Beyond the Visible: How RGB and Hyperspectral Imaging Are Revolutionizing Plant Analysis
This article explores the synergistic integration of RGB and hyperspectral imaging technologies for advanced plant analysis.
Beyond the Visible: How RGB and Hyperspectral Imaging Are Revolutionizing Plant Analysis
Abstract
This article explores the synergistic integration of RGB and hyperspectral imaging technologies for advanced plant analysis. Aimed at researchers and drug development professionals, it details how this combination bridges the gap between morphological data and deep biochemical insights. The content covers foundational principles, practical methodologies for non-invasive phenotyping and disease detection, strategies to overcome technical hurdles, and validation through comparative performance studies. By synthesizing information across these core intents, the article provides a comprehensive guide for leveraging this powerful multimodal approach to accelerate innovation in plant science and the development of plant-derived therapeutics.
Seeing More Than Meets the Eye: Fundamental Principles of RGB and Hyperspectral Plant Imaging
The Limitation of Human Vision and Standard RGB Imaging
The human visual system and conventional red-green-blue (RGB) imaging form the foundation of traditional plant phenotyping and agricultural assessment. However, these methods are fundamentally constrained by their limited spectral perception, capturing only a fraction of the information contained in light-plant interactions. This technical guide examines the physiological and technological boundaries of these conventional approaches and demonstrates how hyperspectral imaging overcomes these limitations by capturing continuous spectral data, enabling early detection of plant stress, precise nutrient assessment, and advanced growth stage classification. When strategically combined, RGB and hyperspectral imaging create a powerful synergistic toolset for plant researchers, offering both practical visual context and deep biochemical insights.
Human vision operates within a narrow band of the electromagnetic spectrum (approximately 400-700 nanometers), perceiving only three color channels through specialized cone cells. This trichromatic system, while sufficient for everyday tasks, misses critical information contained in near-infrared and ultraviolet ranges that reveal plant physiology and health status. Standard RGB imaging systems mimic this limited human visual perception, capturing only broad wavelength ranges corresponding to red, green, and blue light [1]. In agricultural research and drug development from plant sources, this spectral deficiency presents significant diagnostic limitations, as many biochemical processes exhibit spectral signatures outside this visible range or require finer spectral resolution for accurate quantification.
Fundamental Limitations of Human Vision and RGB Imaging
Physiological and Technological Constraints
The limitations of human vision and RGB imaging in plant research stem from both biological constraints and technological simplifications:
- Limited Spectral Resolution: Human eyes contain three types of cones with overlapping spectral sensitivities, while RGB cameras utilize filters with three broad spectral bands. This provides only generalized color information without the fine spectral resolution needed to identify specific biochemical compounds [1].
- Inability to Detect Pre-Visual Stress: Plant stress from pathogens, nutrient deficiencies, or water deficit induces biochemical changes that precede visible symptoms. These changes often manifest in spectral regions beyond human vision or as subtle spectral shifts indistinguishable by RGB analysis [2] [1].
- Subjectivity and Qualitative Nature: Human visual assessment is subjective and influenced by environmental conditions, observer experience, and physiological factors. Similarly, RGB image analysis often relies on color indices that lack direct correlation with specific biochemical traits [3].
Table 1: Quantitative Comparison of Imaging Modalities for Plant Research
| Parameter | Human Vision | Standard RGB Imaging | Hyperspectral Imaging |
|---|---|---|---|
| Spectral Range | 400-700 nm | 400-700 nm | 400-2500 nm (depending on sensor) |
| Number of Bands | 3 (cones) | 3 (R, G, B) | 100-250+ contiguous bands |
| Spatial Resolution | ~60 pixels per degree (visual acuity) | Limited only by sensor | Limited only by sensor |
| Early Stress Detection | Only when visible symptoms appear | Limited to visible symptoms | Before visible symptoms appear [2] |
| Biochemical Specificity | Low | Low | High (specific pigment/protein detection) |
| Cost & Accessibility | High (labor-intensive) | Low | Moderate to High |
Practical Implications for Plant Research
The technical limitations of human vision and RGB imaging translate directly into practical constraints for research and drug development:
- Growth Stage Misclassification: Visual classification of closely spaced wheat growth stages (Zadoks Z37, Z39, Z41) faces significant inter-observer variability, whereas hyperspectral imaging with machine learning achieved F1 scores of 0.832 for the same task [3].
- Late Intervention Timeline: By the time stress symptoms become visible to human observers or detectable with RGB imaging, plants may have already experienced significant physiological damage, reducing potential intervention efficacy and compromising research results [1].
- Incomplete Phenotypic Profiling: RGB-based phenotyping captures morphological traits but misses critical biochemical information essential for comprehensive phenotypic characterization in pharmaceutical development from plant sources.
Hyperspectral Imaging: Overcoming Spectral Limitations
Technological Advancements
Hyperspectral imaging (HSI) addresses the fundamental limitations of human vision and RGB imaging by capturing a full spectrum for each pixel in an image. Rather than three broad bands, HSI collects hundreds of narrow, contiguous spectral bands, creating a continuous spectral signature that serves as a unique biochemical fingerprint for each plant component [2]. Recent technological advances have transformed HSI from a specialized laboratory tool to a practical field instrument:
- Miniaturization and Portability: Modern hyperspectral sensors have become compact enough for deployment on drones, handheld devices, and field-based platforms, enabling in-situ measurement [2] [1].
- Snapshot HSI Technology: Next-generation snapshot hyperspectral cameras eliminate the need for scanning motions, capturing full hyperspectral cubes at video rates (25+ Hz) for real-time plant assessment [4] [1].
- AI-Enhanced Analytics: Cloud-based artificial intelligence and machine learning algorithms can process the vast datasets generated by HSI, extracting meaningful biochemical and physiological information [2].
Research Applications Revealing Invisible Plant Properties
The rich spectral data provided by HSI enables researchers to detect plant properties that remain invisible to human vision and standard RGB imaging:
- Early Stress Detection: HSI identifies fungal, viral, or bacterial infections through subtle biochemical changes before visible symptoms manifest, enabling intervention 7-14 days earlier than conventional methods [2].
- Nutrient Deficiency Mapping: Each nutrient deficiency produces a unique spectral signature detectable by HSI, allowing precise mapping of nitrogen, phosphorus, and potassium distribution across individual plants or entire fields [2].
- Growth Stage Classification: HSI with Support Vector Machine classification successfully distinguished subtle pre-anthesis wheat growth stages (Z37, Z39, Z41) with F1 scores of 0.832, far exceeding human visual assessment reliability [3].
- Biochemical Quantification: HSI enables non-destructive measurement of chlorophyll, anthocyanins, water content, and other biochemical traits through specific spectral features, many located outside the visible range [5].
Table 2: Key Spectral Regions for Plant Traits Beyond RGB Capability
| Plant Trait | Key Spectral Regions | Detection Method | Research Application |
|---|---|---|---|
| Chlorophyll Content | 450-670 nm, 700-750 nm (red edge) | Spectral index calculation | Photosynthetic efficiency assessment |
| Water Stress | 950-970 nm (water absorption) | Depth of water absorption features | Irrigation optimization, drought studies |
| Nitrogen Status | 550-570 nm, 700-720 nm | Spectral shift detection | Nutrient management, fertilizer optimization |
| Cell Structure | 1200-1300 nm, 1600-1700 nm | SWIR reflectance | Plant vigor assessment, disease impact |
| Anthocyanins | 530-560 nm, 670-680 nm | Specific absorption features | Plant stress response, product quality |
Experimental Protocols: Bridging Conventional and Advanced Imaging
Hyperspectral Imaging for Fine-Scale Growth Stage Classification
Objective: To automatically classify closely spaced wheat growth stages (Z37, Z39, Z41) at the individual plant level using hyperspectral imaging, overcoming limitations of human visual assessment [3].
Experimental Setup:
- Plant Material: Wheat cultivar 'Scepter' grown in greenhouse and semi-natural environments with controlled substrate [3].
- Imaging Systems:
- WIWAM hyperspectral imaging system with Specim FX10 camera (400-1000 nm range, 5.5 nm FWHM resolution)
- Top-down imaging from 1.4m height, 512 pixels/line, 2.6mm spatial resolution
- Halogen lighting in closed cabinet to eliminate external variations
- Reference Data: Visual growth stage assessment by trained experts following Zadoks scale
Methodology:
- Image Acquisition: Capture hyperspectral images between 12:00 PM-2:00 PM to minimize diurnal variation
- Calibration: Collect black-and-white reference images after each plant image for radiometric correction
- Spectral Extraction: Isolate leaf regions by masking background elements
- Data Transformation: Apply spectral transformations (Standard Normal Variate, Hyper-hue, Principal Component Analysis)
- Feature Selection: Identify most informative wavelengths (achieved 0.752 F1 score with only 5 wavelengths)
- Classification: Implement Support Vector Machine model with cross-validation
Key Findings: The combined use of multiple spectral transformations outperformed reliance on any single transformation, with SNV transformation demonstrating robust performance under limited training conditions [3].
Protocol for Hyperspectral Analysis of Leaf Color Patterns
Objective: To analyze complex leaf color patterns using hyperspectral reflectance imaging to reveal previously undetectable biochemical features [6].
Experimental Workflow:
- Hyperspectral Image Acquisition: Capture hyperspectral data cubes of plant leaves under controlled illumination
- Background Masking: Implement processing algorithms to isolate leaf regions from background
- Lighting Correction: Apply corrections for uneven lighting across the sample
- Spectral Component Analysis: Identify key spectral components representing distinct biochemical features
- Spatial Projection: Project hyperspectral cubes onto spectral components to highlight specific traits
This protocol enables researchers to move beyond subjective color descriptions to quantitative spectral pattern analysis, revealing features undetectable by human vision or RGB imaging [6].
Synergistic Integration: Combining RGB and Hyperspectral Imaging
AI-Based Spectral Prediction from RGB Images
Emerging research demonstrates the potential for combining the practical advantages of RGB imaging with the analytical power of hyperspectral data through artificial intelligence:
- Spectral Reconstruction: AI models trained on paired RGB and hyperspectral images can predict absorption spectra from standard RGB images, preserving spectral information while reducing cost and acquisition time [7].
- Cost-Effective Deployment: This approach leverages the widespread availability and lower cost of RGB systems while providing access to hyperspectral-grade data for plant trait analysis [7].
- Trait Quantification: AI-derived RGB models can estimate critical plant traits such as chlorophyll and anthocyanin content with strong agreement to hyperspectral reference values [7].
Operational Frameworks for Multi-Modal Imaging
Strategic combination of RGB and hyperspectral imaging in research pipelines creates complementary advantages:
- RGB for Morphological Assessment: Utilize RGB imaging for high-spatial-resolution morphological analysis, plant counting, and canopy structure assessment
- HSI for Biochemical Profiling: Apply hyperspectral imaging for detailed biochemical characterization, stress detection, and nutrient analysis
- Temporal Monitoring: Employ RGB for continuous monitoring due to its faster acquisition rates, with periodic HSI for deep biochemical profiling
The Researcher's Toolkit: Essential Materials and Methods
Table 3: Key Research Reagent Solutions for Plant Imaging Studies
| Item | Function | Application Example |
|---|---|---|
| Specim FX10 Hyperspectral Camera | Captures VNIR spectral range (400-1000 nm) | High-throughput plant phenotyping [5] |
| WIWAM Hyperspectral Imaging System | Provides controlled environment imaging | Growth stage classification studies [3] |
| Standard Normal Variate (SNV) Transformation | Normalizes spectral data for enhanced analysis | Improves robustness in classification models [3] |
| Living Optics Snapshot HSI Camera | Enables video-rate hyperspectral imaging | Real-time plant stress monitoring [1] |
| Supported Vector Machine (SVM) | Machine learning classification algorithm | Fine-scale growth stage classification [3] |
| TraitDiscover Platform | Integrated high-throughput phenotyping | Multi-dimensional plant data collection [5] |
The limitations of human vision and standard RGB imaging in plant research are significant and multifaceted, spanning spectral range, resolution, and biochemical specificity. Hyperspectral imaging technologies effectively address these limitations by providing continuous, high-resolution spectral data that reveals plant properties invisible to conventional methods. For researchers and drug development professionals, the strategic integration of RGB and hyperspectral imaging offers a powerful approach—combining the practical advantages and morphological capabilities of RGB with the deep biochemical insights of hyperspectral analysis. As these technologies continue to advance and become more accessible, they will play an increasingly critical role in accelerating plant research, pharmaceutical development from plant sources, and addressing global agricultural challenges.
Hyperspectral imaging (HSI) has emerged as a transformative technology for plant research, enabling non-invasive analysis of plant physiology, biochemistry, and health status by capturing unique spectral fingerprints. Unlike conventional RGB (Red, Green, Blue) imaging that records only three broad wavelength bands, HSI measures reflected light across hundreds of narrow, contiguous spectral bands, creating a continuous spectrum for each pixel in an image. This detailed spectral data provides researchers with unprecedented capability to detect plant stress, monitor growth stages, and assess chemical composition before visible symptoms appear. This technical guide explores the core principles of HSI, its synergistic application with RGB imaging, and provides detailed experimental protocols for implementing these technologies in plant research applications, with a specific focus on bridging the gap between laboratory research and field deployment.
The fundamental limitation of conventional RGB imaging lies in its simplification of the continuous light spectrum into just three broad bands corresponding to the red, green, and blue receptors of the human eye. While sufficient for representing color perception, this approach discards vast amounts of spectral information that reveal critical details about plant composition and function. Hyperspectral imaging overcomes this limitation by capturing the complete spectral signature of plant materials across a wide range of wavelengths, typically from the visible (400-700 nm) through the near-infrared (700-2500 nm) regions [8].
Each material and biochemical component within plant tissues interacts uniquely with light, absorbing specific wavelengths while reflecting others. This interaction creates a unique spectral signature that serves as a chemical "fingerprint" [9]. For example, chlorophyll strongly absorbs red and blue light while reflecting green and near-infrared wavelengths, creating characteristic absorption features at around 430-450 nm and 650-700 nm, and high reflectance in the NIR region [10]. These subtle spectral variations, often invisible to RGB cameras, become clearly detectable with HSI's fine spectral resolution, enabling researchers to decipher the biochemical language of plants.
Core Technology: HSI vs. RGB Imaging
Fundamental Differences in Data Acquisition
The technological distinction between RGB and hyperspectral imaging begins at the sensor level. RGB cameras utilize a Bayer filter mosaic with separate filters for red, green, and blue channels, effectively averaging light detection across three broad spectral bands [9]. In contrast, hyperspectral imaging systems employ sophisticated spectrographs that disperse incoming light across hundreds of detector elements, capturing narrow, contiguous wavelength bands throughout the electromagnetic spectrum.
This fundamental difference in data acquisition creates a significant disparity in information content. While RGB produces a three-channel image, HSI generates a three-dimensional data structure known as a hypercube, containing two spatial dimensions and one spectral dimension [11] [8]. This hypercube can be visualized as a stack of images, each representing a specific narrow wavelength band, with each pixel containing a complete, continuous spectrum from the imaged scene.
Quantitative Comparison of Capabilities
Table 1: Technical comparison between RGB, Multispectral, and Hyperspectral imaging systems for plant research.
| Parameter | RGB Imaging | Multispectral Imaging | Hyperspectral Imaging |
|---|---|---|---|
| Spectral Bands | 3 (Red, Green, Blue) | 3-10 broad, discrete bands | 50-250+ narrow, contiguous bands |
| Spectral Range | 400-700 nm (Visible) | Typically 400-900 nm | 400-2500 nm (VNIR-SWIR) |
| Spectral Resolution | 50-100 nm | 10-50 nm | 1-10 nm |
| Primary Data Output | 2D color image | Limited spectral indices | Full spectral signature per pixel |
| Information Depth | Morphology, color | General health, limited stress detection | Biochemical composition, early stress identification |
| Weed/Pest Discrimination | Limited | Moderate | High accuracy based on biochemical differences |
| Early Disease Detection | Not possible | Limited, after symptom appearance | Possible before visual symptoms [10] [12] |
| Cost & Complexity | Low | Medium | High |
The practical implication of these technical differences is profound for plant research. While RGB imaging can identify visible symptoms such as color changes or lesions, HSI can detect pre-symptomatic stress through subtle biochemical alterations. For instance, HSI can identify nutrient deficiencies, water stress, and pathogen infections days before any visible symptoms manifest [1] [12]. This early detection capability provides a critical window for intervention, potentially preventing significant crop losses and reducing unnecessary pesticide applications.
The Scientific Toolkit: Essential Equipment and Analytical Approaches
Implementing hyperspectral imaging for plant research requires specific hardware, software, and analytical tools. The selection of appropriate equipment depends on the research objectives, scale of analysis, and operational environment.
Research Reagent Solutions and Essential Materials
Table 2: Essential components of a hyperspectral imaging system for plant research.
| Component | Specifications | Function & Importance |
|---|---|---|
| Hyperspectral Camera | VNIR (400-1000 nm) and/or SWIR (900-2500 nm); Spectral resolution: 1-10 nm; Spatial resolution: Varies with platform | Captures spectral data cube; Core component defining data quality and application scope |
| Illumination System | Halogen lights (lab) or calibrated LEDs (field/space) [12]; Uniform, stable broadband source | Provides consistent illumination crucial for reproducible spectral measurements |
| Spectral Calibration Tools | White reference panel (Spectralon); Dark current reference | Enables conversion of raw data to reflectance values; Essential for quantitative analysis |
| Platform & Positioning | Laboratory scanners, UAVs, ground vehicles, or handheld systems [13] | Determines spatial scale and operational environment; Affects spatial resolution and coverage |
| Data Processing Software | Python, ENVI, or specialized platforms (Specim, Living Optics) | Handles large datasets, calibration, and analysis including machine learning algorithms |
| Reference Chemicals | Laboratory standards for pigments (chlorophyll, carotenoids), nutrients | Validates spectral signatures and develops quantitative models |
Integrated System Configuration
Advanced HSI systems often combine multiple imaging modalities to enhance analytical capabilities. For example, NASA's plant health monitoring system for space crop production integrates both reflectance and fluorescence imaging within a single automated platform [12]. This system utilizes two LED line lights—one providing VNIR broadband illumination for reflectance measurements and another providing UV-A (365 nm) excitation for fluorescence imaging—enabling comprehensive plant health assessment through complementary data streams.
For field applications, systems like the Specim AFX series offer turn-key solutions for UAV-based hyperspectral imaging, allowing researchers to create detailed material maps across large agricultural areas [13]. These systems are radiometrically calibrated and optimized for the challenging environmental conditions encountered in outdoor research.
Experimental Protocols: Methodologies for Plant Research Applications
Protocol 1: Early Detection of Pathogen-Induced Stress
Objective: To detect and identify fungal pathogens in cabbage (Brassica oleracea) before visible symptoms appear using hyperspectral imaging.
Materials and Setup:
- Hyperspectral imaging system with spectral range covering 400-1000 nm (VNIR)
- Uniform illumination system (halogen lights or calibrated LEDs)
- White reference panel (Spectralon) for calibration
- Plant samples: Healthy and Alternaria brassicae-inoculated cabbage plants
- Controlled environment growth chamber with stable lighting conditions
Procedure:
- System Calibration: Acquire white reference images using the Spectralon panel and dark current images with the lens covered before each imaging session.
- Image Acquisition: Place plants in the imaging area at a constant distance (e.g., 1.4 m) from the camera. Capture hyperspectral images of both healthy and inoculated plants at 24-hour intervals post-inoculation.
- Spectral Feature Identification: Extract mean spectra from regions of interest (ROIs) on leaves. Identify key spectral regions affected by pathogen infection:
- 400-450 nm: Flavonoid content alterations
- 430-450 nm & 680-700 nm: Chlorophyll degradation
- 470-520 nm: Carotenoid changes
- 520-600 nm: Xanthophyll cycle activity
- 700-750 nm & 718-722 nm: Phenolic compounds/mycotoxins [10]
- Classification Model Development: Apply machine learning algorithms (Support Vector Machines or Random Forest) to spectral data. Use 70% of data for training and 30% for validation.
- Validation: Compare classification results with molecular diagnostic tests to verify pathogen presence.
Expected Outcomes: The system should achieve over 90% classification accuracy for early infection stages, enabling detection before visible symptoms manifest [12].
Protocol 2: Classification of Individual Wheat Growth Stages
Objective: To automatically classify pre-anthesis growth stages (Zadoks Z37, Z39, Z41) in individual wheat plants using hyperspectral imaging.
Materials and Setup:
- Pushbroom hyperspectral camera (e.g., Specim FX10) covering 400-1000 nm range
- Automated conveyor system or translational stage for consistent scanning
- Controlled lighting environment (closed imaging cabinet with halogen lighting)
- Pot-grown wheat plants (Triticum aestivum) at target growth stages
Procedure:
- Plant Cultivation: Grow wheat plants under controlled conditions. Precisely record growth stages according to Zadoks scale through daily visual inspection.
- Hyperspectral Data Collection: Image plants from top-down perspective at constant distance (e.g., 1.4 m). Capture images between 12:00 PM-2:00 PM to minimize diurnal variation.
- Spectral Transformation: Apply mathematical transformations to spectral data:
- Standard Normal Variate (SNV): Normalizes spectral data to reduce scattering effects
- Hyper-hue: Enhances color-related spectral features
- Principal Component Analysis (PCA): Reduces dimensionality while preserving variance
- Feature Selection: Identify most discriminative wavelengths (5-10 bands) for growth stage differentiation using feature selection algorithms.
- Model Training and Validation: Implement Support Vector Machine classifier with cross-validation. Compare performance against RGB-based classification.
Expected Outcomes: The hyperspectral approach should achieve F1 scores of approximately 0.832 for growth stage classification, significantly outperforming RGB-based methods [3].
The following diagram illustrates the workflow for this hyperspectral analysis of wheat growth stages:
Data Processing and Analysis: From Spectral Data to Actionable Insights
Spectral Preprocessing and Transformation
Raw hyperspectral data requires substantial preprocessing before analysis to remove instrumental artifacts and environmental noise. Essential preprocessing steps include:
- Radiometric Correction: Converting raw digital numbers to reflectance values using white and dark reference images
- Geometric Correction: Addressing spatial distortions from camera optics or platform movement
- Noise Reduction: Applying smoothing filters (Savitzky-Golay, median filtering) to reduce spectral noise
- Spectral Transformation: Using mathematical transformations to enhance spectral features:
- Standard Normal Variate (SNV): Normalizes each spectrum to zero mean and unit variance, reducing light scattering effects
- Derivative Analysis: First and second derivatives help resolve overlapping spectral features and eliminate baseline offsets
These preprocessing steps are critical for ensuring that subsequent analysis reflects actual biological variation rather than measurement artifacts.
Machine Learning Integration for Spectral Analysis
The high dimensionality of hyperspectral data makes machine learning approaches particularly valuable for extracting meaningful biological information. Both conventional and deep learning methods have demonstrated success in plant research applications:
Conventional Machine Learning:
- Support Vector Machines (SVM): Effective for classification tasks with limited training data, achieving F1 scores up to 0.832 for wheat growth stage classification [3]
- Random Forests: Robust for regression and classification tasks, handling high-dimensional data well
- Partial Least Squares Regression (PLSR): Useful for quantifying biochemical constituents from spectral data
Deep Learning Approaches:
- Convolutional Neural Networks (CNNs): Can automatically learn relevant spectral features from raw data, reducing need for manual feature engineering
- Spectral-Spatial Networks: Incorporate both spectral and spatial information for improved classification accuracy
The integration of machine learning with HSI has enabled the development of automated systems capable of detecting plant stress, predicting yield, and classifying growth stages with minimal human intervention.
Integrated Data Visualization: Combining Spatial and Spectral Information
The power of hyperspectral imaging lies in its ability to simultaneously preserve spatial and spectral information. This integrated data structure enables researchers to visualize both the distribution and chemical identity of materials within a scene.
The following diagram illustrates the fundamental structure of hyperspectral data and the process of extracting meaningful biological information:
Future Perspectives and Challenges
Despite its significant potential, several challenges remain for widespread adoption of hyperspectral imaging in plant research. The high cost of traditional HSI systems, while decreasing, still presents a barrier for many research institutions [11]. Data management poses another significant challenge, as hyperspectral datasets are large and computationally demanding to process and store. Additionally, technical expertise requirements for operating HSI systems and interpreting results remain substantial.
Future developments are likely to focus on:
- Miniaturization and cost reduction of hyperspectral sensors [1] [13]
- Improved data processing algorithms and user-friendly software interfaces
- Enhanced integration with other sensing modalities (thermal, fluorescence, 3D)
- Development of standardized spectral libraries for different plant species and stress conditions
- Advancements in real-time processing capabilities for field applications
As these technological advancements progress, hyperspectral imaging is poised to become an increasingly accessible and indispensable tool for plant researchers, enabling deeper insights into plant biology and more sustainable agricultural practices.
Hyperspectral imaging represents a paradigm shift in plant research methodology, moving beyond the morphological assessments possible with RGB imaging to enable non-invasive biochemical characterization and early stress detection. By capturing the unique spectral fingerprint of plants across hundreds of narrow, contiguous wavelength bands, HSI provides researchers with an powerful tool for deciphering the complex relationships between plant physiology, environmental conditions, and genetic expression.
The integration of HSI with machine learning analytics and complementary imaging modalities creates a powerful framework for advancing plant science. While challenges remain in cost, data handling, and technical complexity, ongoing technological developments are steadily addressing these limitations. As hyperspectral systems continue to become more accessible, portable, and user-friendly, their application in both controlled environments and field settings will undoubtedly expand, contributing significantly to our understanding of plant biology and the development of more sustainable agricultural systems.
This technical guide explores the fundamental principles and applications of light reflectance spectroscopy for quantifying plant biochemical traits. The interaction between light and plant tissues produces unique spectral signatures that can be decoded to measure photosynthetic pigments, structural components, water content, and nutrients non-destructively. Within the broader context of plant research, this whitepaper demonstrates how hyperspectral imaging provides detailed biochemical insights that complement the spatial and cost advantages of RGB imaging, creating a powerful synergistic framework for advanced phenotyping and precision agriculture.
Plant leaves interact with electromagnetic radiation through specific mechanisms across different spectral regions. In the visible range (400-700 nm), light absorption is primarily dominated by photosynthetic pigments, with chlorophyll absorbing strongly in the blue and red regions while reflecting green light [14]. The near infrared plateau (NIR, 800-1300 nm) is characterized by multiple scattering within the leaf's internal air spaces, making this region highly sensitive to leaf structure and cellular organization [14]. The short-wave infrared (SWIR, 1300-2500 nm) contains absorption features primarily associated with water (at 1450 nm and 1940 nm) and dry matter constituents including proteins, lignins, cellulose, and other carbon-based compounds [15] [14].
Each biochemical constituent exhibits specific absorption features due to vibrational bonds including C—O, O—H, C—H, and N—H bonds, plus overtones and combinations of these vibrations [15]. These unique spectral signatures enable researchers to distinguish between biochemical components despite their overlapping absorption features.
Quantitative Relationships: Biochemical-Spectral Correlations
Table 1: Correlation between Hyperspectral Reflectance and Key Wheat Physiological Traits (Partial Least Squares Regression Models) [16]
| Trait | Correlation Coefficient (R²) | Bias (%) | Relative Error of Prediction | Measurement Significance |
|---|---|---|---|---|
| Vcmax25 (Rubisco activity) | 0.62 | <0.7% | Slightly greater | Photosynthetic capacity |
| J (Electron transport rate) | 0.70 | <0.7% | Slightly greater | Light reaction efficiency |
| SPAD (Chlorophyll) | 0.81 | <0.7% | Similar | Chlorophyll content |
| LMA (Leaf mass per area) | 0.89 | <0.7% | Slightly greater | Leaf thickness/structure |
| Narea (Leaf nitrogen) | 0.93 | <0.7% | Slightly greater | Nitrogen status |
Table 2: Key Spectral Regions for Biochemical Constituents in Vegetation [15] [14]
| Biochemical Constituent | Key Spectral Regions (nm) | Specific Absorption Features (nm) | Chemical Bonds Involved |
|---|---|---|---|
| Nitrogen/Proteins | 1510, 1730, 1940, 2060, 2180, 2240, 2300 | 1690, 1940, 2060, 2180, 2240, 2300 | N-H, C-H, O-H bonds |
| Lignin | 1120, 1200, 1420, 1450, 1690, 1940, 2100 | 1120, 1420, 1690, 2100 | C-H, C-O bonds in phenolics |
| Cellulose | 1200, 1490, 1780, 1820, 2000, 2100, 2280, 2340 | 1200, 1490, 1780, 2100, 2280 | C-H, C-O bonds in polysaccharides |
| Leaf Water Content | 970, 1200, 1450, 1940 | 1450, 1940 | O-H bonds |
| Chlorophyll/Pigments | 430-470, 660-680, 700-750 (red edge) | 531, 570 (PRI) | Porphyrin ring structure |
Experimental Protocols and Methodologies
Hyperspectral Data Acquisition Protocol
Equipment Requirements:
- Field spectroradiometer or hyperspectral imaging system (400-2500 nm range)
- Standard reflectance reference panel (e.g., Spectralon)
- Controlled illumination source (for laboratory measurements)
- Stable measurement platform with standardized geometry
- Environmental monitoring sensors (light, temperature, humidity)
Standardized Measurement Procedure: [16] [14]
- Instrument Calibration: Warm up spectrometer for recommended duration (typically 30-60 minutes). Perform wavelength calibration using standard sources.
- Reference Measurement: Measure spectralon reference panel under identical illumination conditions as sample measurements.
- Sample Presentation: For leaf measurements, maintain consistent orientation (typically adaxial surface up). Use clipping holder or dark background to minimize contamination.
- Spectral Collection: Acquire multiple spectra per sample (minimum 3-5 readings) to account for heterogeneity. Maintain constant distance and viewing geometry.
- Data Quality Assessment: Check for saturation, signal-to-noise ratio, and abnormal spectral shapes indicating measurement artifacts.
- Reflectance Calculation: Convert raw radiance to reflectance by dividing sample radiance by reference panel radiance.
Continuum-Removal and Band-Depth Analysis
For quantitative biochemical estimation, continuum-removal enhances detection of specific absorption features: [15]
- Continuum Definition: Identify convex hull points surrounding absorption features of interest (e.g., 1730 nm, 2100 nm, 2300 nm for nitrogen).
- Continuum Removal: Divide original reflectance values by corresponding continuum line values at each wavelength.
- Band Depth Calculation: Compute absorption band depth as BD(λ) = 1 - R(λ)/Rc(λ), where R(λ) is reflectance at wavelength λ and Rc(λ) is continuum reflectance at same wavelength.
- Normalization: Normalize band depths by either the depth at band center or area under band depth curve to reduce non-foliar influences.
- Statistical Modeling: Apply stepwise multiple linear regression between normalized band depths and chemically determined constituent concentrations.
Band Depth Analysis Workflow: From raw spectra to biochemical quantification
Genetic Application Protocol: Spectral Phenotyping of Plant Populations
Advanced applications include detecting genetic variation through spectral phenotyping: [14]
Experimental Design:
- Plant Materials: Utilize diverse germplasm including wild accessions, recombinant inbred lines (RILs), and transgenic lines with targeted gene modifications.
- Environmental Control: Implement replicated designs across controlled (glasshouse) and field environments to partition genetic and environmental variance.
- Standardized Measurement Timing: Measure leaves at consistent developmental stages (e.g., fully expanded source leaves).
Spectral Data Processing for Genetic Analysis:
- Quality Control: Remove spectra with obvious artifacts, noise, or contamination.
- Spectral Normalization: Apply standard normal variate (SNV) or multiplicative scatter correction to reduce light-scattering effects.
- Population-Level Analysis: Conduct principal component analysis (PCA) to visualize spectral variation among genotypes.
- Heritability Estimation: Calculate broad-sense heritability for spectral features using mixed models.
- Genome-Wide Association: Correlate specific spectral features with genetic markers to identify loci controlling biochemical traits.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Tools for Plant Reflectance Spectroscopy
| Item | Function/Purpose | Technical Specifications | Application Context |
|---|---|---|---|
| Field Spectroradiometer | Measures reflected radiance across spectral range | 350-2500 nm range, 3-10 nm resolution, fiber optic input | Field and laboratory measurements |
| Hyperspectral Imaging System | Spatial-spectral data acquisition | VNIR (400-1000 nm) and/or SWIR (1000-2500 nm) cameras | High-throughput phenotyping, spatial mapping |
| Spectralon Reference Panel | Provides baseline reflectance (~99%) for calibration | Labsphere Spectralon or equivalent | Essential for reflectance calculation |
| Controlled Illumination Source | Standardized lighting conditions | Tungsten-halogen, uniform light field | Laboratory measurements |
| Leaf Clips | Fixed geometry for repeated measurements | With or without integrated light source | Standardized leaf-level measurements |
| Spectral Library Data | Reference spectra for known materials | USGS, JPL, ASTER spectral libraries | Material identification, validation |
| Chemical Analysis Kits | Ground truth biochemical data | Nitrogen (Kjeldahl), lignin (ACBr), chlorophyll (extraction) | Model calibration, validation |
| ENVI/SPECPR Software | Spectral data processing and analysis | Continuum removal, derivative analysis, classification | Data processing, model development |
Synergistic Integration: RGB and Hyperspectral Imaging in Plant Research
While hyperspectral imaging provides detailed biochemical information through continuous spectral sampling across hundreds of bands, RGB imaging offers complementary advantages through its accessibility, spatial resolution, and cost-effectiveness. The integration of these technologies creates a powerful framework for plant research.
RGB Imaging Applications and Hidden Potential:
- Spatial Analysis: RGB cameras provide high-resolution structural information for assessing plant architecture, leaf area, and growth patterns [1].
- Color-Based Indices: Despite limited to three broad bands, RGB imagery can extract vegetation indices when calibrated properly [17].
- Hidden Contrast Enhancement: Advanced processing of RGB images can reveal contrast variations beyond human visual perception, extracting analytical information about stress patterns and field variability [17].
- Temporal Monitoring: Cost-effectiveness enables high-frequency monitoring for phenological studies and growth trajectory analysis.
Hyperspectral Advantages for Biochemistry:
- Early Stress Detection: Identification of drought, nutrient deficiency, pests, and disease before visible symptoms appear [1].
- Quantitative Biochemistry: Precise measurement of nitrogen, lignin, cellulose, and photosynthetic parameters through specific spectral features [16] [15].
- Genetic Studies: Detection of spectral traits linked to genetic variation, enabling high-throughput phenotyping for breeding programs [14].
RGB-Hyperspectral Synergy: Complementary technologies for comprehensive plant assessment
Light reflectance spectroscopy provides a powerful, non-destructive approach for quantifying plant biochemical traits across multiple scales from individual leaves to canopies. The fundamental interactions between light and biochemical constituents create detectable spectral signatures that can be decoded through rigorous experimental protocols and analytical methods. The continuous spectrum measured by hyperspectral imaging enables precise quantification of nitrogen, lignin, cellulose, water content, and photosynthetic parameters, while RGB imaging offers complementary spatial and temporal monitoring capabilities. Together, these technologies form an integrated framework that advances plant phenotyping, precision agriculture, and ecological research by bridging the gap between visible traits and underlying biochemical composition. As spectroscopic technologies continue to evolve toward more portable, affordable, and automated systems, the application of light reflectance for decoding plant biochemistry will expand, enabling researchers to address critical challenges in food security, environmental sustainability, and plant-based product development.
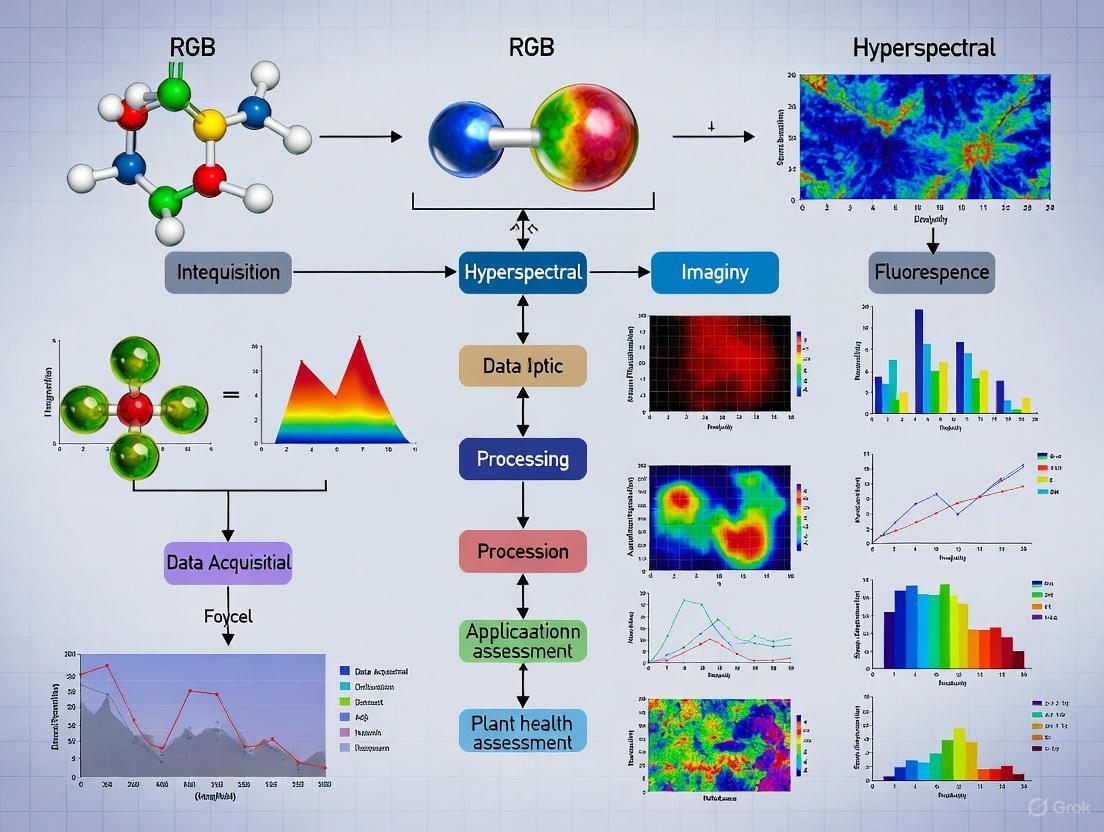
In plant research, the transition from traditional RGB (Red, Green, Blue) imaging to hyperspectral analysis represents a fundamental shift in observational capability. Human vision, and by extension conventional RGB cameras, is limited to perceiving reflected light in three broad wavelength bands, providing information primarily about color and morphology. While useful for identifying visible symptoms, this approach cannot detect the subtle biochemical changes that occur in plants during early stress or disease development [2] [1]. Hyperspectral imaging (HSI) shatters this limitation by capturing light across hundreds of narrow, contiguous spectral bands, creating a detailed data structure known as a hypercube [11]. This hypercube contains both spatial information (x, y) and extensive spectral data (λ) for each pixel, enabling researchers to quantify biochemical and physiological changes in plants before visible symptoms appear [2] [18].
The true power of modern plant phenotyping and disease detection lies not in choosing between RGB and hyperspectral modalities, but in strategically combining them. RGB imaging provides high-spatial-resolution morphological data at lower cost and computational requirements, while HSI delivers unparalleled biochemical insight through high spectral resolution [19] [20]. This technical guide explores the theoretical foundations, practical methodologies, and analytical frameworks for integrating these complementary technologies, providing researchers with a comprehensive toolkit for advancing plant science and agricultural innovation.
Technical Foundations: From RGB to the Hyperspectral Data Cube
RGB Imaging: Spatial Resolution and Morphological Analysis
RGB imaging captures reflected light in three broad wavelength bands corresponding to human visual perception (approximately 400-500 nm for blue, 500-600 nm for green, and 600-700 nm for red). The resulting data structure is a two-dimensional array of pixels, with each pixel containing three intensity values representing these color channels [21]. In plant research, RGB imaging excels at quantifying morphological traits such as leaf area, plant architecture, and visible symptom progression [19]. Its advantages include relatively low hardware costs, straightforward data processing, and high spatial resolution, making it suitable for high-throughput phenotyping applications where visible traits are the primary interest [22] [23].
The fundamental limitation of RGB imaging stems from its spectral poverty. With only three data points per pixel spectrum, it cannot resolve the subtle spectral signatures associated with biochemical changes during early stress responses. Additionally, RGB data are sensitive to varying illumination conditions, requiring careful standardization for quantitative comparisons [19].
Hyperspectral Imaging: Spectral Dimension and Biochemical Insight
Hyperspectral imaging fundamentally expands the data dimensionality by capturing reflected light across hundreds of narrow, contiguous spectral bands, typically ranging from the visible to short-wave infrared regions (400-2500 nm) [11] [21]. The resulting data structure is a three-dimensional hypercube with two spatial dimensions (x, y) and one spectral dimension (λ), where each pixel contains a complete spectral signature representing the biochemical composition of that specific location [11].
This rich spectral data enables the detection of subtle changes in plant physiology long before they become visible to the human eye or RGB sensors. Specific molecular bonds and compounds, including chlorophylls, carotenoids, water, and other biochemical constituents, interact with light at characteristic wavelengths, creating unique absorption features in the spectral profile [21]. By analyzing these spectral fingerprints, researchers can detect early responses to biotic and abiotic stresses, often with 60-90% accuracy, and up to 95% in controlled conditions [21] [18].
Table 1: Comparative Analysis of RGB and Hyperspectral Imaging Technologies
| Parameter | RGB Imaging | Multispectral Imaging | Hyperspectral Imaging |
|---|---|---|---|
| Spectral Bands | 3 broad bands (R, G, B) | 3-10 discrete bands | Hundreds of narrow, contiguous bands |
| Spectral Range | 400-700 nm (Visible) | Visible to NIR | UV to SWIR (250-2500 nm) |
| Spatial Resolution | High | Medium to High | Typically lower due to data volume |
| Data Dimensionality | 2D + 3 channels | 2D + limited spectral data | 3D hypercube (x, y, λ) |
| Primary Applications | Morphological assessment, visible symptom detection | Broad stress detection, vegetation indices | Early stress detection, biochemical analysis, pathogen identification |
| Cost Considerations | $500-$2,000 | $2,000-$10,000 | $20,000-$50,000+ |
| Data Processing Complexity | Low | Medium | High |
Table 2: Quantitative Performance Comparison for Disease Detection
| Performance Metric | RGB with Deep Learning | Hyperspectral Imaging |
|---|---|---|
| Laboratory Accuracy | 95-99% | 95-99% |
| Field Deployment Accuracy | 70-85% | 80-90% |
| Early Detection Capability | Limited to visible symptoms | Pre-symptomatic (3-7 days before visual symptoms) |
| Multiple Infection Classification | Low accuracy (approximately 53% with CNNs) | High accuracy (81% with EfficientNet 2D CNN) |
| Cross-Specificity | Limited, confounded by multiple stressors | High, can distinguish between similar diseases |
Integrated Experimental Protocols
Multi-Modal Image Registration Pipeline
The fusion of RGB and hyperspectral data requires precise pixel-level registration to ensure spatial correspondence between modalities. The following protocol, adapted from successful implementations, enables robust multi-modal image registration [20]:
Materials and Equipment:
- RGB camera (e.g., high-resolution scientific-grade with global shutter)
- Hyperspectral imaging system (e.g., Specim FX series pushbroom scanner)
- Chlorophyll fluorescence imager (e.g., PhenoVation Plant Explorer XS)
- Calibration targets (e.g., checkerboard pattern, spectralon reference panel)
- Computational infrastructure for data processing
Procedure:
System Calibration:
- Perform individual camera calibration for each sensor using checkerboard targets
- Calculate lens distortion parameters and apply correction
- Verify subpixel alignment accuracy (target mean reprojection error <0.5 pixels)
Data Acquisition:
- Acquire RGB images under consistent illumination conditions
- Collect hyperspectral data using pushbroom scanner with consistent movement speed
- Capture chlorophyll fluorescence kinetics using standardized excitation protocols
- Include reference standards in each imaging session for radiometric correction
Image Registration:
- Select reference image (typically high-contrast RGB or ChlF)
- Apply affine transformation to align multimodal datasets
- Implement normalized cross-correlation (NCC) for similarity measurement
- Utilize phase-only correlation (POC) for robustness to intensity variations
- Apply feature-based methods (ORB, RANSAC) for coarse alignment
Validation:
- Quantify overlap ratio using convex hull analysis (target >95%)
- Verify registration accuracy across multiple regions of interest
- Assess impact on downstream analytical results
This protocol has demonstrated overlap ratios of 98.0±2.3% for RGB-to-ChlF and 96.6±4.2% for HSI-to-ChlF registration in Arabidopsis thaliana studies, and 98.9±0.5% for RGB-to-ChlF and 98.3±1.3% for HSI-to-ChlF in Rosa × hybrida infection assays [20].
Hyperspectral Analysis of Multiple Co-Infections
The following experimental protocol details the procedure for hyperspectral imaging and classification of multiple concurrent infections in wheat, as demonstrated in recent research [18]:
Plant Material and Growth Conditions:
- Wheat variety 'Vuka' (susceptible to yellow rust, mildew, and Septoria)
- Growth conditions: 17/11°C for 16/8 h light/dark cycle for 10 days post-sowing
- Pathogen isolates: Yellow rust (WYR 19/215), Mildew (NIAB 21-001), Septoria (R13 and R16)
Infection Protocol:
- Single infections: Inoculate at 10 days post-sowing with adjusted temperatures for each pathogen
- Combined infections: Stagger inoculations to synchronize symptom development
- Yellow rust + mildew: YR at day 10, mildew at day 13
- Yellow rust + Septoria: Septoria at day 10, YR at day 17
Hyperspectral Image Acquisition:
- Imaging system: Pushbroom hyperspectral scanner (400-1000 nm range)
- Spatial resolution: Adjust to capture leaf-level details (approximately 0.1 mm/pixel)
- Spectral resolution: 3-5 nm bandwidth across visible and NIR regions
- Standardization: Include white reference and dark current measurement for each session
Data Processing Pipeline:
- Preprocessing: Radiometric correction, noise reduction, and spectral normalization
- Feature extraction: Identify disease-specific spectral bands (e.g., 689 nm and 753 nm for early infection)
- Classification: Train convolutional neural networks (EfficientNet 2D CNN) on labeled data
- Validation: Assess accuracy using k-fold cross-validation and independent test sets
This methodology achieved 81% overall classification accuracy for single and concurrent infections, with 72% accuracy specifically for combined yellow rust and mildew infections [18].
Diagram 1: Multi-modal plant imaging workflow. This workflow integrates RGB, hyperspectral, and chlorophyll fluorescence data through precise image registration and analysis.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Essential Research Equipment for Multi-Modal Plant Imaging
| Equipment Category | Specific Examples | Technical Specifications | Primary Research Function |
|---|---|---|---|
| Hyperspectral Imaging Systems | Specim FX10/FX17, Living Optics camera | VNIR (400-1000 nm) or SWIR (1000-2500 nm) range; Spectral resolution: 3-12 nm | Capture detailed spectral signatures for biochemical analysis |
| RGB Imaging Systems | Scientific-grade CCD/CMOS cameras | High spatial resolution (20+ MP); Global shutter; Controlled illumination | High-resolution morphological assessment and visible symptom documentation |
| Chlorophyll Fluorescence Imagers | PhenoVation Plant Explorer XS | Modulated measuring light; Saturation pulse capability; Multiple fluorescence parameters | Quantify photosynthetic efficiency and early stress responses |
| Laboratory Scanners | Specim LabScanner 40×20 | Automated scanning stages; Integrated illumination; Calibration targets | Standardized hyperspectral data acquisition in controlled environments |
| Field Deployment Systems | Specim AFX series for UAV/drone | Lightweight design; Robust mounting; GPS synchronization | Airborne hyperspectral data collection for large-scale field studies |
| Data Processing Platforms | Python with scikit-learn, TensorFlow | HSI-specific libraries (Hyperspy, ENVI); GPU acceleration | Data preprocessing, analysis, and machine learning model development |
Data Analysis Framework: From Raw Data to Actionable Insights
Hyperspectral Data Preprocessing and Feature Extraction
Raw hyperspectral data requires extensive preprocessing to extract meaningful biological information. The standard workflow includes:
Radiometric Correction:
- Dark current subtraction to remove sensor noise
- White reference normalization using spectralon panels
- Conversion to reflectance values for comparative analysis
Spectral Preprocessing:
- Savitzky-Golay smoothing to reduce high-frequency noise
- Standard Normal Variate (SNV) transformation to minimize scattering effects
- First and second derivative analysis to enhance subtle spectral features
- Continuum removal to emphasize absorption features [21] [23]
Feature Extraction:
- Vegetation indices calculation (NDVI, PRI, etc.) for specific physiological traits
- Spectral Disease Indices (SDIs) development for pathogen-specific detection
- Principal Component Analysis (PCA) for dimensionality reduction
- Identification of key wavelength regions for specific stresses:
- 550-600 nm: Pigment composition changes
- 690 nm: Chlorophyll fluorescence and photosynthetic activity
- 1400-1450 nm: Water content and hydric stress
- 753 nm: Early stress response detection [21]
Machine Learning Integration for Disease Classification
The high dimensionality of hyperspectral data makes it particularly suitable for machine learning approaches. Recent advances demonstrate compelling performance:
Algorithm Selection:
- Random Forest: Effective for feature selection and classification
- Convolutional Neural Networks (CNNs): Superior for spatial-spectral feature extraction
- EfficientNet with 2D convolution: 81% accuracy for multiple infection classification
- Vision Transformers: Emerging approach for capturing long-range dependencies [22] [18]
Model Training Strategies:
- Transfer learning to overcome limited dataset sizes
- Data augmentation through spectral transformations
- Attention mechanisms to focus on diagnostically relevant regions
- Ensemble methods to improve generalization across environmental conditions
Diagram 2: Hyperspectral data analysis pipeline. This pipeline transforms raw hypercubes into actionable insights through preprocessing, feature extraction, and machine learning.
The strategic integration of RGB and hyperspectral imaging technologies represents a paradigm shift in plant research methodology. While RGB provides cost-effective morphological data with high spatial resolution, hyperspectral imaging delivers unparalleled biochemical insight through spectral analysis. The combination enables researchers to correlate visible symptoms with their underlying physiological causes, creating a more comprehensive understanding of plant health and stress responses.
Current research demonstrates the practical viability of this integrated approach, with successful applications in disease detection, nutrient stress monitoring, and plant phenotyping. As both hardware and analytical methods continue to advance—with improvements in sensor miniaturization, computational efficiency, and machine learning algorithms—the fusion of morphological and spectral data will become increasingly accessible to researchers across diverse agricultural and botanical disciplines. This multimodal approach promises to accelerate breeding programs, enhance sustainable agricultural practices, and provide new insights into plant-pathogen interactions, ultimately contributing to global food security in the face of climate change and emerging plant diseases.
In plant research, the transition from traditional Red-Green-Blue (RGB) imaging to advanced hyperspectral imaging represents a fundamental shift from superficial visual assessment to deep physiological probing. RGB imaging, which captures reflectance in three broad visible wavelength bands (approximately 650 nm, 520 nm, and 475 nm), has long been the workhorse for digital phenotyping, providing excellent data on morphological traits such as leaf area, plant architecture, and visible color changes [24] [25]. However, this technology operates with the same fundamental limitation as the human eye: it can only perceive what is already visually apparent. By the time stress symptoms become visible in the RGB spectrum, physiological damage has often already progressed significantly, compromising research interventions and agricultural management [22] [25].
Hyperspectral imaging (HSI) shatters this limitation by capturing reflected light across hundreds of narrow, contiguous spectral bands, typically ranging from the visible spectrum into the near-infrared (NIR) and short-wave infrared (SWIR) regions (approximately 400-2500 nm) [2] [26]. This creates a continuous spectral signature for each pixel in an image, forming a rich three-dimensional data cube that contains both spatial and spectral information. This technological difference is not merely incremental; it is transformative, enabling researchers to detect subtle changes in plant biochemistry that precede visible symptoms by days or even weeks [22]. This whitepaper delineates the critical blind spots of RGB imaging that hyperspectral technology illuminates, framing this analysis within the compelling thesis that the synergistic combination of both modalities provides the most powerful approach for comprehensive plant research.
The Technical Divide: A Comparative Foundation
The core distinction between these imaging modalities lies in their spectral resolution and range. RGB imaging is limited to three broad bands in the visible spectrum, making it highly effective for quantifying what is visible to the human eye but incapable of probing biochemical composition. In contrast, hyperspectral imaging captures a full spectral profile, with modern systems covering from 350-1000 nm (VNIR) and extending to 2500 nm (SWIR) at resolutions as fine as 1-3 nm [26] [27]. This allows for the identification of specific molecular absorption features related to plant pigments, water content, structural compounds, and other biochemical constituents [28].
Table 1: Fundamental Technical Comparison Between RGB and Hyperspectral Imaging
| Parameter | RGB Imaging | Hyperspectral Imaging |
|---|---|---|
| Spectral Bands | 3 broad bands (Red, Green, Blue) [25] | 50-250+ narrow, contiguous bands [2] |
| Spectral Range | ~400-700 nm (Visible light only) [25] | ~350-2500 nm (VIS, NIR, SWIR) [26] [28] |
| Primary Data Output | 2D image with color information | 3D hypercube (x, y, λ) with spectral signatures [26] |
| Key Measurables | Morphology, color, texture | Biochemical composition, water content, pigment ratios [28] [27] |
| Cost & Accessibility | Low cost, highly accessible [22] | High cost ($20,000-$50,000+), requires expertise [22] |
Critical Blind Spots in RGB Imaging and Hyperspectral Solutions
Pre-Symptomatic Disease and Stress Detection
Perhaps the most significant blind spot of RGB imaging is its inability to detect plant stress before visible symptoms manifest. Research indicates that physiological and biochemical changes within plant tissues, such as alterations in cell structure and pigment composition, occur significantly before these changes become visible as discoloration, lesions, or wilting [22]. Hyperspectral imaging fills this void by detecting subtle spectral shifts associated with these early physiological responses.
A systematic review of disease detection methods revealed that while RGB-based deep learning models can achieve 95-99% accuracy in laboratory conditions, their performance drops dramatically to 70-85% in field deployments due to environmental variability and the challenge of identifying early infections [22]. Hyperspectral systems, particularly those leveraging Transformer-based architectures like SWIN, demonstrate superior robustness, maintaining around 88% accuracy in real-world conditions for identifying diseases like powdery mildew before symptom visibility [22]. This early detection capability, sometimes occurring days before visual symptoms, provides a critical window for intervention that can prevent significant crop loss and reduce unnecessary pesticide applications [2].
Quantification of Photosynthetic Pigments and Biochemical Constituents
While RGB imaging can detect gross color changes associated with chlorophyll degradation (e.g., yellowing leaves), it cannot accurately quantify specific pigment concentrations or distinguish between different photosynthetic pigments. Hyperspectral imaging directly targets the specific absorption features of key plant pigments across the electromagnetic spectrum.
A landmark study on Ginkgo biloba involving 3,460 seedlings from 590 families demonstrated the power of hyperspectral imaging combined with machine learning (Adaptive Boosting algorithm) to non-destructively quantify chlorophyll a, chlorophyll b, and carotenoids with remarkable accuracy (R² > 0.83, RPD > 2.4) [27]. The study implemented a sophisticated analytical workflow:
- Hyperspectral Data Acquisition: A portable Image-λ-V10E-HR hyperspectral imager (350-1000 nm range, 176 spectral channels) captured spectral data from all samples in a controlled darkroom environment with halogen lamp illumination to ensure consistency [27].
- Phased Optimization Strategy: The methodology involved: (1) screening preprocessing methods (raw reflectance, normalization, derivatives), finding normalization most effective; (2) comparing machine learning algorithms (PLSR, RF, AdaBoost); and (3) applying feature wavelength selection (SPA, CARS) to optimize the model [27].
- Biochemical Validation: Following spectral capture, leaf discs were destructively sampled for conventional biochemical analysis to create the ground-truth dataset for model training and validation [27].
This non-destructive approach enables large-scale, dynamic monitoring of pigment remodeling during critical physiological transitions, such as autumn senescence—a capability far beyond the reach of RGB imaging or traditional destructive sampling methods [27].
Figure 1: Experimental workflow for non-destructive pigment quantification in Ginkgo biloba using hyperspectral imaging and machine learning, as detailed in [27].
Early Water and Nutrient Stress Identification
Drought and nutrient deficiencies trigger specific biochemical responses in plants that hyperspectral imaging can identify before these stresses manifest as wilting or chlorosis in RGB images. The short-wave infrared (SWIR) region (894-2504 nm) is particularly sensitive to water content and molecular bonds in compounds like nitrogen-based proteins [28].
Advanced research now involves sensor fusion to create a more comprehensive picture of plant health. A study on strawberry plants developed separate hyperspectral imaging systems for the VIS-NIR (397-1003 nm) and SWIR (894-2504 nm) regions, then fused the data to significantly improve the identification of drought stress [28]. The fusion process required sophisticated image registration and alignment techniques to create a composite image combining the enhanced spectral information from both sensors. This fused data more accurately differentiated between control, recoverable, and non-recoverable plants before the emergence of visually apparent indicators [28]. The VIS-NIR region is sensitive to photosynthetic pigments, while the SWIR region provides stronger features related to water and protein content, demonstrating the value of a broad spectral range for stress detection [28].
Fine-Scale Growth Stage Classification
RGB imaging struggles to differentiate between closely related growth stages, particularly in the pre-anthesis phase where morphological changes are subtle. Research on genetically modified wheat classification highlights this limitation, demonstrating the superiority of hyperspectral imaging for distinguishing between fine-scale Zadoks growth stages Z37, Z39, and Z41 (flag leaf just visible to flag leaf sheath extending) [3].
The experimental protocol involved:
- Data Collection: Capturing hyperspectral and RGB images of individual wheat plants in both controlled greenhouse and semi-natural environments using a WIWAM hyperspectral imaging system with a Specim FX10 camera (400-1000 nm range) [3].
- Spectral Transformations: Applying spectral transformations like Standard Normal Variate (SNV), Hyper-hue, and Principal Component Analysis (PCA) to enhance spectral features [3].
- Machine Learning Classification: Using Support Vector Machine (SVM) classification, which achieved an F1 score of 0.832 for growth stage classification, significantly outperforming RGB-based methods [3].
Notably, after feature selection, the model maintained high accuracy (F1 score of 0.752) using only five key wavelengths, demonstrating that specific spectral features are critically responsible for distinguishing these subtle growth stages—features completely absent in RGB data [3].
Table 2: Quantitative Performance Comparison for Key Agricultural Applications
| Application | RGB Performance Limitations | Hyperspectral Performance Advantages |
|---|---|---|
| Early Disease Detection | 70-85% accuracy in field conditions [22] | 88% accuracy with SWIN Transformer; pre-symptomatic detection [22] |
| Pigment Quantification | Limited to color change detection; cannot quantify specific pigments [24] | Quantifies Chl a, Chl b, Carotenoids (R² > 0.83) [27] |
| Water Stress Detection | Relies on visible wilting; late detection [28] | Identifies water content changes in SWIR region before wilting [28] |
| Growth Stage Classification | Poor accuracy for fine-scale stages (Z37-Z41) [3] | SVM classification achieves F1 score of 0.832 [3] |
| Nutrient Deficiency | Detects advanced chlorosis only | Identifies specific nutrient deficiencies via unique spectral signatures [2] |
The Synergistic Approach: Data Fusion for Enhanced Classification
The limitations of both modalities point toward a synergistic solution. While hyperspectral imaging provides superior biochemical insight, its high cost, computational complexity, and sometimes lower spatial resolution present practical challenges [22] [26]. RGB imaging remains more accessible, cost-effective, and superior for certain morphological analyses [24]. The most powerful approach, therefore, combines the strengths of both.
A compelling case study on vegetable soybean freshness classification demonstrates this principle effectively. Researchers developed a novel ResNet-R&H model that incorporates fused data from both RGB and hyperspectral images [29]. The fusion process involved:
- Hyperspectral Data Extraction: Using ENVI software to extract spectral information.
- RGB Image Reconstruction: Applying downsampling technology to standardize RGB images.
- Data Concatenation: Transforming 1D hyperspectral data into 2D space and concatenating it with RGB image data in the channel direction [29].
The results were striking: the fused-data model achieved a testing accuracy of 97.6%, a significant enhancement of 4.0% and 7.2% compared to using only hyperspectral or RGB data, respectively [29]. This demonstrates that the spatial and textural information from RGB images complements the biochemical information from hyperspectral data, creating a more robust and accurate classification system.
Figure 2: Data fusion workflow for vegetable soybean freshness classification, combining RGB and hyperspectral inputs to achieve superior accuracy [29].
The Scientist's Toolkit: Essential Research Reagent Solutions
Implementing a hyperspectral or multimodal imaging research program requires specific hardware, software, and analytical tools. The following table details key solutions mentioned across the cited research.
Table 3: Essential Research Reagent Solutions for Hyperspectral Plant Phenotyping
| Solution Category | Specific Tool / Platform | Research Application & Function |
|---|---|---|
| Hyperspectral Imagers | Specim FX10 camera (400-1000 nm) [3] | Wheat growth stage classification in controlled environments [3] |
| Specim IQ camera (397-1000 nm) [30] | Plant phenotyping in fabricated ecosystems (EcoFABs) [30] | |
| Image-λ-V10E-HR (350-1000 nm) [27] | Portable field-based pigment quantification in Ginkgo seedlings [27] | |
| Imaging Systems | WIWAM Hyperspectral Imaging System [3] | Integrated system with LemnaTec 3D Scanalyzer for high-throughput phenotyping [3] |
| Line-scan HSI systems (VIS-NIR & SWIR) [28] | Custom-assembled systems for drought stress detection in strawberries [28] | |
| Analytical Software | ENVI Software [29] | Industry-standard software for hyperspectral data extraction and analysis [29] |
| SpecVIEW (v2.9.3.8) [27] | Control software for hyperspectral image acquisition and calibration [27] | |
| Machine Learning Algorithms | Support Vector Machine (SVM) [3] | Classification of fine-scale wheat growth stages (Z37, Z39, Z41) [3] |
| Adaptive Boosting (AdaBoost) [27] | High-accuracy non-destructive prediction of photosynthetic pigments [27] | |
| ResNet-based Models [29] | Deep learning architecture for fused RGB-HSI data classification [29] | |
| Sparse Mixed-Scale Networks (SMSNets) [30] | Convolutional neural networks for hyperspectral image segmentation with minimal training data [30] |
The "blind spots" of RGB imaging in plant research are not merely minor technical limitations but fundamental gaps in our ability to understand and monitor plant physiology at a biochemical level. Hyperspectral imaging effectively illuminates these blind spots by enabling pre-symptomatic stress detection, precise pigment quantification, early water and nutrient deficiency identification, and fine-scale growth stage classification. The evidence from contemporary research overwhelmingly supports a integrated approach where the morphological strengths of RGB imaging are combined with the biochemical probing capabilities of hyperspectral sensing. This multimodal paradigm, facilitated by advanced machine learning and data fusion techniques, represents the future of precision plant science, offering unprecedented insights into plant health, productivity, and physiology from the laboratory to the field.
From Lab to Field: Methodologies and Real-World Applications in Plant Science
Non-invasive plant phenotyping has emerged as a transformative discipline in agricultural research, enabling the precise quantification of plant growth, health, and development without disturbing the organism or its environment. By leveraging advanced imaging technologies, researchers can capture detailed phenotypic traits throughout the plant lifecycle, facilitating breakthroughs in plant breeding, stress response analysis, and precision agriculture. This technical guide focuses specifically on the synergistic integration of RGB (Red, Green, Blue) and hyperspectral imaging technologies, which together provide complementary data streams that significantly enhance research capabilities across diverse agricultural applications.
The fundamental advantage of combining these imaging modalities lies in their ability to capture different aspects of plant physiology and biochemistry. RGB imaging excels in quantifying morphological and structural traits with high spatial resolution, while hyperspectral imaging detects biochemical and physiological changes through detailed spectral signatures across hundreds of narrow, contiguous wavelength bands [9] [31]. This multi-modal approach enables researchers to correlate visual characteristics with underlying biochemical processes, providing a more comprehensive understanding of plant status than either technology could deliver independently.
Recent technological advancements have made both RGB and hyperspectral imaging more accessible to research institutions. The development of compact, automated phenotyping systems like PhenoGazer, which integrates hyperspectral spectrometers with multiple Raspberry Pi cameras and LED lighting systems, demonstrates the trend toward integrated solutions that capture complementary data types simultaneously [32]. Similarly, NASA's hyperspectral plant health monitoring system for space crop production exemplifies how these technologies can be deployed in controlled environments for precise health assessment [33]. These systems represent a paradigm shift from manual, destructive sampling approaches toward automated, non-invasive monitoring that supports higher-throughput phenotyping with minimal human intervention.
Technological Foundations: RGB and Hyperspectral Imaging
RGB Imaging Technology
RGB imaging represents the fundamental approach to digital plant phenotyping, capturing reflected light in three broad wavelength bands corresponding to red (approximately 650 nm), green (520 nm), and blue (475 nm) [31]. This technology emulates human vision but with greater consistency and quantitative capabilities. Modern RGB sensors deployed in phenotyping applications range from standard digital cameras to specialized scientific imaging systems with calibrated output.
The primary strength of RGB imaging lies in its high spatial resolution and cost-effectiveness for quantifying morphological traits. Applications include measuring plant architecture, leaf area, growth rates, and visible symptoms of stress or disease [24]. However, standard RGB imaging is limited to the visible spectrum (400-700 nm) and cannot detect biochemical changes or pre-visual stress indicators [9]. Despite this limitation, recent advances in image analysis algorithms, particularly those incorporating machine learning, have expanded the utility of RGB imaging for certain physiological assessments when combined with appropriate validation methods.
Hyperspectral Imaging Technology
Hyperspectral imaging represents a significant technological advancement beyond conventional RGB imaging by capturing spectral information across hundreds of narrow, contiguous bands spanning extended wavelength ranges [8]. While typical systems cover the visible to near-infrared (VNIR, 400-1000 nm), advanced systems extend into short-wave infrared (SWIR, 1000-2500 nm) ranges, enabling detection of a wider array of biochemical properties [31].
The core principle of hyperspectral imaging is that each material possesses a unique spectral signature or "fingerprint" based on its chemical composition and how it interacts with electromagnetic radiation [9]. This signature is represented as a spectral reflectance curve, which plots reflectance values against wavelengths. Plants under different stress conditions exhibit characteristic alterations in these spectral profiles, enabling early detection before visible symptoms manifest [34].
Hyperspectral data is structured as a three-dimensional "hypercube" comprising two spatial dimensions and one spectral dimension [8]. This rich dataset allows researchers to identify subtle changes in plant physiology through various analytical approaches, including spectral indices, machine learning classification, and spectral unmixing techniques. The technology's ability to detect pre-symptomatic stress responses, nutrient deficiencies, and pathogen infections makes it particularly valuable for precision agriculture and plant breeding applications [35].
Table 1: Comparative Analysis of RGB and Hyperspectral Imaging Technologies
| Parameter | RGB Imaging | Hyperspectral Imaging |
|---|---|---|
| Spectral Bands | 3 broad bands (R, G, B) [31] | Hundreds of narrow, contiguous bands [9] |
| Spectral Range | 400-700 nm (visible spectrum) [31] | Typically 400-2500 nm (VNIR-SWIR) [8] |
| Spatial Resolution | High | Variable, often lower due to spectral data volume |
| Information Captured | Morphological features, color, texture [24] | Biochemical composition, pigment content, water status [36] |
| Early Stress Detection | Limited to visible symptoms | Pre-visual detection possible [34] |
| Cost & Accessibility | Low to moderate | Moderate to high |
| Data Volume | Moderate | Very high (3D hypercubes) [8] |
Integrated Experimental Design and Methodologies
System Configuration and Hardware Integration
Designing an effective integrated phenotyping system requires careful consideration of hardware components, illumination conditions, and spatial configuration. The PhenoGazer system exemplifies this approach, combining a portable hyperspectral spectrometer with eight fiber optics, four Raspberry Pi cameras, and specialized blue LED lights within an automated movable rack system [32]. This configuration enables comprehensive assessment throughout the crop growth cycle, with the upper rack carrying spectrometer fiber optics and RGB cameras for daytime hyperspectral reflectance and morphological imaging, while the lower rack equipped with blue LED lights captures chlorophyll fluorescence at night [32].
Illumination represents a critical factor in system design. Controlled artificial lighting eliminates environmental variability and ensures consistent data acquisition. Halogen lights have traditionally been used for hyperspectral reflectance imaging but generate significant heat, which can affect tender plants [33]. Recent systems increasingly utilize LED technology, which offers cooler operation and specific wavelength capabilities. For instance, NASA's hyperspectral system employs LED line lights providing VNIR broadband and UV-A (365 nm) illumination for sequential reflectance and fluorescence measurements [33]. The integration of both illumination types within a single automated imaging cycle demonstrates advanced system design for comprehensive plant assessment.
Spatial configuration must align with research objectives. Laboratory-based systems typically employ close-range imaging (centimeters to meters) with high spatial resolution, while field-based systems may utilize unmanned aerial vehicles (UAVs) or ground vehicles covering larger areas with lower spatial resolution [8]. The sensor-to-sample arrangement varies accordingly, with stationary plants and moving sensors common in controlled environments, while moving samples with stationary sensors suit conveyor-based systems [33].
Data Acquisition Protocols
Standardized data acquisition protocols ensure consistency and reproducibility in integrated phenotyping studies. The protocol developed for NASA's plant health monitoring system illustrates a rigorous approach, acquiring both hyperspectral reflectance and fluorescence images sequentially during one imaging cycle [33]. Reflectance imaging utilizes VNIR broadband illumination, while fluorescence imaging employs UV-A (365 nm) excitation, with automated translation of the imaging assembly over stationary plants to ensure complete coverage.
Temporal resolution represents another critical consideration. Growth and stress responses develop over time, requiring appropriate sampling intervals. For drought stress detection in lettuce, NASA's system demonstrated capability to identify stress within the first four days of treatment, before visible symptoms appeared [33]. Similarly, phenotyping of soybean plants under different conditions (well-watered, droughted, diseased) required continuous monitoring throughout the growth cycle to capture dynamic responses [32].
Calibration procedures must be implemented consistently to ensure data quality. These include capturing white and dark reference images for hyperspectral data normalization [3], geometric calibration for spatial measurements, and radiometric calibration for quantitative reflectance analysis. The use of standardized reference targets within the imaging scene facilitates cross-comparison between imaging sessions and different sensor systems.
Data Processing and Analysis Methods
The integration of RGB and hyperspectral data requires sophisticated processing pipelines to extract meaningful biological information. The initial stage involves preprocessing operations specific to each data type. For RGB images, this typically includes background segmentation, color normalization, and morphological filtering [24]. For hyperspectral data, preprocessing encompasses radiometric calibration, spectral smoothing, and correction for illumination effects using standardization approaches like Standard Normal Variate (SNV) transformation [3].
Data fusion represents a critical step in leveraging the complementary strengths of both imaging modalities. Spatial registration aligns corresponding regions across datasets, enabling correlative analysis. Feature extraction then identifies relevant characteristics: shape, texture, and color metrics from RGB images; spectral indices, absorption features, and principal components from hyperspectral data [3]. The combined feature set provides a comprehensive representation of plant status encompassing both structural and biochemical attributes.
Machine learning approaches have demonstrated remarkable effectiveness for analyzing these complex multimodal datasets. Support Vector Machine (SVM) classifiers achieved over 90% accuracy for early detection of drought stress in lettuce using VNIR reflectance spectra [33]. Similarly, optimized discriminant classifiers successfully distinguished closely spaced wheat growth stages (Z37, Z39, Z41) using hyperspectral data with selected feature wavelengths [3]. These models benefit from the rich feature sets derived from fused RGB and hyperspectral data, capturing subtle patterns indicative of physiological status.
Table 2: Experimental Protocols for Different Research Applications
| Research Application | Imaging Protocol | Key Measurements | Analysis Methods |
|---|---|---|---|
| Growth Stage Classification [3] | Top-view imaging daily from Z37 to Z41; Hyperspectral (400-1000 nm) + RGB | Spectral reflectance at key wavelengths; Morphological development | SVM classification with SNV transformation; Feature selection with 5 optimal wavelengths |
| Early Stress Detection [33] | Reflectance + fluorescence imaging cycle; Well-watered vs. drought treatment | Chlorophyll fluorescence; Vegetation indices; Canopy temperature | Machine learning classifiers; Spectral indices analysis; Accuracy assessment (>90% achieved) |
| Disease Identification [31] | Regular monitoring pre- to post-symptomatic; Laboratory and field settings | Spectral shifts in specific regions; Lesion development; Pigment changes | Spectral angle mapping; Disease-specific indices; Classification algorithms |
| Nutrient Management [35] | Multi-temporal imaging across nutrient treatments | Chlorophyll-related indices; Leaf area; Growth rates | SPAD correlation analysis; Vegetation indices; Growth modeling |
Applications in Plant Research
Growth Stage Monitoring and Development Trajectory
The integrated use of RGB and hyperspectral imaging has revolutionized growth stage monitoring by enabling precise classification of developmental phases based on both morphological and biochemical cues. Research on wheat growth stages demonstrates this capability, where hyperspectral imaging combined with machine learning achieved F1 scores of 0.832 for classifying closely spaced pre-anthesis stages (Z37, Z39, Z41) - a task challenging for visual assessment or RGB imaging alone [3]. The flag leaf stages (Z37-Z41) represent critical transitions from vegetative to reproductive growth, and accurate classification enables prediction of flowering times essential for regulated biotechnology field trials.
RGB imaging contributes high-resolution morphological data on flag leaf emergence, ligule visibility, and leaf sheath extension - visual indicators of growth stages. Simultaneously, hyperspectral imaging detects subtle biochemical changes associated with developmental transitions, including shifts in pigment composition, water content, and cell structure [3]. The combination provides redundant validation for growth stage classification while capturing ancillary data on plant health and physiological status.
The practical implications are significant for breeding programs and regulatory compliance. Australian regulations require researchers to identify the first plant expected to flower at least 14 days in advance, while US regulations mandate prediction within seven days of anticipated flowering [3]. Integrated imaging approaches automate this process, reducing labor-intensive visual inspections and enabling scalability of field trials for genetically modified crops.
Early Stress and Disease Detection
The combined imaging approach excels in detecting plant stress responses before visible symptoms manifest, creating a critical window for intervention. Hyperspectral imaging identifies biochemical and physiological changes, such as chlorophyll breakdown, anthocyanin accumulation, and altered water content, while RGB imaging monitors the progression of visible symptoms once they appear [34].
For drought stress detection in lettuce, NASA's hyperspectral system achieved classification accuracies exceeding 90% within the first four days of water stress treatment, before any visible symptoms or size differences were evident [33]. This pre-visual detection capability enables early intervention to mitigate stress impacts. Similarly, disease detection applications have demonstrated sensitivity to metabolic changes during early infection stages, before visible symptoms develop [31]. This is particularly valuable for containment of rapidly spreading pathogens in field conditions.
The integration of both imaging modalities creates a comprehensive stress response profile: hyperspectral data reveal the underlying physiological disruptions, while RGB data document visible manifestations and spatial progression. This combined approach supports precision agriculture through targeted interventions, reducing resource inputs while maintaining crop health and productivity.
Nutrient Management and Photosynthetic Efficiency
Integrated phenotyping provides sophisticated approaches to nutrient management by linking visual plant characteristics with biochemical status. RGB imaging tracks growth responses to nutrient availability through morphological metrics like leaf area, canopy development, and color changes [24]. Hyperspectral imaging detects subtle spectral shifts associated with nutrient deficiencies before they become visually apparent, enabling proactive adjustment of fertilization regimes [35].
Chlorophyll fluorescence imaging, often incorporated into advanced phenotyping systems, directly assesses photosynthetic efficiency - a sensitive indicator of plant health. The PhenoGazer system utilizes blue LED lights to induce chlorophyll fluorescence at night, providing complementary data to daytime reflectance measurements [32]. This combination offers insights into both the structural components (through reflectance) and functional aspects (through fluorescence) of photosynthetic apparatus.
The practical applications extend to precision nutrient management, where imaging data inform variable-rate fertilization strategies based on actual crop needs rather than predetermined schedules. This approach optimizes resource use, reduces environmental impact from excess fertilizer application, and maintains optimal plant nutrition throughout the growth cycle [34].
Essential Research Toolkit
Core Instrumentation and Platforms
Implementing integrated RGB-hyperspectral phenotyping requires specific instrumentation tailored to research scale and environment. For controlled environments, systems like the WIWAM hyperspectral imaging system integrated with LemnaTec 3D Scanalyzer provide automated, high-throughput phenotyping with controlled illumination [3]. These systems typically include hyperspectral cameras (e.g., Specim FX10 covering 400-1000 nm), scientific RGB cameras, and integrated lighting in an enclosed cabinet to eliminate environmental variability.
For field-based applications, platforms range from handheld devices to UAV-mounted systems. Handheld hyperspectral imagers enable leaf-level measurements with geo-referencing for field mapping [33]. UAV platforms provide larger spatial coverage with moderate spatial resolution, carrying miniaturized sensors like the Headwall Hyperspec or Cubert UHD 185 Firefly cameras [8]. Ground-based vehicles offer intermediate solutions, with higher payload capacity for multiple sensors while covering field-scale areas.
Emerging integrated systems like PhenoGazer demonstrate the trend toward combining multiple sensing modalities in unified platforms [32]. These systems coordinate RGB cameras, hyperspectral spectrometers, and specialized illumination (including blue LEDs for chlorophyll fluorescence) within automated movable racks, enabling comprehensive assessment throughout diurnal cycles and growth periods.
The computational demands of integrated phenotyping necessitate robust analytical infrastructure. Hyperspectral data alone generates substantial volumes, with 3D hypercubes containing hundreds of spectral bands per spatial pixel [8]. Combined RGB-hyperspectral datasets require significant storage capacity, processing power, and specialized software for analysis.
Spectral analysis tools form the foundation for hyperspectral data processing. These include algorithms for spectral preprocessing (normalization, smoothing, derivative analysis), feature selection (identifying informative wavelengths), and classification (matching spectral patterns to biological conditions). The research on wheat growth stages demonstrated that effective wavelength selection could maintain classification accuracy (F1 scores of 0.752) with only five optimal wavelengths, significantly reducing data dimensionality [3].
Machine learning frameworks have become essential for analyzing complex multimodal datasets. Support Vector Machines (SVM), Random Forests, and Convolutional Neural Networks (CNNs) have all demonstrated success in plant phenotyping applications [33] [3]. These algorithms benefit from the complementary information provided by RGB and hyperspectral data, often achieving higher accuracy than single-modality approaches.
Table 3: Research Reagent Solutions for Integrated Plant Phenotyping
| Category | Item | Technical Specifications | Research Application |
|---|---|---|---|
| Hyperspectral Cameras | Specim FX10 [3] | 400-1000 nm; 5.5 nm FWHM; 512 pixels/line | VNIR spectral analysis for stress detection |
| Specim FX17 [9] | 900-1700 nm; SWIR capability | Enhanced discrimination of biochemical constituents | |
| RGB Imaging Systems | Allied Vision Technologies GT330 [3] | High spatial resolution; Color calibration | Morphological trait extraction |
| Raspberry Pi cameras [32] | Compact; Programmable; Multi-carray arrangements | Multi-angle phenotyping in distributed systems | |
| Illumination Systems | Metaphase Technologies LED line lights [33] | Multiple wavelengths (428, 650, 810, 850, 915, 365 nm); Digital dimming control | Standardized illumination for reflectance/fluorescence |
| Blue LED arrays [32] | Specific blue wavelengths | Chlorophyll fluorescence induction | |
| Platforms & Integration | WIWAM Hyperspectral Imaging System [3] | Integrated with LemnaTec Scanalyzer; Automated conveyor | High-throughput controlled environment phenotyping |
| PhenoGazer [32] | Portable hyperspectral + 8 fiber optics + 4 RGB cameras | Multi-modal phenotyping in walk-in growth chambers | |
| Reference Materials | Spectralon targets | >99% reflectance; Diffuse reflection standard | White reference for spectral calibration |
| Color calibration charts | Known reflectance values; Color standards | RGB camera calibration and color accuracy |
Implementation Framework and Technical Considerations
System Design and Workflow Integration
Successful implementation of integrated RGB-hyperspectral phenotyping requires thoughtful system design aligned with research objectives. The fundamental decision involves choosing between simultaneous and sequential data acquisition. Simultaneous capture ensures perfect temporal alignment but requires sophisticated optical arrangements to co-register sensors. Sequential acquisition simplifies hardware design but necessitates careful synchronization and registration during data processing [32].
The workflow encompasses multiple stages from experimental planning through data interpretation. Initial system calibration establishes baseline performance with standardized references. Data acquisition follows standardized protocols for imaging geometry, illumination, and timing. Processing pipelines then extract relevant features from each modality before data fusion creates integrated datasets. Analytical methods translate these fused datasets into biological insights, validated against ground-truth measurements.
Scalability represents a critical consideration in system design. Laboratory systems offer high precision but limited throughput, while field systems provide larger coverage with potentially reduced resolution. The research purpose should dictate this balance - breeding programs may prioritize throughput for screening large populations, while physiological studies may emphasize detailed characterization of fewer specimens [3].
Data Management and Analytical Challenges
The integrated approach generates substantial data management challenges. A single hyperspectral hypercube can contain hundreds of megabytes, while time-series data across multiple plants and conditions quickly scales to terabytes. Effective data management requires structured storage, metadata standards, and efficient retrieval systems to support analysis.
Analytical challenges include data dimensionality reduction, since hyperspectral data contains大量redundant information. Feature selection methods identify optimal wavelength subsets that maintain classification accuracy while reducing computational demands [3]. Data fusion techniques must address scale differences between high-spatial-resolution RGB and potentially lower-spatial-resolution hyperspectral data, requiring sophisticated registration algorithms.
Validation remains essential for translating sensor data into biological understanding. Ground-truth measurements, whether through destructive sampling, manual ratings, or reference instruments like SPAD meters, establish the relationship between sensor readings and physiological status [34]. This validation enables accurate interpretation of imaging data and builds confidence in the non-invasive approach.
The integration of RGB and hyperspectral imaging technologies represents a powerful approach to non-invasive plant phenotyping, combining complementary data streams to provide comprehensive insights into plant health and development. This multimodal strategy leverages the high spatial resolution and morphological capabilities of RGB imaging with the biochemical sensitivity and pre-visual detection capabilities of hyperspectral analysis. Together, they enable researchers to correlate visual characteristics with underlying physiological processes, creating a more complete understanding of plant status than either modality could provide independently.
Current research demonstrates the effectiveness of this integrated approach across diverse applications, from growth stage classification and stress detection to nutrient management and disease monitoring. The development of automated systems like PhenoGazer and NASA's hyperspectral monitoring platform illustrates the trend toward streamlined, high-throughput phenotyping solutions that minimize human intervention while maximizing data quality [32] [33]. These systems support both controlled environment and field-based research, scaling from detailed physiological studies to breeding program applications.
Future advancements will likely focus on increasing accessibility through reduced costs, improved computational efficiency, and enhanced user interfaces. The development of optimized feature selection methods, as demonstrated in wheat growth stage classification where five key wavelengths maintained high accuracy [3], will help reduce data dimensionality and processing demands. Similarly, advances in machine learning will improve classification accuracy while requiring less specialized expertise for implementation. As these technologies continue to evolve, the integrated use of RGB and hyperspectral imaging will play an increasingly central role in plant research, supporting advancements in crop improvement, sustainable agriculture, and food security.
The increasing prevalence of agricultural stressors driven by climate change poses significant threats to global food security. Drought, nutrient deficiency, and pathogen infection collectively cause substantial yield reductions, with drought alone potentially reducing maize yields by up to 40% [37]. In this context, advanced imaging technologies have emerged as powerful tools for detecting plant stress before visible symptoms manifest. While traditional RGB imaging provides structural information, hyperspectral imaging captures detailed spectral data that reveals biochemical and physiological changes in plants. The integration of these complementary technologies—combining the accessibility of RGB with the rich spectral information of hyperspectral imaging—represents a transformative approach for plant research, enabling pre-symptomatic stress detection and intervention [13] [1] [38].
Technical Foundation of Plant Stress Detection
The Physiological Basis of Stress Indicators
Plants respond to stress through complex physiological changes that affect their light interaction properties. Photosynthesis is particularly vulnerable to environmental stressors, with photosystem II (PSII) often being the primary target [37]. Under stress conditions, the normal flow of electrons through photosynthetic pathways is disrupted, leading to:
- Increased reactive oxygen species (ROS) generation including singlet oxygen (^1O₂), hydrogen peroxide (H₂O₂), and superoxide anion radicals (O₂•⁻) [37]
- Stomatal closure and reduced CO₂ uptake, particularly under drought stress [37] [39]
- Altered chlorophyll fluorescence patterns as protective mechanisms dissipate excess light energy [37]
- Biochemical composition changes including pigment degradation and accumulation of compatible solutes [39]
These physiological alterations create detectable signatures across various spectral regions, forming the basis for imaging-based stress detection methodologies.
Hyperspectral versus RGB Imaging: A Comparative Analysis
The complementary strengths of hyperspectral and RGB imaging technologies provide researchers with a powerful toolkit for comprehensive plant stress assessment.
Table 1: Comparison of RGB and Hyperspectral Imaging Technologies
| Feature | RGB Imaging | Hyperspectral Imaging |
|---|---|---|
| Spectral Bands | 3 broad bands (Red, Green, Blue) [38] | Numerous narrow, contiguous bands (>20 channels) [13] [38] |
| Information Captured | Structural information, color patterns [1] | Biochemical composition, physiological status [13] [1] |
| Spatial Resolution | Typically high | Varies, often lower in specialized systems [38] |
| Cost & Accessibility | Low cost, widely available [38] | Higher cost, specialized equipment required [38] |
| Primary Strengths | Rapid structural assessment, ease of use | Early stress detection, material identification [13] |
| Data Density | Lower information density [38] | Higher information density, enabling spectral reconstruction [38] |
Advanced Detection Methodologies
Chlorophyll Fluorescence Imaging
Chlorophyll a fluorescence analysis represents one of the most sensitive methods for early stress detection, capable of identifying biotic stress as early as 15-30 minutes after insect herbivory or pathogen application [37]. This non-destructive technique monitors the quantum efficiency of PSII photochemistry, providing insights into the photosynthetic apparatus's functional status.
Key Chlorophyll Fluorescence Parameters:
- Fᵥ/Fₘ: Maximum quantum efficiency of PSII photochemistry
- qp: Fraction of open PSII reaction centers - identified as the most accurate indicator for early stress detection [37]
- ΦPSII: Actual quantum yield of PSII electron transport
- NPQ: Non-photochemical quenching dissipating excess energy as heat
Experimental Protocol for Chlorophyll Fluorescence Imaging:
- Dark-adapt leaves for 20-45 minutes depending on species and prior light exposure
- Apply measuring light (ML) to determine minimal fluorescence (F₀)
- Apply saturating light pulse to measure maximum fluorescence (Fₘ)
- Calculate variable fluorescence: Fᵥ = Fₘ - F₀
- Expose to actinic light (AL) to simulate photosynthetic conditions
- Monitor fluorescence parameters during light adaptation
- Analyze spatial heterogeneity of fluorescence parameters across leaf surface
The fraction of open PSII reaction centers (qp) has been established as particularly valuable for early stress detection, showing significant changes before visible symptoms appear [37].
Hyperspectral Imaging for Pre-Symptomatic Detection
Hyperspectral imaging extends beyond traditional spectroscopy by combining spatial and spectral information, enabling rapid scanning of non-homogeneous samples [13]. This technology detects subtle spectral signatures associated with stress-induced biochemical changes long before they become visible.
Key Stress Indicators Detectable via Hyperspectral Imaging:
- Pigment composition alterations: Chlorophyll degradation, anthocyanin accumulation
- Water content variations: Spectral shifts in water absorption bands
- Cell structure modifications: Changes affecting light scattering properties
- Nutrient-related patterns: Spectral signatures associated with nutrient deficiencies
Experimental Workflow for Laboratory Hyperspectral Imaging:
- Sample Preparation: Mount plant samples ensuring uniform presentation
- System Setup: Configure hyperspectral sensor, scanner, illumination, and software [13]
- Data Acquisition: Capture hyperspectral cubes across relevant wavelength ranges
- Calibration: Convert raw data to reflectance using reference standards
- Spectral Analysis: Identify stress-specific spectral features and develop classification models
- Validation: Correlate spectral findings with physiological measurements
dot code for Hyperspectral Imaging Workflow for Stress Detection
RGB to Hyperspectral Reconstruction
Emerging deep learning approaches now enable the reconstruction of hyperspectral information from standard RGB images, bridging the gap between accessibility and information richness [38]. This computational approach addresses the limitations of specialized HSI hardware, including cost, size, and spatial resolution constraints [38].
Deep Learning Architectures for Spectral Reconstruction:
- PixelFeatureNet: Processes individual pixels without spatial context using fully connected layers
- LocalFeatureNet: Incorporates local neighborhood information via convolutional layers
- U-Net: Leverages encoder-decoder structure with skip connections for medical imaging
- MST++: Employs transformer-based architecture with spectral-wise self-attention mechanisms
Performance Metrics for Reconstruction Quality:
- RMSE (Root Mean Square Error): Measures pixel-wise reconstruction accuracy
- SAM (Spectral Angle Mapper): Assesses spectral fidelity independent of illumination
- PSNR (Peak Signal-to-Noise Ratio): Evaluates image quality preservation
- SSIM (Structural Similarity Index): Quantifies structural information retention
Transformer-based models like MST++ have demonstrated superior performance in reconstructing both visible and extended spectral ranges, effectively predicting critical spectral profiles for stress identification [38].
Stress-Specific Detection Signatures
Drought Stress Detection
Drought stress triggers complex physiological responses including stomatal closure, reduced CO₂ uptake, hormonal imbalances, and oxidative damage through increased ROS production [37] [39]. These changes create distinctive spectral signatures detectable through imaging technologies.
Table 2: Drought Stress Detection Parameters and Methods
| Detection Method | Key Measurable Parameters | Detection Timeframe | Accuracy Indicators |
|---|---|---|---|
| Chlorophyll Fluorescence | qp (open PSII centers), NPQ, ΦPSII [37] | Onset of stress [37] | qp as most accurate indicator [37] |
| Hyperspectral Imaging | Water absorption bands, pigment ratios | Pre-visual (1-3 days before symptoms) | Spectral changes in NIR and SWIR regions |
| Thermal Imaging | Canopy temperature, stomatal conductance | Early stress phase | Temperature elevation >2°C above baseline |
| RGB-Based Reconstruction | Derived spectral features, color纹理 changes | Early to moderate stress | Correlation with reference HSI (RMSE <0.05) |
Nutrient Deficiency Identification
Nutrient deficiencies cause specific biochemical changes that alter spectral properties across various wavelength regions, enabling precise identification through hyperspectral analysis.
Nitrogen Deficiency:
- Chlorophyll reduction causing blue shift in red edge position
- Increased visible reflectance due to decreased absorption
- Characteristic changes in carotenoid to chlorophyll ratio
Phosphorus Deficiency:
- Leaf thickening and biochemical changes affecting NIR reflectance
- Anthocyanin accumulation creating specific absorption features
- Interaction with water absorption bands in SWIR region
Potassium Deficiency:
- Effects on stomatal regulation influencing thermal properties
- Alterations in leaf turgor and cell structure affecting light scattering
- Distinctive spatial patterns of symptom development
Pathogen and Disease Detection
Plant pathogens induce complex interactions that generate unique spectral fingerprints, often detectable before lesion formation or sporulation.
Early Detection Timeframes:
- Chlorophyll fluorescence changes detectable within 30 minutes of Botrytis cinerea application [37]
- Hyperspectral signatures identifiable 24-48 hours before visual symptoms
- RGB-based reconstruction enabling disease detection at early infection stages
Key Pathogen Detection Strategies:
- Spectral Feature Analysis: Identification of pathogen-specific absorption features
- Temporal Pattern Recognition: Monitoring spectral changes over time
- Spatial Heterogeneity Assessment: Analyzing patchiness of stress responses
- Multi-Model Integration: Combining spectral, fluorescence, and thermal data
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Materials for Plant Stress Imaging
| Research Material | Function/Application | Technical Specifications |
|---|---|---|
| Hyperspectral Imaging Systems | Capture spatial-spectral data cubes for material identification [13] | Spectral range (400-2500 nm), spatial resolution, SNR >500:1 [13] |
| Pulse-Amplitude Modulation (PAM) Fluorometer | Quantify chlorophyll fluorescence parameters [37] | Measuring light source, saturating pulse capability, detection sensitivity |
| Spectral Calibration Standards | Convert raw data to reflectance/radiance [13] | Known reflectance properties (typically 2-99%), spectral stability |
| Controlled Environment Chambers | Maintain standardized conditions for experiments | Temperature control (±1°C), humidity control (±5%), programmable lighting |
| Deep Learning Frameworks | RGB to hyperspectral reconstruction [38] | Support for CNN/transformer architectures, GPU acceleration |
| Reference Biochemical Assays | Validate spectral findings with physiological data | Chlorophyll extraction, ELISA for pathogens, ion content analysis |
Integrated Experimental Framework
The combination of RGB and hyperspectral imaging creates a powerful synergistic relationship for comprehensive plant stress assessment. RGB imaging provides broad accessibility, high spatial resolution, and rapid data acquisition, while hyperspectral imaging delivers detailed biochemical information and early detection capabilities [1] [38].
dot code for RGB and Hyperspectral Imaging Integration Logic
Implementation Workflow for Integrated Stress Monitoring:
- Baseline Establishment: Capture reference RGB and hyperspectral data from healthy plants
- Continuous RGB Monitoring: Deploy accessible RGB systems for frequent temporal assessment
- Targeted HSI Validation: Apply hyperspectral imaging to suspicious areas identified via RGB
- Model Training: Develop reconstruction algorithms using paired RGB-HSI datasets
- Multi-Scale Integration: Combine laboratory, field, and airborne imaging data [13]
- Decision Support: Generate actionable insights for precision treatment applications
This integrated framework enables researchers to leverage the complementary strengths of both imaging modalities, achieving both broad spatial coverage and detailed spectral analysis for comprehensive plant health assessment.
The integration of RGB and hyperspectral imaging technologies represents a paradigm shift in plant stress detection, moving from reactive symptom management to proactive physiological monitoring. By combining the accessibility and high spatial resolution of RGB systems with the rich biochemical information provided by hyperspectral imaging, researchers can achieve unprecedented capabilities for early stress identification. The emerging capability to reconstruct hyperspectral information from RGB images through advanced deep learning approaches further enhances the practicality and scalability of these methodologies. As these technologies continue to evolve, their integration will play an increasingly vital role in addressing the pressing challenges of global food security threatened by climate change, pathogen evolution, and resource limitations.
Pre-Symptomatic Disease Identification During the Incubation Period
The incubation period in plant pathology presents a critical diagnostic challenge, representing the time between pathogen infection and the appearance of visible symptoms. During this phase, covert physiological changes occur within plant tissues that evade conventional visual inspection yet offer the most promising window for intervention. Traditional disease management strategies, which typically react to visible symptoms, face significant limitations as infections become established and control measures less effective by the time symptoms manifest [40]. This diagnostic gap has driven the exploration of advanced imaging technologies capable of detecting pre-symptomatic infection signatures through subtle alterations in plant physiology, biochemistry, and structural integrity.
The integration of RGB and hyperspectral imaging represents a transformative approach to plant disease diagnostics, combining practical accessibility with deep physiological profiling. While RGB imaging captures visible symptoms in three broad wavelength bands (approximately 475 nm, 520 nm, and 650 nm), hyperspectral imaging extends this capability across hundreds of contiguous spectral bands from visible to near-infrared regions (400-1000 nm and beyond) [25]. This multi-modal framework enables researchers to correlate visible symptom progression with underlying physiological changes detectable only through spectral analysis, creating a comprehensive diagnostic picture from pre-symptomatic to symptomatic disease stages.
Technological Foundations of Pre-Symptomatic Detection
Hyperspectral Imaging Mechanisms
Hyperspectral imaging systems generate three-dimensional data structures known as hypercubes, comprising two spatial dimensions and one spectral dimension [18]. Each pixel within a hyperspectral image contains a complete reflectance spectrum, providing a detailed fingerprint of the biochemical and biophysical properties of the imaged tissue. This rich spectral data enables the identification of pre-symptomatic infection markers through several key mechanisms:
- Chlorophyll Alteration Detection: Pathogen activity often disrupts chloroplast function and chlorophyll composition, creating detectable shifts in the red edge region (680-750 nm) before visible chlorosis appears [18] [41].
- Water Stress Signatures: Fungal hyphae or bacterial colonies can interfere with water transport and regulation, altering reflectance in water absorption bands (970 nm, 1200 nm, 1450 nm) [18].
- Cell Structure Changes: Subcellular damage and membrane disruption modify light scattering properties, particularly in near-infrared regions (750-1300 nm) where healthy plant tissues typically show high reflectance [41].
- Biochemical Defense Responses: Plants activate defense mechanisms during incubation, producing secondary metabolites with distinctive spectral signatures [42].
RGB Imaging Limitations and Complementary Value
Conventional RGB imaging remains constrained for pre-symptomatic detection by its limitation to the visible spectrum (approximately 400-700 nm), where biochemical changes typically become apparent only after substantial tissue damage has occurred [22] [25]. However, when integrated with hyperspectral data, RGB imagery provides essential spatial context and enables correlation between early spectral markers and subsequent visual symptom development. This complementary relationship forms the foundation for effective multi-modal disease diagnostics.
Experimental Methodologies for Incubation Period Detection
Hyperspectral Imaging Protocol for Pre-Symptomatic Pathogen Detection
The following protocol outlines a standardized approach for detecting plant diseases during the incubation period using hyperspectral imaging, adaptable for various pathosystems.
Plant Material Preparation and Pathogen Inoculation
- Plant Selection: Select uniform, healthy plants of susceptible varieties. For wheat studies, cultivar 'Vuka' has proven effective for pathogens like yellow rust, mildew, and Septoria [18].
- Growth Conditions: Maintain plants in controlled environments with species-appropriate day/night temperatures (e.g., 17/11°C for wheat) and photoperiods (e.g., 16/8 hours light/dark) until inoculation [18].
- Pathogen Inoculation: Apply pathogen suspensions using standardized methods:
- Multiple Infection Synchronization: For co-infection studies, stagger inoculation times to align symptom development (e.g., inoculate with yellow rust 7 days after Septoria for concurrent symptom expression) [18].
Hyperspectral Image Acquisition
- Imaging System Setup: Use a pushbroom or snapshot hyperspectral imaging system covering visible to near-infrared ranges (400-1000 nm or 400-2500 nm depending on application).
- Standardized Imaging Conditions:
- Maintain consistent illumination using stabilized halogen lights positioned at 45° angles to minimize specular reflectance.
- Include standard reflectance panels in each capture for radiometric calibration.
- Position leaves adaxial side up in Petri dishes with minimal background interference [18].
- Maintain fixed distance between sensor and sample (typically 30-50 cm depending on lens).
- Temporal Imaging Schedule: Capture images at regular intervals post-inoculation (e.g., daily) to track spectral changes throughout incubation period.
- Spectral Range Selection: Focus on diagnostically relevant regions while excluding noisy bands (typically 450-1000 nm after removing noisy edges) [41].
Data Preprocessing and Analysis
- Radiometric Correction: Convert raw digital numbers to reflectance values using reference panel data.
- Geometric Correction: Correct for spatial distortions using calibration frames.
- Region of Interest (ROI) Selection: Define multiple ROIs representing healthy and potentially infected tissues, excluding veins and damaged areas.
- Noise Reduction: Apply Savitzky-Golay smoothing or similar algorithms to reduce spectral noise while preserving meaningful features.
- Data Normalization: Use standard normal variate (SNV) or multiplicative scatter correction to minimize lighting and scattering effects.
The following workflow diagram illustrates the complete experimental process from sample preparation to disease classification:
RGB to Hyperspectral Image Conversion Protocol
For research settings with budget constraints, converting existing RGB images to simulated hyperspectral images (SHSI) provides an alternative approach to spectral analysis:
- Feature Extraction: Process RGB images through pre-trained deep learning models (ResNet50 demonstrates superior performance over VGG16 and EfficientNet-B7 for this application) [25].
- SHSI Generation: Use neural network generators to transform extracted features into simulated hyperspectral data cubes.
- Validation: Compare SHSI-derived vegetation indices (e.g., NDVI) with actual hyperspectral measurements using mean absolute error (MAE) and root mean squared error (RMSE) metrics [25].
- Disease Assessment: Apply spectral analysis techniques to SHSI data for disease detection and severity assessment.
Quantitative Data and Detection Performance
Spectral Signature Changes During Incubation Period
The table below summarizes key spectral features associated with pre-symptomatic disease detection across various pathosystems:
Table 1: Spectral Features for Pre-Symptomatic Disease Detection
| Wavelength Range | Physiological Association | Pathosystem Example | Detection Timing | Accuracy/Performance |
|---|---|---|---|---|
| 550-600 nm | Chlorophyll degradation | Tobacco TMV [41] | 2 days post-infection | >95% with LS-SVM classifiers |
| 689 nm & 753 nm | Early infection indicators | Multiple species [21] | Pre-symptomatic | Critical wavelengths for early detection |
| 670-700 nm | Chlorophyll-sensitive region | Sugar beet Fusarium [42] | Pre-symptomatic | Optimal for disease identification |
| 750-1000 nm | Cell structure collapse | Tobacco TMV [41] | 2-4 days post-infection | Reflectance decrease in infected tissue |
| 830-1000 nm | Near-infrared critical region | Sugar beet fungi [42] | Pre-symptomatic | Essential for disease type classification |
| 1400-1450 nm | Water stress indicators | Multiple species [21] | Early infection | Detect water status changes |
Detection Performance Across Imaging Modalities
Table 2: Performance Comparison of Imaging Modalities for Disease Detection
| Imaging Modality | Early Detection Capability | Laboratory Accuracy | Field Accuracy | Cost Range (USD) | Key Limitations |
|---|---|---|---|---|---|
| Hyperspectral Imaging | Excellent (pre-symptomatic) | 95-99% [22] | 70-85% [22] | $20,000-50,000 [22] | High cost, complex data processing |
| RGB Imaging | Poor (symptomatic stage) | 90-98% [22] | 50-75% [22] | $500-2,000 [22] | Limited to visible symptoms |
| Thermal Imaging | Good (stress indicators) | 80-90% [40] | 70-80% [40] | $1,000-5,000 | Environmental sensitivity |
| Simulated HSI from RGB | Moderate | 85-92% [25] | Under investigation | Lower cost | Dependent on model training |
Classification Algorithms for Pre-Symptomatic Detection
The table below compares machine learning approaches for detecting diseases during the incubation period:
Table 3: Algorithm Performance for Pre-Symptomatic Disease Classification
| Algorithm | Application Context | Key Features | Reported Accuracy | Advantages |
|---|---|---|---|---|
| EfficientNet (2D CNN) | Wheat multiple infections [18] | Hyperspectral image classification | 81% overall (72% for combined infections) | Balanced accuracy with computational efficiency |
| k-Nearest Neighbors (KNN) | Sugar beet soilborne diseases [42] | Multi-objective disease assessment | 97-100% identification, 99% classification | Highest performance in ICQP framework |
| Random Forest | Tobacco TMV detection [41] | Effective wavelength classification | >90% with selected wavelengths | Robust with high-dimensional data |
| LS-SVM/ELM | Tobacco TMV detection [41] | Data fusion (spectral + texture) | 95% with pre-symptomatic detection | Excellent with limited samples |
| YOLO-ESC | Plant disease detection [43] | Real-time object detection | 4.1% improvement over YOLOv8n | Enhanced multi-scale detection |
| SWIN Transformer | Plant disease benchmarks [22] | Real-world dataset processing | 88% accuracy vs. 53% for traditional CNNs | Superior robustness in field conditions |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Essential Research Materials for Pre-Symptomatic Disease Detection Studies
| Category | Specific Items | Technical Specifications | Application Purpose |
|---|---|---|---|
| Imaging Systems | Pushbroom hyperspectral camera | 400-1000 nm range, 204+ bands [42] | Primary spectral data acquisition |
| RGB camera | 1080P resolution, calibrated color | Reference imaging and symptom documentation | |
| Thermal imaging camera | 8×8 infrared sensor array [40] | Temperature variation monitoring | |
| Reference Materials | Reflectance calibration panel | 99%, 50%, and 5% reflectance standards | Radiometric calibration |
| Dark reference attachment | Light-tight enclosure | Dark current measurement | |
| Pathogen Materials | Fungal spore suspensions | 5×10⁵ spores/mL concentration [18] | Controlled inoculation |
| Virulent pathogen isolates | Characterized strains (e.g., WYR 19/215) [18] | Standardized infection protocols | |
| Computational Tools | Deep learning frameworks | TensorFlow, PyTorch | Model development and training |
| Spectral analysis software | ENVI, Python spectral libraries | Data preprocessing and analysis | |
| Experimental Supplies | Growth chamber facilities | Controlled temperature, humidity, light | Standardized plant growth |
| Sterile Petri dishes | 9 cm diameter with affixing medium [18] | Leaf sample presentation |
Integrated Data Fusion Framework
The combination of RGB and hyperspectral data creates a powerful diagnostic system that leverages the strengths of both modalities. The following diagram illustrates how information flows through this integrated framework:
This integrated framework enables:
- Early detection through hyperspectral identification of physiological changes
- Symptom validation via RGB documentation of visual disease progression
- Stress monitoring through thermal profiling of transpiration alterations
- Comprehensive assessment combining identification, severity quantification, and localization
Pre-symptomatic disease identification during the incubation period represents a paradigm shift in plant disease management, moving from reactive treatment to proactive intervention. The strategic integration of RGB and hyperspectral imaging creates a powerful diagnostic framework that captures both visible symptoms and underlying physiological changes, enabling researchers to identify infections before significant damage occurs. As these technologies continue to evolve—with advancements in sensor miniaturization, algorithmic efficiency, and cost reduction—their implementation in both research and commercial agriculture promises to significantly reduce crop losses and enhance global food security. The experimental methodologies and technical frameworks presented in this guide provide researchers with comprehensive tools to advance this critical field of study.
Chemotype Classification for Medicinal Plants and Quality Control
In the realm of medicinal plant science, chemotype classification serves as a fundamental discipline for ensuring the efficacy, safety, and quality of plant-derived therapeutics. A chemotype refers to a chemically distinct population within a plant species that produces a specific profile of secondary metabolites, which are often the primary bioactive compounds responsible for pharmacological effects. Unlike genotype variations, which involve genetic differences, chemotypic variations arise from differences in the expression of biochemical pathways. The precise identification of these chemical profiles is critical for medicinal plants, as their therapeutic value is directly linked to the presence and concentration of specific active compounds. Variations in these profiles can significantly alter a plant's medicinal properties, making accurate chemotyping an essential component of quality control in the production of herbal medicines and plant-based pharmaceuticals [44].
The integration of advanced analytical technologies has revolutionized chemotype classification. Traditional methods relied heavily on morphological identification, which could be unreliable due to phenotypic plasticity and environmental influences. Modern approaches now leverage sophisticated instrumentation including chromatography, spectroscopy, and molecular techniques to establish definitive chemical fingerprints. Furthermore, the emergence of imaging technologies, particularly hyperspectral imaging, offers a non-destructive, rapid, and comprehensive method for linking spatial characteristics with biochemical composition. This whitepaper explores the methodologies for chemotype classification and demonstrates how the synergistic combination of RGB and hyperspectral imaging creates a powerful framework for advanced medicinal plant research and quality assurance [44] [1].
Fundamentals of Chemotaxonomy
Chemotaxonomy, the science of using chemical characteristics to classify and identify plants, provides the theoretical foundation for chemotype classification. This discipline is predicated on the understanding that the production of secondary metabolites—such as alkaloids, flavonoids, terpenoids, and phenolic compounds—is genetically regulated and can therefore reflect evolutionary relationships and intraspecific diversity. These compounds are not directly involved in primary plant growth or development but serve ecological roles in defense and signaling. For medicinal plants, they are the primary sources of therapeutic activity [44].
Key Secondary Metabolites in Chemotype Classification
The following table summarizes the major classes of secondary metabolites central to chemotyping, their primary functions, and their roles in quality control.
Table 1: Key Secondary Metabolites in Medicinal Plant Chemotyping
| Metabolite Class | Pharmacological Activities | Role in Chemotyping & Quality Control | Example Medicinal Plants |
|---|---|---|---|
| Alkaloids | Analgesic, Anticancer, Antimalarial | Chemotaxonomic markers; quality control via potency and toxicity assessment | Opium poppy (Papaver somniferum), Cinchona |
| Flavonoids | Antioxidant, Anti-inflammatory, Cardioprotective | Profile determines chemotype; indicator of plant stress and processing | Milk Thistle (Silybum marianum), Ginkgo |
| Terpenoids | Anticancer, Antimicrobial, Anti-parasitic | Chemotype differentiation based on volatile oil composition; authenticity verification | Cannabis (Cannabis sativa), Tea Tree |
| Phenolic Acids & Tannins | Antioxidant, Astringent, Wound Healing | Quantification for standardization of herbal products | Green Tea, Oak, Witch Hazel |
The stability of these chemical profiles, while genetically controlled, can be influenced by environmental factors, harvest time, and post-harvest processing. Therefore, a robust chemotyping system must account for these variables to ensure consistent quality. Chemotaxonomy excels in differentiating between closely related species and cryptic species that are morphologically identical but chemically distinct, a common challenge in medicinal plant authentication with direct implications for drug safety and efficacy [44] [45].
Analytical Techniques for Chemotyping
A suite of analytical techniques is employed to determine the chemical profiles that define a chemotype. The choice of technique depends on the required sensitivity, resolution, and the specific class of metabolites being analyzed.
Chromatographic and Spectroscopic Methods
Chromatography forms the backbone of quantitative chemotype analysis, separating complex mixtures into individual components.
- High-Performance Liquid Chromatography (HPLC): Widely used for the quantification of non-volatile secondary metabolites like flavonoids and alkaloids. It allows for the precise measurement of individual compound concentrations, which is essential for standardizing herbal products [44].
- Gas Chromatography-Mass Spectrometry (GC-MS): Ideal for the separation and identification of volatile and semi-volatile compounds, such as terpenoids and essential oils. The coupled mass spectrometer provides definitive identification of each separated compound [44].
- Liquid Chromatography-Mass Spectrometry (LC-MS): This is a high-throughput technique that combines the separation power of LC with the identification capabilities of MS. Advanced systems like LC-MS-Qtof (Quadrupole Time-of-Flight) provide high mass accuracy and are indispensable for discovering novel bioactive compounds and for comprehensive metabolomic profiling [44].
Spectroscopic methods provide complementary information:
- Nuclear Magnetic Resonance (NMR) Spectroscopy: Particularly useful for the structural elucidation of unknown compounds. It provides a highly reproducible "fingerprint" of a plant extract, useful for both identification and quantification in a non-targeted manner [44].
- Fourier-Transform Infrared (FTIR) Spectroscopy: A rapid technique that probes the functional groups in a sample, providing a spectral fingerprint that can be used for preliminary classification and authentication [44].
Molecular and Data Analysis Techniques
- DNA Barcoding: This molecular technique uses short, standardized gene regions (e.g., rbcL, matK) to identify plant species at a genetic level. It is often used in conjunction with chemotyping to resolve taxonomic ambiguities and authenticate plant material, especially when morphological features are absent [45].
- Multivariate Analysis: The complex data generated by analytical instruments are processed using statistical techniques like Principal Component Analysis (PCA) and Cluster Analysis (CA). These methods reduce dimensionality and visualize patterns, allowing researchers to group plants with similar chemical profiles and objectively define chemotypes [44].
Table 2: Standard Experimental Protocols for Key Chemotyping Techniques
| Technique | Sample Preparation | Key Experimental Parameters | Data Output & Analysis |
|---|---|---|---|
| HPLC for Flavonolignans | Dry plant material ground and extracted with methanol via sonication. | C18 column; Mobile phase: water-acetonitrile gradient; Flow rate: 1.0 mL/min; Detection: UV at 288 nm. | Retention times and peak areas of silybin, silychristin, etc.; quantification against standards. |
| GC-MS for Terpenes | Hydro-distillation to obtain essential oil; dilution in hexane. | Non-polar capillary column; Temperature program: 60°C to 250°C; Ionization: EI. | Mass spectrum identification against libraries (NIST); relative percentage of terpene components. |
| LC-MS Metabolomics | Freeze-dried tissue extracted with methanol:water:formic acid. | C18 column; gradient elution; MS detection in positive/negative mode. | High-resolution mass data; metabolite identification using databases; multivariate stats (PCA). |
| DNA Barcoding | Genomic DNA extraction from silica-gel-dried leaves. | PCR amplification of matK and rbcL gene regions; Sanger sequencing. | Sequence alignment and comparison with reference databases (e.g., BOLD). |
The Imaging Revolution: RGB and Hyperspectral Data Fusion
While traditional analytical methods are highly accurate, they are often destructive, time-consuming, and limited to small samples. Imaging technologies offer a paradigm shift, enabling rapid, non-destructive, and spatial analysis of biochemical properties.
From RGB to Hyperspectral Imaging
RGB imaging, which captures light in three broad bands (red, green, blue), recreates a scene as perceived by the human eye. It is excellent for capturing structural information and can be used for basic plant health assessment and morphological phenotyping. However, its ability to quantify chemical changes is severely limited [1].
Hyperspectral imaging (HSI) bridges this gap. It captures light across hundreds of narrow, contiguous wavelength bands for every pixel in an image, generating a continuous spectrum for each point [2]. This creates a detailed three-dimensional data cube (x, y, λ) containing both spatial and spectral information. The resulting spectral signatures are unique to the biochemical composition of the material, allowing for the detection and mapping of specific compounds [2] [1].
The fusion of these technologies creates a powerful synergy. RGB images provide a high-spatial-resolution structural context, while hyperspectral data delivers deep biochemical insight. The combination allows researchers to correlate visual features with underlying chemical profiles directly on the plant tissue, without destruction.
Reconstructing Hyperspectral Data from RGB Images
A significant technological advancement is the reconstruction of hyperspectral images from standard RGB images, known as Spectral Super-Resolution (SSR). This is an active area of research in computer vision. Deep learning models, such as the recently proposed MSS-Mamba architecture, are trained to learn the complex mapping between a 3-band RGB image and its corresponding high-dimensional hyperspectral data cube [46].
This approach offers a low-cost alternative to physical hyperspectral imaging. By using a transformer-based model with a Continuous Spectral–Spatial Scan (CS3) mechanism, MSS-Mamba can effectively model long-range dependencies in the spectral and spatial domains, recovering a high-fidelity hyperspectral image from a single RGB input [46]. This makes the benefits of hyperspectral analysis more accessible, as it can be applied to existing RGB image databases or captured with standard cameras.
Diagram 1: SSR from RGB with MSS-Mamba
Integrated Workflow for Chemotype Classification and Quality Control
The integration of chemical analysis, genetics, and imaging provides the most robust framework for chemotype classification. The following workflow outlines the process from sample collection to final classification, highlighting the role of imaging at each stage.
Diagram 2: Integrated Chemotyping Workflow
- Sample Collection and Imaging: Fresh or dried plant material is first subjected to RGB and hyperspectral imaging. The RGB image documents morphology, while the HSI cube captures the initial spectral fingerprint.
- Data Fusion and Model Application: The spatial detail from the RGB image is fused with the spectral detail from the HSI. A pre-trained machine learning model analyzes the fused data to predict the chemical profile (e.g., concentration of key metabolites) and provide a preliminary chemotype classification.
- Laboratory Validation: The predictions are validated using gold-standard laboratory methods. A subsample of the plant material is analyzed via HPLC or GC-MS for quantitative chemical profiling. DNA barcoding is performed to confirm species identity, a critical step for authentication.
- Definitive Classification and QC: Data from all sources is integrated to make a final chemotype classification. This comprehensive report forms the basis for quality control, ensuring the plant material meets the required chemical specifications for its intended use.
Case Study: Chemotyping inSilybum marianum
The application of this integrated approach is effectively illustrated in the study of milk thistle (Silybum marianum), a medicinal plant prized for its hepatoprotective flavonolignan complex, silymarin.
Research on a wide germplasm collection has identified three stable chemotypes in S. marianum based on the relative concentrations of flavonolignans like silybin, silychristin, and silydianin [45]:
- Chemotype A: Characterized by high levels of both silychristin and silybin.
- Chemotype B: Dominated by silydianin.
- Chemotype C: An intermediate type, resulting from hybridization between A and B.
Crucially, while total silymarin content can be affected by environment, the relative proportions that define the chemotype are genetically regulated and exhibit high phenotypic stability [45]. In this study, chemical analysis (HPLC) was combined with DNA barcoding using the ITS2 region to correctly differentiate S. marianum from the closely related S. eburneum, which was found to possess a distinct, stable Chemotype D where isosilychristin is the predominant component [45]. This precise classification is vital for breeding programs and for guaranteeing consistent therapeutic outcomes in medicinal products derived from milk thistle.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Chemotyping Studies
| Item Category | Specific Examples | Function in Research |
|---|---|---|
| Chromatography Standards | Silybin, Silychristin, Geraniol, Caffeine, Morphine | Authentic chemical standards used for calibration, identification, and quantification of target metabolites in plant extracts. |
| DNA Extraction & PCR Kits | CTAB extraction buffers, DNeasy Plant Kits, PCR master mixes, primers for matK, rbcL, ITS2 | For isolating high-quality plant genomic DNA and amplifying specific barcode regions for species authentication. |
| Hyperspectral Imaging Systems | Portable snapshot cameras (e.g., Living Optics), line-scanner systems, UAV-mounted sensors | To capture spatial-spectral data cubes from plant samples in the lab or field for non-destructive chemical analysis. |
| Solvents & Reagents | HPLC-grade methanol, acetonitrile, water; derivatization agents for GC-MS | For the extraction, separation, and analysis of plant metabolites using chromatographic and spectroscopic techniques. |
| Data Analysis Software | Python/R libraries (scikit-learn, hyperSpec), VOSviewer, CiteSpace, SIMCA | For processing complex chemical and spectral data, performing multivariate statistics, and conducting bibliometric analysis. |
Chemotype classification is an indispensable tool for modernizing and securing the medicinal plant supply chain. By moving beyond morphological assessment to a precise, chemistry-based classification system, it ensures the standardized quality of herbal drugs, supports the discovery of novel bioactive compounds, and guides breeding programs for improved cultivars. The integration of advanced chemical profiling techniques with cutting-edge imaging technologies, particularly the fusion of RGB and hyperspectral data, creates a powerful, non-destructive, and high-throughput platform for analysis. The emerging capability to reconstruct hyperspectral information from low-cost RGB images using deep learning models like MSS-Mamba promises to make this powerful technology more accessible, further accelerating research and ensuring that the profound benefits of medicinal plants can be harnessed safely, effectively, and consistently for drug development and global health.
High-Throughput Screening in Breeding and Genetic Research
High-throughput screening (HTS) technologies are revolutionizing plant breeding and genetic research by enabling rapid, non-destructive, and precise phenotypic characterization of large populations. This technical guide explores the transformative potential of integrating RGB and hyperspectral imaging technologies—a synergistic approach that combines detailed morphological data with rich biochemical information. We provide a comprehensive analysis of the fundamental principles, detailed experimental protocols, and practical implementation frameworks that empower researchers to extract meaningful biological insights from complex phenotypic data. By bridging the gap between genotype and phenotype, these advanced imaging systems accelerate the development of improved crop varieties with enhanced yield, stress resilience, and quality traits, ultimately addressing global food security challenges in the face of climate change.
High-throughput plant phenotyping (HTP) has emerged as a critical discipline that addresses the longstanding bottleneck in plant breeding and genetic research: the detailed characterization of complex traits across large populations. Where traditional phenotyping methods rely on manual, often destructive measurements that are labor-intensive, time-consuming, and subject to human bias [47], HTP technologies automate this process through integrated imaging sensors and analytical platforms. These systems enable non-invasive, continuous monitoring of plant growth, structure, and physiology throughout the development cycle, providing unprecedented insights into gene function and plant-environment interactions [11].
The integration of multiple imaging modalities represents a paradigm shift in phenotypic screening. While RGB (Red, Green, Blue) imaging provides high-resolution morphological data including plant structure, leaf area, and color information [9], hyperspectral imaging (HSI) captures hundreds of contiguous spectral bands across a wide range of the electromagnetic spectrum (typically 400-2500 nm), revealing biochemical composition and physiological status that are invisible to the human eye or conventional cameras [11] [48]. This complementary approach generates multidimensional datasets that capture both structural and functional traits, enabling researchers to establish deeper correlations between genetic makeup and phenotypic expression.
Technological Foundations: RGB and Hyperspectral Imaging
RGB Imaging in Plant Phenotyping
RGB imaging systems mimic human vision by capturing reflected light in three broad wavelength bands corresponding to red, green, and blue colors. In plant phenotyping applications, RGB cameras provide high-spatial-resolution data for quantifying morphological and architectural traits. Advanced analysis of RGB images enables measurement of parameters including leaf area, plant biomass, growth rates, leaf angle distribution, and canopy coverage [49]. The strengths of RGB imaging include its cost-effectiveness, relatively simple data processing requirements, and ability to provide intuitively interpretable visual data. However, its limitation lies in capturing only superficial color information without insight into underlying biochemical processes [9].
Hyperspectral Imaging in Plant Phenotyping
Hyperspectral imaging operates on a fundamentally different principle, capturing both spatial and spectral information simultaneously to form a three-dimensional data structure known as a "hypercube" [11]. This hypercube contains complete spectral signatures for each pixel in the image, typically spanning hundreds of narrow, contiguous bands across the visible, near-infrared (NIR), and short-wave infrared (SWIR) regions [11]. Each material possesses a unique spectral signature based on its chemical composition and how it interacts with electromagnetic radiation [9]. In plants, these spectral fingerprints reveal information about pigment content, water status, nitrogen levels, and other biochemical constituents that serve as early indicators of plant health, stress response, and physiological status [48].
The key advantage of hyperspectral imaging is its ability to detect subtle changes in plant physiology before visible symptoms manifest [48]. For instance, water stress can be identified days before wilting becomes visible through changes in water absorption bands in the SWIR region, while nutrient deficiencies alter spectral signatures in specific wavelengths associated with photosynthetic pigments and protein content [48].
Comparative Analysis of Imaging Modalities
Table 1: Technical comparison of RGB and hyperspectral imaging technologies
| Parameter | RGB Imaging | Hyperspectral Imaging |
|---|---|---|
| Spectral Bands | 3 broad bands (Red, Green, Blue) [9] | Hundreds of contiguous narrow bands (e.g., 400-1000 nm VNIR, 900-1700 nm NIR) [9] [11] |
| Spatial Resolution | High (depends on sensor and optics) | Variable, typically lower due to spectral data acquisition |
| Information Depth | Surface morphology and color [9] | Biochemical composition and physiological status [9] [48] |
| Data Volume | Relatively low (3 values per pixel) | Very high (hundreds of values per pixel) [48] |
| Primary Applications | Growth monitoring, shape analysis, disease lesion identification | Early stress detection, nutrient status assessment, quality trait prediction [11] [48] |
| Cost Considerations | Lower cost, widely accessible | Higher sensor cost, requires specialized expertise [48] |
| Data Processing Complexity | Moderate | High, requires specialized spectral analysis tools [48] |
Table 2: Complementary strengths of RGB-hyperspectral integration in plant research
| Research Application | RGB Contribution | Hyperspectral Contribution | Synergistic Benefit |
|---|---|---|---|
| Disease Detection | Lesion identification, symptom progression tracking [9] | Pre-symptomatic detection, pathogen differentiation [11] [48] | Complete disease cycle monitoring from infection to symptom development |
| Nutrient Management | Overall plant vigor assessment, growth monitoring | Specific nutrient deficiency identification (N, P, K) [48] | Precise fertilizer application based on both growth status and nutrient needs |
| Stress Response | Canopy structure changes, wilting observation | Early stress indicators through spectral shifts [49] [48] | Comprehensive stress resilience profiling across severity levels |
| Yield Prediction | Morphological yield components (ear size, panicle architecture) [47] | Physiological yield determinants (photosynthetic efficiency, water status) [49] | More accurate and earlier yield forecasts |
| Quality Traits | Seed color, size, and shape analysis [47] | Biochemical composition (protein, oil, starch content) [9] | Integrated assessment of both visual and nutritional quality |
Experimental Protocols for Integrated Imaging
Protocol 1: High-Throughput Screening of Radiation-Induced Mutants
This protocol, adapted from studies on Arabidopsis thaliana mutants, demonstrates an automated pipeline for identifying phenotypic variants in populations generated through radiation mutagenesis (heavy ion beams and γ-radiation) [50].
Materials and Plant Preparation
- Plant Materials: Wild-type and mutant populations (e.g., M3 generation Arabidopsis thaliana)
- Growth System: Controlled environment growth chambers with standardized conditions (22±1°C, 60% RH, 18/6h photoperiod)
- Cultivation Media: Murashige and Skoog medium (0.8% agar, 1.5% sucrose) in specialized multi-well plates compatible with HTS systems
- Sterilization: 10% sodium hypochlorite for 15 minutes followed by sterile water rinsing [50]
Imaging System Configuration
- Platform: High-throughput plant imaging system (e.g., Scanalyzer HTS, LemnaTec)
- Sensors: Integrated RGB and hyperspectral cameras (e.g., Specim FX10 for VNIR: 400-1000 nm)
- Lighting: Controlled illumination cabinet with halogen lights to ensure consistent spectral acquisition
- Spatial Resolution: Camera positioned 1.4 m above plants, achieving approximately 2.6 mm/pixel resolution [3]
Data Acquisition Protocol
- Synchronized Imaging: Capture co-registered RGB and hyperspectral images daily throughout plant development cycle
- Reference Standards: Include spectral calibration panels in each imaging session
- Environmental Control: Maintain consistent imaging timing (12:00-2:00 PM) to minimize diurnal variation effects
- Data Management: Implement automated file naming and storage system for large datasets (approximately 10 GB for 4,635 image datasets) [50]
Data Analysis Pipeline
- Image Preprocessing: Background removal, flat-field correction for illumination uniformity
- Feature Extraction:
- RGB: Plant area, compactness, leaf count, color indices
- Hyperspectral: Vegetation indices (NDVI, PRI, Chlorophyll Index), spectral signatures across 400-1000 nm range
- Multivariate Analysis: Principal Component Analysis (PCA) to reduce dimensionality and identify spectral patterns
- Machine Learning Classification: Logistic Growth Curve analysis and scatter matrix clustering to identify phenotypic outliers [50]
Validation Methods
- Confirmation through manual phenotyping by trained researchers
- Physiological validation using chlorophyll fluorescence measurements
- Subcellular structural analysis to confirm phenotypic observations [50]
Protocol 2: Wheat Growth Stage Classification for Flowering Time Prediction
This protocol details an approach for fine-scale classification of wheat growth stages (Zadoks Z37, Z39, Z41) using hyperspectral imaging to predict flowering time—a critical requirement for regulated field trials of genetically modified crops [3].
Plant Growth Conditions
- Cultivar: Wheat (Triticum aestivum) 'Scepter' grown in greenhouse and semi-natural environments
- Growth Substrate: 1:1:1 (v/v/v) mixture of sand, clay, and UC soil mix
- Environmental Conditions: Greenhouse maintained at 18°C day/13°C night with controlled irrigation [3]
Hyperspectral Image Acquisition
- Imaging System: WIWAM hyperspectral system integrated with LemnaTec 3D Scanalyzer
- Camera: Specim FX10 with VNIR-2 spectrograph (400-1000 nm range, 5.5 nm FWHM resolution)
- Configuration: Top-down view with camera 1.4 m above plants, 38° lens
- Lighting: Enclosed cabinet with halogen lights to eliminate external light variation
- Calibration: Black and white reference images captured immediately after each plant scan [3]
Spectral Data Preprocessing
- Spectral Transformations:
- Standard Normal Variate (SNV) to remove scatter effects
- Hyper-hue transformation for illumination invariance
- Principal Component Analysis for dimensionality reduction
- Feature Selection: Identify most informative wavelengths for classification
- Data Partitioning: Split dataset into training and validation subsets [3]
Classification Modeling
- Algorithm: Support Vector Machine (SVM) with optimized kernel parameters
- Input Features: Spectral reflectance values from preprocessed hypercubes
- Output Classes: Three growth stages (Z37, Z39, Z41)
- Performance Metrics: F1 scores (achieving 0.832 with combined transformations) [3]
Implementation Considerations
- Model optimization with reduced feature sets (5 wavelengths maintained F1 score of 0.752)
- Evaluation of model performance under limited training data conditions
- Assessment of generalizability across different environmental conditions [3]
Integrated imaging workflow for high-throughput screening
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of high-throughput screening with RGB and hyperspectral imaging requires careful selection of specialized equipment, software tools, and analytical resources. The following table summarizes key components of an integrated plant phenotyping toolkit.
Table 3: Essential research reagents and solutions for high-throughput imaging
| Category | Specific Products/Models | Technical Specifications | Primary Function |
|---|---|---|---|
| Hyperspectral Cameras | Specim FX10 [9] [5] | VNIR (400-1000 nm), 5.5 nm spectral resolution | Captures spectral signatures in visible and near-infrared range |
| Hyperspectral Cameras | Specim FX17 [9] [5] | NIR (900-1700 nm) | Extends spectral range to short-wave infrared for chemical analysis |
| RGB Imaging Systems | Allied Vision Technologies GT330 [3] | High spatial resolution color imaging | Provides morphological data and visual reference |
| Platform Systems | PlantScreen Modular System [49] | Integrated multi-sensor platform with automation | Enables high-throughput screening with minimal manual intervention |
| Growth Systems | FytoScope FS-WI [49] | Walk-in chamber with controlled environment | Standardizes plant growth conditions for reproducible experiments |
| Analysis Software | LemnaTec Scanalyzer [50] | Image processing and data management | Handles large datasets and extracts phenotypic features |
| Spectral Libraries | Vegetation spectral databases [11] | Reference spectra for healthy/stressed plants | Provides baseline for spectral analysis and classification |
| Calibration Standards | Spectralon reference panels [3] | Known reflectance properties | Ensures radiometric accuracy across imaging sessions |
Data Analysis and Computational Approaches
The integration of RGB and hyperspectral imaging generates complex, high-dimensional datasets that require sophisticated computational approaches for meaningful biological interpretation. Effective analysis pipelines typically incorporate multiple techniques:
Preprocessing and Quality Control Raw spectral data requires substantial preprocessing to remove artifacts and ensure data quality. Standard procedures include radiometric correction to account for sensor irregularities, geometric correction to maintain spatial accuracy, and atmospheric compensation for field applications [48]. Spectral normalization techniques such as Standard Normal Variate (SNV) transformation are particularly effective for reducing lighting variability and enhancing spectral features relevant to biological classification [3].
Feature Extraction and Dimensionality Reduction Hyperspectral data cubes contain hundreds of spectral bands, many of which may be highly correlated. Principal Component Analysis (PCA) is widely used to reduce dimensionality while preserving essential spectral information [50] [3]. For RGB data, feature extraction focuses on morphological descriptors such as leaf area, compactness, and color histograms [49]. Advanced approaches incorporate wavelength selection algorithms to identify minimal spectral feature sets that maintain classification accuracy—critical for developing cost-effective field applications [3].
Machine Learning for Phenotype Classification Both conventional machine learning and deep learning approaches have demonstrated success in plant phenotyping applications. Support Vector Machines (SVM) achieve high accuracy (F1 scores >0.83) for fine-scale growth stage classification when applied to properly transformed spectral data [3]. Random Forest algorithms effectively handle high-dimensional data for trait prediction, while neural networks can model complex nonlinear relationships between spectral features and physiological traits [11] [49]. The integration of temporal data through Logistic Growth Curve analysis further enhances the detection of developmental phenotypes [50].
Computational workflow for imaging data analysis
Applications in Plant Breeding and Genetic Research
Early Stress Detection and Resilience Breeding
The combined RGB-hyperspectral approach enables identification of plant stress responses days or weeks before visible symptoms appear. Hyperspectral imaging detects subtle changes in photosynthetic efficiency, water content, and pigment composition that serve as early indicators of drought stress, nutrient deficiencies, and pathogen infection [48]. For example, water stress alters spectral reflectance in specific SWIR regions associated with water absorption, allowing detection before visible wilting [48]. This early detection capability is invaluable for screening large breeding populations for stress resilience, significantly accelerating development of climate-resilient crop varieties.
Precision Phenotyping for Mutant Screening
Radiation-induced mutagenesis generates valuable genetic diversity for crop improvement, but identifying desirable mutants traditionally requires manual screening of thousands of plants. Integrated imaging systems automate this process through quantitative phenotyping. Studies on Arabidopsis thaliana demonstrate that machine-based screening achieves higher accuracy and efficiency compared to human assessment, with accuracy rates exceeding 80% for identifying phenotypic variants in complex populations [50]. The combination of RGB-derived morphological data and hyperspectral biochemical profiles enables comprehensive characterization of mutant phenotypes, connecting genetic variation to functional traits.
Growth Stage Monitoring and Yield Prediction
Accurate monitoring of developmental transitions is essential for breeding programs, particularly for regulated field trials of genetically modified crops that require precise flowering time predictions. Hyperspectral imaging combined with machine learning classifiers can distinguish subtle growth stages (e.g., Zadoks Z37, Z39, Z41 in wheat) with F1 scores up to 0.832 [3]. Temporal phenomic models incorporating time-series imaging data can predict harvest-related traits such as total biomass dry weight (R² = 0.97) and spike weight (R² = 0.93) with high accuracy early in development [49]. This predictive capability enables breeders to select for yield potential before maturity, significantly reducing breeding cycle times.
Nutritional Quality and Seed Trait Analysis
Hyperspectral imaging extends phenotyping beyond agronomic traits to include quality parameters. The technology can estimate seed composition traits including oil, protein, and carbohydrate content based on spectral signatures [9] [11]. For instance, the spectral feature at 930 nm related to oil content provides a precise signature for sorting almonds from shells [9]. This capability enables breeders to incorporate nutritional quality traits into selection programs, developing crops with enhanced nutritional profiles alongside improved yield and stress tolerance.
Implementation Challenges and Future Perspectives
Despite its transformative potential, implementing integrated RGB-hyperspectral screening presents significant challenges that must be addressed for widespread adoption.
Technical and Computational Barriers The enormous data volumes generated by hyperspectral imaging (often terabytes per experiment) create substantial storage and processing demands [48]. specialized computational infrastructure and expertise are required for efficient data management and analysis. Additionally, sensor costs remain prohibitive for many research programs, particularly in developing regions or for orphan crops [48]. Future developments in miniaturized sensors, edge computing, and cloud-based analysis platforms will help overcome these barriers, making the technology more accessible to diverse research communities.
Standardization and Reproducibility Variability in imaging protocols, environmental conditions, and analysis methods complicates cross-study comparisons and meta-analyses. Establishing standardized trait ontologies, imaging protocols, and data formats is essential for building shared phenotyping resources [47]. Initiatives such as the "Seed Identification Card" model developed by the Rural Development Administration (RDA) in Korea demonstrate the value of standardized phenotypic descriptors for enhancing the utility of genetic resources [47].
Future Directions Emerging applications in orphan crops represent a promising frontier for high-throughput phenotyping [47]. These underutilized species often possess valuable stress tolerance and nutritional traits but have received limited breeding attention due to resource constraints. Affordable imaging technologies could accelerate their genetic improvement, enhancing agricultural biodiversity and resilience. Additionally, the integration of artificial intelligence and machine learning is evolving from descriptive phenotyping to prescriptive breeding, enabling prediction of plant performance under specific environmental scenarios and accelerating development of tailored crop varieties [47].
The integration of RGB and hyperspectral imaging technologies represents a paradigm shift in high-throughput screening for plant breeding and genetic research. This synergistic approach combines detailed morphological information with rich biochemical data, providing unprecedented insights into plant function and performance. The experimental protocols and analytical frameworks presented in this guide provide researchers with practical tools for implementing these technologies in diverse research programs. As these methods become more accessible and standardized, they will play an increasingly vital role in accelerating crop improvement and addressing global food security challenges in a changing climate.
Navigating Technical Challenges and Optimizing Your Imaging Workflow
Addressing the High Dimensionality and Data Volume of Hyperspectral Imagery
Hyperspectral imaging (HSI) is a powerful analytical technique that captures spatial and spectral information across hundreds of contiguous, narrow wavelength bands, generating a detailed spectral signature for each pixel in an image [51]. This detailed data enables precise identification and characterization of materials, including the subtle biochemical properties of plants [2]. However, the richness of this information comes with a significant challenge: the extreme dimensionality and vast data volume of hyperspectral datacubes [52]. These characteristics pose substantial obstacles for data storage, transmission, and computational processing, especially in time-sensitive agricultural and plant research applications [1].
The core of this challenge lies in the data structure itself. A single hyperspectral image is a three-dimensional datacube, combining two spatial dimensions (x, y) with one spectral dimension (λ) [51]. While multispectral imaging might capture 3-10 broad bands, hyperspectral systems can capture 50 to over 250 narrow bands, leading to data sizes that are orders of magnitude larger than conventional RGB imagery [2]. Furthermore, adjacent bands in a hyperspectral datacube are often highly correlated, resulting in substantial information redundancy [52]. For research focusing on plants, this high-dimensional data space is both a blessing and a curse. It allows for the detection of early-stage stress, nutrient deficiencies, and diseases before they become visible to the naked eye [2] [1], but it also necessitates sophisticated data reduction techniques to make analysis feasible and efficient.
Framed within the context of combining RGB and hyperspectral imaging for plant research, addressing the data volume challenge becomes imperative. RGB imaging provides high-spatial-resolution structural information at a low computational cost, while HSI delivers unparalleled spectral resolution for biochemical analysis. The synergy of these modalities creates a comprehensive picture of plant health [3]. However, to harness this synergy effectively, researchers must employ strategies to reduce the data burden of HSI without losing critical spectral information. This guide provides an in-depth technical overview of the methods and protocols that enable researchers to overcome the data challenges of hyperspectral imagery, thereby unlocking the full potential of combined imaging approaches in plant science.
Core Challenges in Hyperspectral Data Management
The management and analysis of hyperspectral data are primarily hindered by two interconnected issues: the "curse of dimensionality" and the practical burdens of massive data volume. In high-dimensional spaces, data becomes sparse, and conventional statistical and machine learning algorithms require exponentially more samples to maintain accuracy, a phenomenon known as the Hughes effect [51]. This sparsity can degrade the performance of classification models when applied to the original, high-dimensional spectral space.
The sheer data volume also imposes immediate practical constraints. Storing raw hyperspectral datacubes from field or laboratory studies demands significant digital capacity. Transferring these large files from field collection sites to computational resources for analysis can be slow, creating bottlenecks in research workflows [1]. Furthermore, processing hundreds of spectral bands for each pixel is computationally intensive, often requiring specialized hardware and preventing real-time or near-real-time analysis, which is crucial for applications like precision agriculture and dynamic phenotyping [52] [3].
The table below summarizes the key differences between RGB, Multispectral, and Hyperspectral imaging, highlighting the source of the data challenge.
Table 1: Comparison of RGB, Multispectral, and Hyperspectral Imaging Modalities
| Feature | RGB Imaging | Multispectral Imaging | Hyperspectral Imaging |
|---|---|---|---|
| Spectral Bands | 3 broad bands (Red, Green, Blue) [51] | 3-10 discrete, broad bands [2] | 50-250+ narrow, contiguous bands [2] [51] |
| Spectral Resolution | Low | Medium | Very High (e.g., 5.5 nm FWHM [3]) |
| Data Volume per Image | Low | Moderate | Very High |
| Primary Information | Spatial structure, color [1] | Selected spectral indices (e.g., NDVI) [2] | Full spectral signature for biochemical analysis [2] [51] |
| Key Strength in Plant Research | Morphological assessment, plant counting [1] | Cost-effective health monitoring | Early stress detection, detailed biochemical composition [1] |
Technical Approaches to Dimensionality and Data Reduction
A suite of technical strategies has been developed to mitigate the challenges of hyperspectral data. These methods can be broadly categorized into dimensionality reduction techniques, which reduce the number of spectral variables, and data volume compression techniques, which minimize the physical storage size of the datacube.
Dimensionality Reduction Techniques
Dimensionality reduction (DR) is a critical pre-processing step in HSI analysis aimed at projecting the high-dimensional data into a more manageable, lower-dimensional space while preserving its essential information [53]. These techniques are generally divided into two classes: feature extraction and band selection.
Feature Extraction
Feature extraction methods transform the original high-dimensional data into a new set of reduced features. These new features are a linear or non-linear combination of the original spectral bands.
- Principal Component Analysis (PCA): A classic linear technique that projects the data onto a new set of orthogonal axes (principal components) ordered by the amount of variance they explain from the original data. This allows a small number of components to capture most of the data's information [52] [53].
- Linear Discriminant Analysis (LDA): A supervised method that seeks a projection that maximizes the separability between predefined classes [51] [53].
- Manifold Learning: Non-linear methods like Locality Preserving Projections (LPP) attempt to uncover the intrinsic low-dimensional structure (manifold) embedded in the high-dimensional spectral space [53].
- Tensor-Based Methods: Advanced techniques, such as Spatial-Spectral Neighborhood Tensor Graph Embedding (SNTGE), treat the HSI as a third-order tensor (row × column × band) to directly leverage its inherent spatial-spectral structure, often leading to improved feature extraction for classification [53].
Band Selection
Band selection techniques aim to identify and retain a subset of the most informative original spectral bands, thereby eliminating redundancy while maintaining the physical interpretability of the data.
- Standard Deviation (STD)-based Selection: This simple yet powerful unsupervised method selects bands with the highest standard deviation across the dataset. The underlying assumption is that a high variance indicates rich information content. A 2025 study demonstrated that STD-based selection could reduce data size by up to 97.3% while maintaining a classification accuracy of 97.21% on biomedical tissue samples, outperforming more complex methods in stability and efficiency [52].
- Information-Theoretic Methods: Approaches based on Mutual Information (MI) and Shannon entropy select bands that are most relevant for class discrimination and contain the highest information content [52] [54]. A hybrid method combining MI with a Minimum Noise Fraction transform has been shown to achieve classification accuracies over 99% on some datasets [52].
- Clustering-Based Selection: Algorithms like Data Gravitation and Weak Correlation Ranking (DGWCR) group highly correlated spectral bands into clusters and then select a representative band from each cluster, effectively reducing redundancy [52].
Table 2: Comparison of Representative Dimensionality Reduction Techniques
| Method | Type | Supervision | Key Principle | Advantages | Limitations |
|---|---|---|---|---|---|
| PCA [52] [53] | Feature Extraction | Unsupervised | Maximizes variance in projected data | Simple, fast, effective for redundancy removal | Assumes linear relationships, may lose discriminative info |
| LDA [51] [53] | Feature Extraction | Supervised | Maximizes inter-class separation | Enhances class separability | Requires labeled data, can overfit |
| STD-based [52] | Band Selection | Unsupervised | Selects highest variance bands | Simple, fast, preserves original bands, highly efficient | May select noisy bands |
| MI-based [52] | Band Selection | Supervised | Selects most class-relevant bands | High classification performance | Complex, requires labels, computationally heavy |
| SNTGE [53] | Feature Extraction | Unsupervised | Tensor graph preserving spatial-spectral structure | Leverages spatial context, high classification accuracy | Complex implementation and computation |
Data Volume and Compression Strategies
Beyond dimensionality reduction, other strategies address the sheer volume of HSI data.
- Sensor Miniaturization and Onboard Processing: The development of compact, robust hyperspectral cameras, such as snapshot sensors, enables data collection closer to the target (e.g., handheld or on ground vehicles), which can reduce the spatial scale and therefore the data size of a single scan [1] [13]. Furthermore, cameras like the Specim IQ come with built-in processing capabilities, allowing for initial analysis and compression directly on the device [13].
- Event-Based Sensing: An emerging approach that moves beyond traditional frame-based acquisition. Instead of capturing full frames at a fixed rate, event-based sensors record changes in per-pixel intensity asynchronously, potentially leading to significant data reduction for dynamic scenes [55].
- Cloud and AI-Driven Analytics: The vast volume of HSI data is increasingly managed through cloud-based platforms that leverage artificial intelligence (AI) and machine learning for near-real-time interpretation. This outsources the computational burden to powerful remote servers [2].
Experimental Protocols for Plant Research
The integration of RGB and hyperspectral imaging, coupled with robust dimensionality reduction, provides a powerful toolkit for advanced plant phenotyping. The following protocol, inspired by a 2025 study on wheat growth stage classification, offers a detailed methodology for a typical plant research application [3].
Case Study: Classification of Individual Wheat Growth Stages
Objective: To automatically classify individual wheat plants into three closely spaced pre-anthesis growth stages (Zadoks Z37, Z39, Z41) using hyperspectral and RGB data to reduce reliance on manual, labor-intensive visual inspections [3].
Experimental Workflow:
The following diagram illustrates the end-to-end workflow for this experiment, from plant preparation to model validation.
Plant Growth and Preparation
- Cultivar: Use a commercially relevant wheat cultivar like 'Scepter'.
- Growing Conditions:
- Controlled Environment (Greenhouse): Grow plants in pots with a standardized soil mixture (e.g., 1:1:1 sand, clay, UC soil mix). Maintain controlled temperature (e.g., 18°C day/13°C night) and irrigation schedules [3].
- Semi-Natural Environment: Grow plants in larger outdoor trays with the same soil mixture, relying primarily on rainfall with supplemental watering. This mimics field conditions and tests model generalizability [3].
- Experimental Design: Employ staggered planting dates to capture phenotypic variability across different environmental conditions [3].
Data Acquisition
- Hyperspectral Imaging:
- Sensor: Use a push-broom hyperspectral camera like the Specim FX10, which covers the visible and near-infrared (VNIR: 400–1000 nm) range with a high spectral resolution (e.g., 5.5 nm FWHM) [3].
- Setup: In a controlled environment, use an automated system (e.g., a LemnaTec 3D Scanalyzer) inside an enclosed cabinet with halogen lighting for uniformity. Position the camera top-down, approximately 1.4 m above the plants. For semi-natural environments, use a tripod or similar stable platform [3].
- Calibration: Capture white and dark reference images immediately before or after each session for radiometric correction [3].
- RGB Imaging: Capture high-resolution color images of the same plants in the controlled environment using standardized digital cameras (e.g., Allied Vision Technologies GT330) [3].
Data Preprocessing
- Hyperspectral Data Calibration: Convert raw digital numbers to reflectance values using the white and dark reference images [3].
- Spectral Transformation: Apply transformations to the calibrated spectral data to enhance features and reduce scattering effects. Common methods include:
- Standard Normal Variate (SNV): Normalizes each individual spectrum to have a mean of zero and a standard deviation of one [3].
- Hyper-hue: A transformation that can improve spectral distinctions [3].
- Principal Component Analysis (PCA): Used here as a transformation for feature enhancement rather than just reduction [3].
Feature Engineering and Dimensionality Reduction
- Feature Selection: Systematically compare the performance of the three spectral transformations (SNV, Hyper-hue, PCA). The SNV transformation has been shown to be particularly robust, especially when training data is limited [3].
- Dimensionality Reduction: Perform band selection to identify the most informative wavelengths for the classification task. The study demonstrated that after feature selection, a high classification accuracy (F1 score of 0.752) could be maintained with only five key wavelengths, drastically reducing data dimensionality and computational load [3].
Model Training and Classification
- Algorithm: Utilize a Support Vector Machine (SVM) classifier. SVMs are well-suited for high-dimensional data and often provide strong performance with limited samples [3].
- Training: Train the SVM model on the transformed and reduced hyperspectral features, with growth stages (Z37, Z39, Z41) as the target labels.
- Performance Evaluation: Evaluate the model using metrics like F1 score, which balances precision and recall. The combined use of multiple spectral transformations has been shown to achieve high F1 scores (e.g., 0.832), outperforming reliance on any single transformation or RGB data alone [3].
The Researcher's Toolkit
Table 3: Essential Research Reagents and Equipment for Hyperspectral Plant Phenotyping
| Item | Function/Description | Example from Literature |
|---|---|---|
| Hyperspectral Camera (VNIR) | Captures spectral data in the visible and near-infrared range (400-1000 nm), key for plant pigment and water content analysis. | Specim FX10 camera [3] |
| RGB Camera | Provides high-resolution spatial and color information for morphological assessment and coregistration with HSI data. | Allied Vision Technologies GT330 [3] |
| Automated Gantry System | Enables precise, high-throughput imaging of plants in controlled conditions by moving the sensor in a predefined pattern. | LemnaTec 3D Scanalyzer [3] |
| Standardized Reference Targets | Used for radiometric calibration; white reference for correction, dark reference for sensor noise. | Halon white reference & dark current image [3] |
| Controlled Growth Chambers | Provides a stable environment for plant growth, ensuring consistent conditions for phenotypic expression. | Greenhouse with controlled temp/light [3] |
| Data Processing Software | Platform for applying calibration, dimensionality reduction, transformations, and machine learning models. | Python with scikit-learn, custom scripts [52] [3] |
The high dimensionality and voluminous nature of hyperspectral imagery present significant but surmountable challenges. As detailed in this guide, a combination of strategic approaches—including sophisticated dimensionality reduction techniques like STD-based band selection and spatial-spectral tensor methods, alongside advancements in sensor technology and data processing architectures—effectively mitigates these issues. When framed within the context of plant research, the synergy between RGB and hyperspectral imaging is clear: RGB provides the high-fidelity spatial context, while HSI, once distilled through these reduction techniques, delivers the deep biochemical insights. The experimental protocol for wheat growth stage classification demonstrates that it is possible to reduce data size by over 97% without sacrificing critical classification accuracy, enabling scalable, automated phenotyping. By adopting these methodologies, researchers and drug development professionals can leverage the full, non-destructive power of hyperspectral imaging to advance plant science, improve crop yields, and strengthen biosafety in an efficient and computationally feasible manner.
Overcoming the Curse of Dimensionality and Limited Labeled Training Samples
In plant sciences, non-invasive imaging technologies are fundamental for unlocking phenotypic secrets. While traditional RGB imaging captures morphological structure, hyperspectral imaging (HSI) generates a detailed spectral signature for each pixel, creating a three-dimensional hypercube (x, y, λ) that encodes rich physico-chemical information [11]. This high spectral resolution enables the detection of subtle physiological changes related to plant water status, structural compounds, and stress responses long before they become visibly apparent [1] [56].
However, this powerful capability comes with a significant computational challenge: the Curse of Dimensionality. A single hyperspectral data cube can contain hundreds of contiguous narrow bands, creating a high-dimensional feature space where the distance between data points becomes less meaningful and the risk of model overfitting intensifies [57]. This problem is compounded by the Limited Availability of Labeled Training Samples, as manually annotating hyperspectral data is costly, labor-intensive, and requires specialized expertise [57] [58]. This combination creates a critical bottleneck for applying deep learning to HSI analysis.
Framed within the broader benefits of combining RGB and HSI for plant research, this whitepaper explores technical solutions to these challenges. The fusion of RGB's high spatial clarity with HSI's rich spectral data, coupled with advanced machine learning strategies, paves the way for more robust and accessible plant phenotyping platforms.
Technical Challenges in Hyperspectral Data Analysis
The Curse of Dimensionality: A Spectral Perspective
The curse of dimensionality in HSI manifests through several specific problems:
- Data Sparsity: The volume of feature space grows exponentially with dimensionality, causing the available training samples to become sparse, making it difficult to learn reliable model parameters [57].
- Increased Computational Cost: Processing and storing hundreds of spectral bands per pixel demands significant computational resources and memory, complicating real-time analysis [58].
- Hughes Phenomenon: For a fixed number of training samples, the predictive power of a classifier can decrease as the dimensionality increases beyond a certain point [57].
The Label Scarcity Problem in Plant Phenotyping
The acquisition of labeled data for HSI in plant research is particularly challenging. Annotations often require destructive harvesting for ground-truth validation (e.g., measuring root dry mass or leaf nutrient content) [19], or depend on expert knowledge for stressor identification (e.g., distinguishing root pathogen from herbivore damage) [56]. This limits the scale of datasets available for supervised learning.
Technical Solutions and Methodologies
Dimensionality Reduction and Feature Selection
A direct approach to mitigating dimensionality is to reduce the feature space while preserving critical information.
Table 1: Dimensionality Reduction Techniques for Hyperspectral Plant Data
| Technique Category | Specific Method | Application in Plant Phenotyping | Key Benefit |
|---|---|---|---|
| Machine Learning-based Band Selection | Recursive Feature Elimination (RFE) [59] | Optimized selection of NIR, SWIR1, and SWIR2 bands for early stress detection. | Data-driven; creates targeted, interpretable vegetation indices (e.g., MLVI, H_VSI). |
| Spectral Index Development | Novel Hyperspectral Indices (e.g., H_VSI) [59] | Detects water and structural stress 10-15 days earlier than NDVI. | Reduces hundreds of bands to a single, highly informative value. |
| Deep Learning-based Feature Extraction | Lightweight 1D-CNNs [58] | Onboard satellite processing for real-time classification of land cover and crop stress. | Automatically extracts relevant nonlinear features without manual preprocessing. |
Leveraging Unlabeled Data with Self-Supervised Learning
Self-supervised learning (SSL) has emerged as a powerful paradigm for learning meaningful representations from unlabeled data, thereby overcoming the labeled data bottleneck.
Contrastive Learning (CL) is a particularly effective SSL approach. Its core principle is to learn representations by pulling "positive" samples (different augmented views of the same data instance) closer in the feature space while pushing "negative" samples (views from different instances) apart [57]. The typical workflow for applying contrastive learning to hyperspectral plant data involves:
- Data Augmentation: Creating multiple augmented views of each unlabeled hyperspectral patch or pixel spectrum. Transformations can include spectral jittering, random masking of bands, and spatial cropping.
- Representation Learning: Training an encoder network using a contrastive loss function (e.g., NT-Xent) on the augmented unlabeled data. This step teaches the model to be invariant to irrelevant noise and to focus on intrinsic spectral-spatial features.
- Fine-tuning: The pre-trained encoder is then fine-tuned on a downstream task (e.g., stress classification) using the limited set of available labeled samples. This process yields significantly better performance than training a model from scratch on the small labeled dataset [57].
Empirical results have demonstrated that classifiers fine-tuned with a contrastive learning-based encoder maintain competitive performance even when the amount of labeled training data is reduced by 50% [57].
Fusion of RGB and Hyperspectral Imaging
Combining the strengths of RGB and hyperspectral imaging presents a practical strategy to alleviate both dimensionality and data scarcity issues.
- Spectral Super-Resolution (SSR): This technique reconstructs a high-spectral-resolution HSI from a low-cost, readily available RGB image [46]. Advanced models like MSS-Mamba use a multi-scale spectral–spatial sequence learning approach to infer the detailed spectral signature from the three RGB channels, effectively generating synthetic yet high-fidelity hyperspectral data [46].
- Spatial-Spectral Fusion: Conversely, hyperspectral images can be fused with high-spatial-resolution multispectral (MS) or RGB images to enhance spatial detail. Methods like the Retractable Spatial–Spectral Transformer (RSST) integrate the fine spectral resolution of HSI with the sharp spatial details of MSI/RGB, generating a unified image with high resolution in both domains [60].
The following diagram illustrates a technical workflow that integrates RGB and hyperspectral data, leveraging self-supervised learning to overcome data limitations.
Experimental Protocols for Plant Root Phenotyping
This section provides a detailed methodology for a key application: hyperspectral root phenotyping in soil-filled rhizoboxes, which effectively demonstrates the fusion of RGB and HSI to overcome segmentation and labeling challenges [61] [19].
Rhizobox Setup and Plant Growth
- Rhizobox Design: Construct rhizoboxes (e.g., 30 cm wide × 100 cm high × 1 cm deep) with a grey PVC back-plate and side frames. The front should consist of a mineral glass window fixed by metal angle bars to allow for root observation [61] [19].
- Soil Substrate: Fill rhizoboxes with natural soil (e.g., sieved to < 2 mm). Using substrates with different colors (e.g., dark topsoil vs. bright subsoil) is useful for comparing segmentation performance. Adjust soil moisture to a target matrix potential (e.g., -100 cm for well-watered, -1000 cm for water-limited) [61].
- Planting and Growth: Plant pre-soaked seeds (e.g., Triticum durum) directly into the soil. Position the boxes at a 45° angle in a growth chamber to encourage root growth along the glass surface. Darken the glass surface between imaging sessions to minimize light exposure and algae growth [61].
Image Acquisition Workflow
The imaging protocol involves sequential data capture using both RGB and hyperspectral systems.
A. RGB Imaging Protocol [19]:
- Setup: Use a custom-made or commercial RGB camera system with uniform, cross-polarized lighting to minimize glare from the glass surface.
- Acquisition: Capture high-resolution images of the rhizobox at regular intervals (e.g., twice weekly). Ensure consistent camera position and lighting conditions across all sessions.
- Analysis: Use automated or semi-automated root segmentation software (e.g., based on color thresholding and machine learning) to quantify root system architecture traits like total root length, depth, and branching density.
B. Hyperspectral Imaging Protocol [61]:
- Setup: Utilize a push-broom hyperspectral imaging system (e.g., ImSpector N17E spectrograph with a NIR camera) covering the 1000–1700 nm spectral range with 256 bands. The system should be mounted on a motorized positioning stage for precise movement.
- Calibration: Before scanning each rhizobox, acquire images of a white reference (Spectralon tile) and a dark reference (with the lens covered) under the same integration time to correct for sensor dark current and uneven illumination.
- Acquisition: Move the imaging system vertically along the rhizobox in 3 cm strides with 10% overlap, continuously capturing line scans. The spatial resolution should be high (e.g., 0.1 mm). Adjust camera integration time to utilize ~85% of the dynamic range without saturation.
- Data Construction: Compose the full rhizobox image by stitching the individual strides during post-processing.
Data Processing and Analysis Pipeline
- Hyperspectral Data Preprocessing: Apply white and dark reference correction to convert raw digital numbers to reflectance. Further preprocess images with log-linearization and asymptotic least squares correction to enhance spectral features [61].
- Root Segmentation:
- RGB-based: Rely on color (RGB) thresholding and edge detection algorithms.
- Spectral-based: Apply automated segmentation algorithms like fuzzy clustering or multilevel thresholding on the preprocessed hyperspectral data cube. This method often provides superior performance when root-soil color contrast is poor [61].
- Spectral Signature Analysis: Extract mean reflectance spectra from different root regions (e.g., center vs. border, main axes vs. laterals). Analyze differences in absorption features related to water (e.g., ~1200 nm, ~1450 nm) and structural compounds (e.g., lignin, cellulose) to infer functional properties [61].
- Chemometric Mapping: Use advanced statistical models (chemometrics) to translate spectral data into quantitative maps of root zone properties, such as water content, by analyzing the difference spectra between key absorption regions [61] [19].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Materials and Equipment for Fused RGB-Hyperspectral Root Phenotyping
| Item Name | Function/Application | Technical Notes |
|---|---|---|
| Soil-Filled Rhizobox | Provides a near-natural growth environment for 2D root system observation. | Inner space typically 1-3 cm; front glass must be transparent in both visible and NIR ranges [61] [19]. |
| Snapshot Hyperspectral Camera | Captures full hyperspectral data cube instantaneously without scanning. | Ideal for dynamic or field-based studies; reduces motion artifacts [1]. |
| Push-Broom HSI System | High-resolution spectral imaging for detailed lab-based root analysis. | Requires precise movement of camera or sample; delivers high spatial/spectral fidelity [61]. |
| Spectralon White Reference | Calibration standard for converting raw sensor data to reflectance. | Critical for accurate and reproducible spectral measurements across time [61]. |
| Controlled Irrigation System | Maintains precise soil moisture levels for abiotic stress studies. | Can be automated with soil moisture sensors for consistent treatment application [56]. |
| Machine Learning-Optimized Vegetation Indices (e.g., MLVI, H_VSI) | Data-reduced spectral indices for early stress detection. | Developed using RFE; more sensitive than traditional indices like NDVI [59]. |
The curse of dimensionality and the scarcity of labeled samples are significant but surmountable hurdles in plant hyperspectral imaging. As detailed in this guide, a combination of strategic approaches—including machine learning-based feature selection, self-supervised learning to leverage unlabeled data, and the synergistic fusion of RGB and hyperspectral data—provides a powerful framework to overcome these challenges. The experimental protocol for root phenotyping serves as a concrete example of how these principles are applied in practice, enabling researchers to extract robust, functional insights from complex spectral data. By adopting these advanced methodologies, plant scientists can accelerate the development of high-throughput, precision phenotyping platforms that enhance our understanding of plant health and stress responses.
Computational spectral reconstruction represents a paradigm shift in hyperspectral imaging, enabling the derivation of detailed spectral information from conventional RGB images. This technical guide elucidates the core principles, methodologies, and applications of spectral reconstruction techniques, with a specific focus on plant science research. By transforming standard color images into rich hyperspectral data cubes, these computational methods facilitate advanced analysis of plant health, physiology, and biochemistry without the prohibitive costs of specialized hardware. We provide an in-depth examination of reconstruction algorithms, performance metrics, and experimental protocols, underscoring the transformative potential of combining ubiquitous RGB imaging with hyperspectral analytical capabilities for agricultural innovation and drug development from plant sources.
Hyperspectral imaging (HSI) captures light intensity across hundreds of narrow, contiguous wavelength bands, generating a continuous spectral signature for each pixel in an image [9]. This detailed spectral data enables the identification of materials based on their chemical composition, far beyond the capabilities of standard RGB cameras which are limited to three broad spectral channels (red, green, and blue) [9]. In plant research, these spectral fingerprints can reveal critical information about plant physiology, biochemistry, and health status, often before visible symptoms manifest [2].
However, the adoption of hyperspectral imaging has been constrained by significant limitations. Traditional hyperspectral imagers are typically expensive, complex systems requiring precise optical components and scanning mechanisms, making them inaccessible for many research applications [62]. Furthermore, the sequential scanning process of many systems renders them unsuitable for capturing dynamic phenomena in real-time [63].
Computational spectral reconstruction addresses these limitations by mathematically inferring hyperspectral information from widely available RGB images [62]. This approach leverages the fact that RGB images are essentially projections of the full spectral information onto three color channels, and with appropriate algorithms, significant portions of the original spectral data can be recovered [64]. For plant researchers, this technology promises to democratize access to spectral analysis by utilizing existing RGB imaging systems, from laboratory cameras to UAV-based and even smartphone-based sensors.
Fundamentals of Spectral Reconstruction
The Image Formation Model
The mathematical foundation for spectral reconstruction begins with the image formation model that describes how hyperspectral data is compressed into an RGB image. According to the Lambertian assumption, the relationship between hyperspectral images (HSIs) and RGB images can be expressed as:
[ Ic \left( x,y \right) = \int{w1}^{w2} R\left( x,y,w \right) L \left( w \right) S_c \left( w \right) d w ]
Where:
- ( I_c ) represents each channel of an RGB image (where c = R, G, B)
- ( S_c\left( w \right) ) denotes the spectral response function of the camera
- ( R\left( x,y,w \right) ) is the spectral reflectance of the objects
- ( L \left( w \right) ) is the illumination spectrum
- ( w1 ) and ( w2 ) define the spectral range [62]
When the illumination spectrum is known or calibrated, the hyperspectral image ( H \left( x, y, w\right) ) can be defined as the product of the scene's spectral reflectance and the illumination spectrum, simplifying the equation to:
[ Ic \left( x,y \right) = \int{w1}^{w2} H \left( x, y, w \right) S_c \left( w \right) d w ]
The challenge of spectral reconstruction is to invert this forward model—recovering the multi-channel datacube ( H\left( x, y, w \right) ) from the captured 3-channel RGB image ( I_c ) [62]. This constitutes an ill-posed inverse problem, as infinitely many possible spectral configurations could produce the same RGB values, necessitating the use of priors or learning-based approaches to constrain the solution space.
Key Datasets for Development and Benchmarking
The advancement of spectral reconstruction methods relies on standardized hyperspectral datasets for training and validation. The table below summarizes five principal open-source datasets used in the computational spectral imaging community.
Table 1: Key Hyperspectral Datasets for Spectral Reconstruction Research
| Dataset Name | Data Volume | Spatial Resolution | Spectral Channels | Spectral Range & Interval | Key Characteristics |
|---|---|---|---|---|---|
| CAVE [62] | 32 images | 512 × 512 pixels | 31 bands | 400-700 nm, 10 nm interval | Captured with tunable filter; various indoor objects under controlled illumination |
| ICVL [62] | 203 images | 1392 × 1300 pixels | 31 bands | 400-700 nm, 10 nm interval | Diverse indoor/outdoor scenes; collected with line-scanning camera |
| BGU-HS [62] | 286 images | 1392 × 1300 pixels | 31 bands | 400-700 nm, 10 nm interval | Largest natural HSI dataset; expanded for NTIRE-2018 challenge |
| ARAD-HS [62] | 510 images | 512 × 482 pixels | 31 bands | 400-700 nm, 10 nm interval | Collected with portable Specim-IQ camera; diverse scenes for NTIRE-2020 |
| KAUST-HS [62] | 409 images | 512 × 512 pixels | 31 bands | 400-730 nm, 10 nm interval | Reflectance HSIs calibrated with white board; diverse indoor/outdoor scenes |
These datasets have been instrumental in driving progress through community challenges such as NTIRE-2018 and NTIRE-2020, which have established standardized benchmarks for comparing reconstruction algorithms [62].
Performance Evaluation Metrics
The quality of reconstructed hyperspectral images is typically assessed using spatial and spectral accuracy metrics that compare reconstructions with ground truth measurements. The three predominant metrics are:
Mean Relative Absolute Error (MRAE): [ \text{MRAE} = \frac{1}{N}\sum{i=1}^{N}{\left( \frac{\left| H{GT}^{i}-H{SR}^{i} \right|}{H{GT}^{i}} \right)} ] This metric calculates the average relative absolute error across all pixels, providing a normalized measure of reconstruction accuracy [62].
Root Mean Square Error (RMSE): [ \text{RMSE} = \sqrt{ \frac{1}{N}\sum{i=1}^{N} \left( H{GT}^{i}-H_{SR}^{i} \right)^2 } ] RMSE quantifies the absolute difference between reconstructed and ground truth values, with higher penalties for larger errors [62].
Spectral Angle Mapper (SAM): SAM measures the spectral similarity by computing the angle between reconstructed and ground truth spectral vectors, thereby evaluating the preservation of spectral shape regardless of overall brightness [62].
Computational Reconstruction Methods
Taxonomy of Reconstruction Approaches
Spectral reconstruction methods can be broadly categorized into three main paradigms: traditional model-based approaches, modern deep learning-based methods, and hybrid frameworks that combine elements of both.
Table 2: Computational Spectral Reconstruction Methodologies
| Method Category | Core Principle | Representative Techniques | Advantages | Limitations |
|---|---|---|---|---|
| Prior-Based/Model-Based Methods [62] [63] | Explores statistical priors in HSIs (sparsity, spatial similarity, spectral correlation) to constrain solution space | Sparsity-based regularization [62], Low-rank constraints [63], Total variation regularization [63] | Theoretical interpretability; robustness in data-scarce scenarios [62] | High computational cost; limited adaptability to complex real-world conditions [63] |
| Data-Driven/Deep Learning Methods [62] [63] | Learns mapping from RGB to hyperspectral domains using large datasets without handmade priors | CNN architectures [62], Generative Adversarial Networks (GANs) [62], Transformer-based models (MST++) [64] | State-of-the-art accuracy; automated feature extraction; fast inference [62] [64] | Black-box nature; limited generalizability; substantial training data requirements [63] [64] |
| Hybrid Methods [63] | Integrates iterative optimization schemes with trainable neural network components | Deep unrolling pipelines (DUN) [63], Physics-guided neural networks | Balances interpretability with performance; incorporates physical constraints [63] | Implementation complexity; potential trade-offs between components |
Specialized Reconstruction Architectures
Recent advances have introduced specialized neural architectures designed to address specific challenges in spectral reconstruction:
Spatial-Spectral Cross-Attention-Driven Network (SSCA-DN): This novel approach employs a multi-scale feature aggregation module and spectral-wise transformer to simultaneously model long-range spectral dependencies and spatial details [65]. By using spatial and spectral attention mechanisms to interactively guide reconstruction, SSCA-DN effectively captures spatial-spectral cross-correlations while considering multi-scale features [65].
Transformer-Based Architectures: Models like MST++, which won the NTIRE 2022 challenge, leverage self-attention mechanisms to capture global dependencies in spectral data, demonstrating improved performance over earlier CNN-based approaches [64].
Unfolding Networks: These methods mathematically "unfold" traditional iterative optimization algorithms into deep network layers, combining the interpretability of model-based approaches with the adaptive learning capabilities of deep neural networks [63].
Applications in Plant Research
Plant Science Use Cases
The integration of computational spectral reconstruction with plant research addresses critical challenges in agricultural science and plant-based drug development:
Crop Health Monitoring & Early Disease Detection: Reconstructed hyperspectral data enables identification of fungal, viral, or bacterial infections by revealing biochemical and physiological changes invisible to conventional sensors, allowing intervention before yield compromise [2].
Nutrient and Water Stress Management: Each nutrient deficiency produces a unique spectral signature detectable through reconstructed hyperspectral imagery, guiding variable rate fertilizer applications and precision irrigation [2].
Herbicide Efficacy Assessment: Research demonstrates that hyperspectral sensing combined with machine learning can quantify herbicide-induced stress in plants with precision approaching trained weed scientists, potentially automating efficacy evaluation [66].
High-Throughput Phenotyping: Automated systems integrating hyperspectral imaging with robotic arms enable large-scale plant analysis, capturing physiological changes in response to environmental stresses and accelerating breeding programs [67].
Yield Prediction and Supply Chain Optimization: By aggregating rich hyperspectral data across entire fields, AI models can predict yields with unprecedented accuracy, enhancing harvest planning and supporting traceability in food supply chains [2].
Experimental Protocol: Hyperspectral Analysis of Plant Leaves
For researchers implementing spectral reconstruction for plant analysis, the following protocol provides a methodological framework:
Image Acquisition Setup:
- Configure controlled lighting conditions to minimize shadows and specular reflections
- Position RGB camera perpendicular to plant samples to minimize spatial distortion
- Include color reference charts in frame for potential post-processing calibration
- Maintain consistent camera settings (ISO, aperture, shutter speed) across all captures
Background Masking and Leaf Region Extraction:
- Apply segmentation algorithms (e.g., threshold-based, U-Net) to isolate leaves from background
- Generate binary masks to identify regions of interest
- Remove non-leaf elements (soil, pots, markers) from analysis
Spectral Reconstruction:
- Preprocess RGB images (flat-field correction, noise reduction)
- Apply trained reconstruction model (e.g., CNN, transformer-based) to generate hyperspectral data cubes
- Validate reconstruction quality using reference measurements if available
Spectral Component Analysis:
- Identify key spectral components related to biochemical features of interest
- Project hyperspectral cubes onto these components to highlight specific spectral traits
- Analyze spatial distribution of spectral features across leaf surfaces [6]
Data Interpretation and Model Integration:
- Apply vegetation indices (e.g., NDVI, PRI) to reconstructed spectra
- Utilize machine learning classifiers (e.g., random forest) for stress detection or trait quantification [66]
- Correlate spectral features with physiological measurements for validation
Research Toolkit: Essential Materials and Solutions
Table 3: Research Reagent Solutions for Plant Spectral Imaging Experiments
| Item | Function/Benefit | Application Context |
|---|---|---|
| Standard Reference Charts | Provides color calibration for reconstruction algorithms; ensures measurement consistency across sessions | Essential for quantitative studies; enables cross-study comparisons |
| Portable Hyperspectral Validation Sensors (e.g., Spectroradiometer) [66] | Ground-truth measurement for validating reconstructed spectra; assesses algorithm accuracy | Critical for method development and validation; field measurements |
| Controlled Illumination Systems | Standardizes lighting conditions; minimizes shadows and specular reflections | Laboratory imaging setups; phenotyping platforms [67] |
| Leaf Clips and Positioning Fixtures | Maintains consistent leaf orientation and distance from camera; reduces motion artifacts | High-precision leaf-level measurements; time-series studies |
| Data Processing Software (e.g., LemnaGrid [67]) | Enables image analysis without traditional coding; streamlines processing pipelines | Accessible for domain experts without programming background |
| Random Forest Machine Learning Algorithms [66] | Analyzes vegetation indices; classifies plant stress responses | Herbicide efficacy studies; stress detection models |
Workflow Visualization
Future Outlook and Challenges
The field of computational spectral reconstruction faces several significant challenges that represent opportunities for future research:
Generalization Across Conditions: Current models often struggle with variations in illumination, camera sensors, and environmental conditions, limiting their real-world applicability [64]. Developing domain adaptation techniques and invariant representations remains an active research area.
Spectral Accuracy vs. Computational Efficiency: Trade-offs exist between reconstruction accuracy and processing speed, particularly for real-time applications [63]. Optimized network architectures and efficient inference algorithms are needed for field deployment.
Standardization and Validation: The lack of standardized protocols for evaluating reconstructed spectral data hinders comparative assessment across studies [64]. Community-wide benchmarks and validation methodologies would accelerate progress.
Physical Consistency: While data-driven approaches achieve impressive metric scores, questions remain about their ability to accurately reproduce physically plausible spectra rather than just statistically likely ones [64].
The global hyperspectral imaging market in agriculture is projected to exceed $400 million by 2025, with over 60% of precision agriculture systems expected to utilize hyperspectral imaging for crop monitoring [2]. Computational spectral reconstruction will play a pivotal role in achieving these projections by making hyperspectral analysis more accessible and cost-effective.
For the plant research community, these advancements promise new capabilities for non-invasive plant phenotyping, stress detection, and chemical characterization at scale. As reconstruction algorithms continue to mature, the combination of ubiquitous RGB imaging and computational spectral reconstruction may ultimately democratize hyperspectral analysis, transforming it from a specialized technique to a mainstream tool for agricultural innovation and plant-based drug discovery.
Computational spectral reconstruction represents a transformative approach to hyperspectral imaging, enabling researchers to derive rich spectral information from conventional RGB images. For plant scientists and drug development professionals, this technology offers a pathway to sophisticated spectral analysis without prohibitive hardware investments. By understanding the fundamental principles, methodological approaches, and application protocols outlined in this technical guide, researchers can effectively leverage these techniques to advance plant health monitoring, stress detection, and biochemical characterization. As reconstruction algorithms continue to evolve, the synergy between accessible RGB imaging and computational spectral recovery will undoubtedly open new frontiers in plant research and agricultural innovation.
Combining red–green–blue (RGB) and hyperspectral imaging (HSI) has emerged as a transformative approach in plant research, enabling simultaneous assessment of morphological, physiological, and biochemical traits. While RGB imaging captures high-spatial-resolution data on plant structure and color, HSI provides high-dimensional spectral data for quantifying pigments, water content, and stress responses [68] [69]. This integration bridges the gap between phenotypic observations and underlying genetic and environmental interactions, supporting advancements in precision agriculture, stress phenotyping, and crop breeding. This guide explores sensor technologies, platforms, and methodologies for deploying RGB-HSI systems across controlled and field environments.
Fundamentals of RGB and Hyperspectral Imaging
Technical Comparisons
HSI captures hundreds of narrow, contiguous spectral bands (e.g., 400–1000 nm or 900–1700 nm), enabling detailed material identification based on chemical composition. In contrast, RGB imaging relies on three broad visible bands (red, green, blue), limiting its discriminative power to color and shape [9]. For example, HSI can distinguish plant stress indicators like chlorophyll fluorescence or water content imperceptible to RGB sensors [69] [3].
Table 1: RGB vs. Hyperspectral Imaging for Plant Phenotyping
| Feature | RGB Imaging | Hyperspectral Imaging |
|---|---|---|
| Spectral Bands | 3 broad bands (red, green, blue) | 100+ narrow, contiguous bands (e.g., 400–1000 nm) [9] |
| Primary Data | Color, texture, morphology | Spectral signatures for biochemical traits [69] |
| Key Applications | Biomass estimation, growth staging | Chlorophyll content, nitrogen assessment, stress detection [68] [69] |
| Limitations | Insensitive to biochemical changes | High cost, data complexity, computational demands [68] [70] |
| Cost & Accessibility | Low-cost, widely available | Expensive; requires specialized processing [71] |
Synergistic Benefits
- Enhanced Stress Detection: Hybrid systems capture both structural (RGB) and functional (HSI) traits, improving early detection of drought, disease, or nutrient deficiency [32] [69].
- Multi-Trait Phenotyping: RGB-derived parameters (e.g., leaf area) complement HSI-based indices (e.g., NDVI), enabling comprehensive growth monitoring [68] [3].
- Data Fusion: Pixel-level registration of RGB, HSI, and chlorophyll fluorescence data allows correlative analysis of photosynthetic efficiency and pigment dynamics [20].
Sensor Platforms and Configurations
Laboratory Scanners
Controlled-environment systems like PhenoGazer integrate HSI spectrometers, RGB cameras, and automation for high-throughput phenotyping. Key features include:
- Automated Racks: Moveable upper/lower racks for continuous daytime (reflectance) and nighttime (chlorophyll fluorescence) imaging [32].
- Sensor Fusion: Combines eight HSI fiber optics, four RGB cameras, and blue LED lights for multi-modal data acquisition [32].
- Environmental Monitoring: Includes PAR, soil moisture, and temperature sensors for contextual data [32].
Table 2: Laboratory vs. UAV-Based Imaging Systems
| Platform | Sensors | Spatial Resolution | Key Advantages | Example Use Cases |
|---|---|---|---|---|
| Laboratory Scanners | HSI spectrometer, RGB cameras, LEDs | Sub-millimeter [32] | Controlled conditions, high precision | Stress response studies [32] |
| UAVs (Multispectral) | RGB + narrow-band multispectral (e.g., Parrot Sequoia) | 1–10 cm/pixel [71] | Rapid coverage, low cost | Broad-scale chlorophyll mapping [71] |
| UAVs (Hyperspectral) | Push-broom HSI (e.g., Headwall Nano) | 1–5 cm/pixel [71] | High spectral resolution for robust modeling | Cyanobacteria monitoring [71] |
Unmanned Aerial Vehicles (UAVs)
UAVs bridge field and lab scales, enabling scalable phenotyping.
- Multispectral UAVs: Systems like DJI Mavic 3 Multispectral offer RGB and four narrow bands (green, red, red-edge, NIR), suitable for vegetation indices (e.g., NDVI) [72] [73].
- Hyperspectral UAVs: Push-broom systems (e.g., Headwall Nano-Hyperspec) require precise georeferencing but provide lab-quality spectral data for complex traits like phycocyanin [71].
- Integration Workflows: Protocols like DroneScape use dual gimbals (e.g., DJI Matrice 350) to simultaneously capture RGB and 10-band multispectral imagery, aligned post-flight [73].
Handheld and Proximal Systems
Portable spectrometers and handheld HSI systems (e.g., Specim FX10) enable leaf-scale measurements. These are ideal for validating UAV or lab data and targeting specific plant organs [9] [3].
Experimental Protocols for Multi-Modal Phenotyping
Laboratory Protocol: Multi-Trait Phenotyping
Objective: Simultaneously assess growth, chlorophyll, and water status using RGB-HSI fusion [32] [20].
Workflow:
- System Setup:
- Install HSI spectrometer (400–1000 nm) and RGB cameras on automated racks.
- Add blue LEDs for nighttime chlorophyll fluorescence induction [32].
- Data Acquisition:
- Capture RGB images during the day for morphology.
- Acquire HSI reflectance (500–900 nm) and fluorescence (690–750 nm) [20].
- Image Registration:
- Use affine transformation to align RGB, HSI, and fluorescence images [20].
- Trait Extraction:
- RGB: Leaf area, biomass via pixel classification.
- HSI: Calculate NDVI, chlorophyll indices (e.g., LCI), water bands [68].
Diagram Title: Laboratory RGB-HSI Phenotyping Workflow
UAV Protocol: Field-Based Crop Monitoring
Objective: Map chlorophyll-a and cyanobacteria in ponds or fields using multispectral/hyperspectral UAVs [71].
Workflow:
- Pre-Flight:
- Data Collection:
- Capture synchronized RGB and multispectral/hyperspectral imagery.
- Log GPS/IMU data for georeferencing [71].
- Processing:
- Generate orthomosaics and extract reflectance values.
- Apply bio-optical models (e.g., chlorophyll-a: 660–690 nm/700–730 nm) [71].
The Scientist’s Toolkit
Table 3: Essential Reagents and Materials for Plant Phenotyping
| Item | Function | Example Use Case |
|---|---|---|
| Calibrated Reflectance Panel | Converts sensor data to reflectance; ensures spectral accuracy [73] | UAV field surveys [73] |
| Blue LED Illumination | Induces chlorophyll fluorescence for nighttime imaging [32] | Laboratory stress phenotyping [32] |
| Hyperspectral Imaging Software | Processes hypercubes; extracts vegetation indices [3] | Growth stage classification [3] |
| RTK GPS Base Station | Provides centimeter-accurate georeferencing for UAV imagery [73] | Precision agriculture mapping [73] |
| Multi-Well Plates | High-throughput screening of plant responses under controlled conditions [20] | Phenotyping Arabidopsis thaliana [20] |
Data Processing and Analysis
Image Registration and Fusion
- Challenges: Multi-modal data (RGB, HSI, fluorescence) exhibit spatial mismatches due to sensor positioning [20].
- Solutions:
Machine Learning for Trait Prediction
- Feature Selection: Hyperspectral data reduction via Principal Component Analysis (PCA) or wavelength selection (e.g., 5 bands for growth staging) [3].
- Models:
Diagram Title: Hyperspectral Data Analysis Pipeline
Integrating RGB and hyperspectral imaging unlocks multi-scale, multi-trait phenotyping capabilities essential for modern plant research. Laboratory systems provide granular insights under controlled conditions, while UAVs and handheld devices enable scalable field deployment. Success hinges on selecting platform-appropriate sensors, implementing robust registration protocols, and leveraging machine learning for data fusion. As sensors evolve, combining RGB-HSI with emerging technologies (e.g., LiDAR) will further illuminate plant-environment interactions.
Data Fusion and Machine Learning for Enhanced Analysis and Interpretation
The integration of RGB and hyperspectral imaging, powered by advanced machine learning algorithms, is revolutionizing plant research by enabling comprehensive, non-destructive analysis of physiological and biochemical traits. This technical guide examines the architectures, methodologies, and applications of data fusion techniques that combine the high-resolution spatial information from RGB images with the detailed spectral data from hyperspectral imaging. We present quantitative evidence demonstrating that fused data approaches significantly outperform single-modality analyses across various plant science applications, from quality assessment to disease detection. The implementation of these technologies provides researchers with powerful tools for enhanced phenotypic characterization, early stress detection, and precision agriculture applications.
RGB Imaging Fundamentals
Red, Green, Blue (RGB) imaging captures reflected light in three broad wavelength bands corresponding to human visual perception (approximately 400-700 nm). This technology provides high-resolution spatial information about plant morphology, color, and texture, making it ideal for assessing visual quality attributes and morphological traits. In plant research, RGB imaging has been widely deployed for tasks including growth monitoring, yield estimation, and visual quality assessment of agricultural products [29]. The widespread availability, relatively low cost, and straightforward data processing requirements of RGB cameras have facilitated their adoption in various plant phenotyping applications. However, the limitation of RGB imaging lies in its inability to detect biochemical changes or subtle physiological alterations that precede visible symptoms.
Hyperspectral Imaging Fundamentals
Hyperspectral imaging (HSI) captures reflected light across hundreds of narrow, contiguous spectral bands, typically ranging from the visible to near-infrared regions (400-1000 nm or beyond) [74]. This technology generates a three-dimensional datacube (hypercube) with two spatial dimensions and one spectral dimension, containing complete spectral information for each pixel in the image [75]. The rich spectral data enables detection of subtle changes in plant biochemical composition, including pigment content, water status, and nutrient levels, often before visible symptoms manifest [21] [76]. Hyperspectral imaging has demonstrated exceptional capability in predicting nutrient content [29], detecting plant diseases [75], and classifying fine-scale growth stages [3]. The primary limitations of HSI include higher equipment costs, computational complexity, and challenges in data management due to large file sizes.
Data Fusion Methodologies and Architectures
Technical Challenges in Data Fusion
The integration of RGB and hyperspectral data presents significant technical challenges stemming from fundamental differences in data dimensionality and structure. RGB images are typically 2D arrays with three color channels, while hyperspectral data constitutes a 3D hypercube with hundreds of spectral bands [29]. Additional challenges include spatial resolution mismatches, varying signal-to-noise ratios, and alignment difficulties between modalities. Successful data fusion requires addressing these issues through specialized preprocessing, feature extraction, and alignment techniques to ensure meaningful integration of complementary information.
Data-Level Fusion Approaches
Data-level fusion involves the direct integration of raw or preprocessed data from multiple sensors before feature extraction. For RGB and hyperspectral fusion, this typically requires transforming the 1D spectral data from HSI into a 2D spatial representation that can be concatenated with RGB data in the channel direction [29]. In the ResNet-R&H model developed for vegetable soybean freshness classification, researchers used downsampling technology to reconstruct RGB images and transformed one-dimensional hyperspectral data into two-dimensional space, allowing the data to be overlaid and concatenated in the channel direction [29]. This approach generates fused data that preserves both the high-resolution spatial information from RGB and the detailed spectral signatures from HSI, creating an enriched input for subsequent analysis.
Feature-Level Fusion Strategies
Feature-level fusion involves extracting relevant features from each modality separately, then combining these features into a unified representation. This approach typically employs dedicated feature extraction pipelines for each data type, followed by feature concatenation or more sophisticated integration methods. For plant disease detection, feature-level fusion might combine texture features from RGB images with spectral indices from HSI data [21]. The advantage of this approach is the ability to optimize feature extraction for each modality independently while reducing dimensionality before fusion. Common feature extraction techniques include vegetation indices calculation from spectral data (e.g., CTR2, LLSI, and spectral disease indices) [21] and convolutional neural network (CNN) features from RGB images.
Decision-Level Fusion Techniques
Decision-level fusion maintains separate processing pipelines for each modality until the final classification or regression stage, where outputs from individual models are combined. This approach was demonstrated in tea green leafhopper damage classification, where separate models were developed for RGB and hyperspectral data [77]. The RGB classification utilized WT-VGG16 architecture with 80.0% accuracy, while the hyperspectral classification employed SPA-LSTM achieving 95.6% accuracy [77]. Decision-level fusion combines these outputs through voting schemes, weighted averaging, or meta-classifiers. This approach offers flexibility in model selection for each modality and robustness to sensor-specific failures, though it may miss subtle cross-modal interactions captured by earlier fusion strategies.
Machine Learning Architectures for Fused Data
Deep Learning Models for Multimodal Data
Convolutional Neural Networks (CNNs) have emerged as the predominant architecture for analyzing fused RGB-hyperspectral data due to their ability to automatically learn hierarchical features from spatial and spectral dimensions. The ResNet (Residual Network) architecture has demonstrated particular effectiveness for fused data analysis, as evidenced by the ResNet-R&H model achieving 97.6% accuracy in vegetable soybean freshness classification [29]. The residual connections in ResNet facilitate training of deeper networks while mitigating vanishing gradient problems, making them well-suited for capturing complex relationships between multi-channel data [29]. For hyperspectral-only classification, 3D CNNs can simultaneously extract spatial and spectral features, while 2D CNNs applied to spectral subsets have also shown strong performance, with EfficientNet-B0 achieving 81% accuracy for multiple wheat infection classification [75].
Traditional Machine Learning with Feature Engineering
Traditional machine learning algorithms combined with carefully engineered features remain competitive for specific applications, particularly when training data is limited. Support Vector Machines (SVM) have demonstrated excellent performance in wheat growth stage classification, achieving F1 scores of 0.832 when combined with appropriate spectral transformations like Standard Normal Variate (SNV), Hyper-hue, or Principal Component Analysis [3]. Feature selection methods such as Competitive Adaptive Reweighted Sampling (CARS) can identify minimal wavelength sets (as few as 5 wavelengths) that maintain high classification accuracy (F1 score 0.752) while significantly reducing dimensionality [3]. Partial Least Squares Regression (PLSR) combined with feature selection has also proven effective for nutrient prediction from spectral data [74].
Hybrid and Sequential Models
Hybrid architectures that combine different neural network components or integrate deep learning with traditional machine learning have shown promise for specialized tasks. Long Short-Term Memory (LSTM) networks effectively model sequential dependencies in spectral data, achieving 95.6% accuracy for tea pest damage classification when combined with Successive Projections Algorithm (SPA) feature selection [77]. For hyperspectral image reconstruction from RGB inputs, specialized architectures including MIRNet, HRNet, MPRNet, and Restormer have demonstrated strong performance, with MIRNet achieving PSNR of 33.29 dB in reconstruction tasks [74]. These reconstructed hyperspectral images enabled accurate prediction of needle nutrient content with R² values of 0.8523, 0.7022, and 0.8087 for nitrogen, phosphorus, and potassium, respectively [74].
Quantitative Performance Comparison
Table 1: Performance Metrics of RGB, Hyperspectral, and Fused Data Approaches
| Application | Model | Data Modality | Accuracy | Other Metrics |
|---|---|---|---|---|
| Vegetable Soybean Freshness Classification | ResNet-R&H | RGB & Hyperspectral Fusion | 97.6% | - |
| Vegetable Soybean Freshness Classification | ResNet18 | Hyperspectral Only | 93.6% | - |
| Vegetable Soybean Freshness Classification | ResNet18 | RGB Only | 90.4% | - |
| Tea Green Leafhopper Damage Classification | SPA-LSTM | Hyperspectral Only | 95.6% | - |
| Tea Green Leafhopper Damage Classification | WT-VGG16 | RGB Only | 80.0% | - |
| Multiple Wheat Infection Classification | EfficientNet-B0 (2D) | Hyperspectral Only | 81% | - |
| Wheat Growth Stage Classification | SVM with Feature Selection | Hyperspectral with Transformations | F1: 0.832 | - |
| Wheat Growth Stage Classification | SVM with 5 Wavelengths | Hyperspectral with Feature Selection | F1: 0.752 | - |
| Pine Needle Nutrient Prediction (N) | PLSR | Reconstructed Hyperspectral | R²: 0.8523 | - |
| Pine Needle Nutrient Prediction (P) | PLSR | Reconstructed Hyperspectral | R²: 0.7022 | - |
| Pine Needle Nutrient Prediction (K) | PLSR | Reconstructed Hyperspectral | R²: 0.8087 | - |
Table 2: Performance Comparison of Hyperspectral Reconstruction Models
| Reconstruction Model | PSNR (dB) | Spectral Range | Resolution | Key Applications |
|---|---|---|---|---|
| MIRNet | 33.29 | 400-1000 nm | 3.4 nm | Nutrient prediction |
| HRNet | 26.89 | 400-1000 nm | 3.4 nm | Nutrient prediction |
| MPRNet | 33.50 | 400-1000 nm | 3.4 nm | Nutrient prediction |
| Restormer | 33.40 | 400-1000 nm | 3.4 nm | Nutrient prediction |
Experimental Protocols and Methodologies
Plant Material Preparation and Imaging
Vegetable Soybean Freshness Classification Protocol
Vegetable soybean pods of genotype 'Zhenong 6' were harvested at the R6 stage and stored in a controlled environment greenhouse maintaining 24°C, 60% humidity, 400 ppm CO2 concentration, and a 12/12-hour light/dark photoperiod [29]. RGB images were captured using a Canon EOS 200D II camera with specialized lens configuration (f=18-55mm, focal length 0.25 m, shutter speed 1/4000). Hyperspectral images were collected concurrently, with spectral information extracted using ENVI software [29]. For physicochemical analysis, seed hardness was measured using a TA.XT Plus texture analyzer with cylinder stainless probe (diameter 2mm), while chemical compositions including moisture content, free amino acids, soluble sugar, protein, and oil content were determined through standardized laboratory methods [29].
Multiple Wheat Infection Classification Protocol
Wheat variety 'Vuka' was sown in 7.5 cm diameter pots and grown in disease-free growth rooms at 17/11°C for 16/8 h light/dark cycles for 10 days [75]. Pathogen inoculations included yellow rust (isolate WYR 19/215), mildew (NIAB 21-001 isolate), and Septoria (R13 and R16 inoculum). For multiple infections, inoculations were timed to synchronize symptom development: yellow rust and mildew inoculations at 10 and 13 days, respectively; yellow rust and Septoria inoculations at 10 and 17 days, respectively [75]. Hyperspectral imaging used a VideometerLab 4 camera, generating hypercubes of 2192×2192×19 dimensions covering 375-970 nm range. A total of 1447 images were acquired for model training and validation [75].
Wheat Growth Stage Classification Protocol
Wheat cultivar 'Scepter' was grown in both greenhouse and semi-natural environments using standardized growth substrate (1:1:1 sand, clay, and University of California soil mix) [3]. Greenhouse plants were maintained at 18°C day/13°C night with controlled irrigation. Hyperspectral imaging used a WIWAM system with Specim FX10 camera (400-1000 nm range, 5.5 nm FWHM resolution) positioned 1.4 m above plants, capturing top-view images at 512 pixels per line with 2.6 mm spatial resolution [3]. Images were collected between 12:00 PM to 2:00 PM with black-and-white reference images captured for calibration.
Data Preprocessing Workflows
Hyperspectral Data Preprocessing
Raw hyperspectral data requires extensive preprocessing to correct for sensor artifacts, illumination variations, and environmental effects. Standard workflow includes acquiring and cleaning images, followed by data extraction and processing [21]. Key steps include:
- Radiometric Correction: Converting raw digital numbers to reflectance values using white and dark reference measurements
- Geometric Correction: Addressing spatial distortions from sensor optics
- Noise Reduction: Applying spectral and spatial filtering to reduce random noise
- Normalization: Standardizing data across imaging sessions using techniques like Standard Normal Variate (SNV) transformation [3]
Failure to implement proper preprocessing can lead to wavelength shifts and reflectance variations, significantly impacting classification accuracy [21].
RGB Data Preprocessing
RGB image preprocessing typically includes:
- Color Calibration: Using standard color charts to ensure color accuracy
- Geometric Correction: Lens distortion compensation
- Background Segmentation: Isolating plant material from background using masking techniques [6]
- Image Enhancement: Contrast adjustment and noise reduction
Data Fusion Preprocessing
For effective data fusion, additional preprocessing steps include:
- Spatial Alignment: Ensuring pixel-level registration between RGB and hyperspectral images
- Resolution Matching: Downsampling or upsampling to achieve consistent spatial resolution
- Dimensionality Transformation: Converting 1D spectral data to 2D spatial representation for channel concatenation [29]
Visualization of Data Fusion Workflows
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Equipment and Software for RGB-Hyperspectral Fusion Research
| Category | Item | Specification/Example | Function in Research |
|---|---|---|---|
| Imaging Hardware | RGB Camera | Canon EOS 200D II [29] | Captures high-resolution spatial and color information |
| Hyperspectral Camera | VideometerLab 4 [75], Specim FX10 [3], Gaifields Pro-V10 [74] | Captures spectral datacubes across visible and NIR ranges | |
| Imaging Chamber | Controlled lighting environment [3] | Ensures consistent illumination conditions | |
| Calibration Targets | White and black reference panels [3] | Enables radiometric calibration of images | |
| Software & Algorithms | Image Processing | ENVI [29] | Extracts and processes hyperspectral information |
| Deep Learning Frameworks | PyTorch, TensorFlow | Implements ResNet, EfficientNet, VGG architectures | |
| Traditional ML | Scikit-learn, MATLAB | Implements SVM, PLSR, feature selection algorithms | |
| Data Visualization | Custom scripts, ColorBrewer [78] | Creates accessible visualizations and color schemes | |
| Laboratory Equipment | Texture Analyzer | TA.XT Plus (Stable Micro Systems) [29] | Measures mechanical properties like seed hardness |
| Chemical Analysis | Hitachi 8900 amino acid analyzer [29] | Quantifies biochemical composition for validation | |
| Sample Preparation | Standardized growth chambers [29] [75] | Maintains consistent plant growth conditions | |
| Field Equipment | Drone Platforms | UAV-mounted sensor systems [21] | Enables large-scale field imaging |
| Portable Spectrometers | Field-deployable HSI systems | Allows in-situ spectral measurements |
The integration of RGB and hyperspectral imaging through advanced machine learning techniques represents a paradigm shift in plant research methodology. The quantitative evidence presented demonstrates that fused data approaches consistently outperform single-modality analyses, with accuracy improvements of 4.0-7.2% in classification tasks [29]. The complementary nature of these modalities - combining rich spatial information from RGB with detailed biochemical signatures from HSI - enables researchers to capture comprehensive phenotypic information non-destructively.
Future developments in this field will likely focus on reducing the cost and complexity barriers associated with hyperspectral imaging through reconstruction techniques from RGB inputs [74], optimizing feature selection to minimize data dimensionality while maintaining accuracy [3], and developing standardized protocols for multi-sensor data fusion. As these technologies become more accessible and computationally efficient, they have the potential to transform precision agriculture, high-throughput phenotyping, and sustainable crop management practices through enhanced analysis and interpretation capabilities.
Proof of Performance: Validating and Comparing Imaging Technologies
In plant research, the choice of imaging technology directly dictates the depth and quality of insights one can extract. While traditional red-green-blue (RGB) imaging captures what the human eye can see, multispectral and hyperspectral imaging extend vision into realms of light interaction that reveal the internal physiology and health status of plants. This technical guide provides a direct performance comparison of these three imaging modalities, framed within the context of advancing plant science. Specifically, we demonstrate how the combination of high-resolution RGB data with rich spectral data from hyperspectral imaging creates a powerful synergy for research, enabling both detailed morphological analysis and deep chemical characterization.
The fundamental difference between these technologies lies in their approach to sampling the electromagnetic spectrum. RGB imaging captures three broad bands corresponding to red, green, and blue light [79]. Multispectral imaging (MSI) collects data at several (typically 3-10) specific, discrete wavelength bands, which can include non-visible ranges like near-infrared (NIR) [80]. Hyperspectral imaging (HSI), by contrast, captures hundreds of narrow, contiguous spectral bands, creating a continuous spectrum for each pixel in the image [81] [82]. This creates a detailed data cube where each spatial location (x, y) is associated with a full light spectrum, enabling precise material identification based on unique spectral signatures [13] [81].
Core Technical Specifications and Performance Comparison
The performance disparities between RGB, multispectral, and hyperspectral imaging stem from their fundamental design parameters, including spectral resolution, bandwidth, and data structure.
Table 1: Fundamental Technical Specifications of RGB, Multispectral, and Hyperspectral Imaging
| Parameter | RGB Imaging | Multispectral Imaging (MSI) | Hyperspectral Imaging (HSI) |
|---|---|---|---|
| Number of Bands | 3 discrete bands (Red, Green, Blue) [79] | 3-10 discrete bands [80] | 100+ contiguous bands (often 200-300+) [81] [80] |
| Bandwidth (Spectral Resolution) | Very broad (~100 nm each) [79] | Broad (50-200 nm) [80] | Very narrow (1-15 nm) [80] |
| Spectral Coverage | Visible light only (400-700 nm) [82] | Selected bands in visible and/or non-visible (e.g., NIR) [80] | Continuous spectrum from visible to infrared (e.g., 400-2500 nm) [81] |
| Data Output | Single 2D image [82] | Multiple 2D images (one per band) [82] | 3D Data Hypercube (x, y, wavelength) [82] |
| Data Volume | Low (MB range) | Medium (MB - GB range) [80] | Very High (TB range for large areas) [80] |
| Primary Data Strength | High spatial resolution, morphological features | Targeted spectral information for specific indices | Complete spectral signature for each pixel [81] |
Table 2: Performance Comparison in Plant Research Applications
| Aspect | RGB Imaging | Multispectral Imaging (MSI) | Hyperspectral Imaging (HSI) |
|---|---|---|---|
| Spatial Resolution | Generally very high | Generally high [79] | Often lower due to trade-offs with spectral detail [79] |
| Early Stress Detection | Limited to visible symptoms | Can detect stress after physiological changes | Can detect pre-visual stress at a biochemical level [1] |
| Material Identification | Based on color and morphology | Can distinguish broad material classes | Precise identification of materials and chemicals [79] |
| Quantification Power | Low; qualitative or semi-quantitative | Medium; good for established indices | High; enables detailed chemical mapping [13] |
| Pathogen/Disease Detection | Possible only when visual symptoms appear | Can detect specific diseases with known spectral responses | Can differentiate between pathogen types and infection degrees [83] |
| Processing Complexity | Low | Medium | High; requires specialized algorithms [79] |
| Cost & Accessibility | Low cost and highly accessible | Cost-effective; increasingly accessible [79] [80] | High cost; requires specialized expertise [79] [80] |
The tables illustrate a clear trade-off: as spectral resolution and analytical power increase, so do data complexity, processing requirements, and cost. Hyperspectral imaging's key advantage is its continuous sampling, which produces smooth spectral curves that are sensitive to subtle spectral features caused by molecular vibrations and chemical composition [81]. This is critical for detecting pre-visual plant stress. Multispectral systems, with their discrete bands, produce spectra that are "stair-stepped" and may miss these subtle features if the bands are not perfectly positioned [81]. RGB's three broad bands provide almost no discriminative power for biochemical analysis beyond basic color.
Experimental Protocols for Plant Imaging
To illustrate the practical application and performance differences, we detail two experimental protocols from recent research. The first demonstrates the fusion of RGB and hyperspectral data, while the second showcases a pure hyperspectral approach for complex vegetation mapping.
Protocol 1: Fused RGB-HSI for Fungal Infection Detection in Apples
This protocol, adapted from a 2023 study, successfully combined RGB and HSI to detect black root mold (BRM) infection degrees in apples, achieving a 96% accuracy in prediction sets [83].
1. Sample Preparation:
- Obtain 230 apple samples with uniform ripeness, shape, and color.
- Inoculate 178 samples with Rhizopus Stolonifer (RS).
- Store inoculated samples at 25°C and 99% relative humidity for up to 5 days.
- Label infection degrees based on time since infection: Healthy (0 days), Mild (2 days), Moderate (3 days), Severe (5 days).
2. Image Acquisition Setup:
- RGB Imaging: Use a digital imaging instrument (e.g., MV-CE060-10UC). Set the sample-to-lens distance to 500 mm and use an 8 mm focal length lens to ensure the entire sample is within the 450 mm × 300 mm field of view [83].
- HSI System: Use a visible/NIR hyperspectral imager (e.g., SOC710VP) in a dark box with tungsten halogen lamps. Set the distance between the carrier table and lens to 50 cm. The spectral range should cover 400-1000 nm [83].
3. Data Acquisition and Pre-processing:
- Capture RGB and HSI images for all samples.
- For HSI, perform black and white correction using the formula:
R = (R0 - B) / (W - B) * 100%, whereR0is the raw image,Wis a white reference image, andBis a dark reference image [83]. - Separate apples from the background in both RGB and HSI using threshold segmentation to create Region of Interest (ROI) masks.
4. Feature Extraction:
- From RGB: Extract statistic features (color moments: mean, variance, skewness) and deep learning-based network features using a Convolutional Neural Network (CNN) [83].
- From HSI: Extract mean reflectance spectra from the ROI. Use an algorithm like Random Frog (RFrog) to identify Effective Wavelengths (EWs). The study found 790, 865, and 891 nm to be critical. Extract the monochrome images at these EWs and superpose them. Extract the same statistic and network features from these EW images as from the RGB images [83].
5. Data Fusion and Modeling:
- Fuse the extracted statistic and network features from both the RGB and HSI-EW images.
- Use machine learning classifiers like Random Forest (RF), Support Vector Machine (SVM), or K-Nearest Neighbor (KNN) to build a classification model identifying the infection degree. The fusion model achieved superior performance compared to using either data source alone [83].
Protocol 2: Drone-Based HSI for Monitoring Moss and Lichen in Antarctica
This 2025 protocol highlights the use of HSI in extreme environments for fine-scale vegetation mapping, achieving up to 99.8% accuracy with a UNet model [84].
1. Field Data Collection:
- Conduct the study in a targeted area (e.g., Antarctic Specially Protected Area 135).
- Collect ground-based HSI data and high-resolution RGB imagery for validation and labeling.
- Use a Uncrewed Aerial Vehicle (UAV) equipped with a hyperspectral sensor (e.g., Specim AFX series) and a Real-Time Kinematic (RTK) Global Navigation Satellite System (GNSS) for precise geolocation [84].
2. Aerial Data Acquisition:
- Execute pre-planned flight paths with the UAV to capture hyperspectral data cubes over the study area.
- Ensure optimal sunny conditions for collecting reflectance data, though artificial illumination is an option [13].
3. Data Processing and Labeling:
- Convert raw data to reflectance using known reflectance reference panels in the scene or atmospheric correction [13].
- Use the high-resolution RGB imagery and ground observations to delineate and label geometric shapes for each class (e.g., healthy moss, stressed moss, lichen, rock, ice) in the hyperspectral data [84].
4. Model Training and Analysis:
- For full models, extract features including Principal Component Analysis (PCA) components, novel spectral indices (e.g., NDMLI, HSMI, MTHI), and statistical features.
- For light models, use only spectral indices derived from eight key wavelength bands (404, 480, 560, 655, 678, 740, 888, and 920 nm) [84].
- Train and compare multiple machine learning models, including gradient boosting (XGBoost, CatBoost) and convolutional neural networks (G2C-Conv2D, G2C-Conv3D, UNet) on both full and light feature sets [84].
- The study found that common indices like NDVI were inadequate, while custom indices were highly effective [84].
Workflow Visualization: Fused RGB-Hyperspectral Analysis
The following diagram illustrates the logical workflow for a plant study that synergistically combines RGB and hyperspectral imaging, as demonstrated in the experimental protocols.
Fused RGB-HSI Plant Analysis Workflow
This workflow highlights the complementary nature of the two imaging technologies. RGB data contributes high-resolution spatial and morphological context, while hyperspectral data provides deep spectral and chemical information. Their fusion creates a comprehensive dataset that enables more powerful and accurate machine learning models for plant phenotyping.
The Researcher's Toolkit: Essential Equipment and Reagents
Selecting the appropriate tools is critical for designing a successful spectral imaging study. The following table details key equipment and software solutions used in the featured experiments and the broader field.
Table 3: Essential Research Toolkit for Plant Spectral Imaging
| Category | Item | Function & Application Notes |
|---|---|---|
| Imaging Hardware | Hyperspectral Camera (Pushbroom/Snapshot)e.g., Specim FX/AFX series, SOC710VP | Captures the 3D hypercube. Selection depends on spectral range (VNIR: 400-1000 nm; SWIR: 900-1700 nm) required for target plant chemicals [13] [83]. |
| High-Resolution RGB Camerae.g., MV-CE060-10UC | Provides high-spatial-resolution morphological reference images for coregistration and analysis [83]. | |
| Uncrewed Aerial Vehicle (UAV/Drone) | Platform for airborne imaging, enabling coverage of large field areas or difficult terrain [84]. | |
| Calibration TargetsWhite Reference, Dark Current | Essential for converting raw digital numbers to radiometrically calibrated reflectance data [13] [83]. | |
| Software & Algorithms | Machine Learning Librariese.g., Scikit-learn, XGBoost, CatBoost | For building classification and regression models to interpret spectral data [84]. |
| Deep Learning Frameworkse.g., TensorFlow, PyTorch | Used for complex models like CNN (e.g., UNet) for semantic segmentation of hyperspectral data [84] [83]. | |
| Spectral Analysis Softwaree.g., ENVI, Python (NumPy, SciPy) | For processing the hypercube, performing spectral unmixing, and extracting indices. | |
| Experimental Materials | Controlled Environment Chamber | For standardizing growth conditions and imaging parameters (light, temperature, humidity) [83]. |
| Stable Lighting SystemTungsten Halogen Lamps, LED Panels | Provides consistent, uniform illumination crucial for reproducible reflectance measurements [83]. | |
| Precision Geolocation SystemGNSS RTK | Provides centimeter-accurate positioning for precise spatial registration of aerial imagery [84]. |
The direct performance comparison reveals that RGB, multispectral, and hyperspectral imaging are not mutually exclusive technologies but exist on a continuum of trade-offs between spatial detail, spectral insight, cost, and complexity. RGB imaging remains invaluable for capturing high-resolution morphological data. Multispectral imaging offers a balanced, cost-effective solution for applications where the key spectral responses are well-defined. Hyperspectral imaging stands apart as the most powerful tool for detecting pre-visual stress, identifying specific chemicals and pathogens, and dealing with complex or unknown spectral profiles.
The future of advanced plant research lies in the strategic combination of these modalities. As demonstrated, fusing high-resolution RGB imagery with rich hyperspectral data cubes creates a synergistic effect that is greater than the sum of its parts. This approach provides researchers with a complete picture, from the macroscopic structure of a plant down to its biochemical makeup, enabling earlier interventions, more precise phenotyping, and ultimately, a deeper understanding of plant health and function.
The accurate, non-destructive estimation of chlorophyll content, represented by SPAD values, is crucial for monitoring plant health, stress responses, and photosynthetic efficiency. This technical guide examines a structured approach for SPAD estimation under varying environmental conditions, focusing on a study of the endangered tropical tree species Hopea hainanensis under different shade levels. The research demonstrates that integrating vegetation indices (VIs) derived from multispectral (MS) and RGB imaging with machine learning algorithms enables highly accurate SPAD prediction, with the optimal model achieving an R² of 0.9389 for modeling and 0.8013 for test samples [85]. This case study reinforces the core thesis that combining the cost-effectiveness and accessibility of RGB imaging with the rich spectral information of hyperspectral and multispectral technologies creates a powerful, versatile framework for plant phenotyping across diverse research and agricultural applications.
Chlorophyll, a key pigment in photosynthesis, is a sensitive indicator of plant physiological status, responding dynamically to environmental factors such as light intensity, water availability, and nutrient levels. The SPAD meter provides a portable, non-destructive method for measuring relative chlorophyll content, but its manual operation limits scalability for large-scale studies [85]. High-throughput phenotyping (HTP) using remote sensing imagery offers a solution. However, a significant challenge lies in developing robust estimation models that remain accurate across varying environmental conditions, such as the different shade levels experienced by understory species like Hopea hainanensis [85]. This case study details an experimental framework that addresses this challenge by fusing multi-source image data and machine learning, providing a replicable protocol for researchers.
Experimental Protocol: SPAD Estimation inHopea hainanensis
Plant Material and Environmental Design
- Species: Two-year-old seedlings of the endangered tropical tree Hopea hainanensis [85].
- Experimental Site: Haikou, Hainan, China (tropical monsoon climate) [85].
- Environmental Variable: Four orthographic shade levels (0%, 25%, 50%, and 75%) were established using shade nets to simulate different light environments [85].
- Experimental Design: 200 seedlings were allocated with 50 replicates per shade level, spaced 1.2 m apart in the field [85].
- Key Finding: Quantitative analysis confirmed that SPAD values changed significantly (p < 0.01) with shade level, with the 50% shade treatment being most conducive to chlorophyll accumulation [85].
Data Acquisition and Pre-processing
Data were collected during a key growth period (August 2021). The workflow integrated multiple data streams to ensure comprehensive phenotyping.
Figure 1: Experimental workflow for SPAD estimation under different shade conditions.
SPAD Measurement: Using a portable plant nutrient meter (TYS-4N), three leaves per seedling were randomly selected, and their SPAD values were measured and averaged to ensure representativeness [85].
RGB Image Acquisition: Canopy images were captured approximately 2 meters above the ground using a digital camera (Canon EOS 4000D) in semi-automatic aperture priority mode [85].
Multispectral Image Acquisition: A multispectral camera (MicaSense Edge 3) with five narrowband sensors was used. The spectral bands and their specifications are shown in Table 1 [85].
Image Processing: Regions of interest were defined through visual interpretation. For both RGB and MS images, this step was critical for extracting accurate color and spectral information from the plant material while excluding background [85].
Feature Extraction and Selection
Vegetation indices were calculated from the image data to serve as predictors for SPAD values.
RGB Vegetation Indices: Indices were derived from the red, green, and blue channel values. The Red-Green Ratio Index (RGRI) showed the strongest individual correlation with SPAD values [85].
Multispectral Vegetation Indices: Indices were calculated using the five available bands (Blue, Green, Red, Red Edge, and Near-IR) [85].
Feature Selection: The Lasso algorithm was employed to select the most informative VIs and eliminate multicollinearity, ensuring that all selected features had a variance inflation factor (VIF) below 10 [85].
Modeling Approaches and Statistical Analysis
Four different modeling approaches were compared to identify the optimal strategy for SPAD estimation:
- Ordinary Least Squares Regression (OLR): A standard linear regression approach.
- Linear Mixed Effects Model (LMM): Incorporated random effects for shade level, allowing the model to adapt to different shade conditions.
- Random Forest (RF): An ensemble machine learning method based on decision trees.
- Support Vector Regression (SVR): A machine learning algorithm effective for nonlinear regression.
Model performance was evaluated using the coefficient of determination (R²), root mean square error (RMSE), and mean absolute percentage error (MAPE) [85].
Quantitative Results and Performance Comparison
Model Performance Metrics
Table 1: Performance comparison of SPAD estimation models using different algorithms and data sources.
| Data Source | Modeling Algorithm | R² (Modeling) | R² (Test) | Key Features |
|---|---|---|---|---|
| Multispectral VIs | Random Forest (RF) | 0.9389 | 0.8013 | Multiple VIs, no multicollinearity (VIF<10) [85] |
| Multispectral VIs | Support Vector Regression (SVR) | High | High | Multiple VIs, no multicollinearity [85] |
| Multispectral VIs | Linear Mixed Effect Model (LMM) | High | -- | Dummy variables/random effects for shade [85] |
| RGB VIs | Random Forest (RF) | Lower than MS | -- | Multiple VIs selected by Lasso [85] |
| RGB VIs | Ordinary Least Squares (OLR) | Lower than ML | -- | Single VI (e.g., RGRI) [85] |
Vegetation Indices and Sensor Bands
Table 2: Key sensor specifications and vegetation indices for chlorophyll estimation.
| Category | Parameter / Index | Specification / Formula | Relevance to SPAD |
|---|---|---|---|
| Multispectral Bands [85] | Blue | 475 nm center (20 nm FWHM) | Chlorophyll absorption |
| Green | 560 nm center (20 nm FWHM) | Leaf reflectance | |
| Red | 668 nm center (10 nm FWHM) | Chlorophyll absorption | |
| Red Edge | 717 nm center (10 nm FWHM) | Sensitive to chlorophyll content | |
| Near-IR | 840 nm center (40 nm FWHM) | Leaf structure & health | |
| RGB VIs [85] [86] | RGRI (Red-Green Ratio Index) | R / G | Strongest individual correlation |
| Kawashima Index | (R - B) / (R + B) | Correlated with SPAD in multiple species | |
| Normalized Red Index | R / (R + G + B) | Estimator of chlorophyll content | |
| Green-Red Ratio (G:R) | G / R | Found superior in some studies (e.g., lettuce) |
The Integrated Imaging Framework: RGB and Hyperspectral Synergy
The case study demonstrates that while multispectral VIs generally outperform RGB VIs for SPAD estimation, RGB imaging remains a highly viable and cost-effective alternative, especially when combined with robust machine learning models [85]. This supports the broader thesis that combining RGB with more advanced spectral imaging creates a powerful, multi-tiered phenotyping framework.
Advantages of RGB Imaging: RGB sensors offer a low-cost, accessible, and high-resolution option for plant phenotyping. The developed GreenLeafVI FIJI plugin, for instance, facilitates high-throughput, image-based chlorophyll analysis using only RGB images, making the technique widely accessible [86]. RGB imaging is particularly effective for estimating morphological traits [24].
Advantages of Hyperspectral/Multispectral Imaging: These technologies provide rich spectral data beyond the visible range, capturing detailed biochemical information. They generally achieve higher accuracy for estimating physiological traits like chlorophyll and nitrogen content [85] [87] [88]. The Red Edge and Near-IR bands are particularly sensitive to plant physiological status and are not available in standard RGB cameras [85].
The Integrated Approach: Combining these technologies allows researchers to leverage the strengths of each. RGB images can be used for high-throughput screening and morphological assessment, while hyperspectral/multispectral data can be deployed for more detailed biochemical analysis when higher precision is required. This synergy is exemplified by systems like MADI [89] and PhenoGazer [32] [90], which integrate multiple imaging modalities (RGB, thermal, hyperspectral, chlorophyll fluorescence) for a holistic view of plant health and stress responses.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key equipment and software for image-based chlorophyll estimation.
| Category | Item | Specification / Example | Primary Function |
|---|---|---|---|
| Imaging Hardware | RGB Camera | Canon EOS 4000D [85] | Captures high-resolution visible spectrum images for color-based analysis. |
| Multispectral Camera | MicaSense Edge 3 (5 bands) [85] | Captures specific non-visible wavelengths (e.g., Red Edge, NIR) for advanced VIs. | |
| Hyperspectral Imager | Portable spectrometer with fiber optics [87] | Captures continuous, high-resolution spectral data for detailed biochemical profiling. | |
| Chlorophyll Meter | TYS-4N [85] or SPAD-502 [86] | Provides ground-truth SPAD values for model calibration and validation. | |
| Environmental Control | Shade Nets | Variable density (e.g., 0%, 25%, 50%, 75%) [85] | Manipulates light intensity as an experimental environmental variable. |
| Environmental Sensors | PAR, soil moisture, temperature sensors [90] | Logs concurrent environmental metadata to contextualize phenotypic data. | |
| Software & Analysis | Image Analysis Plugin | GreenLeafVI for FIJI/ImageJ [86] | High-throughput analysis of RGB images for chlorophyll content estimation. |
| Feature Selection Algorithm | Lasso algorithm [85] | Selects most informative vegetation indices and reduces multicollinearity. | |
| Machine Learning Library | -- (For RF, SVR, etc.) [85] | Builds predictive models linking spectral features to SPAD values. |
Advanced Data Integration and Modeling Workflow
The most robust SPAD estimation models account for complex interactions between spectral data and environmental conditions. The following diagram outlines an advanced workflow that integrates these elements.
Figure 2: Advanced data integration and modeling workflow for robust SPAD estimation.
Data Fusion and Feature Engineering: This critical step involves combining image-derived features (VIs) with environmental metadata. Studies show that integrating environmental variables such as shade level, temperature, and soil moisture with image data significantly improves the performance of machine learning models for stress classification and trait prediction compared to image-only approaches [91].
Advanced Feature Selection: For high-dimensional hyperspectral data, advanced algorithms like Dynamic Reptile Search Algorithm-enhanced CARS (DRSA-CARS) can improve feature selection. This method has been shown to reduce feature dimensionality by up to 75.7% for SPAD estimation while improving prediction accuracy (R²) by 24.4% [87].
Model Training with Environmental Adaptation: Models that incorporate environmental factors directly, either as dummy variables or random effects (as in LMM), show a greatly improved ability to adapt to different growing conditions, addressing a key limitation of many standard estimation models [85].
This case study establishes a robust methodology for non-destructive SPAD estimation that remains effective across varying environmental conditions. The integration of RGB and multispectral/hyperspectral imaging, coupled with machine learning, provides a scalable and accurate solution for high-throughput plant phenotyping. Future efforts in this field should focus on the development of standardized data fusion protocols, the creation of large, multi-species benchmark datasets [88], and the creation of user-friendly software tools to make these advanced methodologies more accessible to the broader plant science community [86]. This integrated imaging approach is pivotal for advancing our understanding of plant responses to environmental stresses and for accelerating breeding programs aimed at developing more resilient crops.
The integration of RGB and hyperspectral imaging is transforming plant research by combining accessibility with deep spectral insight. This synergy provides researchers with a powerful, multi-scale toolkit for quantifying plant health, enabling applications from precise phenotyping to accelerated drug development from plant-based compounds. While RGB imaging offers high spatial resolution and widespread availability, hyperspectral imaging (HSI) captures hundreds of contiguous spectral bands to reveal biochemical changes invisible to conventional cameras [2] [9]. This technical guide quantifies the measurable gains achieved by combining these technologies, focusing on detection accuracy, early intervention timelines, and model robustness within plant science research.
Detection Accuracy: A Quantitative Comparison
The core advantage of hyperspectral imaging lies in its superior spectral resolution, which enables the identification of unique biochemical signatures associated with plant health, stress, and disease. This section provides a quantitative comparison of detection accuracy between RGB and hyperspectral imaging across various plant research applications.
Key Performance Metrics
Table 1: Detection Accuracy of RGB vs. Hyperspectral Imaging in Plant Research
| Application | Plant/Disease Model | RGB Accuracy (Best Reported) | Hyperspectral Accuracy (Best Reported) | Citation |
|---|---|---|---|---|
| Pest Damage Classification | Tea Green Leafhopper | 80.0% (WT-VGG16 Model) | 95.6% (SPA-LSTM Model) | [77] |
| Disease Classification (Single) | Wheat Foliar Diseases | ~85% (Estimated from literature) | Up to 98.09% | [18] [92] |
| Disease Classification (Multiple Co-infections) | Wheat (Yellow Rust & Mildew) | Low accuracy on co-infections | 72.0% (EfficientNet 2D-CNN) | [18] |
| Counterfeit Detection | N/A | Limited by spectral bands | 99.03% F1-Score (for alcohol) | [92] |
| Food Quality Control | Pine Nuts | Limited by spectral bands | 100% Classification Accuracy | [92] |
Analysis of Performance Gaps
The data in Table 1 demonstrates a consistent and significant accuracy advantage for hyperspectral imaging. The ~15% performance gap in pest damage classification and near-perfect scores in material identification underscore HSI's capacity to detect subtle biochemical changes. RGB models, while effective for tasks like basic species classification, struggle with spectral ambiguity, where different materials appear visually identical but are chemically distinct [9]. Hyperspectral imaging overcomes this by capturing a unique spectral fingerprint for each material. For instance, as shown in Figure 1, an RGB camera cannot reliably distinguish an almond from its shell, whereas a hyperspectral camera easily identifies them based on an oil absorption feature at 930 nm [9].
Early Intervention Timelines: Capturing the Pre-Symptomatic Window
A critical gain from hyperspectral imaging is the dramatic extension of the intervention timeline, allowing researchers to identify stress long before visible symptoms appear.
The Science of Pre-Symptomatic Detection
Plant stress from pathogens, pests, or nutrient deficiency triggers biochemical and physiological changes that alter light interaction. These include:
- Chlorophyll Degradation: Affects absorption in the 600-735 nm range, impacting photosynthesis [18].
- Water Content Changes: Alters reflectance in bands beyond 750 nm and in the 1400-1450 nm region [18] [21].
- Cellular Structure Disruption: Changes leaf reflectance properties across multiple wavelengths [1].
These changes create unique spectral signatures that HSI can detect during the pre-symptomatic or early symptomatic stages. Research indicates that specific wavelength bands, such as 689 nm and 753 nm, are particularly critical for early infection identification [21].
Experimental Workflow for Early Disease Detection
The following diagram illustrates a typical experimental workflow for capturing and analyzing this pre-symptomatic window.
Workflow for Early Disease Detection
Quantifying the Timeline Advantage
Table 2: Early Intervention Timelines for Wheat Pathogens Using HSI
| Pathogen | Lifestyle | Symptomatic Latent Period | Pre-Symptomatic Detection via HSI (Estimated) | Key Spectral Bands for Early Detection |
|---|---|---|---|---|
| Yellow Rust (P. striiformis) | Obligate Biotroph | ~10 days [18] | ~3-7 days post-inoculation | 550-600 nm, ~690 nm, ~753 nm [18] [21] |
| Mildew (B. graminis) | Obligate Biotroph | 5-10 days [18] | ~2-5 days post-inoculation | 550-600 nm, ~690 nm, ~753 nm [18] [21] |
| Septoria (Z. tritici) | Hemibiotroph | 14-28 day latent phase [18] | ~7-14 days post-inoculation | ~695 nm, 720 nm, 1400-1450 nm [18] [21] |
This accelerated timeline enables interventions several days before visible symptoms, such as pustules or necrosis, manifest. This is crucial for research into antifungal compounds or for breeding resistance traits, allowing for more precise monitoring of treatment efficacy.
Model Robustness: Handling Complexity and Real-World Variation
Robustness is the ability of a model to maintain high performance when faced with novel data, varying environmental conditions, or complex biological scenarios. The fusion of RGB and hyperspectral data significantly enhances model robustness.
Robustness in Complex Biological Scenarios
A key challenge in plant disease diagnosis is the presence of multiple concurrent infections, which can confound classifiers. A 2025 study on wheat diseases demonstrated that hyperspectral imaging, combined with deep learning, could classify concurrent infections with significant accuracy [18]. The EfficientNet model with 2D convolution input achieved 72% accuracy in detecting leaves co-infected with yellow rust and mildew [18]. This is a notable result, as the study found that the spectral signature of a pathogen can change when another pathogen is present, indicating that models are detecting complex, emergent interactions rather than simply adding two independent signatures.
Enhancing Robustness Through Data Fusion and Reconstruction
The integration of RGB and HSI data creates more robust models. Two advanced methodologies are at the forefront:
Multi-Modal Data Fusion: Combining RGB's high spatial detail with HSI's rich spectral information in a single model. For example, a study on tea pest damage used separate but complementary models for each data type, with the HSI model (95.6% accuracy) providing a significant boost over the RGB model (80.0% accuracy) [77]. A fused system would leverage the strengths of both.
RGB-to-HSI Reconstruction: To address the cost and complexity of HSI systems, novel deep learning methods are being developed to reconstruct hyperspectral images from standard RGB inputs. The MSS-Mamba model, based on state space models, excels at this "spectral super-resolution" task by efficiently modeling long-range dependencies in spectral-spatial data with linear complexity [46]. This allows for the generation of high-fidelity spectral data from inexpensive RGB captures, making spectral analysis more accessible and robust against hardware limitations. Furthermore, dual-illumination methods using RGB sensors have been shown to improve the accuracy of spectral reflectance reconstruction by providing complementary spectral cues, enhancing the model's ability to infer intrinsic material properties [93].
The Scientist's Toolkit: Essential Research Reagent Solutions
This section details the key hardware, software, and analytical reagents required to implement the protocols and achieve the quantified gains discussed in this guide.
Table 3: Essential Research Reagents and Tools for RGB-HSI Plant Research
| Category | Item | Function & Specification Guidelines |
|---|---|---|
| Imaging Hardware | Hyperspectral Imager (Pushbroom/Snapshot) | Captures hypercubes. Select based on spectral range (e.g., VNIR: 400-1000 nm for pigments), resolution, and platform (lab, field, UAV) [21] [13]. |
| High-Resolution RGB Camera | Provides high-spatial-resolution reference imagery. Essential for data fusion and spatial context. | |
| Calibration Targets (Dark, White) | Critical for HSI data preprocessing to correct for sensor drift and convert raw data to reflectance [21]. | |
| Controlled Illumination System | Ensures consistent, uniform lighting for lab-based imaging to avoid spectral shadows and artifacts. | |
| Software & Algorithms | Data Preprocessing Platform | Tools for radiometric calibration, noise reduction, and image normalization (e.g., Python, ENVI, SPECIM software) [21] [13]. |
| Machine Learning Frameworks | Libraries (e.g., TensorFlow, PyTorch) for developing and training custom CNN, LSTM, or Transformer models for classification and reconstruction [18] [46]. | |
| Spectral Analysis Software | For calculating vegetation indices (NDVI, LLSI), Spectral Disease Indices (SDIs), and analyzing spectral libraries [21]. | |
| Biological Materials | Standardized Plant Cultivars | Use genetically uniform plant lines (e.g., wheat 'Vuka') to minimize biological variation and isolate treatment effects [18]. |
| Characterized Pathogen Strains | Use well-defined pathogen isolates (e.g., from culture collections) with known virulence for reproducible inoculations [18]. | |
| Growth & Inoculation Supplies | Growth chambers, inoculation towers, spore suspension materials, and hydrofluoroether solutions for uniform pathogen application [18]. |
Logical Pathway for Technology Selection
The following diagram outlines the decision-making pathway for selecting the appropriate imaging and analysis strategy based on research goals and constraints.
Pathway for Imaging Strategy Selection
The quantitative evidence presented in this guide firmly establishes the significant gains offered by hyperspectral imaging, particularly when combined with RGB data, for plant research. These gains are measurable in:
- Substantially elevated detection accuracy, often exceeding 95%, for pests, diseases, and quality traits.
- An dramatically extended intervention timeline, enabling detection days or weeks before visual symptoms appear, as quantified in wheat pathogen studies.
- Enhanced model robustness, allowing for accurate diagnosis in complex real-world scenarios like multiple co-infections and providing pathways to make spectral analysis more accessible via reconstruction algorithms.
As hyperspectral systems continue to become more compact, affordable, and integrated with powerful AI, their adoption will accelerate [2] [92]. The future of plant research lies in strategically combining the deep spectral interrogation of HSI with the spatial ubiquity of RGB to build more resilient crops, develop novel plant-based therapeutics, and advance our fundamental understanding of plant biology.
The quest for non-destructive, high-throughput plant phenotyping is a cornerstone of modern agricultural research. While traditional RGB imaging provides valuable structural data, the integration of hyperspectral imaging unlocks a deeper, chemical-level understanding of plant physiology by measuring unique spectral signatures across hundreds of contiguous bands [9]. This technical guide benchmarks popular Vegetation Indices (VIs) derived from such spectral data, evaluating their effectiveness for estimating specific plant traits. Our analysis, framed within a broader thesis on the benefits of combining RGB and hyperspectral technologies, reveals that index performance is highly context-dependent, influenced by the target plant trait, species, and environmental conditions. We provide structured quantitative comparisons, detailed experimental protocols, and actionable insights to guide researchers in selecting and applying the most effective VIs for their specific applications, thereby enhancing the precision and reliability of plant phenotyping and drug development research.
The limitation of conventional RGB imaging, which characterizes objects based primarily on shape and color using only three broad visible bands, is its inability to quantify chemical composition [9]. Hyperspectral technology addresses this by capturing hundreds of narrow, contiguous spectral bands, creating a detailed fingerprint that can be linked to specific biochemical and biophysical properties [9] [1]. This capability is paramount for early stress detection, as hyperspectral cameras can identify issues like nutrient deficiency or drought before they become visible to the human eye [1].
Vegetation Indices are mathematical combinations of reflectance values at specific wavelengths designed to highlight these properties. They serve as a critical bridge between raw, complex spectral data and interpretable, actionable biological insights. Effectively benchmarking these indices is therefore essential for advancing precision agriculture and the systematic discovery of plant-derived compounds.
Quantitative Benchmarking of Vegetation Indices
The performance of a Vegetation Index is not universal; it varies significantly with the plant functional type (PFT) and the specific trait being measured. The tables below summarize key findings from recent studies on VI effectiveness for estimating gross primary productivity (GPP) and pigment content.
Table 1: Performance of Vegetation Indices for Gross Primary Productivity (GPP) Estimation Across Plant Functional Types (PFTs) [94]
| Vegetation Index | Full Name | Overall Performance (R²) | Key Strengths and PFT-Specific Advantages |
|---|---|---|---|
| NIRv | Near-infrared reflectance of vegetation | 0.60 | Highest overall model performance for urban vegetation GPP; least susceptible to saturation. |
| kNDVI | Kernel Normalized Difference Vegetation Index | N/R | Unique advantages for Deciduous Broadleaf Forest (DBF) and Evergreen Needle-leaf Forest (ENF). |
| EVI | Enhanced Vegetation Index | Stronger than NDVI | Stronger correlation with GPP dynamics than NDVI in most PFTs. |
| NDVI | Normalized Difference Vegetation Index | Weaker than EVI/NIRv/kNDVI | Lower performance in linear VI-GPP relationships; prone to saturation. |
| Note | Environmental factors (temperature, shortwave radiation, vapor pressure) significantly improved GPP estimation for Evergreen Broadleaf Forest (EBF), ENF, and Savanna (SAV). |
Table 2: Sensitivity of VI-Based Pigment Assessment to Methodological Variations [95]
| Experimental Factor | Impact on Pigment Content Estimation Error | Notes and Recommendations |
|---|---|---|
| Shift in Central Wavelength | 42% - 77% relative error | Even minor shifts of <20 nm can induce large errors; highlights need for precise sensor calibration. |
| Choice of VI Formula | 36% - 86% relative error | Selecting an inappropriate index or regression model is a major source of inaccuracy. |
| Change in Bandwidth | 2% - 5% relative error | Has a much smaller impact compared to central wavelength and formula choice. |
| General Note | Comparing results from different sensors or platforms is unreliable unless channel parameters and calibration details are explicitly stated. Standardization is vital. |
Experimental Protocols for VI Benchmarking
To ensure reproducible and reliable results, researchers must adhere to rigorous experimental methodologies. The following protocols are essential for benchmarking vegetation indices.
Rhizobox-Based Plant Growth and Stress Induction
For controlled root and shoot imaging, a rhizobox system is recommended [19].
- Rhizobox Design: Construct boxes with a transparent front window (e.g., 6mm mineral glass) and a grey PVC frame, with an internal space of 10-30mm to control soil volume and weight.
- Substrate Preparation: Use a sieved (≤2mm) natural soil. Pre-wet the substrate to a specified water content (e.g., 0.12-0.18 cm³ cm⁻³) to avoid structure degradation during filling.
- Packing and Calibration: Fill rhizoboxes horizontally to prevent particle segregation. Gently compact soil to achieve a target bulk density representative of field conditions (e.g., 1.3 g cm⁻³).
- Water Regime: Calculate and apply water to achieve a target soil water content, for instance, 80% of Plant Available Water (PAW), to induce controlled water stress for experimental treatments.
Hyperspectral and RGB Data Acquisition
A dual-sensor approach provides complementary data streams [19].
- Hyperspectral Imaging: Use a hyperspectral imager (e.g., covering 400-1700 nm) to capture the full spectral signature of the plant-soil system. This data is used for calculating a wide array of VIs and for advanced analyses like water content mapping.
- RGB Imaging: Capture simultaneous high-resolution color images. These are valuable for quantifying root and shoot development over time and provide a visual reference.
- Spatial Registration: Ensure hyperspectral and RGB images are spatially aligned to facilitate direct comparison and data fusion.
Reference (Ground Truth) Data Collection
VI models must be calibrated and validated against direct, laboratory-based measurements.
- Pigment Extraction: For chlorophyll and carotenoid benchmarking, extract pigments from leaf discs using solvents like dimethylformamide. Perform technical replicates for reliability [95].
- Spectrophotometry: Quantify pigment concentrations from the extracts using a laboratory spectrophotometer, applying established equations (e.g., Lichtenthaler's formulae).
- Data Correlation: Statistically correlate the laboratory-measured pigment values with the VIs calculated from the hyperspectral imagery to build and validate calibration models.
Data Processing and VI Calculation Workflow
The journey from raw data to biological insight involves a structured pipeline, which can be visualized as a workflow as below.
Diagram 1: Spectral Data to Trait Prediction Workflow
Success in spectral phenotyping relies on a combination of hardware, software, and biological materials. The following table details key components of the experimental toolkit.
Table 3: Essential Research Reagents and Resources for VI Benchmarking
| Category / Item | Specific Examples | Function and Application |
|---|---|---|
| Imaging Hardware | Hyperspectral Imagers (e.g., Specim FX10, FX17); RGB Cameras | Captures spectral fingerprints (HSI) and structural/color data (RGB) for analysis. |
| Plant Growth System | Custom Rhizoboxes with glass front | Enables non-destructive, in-situ monitoring of root system development in natural soil. |
| Reference Analysis Tools | Laboratory Spectrophotometer | Provides accurate ground truth data for pigment concentration (Chl, Car) to calibrate VI models. |
| Data Visualization Software | Blender; HTMLview/Blenderview [96] | Creates engaging 3D models and interactive visualizations of high-dimensional data (e.g., transcriptomic). |
| Standardized Substrate | Sieved field soil (e.g., silt loam) | Provides a controlled yet natural growth medium for plant experiments. |
| Chemical Reagents | Solvents (e.g., dimethylformamide, acetone) | Used for destructive sampling and extraction of pigments for ground truth validation. |
Benchmarking vegetation indices reveals a clear, nuanced picture: there is no single "best" index for all situations. The Near-infrared reflectance of vegetation (NIRv) has demonstrated superior performance for estimating gross primary productivity in urban vegetation [94], while indices like kNDVI show unique advantages for specific forest types [94]. However, the accuracy of any VI is critically dependent on precise instrument calibration and appropriate model selection, as minor shifts in spectral parameters can induce errors exceeding 40% [95]. The integration of RGB and hyperspectral imaging creates a powerful synergy, combining detailed structural information with deep biochemical insights. This multi-modal approach, supported by rigorous benchmarking and standardized protocols, provides researchers and drug development professionals with a robust framework for non-destructive plant phenotyping, ultimately accelerating research in sustainable agriculture and plant-derived pharmaceutical compounds.
The quest for precise, non-destructive methods to monitor plant health and physiological status is a cornerstone of modern agricultural research. Optical imaging techniques provide powerful tools for this purpose, yet researchers face a fundamental trade-off: the choice between technologically simple, cost-effective systems and those offering deep, granular biochemical information. This analysis examines this trade-off by comparing conventional RGB imaging with hyperspectral imaging (HSI), and explores how their combination creates a synergistic toolset for plant sciences. RGB imaging, which measures reflectance in three broad visible bands (Red, Green, Blue), is technically accessible and low-cost [97]. In contrast, HSI captures hundreds of contiguous, narrow spectral bands, generating a detailed spectrum for each pixel in an image [98] [11]. This allows for non-destructive quantification of plant biochemical and physiological traits based on their spectral signatures [61] [74]. The core dilemma lies in balancing the unparalleled information depth of HSI against its higher system complexity and cost, a challenge that emerging technological bridges are beginning to resolve.
Technical Comparison of RGB and Hyperspectral Imaging
The fundamental difference between RGB and hyperspectral imaging lies in their respective spectral resolutions and the resulting information content. An RGB camera, equipped with a Bayer filter, captures only three wide spectral bands corresponding to red, green, and blue light [97]. A hyperspectral sensor, however, measures up to several hundred narrow bands (a few nanometers wide) across a much broader range of the electromagnetic spectrum, which may include the visible, near-infrared (NIR), and short-wave infrared (SWIR) regions [98]. The output of HSI is a three-dimensional data array known as a hypercube, containing two spatial dimensions and one spectral dimension [11].
This difference in data acquisition directly translates to a disparity in information depth. RGB data is primarily limited to assessing morphological parameters and color-based indices related to visible pigments like chlorophylls and carotenoids [97]. HSI, by contrast, can probe a wide array of plant properties due to the specific absorption features of biochemical components. For instance, water content influences the short-wave infrared range, cellular structure affects the near-infrared, and photosynthetic pigments alter the visible spectrum [98]. This enables HSI to identify and quantify disease severity, nutrient status (e.g., nitrogen), water content, and other physiological traits, often during the incubation period of a stressor when symptoms are not yet visible to the human eye [98] [74].
Table 1: Technical and Operational Comparison of RGB and Hyperspectral Imaging Systems
| Feature | RGB Imaging | Hyperspectral Imaging |
|---|---|---|
| Spectral Bands | 3 broad bands (Red, Green, Blue) [97] | Hundreds of narrow, contiguous bands [98] |
| Information Depth | Morphology, color indices, visible pigments [97] | Biochemistry, water content, nutrient status, early stress detection [98] [74] |
| Primary Strengths | Low cost, high accessibility, simple operation, high spatial resolution, rapid data processing [97] [99] | High spectral information depth, non-destructive chemical sensing, early disease detection [98] [11] |
| Data Complexity | Low (2D array of 3 values per pixel) | High (3D hypercube, large data volumes) [11] |
| System Cost | Low (consumer-grade cameras sufficient) [99] | High (specialized cameras cost \$10,000+) [99] |
| Operational Complexity | Low; platforms include smartphones, UAVs [97] | High; often requires precise scanning, stable platforms, and calibration [98] [20] |
Experimental Workflows and Imaging Pipelines
The application of these imaging technologies in research requires distinct yet sometimes interconnected workflows. The following diagrams illustrate the generalized processes for HSI-based root phenotyping and the emerging approach of fusing RGB and HSI data.
Detailed Methodologies for Key Applications
Hyperspectral Root Phenotyping in Soil: This protocol enables non-destructive root system architecture analysis and chemometric characterization [61]. Plants are grown in thin, soil-filled rhizoboxes with a transparent front. The imaging system typically consists of a push-broom hyperspectral camera (e.g., 1000-1700 nm range) with a line illumination source, mounted on a motorized positioning system [61]. The camera acquires line scans which are later composed into a full image. Critical preprocessing steps include normalizing the data using white and dark reference scans, log-linearization to correct for non-uniform illumination, and Asymptotic Least Squares (ALS) correction to remove scattering effects [61]. Automated root segmentation from the soil background is achieved via fuzzy clustering or multilevel thresholding of the preprocessed spectral data. Subsequently, the extracted spectral signatures from root pixels are analyzed to infer physico-chemical properties, such as water content based on specific water absorption bands, or root decay by tracking changes in structural carbon features [61].
Multi-Modal Image Registration for Data Fusion: This methodology allows for the pixel-level fusion of information from RGB, hyperspectral, and chlorophyll fluorescence (ChlF) sensors, leveraging the strengths of each [20]. The process begins with individual calibration of each camera to correct for lens distortion. A key step is selecting an optimal reference image (e.g., a high-contrast ChlF image) to which other modalities are aligned. Automated registration algorithms, such as Phase-Only Correlation (POC) or Normalized Cross-Correlation (NCC), are then used to compute an affine transformation matrix that aligns the "moving" image (e.g., HSI) with the reference [20]. Due to potential non-linear distortions, an additional fine registration step applied to individual plant objects within the image is often necessary to achieve a high overlap ratio (>96%) [20]. This precise alignment enables the creation of multi-modal data cubes where each spatial location contains data from all sensors, vastly enriching the feature set for machine learning models.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of plant imaging studies requires careful selection of materials and computational tools. The following table details key solutions and their functions.
Table 2: Key Research Reagent Solutions for Plant Imaging Studies
| Item Name | Function/Application in Research |
|---|---|
| Soil-Filled Rhizoboxes | Provides a near-natural growth environment for roots while allowing optical access for non-destructive, repeated imaging [61]. |
| Spectralon White Reference Tile | Used for calibrating hyperspectral images by providing a >99% reflective standard, correcting for uneven illumination and system noise [61]. |
| Hyperspectral Imaging Sensors (e.g., Specim, Headwall) | Push-broom or snapshot cameras that capture high-resolution spectral data cubes; selection depends on required spectral range (VNIR vs. SWIR) and platform [98]. |
| Controlled Illumination Systems (Halogen Line Lights) | Provides consistent, homogeneous lighting crucial for reproducible spectral measurements, minimizing shadows and specular reflection [61]. |
| Deep Learning Models (e.g., MSS-Mamba, Restormer, WASSAT) | Algorithms for reconstructing hyperspectral images from RGB inputs or analyzing complex spectral-spatial data, overcoming the cost barrier of HSI hardware [46] [74] [99]. |
| Chemometric Algorithms (e.g., PLSR, CARS) | Statistical and machine learning methods (Partial Least Squares Regression, Competitive Adaptive Reweighted Sampling) for linking spectral features to quantitative plant traits like nutrient content [74]. |
Quantitative Cost-Benefit Analysis
The decision between imaging modalities is ultimately guided by the specific research question, budget, and operational constraints. The following table synthesizes key quantitative comparisons to inform this decision.
Table 3: Quantitative Cost-Benefit Analysis of Imaging Approaches
| Analysis Criterion | RGB Imaging | Hyperspectral Imaging | RGB-HSI Fusion (Reconstruction) |
|---|---|---|---|
| Financial Outlay | Low (< few hundred USD) [99] | Very High (> \$10,000 USD) [99] | Low (leverages existing RGB hardware) [74] |
| Spectral Resolution | 3 bands [97] | Hundreds of bands (e.g., 176 bands @ 3.4 nm) [74] | Reconstructed HSI (e.g., 176 bands) [74] |
| Nutrient Prediction (R²) | Not directly possible | N: 0.90, P: 0.68, K: 0.84 [74] | N: 0.85, P: 0.70, K: 0.81 [74] |
| Disease Classification | 80.0% accuracy (tea leafhopper) [77] | 95.6% accuracy (tea leafhopper) [77] | N/A |
| Data Processing Demand | Low to Moderate | Very High (large hypercubes) [11] | High (DL model inference) [46] |
| Operational Scalability | High (UAVs, smartphones) [97] | Low to Moderate (platform stability critical) [98] | High (potential for field deployment) [74] |
The cost-benefit analysis between RGB and hyperspectral imaging reveals a clear trade-off. HSI provides unrivalled information depth for non-destructive biochemical sensing and early stress detection, but its high cost and complexity can be prohibitive [11] [99]. RGB imaging offers an accessible, scalable alternative for morphological and some color-based physiological assessments, but lacks the granularity for detailed chemical analysis [97]. The most promising path forward lies in combining these modalities not just at the data level through registration [20], but also through the emerging paradigm of hyperspectral image reconstruction. Deep learning models that generate high-fidelity hyperspectral data from standard RGB inputs [46] [74] represent a paradigm shift, potentially offering a favorable balance by lowering the cost and complexity barrier while preserving critical spectral information. This approach, alongside continued development of robust multi-modal fusion pipelines, will democratize advanced plant phenotyping, accelerating research in crop breeding, precision agriculture, and plant physiology.
Conclusion
The combination of RGB and hyperspectral imaging represents a paradigm shift in plant analysis, moving from subjective visual assessment to objective, data-driven insight. RGB provides accessible morphological context, while hyperspectral imaging unlocks a deep, non-invasive window into plant physiology and chemistry. This synergy enables the early detection of stresses and diseases, precise chemotyping for medicinal plants, and high-throughput phenotyping that is crucial for modern breeding programs. Future directions point toward the wider adoption of portable and snapshot hyperspectral systems, more sophisticated data fusion algorithms, and advanced machine learning models that can fully exploit this rich multimodal data. For biomedical and clinical research, particularly in the realm of plant-derived pharmaceuticals, this integrated approach promises stricter quality control, accelerated compound discovery, and a more profound understanding of how growth conditions influence bioactive chemical profiles, ultimately leading to more standardized and efficacious plant-based therapeutics.
References
- 1. Top 3 Benefits of Hyperspectral Imaging for Agriculture ... [https://www.livingoptics.com/top-3-benefits-of-hyperspectral-imaging-for-agriculture-research/]
- 2. Hyperspectral Imaging in Agriculture Market: 2025 Advances [https://farmonaut.com/remote-sensing/hyperspectral-imaging-in-agriculture-market-2025-advances]
- 3. Hyperspectral-based classification of individual wheat ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12606948/]
- 4. Video‐rate tunable colour electronic paper with human ... [https://www.nature.com/articles/s41586-025-09642-3]
- 5. Transformative Impact of Hyperspectral Imaging on Plant ... [https://www.specim.com/case-phenotrait/]
- 6. Hyperspectral reflectance imaging and spectral component ... [https://www.sciencedirect.com/science/article/pii/S2666166725002606]
- 7. AI-based absorption spectrum prediction of plants from ... [https://spie.org/photonics-west/presentation/AI-based-absorption-spectrum-prediction-of-plants-from-RGB-imaging/13887-22]
- 8. Hyperspectral imaging and its applications: A review - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11253060/]
- 9. Hyperspectral Technology vs. RGB [https://www.specim.com/hyperspectral-technology-vs-rgb/]
- 10. Application of Hyperspectral Imaging for Early Detection ... [https://www.mdpi.com/2073-4395/15/7/1516]
- 11. Applications of hyperspectral imaging in plant phenotyping [https://www.sciencedirect.com/science/article/abs/pii/S1360138521003460]
- 12. A hyperspectral plant health monitoring system for space ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2023.1133505/full]
- 13. Benefits of hyperspectral imaging for research [https://www.specim.com/technology/benefits-of-hsi-for-research/]
- 14. Evaluating potential of leaf reflectance spectra to monitor plant ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-023-01089-9]
- 15. Spectroscopic Determination of Leaf Biochemistry Using ... [https://www.sciencedirect.com/science/article/abs/pii/S0034425798000844]
- 16. Hyperspectral reflectance as a tool to measure biochemical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5853784/]
- 17. How RGB can reveal hidden contrast in ag imaging [https://www.linkedin.com/posts/kevin-shearer-a3522a116_agtech-precisionag-rgb-activity-7346356880799121408-6y8o]
- 18. Hyperspectral image analysis for classification of multiple ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12595906/]
- 19. RGB and Spectral Root Imaging for Plant Phenotyping and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5614140/]
- 20. Automated image registration of RGB, hyperspectral and ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-024-01296-y]
- 21. Ultimate Guide to Spectral Data for Plant Disease Detection [https://anvil.so/post/ultimate-guide-to-spectral-data-for-plant-disease-detection]
- 22. Recent advances in plant disease detection: challenges and ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-025-01450-0]
- 23. Application of Non-Destructive Technology in Plant ... [https://www.mdpi.com/2077-0472/15/15/1670]
- 24. Application of RGB-Imaging techniques for high-throughput ... [https://agrifoodscience.com/index.php/TURJAF/article/view/7951]
- 25. Unlocking the potential of simulated hyperspectral imaging ... [https://www.frontiersin.org/journals/agronomy/articles/10.3389/fagro.2024.1435234/full]
- 26. Advances in Hyperspectral and Diffraction Imaging for ... [https://www.mdpi.com/2077-0472/15/16/1775]
- 27. Large-scale non-destructive crown-level assessment of ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-025-01439-9]
- 28. Hyperspectral imaging VIS-NIR and SWIR fusion for ... [https://www.sciencedirect.com/science/article/abs/pii/S0168169925008087]
- 29. ResNet incorporating the fusion data of RGB & ... [https://www.nature.com/articles/s41598-024-51668-6]
- 30. Hyperspectral segmentation of plants in fabricated ... [https://www.frontiersin.org/journals/high-performance-computing/articles/10.3389/fhpcp.2025.1547340/full]
- 31. Hyperspectral image analysis techniques for the detection and ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-017-0233-z]
- 32. PhenoGazer: A high-throughput phenotyping system to ... [https://www.sciencedirect.com/science/article/pii/S2643651525000536]
- 33. A hyperspectral plant health monitoring system for space ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10352677/]
- 34. Non-destructive inspection for plant stress detection [https://sensing.konicaminolta.eu/mi-en/solutions/agriculture/disease-identification]
- 35. Hyperspectral imaging in agriculture and vegetation [https://www.specim.com/hyperspectral-imaging-applications/agriculture-and-vegetation/]
- 36. Smarter crop monitoring with hyperspectral imaging [https://www.photonfocus.com/smarter-crop-monitoring-with-hyperspectral-imaging/]
- 37. Early-Stage Detection of Biotic and Abiotic Stress on Plants by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10452221/]
- 38. RGB to hyperspectral: Spectral reconstruction for enhanced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11665794/]
- 39. A comprehensive review of crop stress detection [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1638675/full]
- 40. Pre-Symptomatic Disease Detection in the Vine, ... [https://www.mdpi.com/2073-4395/11/9/1682]
- 41. Hyperspectral Imaging for Presymptomatic Detection of ... [https://www.nature.com/articles/s41598-017-04501-2]
- 42. All-in-one machine learning framework for early detection ... [https://www.sciencedirect.com/science/article/pii/S2772375525008640]
- 43. Enhanced multiscale plant disease detection with the ... [https://www.nature.com/articles/s41598-025-89034-9]
- 44. Chemotaxonomy, an Efficient Tool for Medicinal Plant ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12299962/]
- 45. Exploring the chemotypic variability of Silybum marianum ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1584104/full]
- 46. Reconstructing Hyperspectral Images from RGB Images by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12468563/]
- 47. High-Throughput Imaging for Precision Breeding and ... [https://www.jales.org/articles/xml/DQxK/]
- 48. Crop Monitoring with Hyperspectral Imaging for Precision ... [https://www.leher.ag/blog/crop-monitoring-hyperspectral-imaging-precision-agriculture]
- 49. Prediction of harvest-related traits in barley using high- ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1686506/full]
- 50. Advancing radiation-induced mutant screening through high ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-025-01367-8]
- 51. Hyperspectral Imaging [https://arxiv.org/html/2508.08107v1]
- 52. Dimensionality reduction in hyperspectral imaging using ... [https://www.nature.com/articles/s41598-025-21738-4]
- 53. Dimensionality Reduction of Hyperspectral Imagery Based ... [https://www.sciencedirect.com/science/article/abs/pii/S0016003225003552]
- 54. Dimension Reduction and Entropy-Based Hyperspectral ... [https://www.frontiersin.org/journals/remote-sensing/articles/10.3389/frsen.2025.1702834/full]
- 55. Addressing data size challenges of hyperspectral imaging ... [https://www.spiedigitallibrary.org/conference-proceedings-of-spie/13464/1346409/Addressing-data-size-challenges-of-hyperspectral-imaging-through-event-based/10.1117/12.3053794.full]
- 56. Raspberry plant stress detection using hyperspectral imaging [https://pmc.ncbi.nlm.nih.gov/articles/PMC10020142/]
- 57. Enhancing Hyperspectral Image Prediction with ... [https://arxiv.org/html/2410.07790v1]
- 58. AI Deep Learning Advances Hyperspectral Imaging for ... [https://www.spectroscopyonline.com/view/ai-deep-learning-advances-hyperspectral-imaging-for-earth-observation]
- 59. MLVI-CNN: a hyperspectral stress detection framework ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1631928/full]
- 60. Hyperspectral and Multispectral Remote Sensing Image ... [https://www.mdpi.com/2072-4292/17/12/1973]
- 61. Hyperspectral imaging: a novel approach for plant root ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-018-0352-1]
- 62. A survey on computational spectral reconstruction methods ... [https://www.nature.com/articles/s41598-022-16223-1]
- 63. Recent Advancements in Hyperspectral Image Reconstruction ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12158250/]
- 64. Spectral Reconstruction from RGB Imagery: A Potential ... [https://www.mdpi.com/1424-8220/24/11/3666]
- 65. Computational spectral imaging reconstruction via a spatial ... [https://opg.optica.org/josaa/abstract.cfm?uri=josaa-42-2-139]
- 66. Hyperspectral imaging pushes weed science a step further [https://uaex.uada.edu/media-resources/news/2025/june/06-16-2025-ark-hyperspectral-weed-research.aspx]
- 67. Hyperspectral Imaging Meets High-Throughput Plant ... [https://www.specim.com/plant-phenotyping/]
- 68. Comparison of UAV RGB Imagery and Hyperspectral ... [https://www.mdpi.com/2072-4292/14/15/3811]
- 69. High-throughput phenotyping using digital and hyperspectral ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7382386/]
- 70. Hyperspectral Sensors and Autonomous Driving [https://arxiv.org/html/2508.19905v1]
- 71. Comparing Unmanned Aerial Multispectral and ... [https://www.mdpi.com/2504-446X/7/7/410]
- 72. Top Drones for Building & Structural Inspections in 2025 [https://advexure.com/blogs/news/top-drones-for-building-structural-inspections-in-2025?srsltid=AfmBOopFX3w_PFE7mGWhvMp_uds58c8Q1rvRxRz99KkKOUVdphTNRVRm]
- 73. DroneScape: Collection of RGB and 10-Band Multispectral ... [https://www.protocols.io/view/dronescape-collection-of-rgb-and-10-band-multispec-dyia7uae.html]
- 74. Reconstructed hyperspectral imaging for in-situ nutrient ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12375653/]
- 75. Hyperspectral image analysis for classification of multiple ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-025-01461-x]
- 76. Hyperspectral imaging to characterize the vegetative tissue ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1515998/full]
- 77. A deep learning model based on RGB and hyperspectral ... [https://www.sciencedirect.com/science/article/pii/S2772375525000516]
- 78. 9 Best Practices for Data Visualization That Work in 2025 [https://www.widgetly.co/blog/best-practices-for-data-visualization]
- 79. Multispectral Vs. Hyperspectral Imaging: Differences And ... [https://eos.com/blog/multispectral-vs-hyperspectral-imaging/]
- 80. Multispectral Imaging & Hyperspectral Imaging [https://va-imaging.com/blogs/machine-vision-knowledge-center/multispectral-imaging-hyperspectral-imaging]
- 81. Hyperspectral vs multispectral cameras [https://www.specim.com/technology/hyperspectral-vs-multispectral-cameras/]
- 82. Hyperspectral and Multispectral Imaging [https://www.edmundoptics.com/knowledge-center/application-notes/imaging/hyperspectral-and-multispectral-imaging/?srsltid=AfmBOooPSPtM3Rv53FVPcEnSYhkuPUy_a0q9gU1G6_7Ynh71z1PCx4Kn]
- 83. Statistic and Network Features of RGB and Hyperspectral ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10137991/]
- 84. Drone hyperspectral imaging and artificial intelligence for ... [https://www.nature.com/articles/s41598-025-11535-4]
- 85. Performance comparison of RGB and multispectral ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9355326/]
- 86. GreenLeafVI: A FIJI Plugin for High‐Throughput Analysis of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12535850/]
- 87. Non-destructive estimation of SPAD and biomass in ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1640779/full]
- 88. A multi-spectral and hyperspectral image dataset for ... [https://www.nature.com/articles/s41598-025-85714-8]
- 89. New imaging platform tracks plant stress in real time [https://e3.eurekalert.org/news-releases/1098266]
- 90. Publication : USDA ARS [https://www.ars.usda.gov/research/publications/publication/?seqNo115=419295]
- 91. Integration of Environmental Metadata with Image-Based ... [https://papers.ssrn.com/sol3/papers.cfm?abstract_id=5350101]
- 92. Modern Trends and Recent Applications of Hyperspectral ... [https://www.mdpi.com/2227-7080/13/5/170]
- 93. Spectral reflectance imaging with dual-illumination and ... [https://opg.optica.org/oe/fulltext.cfm?uri=oe-33-21-44191]
- 94. Comparing the performance of vegetation indices for ... [https://www.sciencedirect.com/science/article/pii/S1574954125000329]
- 95. Variation in Assessment of Leaf Pigment Content from ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12608226/]
- 96. Application of Open-Source Digital Resources for 3D ... [https://pubmed.ncbi.nlm.nih.gov/40958711/]
- 97. RGB Imaging as a Tool for Remote Sensing of ... [https://www.mdpi.com/2223-7747/13/9/1262]
- 98. A review of hyperspectral image analysis techniques for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8983301/]
- 99. Everyday Cameras Transformed Into Crop Analysis Tools [https://www.technologynetworks.com/analysis/news/everyday-cameras-transformed-into-crop-analysis-tools-407218]