Beyond SpCas9: A Comparative Guide to CRISPR-Cas9 Variants for Precision Plant Genome Editing
This article provides a comprehensive comparative analysis of CRISPR-Cas9 variants and their applications in plant genome editing.
Beyond SpCas9: A Comparative Guide to CRISPR-Cas9 Variants for Precision Plant Genome Editing
Abstract
This article provides a comprehensive comparative analysis of CRISPR-Cas9 variants and their applications in plant genome editing. Tailored for researchers and plant biotechnologists, it explores the foundational characteristics of natural and engineered Cas9 proteins, their specific methodological applications in enhancing crop traits, and strategies for optimizing editing efficiency while minimizing off-target effects. The content synthesizes current data on variant performance, delivery methods, and validation techniques, offering a practical guide for selecting the optimal CRISPR tools for plant engineering projects aimed at improving agricultural sustainability and food security.
The CRISPR-Cas9 Toolkit: From Natural Diversity to Engineered Precision
In the rapidly evolving field of plant genome editing, the CRISPR system from Streptococcus pyogenes (SpCas9) has established itself as a foundational technology and versatile workhorse. Since its adaptation as a genome editing tool, SpCas9 has revolutionized plant biotechnology, enabling unprecedented precision in crop improvement efforts aimed at addressing global challenges such as food security and climate change [1]. While numerous CRISPR systems and variants have emerged in recent years, SpCas9 remains the most widely adopted and characterized platform in plant research applications.
This guide provides an objective comparison of SpCas9's performance against other genome editing technologies, presenting detailed experimental data and methodologies to help researchers select appropriate tools for their specific plant editing projects. By examining structural features, editing efficiencies, practical applications, and experimental considerations, we aim to provide a comprehensive resource for scientists engaged in plant genome engineering and crop improvement programs.
Structural Mechanisms and Technological Evolution
SpCas9 Architecture and Functional Domains
The molecular architecture of SpCas9 reveals the structural basis for its functionality as a programmable nuclease. SpCas9 is a multi-domain protein comprising seven structural domains: REC1, REC2, REC3, BH (bridge helix), Pi (PAM interaction), HNH, and RuvC [1]. These domains are organized into two primary lobes: the recognition lobe (REC) and the nuclease lobe (NUC). The REC lobe, consisting of REC1, REC2, and REC3 domains, facilitates sgRNA binding to target DNA, while the NUC lobe contains the catalytic centers for DNA cleavage [1].
The HNH and RuvC domains serve as the two catalytic centers responsible for DNA cleavage. The HNH domain cleaves the DNA strand complementary to the sgRNA, while the RuvC domain cleaves the non-targeted strand, resulting in a double-strand break (DSB) [1]. The Pi domain plays a critical role in recognizing and engaging the protospacer adjacent motif (PAM) sequence, a crucial step in target recognition [1]. The bridge helix (BH) serves as a structural connector between the REC and NUC lobes, mediating conformational transitions during Cas9 activation [1].
The SpCas9 system utilizes a single guide RNA (sgRNA), an engineered fusion of the naturally occurring crRNA and tracrRNA [1]. The sgRNA contains a 20-nucleotide spacer sequence at its 5' end that determines target specificity through complementary base pairing with the DNA protospacer. The PAM-proximal region (nucleotides 14-20), known as the "seed" sequence, exhibits minimal tolerance for mismatches, while the PAM-distal region (nucleotides 1-13) can accommodate some sequence variation [1].
Chronological Development of SpCas9-Based Editing Tools
The SpCas9 system has evolved significantly since its initial development, with successive innovations expanding its capabilities and applications in plant genome editing. Base editing and prime editing technologies represent major advancements that build upon the foundational SpCas9 architecture [1]. Base editors utilize catalytically impaired SpCas9 (nickase or dead Cas9) fused to deaminase enzymes, enabling direct conversion of one base pair to another without inducing double-strand breaks [1]. Prime editors further extend this capability by combining reverse transcriptase with SpCas9 nickase and a prime editing guide RNA (pegRNA), allowing for precise nucleotide substitutions, small insertions, and deletions without donor DNA templates [1].
Recent methodological innovations, including dual pegRNA strategies and site-specific integrases, have significantly expanded the potential of prime editing for precise gene insertions in plants [1]. The continued refinement of SpCas9-based tools, informed by growing understanding of DNA repair mechanisms in plants, continues to open new avenues for precise genome manipulation in crop species.
Comparative Performance Analysis of Genome Editing Technologies
Editing Efficiency Across Plant Species
The performance of SpCas9 has been systematically evaluated against alternative genome editing technologies across multiple plant species. Recent benchmarking studies provide quantitative data on editing efficiencies, enabling objective comparison between different platforms.
Table 1: Editing Efficiency Comparison Across CRISPR Systems in Plants
| Editing System | Plant Species | Target Gene | Editing Efficiency | Key Findings | Citation |
|---|---|---|---|---|---|
| SpCas9 | Nicotiana benthamiana | Multiple targets | 0.1% to >30% (protoplasts) | High variability based on sgRNA design | [2] |
| ttLbUV2 (Cas12a) | Arabidopsis thaliana | GL1 | 20.8% to 99.1% | High efficiency with minimal target bias | [3] |
| LbCas12a-RRVL | Arabidopsis thaliana | ECA3-1, GL2, TT4 | Higher than ttLbUV2 | Minimal practical differences | [3] |
| Cas12i3V1 | Arabidopsis thaliana | 4 of 6 targets | Relatively high efficiency | Promising new tool for plant editing | [3] |
| AsCas12f variants | Arabidopsis thaliana | Multiple targets | Poor or no detectable editing | Requires further optimization | [3] |
The data reveal that optimized Cas12a variants such as ttLbUV2 can achieve editing efficiencies exceeding 99% in some targets, comparable to high-performance SpCas9 systems [3]. However, SpCas9 maintains advantages in predictability and established protocols, with extensive datasets available to inform sgRNA design across diverse plant species. The performance of newer systems like Cas12i shows promise but requires further validation across a broader range of plant species.
Multiplex Editing Capabilities
A key advantage of SpCas9 in plant applications is its capacity for multiplex genome editing, enabling simultaneous modification of multiple genetic loci. Comparative studies demonstrate that SpCas9 efficiently processes tandemly arrayed sgRNAs, enabling coordinated editing of multiple targets [3].
In direct comparisons of multiplexing capabilities, optimized LbCas12a systems (ttLbUV2) demonstrated highly efficient simultaneous mutation of homologous genes CHLI1 and CHLI2 using a single crRNA, with efficiencies reaching 85.4% to 99.3% across different target pairs [3]. The study also confirmed that the order of crRNAs in the tandem array does not significantly affect editing efficiency, with 97.8% and 96.1% efficiency observed when crRNA order was swapped [3].
SpCas9 systems have demonstrated particular strength in large-scale multiplexed applications, as evidenced by a study in Salvia miltiorrhiza that established a high-efficiency editing platform enabling the creation of 1,664 homozygous or biallelic mutants [4]. The success was contingent on identifying optimal transformation conditions, including selection of the inbred line bh2-7 and specific Agrobacterium strains (K599 and C58C1) [4].
Structural Variant Generation
The capacity to generate structural variants represents an emerging application of genome editing technologies in plants. While early applications focused primarily on gene knockouts, recent advances enable more complex genome rearrangements including deletions, duplications, inversions, and translocations [4].
SpCas9 systems have been successfully employed to generate targeted deletions in plants, with efficiency influenced by factors including distance between target sites and specific genomic context. Newer CRISPR systems, particularly those utilizing Cas12a, offer alternative mechanisms for generating structural variations due to their distinct DNA cleavage patterns that produce sticky ends rather than the blunt ends characteristic of SpCas9 [3].
Table 2: Comparison of Editing Tool Characteristics for Plant Genome Engineering
| Characteristic | SpCas9 | Cas12a (Cpf1) | TALENs | ZFNs |
|---|---|---|---|---|
| PAM Requirement | NGG | TTTV | None | None |
| Protein Size | ~1368 aa | ~1300 aa | Large (~3000 aa) | ~1000 aa |
| Guide RNA | sgRNA (∼100 nt) | crRNA (∼42 nt) | Protein-DNA binding | Protein-DNA binding |
| Cleavage Type | Blunt ends | Sticky ends | Sticky ends | Sticky ends |
| Multiplexing | Good (tandem sgRNAs) | Excellent (crRNA arrays) | Limited | Limited |
| Ease of Design | Simple | Simple | Complex | Very complex |
| Development Time | Days | Days | Weeks | Months |
| Off-target Effects | Moderate | Lower | Low | Moderate |
| Delivery Challenge | Moderate (size) | Moderate (size) | High (size) | Moderate |
Advanced Applications and Specialized Uses
CRISPR Activation and Interference Systems
The development of catalytically inactive SpCas9 (dCas9) has enabled the creation of powerful transcriptional control systems that modulate gene expression without altering DNA sequences. CRISPR activation (CRISPRa) systems employ dCas9 fused to transcriptional activators to achieve targeted gene upregulation [5].
In plant systems, CRISPRa has been successfully applied to enhance disease resistance through endogenous gene activation. For example, in tomato, CRISPRa-mediated upregulation of SlPR-1 enhanced defense against Clavibacter michiganensis infection [5]. Similarly, epigenetic reprogramming of SlWRKY29 in Micro-Tom tomato established a transcriptionally permissive chromatin state that enhanced somatic embryo induction and maturation [5].
A recently developed CRISPR-dCas9 toolkit fused with a stress-responsive membrane-bound NAC domain enables heat-inducible, reversible gene regulation in solanaceous plants [4]. This system demonstrates the potential for conditional control, where dCas9 relocates to the nucleus upon heat stress, activating or repressing target genes to enhance traits like tomato resistance to bacterial specks and improved heat tolerance [4].
Virus-Mediated Delivery Systems
Innovative delivery approaches represent an active area of development for SpCas9 systems in plants. Virus-induced genome editing (VIGE) systems have been engineered to deliver CRISPR components efficiently without stable transformation [4].
The tobacco rattle virus (TRV) has been successfully engineered to deliver compact editing systems, including the TnpB enzyme ISYmu1 and guide RNA, into Arabidopsis thaliana, enabling transgene-free editing in a single step [4]. Similarly, heritable genome editing in tomatoes has been achieved using a TRV-based VIGE system delivering mobile RNA-fused gRNAs to SpCas9-expressing lines, with heritable editing rates reaching up to 100% under optimized conditions including reduced light intensity [4].
These virus-mediated delivery strategies address a significant bottleneck in plant genome editing by enabling genotype-independent editing without the need for tissue culture, expanding the range of plant species accessible to genetic improvement [4].
Experimental Design and Methodological Considerations
Editing Efficiency Quantification Methods
Accurate detection and quantification of editing outcomes is crucial for evaluating SpCas9 performance in plant systems. Recent benchmarking studies have systematically compared methods for quantifying genome editing efficiency [2].
Table 3: Comparison of Methods for Quantifying CRISPR Edits in Plants
| Method | Sensitivity | Accuracy | Cost | Throughput | Best Applications |
|---|---|---|---|---|---|
| AmpSeq | Very High | Very High | High | Medium | Gold standard, heterogeneous populations |
| PCR-RFLP | Low to Medium | Medium | Low | High | Rapid screening, high-efficiency edits |
| T7E1 | Low to Medium | Medium | Low | High | Preliminary efficiency assessment |
| Sanger + ICE/TIDE | Medium | Medium | Medium | Medium | Low-frequency edits, single targets |
| PCR-CE/IDAA | High | High | Medium | High | Multiplex editing, large indels |
| ddPCR | High | High | Medium | Medium | Specific variant detection |
Targeted amplicon sequencing (AmpSeq) is widely considered the gold standard due to its sensitivity, accuracy, and reliability in detecting editing events in highly heterogeneous plant populations [2]. However, techniques such as PCR-capillary electrophoresis/InDel detection by amplicon analysis (PCR-CE/IDAA) and droplet digital PCR (ddPCR) also demonstrate high accuracy when benchmarked against AmpSeq, offering alternatives that balance cost, throughput, and sensitivity [2].
For SpCas9 editing quantification, factors including plant ploidy, sequence variation between homeologs, and editing efficiency should inform method selection. Polyploid species present particular challenges for accurate edit quantification due to the presence of multiple gene copies that may be edited at different frequencies [2].
Research Reagent Solutions
Successful implementation of SpCas9 genome editing in plants requires carefully selected molecular tools and delivery systems. The following essential reagents represent critical components for establishing an effective plant editing pipeline.
Table 4: Essential Research Reagents for SpCas9 Plant Genome Editing
| Reagent Category | Specific Examples | Function | Considerations |
|---|---|---|---|
| Cas9 Expression System | pIZZA-BYR-SpCas9 [2] | High-level Cas9 expression | Utilizes geminiviral replicon for enhanced expression |
| sgRNA Expression | pBYR2eFa-U6-sgRNA [2] | sgRNA transcription | Arabidopsis U6-26 promoter for Pol III-driven expression |
| Delivery Vector | Dual geminiviral replicon (GVR) system [2] | Component delivery | Based on Bean yellow dwarf virus (BeYDV) |
| Editing Detection | AmpSeq, PCR-CE/IDAA, ddPCR [2] | Quantifying editing efficiency | Method choice depends on sensitivity requirements |
| Plant Transformation | Agrobacterium strains K599, C58C1 [4] | DNA delivery | Strain selection affects efficiency in different species |
| Regeneration Promoters | WUS, STM, IPT [4] | Enhanced plant regeneration | Co-delivered with editing components |
Experimental Workflow for Plant Genome Editing
A standardized workflow for SpCas9-mediated plant genome editing ensures consistent results and enables reliable comparison across experiments. The following diagram illustrates a generalized approach that can be adapted for specific plant species and experimental goals.
SpCas9 continues to serve as the versatile workhorse of plant genome editing, offering a balance of efficiency, reliability, and well-characterized performance across diverse plant species. While emerging technologies like optimized Cas12a variants demonstrate competitive editing efficiencies in specific applications, SpCas9 maintains advantages in protocol standardization, extensive validation data, and versatility through its various engineered derivatives.
The future of SpCas9 in plant genome editing will likely focus on enhanced specificity variants, expanded PAM recognition, and integration with other technologies such as recombinases and transposons for more complex genetic engineering applications. As the field progresses, SpCas9-based systems will continue to evolve, maintaining their central position in the plant biotechnology toolkit while providing a robust platform for addressing global agricultural challenges.
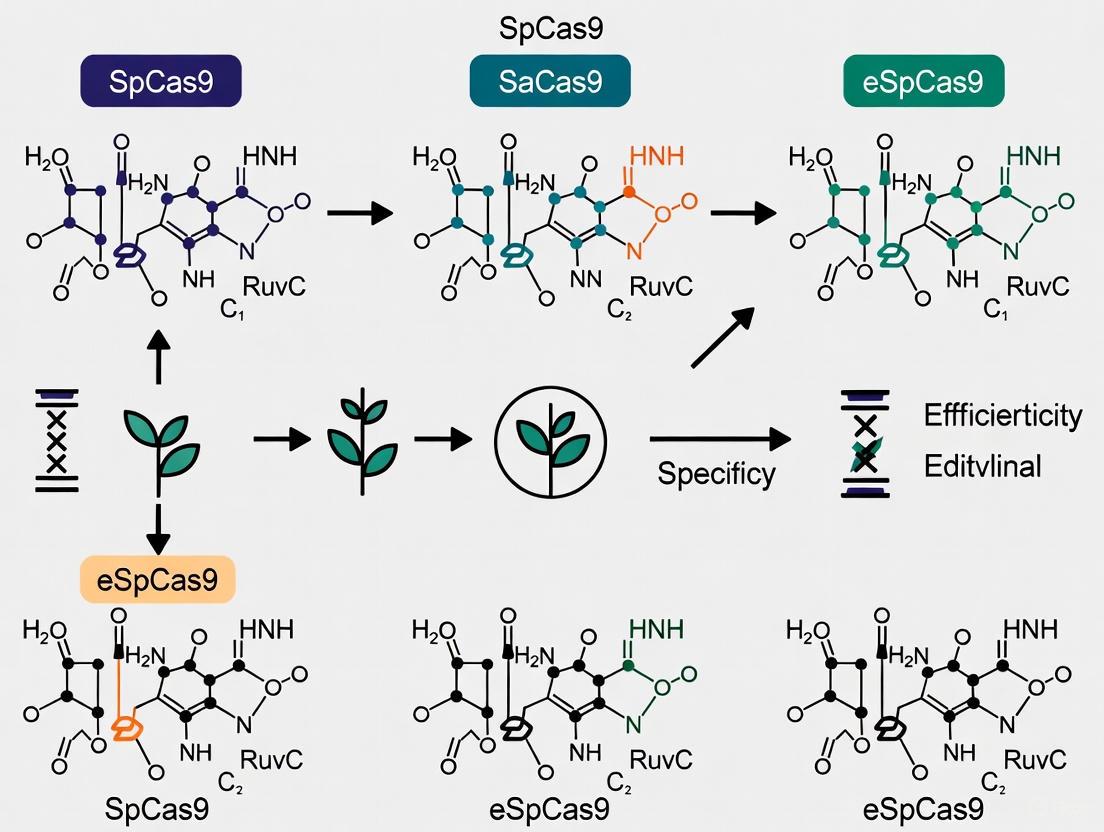
The CRISPR-Cas9 system, derived from microbial adaptive immune systems, has revolutionized genetic engineering in plant research. While the Cas9 from Streptococcus pyogenes (SpCas9) is widely used, its limitations regarding size and protospacer adjacent motif (PAM) requirements have driven the exploration of natural Cas9 orthologs. Staphylococcus aureus* Cas9 (SaCas9), Streptococcus thermophilus Cas9 (StCas9), and Neisseria meningitidis Cas9 (NmCas9) offer distinct advantages in terms of compact size, unique PAM recognition, and editing precision. This guide provides a comparative analysis of these three natural variants to inform their application in plant editing research.
At a Glance: Comparative Profiles of Natural Cas9 Variants
The table below summarizes the core characteristics of SaCas9, StCas9, and NmCas9 to help researchers make an initial selection based on common experimental needs.
| Feature | SaCas9 | StCas9 | NmCas9 |
|---|---|---|---|
| Origin | Staphylococcus aureus | Streptococcus thermophilus | Neisseria meningitidis |
| Size (aa) | 1,053 [6] | 1,121 [7] | 1,082 [7] |
| PAM Sequence | 5'-NNGRRT-3' or 5'-NNGRR(N)-3' [6] | 5'-NNAGAAW-3' [7] | 5'-NNNNGATT-3' [7] |
| Guide RNA | sgRNA [6] | crRNA & tracrRNA [7] | sgRNA [7] |
| Key Advantage | Small size ideal for viral delivery [6] | High specificity; targets complex PAMs [8] | Long PAM for enhanced specificity [7] |
| Plant Applications | Tobacco, potato, rice [6] | Cereal crops [7] | Cereal crops [7] |
Performance and Experimental Data
Quantitative data on editing efficiency and specificity are critical for selecting the appropriate Cas9 variant.
Table: Documented Performance Metrics in Plant and Human Cells
| Variant | Editing Efficiency | Specificity (Off-Target Effects) | Experimental Context |
|---|---|---|---|
| SaCas9 | High indel generation efficiency in plants [6] | Engineered high-fidelity variant (SaCas9-HF) available [6] | Gene editing in tobacco, potato, and rice; study of plant-pathogen interactions [6] |
| StCas9 | Used successfully in cereal crops [7] [8] | Not specifically reported; recognized for high specificity [8] | Genome editing in cereal crops [7] |
| NmCas9 | Used successfully in cereal crops [7] | Not specifically reported | Genome editing in cereal crops [7] |
Experimental Workflow for Cas9 Application in Plants
The following diagram illustrates a generalizable protocol for implementing CRISPR-Cas9 genome editing in plants, from design to validation. This workflow integrates key steps from established plant genomics protocols [7].
CRISPR-Cas9 Plant Editing Workflow: This diagram outlines the key stages of a genome editing experiment in plants, from initial guide RNA design to final validation of edited lines [7].
The Scientist's Toolkit: Essential Research Reagents
Successful genome editing requires a suite of reliable reagents and tools. The table below lists essential components for experiments utilizing natural Cas9 variants.
| Reagent/Tool | Function | Application Notes |
|---|---|---|
| Binary Plasmid Vector | Carries expression cassettes for Cas9 and gRNA into plant cells [7] | Must be compatible with the chosen Cas9 variant and plant transformation method. |
| Cas9 Nuclease | The effector protein that creates double-stranded breaks in target DNA [6] | Available as purified protein for RNP delivery or as a coding sequence (DNA/mRNA). |
| Guide RNA (gRNA) | A synthetic RNA that directs Cas9 to the specific target genomic locus [7] | Design is critical for efficiency and specificity; sequence must be validated. |
| Agrobacterium tumefaciens | A common bacterium used to deliver genetic material into plant cells [7] | Used for stable transformation; requires a binary vector system. |
| Viral Delivery Vector | Engineered virus (e.g., Tobacco Rattle Virus) to deliver editing machinery [9] | Useful for difficult-to-transform species; can achieve systemic editing. |
| Web-Based gRNA Design Tools | In silico tools for selecting optimal gRNA targets and predicting off-target sites [7] | Tools like CRISPR-P 2.0, Cas-Designer, and CHOPCHOP support major crops. |
Selection Guide: Matching Cas9 Variants to Research Goals
The final choice of a Cas9 variant depends heavily on the specific requirements of the research project. The following diagram provides a decision-making framework.
Cas9 Variant Selection Guide: This flowchart assists in selecting the most suitable natural Cas9 variant based on delivery method and target site PAM sequence [7] [6].
The exploration of natural Cas9 diversity beyond SpCas9 provides plant researchers with a powerful and expanded toolkit. SaCas9 stands out for applications where a small size is critical for delivery. StCas9 and NmCas9 offer alternative PAM recognition, increasing the range of genomic sites that can be targeted. The choice among them should be guided by the specific target genomic sequence, the delivery method constraints, and the desired balance between efficiency and specificity. As the field progresses, the integration of these natural variants, along with newly engineered editors and AI-designed nucleases [10], will continue to push the boundaries of precision genome editing in plant research.
The CRISPR-Cas9 system has revolutionized plant genome editing, yet its targeting capacity is constrained by a critical molecular requirement: the protospacer adjacent motif (PAM). This short DNA sequence flanking a guide RNA-programmed target site is essential for Cas9 recognition and activation. The widely used Streptococcus pyogenes Cas9 (SpCas9) primarily recognizes a 5'-NGG-3' PAM, severely limiting the sequence space accessible for editing. With only 1 in 16 random DNA sequences containing an NGG PAM, researchers have engineered novel Cas9 variants with relaxed PAM requirements to dramatically expand the targeting potential for plant research and crop improvement. This guide provides a comprehensive comparison of these engineered Cas variants, empowering researchers to select optimal tools for their specific applications.
Comparative Analysis of Cas9 Variants
The development of PAM-flexible Cas9 variants has significantly expanded the genomic target space for plant genome editing. The table below summarizes the key characteristics of major Cas9 variants used in plant research.
Table 1: Comparison of Cas9 Variants for Plant Genome Editing
| Cas9 Variant | Original PAM Requirement | Engineered PAM Compatibility | Key Features & Applications in Plants | Reported Editing Efficiency |
|---|---|---|---|---|
| SpCas9 (Wild-type) | 5'-NGG-3' | N/A | Widely adopted; established workflows; reliable performance | Varies significantly by target site and organism [2] |
| xCas9 | 5'-NGG-3' | NG, GAA, GAT, GAG | Expanded PAM recognition in rice; used with tRNA-esgRNA systems for enhanced efficiency | Efficient mutations at GAD PAM sites in rice [11] |
| SpRY | 5'-NGG-3' | NRN > NYN (Near-PAMless) | Broadest PAM compatibility; engineered from SpCas9 PID mutations | Robust editing across diverse PAMs; slightly reduced efficiency compared to SpCas9 [12] |
| SpRYc (Chimeric) | N/A | NRN & NYN (Comprehensive NNN coverage) | Combines SpRY PID with Sc++ N-terminus; maintains high specificity | Competes with SpRY on NYN loci; superior to SpCas9 on non-NGG sites [12] |
| Sc++ | 5'-NGG-3' | 5'-NNG-3' | Positive-charged loop enables relaxed second base requirement; high fidelity | Efficient NNG editing with reduced off-target effects [12] |
Experimental Data and Performance Metrics
Targeting Range and Efficiency
The engineering of PAM-flexible Cas9 variants has dramatically increased the theoretical targeting space in plant genomes. While wild-type SpCas9 can target approximately 9.6% of genomic sites, xCas9 expands this to 36.5%, and SpRY achieves near-complete coverage at 98.5% of sites [12]. In practical applications, xCas9 has demonstrated efficient gene mutations at GAD (where D is A, T, or G) PAM sites in rice plants, with the tRNA-esgRNA system significantly enhancing editing efficiency [11].
SpRYc, a chimeric enzyme combining properties of SpRY and Sc++, exhibits particularly broad PAM compatibility while maintaining editing precision. Experimental data comparing SpRYc to SpCas9 and SpRY across sixteen genomic loci representing every two-base PAM combination demonstrated that SpRYc generates modifications at all tested genomic loci, performing comparably to SpRY and more optimally on select 5'-NYN-3' loci [12].
Precision and Specificity
Beyond expanding targeting range, engineered Cas variants must maintain high specificity to minimize off-target effects. GUIDE-Seq analysis reveals that SpRYc exhibits nearly four-fold lower off-target activity with VEGFA-targeting guide RNA and two-fold lower off-target activity at EMX1 sites compared to SpRY [12]. This enhanced specificity is attributed to Sc++'s intrinsic high-fidelity characteristics inherited by the chimeric enzyme.
Experimental Protocols for PAM-Flexible Editing
Transient Expression in Plant Systems
Transient expression in Nicotiana benthamiana leaves provides a robust alternative to stable transformation for rapidly testing CRISPR components. A modified dual geminiviral replicon (GVR) system based on the Bean yellow dwarf virus enables efficient co-expression of Cas9 variants and sgRNAs [2]. The protocol involves:
- Vector Construction: Clone sgRNAs into appropriate expression vectors (e.g., pBYR2eFa-U6-sgRNA for geminiviral systems)
- Agroinfiltration: Co-infiltrate Nicotiana benthamiana leaves with Agrobacterium strains carrying Cas9 and sgRNA vectors
- Genomic DNA Extraction: Harvest infiltrated tissue 7 days post-infiltration for DNA analysis
- Editing Efficiency Quantification: Utilize appropriate detection methods (detailed in subsequent sections)
Stable Transformation in Rice
For stable integration in monocot systems like rice, the following protocol has proven effective for xCas9-mediated editing [11]:
- Vector Assembly: Construct tRNA-esgRNA arrays with multiple targets under control of U3/U6 promoters
- Rice Transformation: Introduce constructs into embryogenic calli via Agrobacterium tumefaciens strain EHA105
- Selection & Regeneration: Culture on hygromycin-containing media for 4 weeks, then transfer to regeneration media
- Mutant Identification: Screen T0 plants via PCR and sequencing; use tools like dsDecode for mutation analysis
Engineering Strategy for PAM-Flexible Cas9 Variants
Detection and Quantification Methods
Accurate detection and quantification of editing outcomes is crucial for evaluating PAM-flexible Cas9 performance. The table below compares major detection methods used in plant editing research.
Table 2: Methods for Detecting and Quantifying CRISPR Edits in Plants
| Method | Detection Principle | Sensitivity | Advantages | Limitations |
|---|---|---|---|---|
| Amplicon Sequencing (AmpSeq) | High-throughput sequencing of target loci | Highest sensitivity (<0.1%) | Considered "gold standard"; comprehensive mutation profiling | Higher cost; longer turnaround; specialized facilities needed [2] |
| PCR-CE/IDAA | PCR amplification with capillary electrophoresis | High sensitivity | Accurate; benchmarked well against AmpSeq | Limited to smaller indels; specific equipment required [2] |
| ddPCR | Droplet digital PCR with fluorescent probes | High sensitivity (absolute quantification) | Precise efficiency measurement; high throughput | Requires specific probe design; limited multiplexing [2] |
| T7E1 Assay | Enzyme cleavage of mismatched heteroduplexes | Moderate sensitivity | Low cost; simple protocol | Semi-quantitative; lower accuracy [2] |
| PCR-RFLP | Restriction fragment length polymorphism | Moderate sensitivity | Inexpensive; accessible | Dependent on native restriction sites; less quantitative [2] |
| Qualitative/Quantitative PCR | Primer/probe-based detection of Cas transgenes | 0.1% (qualitative), 14 copies (qPCR) | Specific; sensitive; regulatory applications | Detects presence rather than editing efficiency [13] |
Workflow for Detection and Quantification of CRISPR Edits in Plants
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of PAM-flexible editing requires specific reagents and vectors optimized for plant systems. The table below details essential research solutions for plant genome editing workflows.
Table 3: Essential Research Reagents for Plant CRISPR Editing
| Reagent / Solution | Function & Application | Examples & Specifications |
|---|---|---|
| Cas9 Expression Vectors | Delivery of Cas9 variants to plant cells | pIZZA-BYR-SpCas9 (geminiviral), pMDC32 (binary), pRGEB (modular) |
| sgRNA Cloning Systems | Guide RNA expression cassettes | pYPQ vectors (Golden Gate), pBYR2eFa-U6-sgRNA, pCAMBIA |
| tRNA-esgRNA Systems | Enhanced sgRNA expression for challenging PAMs | xCas9 tRNA-esgRNA arrays for GA PAM editing in rice [11] |
| Plant Codon Optimization | Enhanced expression in plant systems | Rice-optimized xCas9; N. benthamiana-optimized SpRY |
| Detection Primers/Probes | Specific amplification of target loci | Cpf1-specific primers for qualitative PCR (LOD: 0.1%) and qPCR (LOD: 14 copies) [13] |
| Plant Transformation Vectors | Agrobacterium-mediated delivery | pMDC32Cas9NktPDS (banana), pCAMBIA (rice), pGreen (tomato) |
The engineering of PAM-flexible Cas9 variants represents a transformative advancement in plant genome editing, dramatically expanding the accessible genomic target space. While each variant offers distinct advantages, researchers must consider the tradeoffs between PAM flexibility, editing efficiency, and specificity when selecting tools for specific applications. xCas9 provides reliable performance with moderately expanded PAM recognition, while SpRY and SpRYc offer near-complete targeting freedom with some efficiency tradeoffs. As detection methods continue to improve in sensitivity and accessibility, and regulatory frameworks evolve to address gene-edited crops, these PAM-flexible systems will play an increasingly vital role in accelerating crop improvement and fundamental plant research.
The advent of CRISPR-Cas9 technology has revolutionized genetic engineering, offering unprecedented precision in manipulating genomes for research and therapeutic applications. A critical hurdle in deploying this technology, particularly for in vivo applications, is the efficient delivery of CRISPR components into target cells. Viral vectors, especially adeno-associated viruses (AAVs), have emerged as leading delivery vehicles due to their favorable safety profile and high transduction efficiency. However, their limited packaging capacity of approximately 4.7 kilobases (kb) poses a significant challenge for delivering the commonly used Streptococcus pyogenes Cas9 (SpCas9), which alone spans about 4.2 kb, leaving insufficient space for essential regulatory elements and guide RNAs. This review provides a comparative analysis of compact Cas9 variants and novel CRISPR systems developed to overcome this packaging limitation, presenting key experimental data and protocols to guide researchers in selecting the optimal nuclease for viral vector-based delivery in plant and biomedical research.
The Viral Vector Packaging Challenge and Compact CRISPR Systems
Recombinant AAV (rAAV) vectors are among the most promising platforms for in vivo CRISPR delivery due to their non-pathogenic nature, low immunogenicity, and sustained transgene expression [14]. However, their <4.7 kb packaging capacity necessitates the use of smaller Cas orthologs or innovative splitting strategies [14] [15]. To address this, researchers have explored several avenues: identifying naturally compact Cas proteins, engineering smaller variants, and developing dual-vector systems. The most common strategy involves using Cas9 orthologs from other bacteria that are inherently smaller than SpCas9, enabling packaging of the entire CRISPR system—nuclease, guide RNA, and promoters—within a single viral vector [14] [16]. This "all-in-one" configuration simplifies production and ensures coordinated delivery to the same cell.
Table 1: Compact Cas Variants for Viral Vector Delivery
| Cas Protein | Species of Origin | Size (amino acids) | PAM Sequence | Packaging Compatibility | Reported Editing Efficiency |
|---|---|---|---|---|---|
| SpCas9 | Streptococcus pyogenes | ~1368 | NGG | Requires dual-vector or split systems | High (benchmark) |
| SaCas9 | Staphylococcus aureus | ~1053 | NNGRRT | Single AAV vector | Up to 40% in mouse liver [14] |
| CjCas9 | Campylobacter jejuni | ~984 | NNNNRYAC | Single AAV vector | Efficient in retinal studies [14] |
| Nme2Cas9 | Neisseria meningitidis | ~1082 | NNNNGATT | Single AAV vector | Effective for base editing [14] |
| Cas12f (Cas14) | Uncultured Archaea | ~400-700 | T-rich | Single AAV with ample space | Demonstrated in preclinical models |
| IscB | Putative Cas9 ancestor | ~400-600 | Varies by variant | Single AAV with ample space | 15% editing in mouse liver [14] |
| TnpB | Putative Cas9 ancestor | ~400-600 | Varies by variant | Single AAV with ample space | Up to 56% editing in mouse liver [14] |
For applications requiring the full-length SpCas9, dual rAAV vector systems have been developed where the Cas9 nuclease and its gRNA are delivered on separate vectors [14]. While this resolves the size constraint, it often requires high viral doses and raises concerns about co-transduction efficiency and potential immune responses [15]. More recently, putative ancestors of modern Cas proteins, such as IscB and TnpB, have been identified as ultra-compact tools for genome editing [14]. Their small molecular size offers enhanced compatibility with AAV packaging constraints and may present a reduced immunogenicity profile, making them promising candidates for future therapeutic applications [14].
Comparative Performance Data of Compact Cas Variants
Editing Efficiency and Specificity
Compact Cas variants have demonstrated robust editing efficiencies in various preclinical models. For instance, subretinal delivery of rAAV8 vectors encoding the engineered compact variant CasMINI_v3.1/ge4.1 achieved transduction efficiencies exceeding 70% in GFP+ retinal cells of a retinitis pigmentosa mouse model [14]. This treatment led to a significant improvement in cone photoreceptor function within one month post-injection [14]. Similarly, systemic delivery of rAAV9 vectors encoding Nme2-ABE8e, a compact base editor, successfully corrected the Fah mutation in a mouse model of hereditary tyrosinemia type 1 (HT1) [14]. Although the overall editing efficiency was relatively low (0.34%), the treatment restored 6.5% FAH-positive hepatocytes, exceeding the therapeutic threshold and demonstrating clinical potential [14].
The hypercompact IscB and TnpB systems have shown particularly promising results. In a tyrosinemia mouse model, systemic delivery of rAAV8 vectors encoding EnIscB–ωRNA-based ABE achieved 15% editing efficiency and restored Fah expression [14]. In a separate study, intramuscular injection of an rAAV9 vector encoding IscB.m16*-CBE resulted in 30% exon skipping and recovery of dystrophin expression in a humanized mouse model of Duchenne muscular dystrophy (hDMD) [14]. Furthermore, a self-complementary AAV9 vector encoding TnpB targeting Pcsk9 achieved up to 56% editing in the liver and significantly reduced blood cholesterol levels after systemic injection [14].
Table 2: Experimental Performance of Compact Cas Systems in Preclinical Models
| Cas System | Delivery Method | Disease Model | Target Gene | Editing Efficiency | Functional Outcome |
|---|---|---|---|---|---|
| CasMINI_v3.1/ge4.1 | rAAV8, subretinal | RhoP23H/+ mouse (Retinitis Pigmentosa) | Nr2e3 | >70% transduction | Improved cone function |
| Nme2-ABE8e | rAAV9, systemic | FahPM/PM mouse (Hereditary Tyrosinemia) | Fah | 0.34% (6.5% FAH+ hepatocytes) | Metabolic correction |
| EnIscB-ωRNA ABE | rAAV8, systemic | Fah mouse (Tyrosinemia) | Fah | 15% | Fah expression restored |
| IscB.m16*-CBE | rAAV9, intramuscular | hDMD mouse (Duchenne Muscular Dystrophy) | Dystrophin exon | 30% exon skipping | Dystrophin recovery |
| TnpB | scAAV9, systemic | Wild-type mouse | Pcsk9 | Up to 56% | Reduced cholesterol |
Specificity and Off-Target Considerations
While compact Cas variants solve the delivery problem, their specificity profiles vary. The targeting accuracy of CRISPR systems is influenced by both the protospacer adjacent motif (PAM) sequence requirement and the base pairing between the guide RNA and target DNA [17]. For example, SaCas9 recognizes the PAM sequence "NNGRRT," which is longer and less frequent in genomes than the "NGG" PAM of SpCas9, potentially reducing off-target sites but also narrowing the targetable range [17]. The seed region—the PAM-proximal 10–12 nucleotides of the sgRNA—is particularly crucial for specific recognition and cleavage [17]. Mismatches in this region can significantly reduce off-target effects.
To mitigate off-target activity, several strategies can be employed with compact Cas variants, including using truncated sgRNAs, paired nickases, and high-fidelity engineered variants [17]. These approaches enhance specificity while maintaining the packaging advantages of smaller Cas proteins.
Experimental Protocols for Evaluating Cas Variants
Protocol 1: In Vivo Efficacy Assessment of AAV-Delivered Compact Cas Systems
Objective: To evaluate the genome editing efficiency and functional recovery of a compact Cas variant delivered via AAV in a mouse disease model.
Materials:
- Animal Model: Disease-specific mouse model (e.g., FahPM/PM for tyrosinemia)
- Viral Vector: rAAV encoding compact Cas variant and gRNA (e.g., rAAV9-Nme2-ABE8e)
- Control: rAAV encoding a non-targeting gRNA
- Reagents: DNA extraction kit, PCR reagents, NGS library prep kit, tissue staining antibodies
Methodology:
- Vector Administration: Administer rAAV via appropriate route (e.g., systemic via tail vein for liver targets, subretinal for retinal targets) at a standardized titer (e.g., 1×10^12 vg/mouse).
- Tissue Collection: Harvest target tissues (e.g., liver, retina) at predetermined timepoints (e.g., 2, 4, 8 weeks post-injection).
- DNA Extraction: Isolate genomic DNA from homogenized tissues using a commercial kit.
- Editing Efficiency Analysis:
- Amplify the target genomic region by PCR.
- Perform next-generation sequencing (NGS) on the amplified products.
- Analyze sequencing data using computational tools (e.g., CRISPResso2) to quantify insertion/deletion (indel) frequencies or base conversion rates.
- Functional Assessment:
- For metabolic diseases: Perform immunohistochemistry on tissue sections to detect protein restoration (e.g., FAH staining in hepatocytes).
- For retinal diseases: Conduct electroretinography (ERG) to measure photoreceptor function.
- For muscular diseases: Analyze dystrophin expression by Western blot and muscle strength.
This protocol was adapted from studies demonstrating the therapeutic efficacy of Nme2-ABE8e in hereditary tyrosinemia and CasMINI in retinitis pigmentosa [14].
Protocol 2: Off-Target Analysis Using Digenome-Seq
Objective: To identify genome-wide off-target sites of a compact Cas variant with high sensitivity.
Materials:
- Purified genomic DNA from target cell type
- Recombinant compact Cas protein (e.g., SaCas9, CjCas9)
- In vitro transcribed gRNA
- NGS platform and associated reagents
Methodology:
- In Vitro Cleavage: Incubate 1-5 μg of genomic DNA with the Cas9-gRNA ribonucleoprotein (RNP) complex in an appropriate reaction buffer.
- DNA Purification: Extract and purify the DNA after cleavage.
- Whole-Genome Sequencing: Prepare sequencing libraries from the cleaved DNA and perform high-coverage whole-genome sequencing (WGS).
- Bioinformatic Analysis:
- Map sequencing reads to the reference genome.
- Identify cleavage sites by detecting DNA fragments with identical 5' ends.
- Compare these sites with in silico predicted off-target sites based on sequence similarity to the gRNA.
Digenome-seq is particularly valuable for its sensitivity in detecting off-target effects with low false-positive rates and has been widely applied to characterize both SpCas9 and smaller variants [17].
Research Reagent Solutions Toolkit
Table 3: Essential Research Reagents for Viral CRISPR Delivery Studies
| Reagent/Category | Specific Examples | Function in Research |
|---|---|---|
| Viral Vectors | rAAV serotypes (AAV8, AAV9), Lentiviral vectors | Delivery of CRISPR components to target cells |
| Compact Cas Variants | SaCas9, CjCas9, Nme2Cas9, CasMINI, IscB, TnpB | Genome editing nucleases sized for viral packaging |
| Guide RNA Cloning Systems | U6-promoter driven sgRNA vectors, crRNA/tracrRNA for RNP formation | Target specificity for CRISPR systems |
| Cell Culture Models | HEK293T cells, Primary cells from disease models, iPSCs | In vitro testing of editing efficiency and specificity |
| Animal Models | Mouse disease models (e.g., FahPM/PM, RhoP23H/+) | In vivo validation of therapeutic genome editing |
| Analysis Kits | NGS library preparation kits, DNA extraction kits, Immunohistochemistry reagents | Evaluation of editing outcomes and functional recovery |
| Bioinformatics Tools | CRISPResso2, CCTop, Cas-OFFinder | Analysis of NGS data and prediction of off-target sites |
The development of compact Cas variants has significantly advanced the field of viral vector-mediated CRISPR delivery, enabling efficient in vivo genome editing for both research and therapeutic applications. While each variant presents a unique combination of size, PAM requirement, editing efficiency, and specificity, the expanding toolkit now offers researchers multiple options to match their specific experimental needs. For plant research, where delivery challenges may differ but size constraints persist in certain vector systems, these compact variants provide valuable alternatives to traditional SpCas9. As the field progresses, further engineering and characterization of these systems will continue to enhance their precision and expand their applicability across biological disciplines.
The field of plant genome editing has been revolutionized by CRISPR-Cas9 technology, but limitations in targeting scope, specificity, and efficiency persist. The protospacer adjacent motif (PAM) requirement of the widely used Streptococcus pyogenes Cas9 (SpCas9), primarily the NGG sequence, constrains the targetable genomic space [18]. Furthermore, off-target effects can compromise experimental results and therapeutic applications [19] [20] [21]. To overcome these challenges, researchers have pursued various strategies, including mining natural CRISPR diversity and engineering existing proteins. Recently, a new approach has emerged: using artificial intelligence (AI) to design novel gene editors from scratch. This article objectively compares the performance of these different strategies, focusing on the AI-designed OpenCRISPR-1 and other engineered Cas9 variants, within the context of plant editing research. We summarize quantitative data from key studies and provide detailed methodologies to help researchers select the most appropriate tools for their specific applications.
Performance Comparison of Cas9 Variants
The quest for better gene editors has yielded several variants with distinct characteristics. The following tables summarize the key performance metrics of these systems, based on recent high-throughput evaluations.
Table 1: On-target and Off-target Performance of Cas9 Variants in Human Cells
| Cas9 Variant | Origin | PAM Requirement | Median On-Target Indel Rate (%) | Off-Target Profile (Relative to SpCas9) | Key Characteristics |
|---|---|---|---|---|---|
| SpCas9 (Wild-type) | Streptococcus pyogenes | NGG | 48.3 [22] | Baseline (6.1% median indel rate) [22] | Robust activity; broad off-target effects [19] [20] [21] |
| OpenCRISPR-1 | AI-generated (Profluent) | NGG [23] | 55.7 [22] | 95% reduction in off-target editing [22] | "Drop-in" replacement for SpCas9; potentially lower immunogenicity [23] [24] |
| FrCas9 | Faecalibaculum rodentium | NNTA [20] | Higher than SpCas9 (e.g., 32,408 vs. 14,297 reads at RNF2-1 locus) [20] | Substantially fewer off-target sites [19] [20] | High specificity; enhanced efficiency; benefits from TREX2 fusion [19] [20] |
| eCas9-NG | Engineered (SpCas9-derived) | NG [18] | Data not fully quantified | Lower off-target possibility [18] | Expanded PAM recognition; high-fidelity mutations [18] |
| xCas9 | Engineered (SpCas9-derived) | NG, GAA, etc. [18] | Low editing activities (e.g., 6.1% at TGG PAM in rice) [18] | Data not fully quantified | Broad PAM recognition; lower efficiency in plants [18] |
Table 2: Performance in Plant Systems (Rice) for Base Editing
| Base Editor Construct | Underlying Nuclease | Editing Type | Example Editing Efficiency at Canonical PAM (GGG) | Example Editing Efficiency at Non-Canonical PAM |
|---|---|---|---|---|
| Cas9n-CBE | SpCas9 (nickase) | C-to-T | 54.8% [18] | Not effective at non-canonical PAMs [18] |
| Cas9n-NG-CBE | Cas9-NG (nickase) | C-to-T | 50% [18] | 33.3% at CGA PAM; 13.3% at TGC PAM [18] |
| eCas9n-NG-CBE | eCas9-NG (nickase) | C-to-T | Lower than Cas9n-NG-CBE [18] | 22.7% at TGC PAM [18] |
| xCas9n-CBE | xCas9 (nickase) | C-to-T | No activity detected at tested TGN PAMs [18] | Ineffective in rice [18] |
| ABE7.10 Editors | Various Cas9 nickases | A-to-G | Low (e.g., 6.5% for Cas9n-NG-ABE at GGG) [18] | Very low or no editing detected [18] |
Experimental Protocols for Evaluating Genome Editors
Robust experimental validation is crucial for comparing genome editors. The following section details key methodologies cited in the performance data.
On-target and Genome-wide Off-target Analysis (GUIDE-seq & AID-seq)
GUIDE-seq (Genome-wide, Unidentified Double-strand Break Evaluation by Sequencing) was used to compare FrCas9, SpCas9, and OpenCRISPR-1 [19] [20].
- Oligonucleotide Tag Integration: Cells are co-transfected with the Cas9/sgRNA expression constructs and a blunt, double-stranded oligodeoxynucleotide (dsODN) tag.
- Tag Capture at DSBs: When a double-strand break (DSB) occurs, the dsODN is integrated into the genome via the non-homologous end joining (NHEJ) pathway.
- Library Preparation & Sequencing: Genomic DNA is extracted, sheared, and used to prepare a sequencing library. PCR enrichment is performed using primers specific to the integrated dsODN tag.
- Data Analysis: Sequenced reads are mapped to the reference genome to identify all DSB sites, both on-target and off-target [20].
AID-seq (Adapter-mediated Off-target Identification by Sequencing) provides a highly sensitive, amplification-based method.
- Digestion and Adapter Ligation: Genomic DNA is extracted from edited cells and digested with a restriction enzyme. Double-stranded adapters are ligated to the resulting fragments.
- Target-specific PCR: Two rounds of PCR are performed using adapter-specific and target-specific primers to amplify fragments containing potential off-target sites.
- High-Throughput Sequencing: The final PCR products are sequenced on a high-throughput platform.
- Bioinformatic Analysis: Reads are analyzed to quantify cutting efficiency at thousands of target sites simultaneously, providing a comprehensive profile of on-target and off-target activity [20].
Editing Efficiency Quantification in Plants
For plant editing, targeted amplicon sequencing (AmpSeq) is considered the gold standard for quantifying edits [2].
- Target Amplification: PCR is performed using primers flanking the target genomic region from isolated plant DNA.
- Library Preparation and NGS: The PCR amplicons are prepared into a sequencing library and analyzed on a next-generation sequencing (NGS) platform.
- Variant Analysis: The resulting sequences are aligned to a reference, and insertion/deletion (indel) frequencies are calculated by analyzing the proportion of sequences with mutations at the target site [2].
Alternative methods include:
- T7 Endonuclease 1 (T7E1) Assay: PCR amplicons from the target site are denatured and reannealed, creating heteroduplexes if indels are present. T7E1 enzyme cleaves these heteroduplexes, and the cleavage products are visualized by gel electrophoresis to estimate editing efficiency [2].
- PCR-Capillary Electrophoresis/InDel Detection by Amplicon Analysis (PCR-CE/IDAA): This method uses a fluorescently labeled PCR primer to amplify the target region. The amplicons are separated by capillary electrophoresis, and the resulting electropherogram reveals different fragment sizes corresponding to indels, allowing for precise quantification [2].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Resources for CRISPR Plant Research
| Reagent / Resource | Function in Research | Example/Note |
|---|---|---|
| OpenCRISPR-1 Sequence | AI-generated Cas9 protein for genome editing. | Freely available via GitHub and AddGene; acts as a "drop-in" replacement for SpCas9 in many protocols [23] [24]. |
| CRISPR-Cas Atlas | Extensive dataset for training AI models and bioinformatic mining. | Open-sourced by Profluent; contains over one million CRISPR operons [22] [24]. |
| CRISPOR Tool | In-silico tool for sgRNA design and off-target prediction. | Web-based tool (crispor.gi.ucsc.edu) used for selecting targets with predicted efficiency scores [2]. |
| Dual Geminiviral Replicon (GVR) System | Enables high-level transient expression of CRISPR components in plant leaves. | Based on Bean yellow dwarf virus (BeYDV) for co-expressing SpCas9 and sgRNAs in Nicotiana benthamiana [2]. |
| AID-seq & GUIDE-seq Kits | For genome-wide, unbiased identification of off-target effects. | Critical for safety and specificity profiling of novel editors like FrCas9 and OpenCRISPR-1 [19] [20]. |
| CBE4 & ABE7.10 Base Editor Plasmids | For achieving targeted C-to-T or A-to-G base conversions. | Can be fused to nickase variants of Cas9 (e.g., Cas9-NG, eCas9-NG) to expand target scope [18]. |
Discussion and Future Perspectives
The comparative data reveals a diverse landscape of CRISPR-Cas9 variants, each with distinct advantages. OpenCRISPR-1 represents a breakthrough as the first functional AI-designed editor. Its primary advantage is its status as a highly specific, "drop-in" replacement for SpCas9 that requires minimal protocol adjustments while offering a significantly improved off-target profile [23] [22]. In contrast, FrCas9 demonstrates exceptional on-target efficiency and specificity in comparative studies, positioning it as a powerful candidate for sensitive therapeutic applications where minimizing off-target effects is paramount [19] [20]. For plant researchers seeking to target genomic sites beyond the traditional NGG PAM, engineered variants like Cas9-NG and eCas9-NG are proven and effective tools, enabling efficient editing at NG PAM sites in systems like rice [18].
The emergence of AI-designed proteins like OpenCRISPR-1 signals a shift in protein engineering. This approach uses large language models trained on massive biological datasets to generate novel protein sequences with desired functions, moving beyond the constraints of natural evolution and traditional engineering [22] [25] [24]. As these AI models and high-throughput screening platforms continue to evolve, the future of plant genome editing will likely see an increase in bespoke editors tailored for specific applications, such as targeting particularly challenging genomic loci or exhibiting minimal immunogenicity for clinical use [25] [24]. This will empower researchers to address complex biological questions and develop more precise tools for crop improvement with greater efficiency and safety.
From Lab to Field: Practical Applications of Cas9 Variants in Crop Improvement
The successful application of CRISPR-Cas technology in plant research is highly dependent on the efficient delivery of editing reagents into plant cells. Among the most prominent delivery strategies are Agrobacterium-mediated transformation, plasmid DNA transfection, and ribonucleoprotein (RNP) complex delivery. Each method presents a unique combination of editing efficiency, specificity, and practical implementation considerations. This guide provides an objective comparison of these three fundamental approaches, drawing on recent experimental data to help researchers select the most appropriate method for their plant genome editing projects.
The following table summarizes the core characteristics and performance metrics of the three delivery methods, based on recent comparative studies.
Table 1: Comparative Performance of CRISPR-Cas9 Delivery Methods in Plants
| Delivery Method | Mechanism of Delivery | Editing Efficiency | Off-Target & Unwanted Effects | Key Advantages | Key Limitations |
|---|---|---|---|---|---|
| Agrobacterium-mediated Transformation [26] [27] | Uses disarmed Agrobacterium tumefaciens to transfer T-DNA containing CRISPR cassettes into the plant genome. | High number of mutations; can result in chimeric plants with mixed genotypes requiring segregation [26]. | Potential for random T-DNA integration; can induce host defense responses [27]. | Well-established protocol; suitable for stable transformation and regeneration of whole plants. | Species-dependent efficiency; can be time-consuming due to required tissue culture and segregation. |
| Plasmid Transfection [26] [28] | Direct delivery of plasmid DNA encoding Cas9 and gRNA into plant cells, often via PEG-mediated transfection of protoplasts. | Can achieve high editing rates (e.g., up to 97% mutagenesis in pea protoplasts) [28]. | Unwanted plasmid DNA integration into the host genome occurs frequently (~30%) [26]. | Rapid validation of gRNA efficiency; high throughput screening in protoplasts. | Requires efficient protoplast isolation and regeneration; unwanted plasmid integration is a concern. |
| RNP Complex Delivery [26] [29] [30] | Direct delivery of pre-assembled, purified Cas9 protein and gRNA complexes into protoplasts or via biolistics. | High editing efficiency; demonstrated in chicory, conifers, and onion cells (e.g., 4.5x increase with improved biolistics) [26] [29] [30]. | No off-target mutations detected in controlled studies; significantly reduced risk of DNA vector integration [26]. | DNA-free editing, avoiding foreign DNA integration; immediate activity and rapid degradation reducing off-target risks; high public acceptance. | Requires technical expertise in protein handling and protoplast culture; regeneration from protoplasts can be challenging. |
Detailed Experimental Protocols
To ensure reproducibility, this section outlines the core methodologies from key studies comparing these delivery systems.
Agrobacterium-mediated Stable Transformation
A comparative study in root chicory used Agrobacterium tumefaciens to deliver CRISPR/Cas9 reagents targeting the CiGAS genes [26].
- Vector Design: The Cas9 gene and sgRNA expression cassette were cloned into a binary T-DNA vector.
- Plant Transformation: Chicory explants were co-cultivated with Agrobacterium strain LBA4404. Transformed tissues were selected on appropriate antibiotics.
- Regeneration and Analysis: Transgenic plants were regenerated from selected calli. Genomic DNA was extracted from regenerated plants and analyzed by sequencing to identify mutations in the target genes. A key finding was the frequent occurrence of chimerism, where a single plant contained a mixture of different edited and unedited cells, complicating analysis and requiring segregation in subsequent generations [26].
Plasmid Transfection in Protoplasts
PEG-mediated plasmid transfection offers a rapid, transient system for testing editing efficiency, as optimized in pea [28].
- Protoplast Isolation: Protoplasts are isolated from plant tissues (e.g., leaves) by enzymatic digestion of the cell wall using a solution containing cellulase and macerozyme.
- Transfection: The plasmid DNA (e.g., 20 µg) is mixed with purified protoplasts in the presence of PEG (e.g., 20% concentration) and incubated for a short period (e.g., 15 minutes).
- Analysis: DNA is extracted from transfected protoplasts after 24-48 hours. Editing efficiency is quantified using methods like amplicon sequencing or the T7E1 assay. This high-throughput platform allows for rapid validation of multiple gRNAs before committing to stable transformation [28].
RNP Delivery and Advanced Biolistics
RNP delivery can be achieved via PEG-mediated protoplast transfection or particle bombardment.
- PEG-mediated RNP Transfection: The protocol is similar to plasmid transfection, but the cargo is a pre-complexed mixture of purified Cas9 protein and in vitro-transcribed sgRNA [26] [29]. This was successfully used in chicory and conifer protoplasts, yielding precise edits without foreign DNA [26] [29].
- Biolistic RNP Delivery (Flow Guiding Barrel): A recent technological advancement, the Flow Guiding Barrel (FGB), significantly improves biolistic delivery [30].
- RNP Complex Assembly: Cas9 protein and sgRNA are pre-assembed into RNP complexes in vitro.
- Particle Coating: The RNP complexes are adsorbed onto microscopic gold or tungsten particles.
- Bombardment: The coated particles are accelerated into plant tissues (e.g., onion epidermis, maize embryos) using a gene gun equipped with the FGB device. The FGB optimizes gas and particle flow, increasing delivery efficiency and consistency, and has been shown to boost RNP editing efficiency by 4.5-fold in onion cells [30].
The Scientist's Toolkit: Essential Reagents and Solutions
The following table catalogs key materials required for implementing the discussed delivery methods.
Table 2: Key Research Reagent Solutions for CRISPR Delivery in Plants
| Reagent / Solution | Function / Application | Example Use Cases |
|---|---|---|
| Cellulase R-10 & Macerozyme R-10 [28] | Enzymatic digestion of plant cell walls for protoplast isolation. | Essential for preparing protoplasts for PEG-mediated plasmid or RNP transfection. |
| Polyethylene Glycol (PEG) Solution [28] | Facilitates the fusion of cell membranes and the uptake of macromolecules into protoplasts. | Used as the primary transfection agent in protoplast-based delivery of plasmids and RNPs. |
| Binary Vectors (e.g., pCAMBIA) [27] | Plant transformation vectors containing T-DNA borders for Agrobacterium-mediated gene transfer. | Standard for stable integration of CRISPR cassettes into the plant genome via Agrobacterium. |
| Gold Microcarriers [30] | Microscopic particles used as projectiles to deliver genetic material directly into cells via biolistics. | Coated with plasmid DNA or RNP complexes for bombardment into plant tissues. |
| Flow Guiding Barrel (FGB) [30] | A 3D-printed device that optimizes gas and particle flow in biolistic gene guns. | Enhances the efficiency and consistency of RNP and DNA delivery via particle bombardment. |
| Acetosyringone [27] | A phenolic compound that induces the vir genes of the Agrobacterium Ti plasmid. | Added to co-cultivation media to enhance the efficiency of T-DNA transfer. |
Quantification of Editing Outcomes
Accurately measuring editing efficiency is critical for evaluating and comparing delivery methods. A comprehensive benchmarking study compared several quantification techniques [2].
- High-Accuracy Methods: Targeted amplicon sequencing (AmpSeq) is considered the "gold standard" due to its high sensitivity and accuracy, providing base-resolution data on editing outcomes. PCR-capillary electrophoresis (PCR-CE/IDAA) and droplet digital PCR (ddPCR) were also found to be highly accurate when benchmarked against AmpSeq [2].
- Traditional Methods: Techniques like the T7 endonuclease 1 (T7E1) assay and PCR-restriction fragment length polymorphism (RFLP) are useful for initial screening but are generally less sensitive and quantitative than sequencing-based methods [2].
- Sanger Sequencing with Deconvolution: While accessible, its accuracy can be affected by factors like the base-calling algorithm used. For reliable quantification of editing efficiency, especially in heterogeneous samples, AmpSeq is highly recommended [2].
The choice between Agrobacterium, plasmid, and RNP delivery methods is multifaceted, requiring researchers to balance priorities among editing efficiency, precision, regulatory considerations, and species-specific practicality.
- Agrobacterium-mediated transformation remains the cornerstone for generating stable transgenic plants but carries a higher risk of vector DNA integration and can produce complex, chimeric genotypes.
- Plasmid transfection provides a powerful platform for rapid, high-throughput validation of gRNA efficiency in protoplasts but shares the risk of DNA integration and depends on efficient protoplast regeneration.
- RNP delivery stands out for its high precision, minimal off-target effects, and ability to produce transgene-free edited plants. Its efficiency is being continually enhanced by advancements like improved biolistics with the FGB device [30].
For a research project prioritizing speed and the generation of transgene-free plants, RNP delivery is often the most suitable choice. When stable integration is the ultimate goal, Agrobacterium is the established method, though it requires careful screening. Plasmid-based protoplast systems serve as an invaluable intermediate screening step. The ongoing refinement of these delivery technologies, coupled with accurate editing quantification, continues to expand the frontiers of plant genome engineering.
Cereal crops, including wheat, rice, barley, and maize, form the foundation of global food security. However, many crucial agronomic traits—such as yield, drought tolerance, and disease resistance—are controlled by complex genetic networks rather than single genes [31]. Multiplexed genome editing, which enables simultaneous modification of multiple genetic targets, has emerged as a transformative approach for engineering these polygenic traits in cereals. By allowing researchers to address genetic redundancy and pathway engineering, multiplexing overcomes limitations of single-gene editing, particularly in polyploid species like wheat that contain multiple copies of essential genes [31] [32].
The CRISPR-Cas system, particularly CRISPR-Cas9, has become the platform of choice for multiplexed editing due to its simplicity and versatility [33] [34]. Unlike earlier technologies such as zinc finger nucleases (ZFNs) and transcription activator-like effector nucleases (TALENs), which require complex protein engineering for each new target, CRISPR systems can be redirected to different genomic loci simply by designing complementary guide RNAs (gRNAs) [33]. This molecular programmability makes CRISPR systems uniquely suited for multiplexing, as evidenced by studies demonstrating simultaneous editing of up to 107 target sites in plants [35]. For cereal researchers, this capability is revolutionizing functional genomics and crop improvement strategies, enabling sophisticated engineering projects that were previously impractical or impossible.
Comparative Analysis of Multiplexing Platforms and Architectures
Multiplexing System Performance
Multiplexed editing systems employ distinct architectural strategies for expressing multiple guide RNAs, each with characteristic advantages and limitations for cereal applications.
Table 1: Comparison of Multiplex gRNA Expression Systems in Cereals
| System Architecture | Mechanism | Example Applications | Editing Efficiency Range | Key Advantages |
|---|---|---|---|---|
| Individual Pol III Promoters | Multiple separate gRNA expression cassettes | Gene family editing in Arabidopsis [31] | 0-94% across targets [31] | High fidelity; predictable processing |
| tRNA-gRNA Arrays | Endogenous tRNA-processing machinery (RNase P/Z) cleaves gRNAs from transcript | Multiplex editing in wheat, barley [34] [36] | Superior efficiency in wheat/barley vs. ribozyme systems [36] | Compatible with Pol II promoters; modular design |
| Ribozyme-gRNA Arrays | Self-cleaving hammerhead/hepatitis delta virus ribozymes flank gRNAs | Gene editing in rice, wheat [34] | Similar efficiency to tRNA in rice [36] | No auxiliary proteins needed; works across diverse species |
| Cas12a crRNA Arrays | Cas12a processes its own pre-crRNA transcripts via recognition of hairpin structures | Multiplex regulation in plants [34] | Efficient processing of long arrays [34] | Simplified delivery; compact array design |
Cas Protein Variants for Cereal Multiplexing
The selection of Cas proteins significantly influences multiplexing efficiency, specificity, and targeting range in cereal genomes.
Table 2: Cas Variants for Multiplexed Cereal Genome Editing
| Cas Variant | PAM Requirement | Processing Capability | Best Applications in Cereals | Delivery Considerations |
|---|---|---|---|---|
| SpCas9 | NGG | Requires exogenous processing systems (tRNAs, ribozymes, Csy4) | High-efficiency knockout of gene families; polyploid gene targeting | Standard binary vectors; viral delivery limited by size |
| Cas12a | T-rich (TTTV) | Self-processes crRNA arrays | AT-rich genomes; simplified multiplex vector construction | Smaller size beneficial for viral delivery |
| nCas9 (H840A) | NGG | Nickase activity for base editing | Precision editing with reduced off-target effects | Compatible with BE, PE systems |
| dCas9 | NGG | Catalytically dead; transcriptional regulation | Multiplexed gene regulation (CRISPRi/a) | Epigenetic engineering without DNA cleavage |
| Compact Cas variants (AsCas12f) | T-rich | Varies by specific nuclease | Viral delivery (e.g., PVX) for systemic editing [36] | ~1/3 size of SpCas9; enables full RNP delivery via viruses |
Experimental Framework for Cereal Multiplex Editing
Workflow for Implementing Multiplexed Editing
The following diagram illustrates the comprehensive workflow for designing and implementing multiplexed genome editing experiments in cereal crops:
gRNA Design Pipeline for Complex Cereal Genomes
Designing effective gRNAs for cereals requires specialized approaches to address polyploidy and repetitive genomes:
Case Study: Engineering Wheat Flowering Time via Multiplex Promoter Editing
Experimental Protocol: Ppd-1 Gene Editing in Wheat
A recent study demonstrates the power of multiplex editing for engineering complex traits in wheat by targeting flowering time genes [37]. The following detailed protocol outlines the methodology:
Target Selection and gRNA Design:
- Biological Rationale: Wheat Ppd-1 genes (Ppd-A1, Ppd-B1, Ppd-D1) control photoperiod sensitivity. Natural mutations in promoter regions containing CHE transcription repressor binding sites cause early heading, adapting wheat to different environments [37].
- gRNA Design Strategy: Ten gRNAs were designed to flank CHE binding sites in Ppd-1 promoters. In silico analysis identified gRNAs with high predicted efficiency against Ppd-D1 and Ppd-B1, but not Ppd-A1, leading to focused editing on the former two genes [37].
- In Vitro Validation: Ribonucleoprotein (RNP) complexes with gRNA18 and gRNA21 showed 96% and 94% cleavage efficiency respectively in vitro, though efficiency decreased to 37% and 12% in protoplast assays, highlighting importance of pre-validation [37].
Vector Construction and Transformation:
- Assembly Method: Selected gRNAs were combined with classic sgRNA backbone and wheat U3 promoter, then integrated with SpCas9 and BAR cassettes using the MoClo Kit [37].
- Transformation Protocol: Embryogenic callus from 931 immature embryos of wheat line 'Velut' was transformed via biolistic delivery using PDS-1000/He Gun. Selection employed phosphinothricin-containing media [37].
- Regeneration: 133 independent T0 plantlets were regenerated from selection media, with molecular analysis conducted to identify successful editing events.
Molecular Analysis and Selection:
- Mutation Detection: Next-generation sequencing (NGS) and Sanger sequencing identified mutations in target regions. From 133 plantlets, 46 (35%) contained mutations—26 with plasmid insertions, and 20 without plasmid (transient expression) [37].
- Mutation Spectrum: The majority were 1 bp indels, though deletions of 4-17 bp were common. Seven plants contained large deletions (219-266 bp) removing both CHE repressor binding sites [37].
- Transgene-Free Identification: 20 plantlets contained mutations but lacked plasmid integration, enabling immediate selection of transgene-free edited lines, a significant advantage for regulatory compliance [37].
Phenotypic and Molecular Validation
Gene Expression Analysis:
- Under short-day conditions, edited T0 plants and T2 lines showed altered Ppd-1 expression patterns directly correlated with specific mutation types [37].
- Deletions spanning CHE transcription repressor binding sites demonstrated significantly modified diurnal expression patterns, confirming the functional role of these cis-elements [37].
Heading Time Assessment:
- T1 and T2 families with mutations affecting the "core region" containing CHE binding sites initiated heading significantly earlier than non-mutated controls under field conditions [37].
- The study successfully generated early-heading wheat lines with commercial potential for regions with shorter growing seasons [37].
Research Reagent Solutions for Cereal Multiplex Editing
Table 3: Essential Research Reagents for Cereal Multiplex Editing
| Reagent/Category | Specific Examples | Function in Multiplex Editing | Cereal-Specific Considerations |
|---|---|---|---|
| gRNA Cloning Systems | pYPQ vectors [38]; MoClo Toolkit [37] | Modular assembly of multiplex gRNA arrays | Wheat U3/U6 promoters show superior activity in cereals |
| Cas9 Expression Systems | pMDC32_Cas9 [38]; pJHY1 [32] | Constitutive or inducible nuclease expression | Maize ubiquitin promoter often provides strong expression |
| Delivery Vectors | pRBCS-2B:Cas9 [32]; L241780Ppd_gRNA [37] | Agrobacterium or biolistic delivery | Species-specific optimization required (e.g., rice vs wheat) |
| Processing Enzymes | tRNA processing machinery; Csy4; Ribozymes [34] | Intracellular liberation of individual gRNAs from polycistronic transcripts | tRNA systems show superior efficiency in wheat/barley [36] |
| Selection Markers | BAR gene (phosphinothricin resistance) [37] | Selection of transformed events | Herbicide resistance enables field selection in some systems |
| Validation Tools | WheatCRISPR [32]; CRISPR-GE | gRNA design and off-target prediction | Wheat-specific tools account for polyploid genome complexity |
Comparative Performance Analysis and Technical Considerations
Efficiency Metrics Across Cereal Species
Multiplex editing efficiency varies significantly across cereal species and transformation methods:
- Wheat (Biolistic Transformation): The Ppd-1 editing case study achieved 35% mutation rate in T0 plants, with 15% (20/133) resulting from transient expression without stable T-DNA integration [37].
- Rice (Agrobacterium-mediated): Higher efficiency typically observed, with tRNA and ribozyme systems performing similarly in rice, unlike the tRNA superiority seen in wheat and barley [36].
- Regeneration Challenges: Multiplex editing in cereals faces species-specific and genotype-dependent regeneration barriers, particularly for complex edits requiring precise homologous recombination [36].
Advanced Editing Tool Evolution
Beyond standard CRISPR-Cas9 systems, advanced editors offer enhanced precision for cereal applications:
Base Editing Systems:
- Cytosine Base Editors (CBEs): Enable C•G to T•A conversions without double-strand breaks using deaminase enzymes fused to nCas9 [39] [40].
- Adenine Base Editors (ABEs): Mediate A•T to G•C conversions through engineered TadA deaminase variants [39].
- Cereal Applications: Base editors effectively create single-base changes for trait modification while minimizing complex indel formations common with nuclease editing [39].
Prime Editing Systems:
- PE1/PE2/PE3 Evolution: Successive generations show improved editing efficiency (from 10-20% to 30-50% in human cells) through RT optimization and dual-nicking strategies [40].
- PE4/PE5 Enhancements: Incorporation of MMR inhibition (MLH1dn) increases efficiency to 50-80% by preventing mismatch repair of edited strands [40].
- Cereal Relevance: Prime editing enables precise base conversions, small insertions, and deletions without donor templates, offering particular value for allele-specific editing in polyploid cereals [40].
Multiplexed genome editing represents a paradigm shift in cereal crop engineering, moving beyond single-gene manipulation to system-level genetic redesign. The comparative analysis presented here demonstrates that optimal multiplexing strategies must be tailored to specific cereal species, considering their distinct genomic architectures, transformation efficiencies, and regeneration capabilities. For polyploid wheat, tRNA-based processing systems combined with careful gRNA design to target all homoeologs simultaneously provides the most reliable approach [32] [36]. For diploid cereals like rice, both tRNA and ribozyme systems show comparable efficiency, offering greater flexibility in vector design [36].
Future developments in multiplex editing will likely focus on enhancing precision editing capabilities through prime editing systems [40], improving delivery methods to overcome transformation bottlenecks [36], and developing more sophisticated computational tools for predicting editing outcomes across complex cereal genomes. As these technologies mature, multiplexed editing will become increasingly central to cereal improvement programs, enabling sophisticated engineering of complex trait networks that underlie agricultural productivity and sustainability.
Bacterial blight, caused by Xanthomonas oryzae pv. oryzae (Xoo), represents a major threat to global rice production, particularly in Southeast Asia and West Africa [41]. This disease can cause yield losses of 10%-20%, significantly impacting food security and agricultural economies [42] [43]. Traditional breeding approaches for developing resistant varieties face limitations due to the complex molecular interplay between pathogen virulence factors and host susceptibility genes.
The discovery of Transcription Activator-Like Effectors (TALEs) as key bacterial virulence factors and their targeting of specific host genes has revolutionized our understanding of bacterial blight pathogenesis [41] [43]. TALEs function as eukaryotic transcription factors that bind to specific effector binding elements (EBEs) in the host genome, inducing the expression of susceptibility (S) genes that benefit the pathogen [42] [43].
Among these S genes, members of the OsSWEET family—particularly OsSWEET11, OsSWEET13, and OsSWEET14—encode sugar transporters that pathogens activate to divert nutritional resources from the plant [41] [43]. This case study examines how CRISPR/Cas9-mediated genome editing of OsSWEET genes has emerged as a powerful strategy for developing durable bacterial blight resistance in rice, comparing the efficacy of different approaches across genetic backgrounds.
Molecular Mechanisms of Susceptibility and Editing Strategies
TALE-SWEET Interaction Pathway
The molecular pathogenesis of bacterial blight centers on the manipulation of host sugar transport mechanisms. OsSWEET genes encode bidirectional sugar transporters localized to the plasma membrane, responsible for sucrose and glucose transport [41]. During infection, Xoo delivers TALEs into the plant cell through its type III secretion system [41].
These TALEs translocate to the nucleus and bind to EBEs in the promoters of specific OsSWEET genes. The binding specificity is determined by repeat variable diresidues (RVDs) within the TALE central repeat domain, with different RVD combinations recognizing different DNA sequences [41]. This induction leads to increased expression of SWEET sugar transporters, resulting in the efflux of sugars to the apoplast where the pathogen accesses these nutrients for growth and propagation [41].
Table 1: Major OsSWEET Susceptibility Genes and Their Corresponding TAL Effectors
| Susceptibility Gene | Targeting TAL Effectors | Geographic Distribution | Functional Role |
|---|---|---|---|
| OsSWEET11 | PthXo1 | Asian strains | Sucrose/glucose transporter |
| OsSWEET13 | PthXo2 and variants | Asian strains | Sucrose/glucose transporter |
| OsSWEET14 | AvrXa7, PthXo3, TalC, Tal5, TalF | Asian and African strains | Sucrose/glucose transporter |
The following diagram illustrates the molecular mechanism of TALE-mediated susceptibility and the strategic approaches for developing resistance through genome editing:
Strategic Approaches to Gene Editing for Disease Resistance
Two primary genome editing strategies have been employed to disrupt the TALE-SWEET interaction and confer bacterial blight resistance:
Promoter Editing Approach: This strategy involves modifying the EBEs in the promoter regions of OsSWEET genes to prevent TALE binding while preserving normal gene function and regulation. Research has demonstrated that editing the EBEs of OsSWEET14 can confer resistance to specific Xoo strains depending on which TALE binding sites are modified [42]. For instance, an 18-bp deletion in the promoter region overlapping with AvrXa7, PthXo3, and Tal5 EBEs was identified in African rice varieties and designated as the recessive resistance gene xa41(t) [41].
Coding Sequence Knockout Approach: This alternative strategy completely disrupts the OsSWEET gene function by introducing frameshift mutations in the coding region. CRISPR/Cas9 has been successfully used to generate knockout mutations in OsSWEET14, resulting in broad-spectrum resistance to both Asian and African Xoo strains [41] [44]. Interestingly, different rice genetic backgrounds show varying responses to OsSWEET14 knockout, with the Zhonghua 11 background demonstrating strong resistance to African Xoo strains where the Kitaake background showed only moderate resistance [41].
Experimental Comparison of Editing Strategies
Quantitative Assessment of Resistance Performance
The efficacy of different gene editing strategies has been systematically evaluated across multiple studies, with varying outcomes based on the approach, genetic background, and targeting specificity. The following table summarizes the performance of major editing strategies reported in recent research:
Table 2: Performance Comparison of Major OsSWEET Gene Editing Strategies
| Editing Strategy | Rice Cultivar | Target Gene/Region | Resistance Performance | Agronomic Impact |
|---|---|---|---|---|
| Coding Sequence Knockout | Zhonghua 11 | OsSWEET14 coding region | Strong resistance to Asian Xoo strains and African strain AXO1947 [41] | Increased plant height, no yield penalty [44] |
| Promoter Editing | TBR225 | OsSWEET14 promoter (AvrXa7, PthXo3, TalF EBEs) | Complete resistance to one Vietnamese Xoo strain, significantly reduced susceptibility to another [42] | No significant difference in agronomic traits [42] |
| Multiplex Promoter Editing | Guihong 1 & Zhonghua 11 | OsSWEET11, OsSWEET14, and OsSULTR3;6 promoters | Significantly enhanced resistance to Xoo and Xoc strains; lesion length reduced by 66-93% [43] | No morphological differences, unchanged agronomic traits [43] |
| Promoter Editing | Kitaake | OsSWEET14 promoter (TalC EBE) | Susceptible to African strains depending on TalC for virulence [41] | Not specified |
Detailed Experimental Protocols
CRISPR/Cas9 Vector Construction and Plant Transformation
The experimental workflow for developing disease-resistant rice lines through OsSWEET gene editing involves several critical steps, each requiring optimization for successful outcomes:
Vector Design and Construction: Studies have utilized different CRISPR/Cas9 systems for editing OsSWEET genes. In the Zhonghua 11 background, researchers built a CRISPR/Cas9 construct targeting two sites in the OsSWEET14 coding region—Target I located in the 1st exon and Target II in the 3rd exon [41] [44]. The CRISPR cassettes were introduced into rice plants using Agrobacterium tumefaciens-mediated transformation, followed by selection of transformed cells and regeneration into whole plants [2].
For promoter editing approaches, guide RNAs were designed to target specific EBEs recognized by major TALEs. In the TBR225 variety, researchers designed sgRNAs to target the AvrXa7, PthXo3, and TalF EBEs in the OsSWEET14 promoter [42]. Multiplex editing strategies have also been successfully implemented, simultaneously targeting EBEs in multiple susceptibility genes including OsSWEET11, OsSWEET14, and OsSULTR3;6 [43].
Selection and Regeneration: Following transformation, regenerated plants were screened for desired mutations using PCR and sequencing. In the Zhonghua 11 study, researchers obtained nine different heritable OsSWEET14 mutant alleles through this process [41]. Six of these alleles contained frameshift mutations expected to completely disrupt gene function, while three contained in-frame mutations [41].
The following diagram illustrates the complete experimental workflow from vector construction to the evaluation of edited lines:
Molecular Analysis and Resistance Evaluation
Genotyping and Mutation Characterization: Researchers employed polymerase chain reaction (PCR) and sequencing to detect modifications in rice transformants [41]. In the Zhonghua 11 study, this analysis revealed that two lines (CR-S14-2 and CR-S14-6) harbored homozygous mutant alleles in the T0 generation, while four other lines contained biallelic mutant alleles [41]. Advanced quantification methods such as targeted amplicon sequencing (AmpSeq) have been recognized as the "gold standard" for accurately detecting and quantifying CRISPR edits due to their high sensitivity and reliability [2].
Resistance Phenotyping: Edited rice lines were evaluated for disease resistance through inoculation with various Xoo strains. Lesion length measurements provided quantitative assessment of resistance levels. For example, in the multiplex editing study, lesion lengths in edited lines inoculated with Xoc strain GX01 and Xoo strains K74 and PXO99A were reduced by 66-93% compared to wild-type plants [43].
Gene Expression Analysis: Quantitative RT-PCR was used to measure the expression of susceptibility genes in edited lines after pathogen inoculation. Studies consistently showed that successful editing resulted in significantly reduced induction of OsSWEET genes following infection, confirming that the edited promoters escaped TALE induction [42] [43].
Off-Target Analysis: Comprehensive off-target potential assessment was performed by predicting putative off-target sites using bioinformatics tools like CRISPR-P and CRISPR-GE [43]. In one study, 46 putative off-target sites were analyzed through PCR amplification and sequencing, with results showing only single-nucleotide polymorphisms but no CRISPR-induced mutations [43].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for OsSWEET Gene Editing Experiments
| Reagent/System | Specific Application | Function and Importance |
|---|---|---|
| CRISPR/Cas9 System | Targeted mutagenesis | Introduction of double-strand breaks at specific genomic loci [41] [43] |
| Dual Geminiviral Replicon System | Transient expression | Rapid testing of editing efficiency in plant leaves [2] |
| Endogenous Promoters | Driving Cas9 expression | Enhanced editing efficiency in specific cultivars; LarPE004 promoter showed high activity in larch [45] |
| Ribonucleoproteins (RNPs) | DNA-free editing | Direct delivery of preassembled Cas9-gRNA complexes; eliminates transgene integration [46] [47] |
| Amplicon Sequencing | Mutation quantification | Gold standard for sensitive detection and accurate quantification of editing efficiency [2] |
| Agrobacterium tumefaciens | Stable transformation | Delivery of CRISPR cassettes for stable integration and heritable edits [41] [46] |
This case study demonstrates that CRISPR/Cas9-mediated editing of OsSWEET genes represents a powerful strategy for developing bacterial blight-resistant rice varieties. Both promoter editing and coding sequence knockout approaches have proven effective, with the optimal strategy depending on the specific genetic background, target pathogens, and agronomic requirements.
The research reviewed reveals several critical factors for success. First, genetic background significantly influences resistance outcomes, as demonstrated by the strong resistance in Zhonghua 11 compared to the moderate resistance in Kitaake following OsSWEET14 knockout [41]. Second, multiplex editing targeting multiple susceptibility genes simultaneously provides broader resistance spectrum against diverse pathogen strains [43]. Third, both editing strategies can achieve disease resistance without compromising agronomic performance, making them suitable for crop improvement.
As genome editing technologies continue to advance, with the development of more precise base editors and improved delivery systems, the precision and efficiency of creating disease-resistant crops will further improve. The successful case studies of OsSWEET editing provide a valuable roadmap for implementing similar strategies in other crops facing pathogen threats.
Biofortification represents a transformative strategy to combat micronutrient deficiencies, or "hidden hunger," by enhancing the nutritional value of staple crops through agricultural and nutritional sciences [48]. For researchers and scientists focused on crop biotechnology, the advent of clustered regularly interspaced short palindromic repeats (CRISPR)-associated protein (Cas) systems has revolutionized this field, enabling precise genetic modifications that were previously unattainable with conventional breeding alone [49]. This guide provides an objective comparison of different Cas9-derived editing technologies within the context of nutritional biofortification, evaluating their performance through experimental data, protocols, and practical implementation considerations.
The fundamental advantage of genome editing over traditional transgenic approaches lies in its ability to make precise changes without necessarily integrating foreign genetic material, potentially streamlining regulatory pathways and addressing consumer concerns associated with genetically modified organisms (GMOs) [49]. As global food security challenges intensify due to climate change and population growth, biofortified crops developed through advanced genome editing have emerged as crucial tools for improving nutritional outcomes, particularly in low- and middle-income countries [50].
Evolution of CRISPR-Cas Systems for Precision Editing
The CRISPR-Cas system originated as an adaptive immune mechanism in prokaryotes, with the type II system from Streptococcus pyogenes (SpCas9) becoming the most widely adopted for biotechnological applications due to its simplicity and robustness [1]. The canonical SpCas9 creates double-strand breaks (DSBs) in DNA at sites specified by a guide RNA, which are then repaired through non-homologous end joining (NHEJ) or homology-directed repair (HDR) mechanisms [38]. While powerful, this approach has limitations for biofortification applications, including potential off-target effects and limited precision for specific nucleotide conversions [51].
Table 1: Chronological Development of Key Genome Editing Technologies
| Technology | Year Introduced | Key Mechanism | Primary Editing Outcome | Advantages for Biofortification |
|---|---|---|---|---|
| Zinc Finger Nucleases (ZFNs) | 1990s | Protein-DNA binding domain fused to FokI nuclease | DSBs at target sites | First programmable nucleases; enabled targeted genome modification |
| TALENs | 2009 | Transcription activator-like effector domain fused to FokI nuclease | DSBs at target sites | Higher specificity than ZFNs; modular DNA-binding domain |
| CRISPR-Cas9 | 2012 | RNA-guided Cas9 nuclease | DSBs at target sites | Easier reprogramming; higher efficiency; multiplexing capability |
| Base Editors (BEs) | 2016 | Cas9 nickase fused to deaminase enzymes | Direct nucleotide conversion without DSBs | Higher precision; reduced indel formation; C•G to T•A or A•T to G•C conversions |
| Prime Editors (PEs) | 2019 | Cas9 nickase fused to reverse transcriptase | All possible base-to-base conversions, small insertions/deletions without DSBs | Greatest precision; broad editing scope; minimal byproducts |
| AI-Designed Editors | 2024 | Protein language models generating novel Cas proteins | Diverse editing outcomes with novel PAM specificities | Bypasses evolutionary constraints; optimized properties for specific applications |
Recent advancements have substantially expanded the CRISPR toolbox beyond the original SpCas9 system. The development of base editing represented a significant leap forward by enabling direct chemical conversion of one base pair to another without requiring DSBs [51]. This technology harnesses deaminase enzymes fused to Cas9 nickases to mediate precise nucleotide conversions, with cytosine base editors (CBEs) facilitating C•G to T•A transitions and adenine base editors (ABEs) enabling A•T to G•C transitions [51].
The more recent innovation of prime editing has further expanded capabilities by functioning as a "search-and-replace" genome editing technology that can introduce all possible base-to-base conversions, small insertions, and deletions without requiring DSBs or donor DNA templates [51]. Prime editors consist of a Cas9 nickase (H840A) fused to an engineered reverse transcriptase (RT) programmed with a prime editing guide RNA (pegRNA) that specifies both the target site and encodes the desired edit [51].
Most recently, artificial-intelligence-enabled design has emerged as a powerful approach to bypass evolutionary constraints and generate editors with optimal properties. Using large language models trained on biological diversity, researchers have successfully designed novel gene editors such as OpenCRISPR-1, which exhibits compatibility with base editing while being 400 mutations away from any natural sequence [10].
Diagram 1: Evolution of genome editing technologies showing transition from DSB-dependent to DSB-independent systems.
Comparative Analysis of Editing Technologies for Biofortification
Performance Metrics Across Editing Platforms
Different genome editing platforms offer distinct advantages and limitations for biofortification applications. The selection of an appropriate system depends on multiple factors, including the desired nutritional trait, target crop species, and specific genetic changes required.
Table 2: Performance Comparison of Major Genome Editing Technologies for Biofortification Applications
| Technology | Editing Efficiency Range | Precision Level | Multiplexing Capacity | Off-Target Effects | Primary Applications in Biofortification |
|---|---|---|---|---|---|
| CRISPR-Cas9 | 0.8-95% depending on crop and target [38] | Moderate (indels at target site) | High (multiple gRNAs) | Moderate to high (DSB-dependent) | Gene knockouts for anti-nutrient reduction; trait stacking |
| Base Editors | 10-70% (varies by base conversion type) [51] | High (specific nucleotide conversion) | Moderate | Low to moderate (bystander editing possible) | Precise amino acid changes in storage proteins; metabolic pathway engineering |
| Prime Editors | 1-50% (version and target dependent) [51] | Very high (custom sequence changes) | Low with current systems | Very low (nickase-based) | Creation of novel nutritional alleles; restoration of lost functions |
| AI-Designed Editors | Comparable or improved relative to SpCas9 [10] | To be fully characterized | Demonstrated for Cas9-like effectors | Improved specificity reported | Novel PAM recognition; optimized performance in challenging contexts |
Applications in Staple Crop Biofortification
Each editing technology has demonstrated unique strengths for specific biofortification applications. CRISPR-Cas9 has proven highly effective for gene knockout strategies, such as disrupting genes encoding anti-nutritional factors or repressors of nutrient accumulation. For example, in East African highland bananas, CRISPR-Cas9-mediated editing of the phytoene desaturase (PDS) gene achieved up to 100% editing efficiency in the cultivar Nakitembe and 94.6% in NAROBan5, demonstrating the potential for manipulating carotenoid pathways [38].
Base editors excel in applications requiring precise nucleotide changes, such as modifying enzymatic active sites to alter substrate specificity or creating missense mutations that enhance protein nutritional quality. The compatibility of AI-designed editors like OpenCRISPR-1 with base editing further expands these possibilities [10].
Prime editing offers the broadest versatility for creating specific sequence modifications, including all possible base substitutions, small insertions, and deletions. This capability is particularly valuable for biofortification applications that require the introduction of novel nutritional traits not present in the natural gene pool, such as recreating beneficial alleles from wild relatives or engineering completely new metabolic capabilities [51].
Experimental Protocols for Biofortification Research
CRISPR-Cas9 Mediated Genome Editing in Triploid Crops
The following protocol details the successful genome editing approach used for East African highland bananas (Musa-AAA), as demonstrated in recent research [38]. This methodology is particularly relevant for challenging polyploid species and provides a framework for biofortification applications.
Vector Design and Construction:
- Target Selection: Identify target genes through genomic analysis. For biofortification, focus on genes involved in nutrient biosynthesis, transport, or storage. The phytoene desaturase (PDS) gene serves as an excellent visual marker for establishing editing protocols.
- sgRNA Design: Design sgRNAs with 20-nucleotide spacer sequences targeting conserved regions. For the banana study, two sgRNAs were designed from the Nakitembe PDS gene, with oligonucleotide pairs synthesized with appropriate adaptor sequences.
- Vector Assembly: Clone sgRNAs individually into sgRNA expression plasmids (pYPQ131C and pYPQ132C), then multiplex into pYPQ142 via Golden Gate cloning. Recombine the resulting cassette with a Cas9 entry vector (pYPQ167) and binary vector (pMDC32) to generate the final construct (pMDC32Cas9NktPDS).
Plant Transformation and Regeneration:
- Transformation: Transform embryonic cell suspensions (ECS) using Agrobacterium tumefaciens strain AGL1 harboring the CRISPR-Cas9 construct.
- Selection and Regeneration: Culture transformed cells on selective media containing appropriate antibiotics. Subculture regularly every 2 weeks for approximately 5 months until somatic embryos develop.
- Plant Regeneration: Transfer embryogenic calli to regeneration medium and maintain under 16/8h light/dark conditions at 26±2°C until complete plantlets develop.
Validation and Analysis:
- Phenotypic Screening: Monitor for visual phenotypes indicative of successful editing, such as albinism in PDS-edited lines.
- Molecular Validation: Extract genomic DNA and perform PCR amplification of target regions. Use sequence analysis to confirm editing events and characterize mutation types.
- Nutritional Analysis: For biofortification targets, quantify nutrient levels using appropriate biochemical methods (e.g., HPLC for carotenoids, ICP-MS for minerals).
Diagram 2: Experimental workflow for CRISPR-mediated biofortification of staple crops.
Prime Editing Implementation Protocol
Prime editing requires specialized experimental approaches distinct from standard CRISPR-Cas9 workflows. The following protocol is adapted from recent advances in prime editing systems [51].
Prime Editor Design:
- pegRNA Design: Design prime editing guide RNAs (pegRNAs) containing:
- A spacer sequence (approximately 20 nt) specifying the target site
- A reverse transcriptase template (RTT) encoding the desired edit (typically 10-25 nt)
- A primer binding site (PBS) (approximately 13 nt)
- Stabilization Modifications: Incorporate structured RNA motifs (evopreQ1, mpknot, xr-pegRNA, or G-quadruplex) at the 3' end of pegRNAs to protect against degradation and improve editing efficiency.
- PE Protein Selection: Select appropriate prime editor protein (PE1, PE2, PE3, or PE3b) based on the application. PE3 systems incorporating an additional nicking sgRNA generally show higher efficiency but may increase indel formation.
Delivery and Validation:
- Delivery Method: Choose delivery method based on target crop species. For plants, Agrobacterium-mediated transformation of embryonic tissues remains most common, though ribonucleoprotein (RNP) delivery is emerging as an alternative.
- Screening: Screen for precise edits using restriction fragment length polymorphism (RFLP) analysis, high-resolution melting (HRM) curve analysis, or Sanger sequencing with decomposition tools.
- Specificity Validation: Assess off-target effects through whole-genome sequencing or targeted sequencing of predicted off-target sites.
Research Reagent Solutions for Genome Editing
Successful implementation of biofortification research requires access to specialized reagents and tools. The following table details essential materials and their applications.
Table 3: Essential Research Reagents for Crop Biofortification Studies
| Reagent Category | Specific Examples | Function and Application | Considerations for Biofortification Research |
|---|---|---|---|
| CRISPR-Cas Systems | SpCas9, LbCas12a, AI-designed OpenCRISPR-1 [10] | Target DNA cleavage or modification | Select based on PAM requirements, size constraints, and efficiency in target crop |
| Editing Plasmids | pYPQ131C, pYPQ132C, pMDC32 [38] | Delivery of editing components | Modular systems enable rapid testing of multiple guide RNAs and editing configurations |
| Transformation Tools | Agrobacterium strains (AGL1, EHA105), biolistic equipment | Delivery of editing constructs into plant cells | Optimization required for each crop species; genotype-independent methods needed for broader application |
| Selection Markers | Antibiotic resistance (hygromycin, kanamycin), visual markers (GFP, RFP) | Identification of successfully transformed events | Consider regulatory requirements; excision systems or marker-free approaches may be preferred |
| Validation Reagents | PCR enzymes, sequencing primers, restriction enzymes | Molecular confirmation of editing events | Digital PCR and next-generation sequencing provide highest sensitivity for detecting editing events |
| Cell Culture Media | Embryogenic callus induction and maintenance media | Support of plant regeneration from transformed cells | Composition must be optimized for each crop species and genotype |
The comparative analysis of Cas9 variants and related editing technologies reveals a rapidly evolving landscape with significant implications for nutritional biofortification of staple crops. While standard CRISPR-Cas9 systems remain highly effective for gene knockout applications, base editors and prime editors offer increasingly precise alternatives for sophisticated metabolic engineering approaches. The recent emergence of AI-designed editors further expands the toolbox, potentially overcoming limitations of naturally derived systems.
For research and development professionals, the selection of an appropriate editing platform must consider multiple factors, including the specific nutritional trait target, regulatory environment, and implementation timeline. CRISPR-Cas9 provides a robust, well-characterized system for most initial biofortification efforts, particularly those involving gene knockouts. Base editors offer enhanced precision for specific nucleotide conversions, while prime editors represent the cutting edge for customized sequence changes. AI-designed systems show promise for overcoming limitations of natural systems but require further characterization.
Future directions in the field will likely focus on improving editing efficiency in challenging crop species, developing novel delivery methods that avoid tissue culture bottlenecks, and stacking multiple nutritional traits in single varieties. As global climate patterns continue to shift, the integration of climate resilience with enhanced nutrition will become increasingly important. The ongoing refinement of regulatory frameworks for gene-edited crops will also play a crucial role in determining how quickly these technologies can transition from research laboratories to fields and markets, ultimately impacting global nutrition security.
Abiotic stresses, particularly drought and salinity, pose significant threats to global crop productivity and food security. The development of climate-resilient crops is a critical priority for agricultural biotechnology. Among the most promising tools for this endeavor are CRISPR-Cas genome editing systems, which enable precise modifications of plant genomes to enhance stress tolerance. This guide provides a comparative analysis of different Cas variants and their applications in developing drought and salinity tolerance in crops, supported by experimental data and methodologies.
CRISPR-Cas Variants: Expanding the Toolkit for Stress Tolerance
The foundational CRISPR-Cas9 system from Streptococcus pyogenes (SpCas9) has been widely utilized for plant genome editing. However, its utility can be limited by its protospacer adjacent motif (PAM) requirement (NGG), potential off-target effects, and the nature of induced mutations. To overcome these limitations, several engineered variants and orthologues have been developed, each with distinct advantages for specific applications in abiotic stress engineering [52] [53].
Key Cas Variants for Plant Abiotic Stress Research:
- SpCas9 (Wild-type): The standard nuclease with NGG PAM requirement; widely used for gene knockouts but limited by PAM constraints [52].
- Cas9-NG: An engineered variant that recognizes relaxed NG PAM sites, significantly expanding the targetable genomic space [18].
- eCas9-NG: A high-fidelity version of Cas9-NG with reduced off-target effects, combining expanded PAM recognition with improved specificity [18].
- xCas9: Recognizes a broad range of PAM sequences including NG, GAA, and GAT, though with reported lower editing efficiency in plants [18].
- Cas12a (Cpf1): Utilizes T-rich PAM sequences (TTTV), creates staggered DNA cuts, and is particularly useful for editing AT-rich genomic regions [54].
- Exonuclease-fused Cas Systems: Cas9 or Cas12a fused with exonucleases (e.g., T5 Exo, TREX2) to induce larger, more precise deletions suitable for studying regulatory elements [54].
Table 1: Comparison of Key Cas Variants for Plant Abiotic Stress Research
| Cas Variant | PAM Requirement | Key Features | Editing Efficiency in Plants | Best Applications for Stress Tolerance |
|---|---|---|---|---|
| SpCas9 | NGG | Gold standard, well-established protocols | High (76.5% at NGG sites) [18] | Single gene knockouts, multiplex editing |
| Cas9-NG | NG | Expanded targeting scope | Moderate to high (9.1%-45.5% at non-canonical sites) [18] | Targeting genes with limited NGG sites |
| eCas9-NG | NG | Reduced off-target effects, expanded targeting | Lower than Cas9-NG (5.5%-8.3%) but higher specificity [18] | Applications requiring high precision |
| xCas9 | NG, GAA, GAT | Broad PAM recognition | Low (6.1% at TGG PAM) [18] | Limited applications in plants |
| Cas12a | TTTV | T-rich PAM, staggered cuts, shorter crRNA | Moderate (41-85%) [54] | Editing AT-rich genomic regions |
| T5-Exo-Cas9 | NGG | Generates large deletions (>50 bp) | High (>75%) with shifted mutation spectrum [54] | Regulatory element dissection |
Comparative Performance Data in Crop Systems
Editing Efficiency and Specificity
Direct comparison of Cas variants in rice reveals significant differences in performance. In studies targeting the OsWaxy gene, wild-type SpCas9 demonstrated the highest efficiency (76.5%) at canonical NGG PAM sites. Cas9-NG showed substantial activity (9.1%-45.5%) across non-canonical PAM sites (TGA, TGT, TGC), while xCas9 exhibited minimal editing (6.1%) even at TGG PAM sites [18]. The newly developed eCas9-NG variant, while showing reduced efficiency (5.5%-8.3%) compared to Cas9-NG, offers enhanced specificity with no detected off-target effects in analyzed plants [18].
Mutation Profile Engineering
Standard Cas9 and Cas12a systems predominantly produce small insertions or deletions (indels) of 1-13 bp, which are often sufficient for gene knockouts but inadequate for targeting larger regulatory elements. The fusion of exonucleases to Cas systems has successfully shifted this mutation spectrum. In soybean, T5-Exo-Cas9 fusions generated large deletions (>50 bp) at 12% frequency and moderate deletions (26-50 bp) at 27% frequency, while TREX2-Cas9 fusions favored small-sized deletions (11-25 bp) at 67% frequency [54]. This capability is particularly valuable for targeting cis-regulatory elements, microRNAs, and transcription factor binding sites that typically range from 10-40 bp [54].
Table 2: Deletion Size Distribution by Cas Variant in Soybean Hairy Root System [54]
| Editing System | Micro (1-10 bp) | Small (11-25 bp) | Moderate (26-50 bp) | Large (>50 bp) | Overall Efficiency |
|---|---|---|---|---|---|
| Cas9 | 84% | 2.5% | 2% | 0.5% | >75% |
| T5-Exo-Cas9 | 31% | 30% | 27% | 12% | >75% |
| TREX2-Cas9 | 9% | 67% | 18% | 6% | >75% |
| Cas12a | 71% | 21% | 4.7% | 0.3% | 41-85% |
| T5-Exo-Cas12a | 15% | 30% | 55% | 5% | 41-85% |
Applications in Drought and Salinity Tolerance
Success Cases in Drought Tolerance
Potato CBP80 Editing: CRISPR/Cas9 editing of the StCBP80 gene in tetraploid potato cv. Spunta resulted in enhanced drought tolerance through improved stomatal regulation. Eight independent partially edited lines (2-3 alleles edited) were obtained and characterized under restricted water conditions. The edited lines exhibited reduced transpiration rates, improved leaf area index, and lower yield penalties under drought stress. Molecular analysis confirmed differential expression of drought-responsive genes (P5CS, PDH, and MYB33), supporting CBP80's role in stress response modulation [55].
Experimental Protocol: The editing construct was delivered via Agrobacterium-mediated transformation of potato explants. The sgRNA was designed to target specific exons of the StCBP80 gene (LOC102588913). Transgenic plants were regenerated and screened for mutations. Drought phenotyping was conducted under controlled water restriction, measuring physiological parameters (transpiration rate, leaf area index) and yield components. Gene expression analysis was performed using quantitative real-time PCR on drought-responsive genes [55].
Tomato HyPRP1 Domain Editing: Precise domain editing of the SlHyPRP1 gene in tomato using CRISPR-Cas9 multiplexing generated variants with eliminated functional domains (PRD and/or 8CM). The edited lines exhibited multi-stress tolerance, including improved performance under drought conditions. The PRD removal variant (PR1v1) showed continuous growth after 5 days of water cutoff and improved germination under osmotic stress (up to 200 mM mannitol) [56].
Success Cases in Salinity Tolerance
Rice OsRR22 Knockout: CRISPR/Cas9-mediated knockout of the OsRR22 gene, encoding a B-type response regulator transcription factor, significantly enhanced salinity tolerance in rice. The editing system utilized the pYLCRISPR/Cas9Pubi-H vector with hpt selection. Nine mutant plants with six mutation types were identified from 14 T0 transgenic plants. Homozygous T2 mutant lines showed significantly enhanced salinity tolerance at the seedling stage without adverse effects on agronomic traits [57].
Experimental Protocol: The Cas9-OsRR22-gRNA expressing vector was constructed using the pYLCRISPR/Cas9Pubi-H system. The target sequence (19-20 bp upstream of PAM) was selected with minimal off-target potential. Transformation was performed via Agrobacterium-mediated method in rice cultivar WPB106. Mutant plants were identified through sequencing of the target region. Salinity tolerance was assessed at seedling stage using nutrient solution culture with salt stress [57].
Tomato HyPRP1 Editing for Salt Tolerance: In our previous study, CRISPR-Cas9 multiplex editing of SlHyPRP1 generated precise deletions of its functional domains. The variants with removed PRD domain showed the highest level of salinity tolerance, demonstrating the value of domain-specific editing rather than complete gene knockouts for optimizing stress tolerance traits [56].
Signaling Pathways in Plant Stress Response
The following diagrams illustrate key molecular pathways involved in drought and salinity stress responses that have been successfully targeted using CRISPR-based approaches.
Diagram 1: Stress Response Pathways and CRISPR Targets. This diagram illustrates key molecular pathways involved in drought and salinity stress responses. ABA (abscisic acid) serves as a central signaling molecule that regulates multiple downstream effectors. CBP80, AREB1, MYB33, HyPRP1, and OsRR22 represent successful CRISPR editing targets for enhancing stress tolerance.
Experimental Workflow for Stress Tolerance Engineering
Diagram 2: Experimental Workflow for Engineering Stress Tolerance. This diagram outlines the key steps in developing stress-tolerant crops using CRISPR-Cas systems, from target identification to phenotypic characterization.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for CRISPR-Based Stress Tolerance Engineering
| Reagent/Category | Specific Examples | Function/Application | Considerations for Stress Research |
|---|---|---|---|
| Cas Expression Systems | pYLCRISPR/Cas9Pubi-H [57], exonuclease-fusion vectors [54] | Delivery of Cas nucleases with various promoters | Selection of appropriate Cas variant based on PAM requirements and editing goals |
| Plant Transformation | Agrobacterium strains (e.g., K599 for hairy roots [54]), RNP delivery | Introduction of editing components into plant cells | Hairy root systems enable rapid testing; stable transformation for whole plants |
| Selection Markers | Hygromycin (hpt), GFP screening [54] [57] | Identification of successfully transformed tissues | Consider marker-free approaches for regulatory compliance |
| sgRNA Design Tools | CRISPR-GE [18], target specificity validation | Design of specific guide RNAs with minimal off-target potential | Essential for polyploid crops with homeoalleles |
| Mutation Detection | Deep amplicon sequencing [54], Sanger sequencing with decoding [57] | Identification and characterization of induced mutations | Long-range PCR useful for detecting large deletions [54] |
| Phenotyping Platforms | Controlled drought stress systems, salinity screening protocols [57] [55] | Assessment of stress tolerance phenotypes | Include physiological (transpiration) and molecular (gene expression) metrics |
The expanding CRISPR-Cas toolkit offers diverse solutions for engineering drought and salinity tolerance in crops. Wild-type SpCas9 remains highly efficient for standard knockout applications, while Cas9-NG provides broader targeting scope with maintained efficiency. For applications requiring enhanced specificity, eCas9-NG offers reduced off-target effects, and for generating larger deletions targeting regulatory elements, exonuclease-fused systems show particular promise. The choice of Cas variant should be guided by the specific target genes, desired mutation types, and crop system requirements. As these tools continue to evolve, their precision and applicability for developing climate-resilient crops will further expand, contributing to global food security under changing environmental conditions.
Maximizing Efficiency and Minimizing Risks: Optimization Strategies for Plant Systems
The application of CRISPR-Cas systems in plant genome editing has revolutionized functional genomics and crop improvement strategies. However, the occurrence of off-target effects—unintended edits at genomic sites with similarity to the target sequence—remains a significant concern for researchers seeking precise genetic modifications. These off-target activities can confound experimental results, introduce unintended phenotypic changes, and raise safety concerns for agricultural applications. The CRISPR-Cas9 system from Streptococcus pyogenes (SpCas9), while widely used as the workhorse nuclease in many laboratories, demonstrates considerable off-target editing due to its tolerance for mismatches between the guide RNA and target DNA [6] [58].
To address these limitations, several high-fidelity Cas variants have been engineered specifically to reduce off-target effects while maintaining robust on-target activity. This comparison guide focuses on two advanced high-fidelity variants—eSpOT-ON (an engineered version of PsCas9) and hfCas12Max (an engineered Cas12i variant)—which represent significant advancements in precision genome editing for plant research. These variants employ distinct molecular strategies to enhance specificity, offering researchers valuable tools for applications demanding high precision, such as functional gene characterization in complex plant genomes and the development of precisely edited crop varieties with improved agronomic traits.
Understanding Off-Target Effects in CRISPR Systems
Off-target effects in CRISPR systems occur when the Cas nuclease cleaves DNA at sites other than the intended target sequence. The mechanisms underlying these erroneous edits involve several factors, including mismatch tolerance between the guide RNA and target DNA, genomic context, cellular enzyme concentration, and the energetics of RNA-DNA hybrid formation [58]. Traditional SpCas9 can recognize non-canonical PAM sequences (such as 5'-NAG-3' and 5'-NGA-3' in addition to its canonical 5'-NGG-3' PAM), further expanding its potential off-target landscape [6].
In plant genome editing, the detection and quantification of off-target effects present unique challenges due to factors such as polyploidy, high frequencies of sequence variations between homeologs, and the presence of repetitive DNA [2]. Accurate measurement of editing specificity is essential for evaluating novel high-fidelity variants, with targeted amplicon sequencing (AmpSeq) currently considered the gold standard for sensitive detection of low-frequency off-target events [2].
High-Fidelity Variants: Molecular Solutions for Enhanced Precision
eSpOT-ON (Engineered PsCas9)
Development and Engineering Strategy eSpOT-ON is a commercially available high-fidelity nuclease developed by AstraZeneca through extensive engineering of the wild-type Parasutterella secunda Cas9 (PsCas9) [6]. While the native PsCas9 nuclease exhibited high on-target activity, it lacked the fidelity required for therapeutic applications. Researchers systematically tested mutations across multiple domains of the nuclease, focusing on the RuvC, wedge (WED), and PAM-interacting domains, to create an engineered PsCas9 variant (commercially termed eSpOT-ON) with superior specificity profiles [6].
Key Characteristics and Performance eSpOT-ON achieves exceptionally low off-target editing while retaining robust on-target activity, addressing a common trade-off in high-fidelity nuclease development where reduced off-target effects often come at the expense of on-target efficiency [6]. The system incorporates optimized guide RNAs with reduced size, which enhances molecular stability and improves interaction between the nuclease and guide RNA, further boosting editing efficiency [6]. eSpOT-ON is available in both recombinant protein and mRNA formats, facilitating flexible delivery strategies for plant research applications.
hfCas12Max
Development and Engineering Strategy hfCas12Max was engineered by HuidaGene Therapeutics from a Cas12i nuclease (type V CRISPR-Cas family) using their HG-PRECISE platform [6]. This engineering process focused on modifying the natural Cas12i enzyme to enhance its gene editing capabilities while reducing unwanted off-target activity. The resulting high-fidelity nuclease maintains the characteristic staggered-end DNA cuts produced by other Cas12 family nucleases, which differ from the blunt-end cuts generated by Cas9 enzymes [6].
Key Characteristics and Performance hfCas12Max recognizes a simple 5'-TN-3' PAM sequence, significantly expanding the range of targetable genomic sites compared to SpCas9 (which requires 5'-NGG-3') [6]. This broad PAM recognition profile enables researchers to target genomic regions inaccessible to many other Cas nucleases. With a compact size of only 1080 amino acids and requiring only a crRNA (which is shorter than standard sgRNAs), hfCas12Max offers practical advantages for delivery via lipid nanoparticles (LNPs) or viral vectors [6]. This variant has already been incorporated into clinical development programs, including HG302, a CRISPR-based therapeutic for Duchenne muscular dystrophy [6].
Comparative Analysis of High-Fidelity Variants
Table 1: Comparison of Key Characteristics Between High-Fidelity Variants
| Characteristic | eSpOT-ON (ePsCas9) | hfCas12Max | Standard SpCas9 |
|---|---|---|---|
| Parent Nuclease | Parasutterella secunda Cas9 | Cas12i (Type V) | Streptococcus pyogenes Cas9 |
| Engineering Approach | Domain-specific mutations (RuvC, WED, PAM-interacting) | HG-PRECISE platform engineering | Wild-type |
| PAM Requirement | Specific PAM not detailed in available literature | 5'-TN-3' | 5'-NGG-3' |
| Cut Type | Blunt ends (characteristic of Cas9) | Staggered ends (characteristic of Cas12) | Blunt ends |
| Size | Not specified | 1080 amino acids | 1368 amino acids |
| Guide RNA | Optimized, reduced-size gRNA | crRNA (shorter than standard sgRNAs) | sgRNA (crRNA+tracrRNA) |
| Key Advantage | Exceptional low off-target editing with maintained high on-target activity | High fidelity with expanded targeting range due to simple PAM | Well-characterized, widely used |
Table 2: Performance Metrics and Applications
| Performance Aspect | eSpOT-ON | hfCas12Max | Standard SpCas9 |
|---|---|---|---|
| On-target Efficiency | Robust activity maintained | Enhanced editing capabilities | Variable depending on gRNA |
| Off-target Reduction | Exceptionally low off-target editing | Significant reduction in unwanted editing | High off-target potential |
| Plant Applications | Suitable for plant editing systems | Demonstrated utility in plant systems | Extensive history in plants |
| Delivery Format | Recombinant protein, mRNA | Compatible with LNPs, AAVs | Multiple formats available |
| Therapeutic Status | Preclinical development | In clinical pipeline (HG302 for DMD) | Clinical trials ongoing |
Experimental Protocols for Evaluating High-Fidelity Variants in Plants
Protoplast-Based Validation of Editing Efficiency
The evaluation of high-fidelity Cas variants in plant systems requires robust experimental protocols that can accurately quantify both on-target efficiency and off-target effects. Protoplast-based transient expression systems provide a rapid validation platform before undertaking stable transformation, which is particularly valuable for assessing novel CRISPR variants [59].
Optimized Protoplast Isolation and Transfection Protocol [59]:
Plant Material Preparation: Use 10-14 day old rice seedlings or 4-5 week old Arabidopsis leaves as starting material.
Tissue Digestion:
- Incubate finely cut leaf strips in enzyme solution containing 0.6M mannitol for 5 hours to digest cell walls.
- Include a sucrose gradient step to separate viable protoplasts from debris and non-viable cells, significantly improving viability (from 50% to 80-91% in rice).
Transfection Optimization:
- Adjust plasmid DNA concentration to 20-30µg for optimal transfection efficiency.
- Use 20-minute incubation period with PEG-Ca2+ solution.
- Prefer smaller plasmid sizes (<10kb) for higher transfection efficiency.
Efficiency Validation:
- For CRISPR-deletion strategies using dual sgRNAs, editing success can be detected via simple agarose gel electrophoresis.
- Confirm precise editing outcomes using Sanger sequencing of PCR products.
This protocol enables researchers to validate sgRNA cleavage efficiency and compare nuclease performance within one week, significantly accelerating the testing of high-fidelity variants like eSpOT-ON and hfCas12Max before committing to lengthy stable transformation experiments [59].
Methods for Quantifying Off-Target Effects
Accurate detection of off-target effects is crucial for evaluating high-fidelity variants. Multiple methods are available with varying sensitivities and requirements [2]:
Table 3: Methods for Quantifying Genome Editing Efficiency and Specificity
| Method | Principle | Sensitivity | Advantages | Limitations |
|---|---|---|---|---|
| Targeted Amplicon Sequencing (AmpSeq) | High-throughput sequencing of PCR amplicons from target sites | Very high (detects low-frequency edits) | Considered gold standard, quantitative | Higher cost, specialized equipment |
| PCR-RFLP | Restriction enzyme digestion of PCR products from target sites | Moderate | Simple, low-cost | Limited sensitivity, requires specific restriction site |
| T7 Endonuclease 1 (T7E1) Assay | Enzyme cleavage of mismatched DNA heteroduplexes | Moderate | Widely used, no specialized equipment | Semi-quantitative, intermediate sensitivity |
| Droplet Digital PCR (ddPCR) | Partitioned PCR enabling absolute quantification of edits | High | Accurate quantification, high sensitivity | Specialized equipment required |
| PCR-Capillary Electrophoresis/IDAA | Fragment size analysis by capillary electrophoresis | High | Accurate size determination of indels | Specialized equipment required |
Among these methods, AmpSeq, PCR-CE/IDAA, and ddPCR show the highest accuracy when benchmarked against AmpSeq as the gold standard [2]. For comprehensive assessment of high-fidelity variants, researchers should employ at least one high-sensitivity method (such as AmpSeq) to confidently detect low-frequency off-target events that might be missed by less sensitive approaches.
Table 4: Key Research Reagent Solutions for High-Fidelity CRISPR Experiments
| Reagent/Resource | Function | Application Notes |
|---|---|---|
| High-quality Synthetic Guide RNA | Directs Cas nuclease to target sequence | Critical for achieving high editing efficiency and reducing off-target effects [6] |
| Protoplast Isolation Solutions | Enzymatic digestion of cell walls, osmotic stabilization | 0.6M mannitol concentration optimal for rice; sucrose gradient improves viability [59] |
| PEG Transfection Solution | Facilitates DNA/RNP delivery into protoplasts | 20-minute incubation optimal; concentration must be carefully optimized [59] |
| Next-generation Sequencing Kits | AmpSeq library preparation for off-target assessment | Essential for comprehensive evaluation of editing specificity [2] |
| Ribonucleoprotein (RNP) Complexes | Pre-complexed Cas protein and guide RNA | Higher editing efficiency, reduced off-target effects compared to plasmid delivery [6] |
| Cell Viability Stains (FDA, Evans Blue) | Assessment of protoplast viability and integrity | Critical for normalizing transfection efficiency and editing outcomes [59] |
Decision Framework: Selecting the Appropriate High-Fidelity Variant
The choice between eSpOT-ON, hfCas12Max, and other high-fidelity variants depends on several experimental factors:
Select eSpOT-ON when:
- Maximum reduction of off-target effects is the primary concern
- Maintaining high on-target efficiency is critical
- Working with genetically complex plant systems where even low-frequency off-targets could confound results
Choose hfCas12Max when:
- Targeting genomic regions with simple 5'-TN-3' PAM sequences
- Delivery size constraints are a consideration (e.g., viral vector packaging)
- Staggered-end cuts are preferred for downstream applications
General considerations for plant research:
- Assess the PAM requirements for your target genes of interest
- Consider the delivery method (RNP, mRNA, plasmid) compatible with your plant system
- Evaluate the availability of validated protocols for your specific plant species
- Factor in the need for comprehensive off-target assessment using sensitive detection methods
The development of high-fidelity CRISPR variants like eSpOT-ON and hfCas12Max represents significant progress in addressing the challenge of off-target effects in plant genome editing. These engineered nucleases employ distinct strategies to enhance specificity while maintaining efficient on-target activity, providing researchers with valuable tools for precise genetic manipulation.
Future advancements in this field will likely include the continued refinement of existing high-fidelity variants through additional protein engineering, the exploration of novel Cas nucleases from diverse microbial sources, and the integration of artificial intelligence approaches to guide nuclease design and gRNA selection [60]. AI-driven tools are already being applied to predict gRNA activity, off-target profiles, and editing outcomes, further enhancing the precision and efficiency of CRISPR experiments [60].
As these technologies mature, researchers can expect an expanding toolkit of high-fidelity editing options tailored to specific plant species and experimental requirements, ultimately accelerating both basic plant research and the development of improved crop varieties with precisely engineered traits.
The CRISPR/Cas9 system from Streptococcus pyogenes (SpCas9) has become the most widely adopted tool for genome engineering in plants, enabling targeted gene modifications across many crop species [11] [61]. However, a significant limitation of the wild-type SpCas9 system is its requirement for a specific protospacer adjacent motif (PAM) sequence (5'-NGG-3') immediately adjacent to the target site [11] [61] [6]. This constraint restricts the potential targetable sites within plant genomes, particularly in AT-rich regions where NGG PAMs are less frequent.
To address this limitation, researchers have developed engineered SpCas9 variants with altered PAM specificities. Two of the most promising variants are xCas9 and Cas9-NG, both designed to recognize relaxed PAM sequences while maintaining DNA targeting specificity [62] [18]. xCas9, developed through phage-assisted continuous evolution, contains seven mutations (A262T, R324L, S409I, E480K, E543D, M694I, E1219V) that broaden PAM recognition to include NG, GAA, and GAT sequences [62] [63]. Cas9-NG, created by structure-guided engineering, contains a single mutation (D1135L) with additional mutations in the PI domain (R1335E/L1111R/R1335Q/T1337R) that enable robust targeting of NG PAMs [62] [18].
This guide provides a comprehensive comparison of these PAM-expanded Cas9 variants, focusing on their performance in plant systems to help researchers select the appropriate tools for their genome editing applications.
Technical Comparison of xCas9 and Cas9-NG Variants
Molecular Characteristics and PAM Recognition Profiles
xCas9 (xCas9 3.7) incorporates seven amino acid mutations (A262T/R324L/S409I/E480K/E543D/M694I/E1219V) that alter PAM interaction, enabling recognition of a broader range of sequences including NG, GAA, and GAT [62] [63]. The variant was developed through phage-assisted continuous evolution to enhance PAM compatibility while maintaining high DNA specificity [62].
Cas9-NG contains a D1135L mutation that disrupts the interaction with the distal guanine in the NGG PAM, combined with additional mutations in the PAM-interacting (PI) domain (R1335E/L1111R/R1335Q/T1337R) that enable efficient targeting of NG PAMs (where N = A, T, C, or G) [62] [18]. Unlike xCas9, Cas9-NG does not efficiently recognize GAA and GAT PAMs [62].
Table 1: Molecular Characteristics of PAM-Expanded Cas9 Variants
| Characteristic | xCas9 | Cas9-NG | Wild-Type SpCas9 |
|---|---|---|---|
| Key Mutations | A262T, R324L, S409I, E480K, E543D, M694I, E1219V | D1135L + PI domain mutations | None |
| Canonical PAM | NGG | NGG | NGG |
| Expanded PAM Recognition | NG, GAA, GAT | NG | NAG (weak) |
| Size (aa) | ~1368 | ~1368 | ~1368 |
| PAM Interaction | Broadened specificity | Relaxed specificity | Stringent specificity |
Performance Comparison in Plant Systems
Comparative studies in rice and Arabidopsis reveal distinct performance patterns for these variants. The following diagram illustrates the general workflow for evaluating these Cas9 variants in plants, from vector construction to analysis of editing outcomes:
Figure 1: Experimental Workflow for Evaluating Cas9 Variants in Plants
Table 2: Editing Efficiencies of Cas9 Variants at Different PAM Sites in Rice
| PAM Type | Example PAM | xCas9 Efficiency | Cas9-NG Efficiency | SpCas9 Efficiency |
|---|---|---|---|---|
| NGG (Canonical) | TGG | ~Equivalent to WT [62] | Significantly reduced (27.3% vs 76.5%) [18] | High (76.5%) [18] |
| NGT | TGT | Limited activity [62] | 9.1%-45.5% [18] | Ineffective |
| NGA | TGA | Limited activity [62] | 9.1%-45.5% [18] | Ineffective |
| NGC | TGC | Limited activity [62] | 9.1%-45.5% [18] | Ineffective |
| GAA | GAA | Limited in KO; improved with tRNA-esgRNA [11] [61] | Not efficient [62] | Ineffective |
| GAT | GAT | Limited in KO; improved with tRNA-esgRNA [11] [61] | Not efficient [62] | Ineffective |
In Arabidopsis, both variants show reduced efficiency compared to rice. xCas9 edited only 9.5% of transgenic lines at NGG PAMs and 10% at NGA PAMs, while Cas9-NG showed 3.4-13% efficiency at NG PAMs, with one NGA site reaching 48% efficiency [64].
Optimization Strategies for Enhanced Performance
RNA Expression Systems
A critical advancement for improving xCas9 performance in plants involves the implementation of the tRNA-enhanced sgRNA (esgRNA) system [11] [63] [61]. This approach utilizes cellular enzymes RNase P and RNase Z to process precise 20-nucleotide target sequences from precursor transcripts, resulting in significantly improved editing efficiency at non-canonical PAM sites [63] [61].
Studies demonstrate that xCas9 combined with tRNA-esgRNA systems enables efficient gene mutagenesis at GAA, GAT, and GAG PAM sites in rice, where previous systems failed [11] [61]. For example, xCas9 with tRNA-esgRNA achieved base editing at GAA PAM sites with frequencies up to 44.4% in T0 plants, compared to no editing with conventional sgRNA systems [63].
Base Editor Development
Both xCas9 and Cas9-NG have been successfully incorporated into cytosine base editor (CBE) and adenine base editor (ABE) platforms to expand the targeting scope of precise base editing in plants:
xCas9n-epBE: An efficient cytosine base editor created by fusing xCas9 nickase (D10A) with PmCDA1 cytidine deaminase and UGI, which enables C-to-T conversion at GA and NG PAM sites in rice [63]. This system achieved base editing frequencies ranging from 5% to 70% at various non-canonical PAM sites [63].
Cas9n-NG-CBE: A Cas9-NG-based cytosine base editor that shows robust activity at NG PAM sites with editing windows primarily at positions C3-C8, achieving C-to-T conversion efficiencies of 5.6%-50% at various NG PAM sites in rice [18].
Adenine Base Editors: Both xCas9 and Cas9-NG have been used in ABE systems, though with lower efficiency than CBEs in plants. Cas9n-NG-ABE showed minimal activity (6.5% at GGG PAM), indicating need for further optimization [18].
Table 3: Optimized Systems for PAM-Expanded Genome Editing in Plants
| Application | System | Key Components | Performance | Optimal PAMs |
|---|---|---|---|---|
| Gene Knockout | xCas9 + tRNA-esgRNA | xCas9 + tRNA-processed sgRNA | Efficient editing at GAA, GAT, GAG PAMs [11] [61] | GAA, GAT, GAG |
| Gene Knockout | Cas9-NG | Cas9-NG + conventional sgRNA | High efficiency at NG PAMs (9.1-45.5%) [18] | NGT, NGA, NGC |
| Cytosine Base Editing | xCas9n-epBE | xCas9n + PmCDA1 + UGI + tRNA-esgRNA | 5-70% efficiency at GA PAMs [63] | GAA, GAT, GAC, GAG |
| Cytosine Base Editing | Cas9n-NG-CBE | Cas9n-NG + rAPOBEC1 + UGI | 5.6-50% efficiency at NG PAMs [18] | NGG, NGT, NGA |
| Adenine Base Editing | xCas9n-ABE | xCas9n + ecTadA ecTadA* | Very low efficiency in plants [18] | Limited |
| Adenine Base Editing | Cas9n-NG-ABE | Cas9n-NG + ecTadA ecTadA* | Minimal activity (6.5% at best) [18] | Limited |
Experimental Protocols for Plant Genome Editing with PAM-Expanded Variants
Vector Construction for xCas9 and Cas9-NG Systems
The following protocol outlines the key steps for constructing plant transformation vectors for PAM-expanded Cas9 variants, based on established methods [11] [63] [61]:
Cas9 Variant Cloning:
- For xCas9: Introduce seven mutations (A262T, R324L, S409I, E480K, E543D, M694I, E1219V) into a plant-codon-optimized SpCas9 backbone using site-directed mutagenesis or gene synthesis [11] [61].
- For Cas9-NG: Introduce the D1135L mutation combined with PI domain mutations (R1335E/L1111R/R1335Q/T1337R) into SpCas9 [18].
RNA Expression Cassette Assembly:
- For standard sgRNA: Clone target sequences (20nt) under plant U6 or U3 promoters [61].
- For enhanced tRNA-esgRNA system: Insert tRNA-sgRNA fusion sequences designed to be processed by endogenous RNases to generate precise sgRNAs [11] [63]. Multiple sgRNAs can be assembled in tandem for multiplexed editing.
Binary Vector Assembly:
- Assemble expression cassettes in T-DNA binary vectors suitable for Agrobacterium-mediated transformation.
- Include plant selectable markers (e.g., hygromycin resistance) and appropriate terminators.
Plant Transformation and Analysis
The transformation and analysis workflow follows established protocols for rice and other model plants [11] [63] [61]:
Plant Transformation:
- Introduce binary vectors into Agrobacterium tumefaciens strain EHA105.
- Infect embryogenic calli induced from mature seeds.
- Culture on selection medium (e.g., 50 μg/mL hygromycin) for 4 weeks to obtain resistant calli.
- Regenerate shoots on regeneration medium for approximately 1 month.
- Root developed shoots to obtain T0 plants.
Mutation Analysis:
- Extract genomic DNA from T0 plants or resistant calli.
- PCR-amplify target regions using specific primers.
- Analyze mutations by:
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagents for PAM-Expanded Genome Editing in Plants
| Reagent/Solution | Function | Examples/Specifications |
|---|---|---|
| xCas9 Expression Vector | Expresses xCas9 variant with broad PAM recognition | Contains xCas9 3.7 with 7 mutations; driven by constitutive promoters like OsUbq or 35S [63] |
| Cas9-NG Expression Vector | Expresses Cas9-NG variant for NG PAM targeting | Contains D1135L and PI domain mutations; compatible with standard sgRNAs [18] |
| tRNA-esgRNA Vectors | Enhanced sgRNA expression system | Produces precise sgRNAs through cellular processing; significantly boosts xCas9 efficiency [11] [63] |
| Base Editor Constructs | For precise base editing at expanded PAM sites | xCas9n-epBE (xCas9n-PmCDA1-UGI) or Cas9n-NG-CBE (Cas9n-NG-rAPOBEC1-UGI) [63] [18] |
| Plant Codon-Optimized Cas9 | Improves expression in plant systems | Sequence optimization based on plant codon usage preferences [11] [61] |
| Binary Vectors for Plant Transformation | T-DNA vectors for Agrobacterium delivery | Contain plant selection markers (hygromycin, basta) and appropriate terminators [11] |
| Agrobacterium Strains | For plant transformation | EHA105 for rice and other monocots; GV3101 for Arabidopsis [11] [64] |
Based on comprehensive benchmarking in plant systems, the following guidelines emerge for selecting and implementing PAM-expanded Cas9 variants:
For targeting NG PAM sites: Cas9-NG demonstrates superior performance with editing efficiencies ranging from 9.1% to 45.5% at NGT, NGA, and NGC PAMs in rice [62] [18]. The variant is particularly effective in AT-rich genomic regions where NG PAMs are abundant.
For targeting GA PAM sites: xCas9 combined with tRNA-esgRNA systems enables efficient editing at GAA, GAT, and GAG PAMs, achieving base editing frequencies of 5%-70% in rice [63]. This system is preferred for targeting specific sequences with GA-rich PAM contexts.
For maximal on-target efficiency with canonical PAMs: Wild-type SpCas9 remains the most efficient option for NGG PAM sites, with Cas9-NG showing significantly reduced activity (27.3% vs 76.5%) and xCas9 showing equivalent or slightly reduced efficiency at NGG PAMs [62] [18].
For enhanced specificity applications: The eCas9-NG variant, which incorporates high-fidelity mutations (K848A/K1003A/R1060A) into Cas9-NG, provides reduced off-target editing while maintaining NG PAM recognition, though with somewhat lower efficiency [18].
The development of xCas9 and Cas9-NG variants significantly expands the targeting scope of CRISPR-based genome editing in plants. While current implementations show varying efficiencies across different PAM types and plant species, ongoing optimization of expression systems and editor architectures continues to enhance their utility. Researchers should select variants based on their specific PAM requirements and implement appropriate RNA expression systems to maximize editing efficiency for their target applications.
Plant genomes exhibit remarkable diversity in their genomic GC content, which can significantly influence the efficiency and outcome of CRISPR-Cas9 genome editing. The GC content of a target site—the percentage of nitrogenous bases that are either guanine (G) or cytosine (C)—affects DNA melting temperature, secondary structure formation, and chromatin accessibility. These biophysical properties directly impact how effectively the Cas9-sgRNA complex can bind to its target DNA sequence [66] [67]. Research has demonstrated that sgRNAs with very low or very high GC content often exhibit reduced editing efficiency, with optimal performance typically observed in intermediate ranges of 40-60% GC [67]. The challenge is particularly pronounced in plants with GC-rich genomes, where target site accessibility may be limited by compact chromatin structures. Understanding and addressing these constraints is essential for developing effective editing strategies across diverse plant species, from Arabidopsis to GC-rich monocots.
Comparative Analysis of CRISPR Systems and GC Content Tolerance
Standard CRISPR-Cas9 Systems
The protospacer adjacent motif (PAM) requirement of standard Streptococcus pyogenes Cas9 (SpCas9) is 5'-NGG-3', which inherently biases targeting toward GC-rich regions [1]. This PAM constraint, combined with the need for optimal sgRNA binding, means that SpCas9 efficiency is highly dependent on GC content. The sgRNA spacer sequence can be divided into two functional segments: a PAM-distal region (nucleotides 1-13) that can tolerate some sequence variation, and a PAM-proximal "seed" region (nucleotides 14-20) where mismatches typically disrupt Cas9 binding [1]. This seed region requirement means that high GC content in this segment is particularly critical for successful editing. Studies across multiple plant species have confirmed that editing efficiency drops significantly when GC content falls outside the 40-60% optimal range [67].
Table 1: Performance of Cas9 Variants Under Different GC Content Conditions
| Editor/Variant | PAM Requirement | Optimal GC Range | Editing Efficiency in High GC | Primary Applications |
|---|---|---|---|---|
| SpCas9 (Standard) | NGG | 40-60% | Moderate (varies by species) | Gene knockouts, multiplex editing |
| dCas9 | NGG | 40-60% | Moderate | Gene repression, epigenome editing |
| nCas9 | NGG | 40-60% | Moderate | Base editing, prime editing |
| CBE (CBE4max) | NGG | 45-70% | High (up to 89% efficiency) | C-to-T conversions, crop improvement |
| ABE | NGG | 45-70% | High | A-to-G conversions, trait development |
| CGBE | NGG | 45-70% | Moderate to High | C-to-G conversions, herbicide resistance |
| evoFERNY-BE4max | NGG | 50-80% | Very High (70% at HEK3 locus) | GC-rich target sites, specialized applications |
Base Editing Systems and GC Optimization
Base editing technologies represent a significant advancement for applications requiring precise single-nucleotide changes, with distinct considerations for GC content. Cytosine base editors (CBEs) and adenine base editors (ABEs) have demonstrated particularly robust activity in GC-rich contexts, with optimized systems achieving efficiencies up to 89% in plant systems [68]. The editing window for these systems—typically 4-5 nucleotides in the spacer region—means that the local GC environment surrounding the target base critically influences deaminase activity [68] [40].
Recent engineering efforts have specifically addressed GC-rich challenges through multiple strategies. The evolution of novel deaminases such as evoFERNY-BE4max has demonstrated remarkable improvements in GC-rich contexts, achieving 70% editing efficiency at the challenging HEK3 locus compared to just 2.3% with previous editors [68]. Similarly, the development of TadA-derived cytidine deaminases (TadCBE) has provided editors with maintained or enhanced activity in high-GC regions while offering reduced off-target effects and smaller construct size [68]. For C-to-G transversion editing, CGBE systems have been optimized through the incorporation of cold-adapted uracil DNA glycosylase (coUNG) from Gadus morhua, which demonstrated 1.71- to 2.54-fold increases in editing efficiency compared to systems using human UNG [69].
Table 2: Experimental Efficiency Metrics Across GC Ranges in Plant Systems
| Editing System | Low GC (<40%) Efficiency | Medium GC (40-60%) Efficiency | High GC (>60%) Efficiency | Notable Plant Applications |
|---|---|---|---|---|
| SpCas9 (Standard) | 5-25% (high variability) | 40-85% (optimal range) | 10-45% (species-dependent) | Banana PDS editing [38] |
| CBE (BE4max) | 15-30% | 50-90% | 20-60% | Rice, wheat, maize improvement [68] |
| ABE | 10-35% | 45-80% | 15-50% | Herbicide resistance [68] |
| CGBE (CDc-CGBEco) | 8-22% | 26-52% | 12-30% | Soybean, tobacco, rice [69] |
| evoAPOBEC1-BE4max | 25-45% | 55-90% | 40-75% | GC-rich sites in multiple crops [68] |
Experimental Protocols for GC-Rich Targets
High-Efficiency Base Editing in GC-Rich Contexts
The following protocol, adapted from recent studies optimizing base editing for high-GC plant genomes, provides a framework for achieving efficient editing in challenging contexts [68] [69]:
Vector Construction:
- Backbone Selection: Utilize plant-optimized binary vectors such as pMDC32 or pHM1 for broad host range applications. For high-GC targets, incorporate the CBE4max architecture or similar optimized systems.
- Promoter Optimization: For dicots, use the UBQ10 promoter; for monocots, employ the maize UBIQUITIN promoter to drive Cas9/nCas9 expression. These promoters provide strong, constitutive expression necessary for challenging edits.
- Nuclear Localization Signals (NLS): Implement a bipartite NLS (bpNLS) at both N- and C-termini of the base editor to enhance nuclear import, particularly critical for GC-rich targets with potentially reduced accessibility.
- sgRNA Scaffold: Employ modified sgRNA scaffolds (e.g., gRNA 2.0) with stem-loop optimizations to enhance stability and binding in GC-rich contexts.
Plant Transformation and Selection:
- Delivery Method: Use Agrobacterium-mediated transformation (strain AGL1 or EHA105) for dicots and embryogenic cell suspension transformation for monocots.
- Selection Strategy: Apply appropriate antibiotic selection (hygromycin for pMDC32-based vectors) with progressive increase in concentration over 4-8 weeks.
- Regeneration: Transfer putative transformants to regeneration medium containing plant growth regulators (auxins and cytokinins) appropriate for the target species.
Editing Validation:
- Primary Screening: Conduct band-shift PCR or restriction fragment length polymorphism analysis for initial identification of edited events.
- Sequencing Confirmation: Perform Sanger sequencing of PCR amplicons spanning the target region, complemented by deep amplicon sequencing to quantify editing efficiency and detect byproducts.
- Phenotypic Assessment: For visible phenotypes like the PDS gene, document albinism or variegation as preliminary evidence of successful editing [38].
GC-Rich Genome Editing Workflow
Multiplexed Editing in GC-Rich Regions
For complex traits controlled by multiple genes or regulatory elements, multiplexed editing in GC-rich regions requires specialized approaches:
sgRNA Design Considerations:
- GC-Balanced Spacers: Design sgRNAs with GC content between 40-60% for each target, avoiding extreme values that compromise efficiency.
- Seed Region Optimization: Ensure perfect complementarity in the seed region (positions 14-20) adjacent to PAM, with particular attention to GC distribution.
- Structural Prediction: Utilize computational tools (e.g., CRISPR-P, CCTop) to predict secondary structures that might impede access to GC-rich targets.
Experimental Implementation:
- Vector Assembly: Employ Golden Gate cloning for multiplex constructs, assembling individual sgRNA expression cassettes into a single binary vector [38].
- Expression Optimization: Use polymerase II promoters for coordinated expression or polymerase III promoters (U6, U3) for individual sgRNA control.
- Efficiency Balancing: For targets with varying GC content, position sgRNAs with lower predicted efficiency (due to suboptimal GC) closer to the left side of polycistronic tRNA-gRNA arrays to capitalize on higher expression of upstream units.
The Scientist's Toolkit: Essential Reagents for GC-Rich Genome Editing
Table 3: Key Research Reagent Solutions for Plant Genome Editing
| Reagent/Category | Specific Examples | Function/Application | GC-Rich Considerations |
|---|---|---|---|
| Base Editor Systems | CBE4max, evoFERNY-BE4max, CDc-CGBEco | Precision nucleotide conversion without double-strand breaks | Enhanced performance in high-GC contexts; specialized deaminases for challenging targets |
| Optimized Cas Variants | nCas9 (D10A), dCas9 (D10A/H840A) | Nickase activity for base editing; binding without cleavage | Maintains targeting capability in GC-rich regions with reduced DNA damage response |
| Vector Backbones | pMDC32, pHM1, pYPQ series | Delivery of editing components to plant cells | Broad host range compatibility; optimized for diverse plant species with varying GC content |
| Promoter Systems | UBQ10, maize UBIQUITIN, RecA | Drive expression of editing components | Strong, constitutive expression necessary for efficient editing in compact chromatin |
| Terminator Sequences | NosT, OCS terminator | Proper transcription termination | Enhances expression stability across genomic contexts |
| Selection Markers | Hygromycin resistance, sacB counter-selection | Identification of transformed events | Allows for efficient recovery of edited lines despite potential reduced transformation efficiency in GC-rich regions |
| sgRNA Scaffolds | gRNA 2.0, modified structures | Guide Cas proteins to target sites | Enhanced stability and binding affinity in GC-rich environments |
The optimization of genome editing tools for varying GC contexts represents an ongoing challenge with significant implications for crop improvement. While current base editing systems have made substantial progress in addressing GC-rich targets, further advances in editor architecture, delivery methods, and computational prediction will continue to expand the targeting scope. The integration of machine learning approaches for sgRNA efficiency prediction in species-specific genomic contexts shows particular promise for addressing current limitations [67] [70]. As these technologies mature, researchers will be increasingly equipped to address previously intractable targets in plant genomes, accelerating the development of crops with enhanced climate resilience, nutritional quality, and sustainable production characteristics.
In CRISPR-based genome editing systems, the guide RNA (gRNA) serves as the essential targeting component that directs CRISPR-associated (Cas) nucleases to specific genomic loci. The design of the gRNA sequence fundamentally determines both the efficiency (on-target activity) and specificity (minimization of off-target effects) of any CRISPR experiment [32] [71]. While all CRISPR systems require some form of guide RNA, the most commonly used format is the single guide RNA (sgRNA), an engineered fusion of the CRISPR RNA (crRNA) containing the target-specific sequence and the trans-activating crRNA (tracrRNA) that serves as a scaffold for Cas protein binding [1] [71]. For plant research, particularly in complex polyploid crops like wheat, gRNA design presents unique challenges due to larger genomes with higher proportions of repetitive DNA and homeologous genes across subgenomes, making specificity a paramount concern [32]. This guide examines the principles underlying high-functioning gRNA design, comparing various optimization strategies and their applications in plant editing research.
Core Principles of gRNA Design
Fundamental Components and Design Considerations
Effective gRNA design requires careful attention to several sequence-specific and structural factors:
PAM Specificity: The Protospacer Adjacent Motif (PAM) sequence is essential for Cas protein recognition and binding. Different Cas proteins recognize distinct PAM sequences; for example, the commonly used SpCas9 from Streptococcus pyogenes requires a 5'-NGG-3' PAM immediately downstream of the target sequence [1] [71]. The PAM itself is not part of the gRNA sequence but must be present in the target DNA.
Seed Region Importance: The 10-12 nucleotides proximal to the PAM sequence, known as the "seed region," are particularly critical for target recognition and cleavage efficiency. Mismatches in this region typically disrupt Cas9 binding more significantly than mismatches in the distal region [1].
GC Content Optimization: The GC content of the gRNA spacer sequence significantly impacts stability and performance. Optimal GC content typically falls between 40-80%, with extreme values (either too high or too low) potentially reducing editing efficiency [71].
Specificity Considerations: The gRNA sequence should be unique within the genome to minimize off-target effects. This is especially crucial in polyploid plants like wheat, where similar sequences may exist across multiple subgenomes [32].
Physical and Structural Properties Affecting gRNA Function
Beyond the primary sequence, the structural properties of gRNAs influence their functionality:
Secondary Structure Stability: The folding stability of both the spacer sequence and the scaffold region affects gRNA-Cas protein binding and target DNA recognition. gRNAs with propensity to form stable internal secondary structures may exhibit reduced editing efficiency [32].
Gibbs Free Energy: The thermodynamic properties of gRNA-DNA duplex formation can predict on-target efficiency, with optimal binding energies typically associated with higher success rates [32].
gRNA Length: While standard SpCas9 gRNAs typically employ 20-nucleotide spacers, fine-tuning the length (commonly between 17-23 nucleotides) can optimize activity and specificity for particular applications [71] [72].
Comparative Analysis of gRNA Optimization Strategies
Recent advances have produced multiple strategies to enhance gRNA performance. The table below summarizes the key approaches, their mechanisms, and comparative performance characteristics.
Table 1: Comparison of gRNA Optimization Strategies for Enhanced Specificity and Efficiency
| Strategy | Mechanism of Action | Specificity Improvement | Efficiency Impact | Primary Applications |
|---|---|---|---|---|
| Hybrid DNA-RNA gRNAs [73] | Strategic substitution of RNA nucleotides with DNA in spacer region | Dramatically reduced off-target editing; reduced bystander editing in base editing | Maintains or increases on-target editing; reduces unwanted bystander edits | Adenine base editing therapies; correction of point mutations |
| Circular gRNAs (cgRNAs) [72] | Covalently closed circular structure resistant to exonuclease degradation | Slightly reduced specificity observed in some transcriptional activation contexts | 1.2-2.5-fold enhancement in editing efficiency; prolonged activity duration | Cas12f systems; gene activation; base editing with compact Cas variants |
| Engineered Scaffolds [1] [72] | Optimization of tracrRNA sequence and structure | Improved specificity through enhanced Cas protein binding | Increased editing efficiency across diverse targets | Multiple CRISPR systems including Cas9, Cas12f |
| Computational Design Tools [74] [32] | Genome-wide specificity prediction and off-target assessment | Significant reduction in potential off-target sites | Maintenance of high on-target activity with optimized sequences | Design of high-specificity gRNA libraries; polyploid crops |
Hybrid DNA-RNA gRNAs for Base Editing Applications
The strategic replacement of specific RNA nucleotides with DNA in the gRNA spacer sequence represents a particularly promising approach for improving base editor specificity. Research on adenine base editing therapies for phenylketonuria (PKU) and pseudoxanthoma elasticum (PXE) demonstrated that hybrid gRNAs with specific DNA substitutions at positions 3-10 of the spacer sequence dramatically reduced off-target editing while maintaining high on-target correction rates [73]. In these studies, the most effective hybrid gRNAs reduced off-target editing from 1.3% to nearly undetectable levels while simultaneously reducing unwanted bystander edits at the on-target site from 4.4% to approximately 1% [73]. This dual benefit of enhanced specificity and reduced bystander editing makes hybrid gRNAs particularly valuable for therapeutic applications where precision is critical.
Circular gRNAs for Enhanced Stability and Efficiency
Engineering circular gRNAs (cgRNAs) represents an innovative approach to address the limited half-life of conventional linear gRNAs. The covalently closed circular structure provides enhanced resistance to exonuclease degradation, resulting in significantly greater stability and prolonged activity [72]. In direct comparisons, cgRNAs exhibited 194.6-fold higher expression levels than linear gRNAs and maintained functional activity for extended durations (up to 7 days versus 6 days for normal gRNAs) [72]. This enhanced stability translated to 1.9-19.2-fold improvements in gene activation efficiency and 1.2-2.5-fold enhancements in adenine base editing efficiency when combined with the compact Cas12f system [72]. The optimization of cgRNA components—including spacer length (23 nucleotides proving optimal) and incorporation of flexible poly-AC RNA linkers—further enhanced system performance [72].
Computational Design and Specificity Analysis
Computational tools for gRNA design have evolved significantly to address specificity concerns. GuideScan2 represents a recent advancement in this area, utilizing a memory-efficient algorithm based on the Burrows-Wheeler transform to enable comprehensive genome-wide gRNA design and specificity analysis [74]. This tool allows researchers to identify and avoid gRNAs with numerous potential off-target sites, which is particularly important for plant genomes with high repetition rates. Studies have demonstrated that GuideScan2's specificity scores show significant correlation with experimentally measured specificities (Spearman correlation 0.44, p < 0.001) [74]. The confounding effects of low-specificity gRNAs in CRISPR screens underscore the importance of these computational approaches; in published CRISPRi screens, genes targeted by low-specificity gRNAs were systematically less likely to be identified as hits, potentially due to dilution of the editing machinery across multiple off-target sites [74].
Experimental Protocols for gRNA Evaluation
Protocol for Evaluating Hybrid gRNA Specificity and Efficiency
This protocol is adapted from studies demonstrating reduced off-target editing with DNA-substituted gRNAs in base editing applications [73]:
gRNA Design and Synthesis:
- Design standard RNA gRNAs and hybrid versions with single, double, or triple DNA nucleotide substitutions at positions 3-10 in the spacer sequence.
- Synthesize gRNAs using solid-phase chemical synthesis with appropriate DNA nucleotide incorporation.
Cell Transfection:
- Culture human hepatocyte lines (e.g., HuH-7) homozygous for the target variant.
- Transfect cells with ABE8.8 mRNA in combination with standard or hybrid gRNAs using lipid nanoparticles.
Editing Assessment:
- Perform targeted amplicon sequencing of the on-target site 72 hours post-transfection.
- Analyze sequencing data for corrective editing efficiency and bystander editing rates.
- Utilize ONE-seq (OligoNucleotide Enrichment and sequencing) or similar methods to identify potential off-target sites.
- Perform hybrid capture sequencing of all candidate off-target sites with ONE-seq scores >0.01.
Validation:
- Confirm top-performing hybrid gRNAs in humanized mouse models of target diseases.
- Assess phenotypic reversion and biochemical correction in addition to editing metrics.
Protocol for Circular gRNA Implementation
This protocol outlines the development and testing of circular gRNAs for enhanced stability and efficiency, particularly with compact Cas systems like Cas12f [72]:
cgRNA Construction:
- Design cgRNA expression cassettes using the Tornado system with ribozyme-flanked gRNA sequences.
- Incorporate flexible RNA linkers (5-10 nucleotide poly-AC sequences) between ribozymes and gRNA components.
- Test spacer lengths ranging from 19-23 nucleotides to identify optimal configuration.
Stability Assessment:
- Transfert cgRNA plasmids into reporter cell lines expressing dCas12f-VPR fusion protein.
- Treat cells with actinomycin D to inhibit transcription and assess RNA stability over time.
- Perform RT-PCR with outward-facing primers to verify circularization and quantify expression levels.
Efficiency Evaluation:
- Measure gene activation efficiency via fluorescence-activated cell sorting (FACS) in reporter cell lines.
- Conduct dose-response analyses (8-500 ng in 24-well plates) and time-course experiments (1-7 days).
- Compare cgRNA performance against normal and linear gRNA controls.
Endogenous Gene Activation:
- Test optimized cgRNAs on endogenous gene targets in multiple cell lines (HEK293T, MCF7, THP1).
- Perform RNA-seq to assess both on-target activation and genome-wide specificity.
gRNA Design Workflow and Strategic Implementation
The following diagram illustrates the comprehensive workflow for designing and optimizing high-specificity gRNAs, integrating multiple strategies discussed in this guide:
Essential Research Reagent Solutions
The table below catalogues key reagents and tools essential for implementing advanced gRNA design strategies in plant genome editing research.
Table 2: Research Reagent Solutions for Advanced gRNA Design and Validation
| Reagent/Tool | Function | Application Notes |
|---|---|---|
| GuideScan2 Software [74] | Genome-wide gRNA design and specificity analysis | Enumerates potential off-targets; 50x memory improvement over original GuideScan; essential for polyploid crops |
| Hybrid DNA-RNA gRNAs [73] | Reduce off-target and bystander editing in base editors | Position-specific DNA substitutions (positions 3-10) most effective; compatible with mRNA-LNP delivery |
| Circular gRNA Expression System [72] | Enhance gRNA stability and longevity | Tornado system with ribozyme flanking; optimal with 23-nt spacer and AC5/AC10 linkers; particularly beneficial for Cas12f |
| WheatCRISPR Software [32] | Species-specific gRNA design for polyploid crops | Accounts for homeologous genes across A, B, D subgenomes; essential for minimizing off-targets in wheat |
| ONE-seq Assay [73] | Comprehensive identification of off-target sites for base editors | Specifically tailored for base editing systems rather than conventional CRISPR-Cas9; more accurate than GUIDE-seq for BE applications |
| High-Specificity gRNA Libraries [74] | Pre-designed libraries with minimized off-target potential | 6 gRNAs per gene with safe-harbor-targeting and non-targeting controls; reduces confounding effects in screens |
The strategic design of guide RNAs represents a critical determinant of success in plant genome editing applications. For researchers working with complex plant genomes, particularly polyploid species like wheat, a multi-faceted approach incorporating computational design tools, novel gRNA architectures, and rigorous validation protocols provides the most reliable path to achieving high specificity and efficiency. The continuing evolution of gRNA optimization strategies—from hybrid DNA-RNA designs to circular gRNAs—offers powerful solutions to the persistent challenges of off-target editing and variable efficiency. By systematically implementing these principles and leveraging the appropriate reagent solutions, plant researchers can maximize editing precision while minimizing unintended effects, accelerating the development of improved crop varieties through precision genome editing.
Efficient delivery of CRISPR-Cas systems into plant cells remains a significant bottleneck in agricultural biotechnology. While CRISPR technology has revolutionized plant genome editing, enabling precise genetic modifications for crop improvement, the delivery of editing components faces unique physiological barriers not present in animal systems. The plant cell wall presents a primary obstacle, physically blocking the entry of large biomolecules and nanoparticle complexes. Furthermore, the absence of conserved endocytic pathways complicates intracellular trafficking, and the central vacuole in many plant cells can sequester or degrade delivered cargo. Tissue culture steps introduce additional complications, including genotype-dependent regeneration efficiency and somaclonal variation. This guide objectively compares current delivery systems—focusing on nanoparticle-based approaches and tissue culture optimization—by analyzing experimental data to provide researchers with validated protocols for enhancing editing efficiency in plants.
Comparative Analysis of Genome Editing Systems
CRISPR-Cas Variants and Architectures
Table 1: Comparison of Key CRISPR-Cas Systems for Plant Editing
| System | Key Components | Editing Type | Typical Efficiency in Plants | Primary Applications | Key Advantages | Main Limitations |
|---|---|---|---|---|---|---|
| CRISPR-Cas9 | Cas9 nuclease, sgRNA | DSBs, knockouts | 10-90% (species-dependent) [75] | Gene knockout, large deletions | Well-established, high efficiency | Off-target effects, PAM restriction |
| Base Editors | Cas9 nickase, deaminase | Single nucleotide conversion | 5-50% [40] | Point mutations, SNP correction | No DSBs, reduced off-targets | Limited to specific base changes, bystander edits |
| Prime Editors | Cas9 nickase-RT fusion, pegRNA | All single-base changes, small insertions/deletions | 10-70% (PE1-PE7 variants) [40] | Precise editing, transition/transversion | No DSBs, highly versatile, >90% of known pathogenic mutations | Complex pegRNA design, lower efficiency in some systems |
| Cas12 Systems | Cas12a, crRNA | DSBs, multiplex editing | 15-60% [75] | Multiplex editing, transcriptional regulation | Shorter crRNAs, staggered cuts, multiplexing | Lower efficiency than Cas9 in some plants |
The evolution of prime editing systems demonstrates significant progress in overcoming precision limitations. Initial PE1 systems showed modest efficiency (~10-20% in HEK293T cells), while subsequent PE2 systems incorporating engineered reverse transcriptase improved this to 20-40% [40]. The PE3 system introduced an additional sgRNA to nick the non-edited strand, boosting efficiency to 30-50% [40]. More recently, PE4 and PE5 systems incorporating dominant-negative MLH1 to suppress mismatch repair achieved 50-80% efficiency, while PE6 and PE7 systems with compact reverse transcriptase variants and stabilized pegRNAs reached 70-95% efficiency in mammalian systems [40]. While these efficiencies are reported in mammalian cells, they represent the potential trajectory for plant prime editing optimization.
Delivery System Performance Metrics
Table 2: Delivery System Efficiency Comparison in Plant Systems
| Delivery Method | Cargo Format | Model Plant | Editing Efficiency | Regeneration Efficiency | Throughput | Key Experimental Findings |
|---|---|---|---|---|---|---|
| Agrobacterium-mediated | DNA, RNA | Tobacco, Rice | 30-95% [75] | Medium-High | Medium | Most established method; T-DNA integration; genotype dependence |
| Biolistic Particle Delivery | DNA, RNP | Maize, Wheat | 10-80% [75] | Low-Medium | Low | Direct physical delivery; cell damage; high copy integration |
| Nanoparticle-mediated | DNA, RNP | Maize, Tomato | 15-60% [75] | Medium | High | Protects cargo; customizable; emerging technology |
| PEG-mediated Protoplast Transfection | RNP | Lettuce, Arabidopsis | 20-90% [75] | Low (regeneration challenging) | High | High editing efficiency; difficult regeneration |
| Virus-Based Systems | DNA, RNA | Nicotiana, Solanaceae | 40-95% [75] | Not applicable | High | Systemic movement; no integration needed; limited cargo size |
Nanoparticle delivery systems show particular promise for RNP delivery, which offers transient activity that minimizes off-target effects. Lipid nanoparticles (LNPs) have demonstrated efficient RNP encapsulation and delivery, with studies showing tissue-specific editing in animal models [76]. In plants, nanoparticle-mediated delivery protects CRISPR components from degradation and enhances cellular uptake, significantly improving trait enhancement in maize [75]. The editing efficiency varies based on nanoparticle composition, with cationic lipids and polymers facilitating better endosomal escape through the proton sponge effect [77].
Experimental Protocols and Methodologies
Nanoparticle-Mediated Delivery Protocol
Protocol: LNP-RNP Delivery for Plant Protoplasts
Materials: Cas9 protein, sgRNA, cationic lipids (DOTAP, DOPE), microfluidic device, plant protoplasts, tissue culture media.
Step 1: RNP Complex Formation
- Incubate purified Cas9 protein (5μM) with sgRNA (7.5μM) in nuclease-free buffer at room temperature for 15 minutes to form RNP complexes [76].
Step 2: Nanoparticle Formulation
- Prepare lipid mixture (ionizable cationic lipid:phospholipid:cholesterol:PEG-lipid = 50:10:38.5:1.5 mol%) in ethanol [77].
- Use microfluidic device to mix lipid solution with aqueous RNP solution at 3:1 flow rate ratio (total flow rate 12 mL/min) [76].
- Dialyze resulting LNPs against PBS pH 7.4 overnight to remove ethanol.
Step 3: Characterization and Quality Control
Step 4: Protoplast Transfection
- Incubate 10^5 protoplasts with LNP-RNPs (50μg total lipid) in 1mL osmoticum for 30 minutes.
- Add PEG solution (40% final concentration) for 5 minutes to enhance uptake [75].
- Wash carefully and culture in regeneration media.
Validation: Assess editing efficiency 48-72 hours post-transfection using T7EI assay or sequencing. Expected efficiency: 40-60% in amenable species [75].
Tissue Culture Optimization Strategy
Integrated Workflow for Regeneration of Edited Plants
The experimental workflow for regeneration highlights critical optimization points that significantly impact editing success. Genotype-specific media requirements necessitate small-scale trials for each plant variety, as responses to hormone combinations vary substantially [75]. Selection timing proves crucial—initiating selection too early reduces survival of edited cells, while delayed selection permits chimerism. The hormone balance during regeneration must be precisely calibrated, with cytokinin-rich media promoting shoot formation and auxin-rich media supporting root development [75].
Research Reagent Solutions
Table 3: Essential Reagents for Delivery and Tissue Culture Optimization
| Reagent/Category | Specific Examples | Function | Application Notes |
|---|---|---|---|
| CRISPR Nucleases | SpCas9, Cas12a, HiFi Cas9 | DNA recognition and cleavage | High-fidelity variants reduce off-targets; Cas12a for multiplexing [78] |
| Editing Reagents | Base editors (ABE, CBE), Prime editors | Precision editing without DSBs | PE systems require optimized pegRNA design [40] |
| Delivery Materials | Cationic lipids (DOTAP), Polyethyleneimine (PEI), Gold nanoparticles | Cargo encapsulation and delivery | LNPs suitable for RNP delivery; size <100nm optimal [77] [75] |
| Tissue Culture Media | MS Media, B5 Vitamins, Plant Growth Regulators | Support cell growth and regeneration | Hormone combinations must be genotype-specific [75] |
| Selection Agents | Antibiotics (Kanamycin), Herbicides (Bialaphos) | Selection of transformed cells | Concentration must be determined empirically [75] |
| Analytical Tools | T7 Endonuclease I, Surveyor Assay, PCR Reagents | Detection of editing events | Next-generation sequencing provides most accurate efficiency data [79] |
The optimal delivery strategy for plant genome editing depends on the specific research objectives, plant species, and available resources. Nanoparticle-based RNP delivery offers advantages for reducing off-target effects and overcoming regulatory concerns regarding foreign DNA integration. However, Agrobacterium-mediated transformation remains the most efficient method for many dicot species. For challenging transformations, a combined approach using nanoparticle delivery followed by optimized tissue culture protocols can maximize success. As the field advances, integrating these delivery platforms with high-fidelity editing systems and genotype-independent regeneration protocols will ultimately overcome the persistent barriers in plant genetic engineering. Researchers should prioritize establishing efficient tissue culture systems for their target species before investing in advanced editing approaches, as regeneration efficiency often proves to be the ultimate limiting factor rather than editing efficiency itself.
Benchmarking Performance: Comparative Analysis of Editing Outcomes
The CRISPR-Cas9 system has revolutionized plant genetic research and crop improvement, with the editing efficiency of its core component, the Cas9 nuclease, being a critical determinant of success [53]. However, the foundational wild-type Streptococcus pyogenes Cas9 (SpCas9) presents limitations, including a constrained protospacer adjacent motif (PAM) recognition and potential for off-target effects [80]. To overcome these challenges, numerous Cas9 variants have been engineered, leading to a complex landscape of options for plant researchers. This guide provides a objective, data-driven comparison of these variants, focusing on their editing efficiencies, specificities, and PAM compatibilities within plant systems. Such a comparison is essential for selecting the optimal Cas9 variant for specific experimental or breeding objectives in plants, thereby accelerating functional genomics and the development of improved crop varieties.
At a Glance: Key Cas9 Variants and Their Characteristics
The table below summarizes the core features and performance metrics of major Cas9 variants relevant to plant genome editing.
Table 1: Head-to-Head Comparison of Cas9 Variants for Plant Genome Editing
| Cas9 Variant | Key Mutations/Features | PAM Compatibility | Editing Efficiency | Specificity (Off-Target Reduction) | Primary Application in Plants |
|---|---|---|---|---|---|
| Wild-Type SpCas9 | N/A | NGG | High, but variable across loci [2] | Baseline off-target activity [80] | Standard gene knockouts [53] |
| High-Fidelity Variants | |||||
| › eSpCas9(1.1) | K848A, K1003A, R1060A [81] | NGG | Reduced compared to SpCas9 [81] [80] | High [80] | Applications requiring high specificity [80] |
| › SpCas9-HF1 | N497A, R661A, Q695A, Q926A [81] | NGG | Reduced compared to SpCas9 [80] | High [80] | Applications requiring high specificity [80] |
| › HypaCas9 | N692A, M694A, Q695A, H698A [81] | NGG | Comparable to SpCas9 at some sites [81] | High [80] | Balanced high efficiency and fidelity [81] |
| PAM-Flexible Variants | |||||
| › xCas9(3.7) | --- | NG, GAA, GAT [80] | Lower than SpCas9 at NGG sites [80] | Moderate [80] | Expanding targeting scope [80] |
| › SpCas9-NG | --- | NG [80] | Lower than SpCas9 at NGG sites [80] | Moderate [80] | Expanding targeting scope [80] |
| › SpRY | --- | NRN > NYN (near PAM-less) [80] | Lower than SpCas9 at NGG sites [80] | Lower (increased off-targets) [80] | Maximum targeting freedom [80] |
| AI-Engineered Variants | |||||
| › AncBE4max-AI-8.3 | Multiple AI-predicted mutations [82] | NGG | 2-3 fold increase over AncBE4max base editor [82] | Data pending | High-efficiency base editing [82] |
| Loop-Engineered Variants | |||||
| › AtCas9-Z7 | Mesophilic loop transplantation [83] | NNG (Expanded) | Highly improved over WT AtCas9 [83] | Data pending | Editing in challenging conditions [83] |
Quantitative Data from Plant and Mammalian Systems
Efficiency and Specificity at NGG PAMs
A systematic evaluation of high-fidelity variants in HEK293T cells revealed that while most exhibit significantly reduced off-target activity, their on-target efficiency can be compromised [80]. For instance, at the EMX1 target site, SpCas9 achieved 43.6% editing, whereas eSpCas9(1.1) and SpCas9-HF1 showed reduced efficiencies of 35.8% and 29.4%, respectively. In contrast, HypaCas9 maintained robust on-target cleavage (42.6%) while also demonstrating high fidelity [80]. This trade-off between efficiency and specificity is a critical consideration for plant researchers.
Performance in Plant Base Editing Systems
In rice, the choice of cytidine deaminase fused to Cas9 nickase significantly impacts efficiency. A PmCDA1-based editor (SpCas9-pBE) demonstrated 3 to 10-fold higher editing efficiency than an rAPOBEC1-based editor (SpCas9-rBE) across seven targets, with efficiencies ranging from 5% to 100% [81]. When high-fidelity variants were applied to base editing in rice, their performance varied drastically: HypaCas9-pBE maintained high efficiency (53-87% at tested sites), SpCas9-HF2-pBE showed reduced activity, and eSpCas9(1.1)-pBE showed no detectable base-editing ability at the targets assessed [81].
PAM Flexibility and Associated Trade-Offs
The development of PAM-flexible variants like SpG (NGN PAM) and SpRY (NRN>NYN PAM) has greatly expanded the targeting scope of CRISPR in plants [80]. However, this expanded range comes with a cost. These variants generally exhibit lower editing efficiency at their non-canonical PAMs compared to SpCas9 at NGG sites. Furthermore, they display increased off-target activity, with SpRY showing the highest level of promiscuity due to its relaxed PAM requirement [80].
Detailed Experimental Protocols for Efficiency Assessment
To ensure the reliability and reproducibility of the data presented in this guide, understanding the underlying experimental methodologies is essential. The following workflows are commonly used for benchmarking Cas9 variants.
Transient Expression in Plant Leaves
This protocol uses a modified dual geminiviral replicon (GVR) system for rapid, high-level co-expression of Cas9 and sgRNAs in Nicotiana benthamiana leaves, bypassing the need for stable transformation [2].
Diagram 1: Workflow for transient expression assay in plants.
Key Steps:
- Vector Construction: The genes encoding the SpCas9 variant and the single guide RNA (sgRNA) are cloned into separate Bean Yellow Dwarf Virus (BeYDV)-based geminiviral replicon vectors. The Cas9 is typically driven by the Cauliflower Mosaic Virus 35S (CaMV 35S) promoter, while the sgRNA is expressed under a U6 promoter [2].
- Plant Agroinfiltration: The two binary vectors are transformed into Agrobacterium tumefaciens. The bacterial cultures are then mixed and infiltrated into the leaves of young N. benthamiana plants to deliver the T-DNAs into plant cells [2].
- Tissue Harvest and DNA Extraction: After a 7-day incubation period to allow for gene expression and genome editing, the infiltrated leaf tissue is harvested. Genomic DNA is extracted from a pooled sample of this tissue [2].
- Efficiency Quantification: The target genomic region is amplified by PCR and subjected to one of several quantification methods, such as targeted amplicon sequencing (AmpSeq), which is considered the "gold standard" for its sensitivity and accuracy [2].
High-Throughput Specificity Assessment (PEM-seq)
Primer-extension-mediated sequencing (PEM-seq) is a powerful method to comprehensively profile editing outcomes, including off-target effects and genomic rearrangements, in mammalian cells [80]. While demonstrated in HEK293T cells, the principles are applicable to plant research.
Key Steps:
- Cell Transfection and Sorting: HEK293T cells are co-transfected with plasmids expressing the Cas9 variant (fused to mCherry) and the sgRNA (fused to GFP). After 72 hours, successfully transfected cells are isolated using fluorescence-activated cell sorting (FACS) based on mCherry and GFP signals [80].
- Library Preparation and Sequencing: Genomic DNA is extracted from the sorted cells. A biotinylated primer is used for in situ primer extension from the Cas9 target site, followed by pull-down, circularization, and amplification to create a sequencing library that captures fragments with double-strand breaks [80].
- Data Analysis: High-throughput sequencing data is analyzed to identify:
- Small Indels: At the on-target and potential off-target sites.
- Translocations: Resulting from erroneous repair of concurrent double-strand breaks.
- Large Deletions: At the target site [80].
The Scientist's Toolkit: Essential Research Reagents
The following table lists key reagents and materials required for conducting comparative studies of Cas9 variants in plant systems, as derived from the cited methodologies.
Table 2: Essential Reagents for Cas9 Variant Benchmarking in Plants
| Reagent / Material | Function / Description | Example Use Case |
|---|---|---|
| Geminiviral Replicon Vectors | Binary vectors for high-level transient expression of Cas9 and sgRNAs in plant cells [2]. | High-efficiency transient expression in N. benthamiana [2]. |
| Agrobacterium tumefaciens | A bacterial strain used to deliver T-DNA containing CRISPR constructs into plant cells. | Standard delivery method for stable transformation or transient expression [2]. |
| Next-Generation Sequencing (NGS) | Enables high-sensitivity quantification of editing outcomes and off-target detection [2] [80]. | Targeted amplicon sequencing (AmpSeq) for gold-standard efficiency measurement [2]. |
| Droplet Digital PCR (ddPCR) | Provides absolute quantification of editing events with high precision without the need for NGS [2]. | Accurate quantification of editing efficiency in heterogeneous samples [2]. |
| T7 Endonuclease I (T7E1) | Enzyme that cleaves mismatched heteroduplex DNA, allowing for indirect detection of indels [81]. | Low-cost, rapid initial assessment of editing activity [81]. |
| sgRNA Expression Constructs | Plasmids containing the U6 promoter for Pol III-driven expression of guide RNAs. | Co-delivery with Cas9 vectors to form the functional editing complex. |
| Plant Culture Media & Selectable Markers | Media for callus induction, regeneration, and antibiotics/herbicides for selecting transformed events. | Stable transformation of rice and other crops [81]. |
The landscape of Cas9 variants offers plant researchers a diverse toolkit tailored to different experimental needs. The choice of variant involves a careful balance between editing efficiency, target specificity, and PAM compatibility. High-fidelity variants like HypaCas9 are excellent for applications where minimizing off-target effects is paramount, even with a potential slight reduction in efficiency. For projects requiring maximal targeting scope, PAM-flexible variants like SpRY are invaluable, though researchers must be vigilant about their increased off-target potential. Emerging engineering strategies, particularly AI-guided design [82] and loop engineering [83], show great promise in breaking these trade-offs by creating variants with simultaneously enhanced efficiency, specificity, and range. The optimal Cas9 variant is ultimately dictated by the specific research question, target sequence, and desired outcome in the plant system of interest.
The application of CRISPR-Cas systems has revolutionized plant functional genomics and precision breeding, yet concerns regarding off-target effects persist within the scientific community. While early studies in mammalian systems reported significant off-target activity, emerging evidence in plants suggests a different landscape where off-target mutations are considerably rarer [84] [85]. This discrepancy underscores the critical importance of robust, comprehensive assessment methods to accurately characterize editing specificity across diverse plant species and CRISPR systems. Among the available detection technologies, whole-genome sequencing (WGS) has emerged as the gold standard for unbiased, genome-wide evaluation of off-target effects, providing researchers with a complete picture of unintended mutations resulting from genome editing procedures [86] [84].
The specificity challenge is further complicated by the diversity of CRISPR systems now available. While early plant editing efforts primarily utilized Streptococcus pyogenes Cas9 (SpCas9), recent advances have introduced numerous alternatives, including high-fidelity Cas9 variants, Cas12a nucleases, and prime editing systems, each with distinct specificity profiles [87] [40] [88]. This evolving toolkit necessitates systematic comparison to guide researchers in selecting appropriate systems for their applications. Within this context, WGS provides the comprehensive data required for rigorous comparison of these systems, enabling evidence-based decisions in experimental design.
This review provides a comprehensive analysis of WGS methodology for off-target detection in plant systems, benchmarking its performance against alternative detection methods, and presenting experimental evidence of specificity profiles across diverse CRISPR systems. By synthesizing recent findings from multiple plant species, we aim to establish clear guidelines for researchers seeking to validate the precision of their genome editing approaches.
Methodological Comparison: Off-Target Detection Technologies
Multiple methods have been developed to identify off-target effects of CRISPR systems, each with distinct advantages, limitations, and appropriate use cases. Table 1 summarizes the key characteristics of major detection methodologies.
Table 1: Comparison of Off-Target Detection Methods
| Method | Principle | Advantages | Disadvantages | Plant Studies Using Method |
|---|---|---|---|---|
| Whole-Genome Sequencing (WGS) | Sequences entire genome of edited and control plants | Unbiased, comprehensive detection of all mutation types | Higher cost, computational complexity, requires reference genome | Grapevine [85], Physcomitrium patens [84] |
| GUIDE-seq | Captures double-strand breaks via integration of double-stranded oligodeoxynucleotides | Highly sensitive, low false positive rate | Limited by transfection efficiency, not widely used in plants | Primarily mammalian systems [86] |
| CIRCLE-seq / Digenome-seq | In vitro Cas9 digestion of purified genomic DNA followed by sequencing | Highly sensitive, cell-free system | In vitro conditions may not reflect cellular context | Maize [85] |
| In Silico Prediction | Computational prediction of off-target sites based on sequence similarity | Fast, inexpensive, convenient | High false positive and negative rates, misses sgRNA-independent effects | Tomato [89] |
| Targeted Amplicon Sequencing | Deep sequencing of predicted off-target sites | Cost-effective for validating suspected sites | Limited to known/predicted sites, incomplete genome coverage | Tomato [89] |
WGS stands apart from other methods due to its completely unbiased nature, capable of detecting off-target mutations regardless of their sequence similarity to the target site or prior prediction. Unlike GUIDE-seq and CIRCLE-seq, which detect double-strand breaks or in vitro cleavage patterns, WGS directly identifies accumulated mutations in the edited genome, providing a comprehensive record of all variant types, including single nucleotide polymorphisms (SNPs), insertions and deletions (indels), and even structural variations [86] [84]. This comprehensive detection comes with increased computational demands and cost compared to targeted approaches, but provides the most definitive assessment of editing specificity.
The detection workflow for WGS-based off-target analysis involves multiple critical steps, from experimental design through bioinformatic analysis, as illustrated below:
Figure 1: WGS Off-Target Detection Workflow. Critical experimental considerations (yellow) and essential resources (green) are highlighted at relevant workflow stages.
WGS in Practice: Key Experimental Designs and Findings in Plants
Recent applications of WGS for specificity assessment in diverse plant species have yielded crucial insights into the true off-target rates of CRISPR systems. These studies consistently demonstrate that CRISPR-Cas9 exhibits high specificity in plants, with off-target mutations being exceptionally rare when proper experimental controls are implemented.
In a comprehensive study comparing CRISPR-Cas9 and TALEN systems in Physcomitrium patens, researchers performed WGS on edited plants and found "a similar number of differences for the two editing strategies compared to control non-transfected plants, with an average of 8.25 SNVs and 19.5 InDels for the CRISPR-edited plants, and an average of 17.5 SNVs and 32 InDels for the TALEN-edited plants" [84]. Crucially, the study noted that "a comparable number of SNVs and InDels could be detected in the PEG-treated control plants," indicating that the observed mutations were likely attributable to the transformation process or spontaneous mutations rather than off-target activity of the nucleases [84].
Similarly, WGS analysis of CRISPR-Cas9-edited grapevine plants revealed exceptionally high specificity. Researchers sequenced seven edited plants targeting the VvbZIP36 gene and identified "between 202,008 and 272,397 single nucleotide polymorphisms (SNPs) and between 26,391 and 55,414 insertions/deletions (indels) in the seven Cas9-edited grapevine plants compared with the three WT plants" [85]. However, after extensive analysis of 3,272 potential off-target sites, "only one off-target indel mutation was identified from the WGS data and validated by Sanger sequencing" [85]. This finding demonstrates that while numerous genetic variants exist between edited and control plants, truly CRISPR-induced off-target mutations are extremely rare.
Table 2 summarizes quantitative findings from recent WGS studies in plants:
Table 2: WGS Off-Target Detection Results in Plant Studies
| Plant Species | Editing System | Sequencing Depth | Total Detected Variants | Validated Off-Targets | Reference |
|---|---|---|---|---|---|
| Physcomitrium patens | CRISPR-Cas9 | Not specified | Avg. 8.25 SNVs, 19.5 InDels | None significant vs control | [84] |
| Physcomitrium patens | TALEN | Not specified | Avg. 17.5 SNVs, 32 InDels | None significant vs control | [84] |
| Grapevine | CRISPR-Cas9 | Not specified | 202,008-272,397 SNPs, 26,391-55,414 InDels | 1 validated off-target | [85] |
| Tomato | CRISPR-Cas9 | Amplicon sequencing | On-target efficiency: 4.3-64.2% | Off-targets at 1-2 mismatch sites | [89] |
The high specificity observed in these WGS studies contrasts with early concerns about CRISPR off-target effects in plant systems. The evidence suggests that the plant cellular environment, DNA repair mechanisms, or delivery methods may naturally limit off-target effects compared to mammalian systems. Furthermore, these findings highlight the critical importance of including proper controls (non-transfected and transformation-treated plants) in WGS experiments to distinguish true off-target mutations from background genetic variation and tissue culture-induced mutations.
Implementing robust WGS-based off-target assessment requires specific experimental and bioinformatic resources. The following toolkit summarizes essential components for successful specificity analysis:
Table 3: Research Reagent Solutions for WGS Off-Target Analysis
| Category | Specific Resource | Function and Importance |
|---|---|---|
| Sequencing Platforms | Illumina NovaSeq, HiSeq, or NextSeq | Generate high-quality short reads with sufficient depth (typically 30-50x) for variant calling |
| Bioinformatics Tools | BWA (Burrows-Wheeler Aligner) | Align sequencing reads to reference genome with high accuracy and efficiency |
| Bioinformatics Tools | GATK (Genome Analysis Toolkit) | Industry-standard variant discovery with filtering and annotation capabilities |
| Reference Materials | Species-specific reference genome | Essential reference for read alignment and variant calling; quality impacts detection accuracy |
| Experimental Controls | Wild-type non-transformed plants | Baseline for distinguishing natural genetic variation from editing-induced mutations |
| Experimental Controls | Transformation-treated plants | Control for mutations induced by tissue culture, transformation, or delivery method |
| Validation Tools | Sanger sequencing | Independent validation of putative off-target mutations identified through WGS |
The quality of the reference genome significantly impacts the reliability of WGS-based off-target detection. As noted in the grapevine study, genetic variants between the experimental cultivar and the reference genome can create "243 newly generated off-target sites caused by genetic variants between the Thompson Seedless cultivar and the grape reference genome (PN40024)" [85]. Researchers should therefore prioritize using the most complete and contiguous reference genome available for their specific cultivar or generate a custom reference when possible.
Comparing CRISPR Systems: Specificity Evidence from Plant Studies
Beyond standard SpCas9, recent research has characterized the specificity profiles of alternative CRISPR systems in plants. The evidence from multiple studies enables comparative analysis of off-target tendencies across platforms.
Protoplast-based studies in tomato revealed that "off-target mutations were found for 13 out of 89 sgRNAs and only occurred at positions with one or two mismatches (at 14 and 3 sites, respectively)" with no off-target mutations detected at sites with three or more mismatches [89]. This highlights the importance of careful sgRNA design to minimize potential off-target sites with high sequence similarity.
Comparative analysis of delivery methods in the same tomato study found that "the use of RNPs led to a significant decrease in relative off-target frequencies at 6 out of 17, no significant difference at 9, and an increase at 2 sites" [87]. This suggests that ribonucleoprotein (RNP) delivery can modestly reduce but not eliminate off-target effects in some contexts.
Novel Cas9 orthologs with different PAM requirements also demonstrate favorable specificity profiles. The recently characterized LrCas9 from probiotic Lactobacillus rhamnosus, which recognizes a 5'-NGAAA-3' PAM, was shown to mediate efficient editing "with high-fidelity" in rice, wheat, tomato, and Larix cells [88]. The distinct PAM preference of such orthologs naturally limits the potential off-target space compared to SpCas9.
Prime editing systems represent a significant advancement for precision editing, as they "minimize unintended editing and bystander editing" associated with earlier base editing platforms [40]. The development of optimized prime editors like PE5 and PE6 has further enhanced specificity while maintaining efficiency [40]. The relationship between different CRISPR systems and their editing characteristics can be visualized as follows:
Figure 2: CRISPR System Evolution and Specificity Profiles. The development of newer editing platforms (red nodes) has progressively addressed specificity limitations of earlier systems.
Temperature sensitivity represents another factor influencing specificity, particularly for Cas12a systems. Research comparing Cas9 and Cas12a proteins found that "Cas9 and Cas12a RNP delivery resulted in genome editing frequencies (8.7–41.2%) at various temperature conditions, 22°C, 26°C, and 37°C, with no significant temperature sensitivity" [87]. This stability across temperatures is advantageous for maintaining consistent specificity profiles under standard plant growth conditions.
The comprehensive evidence from WGS studies across multiple plant species consistently demonstrates that CRISPR systems, when appropriately designed and implemented, exhibit high specificity with minimal off-target effects. The rare off-target mutations that do occur predominantly manifest at sites with high sequence similarity to the intended target (1-2 mismatches), emphasizing the critical importance of careful sgRNA design and comprehensive in silico prediction before embarking on editing experiments.
WGS remains the definitive method for specificity assessment due to its unbiased, genome-wide coverage, though it requires careful experimental design including proper controls to distinguish true off-target effects from background mutation. As CRISPR technology continues to evolve with novel editors like prime editing systems and Cas9 orthologs with distinct PAM preferences, WGS will play an increasingly important role in characterizing the specificity profiles of these next-generation tools.
For researchers planning genome editing experiments in plants, we recommend selecting high-specificity systems like high-fidelity Cas9 variants or prime editors for applications demanding utmost precision, implementing WGS-based validation for clinical or commercial applications where comprehensive specificity assessment is essential, prioritizing RNP delivery when possible to reduce off-target effects in susceptible systems, and designing sgRNAs with computational tools that minimize potential off-target sites with 1-2 mismatches, particularly in the PAM-proximal region.
As the plant genome editing field progresses toward more complex applications including multiplex editing and chromosome engineering, WGS will continue to provide the critical specificity data needed to ensure both scientific rigor and regulatory compliance.
The advent of CRISPR-Cas9 technology has revolutionized plant biotechnology, offering unprecedented precision for crop improvement. However, traditional delivery methods that rely on DNA vectors for introducing CRISPR components face significant challenges, including random transgene integration, persistent foreign DNA in the genome, and stringent regulatory oversight. DNA-free genome editing using preassembled ribonucleoprotein (RNP) complexes of Cas9 protein and guide RNA has emerged as a powerful alternative that eliminates these concerns by delivering editing machinery transiently without DNA integration [90]. This approach is particularly valuable for vegetatively propagated, highly heterozygous, or recalcitrant plant species where transgene segregation is impractical, enabling researchers to preserve elite genetic backgrounds while introducing targeted mutations [91] [92].
This guide provides a comprehensive comparison of DNA-free RNP systems against alternative editing approaches, with detailed experimental data and protocols to inform researchers' experimental design.
Technical Comparison: RNP Delivery Versus Alternative Editing Strategies
Table 1: Comparative analysis of major genome editing delivery approaches
| Editing Approach | Key Components | Delivery Methods | Editing Efficiency | Transgene Risk | Regulatory Status | Ideal Applications |
|---|---|---|---|---|---|---|
| DNA-Free RNP | Preassembled Cas protein + gRNA | PEG-transfection, Biolistics, Nanoparticles | Variable (e.g., ~19% in raspberry) [91] | None | Favorable in many jurisdictions [90] | Species with heterozygous backgrounds, functional genomics |
| Plasmid-Based CRISPR | DNA encoding Cas9 + gRNA | Agrobacterium, Biolistics | High, but variable | Stable integration possible | Often regulated as GMO | High-throughput transformations, HDR editing |
| Base Editing | Cas9 nickase-deaminase fusion + gRNA | DNA, RNA, or RNP delivery | High for specific conversions | Depends on delivery method | Emerging framework | Precision point mutations, therapeutic corrections |
| Prime Editing | Cas9 nickase-reverse transcriptase + pegRNA | Plasmid or RNP delivery | Moderate, improving with new systems | Depends on delivery method | Evolving | Versatile precise edits without donor templates |
Critical Performance Metrics
Specificity and Off-Target Effects: RNP complexes exhibit reduced off-target effects compared to DNA-based delivery systems due to their transient cellular presence and rapid degradation after editing [92] [90]. The shorter exposure time of the genome to active nucleases minimizes unintended mutations, a crucial advantage for therapeutic applications and commercial crop development where genomic purity is paramount [93].
Regulatory Considerations: The absence of foreign DNA integration makes RNP-edited plants indistinguishable from natural mutants in many regulatory frameworks. Countries including the USA, Japan, and Australia have excluded some transgene-free edited crops from GMO regulations, significantly streamlining the path to commercialization [90].
Technical Limitations and Challenges: A significant constraint of RNP systems is the inability to implement selection markers, necessitating laborious screening to identify successfully edited events, especially when edits don't produce visible phenotypes [93]. Additionally, plant regeneration from protoplasts remains a major bottleneck for many species, particularly woody plants, limiting the broad application of this otherwise powerful technology [92] [90].
Experimental Data and Protocols for RNP-Based Editing
Case Study: DNA-Free Genome Editing in Raspberry
A landmark study demonstrated the first successful DNA-free genome editing in raspberry (Rubus idaeus), achieving 19% editing efficiency at the phytoene desaturase (PDS) gene locus through RNP-mediated transfection of protoplasts [91].
Table 2: Quantitative outcomes from raspberry RNP editing experiment
| Experimental Parameter | Specification | Result/Outcome |
|---|---|---|
| Target Gene | Phytoene desaturase (PDS) | Visual albino phenotype confirmation |
| Protoplast Source | Stem-derived cultures | High-yielding isolation |
| RNP Delivery Method | PEG-mediated transfection | Successful intracellular delivery |
| Editing Efficiency | Amplicon sequencing analysis | 19% at target loci |
| Mutation Detection | Multiple techniques | Amplicon sequencing most sensitive |
| Regeneration Status | Protoplast to whole plant | Protocol development critical |
Detailed RNP Editing Workflow
The following diagram illustrates the complete experimental workflow for DNA-free genome editing in plants, from protoplast isolation to regenerated edited plants:
Critical Protocol Steps and Optimization
Protoplast Isolation and Viability: The raspberry protocol used stem-derived tissues from the cultivar BWP102, emphasizing the importance of selecting highly vigorous source material [91]. Enzymatic cell wall digestion must be optimized for each species and tissue type to maximize protoplast yield while maintaining membrane integrity for subsequent transfection.
RNP Assembly and Delivery: Commercial Cas9 proteins and chemically synthesized guide RNAs were combined to form preassembled RNP complexes, then delivered via PEG-mediated transfection [91]. PEG concentration and exposure time require optimization to balance transfection efficiency with cytotoxicity.
Mutation Detection and Analysis: The study highlighted that amplicon sequencing was significantly more sensitive for detecting indels than other methods, crucial for accurately assessing editing efficiency in low-efficiency experiments [91].
Table 3: Key research reagents for DNA-free RNP editing experiments
| Reagent/Category | Specification | Research Function |
|---|---|---|
| Cas9 Nuclease | Wild-type or high-fidelity variants | Target DNA cleavage |
| Guide RNA | Synthetic, chemical modification possible | Target sequence recognition |
| Protoplast Isolation Enzymes | Cellulase, pectinase, macerozyme | Plant cell wall digestion |
| Transfection Agent | Polyethylene glycol (PEG) | Membrane permeabilization |
| Cell Culture Media | Osmotically adjusted with sugars | Protoplast viability maintenance |
| Regeneration Media | Hormone-balanced formulations | Whole plant development |
| Detection Reagents | PCR, electrophoresis, sequencing | Edit verification |
Advanced Delivery Systems Beyond PEG
While PEG-mediated protoplast transfection remains the most reported RNP delivery method in plants, emerging technologies offer promising alternatives:
Nanoparticle-Based Systems: Carbon nanotubes and other nanocarriers can facilitate RNP delivery through stomatal pores or cell walls without requiring protoplast isolation [90].
Cell-Penetrating Peptides (CPPs): CPPs conjugated to RNPs enhance cellular uptake across intact membranes, potentially enabling editing of tissues and embryos without dissociation into single cells [94].
Biolistic Delivery: Gold or tungsten particles coated with RNPs can be bombarded directly into intact cells or tissues, though this method may cause more cellular damage [90].
Cas9 Variant Selection Guide for RNP Editing
The choice of Cas9 variant significantly influences editing outcomes in RNP experiments. Different variants offer distinct advantages in terms of targeting range, specificity, and complex stability.
Structural and Functional Relationships
The structural domains of SpCas9 directly impact its functionality in RNP applications. Key domains include the REC lobe (REC1, REC2, REC3) for sgRNA binding and target recognition, and the NUC lobe containing HNH and RuvC nuclease domains for DNA cleavage, with the PAM-interacting (PI) domain conferring sequence specificity [1]. Understanding these relationships enables researchers to select appropriate Cas9 variants or engineered versions:
Wild-Type SpCas9: Recognizes NGG PAM sequences, offers robust activity, and is widely available from commercial sources for RNP assembly [1].
High-Fidelity Variants: Engineered versions with reduced off-target effects while maintaining on-target efficiency, valuable for applications requiring exceptional specificity [1].
Cas12a (Cpf1) Systems: Alternative CRISPR nucleases recognizing T-rich PAM sequences (TTTV), expanding targeting range and producing staggered DNA ends with potential advantages for certain editing outcomes [1].
Compact Cas Proteins: Smaller nucleases like CasΦ (786 aa) and IscBs (∼400 aa) enable easier delivery and packaging, though they are less characterized in plant RNP systems [93] [95].
DNA-free RNP delivery represents a transformative approach for plant genome editing, particularly valuable for species where transgene persistence presents biological or regulatory challenges. While technical hurdles in plant regeneration persist, ongoing advancements in delivery technologies, Cas protein engineering, and protoplast culture systems continue to expand the applications of this precise editing platform. As regulatory frameworks increasingly distinguish between transgenic and transgene-free editing, RNP methodologies offer a pathway to commercialize improved crop varieties with targeted genetic enhancements.
Structural versus Language Model-Based Protein Design Validation
The precision of gene-editing technologies like CRISPR/Cas9 has revolutionized plant biotechnology, enabling the development of crops with enhanced yield, disease resistance, and nutritional profiles [53]. The efficacy of these tools is fundamentally governed by the Cas9 protein, and engineering superior Cas9 variants with higher specificity, efficiency, or novel functionality is a central pursuit in agricultural research [96]. Validating the performance of these newly designed protein variants relies on two powerful but philosophically distinct computational paradigms: structure-based modeling and language model-based prediction. This guide provides an objective comparison of these methodologies, detailing their experimental protocols, performance data, and practical applications for plant editing research.
Structure-Based Protein Design Validation
Structure-based methods rely on biophysical simulations to model how amino acid changes affect a protein's three-dimensional structure and energetic landscape.
- Core Principle: These approaches operate on the principle that a protein's function is determined by its structure, which in turn is dictated by its sequence and the biophysical properties of its constituents [97]. They use force fields to calculate energetic terms—such as van der Waals forces, hydrogen bonding, and solvation energy—to score the stability and fitness of protein variants [98].
- Representative Tool: The Rosetta software suite is a prominent example. It can model the structures of millions of protein sequence variants and extract dozens of biophysical attributes to assess variant fitness [98].
- Workflow: The typical process involves generating a structure (from experiments or prediction with tools like AlphaFold), introducing mutations in silico, and then using a scoring function to evaluate the structural and energetic consequences of the change [97] [98].
Language Model-Based Protein Design Validation
Protein Language Models (pLMs) learn the evolutionary principles of proteins from massive databases of natural protein sequences, treating amino acid sequences as texts in a foreign language [99].
- Core Principle: pLMs are trained with self-supervised objectives, such as predicting a masked amino acid in a sequence. Through this, they learn the complex contextual relationships between residues and internalize the "grammar" of evolutionarily fit proteins [100] [101]. They can predict the effect of a mutation without an explicit structural model, based solely on the sequence.
- Representative Models: Models like ESM-2 (Evolutionary Scale Modeling) are transformer-based pLMs that generate context-aware representations for each residue in a sequence, which can be fine-tuned for variant effect prediction [98] [99].
- Advanced Hybrids: Newer frameworks like METL (Mutational Effect Transfer Learning) pretrain transformers on biophysical simulation data from Rosetta, thereby uniting machine learning with biophysical modeling. This approach aims to capture fundamental biophysical relationships before being fine-tuned on experimental data [98].
The diagram below illustrates the logical relationship and workflow between these two validation paradigms.
Comparative Performance Data
The following tables summarize quantitative comparisons between structure-based and language model-based validation methods, drawing from benchmark studies.
Table 1: Overall Performance on General Protein Engineering Tasks
| Method | Core Approach | Strengths | Limitations | Representative Performance |
|---|---|---|---|---|
| Structure-Based (e.g., Rosetta) | Biophysical simulation & energetic scoring | High interpretability; strong generalization from small data; models physical mechanisms [98]. | Computationally expensive; accuracy limited by force field quality [97]. | Spearman ρ ~0.85 on Rosetta total score prediction (in-distribution) [98]. |
| Evolutionary pLMs (e.g., ESM-2) | Learning evolutionary constraints from sequences | No explicit structure needed; very fast inference; powerful on natural proteins [100] [99]. | Can overlook biophysics; performance may drop on novel, non-natural sequences [102]. | Top performer on many deep mutagenesis benchmarks, outperforming other pLMs [100]. |
| Biophysics pLMs (e.g., METL) | Pretraining on simulation data, then fine-tuning | Excels with small training sets; strong at position extrapolation; integrates biophysics [98]. | Pretraining can overfit to base proteins used for data generation [98]. | Designed functional GFP variants from only 64 training examples [98]. |
Table 2: Performance on Specific Experimental Assays (Sample Data from METL Study) [98]
| Protein | Assay Type | METL-Local (Spearman ρ) | ESM-2 (Spearman ρ) | Rosetta Total Score (Spearman ρ) |
|---|---|---|---|---|
| GFP | Fluorescence | 0.70 (on small data) | 0.65 (requires more data) | 0.58 |
| GB1 | Stability/Binding | 0.68 (on small data) | 0.55 (requires more data) | 0.51 |
| TEM-1 | Drug Resistance | 0.61 | 0.72 | 0.45 |
Experimental Protocols for Key Validation Experiments
Protocol for Deep Mutational Scanning (DMS) Validation
This experimental protocol is a gold standard for empirically validating computationally predicted variant effects [100].
- Library Construction: Create a comprehensive library of plasmid DNA encoding thousands of single-amino-acid variants of the target protein (e.g., a Cas9 variant).
- Selection Pressure: Express the variant library in an appropriate host system (e.g., plant cells, yeast, bacteria) under a defined functional selection. For Cas9, this could be based on its editing efficiency or specificity.
- Sequencing and Count Analysis: Use next-generation sequencing (NGS) to quantify the abundance of each variant before and after selection.
- Fitness Score Calculation: Compute an enrichment score for each variant based on its frequency change. This score serves as the experimental ground-truth fitness measurement [100].
- Computational Correlation: Correlate the experimental fitness scores with the in silico scores generated by structure-based models or pLMs to evaluate predictive accuracy, typically using a metric like Spearman's rank correlation coefficient.
Protocol for In-Planta Editing Efficiency Assay
This protocol directly tests the performance of computationally designed Cas9 variants in a plant system [53] [96].
- Vector Delivery: Introduce expression vectors for the wild-type and novel Cas9 variants, along with their target guide RNAs, into plant cells. This can be achieved via Agrobacterium-mediated transformation or biolistics.
- Regeneration and Selection: Regenerate whole plants from the transformed cells under appropriate antibiotic or herbicide selection to obtain transgenic lines.
- Genomic DNA Extraction: Isolate genomic DNA from the transgenic plant tissue.
- Target Locus Amplification: Use polymerase chain reaction (PCR) to amplify the genomic region targeted by the Cas9 variant.
- Editing Analysis:
- Restriction Fragment Length Polymorphism (RFLP): If the edit disrupts a restriction site, digest the PCR product and analyze via gel electrophoresis.
- Sanger Sequencing: Sequence the PCR products and use decomposition software (e.g., TIDE, DECODR) to quantify the frequency of insertions and deletions (indels).
- High-Throughput Sequencing: For the most accurate quantification, subject the PCR amplicons to NGS to precisely determine the spectrum and frequency of all mutation types [96].
The workflow for this plant-based validation is detailed below.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Tools for Cas9 Validation in Plants
| Category | Item | Function & Description |
|---|---|---|
| Computational Tools | Rosetta | Suite for protein structure prediction, design, and docking; used for biophysical scoring [98]. |
| ESM-2 pLM | A large protein language model that can be fine-tuned for zero-shot prediction of variant effects [98]. | |
| AlphaFold2 | Highly accurate protein structure prediction tool; provides reliable structural models for analysis [101]. | |
| CRISPR Reagents | Cas9 Expression Vector | A plant-optimized plasmid for expressing the Cas9 gene, often codon-optimized for the target plant species [53]. |
| sgRNA Expression Cassette | A plasmid or fragment containing the sgRNA sequence under a U6 or U3 pol III promoter for expression in plants [96]. | |
| Transformation & Analysis | Agrobacterium tumefaciens Strain (e.g., GV3101) | A common vector for delivering CRISPR/Cas9 constructs into plant genomes via stable transformation [53]. |
| Plant Cell Culture Media | Specific media (e.g., MS Media) for regenerating whole plants from transformed cells [53]. | |
| NGS Library Prep Kit | Commercial kit for preparing sequencing libraries from PCR amplicons of the target site for high-throughput editing analysis [96]. |
Both structure-based and language model-based validation methods are powerful assets for protein engineers in plant research. Structure-based models offer interpretability and are robust in data-scarce scenarios, making them ideal for initial screening and when designing highly novel variants. In contrast, language models leverage evolutionary information for high-throughput, accurate predictions, especially for variants within natural sequence space. The emerging trend of hybrid models like METL, which marry biophysical simulation with deep learning, represents a promising future direction, potentially offering the strengths of both paradigms. The choice of method should be guided by the specific research context: the availability of experimental data, the novelty of the designed protein, and the required balance between interpretability and sheer predictive power.
The application of CRISPR-Cas technology in plant biology represents a paradigm shift in agricultural science, enabling precise genetic modifications that were previously unattainable. As research institutions and commercial developers increasingly deploy these tools to enhance crop resilience, nutritional quality, and yield, understanding the intricate relationship between technical choices and regulatory consequences becomes paramount [103]. The selection of specific Cas variants directly influences not only experimental success but also how resulting plants are classified under global biosafety frameworks.
Different CRISPR systems and their implementation strategies leave distinct molecular signatures that regulatory bodies examine when determining whether a genetically edited plant falls under traditional Genetically Modified Organism (GMO) regulations or qualifies for exemptions. This comparative guide examines how key Cas9 variants—including SpCas9, SaCas9, and engineered alternatives—perform across critical parameters that influence both experimental outcomes and regulatory positioning. By objectively analyzing these variants alongside emerging technologies like prime editing, this guide provides plant researchers with a strategic framework for selecting editing tools that align with both experimental goals and compliance requirements in target markets.
Comparative Analysis of Cas Variants for Plant Research
Technical and Regulatory Characteristics of CRISPR Systems
The table below summarizes the key attributes of major CRISPR systems with implications for both experimental utility and regulatory status.
Table 1: Comparative Analysis of CRISPR Systems for Plant Genome Editing
| CRISPR System | PAM Requirement | Size (aa) | Editing Signature | Regulatory Considerations | Primary Applications in Plants |
|---|---|---|---|---|---|
| SpCas9 (Streptococcus pyogenes) | 5'-NGG-3' [6] | 1368 [6] | Double-strand breaks, indels [104] | Traditional GMO framework in some jurisdictions; potential exemption if no foreign DNA remains [104] | Gene knock-outs, trait stacking [104] |
| SaCas9 (Staphylococcus aureus) | 5'-NNGRRT-3' [6] | 1053 [6] | Double-strand breaks, indels | Favorable for delivery; smaller size may reduce regulatory scrutiny in some cases | Plant pathogen resistance, stress tolerance [6] |
| ScCas9 (Streptococcus canis) | 5'-NNG-3' [6] | ~1368 (89.2% homology to SpCas9) [6] | Double-strand breaks, indels | Expanded targeting may require additional off-target analysis | Not specified in search results |
| Cas12a (Cpf1) | 5'-TTT(A/C/G)-3' [105] | ~1300 | Staggered cuts, easier multiplexing | Different cutting pattern may influence regulatory assessment | Not specified in search results |
| Prime Editors | Varies with nCas9 used [40] | ~Fused nCas9-RT | Precise edits without double-strand breaks [40] | Strong case for non-GMO classification due to precision and absence of donor DNA [40] | Precise nucleotide conversions, small insertions/deletions [40] |
| hfCas12Max (Engineered) | 5'-TN-3' [6] | 1080 [6] | Staggered cuts, high fidelity | Reduced off-target effects may simplify safety documentation | Therapeutic development (potential plant applications) [6] |
Quantitative Performance Metrics
The following table compares experimental performance data for key Cas variants, informing both practical selection and regulatory risk assessment.
Table 2: Experimental Performance Metrics of CRISPR Systems
| CRISPR System | Editing Efficiency | Off-Target Risk | Specificity Enhancements | Delivery Considerations |
|---|---|---|---|---|
| SpCas9 | High in multiple plant species [104] | Moderate without optimization [6] | High-fidelity variants available [6] | Large size challenges viral delivery [6] |
| SaCas9 | High efficiency in plants (e.g., tobacco, potato, rice) [6] | Lower than SpCas9 in comparative studies [6] | SaCas9-HF variant available [6] | AAV-compatible [6] |
| Prime Editors | 20-50% in mammalian cells (PE3 system) [40] | Very low (no double-strand breaks) [40] | Engineered RT reduces off-target effects [40] | Large size challenges delivery [40] |
| hfCas12Max | Enhanced on-target editing [6] | Significantly reduced off-targets [6] | Engineered for high fidelity [6] | AAV and lipid nanoparticle compatible [6] |
Cas Variant Selection Framework for Compliance and Efficiency
The decision pathway below illustrates the strategic selection of Cas variants based on both experimental goals and regulatory considerations.
CRISPR Selection Pathway for Plant Research
Experimental Protocols and Validation Methods
gRNA Design and Optimization Protocol
Effective CRISPR experiments begin with strategic gRNA design, which significantly impacts both on-target efficiency and off-target risks—key considerations for regulatory approval.
- Target Selection: Identify 20nt target sequence immediately upstream of the PAM site specific to your chosen Cas variant (e.g., NGG for SpCas9, NNGRRT for SaCas9) [105]. The target sequence must be unique within the genome to minimize off-target effects.
- Efficiency Prediction: Utilize established algorithms such as Rule Set 3 (for on-target efficiency) and Cutting Frequency Determination (CFD for off-target potential) [105]. These tools analyze thousands of experimentally validated gRNAs to predict performance.
- Experimental Validation: For regulatory compliance, thoroughly characterize editing outcomes through:
- Sanger Sequencing: Confirm intended edits at the target locus
- Whole Genome Sequencing: Identify potential off-target effects in regions with sequence similarity
- Phenotypic Screening: Verify expected trait modifications in edited plants
Online design tools such as CRISPick, CHOPCHOP, and GenScript's sgRNA Design Tool implement these scoring methods and provide comprehensive gRNA rankings based on multiple parameters [105].
Molecular Analysis for Regulatory Documentation
Comprehensive molecular characterization is essential for regulatory submissions and varies depending on the Cas system employed.
- PCR Amplification: Amplify target regions using primers flanking the edit site (150-200bp on each side)
- Sequence Verification: Confirm precise edits through Sanger sequencing; for prime editing, verify exact nucleotide changes without bystander edits [40]
- Off-Target Assessment: Employ tools like Lindel or inDelphi to predict insertion and deletion patterns resulting from CRISPR-mediated cleavage [105]
- Vector Backbone Detection: Test for absence of plasmid backbone integration—critical for classification as non-transgenic in some jurisdictions [104]
For prime editing systems, additional validation should include:
- Flap Resolution Analysis: Verify proper resolution of the branched intermediate structure characteristic of prime editing [40]
- MMR Suppression Assessment: For PE4 and PE5 systems, evaluate efficiency of mismatch repair inhibition [40]
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for CRISPR-Based Plant Research
| Reagent / Tool | Function | Implementation Example |
|---|---|---|
| Cas9 Expression Vector | Expresses Cas nuclease in plant cells | SpCas9 for broad targeting; SaCas9 for size-constrained applications [6] |
| gRNA Expression Construct | Guides Cas to specific genomic loci | Designed with 20nt spacer sequence complementary to target [105] |
| Prime Editing System | Enables precise edits without double-strand breaks | PE2 for basic prime editing; PE3 with additional nicking sgRNA for enhanced efficiency [40] |
| Delivery Vehicle | Introduces editing components into plant cells | Gold particle bombardment, Agrobacterium-mediated transformation, or viral vectors [104] |
| Selective Markers | Identifies successfully transformed plant cells | Antibiotic resistance genes (hygromycin, kanamycin) or visual markers (GFP) |
| gRNA Design Tools | Predicts gRNA efficiency and specificity | CRISPick (Rule Set 3), CHOPCHOP, or GenScript sgRNA Design Tool [105] |
Regulatory Positioning Through Editing Tool Selection
Global Regulatory Trends and Technology Implications
The evolving global regulatory landscape for gene-edited plants increasingly distinguishes between different technical approaches, creating strategic opportunities for researchers.
- SDN-1 and SDN-2 Classification: Cas9 variants that create double-strand breaks resulting in small deletions (indels) without introducing foreign DNA may qualify for expedited regulatory status in several countries, including the United States, Japan, and Argentina [104].
- The Prime Editing Advantage: Prime editing systems, which function without double-strand breaks and can introduce precise changes without donor DNA templates, present a compelling case for non-GMO classification in markets with restrictive GMO regulations [40].
- Trait-Based vs. Process-Based Regulation: An emerging trend shifts focus from the engineering process to the characteristics of the final product, potentially benefiting precise editing technologies that produce changes indistinguishable from natural mutations [103].
The experimental workflow below outlines the key steps for developing gene-edited plants with regulatory compliance as a core consideration.
Experimental Workflow for Compliant Plant Gene Editing
The selection of CRISPR-Cas systems for plant genome editing represents a critical decision point with profound implications for both research success and regulatory positioning. Our comparative analysis demonstrates that while SpCas9 remains the workhorse for routine gene knockouts, emerging alternatives like SaCas9 (for delivery-constrained applications) and prime editing systems (for precision and regulatory advantages) offer compelling benefits for specific use cases. The evolving global regulatory landscape increasingly rewards editing strategies that minimize off-target effects and avoid the introduction of foreign DNA, making high-fidelity variants and precision editing tools particularly valuable for crop development programs targeting markets with restrictive GMO policies. As plant biodesign enters a new era of sophistication, strategic selection of CRISPR tools based on both technical and regulatory parameters will accelerate the development of sustainable agricultural solutions while navigating complex compliance requirements.
Conclusion
The expanding repertoire of Cas9 variants represents a transformative toolkit for plant biotechnology, offering researchers unprecedented precision and flexibility. The comparative analysis reveals that variant selection must be guided by specific project requirements: smaller variants like SaCas9 enable efficient delivery, PAM-relaxed enzymes like Cas9-NG expand targetable sites, and high-fidelity editors minimize off-target risks in complex genomes. Future directions will focus on AI-designed editors with customized properties, improved delivery mechanisms for recalcitrant species, and integration of base and prime editing systems for single-nucleotide precision. As regulatory frameworks evolve, these advanced CRISPR tools promise to accelerate the development of climate-resilient, nutrient-dense crops, fundamentally advancing global food security efforts. The convergence of computational design, optimized delivery, and precise editing heralds a new era of precision plant breeding.
References
- 1. A long journey towards genome editing technologies in ... [https://www.frontiersin.org/journals/genome-editing/articles/10.3389/fgeed.2025.1663352/full]
- 2. Comprehensive benchmarking of genome editing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12144422/]
- 3. Optimization of CRISPR/Cas12 tools for plant genome editing [https://pmc.ncbi.nlm.nih.gov/articles/PMC12418774/]
- 4. CARBON Newsletter (29 April 2025) - Your Latest ... [https://crisprmedicinenews.com/news/carbon-newsletter-29-april-2025-your-latest-news-about-crispr-in-agrobio/]
- 5. CRISPR activation: identifying and using novel genes for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12450905/]
- 6. How to Choose the Right Cas Variant for Every CRISPR ... [https://www.synthego.com/guide/how-to-use-crispr/cas-nuclease-variants/]
- 7. CRISPR/Cas9‐Mediated Gene Knockout in Cereal Crops - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12461677/]
- 8. CRISPR for Crop Improvement: An Update Review [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2018.00985/full]
- 9. Researchers Say New Method Speeds Up Crop Editing [https://www.seedworld.com/us/2025/04/24/researchers-say-new-method-speeds-up-crop-editing/]
- 10. Design of highly functional genome editors by modelling ... [https://www.nature.com/articles/s41586-025-09298-z]
- 11. Genome Engineering in Plant Using an Efficient CRISPR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8525348/]
- 12. PAM-flexible genome editing with an engineered chimeric ... [https://www.nature.com/articles/s41467-023-41829-y]
- 13. Qualitative and Quantitative Detection of CRISPR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10572612/]
- 14. innovations and challenges in rAAV vector-based CRISPR ... [https://www.nature.com/articles/s41434-025-00573-2]
- 15. Viral Vectors for the in Vivo Delivery of CRISPR Components [https://pmc.ncbi.nlm.nih.gov/articles/PMC9133430/]
- 16. CRISPR Delivery Methods: Cargo, Vehicles, and Challenges [https://www.synthego.com/blog/delivery-crispr-cas9/]
- 17. Methods for detecting off-target effects of CRISPR/Cas9 [https://www.sciencedirect.com/science/article/abs/pii/S0734975025002368]
- 18. Engineered Cas9 variant tools expand targeting scope of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7206991/]
- 19. Systematic high-throughput evaluation reveals FrCas9's superior... [https://pubmed.ncbi.nlm.nih.gov/40138428/]
- 20. Systematic high-throughput evaluation reveals FrCas9's ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11939069/]
- 21. CRISPR/Cas9 therapeutics: progress and prospects [https://www.nature.com/articles/s41392-023-01309-7]
- 22. OpenCRISPR-1: Generative AI Meets CRISPR [https://crisprmedicinenews.com/news/opencrispr-1-generative-ai-meets-crispr/]
- 23. GitHub - Profluent-AI/ OpenCRISPR : AI-generated gene editing systems [https://github.com/Profluent-AI/OpenCRISPR]
- 24. Profluent Announces Publication of Generative AI ... [https://crisprmedicinenews.com/press-release-service/card/profluent-announces-publication-of-generative-ai-research-in-nature-with-new-results-for-opencrispr/]
- 25. Revolutionizing CRISPR technology with artificial intelligence [https://pmc.ncbi.nlm.nih.gov/articles/PMC12322281/]
- 26. A comparison of three different delivery methods for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10131283/]
- 27. Engineering Agrobacterium for improved plant transformation [https://pmc.ncbi.nlm.nih.gov/articles/PMC11885899/]
- 28. Efficient protoplast isolation and transfection for CRISPR ... [https://link.springer.com/article/10.1007/s11240-025-03198-w]
- 29. A Protoplast System for CRISPR-Cas Ribonucleoprotein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11990275/]
- 30. Enhancing biolistic plant transformation and genome ... [https://www.nature.com/articles/s41467-025-60761-x]
- 31. harnessing multiplex editing for polygenic trait engineering ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12527382/]
- 32. Comprehensive approaches to design efficient gRNA for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12259558/]
- 33. Applications of multiplexed CRISPR–Cas for genome ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12322214/]
- 34. Multiplexed CRISPR technologies for gene editing and ... [https://www.nature.com/articles/s41467-020-15053-x]
- 35. Applications of multiplex genome editing in higher plants [https://www.sciencedirect.com/science/article/abs/pii/S0958166918301496]
- 36. Editorial: Engineering future crops through genome editing [https://pmc.ncbi.nlm.nih.gov/articles/PMC12605012/]
- 37. Fine tuning wheat heading time through genome editing of ... [https://www.nature.com/articles/s41598-025-25295-8]
- 38. Efficient CRISPR/Cas9-mediated genome editing of ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1677409/full]
- 39. Plant base editing: a decade of progress and future ... [https://www.sciencedirect.com/science/article/pii/S2662173825002103]
- 40. Emerging trends in prime editing for precision genome ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12322275/]
- 41. CRISPR/Cas9-mediated mutation of OsSWEET14 in rice cv ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7333420/]
- 42. Improved bacterial leaf blight disease resistance in the major ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0255470]
- 43. Engineering Resistance to Bacterial Blight and ... - Rice [https://thericejournal.springeropen.com/articles/10.1186/s12284-021-00482-z]
- 44. CRISPR/Cas9-mediated mutation of OsSWEET14 in rice cv ... [https://bmcplantbiol.biomedcentral.com/articles/10.1186/s12870-020-02524-y]
- 45. Development and activity evaluation of a highly efficient ... [https://www.sciencedirect.com/science/article/pii/S2468014125001190]
- 46. A comparison of three different delivery methods for ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2023.1111110/full]
- 47. A Comparative Study on CaMLO2-Gene-Editing Efficiency ... [https://www.mdpi.com/1422-0067/24/23/16775]
- 48. Biofortification strategies for enhancing crop nutritional value [https://link.springer.com/article/10.1007/s44372-025-00276-3]
- 49. CRISPR-Cas9 Application in Crop Biotechnology [https://nhsjs.com/2025/crispr-cas9-application-in-crop-biotechnology-a-decade-of-research-2014-to-2024-toward-food-security-in-developing-countries/]
- 50. Unlocking Opportunities and Overcoming Challenges in ... [https://pubmed.ncbi.nlm.nih.gov/39940376/]
- 51. Emerging trends in prime editing for precision genome ... [https://www.nature.com/articles/s12276-025-01463-8]
- 52. CRISPR-Cas orthologues and variants - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC5569134/]
- 53. CRISPR Variants for Gene Editing in Plants: Biosafety Risks and Future Directions - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10671001/]
- 54. Exonuclease-fused CRISPR-cas system enhances targeted genome editing for functional genomics in soybean | BMC Plant Biology | Full Text [https://bmcplantbiol.biomedcentral.com/articles/10.1186/s12870-025-07230-1]
- 55. CRISPR/Cas9 editing of CBP80 enhances drought ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1598947/full]
- 56. CRISPR-Cas9-based precise engineering of SlHyPRP1 ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2023.1186932/full]
- 57. Enhanced rice salinity tolerance via CRISPR/Cas9- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7413041/]
- 58. Off-target interactions in the CRISPR-Cas9 Machinery [https://pubmed.ncbi.nlm.nih.gov/40688512/]
- 59. accelerating Cas9/sgRNA cleavage efficiency validation in ... [https://link.springer.com/article/10.1007/s42994-024-00139-7]
- 60. Revolutionizing CRISPR technology with artificial intelligence [https://www.nature.com/articles/s12276-025-01462-9]
- 61. Genome Engineering in Plant Using an Efficient CRISPR ... [https://www.frontiersin.org/journals/genome-editing/articles/10.3389/fgeed.2020.618385/full]
- 62. Improving Plant Genome Editing with High-Fidelity xCas9 ... [https://www.sciencedirect.com/science/article/pii/S1674205219301248]
- 63. Expanding the base editing scope to GA and relaxed NG ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7061872/]
- 64. Engineered xCas9 and SpCas9‐NG variants broaden PAM ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6737014/]
- 65. Genome-wide evaluation of gene editing outcomes using ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2024.1496861/full]
- 66. CRISPR/dCas9 platforms in plants - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC6920162/]
- 67. Challenges and Opportunities in the Application of CRISPR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12503735/]
- 68. Advances and Applications of Plant Base Editing Technologies [https://pmc.ncbi.nlm.nih.gov/articles/PMC12524351/]
- 69. Improving plant C-to-G base editors with a cold-adapted ... [https://www.sciencedirect.com/science/article/abs/pii/S0167779925000861]
- 70. Critical considerations and computational tools in plant ... [https://www.sciencedirect.com/science/article/pii/S2405844024171666]
- 71. The Complete Guide to Understanding CRISPR sgRNA [https://www.synthego.com/guide/how-to-use-crispr/sgrna/]
- 72. Engineered circular guide RNAs enhance miniature ... [https://www.nature.com/articles/s41467-025-58367-4]
- 73. Improved specificity and efficiency of in vivo adenine base ... [https://www.nature.com/articles/s41551-025-01545-y]
- 74. Genome-wide CRISPR guide RNA design and specificity ... [https://genomebiology.biomedcentral.com/articles/10.1186/s13059-025-03488-8]
- 75. Recent advances of CRISPR-based genome editing for ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2024.1478398/full]
- 76. CRISPR/Cas9 Delivery Systems to Enhance Gene Editing ... [https://www.mdpi.com/1422-0067/26/9/4420]
- 77. Nanoparticle based delivery of CRISPR/Cas9 genome editing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6398936/]
- 78. High-fidelity CRISPR-Cas9 variants with undetectable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4851738/]
- 79. CRISPR-Cas9 induces large structural variants at on-target ... [https://www.nature.com/articles/s41467-022-28244-5]
- 80. In-depth assessment of the PAM compatibility and editing activities of... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8421146/]
- 81. Multiplex nucleotide editing by high-fidelity Cas variants with... 9 [https://bmcplantbiol.biomedcentral.com/articles/10.1186/s12870-019-2131-1]
- 82. AI-guided Cas9 engineering provides an effective strategy ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12583830/]
- 83. Loop Engineering of AtCas9 for effective and broad ... [https://www.sciencedirect.com/science/article/pii/S2772892725000604]
- 84. Genome-wide specificity of plant genome editing by both ... [https://www.nature.com/articles/s41598-022-13034-2]
- 85. Whole-genome sequencing reveals rare off-target ... [https://www.nature.com/articles/s41438-021-00549-4]
- 86. Off-target effects in CRISPR/Cas9 gene editing - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10034092/]
- 87. Efficiency, Specificity and Temperature Sensitivity of Cas9 ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8790294/]
- 88. Efficient plant genome engineering using a probiotic ... [https://www.nature.com/articles/s41467-023-41802-9]
- 89. High-throughput sgRNA testing reveals rules for Cas9 ... [https://www.frontiersin.org/journals/genome-editing/articles/10.3389/fgeed.2023.1196763/full]
- 90. Non-GM Genome Editing Approaches in Crops [https://www.frontiersin.org/journals/genome-editing/articles/10.3389/fgeed.2021.817279/full]
- 91. DNA-free CRISPR genome editing in raspberry (Rubus ... [https://www.frontiersin.org/journals/genome-editing/articles/10.3389/fgeed.2025.1589431/full]
- 92. CRISPR RNP-Mediated Transgene-Free Genome Editing ... [https://pubmed.ncbi.nlm.nih.gov/40923633/]
- 93. Advances in gene editing without residual transgenes in plants [https://pmc.ncbi.nlm.nih.gov/articles/PMC8968301/]
- 94. Developing an Efficient DNA-Free, Non-Transgenic ... [https://avf.org/research-summary/developing-an-efficient-dna-free-non-transgenic-genome-editing-methodology-in-grapevine-2/]
- 95. CMN Weekly (28 November 2025) - Your Weekly CRISPR ... [https://crisprmedicinenews.com/news/cmn-weekly-28-november-2025-your-weekly-crispr-medicine-news/]
- 96. Comparison of CRISPR/Cas9 and Cas12a for gene editing ... [https://www.sciencedirect.com/science/article/pii/S2211926424004089]
- 97. Experimentally-Driven Protein Structure Modeling - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7214187/]
- 98. Biophysics-based protein language models for ... [https://www.nature.com/articles/s41592-025-02776-2]
- 99. A Comprehensive Review of Protein Language Models [https://arxiv.org/html/2502.06881v1]
- 100. Structure-Informed Protein Language Models are Robust ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10418537/]
- 101. Evaluating the advancements in protein language models for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11790633/]
- 102. Pretrained protein language models choose between ... [https://pubmed.ncbi.nlm.nih.gov/41256629/]
- 103. Programmable genome engineering and gene ... [https://www.sciencedirect.com/science/article/pii/S2590346225001890]
- 104. CRISPR in Agriculture: 2024 in Review [https://innovativegenomics.org/news/crispr-in-agriculture-2024/]
- 105. A Guide to Efficient CRISPR gRNA Design: Principles and ... [https://www.genscript.com/a-guide-to-efficient-crispr-grna-design-principles-and-design-tools.html]