources [19]. This approach stands in stark contrast to traditional "open-loop" life support that depends on consumables from Earth.
The fundamental challenge in CELSS operation involves achieving and maintaining a balanced state where carbon (C), nitrogen (N), and other essential elements are efficiently recycled without accumulation of waste or depletion of resources. In these systems, plants serve dual roles: they produce food and oxygen through photosynthesis while consuming carbon dioxide and other waste products [18]. Materially closed microbial ecosystems further enhance this recycling, functioning as reliable, self-sufficient systems for processing carbon compounds under constant energy input [19]. The successful integration of these biological and technological components is paramount for creating sustainable habitats beyond Earth, making research into carbon and nitrogen cycling a critical scientific endeavor for the future of space exploration.
Quantitative Data on Carbon and Nitrogen Cycling
Carbon to Nitrogen Ratios and Nutrient Cycling
Table 1: Carbon to Nitrogen (C:N) Ratios and Their Impacts on Nutrient Cycling
| Material/Parameter | C:N Ratio | Impact on Decomposition and Nutrient Availability |
|---|---|---|
| Soil Microorganisms | 24:1 [20] | Optimal ratio for microbial growth and function. |
| Materials with High C:N | > 24:1 [20] | Cause temporary nitrogen immobilization; microbes scavenge soil N, reducing availability for plants. |
| Materials with Low C:N | < 24:1 [20] | Result in net nitrogen mineralization; surplus N is released, increasing availability for plants. |
| Management Strategy | Combine high and low C:N materials [20] | Incorporating low C:N cover crops enhances N availability from high C:N residues. |
Carbon-Nitrogen Interaction under Elevated CO₂
Table 2: Effects of Elevated CO₂ on Carbon-Nitrogen Coupling in Plants
| Process/Parameter | Effect under Elevated CO₂ | Consequence for Agroecosystems |
|---|---|---|
| Photosynthesis & Biomass | Promoted/Increased [20] | Augments nitrogen requirement, potentially diluting nutrient concentration in plant tissues. |
| Crop Nutritional Quality | Decreased [20] | Triggered nutrient dilution effects can reduce the nutritional value of food crops. |
| Nitrogen Limitation | Amplified [20] | Modified belowground processes, altering the ecosystem's nitrogen cycle. |
| Greenhouse Gas Processes | Impacted [20] | Can lead to enhanced nitrous oxide (N₂O) emissions, affecting the system's environmental impact. |
Experimental Protocols for CELSS Research
Protocol 1: Assessing Nutrient Release from Crop Residues
Objective: To quantify the rate of decomposition and nitrogen mineralization/immobilization from various plant residues with different C:N ratios in a controlled, soil-like substrate.
Materials:
- Plant Residues: Samples of wheat straw (high C:N), clover shoots (low C:N), and other relevant crop wastes.
- Growth Substrate: A standardized, inert substrate (e.g., calcined clay) inoculated with a diverse microbial consortium.
- Containers: Sealed, aerated microcosm units (≥ 1 L volume).
- Analytical Equipment: Elemental analyzer for C and N content, pH meter, soil nutrient extraction kits (e.g., for ammonium and nitrate).
Methodology:
- Preparation: Oven-dry (60°C) and finely grind plant residue samples. Determine the initial C and N content to calculate the C:N ratio [20].
- Incubation Setup: Thoroughly mix a known weight of each residue type with the pre-moistened growth substrate in separate microcosms. Include a substrate-only control.
- Monitoring: Incubate microcosms under constant temperature (e.g., 25°C). Maintain moisture at field capacity. Destructively harvest replicate microcosms at 0, 1, 2, 4, 8, and 12 weeks.
- Analysis: For each harvest, extract and analyze the substrate for inorganic nitrogen (NH₄⁺ and NO₃⁻) concentrations. Measure total C and N at selected intervals to track mass balance.
- Data Interpretation: Plot inorganic N over time. Net mineralization is indicated by a steady increase in NH₄⁺ and NO₃⁻, while immobilization is indicated by a decrease or stable, low levels compared to the control.
Protocol 2: System-Level Integration of Crop-Livestock Synergies
Objective: To evaluate the efficiency of a closed-loop recycling system integrating plant cultivation and organic waste (manure) production for nutrient recovery.
Materials:
- Experimental System: Sealed, modular chambers for plant growth and waste processing.
- Crop Units: Hydroponic or aeroponic systems for high-yield crops (e.g., lettuce, wheat).
- Waste Processing Units: Bioreactors for aerobic composting or anaerobic digestion of simulated or real organic waste.
- Monitoring Sensors: CO₂ sensors, O₂ sensors, dissolved oxygen and pH probes for liquid systems.
- Analysis Kits: For quantifying total nitrogen, ammonium, nitrate, and phosphate in solid and liquid samples.
Methodology:
- System Baseline: Establish plant growth units and characterize the nutrient content of the initial growth solution. Characterize the initial C:N ratio of the organic waste input [21] [20].
- Integration Loop: Harvest plant residues and introduce them into the waste processing unit. Subsequently, apply the processed effluent (compost tea or digestate) from the waste unit back to the plant growth system as a nutrient supplement.
- Data Collection: Continuously monitor atmospheric O₂ and CO₂ levels. Regularly sample and analyze the nutrient solutions and solid wastes for key elements (C, N, P, K) and pH.
- Performance Metrics: Calculate mass balances for carbon and nitrogen. Determine closure efficiency as (1 - [mass of supplemental input / total system mass]) × 100% for each element. Monitor plant growth rates and biomass yield as indicators of system health and productivity [21] [18].
Signaling and Metabolic Pathways in Carbon-Nitrogen Interaction
The coordination of carbon (C) and nitrogen (N) metabolism is a fundamental requirement for plant growth and is governed by a complex signaling network. Plants must balance their investments in carbon skeletons (from photosynthesis) and nitrogen assimilation to synthesize amino acids, nucleotides, and chlorophyll [22]. This integration occurs at multiple levels, from metabolism to gene expression, and is regulated by environmental factors such as light, CO₂ concentration, and nutrient availability.
Diagram 1: C-N Metabolic and Signaling Integration in C3 Plants.
Research Reagent Solutions and Essential Materials
Table 3: Key Research Reagents and Materials for CELSS Experimentation
| Item | Function/Application |
|---|---|
| Hydroponic/Aeroponic Growth Systems | Enables soil-free plant cultivation for precise nutrient control and space-efficient food production in CELSS prototypes [18]. |
| Bioreactors (e.g., for MELiSSA Project) | Mimics self-sustaining ecosystems using compartments of algae, bacteria, and plants to close the life support loop [18]. |
| ¹⁵N-Labeled Fertilizers | Tracer compounds used to quantify nitrogen use efficiency (NUE) and track the fate of N from different sources (e.g., urine, feces, mineral fertilizer) through the ecosystem [20]. |
| Molecular Sieves & Catalytic Converters | Key components of air purification systems for removing CO₂ and regenerating O₂ through chemical processes [18]. |
| Microbial Consortia | Engineered communities of bacteria and fungi crucial for decomposing waste materials and facilitating nutrient recycling in closed systems [19]. |
| Nutrient Solution Kits | Pre-mixed or customizable solutions containing essential macro and micronutrients (e.g., N, P, K, Ca, Mg, Fe) for plant growth in hydroponic systems. |
| CO₂ and O₂ Sensors | Critical for real-time monitoring of atmospheric gas composition within the closed system to ensure balance between plant and human needs [18]. |
Integrated Workflow for CELSS Experimentation
A systematic approach is required to move from fundamental research to the implementation of a fully functional Closed Ecological Life Support System. The workflow integrates plant physiology, waste processing, and system-level engineering, with continuous monitoring and balancing of carbon and nitrogen flows being the central theme.
Diagram 2: CELSS Development and Testing Workflow.
The Strategic Geopolitical Landscape of BLSS Development
The development of Bio-regenerative Life Support Systems (BLSS) represents a critical strategic capability for sustained human exploration beyond low-Earth orbit. Within a BLSS, the cultivation of higher plants is crucial as they contribute to all major functional aspects, including food production, carbon dioxide reduction, oxygen production, water recycling, and waste management [23]. These systems aim to decrease resupply mass by regenerating essential resources for humans through biological processes, making them indispensable for long-duration space missions and eventual habitats on the Moon and Mars [23]. The geopolitical landscape of 2025, characterized by escalating trade tensions and technological competition, directly influences the global collaboration and resource allocation necessary for advancing these complex biological systems [24].
The strategic value of BLSS technology extends beyond space exploration, offering potential applications for terrestrial sustainability and environmental management. However, global supply chain vulnerabilities, resource nationalism, and technology transfer restrictions create significant headwinds for multinational research initiatives. Recent analyses indicate that unmanaged decoupling in technology and trade between major powers could severely impact global research collaborations, affecting supply chains for critical components and biological materials essential for BLSS development [24]. Furthermore, Russia's unpredictable geopolitical maneuvers add another layer of uncertainty, potentially disrupting international scientific partnerships and data sharing agreements that have historically advanced life support research [24].
Geopolitical Risk Assessment and Strategic Implications
Analysis of Critical Geopolitical Factors
The successful development of BLSS requires navigating an increasingly complex geopolitical terrain. Supply chain resilience emerges as a paramount concern, particularly for specialized components and biological specimens that may be subject to export controls or sourcing limitations from politically volatile regions. According to recent risk assessments, companies operating in or dependent on regions influenced by geopolitical instability may encounter operational risks, including supply chain interruptions and compliance issues with international regulations [24]. The semiconductor supply crisis recently highlighted how tensions around strategic regions could ripple across industries relying on these critical components, offering a cautionary tale for BLSS development dependencies [24].
International collaboration frameworks for space exploration face increasing strain as terrestrial geopolitical tensions escalate. The redistribution of global influence and emerging multipolar world order creates both challenges and opportunities for BLSS development. While historical space exploration has benefited from international cooperation, current trends suggest that future BLSS initiatives may develop along parallel, competing technological pathways rather than through unified global efforts. Research indicates that only 30% of CEOs have full visibility into their company's exposure to political risk across operations, markets and suppliers, highlighting the preparedness gap that also affects complex research initiatives like BLSS development [25].
Strategic Mitigation Approaches
Table: Geopolitical Risk Mitigation Strategies for BLSS Development Programs
| Risk Category | Impact on BLSS Development | Mitigation Strategy |
|---|---|---|
| Trade Disruptions | Delay in critical component acquisition; increased costs | Supplier diversification across multiple geopolitical regions; maintain strategic inventory of high-risk items |
| Technology Transfer Restrictions | Limited access to advanced environmental control systems; restricted data sharing | Develop modular architecture with swappable subsystems; invest in dual-use technologies with terrestrial applications |
| Political Instability in Partner Nations | Disruption of international research collaborations; funding uncertainty | Establish distributed research facilities across multiple jurisdictions; create contingency partnerships |
| Export Controls on Biological Materials | Restrictions on plant genetic resources; limited access to microbial communities | Develop in-situ resource utilization capabilities; establish protected genetic repositories for essential species |
Proactive scenario planning plays a pivotal role in preparing BLSS development for potential geopolitical disruptions. By envisioning various future scenarios, research organizations can identify vulnerabilities and develop contingency plans [24]. This forward-thinking approach enables institutions to anticipate challenges and respond effectively when uncertainties arise. Sigma7's holistic methodology integrates scenario planning with business strategy, ensuring a comprehensive approach to risk management that can be adapted for BLSS research initiatives [24].
Building research supply chain resilience represents another critical strategy. Diversifying suppliers of critical components avoids reliance on high-risk regions [24]. Similarly, establishing redundant capabilities for essential biological systems through distributed research networks enhances stability when geopolitical events disrupt particular nodes. The implementation of robust risk management plans, such as developing flexible collaboration strategies, helps sustain operations amidst uncertainty [24].
Experimental Protocols for BLSS Plant Cultivation Research
Protocol: Evaluation of Plant Growth Orientation Under Altered Environmental Conditions
Purpose and Scope
This protocol describes a methodology to investigate growth patterns and morphological development of candidate plant species under orientation conditions that simulate altered gravity environments. The research aims to confirm the possibility of plant culture under microgravity in space by examining growth rates and morphological characteristics of plants cultivated in inverted orientations [26].
Materials and Equipment
- Plant Species: Lettuce (Lactuca sativa) and turnips (Brassica rapa)
- Growth Apparatus: Custom-designed growth chambers with upper and lower rooting beds arranged around fluorescent lamp arrays
- Lighting System: Fluorescent lamps providing Photosynthetic Photon Flux Density (PPFD) of approximately 300 μE/m²/s at plant canopy level
- Environmental Monitoring: CO₂ and O₂ sensors for gas exchange measurements
- Data Collection Equipment: Digital scales for fresh weight measurement, digital calipers for morphological measurements
Procedure
- Experimental Setup: Configure growth chambers with rooting beds positioned both above and below the fluorescent lamp arrays [26].
- Plant Establishment: Germinate test species under standardized conditions and transplant to both upper and lower rooting beds at equivalent developmental stages.
- Orientation Treatment: Grow plants normally on the lower bed and upside down on the upper bed [26].
- Environmental Maintenance: Maintain consistent PPFD of 300 μE/m²/s at the plant canopy through regular adjustment of lamp positioning [26].
- Data Collection:
- Record fresh weights of aerial tissues (30 days post-treatment for lettuce; 25 days for turnips)
- Document morphological characteristics including stem orientation, leaf expansion patterns, and root development
- Measure CO₂ absorption and O₂ release rates to estimate gas exchange capabilities [26]
- Data Analysis: Compare growth rates and morphological development between standard and inverted orientations using appropriate statistical methods.
Troubleshooting and Notes
- Ensure consistent moisture levels in both upper and lower rooting beds to prevent hydration disparities
- Regularly rotate plant positions within treatments to minimize edge effects
- The PPFD of approximately 300 μE/m²/s has been established as sufficient to ensure vegetable crops such as lettuce and turnips grow normally toward the light source regardless of gravity direction [26]
Protocol: High-Density Plant Cultivation for Spatial Efficiency
Purpose and Scope
This protocol outlines a methodology for maximizing plant production within limited spatial confines, a critical consideration for BLSS applications in space-constrained environments. The approach utilizes vertically arranged growth panels to increase cultivation density without compromising individual plant health or productivity [26].
Materials and Equipment
- Growth Structure: Modular units composed of vertical panels supporting rooting beds arranged on two sides of fluorescent lamp arrays [26]
- Irrigation System: Automated nutrient delivery system with individual zone control
- Support Materials: Appropriate substrate or hydroponic growing medium
- Environmental Control: Capability to maintain optimal temperature, humidity, and atmospheric composition
Procedure
- System Assembly: Construct vertical growth units with rooting beds on both sides of central lighting arrays [26].
- Planting Configuration: Establish crops at a planting density of 25 plants/m² on each vertical surface [26].
- Spatial Optimization: Position plants to grow horizontally toward the lamps, maximizing light interception while minimizing mutual shading [26].
- System Monitoring:
- Track growth rates and development stages across different vertical positions
- Measure photosynthetic parameters at various canopy levels
- Monitor resource utilization (water, nutrients) per unit of biomass produced
- Productivity Assessment: Calculate total biomass production per unit floor area and compare against traditional single-level cultivation approaches.
Expected Outcomes
Based on established parameters, each vertical unit should support approximately 200 plants, achieving a productivity of 100 plants per m² of floor area while maintaining healthy growth and development [26].
Protocol: Atmospheric Purification Capacity of Plant Systems
Purpose and Scope
This protocol describes methods to quantify the capacity of mixed plant canopies to remove toxic impurities from closed atmospheric systems, specifically evaluating the processing of sulfur dioxide (SO₂) as a model contaminant [27]. The research examines both passive absorption by biologically inactive system components and active metabolic processing by plants.
Materials and Equipment
- Test Chamber: Sealed environment with controlled atmospheric conditions
- Contaminant Introduction System: Precision equipment for pulsing and continuous supply of test gases
- Analytical Instrumentation: Gas chromatographs or specific electrodes for SO₂ quantification
- Photosynthesis Measurement: Equipment for monitoring photosynthetic rates under contaminant exposure
Procedure
- System Establishment: Create mixed canopies of agricultural plants within sealed test chambers at defined planting densities [27].
- Contaminant Exposure:
- Pulse Introduction: Administer discrete SO₂ injections at varying concentrations
- Continuous Introduction: Maintain constant SO₂ infusion at controlled rates [27]
- Absorption Kinetics:
- Monitor atmospheric SO₂ concentrations over time following pulse introduction
- Measure steady-state SO₂ levels during continuous introduction
- Record intensity of visible photosynthesis of plant communities throughout exposure periods [27]
- Data Modeling:
- Characterize passive absorption using kinetics with saturation models
- Describe active absorption using enzymatic kinetics equations [27]
- Threshold Determination: Identify contaminant introduction rates that do not essentially damage biological system components [27].
Interpretation Guidelines
The parameters of the mathematical model describing removal of toxic substances from closed ecosystem atmosphere can be used to characterize integrally the so-called absorptive properties of the system [27].
Quantitative Performance Metrics for BLSS Subsystems
Table: Quantitative Growth and Gas Exchange Metrics for BLSS Candidate Species
| Plant Species | Growth Period (days) | Biomass Production (g fresh weight/plant) | CO₂ Absorption Rate | O₂ Release Rate | Light Requirement (PPFD) |
|---|---|---|---|---|---|
| Lettuce | 30 | 42-46 (aerial tissues) | Documented in study [26] | Documented in study [26] | ~300 μE/m²/s [26] |
| Turnips | 25 | 66 (leaves), 42 (swollen root) | Documented in study [26] | Documented in study [26] | ~300 μE/m²/s [26] |
Table: System-Level Performance Parameters for BLSS Implementation
| Parameter | Specification | Significance |
|---|---|---|
| Planting Density | 25 plants/m² [26] | Determines spatial efficiency of food production |
| Vertical Farming Capacity | 200 plants/unit; 100 plants/m² floor area [26] | Enables high-density cultivation in space-constrained environments |
| Toxic Substance Removal | Combination of passive (saturation kinetics) and active (enzymatic kinetics) processing [27] | Critical for maintaining atmospheric quality in closed systems |
| Orientation Independence | Normal growth regardless of gravity vector when sufficient light provided [26] | Enables flexible system architecture in microgravity or partial gravity environments |
Visualization of BLSS Functional Relationships
BLSS System Interdependencies Diagram
BLSS Experimental Workflow Diagram
The Scientist's Toolkit: Essential Research Materials for BLSS Investigations
Table: Critical Research Reagents and Materials for BLSS Experimentation
| Item Category | Specific Examples | Research Function | Geopolitical Considerations |
|---|---|---|---|
| Plant Genetic Resources | Lettuce (Lactuca sativa), Turnips (Brassica rapa), Other candidate species [26] | Primary producers for food, oxygen regeneration, and carbon sequestration | Diversified sourcing to mitigate single-point failure risks; potential export restrictions on specialized cultivars |
| Atmospheric Monitoring Equipment | CO₂ and O₂ sensors; SO₂ quantification systems [27] | Real-time measurement of gas exchange rates and contaminant processing | Dependency on specialized manufacturers concentrated in specific geopolitical regions |
| Growth Substrate Components | Hydroponic nutrients; solid growth media; soil substitutes | Rooting support and mineral nutrient delivery | Supply chain vulnerabilities for specialized components; potential for in-situ resource utilization |
| Lighting Systems | Fluorescent lamps; LED arrays with specific spectra [26] | Energy source for photosynthesis; growth and morphological development | Critical dependency on semiconductor components subject to trade disruptions |
| Environmental Control Systems | Temperature regulation; humidity control; atmospheric composition management | Maintenance of optimal growth conditions | Specialized components potentially subject to technology transfer restrictions |
| Biological Sampling Kits | Biomass measurement tools; tissue collection apparatus; preservation materials | Quantitative analysis of growth parameters and physiological status | Limited availability of specialized disposable items during supply chain disruptions |
The development of Bio-regenerative Life Support Systems represents a critical strategic capability that intersects with complex geopolitical realities. As research advances, maintaining awareness of supply chain vulnerabilities, international collaboration frameworks, and technology transfer environments will be essential for sustainable progress. The experimental protocols and quantitative metrics provided herein offer methodologies for advancing BLSS capabilities while acknowledging the geopolitical context that necessarily shapes implementation pathways. By integrating robust scientific methodologies with strategic geopolitical awareness, the research community can navigate the complex landscape of BLSS development more effectively, ultimately enabling sustained human presence beyond Earth through biologically-based life support systems.
Implementing Plant Cultivation Systems: From Hydroponics to Whole-System Architecture
Closed Ecological Life Support Systems (CELSS) are fundamental for long-duration human space exploration, as they aim to recycle vital resources and produce food sustainably. In these environments, the efficient cultivation of plants is paramount. This document provides detailed application notes and experimental protocols for three advanced plant cultivation systems—Hydroponic, Aeroponic, and Porous Ceramic Substrate systems—evaluating their integration into CELSS. The content is structured to provide researchers and life support engineers with a comparative quantitative analysis and reproducible methodologies for system implementation and testing.
Hydroponic Systems
Hydroponic systems cultivate plants without soil by suspending root systems in a nutrient-rich aqueous solution [28]. Essential macronutrients (e.g., nitrogen, phosphorus, potassium) and micronutrients are dissolved in the water, making them readily available for plant uptake [29]. Common setups include the Nutrient Film Technique (NFT), where a thin film of solution flows over the roots; Deep Water Culture (DWC), where roots are submerged in an oxygenated solution; and drip systems [28]. These systems are noted for enabling faster plant growth and higher yields compared to traditional soil-based agriculture, primarily due to the direct availability of nutrients and water [28] [30]. A key advantage for CELSS is their high water efficiency, as the nutrient solution can be recirculated, reducing consumption by up to 95% compared to traditional farming [30].
Aeroponic Systems
Aeroponics represents a further advancement in soilless cultivation by suspending plant roots in an air or mist environment [31]. The roots are periodically misted with a hydro-atomized, nutrient-dense solution, providing unparalleled access to oxygen and nutrients [28] [31]. This method is categorized into high-pressure systems, which produce a fine mist (20-50 micrometers) for optimal absorption, and low-pressure systems [28] [31]. The primary benefits for CELSS include extreme resource efficiency, using up to 95% less water than traditional methods, and promoting faster growth rates due to the highly oxygenated root zone [28] [29]. Its closed-loop nature also minimizes the risk of pathogen transmission between plants [31].
Porous Ceramic Substrate Systems
Porous ceramic substrates are a type of inorganic growing medium that provides physical support, water retention, and aeration to plant roots. These substrates are typically manufactured from baked clay, forming stable, porous pellets [32]. Their intrinsic porosity allows them to act as a reservoir for moisture and nutrients while ensuring adequate air spaces to prevent root anoxia. A significant advantage is their chemical inertness and neutral pH, which prevents them from altering the nutrient solution's chemistry [32]. Furthermore, their durability allows them to be sterilized and reused indefinitely, making them a sustainable option for long-duration space missions where resource resupply is limited [32].
Table 1: Quantitative Comparison of Cultivation System Architectures
| Parameter | Hydroponic Systems | Aeroponic Systems | Porous Substrate (Clay Pellets) |
|---|---|---|---|
| Water Usage Efficiency | High (Recirculating system) [28] | Very High (Up to 95% less than traditional farming) [28] [29] | Moderate (Dependent on irrigation cycle) |
| Nutrient Delivery Efficiency | High (Direct root contact) [28] | Very High (Direct root absorption from mist) [31] | High (Solution held in substrate porosity) [32] |
| Root Zone Oxygenation | Moderate (Requires active oxygenation in DWC) | Very High (Roots suspended in air) [31] | Good (Porous structure maintains air spaces) [32] |
| Typical Growth Rate | Faster than substrate [28] | Fastest among systems [28] | Good |
| System Redundancy | High (Buffer of nutrient solution) | Low (Vulnerable to pump failure) [28] | High (Substrate holds water and nutrients) |
| Suitability for CELSS* Nutrient Cycling | Excellent (Closed-loop recycling possible) [33] | Excellent (Precise, minimal waste) [33] | Good (Can be integrated into recirculating systems) |
| Mass and Volume Requirements | Moderate | Low (Vertical stacking possible) [29] | High (Mass of substrate) |
CELSS: Closed Ecological Life Support Systems
Experimental Protocols for CELSS Research
Protocol: Comparative Growth and Yield Analysis in a Controlled Environment
This protocol is designed to quantitatively evaluate the performance of the three system architectures under the resource constraints typical of a CELSS.
1. Objective: To measure and compare the growth rate, yield, water consumption, and nutrient use efficiency of a model crop (e.g., lettuce, Lactuca sativa) across hydroponic, aeroponic, and porous ceramic substrate systems.
2. Materials:
- Growth Chambers: Environmentally controlled chambers with adjustable LED lighting, temperature, humidity, and CO₂.
- Cultivation Systems:
- Hydroponic: NFT or DWC system.
- Aeroponic: High-pressure misting system with pump (≥80 PSI), misting nozzles, and timer.
- Porous Substrate: Pots filled with pre-washed, sterile clay pellets (e.g., LECA) connected to a drip or sub-irrigation system.
- Plant Material: Sterilized seeds of a uniform lettuce cultivar.
- Nutrient Solution: Standardized Hoagland's solution.
- Data Collection Tools: pH and Electrical Conductivity (EC) meters, digital scale, calipers, spectrophotometer for nutrient analysis, and a photosynthesis meter.
3. Methodology:
- System Setup & Calibration:
- Assemble each system within the growth chambers.
- Calibrate the aeroponic timer for a cycle of 15 seconds ON / 3 minutes OFF to ensure roots remain moist but not saturated [34].
- Calibrate the hydroponic and substrate system pumps for continuous or frequent irrigation.
- Fill all reservoirs with the standardized nutrient solution (pH 5.5-6.0, EC 1.2-2.0 mS/cm).
- Plant Establishment & Acclimation:
- Germinate seeds in sterile rock wool cubes [29].
- Upon seedling development, transplant them into their respective systems, ensuring uniformity in plant size and health across all groups.
- Environmental & Resource Monitoring:
- Maintain constant environmental conditions: light intensity, photoperiod (e.g., 16h light/8h dark), temperature (22±2°C), relative humidity (60-70%), and CO₂ (600-800 ppm).
- Monitor and adjust reservoir pH and EC daily.
- Record the volume of water and nutrient solution added to each system to maintain levels.
- Data Collection:
- Biomass: Destructively harvest a subset of plants at 14, 28, and final harvest (42 days). Measure fresh and dry weight of shoots and roots.
- Morphology: Measure leaf area, root length, and count leaf number weekly on non-destructive samples.
- Resource Use Efficiency: Calculate Water Use Efficiency (WUE) as grams of fresh biomass per liter of water consumed.
- Physiological Analysis: At final harvest, analyze plant tissue for nutrient content (N, P, K, Ca, Mg) and secondary metabolites (e.g., phenolics, antioxidants).
4. Data Analysis: Perform statistical analysis (e.g., ANOVA with post-hoc tests) on all quantitative data to identify significant differences (p < 0.05) between the systems for all measured parameters.
Protocol: Stress Resilience to Nutrient and Water Perturbations
This protocol tests the robustness of each system, a critical factor for the reliability of a CELSS.
1. Objective: To assess the tolerance of plants grown in each system architecture to transient nutrient deficiency and water interruption.
2. Methodology:
- Establish Control Groups: Grow plants as described in Protocol 3.1 for 30 days under optimal conditions.
- Impose Stress Treatments:
- Nutrient Stress: Replace the nutrient solution in the reservoir with a 1/4 strength solution for a period of 7 days.
- Water/Delivery Stress: Halt all irrigation/misting in the systems for a period of 12 hours.
- Post-Stress Recovery & Assessment:
- After the stress period, resume optimal nutrient and irrigation protocols.
- Monitor and record visual symptoms of stress (wilting, chlorosis) at 0, 6, 24, and 48 hours after stress induction and after recovery.
- Measure photosynthetic efficiency (Fv/Fm ratio) using a chlorophyll fluorometer before, immediately after, and 48 hours after the stress period.
- At the end of the recovery period, measure the final biomass and compare it to non-stressed controls.
System Architecture and Workflow Visualization
CELSS Cultivation System Integration Logic
Aeroponic Misting Control Logic
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Reagents for CELSS Cultivation Research
| Item Name | Function/Application | Technical Notes |
|---|---|---|
| Hoagland's Solution | Standardized nutrient solution for plant growth. | Provides all essential macro and micronutrients. Easily modifiable to induce nutrient stress. |
| Rock Wool Cubes | Inert, sterile substrate for seed germination and seedling establishment. | Excellent water-to-air ratio. Soak to adjust pH to 5.5 before use [29]. |
| LECA (Clay Pellets) | Porous ceramic substrate for physical plant support. | Chemically inert, reusable, and provides good aeration. Sterilize between uses [32]. |
| pH & EC Meters | Monitoring acidity/alkalinity and nutrient concentration (Electrical Conductivity) of solutions. | Critical for maintaining optimal nutrient bioavailability (pH 5.5-6.5) [28]. |
| High-Pressure Diaphragm Pump | Generating fine mist (20-50 µm droplet size) in high-pressure aeroponic systems. | Essential for creating the optimal root zone environment in aeroponics [31]. |
| Programmable Timer | Automating irrigation and misting cycles. | Allows for precise control of nutrient delivery intervals and duration, crucial for aeroponics [29]. |
| Hydroponic-Grade Mineral Nutrients | Sourcing pure, soluble forms of essential plant nutrients (N, P, K, Ca, Mg, S, Micronutrients). | Ensures no clogging of systems and optimal plant uptake. Vital for precise nutrient cycling studies [33]. |
Application Notes
The Role of Dwarf Varieties in Closed Ecological Life Support Systems
In closed ecological life support systems, the optimization of space, resources, and yield is paramount. Dwarf crop varieties represent a critical technology for such environments, as their compact architecture allows for higher planting densities and more efficient use of limited volume. The development of 'USU-Apogee', a full-dwarf hard red spring wheat, exemplifies this approach. This cultivar was specifically bred for controlled environments, standing at just 45-50 cm tall, which is 10-15 cm shorter than other previously used genotypes like 'Yecora Rojo'. This reduced height is coupled with a 10-30% yield advantage, making it a superior candidate for space-based food production [35] [36]. The selection for such dwarf varieties mitigates issues like lodging (falling over) and improves the harvest index—the proportion of total biomass converted into edible grain.
Beyond wheat, the principles of the Green Revolution are being applied to other staples. Research into semi-dwarf maize demonstrates how architectural optimization can enhance lodging resistance and performance under high-density planting, which is a likely scenario in a life support system [37]. The core physiological mechanism often involves the manipulation of plant growth hormones, particularly gibberellins (GAs), to create shorter, sturdier plants without compromising productivity [38].
Key Physiological and Microbial Considerations
The selection of crops for closed systems must extend beyond visible traits to include physiological and microbial interactions. Dwarfing traits can significantly alter plant-microbe relationships in the rhizosphere—the soil zone influenced by root secretions. Studies show that tall wheat cultivars sustain a more complex and interconnected rhizosphere bacterial network compared to semi-dwarf cultivars [39]. This suggests that the introduction of dwarfing genes may have inadvertently affected the plant's ability to recruit beneficial microbes.
Furthermore, the miR396-GRF-GIF regulatory module is a key conserved pathway controlling growth in plants. Growth-Regulating Factors (GRFs) are transcription factors that promote cell proliferation and organ growth. Their activity is finely tuned by microRNA396 (miR396), which represses specific GRF genes. Engineering this pathway offers a potential strategy for developing optimized cultivars with enhanced growth and yield [40]. Understanding these molecular networks is crucial for predicting how dwarf varieties will perform and interact with their engineered ecosystem over multiple generations.
Protocols
Protocol 1: Evaluation of Dwarf Wheat Cultivars in a Controlled Environment
This protocol is adapted from methods used in the development and analysis of the 'USU-Apogee' wheat cultivar [35] [36] [39].
Research Reagent Solutions
| Reagent / Material | Function / Explanation |
|---|---|
| High-Yielding Variety (HYV) Seeds | Genetically superior seeds with traits such as dwarfing, high yield potential, and stress resistance [41]. |
| Inorganic Fertilizer (e.g., NH₄NO₃) | Provides essential macronutrients, particularly nitrogen, which is critical for biomass and grain yield [42]. |
| Plant Growth Regulators (PGRs) | Chemicals like Chlormequat that inhibit gibberellin biosynthesis, used to further control plant height and study phenotypic plasticity [42]. |
| PowerSoil DNA Isolation Kit | For standardized extraction of microbial genomic DNA from rhizosphere soil samples [39]. |
| 16S rRNA Gene Primers | For amplifying specific regions of bacterial DNA for subsequent sequencing and community analysis [39]. |
Experimental Workflow
The following diagram outlines the key stages for evaluating dwarf cultivars, from initial growth to final analysis of plant and microbiome data.
Detailed Methodology
Step 1: Controlled Environment Setup
- Planting: Utilize a standardized growth substrate. Pre-germinate seeds and plant at a defined density.
- Experimental Design: Arrange plants in a Completely Randomized Design (CRD) with a minimum of three replicates per cultivar to ensure statistical robustness [42].
- Environmental Control: Maintain precise control over temperature, light intensity, photoperiod, and atmospheric composition to simulate life support system conditions.
Step 2: Plant Growth & Maintenance
- Fertilization: Apply nutrients as a base dressing. Nitrogen (N) is a critical variable; treatments can include multiple N rates (e.g., 0, 2.5, and 45 mg N kg⁻¹ of soil) to assess Nitrogen-Use Efficiency (NUE) [42].
- Watering: Implement a consistent irrigation schedule, as HYV seeds can have different water requirements compared to traditional varieties [41].
Step 3: Phenotypic Data Collection Collect quantitative data at key developmental stages (e.g., flowering, maturity):
- Plant Height: Measure from soil base to the tip of the main stem.
- Tiller/Fertile Tiller Count: Number of primary and grain-producing shoots.
- Yield Components: Record number of panicles, spikelets, and grains per plant. Measure fresh and dry weights of panicles, straw, and grains.
- Root Morphology: Wash root systems and scan them. Use image analysis software to determine traits like Root Diameter and Specific Root Length (SRL) [39].
Step 4: Rhizosphere Sampling
- At a defined growth stage (e.g., flowering), carefully uproot plants.
- Gently shake off loose soil. The tightly attached soil shaken into a sterile bag is defined as the rhizosphere soil [39].
- Homogenize the sample and subsample for DNA extraction, storing at -80°C.
Step 5: Microbiome Analysis
- DNA Extraction: Use a commercial kit (e.g., MoBio PowerSoil) for consistent results [39].
- 16S rRNA Gene Sequencing: Amplify and sequence the bacterial 16S rRNA gene from the extracted DNA using Illumina-based platforms.
- Bioinformatics: Process sequences to identify Operational Taxonomic Units (OTUs). Use statistical tools like PERMANOVA to test for community differences between tall and dwarf cultivars [39].
Step 6: Data Integration and Analysis
- Correlate plant phenotypic data (yield, height, SRL) with microbiome data (community structure, diversity).
- Statistical analysis (e.g., ANOVA, post-hoc tests) should be performed to confirm the significance of observed differences between cultivars [39].
Protocol 2: Molecular Analysis of the Gibberellin Pathway in Dwarf Crops
This protocol is based on research that generated dwarf maize through the manipulation of gibberellin (GA) levels [38].
Key Molecular Pathways
The diagram below illustrates the core gibberellin inactivation pathway that can be manipulated to induce dwarfism.
Detailed Methodology
Step 1: Genetic Transformation
- Gene Selection: The AtGA2ox1 gene from Arabidopsis thaliana, which encodes a gibberellin 2-oxidase, is a well-characterized candidate [38].
- Vector Construction: Clone the AtGA2ox1 gene into an appropriate plant transformation vector under the control of a constitutive or tissue-specific promoter.
- Plant Transformation: Introduce the construct into the target crop (e.g., maize) using established methods like Agrobacterium-mediated transformation [38].
Step 2: Molecular Phenotyping of Transgenic Lines
- Plant Height Assessment: Measure and compare the height of transgenic and wild-type plants under controlled conditions. The goal is a significant reduction (e.g., as achieved in GM maize [38]).
- Gibberellin Profiling:
- Extraction: Harvest shoot tissues and extract GAs using organic solvents.
- Quantification: Use liquid chromatography-mass spectrometry (LC-MS) to quantify levels of bioactive GAs (e.g., GA1) and their inactive products. Successful AtGA2ox1 expression should result in a substantial decline (e.g., 74%) in bioactive GA1 levels [38].
- Chlorophyll Content: Measure leaf chlorophyll concentration. Dwarf genotypes with modulated GA pathways often exhibit increased chlorophyll content [38].
- Histological Analysis: Examine stem cross-sections under a microscope. Dwarf plants may show condensed stem cells and altered vascular bundle structures [38].
Data Presentation
Table 1: Performance Metrics of Dwarf Crops in Selected Studies
| Crop | Cultivar / Line | Key Characteristic | Height Reduction | Yield Impact | Additional Traits | Source |
|---|---|---|---|---|---|---|
| Wheat | 'USU-Apogee' | Full-dwarf cultivar | 45-50 cm tall (10-15 cm shorter than controls) | 10-30% yield increase | Resistance to leaf tip chlorosis | [35] [36] |
| Maize | AtGA2ox1 Transgenic Line | Bioactive GA passivation | Significant reduction (specific height not given) | 10-14% grain yield increase | Higher chlorophyll content; Altered root/shoot ratio | [38] |
| Rice | Dwarf Varieties (e.g., IR-5) | Green Revolution semi-dwarf | Dwarf stature | High yield with N-fertilizer | Reduced lodging risk; Responsive to fertilizers | [41] |
Table 2: Advantages and Challenges of High-Yielding Dwarf Varieties
| Aspect | Description / Example |
|---|---|
| Key Advantages | |
| Higher Harvest Index | More biomass partitioned to grain rather than straw [38] [37]. |
| Lodging Resistance | Short, sturdy stems prevent falling over, especially under high nitrogen or wind [37] [41]. |
| Response to Inputs | Respond well to fertilizer and irrigation, leading to significant yield gains [41]. |
| Suitability for Controlled Environments | Ideal for space-limited, intensive cultivation in life support systems [35]. |
| Considerations & Challenges | |
| Altered Rhizosphere Microbiome | Semi-dwarf cultivars may host less complex bacterial networks than tall cultivars [39]. |
| Input Dependency | Often require more reliable water, fertilizer, and management [41]. |
| Potential Disease Sensitivity | Maybe more sensitive to certain diseases compared to traditional varieties [41]. |
In closed ecological life support systems (CELSS), the efficient recycling of nutrients is paramount for long-duration space missions and extraterrestrial habitation. These systems aim to achieve a high degree of material closure, mirroring Earth's biogeochemical cycles but within constrained artificial environments [6]. Human metabolic waste represents a critical resource stream, containing essential plant nutrients that must be recovered and reused to sustain food production and regenerate breathable air [33]. Nutrient recovery and delivery from human waste streams thus forms a fundamental pillar of bioregenerative life support, transforming what is traditionally considered waste into valuable fertilizers for plant cultivation systems [43]. This application note details protocols and methodologies for closing the nutrient loop within CELSS, with specific focus on technologies and processes applicable to space-based research environments.
Nutrient Composition of Human Waste Streams
Human excreta contains most essential nutrients required for plant growth, though their concentration and availability vary significantly between waste fractions. A detailed understanding of this composition is essential for designing efficient recovery systems.
Table 1: Primary Nutrient Distribution in Human Excreta
| Waste Stream | Nitrogen (N) | Phosphorus (P) | Potassium (K) | Key Characteristics |
|---|---|---|---|---|
| Urine | 80-90% of excreted N | 50-80% of excreted P | 80-90% of excreted K | Low volume, high nutrient concentration, relatively sterile |
| Feces | Remaining 10-20% | Remaining 20-50% | Remaining 10-20% | High organic matter, complex structure, requires stabilization |
| Mixed Wastewater | Variable dilution | Variable dilution | Variable dilution | Dilute nutrient concentration, complex mixture (blackwater/greywater) |
Source: Adapted from [43]
The successful integration of these nutrient streams into CELSS agriculture requires robust recovery pathways. The following workflow outlines the primary stages from waste collection to plant nutrient delivery.
Quantitative Evidence and Recovery Efficiencies
Research into nutrient recovery technologies has yielded substantial data on their potential performance. A large-scale systematic map of the evidence base, encompassing nearly 11,000 articles, reveals the focus and maturity of different recovery pathways [43].
Table 2: Global Research Focus on Nutrient Recovery and Reuse (Evidence from 10,950 articles)
| Research Category | Proportion of Evidence Base | Key Observations and Knowledge Gaps |
|---|---|---|
| Recovery Technologies | 41.9% | Most studied area; includes physical, chemical, and biological methods. |
| Reuse in Agriculture | 53.4% | Dominant application focus; includes crop trials and soil amendment studies. |
| Product Characterization | 4.0% | Relative knowledge gap; more data needed on final product composition and bioavailability. |
| User Acceptance & Perception | 0.7% | Significant knowledge gap; critical for technology adoption in closed environments. |
Furthermore, specific recovery processes can achieve high efficiency. For instance, microalgae-based treatment of wastewater can remove up to 90% of nitrogen and phosphorus from the water stream, simultaneously concentrating these nutrients in harvestable biomass [44]. This biomass can then serve as a potent biofertilizer, completing the cycle from waste to plant nutrient.
Application Protocols for CELSS
Protocol: Nutrient Recovery from Source-Separated Urine via Struvite Precipitation
This protocol describes a chemical method for recovering phosphorus and nitrogen from urine in the form of struvite (MgNH₄PO₄·6H₂O), a slow-release fertilizer ideal for CELSS agriculture.
I. Research Reagent Solutions
Table 3: Essential Reagents for Struvite Precipitation
| Reagent/Material | Function | Notes for CELSS Application |
|---|---|---|
| Source-Separated Urine | Feedstock containing N, P, K | Pre-stored for 24h to hydrolyze urea; collected from sanitation systems. |
| Magnesium Chloride (MgCl₂) | Magnesium ion source for crystal formation | Preferred Mg source due to high solubility and low cost. |
| Sodium Hydroxide (NaOH) | pH adjustment to optimal range (8.5-10) | Essential for shifting equilibrium towards struvite formation. |
| Filter Paper (0.45 μm) | Recovery of precipitated crystals | Standard laboratory filter or membrane filter. |
| Drying Oven | Product dehydration | Stabilizes product for long-term storage and use. |
II. Experimental Workflow
III. Methodology Details
- Urine Preparation: Collect fresh urine using a source-separation sanitation system. Allow the urine to hydrolyze for at least 24 hours to convert urea to ammonium, which increases the ammonium concentration necessary for struvite formation.
- Baseline Analysis: Analyze a sample of the hydrolyzed urine for its initial phosphorus (e.g., as PO₄-P) and nitrogen (e.g., as NH₄-N) concentration. This data is critical for calculating stoichiometric reagent addition.
- Reaction Setup: Place a known volume of urine in a stirred reactor. Slowly add a sodium hydroxide (NaOH) solution (e.g., 1-2 M) under continuous mixing until the pH reaches 9.0.
- Precipitation: Add a magnesium chloride (MgCl₂) solution to the reactor. The molar ratio of Mg:PO₄ should be slightly supra-stoichiometric, typically 1.3:1, to drive the reaction to completion. Continue mixing for 30-60 minutes to allow crystal growth.
- Product Recovery: Filter the contents of the reactor through filter paper to collect the white crystalline precipitate. Rinse the crystals with a small amount of deionized water to remove impurities.
- Product Finishing: Transfer the filter cake to a drying oven and dry at 40°C for approximately 24 hours. The resulting solid can be gently ground and stored as a slow-release phosphorus fertilizer for base substrate amendment in plant growth systems.
Protocol: Valorization of Solid Waste and Biosolids
This protocol describes the stabilization of feces and other organic solids through composting, producing a stable soil amendment that improves substrate structure and provides a slow-release nutrient source.
I. Methodology Details
- Feedstock Preparation: Combine source-separated feces with a carbon-rich bulking agent (e.g., shredded inedible plant biomass, wood chips) at a typical carbon-to-nitrogen (C:N) ratio of 25:1 to 30:1. The bulking agent ensures proper porosity and aeration.
- Composting Reactor: Place the mixed feedstock into a controlled bioreactor capable of maintaining temperature and aeration. In CELSS, this must be a closed system to avoid gaseous emissions into the habitat.
- Process Monitoring: Actively manage the compost pile. Maintain temperatures of 55-65°C for several days to ensure pathogen reduction. Turn the pile or force-aerate it periodically to maintain aerobic conditions. Monitor moisture content, keeping it between 40-60%.
- Curing and Storage: After the active thermophilic phase, allow the compost to cure for several weeks at lower temperatures. This stabilizes the product and completes the decomposition process. The final compost should be dark, crumbly, and have an earthy smell. It can be incorporated into plant growth substrates as an organic amendment.
Experimental Validation and Plant Assays
Validating the efficacy of recovered nutrients is a critical step. This is typically done through controlled plant growth experiments, comparing the performance of recovered products against conventional fertilizers.
I. Research Reagent Solutions
- Test Plants: Amaranth (Amaranthus cruentus), a fast-growing C4 plant with high nutritional value, is an excellent model organism for initial trials [44]. Other candidates include wheat, lettuce, or tomato, depending on CELSS crop selection.
- Growth Substrate: Inert media such as perlite, vermiculite, or rockwool for hydroponic systems, or a standardized soil if using soil-based cultivation.
- Nutrient Solutions: Treatments should include:
- T1: Control with inorganic fertilizer (e.g., Hoagland's solution).
- T2: Recovered product (e.g., struvite, compost leachate, microalgae biomass).
- T3: Blend of recovered products.
- Analysis Kits: Equipment for plant physiological analysis, including chlorophyll meter, portable photosynthesis system, and kits for nutrient analysis in plant tissue.
II. Experimental Workflow for Validation
III. Methodology Details
- Experimental Design: Establish a randomized block design in a growth chamber with controlled environmental conditions (light, temperature, humidity, CO₂). Sow seeds of the test plant in the growth substrate.
- Treatment Application: Once seedlings are established, begin applying the different nutrient solutions. Ensure all treatments, including the control, receive equivalent total amounts of key nutrients (N, P, K) based on prior chemical analysis of the recovered products.
- Physiological Monitoring: Periodically measure photosynthetic rate, stomatal conductance, and chlorophyll content (SPAD values) throughout the growth cycle.
- Harvest and Analysis: At a predetermined developmental stage (e.g., early flowering), harvest plant biomass. Separate shoots and roots for dry weight measurement. Analyze dried leaf tissue for macro- and micronutrient concentration to determine nutrient uptake efficiency.
- Data Interpretation: Compare biomass yield, physiological data, and nutrient uptake across treatments. Successful recovery products will show comparable or superior performance to inorganic controls, demonstrating their viability for CELSS. For example, studies have shown that amaranth plants fertilized with sewage-derived biofertilizers (microalgae, biosolids) can exhibit increased shoot biomass and photosynthetic rates, confirming the efficacy of the recovered nutrients [44].
Integration Strategy for CELSS
The ultimate goal is the seamless integration of these protocols into a functioning closed-loop life support system. This requires a systems-level approach that considers mass balances, control engineering, and operational logistics.
- Mass Balance Modeling: Implement dynamic models to track the flux of key elements (C, N, P, K, water) through the human habitat, waste processing, and plant cultivation units. This allows for predictive management of nutrient inventories and identification of potential bottlenecks [33].
- Hybrid Life Support: Combine bioregenerative approaches (using plants and microbes) with physicochemical systems (for water recycling, air revitalization) to create a robust, redundant architecture. For instance, a CELSS can use hydroponic plant cultivation for primary food production and air regeneration, coupled with a composting reactor for solid waste processing and a struvite reactor for urine treatment [6].
- Contingency Planning: Develop protocols for system perturbations, such as variable crew size, crop failure, or reactor malfunction. This includes maintaining a small buffer stock of critical nutrients or having the capability to process waste via alternative pathways.
Closed Ecological Life Support Systems (CELSS) are advanced bio-regenerative systems designed to sustain human life in space by producing food, regenerating oxygen, recycling water, and managing waste through biological processes, primarily using higher plants [23]. Within these systems, precision environmental control is paramount. It moves beyond traditional farming by creating a tightly managed, data-driven environment where sensors, artificial intelligence (AI), and automated irrigation work in concert to optimize plant growth and system-wide resource cycling [23] [27]. The objective is to achieve maximum efficiency and stability in atmospheric and water composition within a closed system, which is critical for long-duration space missions [27]. These controlled environments require several specialized subsystems to function effectively [23].
Core Sensing and Monitoring Technologies
At the foundation of any precision control system is a sophisticated network of sensors that transform invisible environmental factors into actionable data [45]. These sensors provide the continuous monitoring necessary for maintaining closed-system equilibrium.
Table 1: Core Sensor Technologies for Precision Environmental Control
| Sensor Type | Measured Parameter(s) | Precision Function in CELSS | Typical Specifications/Notes |
|---|---|---|---|
| Soil Moisture Sensors [46] | Volumetric Water Content | Enables precision irrigation by triggering water delivery only when needed, preventing water logging and stress. | IoT-enabled; part of a larger sensor stack. |
| Temperature Sensors [45] | Ambient Air & Root Zone Temperature | Creates thermal maps, identifies microclimates, and automatically adjusts climate controls for optimal metabolic rates. | Can detect slight temperature gradients affecting development. |
| Humidity Sensors [45] | Relative Humidity, Vapor Pressure Deficit (VPD) | Prevents fungal growth by managing moisture in the air while ensuring sufficient transpiration for plant health and nutrient uptake. | VPD is a key measurement for plant health. |
| pH Sensors [45] | Soil or Hydroponic Solution Acidity/Alkalinity | Ensures optimal nutrient availability and uptake; critical for maintaining system health in soilless CELSS agriculture. | Continuous monitoring and adjustment is possible. |
| Multispectral Sensors [46] | Crop Health, Soil Moisture (indirect) | Leveraged via satellite or drone imagery to monitor crop health and field conditions non-invasively across large areas. | Provides data for AI analysis. |
Beyond the sensors listed in Table 1, gas sensors are uniquely critical in a CELSS. They monitor atmospheric components like CO₂ and O₂, which plants cycle, and can even detect and help manage the removal of trace toxic impurities from the sealed atmosphere [27].
AI, Data Integration, and Automated Decision-Making
The data collected from the sensor network is processed and interpreted by Artificial Intelligence (AI) to move from simple monitoring to predictive control and automated decision-making [46] [45].
Predictive Analytics and Machine Learning
Advanced machine learning algorithms analyze historical crop data alongside real-time sensor information to predict optimal growing conditions and potential issues before they become critical [45]. These systems can accurately forecast disease outbreaks or pest infestations, allowing for preemptive intervention. By learning from each growth cycle within the closed system, the AI's predictions and recommendations become increasingly sophisticated and tailored to the specific CELSS environment [45].
Computer Vision for Plant Health
AI-powered cameras provide a layer of visual monitoring that can identify subtle signs of disease, nutrient deficiency, or plant stress that may be invisible to the human eye or basic sensors [45]. These systems can analyze thousands of plants per hour, providing an early warning system that allows for targeted intervention before problems spread and compromise the limited biosphere of a CELSS [45].
Automated and Precision Irrigation Systems
Automated irrigation is the actuator through which sensor and AI decisions are physically manifested, ensuring the precise delivery of water and nutrients.
System Operation and Technologies
An auto irrigation system operates via a closed-loop control system. Soil moisture sensors continuously monitor root zones, and this data is fed into a central controller [46]. This controller, often guided by AI that integrates weather data and evapotranspiration models, then commands automated valves and energy-efficient pumps to dynamically regulate water flow to individual plants or zones [46] [45]. This ensures water is delivered based on real-time plant need, eliminating the wastefulness of timer-based systems. For example, if the system detects adequate soil moisture after a simulated rainfall event, it will automatically skip the next watering cycle, directly conserving water [46].
Quantitative Impact of Automation
The integration of sensors, AI, and automated actuation delivers measurable benefits essential for the resource-conscious context of a CELSS.
Table 2: Estimated Impact of Smart Irrigation and Automation Technologies [46]
| Technology Type | Estimated Water Savings (%) | Estimated Yield Improvement (%) | Estimated Cost Reduction (%) |
|---|---|---|---|
| Traditional Systems (Manual/Timer-based) | 0-10% | 0-5% | 0-3% |
| Smart Sensors + IoT | 15-25% | 10-17% | 13-18% |
| AI-Based Controllers | 25-30% | 18-25% | 20-27% |
| Integrated Automated Systems | 28-35% | 20-30% | 22-30% |
Experimental Protocols for System Validation
Protocol: Calibration of an Integrated Sensor Network
Objective: To ensure accuracy and reliability of all environmental sensors within a CELSS growth chamber.
- Pre-Calibration: Visually inspect all sensors (soil moisture, temperature, humidity, pH, CO₂) for damage or debris. Clean probes as necessary.
- Reference Standards: Place sensors alongside NIST-traceable reference instruments in a stable, controlled environment.
- Data Logging: Simultaneously log data from the sensor network and reference instruments for a minimum of 24 hours across a range of pre-set environmental conditions (e.g., 18°C, 22°C, 26°C; 60% RH, 70% RH, 80% RH).
- Analysis and Adjustment: Calculate the mean and standard deviation for the difference between each sensor and its reference. Apply linear offset corrections or calibration constants to the sensor software as defined by the discrepancy analysis.
- Validation: Verify calibration by introducing a new set of environmental conditions and confirming sensor readings remain within ±2% of reference values.
Protocol: Validating AI-Driven Irrigation Decision Support
Objective: To quantify the efficiency of an AI-controlled irrigation system against a traditional timer-based schedule in a CELSS-relevant crop.
- Experimental Setup: Establish two identical growth chambers with the same crop (e.g., lettuce Lactuca sativa), soil substrate, and light levels (PPFD ~300 μmol/m²/s) [26].
- System Configuration: Chamber A uses an AI-driven system with soil moisture sensors and weather integration. Chamber B uses a fixed-timer set to irrigate twice daily for 10 minutes.
- Monitoring and Metrics: Over a 30-day growth cycle, track:
- Water Usage: Total volume of water delivered to each chamber.
- Plant Biomass: Fresh weight of above-ground biomass at harvest [26].
- Plant Health: Weekly leaf area index and chlorophyll content (via SPAD meter).
- Data Analysis: Perform a t-test to compare the final biomass and water use efficiency (biomass produced per liter of water) between the two chambers. A statistically significant improvement (p < 0.05) in Chamber A demonstrates the AI system's superiority.
System Workflow and Signaling Pathways
The following diagram illustrates the integrated logical workflow of a precision environmental control system within a CELSS context.
The Scientist's Toolkit: Research Reagent and Material Solutions
Table 3: Essential Research Materials for CELSS Precision Agriculture
| Item / Solution | Function / Application |
|---|---|
| 3-in-1 Wi-Fi Hydroponics Kit (e.g., pH, Conductivity, Temperature) [45] | Enables remote, continuous monitoring of key hydroponic solution parameters, crucial for soilless CELSS cultivation. |
| Calibration Buffers (pH 4.0, 7.0, 10.0) | Essential for maintaining the accuracy of pH sensors in hydroponic nutrient delivery systems to ensure optimal nutrient uptake. |
| Nutrient Solution (Hoagland's Solution or equivalent) | A standardized, complete mix of essential macro and micronutrients for consistent plant growth studies in hydroponic CELSS prototypes. |
| Soil Moisture Calibration Standards | Used to correlate sensor output (e.g., dielectric permittivity) with actual volumetric water content for specific growth media. |
| Sterilized Growth Substrate (e.g., Clay Pebbles, Rockwool) | Provides a sterile, inert root support structure for hydroponic experiments, preventing confounding variables from soil microbes. |
| Liquid CO₂ for Enrichment | Used in experimental chambers to study and maintain elevated CO₂ levels, optimizing photosynthesis and contributing to atmospheric balance [27]. |
The pursuit of human exploration beyond Earth necessitates the development of advanced Bioregenerative Life Support Systems (BLSS) to sustainably provide essential resources. These closed-loop systems rely on biological processes to regenerate oxygen, purify water, and produce food, thereby reducing dependence on Earth-based resupply for long-duration missions [47]. This case study examines two premier terrestrial analogs for BLSS research: the Space Analog for the Moon and Mars (SAM) at the University of Arizona's Biosphere 2, and China's Yuegong-1 (Lunar Palace 1) at Beihang University. These facilities enable researchers to validate architecture concepts, demonstrate technologies, and study human performance in controlled, isolated environments that simulate extraterrestrial habitats [48].
SAM Habitat at Biosphere 2
The SAM habitat is a hermetically sealed and pressurized research facility constructed around the original 1987 Biosphere 2 Test Module [49] [50]. With a total area of 1,200 square feet, SAM integrates a greenhouse with hydroponic systems, a workshop, kitchen, common area, and sleeping quarters [49]. A fully functional airlock connects to an indoor Mars yard for simulated extravehicular activities (EVAs) in pressurized suits [50]. SAM distinguishes itself as one of the few analogs capable of maintaining precise atmospheric pressure and composition control, providing a high-fidelity environment for testing mechanical and plant-based life support systems [49] [51]. Research at SAM focuses on developing advanced systems for bioregeneration, CO₂ scrubbing, food cultivation, and resource recycling [50].
Beijing Lunar Palace 1 (Yuegong-1)
The Lunar Palace 1 facility encompasses 160 square meters of self-contained laboratory space in Beijing, comprising a 58m² vegetation area with two plant cultivation cabins and a 42m² living area with three bedrooms, dining room, bathroom, and waste disposal chamber [52] [53]. As China's first BLSS research facility and the third of its kind globally, Lunar Palace 1 employs a bioregenerative approach where oxygen is regenerated through the vegetation compartment, water is recycled internally, and crew waste is composted [52]. The system was designed to produce 55% of food consumed by a three-person crew internally, balanced by reserves [52].
Table 1: Comparative Facility Specifications
| Parameter | SAM Habitat | Lunar Palace 1 |
|---|---|---|
| Total Area | 1,200 ft² (111 m²) | 160 m² |
| Vegetation Area | Test Module greenhouse | 58 m² (two cabins) |
| Living Area | Workshop, kitchen, crew quarters, bathroom | 42 m² (3 bedrooms, dining, bathroom) |
| Key Features | Pressurized, airlock, Mars yard, hydroponics | Waste disposal chamber, bioregenerative system |
| Primary Research Focus | Mechanical & plant-based life support integration | Closed-loop ecological life support |
| Crew Capacity | 1-4 members | 3-4 members |
Experimental Protocols and Methodologies
Plant Cultivation and Bioregenerative Research
Protocol 1: Dwarf Pea Plant Carbon Sequestration Study (SAM Habitat)
Objective: To quantify carbon dioxide removal and oxygen production rates of dwarf pea plants in a sealed environment [54].
Materials:
- 144 dwarf pea plants (Pisum sativum dwarf variety)
- Hydroponic racks with automated nutrient delivery
- Environmental sensors (CO₂, O₂, temperature, humidity)
- Sealed growth chambers within SAM Test Module
- Photosynthetically Active Radiation (PAR) meters
Methodology:
- Establish baseline atmospheric conditions within sealed SAM habitat
- Introduce human participant and 144 dwarf pea plants simultaneously
- Maintain continuous monitoring of gas concentrations via embedded sensors
- Provide controlled lighting, nutrient delivery, and environmental parameters
- Harvest plants on Day 8 for biomass analysis and carbon content measurement
- Continue monitoring for additional week to document atmospheric changes without plants
- Calculate carbon sequestration rates per plant based on gas exchange data [54]
Protocol 2: Multi-Crop BLSS Integration (Lunar Palace 1)
Objective: To evaluate the sustainability of diverse crop species within a closed ecosystem.
Materials:
- Five cereal crops: wheat, corn, soybeans, peanuts, lentils
- Fifteen vegetable species: carrots, cucumbers, water spinach, etc.
- Fruit crops: strawberries
- Protein source: Yellow mealworms (Tenebrio molitor)
- Bioregenerative life support infrastructure
- Waste composting and recycling systems [52]
Methodology:
- Establish optimized growing conditions for each crop species
- Implement crop rotation schedule to ensure continuous production
- Cultivate mealworms on inedible plant biomass and food scraps
- Monitor system closure through regular analysis of gas, water, and nutrient cycles
- Evaluate nutritional adequacy of produced diet for crew members
- Assess waste processing efficiency through biofermentation techniques [52]
Resource Management and System Closure
Protocol 3: Water Recycling and Conservation (SAM Habitat)
Objective: To evaluate water recycling efficiency and conservation strategies in a closed system.
Methodology:
- Allocate fixed water budget (60 gallons for 4 crew members over 6 days)
- Implement multi-stage water filtration and recycling systems
- Conduct daily water quality analysis
- Track consumption patterns across different uses (hydration, hygiene, plant growth)
- Evaluate water recovery rates from atmospheric humidity and waste streams [49]
Protocol 4: Atmospheric Management in Sealed Environments
Objective: To monitor and regulate atmospheric composition in closed habitats.
Methodology:
- Implement continuous monitoring of O₂, CO₂, humidity, and pressure
- Utilize both mechanical systems (CO₂ scrubbers) and biological systems (plants) for atmospheric control
- Track human and plant contributions to atmospheric changes
- Correlate crew activities (exercise, cooking, sleep) with atmospheric fluctuations
- Optimize system parameters to maintain safe and stable atmospheric conditions [49]
Quantitative Research Data and Findings
Table 2: Mission Parameters and Resource Utilization
| Parameter | SAM Inclusion I Mission | Lunar Palace 1 (2014) |
|---|---|---|
| Mission Duration | 6 days | 105 days |
| Crew Size | 4 members | 3 members |
| Water Allocation | 60 gallons (227 liters) total | N/A |
| Water Actual Use | 40 gallons (151 liters) total | N/A |
| Food System | Vegetarian rehydrated/freeze-dried | 55% internally produced |
| Primary Protein | Traditional plant sources | Yellow mealworms |
| Atmospheric Control | Mechanical + plant-based | Bioregenerative |
| Waste Management | Dehydration for mycelium processing | Biofermentation & composting |
Table 3: Crop Performance in BLSS Research
| Crop Species | Growth System | Key Metrics | Research Facility |
|---|---|---|---|
| Dwarf Pea Plants | Hydroponic racks | Carbon sequestration rate, O₂ production, space efficiency | SAM Habitat [54] |
| Wheat | Controlled environment chambers | Oxygen regeneration, calorie production, growth cycle | Lunar Palace 1 [52] |
| Yellow Mealworms | Waste conversion system | Protein yield (75% by weight), feed conversion ratio | Lunar Palace 1 [52] |
| Multi-Crop Systems | Integrated BLSS | Closure percentage, nutritional balance, resilience | Both Facilities |
Signaling Pathways and Experimental Workflows
BLSS Experimental Workflow Comparison
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagents and Experimental Materials
| Item | Function | Application in BLSS Research |
|---|---|---|
| Hydroponic Nutrient Solutions | Provide essential macro/micronutrients for plant growth | SAM dwarf pea cultivation; Lunar Palace crop production [54] [52] |
| Environmental Sensors (CO₂, O₂) | Monitor atmospheric composition in real-time | Both facilities for system closure verification [49] [52] |
| Dwarf Crop Varieties | Space-efficient food production | SAM dwarf peas; Lunar Palace compact cereals [54] [52] |
| Yellow Mealworms (Tenebrio molitor) | Convert inedible biomass to animal protein | Lunar Palace sustainable protein source [52] |
| Biofiltration Media | Water purification and contaminant removal | SAM water recycling systems [49] |
| Mycelium Inoculants | Process food scraps into protein-rich mushrooms | SAM waste-to-food conversion [49] |
| Hydroponic Growth Racks | Maximize production in limited volume | Both facilities for controlled agriculture [54] [52] |
| Braille Identification Markers | Ensure accessibility for visually impaired researchers | SAM habitat inclusivity features [49] |
The research conducted at SAM and Lunar Palace 1 demonstrates significant progress in BLSS development for long-duration space missions. SAM's focus on high-fidelity habitat simulation and Lunar Palace 1's emphasis on closed-loop bioregeneration provide complementary approaches to solving the challenges of sustainable life support. The quantitative data generated through their respective experimental protocols contributes essential parameters for future mission planning, including crop selection criteria, resource allocation models, and system integration strategies. Continued research at these facilities will further refine our understanding of the technical and human factors necessary for establishing permanent human presence beyond Earth, informing both space exploration and terrestrial sustainability initiatives.
Addressing System Failures, Plant Pathologies, and Optimization Strategies
In closed ecological life support systems, the management of plant pathologies is paramount for ensuring system health and food security. Unlike open-field agriculture, these controlled environments present unique challenges, including limited genetic diversity, controlled but potentially conducive microclimates, and the necessity for sustainable, low-toxicity control methods. Plant diseases, caused by fungal, bacterial, viral, and oomycete pathogens, can lead to significant yield reductions, threatening the sustainability of life support systems [55]. This document outlines advanced protocols for disease identification and sustainable control strategies specifically designed for closed research environments, emphasizing the integration of molecular diagnostics, biological control, and epidemiological principles.
Disease Identification and Diagnostic Protocols
Accurate and early diagnosis is the cornerstone of effective plant disease management in confined systems. Traditional visual identification is often insufficient until significant damage has occurred; therefore, leveraging sensitive laboratory methods is crucial for early detection before irreparable losses are encountered [56].
Diagnostic Workflow and Methodologies
A robust diagnostic pipeline integrates both field observations and laboratory confirmations. The following workflow details the systematic approach for identifying pathogenic threats.
Diagram 1: Integrated plant disease diagnostic workflow for closed environments.
The table below summarizes the key laboratory techniques used for pathogen identification, detailing their applications and limitations.
Table 1: Summary of Laboratory Diagnostic Methods for Plant Pathogens
| Method | Principle | Key Application | Key Advantage | Key Limitation |
|---|---|---|---|---|
| Koch's Postulates | Fulfillment of four criteria to establish a microbe as the cause of a specific disease. | Isolating and confirming pathogenicity of a suspected pathogen. | Gold standard for proving disease causality. | Time-consuming; not all pathogens are culturable [56]. |
| Microscopic Examination | Direct visualization of pathogen structures (e.g., spores, hyphae) using light or electron microscopy. | Initial identification of fungal and oomycete pathogens. | Rapid; provides visual confirmation of pathogen presence. | Requires expertise; may not distinguish between species [56]. |
| Serological Methods (e.g., ELISA) | Detection of pathogen-specific antigens using antibodies. | High-throughput testing for viruses and bacteria. | Highly specific; suitable for testing many samples. | Sensitivity can be lower than DNA-based methods [56]. |
| DNA-Based Methods (PCR) | Amplification of pathogen-specific DNA sequences using the polymerase chain reaction. | Highly sensitive and specific detection and identification of all pathogen types. | Extreme sensitivity for early detection; can differentiate strains. | Requires DNA extraction equipment and thermal cyclers [56]. |
Molecular Identification Protocol: PCR and DNA Sequencing
This protocol provides a detailed methodology for identifying plant pathogens using polymerase chain reaction (PCR) and sequencing of the Internal Transcribed Spacer (ITS) region for fungi or the 16S rRNA gene for bacteria [56].
1. DNA Extraction:
- Objective: To obtain high-quality, PCR-grade genomic DNA from plant tissue (symptomatic leaf, stem, or root material).
- Materials:
- Lysis Buffer: A buffer containing CTAB (Cetyltrimethylammonium bromide) to break down plant and pathogen cell walls.
- Chloroform-Isoamyl Alcohol: Used to separate proteins and lipids from nucleic acids.
- Isopropanol: Precipitates the DNA from the aqueous phase.
- RNase A: Enzyme that degrades RNA to purify the DNA extract.
- TE Buffer: Tris-EDTA buffer for resuspending and storing the purified DNA.
- Procedure:
- Grind 100 mg of plant tissue to a fine powder in liquid nitrogen using a sterile mortar and pestle.
- Add 500 µL of pre-warmed (65°C) CTAB lysis buffer and mix thoroughly.
- Incubate the sample at 65°C for 30-60 minutes with occasional mixing.
- Add an equal volume of chloroform-isoamyl alcohol (24:1), mix thoroughly, and centrifuge at 12,000 × g for 10 minutes.
- Transfer the upper aqueous phase to a new tube. Add 0.7 volumes of isopropanol, mix gently, and incubate at -20°C for 30 minutes to precipitate DNA.
- Centrifuge at 12,000 × g for 15 minutes to pellet the DNA. Wash the pellet with 70% ethanol.
- Air-dry the pellet and resuspend in 50 µL of TE buffer containing RNase A. Quantify DNA concentration using a spectrophotometer.
2. PCR Amplification:
- Objective: To amplify a specific region of pathogen DNA for identification.
- Materials:
- Primers: Use universal primers ITS1/ITS4 for fungi or 27F/1492R for bacteria.
- PCR Master Mix: Contains Taq DNA polymerase, dNTPs, MgCl₂, and reaction buffers.
- Thermal Cycler: Instrument for precise temperature cycling.
- Procedure:
- Prepare a 25 µL PCR reaction: 12.5 µL PCR master mix, 1 µL each of forward and reverse primer (10 µM), 2 µL DNA template (10-50 ng), and 8.5 µL nuclease-free water.
- Run the PCR with the following cycling conditions:
- Initial Denaturation: 95°C for 5 minutes.
- Amplification (35 cycles): Denature at 95°C for 30 sec, Anneal at 55°C for 30 sec, Extend at 72°C for 1 minute.
- Final Extension: 72°C for 7 minutes.
3. Gel Electrophoresis and Analysis:
- Objective: To visualize and confirm the success of the PCR amplification.
- Procedure:
- Load 5 µL of the PCR product mixed with loading dye onto a 1.5% agarose gel containing a DNA stain.
- Run the gel at 100V for 30-45 minutes alongside a DNA molecular weight marker.
- Visualize the gel under UV light. A single, bright band of the expected size indicates a successful amplification.
- Purify the PCR product and submit it for Sanger sequencing. Compare the resulting sequence to databases (e.g., GenBank) for definitive pathogen identification.
Disease Control and Management Strategies
Sustainable disease management in closed environments prioritizes biological and integrated methods over chemical interventions to maintain system health and avoid contamination.
Biological Control Agents (BCAs) and Mechanisms
Biological control uses beneficial microorganisms to suppress pathogens, improve plant immunity, or modify the environment [57]. It is a promising alternative to pesticides, offering target-specificity and potential for long-term establishment in a cropping system [57] [58].
Table 2: Major Biological Control Agents (BCAs) and Their Modes of Action
| Biocontrol Agent (BCA) | Example Organisms | Mode of Action | Target Pathogens/Diseases |
|---|---|---|---|
| Fungal Antagonists | Trichoderma harzianum, Ampelomyces quisqualis | Mycoparasitism (direct attack), antibiosis (antimicrobial compounds), competition for space and nutrients [57] [58]. | Soil-borne fungal pathogens; powdery mildews [58]. |
| Bacterial Antagonists | Bacillus subtilis, Pseudomonas chlororaphis, fluorescent pseudomonads | Antibiosis (e.g., lipopeptides, phenazines, hydrogen cyanide), competition for nutrients (e.g., iron via siderophores), induction of systemic resistance (ISR) in the host plant [57] [58]. | Root and foliar pathogens caused by fungi and bacteria; Plasmopara viticola (downy mildew) [57]. |
| Endophytic BCAs | Pseudozyma flocculosa, endophytic Bacillus spp. | Colonize plant tissues without causing disease and produce inhibitory secondary metabolites that deter pathogens [57]. | Powdery mildew; various phytopathogenic microorganisms [57]. |
| Induced Resistance | Application of specific bacterial strains or compounds | Primes the plant's own defense mechanisms (e.g., Systemic Acquired Resistance - SAR), making the host less susceptible to future infection [59] [57]. | Broad-spectrum protection against multiple pathogens. |
Protocol for Screening and Applying Bacterial BCAs
This protocol outlines the process for evaluating and utilizing bacterial BCAs, such as Bacillus subtilis, for disease suppression in a closed growth environment.
1. Screening for Antagonistic Activity: Dual Culture Assay
- Objective: To identify bacterial isolates that inhibit the growth of a target pathogen in vitro.
- Materials: Potato Dextrose Agar (PDA) plates, pure culture of target fungal pathogen, candidate BCA isolate.
- Procedure:
- Place a 5 mm mycelial plug of the target pathogen in the center of a PDA plate.
- Streak the candidate BCA bacteria in parallel lines 2.5 cm away from the fungal plug.
- Incubate the plate at 25°C and observe daily for 5-7 days.
- Measure the zone of inhibition (clear area with no fungal growth) between the BCA and the pathogen. A larger zone indicates stronger antagonistic activity.
2. Preparation of BCA Inoculum
- Objective: To produce a concentrated suspension of the BCA for application.
- Materials: Nutrient broth, centrifuge, sterile water or buffer.
- Procedure:
- Inoculate a flask of sterile nutrient broth with the BCA and incubate with shaking (150 rpm) at 28°C for 48 hours.
- Transfer the culture to a centrifuge tube and pellet the bacterial cells at 8,000 × g for 10 minutes.
- Discard the supernatant and resuspend the pellet in sterile 0.1M MgSO₄ buffer.
- Adjust the concentration of the suspension to 1 × 10⁸ CFU/mL using a spectrophotometer or plate counting.
3. Plant Application and Disease Assessment
- Objective: To evaluate the efficacy of the BCA in reducing disease on plants.
- Materials: Sprayer, growth chamber with plants at a susceptible stage.
- Procedure:
- Divide plants into two groups: treatment and control.
- For the treatment group, thoroughly spray the BCA suspension onto leaves and soil until runoff. For the control group, apply only the sterile buffer.
- After 24 hours, challenge all plants with the target pathogen by spraying with a pathogen spore suspension or introducing infected material.
- Maintain plants in controlled conditions and monitor disease development.
- After 7-14 days, assess disease severity using a standardized scale (e.g., 0-5, where 0=no symptoms and 5=severe symptoms or plant death). Calculate the percent disease control using the formula:
[(Disease Index Control - Disease Index Treated) / Disease Index Control] × 100.
Integrated Disease Management Framework
For long-term stability, a multi-faceted Integrated Pest Management (IPM) approach is essential. This combines all available control strategies within an epidemiological framework to manage disease at the landscape scale of the closed environment [55] [60].
Diagram 2: Components of an Integrated Disease Management (IDM) framework for closed environments.
This framework emphasizes prevention through sanitation and the use of clean planting material, continuous monitoring via diagnostic protocols, and the prioritized use of biological controls. Chemical interventions should be a last resort due to their potential disruptive effects on a closed ecosystem [55] [61].
The Scientist's Toolkit: Essential Research Reagents
The following table catalogs key reagents and materials essential for implementing the diagnostic and control protocols described in this document.
Table 3: Essential Research Reagents for Plant Pathology in Closed Environments
| Reagent/Material | Function/Application | Example Use Case |
|---|---|---|
| CTAB Lysis Buffer | DNA extraction from plant and fungal tissue. Breaks down cell walls and membranes to release nucleic acids. | Protocol 2.2: DNA extraction for PCR-based pathogen diagnosis [56]. |
| Universal PCR Primers (ITS/16S) | Amplification of conserved genomic regions for pathogen identification. | Protocol 2.2: PCR amplification to identify fungal (ITS) or bacterial (16S) pathogens [56]. |
| Agarose | Matrix for gel electrophoresis to separate and visualize DNA fragments by size. | Protocol 2.2: Confirming the presence and size of a PCR amplicon [56]. |
| Selective Media (PDA, NA) | Isolation and cultivation of specific microorganisms (fungi or bacteria). | Protocol 3.2: Culturing target fungal pathogens or bacterial BCAs for dual-culture assays. |
| Trichoderma harzianum | A common fungal BCA used for its mycoparasitic and competitive abilities. | Biological control of soil-borne diseases like damping-off and root rots [57] [58]. |
| Bacillus subtilis | A common bacterial BCA known for producing lipopeptides and inducing systemic resistance. | Biological control of foliar and root diseases; suppression of Plasmopara viticola [57]. |
| Nutrient Broth | Liquid medium for high-density cultivation of bacterial BCAs. | Protocol 3.2: Production of BCA inoculum for plant application trials. |
Plant biology in space is a critical discipline for enabling long-duration human space exploration. Within Bioregenerative Life Support Systems (BLSS), plants provide essential functions including oxygen production, carbon dioxide recycling, water purification, waste management, and fresh food production [62]. Furthermore, plants offer psychological benefits for crew members during extended isolation in space [62] [63]. However, the space environment presents unique challenges, with microgravity being a fundamental factor that induces physiological stress in plants, affecting their growth, development, and molecular processes [64] [63]. Understanding these effects and developing countermeasures is essential for creating sustainable BLSS for future missions to the Moon and Mars.
This document provides application notes and experimental protocols to study plant physiological stress under microgravity conditions. It is structured to support research within the broader context of developing robust plant cultivation systems for closed ecological life support, targeting researchers, scientists, and professionals in gravitational biology and related fields.
Physiological Effects of Microgravity on Plants
Microgravity induces a range of physiological and molecular responses in plants, fundamentally altering their normal growth patterns. The table below summarizes the key effects observed across different organizational levels.
Table 1: Documented Effects of Microgravity on Plant Physiology and Development
| System Level | Observed Effect | Experimental Evidence |
|---|---|---|
| Cellular | Altered cell proliferation and cell cycle regulation | Increased cell division reported in Arabidopsis cell cultures [63] [65] |
| Cellular | Disruption of meristematic competence | Observed in root apical meristem of seedlings [64] [63] |
| Molecular | Reprogramming of gene expression | Upregulation of genes involved in cell wall remodeling, oxidative stress, and defense response [63] [66] |
| Molecular | Changes in auxin polar transport and signaling | Altered localization of PIN-FORMED (PIN) auxin efflux carriers [63] [67] |
| Organ | Impaired gravitropism; altered root and shoot growth angles | Studies on Arabidopsis and rice showing disrupted root bending [68] [64] |
| Organ | Cell wall remodeling and property changes | Transcriptomic studies and physical measurements [66] [65] |
| Whole Plant | Alterations in photosynthesis and metabolism | Transcriptomic changes in photosynthesis-related genes in etiolated plants [66] |
Gravity Perception and Signaling
On Earth, plants perceive gravity primarily through the sedimentation of starch-filled plastids (amyloplasts) within specialized statocytes in the root columella and shoot endodermal cells. This is known as the starch-statolith hypothesis [67]. In microgravity, this sedimentation is disrupted, impairing the initial perception of the gravity vector [64] [67].
The downward movement of statoliths triggers a signal transduction pathway that leads to the asymmetric redistribution of the plant hormone auxin. This gradient is established and maintained by polar auxin transport proteins, primarily the PIN-FORMED (PIN) family of efflux carriers [63] [67]. In microgravity, this polar auxin transport is altered, leading to a loss of auxin asymmetry and consequently, impaired gravitropic curvature [63] [65]. Recent research on rice has elucidated that auxin activates genes responsible for cell wall biosynthesis and reinforcement on the lower side of the root, preventing cell expansion and enabling downward bending [68].
The following diagram illustrates the gravisensing and signaling pathway in plant roots, and how it is disrupted under microgravity conditions.
Experimental Platforms for Microgravity Research
Studying plant responses to microgravity requires specialized platforms, each with distinct capabilities, limitations, and cost considerations. The choice of platform depends on the experiment's specific requirements for duration, gravity quality, sample access, and budget.
Table 2: Comparison of Platforms for Microgravity Plant Research
| Platform | Microgravity Duration | Gravity Level (g) | Key Advantages | Key Constraints | Relative Cost |
|---|---|---|---|---|---|
| Clinostat/RPM | Unlimited (simulated) | Simulated | Ground-based; long-term studies; high sample throughput. | Not true microgravity; constant direction change. | Low [69] |
| Drop Tower | 5-10 seconds | 10⁻⁴ – 10⁻⁸ | High-quality microgravity; rapid turnaround. | Very short duration; high landing acceleration. | Medium [69] |
| Parabolic Flight | 15-30 seconds | 10⁻² – 10⁻³ | Can simulate Moon/Mars gravity; crew-operated experiments. | Low-quality microgravity; repeated hypergravity phases. | Medium-High [69] |
| Sounding Rockets | 4-13 minutes | 10⁻² – 10⁻⁴ | Good quality; recovery of samples possible. | High launch acceleration; short duration. | High [69] |
| International Space Station (ISS) | Days to years | 10⁻² – 10⁻⁵ | Long-term studies; crew interaction; advanced hardware. | High cost; long lead times; launch vibrations. | Very High [62] [69] |
Experimental Protocol: Assessing Gravitropic Response on a Random Positioning Machine (RPM)
Objective: To quantify the disruption of root gravitropism in Arabidopsis thaliana seedlings under simulated microgravity using an RPM.
Background: An RPM is a ground-based facility that randomizes the direction of the gravity vector over time, thereby averaging gravity to near zero and creating simulated microgravity conditions [70] [69].
Materials & Reagents:
- Arabidopsis thaliana seeds (e.g., Col-0 wild-type and relevant mutants such as pin2)
- Random Positioning Machine (RPM)
- Square Petri dishes (120 x 120 mm)
- Plant growth medium (e.g., 1/2 X MS medium with 0.8-1% agar)
- Sterile laminar flow hood
- Growth chamber with controlled light and temperature
- Digital camera or scanner for documentation
- Software for root angle analysis (e.g., ImageJ)
Procedure:
- Preparation:
- Pour plant growth medium into square Petri dishes.
- Sterilize Arabidopsis seeds using standard protocols (e.g., ethanol and bleach sterilization).
- Sowing and Germination:
- Sow sterilized seeds in rows on the surface of the solid growth medium.
- Seal plates with porous tape and stratify in the dark at 4°C for 2-3 days to synchronize germination.
- Transfer plates vertically to a growth chamber. Maintain under controlled conditions (e.g., 22°C, 16/8h light/dark cycle) for 3-4 days until roots are 1-2 cm long.
- RPM Experiment Setup:
- Select seedlings with straight, vertically oriented roots of similar length.
- Mark a subset of plates as "Experimental" and mount them securely onto the center of the RPM platform. Ensure the plant plane is aligned correctly.
- Mark another set of plates as "Static 1g Control" and place them vertically in the same growth chamber, shielded from the RPM's vibration.
- A third set, "Rotational Control," can be placed on a slowly rotating clinostat (1-2 rpm) to account for effects of constant fluid motion.
- Treatment and Data Acquisition:
- Run the RPM according to manufacturer specifications (typical speeds: 1-60 rpm) for a predetermined period (e.g., 24-48 hours).
- Photograph all plates at the start (T=0) and end (T=24/48h) of the experiment. Ensure plates are in the same orientation for imaging.
- Termination and Analysis:
- Measure the root growth angle relative to the gravity vector (vertical) for each seedling using image analysis software.
- Calculate the mean growth angle and standard deviation for each treatment group (RPM, Static 1g, Rotational Control).
- Statistically compare the mean angles using an appropriate test (e.g., Student's t-test or ANOVA) to determine if the RPM-induced simulated microgravity significantly disrupted gravitropic response compared to controls.
The Scientist's Toolkit: Key Research Reagents and Materials
Successful investigation into plant gravitational biology relies on a suite of specialized reagents, biological tools, and hardware.
Table 3: Essential Research Reagents and Solutions for Gravitational Plant Biology
| Reagent / Material | Function / Utility | Example Application |
|---|---|---|
| Arabidopsis thaliana Wild-types | Model plant with fully sequenced genome; well-characterized gravitropic response. | Baseline for physiological and molecular studies [67] [66]. |
| Auxin Transport Mutants | Disrupted auxin signaling or transport. Elucidates auxin's role in microgravity responses. | pin2 (altered root gravitropism) [67] [65]. |
| PIN Protein Antibodies | Immunodetection of auxin efflux carriers. Visualizes protein localization and abundance. | Confocal microscopy to study PIN polarity in roots under microgravity [67]. |
| Transcriptomic Kits (RNA-Seq) | Genome-wide expression profiling. Identifies genes and pathways responsive to microgravity. | Profiling seedlings from BRIC experiments on ISS [66]. |
| Genome Editing Tools (CRISPR/Cas9) | Targeted gene knockout or modification. Creates custom mutants to test gene function. | Engineering plants with enhanced stress resistance for BLSS [64]. |
| Auxin Reporters (e.g., DR5:GFP) | Visualizing auxin response maxima in vivo. Reports spatial and temporal auxin distribution. | Confirming loss of auxin asymmetry in microgravity [67]. |
| Fixatives (e.g., RNAlater, Glutaraldehyde) | Preserves RNA, protein, or cellular structure at specific time points. | Fixing biological samples in orbit for post-flight omics analysis [66]. |
Advanced Molecular Analysis and Future Directions
Modern plant space biology leverages omics technologies to build a systems-level understanding of microgravity adaptation. Transcriptomic studies consistently show that plants in spaceflight undergo cell wall remodeling, oxidative stress, and altered defense responses [66]. A key finding is the "apparent paradox" where significant molecular changes do not always lead to catastrophic organismal failure, suggesting robust adaptive plasticity [63].
Future research will focus on:
- Utilizing CRISPR/Cas9 to develop plants with enhanced resilience to combined space stressors (microgravity, radiation) [64].
- Integrating multiple omics datasets (transcriptomics, proteomics, metabolomics) to model plant adaptation networks.
- Testing selected plant lines in advanced BLSS ground demonstrators and on the ISS to validate their performance for life support functions [62].
The workflow below outlines a comprehensive strategy for developing and validating microgravity-adapted plant lines for BLSS applications.
Maintaining equilibrium in closed ecological life support systems (CELSS) is paramount for ensuring the health of both plants and crew members. System imbalances, particularly in atmospheric composition and nutrient delivery, can rapidly compromise plant growth, gas processing capabilities, and the overall stability of the regenerative system. This application note provides researchers and drug development professionals with a standardized framework for the quantitative monitoring, prevention, and correction of gas fluctuations and nutrient toxicity in CELSS. The protocols detail the integration of real-time sensor data with plant physiological feedback to manage these critical imbalances, thereby supporting the reliability of life support research.
In a CELSS, higher plants are fundamental for providing food, regenerating major gases (CO₂ and O₂), reclaiming water, and purifying the atmosphere by removing toxic impurities produced by other ecological system components [27]. The closure of the system, however, makes it highly susceptible to internal fluctuations. Two of the most critical challenges are gas fluctuations, including the buildup of toxic volatile compounds, and nutrient toxicity within the plant growth subsystems. These imbalances can disrupt plant metabolic functions, reduce biomass yield, and ultimately jeopardize the system's ability to support human life. A quantitative biology approach—involving continuous measurement, statistical analysis, and predictive modeling—is essential to understand and manage these complex dependencies [71].
Quantitative Monitoring and Data Management
A rigorous, data-driven methodology is the cornerstone of identifying and addressing system imbalances. This involves the systematic collection and analysis of both quantitative and qualitative data.
Data Types and Acquisition
- Quantitative Data: These are numerical measurements critical for objective analysis. In CELSS research, this includes Electrical Conductivity (EC), pH, dissolved oxygen, gas concentration levels (e.g., CO₂, O₂, SO₂), and plant morphometric data acquired through high-throughput phenotyping [72] [73].
- Qualitative Data: These are descriptive observations, such as leaf color, leaf curling, or the presence of necrosis. For systematic analysis, these observations can be converted into quantitative data using standardized rating scales (e.g., 0 for no visible symptoms, 3 for severe chlorosis) [72].
Data Structuring and Analysis
Organizing data into clear tables is vital for trend identification. The table below provides a template for logging key nutrient solution parameters.
Table 1: Daily Log for Nutrient Solution and Atmospheric Monitoring
| Date & Time | EC (dS/m) | pH | Nutrient Temp (°C) | Air [CO₂] (ppm) | Air [SO₂] (ppb) | Qualitative Observations |
|---|---|---|---|---|---|---|
| [Timestamp] | 1.2 | 5.8 | 20.5 | 450 | <1 | Leaves appear healthy, deep green |
| [Timestamp] | 1.7 | 5.9 | 20.7 | 442 | 5 | Slight leaf tip chlorosis on lower leaves |
Statistical analysis and visualization tools (e.g., line graphs for EC over time, bar charts for comparative growth analysis) should then be used to identify correlations and trends, such as a rising EC coinciding with the appearance of visual toxicity symptoms [72].
Protocol 1: Managing Gas Fluctuations and Toxic Impurities
Plants in CELSS contribute to atmospheric stabilization not only through gas exchange but also via the active and passive absorption of toxic volatile substances like sulfur dioxide (SO₂) [27].
Experimental Workflow for Gas Exposure and Response
The following diagram outlines a protocol to quantify a plant canopy's capacity to absorb and process a gaseous toxicant.
Materials and Reagents
- Sealed Plant Growth Chamber: Capable of precise environmental control.
- Gas Analyzers: For target gases (e.g., SO₂, CO₂).
- Gas Cylinder: Containing the calibrated mixture of the target toxic gas.
- Data Logger: For continuous sensor data acquisition.
Step-by-Step Procedure
- System Stabilization: Establish a stable, sealed environment with a mature, mixed-plant canopy. Continuously monitor baseline atmospheric composition and photosynthetic rates.
- Gas Introduction: Introduce a precise, pulsed dose of the target gas (e.g., SO₂) into the sealed system.
- Continuous Monitoring: Record the concentration of the target gas at high frequency. Simultaneously, monitor the intensity of visible photosynthesis and document any changes in plant physiology.
- Data Modeling: Fit the gas concentration decay data to kinetic models. Passive absorption often follows saturation kinetics, while active biological processing may fit enzymatic (Michaelis-Menten) kinetics [27].
- Threshold Determination: Identify the maximum continuous infusion rate of the gas that does not cause significant damage to the biological components, thereby defining the system's absorptive capacity.
Protocol 2: Preventing and Correcting Nutrient Toxicity
Nutrient toxicity occurs when excessive amounts of a specific nutrient are present, leading to adverse health effects, blocking the uptake of other essential elements, and potentially causing plant death [74] [75].
Key Symptoms of Nutrient Toxicity
Accurate identification is crucial, as the remedy for a deficiency can exacerbate a toxicity issue.
Table 2: Visual Identification Guide for Common Nutrient Toxicities
| Nutrient | Key Visual Symptoms |
|---|---|
| Nitrogen (N) | Leaves turn light green; stalks become short, slender, and brittle; root systems overly dense [75]. |
| Phosphorus (P) | Stunted, abnormally dark green plants; may show purple pigmentation; can induce Zinc/Copper deficiency [75]. |
| Potassium (K) | Stunted growth, small leaves; chlorosis at leaf tips; purple veins/edges; can block Mg, Mn, Fe, Zn uptake [75]. |
| Calcium (Ca) | Severely stunted growth; crinkled or rosette leaves; interveinal white spots; marginal necrosis [75]. |
| Magnesium (Mg) | Symptoms on younger leaves: interveinal chlorosis, mottled appearance, upward leaf curling [75]. |
| Zinc (Zn) | Rapid yellowing of older leaves; stunted new growth; reddish-brown spots on middle-aged leaves [75]. |
Workflow for Correcting Nutrient Toxicity
The following protocol outlines the emergency response to a diagnosed nutrient toxicity.
Research Reagent and Equipment Solutions
Table 3: Essential Research Toolkit for Hydroponic System Management
| Item | Function/Benefit |
|---|---|
| EC & pH Meters | Provides quantitative data on nutrient solution strength and acidity/alkalinity, crucial for daily monitoring [76] [75]. |
| Calibration Solutions | Ensures accuracy and reliability of EC and pH meter readings. Regular calibration is non-negotiable [76]. |
| High-Quality Water Source | Reverse osmosis (RO) or deionized (DI) water is free of confounding ions, serving as the pure base for nutrient solutions and system flushing [75]. |
| Leachate Collection Saucers | Allows for easy collection of root zone effluent for accurate EC and pH testing, reflecting what the plant is actually experiencing [76]. |
| Sensor Network (WSN) | Wireless sensors monitoring microclimatic conditions (light, temperature, humidity) help correlate environmental fluctuations with nutrient uptake changes [73]. |
Step-by-Step Correction Protocol
- Immediate System Flush: Upon confirmation of toxicity, completely drain the nutrient solution from the hydroponic system. Refill the reservoir with pure, pH-neutral water (pH 7.0). Commercially available flushing agents may be used [75].
- Monitor Recovery: Allow the plants to take up the pure water for several days to a week, which helps leach excess nutrients from the root zone and plant tissues. Monitor for a halt in symptom progression and signs of new, healthy growth [75].
- Gradual Nutrient Reintroduction: After recovery is observed, reintroduce nutrients at a low dose (25-33% of normal strength). Increase the concentration incrementally each week over 3-4 weeks until normal levels are resumed, while vigilantly monitoring for any recurrence of symptoms [75].
Proactive prevention, centered on continuous quantitative monitoring and strict control of nutrient inputs, is the most effective strategy for managing CELSS imbalances. The protocols for gas impurity absorption and nutrient toxicity correction provide a standardized framework for researchers to maintain system health. The future of robust CELSS operation lies in moving from reactive correction to predictive management. This will be enabled by integrating high-throughput phenotyping [73], advanced biosensors for signaling molecules [71], and computational modeling that accounts for biological noise and feedback loops [71] to forecast system behavior and preemptively adjust parameters, ensuring long-term stability for closed ecological life support.
Application Notes & Protocols for Closed Ecological Life Support Systems (CELSS)
Water Use Optimization
Application Note: Regulating Plant Transpiration for Water Conservation
In CELSS, optimizing crop water use is critical for sustaining both plant productivity and crew water supplies. The primary strategy involves inducible limitation of maximum transpiration rates under high vapor pressure deficit (VPD) conditions, which conserves soil water for critical grain filling stages without significantly reducing yield potential [77].
Key Physiological Regulation Mechanisms:
- Root-based hydraulic regulation: Involves both radial and axial water conductance pathways
- Aquaporin-mediated transport: Membrane proteins that facilitate water movement across cells
- Stomatal regulation: Controls vapor diffusion while maintaining carbon fixation
- Root system architecture optimization: Balances water acquisition against carbon investment
Table 1: Transpiration Restriction Impact on Yield Under Drought Conditions
| Crop Species | Yield Improvement with Transpiration Restriction | Experimental Context |
|---|---|---|
| Maize | Significant increase | Severe terminal drought |
| Sorghum | Significant increase | Severe terminal drought |
| Pearl millet | Significant increase | Severe terminal drought |
| Wheat | Significant increase | Severe terminal drought |
| Soybean | Great benefit with little trade-off | Modeling studies |
Protocol: Wastewater Recycling for Vegetable Cultivation
Objective: Utilize biologically treated domestic wastewater as primary nutrient source for leafy vegetable production in CELSS [78].
Materials:
- Biologically treated domestic wastewater
- Hoagland solution (control)
- Ice plant (Mesembryanthemum crystallinum) seeds
- Lettuce (Lactuca sativa) seeds
- Hydroponic cultivation system
- Electrical conductivity (EC) meter
- pH meter
Methodology:
- System Setup: Establish parallel cultivation systems for recycled wastewater and Hoagland solution controls
- Planting: Sow ice plant and lettuce seeds in respective systems
- Environmental Monitoring:
- Maintain wastewater EC at ~6.39 mS·cm⁻¹
- Maintain weakly acidic pH throughout cultivation
- Growth Analysis (60-day period):
- Measure edible biomass yield per plant
- Quantify root development parameters
- Analyze nutrient assimilation rates
- Track sodium uptake efficiency
Expected Outcomes:
- Comparable edible biomass between wastewater and control systems
- Enhanced root development in wastewater-grown lettuce
- Higher sodium assimilation in ice plant (~13.12 m² plant area required to process one person's daily sodium excretion)
Wastewater Recycling Experimental Workflow
Energy Consumption Optimization
Application Note: Reducing Energy Input in Plant Production Systems
Energy balancing reveals significant differences between production approaches. Organic farming systems demonstrate 37-50% lower energy input compared to conventional systems, primarily due to avoidance of energy-intensive synthetic fertilizers and pesticides [79].
Table 2: Energy Use Efficiency Comparison: Organic vs. Conventional Systems
| Parameter | Organic Systems | Conventional Systems |
|---|---|---|
| Average Energy Input | 7.2 GJ ha⁻¹ | 14.0 GJ ha⁻¹ |
| Energy Input Range | 4.0-10.7 GJ ha⁻¹ | 10.0-17.1 GJ ha⁻¹ |
| Fertilization Approach | Biological N₂ fixation, manure | Mineral fertilizers |
| Plant Protection | Biological control, mechanical weeding | Chemical pesticides |
| Energy Efficiency | More efficient in 73.3% of cases | Less efficient in most cases |
Key Energy Reduction Strategies:
- Eliminate Haber-Bosch nitrogen: Biological N₂ fixation saves substantial energy
- Reduce mechanical operations: Optimize tillage intensity and frequency
- Implement precision farming: Target resources to minimize waste
- Utilize renewable energy: Replace fossil fuels in operation
Protocol: Low-Energy Fallow Management for Wheat Precursors
Objective: Implement modular harrow technology to reduce energy consumption during fallow preparation for winter wheat in CELSS crop rotations [80].
Materials:
- Modular fallow harrow with vertical flat-cutting blades
- Tractor with fuel consumption monitoring capability
- Soil moisture sensors
- Depth measurement apparatus
Technical Specifications:
- Tillage depth: 5-6 cm (significantly shallower than conventional 10+ cm)
- Blade configuration: Vertical spikes with horizontal blades
- Optimal parameters:
- Vertical inclination angle (ε): Optimized to reduce resistance
- Horizontal deviation angle (γ): Calculated based on ε
- Constraint: 0.6 ≤ tanε · tanγ ≤ 1.0
Methodology:
- Equipment Calibration:
- Set vertical inclination angle (ε) according to soil conditions
- Calculate corresponding horizontal deviation angle (γ)
- Verify tillage depth not exceeding 6 cm
- Field Operation:
- Conduct fallow tillage using modular harrow
- Monitor tractor fuel consumption continuously
- Measure soil moisture retention at depths 0-5 cm, 5-10 cm, 10-15 cm
- Performance Assessment:
- Compare fuel consumption with conventional disc harrows
- Evaluate weed control efficacy
- Quantify soil moisture preservation
Expected Outcomes:
- 25-35% reduction in fuel consumption compared to conventional tillage
- Improved soil moisture retention in critical upper layer (0-5 cm)
- Effective weed control while minimizing soil disturbance
Space Utilization Optimization
Application Note: Maximizing Cultivation Density in Controlled Environments
Efficient space utilization directly impacts production costs and system scalability in CELSS. Multiple strategies exist to increase plant density without compromising growth conditions [81].
Table 3: Space Utilization Efficiency of Greenhouse Layout Systems
| System Type | Floor Area Utilization | Key Advantages |
|---|---|---|
| Traditional straight row | 60-70% | Simple implementation |
| Peninsular bench layout | >75% | Improved access and space utilization |
| Movable bench system | >90% | Maximum space efficiency |
| Rack growing system | Can double growing space | Creates microclimate zones |
| Hanging basket system | Utilizes overhead space | Adjustable light exposure |
Protocol: High-Density Crop Planning and Bench Management
Objective: Implement strategic crop planning and movable bench systems to maximize production turns and space efficiency in CELSS [82].
Materials:
- Movable bench system or transport trays
- Spreadsheet or greenhouse management software
- Color-coded flag system for crop management
- Enhanced liner propagation materials
Methodology:
- Space Analysis:
- Calculate total inside growing space
- Inventory all benches, floors, and vertical spaces
- Determine space allocation percentages for different crop types
- Crop Plan Development:
- Determine product mix based on nutritional requirements and growth characteristics
- Establish specifications and target yields
- Create timeline with crop weeks clearly mapped
- Assign crops to appropriate spaces based on environmental needs
- Movable Bench Implementation:
- Install rolling benches or transport tray systems
- Establish weekly movement protocols
- Implement overhead trolley systems if space is constrained
- Crop Rotation Management:
- Use enhanced liners for rapid turnover (3-4 week cycles)
- Employ color-coded flags to signal plant status (blue for trim, yellow for spacing, green for movement)
- Conduct weekly team reviews of crop progress and space allocation
Expected Outcomes:
- 80-90% space utilization efficiency versus 60-70% with traditional layouts
- 15-25% increase in production turns annually
- More predictable crop timing and higher quality specifications
Space Optimization Strategy Relationships
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Research Materials for CELSS Resource Efficiency Studies
| Reagent/Material | Function in Research | Application Context |
|---|---|---|
| Hoagland solution | Controlled nutrient baseline for comparison studies | Wastewater recycling experiments |
| Enhanced 72 cell liners | Rapid crop turnover with uniform flowering response | Space utilization optimization |
| Ice plant (M. crystallinum) | Saline wastewater remediation studies | Sodium assimilation research |
| Modular fallow harrow | Reduced energy tillage implementation | Low-energy agriculture protocols |
| Soil moisture sensors | Precise water use efficiency monitoring | Transpiration regulation studies |
| VPD control systems | Regulate transpiration stress responses | Water conservation research |
| Movable bench systems | Maximize spatial efficiency in controlled environments | Density optimization experiments |
| Aquaporin expression assays | Analyze root hydraulic conductance mechanisms | Fundamental water transport research |
Integrated Resource Optimization Protocol
Objective: Simultaneously optimize water, energy, and space utilization in an integrated CELSS crop production module.
System Design Principles:
- Closed-loop integration: Wastewater nutrients feed plant systems, plant biomass supports other CELSS functions
- Multi-scale optimization: Coordinate molecular (aquaporins), organ (root-shoot), and whole-canopy processes
- Dynamic monitoring: Implement real-time tracking of all resource flows
- Adaptive control: Adjust systems based on performance metrics and crew needs
Implementation Framework:
- Establish baseline metrics for all three resource domains
- Implement coordinated optimization protocols
- Monitor system interactions and unintended consequences
- Refine protocols through iterative improvement cycles
Expected Outcomes:
- 30-50% reduction in external resource inputs
- Improved system resilience and redundancy
- Enhanced productivity per unit volume and energy input
- More sustainable long-term life support operation
The protocols and application notes presented herein provide a foundation for optimizing the critical resource domains in closed ecological life support systems. Continued refinement through empirical testing and system integration will enable more ambitious long-duration space missions and advance sustainable agricultural practices on Earth.
The success of Closed Ecological Life Support Systems (CELSS) hinges on the reliable cultivation of higher plants, which provide essential functions including food production, oxygen generation, carbon dioxide reduction, and water recycling [23]. Safeguarding plant health and productivity in the resource-limited and unique conditions of space is therefore paramount. Plants in CELSS environments face a multitude of potential abiotic stresses, such as water deficiency, nutrient imbalance, and atypical light or gravity conditions [26]. The early and accurate detection of stress responses is critical for initiating timely interventions to prevent crop loss and system instability.
High-Throughput Phenotyping (HTP) represents a transformative approach for crop improvement and the functional analysis of quantitative traits [83]. By deploying arrays of non-destructive sensors and automated imaging platforms, HTP enables the precise quantification of plant growth and physiological performance. This data-driven methodology is particularly valuable for evaluating transgenic lines, identifying plants with enhanced tolerance to biotic and abiotic stresses, and characterizing unknown gene functions [83]. Within the context of CELSS, integrating HTP technologies allows for the continuous, automated monitoring of plant status. This facilitates the early detection of stress signatures before they manifest as visible symptoms, enabling preemptive management strategies to maintain optimal gas balance and resource recovery, thereby contributing to the overall stability of the closed atmospheric system [27].
Application Notes: Stress Detection Technologies for CELSS
Plant stress responses manifest across a spectrum, from non-visible cellular changes to visible morphological symptoms. A comprehensive monitoring strategy for CELSS should integrate technologies capable of detecting responses at multiple levels.
Table 1: Technologies for Detecting Visible and Non-Visible Plant Stress Responses
| Stress Response Scale | Detection Technology | Measured Parameters | Relevance to CELSS Monitoring |
|---|---|---|---|
| Non-Visible (Cellular/Subcellular) | Molecular Bioassays (e.g., ELISA, Luminescence) [84] | Stress-related hormones (e.g., ABA), Heat shock proteins, Pathogen signals [84] | Early alarm phase detection; pathogen diagnostics. |
| Metabolomic & Proteomic Profiling (Mass Spectrometry) [84] | Stress-responsive metabolites and proteins, Pathogen toxins [84] | Mechanistic understanding of acclimation and resistance phases. | |
| Transcriptomic Analysis (High-throughput sequencing) [84] | Differential gene expression, Activation of stress-response pathways [84] | Identification of key genetic regulators for engineering tolerance. | |
| Visible (Whole-Plant/Organ) | High-Throughput Phenotyping (HTP) Platforms [83] | 3D Structure, Biomass, Water Content [83] | Primary tool for in-situ, non-destructive health assessment. |
| Chlorophyll Fluorescence Imaging [84] | PSII efficiency (Fv/Fm), Photoinhibition [84] | Quantification of abiotic stress impacts (nutrient, drought, heat). | |
| Hyperspectral & Multispectral Sensing [84] | Spectral reflectance profiles, Vegetation indices | Detection of pre-visual pigment and water content changes. |
The value of HTP was demonstrated in the functional analysis of the polyamine biosynthetic pathway. Tomato plants overexpressing the arginine decarboxylase 2 gene were analyzed using an HTP platform, which revealed significant differences in water content and the ability to recover after drought stress compared to wild-type lines [83]. This underscores the power of HTP for identifying and validating genetic modifications that confer enhanced resilience, a key pursuit for selecting ideal CELSS cultivars.
The following workflow outlines the logical process of applying these technologies for stress diagnosis and intervention within a CELSS.
Experimental Protocols
Protocol for HTP-Based Drought Stress Tolerance Screening
This protocol is adapted from methods used to identify tomato lines with enhanced drought tolerance through polyamine pathway manipulation [83]. It is designed for a controlled growth environment simulating CELSS modules.
I. Plant Material and Growth Conditions
- Plant Material: Use transgenic and wild-type control seeds. For the example, use tomato (Solanum lycopersicum) lines overexpressing the arginine decarboxylase 2 gene [83].
- Growth System: Utilize a controlled-environment growth chamber with programmable light, temperature, and humidity. Employ an HTP platform, such as a 3D Scanalyzer, equipped with RGB, fluorescence, and hyperspectral sensors [83].
- Baseline Conditions: Maintain optimal conditions (e.g., 22-25°C, 60-70% RH, 16/8h photoperiod, PPFD of 300 μmol m⁻² s⁻¹ at the canopy level [26]) until the treatment begins.
II. Drought Stress Application and Phenotyping
- Control Group: Continue daily irrigation at 100% pot capacity.
- Stress Treatment Group: Withhold irrigation completely.
- High-Throughput Imaging:
- Frequency: Image all plants daily from the start of the stress period until recovery.
- Modalities:
- RGB Imaging: Capture top and side views to quantify projected leaf area, plant architecture, and color changes.
- Chlorophyll Fluorescence Imaging: Measure the maximum quantum efficiency of PSII (Fv/Fm) in dark-adapted leaves to assess photoinhibition [84].
- Hyperspectral Imaging: Capture reflectance spectra to model water content and pigment composition.
III. Data Processing and Analysis
- Image Analysis: Extract traits using platform software. Key traits include:
- Biomass Accumulation: Derived from plant volume from RGB images.
- Water Use Efficiency: Calculated as biomass produced per unit water consumed.
- Drought Recovery Index: Calculated as the rate of biomass and Fv/Fm recovery upon re-watering [83].
- Statistical Analysis: Perform analysis of variance (ANOVA) to compare traits between transgenic and wild-type lines under stress and control conditions.
Protocol for Molecular Validation of Stress Responses
This protocol supports HTP data by quantifying key molecular stress markers, providing insight into the underlying physiological mechanisms.
I. Sample Collection
- Collect leaf discs (e.g., 100 mg) from both control and stressed plants at multiple time points (e.g., 0, 3, 5 days post-stress induction).
- Flash-freeze samples in liquid nitrogen and store at -80°C.
II. Chlorophyll Fluorescence Assay (Fv/Fm)
- Dark Adaptation: Attach leaf clips to leaves for at least 30 minutes before measurement.
- Measurement: Use a handheld chlorophyll fluorometer.
- Apply a saturating pulse of light to measure maximum fluorescence (Fm).
- Record the minimum fluorescence (Fo) with a measuring beam.
- Calculate Fv/Fm = (Fm - Fo) / Fm [84].
- Interpretation: A value below ~0.83 indicates the onset of abiotic stress [84].
III. Enzyme-Linked Immunosorbent Assay (ELISA) for Heat Shock Proteins
- Protein Extraction: Grind frozen tissue to a fine powder. Homogenize in extraction buffer and centrifuge to collect the supernatant.
- Procedure:
- Coat a microtiter plate with the protein sample.
- Add primary antibody specific to the target heat shock protein (e.g., HSP70).
- Add enzyme-conjugated secondary antibody.
- Add enzyme substrate to produce a colored reaction.
- Measure absorbance with a plate reader [84].
- Analysis: Quantify HSP concentration by comparing absorbance values to a standard curve.
The following diagram maps the key methodological stages and decision points in the integrated HTP and molecular validation workflow.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for HTP Stress Tolerance Experiments
| Item Name | Function / Application | Specific Example / Note |
|---|---|---|
| 3D HTP Platform (e.g., Scanalyzer) | Automated, non-destructive imaging of plant growth, architecture, and physiology over time [83]. | Integrates multiple sensors (RGB, fluorescence, NIR) for multi-dimensional phenotyping. |
| Chlorophyll Fluorometer | Measures photosynthetic efficiency (Fv/Fm), a sensitive indicator of abiotic stress [84]. | Can be imaging-based (on HTP platform) or handheld for spot measurements. |
| ELISA Kits (e.g., for HSPs) | Quantifies specific stress-responsive proteins (e.g., Heat Shock Proteins) for molecular validation [84]. | Provides high specificity and sensitivity for low-abundance proteins. |
| Antibodies (Primary & HRP-Secondary) | Essential components for immunoassays like ELISA, enabling specific detection of target antigens [84]. | Specificity of the primary antibody is critical for assay success. |
| Luminescence Assay Kits (e.g., for ROS/Ca²⁺) | Quantifies rapid signaling molecules like reactive oxygen species (ROS) and calcium ions [84]. | Useful for studying the initial "alarm phase" of the stress response. |
| Mass Spectrometry System | Enables comprehensive ionomic, metabolomic, and proteomic profiling of plant tissue under stress [84]. | Identifies nutrient imbalances and shifts in metabolic pathways. |
Validating System Performance and Comparing Plant Species & Technologies
In closed ecological life support system (CELSS) research, higher plants are integral to bioregenerative functions, contributing to food production, carbon dioxide reduction, oxygen production, water recycling, and waste management [23]. The viability of long-duration space missions and extraterrestrial habitats depends on the precise quantification of these metabolic fluxes. This document provides a standardized set of quantitative performance metrics and detailed experimental protocols for evaluating and comparing the carbon sequestration and oxygen production capacities of plant species within CELSS.
Quantitative Performance Metrics
The performance of plant species can be evaluated based on their carbon sequestration potential, which is directly linked to their photosynthetic efficiency and biomass accumulation. The tables below summarize key metrics for various plant types and systems.
Table 1: Carbon Sequestration Rates of Selected Landscape Tree Species [85]
| Species Category | Example Species | Daily Net C Sequestration per Unit Leaf Area, wCO₂ (μmol CO₂ m⁻² s⁻¹) | Daily Net C Sequestration per Unit Land Area, WCO₂ (g C m⁻² d⁻¹) | Daily Net C Sequestration of Whole Plant, QCO₂ (kg C plant⁻¹ d⁻¹) |
|---|---|---|---|---|
| Large Arbors (LA) | Populus spp. | High | High | High |
| Pterocarya stenoptera | High | High | High | |
| Platanus acerifolia | High | High | High | |
| Small Arbors (SA) | Viburnum odoratissimum | High | High | High |
| Prunus serratifolia | High | High | High | |
| Syringa oblata | High | High | High | |
| Shrubs (S) | Buxus sinica var. parvifolia | High | High | High |
| Bambusa megistophylla | High | High | High | |
| Ligustrum quihoui | High | High | High |
Table 2: Carbon Sequestration Potential of Different Systems and Crops
| System / Crop Type | Metric | Value | Context & Notes |
|---|---|---|---|
| General Plant Uptake | Global Annual CO₂ Absorption by Plants | 2.6 gigatons [86] | Provides scale for terrestrial biosequestration. |
| Individual Trees | Annual CO₂ Sequestration per Tree | 10 - 48 kg [86] | Varies significantly by species, age, and environment. |
| Afforestation | Annual CO₂ Sequestration per Hectare (1000 trees) | 10 - 48 tons [86] | Illustrates potential of large-scale planting. |
| Green Roofs | Annual CO₂ Sequestration per Square Meter | Up to 5 kg [86] | Offers dual benefits of carbon capture and reduced cooling energy needs. |
| Vertical Hydroponic Farming | Annual CO₂ Sequestration per 1000 m² | 36 - 45 tons [86] | Demonstrates high efficiency of controlled environment agriculture. |
| Perennial Energy Crops (PECs) | Annual Net SOC Storage Change (Herbaceous, e.g., Miscanthus) | 1.14 - 1.88 Mg C ha⁻¹ [87] | Exceeds minimum C-neutrality requirement (0.25 Mg C ha⁻¹ yr⁻¹). |
| Annual Net SOC Storage Change (Woody, e.g., Willow) | 0.63 - 0.72 Mg C ha⁻¹ [87] | Significant below-ground carbon storage in root systems. | |
| Enhanced Weathering (EW) with Agriculture | Projected US CDR Potential by 2050 | 0.16 - 0.30 GtCO₂ yr⁻¹ [88] | Geochemical carbon dioxide removal method using crushed silicate rocks on farmland. |
Key Factors and Experimental Protocols
Key Factors Influencing Carbon Sequestration
Research on landscape trees indicates that photosynthetic rate (Pn), crown area (CA), and leaf area index (LAI) are the key factors affecting comprehensive carbon sequestration potential for large arbors, small arbors, and shrubs [85]. Furthermore, the carbon sequestration rate is not linear throughout a tree's life; it typically follows a sigmoid curve, with rapid growth and high CO₂ absorption rates in the first decade [89].
Protocol: Calculating CO₂ Sequestration for Individual Trees
This protocol allows for the estimation of total lifetime CO₂ sequestration based on tree physical measurements [89].
1. Measure Fundamental Parameters:
- Diameter at Breast Height (D): Measure the tree's diameter at 1.37 meters from the ground (in inches or cm).
- Tree Height (H): Measure the total tree height (in feet or meters).
- Tree Age (A): Determine the age of the tree (in years).
2. Calculate Above-Ground Biomass (AGB):
- Use the allometric equation: AGB (lbs) = 0.25 × D² × H [89], where D is in inches and H is in feet.
3. Calculate Below-Ground Biomass (BGB) and Total Biomass:
- BGB is often estimated as 20% of AGB: BGB = 0.2 × AGB.
- Total green biomass is: Total Biomass (TB) = AGB + BGB = 1.2 × AGB.
4. Determine Dry Biomass and Carbon Weight:
- Average dry matter content is ~72.5%: Total Dry Weight (TDW) = TB × 0.725.
- Carbon constitutes ~50% of dry matter: Total Carbon (TC) = TDW × 0.5.
5. Calculate Sequestered CO₂:
- Convert carbon to CO₂ using the ratio of their molecular weights (44/12 ≈ 3.67): CO₂ weight = TC × 3.67.
- This result represents the total CO₂ sequestered over the tree's lifetime. For the annual rate, divide by the tree's age.
Protocol: Assessing Carbon Sequestration via Assimilation Method
This method quantifies carbon sequestration by measuring the net difference between photosynthetic carbon gain and respiratory carbon loss, providing a dynamic and theoretically accurate assessment [85].
1. Plant Material and Site Selection:
- Select healthy, mature plants of the species under investigation, growing under consistent climatic and management conditions.
- Ensure plants are free from obvious disease and pest damage.
2. Determination of Photosynthetic and Growth Indexes:
- Photosynthetic Rate (Pn): Use a portable photosynthetic system (e.g., LCpro SD) to measure the instantaneous net photosynthetic rate. Conduct measurements on clear, windless days from 8:00 to 18:00 at 2-hour intervals. Select several trees per species and several mature, well-grown leaves per tree. Record multiple instantaneous Pn values once the instrument stabilizes and calculate the average [85].
- Growth Indexes: Measure Diameter at Breast Height (DBH) and tree height (H) using a girth tape and laser rangefinder, respectively. Measure crown diameter in east-west and north-south directions to calculate crown area (CA) [85].
- Leaf Area Index (LAI): Capture canopy photos using a digital camera with a fisheye lens positioned at a set height above the ground. Analyze the images using software like Gap Light Analyzer (GLA) to determine LAI [85].
3. Calculation of Daily Net Carbon Assimilation:
- The daily net assimilation amount of photosynthesis is used to estimate the plant's daily carbon sequestration. Based on the calculation principle of daily assimilation, the carbon sequestration can be derived from the integrated photosynthetic data collected over the day [85]. Specific formulas are applied to calculate:
- Daily net carbon sequestration per unit leaf area (wCO₂)
- Daily net carbon sequestration per unit land area (WCO₂)
- Daily net carbon sequestration of the whole plant (QCO₂)
Visualization of Metabolic Pathways and Experimental Workflow
Plant Metabolic Pathway in a CELSS
This diagram illustrates the primary metabolic pathways through which plants contribute to life support functions, highlighting the interconnected cycles of carbon, oxygen, and water.
Experimental Workflow for Quantifying Carbon Sequestration
This workflow outlines the key steps for conducting a quantitative assessment of a plant species' carbon sequestration potential using the assimilation method.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Equipment for CELSS Plant Research
| Item | Category | Function & Application in CELSS Research |
|---|---|---|
| Portable Photosynthesis System (e.g., LCpro SD) | Instrumentation | Measures instantaneous net photosynthetic rate (Pn), transpiration rate (Er), stomatal conductance (Gs), and intercellular CO₂ concentration (Ci) in real-time under field or controlled conditions [85]. |
| Chlorophyll Meter (e.g., SPAD-502 Plus) | Instrumentation | Provides a rapid, non-destructive estimation of leaf chlorophyll content, which correlates with photosynthetic potential and plant nitrogen status [85]. |
| Digital Camera with Fisheye Lens | Instrumentation | Used to capture hemispherical canopy photographs for subsequent software analysis to determine Leaf Area Index (LAI), a critical structural parameter [85]. |
| Laser Rangefinder / Ultrasonic Hypsometer | Instrumentation | Accurately measures tree height (H) and crown dimensions for calculating crown area (CA) and overall plant biomass estimates [85]. |
| Diameter Tape (D-tape) | Tool | A simple, essential tool for measuring tree diameter at breast height (DBH), a fundamental variable in allometric biomass equations [89]. |
| Gap Light Analyzer (GLA) Software | Software | Analyzes hemispherical canopy photographs to quantify LAI and light transmission through the canopy, informing on light capture efficiency [85]. |
| Allometric Equations | Model | Species-specific mathematical models that relate easily measured variables like DBH and H to harder-to-measure properties like AGB, BGB, and total carbon storage [89]. |
Application Notes
The Role of Crop Species in Closed Ecological Life Support Systems (CELSS)
Within Closed Ecological Life Support Systems (CELSS), higher plants are indispensable for bioregenerative life support. They perform multiple critical functions: food production, oxygen generation, carbon dioxide reduction, water recycling, and waste management [90]. The selection of appropriate plant species is therefore paramount to system stability and crew well-being, impacting both physiological health and psychological morale during long-duration space missions [90]. CELSS are designed to be closed to matter, relying on internal recycling processes, but open to energy, typically from solar sources, driving the system far from thermodynamic equilibrium [33]. This review provides a comparative analysis of selected crop species, with a specific focus on pea cultivars as a model organism, and outlines standardized protocols for their evaluation in CELSS-relevant research.
Key Considerations for Crop Selection and Comparative Analysis
A robust comparative analysis of candidate CELSS crops must extend beyond mere yield metrics. The following facets are critical for a holistic evaluation:
- Resource Recycling Efficiency: A primary challenge for lunar or Martian CELSS is the recycling of key bio-elements. While water and oxygen can be sourced from lunar in-situ resources, elements like Carbon (C), Nitrogen (N), Phosphorus (P), Sulfur (S), and Potassium (K) are rarified and must be efficiently recycled within the system [33]. Crop species vary in their nutrient uptake and incorporation into edible biomass, directly impacting system closure.
- Growth and Morphological Characteristics: Plant architecture, such as the leaf shape in peas, influences light interception and canopy structure. Studies have identified quantitative trait loci (QTLs) like qLeafor-1 and qLeafor-2 on linkage group LG7 that control whether a plant has the afila (leafless, with enhanced tendrils) or tendrilless leaf type, which can affect the plant's light utilization efficiency [91].
- Environmental Response and Genetic Regulation: The growth responses of plants to controlled environmental stresses (e.g., specific light spectra) are a key selection criterion. For instance, the phytochrome-mediated growth responses in dwarf ('Progress No. 9') and tall ('Alaska') pea cultivars show varietal differences in the stability and activity of the Pfr (far-red light-absorbing) form of phytochrome, a key photomorphogenic pigment [92]. This molecular-level understanding is crucial for optimizing growth in artificial lighting conditions.
Table 1: Key Nutritional and Growth Metrics for CELSS Candidate Crops
| Crop Species | Edible Biomass Yield (kg/m²/year) | Caloric Density (kcal/100g) | O2 Production Rate (g/day/plant) | Water Transpiration Rate (L/day/plant) | Notable Traits |
|---|---|---|---|---|---|
| Pea (Pisum sativum) | Data Needed | ~81 | Data Needed | Data Needed | High protein; Genetic models available |
| Leafy Greens (e.g., Lettuce) | Data Needed | ~15 | Data Needed | Data Needed | Rapid cycle; High vitamin content |
| Root Vegetable (e.g., Potato) | Data Needed | ~77 | Data Needed | Data Needed | High calorie; Staple crop |
Table 2: Essential Elements for Human Consumption and Challenges in CELSS Recycling (based on [33])
| Element | Human Daily Requirement | Role in Human Physiology | Status in Lunar Regolith |
|---|---|---|---|
| Carbon (C) | Major component of food | Organic molecules | Scarce |
| Nitrogen (N) | Major component of food | Proteins, nucleic acids | Scarce |
| Potassium (K) | ~3.5 g | Electrolyte, nerve function | Scarce |
| Phosphorus (P) | ~1.5 g | Bone, ATP, nucleic acids | Scarce |
| Iron (Fe) | ~14 mg | Oxygen transport in blood | Available via extraction |
| Calcium (Ca) | ~1 g | Bone structure, signaling | Available via extraction |
Experimental Protocols
Protocol 1: Analysis of Phytochrome-Mediated Growth Responses in Pea Cultivars
This protocol details the methodology for comparing growth responses between dwarf and tall pea cultivars under different light regimes, based on classical experiments [92].
Research Reagent Solutions
Table 3: Key Research Reagent Solutions for Phytochrome Analysis
| Reagent / Material | Function / Application | Exemplary Varieties / Notes |
|---|---|---|
| Dwarf Pea Seeds | Model for compact growth habit; e.g., 'Progress No. 9' [93] | 'Little Marvel' (15 inches tall) [93] |
| Tall Pea Seeds | Model for standard growth; e.g., 'Alaska' [92] | 'Lincoln', 'Green Arrow' (2-3 foot vines) [93] |
| Red Light Source | Activates phytochrome (converts Pr to Pfr) to inhibit elongation | Wavelength ~660 nm |
| Far-Red Light Source | Reverts phytochrome (Pfr to Pr) to reverse red light effect | Wavelength ~730 nm |
| Plant Genomic DNA Kit | Extraction of high-quality DNA for subsequent genetic studies | Used in modern genotyping [91] |
Experimental Workflow
The following diagram outlines the key stages of the phytochrome response experiment.
Step-by-Step Procedure
- Plant Material and Growth Conditions: Sow seeds of dwarf (e.g., 'Progress No. 9') and tall (e.g., 'Alaska') pea cultivars in a standardized growth medium. Grow seedlings in complete darkness at a constant temperature (e.g., 22-25°C) for approximately 7-10 days until the third internode begins to develop [92].
- Light Treatment Application: Divide seedlings into experimental groups.
- Group 1 (Red Light): Expose etiolated seedlings to a single pulse of red light (e.g., 5 minutes, 660 nm).
- Group 2 (Red/Far-Red Reversal): Expose a subset of the red-light-treated seedlings to a subsequent pulse of far-red light (e.g., 5 minutes, 730 nm) immediately or after a defined time interval.
- Group 3 (Dark Control): Maintain a group in complete darkness.
- Growth Measurement: 24 hours post-irradiation, meticulously measure the elongation of the 10-mm sub-apical segments of the third internodes in all groups using digital calipers [92].
- Data Analysis: Calculate the percentage inhibition of elongation in red-light-treated groups compared to the dark control. Quantify the extent of far-red reversal. Compare the time-courses of growth inhibition and recovery between the dwarf and tall cultivars.
Protocol 2: Genetic Mapping of Leaf Shape Traits in Pea
This protocol describes a modern approach using high-throughput sequencing to identify genetic markers linked to leaf shape, an important trait for light capture efficiency in CELSS [91].
Research Reagent Solutions
Table 4: Key Research Reagent Solutions for Genetic Mapping
| Reagent / Material | Function / Application | Notes |
|---|---|---|
| Pea Genotypes (PS002, PS047) | Parents with contrasting afila and tendrilless leaf types [91] | Used to create F2 mapping population |
| Restriction Enzymes (RsaI, HaeIII) | Digest genomic DNA for SLAF-seq library construction [91] | Part of Specific Locus Amplified Fragment Sequencing |
| DNA Ligase & Polymerase | Ligate adapters and amplify libraries for sequencing [91] | Essential for SLAF-seq workflow |
| Illumina HiSeq Platform | High-throughput sequencing of SLAF libraries [91] | Generates the raw SNP data |
Experimental Workflow
The diagram below illustrates the integrated workflow for QTL mapping and Bulked Segregant Analysis (BSA).
Step-by-Step Procedure
- Population Development: Cross two pea genotypes with distinct leaf shapes (e.g., PS002, afila type, and PS047, tendrilless type) to generate an F2 mapping population [91].
- Phenotyping: Systematically evaluate and record the leaf shape (afila vs. tendrilless) for each individual in the F2 population at a defined growth stage.
- Genotyping by SLAF-seq:
- DNA Extraction: Extract high-quality genomic DNA from all F2 individuals and parents.
- Library Prep: Digest DNA with RsaI and HaeIII. Ligate sequence adapters, amplify via PCR, and select fragments of a specific size range (e.g., 264-314 bp) for pair-end sequencing on an Illumina platform [91].
- Data Analysis: Process raw reads to identify and genotype SLAF markers and SNPs. Construct a high-density genetic map using polymorphic markers.
- QTL and BSA Analysis: Perform QTL mapping on the F2 population using the genetic map and phenotypic data. In parallel, conduct BSA by sequencing DNA bulks from individuals with extreme phenotypes. Integrate results from both methods to identify robust SNP markers and QTLs (e.g., qLeafor-1, qLeafor-2) associated with leaf shape [91].
Protocol 3: Cultivation and Nutritional Analysis of Pea Shoots and Tendrils
This protocol covers the cultivation and post-harvest handling of peas, including non-traditional edible parts relevant for maximizing food production in CELSS [93].
Procedure
- Cultivation: Grow pea varieties (shelling, snap, or snow) under controlled environmental conditions. Provide support for vining types if necessary.
- Harvesting of Non-Traditional Parts:
- Tendrils: Harvest young, tender tendrils.
- Shoots: Harvest young pea shoots when they are 12 to 18 inches out of the ground [93].
- Post-Harvest Handling: Consume or analyze shoots and tendrils soon after harvest, as they rapidly lose sweetness and toughen. They can be added raw to salads or stirred into dishes like stir-fries at the end of cooking to preserve texture and nutrients [93].
- Nutritional Analysis: Perform standard nutritional profiling (e.g., for vitamin, mineral, and protein content) on the edible shoots and tendrils to quantify their contribution to a balanced diet.
Validating Drought Tolerance and Resource Use Efficiency with High-Throughput Platforms
Within the context of Closed Ecological Life Support Systems (CELSS), the efficient cultivation of plants is paramount for regenerating resources, stabilizing atmospheric composition, and providing food [23] [27]. Success in these systems depends on selecting and breeding plant varieties that exhibit superior resource use efficiency (RUE) and resilience to abiotic stresses, such as drought, within confined and controlled environments [26]. Traditional phenotyping methods are often destructive, labor-intensive, and insufficient for the large-scale, precise measurements required for CELSS research and development. The advent of High-Throughput Plant Phenotyping Platforms (HT3Ps) offers a transformative solution, enabling non-destructive, automated, and quantitative assessment of complex plant traits from the laboratory to the field [94] [95]. This Application Note details the integration of HT3P technologies for validating drought tolerance and RUE, providing robust protocols and data analysis frameworks essential for advancing the selection of ideal cultivars for bio-regenerative life support.
High-Throughput Plant Phenotyping Platforms (HT3Ps) are automated systems designed to collect massive amounts of phenotypic data from hundreds of plants daily [94]. They leverage a suite of non-invasive sensors to monitor and quantify plant growth, physiology, and biochemistry in response to environmental conditions, thereby bridging the gap between genomics and phenomics for accelerated breeding [95].
Platform Configurations and Operating Modes: HT3Ps can be broadly categorized based on their deployment environment and operational design.
- Indoor Platforms (Greenhouses/Growth Chambers): These systems operate under strictly controlled environmental conditions, allowing for the precise study of plant responses to specific stressors. They typically function in one of two modes:
- Conveyor-type ("Plant-to-Sensor"): Potted plants are automatically transported on a conveyor belt to an imaging chamber for data acquisition before being returned to their growth positions [94].
- Benchtop-type: Imaging sensors are moved over stationary plants, which is often more suitable for smaller-scale or specialized experiments [94].
- Field-Based Platforms: These are deployed in agricultural settings to assess plant performance under real-world conditions.
- Ground-based Proximal Phenotyping: Vehicles or manual carts equipped with sensors are used to monitor crops at close range.
- Aerial Remote Sensing: Unmanned aerial vehicles (UAVs or drones) equipped with various imagers are used for large-scale field phenotyping, enabling the coverage of vast areas and the assessment of canopy-level traits [94].
The following table summarizes the primary sensor technologies used in HT3Ps and the key agronomic traits they measure.
Table 1: Common Sensors in HT3P and Their Applications in Drought and RUE Assessment
| Sensor Type | Key Measurable Parameters | Relevance to Drought Tolerance & RUE |
|---|---|---|
| RGB (Red, Green, Blue) | Plant height, biomass, canopy coverage, leaf area, lodging [94]. | Tracks growth reduction and architectural changes under water deficit. |
| Infrared (IR) / Thermal | Canopy temperature, stomatal conductance, water stress index [95]. | Identifies stomatal closure and increased leaf temperature as early signs of drought stress. |
| Fluorescence (FLUO) | Chlorophyll fluorescence, photosynthetic efficiency (e.g., Fv/Fm) [94]. | Assesses the impact of stress on the photosynthetic apparatus and overall plant health. |
| Hyperspectral / Multispectral | Vegetation indices (e.g., NDVI), chlorophyll content, nitrogen content, water content [94]. | Quantifies pigment composition, nutrient status, and leaf water potential, all critical for RUE. |
| Near-Infrared (NIR) | Water content, soluble solids, internal composition [94]. | Directly measures water status and accumulation of osmolytes like soluble sugars. |
Application Notes & Experimental Protocols
Protocol 1: High-Throughput Screening for Drought Tolerance at the Germination and Seedling Stage
This protocol, adapted from screening methods in lettuce and squash, provides a semi-automated workflow for rapidly identifying drought-tolerant lines in a large germplasm collection [96] [97].
1. Plant Material Preparation:
- Plant Material: A large collection of germplasm (e.g., 3,000+ lines) such as an EMS-mutagenized population or a diverse germplasm panel.
- Planting: Fill multi-celled trays (e.g., 128-cell trays with cells 3 cm x 3 cm x 5 cm) with a standardized, well-draining soil mixture. Compress the soil using an empty tray to ensure uniform filling.
- Sowing: Sow seeds at a uniform depth (e.g., 1/4 inch for lettuce). For pumpkin, seeds can be first imbibed for 16 hours in distilled water (control) or a stress solution before sowing on filter paper [96] [97].
2. Experimental Design and Stress Application:
- Control and Treatment: Establish both well-watered control and drought stress treatments.
- Stress Induction: After germination, withhold water entirely to impose a rapid, severe drought stress. Alternatively, for a more controlled onset, water with a solution containing osmotic agents like PEG to simulate drought.
- Environmental Control: Maintain all plants in a growth chamber or greenhouse with tightly controlled light, temperature, and humidity to minimize confounding environmental variation [97].
3. High-Throughput Phenotyping and Data Collection:
- Imaging Schedule: Use a conveyor-type or benchtop HT3P system to image all plants immediately before stress application and then at regular intervals throughout the stress period (e.g., daily).
- Sensor Suite: Acquire images using RGB, fluorescence, and hyperspectral sensors to capture a wide range of physiological responses.
- Key Phenotypic Traits:
- Relative Water Content (RWC): A key indicator of water status. Destructive sampling may be required for validation.
- Wilting Score: Visually scored or determined via image analysis of canopy structure from RGB images.
- Growth Differential: Calculate the relative difference in projected leaf area or biomass (estimated from top-view images) between stressed and control plants [97].
- Chlorophyll Fluorescence: Use FLUO imaging to monitor the efficiency of photosystem II (Fv/Fm) as a measure of photosynthetic health under stress [94].
4. Data Analysis and Candidate Selection:
- Image Analysis: Utilize integrated software platforms to extract quantitative traits from the image data (e.g., leaf area, chlorophyll fluorescence indices, vegetation indices).
- Selection Criteria: Identify candidate tolerant lines based on a combination of high RWC, minimal wilting, and sustained growth under stress conditions compared to the population mean and sensitive controls. The goal is to narrow the large population down to a manageable number of elite candidates for further, more detailed physiological and genetic analysis [97].
The workflow for this protocol is summarized in the following diagram:
Protocol 2: Root Phenotyping for Resource Use Efficiency
Root system architecture (RSA) is a critical determinant of a plant's ability to efficiently acquire water and nutrients, but it is notoriously difficult to measure. The following protocol outlines methods for high-throughput root phenotyping.
1. Plant Growth Systems:
- Rhizotrons / Rhizoslides: Transparent growth containers filled with soil or a growth medium, allowing for non-destructive, repeated imaging of root development against a transparent surface over time [98].
- Agar Plates: Plants are grown on vertically oriented Petri dishes containing a nutrient-rich agar gel. This allows for complete visualization of the entire root system in 2D and is ideal for studying root architecture traits like primary root length, lateral root density, and root hair development [98].
- 3D Imaging Systems (X-ray CT, MRI): For a more sophisticated, non-destructive 3D assessment of roots in soil, X-ray Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) can be used. These technologies are highly expensive but provide unparalleled detail of root-soil interactions [98].
2. Phenotyping and Image Acquisition:
- 2D Imaging: For rhizotrons and agar plates, use high-resolution, calibrated RGB scanners or cameras to capture root images at regular intervals.
- 3D Imaging: For X-ray CT or MRI, follow the specific scanning protocols and parameter settings (e.g., resolution, exposure time) as defined by the facility and research objectives [98].
3. Image Analysis and Trait Quantification:
- Software Tools: Use specialized root image analysis software (e.g., SmartRoot, RootNav, ChronoRoot) to automatically or semi-automatically trace root systems and extract architectural traits [98].
- Key RSA Traits:
- Root Length Density: Total root length per unit volume of soil.
- Rooting Depth: Maximum depth the root system reaches.
- Root Angle: The angle of seminal or nodal roots, which determines soil exploration breadth versus depth.
- Root Diameter: Average diameter of roots.
- Lateral Root Density: Number of lateral roots per unit length of the main root.
4. Correlation with Resource Uptake:
- Correlate the quantified RSA traits with physiological measurements of RUE, such as Water Use Efficiency (WUE)—measured as biomass accumulated per unit water transpired—or Nutrient Uptake Efficiency under resource-limited conditions [98].
Data Integration and Molecular Validation
Phenotypic data from HT3Ps is most powerful when integrated with genomic information to uncover the genetic basis of complex traits.
1. Quantitative Trait Loci (QTL) Mapping:
- Population: Develop a mapping population, such as Recombinant Inbred Lines (RILs), from a cross between drought-tolerant and drought-sensitive parents.
- Genotyping: Use high-throughput genotyping methods like Genotyping-by-Sequencing (GBS) to identify thousands of single nucleotide polymorphisms (SNPs) across the population. A study in chickpea using this approach identified 3,237 high-quality SNPs for map construction [99].
- Phenotyping: Phenotype the entire RIL population under drought stress using the HT3P protocols described above.
- QTL Analysis: Statistically link the genotypic data (SNPs) with the phenotypic data to identify genomic regions (QTLs) associated with drought tolerance traits. For example, a study in maize identified 22 QTLs for drought-related traits, including one for sugar concentration that explained 52.2% of the phenotypic variation [100].
2. Identification of Candidate Genes:
- Within the confidence intervals of the identified QTLs, search the genome annotation to generate a list of putative candidate genes.
- Filter these genes using in-silico expression profiling (analyzing RNA-seq data from stressed vs. control tissues) and haplotype analysis to pinpoint genes with expression patterns and allelic variations that correlate with the tolerant phenotype [99].
The relationship between high-throughput phenotyping, genomics, and candidate gene identification is illustrated below:
Quantitative Data from Trait Mapping Studies
The following table consolidates exemplary quantitative data from QTL mapping studies for drought tolerance in crops, demonstrating the power of combining high-density genotyping with precise phenotyping.
Table 2: Examples of QTLs for Drought Tolerance Traits Identified via High-Throughput Phenotyping and Genotyping
| Crop | Trait | Number of QTLs Identified | Phenotypic Variance Explained (PVE) Range | Chromosomal Location | Key Findings | Source |
|---|---|---|---|---|---|---|
| Chickpea | Membrane Stability Index (MSI) | 1 | >90% | Linkage Group 7 | A major QTL for a key physiological trait; co-located with a QTL for yield. | [99] |
| Chickpea | Yield under Drought | 1 | >10% | Linkage Group 7 | A robust QTL for maintaining yield under terminal drought. | [99] |
| Maize | Sugar Concentration | 1 | 52.2% | Chromosome 6 | Highlights the role of osmolyte accumulation in drought tolerance. | [100] |
| Maize | Grain Yield | 3 | ~75% (combined) | Chromosomes 1, 5, 9 | Demonstrated the complex, polygenic nature of yield under stress. | [100] |
| Maize | Leaf Surface Area | 9 | 25.8% - 42.2% | Chromosomes 3, 9 | Multiple QTLs control leaf morphology, which affects transpiration. | [100] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for High-Throughput Drought Phenotyping
| Item | Function/Application | Example Usage in Protocol |
|---|---|---|
| EMS (Ethyl Methanesulfonate) | Chemical mutagen used to create genetic diversity for forward genetics screens. | Generation of mutant populations (e.g., 3,751 M2 lines of squash) for screening tolerant mutants [96]. |
| GBS (Genotyping-by-Sequencing) Kit | A reduced-representation sequencing method for high-throughput SNP discovery and genotyping. | Constructing high-density genetic maps for QTL mapping in populations like RILs (e.g., used in chickpea and maize studies) [100] [99]. |
| ApeKI Restriction Enzyme | Enzyme used in GBS library preparation to digest genomic DNA for reduced representation. | Part of the GBS protocol for SNP identification [99]. |
| Agarose/Gel Growth Medium | A transparent, gel-like matrix for supporting plant growth in 2D root phenotyping systems. | Used in agar plates for visualizing and quantifying root system architecture (RSA) [98]. |
| PEG (Polyethylene Glycol) | An osmotic agent used to simulate drought stress by lowering the water potential of the growth medium. | Applying controlled water deficit in laboratory-based screening of germination and seedlings [97]. |
| Rhizotron Vessels | Specialized, transparent containers that allow for non-destructive root imaging against a viewing surface. | For monitoring root development over time in a semi-soil environment [98]. |
Benchmarking Against Conventional and Organic Agricultural Practices
The development of robust Closed Ecological Life Support Systems (CELSS) is fundamental for long-duration human space exploration, enabling the regeneration of essential resources through biological processes. Higher plant cultivation plays a uniquely critical role, contributing to food production, carbon dioxide reduction, oxygen production, water recycling, and waste management [23]. Selecting and optimizing agricultural practices for these controlled environments is therefore paramount. This document establishes Application Notes and Protocols for benchmarking Conventional and Organic agricultural practices within the specific constraints of CELSS. The objective is to provide researchers with standardized methodologies to evaluate these practices based on quantitative crop performance data, resource utilization efficiency, and system integration requirements, thereby informing the design of sustainable bio-regenerative life support systems [23].
Comparative Analysis of Agricultural Practices
The following tables provide a structured comparison of Conventional and Organic practices across key performance metrics relevant to CELSS.
Table 1: Crop Performance and Nutritional Metrics
| Metric | Conventional Practice | Organic Practice | Measurement Protocol |
|---|---|---|---|
| Yield (Fresh Weight) | Typically higher yields per unit area [101] | Generally lower yields compared to conventional [101] | Harvest and weigh edible biomass at maturity. Record time from planting to harvest. |
| Growth Rate | Faster initial growth often observed due to readily available synthetic nutrients. | Slower, more sustained growth patterns. | Non-destructive monitoring via high-throughput phenotyping (e.g., rosette area for lettuce) [73]. |
| Nutritional Content (Antioxidants) | Standard levels. | Tendency for higher levels of certain antioxidants and Vitamin C [102]. | Laboratory analysis of harvested tissue (e.g., HPLC for specific antioxidants). |
| Pesticide Residues | Detectable synthetic pesticide residues [101]. | Lower or non-detectable synthetic pesticide residues [101]. | Standardized chemical residue analysis on produce. |
Table 2: Resource Input and Environmental Impact
| Metric | Conventional Practice | Organic Practice | Measurement Protocol |
|---|---|---|---|
| Fertilizer Input | Synthetic, water-soluble fertilizers (e.g., NH₄NO₃, KNO₃). | Organic amendments (e.g., compost, leguminous cover crops). | Log mass/volume of all inputs. Monitor nutrient solution EC and pH in hydroponic systems. |
| Water Usage & Pollution Risk | Higher risk of water pollution via synthetic fertilizer runoff; efficient water use possible with precision agriculture [101]. | Lower risk of chemical water pollution; promotes water retention via soil organic matter [102]. | Measure total water input vs. biomass output. Analyze leachate for nitrate/phosphate levels. |
| Impact on Soil/Biological Health | Can lead to soil degradation and loss of biodiversity over time [102] [101]. | Enhances soil health, fertility, and promotes biodiversity [102] [101]. | In situ microbial activity assays; root architecture analysis. |
Experimental Protocols for CELSS Benchmarking
High-Throughput Plant Phenotyping for Growth Analysis
Principle: Automated, non-invasive imaging allows for precise quantification of plant growth and performance dynamics, essential for detecting subtle genotypic and treatment effects in CELSS candidate crops [73].
Materials:
- Plant-to-sensor or sensor-to-plant high-throughput phenotyping system (e.g., LemnaTec Scanalyzer, PlantScreen) [73].
- Standardized growth containers and substrate.
- Reference plant lines (e.g., Arabidopsis, lettuce, turnip).
- Automated watering and weighing stations.
Procedure:
- Experimental Design: Employ sufficient randomization and replication to account for micro-environmental inhomogeneities within the growth chamber. Use a randomized complete block design.
- Seed Sowing & Germination: Sow seeds of uniform size and quality to minimize variability. Use simultaneously propagated seed material [73].
- Automated Imaging: Transfer plants to the phenotyping system. Acquire daily top-view and side-view images under standardized lighting conditions.
- Image Analysis: Process images through established pipelines (e.g., IAP, Rosette Tracker) to extract morphological traits [73]:
- Projected Leaf Area (PLA)
- Rosette Diameter
- Compactness
- Biovolume
- Data Integration: Correlate extracted image-derived traits with final destructive harvest data (fresh/dry weight) to build growth models.
Gas Exchange Measurements for Atmospheric Management
Principle: Higher plants in a CELSS must maintain atmospheric balance for the crew by absorbing CO₂ and producing O₂ through photosynthesis. Quantifying these rates is critical for system sizing and modeling [26].
Materials:
- Sealed plant growth chamber with integrated gas sensors (CO₂, O₂).
- Controlled light source (PPFD of ~300 μmol/m²/s is sufficient for lettuce/turnip) [26].
- Data logging system.
- Temperature and humidity control.
Procedure:
- System Calibration: Place a known number of plants (e.g., lettuce at 25 plants/m² density) into the sealed chamber [26]. Allow to acclimate under standard light.
- Baseline Measurement: Seal the chamber and record initial CO₂ and O₂ concentrations.
- Light Period Measurement: Illuminate the plants at the target PPFD. Monitor and log the decrease in CO₂ and increase in O₂ concentration over a defined period (e.g., 1-2 hours).
- Dark Period Measurement: Turn off the light source. Monitor and log the increase in CO₂ and decrease in O₂ due to plant respiration.
- Calculation: Calculate the rates of CO₂ absorption and O₂ release (in μmol/m²/s or per plant) for both light and dark periods.
Multi-Spectral Imaging for Crop Health Assessment
Principle: UAVs or fixed sensors with multispectral cameras can be adapted for CELSS to monitor crop health and spatial uniformity using vegetation indices like the Normalized Difference Vegetation Index (NDVI) [103].
Materials:
- Multispectral camera (capturing Red and Near-Infrared bands).
- Imaging platform (UAV or fixed gantry system).
- Image processing software (e.g., with neural network support like Detectron2) [103].
Procedure:
- Image Acquisition: Capture co-registered images in the Red (R) and Near-Infrared (NIR) bands over the crop canopy.
- Index Calculation: Compute the NDVI for each pixel in the image: NDVI = (NIR - R) / (NIR + R) [103].
- Analysis & Segmentation:
- Validation: Correlate NDVI values with chlorophyll content measured via destructive sampling.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for CELSS Agricultural Research
| Item | Function/Application | Example/Notes |
|---|---|---|
| Controlled Environment Chambers | Precisely regulate temperature, humidity, light (intensity, spectrum, photoperiod), and CO₂ to simulate CELSS conditions. | Phytochambers with programmable LED lighting and CO₂ injection. |
| Hydroponic/Nutrient Film Technique (NFT) Systems | Soilless plant cultivation for efficient water and nutrient recycling in a closed loop. | Systems must be compatible with both synthetic and organic nutrient solutions. |
| Synthetic Nutrient Solutions | Provide readily available inorganic macronutrients (N, P, K) and micronutrients for Conventional practice benchmarks. | Hoagland's solution or similar. Precise control over ionic concentrations. |
| Organic Nutrient Amendments | Provide nutrients through biological mineralization for Organic practice benchmarks. | Compost teas, vermicompost, fish emulsion. Sterilization may be required. |
| High-Throughput Phenotyping System | Automated, non-invasive monitoring of plant growth and development over time [73]. | LemnaTec Scanalyzer, Qubit Phenomics PlantScreen, or custom systems. |
| Multispectral Imaging Sensors | Assess plant health, biomass, and spatial uniformity via vegetation indices (e.g., NDVI) [103]. | Cameras capturing visible and near-infrared (NIR) spectra. |
| Gas Analyzers | Quantify photosynthetic CO₂ uptake and O₂ evolution rates for mass balance calculations [26]. | Infrared gas analyzers (IRGA) for CO₂; galvanic or zirconia sensors for O₂. |
| Neural Network Analysis Software | Automated object detection, segmentation, and counting of plants from image data [103]. | Detectron2, IAP (Image Analysis for Plants). Requires pre-trained models. |
Integrated Workflow for CELSS Agricultural Benchmarking
The following diagram synthesizes the experimental protocols into a cohesive research workflow, from initial setup to data-driven decision making for system design.
Within the domain of closed ecological life support system (CELSS) research, the selection and development of technologies for resource recovery are paramount. Two primary approaches exist: well-established physicochemical (PC) systems and emerging bioregenerative (BR) systems that leverage biological processes [4] [104]. This application note provides a standardized framework for assessing the maturity of these technologies using Technology Readiness Levels (TRLs), a systematic metric originally developed by NASA to evaluate the maturity of a technology from basic research (TRL 1) to full flight-proven operation (TRL 9) [105]. The objective is to equip researchers with clear protocols for conducting TRL assessments, enabling consistent comparison between PC and BR life support subsystems and guiding strategic research investment.
Technology Readiness Level (TRL) Framework
The TRL scale provides a common language for researchers, engineers, and program managers to assess the maturity of a given technology. The following table details the standardized TRL definitions as defined by NASA and adopted by other agencies, contextualized for CELSS research [105].
Table 1: Standard Technology Readiness Level (TRL) Definitions for CELSS Research
| TRL | NASA Definition | CELSS Research Interpretation & Example Activities |
|---|---|---|
| TRL 1 | Basic principles observed and reported | Basic research on plant photosynthesis rates under altered gravity or microbial urea hydrolysis pathways. |
| TRL 2 | Technology concept and/or application formulated | Invention of a new bioreactor concept for nitrification or a novel membrane material for water purification. |
| TRL 3 | Analytical and experimental critical function proof-of-concept | Laboratory experiments validate critical function, e.g., a small bioreactor achieves stable nitrification for 30 days. |
| TRL 4 | Component and/or breadboard validation in laboratory environment | A subsystem (e.g., a nutrient solution bioreactor) is integrated with a plant growth chamber in a lab. |
| TRL 5 | Component and/or breadboard validation in relevant environment | The integrated subsystem from TRL 4 is tested in a ground-based space habitat analog facility. |
| TRL 6 | System/subsystem model or prototype demonstration in a relevant environment | A full-scale prototype of the system is demonstrated in a high-fidelity ground test bed like NASA's HERA or a Moon/Mars analog habitat. |
| TRL 7 | System prototype demonstration in a space environment | The system is successfully operated on the International Space Station (ISS) or a similar platform. |
| TRL 8 | Actual system completed and "flight qualified" | The system is certified for a specific mission, e.g., a flight-ready urine processor for a Lunar Gateway module. |
| TRL 9 | Actual system "flight proven" through successful mission operations | The system has been successfully used in a mission, such as the ISS Water Recovery System. |
The following workflow diagram outlines the key stages and decision points in the TRL assessment process for a technology within a CELSS program.
Figure 1: TRL Assessment Workflow for CELSS Technologies
TRL Assessment of Life Support Subsystems
The core life support functions of a CELSS can be broken down into distinct subsystems. The maturity of PC and BR approaches for these functions varies significantly, largely because PC systems have a long history of deployment on crewed spacecraft like the ISS, while BR systems are predominantly in the research and ground-demonstration phase [4] [104].
Table 2: Comparative TRL Assessment of Physicochemical vs. Bioregenerative Subsystems
| Life Support Function | Physicochemical (PC) Systems | TRL (PC) | Bioregenerative (BR) Systems | TRL (BR) | Key Gaps & Research Needs (BR Systems) |
|---|---|---|---|---|---|
| Air Revitalization | Oxygen Generation Assembly (OGA) via electrolysis; CO₂ removal with zeolites or Sabatier process [104]. | 9 (ISS Operational) | Photosynthesis by higher plants and microalgae (e.g., in MELiSSA loop) for O₂ production and CO₂ consumption [4]. | 4-6 (Ground Demonstrators) | System stability, volumetric efficiency, and impact of space environments on long-term photosynthetic yield [4]. |
| Water Recovery | Water Processor Assembly (WPA) & Urine Processor Assembly (UPA) using distillation, filtration, and catalytic oxidation [104]. | 9 (ISS Operational) | Plant transpiration and microbial/root-zone processing in a soil-like substrate (SLS) or hydroponic bioreactor [106] [7]. | 4-5 (Ground Testing) | Closure of the water loop with complex waste streams; reliability of biological membranes and filters [106]. |
| Nitrogen Recovery | Limited recovery; nitrogen is typically vented as part of waste brine or as N₂ [104]. | 3-4 (Conceptual) | Nitrifying bacteria (e.g., in MELiSSA Comp. III) convert ammonium from urine to nitrate fertilizer for plants [104]. | 4-5 (Ground Testing) | Efficiency of urea/ammonium conversion under space conditions; stability of microbial consortia [104]. |
| Food Production | None; all food is supplied from Earth. | 1 (Not Applicable) | Cultivation of higher plants (crops) and microalgae for human consumption [4] [90]. | 6-7 (ISS & Ground Testing) | Optimization of growth cycles, nutrient density, and edible biomass yield in closed, controlled environments [4] [90]. |
| Solid Waste Processing | Compression, drying, and storage for disposal. Some incineration concepts [107]. | 4-5 (Ground Testing) | Aerobic and anaerobic composting to create a soil-like substrate (SLS) for plant growth [106] [107]. | 3-4 (Lab & Analog Testing) | Pathogen control, process kinetics, and gas exchange management in a closed habitat [107]. |
Protocols for TRL Advancement of Bioregenerative Systems
Protocol: TRL 3-4 Validation of a Nutrient Recycling Bioreactor
This protocol outlines the steps to advance a nutrient recycling bioreactor (e.g., for nitrification) from TRL 3 to TRL 4.
- Objective: To validate the critical function (nitrification) and integrate the bioreactor component with a plant growth chamber in a laboratory environment.
- Materials:
- Bench-scale bioreactor (e.g., packed-bed or stirred-tank).
- Inoculum of nitrifying bacterial culture (e.g., Nitrosomonas, Nitrobacter).
- Synthetic urine or urea-based feed solution.
- Hydroponic plant growth chamber (e.g., with lettuce or wheat).
- Analytical equipment: HPLC, Ion Chromatograph, spectrophotometer.
- Procedure:
- Bioreactor Startup: Inoculate the bioreactor and initiate continuous feeding with synthetic urine at a low loading rate. Monitor ammonium (NH₄⁺) and nitrite (NO₂⁻) levels daily until steady-state conversion to nitrate (NO₃⁻) is achieved.
- Performance Characterization: At steady-state, quantify the nitrification rate, conversion efficiency, and biomass productivity of the bacteria over a minimum of 30 days.
- Integration: Connect the effluent from the stabilized bioreactor directly to the nutrient solution reservoir of the plant growth chamber.
- Integrated System Testing: Operate the coupled system for a minimum of one complete plant growth cycle. Monitor plant health (growth rate, chlorophyll content) and nutrient solution composition (macronutrients, micronutrients, potential phytotoxins).
- Success Criteria:
- The bioreactor maintains >90% conversion of influent nitrogen to nitrate.
- Plants in the integrated system show no significant signs of nutrient deficiency or toxicity compared to a control group grown on a conventional nutrient solution.
- The coupled system operates stably for the duration of the test without catastrophic failure.
Protocol: TRL 5-6 Testing in a Relevant Ground-Based Analog
This protocol describes testing an integrated BR subsystem in a ground-based analog habitat to achieve TRL 5-6.
- Objective: To demonstrate the functionality and robustness of a BR subsystem (e.g., food production compartment) in a simulated mission environment with human operators.
- Materials:
- A dedicated module or container representing the BR subsystem (e.g., a growth chamber with LED lighting, nutrient delivery, and atmosphere control).
- The subsystem must be sized to contribute meaningfully to crew needs (e.g., provide 10-20% of daily caloric or oxygen demand).
- A ground analog facility (e.g., NASA's HERA, NEK, or a dedicated laboratory with mission-like operational protocols).
- Crew members (test subjects).
- Procedure:
- Deployment: Install the BR subsystem within the analog habitat and establish all necessary interfaces (power, data, atmospheric exchange).
- Mission Simulation: Conduct a simulated mission of a pre-defined duration (e.g., 30 to 90 days). During this time, crew members will interact with the system as per defined procedures (planting, harvesting, minor maintenance, data recording).
- Monitoring: Continuously monitor key performance parameters (e.g., O₂ production, CO₂ consumption, water transpiration, biomass yield, system power consumption).
- Contingency Testing: Introduce planned operational faults (e.g., power interruption, nutrient pump failure) to assess system robustness and crew recovery procedures.
- Success Criteria:
- The subsystem operates continuously with >95% uptime.
- Measured outputs (O₂, food, water) meet or exceed pre-determined target levels.
- The crew is able to safely operate and maintain the system with minimal external intervention.
- All data required to design a flight unit is collected.
The Scientist's Toolkit: Key Research Reagents & Materials
The following table details essential materials and reagents for conducting ground-based research on bioregenerative life support systems.
Table 3: Key Research Reagents and Materials for BRSS Experiments
| Item | Function & Application in CELSS Research |
|---|---|
| Simulated Regolith & Soils | JSC-1A (Lunar simulant) or MMS (Mars simulant), often mixed with organic matter to create a "soil-like substrate" (SLS) for studying plant growth and microbial community dynamics in representative media [106] [7]. |
| Synthetic Urine & Waste Streams | Chemically defined recipes that mimic the composition of human urine and other liquid wastes. Used to test and develop nutrient recycling bioreactors without the biohazard risks of real waste [104]. |
| Nitrifying Bacterial Consortia | Defined cultures or enriched communities of ammonia-oxidizing (e.g., Nitrosomonas) and nitrite-oxidizing (e.g., Nitrobacter) bacteria. Essential for developing biological nitrogen recovery systems from urine [104]. |
| Candidate Plant Cultivars | Dwarf or fast-growing species with high edible biomass ratio, such as lettuce (Lactuca sativa 'Outredgeous'), dwarf tomato ('Red Robin'), Mizuna, or wheat ('Apogee'). Selected for space cultivation trials [4] [90]. |
| Controlled Environment Chambers | Growth chambers with precise control over temperature, humidity, light intensity (LED spectrum), and CO₂ concentration. Used to optimize plant growth parameters and simulate spacecraft environments [4] [90]. |
| Hydroponic & Nutrient Film Technique (NFT) Systems | Soilless plant cultivation systems that allow for precise delivery and recycling of water and nutrients. The foundation for most space-based plant growth systems [4] [106]. |
The systematic application of the TRL framework reveals a clear technological landscape: physicochemical systems for air and water revitalization are highly mature (TRL 9), while bioregenerative systems for food production and nutrient recycling are advancing through mid-TRL levels (TRL 4-7) in ground-based research [4] [104]. The future of long-duration, sustainable human presence in space lies in hybrid life support systems that synergistically combine the reliability of proven PC technologies with the resource regeneration and psychological benefits of BR systems [106]. The protocols and assessment tools provided herein are designed to standardize the development path, reduce project risk, and accelerate the maturation of the biological technologies required to make this future a reality.
Conclusion
Bioregenerative Life Support Systems represent a paradigm shift from purely physicochemical to integrated biological systems essential for long-duration space exploration. The synthesis of research confirms that plant cultivation systems can reliably manage air and water quality while providing nutrition, but success hinges on robust crop selection, precise environmental control, and efficient nutrient recycling from waste. The validation of specific cultivars like dwarf peas and the application of high-throughput phenotyping are critical for selecting stress-resilient plants. Future directions must focus on closing the nutrient loop for elements like carbon and nitrogen, comprehensively understanding deep-space radiation effects on plant biology, and developing compact, automated systems for space-limited environments. The advancements in BLSS not only enable human endurance-class missions to the Moon and Mars but also propel innovations in controlled-environment agriculture on Earth, with significant potential applications in pharmaceutical research for producing plant-derived therapeutics in isolated, controlled settings.
References
- 1. | CELSS Knowledge Base CELSS [https://celss.net/tiki-index.php?page=CELSS]
- 2. Controlled ecological - life - Wikipedia support system [https://en.wikipedia.org/wiki/Controlled_ecological_life-support_system]
- 3. What is Celss ? Definition of Celss , Celss Meaning - The Economic... [https://economictimes.indiatimes.com/definition/celss]
- 4. Plant and microbial science and technology as cornerstones to Bioregenerative Life Support Systems in space | npj Microgravity [https://www.nature.com/articles/s41526-023-00317-9]
- 5. BLSS: a contribution to future life support - PubMed [https://pubmed.ncbi.nlm.nih.gov/11537782/]
- 6. | Encyclopedia MDPI Closed Ecological Life Support Systems [https://encyclopedia.pub/entry/13129]
- 7. Survivability and life support in sealed mini-ecosystems with simulated planetary soils | Scientific Reports [https://www.nature.com/articles/s41598-024-75328-x]
- 8. US5614378A - Photobioreactors and closed ... ecological life support [https://patents.google.com/patent/US5614378A/en]
- 9. Design and operation of an algal photobioreactor system [https://pubmed.ncbi.nlm.nih.gov/11537069/]
- 10. - Atomic Rockets Closed Life Support [https://projectrho.com/public_html/rocket/celss.php]
- 11. Hydroponics: current trends in sustainable crop production - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10625363/]
- 12. What Is a Closed-Loop Wastewater Treatment System? - Lakeside Equipment Corporation [https://www.lakeside-equipment.com/what-is-a-closed-loop-wastewater-treatment-system/]
- 13. Biosphere 2 - Wikipedia [https://en.wikipedia.org/wiki/Biosphere_2]
- 14. Using plants and soil microbes to purify indoor air: lessons from N... [https://journals.openedition.org/factsreports/6092]
- 15. Growing Plants in Space - NASA [https://www.nasa.gov/exploration-research-and-technology/growing-plants-in-space/]
- 16. So You Want to Be a Space Farmer… - NASA [https://www.nasa.gov/missions/station/so-you-want-to-be-a-space-farmer/]
- 17. Plants in space - Wikipedia [https://en.wikipedia.org/wiki/Plants_in_space]
- 18. (CELSS Prototypes)... Closed Ecological Life Support Systems [https://diversedaily.com/closed-ecological-life-support-systems-celss-prototypes-experiments-in-recycling-air-water-and-food-for-extended-missions-and-future-space-habitats/]
- 19. in materially Carbon recycling closed ecological life support systems [https://pubmed.ncbi.nlm.nih.gov/2895673/]
- 20. (PDF) Carbon to Nitrogen Ratios in Cropping Systems [https://www.academia.edu/33986658/Carbon_to_Nitrogen_Ratios_in_Cropping_Systems]
- 21. Crop-livestock synergies and by-products recycling : major factors for... [https://link.springer.com/article/10.1007/s13593-023-00908-6]
- 22. Metabolic and signaling aspects underpinning the regulation of plant ... [https://pubmed.ncbi.nlm.nih.gov/20926550/]
- 23. Designing a Closed for Ecological ... Life Support System Plants [https://link.springer.com/rwe/10.1007/978-3-319-09575-2_103-1]
- 24. Navigating Geopolitical Uncertainty: Strategies for 2025 | Sigma7 [https://www.s7risk.com/navigating-geopolitical-uncertainty-strategies-for-2025/]
- 25. Top 10 geopolitical risks in 2025 Geostrategic Outlook | EY - US [https://www.ey.com/en_us/insights/geostrategy/2025-geostrategic-outlook]
- 26. in a Plant ... Cultivation Closed Ecological Life Support System [https://www.jstage.jst.go.jp/article/seitaikogaku1989/2/1/2_1_51/_article]
- 27. of Cultivation in space: their contribution to stabilizing... plants [https://pubmed.ncbi.nlm.nih.gov/11538793/]
- 28. How Do Substrate , Hydroponic , and Aeroponic Systems Compare ... [https://www.miilkiiagrow.com/news/how-do-substrate-hydroponic-and-aeroponic-systems-compare/]
- 29. How Does an Aeroponic Tower Work | Tower Garden | Tower Farms [https://agrotonomy.com/aeroponic-towers/]
- 30. and Hydroponics • ABIBOO Studio architecture [https://abiboo.com/hydroponics-and-architecture/]
- 31. Aeroponics - Wikipedia [https://en.wikipedia.org/wiki/Aeroponics]
- 32. What hydroponic substrate should you choose? [https://www.goplanter.com/what-hydroponic-substrate-should-you-choose/]
- 33. Supplementing Closed Ecological Life Support Systems with In-Situ Resources on the Moon - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8401783/]
- 34. A Smart Aeroponic Chamber: Structure and Architecture for an... [https://link.springer.com/rwe/10.1007/978-1-0716-3993-1_18]
- 35. " Yield Comparisons and Unique Characteristics of the Dwarf Wheat..." [https://digitalcommons.usu.edu/cpl_dwarfcrops/10/]
- 36. comparisons and unique characteristics of the Yield ... dwarf wheat [https://pubmed.ncbi.nlm.nih.gov/11542565/]
- 37. Reinventing a Sustainable Green Revolution by Breeding and... [https://pubmed.ncbi.nlm.nih.gov/40842150/]
- 38. Development of dwarfish and yield -effective GM maize through... [https://link.springer.com/article/10.1007/s11248-019-00172-z]
- 39. influences selection of the rhizosphere microbiome Wheat dwarfing [https://www.nature.com/articles/s41598-020-58402-y?error=cookies_not_supported]
- 40. Growth-regulating factors: conserved and divergent roles in plant growth and development and potential value for crop improvement - PubMed [https://pubmed.ncbi.nlm.nih.gov/36582168/]
- 41. Seeds - Definition, Development and Advantages High Yielding Variety [https://www.vedantu.com/general-knowledge/high-yielding-variety-seeds]
- 42. Growth Regulator and Nitrogen Fertilization Effects on Performance... [https://scialert.net/fulltext/?doi=biotech.2006.268.276]
- 43. of Recovery from plant excreta and domestic... nutrients human [https://pmc.ncbi.nlm.nih.gov/articles/PMC11378796/]
- 44. Biofertilizers from wastewater as a potential... | PLOS One treatment [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0295624]
- 45. A Guide To Automated Farming Systems - Integrating Smart Sensors ... [https://atlas-scientific.com/blog/automated-farming-systems/]
- 46. Smart Agriculture Irrigation System: Auto Automation 2025 [https://farmonaut.com/precision-farming/smart-agriculture-irrigation-system-auto-automation-2025]
- 47. Frontiers | Aquatic bryophytes as biofilters and resource regenerators in... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1667463/full]
- 48. Space Analog for the Moon and Mars - Wikipedia [https://en.wikipedia.org/wiki/Space_Analog_for_the_Moon_and_Mars]
- 49. Becoming a multiplanetary species: Crew completes first mission in... [https://news.arizona.edu/news/becoming-multiplanetary-species-crew-completes-first-mission-pressurized-habitat-biosphere-2]
- 50. – Sam sam [https://samb2.space/sam/]
- 51. SAM Mars Analog | Biosphere 2 [https://biosphere2.org/research/research-initiatives/sam-mars-analog]
- 52. Yuegong-1 - Wikipedia [https://en.wikipedia.org/wiki/Yuegong-1]
- 53. China's 'Lunar Palace' for Space Research Tested on Earth | Space [https://www.space.com/26267-china-lunar-palace-space-research-mission.html]
- 54. Dwarf peas inside Biosphere 2's SAM breathe new life into space habitat research | University of Arizona News [https://news.arizona.edu/news/dwarf-peas-inside-biosphere-2s-sam-breathe-new-life-space-habitat-research]
- 55. Global challenges facing plant pathology: multidisciplinary approaches to meet the food security and environmental challenges in the mid-twenty-first century | CABI Agriculture and Bioscience | Full Text [https://cabiagbio.biomedcentral.com/articles/10.1186/s43170-021-00042-x]
- 56. diagnosis | PDF Plant disease [https://www.slideshare.net/slideshow/plant-disease-diagnosis/60900045]
- 57. Biological Control of Plant Diseases: An Evolutionary and Eco-Economic Consideration - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8541133/]
- 58. Major Biological Control Strategies for Plant Pathogens - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8879208/]
- 59. Conference Information Page – Plant (#PPATH Pathology )... 2025 2025 [https://www.bspp.org.uk/conference-info-plant-pathology-2025-ppath2025-and-early-careers-plant-pathology-2025-ecpp2025/]
- 60. Sustainable agriculture and plant diseases: an epidemiological perspective - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2610107/]
- 61. Scientific evidence for sustainable plant disease protection strategies for the main arable crops in Sweden. A systematic map protocol | Environmental Evidence | Full Text [https://environmentalevidencejournal.biomedcentral.com/articles/10.1186/s13750-018-0141-3]
- 62. and microbial science and technology as cornerstones to... Plant [https://www.nature.com/articles/s41526-023-00317-9?error=cookies_not_supported&code=fc66d5fa-f9b8-4eef-8628-89924d58c2d2]
- 63. Perspectives for plant biology in space and analogue environments | npj Microgravity [https://www.nature.com/articles/s41526-023-00315-x]
- 64. Investigating plant responses to microgravity and adaptations in gravisensitive environments | Environmental Sciences Europe | Full Text [https://enveurope.springeropen.com/articles/10.1186/s12302-024-00851-7]
- 65. Plant responses to real and simulated microgravity - ScienceDirect [https://www.sciencedirect.com/science/article/abs/pii/S2214552420300730]
- 66. Recent transcriptomic studies to elucidate the plant adaptive response to spaceflight and to simulated space environments - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9287483/]
- 67. Plants in Microgravity: Molecular and Technological Perspectives - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9505700/]
- 68. New Research Shows How Plant Roots Bend And Growth Downward... [https://astrobiology.com/2025/09/new-research-shows-how-plant-roots-bend-and-growth-downward-toward-gravity.html]
- 69. Microgravity research in plants: A range of platforms and options allow research on plants in zero or low gravity that can yield important insights into plant physiology - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6607005/]
- 70. Comparison of Microgravity Analogs to Spaceflight in Studies of Plant ... [https://pubmed.ncbi.nlm.nih.gov/31867033/]
- 71. What is quantitative plant biology? | Quantitative Plant Biology | Cambridge Core [https://www.cambridge.org/core/journals/quantitative-plant-biology/article/what-is-quantitative-plant-biology/87D480D2D4B875A6F02477F2EADBFDA1]
- 72. Planting Science - Student Roadmap - Visualizing and Analyzing Data [https://plantingscience.org/studentdoingscience/visualizing-and-analyzing-data]
- 73. Frontiers | Optimizing experimental procedures for quantitative evaluation of crop plant performance in high throughput phenotyping systems [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2014.00770/full]
- 74. : The Hidden Dangers Nutrient Toxicity [https://www.numberanalytics.com/blog/ultimate-guide-nutrient-toxicity-nutrition-immune-system]
- 75. and Correcting Preventing in Hydro Nutrient ... Toxicity Systems [https://www.hamanol.com/hydroponics/preventing-and-correcting-nutrient-toxicity-in-hydro-systems/]
- 76. Quick Fixes for Abnormal EC Level Fluctuations in Hydroponic... [https://www.miilkiiagrow.com/news/quick-fixes-for-abnormal-ec-level-fluctuations-in-hydroponic-nutr/]
- 77. Frontiers | Optimizing Crop Water for Drought and Climate... Use [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2022.824720/full]
- 78. Experimental study on CELSS-based domestic wastewater recycling... [https://pubmed.ncbi.nlm.nih.gov/41144999/]
- 79. Energy-use efficiency of organic and conventional plant production systems in Germany | Scientific Reports [https://www.nature.com/articles/s41598-024-51768-3]
- 80. Reduction in Energy Consumption by Mitigation of Cultivation Resistance Due to the New Fallow Harrow Concept [https://www.mdpi.com/1996-1073/15/22/8500]
- 81. Space Utilization – Wisconsin Energy Efficiency and Renewable Energy [https://fyi.extension.wisc.edu/energy/greenhouses/space-utilization/]
- 82. Increasing Greenhouse Utilization and Turns - Greenhouse Product News [https://gpnmag.com/article/increasing-greenhouse-utilization-and-turns/]
- 83. - High in Throughput Response: Methods... Phenotyping Plant Stress [https://pubmed.ncbi.nlm.nih.gov/29080181/]
- 84. Frontiers | Current methods and future needs for visible and non-visible... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1585413/full]
- 85. Comprehensive evaluation of carbon potential of... sequestration [https://cbmjournal.biomedcentral.com/articles/10.1186/s13021-023-00238-w]
- 86. Towards carbon neutrality: Enhancing CO2 sequestration by plants to reduce carbon footprint - ScienceDirect [https://www.sciencedirect.com/science/article/abs/pii/S0048969725003985]
- 87. Carbon Sequestration by Perennial Energy Crops: Is the Jury Still Out? - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4732603/]
- 88. Transforming US agriculture for carbon removal with enhanced weathering | Nature [https://www.nature.com/articles/s41586-024-08429-2]
- 89. How to calculate CO2 sequestration - EcoMatcher [https://www.ecomatcher.com/how-to-calculate-co2-sequestration/]
- 90. Designing a Closed Ecological Life Support System for Plants, ... [https://link.springer.com/10.1007/978-3-319-09575-2_103-1]
- 91. Frontiers | QTL Mapping Combined With Bulked Segregant Analysis... [https://www.frontiersin.org/journals/genetics/articles/10.3389/fgene.2018.00615/full]
- 92. of phytochrome-mediated Comparative responses in... analysis growth [https://link.springer.com/article/10.1007/bf00384851]
- 93. Growing Peas : How to Plant, Grow, and... | The Old Farmer's Almanac [https://www.almanac.com/plant/peas]
- 94. Frontiers | High-Throughput Plant Phenotyping Platform (HT3P) as a Novel Tool for Estimating Agronomic Traits From the Lab to the Field [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2020.623705/full]
- 95. A Comprehensive Review of High Throughput Phenotyping and Machine Learning for Plant Stress Phenotyping - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9590503/]
- 96. Frontiers | High and evaluation of salt- throughput ... screening tolerant [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1548576/full]
- 97. Semi- High for Potential Throughput - Screening in... Drought tolerance [https://www.jove.com/pl/t/52492/semi-high-throughput-screening-for-potential-drought-tolerance]
- 98. Root Phenotyping for Improved Resource in Crops Use Efficiency [https://link.springer.com/chapter/10.1007/978-981-19-7498-4_10]
- 99. Genetic mapping of quantitative trait loci associated with drought tolerance in chickpea (Cicer arietinum L.) | Scientific Reports [https://www.nature.com/articles/s41598-023-44990-y]
- 100. Molecular mapping of quantitative trait loci for drought tolerance in maize plants - PubMed [https://pubmed.ncbi.nlm.nih.gov/21644206/]
- 101. Analysis of Farming Comparative : Practices vs.... Conventional [https://hub.edubirdie.com/examples/conventional-farming-vs-organic-farming/]
- 102. vs Conventional Farming Organic [https://www.numberanalytics.com/blog/conventional-vs-organic-farming-nutrition]
- 103. Quantitative and Qualitative Analysis of Agricultural Fields Based on Aerial Multispectral Images Using Neural Networks [https://www.mdpi.com/1424-8220/23/22/9251]
- 104. Development of Nitrogen Recycling Strategies for Bioregenerative Life Support Systems in Space - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8548772/]
- 105. Technology readiness level - Wikipedia [https://en.wikipedia.org/wiki/Technology_readiness_level]
- 106. Supplementing Closed Ecological Life Support Systems with In-Situ Resources on the Moon [https://www.mdpi.com/2075-1729/11/8/770]
- 107. Biological and physicochemical methods for utilization of plant ... [https://www.semanticscholar.org/paper/Biological-and-physicochemical-methods-for-of-plant-Zolotukhin-Tikhomirov/b3ad9a9b75ab6264ed639380271a218e4e4a19d3]